- 1The Affiliated Hospital of Qingdao University, Qingdao, China
- 2Qilu Hospital, Shandong University, Jinan, China
- 3The Second Affiliated Hospital of Shandong First Medical University, Tai’an, China
- 4Department of Obstetrics and Gynecology, Qilu Hospital, Shandong University, Jinan, China
- 5Shandong Second Medical University, Weifang, China
Introduction: Invasive Stratified Mucin-producing Carcinoma (ISMC) of the cervix is a newly named cervical adenocarcinoma associated with Human Papilloma virus (HPV). Due to its relative rarity, clinical data, pathological features, and molecular characteristics of ISMC are still under exploration. This study aims to retrospectively analyze the clinical data and pathological features of ISMC patients, summarizing the clinical and pathological morphological characteristics of ISMC. Immunohistochemistry for SLC7A11, SLC3A2, and PD-L1 will be performed on tumor tissues from ISMC patients to preliminarily explore potential therapeutic targets for ISMC.
Methods: We retrospectively reviewed the electronic medical records and pathological slides of 22 ISMC patients, and performed immunohistochemical staining for solute carrier family 7 member 11 (SLC7A11), solute carrier family 3 member 2 (SLC3A2), and programmed death-ligand 1 (PD-L1).
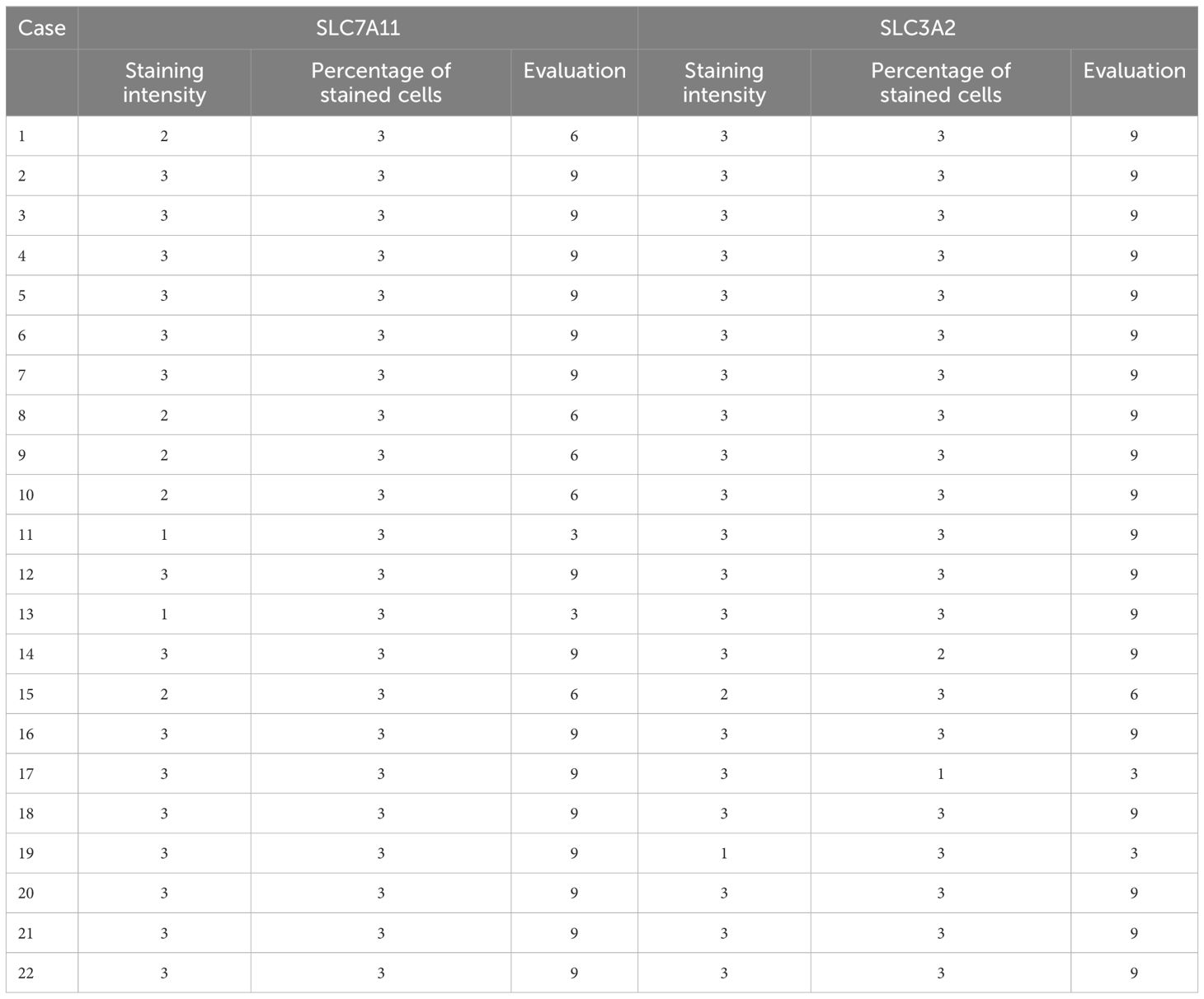
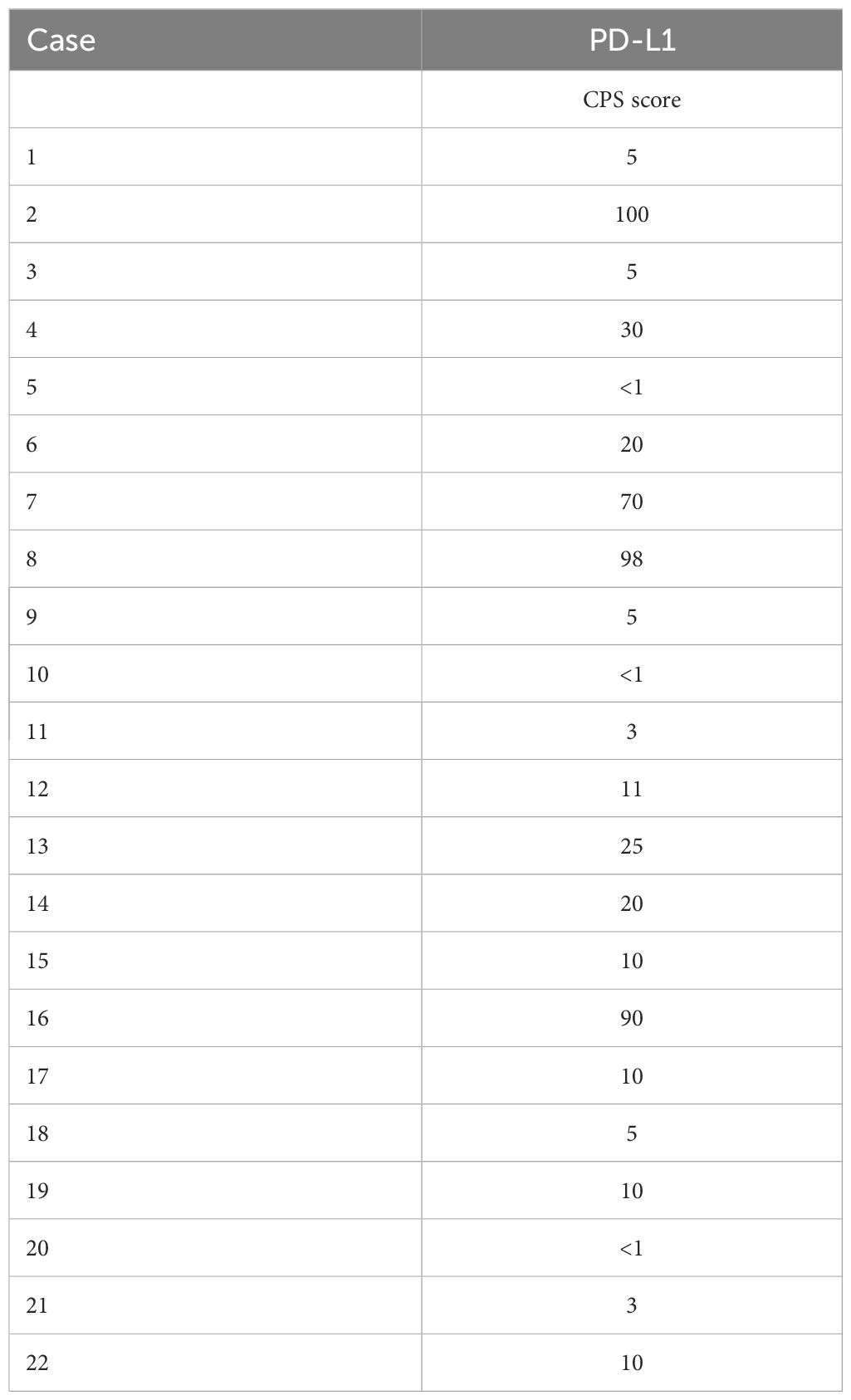
Results: The patients were aged between 31 and 70 years old. The most common symptoms were abnormal vaginal bleeding and unusual vaginal discharge. HPV testing indicated that the infection rate of HPV type 18 was the highest. All patients underwent extensive hysterectomy and pelvic lymph node dissection. The progression-free survival (PFS) ranged from 3 to 112 months, with a postoperative recurrence rate of 22.7% (5/22). ISMC exhibited diverse characteristic microstructures. Immunohistochemistry results showed that the positive rates of SLC7A11 and SLC3A2 were both 91.0% (20/22). The staining intensity of SLC7A11 in frequent ISMC recurrence cases was significantly stronger than in non-recurrent ISMC cases. PD-L1 positivity was observed in 86.4% (19/22) of cases, defined as having a Combined Positive Score(CPS)≥1.
Discussion: ISMC demonstrates a high rate of lymph node metastasis and a high recurrence rate, indicating strong invasiveness. Additionally, ISMC exhibits a wide morphological spectrum. SLC7A11, SLC3A2, and PD-L1 are all highly expressed in ISMC tissues. The high expression of SLC7A11 may indicate a high recurrence rate for ISMC. Immunotherapy with checkpoint inhibitors and iron death-related treatments show potential in the treatment of ISMC, with SLC7A11, SLC3A2, and PD-L1 serving as potential therapeutic targets for ISMC.
Introduction
Invasive Stratified Mucin-producing Carcinoma (ISMC) of the cervix is a newly named type of cervical adenocarcinoma associated with Human Papilloma virus (HPV) (1, 2). Due to the limited availability of publicly documented cases in recent years, clinical data, pathological features, and molecular characteristics of ISMC are still being explored. Currently, there are no guidelines available for the treatment of ISMC. Therefore, it is essential to describe and analyze the characteristics of ISMC.
Ferroptosis was first described in 2012 as a form of programmed cell death caused by iron and lipid peroxidation-dependent reactions (3). As research on ferroptosis has advanced, it has become evident that ferroptosis plays a significant role in the inhibition of tumor progression (4–6). Ferroptosis has been demonstrated to play a significant role in the treatment of malignant tumors through radiotherapy, immunotherapy, and certain chemotherapy methods (7–10). It has been discovered that inhibitors such as ferrostatin and Liproxstatin-1 can effectively inhibit ferroptosis through distinct pathways (11). Furthermore, related drugs are currently in the process of development and testing. Ferroptosis is regulated by several key factors, including SLC7A11, which is a crucial cystine transporter and a member of the solute carrier family 7 (12). The solute carrier family 7 member 11 (SLC7A11, also known as XCT) is the primary transporter of extracellular cystine and serves as the catalytic subunit of the cystine/glutamic acid reverse transporter XC − (13–15). Currently, numerous studies have demonstrated the widespread expression of SLC7A11 in various malignant tumors. It has been found to be linked with the growth, invasion, metastasis, and poor prognosis of malignancy (16–26). Additionally, the targeted treatment strategy of SLC7A11 in cancer therapy has demonstrated its potential in the management of specific malignant tumors (12, 27). The member 2 of the Solute Carrier Family 3 (Recombinant Solute Carrier Family 3, Member 2) forms a complex with SLC7A11 to constitute the cystine/glutamate antiporter protein XC-, and SLC3A2 supports the function of SLC7A11 as a chaperone protein (13, 26). An increasing number of studies have demonstrated the significant role of SLC3A2, similar to SLC7A11, in preventing excessive lipid peroxidation within cells (8, 28). However, the expression of SLC7A11 and SLC3A2 in ISMC is currently unclear. The potential of ferroptosis-related drugs in the treatment of ISMC also requires further exploration.
Targeted therapies targeting the PD-1/PD-L1 pathway have demonstrated promising outcomes in the treatment of various types of cancer (29–32). here is multiple evidence that multiple approaches, including chemotherapy, radiation therapy, and angiogenesis inhibitors, can synergistically target PD-1/PD-L1 through various pathways, such as promoting tumor antigen release, antigen presenting cell function, or effector activity (33–39). Specifically, targeted therapies directed at the PD-1/PD-L1 pathway have demonstrated promising effects in the treatment of cervical cancer, with Pabolizumab being a notable example (40). ISMC is classified as one of the Human Papillomavirus Associated Cervical Adenocarcinoma (HPVA) in the International Endocervical Adenocarcinoma Criteria and Classification (IECC) (2). Correspondingly, the question arises as to whether anti-PD-1 plays a more significant role in HPV-positive tumors, as different clinical trials have yielded varying results (41, 42). In conclusion, further research is needed to investigate the role of targeted therapy of the PD-1/PD-L1 pathway in ISMC.
This paper provides a summary of the clinical and morphological characteristics of Invasive Stratified Mucin-producing Carcinoma (ISMC). The study analyzed data from 22 patients admitted to the Affiliated Hospital of Qingdao University between January 2013 and December 2023, including age of onset, main symptoms, HPV infection, lesion size, diagnosis method, International Federation of Obstetrics and Gynecology (FIGO) stage, concurrent cervical-related diseases, mode of operation, chemoradiation, recurrence, progression-free survival, and HE staining images of pathological tissue. In addition to previous studies on ISMC, this research further explores the expression of iron death related indicators SLC7A11 and SLC3A2. Furthermore, PD-L1 is included in this study as one potential therapeutic immune checkpoint. The purpose of this study was to summarize the clinical and morphological characteristics of ISMC and explore possible therapeutic targets.
Methods
A total of 419 cases of cervical adenocarcinoma, pathologically diagnosed at the Affiliated Hospital of Qingdao University from January 2013 to December 2023, were included in the study. Since ISMC was recently included in the International Standard and Classification System for Cervical Adenocarcinoma (IECC), some previously diagnosed cases may need to be re-evaluated due to the short time frame of its official inclusion. After re-evaluation, 22 cases of ISMC were identified as the subjects for research.
The data of 22 patients with ISMC were collected and subjected to statistical analysis. The parameters included age of onset, main symptoms, HPV infection, lesion size, diagnostic methods, FIGO stage, complicating cervical-related disease, mode of operation, chemoradiotherapy, recurrence, progression-free survival, and HE staining images of pathological tissue.
The Formalin Fixed and Paraffin Embedded (FFPE) tissues from 22 patients with ISMC were sliced and prepared following the standard protocol. IHC staining of SLC7A11, SLC3A2, and PD-L1 was conducted, with normal cervical tissues selected for control experiments. The staining results were then evaluated.
The expression of SLC7A11 and SLC3A2 is assessed based on the intensity and extent of staining. Staining intensity is classified as 0 (no staining), 1 (mild staining), 2 (moderate staining), or 3 (strong staining), while staining extent is divided into grades based on the percentage of stained cells. The total score is calculated by multiplying the intensity score with the distribution score, with a total score greater than or equal to 4 considered positive and a total score between 0-3 considered negative. The Combined Positive Score (CPS) is used to evaluate PD-L1 expression by dividing the number of PD-L1-stained cells by the total number of viable tumor cells and multiplying by 100. At least 100 viable tumor cells are required for evaluating PD-L1 positivity. IHC staining results for SLC7A11, SLC3A2, and PD-L1 are analyzed to assess their respective expressions.
Results
Clinical characteristics of ISMC patients
The 22 patients ranged in age from 31 to 70 years, with an average age of 47 years. The vast majority of patients presented with symptoms of abnormal vaginal bleeding (19/22), while 3 cases exhibited abnormal fluid discharge from the vagina (3/22), and 2 cases had abnormal vaginal discharge (2/22). In addition, one patient was diagnosed with cervical adenocarcinoma through colposcopic biopsy despite exhibiting no obvious clinical symptoms. This diagnosis was prompted by a positive HPV test for type 18. Twenty patients tested positive for HPV, one tested negative, and one declined to be tested. The specific HPV subtype was identified in 11 cases. Among these, 9 cases were found to have HPV type 18 infection (9/11), 2 cases had HPV type 16 infection (2/11), and 1 case had both HPV types 18 and 45 (1/11). he majority of patients were diagnosed with ISMC through biopsy (19/22), one case was diagnosed through diagnostic conization (1/22), and one case was pathologically diagnosed as high-grade squamous intraepithelial lesion (HSIL) via biopsy, followed by diagnosis through cervical conization (1/22). Additionally, a patient who did not undergo HPV testing and cervical liquid-based cytology testing received vaginal cervical myomectomy due to vaginal bleeding. Postoperative pathology suggested ISMC (1/22). According to the latest staging of the International Federation of Gynecology and Obstetrics (FIGO), there were 15 cases in stage I (15/22), 2 cases in stage II (2/22), and 5 cases in stage IIIC1p (5/22) (Table 1).
ISMC patient treatment
Among the patients (7/22), some received preoperative chemotherapy, with the majority of them undergoing 1-2 cycles of intravenous chemotherapy using a combination of paclitaxel and platinum (5/7). In addition to paclitaxel and cisplatin, a patient has added fluorouracil to the chemotherapy regimen (1/7). Another patient presented with severe vaginal bleeding and underwent paclitaxel intravenous chemotherapy and cisplatin interventional embolization therapy (1/7). All patients underwent comprehensive hysterectomy and pelvic lymph node dissection. It is worth mentioning that some patients underwent laparoscopic surgery (4/22), while others underwent open abdominal surgery (18/22). In addition, the patients underwent bilateral salpingectomy (20/22), bilateral tubal ligation and bilateral ovarian biopsy (2/22), para-aortic lymph node dissection (6/22) and presacral lymph node dissection (2/22). In terms of postoperative treatment, 13 cases underwent adjuvant chemotherapy and radiotherapy (13/22), 5 cases received only paclitaxel + platinum-based chemotherapy (5/22), 1 case underwent only radiotherapy after surgery (1/22), and 3 cases did not receive any additional treatment after surgical intervention (3/22) (Table 2).
Follow-up of ISMC patients
In this study, the progression-free survival (PFS) of all ISMC patients ranged from 3 to 112 months (average 33.95 months), with a 1-year recurrence rate of 4.5% and a 2-year recurrence rate reaching 18.2%. This study included a total of 5 cases of postoperative recurrence. Among them, 2 cases showed elevated tumor markers without any imaging evidence of disease progression, and both patients continued regular follow-up. One patient had pelvic lesions identified on PET-CT and chose to undergo oral traditional Chinese medicine treatment. Additionally, 2 cases of malignant tumors invaded the urinary system and resulted in hydronephrosis; one case experienced three recurrences, with the first two recurrences treated with paclitaxel, cisplatin, and fluorouracil chemotherapy via intravenous infusion. The third recurrence led to hemorrhagic shock caused by vaginal bleeding, requiring a sigmoid colostomy due to rectovaginal fistula. The other case underwent transurethral resection of bladder lesions and percutaneous nephrostomy for drainage before receiving traditional Chinese medicine treatment postoperatively (Table 3).
Macroscopic characteristics of ISMC
Overall morphological features indicate that ISMC presents as ulcerative, cauliflower-like, or polypoid tumors with rough cut surface, and grayish-white or grayish-yellow coloration. The maximum diameter of the tumor is 7-60mm, with an average of 28mm (Table 4).
Microstructural characteristics of ISMC
In the HE stained sections of ISMC cases, a stratified mucin-producing intraepithelial lesion (SMILE) involving the cervical glands can be observed (Figure 1A). Unlike traditional squamous cell carcinoma and adenocarcinoma, this type presents as a mixture of stratified epithelial cells and immature nuclei, similar to high-grade squamous intraepithelial lesion (HSIL), but also shows varying proportions of intracytoplasmic vacuoles containing mucin. The quantity of intracellular adhesive proteins ranges from “rich in adhesive proteins” lesions, which show a large amount of adhesive proteins in most cells, to “deficient in adhesive proteins” lesions, which only display a relatively limited number of cells containing adhesive proteins (Figures 1B, C). Under low magnification, it is clearly observed that the cells producing adhesive proteins are distributed throughout the entire epithelial cells. The presence of invasive nests formed by multi-layered mucous cells with peripheral fence-like patterns is considered as clear evidence of invasion (Figures 1D, E). Neutrophil infiltration can be observed around the cancer nests (Figure 1F). Peripheral to some lesions, there is evidence of nuclear atypia in the stratified epithelium. Within the lesion, mucin vacuoles can be observed within the cytoplasm of cancer cells (Figure 1G). In the lesion, membrane shrinkage and internal invagination can be found, dividing and enclosing the cytoplasm to form vesicular bodies, known as apoptotic bodies (Figure 1H). Some lesions present extravasated mucin pools (Figure 1I). The invasion of cervical intraepithelial neoplasia by SMLIE can be observed in multiple slices, adjacent to the ISMC lesion (Figure 1J). This indirectly proves their progressive relationship. In this study, out of the 22 cases of ISMC, 2 cases were found to be combined with HISL (Figure 1K), and 3 cases were found to be combined with Adenocarcinoma In Situ (AIS) (Figure 1L). These findings were all demonstrated in HE sections.
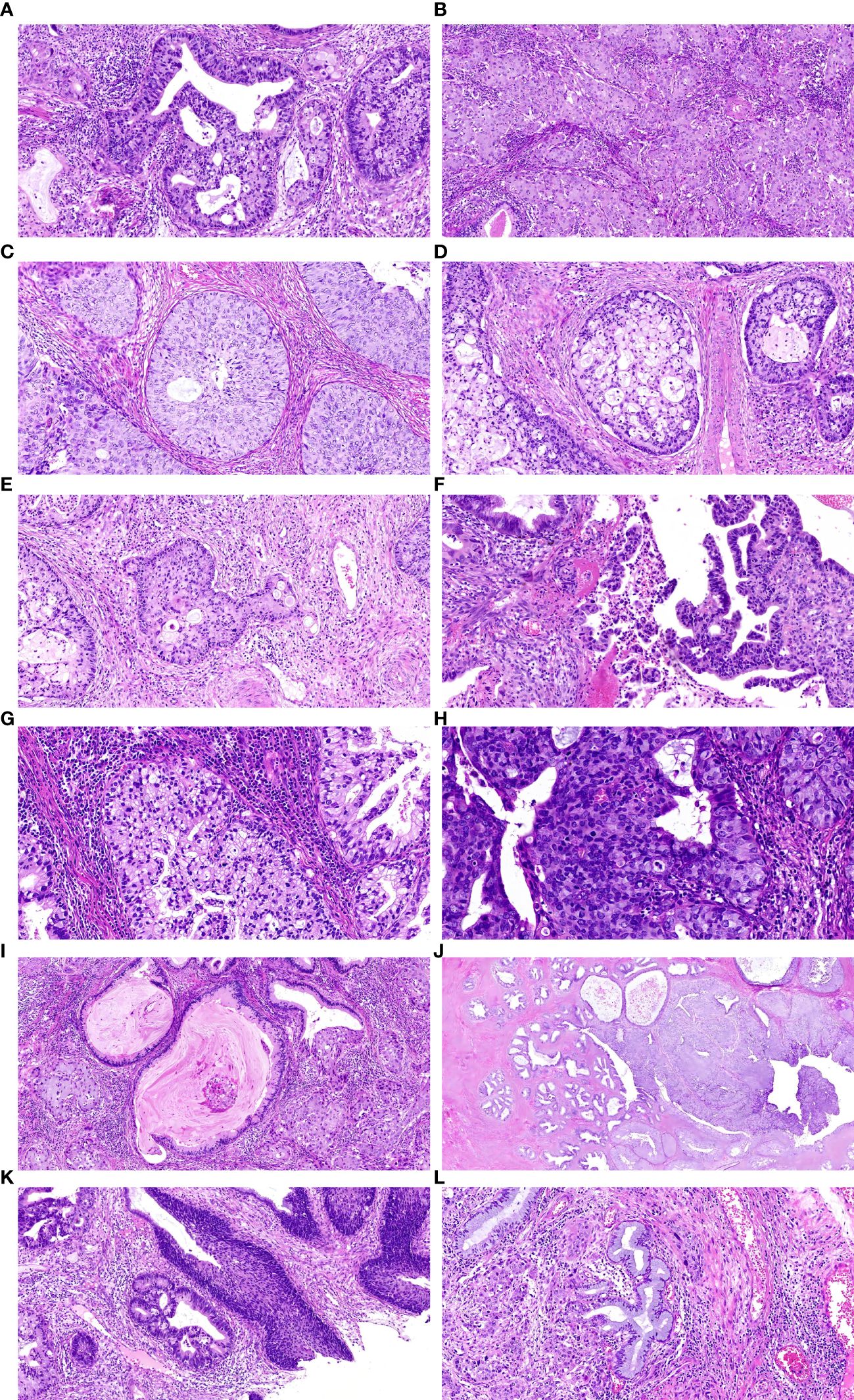
Figure 1. (A) shows a typical SMILE lesion, (B) displays a “rich in mucin” SMILE with abundant mucins in the cytoplasm. (C) illustrates a “mucin-poor” SMILE with relatively low levels of mucins present in the cytoplasm, (D) presents a typical ISMC lesion. (E) demonstrates infiltrative mucinous carcinoma cells nests exhibiting glandular-like changes within surrounding cell nuclei, (F) showcases abundant presence of mucins within the cytoplasm along with infiltration by peripheral neutrophils. (G) presents stratified epithelium displaying nuclear atypia and intracytoplasmic vacuoles filled with mucus, (H) represents apoptotic bodies, (I)indicates an extracellular pool of mucus, (J) shows SMILE involving endocervical glands on the left transitioning into an ISMC lesion on the right, (K) displays combination of ISMC and HSIL (L) revealing concurrent development of original site adenocarcinoma within an ISMC case.
Immunohistochemical staining results of SLC7A11 and SLC3A2
The expression of SLC7A11 demonstrated a high positivity rate, reaching 91.0% (20/22) (Table 5). Its expression was observed in the cytoplasm and cell membrane, with no apparent staining in the cell nucleus (Figure 2). Similarly, the expression rate of SLC3A2 also reached 91.0% (20/22) (Table 5), and its expression was localized to the cell membrane, with less prominent staining in the cell nucleus and cytoplasm (Figure 2).

Figure 2. ISMC tissue staining for SLC7A11 and SLC3A2 is depicted in the upper left and upper right, while staining of SLC7A11 and SLC3A2 in normal cervical tissue is shown in the lower left and lower right.
Comparison of immunohistochemical staining results for SLC7A11 and SLC3A2 in frequent recurrent ISMC cases versus non-recurrent ISMC cases
Ever since Bannai and Kitamura first identified it in 1980, there has been a significant rise in the number of studies showing the extensive presence of SLC7A11 in different types of cancer, along with its various impacts on cancer development, spread, migration, and poor prognosis (16–26, 43–47). In this study, a comparison of the staining for SLC7A11 and SLC3A2 was conducted between ISMC cases with three recurrences and those without recurrence. The staining intensity of SLC7A11 in frequent ISMC recurrence cases was significantly stronger than that in non-recurrent ISMC cases, while the difference in staining intensity of SLC3A2 between the two was not significant (Figure 3).

Figure 3. Comparison of tissue staining for SLC7A11 (top left) and SLC3A2 (top right) in cases of frequent recurrent ISMC is contrasted with tissue staining for SLC7A11 (bottom left) and SLC3A2 (bottom right) in non-recurrent ISMC cases.
Immunohistochemical staining results for PD-L1
The immunohistochemistry results revealed that the positive rate of PD-L1 (CPS≥1) was 86.4% (19/22), with a maximum CPS score of 100 (Table 6; Figure 4). PD-L1 was found to be located in both tumor cells and tumor-associated immune cells within the tumor stroma.
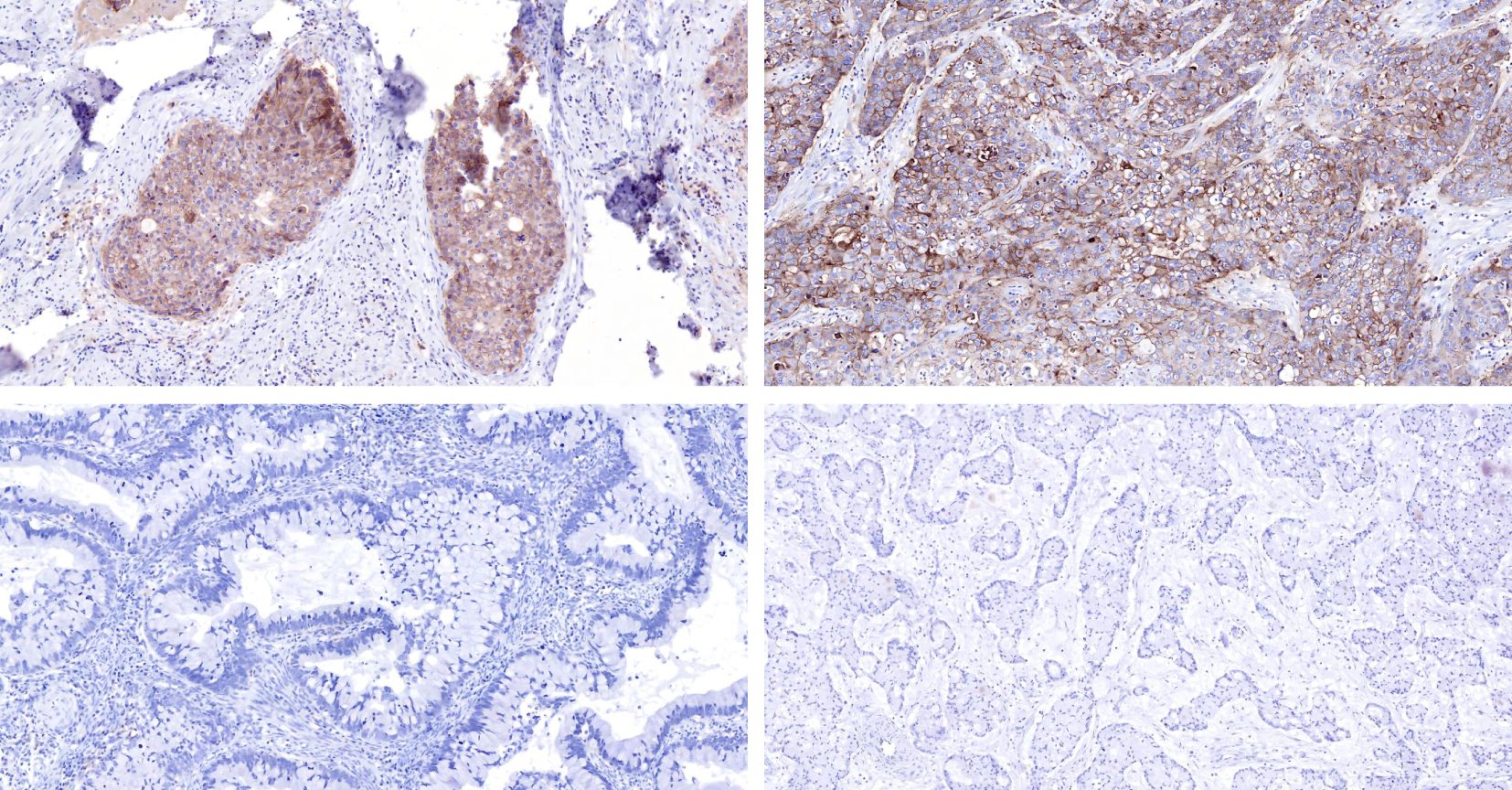
Figure 4. Comparison is made between tissue staining of PD-L1 CPS scores of 100 (top left) and 98 (top right) in ISMC cases, and tissue staining of PD-L1 CPS scores <1 (bottom left, bottom right) in ISMC cases.
Discussion
The ISMC has been recently discovered as a type of cervical adenocarcinoma associated with HPV. Prior to ISMC being officially included in IECC, these instances were often incorrectly diagnosed as adenocarcinoma, squamous cell carcinoma, or different forms of cervical intraepithelial neoplasia (48). At present, there is limited comprehensive statistical information available regarding the prevalence of IECC. An examination of 200 instances of cervical squamous cell carcinoma and adenocarcinoma found that ISMC represents 5% of cases (49). During the same time period, our hospital identified a total of 419 cases of cervical adenocarcinoma, with ISMC accounting for 5.3% (22/419) of the cases. Previous studies have shown that ISMC displays more aggressive clinical and pathological features when compared to conventional HPV-related cervical adenocarcinoma (50–52). The study found that 22.7% (5/22) of ISMC cases had lymph node metastasis, demonstrating its significant invasiveness and emphasizing the importance of precise diagnosis for ISMC.
As a result of the small number of documented cases and lack of clear understanding regarding its natural progression, there is currently no widely accepted standard for treating and predicting the outcome of ISMC. Additionally, there is limited available evidence to use as a reference. The clinical staging, rather than the histological subtype, dictates the prognosis and treatment for patients with cervical cancer. The survival rates differ greatly depending on the stage of the disease. Patients with cervical adenocarcinoma show varying survival rates at different stages, including a 5-year survival rate of 89.8% for stage I, 79.1% for stage II, 47.7% for stage III, and 42.4% for stage IV (53). The majority of research has indicated that tumor size, tumor grade, and lymph node metastasis are all considered as separate prognostic factors. Furthermore, there is also evidence to suggest that marital status may be an independent prognostic factor (53). In the initial phases of the illness (IA1 to IB1), local surgical techniques such as conization, radical hysterectomy, and evaluation of lymph node status are frequently employed as primary treatment approaches (53). Radiotherapy is a potential option for patients who are not eligible for surgery or refuse surgical treatment. The typical course of treatment for individuals with stage IB2-IVA cervical cancer includes simultaneous cisplatin-based chemoradiotherapy in conjunction with brachytherapy (54). The therapy is suitable for individuals with glandular and squamous conditions, and additional research using clinical samples is needed to assess its effectiveness in preventing the spread of cancer triggered by aggressive laminin.
The age at which ISMC patients in this study first experienced symptoms varied from 31 to 70 years old. A different study on ISMC found that the average age of onset ranged from 37 to 75 years old, with a mean age of 50.5 years, suggesting similar findings in terms of the distribution of ages (49). This indicates that there is a potential for ISMC to occur in young women as well. The prevailing agreement is that ISMC is strongly linked to high-risk HPV, specifically types 18 and 16 (49, 55). This research supports the above perspective by showing that 10 out of 11 cases were infected with HPV 18, and only 2 out of 11 cases were infected with HPV 16, when specific HPV genotypes could be identified. HPV is present in most of the cases that are publicly accessible (2, 50, 55). Not all instances of ISMC can be identified through HPV infection. In this research, one out of 21 cases showed a negative result for HPV, and similar occurrences of undetected HPV have also been documented in other studies (3 out of 12) (56). The IECC defines ISMC as cervical cancer related to HPV, and current evidence supports this idea. However, more research is needed to understand the underlying mechanisms. It is also important to investigate why some cases of HPV go undetected, as the limited sensitivity of HPV detection alone may not fully explain this phenomenon. The prognostic significance of different high-risk HPV genotypes, particularly HPV16, 18, and 45, in relation to cervical cancer remains uncertain. Some research has indicated positive outcomes for cervical cancer associated with HPV16 and/or 18 positivity (57–61), while others have suggested a less favorable prognosis (62–72). Furthermore, certain studies have found that high-risk HPV genotypes do not hold prognostic value (73–76). The notable distinctions in clinical characteristics between cervical squamous cell carcinoma and cervical adenocarcinoma, as well as HPV-related and non-HPV-related endocervical adenocarcinomas, underscore the significance of acknowledging inherent prejudices in research investigations. Given the substantial differences in clinical behavior between these histological types and etiologies of cervical cancer, it may be optimal to investigate the prognostic significance of high-risk HPV genotypes based on these factors. Due to the infrequent occurrence of ISMC cases, there is a restricted amount of documented cases accessible. Our objective is to gather a sufficient quantity of ISMC cases for the purpose of analyzing the predictive importance of high-risk HPV genotypes.
It has been documented that PD-L1 expression is observed in tumor cells and different types of immune cells in both cervical squamous cell carcinoma and adenocarcinoma (77). Compared to cervical endometrial adenocarcinoma, squamous cell carcinoma exhibits a higher rate of PD-L1 positivity and an increased presence of immune cells expressing PD-L1. Furthermore, studies suggest that patients with tumor-associated macrophages showing PD-L1 expression in cervical endometrial adenocarcinoma experience a significantly shorter period of progression-free survival compared to those with tumors lacking PD-L1 expression (77). Recent studies of 27 instances of endometrial adenocarcinoma samples found that positive PD-L1 expression was observed in tumor cells in 3.7% of cases, while immune cells showed positive PD-L1 expression in 7.4% of cases (78). The research on cervical adenocarcinoma found no link between the presence of high-risk HPV genotypes and the levels of PD-L1 expression (79). While the occurrence of PD-L1 expression is infrequent, its detection in either tumor cells or immune cells is strongly associated with unfavorable outcomes for individuals diagnosed with cervical adenocarcinoma (78). The presence of PD-L1 expression could indicate the tumor’s defensive reaction to immune attack by compensatory upregulation of PD-L1 or adaptive immune resistance (80, 81). Overall clinical outcomes often depend on the balance between tumor-induced immune responses and immunosuppressive mechanisms, such as the interaction of programmed cell death protein 1/PD-L1. The simultaneous increase in CD8+ tumor-infiltrating lymphocytes and PDL1 expression in cervical adenocarcinoma suggests a tendency towards immune resistance (82), leading to more aggressive tumor behavior. In this research, PD-L1 expression in ISMC patients was evaluated using the CPS system. The results of immunohistochemistry showed that 86.4% (19/22) of patients had a positive rate of PD-L1 (CPS≥1), with the highest CPS score being 98. PD-L1 was detected in both tumor cells and tumor-associated immune cells within the tumor stroma. This finding suggests that high levels of PD-L1 expression may be associated with aggressive behavior and a worse prognosis in ISMC. The trial approved by the FDA in the United States has shown that pembrolizumab, an anti-PD-1 inhibitor, is effective in treating advanced cervical cancer patients with PD-L1 positive status (CPISMC 1) and leads to improved outcomes for patients with recurrent or metastatic cervical cancer (83). These results indicate that PD-1/PD-L1 immunotherapy may be beneficial for these patients.
SLC7A11 serves as the catalytic component of the amino acid transportation system XC-, which is accountable for taking in extracellular cystine molecules and converting them into cysteine, ultimately producing glutathione (84). GSH acts as a strong remover of lipid peroxidation and is an essential coenzyme for the activity of glutathione peroxidase 4 (Gpx4). It has a crucial function in eliminating phospholipid peroxides and protecting cells from ferroptosis (85). As a result, the SLC7A11-GSH system acts as the primary defense mechanism against ferroptosis and plays a crucial role in regulating cellular iron balance (15). Inhibiting SLC7A11 may hinder the production of cysteine and the synthesis of glutathione, potentially resulting in cell death caused by excessive accumulation of lipid peroxidation due to high levels of iron (86). Cysteine can be partially replenished through the sulfurization pathway or alternative non-selective amino acid transporters (87). Cancer cells require a large amount of cysteine and glutathione to neutralize the high levels of reactive oxygen species in their cells, resulting in an upregulation of SLC7A11 as a component of their reliance on nutrients (12, 88). SLC7A11 has been found to be overexpressed in many types of human cancers, and specifically inhibiting SLC7A11 shows a high level of responsiveness (89). The inhibition of the SLC7A11-GSH pathway has been shown to have a significant anti-tumor effect in various types of human cancers (90). Our study indicates that gaining a deeper understanding of the functions of cysteine and SLC7A11 in controlling the redox activity proteins and their interactions could present a promising strategy for treating cancer. The high rate of SLC7A11 expression in ISMC, which aligns with its significant invasiveness, metastasis, and unfavorable prognosis, offers new perspectives on the currently uncertain treatment of ISMC. It is worth noting that there are indications that the overexpression of SLC7A11 in cancerous tumors can increase the activity of the PD-L1 pathway, resulting in the infiltration of tumor-associated macrophages (TAM) and myeloid-derived suppressor cells (MDSC) within the tumor. Inhibiting this oncogenic pathway could potentially serve as an effective therapeutic approach for preventing SLC7A11-mediated metastasis of malignant tumors (91).
SLC3A2, also referred to as CD98, CD98hc or 4F2hc, is a type II transmembrane protein with a molecular weight of 80 kDa that is mainly positioned facing the outside of the cell (92). SLC3A2 is essential for regulating ferroptosis, apoptosis, and proliferation in cancer cells by interacting with XCT (90, 93, 94). The increased expression of SLC3A2 has been associated with the initiation and advancement of various forms of cancer because of its wide range of molecular activities. Previous studies have indicated that SLC3A2 plays a role in the progression of oral cancer by promoting cell growth and suppressing programmed cell death in individuals (94). Our research aims to fill the gap in literature regarding the expression of SLC3A2 in ISMC, and also illustrates the role of the XCT system, consisting of SLC7A11 and SLC3A2, in the iron death process. These findings suggest that iron death-related therapy may be a potential treatment for ISMC, indicating that this mechanism is present in these cells.
Our research has certain limitations, as a considerable number of ISMC cases had already undergone surgery before the formal introduction of the ISMC concept, which prevented us from obtaining the latest tissues and cells of ISMC. Therefore, we were unable to conduct further in vivo and in vitro experiments. Due to the rarity of ISMC, our study had a limited number of cases, which restricted us from conducting some statistical studies, such as investigating whether HPV18 infection is associated with the expression of SLC7A11, SLC3A2 and PD-L1. If given the opportunity, we will make efforts to increase the number of ISMC cases and collect new ISMC tissues for improvement.
In summary, this research has analyzed clinical data and pathological characteristics of cervical endometrioid adenocarcinoma. Due to the varied appearance of this cancer type, a thorough examination is crucial for an accurate diagnosis, especially for less experienced pathologists. Even seasoned pathologists may face difficulties in distinguishing cervical endometrioid adenocarcinoma from other types. The frequent occurrence of lymph node and distant metastasis, as well as postoperative recurrence, indicates the aggressive nature and poor prognosis associated with cervical endometrioid adenocarcinoma. Based on the findings of the immunohistochemistry study, it is possible that ISMC patients could benefit from immune checkpoint inhibitors that target PD-L1/PD-1 and ferroptosis-related therapy aimed at SLC7A11.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding authors.
Ethics statement
The studies involving humans were approved by Medical Ethics Committee of Affiliated Hospital of Qingdao University. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
CL: Data curation, Writing – original draft, Writing – review & editing. WZ: Data curation, Writing – original draft. XH: Investigation, Writing – original draft. XD: Investigation, Writing – original draft. HZ: Funding acquisition, Resources, Writing – review & editing. QY: Funding acquisition, Writing – review & editing. TL: Writing – review & editing. CZ: Writing – original draft.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Park JJ, Sun D, Quade BJ, Flynn C, Sheets EE, Yang A, et al. Stratified mucin-producing intraepithelial lesions of the cervix: adenosquamous or columnar cell neoplasia? Am J Surg Pathol. (2000) 24:1414–9. doi: 10.1097/00000478-200010000-00012
2. Stolnicu S, Barsan I, Hoang L, Patel P, Terinte C, Pesci A, et al. International endocervical adenocarcinoma criteria and classification (Iecc): A new pathogenetic classification for invasive adenocarcinomas of the endocervix. Am J Surg Pathol. (2018) 42:214–26. doi: 10.1097/pas.0000000000000986
3. Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. (2012) 149:1060–72. doi: 10.1016/j.cell.2012.03.042
4. Jiang L, Kon N, Li T, Wang SJ, Su T, Hibshoosh H, et al. Ferroptosis as a P53-mediated activity during tumour suppression. Nature. (2015) 520:57–62. doi: 10.1038/nature14344
5. Zhang Y, Shi J, Liu X, Feng L, Gong Z, Koppula P, et al. Bap1 links metabolic regulation of ferroptosis to tumour suppression. Nat Cell Biol. (2018) 20:1181–92. doi: 10.1038/s41556-018-0178-0
6. Chu B, Kon N, Chen D, Li T, Liu T, Jiang L, et al. Alox12 is required for P53-mediated tumour suppression through a distinct ferroptosis pathway. Nat Cell Biol. (2019) 21:579–91. doi: 10.1038/s41556-019-0305-6
7. Guo J, Xu B, Han Q, Zhou H, Xia Y, Gong C, et al. Ferroptosis: A novel anti-tumor action for cisplatin. Cancer Res Treat. (2018) 50:445–60. doi: 10.4143/crt.2016.572
8. Wang W, Green M, Choi JE, Gijón M, Kennedy PD, Johnson JK, et al. Cd8(+) T cells regulate tumour ferroptosis during cancer immunotherapy. Nature. (2019) 569:270–4. doi: 10.1038/s41586-019-1170-y
9. Lang X, Green MD, Wang W, Yu J, Choi JE, Jiang L, et al. Radiotherapy and immunotherapy promote tumoral lipid oxidation and ferroptosis via synergistic repression of slc7a11. Cancer Discovery. (2019) 9:1673–85. doi: 10.1158/2159-8290.Cd-19-0338
10. Friedmann Angeli JP, Krysko DV, Conrad M. Ferroptosis at the crossroads of cancer-acquired drug resistance and immune evasion. Nat Rev Cancer. (2019) 19:405–14. doi: 10.1038/s41568-019-0149-1
11. Xie Y, Hou W, Song X, Yu Y, Huang J, Sun X, et al. Ferroptosis: process and function. Cell Death differentiation. (2016) 23:369–79. doi: 10.1038/cdd.2015.158
12. Koppula P, Zhuang L, Gan B. Cystine transporter slc7a11/xct in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell. (2021) 12:599–620. doi: 10.1007/s13238-020-00789-5
13. Lim JC, Donaldson PJ. Focus on molecules: the cystine/glutamate exchanger (System X(C)(-)). Exp eye Res. (2011) 92:162–3. doi: 10.1016/j.exer.2010.05.007
14. Conrad M, Sato H. The oxidative stress-inducible cystine/glutamate antiporter, system X (C) (-): cystine supplier and beyond. Amino Acids. (2012) 42:231–46. doi: 10.1007/s00726-011-0867-5
15. Koppula P, Zhang Y, Zhuang L, Gan B. Amino acid transporter slc7a11/xct at the crossroads of regulating redox homeostasis and nutrient dependency of cancer. Cancer Commun (Lond). (2018) 38:12. doi: 10.1186/s40880-018-0288-x
16. Zhao X, Li Y, Wu H. A novel scoring system for acute myeloid leukemia risk assessment based on the expression levels of six genes. Int J Mol Med. (2018) 42:1495–507. doi: 10.3892/ijmm.2018.3739
17. Lanzardo S, Conti L, Rooke R, Ruiu R, Accart N, Bolli E, et al. Immunotargeting of antigen xct attenuates stem-like cell behavior and metastatic progression in breast cancer. Cancer Res. (2016) 76:62–72. doi: 10.1158/0008-5472.Can-15-1208
18. Briggs KJ, Koivunen P, Cao S, Backus KM, Olenchock BA, Patel H, et al. Paracrine induction of hif by glutamate in breast cancer: egln1 senses cysteine. Cell. (2016) 166:126–39. doi: 10.1016/j.cell.2016.05.042
19. Timmerman LA, Holton T, Yuneva M, Louie RJ, Padró M, Daemen A, et al. Glutamine sensitivity analysis identifies the xct antiporter as a common triple-negative breast tumor therapeutic target. Cancer Cell. (2013) 24:450–65. doi: 10.1016/j.ccr.2013.08.020
20. Yin F, Yi S, Wei L, Zhao B, Li J, Cai X, et al. Microarray-based identification of genes associated with prognosis and drug resistance in ovarian cancer. J Cell Biochem. (2019) 120:6057–70. doi: 10.1002/jcb.27892
21. Sugano K, Maeda K, Ohtani H, Nagahara H, Shibutani M, Hirakawa K. Expression of xct as a predictor of disease recurrence in patients with colorectal cancer. Anticancer Res. (2015) 35:677–82.
22. Ji X, Qian J, Rahman SMJ, Siska PJ, Zou Y, Harris BK, et al. Xct (Slc7a11)-mediated metabolic reprogramming promotes non-small cell lung cancer progression. Oncogene. (2018) 37:5007–19. doi: 10.1038/s41388-018-0307-z
23. Zhong W, Weiss HL, Jayswal RD, Hensley PJ, Downes LM, St Clair DK, et al. Extracellular redox state shift: A novel approach to target prostate cancer invasion. Free Radical Biol Med. (2018) 117:99–109. doi: 10.1016/j.freeradbiomed.2018.01.023
24. Zhang L, Huang Y, Ling J, Zhuo W, Yu Z, Luo Y, et al. Overexpression of slc7a11: A novel oncogene and an indicator of unfavorable prognosis for liver carcinoma. Future Oncol (London England). (2018) 14:927–36. doi: 10.2217/fon-2017-0540
25. Kinoshita H, Okabe H, Beppu T, Chikamoto A, Hayashi H, Imai K, et al. Cystine/glutamic acid transporter is a novel marker for predicting poor survival in patients with hepatocellular carcinoma. Oncol Rep. (2013) 29:685–9. doi: 10.3892/or.2012.2162
26. Wada F, Koga H, Akiba J, Niizeki T, Iwamoto H, Ikezono Y, et al. High expression of cd44v9 and xct in chemoresistant hepatocellular carcinoma: potential targets by sulfasalazine. Cancer Sci. (2018) 109:2801–10. doi: 10.1111/cas.13728
27. Hong T, Lei G, Chen X, Li H, Zhang X, Wu N, et al. Parp inhibition promotes ferroptosis via repressing slc7a11 and synergizes with ferroptosis inducers in brca-proficient ovarian cancer. Redox Biol. (2021) 42:101928. doi: 10.1016/j.redox.2021.101928
28. Zhang L, Liu W, Liu F, Wang Q, Song M, Yu Q, et al. Imca induces ferroptosis mediated by slc7a11 through the ampk/mtor pathway in colorectal cancer. Oxid Med Cell Longevity. (2020) 2020:1675613. doi: 10.1155/2020/1675613
29. Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, et al. Improved survival with ipilimumab in patients with metastatic melanoma. New Engl J Med. (2010) 363:711–23. doi: 10.1056/NEJMoa1003466
30. Massard C, Gordon MS, Sharma S, Rafii S, Wainberg ZA, Luke J, et al. Safety and efficacy of durvalumab (Medi4736), an anti-programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin oncology: Off J Am Soc Clin Oncol. (2016) 34:3119–25. doi: 10.1200/jco.2016.67.9761
31. McDermott DF, Sosman JA, Sznol M, Massard C, Gordon MS, Hamid O, et al. Atezolizumab, an anti-programmed death-ligand 1 antibody, in metastatic renal cell carcinoma: long-term safety, clinical activity, and immune correlates from a phase ia study. J Clin oncology: Off J Am Soc Clin Oncol. (2016) 34:833–42. doi: 10.1200/jco.2015.63.7421
32. Hui R, Garon EB, Goldman JW, Leighl NB, Hellmann MD, Patnaik A, et al. Pembrolizumab as first-line therapy for patients with pd-L1-positive advanced non-small cell lung cancer: A phase 1 trial. Ann oncology: Off J Eur Soc Med Oncol. (2017) 28:874–81. doi: 10.1093/annonc/mdx008
33. Xu J, Zhang Y, Jia R, Yue C, Chang L, Liu R, et al. Anti-pd-1 antibody shr-1210 combined with apatinib for advanced hepatocellular carcinoma, gastric, or esophagogastric junction cancer: an open-label, dose escalation and expansion study. Clin Cancer research: an Off J Am Assoc Cancer Res. (2019) 25:515–23. doi: 10.1158/1078-0432.Ccr-18-2484
34. Horn L, Mansfield AS, Szczęsna A, Havel L, Krzakowski M, Hochmair MJ, et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. New Engl J Med. (2018) 379:2220–9. doi: 10.1056/NEJMoa1809064
35. Luke JJ, Lemons JM, Karrison TG, Pitroda SP, Melotek JM, Zha Y, et al. Safety and clinical activity of pembrolizumab and multisite stereotactic body radiotherapy in patients with advanced solid tumors. J Clin oncology: Off J Am Soc Clin Oncol. (2018) 36:1611–8. doi: 10.1200/jco.2017.76.2229
36. Lee SJ, Yang H, Kim WR, Lee YS, Lee WS, Kong SJ, et al. Sting activation normalizes the intraperitoneal vascular-immune microenvironment and suppresses peritoneal carcinomatosis of colon cancer. J immunotherapy Cancer. (2021) 9. doi: 10.1136/jitc-2020-002195
37. Davar D, Dzutsev AK, McCulloch JA, Rodrigues RR, Chauvin JM, Morrison RM, et al. Fecal microbiota transplant overcomes resistance to anti-pd-1 therapy in melanoma patients. Sci (New York NY). (2021) 371:595–602. doi: 10.1126/science.abf3363
38. Ruan H, Hu Q, Wen D, Chen Q, Chen G, Lu Y, et al. A dual-bioresponsive drug-delivery depot for combination of epigenetic modulation and immune checkpoint blockade. Advanced materials (Deerfield Beach Fla). (2019) 31:e1806957. doi: 10.1002/adma.201806957
39. Ott PA, Hu-Lieskovan S, Chmielowski B, Govindan R, Naing A, Bhardwaj N, et al. A phase ib trial of personalized neoantigen therapy plus anti-pd-1 in patients with advanced melanoma, non-small cell lung cancer, or bladder cancer. Cell. (2020) 183:347–62.e24. doi: 10.1016/j.cell.2020.08.053
40. Colombo N, Dubot C, Lorusso D, Caceres MV, Hasegawa K, Shapira-Frommer R, et al. Pembrolizumab for persistent, recurrent, or metastatic cervical cancer. New Engl J Med. (2021) 385:1856–67. doi: 10.1056/NEJMoa2112435
41. Ferris RL, Blumenschein G Jr., Fayette J, Guigay J, Colevas AD, Licitra L, et al. Nivolumab for recurrent squamous-cell carcinoma of the head and neck. New Engl J Med. (2016) 375:1856–67. doi: 10.1056/NEJMoa1602252
42. Cohen EEW, Soulières D, Le Tourneau C, Dinis J, Licitra L, Ahn MJ, et al. Pembrolizumab versus methotrexate, docetaxel, or cetuximab for recurrent or metastatic head-and-neck squamous cell carcinoma (Keynote-040): A randomised, open-label, phase 3 study. Lancet (London England). (2019) 393:156–67. doi: 10.1016/s0140-6736(18)31999-8
43. Polewski MD, Reveron-Thornton RF, Cherryholmes GA, Marinov GK, Aboody KS. Slc7a11 overexpression in glioblastoma is associated with increased cancer stem cell-like properties. Stem Cells Dev. (2017) 26:1236–46. doi: 10.1089/scd.2017.0123
44. Robert SM, Buckingham SC, Campbell SL, Robel S, Holt KT, Ogunrinu-Babarinde T, et al. Slc7a11 expression is associated with seizures and predicts poor survival in patients with Malignant glioma. Sci Trans Med. (2015) 7:289ra86. doi: 10.1126/scitranslmed.aaa8103
45. Shin SS, Jeong BS, Wall BA, Li J, Shan NL, Wen Y, et al. Participation of xct in melanoma cell proliferation in vitro and tumorigenesis in vivo. Oncogenesis. (2018) 7:86. doi: 10.1038/s41389-018-0098-7
46. Shen L, Qian C, Cao H, Wang Z, Luo T, Liang C. Upregulation of the solute carrier family 7 genes is indicative of poor prognosis in papillary thyroid carcinoma. World J Surg Oncol. (2018) 16:235. doi: 10.1186/s12957-018-1535-y
47. Lee JR, Roh JL, Lee SM, Park Y, Cho KJ, Choi SH, et al. Overexpression of cysteine-glutamate transporter and cd44 for prediction of recurrence and survival in patients with oral cavity squamous cell carcinoma. Head Neck. (2018) 40:2340–6. doi: 10.1002/hed.25331
48. Stolnicu S, Hoang L, Zhou Q, Iasonos A, Terinte C, Pesci A, et al. Cervical adenosquamous carcinoma: detailed analysis of morphology, immunohistochemical profile, and outcome in 59 cases. Int J gynecological pathology: Off J Int Soc Gynecological Pathologists. (2023) 42:259–69. doi: 10.1097/pgp.0000000000000921
49. Park E, Kim YT, Kim S, Nam EJ, Cho NH. Immunohistochemical and genetic characteristics of hpv-associated endocervical carcinoma with an invasive stratified mucin-producing carcinoma (Ismc) component. Modern pathology: an Off J United States Can Acad Pathology Inc. (2021) 34:1738–49. doi: 10.1038/s41379-021-00829-3
50. Horn LC, Handzel R, Borte G, Siebolts U, Haak A, Brambs CE. Invasive stratified mucin-producing carcinoma (I-smile) of the uterine cervix: report of a case series and review of the literature indicating poor prognostic subtype of cervical adenocarcinoma. J Cancer Res Clin Oncol. (2019) 145:2573–82. doi: 10.1007/s00432-019-02991-3
51. Stolnicu S, Boros M, Segura S, Horn LC, Parra-Herran C, Oliva E, et al. Invasive stratified mucinous carcinoma (Ismc) of the cervix often presents with high-risk features that are determinants of poor outcome: an international multicenter study. Am J Surg Pathol. (2020) 44:1374–80. doi: 10.1097/pas.0000000000001485
52. Hodgson A, Olkhov-Mitsel E, Howitt BE, Nucci MR, Parra-Herran C. International endocervical adenocarcinoma criteria and classification (Iecc): correlation with adverse clinicopathological features and patient outcome. J Clin Pathol. (2019) 72:347–53. doi: 10.1136/jclinpath-2018-205632
53. Wang M, Yuan B, Zhou ZH, Han WW. Clinicopathological characteristics and prognostic factors of cervical adenocarcinoma. Sci Rep. (2021) 11:7506. doi: 10.1038/s41598-021-86786-y
54. Gadducci A, Guerrieri ME, Cosio S. Adenocarcinoma of the uterine cervix: pathologic features, treatment options, clinical outcome and prognostic variables. Crit Rev oncology/hematology. (2019) 135:103–14. doi: 10.1016/j.critrevonc.2019.01.006
55. Lei R. Invasive stratified mucin-producing carcinoma: A clinicopathological analysis of three cases. Cancer Biol Ther. (2019) 20:1403–7. doi: 10.1080/15384047.2019.1647054
56. Onishi J, Sato Y, Sawaguchi A, Yamashita A, Maekawa K, Sameshima H, et al. Stratified mucin-producing intraepithelial lesion with invasive carcinoma: 12 cases with immunohistochemical and ultrastructural findings. Hum Pathol. (2016) 55:174–81. doi: 10.1016/j.humpath.2016.05.007
57. Dahlgren L, Erlandsson F, Lindquist D, Silfverswärd C, Hellström AC, Dalianis T. Differences in human papillomavirus type may influence clinical outcome in early stage cervical cancer. Anticancer Res. (2006) 26:829–32.
58. Hang D, Jia M, Ma H, Zhou J, Feng X, Lyu Z, et al. Independent prognostic role of human papillomavirus genotype in cervical cancer. BMC Infect Dis. (2017) 17:391. doi: 10.1186/s12879-017-2465-y
59. Lai CH, Chou HH, Chang CJ, Wang CC, Hsueh S, Huang YT, et al. Clinical implications of human papillomavirus genotype in cervical adeno-adenosquamous carcinoma. Eur J Cancer. (2013) 49:633–41. doi: 10.1016/j.ejca.2012.09.008
60. Cuschieri K, Brewster DH, Graham C, Nicoll S, Williams AR, Murray GI, et al. Influence of hpv type on prognosis in patients diagnosed with invasive cervical cancer. Int J Cancer. (2014) 135:2721–6. doi: 10.1002/ijc.28902
61. Onuki M, Matsumoto K, Tenjimbayashi Y, Tasaka N, Akiyama A, Sakurai M, et al. Human papillomavirus genotype and prognosis of cervical cancer: favorable survival of patients with hpv16-positive tumors. Papillomavirus Res (Amsterdam Netherlands). (2018) 6:41–5. doi: 10.1016/j.pvr.2018.10.005
62. de Cremoux P, de la Rochefordière A, Savignoni A, Kirova Y, Alran S, Fourchotte V, et al. Different outcome of invasive cervical cancer associated with high-risk versus intermediate-risk hpv genotype. Int J Cancer. (2009) 124:778–82. doi: 10.1002/ijc.24075
63. Lai CH, Chang CJ, Huang HJ, Hsueh S, Chao A, Yang JE, et al. Role of human papillomavirus genotype in prognosis of early-stage cervical cancer undergoing primary surgery. J Clin oncology: Off J Am Soc Clin Oncol. (2007) 25:3628–34. doi: 10.1200/jco.2007.11.2995
64. Pilch H, Günzel S, Schäffer U, Tanner B, Brockerhoff P, Maeurer M, et al. Human papillomavirus (Hpv) DNA in primary cervical cancer and in cancer free pelvic lymph nodes–correlation with clinico-pathological parameters and prognostic significance. Zentralblatt fur Gynakologie. (2001) 123:91–101. doi: 10.1055/s-2001-12411
65. Kiseleva VI, Mkrtchyan LS, Ivanov SA, Lyubina LV, Bezyaeva GP, Panarina LV, et al. The presence of human papillomavirus DNA integration is associated with poor clinical results in patients with third-stage cervical cancer. Bull Exp Biol Med. (2019) 168:87–91. doi: 10.1007/s10517-019-04654-2
66. Pilch H, Günzel S, Schäffer U, Tanner B, Brockerhoff P, Maeurer M, et al. The presence of hpv DNA in cervical cancer: correlation with clinico-pathologic parameters and prognostic significance: 10 years experience at the department of obstetrics and gynecology of the mainz university. Int J gynecological cancer: Off J Int Gynecological Cancer Soc. (2001) 11:39–48. doi: 10.1046/j.1525-1438.2001.011001039.x
67. Im SS, Wilczynski SP, Burger RA, Monk BJ. Early stage cervical cancers containing human papillomavirus type 18 DNA have more nodal metastasis and deeper stromal invasion. Clin Cancer research: an Off J Am Assoc Cancer Res. (2003) 9:4145–50.
68. Rose BR, Thompson CH, Simpson JM, Jarrett CS, Elliott PM, Tattersall MH, et al. Human papillomavirus deoxyribonucleic acid as a prognostic indicator in early-stage cervical cancer: A possible role for type 18. Am J obstetrics gynecology. (1995) 173:1461–8. doi: 10.1016/0002-9378(95)90633-9
69. Nakagawa S, Yoshikawa H, Onda T, Kawana T, Iwamoto A, Taketani Y. Type of human papillomavirus is related to clinical features of cervical carcinoma. Cancer. (1996) 78:1935–41. doi: 10.1002/(SICI)1097-0142(19961101)78:9<1935::AID-CNCR14>3.0.CO;2-Z
70. Burger RA, Monk BJ, Kurosaki T, Anton-Culver H, Vasilev SA, Berman ML, et al. Human papillomavirus type 18: association with poor prognosis in early stage cervical cancer. J Natl Cancer Institute. (1996) 88:1361–8. doi: 10.1093/jnci/88.19.1361
71. Lombard I, Vincent-Salomon A, Validire P, Zafrani B, de la Rochefordière A, Clough K, et al. Human papillomavirus genotype as a major determinant of the course of cervical cancer. J Clin oncology: Off J Am Soc Clin Oncol. (1998) 16:2613–9. doi: 10.1200/jco.1998.16.8.2613
72. Schwartz SM, Daling JR, Shera KA, Madeleine MM, McKnight B, Galloway DA, et al. Human papillomavirus and prognosis of invasive cervical cancer: A population-based study. J Clin oncology: Off J Am Soc Clin Oncol. (2001) 19:1906–15. doi: 10.1200/jco.2001.19.7.1906
73. van Muyden RC, ter Harmsel BW, Smedts FM, Hermans J, Kuijpers JC, Raikhlin NT, et al. Detection and typing of human papillomavirus in cervical carcinomas in Russian women: A prognostic study. Cancer. (1999) 85:2011–6. doi: 10.1002/(SICI)1097-0142(19990501)85:9<2011::AID-CNCR19>3.0.CO;2-N
74. Silins I, Avall-Lundqvist E, Tadesse A, Jansen KU, Stendahl U, Lenner P, et al. Evaluation of antibodies to human papillomavirus as prognostic markers in cervical cancer patients. Gynecologic Oncol. (2002) 85:333–8. doi: 10.1006/gyno.2002.6628
75. van Bommel PF, van den Brule AJ, Helmerhorst TJ, Gallee MP, Gaarenstroom KN, Walboomers JM, et al. Hpv DNA presence and hpv genotypes as prognostic factors in low-stage squamous cell cervical cancer. Gynecologic Oncol. (1993) 48:333–7. doi: 10.1006/gyno.1993.1058
76. Füle T, Csapó Z, Máthé M, Tátrai P, László V, Papp Z, et al. Prognostic significance of high-risk hpv status in advanced cervical cancers and pelvic lymph nodes. Gynecologic Oncol. (2006) 100:570–8. doi: 10.1016/j.ygyno.2005.09.019
77. Heeren AM, Punt S, Bleeker MC, Gaarenstroom KN, van der Velden J, Kenter GG, et al. Prognostic effect of different pd-L1 expression patterns in squamous cell carcinoma and adenocarcinoma of the cervix. Modern pathology: an Off J United States Can Acad Pathology Inc. (2016) 29:753–63. doi: 10.1038/modpathol.2016.64
78. Karpathiou G, Chauleur C, Mobarki M, Peoc'h M. The immune checkpoints ctla-4 and pd-L1 in carcinomas of the uterine cervix. Pathol Res Pract. (2020) 216:152782. doi: 10.1016/j.prp.2019.152782
79. Zhou F, Chen H, Li M, Strickland AL, Zheng W, Zhang X. The prognostic values of hpv genotypes and tumor pd-L1 expression in patients with hpv-associated endocervical adenocarcinoma. Am J Surg Pathol. (2022) 46:300–8. doi: 10.1097/pas.0000000000001800
80. Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, et al. Colocalization of inflammatory response with B7-H1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Trans Med. (2012) 4:127ra37. doi: 10.1126/scitranslmed.3003689
81. Teng MW, Ngiow SF, Ribas A, Smyth MJ. Classifying cancers based on T-cell infiltration and pd-L1. Cancer Res. (2015) 75:2139–45. doi: 10.1158/0008-5472.Can-15-0255
82. Rivera-Colon G, Chen H, Molberg K, Niu S, Strickland AL, Castrillon DH, et al. Pd-L1 expression in endocervical adenocarcinoma: correlation with patterns of tumor invasion, cd8+ Tumor-infiltrating lymphocytes, and clinical outcomes. Am J Surg Pathol. (2021) 45:742–52. doi: 10.1097/pas.0000000000001633
83. Rischin D, Gil-Martin M, González-Martin A, Braña I, Hou JY, Cho D, et al. Pd-1 blockade in recurrent or metastatic cervical cancer: data from cemiplimab phase I expansion cohorts and characterization of pd-L1 expression in cervical cancer. Gynecologic Oncol. (2020) 159:322–8. doi: 10.1016/j.ygyno.2020.08.026
84. Lim JKM, Delaidelli A, Minaker SW, Zhang HF, Colovic M, Yang H, et al. Cystine/glutamate antiporter xct (Slc7a11) facilitates oncogenic ras transformation by preserving intracellular redox balance. Proc Natl Acad Sci United States America. (2019) 116:9433–42. doi: 10.1073/pnas.1821323116
85. Lin W, Wang C, Liu G, Bi C, Wang X, Zhou Q, et al. Slc7a11/xct in cancer: biological functions and therapeutic implications. Am J Cancer Res. (2020) 10:3106–26.
86. Sato H, Tamba M, Kuriyama-Matsumura K, Okuno S, Bannai S. Molecular cloning and expression of human xct, the light chain of amino acid transport system xc. Antioxidants Redox Signaling. (2000) 2:665–71. doi: 10.1089/ars.2000.2.4-665
87. Zhang HF, Klein Geltink RI, Parker SJ, Sorensen PH. Transsulfuration, minor player or crucial for cysteine homeostasis in cancer. Trends Cell Biol. (2022) 32:800–14. doi: 10.1016/j.tcb.2022.02.009
88. Koppula P, Zhang Y, Shi J, Li W, Gan B. The glutamate/cystine antiporter slc7a11/xct enhances cancer cell dependency on glucose by exporting glutamate. J Biol Chem. (2017) 292:14240–9. doi: 10.1074/jbc.M117.798405
89. Li S, Lu Z, Sun R, Guo S, Gao F, Cao B, et al. The role of slc7a11 in cancer: friend or foe? Cancers. (2022) 14. doi: 10.3390/cancers14133059
90. Liu J, Xia X, Huang P. Xct: A critical molecule that links cancer metabolism to redox signaling. Mol therapy: J Am Soc Gene Ther. (2020) 28:2358–66. doi: 10.1016/j.ymthe.2020.08.021
91. He Q, Liu M, Huang W, Chen X, Zhang B, Zhang T, et al. Il-1β-Induced Elevation of Solute Carrier Family 7 Member 11 Promotes Hepatocellular Carcinoma Metastasis through up-Regulating Programmed Death Ligand 1 and Colony-Stimulating Factor 1. Hepatol (Baltimore Md). (2021) 74:3174–93. doi: 10.1002/hep.32062
92. Montero JC, Calvo-Jiménez E, Del Carmen S, Abad M, Ocaña A, Pandiella A. Surfaceome analyses uncover cd98hc as an antibody drug-conjugate target in triple negative breast cancer. J Exp Clin Cancer research: CR. (2022) 41:106. doi: 10.1186/s13046-022-02330-4
93. Zhao L, Zhou X, Xie F, Zhang L, Yan H, Huang J, et al. Ferroptosis in cancer and cancer immunotherapy. Cancer Commun (Lond). (2022) 42:88–116. doi: 10.1002/cac2.12250
Keywords: invasive stratified mucus of the cervix produces carcinoma, PD-L1, SLC7A11, SLC3A2, immune checkpoint inhibitors, ferroptosis
Citation: Lu C, Zhu W, Han X, Du X, Zhang H, Yao Q, Liu T and Zhang C (2024) Clinicopathological characteristics of invasive stratified mucinous carcinoma of the cervix and the expression and clinical significance of SLC7A11, SLC3A2 and PD-L1. Front. Oncol. 14:1492498. doi: 10.3389/fonc.2024.1492498
Received: 07 September 2024; Accepted: 11 December 2024;
Published: 30 December 2024.
Edited by:
Wenzheng Guo, University of Kentucky, United StatesReviewed by:
Xinzhao Wang, University of Kentucky, United StatesMiaolong He, Tsinghua University, China
Copyright © 2024 Lu, Zhu, Han, Du, Zhang, Yao, Liu and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hui Zhang, d2lsbGlhbXpoQDEyNi5jb20=; Qin Yao, ZHJfeWFvcWluQHFkdS5lZHUuY24=
 Changyu Lu
Changyu Lu Wei Zhu1
Wei Zhu1 Hui Zhang
Hui Zhang Qin Yao
Qin Yao Ting Liu
Ting Liu Ce Zhang
Ce Zhang