- 1Department of Biology, University of Crete, Heraklion, Greece
- 2Botanical Garden, University of Crete, Rethymnon, Greece
- 3Laboratory of Experimental Endocrinology, School of Medicine, University of Crete, Heraklion, Greece
- 4Laboratory of Clinical Virology, School of Medicine, University of Crete, Heraklion, Greece
- 5Clinic of Social and Family Medicine, School of Medicine, University of Crete, Heraklion, Greece
This review performs a comprehensive assessment of the therapeutic potential of three native herbs of Crete (Thymbra capitata (L.) Cav., Salvia fruticosa Mill. and Origanum dictamnus L.), their phytochemical constituents, health benefits and issues relevant to their safety, within a translational context. Issues discussed comprise: 1) Ethnopharmacological uses of the three herbs, reviewed through an extensive search of the literature; 2) Systematic analysis of the major phytochemical constituents of each plant, and their medicinal properties; 3) To what extent could the existing medicinal properties be combined and produce an additive or synergistic effect; 4) Possible safety issues. We conclude with a specific example of the use of a combination of the essential oils of these plants as an effective anti-viral product and the experience gained in a case of a plant-based pharmaceutical development, by presenting the major steps and the continuum of the translational chain.
Introduction
Plants are traditionally used, in different forms, for the treatment of diseases, since the Neolithic Era (Hardy, 2019). Different populations used native flora for the production of preparations, efficient in curing different diseases and conditions (Fabricant and Farnsworth, 2001). The Cretan area (KK, Crete and Karpathos floristic region) is such an example, as its evolutionary history preserved a very diverse flora, with more than 2000 indigenous species, including a high number of endemics (Dimopoulos et al., 2013). Cretan people abundantly use plants and greens in different aspects of their life. Cretan diet, a specific entity of the Mediterranean diet, uses a diversity of plants for culinary purposes (Renaud, 1995 and references herein), while a variety of plants are used as decoctions and infusions for recreational or medical purposes. We systematically analyzed the habits of a rural population in Crete and reported that use of different plant infusions, alone or in combination, resulted in a significant protection from common cold and influenza infections (Lionis et al., 1998). Based on this study, we have focused on three of the most efficient plants (Coridothymus capitatus, Salvia fruticosa and Origanum dictamnus) and developed a supplement, efficient at combatting upper respiratory tract infections (Duikler et al., 2015; Anastasaki et al., 2017).
In this review, we analyze in depth the health benefits of these plants, their most prominent active compounds and their combination. In addition, we review our experience with the development of a nutraceutical product, discussing potential bottlenecks encountered during the process.
The Plants
Taxonomy and Distribution
According to the Euro + Med plantbase, Coridothymus capitatus (L.) Reichenb. fil. is a dwarf-shrub. It is the only member of the monospecific genus Coridothymus. According to the Euro + Med plantbase (Accessed at June 2017), the name Coridothymus capitatus (L.) Reichenb. fil. is synonym of Thymbra capitata (L.) Cav., which is included in Kingdom–Plantae, Division–Tracheophyta, Subdivision–Spermatophytina, Class–Magnoliopsida, Superorder–Asteranae, Order–Lamiales, Family–Lamiaceae Lindl., Genus–Thymbra L.
Accepted name:Thymbra capitata (L.) Cav., Homotypic synonyms: Coridothymus capitatus (L.) Rchb. f., Origanum capitatum (L.) Kuntze, Satureja capitata L., Thymus capitatus (L.) Hoffmanns. & Link.
Salvia fruticosa Mill. is also a member of the Labiatae family. The taxon is included in Kingdom–Plantae, Division–Tracheophyta, Subdivision–Spermatophytina, Class–Magnoliopsida, Superorder–Asteranae, Order–Lamiales, Family–Lamiaceae Lindl., Genus–Salvia L.
Accepted name: Salvia fruticosa Mill, Heterotypic synonyms: Salvia baccifera Etl., Salvia clusii Jacq., Salvia cypria Unger & Kotschy, Salvia fruticosa subsp. cypria (Unger & Kotschy) Holmboe, Salvia fruticosa subsp. thomasii (Lacaita) Brullo and al., Salvia incarnata Etl., Salvia libanotica Boiss. & Gaill., Salvia lobryana Azn., Salvia marrubioides Vahl, Salvia ovata F. Dietr., Salvia sipylea Lam., Salvia subtriloba Schrank, Salvia sypilea Lam., Salvia thomasii Lacaita, Salvia triloba L. f., Salvia triloba subsp. calpeana (Dautez & Debeaux) P. Silva, Salvia triloba var. calpeana Dautez & Debeaux, Salvia triloba subsp. libanotica (Boiss. & Gaill.) Holmboe, Sclarea triloba(L. f.) Raf.
Origanum dictamnus L. is a local endemic of Crete, member of Labiatae family. The taxon is included in Kingdom–Plantae, Division–Tracheophyta, Subdivision–Spermatophytina, Class–Magnoliopsida, Superorder–Asteranae, Order–Lamiales, Family–Lamiaceae Lindl., Genus–Origanum L.
Accepted name: Origanum dictamnus L., Heterotypic synonyms: Amaracus dictamnus (L.) Benth., Majorana dictamnus (L.) Kostel., Heterotypic synonyms: Amaracus tomentosus Moench, Origanum dictamnifolium St.-Lag., Origanum saxatile Salisb.
Coridothymus capitatus is found throughout the Mediterranean region distributed in all Mediterranean countries with the exception of France. Salvia fruticosa is distributed in Italy, Sicily, and the eastern Balkans, including Cyprus, while Origanum dictamnus is local endemic of Crete (Fielding and Turland, 2005).
Methodology
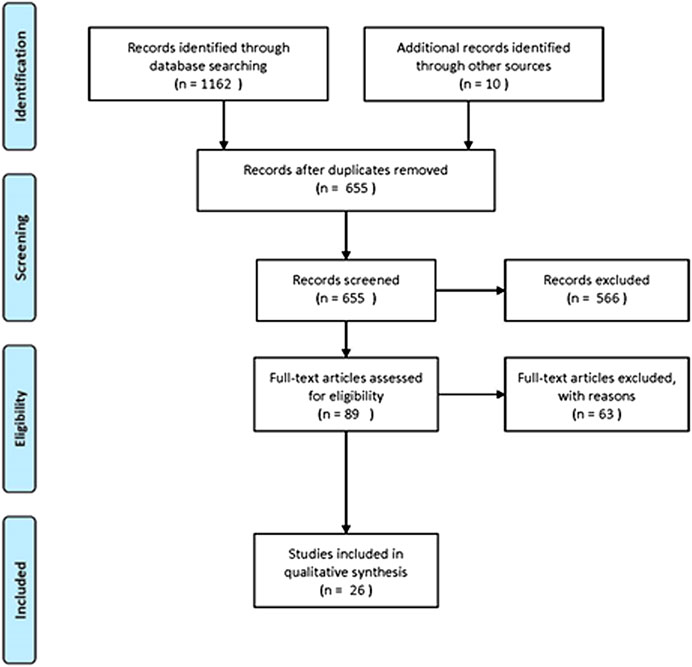
In the context of this review, PubMed, Scopus and Google Scholar were used for the retrieval of the relevant bibliography. The following inclusion criteria were used for the systematic review of the three plants: articles published in English, articles concerning countries of the Mediterranean basin and also including Jordan, Syria, Iran and Iraq, as well as studies with the following keywords in their text: Coridothymus capitatus, Salvia fruticosa and Origanum dictamnus. The PRISMA flow diagram, presenting the results of our search, is depicted in Figure 1. The primary search identified 1,162 articles, plus 10 articles from other sources. After the implementation of the eligibility criteria, 26 articles were finally incorporated. Any evaluation of studies concerning the plant quality was out of scope and has not been included in this review.
Concerning the synergistic interactions of the main constituents of the essential oil (see below) the review was carried out through a literature searching of the same databases using the following keywords in all fields: “synerg* AND (carvacrol OR eucalyptol OR 1,8-cineole OR beta-Caryophyllene).” The selection of manuscripts was based on the following criteria: articles published in English and articles where only documented synergy between pure substances was reported. The primary search identified 4,101 articles, with 1,538 from PubMed, 1,910 from Google Scholar and 653 from Scopus and after the implementation of the eligibility criteria, excluding the repetitions, 25 articles were finally included in the qualitative analysis.
Ethnopharmacological Uses of the Plants
Coridothymus capitatus, Origanum dictamnus and Salvia fruticosa are renown since antiquity for their pharmaceutical properties (Karousou et al., 1998). The first two are included in the Dioscorides book “De Materia Medica.” Salvia fruticosa was introduced in the Iberian Peninsula for cultivation by Greeks and Phoenicians and elements of these cultivations can be discovered today in several parts of the Iberian coast (Rivera et al., 1994). O. dictamnus is also mentioned from the Minoan era, through the centuries from several physicians and philosophers including Asklepios, Euripides, Aristotle, Hippocrates, Theophrastus, Virgil and Galen (Vrachnakis, 2003).
Coridothymus capitatus is known in local cultures with several common names among which Spanish oregano, thyme, headed thyme, conehead thyme, agrio thymari (αγριο θυμάρι), thymari (θυμάρι), tomilho de Creta and tomilho de Dioscórides, Salvia fruticosa is known with the names Greek sage and faskomilo (φασκόμηλο), while Origanum dictamnus is known as diktamo (δίκταμο), dittany of Crete and dictamo de Creta.
The ethnobotanical and ethnopharamacological studies concerning the medical use of Coridothymus capitatus, Salvia fruticosa and Origanum dictamnus, as well as from studies on local herbal markets in Eastern Mediterranean region, are presented in Tables 1–3 respectively, together with the plant part and their mode of preparation/use.
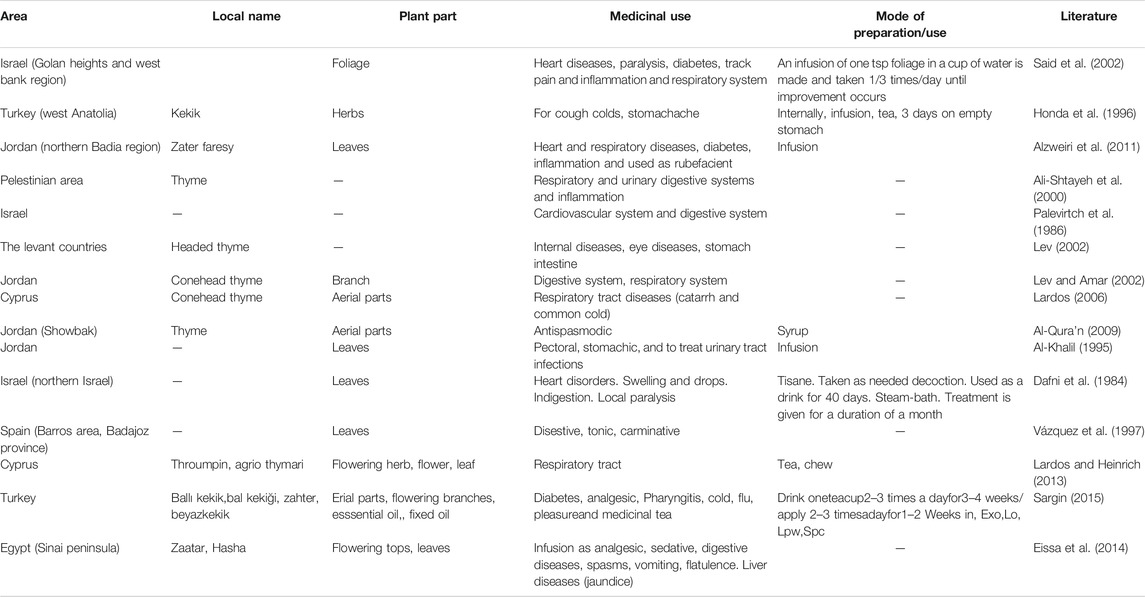
TABLE 1. Medicinal uses of Coridothymus capitatus derived from ethnobotanical and ethnopharmacological studies.
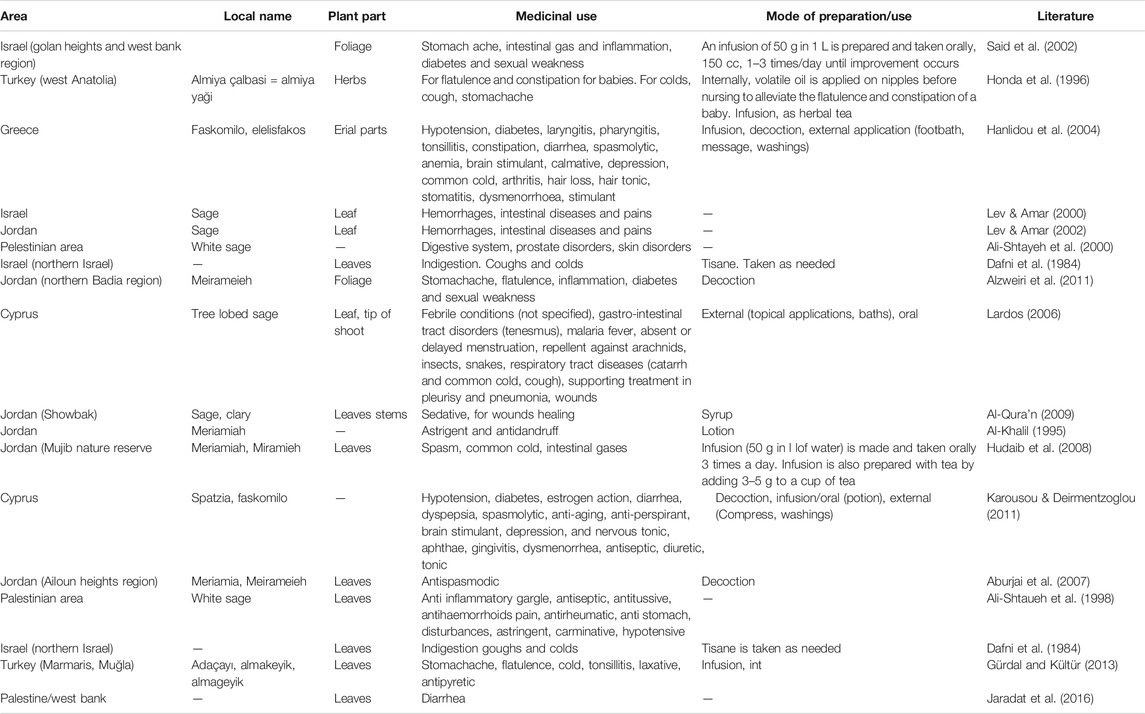
TABLE 2. Medicinal uses of Salvia fruticosa derived from ethnobotanical and ethnopharamacological studies.
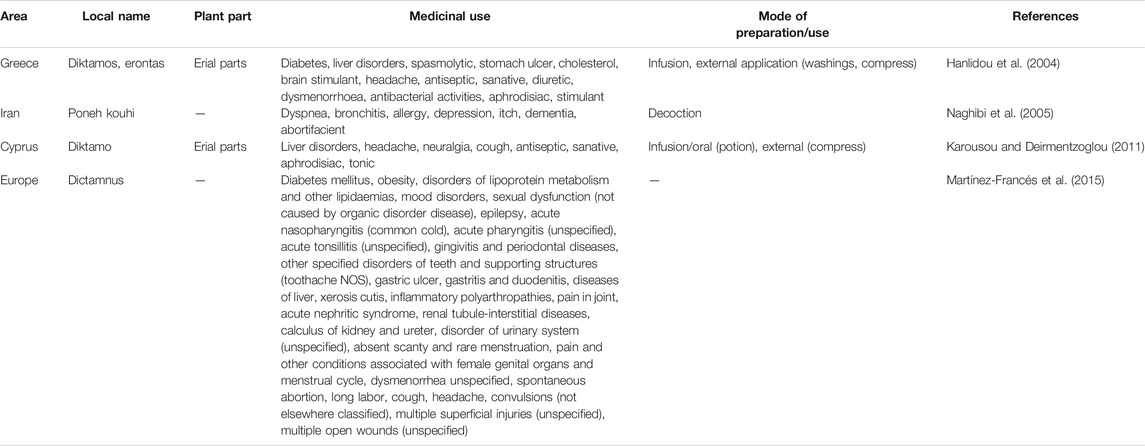
TABLE 3. Medicinal uses of Origanum dictamnus derived from ethnobotanical and ethnopharmacological studies.
Based on this information, medicinal uses, in an ethnobotanical context, suggest that the three species have a highly antimicrobial and anti-inflammatory action. In addition, they are active against various targets and diseases, including diseases of the respiratory, digestive and urinary systems.
Phytochemical Constituents
The essential oils of the three plants is produced from collected plant material, air dried in the dark, at room temperature (25°C) for 10 days. For analysis, after steam distillation, 1 ml of volatile oils were diluted with 2 ml of ether and filtered through anhydrous sodium sulfate to remove water traces and were stored at 4°C. Analysis was performed, as described previously (Duikler et al., 2015), by Gas Chromatography-Mass Spectroscopy (GC-MS, Shimadzu, QP 5050 A), with a MDN-5 column and a Quadrupole Mass Spectrometer as detector, after injection of 2 μL. The carrier gas was helium, the flow rate 0.9 ml/min. The sample was measured in a split mode procedure (1:35). For GS-MS detection an electron ionization system was used with ionization energy at 70 eV. The interested reader should refer to Duikler et al. (2015) for further analytical details.
The chemical constituents of the essential oils of the three plants, as well as their combination (1.5% v/v of pure essential oils in olive oil carrier), used in the context of upper respiratory tract infections are presented in details as Supplemental Material in Duikler et al. (2015). According to this description (see also Figure 2) carvacrol (52.7%) is the main constituent of the mixture, followed by eucalyptol (12.77%) and β-caryophyllene (3.41%). The compounds p-cymene, γ-terpinene, borneol and α-terpineol participate with concentrations 1.32, 1.17, 1.68 and 1.06% respectively, while the rest 15 compounds participate with less than 1% (Figure 2).
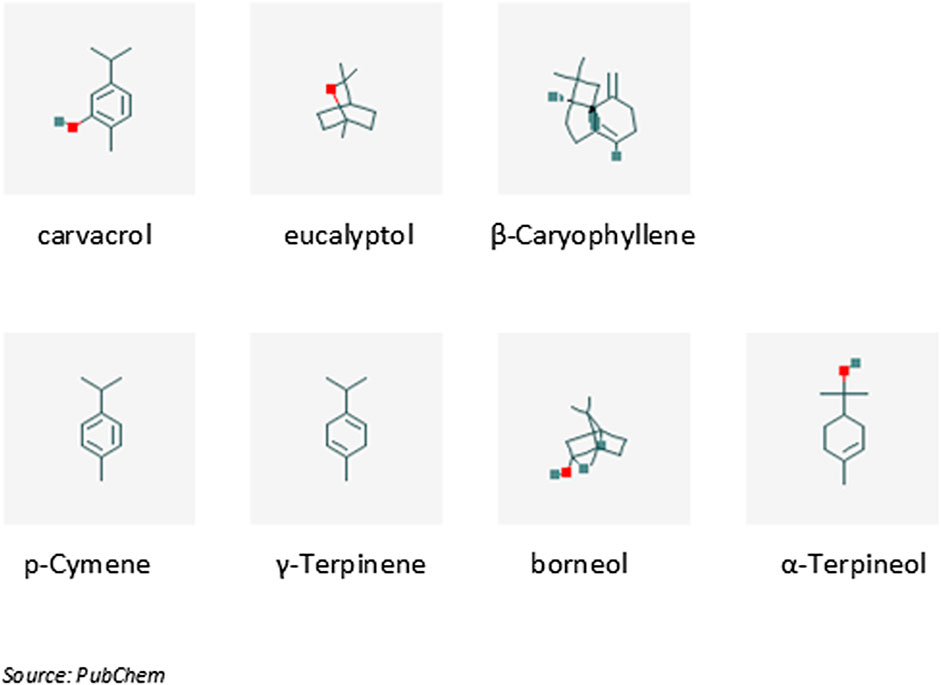
FIGURE 2. The main phytochemical constituents contained in the essential oil of plants discussed in this review. IUPAC names: 2-methyl-5-propan-2-ylphenol (carvacrol), 1,3,3-trimethyl-2-oxabicyclo [2.2.2] octane (eucalyptol), (1R,4E,9S)-4,11,11-trimethyl-8-methylidenebicyclo [7.2.0] undec-4-ene (β-Caryophyllene), 1-methyl-4-propan-2-ylbenzene (p-Cymene), 1-methyl-4-propan-2-ylcyclohexa-1,4-diene (γ-Terpinene), (1S,2R,4R)-1,7,7-trimethylbicyclo [2.2.1] heptan-2-ol (borneol), 2-(4-methylcyclohex-3-en-1-yl)propan-2-ol (α-Terpineol).
Carvacrol (5-isopropyl-2-methylphenol) is an aromatic monoterpene with molecular weight 150.2 g/mol. It is a constituent of many essential oils, especially those found in plants of the family Lamiaceae, where the Origanum vulgare subsp. hirtum (Link) (Kokkini and Vokou, 1989).
Eucalyptol (1,8-cineole) is a cyclic ether monoterpene (molecular weight 154.3 g/mol), which is found as a constituent of plant essential oils (its name is derived from its presence in essential oils of Eucalyptus globosus). It is also considered as a major monoterpene emitted by vegetation into the atmosphere (Guenther et al., 1994).
β-caryophyllene is a natural bicyclic sesquiterpene with molecular weight 204.4 g/mol that is a constituent of many essential oils, such as clove or rosemary oil (Bernardes et al., 2010).
Concerning the other compounds, p-cymene is a naturally occurring organic compound that is characterized as a hydrocarbon related to a monoterpene. Its molecular weight is 134.2 g/mol and it is a constituent of several essential oils, such as the oil of cumin (Allahghadri et al., 2010). γ-terpinene is a terpene with molecular weight 136.3 g/mol. It is a major component of essential oils made from Citrus fruits (Lücker et al., 2002). Borneol is a bicyclic monoterpene with molecular weight 154.3 g/mol containing exactly two rings which are fused to each other. α-Terpineol is a naturally occurring monoterpene alcohol, derived from several sources, including oil of pines. Terpineol, due to its pleasant odor (similar to lilac), is used as an ingredient in perfumes, cosmetics, and flavors (Kim and Chung, 2000).
Uses of Phytochemical Constituents
Apart from the ethnobotanical and ethnopharmacological uses of plants mentioned above, several uses of the aforementioned phytochemical constituents are also documented:
Carvacrol
Carvacrol is the basic constituent of many essential oils, mainly of oregano species (Labiatae) (Kokkini and Vokou, 1989; Kirimer et al., 1995; Baydar et al., 2004; Stefanaki et al., 2016); it is characterized by its strong antioxidant properties (similar to those of vitamin E and ascorbic acid) (Mastelic et al., 2008). It is also known for its pronounced antimicrobial and antibacterial action (Suntres et al., 2015). The antimicrobial activity of several essential oils has been related to their content of carvacrol (García-Beltrán and Esteban, 2016), while, in a number of studies, the antimicrobial and the antibacterial action of the pure compound has also been investigated (Ben Arfa et al., 2006).
The antibacterial action of carvacrol extends to a variety of Gram-positive and negative bacteria (Kim et al., 1995; Rattanachaikunsopon and Phumkhachorn, 2010a; Ravishankar et al., 2010; Rivas et al., 2010). The compound acts as a transmembrane monovalent cation (hydroxyl proton for a potassium cation) exchanger (Suntres et al., 32,015). Therefore, in addition to its hydrophobic characteristics, allowing its accumulation in the membrane, the presence of free hydroxyls is essential for its antibacterial and antimicrobial activity (Ben Arfa et al., 2006).
An anti-inflammatory action of carvacrol is also documented (Landa et al., 2009; Hotta et al., 2010) and relies to the decreased production of inflammatory mediators, including cytokines, prostaglandins, enzymes, nitric oxide (NOS) and reactive oxygen species (ROS) (da Silva Lima et al., 2013). The authors suggest that carvacrol’s anti-inflammatory effects specifically induced IL-10 release, leading, subsequently, to the reduction of IL-1β and prostanoids production. In addition, it was reported that the inhibition of prostaglandin synthesis (Wagner et al., 1986) by carvacrol, is the basis of its antinociceptive activity. Additional mechanisms of antinociceptive actions of carvacrol include its agonistic activity for Transient Receptor Potential Vanilloid 3 (TRPV3), an ionic channel implicated in hyperalgesia and possibly skin sensitization (Xu et al., 2006). In addition, carvacrol activates and promptly desensitizes the specific sensor of environmental irritants TRPA1 ion channel/receptor (Xu et al., 2006).
Eucalyptol (1,8-Cineole)
1,8-cineole (cineole), also known as eucalyptol, is the principal constituent of most Eucalyptus oil-preparations. It is employed in drug preparations, as a percutaneous enhancer and anti-inflammatory agent, decongestant and antitussive, while in aromatherapy it is used as a skin stimulant (Santos and Rao, 2000; Yuan et al., 2006).
Juergens et al. (2004), reported that 1,8-cineol inhibited Th1/Th2-associated cytokine production by human lymphocytes and monocytes. This anti-inflammatory action resulted in a subsequent inhibition of cytokine-induced airway mucus hypersecretion.
1,8 cineole is also known for its analgesic action (Guimarães et al., 2013). Santos and Rao (2000) suggested that the potent anti-inflammatory action of the compound was associated with an outstanding peripheral analgesic effect. They also suggested that 1,8- cineole-induced analgesic effects are not associated to neuronal toxicity, while Khalil et al. (2004) documented that 1,8-cineol, in a concentration-dependent manner, acted directly on sensory nerves and blocked nerve excitability (Lima-Accioly et al., 2006; Guimarães et al., 2013), through a direct activation of TRPM8 channels, which is a specific heat/cold receptor. TRPM8 activation in sensory nerves by 1,8-cineole, produced a specific analgesic effect, in cases of peripheral nerve injury (Proudfoot et al., 2006).
β-Caryophyllene
β-caryophyllene is a widespread plant volatile compound, which is a selective ligand of the CB2 cannabinoid receptor, specifically related to analgesia and inflammation, and devoted of psychotropic actions, mediated by CB1 (Gertsch et al., 2008; Klauke et al., 2014). Therefore, CB2 receptor-selective agonists, like β-caryophyllene, are potential analgesic drug candidates, in various types of pain (such as neuropathic pain) (Guindon and Hohmann, 2008; Kinsey et al., 2010).
p-Cymene
p-cymene is a precursor of carvacrol. It has an antimicrobial action per se, but it is less effective than carvacrol, when used alone (Kiskó and Roller, 2005), because p-cymene lacks a hydroxyl group, which is an important radical for the antimicrobial activity (Ultee et al., 1999; 2002). p-cymene also demonstrated an enhanced antinociceptive action in animal models of neurogenic and inflammatory pain (Guimarães et al., 2013).
γ-Terpinene
The antimicrobial activity of γ-terpinene is debatable, with positive (Sato et al., 2007) and negative (Sivropoulou et al., 1996) reports, against strains of Gram-positive or Gram-negative bacteria.
Borneol
Borneol is a known medicinal substance in Chinese and Indian traditional medicine. Borneol and its derivatives, possess antimicrobial (Corrêa et al., 2012), anti-inflammatory (Ehrnhöfer-Ressler et al., 2013) and antiviral activity. It also posesses a highly stimulatory action at GABAA receptors (Granger et al., 2005; Manayi et al., 2016). GABA is the predominant inhibitory transmitter in the mammalian central nervous system and stimulation of GABAA receptors produces anxiolysis, sedation, anesthesia and myorelaxation (Chebib and Johnston, 2000).
Borneol also expressed topical analgesic action. Wang et al. (2017), providing evidence for a topical analgesic efficacy in humans; TRPM8 was identified as its main molecular target. Moreover, borneol demonstrated anti-thrombotic effects, related to its anticoagulant properties (Li et al., 2008; Chen et al., 2015), possibly related to its potent modulation of the nitrite/nitrate reductase activity (Tang et al., 2009).
α-Terpineol
α-terpineol is characterized by a moderate antibacterial activity, with a broad antibacterial spectrum. It is worth mentioning that a-terpineol showed antibacterial activities against penicillin-resistant bacterial strains (Kotan et al., 2007).
α-terpineol exerts central and peripheral antinociceptive activity (Quintans-Júnior et al., 2011). It also inhibits NF-κB and subsequently down-regulats the expression of proinflammatory IL-1β and IL-6 cytokines (Held et al., 2007; Hassan et al., 2010), mainly explaining the compound’s antinociceptive and anti-inflammatory properties (Oliveira et al., 2012).
α-terpineol also exhibited a gastroprotective action in ethanol-induced gastric ulcers (Souza et al., 2011) and beneficial effects in the cardiovascular system (Ribeiro et al., 2010). The proposed mechanism of action included endothelial NO-related vasorelaxation and activation of the NO–cGMP pathway.
Registered Medical Applications
A series of clinical studies (most of them double-bind placebo-controlled trials, Table 4) evaluated the efficacy of 1,8-cineol (eucalyptol), in several medical issues. 1,8-cineol (eucalyptol) is a licensed medicinal product in Germany, since many years, in intestine-soluble capsules, for the treatment of acute and chronic bronchitis, sinusitis, respiratory infections and rheumatoid-like joint diseases (Juergens et al., 2003; Guimarães et al., 2013).

TABLE 4. Clinical studies concerning the evaluation of efficacy of the main constituents of essential oil preparation from Thymbra capitata (L.) Cav., Salvia fruticosa Mill. and Origanum dictamnus L., in several medical conditions.
Borneol and p-cymene have also been subjects of clinical studies for induced analgesia and treatment of the Fish Tapeworm disease respectively (Vartiainen, 1950; Wang et al., 2017).
Synergy of Phytochemical Constituents
According to a classical definition, synergy occurs when the combined effect of two or more substances is greater than the sum of the individual agents (Breitinger, 2012). When the registered effect is an add up of individual actions, the action is reported as additive. Recently, the definition of synergy has been clarified from two points of view, the pharmacodynamic (enhanced therapeutic actions on the same target) and pharmacokinetic (no direct interaction but a multi-target behavior) (Yang et al., 2014; Zhou et al., 2016). The main reason of employing combinations of active substances with synergistic interactions is to reduce the administered amount of each compound and to increase the biological activity of a preparation/mixture against a specific target. In addition, this strategy diminishes the chance of pharmaceutical resistance of the pathogenic organism (Araújo et al., 2016).
Synergistic interactions were known for specific substances long time ago. For example, Barbaste et al. (2002) proposed a “cascade” reaction from lipophilic to hydrophilic antioxidants, enhancing their biological effects, while Vardar-Ünlü et al. (2003) suggested that the crude essential oil of Thymus pectinatus var pectinatus was more effective as antioxidant (DPPH assay) than its main active components (thymol and carvacrol). Testing the antioxidant activity of thymol, carvacrol and p-cymene, Milos and Makota (2012) showed synergistic effects in any combination of any two of the above compounds. Moreover, p-cymene, the precursor of carvacrol was found to enhance the bactericidal activity of carvacrol when used in combination (Kiskó and Roller, 2005; Rattanachaikunsopon and Phumkhachorn, 2010b). Synergistic effects of p-cymene have also been reported in relation to thymol and γ-terpinene (Dauqan and Abdullah, 2017).
Synergistic interactions of carvacrol, eucalyptol (1,8-cineole) and β-caryophyllene are presented in Table 5. Synergy was also reported between carvacrol and other minor constituents, such as, p-cymene, carvacrol and thymol. Eucalyptol also appeared to have synergistic effects with other constituent, including camphor, terpinene-4-ol and caryophyllene oxide. Additionally, synergy was also reported between minor constituents, such as between camphor and p-cymene.
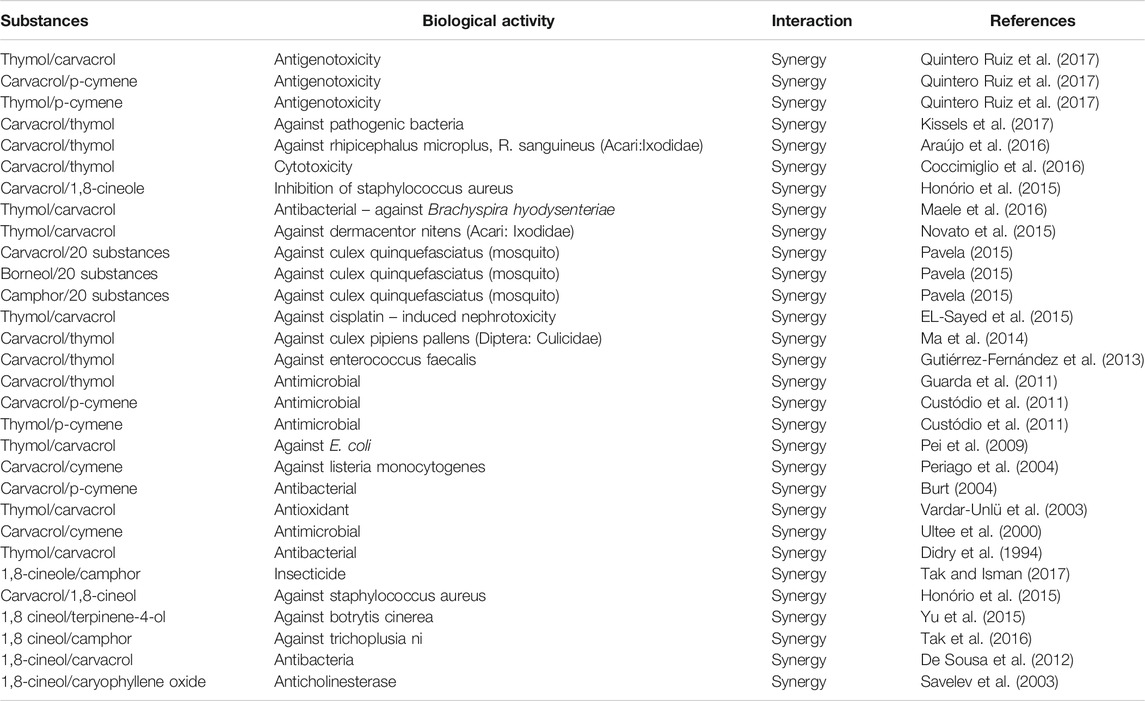
TABLE 5. Synergistic interactions of carvacrol, eucalyptol and β-Caryophyllene. Synergistic interactions of minor constituents of the essential oil reported in the same articles are also reported.
Safety Issues
Carvacrol, eucalyptol and β-caryophyllene, p-cymene, γ-terpinene, borneol and α-terpineol have been approved by the Food and Drug Administration (FDA) for food use (Code for Federal Reguation: 21CFR172.515). Moreover, the European Commission Implementing Regulation (EU No 872/2012) of October 1, 2012, based on the evaluations of EFSA, included carvacrol, eucalyptol and β-caryophyllene, in the Union’s List of Flavorings and Source Materials (Table 6). Their use is therefore permitted, in accordance with good agricultural and manufacturing practices and without given specific restrictions. This EU list takes also into consideration the reports of the Chemical Abstracts Service (CAS), the Joint FAO/WHO Expert Committee on Food additives (JECFA) and the Council of Europe. The rest of the constituents included in the essential oil’s composition of the mixture of Coridothymus capitatus, Salvia fruticosa and Origanum dictamnus (Duikler et al., 2015), are also included in the same list, concerning Food supplements among others, as defined in Directive 2002/46/EC of the European Parliament and the Council, excluding food supplements for infants and young children.
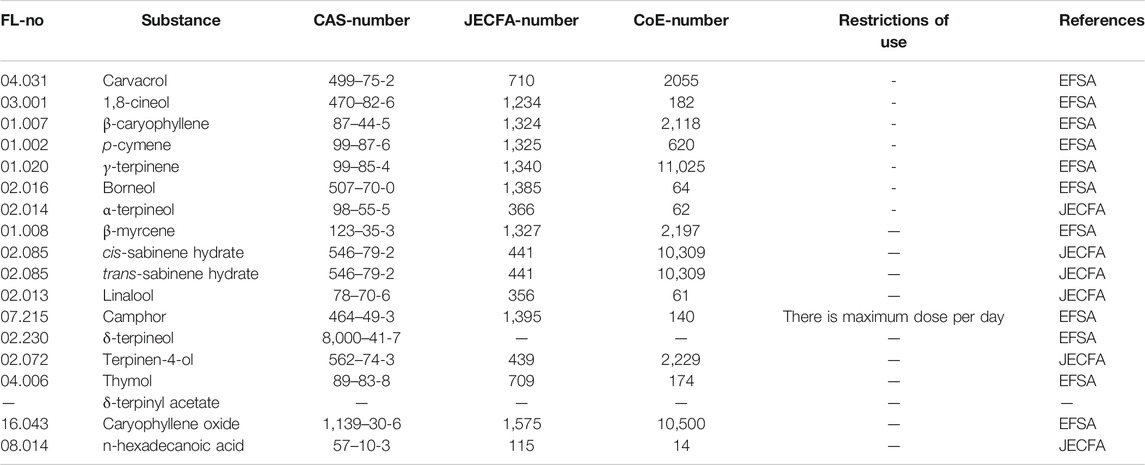
TABLE 6. Chemical constituents of herb essential oil preparations, which are included in the Union List of Flavourings and Source Materials (Commission Implementing Regulation (EU) No 872/2012 of October 1, 2012) based on EFSA and/or JECFA evaluations.
However, in addition to these beneficiary compounds, the extract of the three aromatic plants includes also 0.74 and 0.52% of cis- and trans-thujone respectively (Duijker et al., 2015). Alpha and beta thujone are, according to the Regulation EC No 1334/2008 of the European Parliament and of the Council of December 15, 2008, among the substances which shall not be added as such, to food or food supplements. Maximum concentrations of thujone, naturally present in flavorings and food ingredients with flavoring properties, have been introduced. According to Regulation EC No 1334/2008, “The maximum levels shall not apply where a compound food contains no added flavourings and the only food ingredients with flavoring properties which have been added are fresh, dried or frozen herbs and spices. After consultation with the Member States and the Authority, based on data made available by the Member States and on the newest scientific information, and taking into account the use of herbs and spices and natural flavoring preparations, the Commission, if appropriate, proposes amendments to this derogation.” EMA/HMPC (2010) reported that exposures in the range between 3 and 7 mg/day do not pose special concerns. For higher concentrations, a case-by-case benefit/risk assessment is necessary (Lachenmeier and Uebelacker, 2010; Pelkonen et al., 2013; Sotiropoulou et al., 2016). Finally, Dettling et al. (2004) showed that a single dose of 0.28 mg/kg in men (20 mg/70 kg) and of 0.24 mg/kg (17 mg/70 kg) in women provided “borderline relevance” of adverse effects, mainly related to perturbations in driving, operating machinery, etc.
A Case Study of the Use of Plant Extracts as a Pharmaceutical Product: Lessons Learned
The idea of using herb extracts for the treatment of upper respiratory infections was raised several years ago, when epidemiological observations revealed that people who consumed certain herbs infusions had low rates of respiratory infections and rarely suffered from common colds or influenza infections. The efficacy of the selected herbs was attributed to their antioxidant properties (Lionis et al., 1998). After years of in vitro and in vivo research, an essential oil combination was developed, based on the extracts of the three plants discussed in the present review (Coridοthymus capitatus, Salvia fruticosa and Origanum dictamnus) in extra-virgin olive oil (Duijker et al., 2015); we showed that this combination exerted a synergistic effect against viral upper respiratory tract infections, including influenza (Anastasaki et al., 2017). Moreover, an in vitro study revealed that this combination exhibited a remarkable direct antiviral activity against influenza A/H1N1 viral strains, influenza B and human rhinovirus 14 (HRV14), related to a defective trafficking of influenza A Nucleoprotein (NP) (Tseliou et al., 2019). In this respect, the “one drug, one target, one disease” approach (Zhou et al., 2016) was “violated.” Indeed, the combinatorial use of herbal preparations resulted in an synergistic effect, beyond the reported properties of each plant. Hence, here we address the case of the development and commercialization of a product, containing this essential oils’ combination.
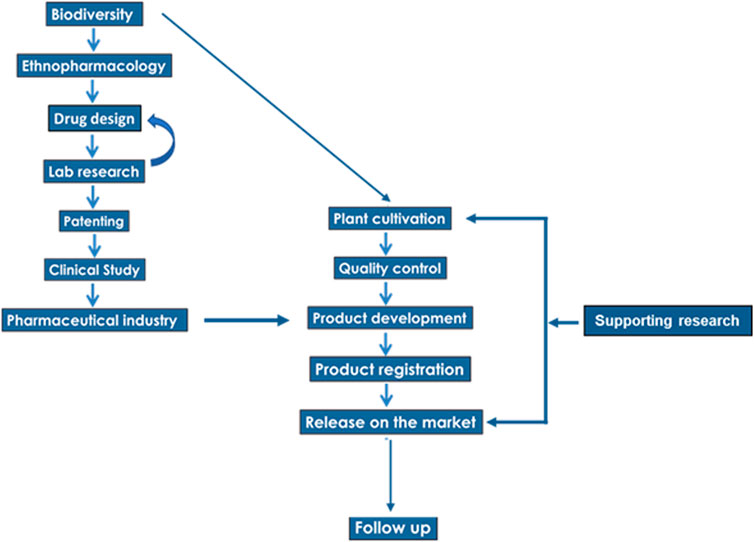
The scheme of the translational chain, from the step of biodiversity to the development of a commercial product, is presented in Figure 3. The first edge of the chain, leading from a plant extract to a final product, relies on the choice of plants, as well as the choice of secondary metabolites, whose biological activity is expected to ensure the desired health benefit (drug design). For example, the preparation containing the essential oils of Thymbra capitata (L.) Cav., Salvia fruticosa Mill. and Origanum dictamnus L. avoided plant extracts rich in alkaloids, as preliminary (Lionis et al., 1988) and clinical (Duijker et al., 2015) or laboratory evidence (Tseliou et al., 2019) documented a potent health benefit of the preparation.
Another part of the continuum is related to the implementation of a clinical study, which is one of the main obstacles in the translational process. In the presented case, it was valuable not only for the demonstration of an antiviral action, but also for the evaluation of the amelioration of the symptoms of the disease, due to supplementary actions of the substances involved in the combination of essential oils.
The supply of raw material is an important part of the translational chain. It is well known that several restrictions rule the natural collections (harvests) and trade of herbs and spices, especially within the framework of the EU environmental policy, as well as within the framework of the United Nations Convention on Biological Diversity. For example, 232 medicinal and aromatic plant species are listed on Appendix II of CITES, which regulates the international trade in endangered species. Worldwide, the increased needs for medicinal plants, in combination with the strengthening of the regulatory framework for their collection and trade, have caused supply constraints of medicinal plants of high ethnopharmacological interest, which introduced a serious constrain in the development and commercialization of plant-derived products (Amirkia and Heinrich, 2014). In the presented case, the plants are cultivated under controlled and monitored conditions, while local farmers have an important income from this agricultural activity. This activity constitutes a viable developmental axis for the local communities.
Recently, a new tool based on Ecological Niche Modeling has been developed, in order to support local farmers in the decision-making process, concerning the suitability of the area where their land is located, for cultivation of medicinal taxa of high ethnopharmacological interest (Bariotakis et al., 2019). A web-based, easy-to-use application was created in the framework of precision agriculture, where the predicted suitability scores for each area of interest can be made accessible to anyone, by the use of its GPS coordinates. So, in our case, the raw material is produced from organic farming, with Global GAP and precision agriculture.
Finally, it should be clear that the continuum in plant-based drug development, is not terminated at the step of release on the market. A follow-up of the final product, after its market release and additional supporting research is necessary. In the example case presented here, a follow up was made through a pragmatic prospective observational study (Anastasaki et al., 2017). In our opinion, this “supporting research” step is necessary as, in many cases, there are further research queries which need clarification even after the pharmaceutical evidence of an essential oil combination. In the example case, the effectiveness of the combination of essential oils in humans was documented (Duijker et al., 2015), but the mode of biological action was not understood. Limited in vitro data was available with ongoing research (Tseliou et al., 2019), providing a possible mechanism, at a cellular level.
In summary, two main lessons emerged from the development of a new pharmaceutical product: The first is resumed in the words “mind the gap,” a well-known phrase from the London underground, as it reveals the necessity of the bridging between successional steps, or successional links of the translational chain. The second concerns the time lag in bridging some successional steps. Indeed, in practice, many subsequent successive steps are fulfilled before others, imposing several loops in the development of the final preparation, which should be treated and resolved accordingly.
However, in spite of the successful use of the three plants, further research is required, in order to decipher additional multi-target action(s), in view of supplementary beneficial effects, which need to be investigated/screened not only in vitro but also in preclinical and clinical studies in the context of Evidence Based Medicine (Stavrou et al., 2013). The results of this screening are expected to clarify whether the ethnopharmacological gap, between reported traditional uses in ethnobotanical studies and the tested properties, is due to a noise of data collection in ethnobotanical practice or reflects underline biological activities, which should be incorporated in the formal therapeutic practice. Moreover, there are no preclinical or clinical evidence about the exposure of sensitive groups (i.e., pregnant women, children, etc.).
A logical subsequent step might be the development a drug, instead of a dietary supplement. A number of items are required for this shift, especially: 1) the repetition of phase I/II trial, with a greater number of participants, together with a detailed pharmacokinetic study of the major active compounds; 2) the performance of a phased III trial. Phase III studies, undertaken in large numbers of patients, often in multiple centers, assess real outcomes in a variety of patients, approximating the global population of patients, who will receive the drug. Their aim is to compare new treatments with existing ones and to demonstrate long-term safety and tolerance (Hobbs and McCarthy, 2009).
Finally, taking into consideration reported variability of the plants’ chemical fingerprint, at least in two of the three native Cretan herbs, which have been used for the development of the active extract (see Karousou et al., 2000; Karousou et al., 2005), it becomes clear that the biological variability in nature does not conform to the requirements for stability in the composition required by market regulations. Thus, further study is suggested concerning the interaction of environmental, chemical, genetic and epigenetic factors, for the quality assurance process. Last but not least, further studies on the cultivation and storage of the above species are required for the standardization and quality control measures, along the whole supply chain.
Author Contributions
EC, SP, CL contributed conception and design of the study, SP and EC wrote the first draft of the manuscript, CL, GS, MK and MB wrote sections of the manuscript. All authors contributed to manuscript revision, read and approved the submitted version.
Conflict of Interest
SP, CL, and EC are inventors of a patent (WO2010GB01836 20,100,929) on the use of the three examined plants for combating upper respiratory tract infections.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be constructed as a potential conflict of interest.
Funding
This work was partially supported by a grant from OLVOS Pharmaceuticals. During the peer review of this manuscript, an additional study of our group was published (Kalyvianaki K, Malamos P, Mastrodimou N, Manoura-Zonou I, Vamvoukaki R, Notas G, Malliaraki N, Moustou E, Tzardi M, Pirintsos S, Lionis C, Sourvinos G, Castanas E and Kampa M. Toxicity evaluation of an essential oil mixture from the Cretan herbs Thyme, greek sage and cretan ddittany, npj Science of Food (2020) 4:20; doi:10.1038/s41538-020-00080-1, suggesting the absence of accute or sub-chronic toxicity of the three herb preparation.
Acknowledgments
We are grateful to Michael Heinrich for his valuable comments on an earlier version of this manuscript.
References
Aburjai, T., Hudaib, M., Tayyem, R., Yousef, M., and Qishawi, M. (2007). Ethnopharmacological survey of medicinal herbs in Jordan, the Ajloun Heights region. J. Ethnopharmacol. 110, 294–304. doi:10.1016/j.jep.2006.09.031. |
Al-Khalil, S. (1995). A survey of plants used in Jordanian traditional medicine. Int. J. Pharmacogn. 33, 317–323. doi:10.3109/13880209509065385. |
Al-Qura’n, S. (2009). Ethnopharmacological survey of wild medicinal plants in Showbak, Jordan. J. Ethnopharmacol. 123, 45–50.
Ali-Shtayeh, M. S., Yaghmour, R. M.-R., Faidi, Y. R., Salem, K., and Al-Nuri, M. A. (1998). Antimicrobial activity of 20 plants used in folkloric medicine in the Palestinian area. J. Ethnopharmacol. 60, 265–271. doi:10.1016/s0378-8741(97)00153-0. |
Ali-Shtayeh, M. S., Yaniv, Z., and Mahajna, J. (2000). Ethnobotanical survey in the Palestinian area: a classification of the healing potential of medicinal plants. J. Ethnopharmacol. 73, 221–232. doi:10.1016/s0378-8741(00)00316-0. |
Allahghadri, T., Rasooli, I., Owlia, P., Nadooshan, M. J., Ghazanfari, T., Taghizadeh, M., et al. (2010). Antimicrobial property, antioxidant capacity, and cytotoxicity of essential oil from cumin produced in Iran. J. Food Sci. 75, H54–H61. doi:10.1111/j.1750-3841.2009.01467.x. |
Altmann, R. E., Campbell, E. L., and Johnston, G. A. R. (2005). (+)- and (−)-borneol: efficacious positive modulators of GABA action at human recombinant α1β2γ2L GABAA receptors. Biochem. Pharmacol. 69, 1101–1111. doi:10.1016/j.bcp.2005.01.002. |
Alzweiri, M., Sarhan, A. A., Mansi, K., Hudaib, M., and Aburjai, T. (2011). Ethnopharmacological survey of medicinal herbs in Jordan, the Northern Badia region. J. Ethnopharmacol. 137, 27–35. doi:10.1016/j.jep.2011.02.007. |
Amirkia, V., and Heinrich, M. (2014). Alkaloids as drug leads — a predictive structural and biodiversity-based analysis. Phytochem. Lett. 10, xlviii–liii. doi:10.1016/j.phytol.2014.06.015
Anastasaki, M., Bertsias, A., Pirintsos, S. A., Castanas, E., and Lionis, C. (2017). Post-market outcome of an extract of traditional Cretan herbs on upper respiratory tract infections: a pragmatic, prospective observational study. BMC Compl. Alternative Med. 17, 466. doi:10.1186/s12906-017-1978-7. |
Andrade e Silva, H. G. (2012). Drug synergy-mechanisms and methods of analysis, toxicity and drug testing. Rijeka: InTech, The German University of Cairo.
Araújo, L. X., Novato, T. P. L., Zeringota, V., Maturano, R., Melo, D., Da Silva, B. C., et al. (2016). Synergism of thymol, carvacrol and eugenol in larvae of the cattle tick, Rhipicephalus microplus, and brown dog tick, Rhipicephalus sanguineus. Med. Vet. Entomol. 30, 377–382. doi:10.1111/mve.12181. |
Ay, G., Bertsias, A., Symvoulakis, E. K., Moschandreas, J., Malliaraki, N., Derdas, S. P., et al. (2015). Reporting effectiveness of an extract of three traditional Cretan herbs on upper respiratory tract infection: results from a double-blind randomized controlled trial. J. Ethnopharmacol. 163, 157–166. doi:10.1016/j.jep.2015.01.030. |
Bariotakis, M., Georgescu, L., Laina, D., Oikonomou, I., Ntagounakis, G., Koufaki, M.-I., et al. (2019). From wild harvest towards precision agriculture: use of ecological Niche modelling to direct potential cultivation of wild medicinal plants in Crete. Sci. Total Environ. 694, 133681. doi:10.1016/j.scitotenv.2019.133681. |
Ben Arfa, A., Combes, S., Preziosi-Belloy, L., Gontard, N., and Chalier, P. (2006). Antimicrobial activity of carvacrol related to its chemical structure. Lett. Appl. Microbiol. 43, 149–154. doi:10.1111/j.1472-765x.2006.01938.x. |
Bernardes, W. A., Lucarini, R., Tozatti, M. G., Bocalon Flauzino, L. G., Souza, M. G. M., Turatti, I. C. C., et al. (2010). Antibacterial activity of the essential oil from Rosmarinus officinalis and its major components against oral pathogens. Z. Naturforsch. C Biosci. 65, 588–593. doi:10.1515/znc-2010-9-1009. |
Burt, S. (2004). Essential oils: their antibacterial properties and potential applications in foods-a review. Int. J. Food Microbiol. 94, 223–253. doi:10.1016/j.ijfoodmicro.2004.03.022. |
Carazza, J., Ribeiro, M. V., Silva, F., Rodrigues Machado, S. M., and Sousa, M. (2011). The essential oils component p-cymene induces proton leak through Fo-ATP synthase and uncoupling of mitochondrial respiration. JEP (J. Environ. Psychol.) 3, 69–76. doi:10.2147/jep.s16387. |
Chebib, M., and Johnston, G. A. R. (2000). GABA-activated ligand gated ion channels: medicinal chemistry and molecular biology. J. Med. Chem. 43, 1427–1447. doi:10.1021/jm9904349. |
Chen, C., Yang, F.-Q., Zhang, Q., Wang, F.-Q., Hu, Y.-J., and Xia, Z.-N. (2015). Natural products for antithrombosis, Evid. base Compl. Alternative Med., 2015, 1, doi:10.1155/2015/876426. |
Coccimiglio, J., Alipour, M., Jiang, Z. H., Gottardo, C., and Suntres, Z. (2016). Antioxidant, antibacterial, and cytotoxic activities of the ethanolic Origanum vulgare extract and its major constituents. Oxid Med Cell Longev 2016. doi:10.1155/2016/1404505. |
Corrêa, P. R. C., Miranda, R. R. S., Duarte, L. P., Silva, G. D. F., Filho, S. A. V., Okuma, A. A., et al. (2012). Antimicrobial activity of synthetic bornyl benzoates againstTrypanosoma cruzi. Pathog. Glob. Health 106, 107–112.doi:10.1179/2047773212y.0000000002. |
Daemon, M., Verge, S., Dumas, M., Soulet, S., Nay, B., Arnaudinaud, V., et al. (2002). Dietary antioxidants, peroxidation and cardiovascular risks. J. Nutr. Health Aging 6, 209–223.
Dafni, A., Yaniv, Z., and Palevitch, D. (1984). Ethnobotanical survey of medicinal plants in northern Israel. J. Ethnopharmacol. 10, 295–310. doi:10.1016/0378-8741(84)90017-5. |
Dauqan, E. M., and Abdullah, A. (2017). Medicinal and functional values of thyme (Thymus vulgaris L.) herb. J. Appl. Biol. Biotechnol. 5, 017–022.
de Sousa, J. P., de Azerêdo, G. A., de Araújo Torres, R., da Silva Vasconcelos, M. A., da Conceição, M. L., and de Souza, E. L. (2012). Synergies of carvacrol and 1,8-cineole to inhibit bacteria associated with minimally processed vegetables. Int. J. Food Microbiol. 154, 145–151. doi:10.1016/j.ijfoodmicro.2011.12.026. |
Dettling, A., Grass, H., Schuff, A., Skopp, G., Strohbeck-Kuehner, P., and Haffner, H. T. (2004). Absinthe: attention performance and mood under the influence of thujone. J. Stud. Alcohol 65, 573–581. doi:10.15288/jsa.2004.65.573. |
Didry, N., Dubreuil, L., and Pinkas, M. (1994). Activity of thymol, carvacrol, cinnamaldehyde and eugenol on oral bacteria. Pharm. Acta Helv. 69, 25–28. doi:10.1016/0031-6865(94)90027-2. |
Dimopoulos, P., Raus, T., Bergmeier, E., Constantinidis, T., Iatrou, G., Kokkini, S., et al. (2013). “An annotated checklist. – Berlin Englera 31,” in Vascular plants of Greece: botanischer Garten und Botanisches Museum Berlin-Dahlem. Athens: Hellenic Botanical Society.
Eissa, T. A. F., Palomino, O. M., Carretero, M. E., and Gómez-Serranillos, M. P. (2014). Ethnopharmacological study of medicinal plants used in the treatment of CNS disorders in Sinai Peninsula, Egypt. J. Ethnopharmacol. 151, 317–332. doi:10.1016/j.jep.2013.10.041. |
El-Sayed, E. M., Abd-Allah, A. R., Mansour, A. M., and El-Arabey, A. A. (2015). Thymol and carvacrol prevent cisplatin-induced nephrotoxicity by abrogation of oxidative stress, inflammation, and apoptosis in rats. J. Biochem. Mol. Toxicol. 29, 165–172. doi:10.1002/jbt.21681. |
EMA/HMPC (2010). Public statement on the use of herbal medicinal products containing thujone. EMA/HMPC/732886/2010.
Euro+Med (2019). Euro+Med PlantBase - the information resource for Euro-Mediterranean plant diversity. Available at: http://ww2.bgbm.org/EuroPlusMed/.Published on the Internet (Accessed June 2017).
Fabricant, D. S., and Farnsworth, N. R. (2001). The value of plants used in traditional medicine for drug discovery. Environ. Health Perspect. 109 (Suppl. 1), 69–75. doi:10.2307/3434847. |
Fielding, J., and Turland, N. (2005). Flowers of Crete. Editor: B. Mathiew (London, England: Kev Royal Botanic Gardens).
Fischer, J., and Dethlefsen, U. (2013). Efficacy of cineole in patients suffering from acute bronchitis: a placebo-controlled double-blind trial. Cough 9, 25. doi:10.1186/1745-9974-9-25. |
García-Beltrán, J. M., and Esteban, M. A. (2016). Properties and applications of plants of Origanum sp. Genus. SM Journal of Biology 2, 1006.
Gertsch, J., Leonti, M., Raduner, S., Racz, I., Chen, J.-Z., Xie, X.-Q., et al. (2008). Beta-caryophyllene is a dietary cannabinoid. Proc. Natl. Acad. Sci. U. S. A. 105, 9099–9104. doi:10.1073/pnas.0803601105. |
Guarda, A., Rubilar, J. F., Miltz, J., and Galotto, M. J. (2011). The antimicrobial activity of microencapsulated thymol and carvacrol. Int. J. Food Microbiol. 146, 144–150. doi:10.1016/j.ijfoodmicro.2011.02.011. |
Guenther, A., Zimmerman, P., and Wildermuth, M. (1994). Natural volatile organic compound emission rate estimates for U.S. woodland landscapes. Atmos. Environ. 28, 1197–1210. doi:10.1016/1352-2310(94)90297-6.
Guimarães, A. G., Quintans, J. S. S., and Quintans-Júnior, L. J. (2013). Monoterpenes with analgesic activity-A systematic review. Phytother Res. 27, 1–15. doi:10.1002/ptr.4686. |
Guindon, J., and Hohmann, A. G. (2008). Cannabinoid CB2 receptors: a therapeutic target for the treatment of inflammatory and neuropathic pain. Br. J. Pharmacol. 153, 319–334. doi:10.1038/sj.bjp.0707531. |
Gürdal, B., and Kültür, Ş. (2013). An ethnobotanical study of medicinal plants in Marmaris (Muğla, Turkey). J. Ethnopharmacol. 146, 113–126.doi:10.1016/j.jep.2012.12.012.
Gutiérrez-Fernández, J., García-Armesto, M. R., Álvarez-Alonso, R., Del Valle, P., de Arriaga, D., and Rúa, J. (2013). Antimicrobial activity of binary combinations of natural and synthetic phenolic antioxidants against Enterococcus faecalis. J. Dairy Sci. 96, 4912–4920. doi:10.3168/jds.2013-6643. |
Hanlidou, E., Karousou, R., Kleftoyanni, V., and Kokkini, S. (2004). The herbal market of Thessaloniki (N Greece) and its relation to the ethnobotanical tradition. J. Ethnopharmacol. 91, 281–299. doi:10.1016/j.jep.2004.01.007. |
Hardy, K. (2019). Paleomedicine and the use of plant secondary compounds in the Paleolithic and Early Neolithic. Evol. Anthropol. 28, 60–71. doi:10.1002/evan.21763. |
Hassan, S. B., Gali-Muhtasib, H., Göransson, H., and Larsson, R. (2010). Alpha terpineol: a potential anticancer agent which acts through suppressing NF-kappaB signalling. Anticancer Res. 30, 1911–1919.
Held, S., Schieberle, P., and Somoza, V. (2007). Characterization of α-terpineol as an anti-inflammatory component of orange juice by in Vitro studies using oral buccal cells. J. Agric. Food Chem. 55, 8040–8046. doi:10.1021/jf071691m. |
Hobbs, M., and McCarthy, M. W. (2009). “Clinical trials,” in Oxford American handbook of clinical pharmacy. Editors M. W. McCarthy, and D. R. Kockler, (New York: Oxford University Press), 115–126.
Honda, G., Yeşilada, E., Tabata, M., Sezik, E., Fujita, T., Takeda, Y., et al. (1996). Traditional medicine in Turkey VI. Folk medicine in west Anatolia: Afyon, Kütahya, Denizli, Muğla, Aydin provinces. J. Ethnopharmacol. 53, 75–87. doi:10.1016/s0378-8741(96)01426-2. |
Hotta, M., Nakata, R., Katsukawa, M., Hori, K., Takahashi, S., and Inoue, H. (2010). Carvacrol, a component of thyme oil, activates PPARα and γ and suppresses COX-2 expression. J. Lipid Res. 51, 132–139. doi:10.1194/jlr.m900255-jlr200. |
Hudaib, M., Mohammad, M., Bustanji, Y., Tayyem, R., Yousef, M., Abuirjeie, M., et al. (2008). Ethnopharmacological survey of medicinal plants in Jordan, Mujib Nature Reserve and surrounding area. J. Ethnopharmacol. 120, 63–71. doi:10.1016/j.jep.2008.07.031. |
Jaradat, N. A., Ayesh, O. I., and Anderson, C. (2016). Ethnopharmacological survey about medicinal plants utilized by herbalists and traditional practitioner healers for treatments of diarrhea in the West Bank/Palestine. J. Ethnopharmacol. 182, 57–66. doi:10.1016/j.jep.2016.02.013. |
Juergens, U. R., Dethlefsen, U., Steinkamp, G., Gillissen, A., Repges, R., and Vetter, H. (2003). Anti-inflammatory activity of 1.8-cineol (eucalyptol) in bronchial asthma: a double-blind placebo-controlled trial. Respir. Med. 97, 250–256. doi:10.1053/rmed.2003.1432. |
Juergens, U. R., Engelen, T., Racké, K., Stöber, M., Gillissen, A., and Vetter, H. (2004). Inhibitory activity of 1,8-cineol (eucalyptol) on cytokine production in cultured human lymphocytes and monocytes. Pulm. Pharmacol. Therapeut. 17, 281–287. doi:10.1016/j.pupt.2004.06.002. |
Jun, Y. S., Kang, P., Min, S. S., Lee, J.-M., Kim, H.-K., and Seol, G. H. (2013). Effect of Eucalyptus oil inhalation on pain and inflammatory responses after total knee replacement: a randomized clinical trial, Evid. base Compl. Alternative Med., 2013, 1, doi:10.1155/2013/502727. |
Karousou, R., and Deirmentzoglou, S. (2011). The herbal market of Cyprus: traditional links and cultural exchanges. J. Ethnopharmacol. 133, 191–203. doi:10.1016/j.jep.2010.09.034. |
Karousou, R., Hanlidou, E., and Kokkini, S. (2000). “The Sage plants of Greece: distribution and infraspecific variation,” inThe Sage. Editor S. E. Kintzios, (Singapore: Harwood Academic Publishers), 27–46.
Karousou, R., Koureas, D. N., and Kokkini, S. (2005). Essential oil composition is related to the natural habitats: Coridothymus capitatus and Satureja thymbra in NATURA 2000 sites of Crete. Phytochemistry 66, 2668–2673. doi:10.1016/j.phytochem.2005.09.020. |
Karousou, R., Vokou, D., and Kokkini, S. (1998). Variation ofSalvia fruticosaEssential oils on the island of Crete (Greece). Plant Biol. 111, 250–254. doi:10.1111/j.1438-8677.1998.tb00705.x. |
Kehrl, W., Sonnemann, U., and Dethlefsen, U. (2004). Therapy for acute nonpurulent rhinosinusitis with cineole: results of a double-blind, randomized, placebo-controlled trial. Laryngoscope 114, 738–742.doi:10.1097/00005537-200404000-00027. |
Kim, J. M., Marshall, M., Cornell, J. A., Iii, J. F. P., and Wei, C. I. (1995). Antibacterial activity of carvacrol, citral, and geraniol against Salmonella typhimurium in culture medium and on fish cubes. J. Food Sci. 60, 1364–1368. doi:10.1111/j.1365-2621.1995.tb04592.x.
Kim, K.-Y., and Chung, H.-J. (2000). Flavor compounds of pine sprout tea and pine needle tea. J. Agric. Food Chem. 48, 1269–1272.doi:10.1021/jf9900229. |
Kim, K. Y., Seo, H. J., Min, S. S., Park, M., and Seol, G. H. (2014). A randomized clinical trial. The effect of 1.8-cineole inhalation on preoperative anxiety. Evidence-Based Complementary Altern. Med. 2014. doi:10.1155/2014/820126 |
Kinsey, S. G., Long, J. Z., Cravatt, B. F., and Lichtman, A. H. (2010). Fatty acid amide hydrolase and monoacylglycerol lipase inhibitors produce anti-allodynic effects in mice through distinct cannabinoid receptor mechanisms. J. Pain 11, 1420–1428. doi:10.1016/j.jpain.2010.04.001. |
Kirimer, N., Başer, K. H. C., and Tümen, G. (1995). Carvacrol-rich plants in Turkey. Chem. Nat. Compd. 31, 37–41. doi:10.1007/bf01167568.
Kiskó, G., and Roller, S. (2005). Carvacrol and p-cymene inactivate Escherichia coli O157: H7 in apple juice. BMC Microbiol. 5, 36. doi:10.1186/1471-2180-5-36. |
Kissels, W., Wu, X., and Santos, R. R. (2017). Short communication: interaction of the isomers carvacrol and thymol with the antibiotics doxycycline and tilmicosin: in vitro effects against pathogenic bacteria commonly found in the respiratory tract of calves. J. Dairy Sci. 100, 970–974. doi:10.3168/jds.2016-11536. |
Klauke, A.-L., Racz, I., Pradier, B., Markert, A., Zimmer, A. M., Gertsch, J., et al. (2014). The cannabinoid CB2 receptor-selective phytocannabinoid beta-caryophyllene exerts analgesic effects in mouse models of inflammatory and neuropathic pain. Eur. Neuropsychopharmacol 24, 608–620. doi:10.1016/j.euroneuro.2013.10.008. |
Kokkini, S., and Vokou, D. (1989). Carvacrol-rich plants in Greece. Flavour Fragrance J. 4, 1–7.doi:10.1002/ffj.2730040102.
Kotan, R., Kordali, S., and Cakir, A. (2007). Screening of antibacterial activities of twenty-one oxygenated monoterpenes. Z. Naturforsch. C Biosci. 62, 507–513. doi:10.1515/znc-2007-7-808. |
Lachenmeier, D. W., and Uebelacker, M. (2010). Risk assessment of thujone in foods and medicines containing sage and wormwood – evidence for a need of regulatory changes? Regul. Toxicol. Pharmacol. 58, 437–443. doi:10.1016/j.yrtph.2010.08.012. |
Landa, P., Kokoska, L., Pribylova, M., Vanek, T., and Marsik, P. (2009). In vitro anti-inflammatory activity of carvacrol: inhibitory effect on COX-2 catalyzed prostaglandin E2 biosynthesisb. Arch Pharm. Res. (Seoul) 32, 75–78. doi:10.1007/s12272-009-1120-6. |
Lardos, A., and Heinrich, M. (2013). Continuity and change in medicinal plant use: the example of monasteries on Cyprus and historical iatrosophia texts. J. Ethnopharmacol. 150, 202–214. doi:10.1016/j.jep.2013.08.026. |
Lardos, A. (2006). The botanical materia medica of the Iatrosophikon-A collection of prescriptions from a monastery in Cyprus. J. Ethnopharmacol. 104, 387–406. doi:10.1016/j.jep.2005.12.035. |
Lev, E., and Amar, Z. (2000). Ethnopharmacological survey of traditional drugs sold in Israel at the end of the 20th century. J. Ethnopharmacol. 72, 191–205. doi:10.1016/s0378-8741(00)00230-0. |
Lev, E., and Amar, Z. (2002). Ethnopharmacological survey of traditional drugs sold in the Kingdom of Jordan. J. Ethnopharmacol. 82, 131–145. doi:10.1016/s0378-8741(02)00182-4. |
Lev, E. (2002). Reconstructed materia medica of the Medieval and Ottoman al-Sham. J. Ethnopharmacol. 80, 167–179.doi:10.1016/s0378-8741(02)00029-6. |
Li, Y.-H., Sun, X.-P., Zhang, Y.-Q., and Wang, N.-S. (2008). The antithrombotic effect of borneol related to its anticoagulant property. Am. J. Chin. Med. 36, 719–727. doi:10.1142/s0192415x08006181. |
Lima, M. d. S., Quintans-Júnior, L. J., de Santana, W. A., Martins Kaneto, C., Pereira Soares, M. B., and Villarreal, C. F. (2013). Anti-inflammatory effects of carvacrol: evidence for a key role of interleukin-10. Eur. J. Pharmacol. 699, 112–117. doi:10.1016/j.ejphar.2012.11.040. |
Lima‐Accioly, P. M., Lavor‐Porto, P. R., Cavalcante, F. S., Magalhães, P. J. C., Lahlou, S., and Morais, S. M.et al. (2006). Essential oil of Croton nepetaefolius and its main constituent, 1,8‐cineole, block excitability of rat sciatic nerve in vitro. Clin. Exp. Pharmacol. Physiol. 33, 1158--1163. doi:10.1111/j.1440-1681.2006.04494.x
Lionis, C., Faresjö, Å., Skoula, M., Kapsokefalou, M., and Faresjö, T. (1998). Antioxidant effects of herbs in Crete. Lancet 352, 1987–1988. doi:10.1016/s0140-6736(05)61333-5. |
Lücker, J., El Tamer, M. K., Schwab, W., Verstappen, F. W. A., van der Plas, L. H. W., Bouwmeester, H. J., et al. (2002). Monoterpene biosynthesis in lemon (Citrus limon). FEBS J. 269, 3160–3171.doi:10.1046/j.1432-1033.2002.02985.x. |
Ma, W.-B., Feng, J.-T., Jiang, Z.-L., Wu, H., Ma, Z.-Q., and Zhang, X. (2014). Fumigant activity of eleven essential oil compounds and their selected binary mixtures against Culex pipiens pallens (Diptera: Culicidae). Parasitol. Res. 113, 3631–3637. doi:10.1007/s00436-014-4028-0. |
Maele, L. V., Heyndrickx, M., De Pauw, N., Verlinden, M., Haesebrouck, F., Martel, A., et al. (2016). In vitro susceptibility of Brachyspira hyodysenteriae to organic acids and essential oil components. J. Vet. Med. Sci. 78, 325–328.
Manayi, A., Nabavi, S. M., Daglia, M., and Jafari, S. (2016). Natural terpenoids as a promising source for modulation of GABAergic system and treatment of neurological diseases. Pharmacol. Rep. 68, 671–679. doi:10.1016/j.pharep.2016.03.014. |
Martínez-Francés, V., Rivera, D., Heinrich, M., Obón, C., and Ríos, S. (2015). An ethnopharmacological and historical analysis of “Dictamnus”, a European traditional herbal medicine. J. Ethnopharmacol. 175, 390–406. doi:10.1016/j.jep.2015.09.011. |
Mastelic, J., Jerkovic, I., Blazevic, I., Poljak-Blaži, M., Borović, S., Ivančić-Baće, I., et al. (2008). Comparative study on the antioxidant and biological activities of carvacrol, thymol, and eugenol derivatives. J. Agric. Food Chem. 56, 3989–3996.
Milos, M., and Makota, D. (2012). Investigation of antioxidant synergisms and antagonisms among thymol, carvacrol, thymoquinone and p-cymene in a model system using the Briggs-Rauscher oscillating reaction. Food Chem. 131, 296–299. doi:10.1016/j.foodchem.2011.08.042.
Naghibi, F., Mosaddegh, M., Mohammadi Motamed, M., and Ghorbani, A. (2005). Labiatae family in folk medicine in Iran: from ethnobotany to pharmacology. Iran. J. Pharm. Res. (IJPR) 4, 63–79.
Novato, T. P. L., Araújo, L. X., de Monteiro, C. M. O., Maturano, R., Senra, T. d. O. S., da Silva Matos, R., et al. (2015). Evaluation of the combined effect of thymol, carvacrol and ( E )-cinnamaldehyde on Amblyomma sculptum (Acari: ixodidae) and Dermacentor nitens (Acari: ixodidae) larvae. Vet. Parasitol. 212, 331–335. doi:10.1016/j.vetpar.2015.08.021. |
Oliveira, M. G., Marques, R. B., Santana, M. F., Santos, A. B., Brito, F. A., and Barreto, E. O.et al. (2012). α‐Terpineol reduces mechanical hypernociception and inflammatory response. Basic & Clin. Pharm. & Toxicol. 111, 120--125. doi:10.1111/j.1742-7843.2012.00875.x
Palevitch, D., Yaniv, Z., Dafni, A., and Simon, J. E. (1986). “Medicinal plants of Israel: an ethnobotanical survey,” in Herbs, spices and medicinal plants. Editor L. A. Craker, (Phoenix, AZ: Oryx Press), 281–345.
Pavela, R. (2015). Acute toxicity and synergistic and antagonistic effects of the aromatic compounds of some essential oils against Culex quinquefasciatus Say larvae. Parasitol. Res. 114, 3835–3853. doi:10.1007/s00436-015-4614-9. |
Pei, R. S., Zhou, F., Ji, B. P., and Xu, J. (2009). Evaluation of combined antibacterial effects of eugenol, cinnamaldehyde, thymol, and carvacrol against E. coli with an improved method. J. Food Sci. 74, 379–383. doi:10.1111/j.1750-3841.2009.01287.x. |
Pelkonen, O., Abass, K., and Wiesner, J. (2013). Thujone and thujone-containing herbal medicinal and botanical products: toxicological assessment. Regul. Toxicol. Pharmacol. 65, 100–107. doi:10.1016/j.yrtph.2012.11.002. |
Periago, P. M., Delgado, B., Fernández, P. S., and Palop, A. (2004). Use of carvacrol and cymene to control growth and viability of Listeria monocytogenes cells and predictions of survivors using frequency distribution functions. J. Food Protect. 67, 1408–1416. doi:10.4315/0362-028x-67.7.1408. |
Proudfoot, C. J., Garry, E. M., Cottrell, D. F., Rosie, R., Anderson, H., Robertson, D. C., et al. (2006). Analgesia mediated by the TRPM8 cold receptor in chronic neuropathic pain. Curr. Biol. 16, 1591–1605. doi:10.1016/j.cub.2006.07.061. |
Quintans‐Júnior, L. J., Oliveira, M. G. B., Santana, M. F., Santana, M. T., Guimarães, A. G., and Siqueira, J. S.et al. (2011) α‐Terpineol reduces nociceptive behavior in mice. Pharm. Biol. 49, 583--586. doi:10.3109/13880209.2010.529616
Rattanachaikunsopon, P., and Phumkhachorn, P. (2010b). Assessment of synergistic efficacy of carvacrol and cymene against Edwardsiella tarda in vitro and in Tilapia (Oreochromis niloticus). Afr. J. Microbiol. Res. 4, 420–425.
Rattanachaikunsopon, P., and Phumkhachorn, P. (2010a). Assessment of factors influencing antimicrobial activity of carvacrol and cymene against Vibrio cholerae in food. J. Biosci. Bioeng. 110, 614–619. doi:10.1016/j.jbiosc.2010.06.010. |
Ravishankar, S., Zhu, L., Reyna-Granados, J., Law, B., Joens, L., and Friedman, M. (2010). Carvacrol and cinnamaldehyde inactivate antibiotic-resistant Salmonella enterica in buffer and on celery and oysters. J. Food Protect. 73, 234–240. doi:10.4315/0362-028x-73.2.234. |
Ribeiro, T. P., Porto, D. L., Menezes, C. P., Antunes, A. A., Silva, D. F., De Sousa, D. P., et al. (2010). Unravelling the cardiovascular effects induced by alpha-terpineol: a role for the nitric oxide-cGMP pathway. Clin. Exp. Pharmacol. Physiol. 37, 811–816. doi:10.1111/j.1440-1681.2010.05383.x. |
Rivera, D., Obon, C., and Cano, F. (1994). The botany, history and traditional uses of three-lobed sage (Salvia fruticosa miller) (Labiatae). Econ. Bot. 48, 190–195. doi:10.1007/bf02908216.
Said, O., Khalil, K., Fulder, S., and Azaizeh, H. (2002). Ethnopharmacological survey of medicinal herbs in Israel, the Golan heights and the west bank region. J. Ethnopharmacol. 83, 251–265. doi:10.1016/s0378-8741(02)00253-2. |
Santos, F. A., and Rao, V. S. N. (2000). Antiinflammatory and antinociceptive effects of 1,8-cineole a terpenoid oxide present in many plant essential oils. Phytother Res. 14, 240–244. doi:10.1002/1099-1573(200006)14:4<240::aid-ptr573>3.0.co;2-x. |
Sargin, S. A. (2015). Ethnobotanical survey of medicinal plants in Bozyazı district of Mersin, Turkey. J. Ethnopharmacol. 173, 105–126.
Sato, K., Krist, S., and Buchbauer, G. (2007). Antimicrobial effect of vapours of geraniol, (R)-(-)-linalool, terpineol,γ-terpinene and 1,8-cineole on airborne microbes using an airwasher. Flavour Fragrance J. 22, 435–437. doi:10.1002/ffj.1818.
Savelev, S., Okello, E., Perry, N. S. L., Wilkins, R. M., and Perry, E. K. (2003). Synergistic and antagonistic interactions of anticholinesterase terpenoids in Salvia lavandulaefolia essential oil. Pharmacol. Biochem. Behav. 75, 661–668. doi:10.1016/s0091-3057(03)00125-4. |
Sivropoulou, A., Papanikolaou, E., Nikolaou, C., Kokkini, S., Lanaras, T., and Arsenakis, M. (1996). Antimicrobial and cytotoxic activities ofOriganumEssential oils. J. Agric. Food Chem. 44, 1202–1205. doi:10.1021/jf950540t. |
Song, H., Schacher, C., and Dethlefsen, U. (2009). Concomitant therapy with Cineole (Eucalyptole) reduces exacerbations in COPD: a placebo-controlled double-blind trial. Respir. Res. 10, 69. doi:10.1186/1465-9921-10-69. |
Sotiropoulou, Ν.-S., Kokkini, M., P.Megremi, S.-F., Daferera, D., Skotti, E., Kimbaris, A., et al. (2016). Determination of Α- and Β-thujone in wormwood and sage infusions of Greek flora and estimation of their average toxicity. Curr. Res. Nutr. Food Sci. 4, 152–160. doi:10.12944/crnfsj.4.special-issue-october.21.
Souma, H., Sağdiç, O., Özkan, G., and Karadoğan, T. (2004). Antibacterial activity and composition of essential oils from Origanum, Thymbra and Satureja species with commercial importance in Turkey. Food Contr. 15, 169–172. doi:10.1016/s0956-7135(03)00028-8.
Stavrou, A., Challoumas, D., and Dimitrakakis, G. (2013). Archibald Cochrane (1909-1988): the father of evidence-based medicine. Interact. Cardiovasc. Thorac. Surg. 18, 121–124. doi:10.1093/icvts/ivt451. |
Stefanaki, A., Cook, C. M., Lanaras, T., and Kokkini, S. (2016). The Oregano plants of Chios Island (Greece): essential oils of Origanum onites L. growing wild in different habitats. Ind. Crop. Prod. 82, 107–113. doi:10.1016/j.indcrop.2015.11.086.
Suntres, Z. E., Coccimiglio, J., and Alipour, M. (2015). The bioactivity and toxicological actions of carvacrol. Crit. Rev. Food Sci. Nutr. 55, 304–318. doi:10.1080/10408398.2011.653458. |
Tak, J.-H., Jovel, E., and Isman, M. B. (2016). Comparative and synergistic activity of Rosmarinus officinalis L. essential oil constituents against the larvae and an ovarian cell line of the cabbage looper, Trichoplusia ni (Lepidoptera: noctuidae). Pest Manag. Sci. 72, 474–480. doi:10.1002/ps.4010. |
Tak, J. H., and Isman, M. B. (2017). Penetration-enhancement underlies synergy of plant essential oil terpenoids as insecticides in the cabbage looper, Trichoplusia ni. Sci. Rep. 7, 42432. doi:10.1038/srep42432. |
Takaishi, V. G., Bezerra, J., Souza, G. T., Carvalho, R. J., Gomes-Neto, N. J., Figueiredo, R. C., et al. (2015). Inhibition of Staphylococcus aureus cocktail using the synergies of oregano and rosemary essential oils or carvacrol and 1, 8-cineole. Front. Microbiol. 6, 1223. doi:10.3389/fmicb.2015.01223. |
Tang, Y., Garg, H., Geng, Y.-J., and Bryan, N. S. (2009). Nitric oxide bioactivity of traditional Chinese medicines used for cardiovascular indications. Free Radic. Biol. Med. 47, 835–840. doi:10.1016/j.freeradbiomed.2009.06.024. |
Tarantilis, R. H. L., Cardoso, M. S. P., Menezes, C. T., Silva, J. P., De Sousa, D. P., and Batista, J. S. (2011) .Gastroprotective activity of α-terpineol in two experimental models of gastric ulcer in rats. Daru: J. Faculty Phar. 19, 277.
Tesche, S., Metternich, F., Sonnemann, U., Engelke, J.-C., and Dethlefsen, U. (2008). The value of herbal medicines in the treatment of acute non-purulent rhinosinusitis. Eur. Arch. Oto-Rhino-Laryngol. 265, 1355. doi:10.1007/s00405-008-0683-z. |
Tseliou, M., Pirintsos, S. A., Lionis, C., Castanas, E., and Sourvinos, G. (2019). Antiviral effect of an essential oil combination derived from three aromatic plants (Coridothymus capitatus (L.) Rchb. f., Origanum dictamnus L. and Salvia fruticosa Mill.) against viruses causing infections of the upper respiratory tract. J. Herb. Med. 17-18, 100288. doi:10.1016/j.hermed.2019.100288.
Ultee, A., Bennik, M. H. J., and Moezelaar, R. (2002). The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. AEM 68, 1561–1568. doi:10.1128/aem.68.4.1561-1568.2002. |
Ultee, A., Kets, E. P. W., and Smid, E. J. (1999). Mechanisms of action of carvacrol on the food-borne pathogen Bacillus cereus. Appl. Environ. Microbiol. 65, 4606–4610. doi:10.1128/aem.65.10.4606-4610.1999. |
Ultee, A., Slump, R. A., Steging, G., and Smid, E. J. (2000). Antimicrobial activity of carvacrol toward Bacillus cereus on rice. J. Food Protect. 63, 620–624. doi:10.4315/0362-028x-63.5.620. |
Vardar-Ünlü, G., Candan, F., Sökmen, A., Daferera, D., Polissiou, M., Sökmen, M., et al. (2003). Antimicrobial and Antioxidant Activity of the Essential Oil and Methanol Extracts ofThymus pectinatusFisch. et Mey. Var.pectinatus(Lamiaceae). J. Agric. Food Chem. 51, 63–67. doi:10.1021/jf025753e. |
Vartiainen, O. (1950). The anthelmintic effects of thymol and p-cymene; a pharmacological and clinical study, with special consideration of the fish tapeworm disease. Ann. Med. Intern. Fenn. 39, 1--87.
Vázquez, F. M., Suarez, M. A., and Pérez, A. (1997). Medicinal plants used in the Barros area, Badajoz province (Spain). J. Ethnopharmacol. 55, 81–85. doi:10.1016/s0378-8741(96)01491-2. |
Wagner, H., and Wierer, R. (1986). In vitro-Hemmung der Prostaglandin-Biosynthese durch etherische Öle und phenolische Verbindungen. Planta Med. 52, 184–187. doi:10.1055/s-2007-969117.
Wang, S., Zhang, D., Hu, J., Jia, Q., Xu, W., Su, D., et al. (2017). A clinical and mechanistic study of topical borneol‐induced analgesia. EMBO Mol. Med. 9, 802–815. doi:10.15252/emmm.201607300. |
Worth, H., and Dethlefsen, U. (2012). Patients with asthma benefit from concomitant therapy with cineole: a placebo-controlled, double-blind trial. J. Asthma 49, 849–853.doi:10.3109/02770903.2012.717657. |
Xu, H., Delling, M., Jun, J. C., and Clapham, D. E. (2006). Oregano, thyme and clove-derived flavors and skin sensitizers activate specific TRP channels. Nat. Neurosci. 9, 628–635. doi:10.1038/nn1692. |
Yang, Y., Zhang, Z., Li, S., Ye, X., Li, X., and He, K. (2014). Synergy effects of herb extracts: pharmacokinetics and pharmacodynamic basis. Fitoterapia 92, 133–147. doi:10.1016/j.fitote.2013.10.010. |
Yu, D., Wang, J., Shao, X., Xu, F., and Wang, H. (2015). Antifungal modes of action of tea tree oil and its two characteristic components againstBotrytis cinerea. J. Appl. Microbiol. 119, 1253–1262. doi:10.1111/jam.12939. |
Yuan, G., Wahlqvist, M., He, G., Yang, M., and Li, D. (2006). Natural products and anti-inflammatory activity. Asia Pac. J. Clin. Nutr. 15, 143.
Keywords: traditional medicine, Southeastern Europe, Mediterranean, Near East, synergy, regulatory affairs, clinical trials, antiviral
Citation: Pirintsos SA, Bariotakis M, Kampa M, Sourvinos G, Lionis C and Castanas E (2020) The Therapeutic Potential of the Essential Oil of Thymbra capitata (L.) Cav., Origanum dictamnus L. and Salvia fruticosa Mill. And a Case of Plant-Based Pharmaceutical Development. Front. Pharmacol. 11:522213. doi: 10.3389/fphar.2020.522213
Received: 21 December 2019; Accepted: 15 October 2020;
Published: 24 November 2020.
Edited by:
Andrei Mocan, Iuliu Hatieganu University of Medicine and Pharmacy, RomaniaReviewed by:
Uroš M. Gašić, University of Belgrade, SerbiaAdriana Trifan, Grigore T. Popa University of Medicine and Pharmacy, Romania
Copyright © 2020 Pirintsos, Bariotakis, Kampa, Sourvinos, Lionis and Castanas. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Elias Castanas, castanas@uoc.gr; Stergios Pirintsos, pirintsos@uoc.gr
 S. A. Pirintsos1,2*
S. A. Pirintsos1,2*