- 1Department of Surgery, Russian Medical Academy of Continuous Professional Education, Moscow, Russia
- 2Department of Surgery, S.P. Botkin City Clinical Hospital, Moscow, Russia
- 3Department of Faculty Surgery №2, I.M.Sechenov First Moscow State Medical University, Moscow, Russia
- 4Department of Gastrointestinal Surgery, Østfold Hospital Trust, Grålum, Norway
- 5Department of Surgery, Fonna Hospital Trust, Odda, Norway
- 6Intervention Centre, Oslo University Hospital – Rikshospitalet, Oslo, Norway
- 7Department of Surgery №1, Yerevan State Medical University after M.Heratsi, Yerevan, Armenia
Objective: This review article summarises the latest evidence for preventive central lymph node dissection in patients with papillary thyroid cancer taking into account the possible complications and risk of recurrence.
Background: Papillary thyroid cancer is the most frequent histological variant of malignant neoplasms of the thyroid gland. It accounts for about 80-85% of all cases of thyroid cancer. Despite good postoperative results and an excellent survival rate in comparison with many other malignant diseases, tumor metastases to the cervical lymph nodes are frequent. Most researchers agree that the presence of obvious metastases in the lymph nodes requires careful lymph node dissection. It was suggested to perform preventive routine lymphadenectomy in all patients with malignant thyroid diseases referred to surgery.
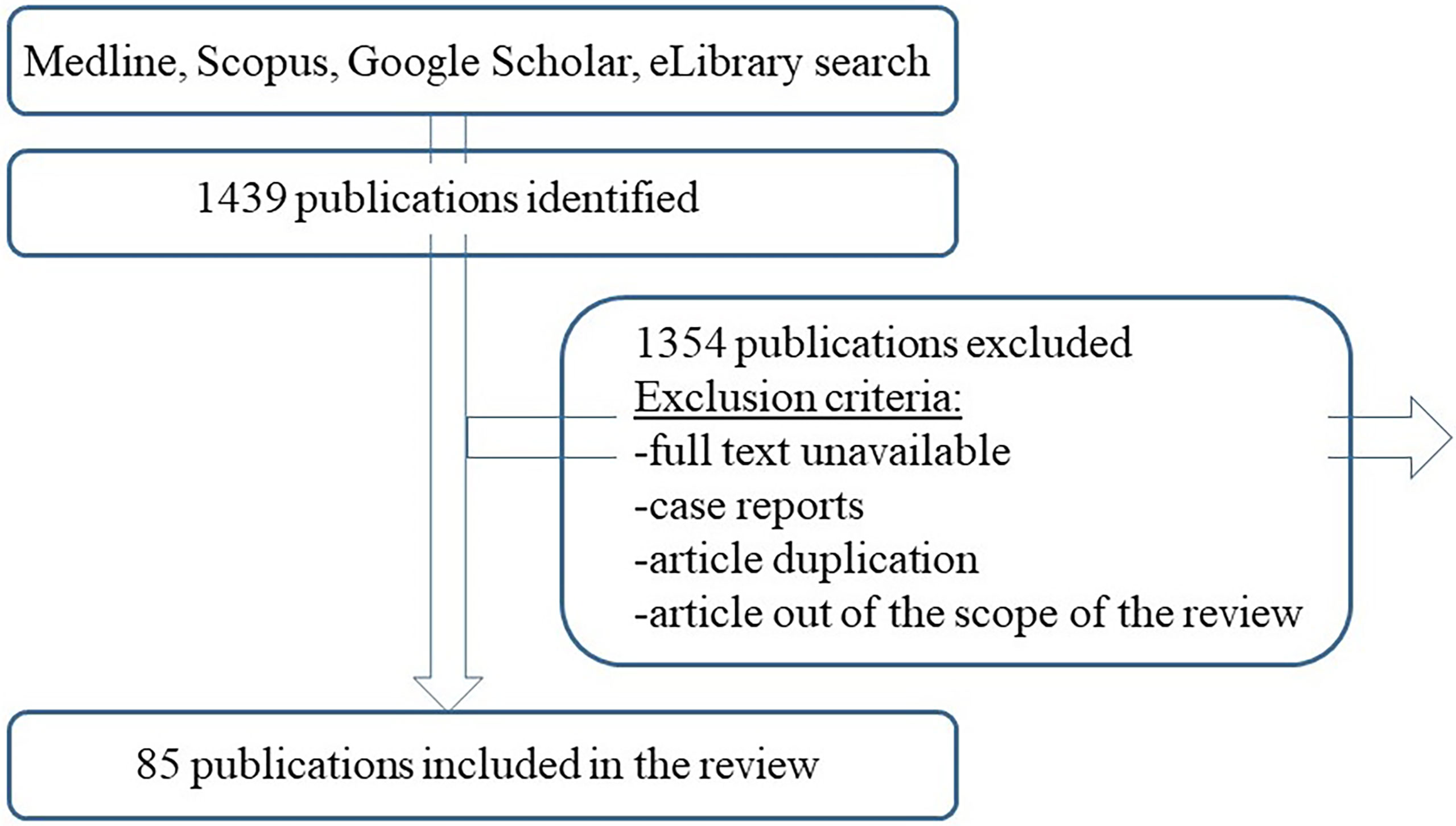
Methods: It was performed the literature review using the “papillary thyroid cancer”, “central lymph node dissection”, “hypocalcemia”, “recurrent laryngeal nerve paresis”, “metastasis”, “cancer recurrence” along with the MESH terms. The reference list of the articles was carefully reviewed as a potential source of information. The search was based on Medline, Scopus, Google Scholar, eLibrary engines. Selected publications were analyzed and their synthesis was used to write the review and analyse the role of preventive central lymph node dissection in patients with papillary thyroid cancer.
Conclusions: The necessity of preventive central lymph node dissection in patients with differentiated papillary thyroid carcinoma is still controversial. There is much evidence that it increases the frequency of transient hypocalcemia. Due to the fact that this complication is temporary, its significance in clinical practice is debatable. It can also be assumed that an extant of surgery in the neck area is associated with an increased risk of recurrent laryngeal nerve injury. However, most studies indicate that this injury is associated more with thyroidectomy itself than with lymph node dissection. Recurrent laryngeal nerve dysfunction is also a temporary complication in the vast majority of cases. At the same time, a large amount of data shows that central lymph node dissection reduces the risk of thyroid cancer recurrence in two times.
Introduction
Papillary thyroid cancer is the most frequent histological variant of malignant neoplasms of the thyroid gland. It accounts for about 80-85% of all cases of thyroid cancer. At the same time, the 10-year survival rate is more than 90% (1–4). Despite good postoperative results and an excellent survival rate in comparison with many other malignant diseases, tumor metastases to the cervical lymph nodes occur on average in 33% of patients (5). The most common area of metastases for papillary thyroid cancer is group VI (central) of the cervical lymph nodes (6). Given the high incidence of metastases to the cervical lymph nodes, prophylactic central lymph node dissection may be a logical procedure to reduce the risk of postoperative tumor recurrence (7). Nevertheless, the accumulated experience in the treatment and research of thyroid cancer has led to the question of the rationality of preventive central lymphadenectomy as a routine procedure (8). This is primarily due to the fact that lymph node dissection increases the risk of possible complications such as hypoparathyroidism and recurrent laryngeal nerve injury (9). Most researchers agree that the presence of obvious metastases in the lymph nodes requires careful lymph node dissection. The necessity of preventive or routine lymphadenectomy for all patients with malignant thyroid diseases remains controversial. The current review provides an analysis of the accumulated knowledge of preventive lymphadenectomy as well as its benefits and disadvantages.
Methods
DDD, SDC, KMI performed the literature search using the “papillary thyroid cancer”, “central lymph node dissection”, “hypocalcemia”, “recurrent laryngeal nerve paresis”, “metastasis”, “cancer recurrence” along with the MESH terms in English and Russian languages with no year limitation. The reference list of the articles was carefully reviewed as a potential source of information. The search was based on Medline, Scopus, Google Scholar, eLibrary engines. Selected publications were analyzed and their synthesis was used to write the review and analyse the role of preventive central lymph node dissection in patients with papillary thyroid cancer (Figure 1).
Central Lymph Node Dissection and its Advantages
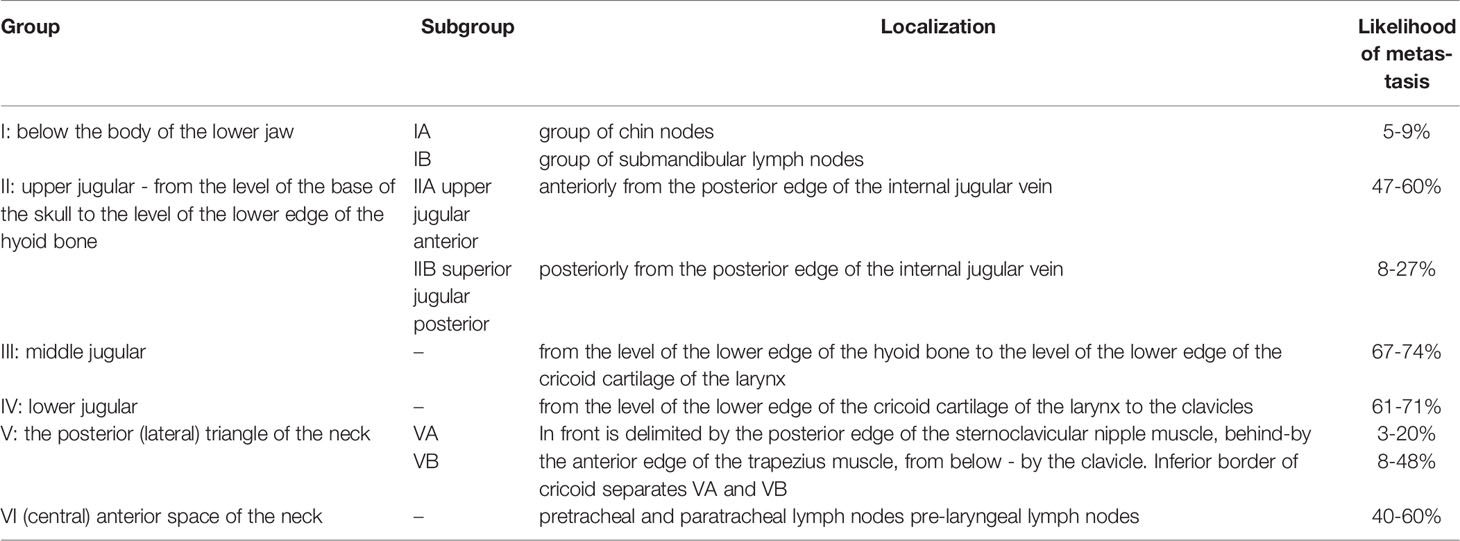
Central lymph node dissection involves the removal of group VI lymph nodes. The dissection is performed from the hyoid bone to the suprasternal notch along the carotid arteries, trachea, and prevertebral fascia (Figure 2, Table 1). Most often metastases are found in this anatomical area on histological examination, at the same time, they are not always visible on ultrasound, CT, or even during surgery (10–13).
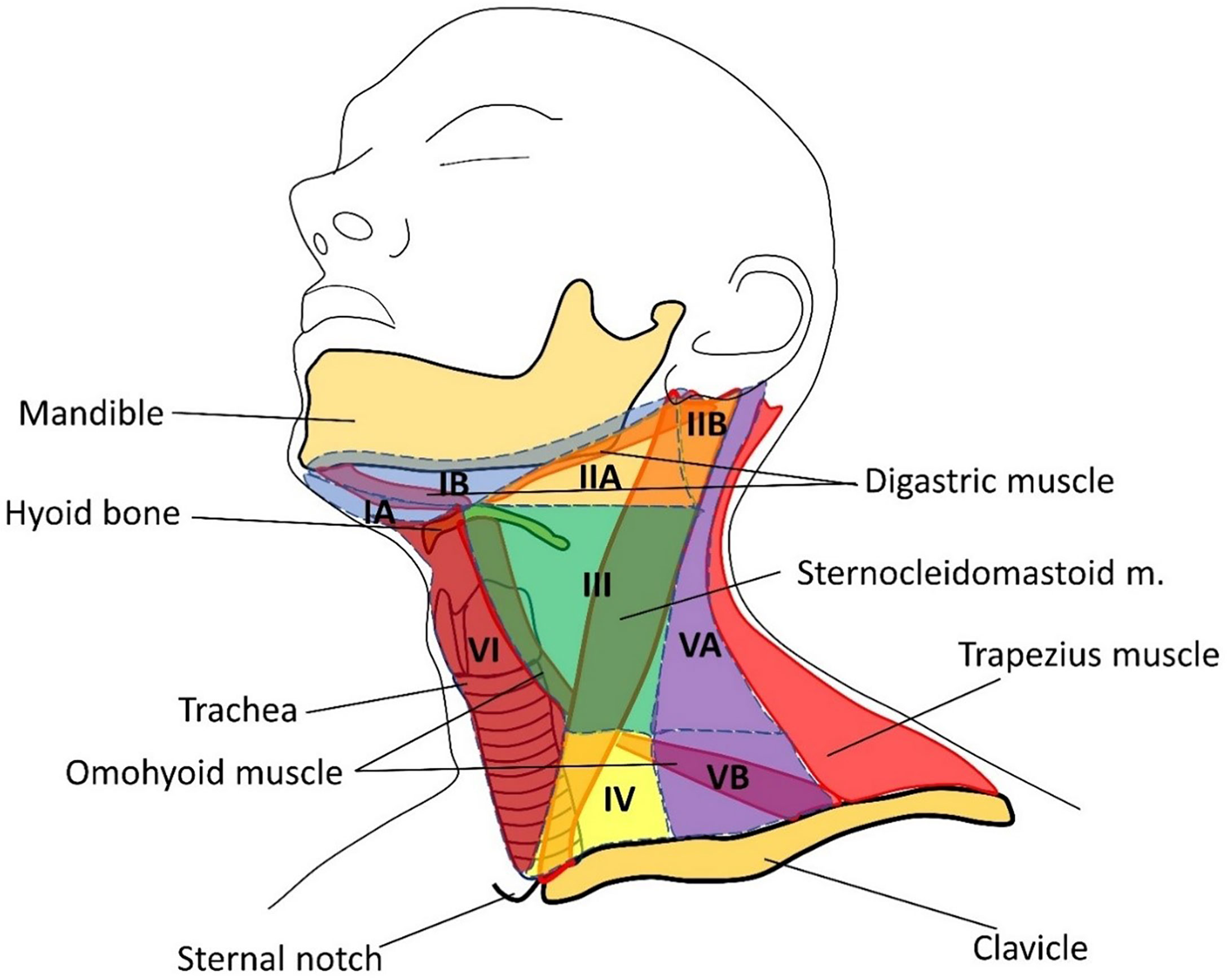
Figure 2 Groups of cervical lymph nodes. I – below the body of the lower jaw; IA: group of chin nodes; IB: group of submandibular lymph nodes; II: upper jugular; IIA: upper jugular anterior; IIB: upper jugular posterior; III: middle jugular; IV: lower jugular; V: posterior (lateral) triangle of the neck; VA: accessory; VB: supraclavicular; VI: anterior space of the neck.
The main advantages of preventive central dissection are: removal of subclinical metastases, improvement of postoperative survival by reducing the frequency of cancer recurrences, reduction in the number of repeated operations for thyroid cancer, significantly more noticeable decrease in thyroglobulin in the postoperative period (2, 14). A meta-analysis of 14 studies involving 4573 patients showed that, on average, lymph node group VI metastases occur in 33% of cases (5). An important factor is also the frequency of metastases outside the central group. A systematic review and meta-analysis of 23 studies, which included 18741 patients, showed that, on average, metastases to the lateral group of lymph nodes occur in 20.9% of patients (15). However, in other studies, the frequency of metastases reaches 40-90% (14, 16, 17). Lymph node metastases are responsible for 75% of locoregional relapses and 50% of deaths (18).
An important reason for performing preventive central lymphadenectomy is the lack of reliable methods for detecting metastases in lymph nodes before operation. The sensitivity of preoperative neck ultrasound to detect lymph nodes is only 46-88%, depending on the size of lymph nodes and technical capabilities of the ultrasound equipment (19–21). CT of the neck also has low rate of sensitivity (74% to 82%) (15). The sensitivity of this method is noticeably reduced in cases when the lymph nodes are less than 9 mm in diameter, which happens in the vast majority of cases (22). According to the results of a meta-analysis of 9 studies involving 1691 patients, the combination of ultrasound examination and CT in detecting metastases to lymph nodes gives a sensitivity of 69% (23).
Another important aspect is the lack of standards for assessing lymph nodes by ultrasound and CT (15, 24). In fact, an increased lymph nodes do not always indicate on their metastatic involvement. In 20% of cases, an increased lymph nodes may be detected against the background of an inflammatory process, mainly Hashimoto’s thyroiditis (25, 26). A meta-analysis of 71 studies, which included 44034 patients, showed that thyroid cancer with or without lymph node involvement is much less common in patients with Hashimoto’s thyroiditis. Hashimoto’s thyroiditis also reduces the risk of cancer recurrence in patients operated on for malignant tumors of the thyroid gland (27, 28).
Staging of thyroid cancer depends on the evaluation of lymph nodes. Many prognostic scales are also accounted for by the involvement of lymph nodes as one of the main criteria of disease severity and negative prognosis (29–31).
Disadvantages of Central Lymph Node Dissection
The main argument in favor of refusing preventive lymphadenectomy is the higher frequency of postoperative complications. The main complications after thyroidectomy with central lymph node dissection are transient and permanent hypoparathyroidism, transient and permanent recurrent laryngeal nerve paresis. According to a meta-analysis of 14 articles that included 4573 patients, the incidence of permanent hypoparathyroidism, recurrent laryngeal nerve paresis and cancer recurrence is 1.1%, 0.5% and 2.8%, respectively (5). The frequency of postoperative hypoparathyroidism is 36.1-42.4%, but it decreases to 1.1-3.9% after 6 months -1 year (32, 33). Risk factors for postoperative hypocalcemia are female, bilateral lymphadenectomy, high ligation of the thyroid arteries, large thyroid gland, low levels of preoperative calcium and parathyroid hormone, autotransplantation of the parathyroid gland (32–35). At the same time, if the parathyroid gland was removed during surgery, its reimplantation with adequate blood supply significantly reduces the risk of hypocalcemia (34, 36). Due to the fact that permanent hypocalcemia is a rare complication, it is difficult to reliably assess the effect of lymphodissection because a small number of cases creates difficulties for adequate statistical analysis. To assess the relationship between lymphodissection and permanent hypocalcemia, an analysis of a large database at the national level is required. The heterogeneity of the data also makes it difficult to perform a meta-analysis that would reliably indicate the effect of central lymph node dissection on the frequency of hypocalcemia (37). The hypocalcemia in the postoperative period has many reasons. A meta-analysis of 23 studies with a total of 877356 patients indicates 12 risk factors for the development of postoperative hypocalcemia, such as hypoparathyroidism, thyroidectomy, hypomagnesemia, vitamin D deficiency, female, thyroid cancer, thyroiditis, retrosternal goiter, parathyroid attachment, central lymph node dissection, lateral lymph node dissection, dissection of surrounding thyroid gland tissues (38).
Seo and co-authors compared 52707 thyroidectomies and 139626 thyroidectomies with central lymph node dissection and found that the frequency of permanent hypocalcemia was higher when performing central lymph node dissection (5.4% compared to 4.6%) (39). Another important finding was the frequency of hypocalcemia depending on the operational activity of the hospital. In hospitals where less than 200 surgical interventions are performed per year, the frequency of hypocalcemia was 6.0-6.5%, in hospitals where 200-799 thyroidectomies are performed per year the frequency of hypocalcemia varied from 3.2% to 7.4%, and in hospitals where more than 800 operations are performed per year – only 3.3% (39). Thus, it is reliably known that the volume of surgical intervention and an experience of surgeons are interrelated with the frequency of postoperative hypocalcemia.
Transient and permanent disorders of recurrent laryngeal nerve function after thyroidectomy remain a serious problem. Postoperative laryngeal paresis occurs in 3.28-27.8% of cases (40–42). In most cases, recurrent laryngeal nerve paresis is transient, because 94.6% of patients have a complete restoration of the voice (43). Lyomasa and co-authors noted that the voice impairment due to injury of the upper laryngeal nerve decreases to zero in the 6th month after surgery in comparison with the first postoperative day. As far as recurrent laryngeal nerve, the authors noted that paresis occurs in 27.8% of patients on the first postoperative day, but decreases to 6.6% after 6 months (41). An important observation is that atypical variants of recurrent laryngeal nerve occur in 24.4% and require a careful identification during surgery (44, 45). In general, the risk of nerve damage is low and more common during repeated operations due to fibrosis of surrounding tissues, which makes it difficult to detect the nerve (46). The question whether central lymph node dissection is a risk factor for transient paresis and laryngeal paralysis remains controvercial because there are radically different data (46–48). Machens and co-authors, analyzing the data of 102 pediatric patients who underwent thyroidectomy with central lymph node dissection, noted an increased rate of only transient laryngeal paresis (6).
Another important problem is cancer metastasis to the lymph nodes along the recurrent laryngeal nerve, which occurs in 8.65% of cases and requires meticulous dissection in that area (49). Manipulations in the nerve area are naturally associated with the risk of its dysfunction in the postoperative period, even if the nerve was not directly injured during the operation. In some patients, thyroid cancer nerve involvement is also noted, which is associated with neuropaxy (50).
Several systematic reviews compared incidence of complications in two groups of patients: those who underwent thyroidectomy and thyroidectomy in combination with central lymph node dissection (Table 2). The majority of authors agree that the risk of transient hypocalcemia increases by 1.5-2.5 times, while the risk of paresis does not differ between the two groups (Table 2).

Table 2 Systematic meta-analyses reviews comparing risk hypocalcemia and recurrent laryngeal nerve paresis after thyroidectomy and thyroidectomy with preventive central lymph node dissection.
It is worth noting that the risk of complications is significantly reduced in hospitals with a large number of surgical interventions per year. Thus, it is rational to perform thyroidectomy with central lymph node dissection in large institutions where a specialist performs at least 50 operations per year (59–62).
Recommendations of Professional Communities and the Risk of Cancer Recurrence
National and international communities have different assessments of the need for central lymph node dissection in thyroid cancer. These recommendations are presented in Table 3.
The majority of surgeons estimate the size of tumor as T3/T4 for recommendation of lymph node dissection. According to Japanese professional communities central lymph node dissection should always preventively be performed. Japanese institutions have accumulated extensive experience in performing preventive lymph node dissections. Out of 4301 patients in whom lymphogenic metastases were not detected preoperatively, preventive lymph node dissection revealed N1a in 2548 (59%) cases. During follow-up cancer recurrence was diagnosed only in 1.2% of cases. At the same time, the 10-year and 20-year survival rates were 99.1 and 98.2%, respectively (71). In fact, there are two approaches: the “western” way of treatment, where preference is given to thyroidectomy followed by radioactive iodine ablation, and the “eastern” way of treatment, where preference is given to preventive lymph node dissection (72, 73).
There were also attempts to find the “optimal” group of patients for whom preventive lymph node dissection is beneficial. The risk group for detecting metastases in lymph nodes in the absence of information about their presence before surgery includes the following factors: male sex, age less than 45 years, multiple tumors, tumor size more than 2 cm, localization in the center of the lobe or at the lower pole, invasion into vessels, spread outside the thyroid gland capsule (74). Other factors may include isthmus location of the tumor, male sex, age less thatn 45 years, tumor adjacent to dorsal membranes, and irregular borders (75, 76). Most meta-analyses up to 2013 indicated a lack of data that central lymph node dissection somehow reduces the risk of locoregional cancer recurrence (Table 4). Further studies have shown that preventive central lymph node dissection reduces the risk of relapse by about half.

Table 4 Meta-analyses assessing the frequency of cancer recurrence depending on the method of surgery.
Comparison of Surgery and Radioactive Iodine Treatment
The role of radioactive iodine (RAI) treatment in differentiated thyroid cancer is controversial. It seems that RAI is a relatively safe treatment option in moderate doses (79). Recent studies demonstrate that in patients with low-risk thyroid cancer undergoing thyroidectomy, a follow-up strategy that did not involve the use of radioiodine was noninferior to an ablation strategy with radioiodine regarding the occurrence of functional, structural, and biologic events at 3 years (80). Follow-up at 6-18 months is based on serum thyroglobulin, thyroglobulin-antibody determination and neck ultrasonography as the cancer recurrence rate is approximately 3% (81). It seems that low-risk thyroid cancer can be monitored effectively as it does not cause significant mortality and morbidity (82). The recent advances in our understanding of thyroid cancer has allowed to expand the definition of “low risk” as demonstrated in Table 5 (64, 82).
Moreover, further research has demonstrated theat several other features can be included as criteria for low-risk: intra-thyroidal encapsulated follicular variant of papillary thyroid cancer, papillary thyroid cancer with: clinical N0 or ≤5 pathologic N1 micro-metastases (<0.2 cm in largest dimension), intra-thyroidal well differentiated follicular thyroid cancer with capsular invasion and no or minimal (<4 foci) vascular invasion, intra-thyroidal papillary microcarcinoma, unifocal or multifocal, including BRAFV600E mutated (if known) (25, 64, 83).
Moreover, the clinical response to RAI therapy for thyroidectomy with central lymph node dissection is not superior to thyroidectomy alone in cN0 papillary thyroid cancer patients (84).
Conclusions
The necessity of preventive central lymph node dissection in patients with differentiated papillary thyroid carcinoma remains controversial. There is much evidence that it increases the frequency of transient hypocalcemia. Due to the fact that this complication is temporary, its significance in clinical practice is debatable. It can also be assumed that an extant of surgery in the neck area is associated with an increased risk of recurrent laryngeal nerve injury, however, most studies indicate that this injury is associated more with thyroidectomy itself than with lymph node dissection. Recurrent laryngeal nerve dysfunction is also a temporary complication in the vast majority of cases. At the same time, a large amount of data shows that central lymph node dissection reduces the risk of thyroid cancer recurrence by about half. It is also important to emphasize that it is rational to perform such operations in medical centers with a large number of thyroid interventions, which significantly reduces the risk of complications. Further research will help to determine an individual approach in the selection of patients for whom central lymph node dissection is beneficial. Taking into account the absence of reliable criteria for determination of metastatic lymph node lesions before operation, low accuracy of ultrasound examination and CT, the risk of cancer recurrence, central lymph node dissection remains an urgent method of thyroid cancer recurrence prevention.
Author Contributions
(I) Conception and design: DD. (II) Administrative support: DD, AS, VS, AK. (III) Literature search and systematisation: DD, KI, SC. (IV) Data analysis and interpretation: All authors. (V) Manuscript drafting: DD, AK, SC, VS. (VI) Critical revision and final approval of manuscript: All authors.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Ito Y, Miyauchi A, Kihara M, Fukushima M, Higashiyama T, Miya A. Overall Survival of Papillary Thyroid Carcinoma Patients: A Single-Institution Long-Term Follow-Up of 5897 Patients. World J Surg (2018) 42(3):615–22. doi: 10.1007/s00268-018-4479-z
2. Mazzaferri EL, Jhiang SM. Long-Term Impact of Initial Surgical and Medical Therapy on Papillary and Follicular Thyroid Cancer. Am J Med (1994) 97(5):418–28. doi: 10.1016/0002-9343(94)90321-2
3. Lundgren CI, Hall P, Dickman PW, Zedenius J. Clinically Significant Prognostic Factors for Differentiated Thyroid Carcinoma: A Population-Based, Nested Case-Control Study. Cancer (2006) 106(3):524–31. doi: 10.1002/cncr.21653
4. Davies L, Welch HG. Thyroid Cancer Survival in the United States: Observational Data From 1973 to 2005. Arch Otolaryngol Head Neck Surg (2010) 136(5):440–4. doi: 10.1001/archoto.2010.55
5. Liu L-S, Liang J, Li J-H, Liu X, Jiang L, Long J-X, et al. The Incidence and Risk Factors for Central Lymph Node Metastasis in Cn0 Papillary Thyroid Microcarcinoma: A Meta-Analysis. Eur Arch Oto Rhino Laryngol (2017) 274(3):1327–38. doi: 10.1007/s00405-016-4302-0
6. Machens A, Hinze R, Thomusch O, Dralle H. Pattern of Nodal Metastasis for Primary and Reoperative Thyroid Cancer. World J Surg (2002) 26(1):22–8. doi: 10.1007/s00268-001-0176-3
7. White ML, Gauger PG, Doherty GM. Central Lymph Node Dissection in Differentiated Thyroid Cancer. World J Surg (2007) 31(5):895–904. doi: 10.1007/s00268-006-0907-6
8. Ahn J-H, Kwak JH, Yoon SG, Yi JW, Yu HW, Kwon H, et al. A Prospective Randomized Controlled Trial to Assess the Efficacy and Safety of Prophylactic Central Compartment Lymph Node Dissection in Papillary Thyroid Carcinoma. Surgery (2021) 171(1):182–9. doi: 10.1016/j.surg.2021.03.071
9. Giordano D, Valcavi R, Thompson GB, Pedroni C, Renna L, Gradoni P, et al. Complications of Central Neck Dissection in Patients With Papillary Thyroid Carcinoma: Results of a Study on 1087 Patients and Review of the Literature. Thyroid (2012) 22(9):911–7. doi: 10.1089/thy.2012.0011
10. Shirley LA, Jones NB, Phay JE. The Role of Central Neck Lymph Node Dissection in the Management of Papillary Thyroid Cancer. Front Oncol (2017) 7(122). doi: 10.3389/fonc.2017.00122
11. Ito Y, Tomoda C, Uruno T, Takamura Y, Miya A, Kobayashi K, et al. Clinical Significance of Metastasis to the Central Compartment From Papillary Microcarcinoma of the Thyroid. World J Surg (2006) 30(1):91–9. doi: 10.1007/s00268-005-0113-y
12. Marshall CL, Lee JE, Xing Y, Perrier ND, Edeiken BS, Evans DB, et al. Routine Pre-Operative Ultrasonography for Papillary Thyroid Cancer: Effects on Cervical Recurrence. Surgery (2009) 146(6):1063–72. doi: 10.1016/j.surg.2009.09.027
13. Patel KN, Yip L, Lubitz CC, Grubbs EG, Miller BS, Shen W, et al. The American Association of Endocrine Surgeons Guidelines for the Definitive Surgical Management of Thyroid Disease in Adults. Ann Surg (2020) 271(3):e21–93. doi: 10.1097/SLA.0000000000003580
14. Popadich A, Levin O, Lee JC, Smooke-Praw S, Ro K, Fazel M, et al. A Multicenter Cohort Study of Total Thyroidectomy and Routine Central Lymph Node Dissection for Cn0 Papillary Thyroid Cancer. Surgery (2011) 150(6):1048–57. doi: 10.1016/j.surg.2011.09.003
15. So YK, Kim MJ, Kim S, Son YI. Lateral Lymph Node Metastasis in Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis for Prevalence, Risk Factors, and Location. Int J Surg (London England) (2018) 50:94–103. doi: 10.1016/j.ijsu.2017.12.029
16. Moo TA, McGill J, Allendorf J, Lee J, Fahey T 3rd, Zarnegar R. Impact of Prophylactic Central Neck Lymph Node Dissection on Early Recurrence in Papillary Thyroid Carcinoma. World J Surg (2010) 34(6):1187–91. doi: 10.1007/s00268-010-0418-3
17. Moo TA, Umunna B, Kato M, Butriago D, Kundel A, Lee JA, et al. Ipsilateral Versus Bilateral Central Neck Lymph Node Dissection in Papillary Thyroid Carcinoma. Ann Surg (2009) 250(3):403–8. doi: 10.1097/SLA.0b013e3181b3adab
18. Grodski S, Cornford L, Sywak M, Sidhu S, Delbridge L. Routine Level Vi Lymph Node Dissection for Papillary Thyroid Cancer: Surgical Technique. ANZ J Surg (2007) 77(4):203–8. doi: 10.1111/j.1445-2197.2007.04019.x
19. Choi YJ, Yun JS, Kook SH, Jung EC, Park YL. Clinical and Imaging Assessment of Cervical Lymph Node Metastasis in Papillary Thyroid Carcinomas. World J Surg (2010) 34(7):1494–9. doi: 10.1007/s00268-010-0541-1
20. Vergez S, Sarini J, Percodani J, Serrano E, Caron P. Lymph Node Management in Clinically Node-Negative Patients With Papillary Thyroid Carcinoma. Eur J Surg Oncol J Eur Soc Surg Oncol Br Assoc Surg Oncol (2010) 36(8):777–82. doi: 10.1016/j.ejso.2010.06.015
21. Wu L-M, Gu H-Y, Qu X-H, Zheng J, Zhang W, Yin Y, et al. The Accuracy of Ultrasonography in the Preoperative Diagnosis of Cervical Lymph Node Metastasis in Patients With Papillary Thyroid Carcinoma: A Meta-Analysis. Eur J Radiol (2012) 81(8):1798–805. doi: 10.1016/j.ejrad.2011.04.028
22. Liu J, Wang Z, Shao H, Qu D, Liu J, Yao L. Improving CT Detection Sensitivity for Nodal Metastases in Oesophageal Cancer With Combination of Smaller Size and Lymph Node Axial Ratio. Eur Radiol (2018) 28(1):188–95. doi: 10.1007/s00330-017-4935-4
23. Suh CH, Baek JH, Choi YJ, Lee JH. Performance of CT in the Preoperative Diagnosis of Cervical Lymph Node Metastasis in Patients With Papillary Thyroid Cancer: A Systematic Review and Meta-Analysis. AJNR Am J Neuroradiol (2017) 38(1):154–61. doi: 10.3174/ajnr.A4967
24. Dolidze DD, Mumladze RB, Vardanyan AV, Vartanyan KF, Vasilyev IT, Levchuk DI, et al. Features of Radionuclide Research After Thyroidectomy at the Thyroid Gland Cancer. Endocrine Surg (2014) 8(4):23–36. doi: 10.14341/serg2014423-36
25. Rosenbaum MA, McHenry CR. Central Neck Dissection for Papillary Thyroid Cancer. Arch Otolaryngol Head Neck Surg (2009) 135(11):1092–7. doi: 10.1001/archoto.2009.158
26. Sywak M, Cornford L, Roach P, Stalberg P, Sidhu S, Delbridge L. Routine Ipsilateral Level VI Lymphadenectomy Reduces Postoperative Thyroglobulin Levels in Papillary Thyroid Cancer. Surgery (2006) 140(6):1000–5. doi: 10.1016/j.surg.2006.08.001
27. Moon S, Chung HS, Yu JM, Yoo HJ, Park JH, Kim DS, et al. Associations Between Hashimoto Thyroiditis and Clinical Outcomes of Papillary Thyroid Cancer: A Meta-Analysis of Observational Studies. Endocrinol Metab (Seoul) (2018) 33(4):473–84. doi: 10.3803/EnM.2018.33.4.473
28. Jin K, Li L, Liu Y, Wang X. The Characteristics and Risk Factors of Central Compartment Lymph Node Metastasis in Cn0 Papillary Thyroid Carcinoma Coexistent With Hashimoto’s Thyroiditis. Gland Surg (2020) 9(6):2026–34. doi: 10.21037/gs-20-699
29. Haigh PI, Urbach DR, Rotstein LE. AMES Prognostic Index and Extent of Thyroidectomy for Well-Differentiated Thyroid Cancer in the United States. Surgery (2004) 136(3):609–16. doi: 10.1016/j.surg.2003.12.009
30. Teo KW, Yuan NK, Tan WB, Parameswaran R. Comparison of Prognostic Scoring Systems in Follicular Thyroid Cancer. Ann R Coll Surgeons England (2017) 99(6):479–84. doi: 10.1308/rcsann.2017.0072
31. Lang BH-H, Lo C-Y, Chan W-F, Lam K-Y, Wan K-Y. Staging Systems for Papillary Thyroid Carcinoma: A Review and Comparison. Ann Surg (2007) 245(3):366–78. doi: 10.1097/01.sla.0000250445.92336.2a
32. Su A, Wang B, Gong Y, Gong R, Li Z, Zhu J. Risk Factors of Hypoparathyroidism Following Total Thyroidectomy With Central Lymph Node Dissection. Medicine (2017) 96(39):e8162. doi: 10.1097/MD.0000000000008162
33. Ru Z, Mingliang W, Maofei W, Qiaofeng C, Jianming Y. Analysis of Risk Factors for Hypoparathyroidism After Total Thyroidectomy. Front Surg (2021) 8(158). doi: 10.3389/fsurg.2021.668498
34. Yazıcıoğlu MÖ, Yılmaz A, Kocaöz S, Özçağlayan R, Parlak Ö. Risks and Prediction of Postoperative Hypoparathyroidism Due to Thyroid Surgery. Sci Rep (2021) 11(1):11876. doi: 10.1038/s41598-021-91277-1
35. Eismontas V, Slepavicius A, Janusonis V, Zeromskas P, Beisa V, Strupas K, et al. Predictors of Postoperative Hypocalcemia Occurring After a Total Thyroidectomy: Results of Prospective Multicenter Study. BMC Surg (2018) 18(1):55. doi: 10.1186/s12893-018-0387-2
36. Hicks G, George R, Sywak M. Short and Long-Term Impact of Parathyroid Autotransplantation on Parathyroid Function After Total Thyroidectomy. Gland Surg (2017) 6(Suppl 1): S75–85. doi: 10.21037/gs.2017.09.15
37. Qin Y, Sun W, Wang Z, Dong W, He L, Zhang T, et al. A Meta-Analysis of Risk Factors for Transient and Permanent Hypocalcemia After Total Thyroidectomy. Front Oncol (2021) 10:614089. doi: 10.3389/fonc.2020.614089
38. Chen Z, Zhao Q, Du J, Wang Y, Han R, Xu C, et al. Risk Factors for Postoperative Hypocalcaemia After Thyroidectomy: A Systematic Review and Meta-Analysis. J Int Med Res (2021) 49(3):300060521996911. doi: 10.1177/0300060521996911
39. Seo GH, Chai YJ, Choi HJ, Lee KE. Incidence of Permanent Hypocalcaemia After Total Thyroidectomy With or Without Central Neck Dissection for Thyroid Carcinoma: A Nationwide Claim Study. Clin Endocrinol (2016) 85(3):483–7. doi: 10.1111/cen.13082
40. Enomoto K, Uchino S, Watanabe S, Enomoto Y, Noguchi S. Recurrent Laryngeal Nerve Palsy During Surgery for Benign Thyroid Diseases: Risk Factors and Outcome Analysis. Surgery (2014) 155(3):522–8. doi: 10.1016/j.surg.2013.11.005
41. Iyomasa RM, Tagliarini JV, Rodrigues SA, Tavares ELM, Martins RHG. Laryngeal and Vocal Alterations After Thyroidectomy. Braz J Otorhinolaryngol (2019) 85(1):3–10. doi: 10.1016/j.bjorl.2017.08.015
42. Shabunin AV, Dolidze DD, Podvyaznikov SO, Mel’nik KV, Mumladze RB, Vardanyan AV, et al. Reduced Low-Traumatic Access Thyroidectomy With Central Neck Dissection. Head Neck Tumors (HNT) (2016) 6(1):46–54. doi: 10.17650/2222-1468-2016-6-1-46-54
43. Chiang FY, Wang LF, Huang YF, Lee KW, Kuo WR. Recurrent Laryngeal Nerve Palsy After Thyroidectomy With Routine Identification of the Recurrent Laryngeal Nerve. Surgery (2005) 137(3):342–7. doi: 10.1016/j.surg.2004.09.008
44. Shen C, Xiang M, Wu H, Ma Y, Chen L, Cheng L. Routine Exposure of Recurrent Laryngeal Nerve in Thyroid Surgery can Prevent Nerve Injury. Neural Regeneration Res 2013 15th June 1 (2013) 8(17):1568–75. doi: 10.3969/j.issn.1673-5374.2013.17.004
45. Pradeep PV, Jayashree B, Harshita SS. A Closer Look at Laryngeal Nerves During Thyroid Surgery: A Descriptive Study of 584 Nerves. Anat Res Int (2012) 2012:490390. doi: 10.1155/2012/490390
46. Varaldo E, Ansaldo GL, Mascherini M, Cafiero F, Minuto MN. Neurological Complications in Thyroid Surgery: A Surgical Point of View on Laryngeal Nerves. Front Endocrinol (Lausanne) (2014) 5(108). doi: 10.3389/fendo.2014.00108
47. Boute P, Merlin J, Biet A, Cuvelier P, Strunski V, Page C. Morbidity of Central Compartment Dissection for Differentiated Thyroid Carcinoma of the Follicular Epithelium. Eur Ann Otorhinolaryngol Head Neck Diseases (2013) 130(5):245–9. doi: 10.1016/j.anorl.2012.10.004
48. Giulea C, Enciu O, Toma EA, Martin S, Fica S, Miron A. Total Thyroidectomy for Malignancy - is Central Neck Dissection a Risk Factor for Recurrent Nerve Injury and Postoperative Hypocalcemia? A Tertiary Center Experience in Romania. Acta Endocrinol (Buchar) (2019) 5(1):80–5. doi: 10.4183/aeb.2019.80
49. Li C, Xiang J, Wang Y. Risk Factors for Predicting Lymph Nodes Posterior to Right Recurrent Laryngeal Nerve (LN-prRLN) Metastasis in Thyroid Papillary Carcinoma: A Meta-Analysis. Int J Endocrinol (2019) 2019:7064328. doi: 10.1155/2019/7064328
50. Grachev NS, Vorozhtsov IN, Babaskina NV, Iaremenko E IU. Estimating the Effectiveness of Surgical Treatment of Children With Thryoid Neoplasms. J Pediatr Surg Anesth Intensive Care (2018) 8(2):33–47. doi: 10.30946/2219-4061-2018-8-2-33-47
51. Chisholm EJ, Kulinskaya E, Tolley NS. Systematic Review and Meta-Analysis of the Adverse Effects of Thyroidectomy Combined With Central Neck Dissection as Compared With Thyroidectomy Alone. Laryngoscope (2009) 119(6):1135–9. doi: 10.1002/lary.20236
52. Shan C-X, Zhang W, Jiang D-Z, Zheng X-M, Liu S, Qiu M. Routine Central Neck Dissection in Differentiated Thyroid Carcinoma: A Systematic Review and Meta-Analysis. Laryngoscope (2012) 122(4):797–804. doi: 10.1002/lary.22162
53. Lang BH-H, Ng S-H, Lau LLH, Cowling BJ, Wong KP, Wan KY. A Systematic Review and Meta-Analysis of Prophylactic Central Neck Dissection on Short-Term Locoregional Recurrence in Papillary Thyroid Carcinoma After Total Thyroidectomy. Thyroid (2013) 23(9):1087–98. doi: 10.1089/thy.2012.0608
54. Wang TS, Cheung K, Farrokhyar F, Roman SA, Sosa JA. A Meta-Analysis of the Effect of Prophylactic Central Compartment Neck Dissection on Locoregional Recurrence Rates in Patients With Papillary Thyroid Cancer. Ann Surg Oncol (2013) 20(11):3477–83. doi: 10.1245/s10434-013-3125-0
55. Zhu W, Zhong M, Ai Z. Systematic Evaluation of Prophylactic Neck Dissection for the Treatment of Papillary Thyroid Carcinoma. Japanese J Clin Oncol (2013) 43(9):883–8. doi: 10.1093/jjco/hyt087
56. Liang J, Li Z, Fang F, Yu T, Li S. Is Prophylactic Central Neck Dissection Necessary for Cn0 Differentiated Thyroid Cancer Patients at Initial Treatment? A Meta-Analysis of the Literature. Acta Otorhinolaryngol Ital (2017) 37(1):1–8. doi: 10.14639/0392-100X-1195
57. Sison C-ZI, Fernando AF, Gutierrez TMDG. Morbidity Outcomes of Prophylactic Central Neck Dissection With Total Thyroidectomy Versus Total Thyroidectomy Alone in Patients With Node Negative Papillary Thyroid Cancer: A Meta-Analysis of Observational Studies. Philippine J Otolaryngol Head Neck Surg (2019) 34(1):6–13. doi: 10.32412/pjohns.v34i1.127
58. Su H, Li Y. Prophylactic Central Neck Dissection and Local Recurrence in Papillary Thyroid Microcarcinoma: A Meta-Analysis. Braz J Otorhinolaryngol (2019) 85(2):237–43. doi: 10.1016/j.bjorl.2018.05.004
59. Shen WT, Ogawa L, Ruan D, Suh I, Kebebew E, Duh QY, et al. Central Neck Lymph Node Dissection for Papillary Thyroid Cancer: Comparison of Complication and Recurrence Rates in 295 Initial Dissections and Reoperations. Arch Surg (Chicago Ill 1960) (2010) 145(3):272–5. doi: 10.1001/archsurg.2010.9
60. Alvarado R, Sywak MS, Delbridge L, Sidhu SB. Central Lymph Node Dissection as a Secondary Procedure for Papillary Thyroid Cancer: Is There Added Morbidity? Surgery (2009) 145(5):514–8. doi: 10.1016/j.surg.2009.01.013
61. Rumiantsev PO. Prophylactic Central Lymph Nodes Dissection (VI Level) in Papillary Thyroid Cancer. Endocrine Surg (2015) 9(1):35–41. doi: 10.14341/serg2015135-41
62. Shabunin AV, Dolidze DD, Podvyaznikov SO, Melnik KV, Mumladze RB, Vardanyan AV, et al. Improved Technique Thyroidectomy With Lateral Modified Radical Neck Dissection. J Modern Oncol (2016) 18(2):72–7. doi: 10.26442/1815-1434_2016.2.72-77
63. Sancho JJ, Lennard TWJ, Paunovic I, Triponez F, Sitges-Serra A. Prophylactic Central Neck Disection in Papillary Thyroid Cancer: A Consensus Report of the European Society of Endocrine Surgeons (ESES). Langenbeck's Arch Surg (2014) 399(2):155–63. doi: 10.1007/s00423-013-1152-8
64. Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients With Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid (2016) 26(1):1–133. doi: 10.1089/thy.2015.0020
66. NCCN. NCCN Clinical Practice Guidelines in Oncology: Thyroid Carcinoma Version 1, Vol. 12Plymouth Meeting: National Comprehensive Cancer Network (2016).
67. Perros P, Boelaert K, Colley S, Evans C, Evans RM, Gerrard Ba G, et al. Guidelines for the Management of Thyroid Cancer. Clin Endocrinol (2014) 81:1–122. doi: 10.1111/cen.12515
68. Yi KH, Lee EK, Kang H-C, Koh Y, Kim SW, Kim IJ, et al. Revised Korean Thyroid Association Management Guidelines for Patients With Thyroid Nodules and Thyroid Cancer. ijt (2016) 9(2):59–126. doi: 10.11106/ijt.2016.9.2.59
69. Takami H, Ito Y, Okamoto T, Yoshida A. Therapeutic Strategy for Differentiated Thyroid Carcinoma in Japan Based on a Newly Established Guideline Managed by Japanese Society of Thyroid Surgeons and Japanese Association of Endocrine Surgeons. World J Surg (2011) 35(1):111–21. doi: 10.1007/s00268-010-0832-6
70. Ito Y, Onoda N, Okamoto T. The Revised Clinical Practice Guidelines on the Management of Thyroid Tumors by the Japan Associations of Endocrine Surgeons: Core Questions and Recommendations for Treatments of Thyroid Cancer. Endocrine J (2020) 67(7):669–717. doi: 10.1507/endocrj.EJ20-0025
71. Ito Y, Miyauchi A, Masuoka H, Fukushima M, Kihara M, Miya A. Excellent Prognosis of Central Lymph Node Recurrence-Free Survival for Cn0m0 Papillary Thyroid Carcinoma Patients Who Underwent Routine Prophylactic Central Node Dissection. World J Surg (2018) 42(8):2462–8. doi: 10.1007/s00268-018-4497-x
72. Ito Y, Miyauchi A. Thyroidectomy and Lymph Node Dissection in Papillary Thyroid Carcinoma. J Thyroid Res (2011) 2011:634170. doi: 10.4061/2011/634170
73. Yasuhiro I, Akira M. Surgical Treatment for Papillary Thyroid Carcinoma in Japan: Differences From Other Countries. Int J Thyroidol (2011) 4(2):75–9.
74. Sun W, Lan X, Zhang H, Dong W, Wang Z, He L, et al. Risk Factors for Central Lymph Node Metastasis in CN0 Papillary Thyroid Carcinoma: A Systematic Review and Meta-Analysis. PloS One (2015) 10(10):e0139021. doi: 10.1371/journal.pone.0139021
75. Shuai Y, Yue K, Duan Y, Zhou M, Fang Y, Liu J, et al. Surgical Extent of Central Lymph Node Dissection for Papillary Thyroid Carcinoma Located in the Isthmus: A Propensity Scoring Matched Study. Front Endocrinol (Lausanne) (2021) 12. doi: 10.3389/fendo.2021.620147
76. Liu J, Fan XF, Yang M, Huang LP. Analysis of the Risk Factors for Central Lymph Node Metastasis of Cn0 Papillary Thyroid Microcarcinoma: A Retrospective Study. Asian J Surg (2021) S1015-9584(21): 603–5. doi: 10.1016/j.asjsur.2021.09.014
77. Zetoune T, Keutgen X, Buitrago D, Aldailami H, Shao H, Mazumdar M, et al. Prophylactic Central Neck Dissection and Local Recurrence in Papillary Thyroid Cancer: A Meta-Analysis. Ann Surg Oncol (2010) 17(12):3287–93. doi: 10.1245/s10434-010-1137-6
78. Liu H, Li Y, Mao Y. Local Lymph Node Recurrence After Central Neck Dissection in Papillary Thyroid Cancers: A Meta Analysis. Eur Ann Otorhinolaryngol Head Neck Dis (2019) 136(6):481–7. doi: 10.1016/j.anorl.2018.07.010
79. Shim SR, Kitahara CM, Cha ES, Kim S-J, Bang YJ, Lee WJ. Cancer Risk After Radioactive Iodine Treatment for Hyperthyroidism: A Systematic Review and Meta-Analysis. JAMA Netw Open (2021) 4(9):e2125072. doi: 10.1001/jamanetworkopen.2021.25072
80. Leboulleux S, Bournaud C, Chougnet CN, Zerdoud S, Al Ghuzlan A, Catargi B, et al. Thyroidectomy Without Radioiodine in Patients With Low-Risk Thyroid Cancer. New Engl J Med (2022) 386(10):923–32. doi: 10.1056/NEJMoa2111953
81. Lamartina L, Leboulleux S, Terroir M, Hartl D, Schlumberger M. An Update on the Management of Low-Risk Differentiated Thyroid Cancer. Endocr Rel Cancer (2019) 26(11):R597–610. doi: 10.1530/ERC-19-0294
82. Iñiguez-Ariza NM, Brito JP. Management of Low-Risk Papillary Thyroid Cancer. Endocrinol Metab (Seoul) (2018) 33(2):185–94. doi: 10.3803/EnM.2018.33.2.185
83. Tuttle RM, Haugen B, Perrier ND. Updated American Joint Committee on Cancer/Tumor-Node-Metastasis Staging System for Differentiated and Anaplastic Thyroid Cancer (Eighth Edition): What Changed and Why? Thyroid (2017) 27(6):751–6. doi: 10.1089/thy.2017.0102
84. Lin B, Qiang W, Wenqi Z, Tianyu Y, Lina Z, Bin J. Clinical Response to Radioactive Iodine Therapy for Prophylactic Central Neck Dissection is Not Superior to Total Thyroidectomy Alone in Cn0 Patients With Papillary Thyroid Cancer. Nucl Med Commun (2017) 38(12):1036–40. doi: 10.1097/MNM.0000000000000756
Keywords: papillary thyroid cancer, preventive central lymph node dissection, hypocalcemia, recurrent laryngeal nerve paresis, metastasis, cancer recurrence
Citation: Dolidze DD, Shabunin AV, Mumladze RB, Vardanyan AV, Covantsev SD, Shulutko AM, Semikov VI, Isaev KM and Kazaryan AM (2022) A Narrative Review of Preventive Central Lymph Node Dissection in Patients With Papillary Thyroid Cancer - A Necessity or an Excess. Front. Oncol. 12:906695. doi: 10.3389/fonc.2022.906695
Received: 28 March 2022; Accepted: 03 June 2022;
Published: 29 June 2022.
Edited by:
Pasquale Cianci, Azienda Sanitaria Localedella Provincia di Barletta Andri Trani (ASL BT), ItalyReviewed by:
Altamura Amedeo, Pia Fondazione di Culto e Religione Card. G. Panico, ItalyGrazisa Scialandrone, Azienda Sanitaria Localedella Provincia di Barletta Andri Trani (ASL BT), Italy
Copyright © 2022 Dolidze, Shabunin, Mumladze, Vardanyan, Covantsev, Shulutko, Semikov, Isaev and Kazaryan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Airazat M. Kazaryan, a2F6YXJ5YW5AZ21haWwuY29t
 David D. Dolidze1,2
David D. Dolidze1,2 Serghei D. Covantsev
Serghei D. Covantsev Airazat M. Kazaryan
Airazat M. Kazaryan