
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Oncol. , 27 March 2025
Sec. Hematologic Malignancies
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1530852
This article is part of the Research Topic Combination Therapies in Acute Myeloid Leukemia (AML). View all 4 articles
The prognosis of MECOMr AML is poor, with a 5-year overall survival (OS) rate of less than 10%. This is mostly attributable to the low efficacy of all available therapies and high relapse rates even after allogeneic stem cell transplantation (alloSCT), which remains the only curative approach. We report upfront sequential alloSCT with venetoclax-based preconditioning as a safe and effective treatment for two newly diagnosed and fit MECOMr AML patients. The sequential alloSCT regimen consisted of triple therapy: preconditioning with decitabine and venetoclax on days 1–5 followed by FLAG-Ida and venetoclax on days 6–10. One or 3 days after preconditioning, the patients underwent busulfan-based myeloablative conditioning and HLA haploidentical or matched related donor stem cell infusion. One month after alloSCT, timely engraftment and complete remission were achieved. At the last follow-up, both patients were in good health and in MRD-negative complete remissions after 11 and 17 months after alloSCT, respectively. The safety and efficacy of upfront sequential alloSCT indicate the need to evaluate this approach for adverse risk of AML in clinical trials.
Ecotropic viral integration site 1 and myelodysplastic syndrome 1 complex rearranged acute myeloid leukemia (MECOMr AML) is a rare and distinct entity, accounting for only 1%–1.6% of all newly diagnosed AML cases. The pathogenesis mostly involves a fusion of GATA2::MECOM (EVI1) due to inversion or translocation of chromosome 3 [inv(3;3)/t(3;3)] resulting in malignant transcriptomic aberrations; however, more than 120 different chromosomal aberrations involving MECOM have been reported (1–6). The prognosis of MECOMr AML is poor, with a 5-year overall survival (OS) rate of less than 10% (2, 3, 5, 7, 8). This is mostly attributable to the low efficacy of all available therapies and high relapse rates even in the setting of allogeneic stem cell transplantation (alloSCT), which remains the only curative approach (2, 3, 5, 7–9). Currently, no targeted therapies directly act upon MECOM leukemogenic rearrangement. There is a high unmet need for more effective treatments and bridging to alloSCT strategies in this poor-risk genomic subgroup of patients with AML. Herein, we report upfront sequential alloSCT with venetoclax-based preconditioning as a safe and effective treatment for two newly diagnosed MECOMr AML patients.
Two patients with newly diagnosed MECOMr AML underwent sequential alloSCT as frontline therapy. Both patients provided informed consent for data sharing, and the treatment was approved by the Institutional Ethics Committee. We collected baseline characteristics, detailed AML genomics, alloSCT-related parameters, response evaluation, measurable residual disease (MRD) data, overall survival, remission duration, time to neutrophil (ANC) and platelet (PLT) engraftment, grades 3–5 non-hematological toxicity according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0
The first part of the frontline sequential triple treatment consisted of cytoreductive hypomethylation/priming with 5-day Dec + Ven (decitabine 20 mg/m2 + venetoclax 400 mg/day, on days 1–5). On day 6, FLAG-Ida + Ven was initiated (fludarabine 30 mg/m2, cytarabine 2,000 mg/m2, G-CSF 5 mcg/kg, and venetoclax 400 mg/day, on days 6–10, idarubicin 8 mg/m2, on days 8–10). After a washout period of 1 or 3 days after preconditioning with Dec + Ven and FLAG-Ida + Ven, both patients underwent a 7-day busulfan-based myeloablative conditioning (MAC) and stem cell infusion with standard-of-care graft-versus-host disease (GVHD) prophylaxis and post-transplant care. (Figure 1)
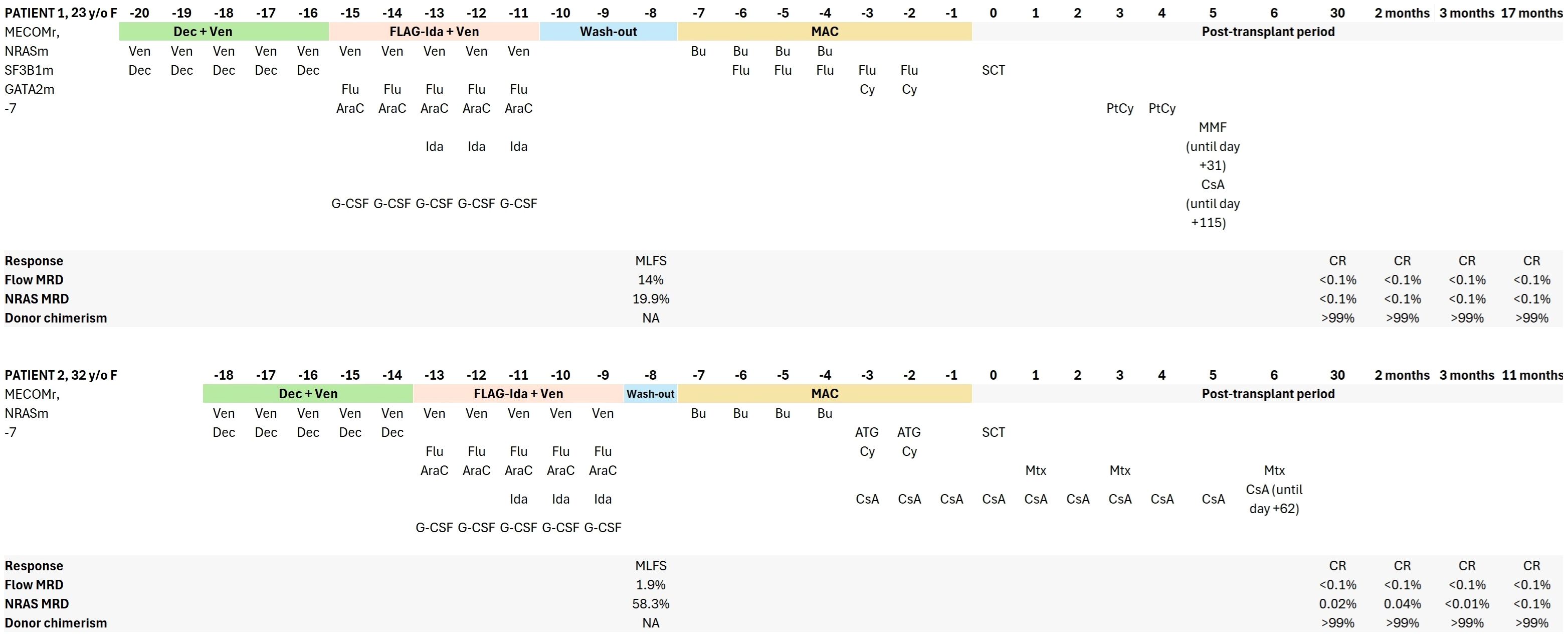
Figure 1. Detailed treatment and response evaluation timeline of patients 1 and 2. F, female; CR, complete remission; MLFS, morphological leukemia-free state; MRD, measurable residual disease; Flu, fludarabine; Dec, decitabine; AraC, cytarabine; Ven, venetoclax; Bu, busulfan; Ida, idarubicin; CsA, cyclosporine A; MMF, mycophenolate mofetil; Cy, cyclophosphamide; PtCy, post-transplant cyclophosphamide; MAC, myeloablative conditioning; ATG, anti-thymocyte globulin; MTX, methotrexate; SCT, stem cell transplantation.
A 23-year-old female, who was fit and previously healthy, was diagnosed with MECOMr AML in June 2023. The detailed patient characteristics and treatments are summarized in Table 1 and Figure 1. Notably, the patient presented with markedly elevated platelet counts and AML-related diabetes insipidus, both clinical features indicative of MECOMr AML. Brain MRI showed infiltration of the infundibulum and absence of the neurohypophyseal signal; however, leukemic cells were not detected in the cerebrospinal fluid. Rapid human leukocyte antigen (HLA) typing identified the patient’s brother as haploidentical; thus, once inv(3)(q21q26.2) was confirmed, preparations for the sequential alloSCT began, and the patient proceeded to the first phase of therapy (Dec + Ven), followed by FLAG-Ida + Ven and MAC. Post-transplant cyclophosphamide (PtCy) was used for T-cell depletion together with cyclosporine A (CsA) and mycophenolate mofetil (MMF)-based GVHD prophylaxis. Pneumonia, sepsis, mucositis, and hand–foot syndrome were reported as grade 3 adverse events during treatment, whereas symptoms of diabetes insipidus have disappeared. Complete remission (CR) with negative MRD and full donor chimerism (99.57%) was confirmed on day +31 after alloSCT. Immunosuppression was tapered and stopped early on day +115 in the absence of GVHD. At the last follow-up (17 months after sequential alloSCT), the patient remained in good health and was in sustained MRD-negative CR with full donor chimerism.
A 32-year-old female, who was fit and previously healthy, was diagnosed with MECOMr AML in November 2023. AML-related symptoms included fever and hematomas. Detailed characteristics and treatment details are summarized in Table 1, Figure 1. Rapid HLA typing identified the patient’s brother as HLA identical; thus, once inv(3)(q21q26.2) was confirmed, sequential alloSCT was scheduled, and the patient proceeded to the first phase of therapy (Dec + Ven), followed by FLAG-Ida + Ven, and MAC. Anti-thymocyte globulin (ATG) was used for T-cell depletion in combination with CsA + methotrexate-based GVHD prophylaxis. The patient experienced treatment-emergent adverse events of febrile neutropenia and mucositis, both of which were evaluated to be grade 3. CR with marginally positive MRD (0.02%) and full donor chimerism (99.5%) was confirmed on day +30 after alloSCT. CsA was tapered and stopped early on day +62 in the absence of GVHD and because of MRD positivity. MRD negativity was confirmed on day +90. At the last follow-up (11 months after sequential alloSCT), the patient remained in good health and sustained MRD-negative CR with full donor chimerism.
We demonstrate two cases of newly diagnosed MECOMr AML patients who had the following typical clinical and genomic features of this AML subtype: diabetes insipidus, elevated PLT counts, dysplastic changes in megakaryopoiesis, monosomy 7, and NRAS, GATA2, SF3B1 co-mutations (2, 4, 10, 11). Rapid AML genomic testing and HLA typing allowed both patients to successfully undergo an intensive, non-conventional approach of frontline sequential alloSCT using a triple “all-at-once” treatment, which, to the best of our knowledge, had not been previously published.
The rationale for this strategy is mainly based on the poor results of standard available therapies for MECOMr AML and the crucial role of alloSCT. In the setting of intensive induction chemotherapy, the reported response rates are only 29%–46% in newly diagnosed patients and less than 20% in the relapsed/refractory (R/R) translating into median overall survival of 5.9–8.4 months only (2, 5, 12). Importantly, alloSCT, especially early in CR1, was associated with significantly better outcomes emphasizing the necessity of fast allotransplantation without exposing the patients to multiple lines of subsequent therapy with possible excessive toxicity and preventing an early relapse (2, 5, 7, 9). Recent reports suggest that the efficacy of hypomethylating agents (HMA) may be similar to intensive chemotherapy with response rates reaching up to 47%, notably lower treatment-related mortality, and comparable survival rates (2, 3, 7). This may be attributed to chemoresistance and the aberrant hypermethylation profile of MECOMr leukemic cells (13). The addition of the BCL-2 inhibitor venetoclax to either HMA or intensive chemotherapy in MECOMr AML is less described, and available data, albeit scarce, do not seem to demonstrate meaningful improvement in efficacy or survival, which may be explained by the hyperexpression of BCL-XL and high rates of co-mutations in the signaling pathways (NRAS, KRAS, PTPN11) (2, 3, 14–18). Investigational approaches for MECOMr AML are currently focused on epigenetic modifiers, inhibition of signaling pathways, and immunotherapy (18).
Based on the aforementioned studies, the first part of our triple therapy consisted of 5-day Dec + Ven to address the aberrant hypermethylation of MECOMr AML, provide cytoreduction, and induce epigenetic priming before intensive chemotherapy. The possible efficacy of HMAs as chemosensitizers when used before chemotherapeutic drugs has been previously described in several reports (19–22). After the 5-day Dec + Ven cytoreductive priming on the next day, both patients proceeded to the FLAG-Ida + Ven regimen, which currently demonstrated unprecedented overall response (99%) and MRD negativity (89%) rates in newly diagnosed AML (23, 24). Notably, both patients achieved morphological blast clearance with positive MRD based on bone marrow evaluation during the short washout period of 1 or 3 days and proceeded to myeloablative conditioning and alloSCT.
Myeloablative conditioning during aplasia after Dec + Ven and FLAG-Ida + Ven may have resulted in additional antileukemic efficacy and, most importantly, prevented the risk of leukemia regrowth before engraftment. Furthermore, immediate alloSCT allowed our patients to achieve timely neutrophil recovery, as FLAG-Ida + Ven is associated with prolonged myelosuppression, whereas the addition of Dec + Ven priming to our regimen could have further extended the aplasia period. However, the cumulative high intensity of sequential triple therapy resulted in deep remissions on day 30 after alloSCT in both patients, and the absence of GVHD allowed us to taper and stop immunosuppression early, thus enhancing the graft-versus-leukemia effect, which is of utmost importance in chemoresistant, adverse-risk AML (25, 26). Because of sustained and prolonged MRD-negative CRs in both cases, we did not initiate maintenance therapy with HMAs or prophylactic donor lymphocyte infusions.
The decision to offer immediate alloSCT without achieving a conventional response is supported by experience from multiple reports and a recently published phase 3 ASAP study demonstrating the efficacy and safety of sequential allotransplantation using FLAMSA (fludarabine, cytarabine, amsacrine) followed by standard conditioning regimens in R/R AML patients with active disease (24–35). Implementing upfront sequential alloSCT with FLAMSA and high-dose melphalan as frontline therapy was described in a subgroup of 10 previously untreated secondary AML patients in a single-center study (36). However, the number and outcomes of MECOMr AML patients enrolled in the trials were not specified. It is important to emphasize that neither of these studies used HMA and venetoclax in their treatment regimens; however, our recently published single-center experience suggests the clinical applicability of decitabine + venetoclax + cytarabine-based preconditioning before standard conditioning regimens in the sequential alloSCT approach for active high-risk R/R AML (37).
Our case report has several limitations. The treatment was administered outside a clinical trial, and due to the rarity of MECOMr AML, we have only described two cases with relatively short follow-up periods. The implementation of sequential HMA + Ven and FLAG-Ida + Ven treatment has not been previously studied, and it remains unknown whether using either of these therapies before sequential alloSCT would have resulted in similar outcomes and whether venetoclax adds efficacy at all in the setting of the MECOMr genotype. Furthermore, whether the HMAs should be used before or in combination with chemotherapy to achieve optimal epigenetic priming and possible chemosensitization remains unclear. Despite the absence of life-threatening adverse events in our young and fit patients, high treatment intensity could be one of the major limitations for further clinical evaluation. In addition, the need for rapid HLA typing and donor identification, as well as donor availability, management of stem cell collection, and logistics, may also restrict the timely applicability of this approach despite the transformative effect of post-transplant cyclophosphamide for haploidentical or mismatched donor alloSCT.
Nevertheless, upfront sequential alloSCT treatment was safe and effective in two patients with MECOMr AML indicating the need to evaluate this challenging approach for newly diagnosed adverse risk AML in a randomized clinical trial.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
The studies involving humans were approved by Ethics Committee of Vilnius University Hospital Santaros Klinikos. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
AŽ: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. MT: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Tang Z, Li Y, Wang X, Chen J, Liu Q, Zhang M, et al. 3q26.2/MECOM rearrangements by pericentric inv(3): diagnostic challenges and clinicopathologic features. Cancers. (2023) 15:458. doi: 10.3390/cancers15020458
2. Richard-Carpentier G, Jones D, Smith A, Brown B, Taylor C, Wilson E, et al. Characteristics and clinical outcomes of patients with acute myeloid leukemia with inv(3)(q21q26.2) or t(3;3)(q21;q26.2). Haematologica. (2023) 108:2331–42. doi: 10.3324/haematol.2022.282030
3. Alhajahjeh A, Patel P, Nguyen T, Lee S, Kim H, Garcia R, et al. Acute myeloid leukemia (AML) with chromosome 3 inversion: biology, management, and clinical outcome. Leuk Lymphoma. (2024) 65:1541–51. doi: 10.1080/10428194.2024.2367040
4. Inv(3)(q21q26) GATA2::MECOM t(3;3)(q21;q26) GATA2::MECOM ins(3;3)(q26;q21q26) GATA2::MECOM . Available online at: https://atlasgeneticsoncology.org/haematological/209001/inv(3)(q21q26) (Accessed November 10, 2024).
5. Sitges M, Romero P, Alvarez S, Martinez L, Hernandez M, Torres A, et al. Acute myeloid leukemia with inv(3)(q21.3q26.2)/t(3;3)(q21.3;q26.2): Study of 61 patients treated with intensive protocols. Eur J Haematol. (2020) 105:138–47. doi: 10.1111/ejh.v105.2
6. Döhner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. (2022) 140(12):1345–77. doi: 10.1182/blood.2022016867
7. Wanquet A, Malard F, Gaugler B, Blaise D, Mohty M, et al. Azacitidine treatment for patients with myelodysplastic syndrome and acute myeloid leukemia with chromosome 3q abnormalities. Am J Hematol. (2015) 90:859–63. doi: 10.1002/ajh.v90.10
8. Polprasert C, Ittiwut C, Tangnuntachai N, Pongstaporn W, Assawamakin A, et al. Clinical characteristics and outcomes of myeloid neoplasms with mecom rearrangement: results from a nationwide multicenter study. Blood. (2023) 142:4213. doi: 10.1182/blood-2023-180100
9. Halaburda K, Labopin M, Brecht A, Cornelissen J, Schmid C, et al. AlloHSCT for inv(3)(q21;q26)/t(3;3)(q21;q26) AML: a report from the acute leukemia working party of the European society for blood and marrow transplantation. Bone Marrow Transplant. (2018) 53:683–91. doi: 10.1038/s41409-018-0165-x
10. Pritzl SL, Matson DR, Juckett MB, Ciske DJ. Concurrent central diabetes insipidus and acute myeloid leukemia. Case Rep Hematol. (2021) 2021:8898671. doi: 10.1155/2021/8898671
11. Harrup R, Pham M, McInerney G. Acute myeloid leukemia with diabetes insipidus and hypophyseal infiltration. Asia Pac J Clin Oncol. (2016) 12:e350–1. doi: 10.1111/ajco.12257
12. Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, et al. Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood. (2010) 116:354–65. doi: 10.1182/blood-2009-11-254441
13. Lugthart S, Figueroa ME, Bindels E, Skrabanek L, Valk PJ, et al. Aberrant DNA hypermethylation signature in acute myeloid leukemia directed by EVI1. Blood. (2010) 117:234. doi: 10.1182/blood-2010-04-281337
14. Pradhan AK, Mohapatra AD, Nayak KB, Chakraborty S. Acetylation of the proto-oncogene EVI1 abrogates Bcl-xL promoter binding and induces apoptosis. PloS One. (2011) 6:e25370. doi: 10.1371/journal.pone.0025370
15. Garciaz S, Hospital M-A, Collette Y, Vey N. Venetoclax resistance in acute myeloid leukemia. Cancers. (2024) 16:1091. doi: 10.3390/cancers16061091
16. Konopleva M, DiNardo CD, Kadia TM, Garcia-Manero G, Borthakur G, Daver N. Efficacy and biological correlates of response in a phase II study of venetoclax monotherapy in patients with acute myelogenous leukemia. Cancer Discovery. (2016) 6:1106–17. doi: 10.1158/2159-8290.CD-16-0313
17. Sango J, Carcamo S, Sirenko M, Maiti A, Mansour H, Ulukaya G, et al. RAS-mutant leukaemia stem cells drive clinical resistance to venetoclax. Nature. (2024) 636(8041):241–50. doi: 10.1038/s41586-024-08137-x
18. Birdwell C, Davis JK, Howard SM, Bhatnagar AS, Myers ML, Klinkebiel RJ. EVI1 dysregulation: impact on biology and therapy of myeloid Malignancies. Blood Cancer J. (2021) 11:64. doi: 10.1038/s41408-021-00457-9
19. Zhang N, Li Y, Wang X, Chen L, Zhang J, Liu H. Decitabine as epigenetic priming with CLAG induce improved outcome of relapsed or refractory acute myeloid leukemia in children. Clin Epigenet. (2024) 16:63. doi: 10.1186/s13148-024-01677-z
20. Leonard SM, Perry T, Woodman CB, Kearns P. Sequential treatment with cytarabine and decitabine has an increased anti-leukemia effect compared to cytarabine alone in xenograft models of childhood acute myeloid leukemia. PloS One. (2014) 9:e87475. doi: 10.1371/journal.pone.0087475
21. Hollenbach PW, Nguyen AN, Brady H, Williams M, Ning Y, Richard N. A comparison of azacitidine and decitabine activities in acute myeloid leukemia cell lines. PloS One. (2010) 5:e9001. doi: 10.1371/journal.pone.0009001
22. Palmieri R, Buckley SA, Othus M, Halpern AB, Percival MM, Scott BL. Randomized phase 1 study of sequential (“primed”) vs. concurrent decitabine in combination with cladribine, cytarabine, G-CSF, and mitoxantrone (CLAG-M) in adults with newly diagnosed or relapsed/refractory acute myeloid leukemia (AML) or other high-grade myeloid neoplasm. Leuk Lymphoma. (2020) 61:1728–31. doi: 10.1080/10428194.2020.1728754
23. Jen WY, Smith J, Lee H, Patel K, Nguyen T, Garcia M. FLAG-IDA + venetoclax in newly diagnosed (ND) or relapsed/refractory (RR) AML. J Clin Oncol. (2024) 42:6519–9. doi: 10.1200/JCO.2024.42.16_suppl.6519
24. DiNardo CD, Lachowiez CA, Takahashi K, Loghavi S, Xiao L, Kadia TM. Venetoclax combined with FLAG-IDA induction and consolidation in newly diagnosed acute myeloid leukemia. Am J Hematol. (2022) 97:1035–43. doi: 10.1002/ajh.26601
25. Cornelissen JJ, Breems DA, van Putten WLJ, Gratwohl A, Passweg JR, Robin M. Comparative analysis of the value of allogeneic hematopoietic stem-cell transplantation in acute myeloid leukemia with monosomal karyotype versus other cytogenetic risk categories. J Clin Oncol Off J Am Soc Clin Oncol. (2012) 30:2140–6. doi: 10.1200/JCO.2011.39.6499
26. Vyas P, Appelbaum FR, Craddock C. Allogeneic hematopoietic cell transplantation for acute myeloid leukemia. Biol Blood Marrow Transplant. (2015) 21:8–15. doi: 10.1016/j.bbmt.2014.10.026
27. Ronnacker J, Urbahn M-A, Reicherts C, Klement S, Bornhäuser M, Röllig C. Early blast clearance during sequential conditioning before allogeneic stem cell transplantation in patients with acute myeloid leukaemia. Br J Haematol. (2024) 205:280–90. doi: 10.1111/bjh.v205.1
28. Stelljes M, Middeke J-M, Bug G, Serve H, Kröger N, Berdel WE, et al. Remission induction versus immediate allogeneic haematopoietic stem cell transplantation for patients with relapsed or poor responsive acute myeloid leukaemia (ASAP): a randomised, open-label, phase 3, non-inferiority trial. Lancet Haematol. (2024) 11:e324–35. doi: 10.1016/S2352-3026(24)00065-6
29. Middeke JM, Stelljes M, Ehninger G, Berdel WE, Serve H, Kröger N. Clofarabine salvage therapy before allogeneic hematopoietic stem cell transplantation in patients with relapsed or refractory AML: results of the BRIDGE trial. Leukemia. (2016) 30:261–7. doi: 10.1038/leu.2015.226
30. Stölzel F, Röllig M, Ehninger G, Platzbecker U, Schetel C. Early intervention with allogeneic hematopoietic cell transplantation during chemotherapy-induced aplasia in patients with high-risk acute myeloid leukemia. Leukemia. (2013) 27:2068–72. doi: 10.1038/leu.2013.142
31. Jondreville L, Daguindau F, Blin N, Henry J, Maury S, Pigneux A, et al. FLAMSA-busulfan-melphalan as a sequential conditioning regimen in HLA-matched or haploidentical hematopoietic stem cell transplantation for high-risk myeloid diseases. Transplant Cell Ther. (2021) 27:915.e1–8. doi: 10.1016/j.jtct.2021.07.018
32. Rodríguez-Arbolí E, Labopin M, Mohty M, Nagler A, Bazarbachi A, Baron F. FLAMSA-Based Reduced-Intensity Conditioning versus Myeloablative Conditioning in Younger Patients with Relapsed/Refractory Acute Myeloid Leukemia with Active Disease at the Time of Allogeneic Stem Cell Transplantation: An Analysis from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Biol Blood Marrow Transplant J Am Soc Blood Marrow Transplant. (2020) 26:2165–73. doi: 10.1016/j.bbmt.2020.07.020
33. Schmid C, Schleuning M, Schwerdtfeger R, Tischer J, Hensel M, Kolb HJ. Long-term survival in refractory acute myeloid leukemia after sequential treatment with chemotherapy and reduced-intensity conditioning for allogeneic stem cell transplantation. Blood. (2006) 108:1092–9. doi: 10.1182/blood-2005-10-4165
34. Schneidawind D, Huber E, Faul C, Vogel W, Kanz L, Bethge WA. Allogeneic hematopoietic cell transplantation with reduced-intensity conditioning following FLAMSA for primary refractory or relapsed acute myeloid leukemia. Ann Hematol. (2013) 92:1389–95. doi: 10.1007/s00277-013-1774-5
35. Schmid C, Schleuning M, Tischer J, Holler E, Haude K-H, Braess J. Early allo-SCT for AML with a complex aberrant karyotype–results from a prospective pilot study. Bone Marrow Transplant. (2012) 47:46–53. doi: 10.1038/bmt.2011.15
36. Saure C, Schroeder T, Zohren F, Groten A, Bruns I, Czibere A. Upfront allogeneic blood stem cell transplantation for patients with high-risk myelodysplastic syndrome or secondary acute myeloid leukemia using a FLAMSA-based high-dose sequential conditioning regimen. Biol Blood Marrow Transplant. (2012) 18:466–72. doi: 10.1016/j.bbmt.2011.09.006
37. Zucenka A, Kevličius L, Smalinskaitė T, Daukėlaitė G, Davainis L, Trociukas I, et al. Individualized sequential allogeneic stem cell transplantation for relapsed or refractory acute myeloid leukemia or high-risk myelodysplastic syndrome patients with active disease. Blood. (2024) 144(Supplement 1):3487. doi: 10.1182/blood-2024-203229
Keywords: acute myeloid leukaemia (AML), allogeneic stem cell transplantation, venetoclax (ABT-199, PubChem CID: 49846579), FLAG-Ida, inv(3)(q21q26.2), MECOM (MDS1 and EV1 complex locus), hypomethylating agents
Citation: Žučenka A and Tamutytė M (2025) Case Report: Decitabine and venetoclax sequentially followed by FLAG-Ida and venetoclax with immediate allogeneic stem cell transplantation in newly diagnosed acute myeloid leukemia with chromosome 3 inversion/MECOM rearrangement. Front. Oncol. 15:1530852. doi: 10.3389/fonc.2025.1530852
Received: 19 November 2024; Accepted: 03 March 2025;
Published: 27 March 2025.
Edited by:
Liren Qian, Fifth Medical Center of the PLA General Hospital, ChinaReviewed by:
Shreyans Gandhi, King’s College Hospital NHS Foundation Trust, United KingdomCopyright © 2025 Žučenka and Tamutytė. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Andrius Žučenka, YW5kcml1cy56dWNlbmthQHNhbnRhLmx0
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.