
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
PERSPECTIVE article
Front. Oncol. , 01 April 2025
Sec. Hematologic Malignancies
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1498855
This article is part of the Research Topic Use of Radiation Therapy for Hematological Malignancies View all 6 articles
Primary cutaneous lymphomas are highly radiosensitive. X-rays work well for localized cutaneous lymphomas. However, if disseminated in the skin and covering larger areas, as is commonly the case with the most common type, mycosis fungoides, x-ray therapy is not suited because the dose to underlying organs exceeds their tolerance. By contrast, electrons have a limited range of penetration, and are ideal for treating superficial lesions. Techniques have been developed to yield a fairly uniform dose to the entire skin surface and treating to a depth of about 1-1½ cm. Total skin electron beam therapy (TSEBT) is probably the most effective skin directed therapy for widespread primary cutaneous lymphomas. For many years the total dose used for mycosis fungoides was 30-36 Gy, given in small fractions. This treatment could only be repeated once. However, total doses of 10-12 Gy have now been shown to offer excellent response rates, and the treatment can be repeated up to 6 times, offering as much or probably even more palliation than the high-dose treatment. Today, most patients are treated with low-dose TSEBT, the higher doses reserved for patients with more resistant disease. Attempts have been made to use photon therapy for total skin irradiation, e.g., tomotherapy. However, even with the most meticulous of techniques there is too much dose in deeper structures, resulting in bone marrow toxicity even with low-dose treatment. This is never seen with electrons, even with high-dose therapy. Further research into optimizing TSEBT and exploring combinations with systemic treatments is ongoing.
Primary cutaneous lymphomas are the second most common primary extranodal lymphomas. The estimated annual incidence of primary cutaneous lymphomas (PCL) is 1/100,000 in Western countries. They must be distinguished from disseminated lymphomas with spread to the skin. Primary cutaneous lymphomas differ significantly from nodal lymphomas and from primary extranodal lymphomas in other locations in several important ways. They tend to remain localized to the skin for a long time, they have a much more indolent course and a much better prognosis than that of lymphomas of similar histological subtype in other locations, and they are treated differently (1–3). Hence, they are kept as separate disease entities in the histopathologic classification of lymphomas, both in the new WHO classification (4) and in the International Consensus Classification (5).
Primary cutaneous lymphomas are most commonly of T-cell origin, and the most common disease entity is mycosis fungoides and Sézary syndrome, a rare leukemic variant, which constitute 60-70% of all primary cutaneous lymphomas (1, 3). Most patients have widespread disease in the skin, but rarely dissemination to lymph nodes or internal organs, and even in these cases the major disease burden is often still in the skin. Except for rare cases of localized skin disease and rare cases of advanced cases treated with allogeneic stem cell transplantation, mycosis fungoides is incurable and treatment is generally palliative (1, 3, 6–8). These patients often live for many years, commonly decades, and the skin disease is often associated with distressing symptoms, e.g., itching, ulceration, and cosmetic problems. Hence, prevention and alleviation of these symptoms is of the greatest importance for maintaining a good quality of life. Skin directed therapies are the mainstay, for patients with early disease stages, patches or thin plaques, given alone, for patients with more advanced skin disease, infiltrated plaques or tumors, supplemented with systemic treatments (1, 3, 6–8).
Like other indolent lymphomas, mycosis fungoides is extremely radiosensitive (9). Doses of 8 Gy achieve complete response rates in > 90% (10). Indeed, mycosis fungoides was probably the first lymphoma reported to be treated with X-rays (11). Kilovolt X-ray therapy works well for small localized cutaneous lesions. However, if the disease is disseminated over larger skin areas, as is commonly the case with mycosis fungoides, it is not suited. The problem with X-rays, even kilovolt, is that if administered over large areas, the dose to the underlying internal organs exceeds their tolerance. By contrast, electrons have a limited range of penetration, limiting their effect to superficial tissues, the depth depending on the energy of the electrons.
The possibility of using artificially accelerated high-energy electrons in radiotherapy was anticipated by physicists in the 1920s (12, 13), but electrons with sufficient energy could not be generated at the time. In the 1940s physicists at the Massachusetts Institute of Technology in Boston constructed a Van de Graaff generator that was able to produce a 2.5 MeV electron beam. The generator was a huge construction, and still the maximum range of electrons in tissue was only around 10 mm (14). By this technology it became possible to treat large skin areas without damaging underlying critical structures (15).
With the introduction of the linear accelerator, it became possible and practical to generate electrons for treatment with energies from 4 to over 20 MeV. Electron beam treatment of localized primary cutaneous lymphomas became and remains the optimal method for radiation therapy, enabling a homogenous dose to skin lesions with full skin dose and reaching the desired depth by judicious use of bolus. The challenge was to develop techniques for treating the whole skin in patients with disease disseminated in the skin, typically cases of mycosis fungoides (16). At Stanford a technique was developed where the patient was treated standing at an extended distance of 3-4 meters from the accelerator, with a dual field technique (angled from the horizontal position to be centered above and below the patient), and in 6 alternating patient positions (17, 18) (see Figure 1). This technique has become widely popular and is used in most centers offering total skin electron beam therapy (TSEBT). It provides a reasonably homogenous treatment of the whole skin (19–24), but supplementary electron fields must be administered to areas (e.g., scalp, perineum, soles) that are shielded during the TSEBT, and lead shielding of thin areas that would otherwise be overdosed (e.g., fingers and toes) should be applied halfway through the treatment. Rotational techniques, where the patient stands on a rotating platform during the treatment, have also been used, with the same need for supplementary electron fields to shielded areas and shielding of thin areas (25, 26). Translational techniques, where the patient is treated at a shorter distance lying alternately in the prone and supine position under the accelerator and being moved in the longitudinal direction are used less often (27, 28). The techniques for TSEBT are quite complex from a physical point of view, using the accelerator in a way that is different from the usual. Modern 3-dimensional treatment planning cannot be applied, and the techniques were developed by dosimetric measurements in phantoms and patients. The technical details were published in an AAPM (American Association of Physicists in Medicine) Report (29). The doses in different areas of the skin will vary depending on patient shape, anatomy, and position. Dose homogeneity is far from what we are normally used to in modern radiation therapy. There will be areas that are underdosed, but as the disease is incurable and the treatment therefore essentially palliative, this is acceptable. There will also be areas that are somewhat overdosed, but as the radiation doses needed in these diseases are fairly low, this is also acceptable. The technique can therefore also be used in other highly radiosensitive hematologic diseases, e.g., leukemia cutis (30). However, the technique is not suitable for solid tumors disseminated in the skin, which require higher doses.

Figure 1. Patient positions for total skin electron beam therapy, 6-field technique. Reprinted from Specht et al., 2015 (9).
Attempts have been made to use modern highly conformal X-ray radiation therapy for total skin irradiation. Helical tomotherapy with the patient in the supine position has been tested in several studies, consistently demonstrating significant hematological toxicity (31–34), which is never seen with TSEBT. Hence, even the most meticulous conformal X-ray technique is unable to limit the radiation to the skin to the same degree that can be achieved with electron beam therapy. TSEBT remains the recommended technique.
When TSEBT was introduced in the 1960s a total dose of 30-36 Gy was generally prescribed for mycosis fungoides, often coined conventional dose TSEBT. This was based on studies showing that the complete remission rate was higher and the duration of remissions longer with this dose than with lower doses (35, 36). The skin is the largest organ in the body; hence the treated volume is nearly 20% of the total body weight. Treating such a large volume to this total dose was quite daring, and the treatment was given in small fractions, usually one fraction per day and with only four treatments per week. As the skin would in many cases become quite red about halfway through the treatment, a break of 1-2 weeks was usually made to avoid severe skin reactions. This made for a total treatment duration of 9-11 weeks. A recent meta-analysis of published data showed for patients treated with this conventional dose a complete response rate in early stage mycosis fungoides of 72% and an overall response rate of 100%, for advanced disease the complete response rate was lower (55%) but the overall response rate was similar (37). Patients will eventually progress, but the duration of the clinical benefit is clearly important, and a good measure of this benefit in mycosis fungoides is the time to next treatment (38). After conventional dose TSEBT it varies according to tumor burden and number or previous treatments, but may be up to a couple of years (35, 38). In most cases some form of maintenance treatment is given after TSEBT in order to maintain the response achieved with TSEBT for as long as possible (16) (see below). Patients with Sézary syndrome with blood involvement have generally not responded well to TSEBT, also because their skin is very thin and vulnerable and therefore does not tolerate irradiation so well, but data seem to indicate that TSEBT may be a useful adjuvant to systemic treatments in patients with significant blood involvement (39).
The toxicity of the treatment is actually quite manageable, rarely > Gr. 2 (37). Acute toxicities are erythema and dry desquamation, but hematologic toxicity is never seen with this treatment, because of the limited depth of penetration of the electrons. Temporary alopecia and loss of finger- and toenails is to be expected (around 70%), and patients are usually unable to sweat properly for 6-12 months. They may also (around 20%) experience variable limb edema during the first year after treatment. Second cancers in the skin, in particular squamous cell carcinomas, are seen with an increased frequency, but it is not possible to define the role of TSEBT in this as patients have always received many other carcinogenic skin directed therapies (40).
The main disadvantages of conventional dose TSEBT are that it takes a long time, which may be very inconvenient as most patients are fairly old and often have to travel to receive the treatment, and that it may be repeated only once in order to keep within the skin tolerance (41).
With the realization that systemic indolent B-cell lymphomas are exquisitely radiosensitive with total doses as low as 4 Gy achieving durable local control in 70% of cases of follicular lymphoma and marginal zone lymphoma (42), we tested a similar dose with TSEBT for mycosis fungoides (43). The result was disappointing with a remission duration of only 2.4 months, confirming that T-cell lymphomas are less radiosensitive than B-cell lymphomas. We then tested a total dose of 10 Gy, 1/3 of our conventional dose TSEBT (44, 45). The response rate was 90%, with 70% complete or very good partial remissions. The median duration of remission was 5.2 months. Many subsequent studies using low-dose regimens of 10-12 Gy have confirmed these results (37, 46–57). The response rates with low-dose TSEBT were excellent. The duration of the responses was shorter than with the conventional dose, but the low-dose treatment could be repeated up to six times, offering as much or probably even more palliation than the conventional dose treatment with less toxicity and more convenience. Results from prospective trials of low dose TSEBT are shown in Table 1. Some form of maintenance treatment is usually given after TSEBT in order to make the response last for as long as possible (16, 58). Maintenance therapy is a highly individualized treatment, based on the experience with respect to response and tolerance with previous treatments in the individual patient. Maintenance therapy may be skin directed (16, 58–61), e.g., psoralen plus ultraviolet A (PUVA) and topical nitrogen mustard, or it may be systemic (16, 58, 61–63), e.g., interferon-alpha, retinoids, or newer drugs such as mogamulizumab, which is being tested in prospective trials. Combining low-dose TSEBT with systemic therapy may be effective also in patients with Sézary syndrome (64, 65). Low-dose TSEBT is today in general the preferred treatment, with higher doses reserved for more radioresistant cases. A typical case of mycosis fungoides with generalized plaques before and after TSEBT is shown in Figure 2.
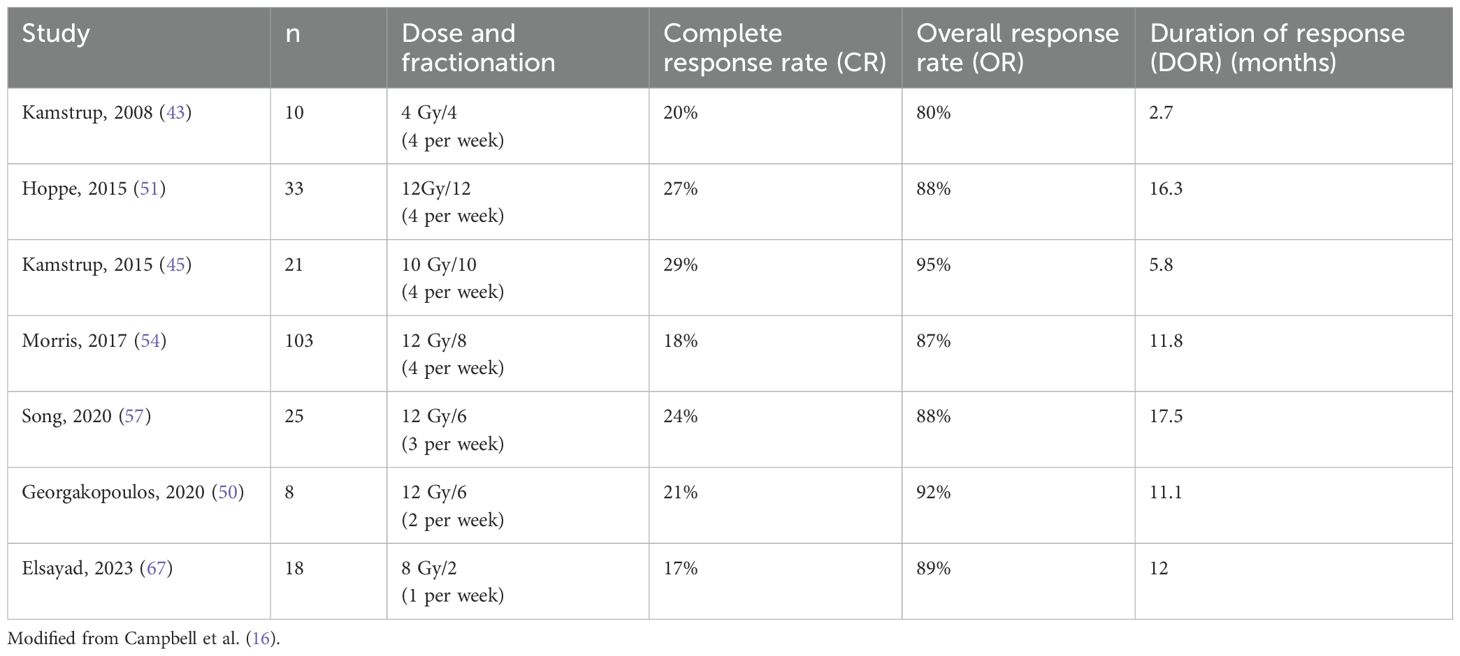
Table 1. Prospective studies of Low Dose Total Skin Electron Therapy (TSEBT) for mycosis fungoides and Sezary syndrome.
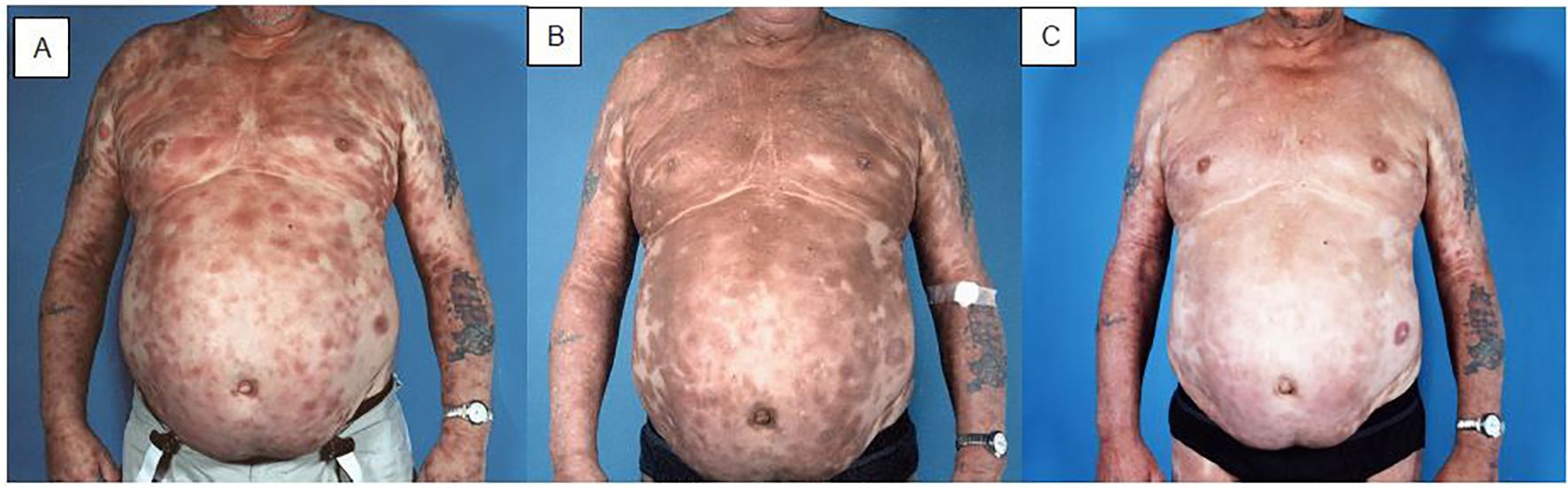
Figure 2. Patient with mycosis fungoides with disseminated plaques before (A), one month after (B), and one year after (C) TSEBT. During and in the weeks after TSEBT the structure of the skin normalizes, but there is still discolouration, which changes from red to brownish. The brown spots become paler and less prominent during the first year after treatment, but there will be some permanent discolouration.
Early in the COVID pandemic, the International Lymphoma Radiation Oncology Group (ILROG) developed and published guidelines for use during the pandemic to minimize patients’ visits to hospitals when receiving radiation therapy for hematological malignancies (66). For TSEBT for mycosis fungoides a schedule of 4 Gy x 2-3 in weekly fractions was recommended, and has since been tested and found effective and well tolerated (67). Hypofractionation may offer advantages for patient living far from the treatment facility. However, each treatment takes quite a long time, which may be problematic for older patients.
At present the only treatment for advanced mycosis fungoides which has shown curative potential is non-myeloablative allogeneic stem cell transplantation, which induces a graft versus-lymphoma effect. A recent meta-analysis showed progression free survival at three years in one-third of patients, but with wide variation between studies (68). For a good outcome of this treatment the patient should be in as good a remission as possible before the transplantation. Conventional dose TSEBT is an effective debulking agent in the skin, and several studies have reported using this treatment as part of the conditioning regimen with promising results (69–71).
TSEBT is an effective and well tolerated treatment for mycosis fungoides and for other hematological malignancies when located in the skin. Electrons have superior physical characteristics which make them particularly suited for treating targets in the skin while sparing underlying normal structures. Hence, no other radiation therapy technique has been able to provide radiation therapy to the whole skin while avoiding toxicity from deeper-lying critical structures, in particular the bone marrow. TSEBT is a special treatment, it is delivered with varying techniques, all of them developed many years ago and by modern standards rather unprecise. Further research into optimization of this treatment will hopefully further improve outcome for patients with hematological malignancies in the skin.
LS: Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Gilson D, Whittaker SJ, Child FJ, Scarisbrick JJ, Illidge TH, Parry EJ, et al. British Association of Dermatologists and U.K. Cutaneous Lymphoma Group guidelines for the management of primary cutaneous lymphomas 2018. Br J Dermatol. (2019) 180:496–526. doi: 10.1111/bjd.2019.180.issue-3
2. Specht L, Skov L. Cutaneous lymphomas. Clin Oncol (R Coll Radiol ). (2019) 31:797–807. doi: 10.1016/j.clon.2019.07.018
3. Willemze R, Hodak E, Zinzani PL, Specht L, Ladetto M. Primary cutaneous lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. (2018) 29:iv30–40. doi: 10.1093/annonc/mdy133
4. Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IBO, Berti E, et al. The 5th edition of the world health organization classification of haematolymphoid tumours: lymphoid neoplasms. Leukemia. (2022) 36:1720–48. doi: 10.1038/s41375-022-01620-2
5. Campo E, Jaffe ES, Cook JR, Quintanilla-Martinez L, Swerdlow SH, Anderson KC, et al. The international consensus classification of mature lymphoid neoplasms: a report from the clinical advisory committee. Blood. (2022) 140:1229–53. doi: 10.1182/blood.2022015851
6. Kamstrup MR, Specht L. Systemic treatment of cutaneous lymphomas. In: Rembielak A, Tagliaferri L, editors. Non-melanoma Skin Cancer: Essentials for Oncologists. CRC press, Taylor & Francis, Boca Raton, Florida (2023). p. 355–64.
7. Latzka J, Assaf C, Bagot M, Cozzio A, Dummer R, Guenova E, et al. EORTC consensus recommendations for the treatment of mycosis fungoides/Sezary syndrome - Update 2023. Eur J Cancer. (2023) 195:113343. doi: 10.1016/j.ejca.2023.113343
8. NCCN Clinical Practice Guidelines in Oncology. Primary Cutaneous Lymphomas Version 2.2024. Ref Type: Online Source (2024).
9. Specht L, Dabaja B, Illidge T, Wilson LD, Hoppe RT. Modern radiation therapy for primary cutaneous lymphomas: field and dose guidelines from the international lymphoma radiation oncology group. Int J Radiat Oncol Biol Phys. (2015) 92:32–9. doi: 10.1016/j.ijrobp.2015.01.008
10. Neelis KJ, Schimmel EC, Vermeer MH, Senff NJ, Willemze R, Noordijk EM. Low-dose palliative radiotherapy for cutaneous B- and T-cell lymphomas. Int J Radiat Oncol Biol Phys. (2009) 74:154–8. doi: 10.1016/j.ijrobp.2008.06.1918
11. Scholz W. Ueber den Einfluss der Röntgenstrahlen auf die Haut in gesundem und krankem Zustande. III. Klinischer Theil. Archiv f Dermatologie u Syphilis. (1902) 59:421–45.
12. Brasch A, Lange F. Aussichten und Möglichkeiten einer Therapie mit schnellen Kathodenstrahlen. Strahlentherapie. (1934) 51:119–28.
13. Coolidge WD, Moore CN. Some experiments with high voltage cathode rays outside of the generating tube. J Franklin Inst. (1926) 202:722–35. doi: 10.1016/S0016-0032(26)90694-7
14. Granke RC, Trump JG, Wright KA. Physical aspects of megavolt electron therapy. Radiology. (1956) 67:553–61. doi: 10.1148/67.4.553
15. Trump JG, Wright KA, Evans WW, Anson JH, Hare HF, Fromer JL, et al. High energy electrons for the treatment of extensive superficial Malignant lesions. Am J Roentgenol. (1953) 69:623–9.
16. Campbell BA, Prince HM, Thursky K, Dabaja B, Hoppe R, Specht L, et al. Breaking down the barriers for patients with cutaneous T-cell lymphoma: current controversies and challenges for radiation oncologists in 2024. Semin Radiat Oncol. (2025) 35:110–25. doi: 10.1016/j.semradonc.2024.08.005
17. Cox RS, Heck RJ, Fessenden P, Karzmark CJ, Rust DC. Development of total-skin electron therapy at two energies. Int J Radiat Oncol Biol Phys. (1990) 18:659–69. doi: 10.1016/0360-3016(90)90075-U
18. Karzmark CJ, Loevinger R, Steele RE, Weissbluth M. A technique for large-field, superficial electron therapy. Radiology. (1960) 74:633–44. doi: 10.1148/74.4.633
19. Anacak Y, Arican Z, Bar-Deroma R, Tamir A, Kuten A. Total skin electron irradiation: evaluation of dose uniformity throughout the skin surface. Med Dosim. (2003) 28:31–4. doi: 10.1016/S0958-3947(02)00235-2
20. Baugh G, Al-Alawi T, Fletcher CL, Mills JA, Grieve RJ. A preliminary comparison of total skin electron treatment techniques to demonstrate the application of a mid-torso phantom for measurement of dose penetration. Br J Radiol. (2011) 84:1125–30. doi: 10.1259/bjr/52924135
21. Ding GX, Chen ZJ, Zhong W, Zhu TC. Skin dose distributions between Stanford and rotational techniques in total skin electron therapy (TSET). Med Phys. (2022) 49:6646–53. doi: 10.1002/mp.v49.10
22. Elsayad K, Moustakis C, Simonsen M, Backer D, Haverkamp U, Eich HT. In-vivo dosimetric analysis in total skin electron beam therapy. Phys Imaging Radiat Oncol. (2018) 6:61–5. doi: 10.1016/j.phro.2018.05.002
23. Misson-Yates S, Naeem M, Palmer I, Holden E, Hedley O, McGovern M, et al. Total skin electron beam therapy rationalization and utility of in vivo dosimetry in a high-volume centre. BJR Open. (2019) 1:20190008. doi: 10.1259/bjro.20190008
24. Pavon EC, Sanchez-Doblado F, Leal A, Capote R, Lagares JI, Perucha M, et al. Total skin electron therapy treatment verification: Monte Carlo simulation and beam characteristics of large non-standard electron fields. Phys Med Biol. (2003) 48:2783–96. doi: 10.1088/0031-9155/48/17/304
25. Heumann TR, Esiashvili N, Parker S, Switchenko JM, Dhabbaan A, Goodman M, et al. Total skin electron therapy for cutaneous T-cell lymphoma using a modern dual-field rotational technique. Int J Radiat Oncol Biol Phys. (2015) 92:183–91. doi: 10.1016/j.ijrobp.2014.11.033
26. Newman NB, Patel CG, Ding GX, Zic JA, Zwerner J, Osmundson EC, et al. Prospective observational trial of low-dose skin electron beam therapy in mycosis fungoides using a rotational technique. J Am Acad Dermatol. (2021) 85:121–7. doi: 10.1016/j.jaad.2020.12.023
27. Anacak Y, Arican Z, Drumea K, Rosenblatt E, Tamir A, Chetver L, et al. Total skin irradiation in mycosis fungoides: comparison between a modified Christie Hospital translational technique and the Stanford technique. Leuk Lymphoma. (2002) 43:2093–7. doi: 10.1080/1042819021000016177
28. Williams PC, Hunter RD, Jackson SM. Whole body electron therapy in mycosis fungoides–a successful translational technique achieved by modification of an established linear accelerator. Br J Radiol. (1979) 52:302–7. doi: 10.1259/0007-1285-52-616-302
29. Karzmark CJ. AAPM Report No. 23. Total skin electron therapy: technique and dosimetry. New York, USA: American Institute of Physics (1988).
30. Bakst RL, Dabaja BS, Specht LK, Yahalom J. Use of radiation in extramedullary leukemia/chloroma: guidelines from the international lymphoma radiation oncology group. Int J Radiat Oncol Biol Phys. (2018) 102:314–9. doi: 10.1016/j.ijrobp.2018.05.045
31. Buglione M, Spiazzi L, Urpis M, Baushi L, Avitabile R, Pasinetti N, et al. Light and shadows of a new technique: is photon total-skin irradiation using helical IMRT feasible, less complex and as toxic as the electrons one? Radiat Oncol. (2018) 13:158. doi: 10.1186/s13014-018-1100-4
32. Haraldsson A, Engleson J, Back SAJ, Engelholm S, Engstrom PE. A Helical tomotherapy as a robust low-dose treatment alternative for total skin irradiation. J Appl Clin Med Phys. (2019) 20:44–54. doi: 10.1002/acm2.2019.20.issue-5
33. Kitaguchi M, Yamashita H, Takenaka R, Okuma K, Nawa K, Nakagawa K. Helical skin radiation therapy including total skin radiation therapy using tomotherapy for primary cutaneous lymphoma with bone marrow suppression as a related adverse event. Pract Radiat Oncol. (2021) 11:e308–21. doi: 10.1016/j.prro.2020.11.007
34. Schaff EM, Rosenberg SA, Olson SJ, Howard SP, Bradley KA. Bone marrow suppression as a complication of total skin helical tomotherapy in the treatment of mycosis fungoides. Radiat Oncol. (2018) 13:67. doi: 10.1186/s13014-018-1013-2
35. Hoppe RT, Fuks Z, Bagshaw MA. The rationale for curative radiotherapy in mycosis fungoides. Int J Radiat Oncol Biol Phys. (1977) 2:843–51. doi: 10.1016/0360-3016(77)90182-1
36. Hoppe RT. Mycosis fungoides: radiation therapy. Dermatol Ther. (2003) 16:347–54. doi: 10.1111/j.1396-0296.2003.01647.x
37. Grandi V, Simontacchi G, Grassi T, Pileri A, Pimpinelli N. Short-term efficacy and safety of total skin electron beam therapy in mycosis fungoides: Systematic review and meta-analysis. Dermatol Ther. (2022) 35:e15840. doi: 10.1111/dth.v35.11
38. Campbell BA, Scarisbrick JJ, Kim YH, Wilcox RA, McCormack C, Prince HM. Time to next treatment as a meaningful endpoint for trials of primary cutaneous lymphoma. Cancers (Basel). (2020) 12. doi: 10.3390/cancers12082311
39. Klein RS, Dunlop JD, Samimi SS, Morrissey KA, Evans KG, Gardner JM, et al. Improvement in peripheral blood disease burden in patients with Sezary syndrome and leukemic mycosis fungoides after total skin electron beam therapy. J Am Acad Dermatol. (2013) 68:972–7. doi: 10.1016/j.jaad.2012.09.056
40. Abel EA, Sendagorta E, Hoppe RT. Cutaneous Malignancies and metastatic squamous cell carcinoma following topical therapies for mycosis fungoides. J Am Acad Dermatol. (1986) 14:1029–38. doi: 10.1016/S0190-9622(86)70128-X
41. Becker M, Hoppe RT, Knox SJ. Multiple courses of high-dose total skin electron beam therapy in the management of mycosis fungoides. Int J Radiat Oncol Biol Phys. (1995) 32:1445–9. doi: 10.1016/0360-3016(94)00590-H
42. Hoskin PJ, Kirkwood AA, Popova B, Smith P, Robinson M, Gallop-Evans E, et al. 4 Gy versus 24 Gy radiotherapy for patients with indolent lymphoma (FORT): a randomised phase 3 non-inferiority trial. Lancet Oncol. (2014) 15:457–63. doi: 10.1016/S1470-2045(14)70036-1
43. Kamstrup MR, Specht L, Skovgaard GL, Gniadecki R. A prospective, open-label study of low-dose total skin electron beam therapy in mycosis fungoides. Int J Radiat Oncol Biol Phys. (2008) 71:1204–7. doi: 10.1016/j.ijrobp.2007.11.039
44. Kamstrup MR, Lindahl LM, Gniadecki R, Iversen L, Skov L, Petersen PM, et al. Low-dose total skin electron beam therapy as a debulking agent for cutaneous T-cell lymphoma: an open-label prospective phase II study. Br J Dermatol. (2012) 166:399–404. doi: 10.1111/j.1365-2133.2011.10670.x
45. Kamstrup MR, Gniadecki R, Iversen L, Skov L, Petersen PM, Loft A, et al. Low-dose (10-Gy) total skin electron beam therapy for cutaneous T-cell lymphoma: an open clinical study and pooled data analysis. Int J Radiat Oncol Biol Phys. (2015) 92:138–43. doi: 10.1016/j.ijrobp.2015.01.047
46. Canu D, Pham-Ledard A, Ouhabrache N, Beylot-Barry M. Changes in total skin electron beam therapy modalities for mycosis fungoides: A single-centre study. Ann Dermatol Venereol. (2022) 149:180–4. doi: 10.1016/j.annder.2022.02.006
47. Chowdhary M, Song A, Zaorsky NG, Shi W. Total skin electron beam therapy in mycosis fungoides-a shift towards lower dose? Chin Clin Oncol. (2019) 8:9. doi: 10.21037/cco.2018.09.02
48. Dault JB, Slade AN, Zhao S, Song S. Comparison of low-dose and standard-dose total skin electron beam therapy in mycosis fungoides. Leuk Lymphoma. (2019) 60:2334–6. doi: 10.1080/10428194.2019.1574008
49. Elsayad K, Kroeger K, Greve B, Moustakis C, Assaf C, Stadler R, et al. Low-dose total skin electron beam therapy: Quality of life improvement and clinical impact of maintenance and adjuvant treatment in patients with mycosis fungoides or Sezary syndrome. Strahlenther Onkol. (2020) 196:77–84. doi: 10.1007/s00066-019-01517-7
50. Georgakopoulos I, Papadavid E, Platoni K, Dilvoi M, Patatoukas G, Kypraiou E, et al. Clinical application of Total Skin Electron Beam (TSEB) therapy for the management of T cell cutaneous lymphomas. The evolving role of low dose (12Gy) treatment schedule. Clin Transl Radiat Oncol. (2019) 15:26–30. doi: 10.1016/j.ctro.2018.12.002
51. Hoppe RT, Harrison C, Tavallaee M, Bashey S, Sundram U, Li S, et al. Low-dose total skin electron beam therapy as an effective modality to reduce disease burden in patients with mycosis fungoides: results of a pooled analysis from 3 phase-II clinical trials. J Am Acad Dermatol. (2015) 72:286–92. doi: 10.1016/j.jaad.2014.10.014
52. Kroeger K, Elsayad K, Moustakis C, Haverkamp U, Eich HT. Low-dose total skin electron beam therapy for cutaneous lymphoma: Minimal risk of acute toxicities. Strahlenther Onkol. (2017) 193:1024–30. doi: 10.1007/s00066-017-1188-8
53. Laughlin BS, van der Walt C, Mangold AR, Breen WG, Rosenthal AC, Lester S, et al. Fine-tuning low-dose total skin electron therapy for optimal management of cutaneous T-cell lymphoma: A comparative analysis of regimens. Adv Radiat Oncol. (2024) 9:101502. doi: 10.1016/j.adro.2024.101502
54. Morris S, Scarisbrick J, Frew J, Irwin C, Grieve R, Humber C, et al. The results of low-dose total skin electron beam radiation therapy (TSEB) in patients with mycosis fungoides from the UK cutaneous lymphoma group. Int J Radiat Oncol Biol Phys. (2017) 99:627–33. doi: 10.1016/j.ijrobp.2017.05.052
55. Rivers CI, Singh AK. Total skin electron beam therapy for mycosis fungoides revisited with adjuvant systemic therapy. Clin Lymphoma Myeloma Leuk. (2019) 19:83–8. doi: 10.1016/j.clml.2018.11.015
56. Smits K, Quint KD, Vermeer MH, Daniels LA, Willemze R, Jansen PM, et al. Total skin electron beam therapy for cutaneous T-cell lymphomas in the Netherlands: A retrospective analysis of treatment outcomes and selection for high or low dose schedule. Clin Transl Radiat Oncol. (2022) 33:77–82. doi: 10.1016/j.ctro.2021.12.001
57. Song A, Gochoco A, Zhan T, Sahu J, Alpdogan O, Porcu P, et al. A prospective cohort study of condensed low-dose total skin electron beam therapy for mycosis fungoides: Reduction of disease burden and improvement in quality of life. J Am Acad Dermatol. (2020) 83:78–85. doi: 10.1016/j.jaad.2020.01.046
58. Dummer R, Assaf C, Bagot M, Gniadecki R, Hauschild A, Knobler R, et al. Maintenance therapy in cutaneous T-cell lymphoma: who, when, what? Eur J Cancer. (2007) 43:2321–9. doi: 10.1016/j.ejca.2007.06.015
59. Kudelka MR, Switchenko JM, Lechowicz MJ, Esiashvili N, Flowers CR, Khan MK, et al. Maintenance therapy for cutaneous T-cell lymphoma after total skin electron irradiation: evidence for improved overall survival with ultraviolet therapy. Clin Lymphoma Myeloma Leuk. (2020) 20:757–67. doi: 10.1016/j.clml.2020.06.020
60. Quiros PA, Jones GW, Kacinski BM, Braverman IM, Heald PW, Edelson RL, et al. Total skin electron beam therapy followed by adjuvant psoralen/ultraviolet-A light in the management of patients with T1 and T2 cutaneous T-cell lymphoma (mycosis fungoides). Int J Radiat Oncol Biol Phys. (1997) 38:1027–35. doi: 10.1016/S0360-3016(97)00127-2
61. Whittaker S, Hoppe R, Prince HM. How I treat mycosis fungoides and Sezary syndrome. Blood. (2016) 127:3142–53. doi: 10.1182/blood-2015-12-611830
62. Elsayad K, Rolf D, Sunderkotter C, Weishaupt C, Muller EC, Nawar T, et al. Low-dose total skin electron beam therapy plus oral bexarotene maintenance therapy for cutaneous T-cell lymphoma. J Dtsch Dermatol Ges. (2022) 20:279–85. doi: 10.1111/ddg.14657
63. Fong S, Hong EK, Khodadoust MS, Li S, Hoppe RT, Kim YH, et al. Low-dose total skin electron beam therapy combined with mogamulizumab for refractory mycosis fungoides and sezary syndrome. Adv Radiat Oncol. (2021) 6:100629. doi: 10.1016/j.adro.2020.11.014
64. Durgin JS, Jariwala NN, Wysocka M, Zhang KK, Maity A, Benoit B, et al. Low-dose total skin electron beam therapy as part of a multimodality regimen for treatment of sezary syndrome: clinical, immunologic, and molecular analysis. JAMA Dermatol. (2021) 157:90–5. doi: 10.1001/jamadermatol.2020.3958
65. Wilson LD, Jones GW, Kim D, Rosenthal D, Christensen IR, Edelson RI, et al. Experience with total skin electron beam therapy in combination with extracorporeal photopheresis in the management of patients with erythrodermic (T4) mycosis fungoides. J Am Acad Dermatol. (2000) 43:54–60. doi: 10.1067/mjd.2000.105510
66. Yahalom J, Dabaja BS, Ricardi U, Ng A, Mikhaeel NG, Vogelius IR, et al. ILROG emergency guidelines for radiation therapy of hematological Malignancies during the COVID-19 pandemic. Blood. (2020) 135:1829–32. doi: 10.1182/blood.2020006028
67. Elsayad K, Weishaupt C, Moustakis C, Danzer MF, Muller EC, Rolf D, et al. Ultrahypofractionated low-dose total skin electron beam in advanced-stage mycosis fungoides and sezary syndrome. Int J Radiat Oncol Biol Phys. (2023) 117:164–70. doi: 10.1016/j.ijrobp.2023.02.052
68. Goyal A, O'Leary D, Foss F. Allogeneic stem cell transplant for treatment of mycosis fungoides and Sezary syndrome: a systematic review and meta-analysis. Bone Marrow Transplant. (2024) 59:41–51. doi: 10.1038/s41409-023-02122-0
69. Duvic M, Donato M, Dabaja B, Richmond H, Singh L, Wei W, et al. Total skin electron beam and non-myeloablative allogeneic hematopoietic stem-cell transplantation in advanced mycosis fungoides and Sezary syndrome. J Clin Oncol. (2010) 28:2365–72. doi: 10.1200/JCO.2009.25.8301
70. Morris SL, Thomas BR, Palanicawandar R, Whittaker S, Child F, Wain M, et al. Long term outcomes of nonmyeloablative allogeneic stem cell transplantation with TSEB TLI and ATG for Mycosis Fungoides and Sezary Syndrome. Bone Marrow Transplant. (2024) 59:874–9. doi: 10.1038/s41409-024-02236-z
Keywords: total skin electron beam therapy, cutaneous lymphomas, mycosis fungoides, radiation therapy, hematological malignancies
Citation: Specht L (2025) Total skin electron beam therapy. Front. Oncol. 15:1498855. doi: 10.3389/fonc.2025.1498855
Received: 19 September 2024; Accepted: 27 January 2025;
Published: 01 April 2025.
Edited by:
Carla Hajj, Memorial Sloan Kettering Cancer Center, United StatesReviewed by:
Brandon Imber, Memorial Sloan Kettering Cancer Center, United StatesCopyright © 2025 Specht. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lena Specht, bGVuYS5zcGVjaHRAcmVnaW9uaC5kaw==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.