- 1Institute for Public Health and Medicine, Center for Food Allergy & Asthma, Northwestern University, Chicago, IL, United States
- 2Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
- 3Department of Pediatrics, Johns Hopkins University School of Medicine, Baltimore, MD, United States
Importance: Food allergy can often cause a significant burden on patients, families, and healthcare systems. The complexity of food allergy management requires a multidisciplinary approach involving different types of healthcare providers, including allergists, dieticians, psychologists, nurses, family practitioners and, of particular relevance for this article, pediatric primary caretakers. Pediatricians may be the first-line healthcare providers for food allergy: strategies for management and guideline adherence have been highlighted.
Observations: This review article summarizes the up-to-date recommendations on the role of pediatricians in the diagnosis, management, and prevention of IgE-mediated food allergy. Early introduction of allergenic foods like peanut is known to be of importance to reduce the development of peanut allergy in infants, and pediatricians are essential for educating and supporting parents in this decision. In scenarios of limited allergist availability, as is often the case among rural, Medicaid and minority populations, pediatricians can assist in the evaluation and management of food allergy, and provide action plans, education and counselling for patients and families.
Conclusions and relevance: Pediatric primary caretakers play a key role in the diagnosis, management, and prevention of IgE-mediated food allergy. As more diagnostic tools and therapies in food allergy become available, the need for a multidisciplinary team is paramount to optimize patient care.
1 Introduction
Food allergy, an immune-mediated adverse reaction to food, is a potentially life-threatening condition associated with a significant economic burden and impact on patients' and caregivers' quality of life (1–6). Immune-mediated food allergies include immunoglobulin E (IgE)-mediated food allergies and mixed-IgE and non-IgE-mediated allergy (7). Most food allergy is IgE-mediated; types of non-IgE-mediated allergy include eosinophilic gastrointestinal diseases and food protein-induced enterocolitis syndrome (FPIES) (6, 7). Since food intolerance is also common and can have a similar clinical presentation to food allergy, a key challenge for healthcare professionals is to distinguish immune-mediated food allergy from food intolerance (7).
With prevalence rates on the rise, IgE-mediated food allergy is estimated to affect up to ∼8% of children and 10.8% of adults in the United States (US) (8, 9). Self-reported prevalence estimates of food allergy are even higher, with >11% of children perceived as food-allergic by their caregivers and nearly 19% of adults believing they have a food allergy (8, 9). In patients with IgE-mediated food allergies, approximately 40% of children and 45% of adults are allergic to multiple foods (8, 9). These discrepancies highlight the need for improved diagnosis, confirmatory testing, preventative strategies and early management of food allergy in order to prevent unnecessary food avoidance and impact on quality of life (9).
Patients may receive support from a variety of different healthcare providers in different scenarios in the management of their food allergies, including primary and secondary care providers, family practitioners, and a wider multidisciplinary team. This review will focus on pediatricians, who are often the first-line healthcare providers (depending on the individual healthcare system and the healthcare access of the patient); they play a vital role in ensuring appropriate preventative measures through early introduction and the diagnosis and management of food allergy (10). However, knowledge gaps exist and variations in adherence to guidelines across pediatric clinics have been highlighted (11–13). This article aims to provide a comprehensive review of the diagnosis, management, and prevention of food allergy by pediatricians and the wider multidisciplinary team, with the aim of improving accurate diagnosis and optimizing evidence-based pediatric food allergy patient care.
2 Diagnosis of food allergy
2.1 Medical history, comorbidities, and family history
In the US, practice recommendations for the diagnosis and management of food allergy were established in the 2010 National Institute of Allergy and Infectious Diseases (NIAID) guidelines (5, 6) and are informed by recent guidelines such as practice parameters developed by the American Academy of Allergy, Asthma & Immunology (AAAAI); the American College of Allergy, Asthma & Immunology (ACAAI); and the Joint Council of Allergy, Asthma & Immunology (JCAAI) (14) and the Global Allergy and Asthma European Network (GA2LEN) guideline (15). For patients with suspected food allergy, a thorough medical history is the first step in making an accurate diagnosis. Such a medical history should include documenting all foods eaten prior to the reaction, severity and duration of symptoms, time elapsed between ingestion of the food and the onset of symptoms, and response to medications (5, 6). An IgE-mediated reaction is suspected when the onset of symptoms is less than 2 h after ingestion (10). Skin manifestations and oral symptoms are usually but not always the first to appear in an IgE-mediated reaction (10). The type of allergen should also be considered when making the diagnosis: reactions to peanuts, tree nuts and shellfish are almost always IgE-mediated, whereas other foods such as milk, wheat and soy could be IgE-mediated, non-IgE-mediated, or mixed (7, 14). It is important to differentiate between IgE- and non-IgE-mediated reactions such as FPIES and food protein-induced allergic proctocolitis (FPIAP), for which there are no biomarkers and diagnosis relies upon recognition of symptom constellation (16, 17). Management of these conditions generally involves removal of the trigger food, which typically resolves FPIES and FPIAP (17), and reintroduction at a later stage under physician supervision for FPIES, as per international consensus guidelines (18).
Food allergic children often present with comorbidities such as atopic dermatitis, asthma and/or allergic rhinitis, as these are linked by the “atopic march” (19, 20). The presence of comorbidities may also affect symptom severity and treatment response, with uncontrolled asthma as a risk factor for severe anaphylaxis (6), although fatalities remain rare (approximately 0.03–0.3 deaths per million person years in the general US population) (21). Food allergy testing should not be a standard part of evaluation of children with respiratory allergies or atopic dermatitis, but a subset of patients with severe, treatment-refractory atopic dermatitis may benefit from food allergy testing if there is a suspicion that allergen ingestion is linked to flares (6, 22).
Atopy in parents can increase the odds of atopic conditions in children, particularly atopic dermatitis (23, 24) but also food allergy (25). However, screening of siblings should not be routinely performed as sibling history is not a major risk factor for developing food allergy (22).
Pollen Food Allergy Syndrome (PFAS), sometimes known as oral allergy syndrome, is an IgE- dependent allergic reaction directed toward fruits, vegetables, and nuts linked to allergic sensitization to pollen, the most frequent symptoms of which is contact urticaria of the oropharyngeal sites (26). Other symptoms of PFAS may include tightness in the throat, difficulty swallowing, dysphonia, nausea, nasal itching, and itching of the ear (27). PFAS complicates the diagnosis and management of food allergies by leading to uncertainty around potential severity of future reactions and indications for prescribing epinephrine, as well as the extent of necessary dietary avoidance (28). The main pan-allergens of relevance for PFAS include three protein families: profilins, pathogenesis-related protein type 10 (PR-10), and nonspecific lipid transfer proteins (LTPs) (26). Bet v 1 (birch) is one of the most studied PR10 proteins (27), and sensitization to Bet v 1 has been shown to be cross-reactive to multiple fruits, vegetables, and nuts e.g., Mal d 1 in apple, Api g 1 in celery, Ara h 8 in peanuts, and Cor a 1 in hazelnuts (29).
2.2 Specific IgE and skin-prick testing
Medical history alone is not sufficient to diagnose food allergy, and it should be supplemented by testing including allergen-specific IgE (sIgE) and/or skin prick testing (SPT) (14). Both SPT and sIgE tests are highly sensitive, but non-specific. Therefore, the use of these tests may lead to false positive results (14, 22). These tests should generally only be undertaken when the patient presents with a history of reaction to a given food (14, 22). If a patient has a clear history of eating peanuts and having a reaction consistent with a food allergic reaction (hives, vomiting, trouble breathing, etc) ordering a specific IgE to peanut is the best next step. Do not order testing on any food that is currently tolerated in their diet and do not order panels. If the sIgE is negative it is a good indication that they do not have that IgE-mediated food allergy. If it is positive, it is important to provide counselling, epinephrine autoinjector, and referral to an allergist. A recent expert consensus for the diagnosis of peanut allergy suggested that diagnostic testing for peanut allergy should be utilized in patients with a moderate-to-high pre-test probability of peanut allergy, depending on patient/caregiver preference and prior to oral food challenge (OFC) (22). To avoid misdiagnosis and unnecessary dietary restrictions that can impact nutrition and quality of life, the expert panel discouraged testing in those with a low pre-test probability (22). Food allergen panel testing or the addition of sIgE testing for foods other than peanut were not recommended, due to their poor positive predictive value, which could contribute to misdiagnosis (22).
SPT is widely used by allergists as a means to detect the presence of sIgE bound to mast cells in the skin (10, 30). A positive test is considered as a wheal diameter of ≥3 mm (30), although this is variable depending on the population and food studied (7). Dermatographism, severe atopic dermatitis and use of antihistamine medications are considered contraindications for SPT testing (10). In general, sIgE levels will be most readily available for pediatricians and may be more likely to correlate with current symptoms (14), although it is important to remember that they cannot predict the severity of future reactions which vary among different patient populations (14, 22). Recent data suggest that, although the utilization of confirmatory SPT and sIgE testing is on the rise, over a third of children with convincing food allergy do not receive a physician diagnosis (31).
2.3 Oral food challenge (OFC)
In some cases, an OFC may be needed to definitively diagnose food allergy (22). OFCs are the best available tests to evaluate the presence of allergic reactivity to foods (32). OFCs may be needed if SPT and/or sIgE results do not correlate with reaction history, or to determine if a specific food allergy has been outgrown (32).
Due to the risk of false positive results with SPT and/or sIgE testing, OFCs can play a vital role in minimizing misdiagnosis (33). However, they must be performed by an experienced allergist, require intensive resources and can cause anaphylaxis (33, 34). Factors that can increase the risk of severe reactions during an OFC include type of allergen (peanut, tree nuts, fish, shellfish and milk are more commonly implicated in fatal and near-fatal anaphylaxis), comorbid uncontrolled asthma, delayed use of epinephrine, and upright posture during the assessment (32). The patient should also be in good health on the day of the OFC; it is not recommended to perform the test in conditions of concurrent illness, poorly controlled asthma, atopic dermatitis or allergic rhinitis, unstable cardiovascular disease, pregnancy or beta-blocker therapy (32). As such, the individual benefits and risks to the patient should be evaluated in a shared decision-making process to determine the value of performing an OFC on a case-by-case basis (32). There is a consensus that better diagnostic tests are needed to minimize the need for OFCs (33–35).
2.4 Other diagnostic tests
Component testing (molecular diagnosis) allows the identification of sIgE for specific proteins, or components, which are available for several allergens including peanut, milk, egg, and several tree nuts (7, 36). Ara h 2 is an example of component testing and is approved for the diagnosis of peanut allergy by the FDA (22, 37). Compared with whole peanut SPT and sIgE tests, Ara h 2 testing is cost-effective and has increased specificity for diagnosis (22). It is important to recognize that component testing is not necessarily needed and that the use of “reflex testing” (testing for multiple allergens irrelevant of reaction history), should be strongly discouraged (22).
Novel techniques such as bead-based epitope assays, basophil activation tests and mast cell tests are also being investigated for use in clinical practice and may help to risk stratify patients in the future, as well as to predict response to immunomodulatory therapy (33, 34). Furthermore, progress in emerging component testing such as Bet v 1 testing or Cor a 9 and Cor a 14 tests may enhance the specificity of testing (27, 38).
2.5 Digital technologies
Digital technologies including clinical decision support systems present a promising new avenue for the enhanced detection and management of food allergy in primary care (39). Tools such as the automated allergy management support system (40) and the food allergy support tool (41) were developed to support the detection and management of IgE-mediated food allergy, respectively, in the primary care setting. Further efforts are needed to ensure compatibility with existing software systems and usability in clinical practice in order to facilitate the implementation of such digital support tools in improving patient care (39).
2.6 Emergency department
The incidence of emergency department (ED) visits for food allergy is increasing, and is estimated to be between 3%–4% for adults and ∼8% for children (42). While the confirmatory diagnosis of food allergy falls out of the realm of acute care, patients suspected of having a food allergy can be identified based on patient history and clinical presentation (42). It is essential that these patients are discharged with an epinephrine prescription and appropriate patient counselling regarding self-administration and food avoidance (42). The NIAID guidelines recommend a follow-up appointment with a primary care physician, with consideration for referral to an allergist (6).
2.7 Barriers to diagnosis
A key barrier to food allergy diagnosis is access to an allergist in certain groups, for example in Medicaid and minority populations (43). Furthermore, a 2014 study indicated that pediatrician confidence in ordering sIgE tests is variable, with only 34.7% adhering to guidelines for appropriate use of diagnostic tests (12). Although the rate of referral to an allergist was 67.3%, there was a significant delay to referral in over half of all patients (12). In cases of good allergist availability, it may be possible to directly refer patients to an allergist based on their reaction history, thus avoiding duplication of testing (Figure 1A). However, the role of the pediatrician may need to be expanded in incidences of delayed or limited access to an allergist (Figure 1B).

Figure 1. Diagnosis and management of IgE-mediated food allergy by pediatricians, in scenarios of good allergist availability (A) or delayed/limited allergist availability (B) *A template and guidance for completing the food allergy anaphylaxis plan is available from the American academy of pediatrics. (44).
2.8 When should an allergist be consulted?
Food allergy significantly impairs quality of life for children with food allergy and their caregivers (1) and leads to increased healthcare resource utilization and costs (45) and substantial population-level psychosocial burden (46). Timely referral to an allergist is of vital importance to reduce the effects of delayed diagnosis on patients. Delay in referral to an allergist can contribute to poor patient experience and increased healthcare resource utilization (47) as well as to the impact of food allergies on patients and caregivers (1). Consultation, and regular follow-up with an allergist should be considered in all children with a convincing diagnosis of IgE-mediated food allergy. Consider the following factors, in particular, for expedited referral:
• Medical history is indicative of anaphylaxis (5, 48)
• Specialized testing such as skin testing for certain food allergens is needed and cannot be accessed from primary care (49)
• OFC testing is needed (48)
• Patient requires repeat instructions on how and when to use injectable epinephrine, especially in those patients with limited adherence and risk-taking behavior (5)
• Food allergy is leading to nutritional concerns (7)
• Shared decision-making is needed regarding treatment options (50)
2.9 Follow-up and re-testing
Following the diagnosis of food allergy, yearly follow-up is recommended with an allergist; this may be more frequent in younger patients and those with risk factors such as nutritional concerns, poorly-controlled asthma, risk-taking behavior, frequent accidental ingestions and/or history of severe reactions (6, 7, 14, 37). Frequency of follow-up is also influenced by comorbidities (e.g., asthma, allergic rhinitis, and atopic dermatitis), availability of allergist services and the type/number of food allergies (37).
The interval for periodic re-testing is allergen-dependent (6). Certain allergens (e.g., milk and egg) are outgrown earlier and more frequently than others (e.g., peanut and tree nuts): annual testing is often utilized for the former, with extended intervals of up to 2–3 years for the latter (6). Re-testing may not be necessary in cases of a recent allergic reaction to the food (6). Long-term food allergy management requires shared decision-making based on the type of allergy, allergy testing information, severity of reaction, and patient preferences (50).
3 Management options
3.1 Avoidance
When food allergy is confirmed, the current mainstay of long-term management remains strict food avoidance; however, accidental exposure is common. Data from the Food Allergy Research and Education (FARE) registry demonstrated that on average, nearly two-thirds of adults and nearly half of children experienced 1 or more food-related allergic reactions per year (51). Data suggest that severe reactions and ED visits are more common in racial minorities and patients from lower income households (52).
3.2 Acute treatment: injectable epinephrine
The first-line acute treatment for food-induced anaphylaxis is injectable epinephrine (53). As noted in the NIAID-Sponsored Expert Panel report, all other treatments have a delayed onset of action and repeat epinephrine dosing remains first-line therapy over adjunctive treatments if there is progression of symptoms or if the response to the initial dose of epinephrine is suboptimal (5). Despite its utility in preventing fatal anaphylaxis, there is evidence that epinephrine is still under-prescribed by healthcare providers and under-used by patients (53). Furthermore, hesitancy to use epinephrine when needed among patients and caregivers can also result in undertreatment of allergic reactions (54). A key role of the pediatrician is to effectively counsel patients with regards to when and how to use epinephrine compared with antihistamines (55). Around 2 in 3 children with physician-confirmed food allergy reported a current epinephrine prescription (19). Notably, the likelihood to carry injectable epinephrine is reduced in children from lower income households (52).
Potentially life-threatening reactions can occur anywhere, and since children spend a large proportion of their time at school, it is also crucial to optimize the management of food allergy in the school setting (56–58). Not only do clinicians play a key role in supporting individual patients to manage their food allergy at school, but they have an opportunity through education and advocacy to help optimize school policies and strategies for managing food allergy on a wider scale (56). In particular, the reduced availability of epinephrine in schools of lower socioeconomic status should be addressed (59).
3.3 Oral immunotherapy
Guidelines that include oral immunotherapy for the treatment of food allergy have been provided by the European Academy of Allergy and Clinical Immunology since 2018 (60), and in January 2020, peanut oral immunotherapy (POIT) was approved by the FDA in patients aged 4–17 years (61). POIT is administered by gradually increasing doses of allergen, and was shown to be effective at inducing desensitization (62) and improving quality of life (63). Despite this, uptake of POIT in the community appears to be limited, with one study indicating that only 10% of eligible patients and caregivers chose to pursue therapy, due to time commitment and adverse effects (64). Furthermore, access to POIT is limited by the availability of centers with trained staff, particularly in clinical practice outside of academic settings (65). Of the 78 responders to a 15-question survey of 780 randomly selected members of the American Academy of Allergy, Asthma & Immunology, 50% were offering POIT at their practice (65), while another survey with responses from 129 private practices among members of the OIT Advisors group, found that 97.5% had treated at least one patient (66). Adverse effects with POIT can vary from mild gastrointestinal symptoms and skin rash/itching, to severe reactions such as eosinophilic esophagitis (EoE) and anaphylaxis (62). Shared decision-making must be prioritized in allergist consultations around POIT and decision aids have been developed to support this process (67).
3.4 Omalizumab for the treatment of food allergy
In February, 2024, omalizumab was approved by the FDA for the treatment of food allergy (68). This monoclonal antibody that specifically blocks the binding of IgE to mast cells and basophils has been approved for the treatment of allergic asthma since 2003, as well as for the treatment of chronic urticaria and chronic rhinosinusitis with nasal polyps. Numerous prior studies have suggested that it could be efficacious for the treatment of food allergy, and this impression was confirmed by the OUtMATCH trial, which demonstrated a high degree of efficacy for patients with multiple food allergies (69–77). Omalizumab is currently approved for the prevention of reactions to small, accidental exposures in all patients with food allergy age 1 year and above.
3.5 Investigational treatments
The treatment landscape for food allergy is rapidly evolving; therapeutic strategies under investigation for allergen immunotherapy include subcutaneous immunotherapy (SCIT), sublingual immunotherapy (SLIT), and epicutaneous immunotherapy (EPIT) (78). Several biologics and small molecules in addition to omalizumab, including BTK inhibitor (acalabrutinib and remibrutinib), anti-IL-33 antibody (etokimab), and anti-TSLP antibody (tezepelumab) are being studied to target different pathways in the allergic response (77, 79–81). Clinical trials are also exploring the combination of treatments, for example anti-IgE in combination with OIT, to improve the safety and time to reach maintenance dose (69).
3.6 Early introduction of allergen
The NIAID guidelines for the diagnosis and management of food allergy were updated in 2017 with an addendum recommending early introduction of peanut in at-risk children, following results from the Learning Early About Peanut Allergy (LEAP) clinical trial (82). At the 4- and 6-month visits, it is important to discuss peanut product introduction. New guidelines suggest all infants should be encouraged to start peanut products as soon as they are ready for solids after the introduction of initial foods like fruits or vegetables. If the infant has severe eczema or if the parents are apprehensive, a specific IgE to peanut or referral to an allergist can be obtained. If this is positive, it is critical to get them in to see an allergist immediately. New data show the longer the delay in introducing peanut products past six months in high-risk infants, the more likely they will have developed a peanut allergy. Early introduction of other allergenic foods might also have benefit. For example, there is some evidence to support early introduction of egg, which is associated with a lower risk of egg allergy, depending on the nature and dose of egg protein exposure (83, 84). Other allergenic foods also show promise through the Enquiring About Tolerance (EAT) study (85) and are currently being studied in the ongoing CANDO study (NCT05258656). The US Department of Agriculture and Department of Health and Human Services’ Dietary Guidelines for Americans recommend introducing potentially allergenic foods (such as peanuts, egg, cow milk products, tree nuts, wheat, crustacean shellfish, fish, and soy) when other complementary foods are introduced to an infant's diet at 4–6 months (86).
Early introduction of food is considered a preference-sensitive clinical scenario and care should be taken to help parents consider their options (50). In order to enable timely introduction, there may be a role for SPT or sIgE testing in infants with moderate to severe eczema or other food allergies, or when caregivers are highly anxious about introducing peanut into their child's diet; however, it is imperative that these results are interpreted appropriately since a positive result may not be diagnostic of food (22).
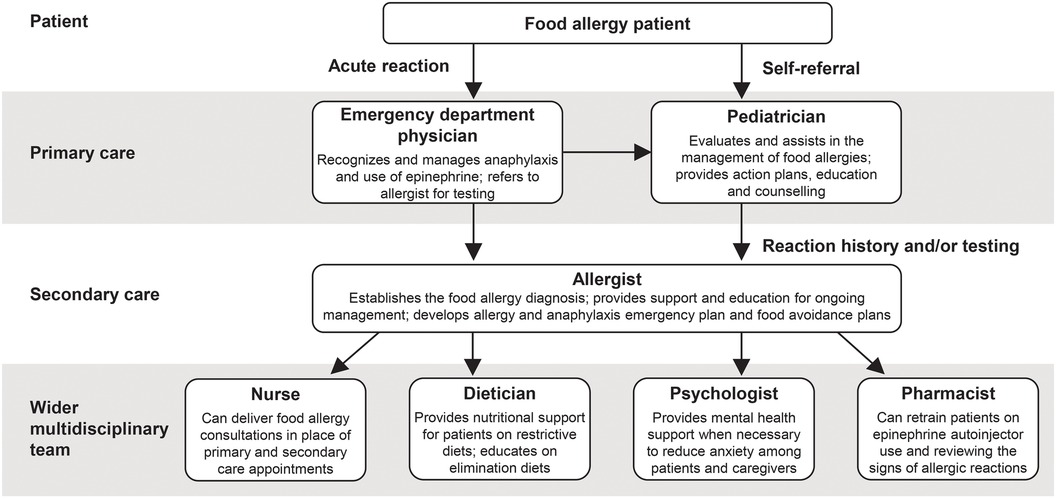
3.7 Multidisciplinary approach
Pediatricians and allergists are central to the effective multidisciplinary management of food allergies. Furthermore, there may be scope for other members of the multidisciplinary team to play a larger role in patient care (Figure 2) to aid pediatricians and allergists in supporting their patients with food allergy treatment:
• A large feasibility trial demonstrated the potential of nurse-led allergy clinics in delivering remote and face-to-face consultations with patients to ease the burden on primary care services (87)
• Dieticians provide crucial nutritional and dietary support to patients on restrictive diets, as well as education on allergen avoidance, and dietician-led services also have potential to reduce primary and secondary care appointments (88)
• Patients with multiple food allergies may benefit most from nutritional counselling (37) and it is recommended by the NIAID for all food allergy patients (6)
• Community pharmacists are able to work collaboratively with pediatricians and allergists to provide ongoing assistance by retraining patients on the use of epinephrine autoinjectors and reviewing the signs of an allergic reaction (89)
• Food allergies can lead to significant psychosocial impact and mental health support may be beneficial to reduce anxiety among patients and caregivers (90)
4 Conclusions
Food allergy leads to significant burden on patients, families, and healthcare systems. Pediatricians play a key role in the prevention, diagnosis, and management of IgE-mediated food allergy, especially in scenarios of limited allergist availability, as is often the case among Medicaid and minority populations. However, allergists should be consulted for all children with food allergy and timely referral is key. As more diagnostic tools and therapies in food allergy become available, the need for a multidisciplinary team is paramount to optimize patient care. A multidisciplinary team comprising nurses, dieticians, and pharmacists, along with paediatricians and allergists, is crucial for accurate diagnosis and appropriate management of food allergy in order to substantially ease the burden on patients, families, and healthcare systems and so that patients can get new treatments as they become available.
4.1 Useful links and resources
• Guidelines for the Diagnosis and Management of Food Allergy in the United States: Summary of the NIAID-sponsored Expert Panel Report (6)
• Addendum Guidelines for the Prevention of Peanut Allergy in the United States: Summary for Clinicians (82)
• iREACH training materials: Early Peanut Product Introduction Tools for Pediatric Clinicians (91)
• AAP Allergy and Anaphylaxis Emergency Plan Template (44)
• EAACI Molecular Allergology User Guide 2.0 (92)
Author contributions
RG: Conceptualization, Methodology, Writing – review & editing. EE: Conceptualization, Methodology, Project administration, Writing – review & editing. RW: Conceptualization, Methodology, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article.
This manuscript was funded by Novartis Pharmaceuticals Corporation.
Acknowledgments
Financial support for medical editorial assistance was provided by Novartis Pharmaceuticals Corporation. We thank Daniela Taylor, Asma S and Sorcha Mc Ginty, PhD, (Novartis Medical and Knowledge Solutions) for their medical editorial assistance with this manuscript.
Conflict of interest
RG declares research support from National Institutes of Health (NIH) (R21 ID # AI135705, R01 ID # AI130348, U01 ID # AI138907); Food Allergy Research & Education (FARE); Novartis; Genentech; Sunshine Charitable Foundation; The Walder Foundation; and the Melchiorre Family Foundation; consulting fees from Novartis; Genentech, Food Allergy Research & Education (FARE), Allergenis LLC; and Aimmune LLC; and ownership interest in Yobee Care, Inc. EE is an employee and stockholder of Novartis Pharmaceuticals Corporation. RW declares grants paid to his institution from NIH; Aimmune; DBV; ALK; Novartis; Genentech; Aravax and Siolta.
The reviewer [RH] declared a shared affiliation with the author [RW] to the handling editor at the time of review.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Warren CM, Otto AK, Walkner MM, Gupta RS. Quality of life among food allergic patients and their caregivers. Curr Allergy Asthma Rep. (2016) 16(5):38. doi: 10.1007/s11882-016-0614-9
2. Warren CM, Jiang J, Gupta RS. Epidemiology and burden of food allergy. Curr Allergy Asthma Rep. (2020) 20(2):6. doi: 10.1007/s11882-020-0898-7
3. Gupta R, Holdford D, Bilaver L, Dyer A, Holl JL, Meltzer D. The economic impact of childhood food allergy in the United States. JAMA Pediatr. (2013) 167(11):1026–31. doi: 10.1001/jamapediatrics.2013.2376
4. Thomsen SF. Epidemiology and natural history of atopic diseases. Eur Clin Respir J. (2015) 2:24642. 2. doi: 10.3402/ecrj.v2.24642
5. Boyce JA, Assa’ad A, Burks AW, Jones SM, Sampson HA, Wood RA, et al. Guidelines for the diagnosis and management of food allergy in the United States: summary of the NIAID-sponsored expert panel report. J Am Acad Dermatol. (2011) 64(1):175–92. doi: 10.1016/j.jaad.2010.11.020
6. National Institute of Allergy and Infectious Diseases (NIAID). Guidelines for the Diagnosis and Management of Food Allergy in the United States. (2010).
7. Gargano D, Appanna R, Santonicola A, De Bartolomeis F, Stellato C, Cianferoni A, et al. Food allergy and intolerance: a narrative review on nutritional concerns. Nutrients. (2021) 13(5):1638. doi: 10.3390/nu13051638
8. Gupta RS, Warren CM, Smith BM, Blumenstock JA, Jiang J, Davis MM, et al. The public health impact of parent-reported childhood food allergies in the United States. Pediatrics. (2018) 142(6):e20181235. doi: 10.1542/peds.2018-1235
9. Gupta RS, Warren CM, Smith BM, Jiang J, Blumenstock JA, Davis MM, et al. Prevalence and severity of food allergies among US adults. JAMA Network Open. (2019) 2(1):e185630. doi: 10.1001/jamanetworkopen.2018.5630
10. Cosme-Blanco W, Arroyo-Flores E, Ale H. Food allergies. Pediatr Rev. (2020) 41(8):403–15. doi: 10.1542/pir.2019-0037
11. Gupta RS, Springston EE, Kim JS, Smith B, Pongracic JA, Wang X, et al. Food allergy knowledge, attitudes, and beliefs of primary care physicians. Pediatrics. (2010) 125(1):126–32. doi: 10.1542/peds.2009-1116
12. Gupta RS, Lau CH, Dyer AA, Sohn MW, Altshuler BA, Kaye BA, et al. Food allergy diagnosis and management practices among pediatricians. Clin Pediatr (Phila). (2014) 53(6):524–30. doi: 10.1177/0009922813518425
13. Krugman SD, Chiaramonte DR, Matsui EC. Diagnosis and management of food-induced anaphylaxis: a national survey of pediatricians. Pediatrics. (2006) 118(3):e554–60. doi: 10.1542/peds.2005-2906
14. Sampson HA, Aceves S, Bock SA, James J, Jones S, Lang D, et al. Food allergy: a practice parameter update—2014. J Allergy Clin Immunol. (2014) 134(5):1016–1025.e43. doi: 10.1016/j.jaci.2014.05.013
15. Muraro A, de Silva D, Halken S, Worm M, Khaleva E, Arasiet S, et al. Managing food allergy: GA(2)LEN guideline 2022. World Allergy Organ J. (2022) 15(9):100687. doi: 10.1016/j.waojou.2022.100687
16. Nowak-Wegrzyn A, Berin MC, Mehr S. Food protein-induced enterocolitis syndrome. J Allergy Clin Immunol Pract. (2020) 8(1):24–35. doi: 10.1016/j.jaip.2019.08.020
17. Cianferoni A. Non-IgE mediated food allergy. Curr Pediatr Rev. (2020) 16(2):95–105. doi: 10.2174/1573396315666191031103714
18. Nowak-Węgrzyn A, Chehade M, Groetch ME, Spergel JM, Wood RA, Allen K, et al. International consensus guidelines for the diagnosis and management of food protein–induced enterocolitis syndrome: executive summary—workgroup report of the adverse reactions to foods committee, American academy of allergy, asthma & immunology. J Allergy Clin Immunol. (2017) 139(4):1111–1126.e4. doi: 10.1016/j.jaci.2016.12.966
19. Sicherer SH, Warren CM, Dant C, Gupta RS, Nadeau KC. Food allergy from infancy through adulthood. J Allergy Clin Immunol Pract. (2020) 8(6):1854–64. doi: 10.1016/j.jaip.2020.02.010
20. Cartledge N, Chan S. Atopic dermatitis and food allergy: a paediatric approach. Curr Pediatr Rev. (2018) 14(3):171–9. doi: 10.2174/1573396314666180613083616
21. Turner PJ, Jerschow E, Umasunthar T, Lin R, Campbell DE, Boyle RJ. Fatal anaphylaxis: mortality rate and risk factors. J Allergy Clin Immunol Pract. (2017) 5(5):1169–78. doi: 10.1016/j.jaip.2017.06.031
22. Greenhawt M, Shaker M, Wang J, Oppenheimer JJ, Sicherer S, Keet C, et al. Peanut allergy diagnosis: a 2020 practice parameter update, systematic review, and GRADE analysis. J Allergy Clin Immunol. (2020) 146(6):1302–34. doi: 10.1016/j.jaci.2020.07.031
23. O’Connor C, Livingstone V, Hourihane JOB, Irvine AD, Boylan G, Murray D. Parental atopy and risk of atopic dermatitis in the first two years of life in the BASELINE birth cohort study. Pediatr Dermatol. (2022) 39(6):896–902. doi: 10.1111/pde.15090
24. Ravn NH, Halling AS, Berkowitz AG, Rinnov MR, Silverberg JI, Egeberg A, et al. How does parental history of atopic disease predict the risk of atopic dermatitis in a child? A systematic review and meta-analysis. J Allergy Clin Immunol. (2020) 145(4):1182–93. doi: 10.1016/j.jaci.2019.12.899
25. Zaslavsky JM, Samady W, Jiang J, Warren CM, Smith BM, Gupta RS. Family history of atopy in food allergy development. J Allergy Clin Immunol. (2019) 143(2, Supplement):AB83. doi: 10.1016/j.jaci.2018.12.258
26. Poncet P, Sénéchal H, Charpin D. Update on pollen-food allergy syndrome. Expert Rev Clin Immunol. (2020) 16(6):561–78. doi: 10.1080/1744666X.2020.1774366
27. Carlson G, Coop C. Pollen food allergy syndrome (PFAS): a review of current available literature. Ann Allergy Asthma Immunol. (2019) 123(4):359–65. doi: 10.1016/j.anai.2019.07.022
28. Giovannini M, Skypala IJ, Caubet JC, Du Toit G, Nowak-Wegrzyn A. Diagnosis and management of pollen food allergy syndrome to nuts. J Allergy Clin Immunol. (2024) 12(3):599–604. doi: 10.1016/j.jaip.2024.01.025
29. Mothes N, Horak F, Valenta R. Transition from a botanical to a molecular classification in tree pollen allergy: implications for diagnosis and therapy. Int Arch Allergy Appl Immunol. (2004) 135(4):357–73. doi: 10.1159/000082332
30. Ansotegui IJ, Melioli G, Canonica GW, Caraballo L, Villa E, Ebisawa M, et al. IgE allergy diagnostics and other relevant tests in allergy, a world allergy organization position paper. World Allergy Organ J. (2020) 13(2):100080. doi: 10.1016/j.waojou.2019.100080
31. Galic I, Warren CM, Jiang J, Tobin MC, Gupta R. Parent report of physician diagnosis in pediatric food allergy: an update. J Allergy Clin Immunol Pract. (2021) 9(1):542–546.e2. doi: 10.1016/j.jaip.2020.09.033
32. Bird JA, Leonard S, Groetch M, Assa’ad A, Cianferoni A, Clark A, et al. Conducting an oral food challenge: an update to the 2009 adverse reactions to foods committee work group report. J Allergy Clin Immunol Pract. (2020) 8(1):75–90.e17. doi: 10.1016/j.jaip.2019.09.029
33. Santos AF, Kulis MD, Sampson HA. Bringing the next generation of food allergy diagnostics into the clinic. J Allergy Clin Immunol Pract. (2022) 10(1):1–9. doi: 10.1016/j.jaip.2021.09.009
34. Alpan O, Wasserman RL, Kim T, Darter A, Shah A, Jones D, et al. Towards an FDA-cleared basophil activation test. Front Allergy. (2022) 3:1009437. doi: 10.3389/falgy.2022.1009437
35. Calvani M, Bianchi A, Reginelli C, Peresso M, Testa A. Oral food challenge. Medicina (Kaunas). (2019) 55(10):651. doi: 10.3390/medicina55100651
36. Foong R-X, Dantzer JA, Wood RA, Santos AF. Improving diagnostic accuracy in food allergy. J Allergy Clin Immunol. (2021) 9(1):71–80. doi: 10.1016/j.jaip.2020.09.037
37. Lloyd M, Loke P, Mack DP, Sicherer SH, Perkin MR, Boyle R, et al. Varying approaches to management of IgE-mediated food allergy in children around the world. J Allergy Clin Immuno. (2023) 11(4):1010–27.e6. doi: 10.1016/j.jaip.2023.01.049
38. Uotila R, Röntynen P, Pelkonen AS, Voutilainen H, Kaarina Kukkonen A, et al. For hazelnut allergy, component testing of Cor a 9 and Cor a 14 is relevant also in birch-endemic areas. Allergy. (2020) 75(11):2977–80. doi: 10.1111/all.14430
39. Dramburg S, Marchante Fernández M, Potapova E, Matricardi PM. The potential of clinical decision support systems for prevention, diagnosis, and monitoring of allergic diseases. Front Immunol. (2020) 11:2116. doi: 10.3389/fimmu.2020.02116
40. Flokstra-de Blok BMJ, Brakel TM, Wubs M, Skidmore B, Kocks JWH, Oude Elberink JNG, et al. The feasibility of an allergy management support system (AMSS) for IgE-mediated allergy in primary care. Clin Transl Allergy. (2018) 8:18. doi: 10.1186/s13601-018-0206-y
41. Otto AK, Dyer AA, Warren CM, Walkner M, Smith BM, Gupta RS. The development of a clinical decision support system for the management of pediatric food allergy. Clin Pediatr (Phila). (2017) 56(6):571–8. doi: 10.1177/0009922816669097
42. Krajewski GS, Krajewski T. Evaluation and management of food allergies in the emergency department. Emerg Med Clin North Am. (2022) 40(1):57–67. doi: 10.1016/j.emc.2021.08.009
43. Dupuis R, Phipatanakul W, Bartnikas LM. Social disparities in early childhood prevention and management of food allergy. J Allergy Clin Immunol. (2023) 151(1):37–41. doi: 10.1016/j.jaci.2022.10.015
44. Wang J, Sicherer SH, SECTION ON ALLERGY AND IMMUNOLOGY , Matsui E, Abramson S, Dinakar C, et al. Guidance on completing a written allergy and anaphylaxis emergency plan. Pediatrics. (2017) 139(3):e20164005. doi: 10.1542/peds.2016-4005
45. Shaker M, Chalil JM, Tran O, Vlahiotis A, Shah H, King T, et al. Commercial claims costs related to health care resource use associated with a diagnosis of peanut allergy. Ann Allergy Asthma Immunol. (2020) 124(4):357–365.e1. doi: 10.1016/j.anai.2020.01.004
46. Warren C, Dyer A, Lombard L, Dunn-Galvin A, Gupta R. The psychosocial burden of food allergy among adults: a US population-based study. J Allergy Clin Immunol Pract. (2021) 9(6):2452–2460.e3. doi: 10.1016/j.jaip.2021.02.039
47. Diwakar L, Cummins C, Lilford R, Roberts T. Systematic review of pathways for the delivery of allergy services. BMJ Open. (2017) 7(2):e012647. doi: 10.1136/bmjopen-2016-012647
48. Kurowski K, Boxer RW. Food allergies: detection and management. Am Fam Physician. (2008) 77(12):1678–86.18619076
49. Anagnostou A. Component resolved diagnostic testing adds clinical utility over existing testing for food allergy-PRO. Ann Allergy Asthma Immunol. (2019) 122(6):576–9. doi: 10.1016/j.anai.2019.03.003
50. Greenhawt M. Shared decision-making in the care of a patient with food allergy. Ann Allergy Asthma Immunol. (2020) 125(3):262–7. doi: 10.1016/j.anai.2020.05.031
51. Fierstein JL, Brown D, Gupta R, Bilaver L. Understanding food-related allergic reactions through a US national patient registry. J Allergy Clin Immunol Pract. (2021) 9(1):206–215.e1. doi: 10.1016/j.jaip.2020.08.011
52. Jiang J, Warren CM, Brewer A, Soffer G, Gupta RS. Racial, ethnic, and socioeconomic differences in food allergies in the US. JAMA Netw Open. (2023) 6(6):e2318162. doi: 10.1001/jamanetworkopen.2023.18162
53. Jones SM, Burks AW. Food allergy. N Engl J Med. (2017) 377(12):1168–76. doi: 10.1056/NEJMcp1611971
54. Fleischer DM, Perry TT, Atkins D, Wood RA, Burks AW, Jones SM, et al. Allergic reactions to foods in preschool-aged children in a prospective observational food allergy study. Pediatrics. (2012) 130(1):e25–32. doi: 10.1542/peds.2011-1762
55. Lau CH, Gupta RS. The pediatrician’s role in the diagnosis and management of food allergy. Pediatr Ann. (2013) 42(7):116–21. doi: 10.3928/00904481-20130619-08
56. Pistiner M, Wang J. Management of food allergy in the school setting: the clinician’s role. In: Gupta R, editors. Pediatric Food Allergy. Cham: Springer Nature Switzerland AG (2020). p. 203–12.
57. Pistiner M, Devore CD. The role of pediatricians in school food allergy management. Pediatr Ann. (2013) 42(8):334–40. doi: 10.3928/00904481-20130723-14
58. Wang J, Bingemann T, Russell AF, Young MC, Sicherer SH. The allergist’s role in anaphylaxis and food allergy management in the school and childcare setting. J Allergy Clin Immunol Pract. (2018) 6(2):427–35. doi: 10.1016/j.jaip.2017.11.022
59. Shah SS, Parker CL, O’Brian E, Davis CM. Disparity in the availability of injectable epinephrine in a large, diverse US school district. J Allergy Clin Immunol. (2014) 2(3):288–293.e1. doi: 10.1016/j.jaip.2013.09.016
60. Pajno GB, Fernandez-Rivas M, Arasi S, Roberts G, Akdis CA, Alvaro-Lozano M, et al. EAACI guidelines on allergen immunotherapy: IgE-mediated food allergy. Allergy. (2018) 73(4):799–815. doi: 10.1111/all.13319
61. US Food and Drug Administration. FDA Approves First Drug for Treatment of Peanut Allergy for Children. Available online at: https://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-treatment-peanut-allergy-children (accessed January 08, 2024).
62. Bird JA, Spergel JM, Jones SM, Rachid R, Assa’ad AH, Wang J, et al. Efficacy and safety of AR101 in oral immunotherapy for peanut allergy: results of ARC001, a randomized, double-blind, placebo-controlled phase 2 clinical trial. J Allergy Clin Immunol Pract. (2018) 6(2):476–485.e3. doi: 10.1016/j.jaip.2017.09.016
63. Hourihane JOB, Beyer K, Abbas A, Fernández-Rivas M, Turner PJ, Blumchen K, et al. Efficacy and safety of oral immunotherapy with AR101 in European children with a peanut allergy (ARTEMIS): a multicentre, double-blind, randomised, placebo-controlled phase 3 trial. Lancet Child Adolesc Health. (2020) 4(10):728–39. doi: 10.1016/S2352-4642(20)30234-0
64. Patrawala S, Ramsey A, Capucilli P, Tuong LA, Vadamalai K, Mustafa SS. Real-world adoption of FDA-approved peanut oral immunotherapy with palforzia. J Allergy Clin Immunol Pract. (2022) 10(4):1120–1122.e1. doi: 10.1016/j.jaip.2021.12.022
65. Anagnostou A, Vickery B. Food oral immunotherapy: a survey among US practicing allergists conducted as a AAAAI leadership institute project and work group report. J Allergy Clin Immunol Pract. (2023) 11(8):2330–4. doi: 10.1016/j.jaip.2023.03.060
66. Wasserman RL, Windom HH, Jones DH. Oral immunotherapy in US allergy practice. J Allergy Clin Immunol Pract. (2023) 11(12):3814–5. doi: 10.1016/j.jaip.2023.09.017
67. Greenhawt M, Shaker M, Winders T, Bukstein DA, Davis RS, Oppenheimer J, et al. Development and acceptability of a shared decision-making tool for commercial peanut allergy therapies. Ann Allergy Asthma Immunol. (2020) 125(1):90–6. doi: 10.1016/j.anai.2020.01.030
68. Author is US Food and Drug Administration. FDA Approves First Medication to Help Reduce Allergic Reactions to Multiple Foods After Accidental Exposure. (2024). Available online at: https://www.fda.gov/news-events/press-announcements/fda-approves-first-medication-help-reduce-allergic-reactions-multiple-foods-after-accidental (accessed April 29, 2024).
69. Zuberbier T, Wood RA, Bindslev-Jensen C, Fiocchi A, Chinthrajah RS, Worm M, et al. Omalizumab in IgE-mediated food allergy: a systematic review and meta-analysis. J Allergy Clin Immunol. (2023) 11(4):1134–46. doi: 10.1016/j.jaip.2022.11.036
70. Andorf S, Purington N, Block WM, Long AJ, Tupa D, Brittain E, et al. Anti-IgE treatment with oral immunotherapy in multifood allergic participants: a double-blind, randomised, controlled trial. Lancet Gastroenterol Hepatol. (2018) 3(2):85–94. doi: 10.1016/S2468-1253(17)30392-8
71. Sindher SB, Kumar D, Cao S, Purington N, Long A, Sampath V, et al. Phase 2, randomized multi oral immunotherapy with omalizumab ‘real life’ study. Allergy. (2022) 77(6):1873–84. doi: 10.1111/all.15217
72. Andorf S, Purington N, Kumar D, Long A, O’Laughlin KL, Sicherer S, et al. A phase 2 randomized controlled multisite study using omalizumab-facilitated rapid desensitization to test continued vs discontinued dosing in multifood allergic individuals. Eclinicalmedicine. (2019) 7:27–38. doi: 10.1016/j.eclinm.2018.12.006
73. Wood RA, Kim JS, Lindblad R, Nadeau K, Henning AK, Dawson P, et al. A randomized, double-blind, placebo-controlled study of omalizumab combined with oral immunotherapy for the treatment of cow’s milk allergy. J Allergy Clin Immunol. (2016) 137(4):1103. doi: 10.1016/j.jaci.2015.10.005
74. MacGinnitie AJ, Rachid R, Gragg H, Little SV, Lakin P, Cianferoni A, et al. Omalizumab facilitates rapid oral desensitization for peanut allergy. J Allergy Clin Immunol. (2017) 139(3):873. doi: 10.1016/j.jaci.2016.08.010
75. Bégin P, Dominguez T, Wilson SP, Bacal L, Mehrotra A, Kausch B, et al. Phase 1 results of safety and tolerability in a rush oral immunotherapy protocol to multiple foods using Omalizumab. Allergy Asthma Clin Immunol. (2014) 10(1):7. doi: 10.1186/1710-1492-10-7
76. Fiocchi A, Vickery BP, Wood RA. The use of biologics in food allergy. Clin Exp Allergy. (2021) 51(8):1006–18. doi: 10.1111/cea.13897
77. Wood RA, Togias A, Sicherer SH, Shreffler WG, Kim EH, Jones SM, et al. Omalizumab for the treatment of multiple food allergies. N Engl J Med. (2024) 390(10):889–99. doi: 10.1056/NEJMoa2312382
78. Hwang DW, Nagler CR, Ciaccio CE. New and emerging concepts and therapies for the treatment of food allergy. Immunotherapy Advances. (2022) 2(1):ltac006. doi: 10.1093/immadv/ltac006
79. Sindher SB, Long A, Chin AR, Hy A, Sampath V, Nadeau KC, et al. Food allergy, mechanisms, diagnosis and treatment: innovation through a multi-targeted approach. Allergy. (2022) 77(10):2937–48. doi: 10.1111/all.15418
80. Fowler J, Lieberman J. Update on clinical research for food allergy treatment. Front Allergy. (2023) 4:1154541. doi: 10.3389/falgy.2023.1154541
81. Fiocchi A, Artesani MC, Riccardi C, Mennini M, Pecora V, Fierr V, et al. Impact of omalizumab on food allergy in patients treated for asthma: a real-life study. J Allergy Clin Immunol Pract. (2019) 7(6):1901–1909.e5. doi: 10.1016/j.jaip.2019.01.023
82. Togias A, Cooper SF, Acebal ML, Assa’ad A, Baker JR Jr, Beck LA, et al. Addendum guidelines for the prevention of peanut allergy in the United States: report of the national institute of allergy and infectious diseases-sponsored expert panel. J Allergy Clin Immunol. (2017) 139(1):29–44. doi: 10.1016/j.jaci.2016.10.010
83. Al-Saud B, Sigurdardóttir ST. Early Introduction of egg and the development of egg allergy in children: a systematic review and meta-analysis. Int Arch Allergy Immunol. (2018) 177(4):350–9. doi: 10.1159/000492131
84. de Silva D, Halken S, Singh C, Muraro A, Angier E, Arasi S, et al. Preventing food allergy in infancy and childhood: systematic review of randomised controlled trials. Pediatr Allergy Immunol. (2020) 31(7):813–26. doi: 10.1111/pai.13273
85. Perkin MR, Logan K, Bahnson HT, Marrs T, Radulovic S, Craven J, et al. Efficacy of the enquiring about tolerance (EAT) study among infants at high risk of developing food allergy. J Allergy Clin Immunol. (2019) 144(6):1606. doi: 10.1016/j.jaci.2019.06.045
86. U.S. Department of Agriculture and U.S. Department of Health and Human Services. Services, Dietary Guidelines for Americans, 2020–2025. (2020).
87. Hammersley V, Kelman M, Morrice L, Kendall M, Mukerjhee M, Harley S, et al. Mixed-methods evaluation of a nurse-led allergy clinic model in primary care: feasibility trial. Clin Transl Allergy. (2022) 12(8):e12180. doi: 10.1002/clt2.12180
88. Collinson A, Waddell L, Freeman-Hughes A, Hickson M. Impact of a dietitian in general practice: paediatric food allergy. J Hum Nutr Diet. (2023) 36(3):707–15. doi: 10.1111/jhn.13130
89. Barnett CW. Need for community pharmacist-provided food-allergy education and auto-injectable epinephrine training. J Am Pharm Assoc. (2005) 45(4):479–85. doi: 10.1331/1544345054475432
90. Feng C, Kim JH. Beyond avoidance: the psychosocial impact of food allergies. Clin Rev Allergy Immunol. (2019) 57(1):74–82. doi: 10.1007/s12016-018-8708-x
91. Gupta R, Jiang J, Iyer A, Hultquist H, Cohn R, Ibrahim K, et al. Pediatric clinicians’ knowledge of infant peanut introduction guidelines following iREACH training modules. J Allergy Clin Immunol. (2022) 149(2):AB38. doi: 10.1016/j.jaci.2021.12.156
Keywords: diagnostic, referral, pediatric clinics, IgE, food allergy
Citation: Gupta RS, Epstein E and Wood RA (2024) The role of pediatricians in the diagnosis and management of IgE-mediated food allergy: a review. Front. Pediatr. 12:1373373. doi: 10.3389/fped.2024.1373373
Received: 19 January 2024; Accepted: 13 May 2024;
Published: 30 May 2024.
Edited by:
Viviana Moschese, University of Rome Tor Vergata, ItalyReviewed by:
Robert Hamilton, Johns Hopkins University, United StatesMaurizio Mennini, Sapienza University of Rome, Italy
Samuele Burastero, San Raffaele Hospital (IRCCS), Italy
© 2024 Gupta, Epstein and Wood. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ruchi S. Gupta, ci1ndXB0YUBub3J0aHdlc3Rlcm4uZWR1
 Ruchi S. Gupta
Ruchi S. Gupta Ellen Epstein
Ellen Epstein Robert A. Wood3
Robert A. Wood3