- 1Division of Head & Neck Tumor Multimodality Treatment, Cancer Center, West China Hospital, Sichuan University, Chengdu, China
- 2Department of Targeting Therapy & Immunology, Cancer Center, West China Hospital, Sichuan University, Chengdu, China
- 3Division of Thoracic Tumor Multimodality Treatment, Cancer Center, West China Hospital, Sichuan University, Chengdu, China
Objectives: To explore the efficacy of epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) on lung adenosquamous cell carcinoma (ASC) with EGFR mutation.
Methods: Efficacy of EGFR-TKIs in the treatment of advanced or recurrent lung ASC with EGFR mutations was assessed retrospectively in 44 patients. Pooled analysis of 74 patients using EGFR-TKIs, including 30 patients selected from 11 publications, was conducted.
Results: In our retrospective research, patients treated with EGFR-TKI in ASC with EGFR mutations had objective response rate (ORR) of 54.5%, disease control rate (DCR) of 79.5%, median progression free survival (mPFS) of 8.8 months, and median overall survival (mOS) of 19.43 months, respectively. A pooled analysis reveals ORR, DCR, mPFS, and mOS are, respectively, 63.4%, 85.9%, 10.00 months, and 21.37 months for ASC patients. In patients with deletions in exon 19 and exon 21 L858R mutations, mPFS (11.0 versus 10.0 months, P=0.771) and mOS (23.67 versus 20.33 months, P=0.973) were similar. Erlotinib or gefitinib-treated patients had an overall survival trend that was superior to that of icotinib-treated patients.
Conclusions: ASC harboring EGFR mutations can be treated with EGFR-TKI in a similar manner to Adenocarcinoma (ADC) harboring EGFR mutations. There is still a need for further investigation to identify the separate roles of ASC’s two components in treating EGFR.
Introduction
Lung adenosquamous cell carcinoma (ASC)has a low incidence of about 0.4%-4%, making it one of the rarest types of lung cancer (1). ASC is characterized by the presence of both glandular and squamous components, each constituting at least 10% of the tumor (2). This dual histology contributes to the aggressive nature of ASC and poses significant therapeutic challenges. The prognosis for ASC patients is generally poorer compared to those with pure adenocarcinoma or squamous cell carcinoma, reflecting its more aggressive biological behavior and limited treatment options. Although immunotherapy improves survival of ASC patient, compared to squamous cell carcinoma and adenocarcinoma, ASC patients have a worse prognosis (3–5).
In patients with non-small cell lung cancer (NSCLC) harboring epidermal growth factor receptor (EGFR) mutations, treatment with EGFR tyrosine kinase inhibitors (EGFR-TKIs) are now norm. Third-generation EGFR-TKIs, such as osimertinib, have become the current standard of care, particularly for patients with EGFR T790M resistance mutations (6, 7). EGFR mutations are predominantly found in adenocarcinoma, but they can also be detected in 54.8% of ASC patients (8). Despite the proven efficacy of EGFR-TKIs in treating EGFR-mutant NSCLC, the evidence for their effectiveness in ASC is limited due to the rarity of the condition and the consequent scarcity of comprehensive studies (9). Current treatment guidelines and clinical trials predominantly focus on adenocarcinoma, leaving a gap in tailored therapeutic strategies for ASC patients with EGFR mutations.
In this context, our study aims to explore the efficacy of EGFR-TKIs in patients with EGFR-mutant ASC through a retrospective analysis and pooled data from published studies. We seek to provide insights into the clinical outcomes and potential benefits of EGFR-TKIs therapy in this unique patient population, thereby addressing a critical gap in the management of ASC.
Patients and methods
Patients
Between January 2009 and April 2022, we collected clinical data on ASC patients treated with EGFR-TKI at West China Hospital. All patients underwent bronchofiberscope or percutaneous lung biopsies, followed by immunohistochemistry (IHC) for pathological confirmation. These retrospective analyses were carried out with informed consent from each patient.
We searched PUBMED for all publications describing the use of EGFR-TKI in advanced or recurrent EGFR mutant ASC patients for further research into its efficacy. There were three subject headings used during the search period of 2005 to 2022: lung cancer, mutation, and EGFR. Journals and publications were not limited by the strategy, but abstracts of conferences were not accepted. An evaluation of EGFR-TKIs used to treat advanced or recurrent ASC patients harboring EGFR mutations was included in this study. The choice was limited to researches published in the English journal. EGFR-TKI therapy was offered to patients who met all three criteria: (1) advanced or recurrent ASC, (2) EGFR mutation, and (3) acceptance of EGFR-TKI therapy (erlotinib 150mg/day, gefitinib 250mg/day, icotinib 125mg tid or dacomitinib 45mg/day). Data, such as EGFR mutation type, EGFR-TKI line, and treatment with EGFR-TKI, were collected as baseline factors. The authors were asked for data that wasn’t included in the article.
Test method for EGFR mutation
In the retrospective data, tissues that were embalmed or freshly harvested were used to extract DNA. The mutations in EGFR were identified using a quantitative PCR analysis using the Amplification Refractory Mutation System.A EGFR mutation was detected using the protocol represented in each study, according to the published data.
Clinical assessments
The Response Evaluation Criteria in Solid Tumors were used to evaluate the efficacy of the EGFR-TKI targeted therapy. There were four types of responses: progressive disease (PD), stable disease (SD), partial response (PR), and complete response (CR). The objective response rate (ORR) is determined by dividing the percentage of patients who were CR or PR by all patients. CR, PR, and SD patients were divided by total patients to determine the disease control rate (DCR). A prognosis of progression free survival (PFS) was calculated from the beginning of treatment to the onset of PD. We also calculated overall survival (OS) from the moment treatment began until death. It was on July 22, 2022, that the last follow-up visit was carried out. In statistical analysis, patients who did not progress or were alive were censored on July 22, 2022.
Statistical methods
Qualitative variables were illustrated as the way of absolute and percentage amounts, while continuous variables were illustrated as medians with ranges. In order to conduct the survival analysis, Kaplan-Meier methods were used. An univariate analysis of log-rank tests was performed in order to determine which prognostic factors affect PFS and OS. A multivariate analysis was conducted by using Cox regression. The significance of P values is determined by using 0.05. Analyses were conducted using SPSS version 22.0.
Results
Patient characteristics
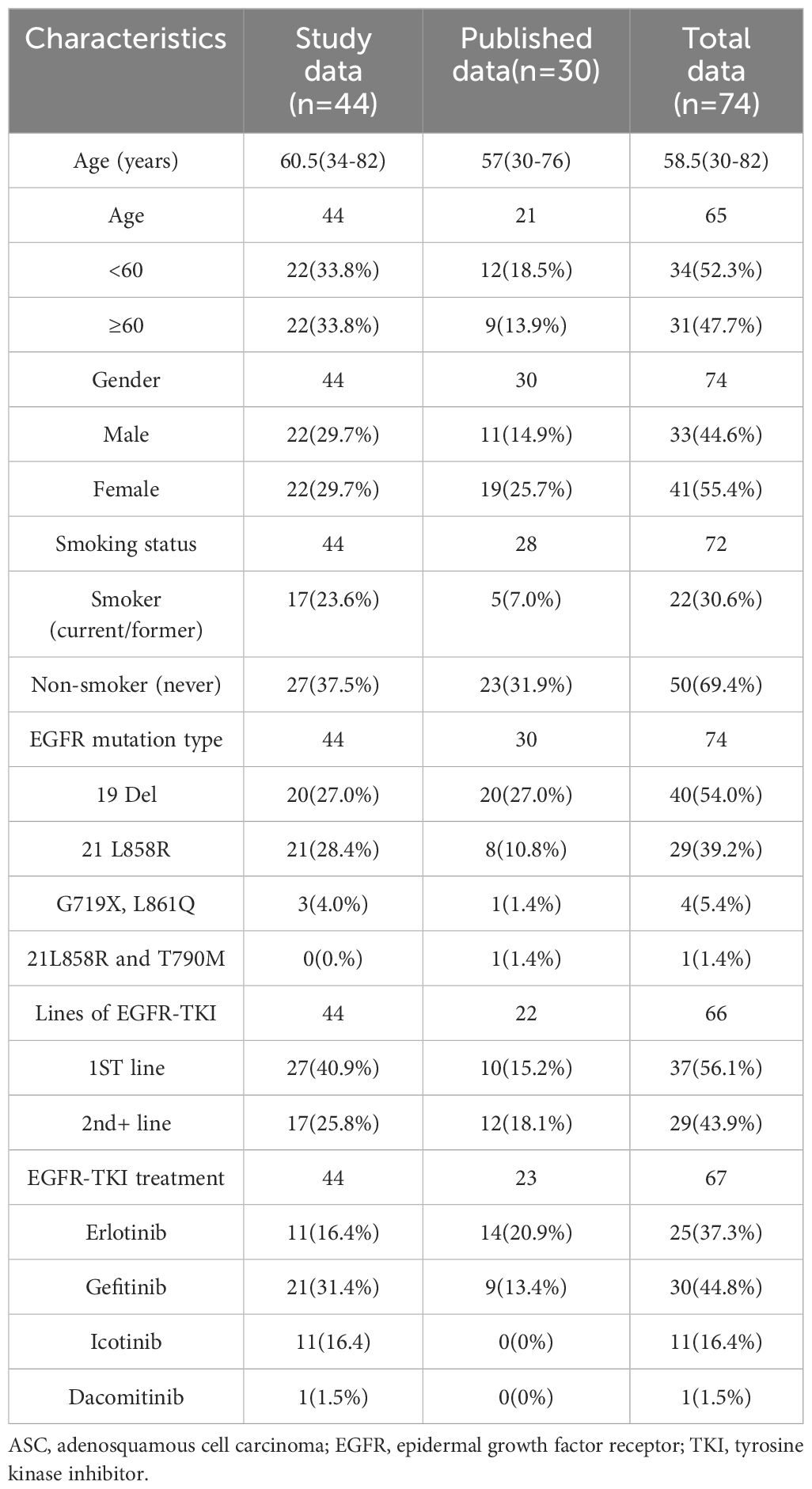
EGFR-TKI treatment was administered to 44 ASC patients with EGFR mutations at the two cancer centers for the purposes of assessing efficacy. Of the 44 ASC patients, there were 22 females and 22 males. Age range was 34-82 years (median 60.5 years). There were 17 patients with a history of smoking. Among the patients, 20 had a mutation in exon 19 (19-DEL), 21 had a mutation in exon 21 (L858R), while 3 had a rare sensitive mutation (G719X, L861Q). As a first-lines treatment, 27 patients were treated with EGFR-TKI, and 17 patients were treated in a second or more line of treatment. There were 11 patients treated with erlotinib, 21 patients treated with gefitinib, 11 patients treated with icotinib, and 1 patient treated with dacomitinib (Table 1).
Efficacy of EGFR-TKI
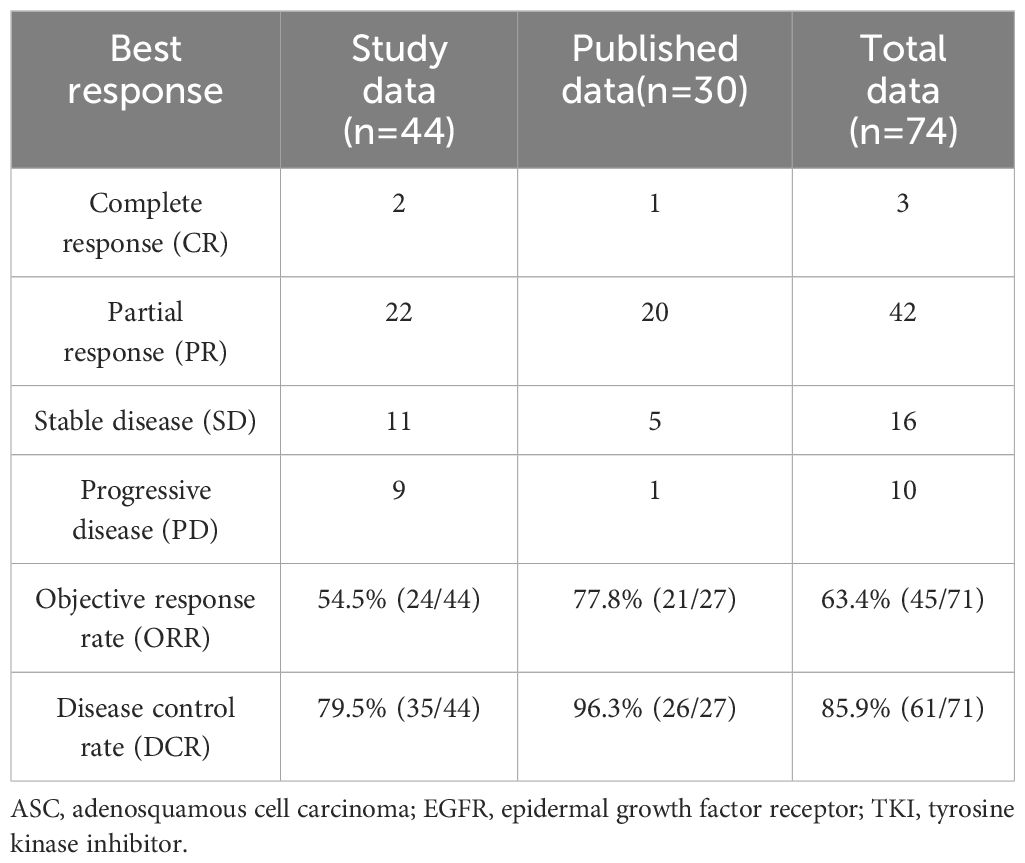
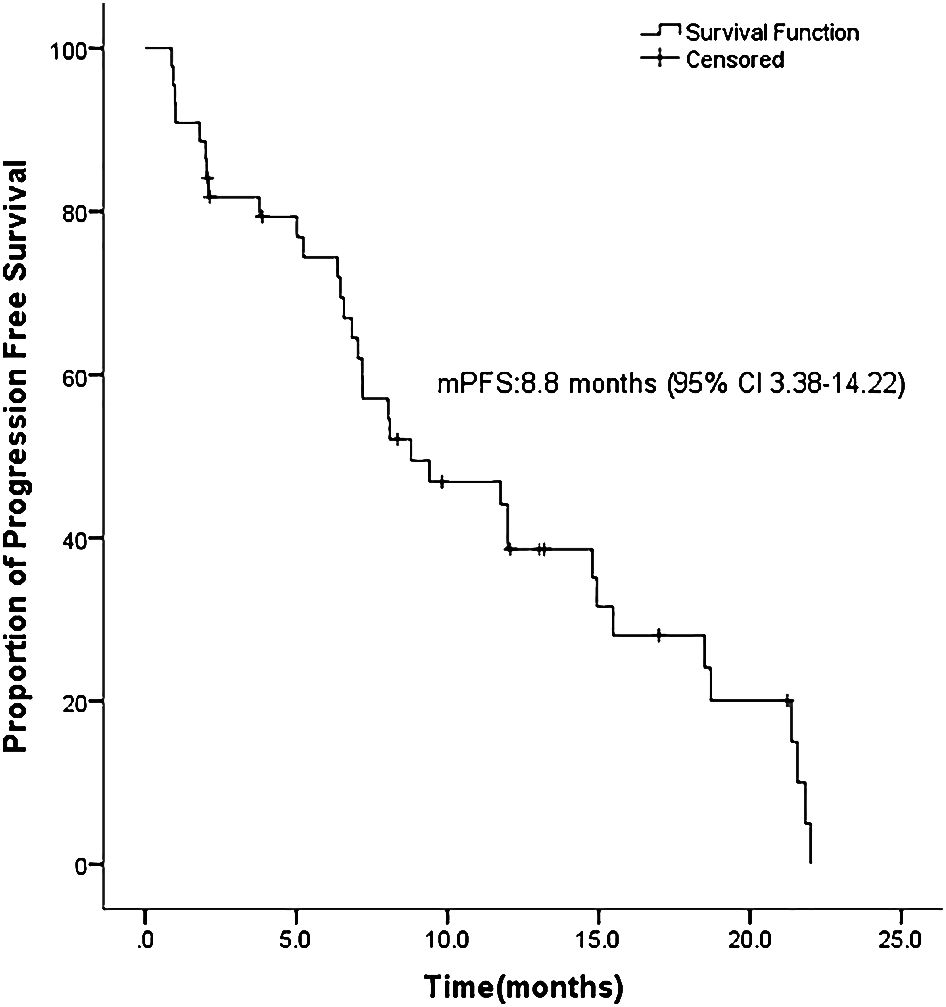
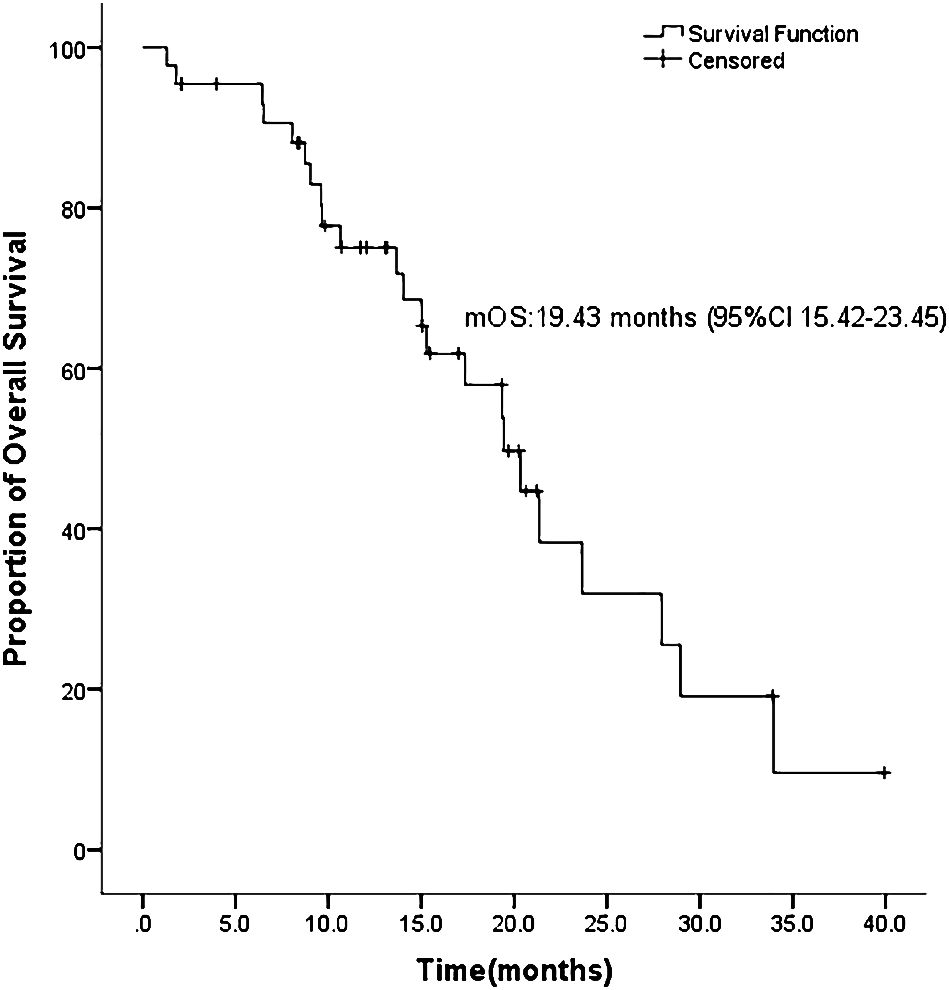
ASCs with EGFR mutations responded to EGFR-TKI with 2 CRs, 22 PRs, 11 SDs, and 9 PDs. The ORR was 54.5% and the DCR was 79.5% for the 44 patients (Table 2). Ten patients had not yet progressed, while 21 patients were still alive on July 22, 2022. Figure 1 shows the mPFS was 8.8 months (95% CI 3.38-14.22), and Figure 2 shows the mOS of 19.43 months (95% CI 15.42-23.45).
Pooled analysis
A total of 30 patients who met the inclusion criteria from eleven research studies were included in this study (10–20). The 11 researches consisted of 8 retrospective studies and 3 case reports. A total of eight of these researches were conducted in East Asian countries. Thirteen patients were from western countries. Definitive data of age, gender, smoking status, EGFR mutation type, lines of EGFR-TKI and EGFR-TKI treatment could be extracted in 21 (70.0%), 30 (100.0%), 28 (93.3%), 30 (100.0%), 22 (73.3%) and 23 (76.7%) of the 30 patients, respectively (Tables 1, 3, 4).
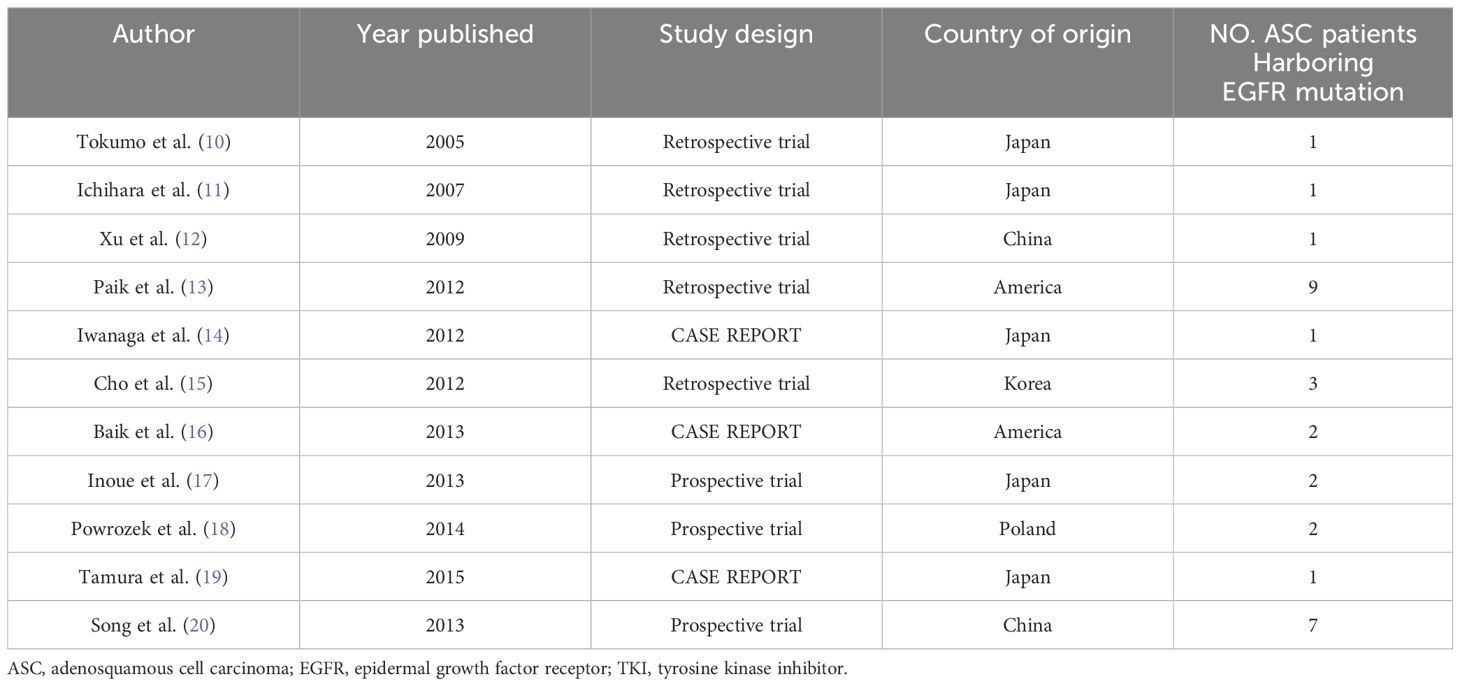
Table 3 The 11 published reports which we could extract the data of recurrent or advanced ASC patients who had EGFR mutation and were treated with EGFR-TKI.
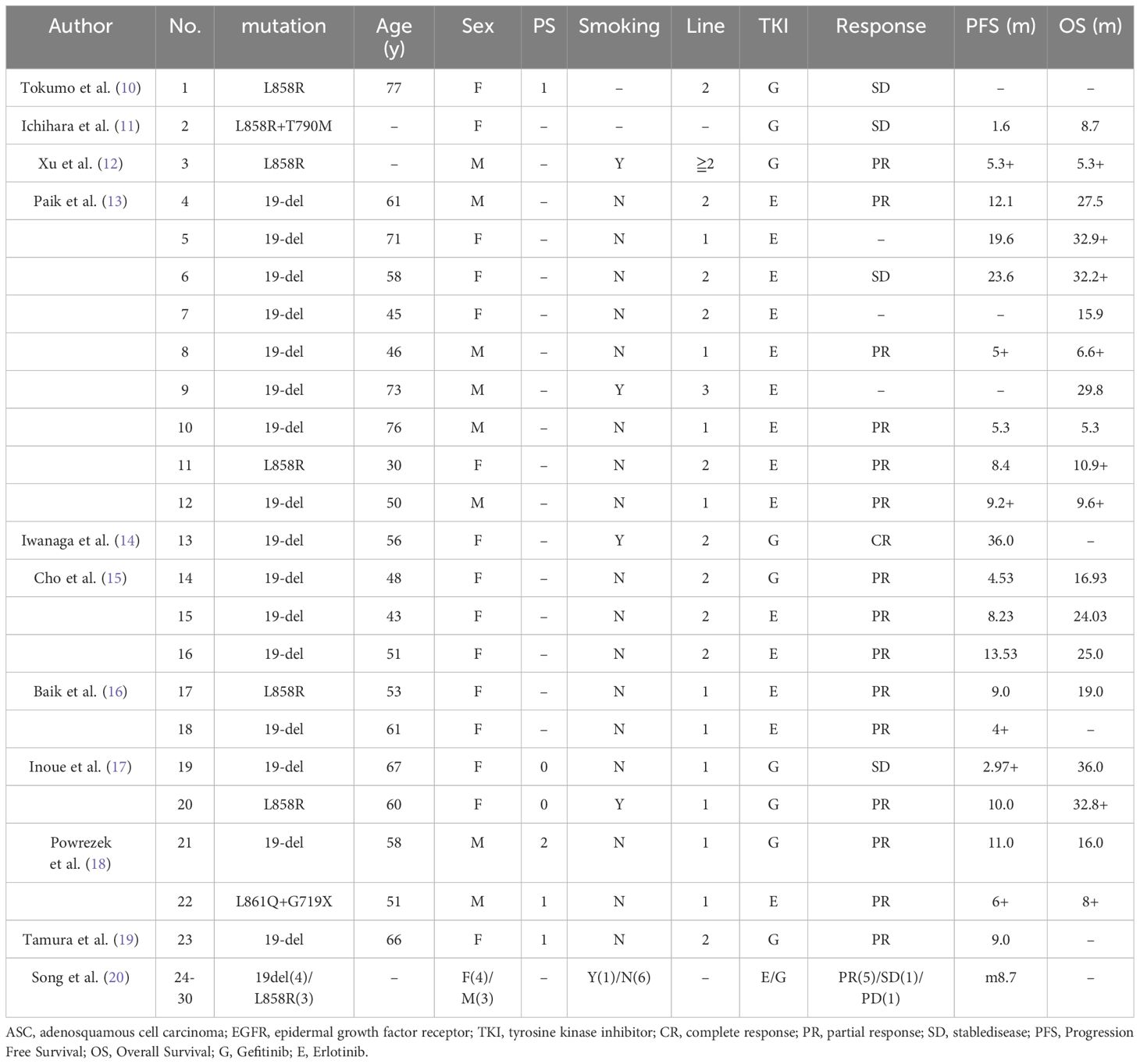
Table 4 Individual patient data of the ASC patients with EGFR mutations extracted from the 11 studies that evaluated the efficacy of EGFR-TKI for ASC patients with EGFR mutations.
Finally, we pooled data from 74 patients. Ages ranged from 30 to 82 years (median 58.5). Gender and smoking history were: male (33/74, 44.6%), female (41/74, 55.4%); never-smoker (50/72, 69.4%), smoker (22/72, 30.6%); There were 40 patients (40/74, 54.0%) with exon 19 deletion, 29 patients (29/74, 39.2%) with L858R mutation and 4 patients (4/74, 5.4%) with rare sensitive mutation (G719X, L861Q). One patient (1.4%) who had both a resistant mutation (T790M) and sensitive mutation (L858R) was excluded in the analysis of PFS and OS. Twenty-five (25/67, 37.3%) patients received erlotinib, 30 patients (30/67, 44.8%) received gefitinib, 11 patients (11/67, 16.4%) received icotinib and 1 patients (1/67, 1.5%) received dacomitinib. In 37 cases (37/66, 56.1%), EGFR-TKI was used as the first line of treatment while in 29 cases (29/66, 43.9%), second or more lines of treatment with EGFR-TKI were used. (Table 1).
There are 27 patients whose tumor responses were identified from published research. In total, 71 patients were evaluated for response. There were three patients with CR, 42 patients with PR, 16 patients with SD, and 10 patients with PD. It had an ORR of 63.4% (45/71) and DCR of 85.9% (61/71) (Table 2).
19 patients with PFS were identified in published research. In total, 63 patients were analyzed for PFS. All patients had a mPFS of 10.00 months (95% CI 6.73-13.27). Exon 19 deletion patients had a mPFS of 11.00 months (95% CI 6.70-15.30), while exon 21 L858R mutation patients had a mPFS of 10.00 months (95% CI 5.89-14.11) (P=0.771). Compared to rare sensitive mutations (G719X, L861Q) patients, exon 19 deletion patients or exon 21 L858R mutation patients had a longer mPFS (11.00 months vs. 2.10 months, P=0.005; 10.00 months vs. 2.10 months, P=0.019). Univariate analysis did not show significant correlations between the data sources, age, gender, smoking status, EGFR-TKI lines, and EGFR-TKI treatment and PFS. Multivariate analysis revealed no significant correlation between clinical features and PFS (Table 5).
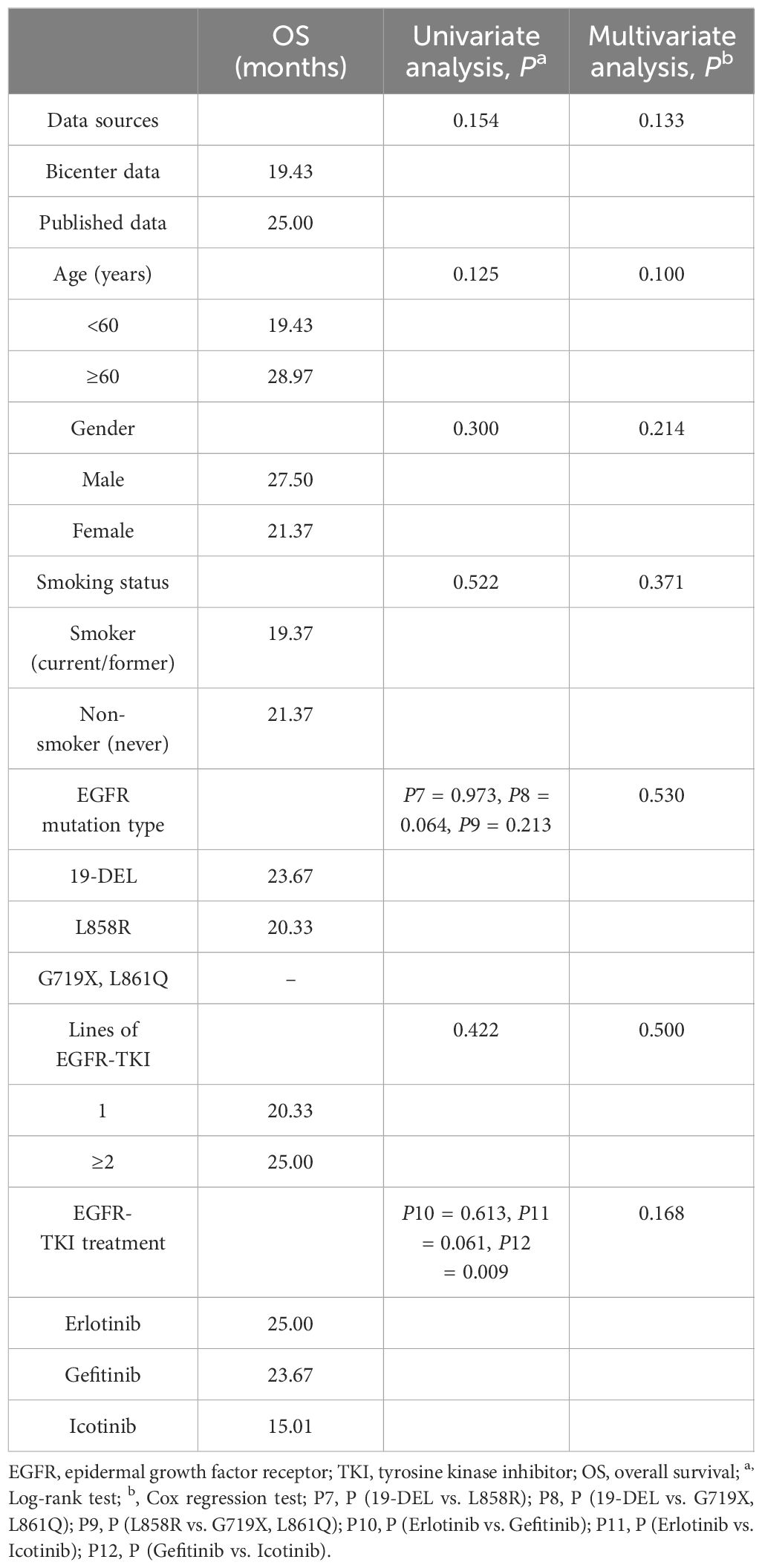
The data of OS was extracted in 18 patients from the published researches. The pooled analysis of OS included 62 patients. The mOS was 21.37 months (95% CI 16.01-26.73). Exon 19 deletion patients had a mOS of 23.67 months, while exon 21 L858R mutation patients had a mOS of 20.33 months (P=0.973). In univariate analysis, erlotinib treatment led to a longer OS compared with icotinib treatment (25.00 months vs. 15.01 months, P=0.061); In univariate analysis, a mOS of 23.67 months was seen in patients treated with gefitinib compared with 15.01 months in patients treated with icotinib (P=0.009); Univariate analyses showed no significant correlation between the data sources, age, gender, smoking status, and lines of EGFR-TKIs and OS. In multivariate analysis, no clinical features were found to be correlated significantly with OS (Table 6).
Discussion
Literature studies concerning EGFR-TKI sensitivity in ASC harboring EGFR mutation are limited due to the low incidence of ASC in lung cancer. As a result, there is not enough evidence to support the efficacy of EGFR-TKI in the treatment of ASC. In our pooled analysis, there was an ORR of 63.4% and DCR of 85.9% in ASC patients treated with EGFR-TKI, and a mPFS of 10.00 months and a mOS of 21.37 months in these patients. Hence, ASC containing mutant EGFR are effectively treated with EGFR-TKI. Meanwhile, EGFR mutation can be detected in 54.8% of ASC patients which demonstrated that the mutation rate is parallel to ADC (8, 21). As EGFR mutations are highly prevalent in ASC patients and EGFR-TKIs are highly effective, we recommend routine EGFR mutation testing for all ASC patients. To our knowledge, our study represents one of the largest studies of EGFR-TKI efficacy in lung ASC patients harboring mutations in EGFR. We believe this data deserves clinical reference.
ADC has been successfully treated with EGFR-TKI in previous clinical studies, with ORRs of 70-85% and mPFS of 8-13 months (7, 22). Previous research has indicated that ASC patients with EGFR mutations achieve mPFS of 9.3 months when treated with first-generation EGFR-TKI (9). As a result of our study, lung ASC had an ORR of 63.4% and a median PFS of 10.00 months. Our study shows that lung ASC with EGFR mutations respond effectively to EGFR-TKI treatment, albeit with a slightly lower efficacy compared to pure adenocarcinomas. The distinct biological behavior of ASC, which includes both squamous and glandular components, might contribute to the differences in treatment outcomes (3). The heterogeneity within the tumor may impact the response to EGFR-TKI, as adenocarcinoma and squamous components may respond differently to treatment. Besides, the variability in the molecular profile of ASC tumors, as compared to pure adenocarcinomas, might also be a factor (8). This variability could influence the tumor’s response to EGFR-TKI therapy. Our study suggests a need for further research to explore the molecular mechanisms behind the differential response of ASC and pure adenocarcinomas to EGFR-TKI therapy.
According to our research, in patients with rare sensitive mutations (G719X, L861Q), the difference in PFS was statistically significant when compared to patients with deletion of exon 19 or exon 21 L858R mutations. However, because there were only 4 patients with rare sensitive mutation, the outcome needed to be further testified by more researches. Further, previous studies have shown that ADC patients with a L858R mutation in exon 21 of EGFR have significantly lower efficacy with EGFR-TKI treatment than patients in exon 19 of EGFR (23). However, patients with deletions in exon 19 and exon 21 L858R mutations had similar PFS (11.0 vs 10.0 months, P=0.771) and OS (23.67 vs. 20.33 months, P=0.973). The cause of this difference needs to further study. Our study primarily focused on the initial efficacy of EGFR-TKI in ASC patients. EGFR-TKI acquired resistance in lung ASC is gradually becoming a research hotspot (24). The progression and resistance mechanisms, including the frequency of T790M mutations, are undoubtedly crucial and future studies focusing on this aspect would indeed be valuable. Bsides, the efficacy in lung ASC of third generation TKI such as osimertinib and ceritinib still needs further study (25, 26). Immunotherapy has shown promising prospects in the treatment of lung ASC (5).
In one published research, 55 ASC patients were demonstrated dual differentiation with varying proportions of ADC and SCC by using the microdissection (27). There is a pity that pathology was unable to determine which of 44 patients carried squamous cell carcinomatous and adenocarcinomatous components. Moreover, some researches discovered that the identical EGFR mutation patterns in the squamous cell carcinomatous and the adenocarcinomatous components in each patient, indicated the monoclonality of the two tumor components in ASC patients (28, 29). This conclusion was also testified by other researches (3). Since the identical EGFR mutation patterns occurred in the squamous cell carcinomatous and the adenocarcinomatous components of ASC, It may be the proportion of two tumor components in EGFR mutant ASC patients that determines the efficacy of EGFR-TKI in EGFR mutant ASC patients. The predominance of one component over the other could potentially affect the treatment outcomes. In addition, the therapeutic advantages on adenocarcinoma components of TKI may generate the withering of the adenocarcinomatous components of ASC, while the squamous cell carcinomatous of ASC gain a quantitative advantage (30). Researchers at our cancer center are investigating how the ratio of these two tumor components and EGFR-TKI efficacy are related.
In NSCLC, especially in metastatic disease, small biopsy samples can make it difficult to accurately differentiate between squamous cell carcinoma and ASC (1). This distinction is crucial as it impacts treatment decisions. Molecular testing, including EGFR mutation analysis, can play a critical role in identifying patients who might benefit from targeted therapies (31). This is particularly relevant in cases where histological classification is uncertain. Given the histological overlap between squamous tumors and ASC, molecular testing provides a more precise approach to identify the tumor’s characteristics, thus guiding appropriate treatment (3). The American Society of Clinical Oncology (ASCO) emphasizes the need for comprehensive molecular profiling in NSCLC. By incorporating molecular testing, clinicians can better tailor treatment strategies to individual patient needs, especially for those with rare or atypical NSCLC subtypes like ASC.
It is necessary to illustrate the limitations of this study. Among the selected published studies, inclusion criteria and test methods for EGFR mutations were different, and clinical traits were not completely described. Moreover, the retrospective nature was another limitation of this research. The low incidence of ASC in lung cancer, however, makes our research quite significant as well.
In conclusion, this study which involved all available data, including data collected from our cancer centers of China and that pooled from previous studies, and identified the clinical profiles of EGFR-TKI application, suggested that EGFR-TKI was found to be an effective treatment in ASC harboring mutations in EGFR. Furthermore, the study recommends that EGFR mutation testing be conducted routinely on all lung ASC patients.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by West China hospital’s Institutional Review Board. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants’ legal guardians/next of kin.
Author contributions
XX: Conceptualization, Data curation, Writing – original draft. WD: Data curation, Writing – original draft. YZ: Supervision, Validation, Writing – original draft. YYL: Investigation, Software, Writing – original draft. MY: Formal analysis, Methodology, Resources, Writing – original draft. YML: Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Acknowledgments
We thank Dr. Baohui Han, MD, Department of Pulmonary, Shanghai Chest Hospital, the People’s Republic of China, Dr. Tomasz POWRÓZEK, MD, Pneumonology, Oncology and Allergology Department, Medical University of Lublin, Poland, Dr. Christina Baik, MD, Thoracic, Head and Neck Medical Oncology, Seattle Cancer Care Alliance, Fred Hutchinson Cancer Research Center, University of Washington, Dr. Akira Inoue, MD, the Department of Respiratory Medicine, Tohoku University Hospital, Seiryomachi, Aobaku, Sendai, Japan and Dr. Young-Chul Kim, MD, Lung cancer clinic, Pulmonary Medicine, Chonnam National University Medical School, Hwasun Hospital, Jeollanam-do, South Korea for providing the data of their patients.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Li C, Lu H. Adenosquamous carcinoma of the lung. OncoTargets Ther. (2018) 11:4829–35. doi: 10.2147/OTT
2. Nicholson AG, Tsao MS, Beasley MB, Borczuk AC, Brambilla E, Cooper WA, et al. The 2021 WHO classification of lung tumors: impact of advances since 2015. J Thorac Oncol. (2022) 17:362–87. doi: 10.1016/j.jtho.2021.11.003
3. Lin G, Li C, Li PS, Fang WZ, Xu HP, Gong YH, et al. Genomic origin and EGFR-TKI treatments of pulmonary adenosquamous carcinoma. Ann Oncol. (2020) 31:517–24. doi: 10.1016/j.annonc.2020.01.014
4. Li C, Zheng XB, Li PS, Wang HJ, Hu J, Wu L, et al. Heterogeneity of tumor immune microenvironment and real-world analysis of immunotherapy efficacy in lung adenosquamous carcinoma. Front Immunol. (2022) 13. doi: 10.3389/fimmu.2022.944812
5. Wei J, Xiang J, Hao Y, Si J, Gu X, Xu M, et al. Clinical outcomes of immune checkpoint inhibitor therapy for advanced lung adenosquamous carcinoma. J Thorac Disease. (2023) 15:260–9. doi: 10.21037/jtd
6. Wu YL, Zhou C, Liam CK, Wu G, Liu X, Zhong Z, et al. First-line erlotinib versus gemcitabine/cisplatin in patients with advanced EGFR mutation-positive non-small-cell lung cancer: analyses from the phase III, randomized, open-label, ENSURE study. Ann Oncol. (2015) 26:1883–9. doi: 10.1093/annonc/mdv270
7. Tian X, Gu T, Lee M-H, Dong Z. Challenge and countermeasures for EGFR targeted therapy in non-small cell lung cancer. Biochim Biophys Acta (BBA) - Rev Cancer. (2022) 1877:188645. doi: 10.1016/j.bbcan.2021.188645
8. Wang H, Liu J, Zhu S, Miao K, Li Z, Qi X, et al. Comprehensive analyses of genomic features and mutational signatures in adenosquamous carcinoma of the lung. Front Oncol. (2022) 12:945843. doi: 10.3389/fonc.2022.945843
9. Hu MJ, Zhang B, Xu JL, Wang SY, Zhao YM, Zhang LL, et al. Clinical outcomes of different generations of EGFR tyrosine kinase inhibitors in advanced lung adenosquamous carcinoma. Mol Diagnosis Ther. (2019) 23:773–9. doi: 10.1007/s40291-019-00425-x
10. Tokumo M, Toyooka S, Kiura K, Shigematsu H, Tomii K, Aoe M, et al. The relationship between epidermal growth factor receptor mutations and clinicopathologic features in non-small cell lung cancers. Clin Cancer Res. (2005) 11:1167–73. doi: 10.1158/1078-0432.1167.11.3
11. Ichihara S, Toyooka S, Fujiwara Y, Hotta K, Shigematsu H, Tokumo M, et al. The impact of epidermal growth factor receptor gene status on gefitinib-treated Japanese patients with non-small-cell lung cancer. Int J Cancer. (2007) 120:1239–47. doi: 10.1002/ijc.22513
12. Xu JM, Han Y, Duan HQ, Gao EM, Zhang Y, Liu XQ, et al. EGFR mutations and HER2/3 protein expression and clinical outcome in Chinese advanced non-small cell lung cancer patients treated with gefitinib. J Cancer Res Clin Oncol. (2008) 135:771–82. doi: 10.1007/s00432-008-0512-1
13. Paik PK, Varghese AM, Sima CS, Moreira AL, Ladanyi M, Kris MG, et al. Response to erlotinib in patients with EGFR mutant advanced non–small cell lung cancers with a squamous or squamous-like component. Mol Cancer Ther. (2012) 11:2535–40. doi: 10.1158/1535-7163.MCT-12-0163
14. Iwanaga K, Sueoka-Aragane N, Nakamura T, Mori D, Kimura S. The long-term survival of a patient with adenosquamous lung carcinoma harboring EGFR-activating mutations who was treated with gefitinib. Internal Med. (2012) 51:2771–4. doi: 10.2169/internalmedicine.51.7428
15. Cho S-H, Park LC, Ji JH, Park S, Hwang DW, Lee JY, et al. Efficacy of EGFR tyrosine kinase inhibitors for non-adenocarcinoma NSCLC patients with EGFR mutation. Cancer Chemother Pharmacol. (2012) 70:315–20. doi: 10.1007/s00280-012-1876-0
16. Baik CS, Pritchard CC, Eaton KD, Chow LQ. EGFR mutations in squamous cell lung cancer in never-smokers. J Thorac Oncol. (2013) 8:e6–7. doi: 10.1097/JTO.0b013e3182762d49
17. Inoue A, Kobayashi K, Maemondo M, Sugawara S, Oizumi S, Isobe H, et al. Updated overall survival results from a randomized phase III trial comparing gefitinib with carboplatin–paclitaxel for chemo-naïve non-small cell lung cancer with sensitive EGFR gene mutations (NEJ002). Ann Oncol. (2013) 24:54–9. doi: 10.1093/annonc/mds214
18. Powrózek T, Krawczyk P, Ramlau R, Sura S, Wojas-Krawczyk K, Kucharczyk T, et al. EGFRgene mutations in patients with adenosquamous lung carcinoma. Asia-Pacific J Clin Oncol. (2014) 10:340–5. doi: 10.1111/ajco.12177
19. Tamura T, Kagohashi K, Satoh H. Successful afatinib therapy after resistance to EGFR-TKI in a patient with advanced adenosquamous cell lung cancer. Oncol Res Treat. (2015) 38:316–7. doi: 10.1159/000431351
20. Song Z, Lin B, Shao L, Zhang Y. Therapeutic efficacy of gefitinib and erlotinib in patients with advanced lung adenosquamous carcinoma. J Chin Med Assoc. (2013) 76:481–5. doi: 10.1016/j.jcma.2013.05.007
21. Wang T, Zhou J, Wang Y, Zheng Q, Lin Z, Li G, et al. Clinicopathological characteristics and prognosis of resectable lung adenosquamous carcinoma: a population-based study of the SEER database. Japanese J Clin Oncol. (2022). 52:1456. doi: 10.1093/jjco/hyac096
22. Nishihara S, Yamaoka T, Ishikawa F, Ohmori T, Ando K, Kusumoto S, et al. Diverse mechanisms of resistance against osimertinib, a third-generation EGFR-TKI, in lung adenocarcinoma cells with an EGFR-activating mutation. Cells. (2022) 11. doi: 10.3390/cells11142201
23. Yang JC-H, Wu Y-L, Schuler M, Sebastian M, Popat S, Yamamoto N, et al. Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol. (2015) 16:141–51. doi: 10.1016/S1470-2045(14)71173-8
24. Wu YH, Zhang K, Guan JX, Wu WB, Zhang J, Chen HG. Treatment with anlotinib after chemotherapy and EGFR-TKI resistance in lung adenosquamous carcinoma with concurrent EGFR and PIK3CA mutations: A case report and literature review. Cancer Manage Res. (2021) 13:7047–53. doi: 10.2147/CMAR.S326094
25. Kim SY, Kim KE, Kim Y, Chung CK. A patient with a lung adenosquamous carcinoma harboring a de novo T790M mutation and huge nonbacterial vegetative growths successfully treated with osimertinib: A case report. Thorac Cancer. (2023) 14:1530–3. doi: 10.1111/1759-7714.14896
26. Mai SX, Wang Y, Wang XM, Yang W, Gao HC, Xu ZN, et al. Neoadjuvant ceritinib treatment in ALK-rearranged locally advanced adenosquamous carcinoma: A case report. Thorac Cancer. (2022) 13:2275–8. doi: 10.1111/1759-7714.14558
27. Cheng C, Luo Z, Xiong W, Shi ZQ, Tan H. Epidemiology and survival outcomes in adenosquamous carcinoma: a population-based study. Int J Colorectal Disease. (2022) 37:1581–92. doi: 10.1007/s00384-022-04198-4
28. Toyooka S, Yatabe Y, Tokumo M, Ichimura K, Asano H, Tomii K, et al. Mutations of epidermal growth factor receptor andK-rasgenes in adenosquamous carcinoma of the lung. Int J Cancer. (2006) 118:1588–90. doi: 10.1002/ijc.21500
29. Shi XH, Wu HW, Lu JL, Duan HL, Liu XG, Liang ZY. Screening for major driver oncogene alterations in adenosquamous lung carcinoma using PCR coupled with next-generation and Sanger sequencing methods. Sci Rep-Uk. (2016) 6:22297. doi: 10.1038/srep22297
30. Ge-Ge L, Cuicui G, Leiqiang L, Yongcang T, Jiangang M, Yiwen O, et al. Case report: A case report and literature review about Pathological transformation of lung adenosquamous cell carcinoma. Front Oncol. (2022) 12. doi: 10.3389/fonc.2022.1029679
Keywords: tyrosine kinase inhibitor, adenosquamous lung carcinoma, EGFR, mutation, lung cancer
Citation: Xia X, Du W, Zhang Y, Li Y, Yu M and Liu Y (2024) Efficacy of epidermal growth factor receptor-tyrosine kinase inhibitor for lung adenosquamous cell carcinoma harboring EGFR mutation: a retrospective study and pooled analysis. Front. Oncol. 14:1354854. doi: 10.3389/fonc.2024.1354854
Received: 13 December 2023; Accepted: 24 June 2024;
Published: 04 July 2024.
Edited by:
Lizza E. L. Hendriks, Maastricht University Medical Centre, NetherlandsReviewed by:
Petros Christopoulos, Heidelberg University Hospital, GermanyYusuke Okuma, National Cancer Center Hospital, Japan
Copyright © 2024 Xia, Du, Zhang, Li, Yu and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yongmei Liu, bHltaTc1QDE2My5jb20=
 Xueming Xia1
Xueming Xia1 Wei Du
Wei Du Min Yu
Min Yu Yongmei Liu
Yongmei Liu