- 1Division of Pediatric Nephrology, Department of Pediatrics, Mayo Clinic, Rochester, MN, United States
- 2Division of Pediatric Nephrology, Department of Pediatrics, Medical University of South Carolina, Charleston, SC, United States
- 3Division of Nephrology, Department of Pediatrics, Stanford University, Stanford, CA, United States
- 4Michigan Institute for Clinical and Health Research, University of Michigan, Ann Arbor, MI, United States
- 5Levine Children's Hospital at Atrium Health, Charlotte, NC, United States
- 6Driscoll Children's Hospital, Corpus Christi, TX, United States
- 7Division of Nephrology and Dialysis, Akron Children's Hospital, Akron, OH, United States
- 8Pediatric Specialists of Virginia, Fairfax, VA, United States
- 9Department of Pediatrics, George Washington University, Washington, DC, United States
- 10Division of Nephrology, Department of Pediatrics, Louisiana State University, New Orleans, LA, United States
- 11Division of Pediatric Nephrology, Department of Pediatrics, McMaster University, Hamilton, ON, Canada
- 12Division of Nephrology, Nationwide Children's Hospital, Columbus, OH, United States
- 13Division of Pediatric Nephrology and Hypertension, Stony Brook Children's Hospital and Renaissance School of Medicine, Stony Brook, NY, United States
- 14Division of Pediatric Nephrology, Department of Pediatrics, University of Michigan, Ann Arbor, MI, United States
Background: Children with nephrotic syndrome (NS) are at high risk for vaccine-preventable infections due to the immunological effects from the disease and concurrent treatment with immunosuppressive medications. Immunizations in these patients may be deferred due to their immunosuppressive treatment which may increase the risk for vaccine-preventable infections. Immunization practices in children with NS continue to vary among pediatric nephrologists. This raises the question of whether children with NS are receiving the recommended vaccinations at appropriate times. Therefore, it is critical to understand the practices and patient education provided by physicians to patients on the topic of vaccinations.
Methods: After informed consent, parents/guardians of 153 pediatric patients (<18 years old) diagnosed with NS from 2005 to 2018 and 50 pediatric nephrologists from 11 participating centers completed anonymous surveys to evaluate immunization practices among pediatric nephrologists, assess the vaccine education provided to families of children with NS, assess the parental knowledge of immunization recommendations, and assess predictors of polysaccharide pneumococcal vaccine adherence. The Advisory Committee on Immunization Practices (ACIP) Immunization 2019 Guideline for those with altered immunocompetence was used to determine accuracy of vaccine knowledge and practices.
Results: Forty-four percent of providers self-reported adherence to the ACIP guidelines for inactive vaccines and 22% to the guidelines for live vaccines. Thirty-two percent of parents/guardians reported knowledge that aligned with the ACIP guidelines for inactive vaccines and 1% for live vaccines. Subjects residing in the Midwest and provider recommendations for vaccines were positive predictors of vaccine adherence (p < 0.001 and p 0.02, respectively).
Conclusions: Vaccine recommendation by medical providers is paramount in vaccine adherence among pediatric patients with NS. This study identifies potential educational opportunities for medical subspecialty providers and family caregivers about immunization recommendations for immunosuppressed patients.
Introduction
Nephrotic syndrome (NS) is caused by renal diseases that affect the permeability of the glomerular filtration barrier resulting in massive proteinuria, including immunoglobulins and complement proteins (1–4). Given the immunological effects from the disease and concurrent treatment with immunosuppressive medications, children with NS are at high risk for severe bacterial infections, especially with encapsulated bacteria (5). Streptococcus pneumoniae is a common encapsulated bacterial pathogen that is known to cause serious infections in children with NS (6, 7). Given the risk for this infectious pathogen, the pneumococcal polysaccharide vaccine (PPSV 23) has been specifically recommended for pediatric patients with certain medical conditions/diseases (i.e., NS, chronic renal failure, and immunosuppression medications). This susceptibility to infection extends beyond bacterial infections and includes serious viral infections such as Varicella, which can result in severe disseminated infections in immunocompromised hosts.
Children with NS have a high burden of healthcare utilization with a mean charge per hospitalization of $26,500 that exceeds many other chronic illnesses (8). Gipson et al. evaluated hospitalization costs in a cohort of children with NS and clearly showed that serious complications of NS, including infection, occur commonly and increase healthcare costs. In this study, 16% of 9,934 discharges in 2006 and 2009 had at least one severe complication (pneumonia, sepsis, peritonitis, thromboembolism, or diabetes) attributable to NS or its treatment. Infection-related complications were the most common including pneumonia, sepsis, or peritonitis. In 2019, Carpenter et al. investigated the prevalence of infection and venous thromboembolism in hospitalized pediatric patients with NS (9). This group demonstrated high rates of infection in hospitalized pediatric NS patients, with Streptococcus pneumoniae being the most common pathogen. Appreciating the infectious susceptibility associated with nephrotic syndrome is critical since improved vaccination practices would potentially aid in the prevention of many of these infections (10, 11).
This raises the question of whether children with NS are receiving the recommended vaccinations at appropriate times. A critical first step in this process is to understand the practices and patient education provided by physicians to patients on the topic of vaccinations. The aims of this are: (1) evaluate the immunization practices among pediatric nephrologists, (2) assess the education provided to families of children with NS by the pediatric nephrology providers, (3) assess the parental knowledge and understanding of these immunization recommendations, and (4) assess predictors of PPSV 23 adherence.
Methods
Parents/guardians of pediatric patients (<18 years old) diagnosed with NS from 2005 to 2018 from 11 institutions in the Pediatric Nephrology Research Consortium and Kidney Research Network participated in the study. Patient inclusion criteria were children <18 years old with primary NS diagnosed between 2005 and 2018 who were seen in the pediatric nephrology practice at the participating center on at least one occasion. All pediatric nephrologists at the participating study centers were also included in the study. A parent/guardian survey was distributed, and a separate pediatric nephrology provider survey was distributed to pediatric nephrologists at each center. This study was performed in line with the principles of the Declaration of Helsinki. Institutional Review Boards of each participating center approved the study. Consent and assent were obtained per institutional guidelines.
Survey and Measures
The parent/guardian survey contained 24 questions which were created by the authors and included demographic data, immunosuppression history, parental knowledge of immunization recommendations and their intent to follow the recommendations, and potentially vaccine-preventable hospitalizations for infections. The provider survey contained 21 questions which were created by the authors and included demographic data, provider immunization recommendation practices, knowledge of the current immunization guidelines, and hospitalizations for infections at any time during their practice that may have been vaccine-preventable. Family caregivers/study coordinators provided redacted copies of patient immunization records. Validation studies of self-administered surveys have shown this type of survey methodology to be a promising tool across medical disciplines (12–14). Surveys were self-administered by the families and providers in the clinic office. The surveys are provided in the Supplementary Material 1, 2.
The Advisory Committee on Immunization Practices (ACIP) Recommended Child and Adolescent Immunization Schedule was used to determine accuracy of parent and provider knowledge of current immunization guidelines (Figure 1) (15). In brief summary, the ACIP recommends inactive vaccines for any pediatric patient who is not receiving immunosuppression medications as well as those on most immunosuppression therapies. There is an exception to consider deferring inactive vaccines for 6 months after receiving B-cell depleting therapy, or may consider administration of inactive vaccines while receiving B-cell depleting therapy with assessment of titers after discontinuation of the B-cell depleting agent. With regards to live vaccines, the ACIP recommends administration of live vaccines in patients who are not on immunosuppression therapy; patients who have received <14 days of high dose corticosteroids; and patients who have been off of Calcineurin inhibitors (CNI)/Mycophenolate Mofetil (MMF) for 2 months, off of cytotoxic agents (i.e., Cyclophosphamide) for 3 months, and off of B-cell depleting agents (i.e., Rituximab) for 6 months. A more detailed summary of this guideline is provided in Supplementary Material 3.
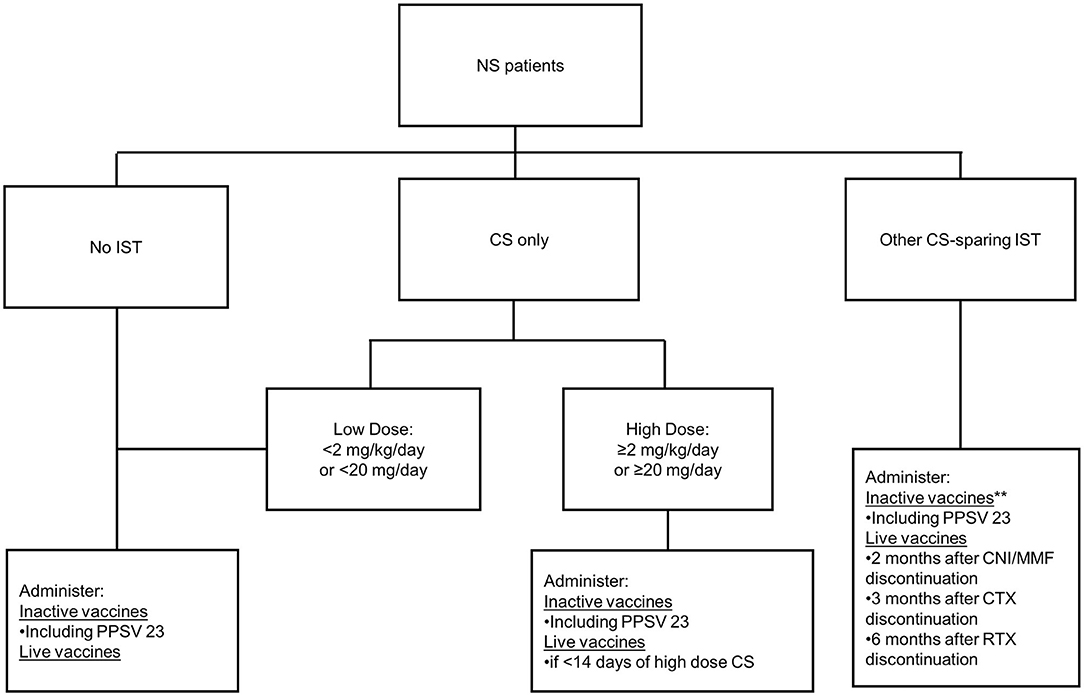
Figure 1. Flow diagram of abbreviated vaccine guidelines. Information for this was adapted from the Advisory Committee on Immunization Practices 2019 vaccine guidelines. Abbreviations: NS, nephrotic syndrome; IST, immunosuppression; CS, corticosteroids; PPSV 23, polysaccharide pneumococcal vaccine; CNI, calcineurin inhibitor; MMF, mycophenolate mofetil; CTX, cyclophosphamide; RTX, rituximab. **Defer 6 months after RTX discontinuation, or administer if no finite endpoint for RTX and assess titers after discontinuation of RTX.
Patient demographic variables included state of residence, patient age, race, parent/guardian level of education, and household income. Provider demographic variables included institution, type of practice (academic vs. private), type of subspecialty training (Pediatric Nephrology, Internal Medicine-Pediatric Nephrology, or Internal Medicine Nephrology), and percent effort allocated to clinical care.
Statistical Analysis
Descriptive statistics included frequencies for categorical variables and mean ± standard deviation (SD) or median (IQR) for continuous variables, as appropriate. Fischer's exact tests were used to compare knowledge and guidelines across regions, and evaluate predictors of PPSV 23 vaccination. Multivariable logistic regression was used to test independent predictors of PPSV 23 vaccination. Variables with unadjusted p < 0.2 entered a multivariable model and were removed in descending order until all remaining terms were significant. For all analyses, p ≤ 0.05 were considered statistically significant.
Results
Study Population Demographics
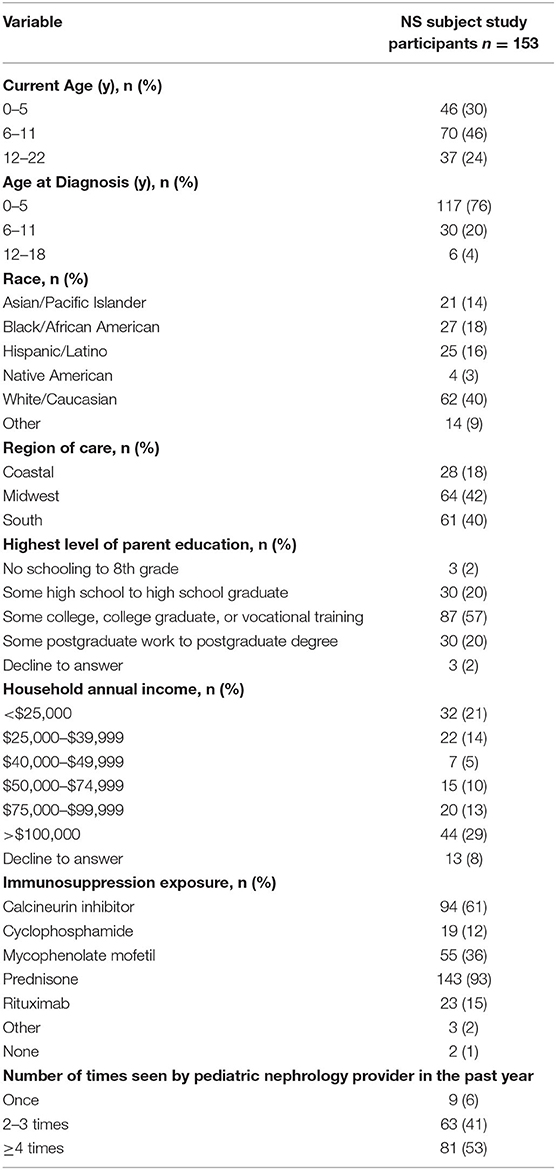
Between November 2015 and December 2018, 175 pediatric patients diagnosed with NS between January 2005 and December 2018, from 11 North American centers, were eligible for the study, of which 153 parents/guardians of pediatric subjects consented to the study and completed the anonymous survey (parent/guardian response rate 87.4%). Fifty-eight pediatric nephrologists at participating centers were provided an anonymous survey, of which 50 (86.2%) completed the survey. All subjects were stratified by region [Coastal (3 centers), Midwest (4 centers), and South (4 centers)]. Due to the small number of study centers on either coast, the current study grouped these centers into one “Coastal” region to avoid identifying a single site and its study participants.
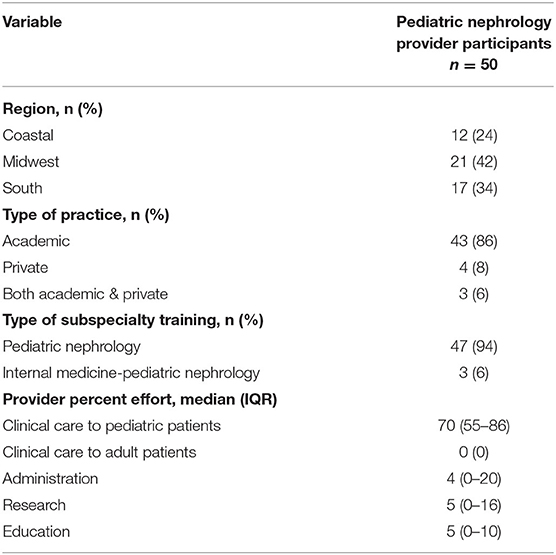
At study enrollment (Table 1a), the median age at NS diagnosis was 3 years of age (IQR 2-5); nearly half of the pediatric patients (46%) were 6–11 years of age at study enrollment; 40% were White; majority of parents/guardians had some post high school education (77%); and nearly all pediatric patients had exposure to immunosuppression (99%). In the pediatric nephrology provider population (Table 1b), the majority practiced in an academic setting (86%); majority trained specifically in Pediatric Nephrology (94%); and all provided care to pediatric patients with the median percent effort allocated to clinical care of 70% (IQR 55–86%).
Pediatric Nephrology Provider Vaccine Recommendations
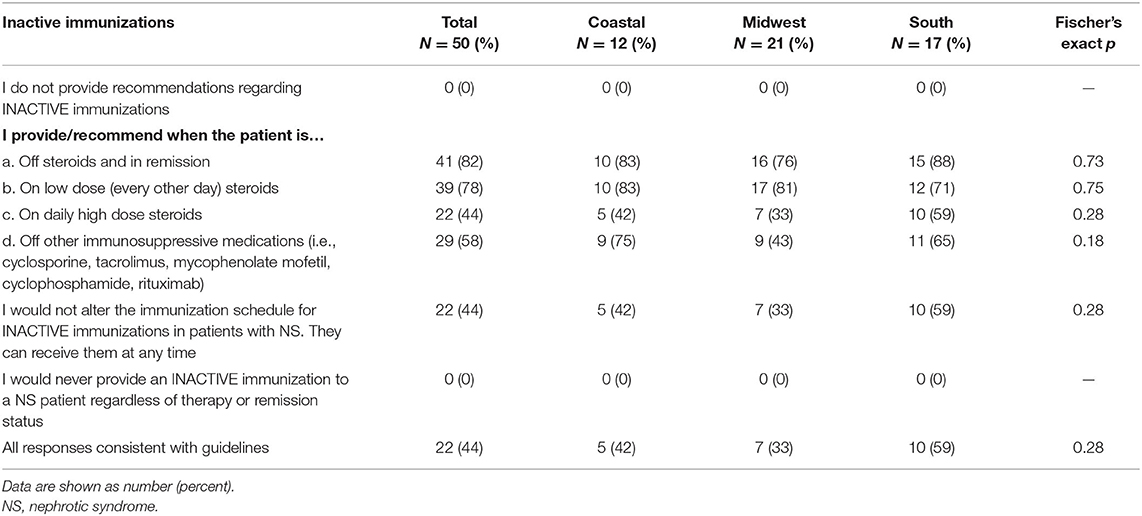
Table 2 displays the pediatric nephrology provider recommendations for inactive vaccines. All providers indicated that they provide recommendations for inactive vaccines to patients. Eighty-two percent recommended inactive vaccines while off of steroids and in remission, and 78% recommended inactive vaccines on low dose steroids. However, only 44% recommended inactive vaccines while on high dose steroids with 58% recommending inactive vaccines when off of other immunosuppressive medications. Only 44% recommended inactive vaccines at any time regardless of immunosuppression status. Overall, 56% of providers had at least one response inconsistent with guidelines.
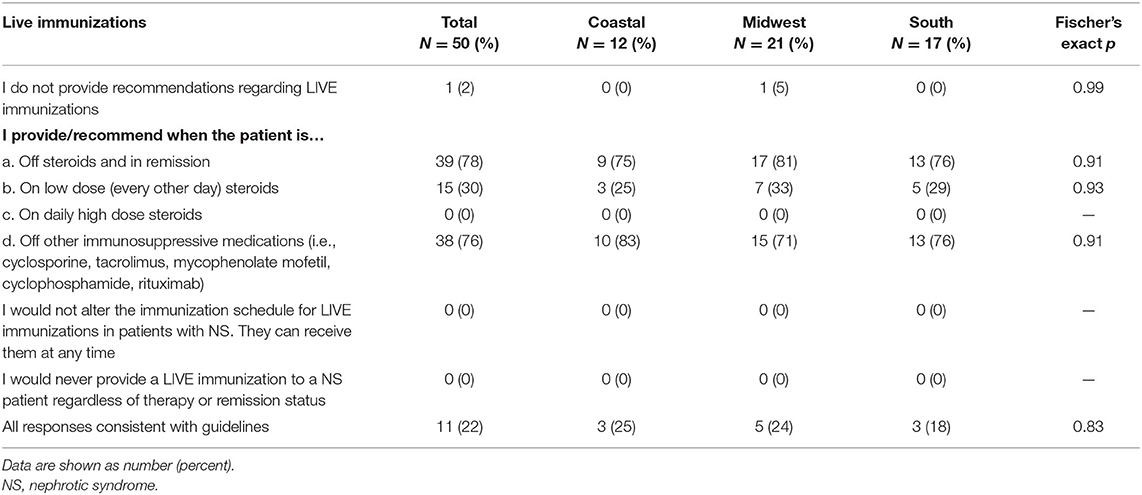
Table 3 displays the pediatric nephrology provider recommendations for live vaccines. Of the 50 providers surveyed, only 1 (2%) indicated they did not provide live vaccine recommendations to patients. Seventy-eight percent recommended live vaccines while off steroids and in remission; however, only 30% recommended live vaccines while on low dose steroids. All providers recommended avoiding live vaccines while on daily high dose steroids, and 76% recommended receiving live vaccines while off other immunosuppressive medication. Overall, only 22% of provider's responses were in complete agreement with the ACIP guidelines for live vaccines. The main deviance from the ACIP guidelines was with respect to low dose steroids. Of the 39 providers that did not follow guidelines, 35 (90%) indicated they would not recommend live vaccines while on low dose steroids.
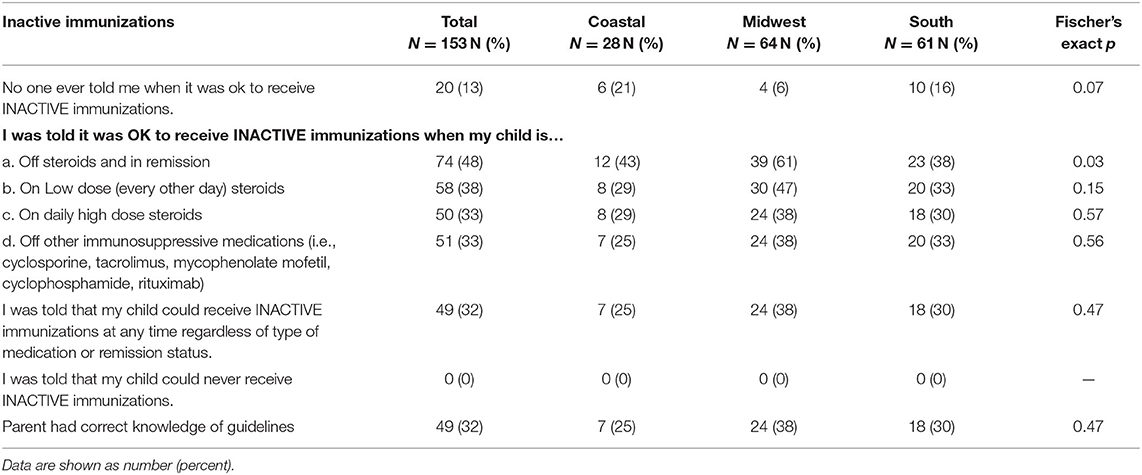
Pediatric NS Parent/Guardian Vaccine Knowledge
Table 4 shows the parent/guardian knowledge of recommended inactive vaccines. Out of 153 respondents, 13% indicated that no one told them when it was acceptable to receive inactive immunizations. Of the remaining, only 48% indicated that inactive vaccines could be administered while off steroids and in remission with 38% indicating that inactive vaccines could be given on low dose steroids. Only 33% indicated that inactive vaccines could be administered while on high dose steroids with 33% indicating that inactive vaccines could be given when off other immunosuppressive medications. Overall, 32% of parents/guardian responses suggested understanding of immunization practices that aligned with ACIP guidelines for inactive vaccines.
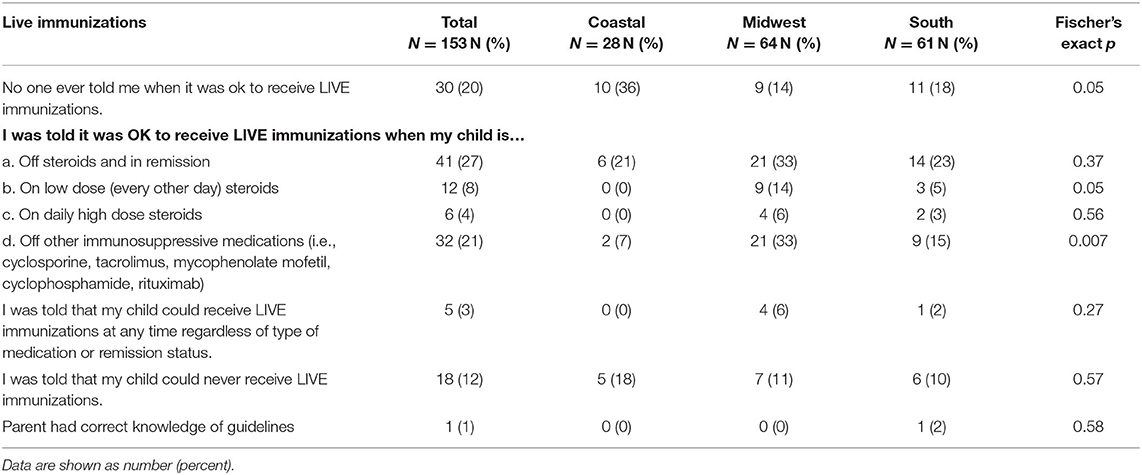
Table 5 shows the parent/guardian knowledge of recommended live vaccines. Twenty percent of parents/guardians indicated that no one told them when it was acceptable to receive live immunizations. Of the remaining, 27% indicated that live vaccines could be administered while off steroids and in remission; 8% indicated that live vaccines could be given while on low dose steroids; and 4% of parents/guardians indicated that live vaccines could be administered on high dose steroids. Twenty-one percent indicated that live vaccines could be administered when off other immunosuppression medications. Overall, 1% of parent/guardian responses suggested an understanding of current live vaccination best practices.
Pediatric NS Patient Immunization Status Validation and Predictors
One-hundred and 52 parents/guardians of pediatric NS subjects (99%) indicated that their child had been vaccinated and one pediatric NS subject (1%) had never been vaccinated. Records of vaccinations were available from 141 (93%) participants for validation of immunization status. Immunization status was considered up to date if the subject received all ACIP recommended vaccines based on immunosuppression status, age at time of survey, and vaccine release date. One-hundred and twenty-two subjects (87%) were up to date on the recommended vaccines. Fifty-three percent received the 7-valent pneumococcal vaccine, 71% received the 13-valent pneumococcal vaccine, and 48% received the PPSV 23.
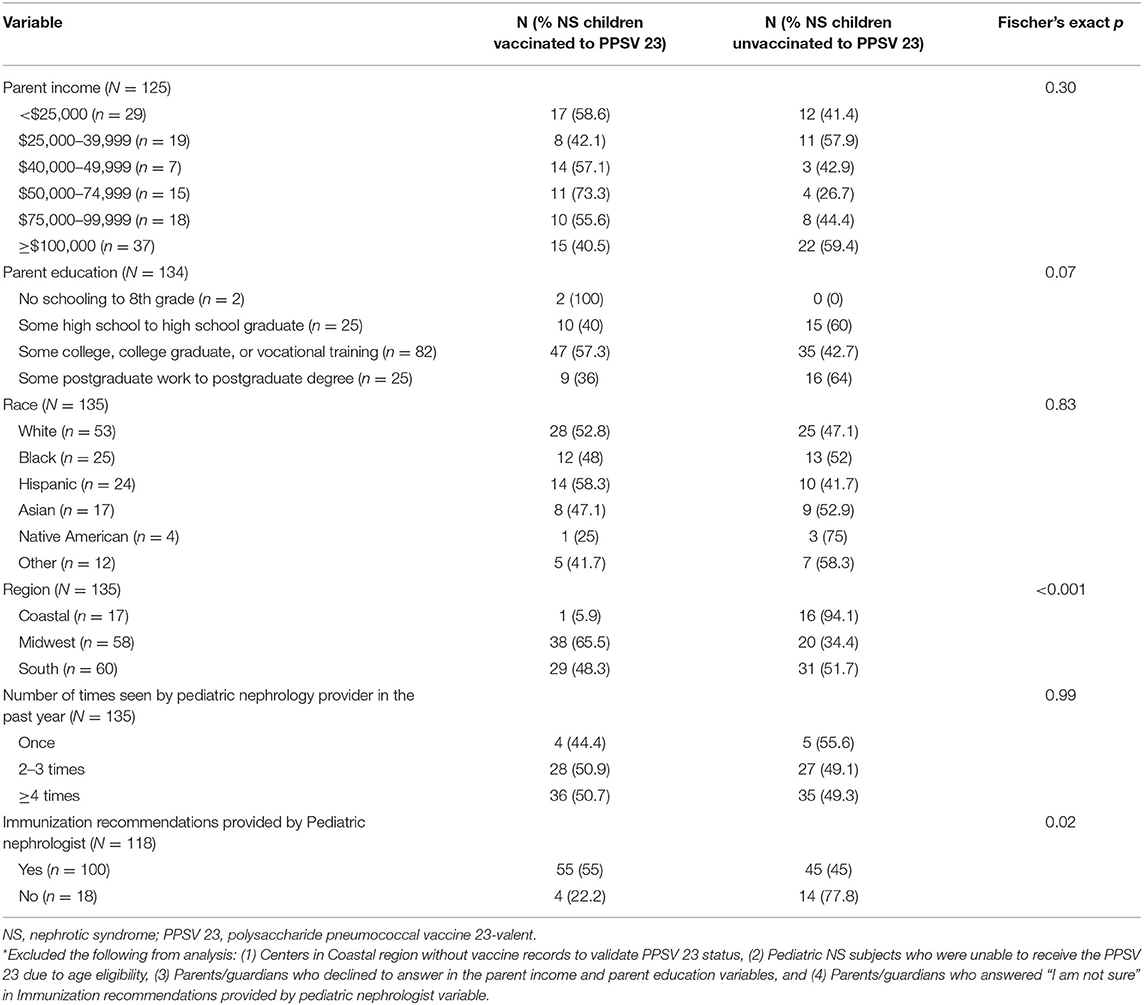
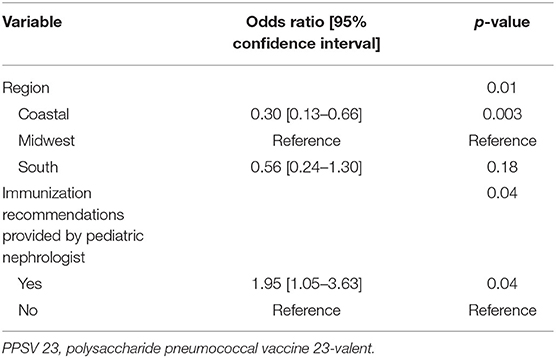
Subjects residing in the Midwest was a positive predictor of subjects receiving the PPSV 23 (p < 0.001). Additionally, receiving immunization recommendations from the pediatric nephrology provider was a positive predictor of subjects receiving the PPSV 23 as well (p 0.02) (Table 6). Multivariable logistic regression of the differences in Table 6 revealed that region and immunization recommendations were independent predictors of vaccinations. Those in the Coastal region were less likely to be vaccinated than the Midwest (OR = 0.30 95% CI = 0.13–0.66) and those with recommendations from the pediatric nephrologist were more likely to be vaccinated than those without recommendations (OR = 1.95 95% CI = 1.05–3.63) (Table 7). When provider responses were reviewed, providers in the Coastal region were less likely to review immunization records on a periodic basis (p < 0.01) and were more likely to state that they were unsure if they would recommend the PPSV23 if the patient had previously received the 7-valent pneumococcal conjugate vaccine (PCV) or PCV-13 (p 0.03) (Supplementary Material 4).
Discussion
While there have been published studies investigating vaccine practices and vaccine adherence patterns, these studies have evaluated the medical provider population alone or the parent population alone. Our study investigates both the subspecialty medical provider and parent population from the same institution and region simultaneously. This study demonstrates gaps in vaccine knowledge of both pediatric nephrology providers and parents of children with NS who are immunosuppressed.
The decision to vaccinate a child can be influenced by parental knowledge, parental understanding of the benefits of vaccination, provider knowledge, and other complex factors related to their child's underlying disease and treatment. Deficiencies in vaccinations directly impact the health of the individual child and of the larger pediatric community if herd protection is not achieved due to poor vaccine adherence. Children most at risk for severe vaccine-preventable infections are those who are immunocompromised, which includes children with NS.
The Center for Disease Control (CDC), ACIP, and the Committee on Infectious Diseases of the American Academy of Pediatrics (AAP) release an updated recommended immunization schedule for healthy children annually (16). This immunization schedule is available to medical providers online (http://www.cdc.gov/vaccines/recs/schedules/child-schedule.htm). In addition to the yearly updated recommendations, health care providers should be aware of guidelines for vaccinating immunocompromised patients, which are readily available online (https://www.cdc.gov/vaccines/hcp/acip-recs/general-recs/immunocompetence.html and https://www.cdc.gov/vaccines/schedules/downloads/child/0-18yrs-child-combined-schedule.pdf). Despite these guidelines and published comprehensive reviews outlining vaccine guidelines in various immunosuppressed states, there remains a discrepancy in vaccine practices among pediatric nephrologists in the management of children with NS on immunosuppressive therapy (17–19).
The discrepancy in vaccine practice amongst providers and low compliance in following ACIP guidelines is likely multifactorial. It could be in part due to the provider's misinterpretation of vaccine guidelines due to the complex immunosuppression regimens that these NS patients may require, or the provider's disagreement of vaccination guidelines based on anecdotal experience with NS relapses and/or belief that immunosuppressed patients may not mount a robust vaccine response. In this study, only 44% of provider responses were in complete alignment with the ACIP guidelines for inactive vaccines and 22% for live vaccines. The CDC general principles for those with altered immunocompetence, state that inactivated vaccines could be deferred during a time of immunosuppression since vaccines may be less effective during this period. However, if an inactivated vaccine is given during the time that a patient is immunosuppressed, then the inactive vaccine may need to be repeated when immune function has improved. Yet, the CDC guidelines emphasize the need for immunocompromised patients to receive certain inactive conjugated and polysaccharide-based vaccines (i.e., pneumococcal vaccines, Haemophilus influenzae type b, and meningococcal vaccines) due to the increased risk for disease if the vaccine is withheld. Live vaccines are typically deferred until immune function has improved due to the risk of uninhibited growth of the attenuated live virus or bacteria in those with altered immunocompetence (20–23). It is clear that the vaccine guidelines for those who are immunosuppressed can be nuanced.
Since 80% of pediatric patients with new onset NS are steroid-sensitive and eventually taper off of steroid therapy, some pediatric nephrologists may opt to withhold vaccines until these patients have return of immune function to help mount a more effective response to the vaccine (24). In our study, 44% of the pediatric nephrologists surveyed reported withholding inactive vaccines when a NS patient is on any dose of steroids due to concern that the patient may not mount an immune response. Ten percent of the pediatric nephrologists surveyed observed a NS relapse after immunization administration. However, published studies have demonstrated vaccine efficacy in NS patients who are immunosuppressed. Aoun and Ulinski exhibited good serologic response to the PPSV 23 in children with NS on high dose prednisone in the short term and in the long term (25). They also demonstrated that the long term pneumococcal antibody response was not impacted by other immunosuppressive agents (26). Hsu et al. demonstrated good vaccine efficacy of the pneumococcal vaccine in children with NS on high dose steroids and no differences in rate of NS relapse (27). In addition, other inactive polysaccharide-based vaccines, such as meningococcal C conjugate vaccine has not been shown to be associated with increased NS relapses (28). Other studies have demonstrated that the Varicella vaccine has been well-tolerated in children with NS and highly immunogenic in those on low-dose, every other day prednisone (29, 30). Despite these groups demonstrating vaccine efficacy without differences in NS relapse rates, there has been a wide variation of vaccine practices as evidenced by two studies conducted in 1993 and 2001 (17, 18). Our study similarly observed varied vaccine practices by providers, which translates to the gap in parental knowledge and understanding of the timing of vaccine administration in their children with NS as seen in the parental/guardian survey responses.
NS can be a difficult disease to treat especially in patients who have a steroid-dependent, frequently-relapsing, or steroid-resistant clinical course. These patients may have no or very brief periods of time off of immunosuppression medications to keep the disease in control. As a result, opportunities to vaccinate with inactive and/or live vaccines become limited if providers choose to withhold immunizations until full recovery of immune function. Withholding vaccines in this high-risk population can lead to an increased risk of vaccine-preventable disease (9). It is known that infection is a common trigger for NS relapses which can result in the need for hospitalization to help manage the complications of the relapse, thus leading to significantly higher hospitalization charges (8).
Factors associated with immunization adherence have been studied by various groups. Our group specifically evaluated the adherence to PPSV23 administration since it is recommended by the ACIP for pediatric patients with NS and immunosuppressed states. Cost of vaccines, lack of parental knowledge of the benefits of vaccines, and lack of vaccine recommendation by medical providers has been reported to adversely impact immunization adherence (31, 32). Our study similarly showed that provider vaccine recommendations were associated with vaccine adherence to PPSV 23. However, we did not investigate the cost of vaccines as a potential factor of immunization adherence in this survey study. Schuller et al. demonstrated an increased likelihood of vaccination if the child's caregiver received higher education, lived in the Northeast, had private insurance, and was of Hispanic race among US children (33). We found that subjects residing in the Midwest were more likely to receive the PPSV 23, which may be due to a higher number of participating study sites in the Midwest region compared to other regions surveyed in North America for our study, and therefore the Midwest region may be more representative than a region with fewer sites. Additionally, parent income, level of parent education, and race were not positive predictors of subjects receiving the PPSV 23 in the current study. The differences in the factors associated with vaccines adherence in our study compared to other published studies may also be explained by our small sample size. Our study also evaluated other possible contributing factors to vaccine adherence including the ability of the subspecialty clinic to provide vaccines (i.e., pneumococcal vaccine) and the communication of vaccine recommendations from the pediatric nephrology provider to the patient's primary care provider (PCP). Seventy-six percent of providers indicated that their subspecialty clinic can administer pneumococcal vaccines. The remaining providers who do not have this capability may defer the responsibility of vaccinations to the PCP. Ninety-two percent of providers indicated that they communicate their vaccine recommendations to their patient's PCP with a letter as the most common modality of communication.
The current study demonstrates the need for a more uniform vaccine practice in the pediatric NS population among pediatric nephrologists, which will in turn better educate and help parents/guardians understand the importance of vaccination and when it is appropriate for their child to receive certain vaccines.
Limitations and Strengths
The limitations of this study include the heterogeneity of survey responses among the parent/guardian cohort and pediatric nephrology providers with regards to knowledge of the ACIP guidelines. The discrepant responses amongst both groups could be explained by comprehension and communication between parent and provider or alternatively by survey interpretation. Since the surveys were anonymous for both cohorts, we were unable to associate the parent/guardian and provider response resulting in the inability to directly correlate responses. While this study evaluated the communication of vaccine recommendations from pediatric nephrologist to PCP, an assessment of the knowledge and practice of the patient's PCP was not evaluated which could have identified another area for improvement of vaccine adherence in this cohort. Lastly, the small sample size of this study may have played a role in the differences in factors associated with vaccine adherence compared to other published studies.
The strengths of this study include the high response rate. This study also investigated the immunization knowledge from both pediatric nephrology providers and parent/guardians of children with NS simultaneously from their shared institutions, which helped to evaluate for any institution/region-specific immunization practices.
Conclusion
Our findings support the growing evidence that vaccine recommendation by medical providers is paramount in vaccine adherence among pediatric patients with NS. The disparate survey responses among the pediatric nephrology providers and parents/guardians likely reflect the individual interpretations of the ACIP guidelines by the medical providers and possibly their anecdotal experiences with treatment of NS. Additionally, some pediatric nephrology practices do not have the capability to administer certain vaccines (i.e., PPSV 23) and therefore rely on vaccinations through the patient's PCP. Ensuring primary care establishment with a PCP as well as being a champion to clarify and communicate vaccination provisioning guidance with the patient's PCP may help to improve vaccine adherence. Future studies assessing the knowledge and practice of vaccine recommendations by PCPs for pediatric patients with NS may also highlight other areas for improvement of vaccine adherence such as communication between healthcare providers to assure vaccinations occur in a timely and complete fashion in the child's primary care office. Lastly, it is important that the ACIP immunization guidelines are reviewed by the pediatric nephrologist and primary care physicians yearly to be informed of any change in the immunization practice recommendations for those with altered immune competence.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
Ethics Statement
The studies involving human participants were reviewed and approved by each participating center. Written informed consent to participate in this study was provided by the participants' legal guardian/next of kin.
Author Contributions
Data analysis was performed by JT and CT. All authors read and approved the final manuscript, contributed to the study conception, design, and data collection.
Funding
Analytic work for this study was supported in-part by the National Center for Advancing Translational Sciences (NCATS Grant Number: UL1TR002240) for the Michigan Institute for Clinical and Health Research.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We are indebted to the patients and families who graciously participated in this research. We express our thanks to all the site coordinators who assisted with patient/provider identification, enrollment, and data collection.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fped.2020.619548/full#supplementary-material
Abbreviations
ACIP, Advisory Committee on Immunization Practices; AAP, American Academy of Pediatrics; CDC, Center for Disease Control; NS, nephrotic syndrome; PPSV 23, pneumococcal polysaccharide vaccine; PCP, primary care provider.
References
1. Peterson PA, Berggard I. Urinary immunoglobulin components in normal, tubular, and glomerular proteinuria: quantities and characteristics of free light chains, IgG, IgA, and fcgamma fragment. Eur J Clin Invest. (1971) 1:255–64.
2. Giangiacomo J, Cleary TG, Cole BR, Hoffsten P, Robson AM. Serum immunoglobulins in the nephrotic syndrome. A possible cause of minimal-change nephrotic syndrome. N Engl J Med. (1975) 293:8–12.
3. Anderson DC, York DL, Rose G, Smith CW. Assessment of serum factor B. serum opsonins, granulocyte chemotaxis, and infection in nephrotic syndrome of children. J Infect Dis. (1979) 140:1–11.
4. Ballow M, Kennedy TL, Gaudio KM, Siegel NJ, McLean RH. Serum hemolytic factor D values in children with steroid-responsive idiopathic nephrotic syndrome. J Pediatr. (1982) 100:192–6.
5. Uncu N, Bülbül M, Yildiz N, Noyan A, Koşan C, Kavukçu S, et al. Primary peritonitis in children with nephrotic syndrome: results of a 5-year multicenter study. Eur J Pediatr. (2010). 169:73–6. doi: 10.1007/s00431-009-0989-x
6. Speck WT, Dresdale SS, McMillan RW. Primary peritonitis and the nephrotic syndrome. Am J Surg. (1974) 127:267–9.
7. Gorensek MJ, Lebel MH, Nelson JD. Peritonitis in children with nephrotic syndrome. Pediatrics. (1988) 81:849–56.
8. Gipson DS, Messer KL, Tran CL, Herreshoff EG, Samuel JP, Massengill SF, et al. Inpatient health care utilization in the United States among children, adolescents, and young adults with nephrotic syndrome. Am J Kidney Dis. (2013) 61:910–7. doi: 10.1053/j.ajkd.2012.12.025
9. Carpenter SL, Goldman J, Sherman AK, Selewski DS, Kallash M, Tran CL, et al. Association of infections and venous thromboembolism in hospitalized children with nephrotic syndrome. Pediatr Nephrol. (2019). 34:261–7. doi: 10.1007/s00467-018-4072-6
10. Fiore AE, Levine OS, Elliott JA, Facklam RR, Butler JC. Effectiveness of pneumococcal polysaccharide vaccine for preschool-age children with chronic disease. Emerg Infect Dis. (1999) 5:828–31.
11. Overturf GD. American academy of pediatrics. Committee on infectious diseases. Technical report: prevention of pneumococcal infections, including the use of pneumococcal conjugate and polysaccharide vaccines and antibiotic prophylaxis. Pediatrics. (2000) 106:367–76. doi: 10.1542/peds.106.2.367
12. Muntner P, Woodward M, Carson AP, Judd SE, Levitan EB, Mann DM, et al. Development and validation of a self-assessment tool for albuminuria: results from the reasons for geographic and racial differences in stroke (REGARDS) study. Am J Kidney Dis. (2011) 58:196–205. doi: 10.1053/j.ajkd.2011.01.027
13. Chehab G, Sander O, Richter J, Acar H, Vordenbäumen S, Brinks R, et al. Validation and evaluation of the German brief index of lupus damage (BILD)–a self-reported instrument to record damage in systemic lupus erythematosus. Lupus. (2013) 22:1050–5. doi: 10.1177/0961203313500369
14. Andy UU, Harvie HS, Smith AL, Propert KJ, Bogner HR, Arya LA. Validation of a self-administered instrument to measure adherence to anticholinergic drugs in women with overactive bladder. Neurourol Urodyn. (2015) 34:424–8. doi: 10.1002/nau.22605
15. ACIP. https://www.cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html. 2019.
16. Robinson CL, Bernstein H, Poehling K, Romero JR, Szilagyi P. Advisory committee on immunization practices recommended immunization schedule for children and adolescents aged 18 years or younger-United States 2020. MMWR Morb Mortal Wkly Rep. (2020) 69:130–2. doi: 10.15585/mmwr.mm6905a3
17. Schnaper HW. Immunization practices in childhood nephrotic syndrome: a survey of North American pediatric nephrologists. Pediatr Nephrol. (1994) 8:4–6.
18. Shroff A, Frank R, Vergara M, Gauthier B, Trachtman H. Prevention of serious bacterial infections in new-onset nephrotic syndrome: a survey of current practices. Clin Pediatr. (2002) 41:47–9. doi: 10.1177/000992280204100110
19. Banerjee S, Dissanayake PV, Abeyagunawardena AS. Vaccinations in children on immunosuppressive medications for renal disease. Pediatr Nephrol. (2016) 31:1437–48. doi: 10.1007/s00467-015-3219-y
20. Sixbey JW. Routine immunizations and the immunosuppressed child. Adv Pediatr Infect Dis. (1987) 2:79–114.
21. Davis LE, Bodian D, Price D, Butler IJ, Vickers JH. Chronic progressive poliomyelitis secondary to vaccination of an immunodeficient child. N Engl J Med. (1977) 297:241–5.
22. Ninane J, Grymonprez A, Burtonboy G, Francois A, Cornu G. Disseminated BCG in HIV infection. Arch Dis Child. (1988) 63:1268–9.
23. Lu Y, Bousvaros A. Varicella vaccination in children with inflammatory bowel disease receiving immunosuppressive therapy. J Pediatr Gastroenterol Nutr. (2010) 50:562–5. doi: 10.1097/MPG.0b013e3181bab351
24. Lombel RM, Gipson DS, Hodson EM. Treatment of steroid-sensitive nephrotic syndrome: new guidelines from KDIGO. Pediatr Nephrol. (2013) 28:415–26. doi: 10.1007/s00467-012-2310-x
25. Aoun B, Wannous H, Azéma C, Ulinski T. Polysaccharide pneumococcal vaccination of nephrotic children at disease onset-long-term data. Pediatr Nephrol. (2010) 25:1773–4. doi: 10.1007/s00467-010-1492-3
26. Ulinski T, Leroy S, Dubrel M, Danon S, Bensman A. High serological response to pneumococcal vaccine in nephrotic children at disease onset on high-dose prednisone. Pediatr Nephrol. (2008) 23:1107–13. doi: 10.1007/s00467-008-0782-5
27. Hsu K, Pelton S, Karumuri S, Heisey-Grove D, Klein J. Population-based surveillance for childhood invasive pneumococcal disease in the era of conjugate vaccine. Pediatr Infect Dis J. (2005). 24:17–23. doi: 10.1097/01.inf.0000148891.32134.36
28. Taylor B, Andrews N, Stowe J, Hamidi-Manesh L, Miller E. No increased risk of relapse after meningococcal C conjugate vaccine in nephrotic syndrome. Arch Dis Child. (2007). 92:887–9. doi: 10.1136/adc.2006.105247
29. Quien RM, Kaiser BA, Deforest A, Polinsky MS, Fisher M, Baluarte HJ. Response to the varicella vaccine in children with nephrotic syndrome. J Pediatr. (1997) 131:688–90.
30. Furth SL, Arbus GS, Hogg R, Tarver J, Chan C, Fivush BA. Varicella vaccination in children with nephrotic syndrome: a report of the Southwest pediatric nephrology study group. J Pediatr. (2003) 142:145–8. doi: 10.1067/mpd.2003.37
31. How CH, Phua See Chun P, Shafi F, Jakes RW. Parental knowledge, attitudes and perception of pneumococcal disease and pneumococcal conjugate vaccines in Singapore: a questionnaire-based assessment. BMC Public Health. (2016) 16:923. doi: 10.1186/s12889-016-3597-5
32. Low MSF, Tan H, Hartman M, Tam CC, Hoo C, Lim J, et al. Parental perceptions of childhood seasonal influenza vaccination in Singapore: a cross-sectional survey. Vaccine. (2017) 35:6096–02. doi: 10.1016/j.vaccine.2017.09.060
Keywords: nephrotic syndrome, children, immunization, immunosuppression, education
Citation: Tran CL, Selewski DT, Oh GJ, Troost JP, Massengill SF, Al-Akash SI, Mahesh S, Amin R, Ashoor IF, Chanchlani R, Kallash M, Woroniecki RP and Gipson DS (2021) Pediatric Immunization Practices in Nephrotic Syndrome: An Assessment of Provider and Parental Knowledge. Front. Pediatr. 8:619548. doi: 10.3389/fped.2020.619548
Received: 20 October 2020; Accepted: 24 December 2020;
Published: 05 February 2021.
Edited by:
Tim Ulinski, Hôpital Armand Trousseau, FranceReviewed by:
Christopher Esezobor, University of Lagos, NigeriaSe Jin Park, Yonsei University, South Korea
Copyright © 2021 Tran, Selewski, Oh, Troost, Massengill, Al-Akash, Mahesh, Amin, Ashoor, Chanchlani, Kallash, Woroniecki and Gipson. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Cheryl L. Tran, dHJhbi5jaGVyeWwyQG1heW8uZWR1
 Cheryl L. Tran
Cheryl L. Tran David T. Selewski
David T. Selewski Gia J. Oh3
Gia J. Oh3 Samhar I. Al-Akash
Samhar I. Al-Akash Rasheda Amin
Rasheda Amin Mahmoud Kallash
Mahmoud Kallash Robert P. Woroniecki
Robert P. Woroniecki