
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pediatr. , 22 July 2022
Sec. Neonatology
Volume 10 - 2022 | https://doi.org/10.3389/fped.2022.882416
This article is part of the Research Topic Congenital And Perinatal Infections: How To Prevent Sequelaes In Neonates And Children View all 11 articles
 Alberto Berardi1*
Alberto Berardi1* Isotta Zinani2
Isotta Zinani2 Luca Bedetti1,3
Luca Bedetti1,3 Eleonora Vaccina2
Eleonora Vaccina2 Alessandra Toschi2
Alessandra Toschi2 Greta Toni2
Greta Toni2 Marco Lecis2
Marco Lecis2 Federica Leone2
Federica Leone2 Francesca Monari4
Francesca Monari4 Michela Cozzolino4
Michela Cozzolino4 Tommaso Zini2
Tommaso Zini2 Alessandra Boncompagni1
Alessandra Boncompagni1 Lorenzo Iughetti2,5
Lorenzo Iughetti2,5 Francesca Miselli1
Francesca Miselli1 Licia Lugli1
Licia Lugli1Objective: To compare two strategies [the neonatal sepsis risk calculator (NSC) and the updated serial clinical observation approach (SCO)] for the management of asymptomatic neonates at risk of early-onset sepsis (EOS) and neonates with mild non-progressive symptoms in the first hours of life.
Methods: This was a single-center, retrospective cohort study conducted over 15 months (01/01/2019–31/03/2020). All live births at ≥34 weeks of gestation were included. Infants were managed using SCO and decisions were compared with those retrospectively projected by the NSC. The proportion of infants recommended for antibiotics or laboratory testing was compared in both strategies. McNemar's non-parametric test was used to assess significant differences in matched proportions.
Results: Among the 3,445 neonates (late-preterm, n = 178; full-term, n = 3,267) 262 (7.6%) presented with symptoms of suspected EOS. There were no cases of culture-proven EOS. Only 1.9% of the neonates were treated with antibiotics (median antibiotic treatment, 2 days) and 4.0% were evaluated. According to NSC, antibiotics would have been administered in 5.4% of infants (absolute difference between SCO and NSC, 3.51%; 95% CI, 3.14–3.71%; p <0.0001) and 5.6% of infants would have undergone “rule out sepsis” (absolute difference between SCO and NSC, 1.63%, 95% CI 1.10–2.05; p <0.0001).
Conclusion: SCO minimizes laboratory testing and unnecessary antibiotics in infants at risk of EOS or with mild non-progressive symptoms, without the risk of a worse neonatal outcome. The NSC recommends almost three times more antibiotics than the SCO without improving neonatal outcomes.
The current management of neonates at risk of early-onset sepsis (EOS) remains controversial, and the suggested approaches are heterogeneous (1). EOS rates have declined substantially due to the widespread use of intrapartum antibiotic prophylaxis (IAP). However, the diagnosis of EOS remains challenging because its initial clinical signs may be ambiguous, diagnostic tests are poorly predictive, and delayed antibiotic treatment can have devastating consequences. Therefore, 30–40 uninfected neonates are exposed to unnecessary antibiotics for each infant subsequently confirmed to have EOS (2). However, perinatal antibiotic exposure is a major concern, given the potential long-term effects of changes in the intestinal flora of uninfected neonates (1, 3) and the consensus on the optimal management of neonates considered at risk of EOS is shifting (4, 5).
The American Academy of Pediatrics suggests three alternative approaches for the use of risk factors (RFs) to identify infants at increased risk of EOS (1). First, the categorical RFs assessment (6); such an approach has some limitations and is associated with higher rates of antibiotic treatment (7). Second, the neonatal sepsis risk calculator (NSC). The NSC has a Bayesian approach to create a multivariate model to predict infant-specific EOS risk, derived and validated from a case-control study of blood culture-proven EOS. NSC permit improved delineation of low-risk babies that can be safely managed with observation. Following the adoption of the most recent NSC laboratory workups, the use of empirical antibiotic administration in the first 24 h of life has decreased significantly (from 5.0 to 2.6%) in a recent US multicenter study (8). However, these low rates of antibiotic treatment have rarely been confirmed outside the USA (the country where NSC was created), and antibiotic treatment may reach 8% of neonates elsewhere (9). Finally, increasing evidence has shown that asymptomatic neonates at risk can be safely managed with serial clinical observation (SCO) without antibiotic treatment (10–12). However, for “less symptomatic” infants, it is difficult to allow adequate time to undergo a physiological transition before deciding whether clinical signs are transient or permanent (4).
This study aimed to assess the impact of an updated SCO approach planned to reduce unnecessary antibiotic therapy in “less symptomatic” full-term and late-preterm neonates. The impact on neonatal outcomes, laboratory testing rates, and neonatal intensive care unit (NICU) admissions were also evaluated. Furthermore, we compared management decisions using the updated SCO with those projected through the virtual application of the NSC.
This was a retrospective study carried out over 15 months (from January 1st, 2019 to March 31th, 2020), in a single, high-volume tertiary care center (Modena University Hospital, Italy), with ~3,000 live births/year. This center advocates a recto-vaginal culture screening strategy at 35–37 weeks gestation (6). The project was approved by the local ethics committee (no. 169/2019/OSS*/AOUMO). All infants born at ≥34 weeks gestation were included in the study. To obtain complete information on the rates of antibiotic treatment in the entire population, we included neonates with malformations, metabolic diseases, or surgical complications. Full maternal data (gestational age, mode of delivery, group B Streptococcus status, RFs for EOS, and duration of IAP) were routinely recorded in neonatal charts. The records were collected anonymously in an Excel format with controlled access, assigning each newborn a progressive numerical code.
This approach is directed at asymptomatic infants with RFs (10). A standardized form, signed by each examiner, were used to detail general wellbeing, skin color, and respiratory signs at standard intervals (at ages 1, 3, 6, 12, 18, 24, 36, and 48 h) (13). All asymptomatic neonates with RFs for EOS are usually managed by midwives, nurses or pediatricians in the mother baby unit, where neonates ≥ 35 weeks' gestation “room in” with their own mother. Asymptomatic neonates with 34 weeks' gestation are usually admitted to intermediate care unit.
Each newborn with symptoms of suspected sepsis is immediately referred to a neonatal care specialist. However, those with mild to moderate disease that requires oxygen support or a high-flow nasal cannula are separated from their mothers and admitted to an intermediate care unit. Severely ill neonates and those undergoing nCPAP or mechanical ventilation are admitted to the NICU. Ampicillin plus an aminoglycoside is administered as empiric therapy for suspected EOS in symptomatic neonates. Before March 2018, white blood cell count (WBC), C-reactive protein (CRP) levels, and blood culture were obtained to rule out sepsis in asymptomatic, chorioamnionitis-exposed neonates or those who developed symptoms of variable severity.
In March 2018, the main neonatal symptoms triggering laboratory evaluation were defined (Table 1A) (14). Neonates with mild, non-progressive symptoms (that can be due to non-infectious diseases, i.e., transient tachypnea of the newborn) who present in the first few hours of life can be reevaluated at 2-h intervals. No laboratory testing is performed, and no antibiotic therapy is given if symptoms remain mild, even after multiple re-evaluations in case of minor criteria. In contrast, the presence of major symptoms (as defined in Table 1A) or worsening of mild symptoms suggest the need for laboratory evaluation. Among patients who undergo a sepsis workup, treatment decisions are left to the discretion of the physician. This updated approach aims to minimize laboratory testing (which, in our experience, strongly influences clinicians' decisions) (10), unnecessary antibiotics, and mother-baby separation.
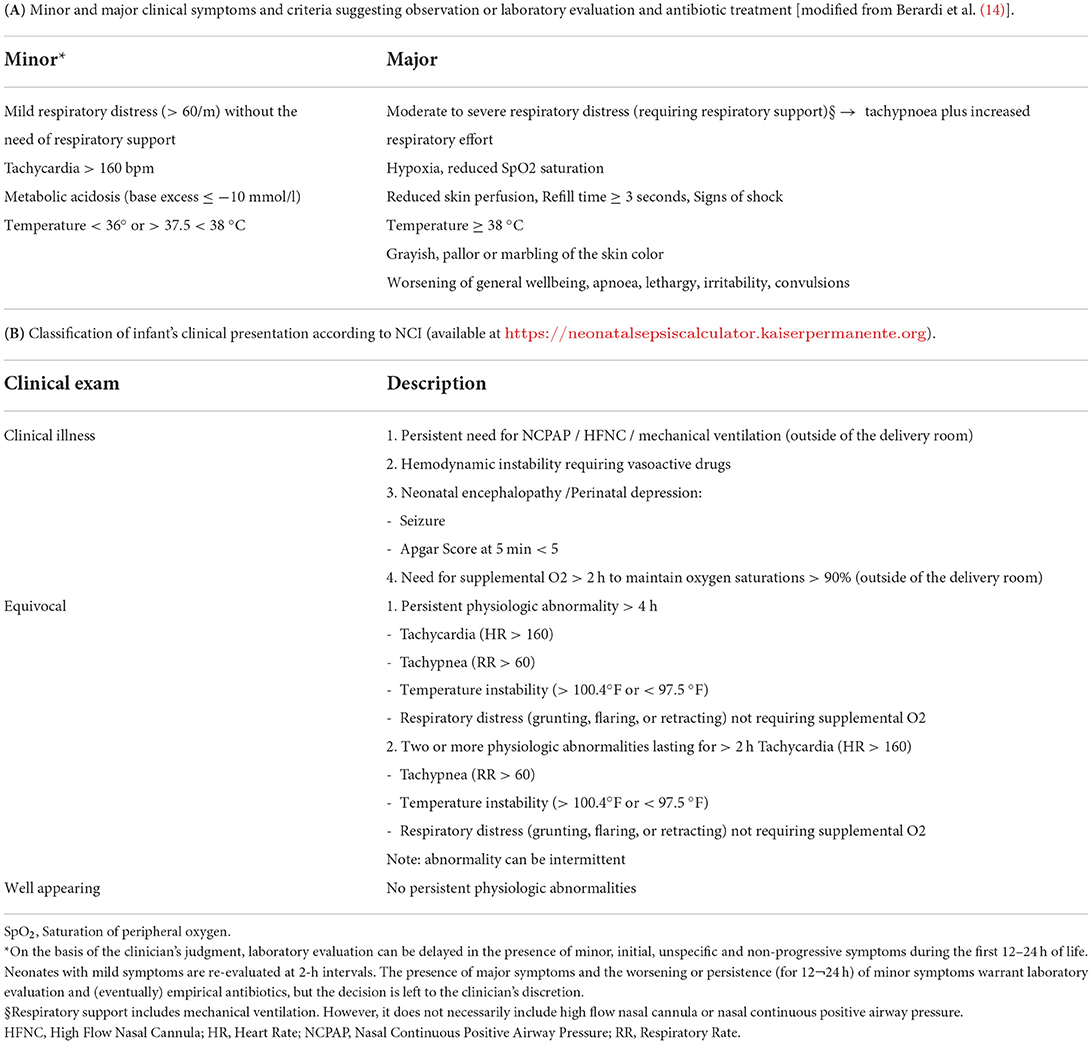
Table 1. Symptoms and classification of infant's clinical presentation according to serial clinical observation and neonatal sepsis calculator.
The NSC is an online tool that quantifies the risk of EOS in infants with a gestational age ≥34 weeks using a pretest probability. Recommendations for antibiotic treatment or neonatal management are derived from an algorithmic framework based on the local incidence of EOS, maternal RFs (gestational age, highest intrapartum temperature, duration of membrane rupture, GBS status, and IAP), and clinical presentation of the infants during the first few hours of life. For each infant, the previous risk of EOS was calculated based on the local incidence of EOS and maternal RFs alone. The prior probability is converted into a posterior probability of EOS in the different categories of infants' clinical presentation (likelihood ratio of 0.41, 5.0, and 21.2 for well-appearing, equivocal, and clinical illness, respectively; Table 1B) (15). The resulting post-test probability of EOS is classified into three risk layers (<0.65, 0.65–1.54, and >1.54 cases/1,000 live births). The management recommendations suggested by the NSC are as follows: (1) no culture, no antibiotics, routine vitals (posterior risk <1/1,000 live births); (2) no culture, no antibiotics, vitals every 4 h for 24 h (posterior risk <1/1,000 live births, but prior risk >1/1,000 live births); (3) blood culture, vitals every 4 h for 24 h (posterior risk 1–3/1,000 live births); (4) strongly consider starting empiric antibiotics, vitals per NICU (posterior risk <3/1,000 live births); and (5) empiric antibiotics, vitals per NICU (posterior risk >3/1,000 live births or clinical signs of illness). In the current study, each infant was retrospectively scored as well as appeared equivocal or clinically ill within 4 h after birth. Recommendations for the management of neonates according to NSC were calculated by assuming an incidence rate of EOS of 0.6/1,000 live births (16).
We used MedCalc version 9.3 (MedCalc Software, https://www.medcalc.org). Continuous variables are expressed as mean ± SD or median and interquartile range (IQR), and categorical data are expressed as numbers (percentages). Categorical and continuous variables were compared between patient groups using the χ2 test, Fisher's exact test, Student's t-test, or Mann–Whitney test, as appropriate. All p-values refer to two-tailed tests of significance; p <0.05 was considered significant. McNemar's non-parametric test was used to assess significant differences in the matched proportions.
During the study period, 3,456 neonates were ≥34 weeks of gestation. Records were available for 3,445 (99.7%) infants, of which 178 were born late preterm and 3,267 were born full-term. The median gestational age was 39.6 weeks and the median birth weight was 3,310 g.
Table 2 shows demographics according to full-term or late-preterm delivery. Vaginal delivery and prenatal vagino-rectal screening were more likely in full-term neonates, while prolonged membrane rupture and IAP were more likely among late preterm neonates. Among the 3,445 infants included in the study, 264 (7.6%) had symptoms of suspected EOS (most were respiratory and already at birth). Table 3 shows the age of presentation of symptoms, NSC scores, and antibiotics administered by comparing full-term and late-preterm neonates. Only 1.9% of the entire cohort was treated with antibiotics (median 2 days), and 4% underwent “rule out sepsis”; 3.1 and 2.3% were admitted to the NICU or intermediate care unit, respectively (neonates who were admitted and were given antibiotics or underwent “rule out sepsis” are reported in the footnote of Table 3).
Among the 200 symptomatic neonates unexposed to antibiotics, 77 (38%) were allowed to “room in” with their mothers within a few hours after birth. The remaining 123 (62%) were admitted to NICU (of which 80 underwent nCPAP) and/or intermediate care unit (of which 67 underwent high-flow nasal cannulation). Symptoms improved substantially within the first 24–48 h of life (median duration 72 h, IQR 24–120). Late-preterm neonates were more likely to have symptoms, undergo SCO, be evaluated, have a higher NSC score, be admitted to the NICU, and be treated with antibiotics. However, the median number of days on antibiotics did not differ between late preterm and full-term neonates.
Figure 1 details neonatal symptoms, Apgar scores and the need for respiratory support among symptomatic neonates. Respiratory symptoms (tachypnea, respiratory distress syndrome, desaturation) were the most common.
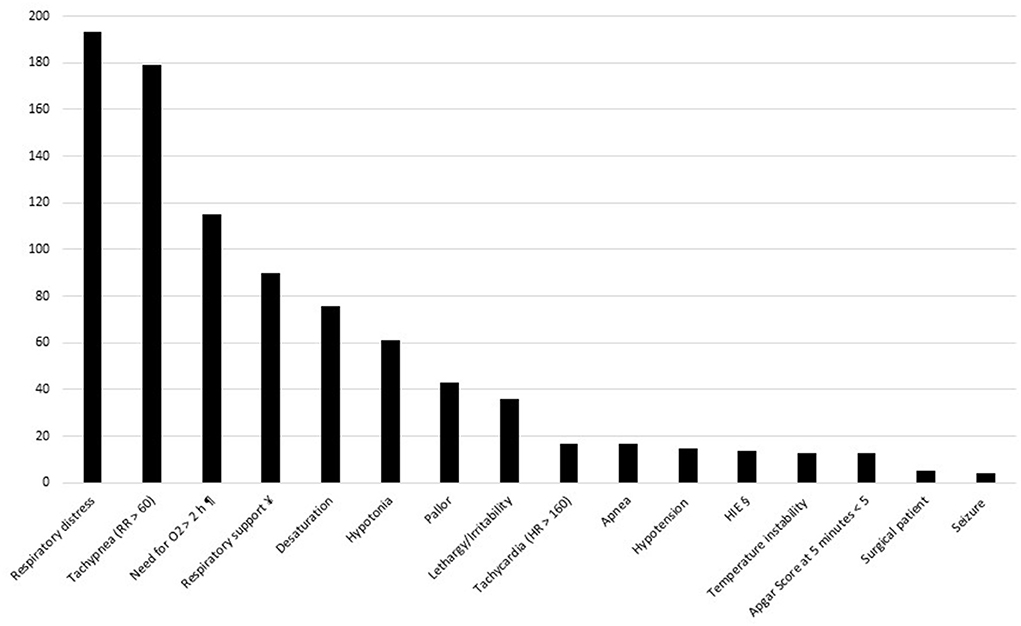
Figure 1. Symptoms and respiratory supports among symptomatic neonates. HIE, hypoxic ischemic encephalopathy. ¶ to maintain SpO2 > 90%. ¥ persistent need for NCPAP / HFNC / mechanical ventilation. § hypoxic ischemic encephalopathy requiring hypothermia.
Among 3,445 infants, the following indications were suggested by NSC: no culture, no antibiotics, routine vitals (n = 3,238), strong consideration of starting empiric antibiotics, vitals per NICU (n = 131); empiric antibiotics, vitals per NICU (n = 55); no culture, no antibiotics, vitals every 4 h for 24 h (n = 13); and blood culture, vitals every 4 h for 24 h (n = 8).
Table 4 compares the number of “rule out sepsis” evaluations (a) and antibiotic treatments (b) in all neonates. Of the 3,380 neonates who did not start on antibiotics according to the SCO, 3,254 would also have avoided antibiotics according to the NSC. The remaining 126 neonates would have been recommended antibiotics by the NSC, but remained well without treatment. In contrast, five neonates would have avoided antibiotics according to NSC (absolute difference 3.5%; 95% CI, 3.1–3.7%, p <0.0001). According to NSC, antibiotics would have been administered to 5.4% of infants. Of the 138 neonates who were evaluated to rule out sepsis according to the SCO, 118 were also evaluated according to the NSC. Seventy-six neonates would have undergone “rule out sepsis” according to the NSC but were not evaluated. In contrast, 20 neonates would not have been evaluated according to as per NSC but were evaluated according to as per SCO (absolute difference, 1.63%; 95% CI, 1.10–2.05; p <0.0001). According to NSC, 5.6% of infants would have undergone “rule out sepsis.”
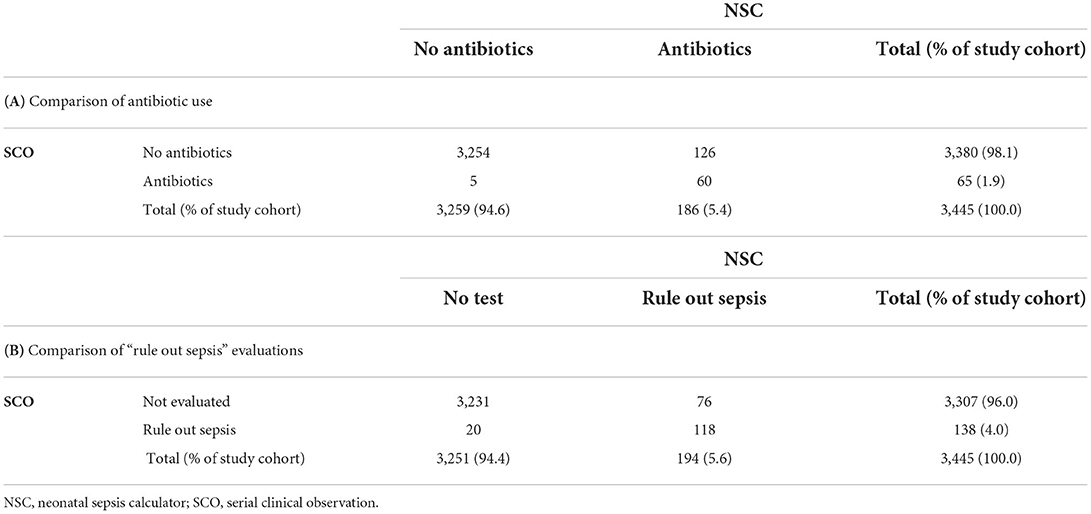
Table 4. Comparison of antibiotic use (A) and “rule out sepsis” evaluations (B) as per SCO vs. recommendations of the NSC in the study population. Differences between proportions were analyzed using McNemar's test.
Table 5 compares the number of “rule out sepsis” evaluations (a) and antibiotic treatments (b) among late-preterm infants. According to NSC, antibiotics would have been administered in 27.0% of late-preterm infants compared to 8.4% according to SCO (absolute difference, 18.6%; 95% CI, 12.3–24.8%; p <0.0001). Similarly, according to NSC, 28.1% of late-preterm infants would have undergone “rule out sepsis,” compared to 18.5% according to SCO (absolute difference, 9.6%; 95% CI, 4.4–14.7%; p <0.0001).
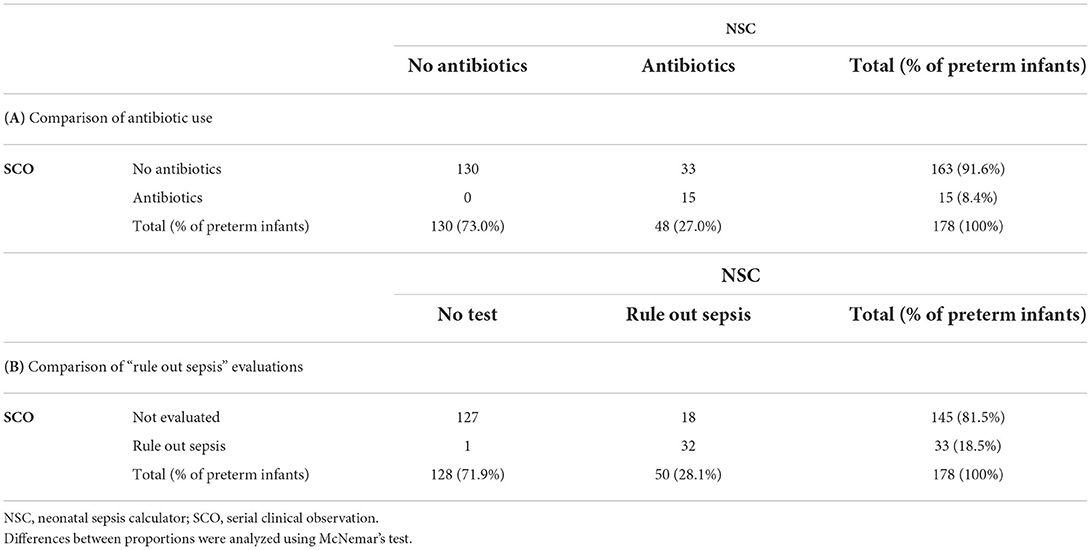
Table 5. Comparison of antibiotic use (A) and “rule out sepsis” evaluations (B) as per SCO vs. recommendations of the NSC among late-preterm infants.
Among the 110 neonates born after an increased maternal intrapartum temperature (≥37.5°C), 20 developed symptoms. Eight of these 20 patients were treated with antibiotics. For the 12 untreated neonates, the NSC would have suggested the following: empiric antibiotics, vitals per NICU (n = 7); blood culture, vitals every 4 h for 24 h (n = 2); no culture, no antibiotics, routine vitals (n = 2); and strongly consider starting empiric antibiotics and vitals per NICU (n = 1). Therefore, 8 of the 12 neonates would have received antibiotics per NSC but remained untreated.
Figure 2 is a box plot showing the distribution of the NSC scores in the three cohorts: (i) all infants in the study population, (ii) infants initiated on antibiotics according to SCO, and (iii) those recommended antibiotics by NSC. The sepsis risk score in the study population was low; infants treated with antibiotics according to SCO had higher sepsis risk scores than those recommended antibiotics by the NSC.
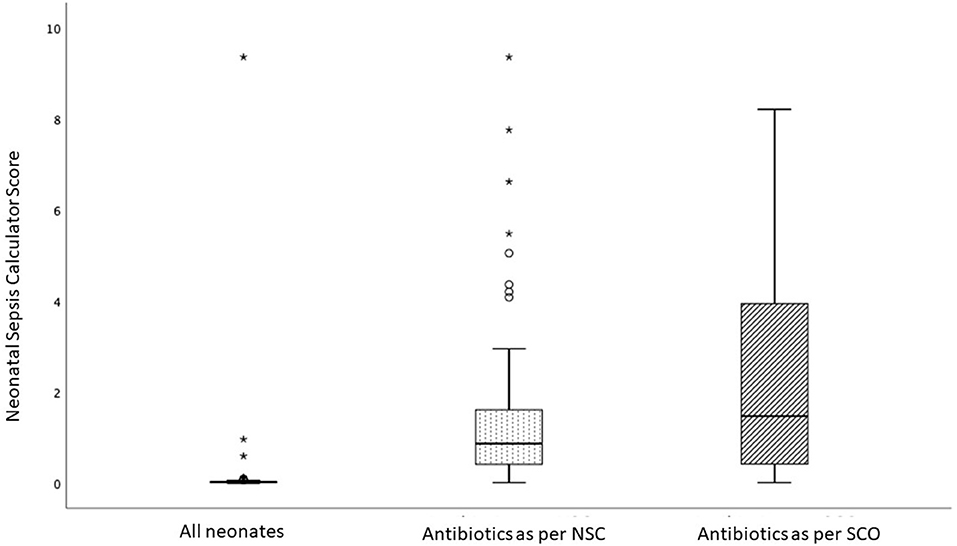
Figure 2. Box-and-Whisker plot comparing the score of the Neonatal Sepsis Calculator (NSC) in three groups: all infants in the study (median score = 0.02; IQR 0.02), infants receiving antibiotics as per NSC (median = 0.86; IQR = 1.22) and infants recommended antibiotics by Serial Clinical Observation (SCO) (median = 0.86; IQR = 1.22). The score in the study population was low; infants recommended antibiotics as per SCO had higher scores compared with infants recommended antibiotics as per NSC. Each box bounds the IQR range divided by the median (solid horizontal line); the lower and upper margins of the box represent the 25th and the 75th centile, respectively. The whiskers extend 1.5 times the IQR from the median. A circle (o) is used to mark outliers with values between 1.5 and 3 box lengths from the upper edge of the box; the asterisk (*) is used for extreme outliers (a value more than 3 times the interquartile range).
No cases of culture-proven sepsis were found. Six infants were administered antibiotics (≥5 days), and all but one were born to GBS-negative mothers. Three of the six infants had an abnormal blood count, three had elevated CRP, and four underwent lumbar puncture (the CSF was sterile in all cases). Their blood cultures were sterile except for one neonate (born to a GBS-positive mother and exposed to inadequate IAP) who developed mild tachypnea (7 h of life) and received ampicillin plus gentamicin. The blood culture yielded Staphylococcus hominis, which was considered a contaminant because the subsequent blood culture was sterile.
None of the 3,445 included infants was readmitted within 30 days of birth with a positive blood culture result. Among neonates who developed symptoms after birth, none worsened or had brain lesions due to delayed antibiotic treatment. Two neonates died: the first was affected by an inherited metabolic disease and the second was affected by a congenital diaphragmatic hernia.
There is a consensus to administer empirical antibiotics to neonates with suspected EOS symptoms, with or without RFs (17). This strategy is based more on historical customs and practices than on evidence. In most babies, non-specific symptoms during the first hours of life are not due to infection. Unnecessary antibiotics may disrupt the neonatal gut microbiome with long-term consequences and increase the resistance to pathogens. Furthermore, intravenous infusion may complicate extravasation, whereas mother-infant separation for EOS evaluation can delay breastfeeding initiation and increase formula supplementation (7, 18). Therefore, strategies to reduce unnecessary antibiotic use are of interest. This is the first comparison between the NSC and the updated SCO approach (13). Indeed, we defined “minor” and “major” clinical symptoms to guide clinicians in the evaluation and treatment of infants with antibiotics.
Approximately ¾ of the symptomatic neonates were not exposed to antibiotics; 38% were allowed to “room in” with their mother within a few hours after birth. Among the remaining 62% who were admitted to the NICU or to the intermediate care unit, some underwent nCPAP or HFNC several hours after birth. Therefore, the persistence of symptoms beyond 2 h after birth may not be a good criterion for the administration of antibiotics, especially if the symptoms do not worsen or the risk of EOS is very low (i.e., infants born out of labor and with intact membranes). “Equivocal symptoms” or “clinical illness” are common in the first hours of life due to the transition to extrauterine life. The developers of NSC suggested that antibiotics “strongly consider” as a safeguard not to discontinue therapy in clinically symptomatic infants, even if the posterior probability is below the threshold for treatment (<3 cases/1,000 live births). However, a comparison with our updated SCO approach shows that this recommendation of NSC would substantially increase antibiotic exposure in uninfected infants without improving neonatal outcomes. In particular, our updated SCO approach reduced antibiotic exposure among late preterm infants by two-thirds compared to NSC. The NSC model is associated with increased postnatal antibiotic exposure, especially among infants with RFs for EOS or those with “transitional” symptoms in the first hours of life. Indeed, in some cases, the NSC model can overestimate the absolute risk of EOS; for example, the NSC assumes the same risk of EOS for neonates unexposed or exposed to inadequate IAP (duration <2 h) (19); however, it is known that the risk of developing EOS in asymptomatic IAP-exposed neonates is very low, regardless of the duration of IAP (20).
The low rate of “rule out sepsis” evaluations we performed in infants with mild symptoms probably contributed to the reduction of unnecessary antibiotics. This finding is consistent with recent studies demonstrating the low predictive value of ancillary tests (21) and the reduced (~30%) antibiotic exposure when CRP is excluded from the diagnostic panel of EOS (22). Furthermore, the importance of a positive blood culture obtained from an asymptomatic newborn infant is unclear (23). In our experience, repeated evaluations of asymptomatic infants may even increase the yield of pathogens from blood cultures (often difficult to interpret), thus giving antibiotics even to infants whose symptoms of EOS would likely never appear.
Only 1.9% of our infants were exposed to antibiotics and most received very short courses. Our approach was of utmost benefit in preterm infants, who often have “transient” symptoms in the first few hours of life, compared with full-term infants. Until recently, up to 35% of late-preterm infants received antibiotics (24), thus separating neonates from their mothers. In contrast, only 8% of our preterm neonates received antibiotics, while 70% were allowed to room in with their mothers. However, the overall neonatal antibiotic exposure rates in preterm and full-term neonates could be 30 times higher than necessary, as the incidence of EOS in our NICU is 0.6/1,000 live births (16). More efforts should be made to better identify the infants to be treated. None of the neonates had worse outcomes due to delayed treatment of an infection. Perhaps the SCO strategy is safer in our center, as adherence to recommendations for GBS prevention is very high (25), whereas SCO would be less effective in centers with low adherence to guidelines.
This study had several important limitations. First, the SCO strategy may be safer in our center, where adherence to recommendations for the prevention of GBS is very high, while it could be less effective where adherence to guidelines is low, or the incidence of EOS is higher. Second, the sample size of the infants in the study was small and we had no cases of culture-proven EOS to define a hypothetical overtreatment index or to confirm the safe management of infected neonates, although this information has already been provided in our previous study (10). In addition, management was based on SCO, whereas the data elements for the NSC were collected retrospectively. This would result in a less accurate identification of the symptoms by which the NSC score was calculated, although newborn charts accurately describe their clinical condition. Finally, the definition of “minor” and “major” symptoms was defined a priori, based on expert opinions in our network. However, a large prospective study including EOS cases and controls may more accurately define which symptoms are most predictive of EOS.
In conclusion, SCO of asymptomatic infants with RFs for EOS or with “mild, non-progressive symptoms” during the first days of life reduces laboratory evaluation, minimizes unnecessary antibiotics, and avoids separation of the mother from her infant without delaying antibiotic treatment of infected infants. Antibiotic overuse is a planetary emergency and we hope that our experience can help reduce neonatal antibiotic exposure.
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
The studies involving human participants were reviewed and approved by Comitato Etico Area Vasta Emilia Nord. Written informed consent from the participants' legal guardian/next of kin was not required to participate in this study in accordance with the national legislation and the institutional requirements.
AB conceptualized the study, drafted the initial manuscript, reviewed and edited it, and supervised the study. IZ and LL contributed to conceptualization, drafted the initial reviewed, and edited the manuscript. LB, EV, AT, GT, ML, FL, FMo, MC, TZ, AB, LI, and FMi contributed to acquisition, analysis and interpretation of data for the work, and reviewed and edited the manuscript. All authors substantially contributed to the work, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
This study was partially supported by the Agenzia Sanitaria Regionale (Emilia-Romagna) Piano Regionale della Prevenzione (2015-2018) C.U.P. n. E43G17000680002.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The handling editor CA declared a past co-authorship with one of the authors AB.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fped.2022.882416/full#supplementary-material
1. Puopolo KM, Benitz WE, Zaoutis TE. Management of neonates born at ≥35 0/7 Weeks' gestation with suspected or proven early-onset bacterial sepsis. Pediatrics. (2018) 142:e20182894. doi: 10.1542/peds.2018-2894
2. Benitz WE, Achten NB. Finding a role for the neonatal early-onset sepsis risk calculator. EClinicalMedicine. (2020) 19:100255. doi: 10.1016/j.eclinm.2019.100255
3. Ficara M, Pietrella E, Spada C, Della Casa Muttini E, Lucaccioni L, Iughetti L, et al. Changes of intestinal microbiota in early life. J Matern Fetal Neonatal Med. (2020) 33:1036–43. doi: 10.1080/14767058.2018.1506760
4. Fleiss N, Hooven TA, Polin RA. Can we back off using antibiotics in the NICU? Semin Fetal Neonatal Med. (2021) 26:101217. doi: 10.1016/j.siny.2021.101217
5. Benitz WE, Wynn JL, Polin RA. Reappraisal of guidelines for management of neonates with suspected early-onset sepsis. J Pediatr. (2015) 166:1070–4. doi: 10.1016/j.jpeds.2014.12.023
6. Verani JR McGee L Schrag SJ Division Division of Bacterial Diseases National Center for Immunization and Respiratory Diseases Centers for Disease Control and Prevention (CDC). Prevention of perinatal group B streptococcal disease–revised guidelines from CDC, 2010. MMWR Recomm Rep Morb. (2010) 59:1–36.
7. Mukhopadhyay S, Dukhovny D, Mao W, Eichenwald EC, Puopolo KM. 2010 perinatal GBS prevention guideline and resource utilization. Pediatrics. (2014) 133:196–203. doi: 10.1542/peds.2013-1866
8. Kuzniewicz MW, Puopolo KM, Fischer A, Walsh EM Li S, Newman TB, Kipnis P, et al. Quantitative, risk-based approach to the management of neonatal early-onset sepsis. JAMA Pediatr. (2017) 171:365–71. doi: 10.1001/jamapediatrics.2016.4678
9. Achten NB, Klingenberg C, Benitz WE, Stocker M, Schlapbach LJ, Giannoni E, et al. Association of use of the neonatal early-onset sepsis calculator with reduction in antibiotic therapy and safety: a systematic review and meta-analysis. JAMA Pediatr. (2019) 173:1032–40. doi: 10.1001/jamapediatrics.2019.2825
10. Berardi A, Fornaciari S, Rossi C, Patianna V, Bacchi Reggiani ML, Ferrari F, et al. Safety of physical examination alone for managing well-appearing neonates ≥ 35 weeks' gestation at risk for early-onset sepsis. J Matern Fetal Neonatal Med. (2015) 28:1123–7. doi: 10.3109/14767058.2014.946499
11. Joshi NS, Gupta A, Allan JM, Cohen RS, Aby JL, Weldon B, et al. Clinical monitoring of well-appearing infants born to mothers with chorioamnionitis. Pediatrics. (2018) 141:e20172056. doi: 10.1542/peds.2017-2056
12. Bain L, Sivakumar D, McCallie K, Balasundaram M, Frymoyer A. A clinical monitoring approach for early onset sepsis: a community hospital experience. Hosp Pediatr. (2022) 12:16–21. doi: 10.1542/hpeds.2021-006058
13. Berardi A, Bedetti L, Spada C, Lucaccioni L, Frymoyer A. Serial clinical observation for management of newborns at risk of early-onset sepsis. Curr Opin Pediatr. (2020) 32:245–51. doi: 10.1097/MOP.0000000000000864
14. Berardi A, Spada C, Ciccia M, Capretti M, Brusa A, Sandri F, et al. Observation on the newborn at risk of early-onset sepsis: the approach of the emilia-romagna region (Italy). Medico E Bambino. (2019) 38:370–6.
15. Kuzniewicz MW, Walsh EM, Li S, Fischer A, Escobar GJ. Development and implementation of an early-onset sepsis calculator to guide antibiotic management in late preterm and term neonates. Jt Comm J Qual Patient Saf. (2016) 42:232–9. doi: 10.1016/S1553-7250(16)42030-1
16. Berardi A, Baroni L, Bacchi Reggiani ML, Ambretti S, Biasucci G, Bolognesi S, et al. The burden of early-onset sepsis in emilia-romagna (Italy): a 4-year, population-based study. J Matern-Fetal Neonatal Med. (2016) 29:3126–31. doi: 10.3109/14767058.2015.1114093
17. van Herk W, el Helou S, Janota J, Hagmann C, Klingenberg C, Staub E, et al. Variation in current management of term and late-preterm neonates at risk for early-onset sepsis: an international survey and review of guidelines. Pediatr Infect Dis J. (2016) 35:494–500. doi: 10.1097/INF.0000000000001063
18. Hooven TA, Randis TM, Polin RA. What's the harm? Risks and benefits of evolving rule-out sepsis practices. J Perinatol. (2018) 38:614–22. doi: 10.1038/s41372-018-0081-3
19. Benitz WE, Achten NB. Technical assessment of the neonatal early-onset sepsis risk calculator. Lancet Infect Dis. (2021) 21:e134–40. doi: 10.1016/S1473-3099(20)30490-4
20. Berardi A, Spada C, Vaccina E, Boncompagni A, Bedetti L, Lucaccioni L. Intrapartum beta-lactam antibiotics for preventing group B streptococcal early-onset disease: can we abandon the concept of “inadequate” intrapartum antibiotic prophylaxis? Expert Rev Anti Infect Ther. (2020) 18:37–46. doi: 10.1080/14787210.2020.1697233
21. Benitz WE. Adjunct laboratory tests in the diagnosis of early-onset neonatal sepsis. Clin Perinatol. (2010) 37:421–38. doi: 10.1016/j.clp.2009.12.001
22. Singh N, Gray JE. Antibiotic stewardship in NICU: De-implementing routine CRP to reduce antibiotic usage in neonates at risk for early-onset sepsis. J Perinatol. (2021) 41:2488–94. doi: 10.1038/s41372-021-01110-w
23. Albers WH, Tyler CW, Boxerbaum B. Asymptomatic bacteremia in the newborn infant. J Pediatr. (1966) 69:193–7. doi: 10.1016/S0022-3476(66)80319-0
24. Wang ML, Dorer DJ, Fleming MP, Catlin EA. Clinical outcomes of near-term infants. Pediatrics. (2004) 114:372–6. doi: 10.1542/peds.114.2.372
Keywords: early-onset sepsis, neonatal early-onset sepsis calculator, newborn, serial clinical observation, neonates, perinatal distress
Citation: Berardi A, Zinani I, Bedetti L, Vaccina E, Toschi A, Toni G, Lecis M, Leone F, Monari F, Cozzolino M, Zini T, Boncompagni A, Iughetti L, Miselli F and Lugli L (2022) Should we give antibiotics to neonates with mild non-progressive symptoms? A comparison of serial clinical observation and the neonatal sepsis risk calculator. Front. Pediatr. 10:882416. doi: 10.3389/fped.2022.882416
Received: 23 February 2022; Accepted: 30 June 2022;
Published: 22 July 2022.
Edited by:
Cinzia Auriti, Bambino Gesù Children's Hospital (IRCCS), ItalyReviewed by:
Fiammetta Piersigilli, Cliniques Universitaires Saint-Luc, BelgiumCopyright © 2022 Berardi, Zinani, Bedetti, Vaccina, Toschi, Toni, Lecis, Leone, Monari, Cozzolino, Zini, Boncompagni, Iughetti, Miselli and Lugli. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Alberto Berardi, YWxiZXJ0by5iZXJhcmRpQHVuaW1vcmUuaXQ=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.