
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Oncol. , 18 October 2022
Sec. Breast Cancer
Volume 12 - 2022 | https://doi.org/10.3389/fonc.2022.871786
This article is part of the Research Topic Dissecting Triple Negative Breast Cancer; Advances and Challenges From the Laboratory to the Clinic View all 8 articles
 Ying Zhong1†
Ying Zhong1† Xinyu Ren2†
Xinyu Ren2† Xi Cao1†
Xi Cao1† Yali Xu1
Yali Xu1 Yu Song1
Yu Song1 Yidong Zhou1
Yidong Zhou1 Feng Mao1
Feng Mao1 Songjie Shen1
Songjie Shen1 Zhe Wang1
Zhe Wang1 Qiang Sun1*
Qiang Sun1*Background: Immunotherapy plays an important role in the treatment of triple-negative breast cancer (TNBC). This study aimed to identify immune-related genes that are associated with the prognosis of patients with TNBC as possible targets of immunotherapy, alongside their related tumor-infiltrating lymphocytes (TILs).
Methods: The clinical data and gene expression profiles of patients with breast cancer were extracted from The Cancer Genome Atlas (TCGA) and Gene Expression Omnibus (GEO) databases and divided into training (n = 1,053) and verification (n = 508) groups. CIBERSORT was used to predict the differences in immune cell infiltration in patient subsets that were stratified according to risk. Gene Ontology (GO) enrichment analysis was used to identify pathways associated with immune-related genes in patient subsets that were stratified according to risk. The clinical data and insulin-like growth factor 2 receptor (IGF2R) expression profiles of patients with breast cancer were extracted from METABRIC. The expression of IGF2R and TILs were evaluated in a cohort containing 282 untreated patients with TNBC. The correlations of IGF2R expression, TILs, and clinicopathological parameters with patient prognosis were analyzed in the whole cohort.
Results: The prognostic model, which was composed of 26 immune-related gene pairs, significantly distinguished between high- and low-risk patients. Univariate and multivariate analyses indicated that the model was an independent prognostic factor for breast cancer. Among the identified genes, the expression of IGF2R significantly distinguished between high- and low-risk patients in TCGA (P = 0.008) and in METABRIC patients (P < 0.001). The expression of IGF2R was significantly associated with clinical risk factors such as TNBC, estrogen receptor (ER)–negative expression, human epidermal growth factor receptor 2 (HER2)–positive expression, and age ≤60 years old in METABRIC patients. In addition, the patients with IGF2R-positive expression had lower disease-free survival (DFS) rates than those with IGF2R-negative expression in the TNBC cohort (67.8% vs. 78.5%, P = 0.023). IGF2R expression also was significantly negatively correlated with TILs, particularly with CD8+ TILs and CD19+ TILs in the cohort of patients with TNBC.
Conclusion: IGF2R can be used as an indicator of a poor prognosis in patients with TNBC and as a potential target and research direction for TNBC immunotherapy in the future.
Breast cancer is the most serious malignant tumor threatening the health of women worldwide. It is the leading global cause of cancer deaths in women and remains incurable when it reaches an advanced stage (1). Approximately 3%–10% of patients with new breast cancer are diagnosed with distant metastasis (2). Metastatic breast cancer remains an almost incurable disease, with an overall survival (OS) period of approximately 3 years and a 5-year survival rate of approximately 25% (3).
Insulin-like growth factor 2 receptor (IGF2R) is a membrane-binding glycoprotein whose main function is transporting lysosomes from the trans-Golgi network to the lysosomes. It plays an important role in cell growth and survival, and its expression is closely related to tumors (4). IGF2R is also associated with a variety of malignancies that include cervical cancer (5), bladder cancer (6), osteosarcoma (7), and mucosal melanoma (8). The loss of IGF2R activity affects tumor growth, apoptosis, angiogenesis, and invasion (9). IGF2R plays an important role in clearing apoptotic cells to maintain the stability of tissue environments (10). IGF2R has been confirmed to be involved in latent transforming growth factor beta (TGFβ) activation in human fibroblasts (11). In addition, TGFβ family cytokines are involved in immune regulation, extracellular matrix synthesis, as well as the proliferation, differentiation, and development regulation of various types of cells (12). Moreover, IGF2R plays a key role in the survival of CD8+ T cells (13) and in the activation and differentiation of T cells (14). Furthermore, some immunological mechanisms and pathways controlled by IGF2R have been discovered (15). However, the role of IGF2R in the immunotherapy of patients with breast cancer requires further study.
Cytotoxic chemotherapy, has long been the main treatment for triple-negative breast cancer (TNBC), and TNBC is more likely to metastasize than other types of breast cancer (16). The proportion of tumor-infiltrating lymphocytes (TILs) in TNBC is much greater than that in hormone receptor (HR)–tumors, and the increased proportion of TILs indicates a better prognosis (17). The cytotoxic T-lymphocyteassociatedantigen 4 (CTLA-4) and programmed death 1 (PD-1)/programmed death ligand 1 (PD-L1) have been observed to block TILs and to promote tumor growth and progression (18). Meanwhile, immune checkpoint inhibitors (ICIs) have been used successfully in the treatment of cancer (19), and immunotherapy has become the first treatment choice for patients with TNBC diagnosed with PD-L1–positive tumors (20). Anti-trophoblast cell surface antigen 2 is an antibody–drug conjugate that has been demonstrated to improve the progression-free survival of patients with metastatic TNBC (21). The poly-ADP ribose polymerase (PARP) inhibitors olaparib and talazoparib have been used in patients with TNBC with the mutant breast cancer gene BRCA who were resistant to chemotherapy (22). In addition, chimeric antigen receptor-positive T cells have been observed to kill tumor endothelial cells and tumor endothelial marker-8–positive TNBC cells by secreting immune-stimulating cytokines, but the relevant research is still in the preclinical stage 23. For patients with TNBC, the clinical benefit of immunotherapy is limited and remains in the research stage. Consequently, tumor immunity needs to be better understood to identify additional immune biomarkers and potential therapeutic targets.
To address these issues, in the present study, we aimed to identify powerful biomarkers for the prediction of ICI responsiveness using data extracted from The Cancer Genome Atlas (TCGA), Gene Expression Omnibus (GEO), and METABRIC databases. We combined these data with those in the immunology database and analysis portal ImmPort to investigate the relevant molecular mechanisms and immune cell relationships. Furthermore, from the breast cancer database of Peking Union Medical College Hospital, we identified the relationships between IGF2R expression and the clinical characteristics of TILs.
This was a retrospective study of the gene expression and the corresponding clinical data of patients included in two independent datasets obtained from publicly available databases. In total, the data from 1,561 patients were analyzed. The expression of 56,737 genes and the survival outcome data of 1,053 patients were obtained from TCGA (https://portal.gdc.cancer.gov/repository). Data on gene expression and DFS of 508 patients were retrieved from the GEO database (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE25066). The expression of the IGF2R gene and the clinical characteristics and survival outcomes of 1,818 patients were acquired from the METABRIC database (http://www.cbioportal.org/datasets).
To construct a prognostic model based on immune-related genes, 2,498 immune-related genes were obtained from the ImmPort database (https://www.immport.org/home) on 30 May 2020. This gene platform includes a list of immunologically relevant genes, curated with functions and Gene Ontology (GO) terms. The ImmuneRegulation web-based tool identified regulators of immune system-specific genes of interest, and the Immcantation framework analyzed high-throughput adaptive immune receptor repertoire sequencing datasets characterizing B-cell and T-cell receptors. In this study, we retained only immune-related genes that were identified in both the GEO and TCGA datasets with a median absolute deviation of >0.5 (24). The relative expression within each immune-related gene pair was compared for each patient in the TCGA dataset. In each pair, if the expression of one gene was larger than that of the other, then the value of the gene pair was considered to be 1; otherwise, the value was considered to be 0. After removing immune-related gene pairs with relatively small variations in expression within the pair (<20%), least absolute shrinkage and selection operator (Lasso) regression was performed for 1,000 simulations, and a prognostic model containing 26 immune-related gene pairs was obtained. This model was used to calculate the risk value of each patient in the TCGA dataset. A receiver operating characteristic curve was established using the risk values, and an optimal cutoff value was determined to distinguish between the low- and high-risk patients.
To further verify the prognostic model based on immune-related gene pairs, the GEO dataset was used as the validation group. The risk value of each patient in the GEO dataset was calculated using the model, and the cutoff value obtained for the training group was used to stratify the GEO patients into high- and low-risk groups. Univariate and multivariate Cox proportional hazards analyses were used to verify whether the model could be used as an independent prognostic factor relative to other clinical features such as age, HR expression, HER2 expression, and American Joint Committee on Cancer (AJCC) stage in the GEO and TCGA datasets.
The CIBERSORT algorithm was used to estimate differences in immune cell infiltration using gene expression data in the high- and low-risk TCGA groups (25). This algorithm uses gene expression data to predict the proportions of 22 types of tumor-infiltrating immune cells, such as T cells, B cells, macrophages, and natural killer cells.
Enrichment analysis of the identified immune-related genes was performed using g:Profiler (26). All GO gene sets were downloaded from the Gene Set Enrichment Analysis website (https://www.gsea-msigdb.org/gsea/index.jsp). Gene sets in the high- and low-risk TCGA groups were compared using the Bioconductor “fgsea” package in R. After 10,000 cycles, significant enrichment pathways were obtained and sequenced. Gene sets with statistical significance were selected with a false discovery rate–adjusted P < 0.05.
The tumor specimens from 282 patients with TNBC at stages I–III were collected. These patients received surgical treatment in our hospital between 2011 and 2014. Patients with stage IV TNBC and patients who received neoadjuvant chemotherapy were excluded. The formalin-fixed paraffin-embedded tumor specimens of these patients were made into tumor microarrays (TMAs). When constructing the TMAs, each tumor specimen included the epithelial components and the tumor stroma after hematoxylin and eosin (HE) staining. The median follow-up time was 69 months (1–104 months). All sections of the TMAs were stained with IGF2R, CD8, and CD19 antibodies, and the frequencies of TILs were evaluated according to the publication A Practical Review for Pathologists and Proposal (27). IGF2R expression was detected by a rabbit monoclonal antibody (#15128, Cell Signaling Technology; dilution, 1:50). CD8 expression was detected by 4B11 (PA0183, prediluted; Leica Microsystems, Shanghai, China). CD19 expression was detected by EP169 (ZA-0569, prediluted; Zhongshan Golden Bridge Biotechnology Co. Ltd., Beijing, China). Two pathologists reviewed all of the samples and scored the immunohistochemical staining independently. The expression of IGF2R was determined by histochemical scoring (H-score), with consideration of the staining intensity and the percentage of positive cancer cells (28). An H-score of 0–49 was classified as the negative group, whereas H-scores of 50–99, 100–199, and 200–300 were classified as 1+, 2+, and 3+, respectively. An H-score classification of 1+, 2+, or 3+ was classified as the positive group. TILs were divided into a low group and a high group, with a median of 5%. CD8+ TILs were also divided into a low group and a high group, with a median of 10%. Finally, CD19+ TILs were divided into a low group and a high group, with a median of 1%. Other pathological features of 282 patients, such as tumor stage, lymph node (LN), tumor grade, and Ki-67, were retrieved from the pathological report of the Beijing Union Medical College Hospital.
Statistical analyses were performed using version 3.6.3 of the R Statistical Software and SPSS 23.0. Comparisons of genes between groups were performed using a t-test. The Kaplan–Meier method and the “survival” package in R were used for survival analysis. Cox proportional hazards regression analysis was used for univariate and multivariate analyses of OS or DFS. The Wilcoxon test was used to compare differences in immune cell infiltration. The chi-squared test was used to compare TILs, CD8+ TILs, and CD19+ TILs, and P < 0.05 was considered statistically significant. Statistical differences were recorded as follows: *P < 0.05, **P < 0.01, and ***P < 0.001.
A total of 56,735 genes and 2,498 unique immune-related genes were obtained from the TCGA and ImmPort databases, respectively. Among them, 1,653 immune-related genes were included in the data obtained from both databases. Then, the immune-related genes from ImmPort and the genes obtained from the GEO dataset were intersected to locate the same genes. Among 606 common immune-related genes, 31,896 immune-related gene pairs were found after removing gene pairs with relatively small internal variations. The immune-related gene pairs from the TCGA dataset were combined with the corresponding clinical data, revealing 69 immune-related gene pairs that were significantly associated with the patient prognosis. Next, the Lasso method for Cox proportional hazards regression analysis was used to construct the prognostic model based on immune-related gene pairs for the training group. Finally, 26 immune-related gene pairs comprising 43 immune-related genes were selected in the model (Table 1).
The prognostic model significantly distinguished between high- and low-risk patients in terms of their OS in the TCGA dataset; the OS of the high-risk patients was significantly shorter than that of the low-risk patients (Figure 1A, P < 0.001). To verify the predictive value of the prognostic model based on immune-related gene pairs, we applied the model to the GEO dataset and stratified the patients into high- and low-risk groups. The DFS values of the two validation groups were similar to those of the training groups; the DFS of the high-risk group was significantly less than that of the low-risk group (Figure 1B, P = 0.026). Next, univariate and multivariate Cox proportional hazards regression analyses were used to study the corresponding clinical data in the TCGA dataset. The prognostic model based on immune-related gene pairs and the AJCC stage were determined to be independent prognostic factors in the TCGA dataset (Supplementary Figures 1A, B). In addition, univariate and multivariate Cox proportional hazards regression analyses were used to analyze the corresponding clinical data in the GEO dataset (Supplementary Figures 1C, D). The prognostic model based on immune-related gene pairs was determined to be an independent prognostic factor in the GEO dataset, according to the univariate Cox proportional hazards regression analysis (Supplementary Figure 1C).
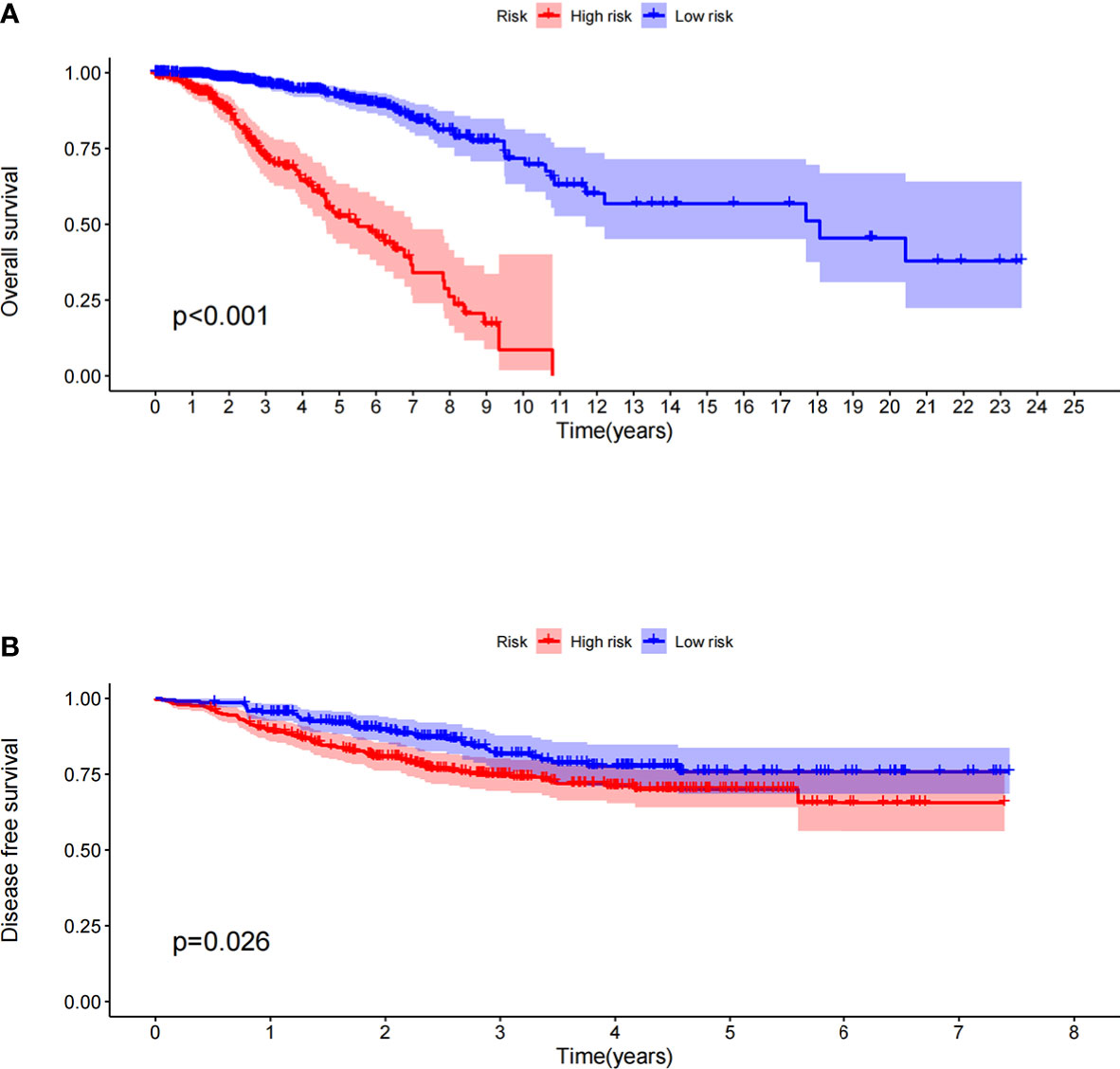
Figure 1 (A) Overall survival between the high- and low-risk patients in the TCGA datasets. (B) Disease-free survival between the high- and low-risk patients in the GEO datasets.
CIBERSORT, which has been applied to many tumor microenvironments (29), was used to predict the infiltration of 21 different immune cell types in the high- and low-risk TCGA groups (Supplementary Figure 2A), including M0 and M2 macrophages, CD8+ T cells, and resting dendritic cells. M0 (Supplementary Figure 2B, P < 0.001) and M2 (Supplementary Figure 2C, P < 0.001) macrophages were highly expressed in the high-risk group, whereas CD8+ T cells (Supplementary Figure 2D, P < 0.001) and naive B cells (Supplementary Figure 2E, P < 0.001) were highly expressed in the low-risk group.
To determine the biological processes and signaling pathways associated with the immune-related gene pairs in the prognostic model, GO enrichment was used to analyze the identified immune-related genes in the TCGA dataset, and pathways with significant differences between high- and low-risk patients were detected (Supplementary Figure 3). CCR chemokine receptor binding, regulation of leukocyte-mediated cytotoxicity, T-cell migration, T-cell receptor complex, and other pathways were determined to be significantly enriched in low-risk patients (Supplementary Figures 4A–F). The enrichment of these pathways in low-risk patients confirmed the importance of immune cells in the treatment and prognosis of patients with breast cancer.
Of the 1,098 patients with breast cancer in the TCGA dataset, those with a higher level of IGF2R had a lower OS compared with those with a lower level of IGF2R (Figure 2A, P < 0.001). In the METABRIC dataset including 1,818 patients with breast cancer, the OS was lower in the patients with a higher IGF2R expression level compared with the patients with a lower IGF2R expression level (Figure 2B, P = 0.008). Of the patients included in the METABRIC dataset, IGF2R expression was greater in the patients with TNBC vs. the other patients (Figure 3A, P < 0.001), in the ER-negative patients vs. the ER-positive patients (Figure 3B, P < 0.001), in the HER2-positive patients vs. the HER2-negative patients (Figure 3C, P < 0.001), in the patients aged ≤60 years old vs. those aged >60 years old (Figure 3D, P < 0.001), and in patients that had undergone chemotherapy vs. those without chemotherapy (Figure 3E, P < 0.001).
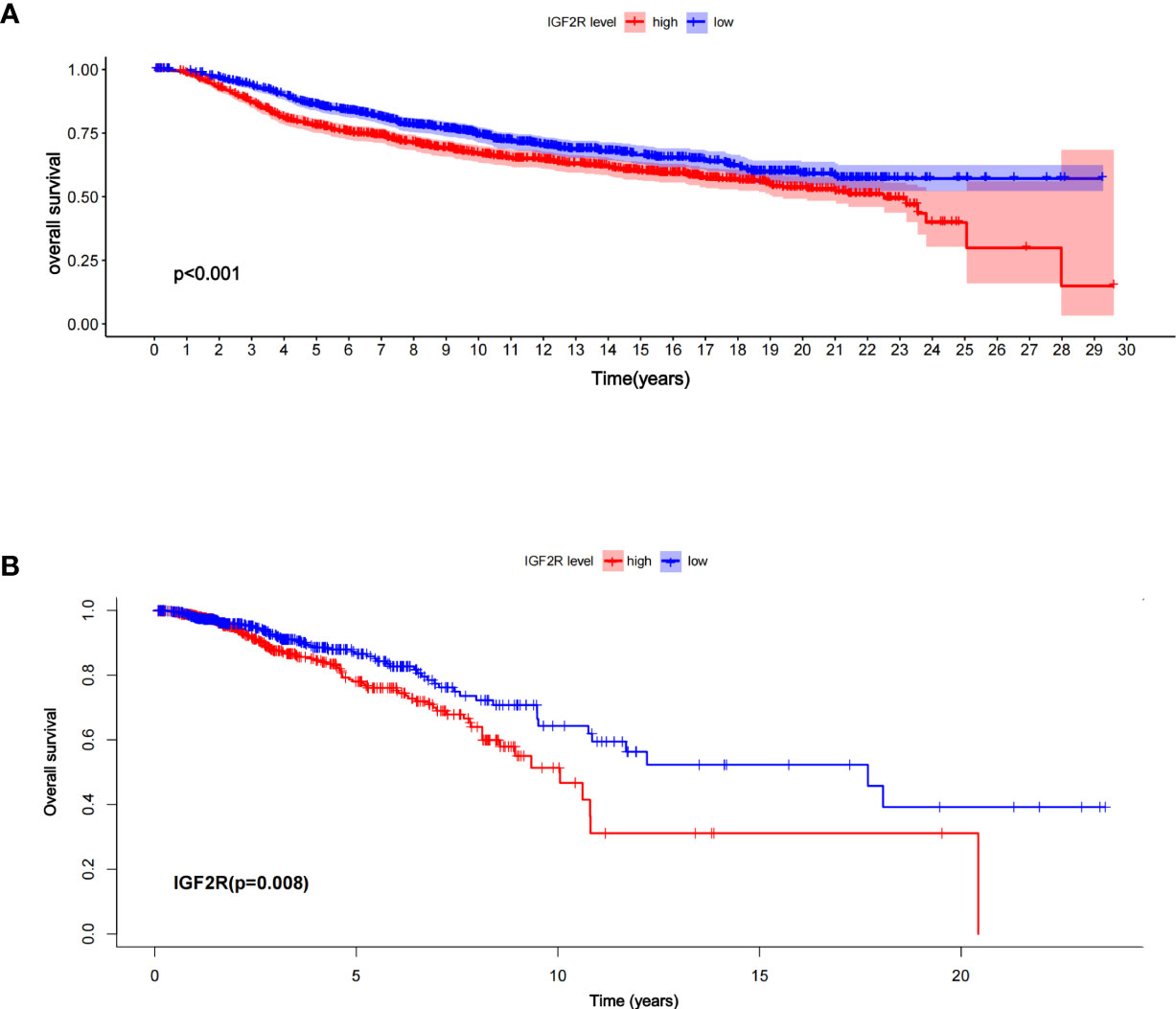
Figure 2 Overall survival between the high- and low-risk patients in (A) the TCGA datasets and (B) the METABRIC, according to IGF2R expression.
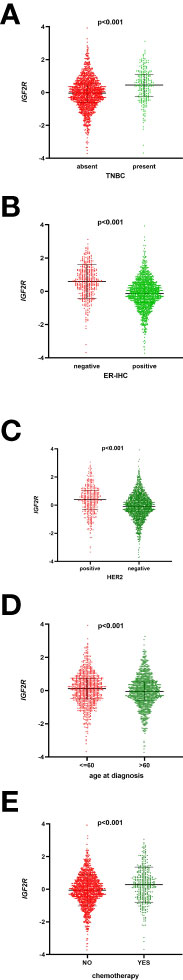
Figure 3 IGF2R expression with other clinical characteristics in the METABRIC datasets. (A) IGF2R expression in patients with TNBC and in other patients. (B) IGF2R expression in ER-negative patients and ER-positive patients. (C) IGF2R expression in HER2-positive patients and HER2-negative patients. (D) IGF2R expression in patients aged ≤60 years old and in patients aged >60 years old. (E) IGF2R expression in patients with chemotherapy and in patients without chemotherapy.
IGF2R staining was evaluated in a cohort of 282 patients with TNBC. There were 159 (56.4%) patients with an H-score of 0, 63 (22.3%) patients with an H-score of 1+, 54 (19.1%) patients with an H-score of 2+, and six (2.1%) patients with an H-score of 3+ (Figure 4). The 4-year DFS of IGF2R-positive TNBC patients was lower compared with that of IGF2R-negative patients (Figure 5; 67.8% vs. 78.5%, P = 0.023). IGF2R expression independently predicted the DFS in univariate and multivariate Cox proportional hazards regression analyses (Figure 6A, P = 0.025; Figure 6B, P = 0.026), whereas the AJCC stage, LN−/+, T-stage, Ki-67, grade, and menopause status of patients did not predict the DFS.
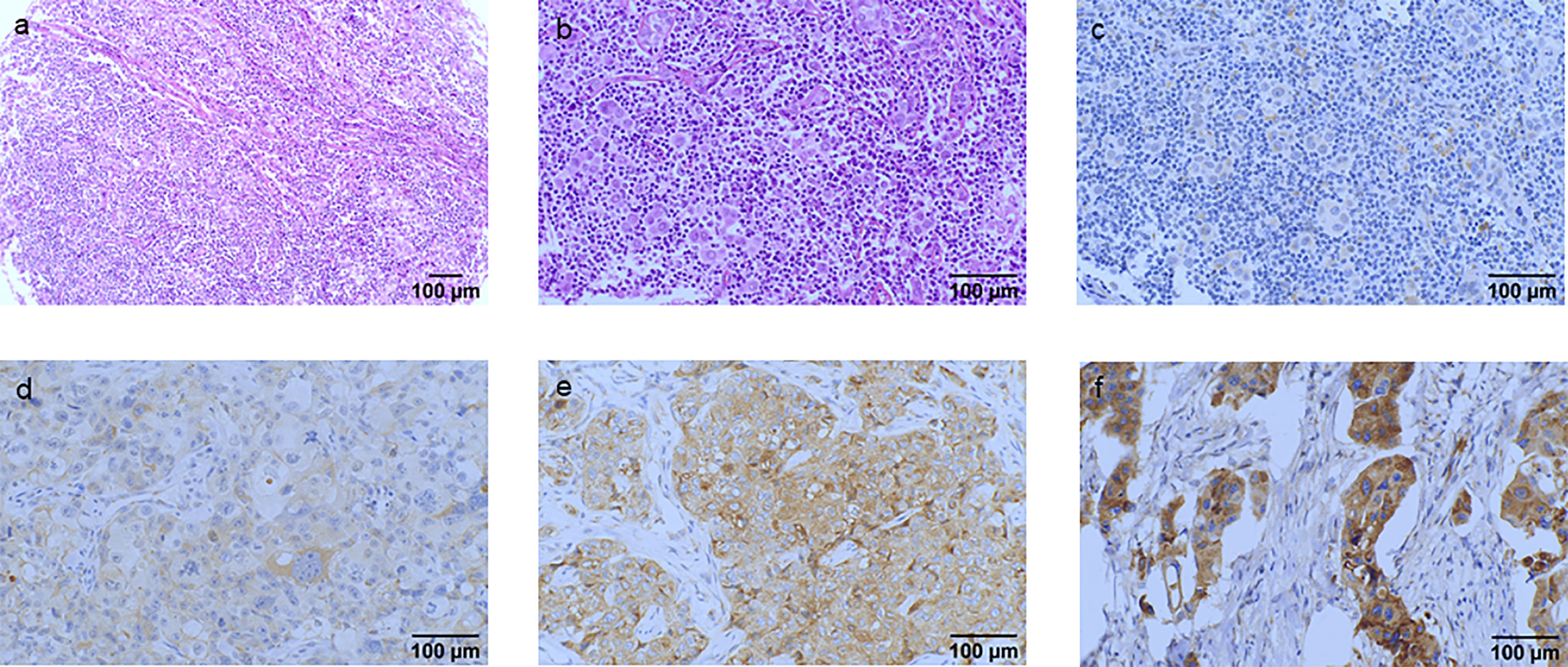
Figure 4 Hematoxylin and eosin (HE) staining and classification of IGF2R expression by the H-score in patients with TNBC. (A) HE staining of the tumor cells (original magnification, ×100). (B) HE staining of the tumor cells (original magnification, ×200). (C) IGF2R-negative expression on tumor cells (original magnification, ×200). (D) IGF2R 1+ expression on tumor cells (original magnification, ×200). (E) IGF2R 2+ expression on tumor cells (original magnification, ×200). (F) IGF2R 3+ expression on tumor cells (original magnification, ×200).
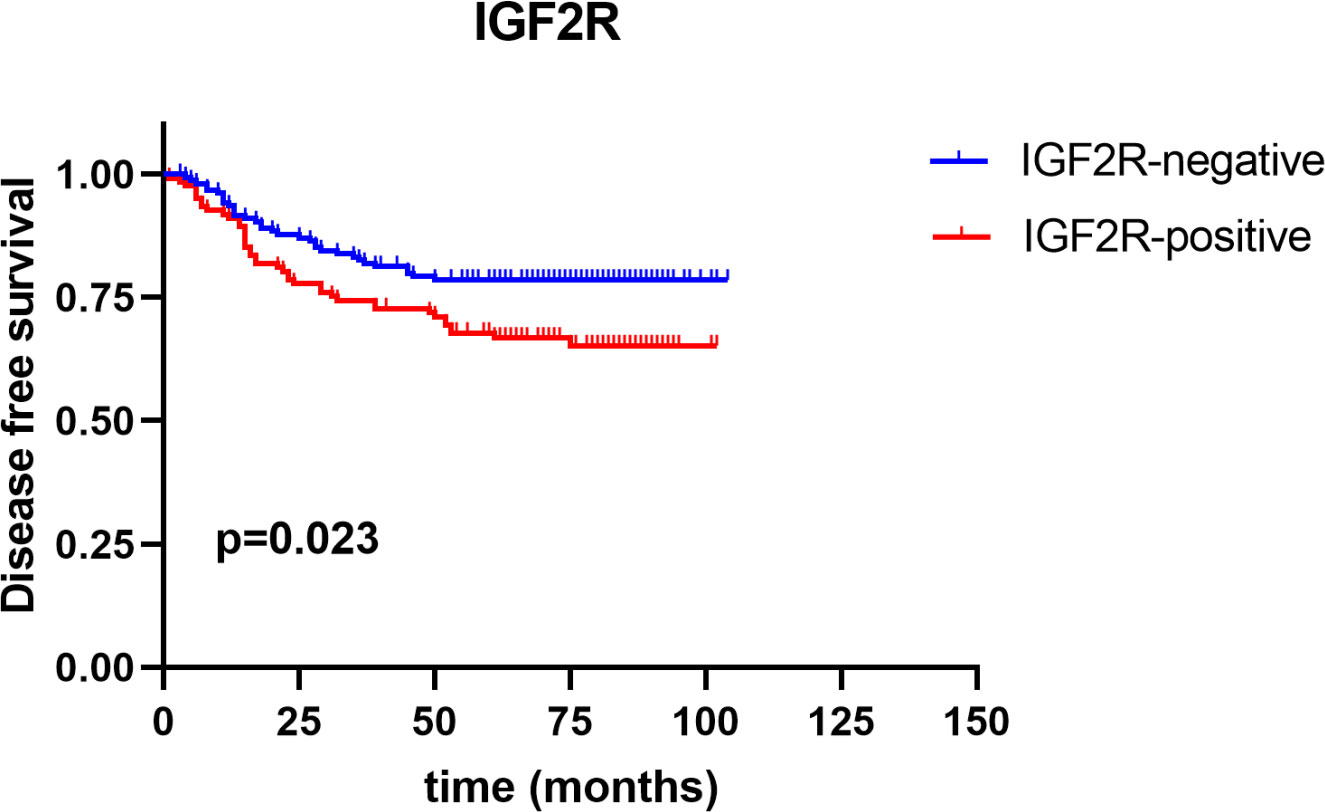
Figure 5 Disease-free survival between IGF2R-positive patients and IGF2R-negative patients in the TNBC cohort.
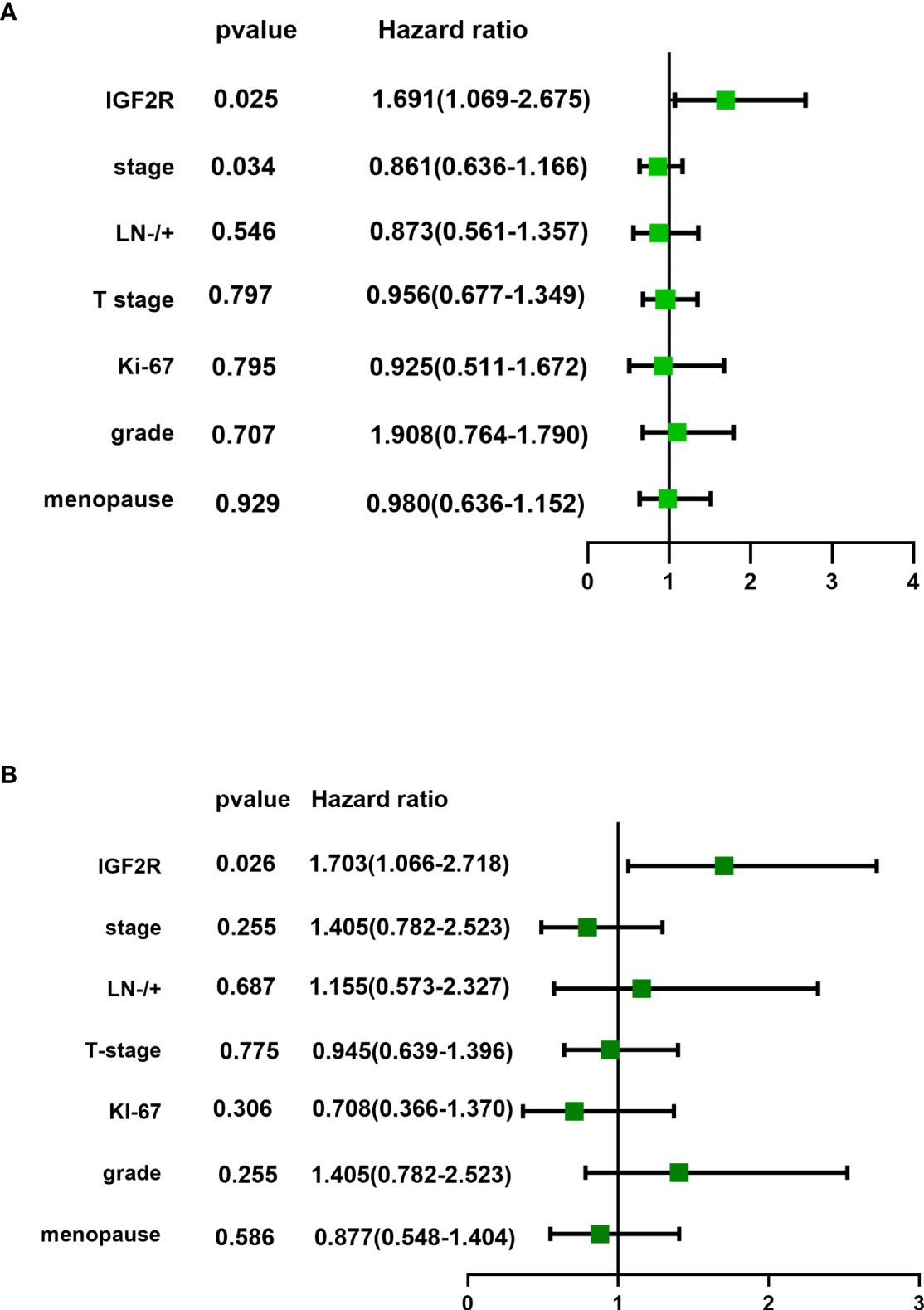
Figure 6 (A) Univariate analysis and (B) multivariate analysis of IFG2R expression and clinical characteristics for patients in the TNBC cohort.
TILs, CD8+ TILs, and CD19+ TILs were detected in the 282 patients with TNBC. High frequencies of TILs, CD8+ TILs, and CD19+ TILs were observed in 97.0%, 44.3%, and 63.5% of patients with TNBC. The percentages of TILs, CD8+ TILs, CD19+ TILs, and IGF2R expression were analyzed (Figure 7). Patients with TNBC with IGF2R-positive expression had lower frequencies of TILs compared with patients with IGF2R-negative expression (Figure 8A, P = 0.046). Moreover, patients with TNBC with IGF2R-positive expression had lower densities of CD8+ TILs and CD19+ TILs compared with those with IGF2R-negative expression (Figures 8B, C, P = 0.031 and P = 0.05).
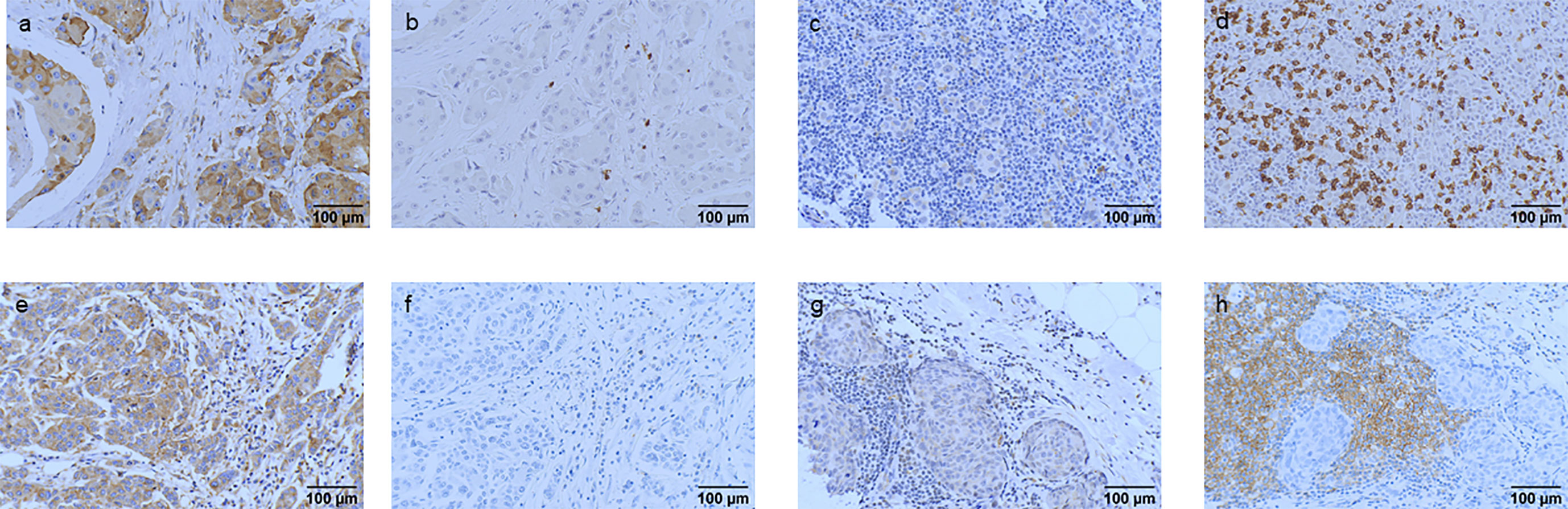
Figure 7 IGF2R expression association with CD8+ TILs and CD19+ TILs. (A, B) The patients with IGF2R-positive expression and a low percentage of CD8+ TILs. (C, D) The patients with IGF2R-negative expression and a high percentage of CD8+ TILs. (E, F) The patients with IGF2R-positive expression and a low percentage of CD19+ TILs. (G, H) The patients with IGF2R-negative expression and a high percentage of CD19+ TILs.
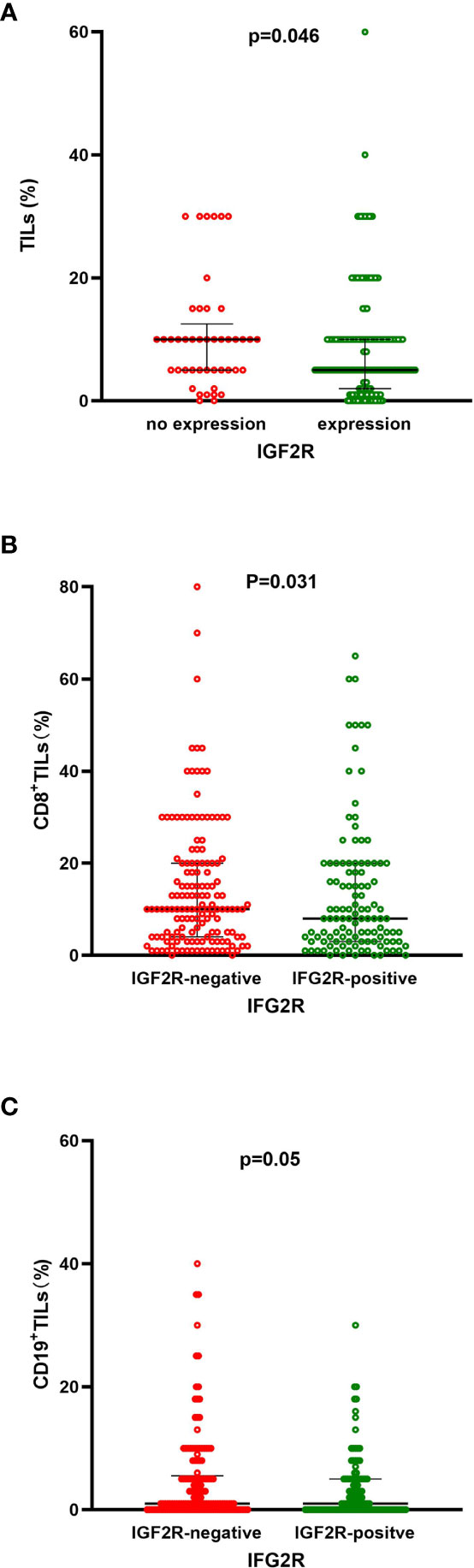
Figure 8 (A) Frequencies of TILs between IGF2R-negative expression and IGF2R-positive expression patients in the TNBC cohort. (B) Frequencies of CD8+ TILs between IGF2R-negative expression and IGF2R-positive expression patients in the TNBC cohort. (C) Frequencies of CD19+ TILs between IGF2R-negative expression and IGF2R-positive expression patients in the TNBC cohort.
In the current study, we demonstrated that a prognostic model constructed with 26 immune-related gene pairs from 43 independent immune-related genes predicted the OS and DFS of patients with breast cancer. Relative sequencing and pairing of genes to create a prognostic model based on immune-related gene pairs have provided reliable results for many types of tumors (30, 31). The prognostic model based on immune-related gene pairs was an independent prognostic factor in the TCGA dataset. The findings from this study revealed that M2 and M0 macrophages were highly expressed in high-risk patients with breast cancer, whereas CD8+ T cells and naive B cells were highly expressed in low-risk patients with breast cancer. In this study, the identified immune-related genes were associated with multiple pathways related to immune cell infiltration, migration, and immune checkpoint enhancement. Among them, regulation of the leukocyte-mediated cytotoxicity pathway is associated with tumor progression and decreased CD8+ infiltration in pancreatic cancer (32). In addition, the T-cell migration pathway has been demonstrated to enhance tumor immunity and increase the efficacy of ICIs in preclinical breast cancer models (33). This pathway can increase the number of T cells in tumors and intratumoral T-cell diversity (34).
Among 43 independent immune-related genes, IGF2R was selected for further research. We found that a high level of IGF2R expression was significantly associated with a poor prognosis in patients included in the TCGA and METABRIC databases. In addition, we observed that IGF2R was closely related with poor clinical characteristics, such as TNBC, premenopause, ER-negative expression, and chemotherapy. Furthermore, we identified that the expression level of IGF2R was significantly associated with a poor prognosis and lower frequencies of TILs and CD8+ TILs in the cohort of patients with TNBC.
IGF2R is a growth inhibitory factor 35. IGF2R deletion or mutation may contribute to the development and progression of cancer 36. The deletion of the IGF2R allele has been shown to be an early event in the etiology of breast cancer (36) as a tumor suppressor (37). In addition, low levels of IGF2R have been associated with a poor prognosis in patients with breast cancer (38). However, IGF2R is overexpressed in HR-negative breast cancer (39). Moreover, IGF2R plays a central role in the differentiation of TNBC subsets. The IGF receptor family also has been related to tumor differentiation and the prognosis of patients with TNBC (40). In fact, IGF2R has been demonstrated to be an unfavorable prognostic factor for patients with ER-negative breast cancer (41). In addition, luminal A and luminal B patients with a high expression of IGF1R and a low expression of IGF2R had significantly higher survival rates than patients with other types of breast cancer (39). Overexpression of IGF2R also has been shown to significantly increase the migration and invasion of MDA-MB-231 cells (42). The level of IGF2R mRNA in MDA-MB-231 cells has been determined to be higher than that in MCF-7 cells (43). Likewise, our study observed that a high expression of IGF2R was associated with a poor prognosis of patients with TNBC.
The development of breast cancer is characterized by an increased infiltration of immune cells in the parenchyma and stroma of a tumor (44). It has been demonstrated that stromal infiltrating lymphocytes (sTILs) have predictive and prognostic value for TNBC and that high percentages of sTILs indicate a better prognosis (44). CD8+ lymphocyte infiltration has been shown to be an independent favorable prognostic indicator in TNBC (45), and a high CD8+ T-cell score is associated with better survival rates in patients with TNBC (46). Improved outcomes of atezolizumab have been observed only in CD8+ and sTILs+ patients (47). The PARP inhibitor olaparib induces CD8+ T-cell infiltration through activation of the cyclic GMP-AMP synthase/stimulator of interferon genes pathway, and CD8+ T-cell depletion severely compromises antitumor efficacy (48). Immunotherapy methods, including PD-1/PD-L1 blocking and chimeric antigen receptor T-cell therapy, have been shown to improve antitumor activity through the proliferation of CD8 + T cells (49). Although PD-1/PD-L1 blocking has triggered great progress in the treatment of TNBC, the benefits are still limited (50–52). Therefore, more immunological targets and therapies through TILs need to be explored to improve the survival rates of patients with TNBC.
The IGF2R polyclonal antibody has been demonstrated to induce the blockade of T-cell differentiation at the CD8− stage and decrease the percentage of CD8+ cells (53). In addition, it has been shown that IGF2R and CD8+ T cells coexist in transplanted hearts and are involved in acute cellular rejection (54). Moreover, the enhancement of IGF2R expression has been revealed to increase apoptosis in CD8α+ dendritic cells, with a consequent reduction in the expression of interleukin (IL)-12 and interferon (IFN)-γ (55), while IL-12 and IFN-α/β provide signal support for CD8+ T memory programming (56). Furthermore, IGF2R is involved in the activation of TGFβ (57). TGFβ enhancement inhibits the proliferation of regulatory T cells (58), decreases the CD8+ T effector cell penetration in tumors (59), and suppresses the immune response (60). This may explain our findings, which show that patients with TNBC with a high expression of IGF2R had a low density of TILs and a low density of CD8+ TILs, thus promoting immune escape and leading to a poor prognosis for these patients.
CD19 is the common therapeutic target of hematological malignancies (61). Anti-CD19 chimeric antigen receptor T-cell therapy directed against B-cell lymphoma has been demonstrated to be efficacious. However, efforts to utilize this approach for breast cancer have delivered only modest improvements (62). The frequencies of CD19+ B cells in breast cancer are greater than those in normal tissues (63). Compared with fibroadenoma, the density of CD19+ B cells in breast cancer is greater and is significantly associated with higher tumor grades and an ER-negative status (64). CD19 is also highly expressed in patients with high-risk breast cancer (65). CD19+ B lymphocytes play an important role in breast cancer through PD-L1 in immune suppression and tumor escape (64). CD19+CD25+ regulatory B cells inhibit TILs and are closely related to the metastasis of breast cancer (66). These studies suggest that CD19+ B cells are a feature of patients with breast cancer with a poor prognosis. In our research, we found that a high percentage of CD19+ TILs was associated with a low expression of IGF2R, whereas a low expression of IGF2R was correlated with a high percentage of TILs. Therefore, we speculate that IGF2R may cause breast cancer immune escape through CD19+ TILs. Future targeting of IGF2R may promote the proliferation of CD19+ TILs by reducing the expression of inflammatory factors, thereby inhibiting the progression of TNBC.
IGF2R inhibits the proliferation of T cells and the infiltration of T cells in a tumor by TGFβ activation. In addition, IGF2R has been shown to promote the secretion of IL-10 by B cells (67), and IL-10 directly activates or expands T cells in a tumor (68). However, IL-10 promotes the depletion of CD8+ T cells in vivo (69) and inhibits the activity of CD8+ T cells (70). The invasion of IL-10 has been demonstrated to activate CD19+ B cells in the pathogen (71). Whether or not IGF2R inhibits the recruitment of TILs through some pathways or cytokines and whether it promotes the depletion of TILs in the tumor might be a direction for future research related to TNBC. An IGF2R inhibitor might be one of the target drugs for the treatment of TNBC in the future.
In summary, by analyzing patients from the TCGA and GEO databases, we found that IGF2R is a gene that is associated with a poor prognosis in patients with breast cancer. In the METABRIC database, the expression of IGF2R distinguished patients with breast cancer with a poor prognosis from those with a more favorable prognosis and was highly expressed in patients with TNBC. Patients with a high expression of IGF2R had a poor prognosis, and a high IGF2R expression was negatively correlated with TILs, CD8+ TILs, and CD19+ TILs in the TNBC cohort. The datasets originated from retrospective studies including patients without ICI therapies. Thus, our prognostic model and IGF2R expression must be more widely validated in prospective cohort studies. In addition, the association of IGF2R, CD8+ TILs, and CD19+ TILs as well as their related immune factors and molecular mechanisms require further verification in vitro and in vivo.
We established a prognostic model based on immunogenomics that reliably predicted the prognosis of patients with breast cancer. We identified that the immune-related gene IGF2R may play an important role in the treatment of TNBC in the future and may provide new targets for immunotherapy. Furthermore, we demonstrated that CD8+ TILs and CD19+ TILs were highly expressed in patients with TNBC with a low expression of IGF2R. Targeting CD8+ TILs and CD19+ TILs combined with IGF2R expression should be investigated in future TNBC treatment research.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
The studies involving human participants were reviewed and approved by Human Research of Peking Union Medical College Hospital. The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
YZ collected and analyzed clinical and gene data. Experiments were performed by YZ, XR, and XC. YX, YDZ, FM, SS, and ZW collected and evaluated the TNBC cohort. YZ, YS, and ZW conducted statistical analysis. QS supported quality management and directed the team. All authors contributed to the article and approved the submitted version.
This study was supported by the CAMS Innovation Fund for Medical Sciences (CIFMS) (2021-I2M-C&T-B-017).
The authors would like to thank the TCGA, GEO, GO, and METABRIC databases and the Department of Pathology of Peking Union Medical College Hospital for the availability of the data. The authors would also like to thank all of the patients who participated in this study.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2022.871786/full#supplementary-material
Supplementary Figure 1 | (A) Univariate analysis and (B) multivariate analysis of the prognostic model based on immune-related gene pairs for patients in the TCGA datasets. (C) Univariate analysis and (D) multivariate analysis of the prognostic model based on immune-related gene pairs for patients in the GEO datasets.
Supplementary Figure 2 | (A) Infiltration of 21 different immune cell types, (B) M0 macrophage infiltration, (C) M2 macrophage infiltration, (D) CD8+ T cell infiltration, and (E) naive B cell infiltration in the high- and low-risk groups by the prognostic model based on immune-related gene pairs in the TCGA datasets.
Supplementary Figure 3 | Pathways identified with significant differences between high- and low-risk groups by the prognostic model based on immune-related gene pairs in the TCGA datasets.
Supplementary Figure 4 | (A) The CCR chemokine receptor binding pathway was significantly enriched in low-risk patients by the prognostic model based on immune-related gene pairs. (B) The regulation of the leukocyte-mediated cytotoxicity pathway was significantly enriched in low-risk patients by the prognostic model based on immune-related gene pairs. (C) The T cell migration pathway was significantly enriched in low-risk patients by the prognostic model based on immune-related gene pairs. (D) The T cell receptor complex pathway was significantly enriched in low-risk patients by the prognostic model based on immune-related gene pairs. (E) The cytokine receptor binding pathway was significantly enriched in low-risk patients by the prognostic model based on immune-related gene pairs. (F) The response to the chemokine pathway was significantly enriched in low-risk patients by the prognostic model based on immune-related gene pairs.
1. Lord SJ, Kiely BE, Pearson SA, Daniels B, O'Connell DL, Beith J, et al. Metastatic breast cancer incidence, site and survival in Australia, 2001-2016: A population-based health record linkage study protocol. BMJ Open (2019) 9:e026414. doi: 10.1136/bmjopen-2018-026414
2. Gonzalez-Angulo AM, Morales-Vasquez F, Hortobagyi GN. Overview of resistance to systemic therapy in patients with breast cancer. Adv Exp Med Biol (2007) 608:1–22. doi: 10.1007/978-0-387-74039-3_1
3. Cardoso F, Spence D, Mertz S, Corneliussen-James D, Sabelko K, Gralow J, et al. Global analysis of advanced/metastatic breast cancer: Decade report (2005-2015). Breast (Edinburgh Scotland) (2018) 39:131–8. doi: 10.1016/j.breast.2018.03.002
4. Brown J, Jones EY, Forbes BE. Interactions of IGF-II with the IGF2R/cation-independent mannose-6-phosphate receptor mechanism and biological outcomes. Vitam. Hormones (2009) 80:699–719. doi: 10.1016/s0083-6729(08)00625-0
5. Takeda T, Komatsu M, Chiwaki F, Komatsuzaki R, Nakamura K., Tsuji K, et al. Upregulation of IGF2R evades lysosomal dysfunction-induced apoptosis of cervical cancer cells via transport of cathepsins. Cell Death Dis (2019) 10:876. doi: 10.1038/s41419-019-2117-9
6. Zhang Z, Mou Z, Xu C, Wu S, Dai X, Chen X, et al. Autophagy-associated circular RNA hsa_circ_0007813 modulates human bladder cancer progression via hsa-miR-361-3p/IGF2R regulation. Cell Death Dis (2021) 12:778. doi: 10.1038/s41419-021-04053-4
7. Karkare S, Allen KJH, Jiao R, Malo ME, Dawicki W, Helal M, et al. Detection and targeting insulin growth factor receptor type 2 (IGF2R) in osteosarcoma PDX in mouse models and in canine osteosarcoma tumors. Sci Rep (2019) 9:11476. doi: 10.1038/s41598-019-47808-y
8. Wróblewska JP, Dias-Santagata D, Ustaszewski A, Wu CL, Fujimoto M, Selim MA, et al. Prognostic roles of BRAF, KIT, NRAS, IGF2R and SF3B1 mutations in mucosal melanomas. Cells (2021) 10:2216. doi: 10.3390/cells10092216
9. Gemma A, Uematsu K, Hagiwara K, Takenoshita S, Kudoh S. [Mechanism of resistance to growth inhibition by transforming growth factor-beta 1 (TGF-beta 1) in primary lung cancer and new molecular targets in therapy]. Gan to kagaku ryoho. Cancer chemother. (2000) 27:1253–9.
10. Ohradanova-Repic A, Machacek C, Donner C, Mühlgrabner V, Petrovčíková E, Zahradníková A Jr, et al. The mannose 6-phosphate/insulin-like growth factor 2 receptor mediates plasminogen-induced efferocytosis. J Leukocyte Biol (2019) 105:519–30. doi: 10.1002/jlb.1ab0417-160rr
11. Leksa V, Godar S, Schiller HB, Fuertbauer E, Muhammad A, Slezakova K, et al. TGF-beta-induced apoptosis in endothelial cells mediated by M6P/IGFII-r and mini-plasminogen. J Cell Sci (2005) 118:4577–86. doi: 10.1242/jcs.02587
12. Massagué J. The transforming growth factor-beta family. Annu Rev Cell Biol (1990) 6:597–641. doi: 10.1146/annurev.cb.06.110190.003121
13. Ahmed KA, Wang L, Griebel P, Mousseau DD, Xiang J. Differential expression of mannose-6-phosphate receptor regulates T cell contraction. J leukocyte Biol (2015) 98:313–8. doi: 10.1189/jlb.2HI0215-049RR
14. Pfisterer K, Forster F, Paster W, Supper V, Ohradanova-Repic A, Eckerstorfer P, et al. The late endosomal transporter CD222 directs the spatial distribution and activity of lck. J Immunol (2014) 193:2718–32. doi: 10.4049/jimmunol.1303349
15. Leksa V, Ilková A, Vičíková K, Stockinger H. Unravelling novel functions of the endosomal transporter mannose 6-phosphate/insulin-like growth factor receptor (CD222) in health and disease: An emerging regulator of the immune system. Immunol Lett (2017) 190:194–200. doi: 10.1016/j.imlet.2017.08.011
16. Newton EE, Mueller LE, Treadwell SM, Morris CA, Machado HL. Molecular targets of triple-negative breast cancer: Where do we stand? Cancers (2022) 14:482. doi: 10.3390/cancers14030482
17. Dieci MV, Mathieu MC, Guarneri V, Conte P, Delaloge S, Andre F, et al. Prognostic and predictive value of tumor-infiltrating lymphocytes in two phase III randomized adjuvant breast cancer trials. Ann Oncol (2015) 26:1698–704. doi: 10.1093/annonc/mdv239
18. Buchbinder EI, Desai A. CTLA-4 and PD-1 pathways: Similarities, differences, and implications of their inhibition. Am J Clin Oncol (2016) 39:98–106. doi: 10.1097/coc.0000000000000239
19. Nathan MR, Schmid P. The emerging world of breast cancer immunotherapy. Breast (2018) 37:200–6. doi: 10.1016/j.breast.2017.05.013
20. Schmid P, Adams S, Rugo HS, Schneeweiss A, Barrios CH, Iwata H, et al. Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N Engl J Med (2018) 379:2108–21. doi: 10.1056/NEJMoa1809615
21. Bardia A, Mayer IA, Diamond JR, Moroose RL, Isakoff S.J, Starodub AN, et al. Efficacy and safety of anti-Trop-2 antibody drug conjugate sacituzumab govitecan (IMMU-132) in heavily pretreated patients with metastatic triple-negative breast cancer. J Clin Oncol Off J Am Soc Clin Oncol (2017) 35:2141–8. doi: 10.1200/jco.2016.70.8297
22. McCann KE, Hurvitz SA. Advances in the use of PARP inhibitor therapy for breast cancer. Drugs context (2018) 7:212540. doi: 10.7573/dic.212540
23. Byrd TT, Fousek K, Pignata A, Szot C, Samaha H, Seaman S, et al. TEM8/ANTXR1-specific CAR T cells as a targeted therapy for triple-negative breast cancer. Cancer Res (2018) 78:489–500. doi: 10.1158/0008-5472.Can-16-1911
24. Guinney J, Dienstmann R, Wang X, de Reyniès A, Schlicker A, Soneson C, et al. The consensus molecular subtypes of colorectal cancer. Nat Med (2015) 21:1350–6. doi: 10.1038/nm.3967
25. Newman AM, Liu CL, Green MR, Gentles AJ, Feng W, Xu Y, et al. Robust enumeration of cell subsets from tissue expression profiles. Nat Methods (2015) 12:453–7. doi: 10.1038/nmeth.3337
26. Raudvere U, Kolberg L, Kuzmin I, Arak T, Adler P, Peterson H, et al. g:Profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update). Nucleic Acids Res (2019) 47:W191–8. doi: 10.1093/nar/gkz369
27. Hendry S, Salgado R, Gevaert T, Russell PA, John T, Thapa B, et al. Assessing tumor-infiltrating lymphocytes in solid tumors: A practical review for pathologists and proposal for a standardized method from the international immunooncology biomarkers working group: Part 1: Assessing the host immune response, TILs in invasive breast carcinoma and ductal carcinoma in situ, metastatic tumor deposits and areas for further research. Adv anatomic Pathol (2017) 24:235–51. doi: 10.1097/pap.0000000000000162
28. Specht E, Kaemmerer D, Sänger J, Wirtz RM, Schulz S, Lupp A, et al. Comparison of immunoreactive score, HER2/neu score and h score for the immunohistochemical evaluation of somatostatin receptors in bronchopulmonary neuroendocrine neoplasms. Histopathology (2015) 67:368–77. doi: 10.1111/his.12662
29. Edin S, Wikberg ML, Dahlin AM, Rutegård J, Öberg Å, Oldenborg PA, et al. The distribution of macrophages with a M1 or M2 phenotype in relation to prognosis and the molecular characteristics of colorectal cancer. PloS One (2012) 7:e47045. doi: 10.1371/journal.pone.0047045
30. Zhou X, Qiu S, Jin D, Jin K, Zheng X, Yang L, et al. Development and validation of an individualized immune-related gene pairs prognostic signature in papillary renal cell carcinoma. Front Genet (2020) 11:569884. doi: 10.3389/fgene.2020.569884
31. Zhang L, Zhu P, Tong Y, Wang Y, Ma H, Xia X, et al. An immune-related gene pairs signature predicts overall survival in serous ovarian carcinoma. OncoTargets Ther (2019) 12:7005–14. doi: 10.2147/ott.S200191
32. Zhuang H, Zhang C, Hou B. GTF2IRD1 overexpression promotes tumor progression and correlates with less CD8+ T cells infiltration in pancreatic cancer. Biosci Rep (2020) 40:BSR20202150. doi: 10.1042/BSR20202150
33. Hollande C, Boussier J, Ziai J, Nozawa T, Bondet V, Phung W, et al. Inhibition of the dipeptidyl peptidase DPP4 (CD26) reveals IL-33-dependent eosinophil-mediated control of tumor growth. Nat Immunol (2019) 20:257–64. doi: 10.1038/s41590-019-0321-5
34. Juric V, O'Sullivan C, Stefanutti E, Kovalenko M, Greenstein A, Barry-Hamilton V, et al. MMP-9 inhibition promotes anti-tumor immunity through disruption of biochemical and physical barriers to T-cell trafficking to tumors. PloS One (2018) 13:e0207255. doi: 10.1371/journal.pone.0207255
35. Chen Z, Ge Y, Landman N, Kang JX. Decreased expression of the mannose 6-phosphate/insulin-like growth factor-II receptor promotes growth of human breast cancer cells. BMC Cancer (2002) 2:18. doi: 10.1186/1471-2407-2-18
36. Chappell SA, Walsh T, Walker RA, Shaw JA. Loss of heterozygosity at the mannose 6-phosphate insulin-like growth factor 2 receptor gene correlates with poor differentiation in early breast carcinomas. Br J Cancer (1997) 76:1558–61. doi: 10.1038/bjc.1997.596
37. Hankins GR, De Souza AT, Bentley RC, Patel MR, Marks JR, Iglehart JD, et al. M6P/IGF2 receptor: A candidate breast tumor suppressor gene. Oncogene (1996) 12:2003–9. doi: 10.7150/jca.23023
38. Esseghir S, Reis-Filho JS, Kennedy A, James M, O'Hare MJ, Jeffery R, et al. Identification of transmembrane proteins as potential prognostic markers and therapeutic targets in breast cancer by a screen for signal sequence encoding transcripts. J Pathol (2006) 210:420–30. doi: 10.1002/path.2071
39. Mountzios G, Aivazi D, Kostopoulos I, Kourea HP, Kouvatseas G, Timotheadou E, et al. Differential expression of the insulin-like growth factor receptor among early breast cancer subtypes. PloS One (2014) 9:e91407. doi: 10.1371/journal.pone.0091407
40. Hessel H, Poignée-Heger M, Lohmann S, Hirscher B, Herold A, Assmann G, et al. Subtyping of triple negative breast carcinoma on the basis of RTK expression. J Cancer (2018) 9:2589–602. doi: 10.7150/jca.23023
41. Liu LY, Chang LY, Kuo WH, Hwa HL, Lin YS, Huang SF, et al. Major functional transcriptome of an inferred center regulator of an ER(-) breast cancer model system. Cancer Inf (2012) 11:87–111. doi: 10.4137/cin.S8633
42. Liu L, Yu D, Shi H, Li J, Meng L. Reduced lncRNA aim enhances the malignant invasion of triple-negative breast cancer cells mainly by activating wnt/β-catenin/mTOR/PI3K signaling. Die Pharmazie (2017) 72:599–603. doi: 10.1691/ph.2017.7547
43. Xie S, Kang JX. Differential expression of the mannose 6-phosphate/ insulin-like growth factor-II receptor in human breast cancer cell lines of different invasive potential. Med Sci monit. Int Med J Exp Clin Res (2002) 8:Br293–300. doi: 10.3390/ijms21186968
44. Goff SL, Danforth DN. The role of immune cells in breast tissue and immunotherapy for the treatment of breast cancer. Clin Breast Cancer (2021) 21:e63–73. doi: 10.1016/j.clbc.2020.06.011
45. Liu S, Lachapelle J, Leung S, Gao D, Foulkes WD, Nielsen TO, et al. CD8+ lymphocyte infiltration is an independent favorable prognostic indicator in basal-like breast cancer. Breast Cancer Res (2012) 14:R48. doi: 10.1186/bcr3148
46. Oshi M, Asaoka M, Tokumaru Y, Yan L, Matsuyama R, Ishikawa T, et al. CD8 T cell score as a prognostic biomarker for triple negative breast cancer. Int J Mol Sci (2020) 21:6969. doi: 10.3390/ijms21186968
47. Emens LA, Molinero L, Loi S, Rugo HS, Schneeweiss A, Diéras V, et al. Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer: Biomarker evaluation of the IMpassion130 study. J Natl Cancer Inst (2021) 113:1005–16. doi: 10.1093/jnci/djab004
48. Pantelidou C, Sonzogni O, De Oliveria Taveira M, Mehta AK, Kothari A, Wang D, et al. PARP inhibitor efficacy depends on CD8(+) T-cell recruitment via intratumoral STING pathway activation in BRCA-deficient models of triple-negative breast cancer. Cancer Discov (2019) 9:722–37. doi: 10.1158/2159-8290.Cd-18-1218
49. Nelson MA, Ngamcherdtrakul W, Luoh SW, Yantasee W. Prognostic and therapeutic role of tumor-infiltrating lymphocyte subtypes in breast cancer. Cancer Metastasis Rev (2021) 40:519–36. doi: 10.1007/s10555-021-09968-0
50. Schmid P, Rugo HS, Adams S, Schneeweiss A, Barrios CH, Iwata H, et al. Atezolizumab plus nab-paclitaxel as first-line treatment for unresectable, locally advanced or metastatic triple-negative breast cancer (IMpassion130): Updated efficacy results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol (2020) 21:44–59. doi: 10.1016/S1470-2045(19)30689-8
51. Cortes J, Cescon DW, Rugo HS, Nowecki Z, Im SA, Yusof MM, et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): A randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet (London England) (2020) 396:1817–28. doi: 10.1016/s0140-6736(20)32531-9
52. Miles D, Gligorov J, André F, Cameron D, Schneeweiss A, Barrios C, et al. Primary results from IMpassion131, a double-blind, placebo-controlled, randomised phase III trial of first-line paclitaxel with or without atezolizumab for unresectable locally advanced/metastatic triple-negative breast cancer. Ann Oncol (2021) 32:994–1004. doi: 10.1016/j.annonc.2021.05.801
53. Kecha O, Brilot F, Martens H, Franchimont N, Renard C, Greimers R, et al. Involvement of insulin-like growth factors in early T cell development: A study using fetal thymic organ cultures. Endocrinology (2000) 141:1209–17. doi: 10.1210/endo.141.3.7360
54. Wei Y, Li J, Huang J, Zhang X, Zhao H, Cui C, et al. Elevation of IGF-2 receptor and the possible underlying implications in end-stage heart failure patients before and after heart transplantation. J Cell Mol Med (2012) 16:1038–46. doi: 10.1111/j.1582-4934.2011.01414.x
55. Suen JL, Wu TT, Li YH, Lee CL, Kuo FC, Yan PS, et al. Environmental factor-mediated transgenerational inheritance of Igf2r hypomethylation and pulmonary allergic response via targeting dendritic cells. Front Immunol (2020) 11:603831. doi: 10.3389/fimmu.2020.603831
56. Xiao Z, Casey KA, Jameson SC, Curtsinger JM, Mescher MF. Programming for CD8 T cell memory development requires IL-12 or type I IFN. J Immunol (2009) 182:2786–94. doi: 10.4049/jimmunol.0803484
57. Gemma A, Hosoya Y, Uematsu K, Seike M, Kurimoto F, Yoshimura A, et al. Mutation analysis of the gene encoding the human mannose 6-phosphate/insulin-like growth factor 2 receptor (M6P/IGF2R) in human cell lines resistant to growth inhibition by transforming growth factor beta(1) (TGF-beta(1)). Lung Cancer (Amsterdam Netherlands) (2000) 30:91–8. doi: 10.1016/s0169-5002(00)00130-6
58. Yang G, Geng XR, Song JP, Wu Y, Yan H, Zhan Z, et al. Insulin-like growth factor 2 enhances regulatory T-cell functions and suppresses food allergy in an experimental model. J Allergy Clin Immunol (2014) 133:1702–1708.e1705. doi: 10.1016/j.jaci.2014.02.019
59. Tauriello DVF, Palomo-Ponce S., Stork D., Berenguer-Llergo A., Badia-Ramentol J., Iglesias M., et al. TGFβ drives immune evasion in genetically reconstituted colon cancer metastasis. Nature (2018) 554:538–43. doi: 10.1038/nature25492
60. Xie J, Shi CW, Huang HB, Yang WT, Jiang YL, Ye LP, et al. Induction of the IL-10-producing regulatory b cell phenotype following trichinella spiralis infection. Mol Immunol (2021) 133:86–94. doi: 10.1016/j.molimm.2021.02.012
61. Moreno-Cortes E, Forero-Forero JV, Lengerke-Diaz PA, Castro JE. Chimeric antigen receptor T cell therapy in oncology - pipeline at a glance: Analysis of the ClinicalTrials.gov database. Crit Rev oncology/hematol. (2021) 159:103239. doi: 10.1016/j.critrevonc.2021.103239
62. Toulouie S, Johanning G, Shi Y. Chimeric antigen receptor T-cell immunotherapy in breast cancer: development and challenges. J Cancer (2021) 12:1212–9. doi: 10.7150/jca.54095
63. Buisseret L, Garaud S, de Wind A, Van den Eynden G, Boisson A, Solinas C, et al. Tumor-infiltrating lymphocyte composition, organization and PD-1/ PD-L1 expression are linked in breast cancer. Oncoimmunology (2017) 6:e1257452. doi: 10.1080/2162402x.2016.1257452
64. Guan H, Lan Y, Wan Y, Wang Q, Wang C, Xu L, et al. PD-L1 mediated the differentiation of tumor-infiltrating CD19(+) b lymphocytes and T cells in invasive breast cancer. Oncoimmunology (2016) 5:e1075112. doi: 10.1080/2162402x.2015.1075112
65. Zhang X, Shen L, Cai R, Yu X, Yang J, Wu X, et al. Comprehensive analysis of the immune-oncology targets and immune infiltrates of n (6)-Methyladenosine-Related long noncoding RNA regulators in breast cancer. Front Cell Dev Biol (2021) 9:686675. doi: 10.3389/fcell.2021.686675
66. Ishigami E, Sakakibara M, Sakakibara J, Masuda T, Fujimoto H, Hayama S, et al. Coexistence of regulatory b cells and regulatory T cells in tumor-infiltrating lymphocyte aggregates is a prognostic factor in patients with breast cancer. Breast Cancer (Tokyo Japan) (2019) 26:180–9. doi: 10.1007/s12282-018-0910-4
67. Geng XR, Yang G, Li M, Song JP, Liu ZQ, Qiu S, et al. Insulin-like growth factor-2 enhances functions of antigen (Ag)-specific regulatory b cells. J Biol Chem (2014) 289:17941–50. doi: 10.1074/jbc.M113.515262
68. Emmerich J, Mumm JB, Chan IH, LaFace D, Truong H, McClanahan T, et al. IL-10 directly activates and expands tumor-resident CD8(+) T cells without de novo infiltration from secondary lymphoid organs. Cancer Res (2012) 72:3570–81. doi: 10.1158/0008-5472.Can-12-0721
69. Damo M, Joshi NS. T(reg) cell IL-10 and IL-35 exhaust CD8(+) T cells in tumors. Nat Immunol (2019) 20:674–5. doi: 10.1038/s41590-019-0389-y
70. Smith LK, Boukhaled GM, Condotta SA, Mazouz S, Guthmiller JJ, Vijay R, et al. Interleukin-10 directly inhibits CD8(+) T cell function by enhancing n-glycan branching to decrease antigen sensitivity. Immunity (2018) 48:299–312.e295. doi: 10.1016/j.immuni.2018.01.006
71. Morris G, Puri BK, Olive L, Carvalho AF, Berk M, Maes M, et al. Emerging role of innate B1 cells in the pathophysiology of autoimmune and neuroimmune diseases: Association with inflammation, oxidative and nitrosative stress and autoimmune responses. Pharmacol Res (2019) 148:104408. doi: 10.1016/j.phrs.2019.104408
Keywords: poor prognosis, triple negative breast cancer, TIL (tumor infiltrating lymphocytes), CD8+ TILs, IGF2R
Citation: Zhong Y, Ren X, Cao X, Xu Y, Song Y, Zhou Y, Mao F, Shen S, Wang Z and Sun Q (2022) Insulin-like growth factor 2 receptor is a key immune-related gene that is correlated with a poor prognosis in patients with triple-negative breast cancer: A bioinformatics analysis. Front. Oncol. 12:871786. doi: 10.3389/fonc.2022.871786
Received: 08 February 2022; Accepted: 27 September 2022;
Published: 18 October 2022.
Edited by:
William M. Gallagher, University College Dublin, IrelandReviewed by:
Marcus Vetter, University Hospital of Basel, SwitzerlandCopyright © 2022 Zhong, Ren, Cao, Xu, Song, Zhou, Mao, Shen, Wang and Sun. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Qiang Sun, YmFrZW5maXNoQDE2My5jb20=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.