
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Oncol. , 17 January 2023
Sec. Breast Cancer
Volume 12 - 2022 | https://doi.org/10.3389/fonc.2022.1094136
 Parynaz Parhizgar1
Parynaz Parhizgar1 Ayad Bahadori Monfared2
Ayad Bahadori Monfared2 Maryam Mohseny2
Maryam Mohseny2 Aliasghar Keramatinia2
Aliasghar Keramatinia2 Seyed Saeed Hashemi Nazari2,3
Seyed Saeed Hashemi Nazari2,3 Syed Azizur Rahman4
Syed Azizur Rahman4 Amina Al Marzouqi5
Amina Al Marzouqi5 Nabeel Al-Yateem5*
Nabeel Al-Yateem5* Alireza Mosavi Jarrahi2,6*
Alireza Mosavi Jarrahi2,6*Objectives: The aim of this study was to estimate the extra risk of second primary cancer among breast cancer patients.
Methods and materials: This is a systematic review. A comprehensive search of literature was performed in PubMed, Web of Science, Cochrane library, and Scopus. The search included all published studies up to October 2022. This systematic review included studies published in the English language that reported the risk of second primary non-breast cancer [i.e., standardized incidence ratio (SIR)] among breast cancer patients older than 15 years. After evaluating the methodological quality of the selected studies, SIRs were pooled with consideration of heterogeneity among studies. The estimates were pooled by age and time since the diagnosis of primary breast cancer for both sexes (male and female). Age was categorized based on before 50 years and after 50 years, and time was categorized as duration of less than and more than 10 years, respectively.
Results: From 2,484 articles, 30 articles were eligible for inclusion in the systematic review and meta-analysis. The studies varied in terms of population, number of cases, study design, setting, and year of implementation of the research. The estimated SIR for men and women was 1.28 (95% CI: 1.18, 1.38) and 1.27 (95% CI: 1.15, 1.39), respectively. Women diagnosed with breast cancer before menopause [SIR: 1.52 (95% CI: 1.34, 1.71) vs. 1.21 (95% CI: 1.08, 1.34)] as well as women after 10 years since their breast cancer diagnosis [1.33 (95% CI: 1.22, 1.431) vs. 1.24 (95% CI: 1.10, 1.37)] were at a higher risk of developing second primary cancer. Among men, while there were no differences in risk based on age, with the increase of time, the risk of second primary cancer was reduced [SIR: 1.22 (95% CI: 1.12, 1.33) vs. 1.00 (95% CI: 0.79, 1.22)].
Conclusion: There is an extra risk of second primary cancer among breast cancer patients. The extra risk should be considered for further screening and preventive measures among this population.
Systematic review registration: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=336062, identifier (CRD42022336062).
Breast cancer is one of the major health problems in the world and ranks number one in mortality among women of older age in many developed and even developing countries (1). Longer survival does not only result in a longer low-quality-of-life among the population of patients but also provides ample opportunity for the occurrence of a second primary malignancy, which somehow is related to the constitutional susceptibility of these patients to malignancy (2). According to Globocan 2020, breast cancer is one of the most common cancers in the world, with age-standardized incidence rates of up to 47.8 per 100,000 and age-standardized mortality rates of up to 13.6 per 100,000 (3). In the recent three decades, better access to health services, breast cancer screening (4), early detection (5), and advances in breast cancer treatment (hormone therapy, surgery, radiotherapy, and chemotherapy) have led to an increase in survival and a reduction in breast cancer deaths (especially in developed countries) (6–8). As patient survival increases, the risk of second primary cancers (also known as second cancers) increases and becomes a burden for patients and healthcare providers. An unexpectedly higher rate of second primary cancer among survivors of breast cancer shed light on an underlying susceptibility among this group of patients (9). The risk of second primary cancer (excluding contralateral breast cancer) has been reported to be as high as 20%–30% among breast cancer survivors (10). While all kinds of cancers are reported as second primary, cancers of the endometrium, ovary, thyroid, lung, soft tissue sarcomas, leukemia, melanoma, stomach, and colon have been reported more frequently (11, 12). The descriptive epidemiology of second primary cancer among survivors at 5, 10, and 15 years is reported to be 3.6%, 8.2%, and 13.9%, respectively (13), and patients less than 50 years of age (also premenopausal ages) are at a higher risk of developing second cancer (10, 12). Several population-based cancer registry studies have assessed the risk of second primary cancers among women diagnosed with primary breast cancer compared with the general population (10, 12, 14–18). Some studies have also examined the risk of developing second cancer based on the type of treatment (19–24), age difference (25–27), and family history (27–29). However, the risk estimates provided by these studies vary widely, and there is no consensus among the studies (in terms of the definition of first and second primary cancers and the inclusion and exclusion criteria). Studies have used different definitions for second primary cancer and different coding systems [such as International Association of Cancer Registries (IARC) and Surveillance, Epidemiology, and End Results (SEER)]. In a study by Molina et al. (7), which covered studies published up to 2013 based on the IARC definition of a second primary cancer, the standardized incidence ratio (SIR) of developing second cancer (excluding second breast cancer) was 1.17 (17% higher than expected). However, this study did not report the risk of each cancer separately. Knowing the risk of a second primary cancer in any of the organs can help policymakers prioritize resources as well as generate solid evidence on the risk of a second primary cancer among breast cancer survivors and will fill the existing gap for administrative purposes. This systematic review aims to investigate the risk and incidence of second primary non-breast malignancies among breast cancer patients (all cancers in men and women) using observational studies.
This systematic review and meta-analysis was developed based on the MOOSE Checklist for Meta-analyses of Observational Studies (30). A comprehensive search of literature was performed in PubMed, Web of Science, Cochrane library, and Scopus. The search included all published studies up to October 2022.
A systematic search was carried out up to October 7, 2022. Initially, relevant keywords were identified using the primary search in PubMed/Medline; then, the search strategy was adapted accordingly for Scopus, Cochrane Library, and Web of Science. Language restrictions were considered in the search process (only English articles). The following selected keywords were used and combined in the selected search databases: multiple primary neoplasm(s), multiple primary cancer, multiple primary malignancies, second cancer, second malignancies, breast cancer, risk, and population-based. The details of the comprehensive search strategy for this systematic review are provided in the supplementary materials. Studies were entered into EndNote X9 software (Thomson Reuters, New York, NY, USA), and duplicate items were detected and removed by this software.
This systematic review included all studies published in the English language that reported the risk of a second primary cancer (i.e., SIR) among breast cancer patients older than 15 years. The second primary cancer was defined based on IARC definition (31) which referred to second primary cancer as more than one cancer diagnosed in the same person which is not dependent on time and not categorized as a recurrence or metastasis; in addition, only one tumor should be present in one organ or paired organ [there are two exceptions (1): Kaposi sarcoma and tumors of the hematopoietic system are considered as one tumor and (2) if tumors with a different morphology are reported in a paired organ, they are counted as two tumors].
Studies that used definitions other than the IARC definition for reporting second primary malignancies (such as those defined by SEER) were excluded. Articles that did not specify the coding definition were excluded if it was apparent that the definition for accepting a second primary cancer did not agree with the IARC definition. Articles reporting a second primary breast cancer usually lack information on reporting second cancer in the same or in the contralateral breast as well as histological reports of tumors. Therefore, all studies which included second primary breast cancer were excluded unless risk estimates for other sites of second primary cancer were reported. Due to the multimodal treatment of breast cancer, studies that examined the risk of second cancer based on treatment type were excluded.
In the selection stage, the two reviewers separately checked the title, abstract, and full text of the articles using the inclusion and exclusion criteria, and any discrepancies between the results were resolved by discussion. The reference lists of the included studies were reviewed to identify other potentially relevant studies. If more than one article were published for one population, the newer or the one with a larger population was included. The data was extracted manually by two independent researchers. Disagreements were resolved through discussion and consensus. If there were deficiencies in the required information of the selected studies, the corresponding author of the article was contacted by email, and additional information about the results of the study was requested. The following information was extracted: first authors, publication date, country of origin, study design, study follow-up period, cases (number of first and second primary cancer), mean age at cancer diagnosis (both first and second primary cancer), gender, definition of second primary cancer, inclusion criteria for their population, and estimated SIR and its associated 95% confidence intervals (CI). The following cancer sites were included in the analysis: skin (melanoma and non-melanoma), head and neck (oral cavity, pharynx, and larynx), esophagus, stomach, colorectal, liver and gallbladder and biliary tract, pancreas, lung and mediastinum, Kaposi sarcoma, leukemia, lymphoma, multiple myeloma, soft tissue sarcoma, bone, prostate, testis, kidney, bladder and urinary tract, brain and nervous system, thyroid, eye, corpus uteri, cervix uteri, ovary, adrenal gland, and parathyroid.
The standard incidence ratio was defined as the incidence of cancer cases in a population over a given period to the incidence of cancer that would be expected over the same period if the study population had the same age-specific rates as the standard population. If the ratio is greater than 1, it is interpreted as extra incidence in the study population. First, we calculated the standard error (SE) for each estimate (SE = √observed/expected2) (32). Then, we calculated pooled risk ratios (SIR) using SIR and SE. Age at primary breast cancer diagnosis was categorized as <50 years and ≥50 years, and the diagnosis of primary breast cancer to the diagnosis of second cancer was categorized as <10 years and ≥10 years. The variations in the categorization of age or time since diagnosis were standardized by pooling the categories to their nearest cutoff point to meet our study’s categorization scheme. Moreover, if the cutoff points of a study overlapped with both cutoff categories of our study, we equally divided the observed and expected values into two cutoff boundaries.
To evaluate the risk of bias in individual studies, we used the SIGN50 Scottish Intercollegiate Network checklist (33). This checklist assesses methodological quality, overall quality (based on design, internal validity, consistency, and accuracy of results), and level of evidence and finally classifies studies into three (low, acceptable, and high) categories. Using the Q-statistic and I2 values, a heterogeneity test was carried out among the included studies. If the I2 value is less than 50%, it was considered as without significant heterogeneity, and we pooled the estimates using the fixed-effect model. An I2 value higher than 50% was considered as obvious heterogeneity, and we pooled estimates using the random-effect model. In addition, subgroup and sensitivity analyses were performed to identify the source of heterogeneity. The effect of a study’s quality on the estimates was measured using meta-regression and subgroup analyses. Publication bias was investigated using funnel plots (34), and Egger’s (35) test.
As shown in Figure 1, the defined search criteria in selected databases retrieved 2,471 potentially relevant articles. Due to duplication, 445 articles were removed. We screened the articles based on the title and the abstract, and 1,942 articles were excluded for different reasons. Furthermore, through a manual search of reference lists, we identified 13 articles. Therefore, 97 articles were left for full-text review, of which 57 articles were excluded: four studies did not use IARC definition (i.e., SEER, etc.), 27 studies did not provide SIR or risk estimate, and four studies did not provide SIR for specific cancer site but reported for total cancer combined. Population or period overlaps were also seen in 22 studies (36–57). Finally, 30 studies (10, 12, 14–17, 27, 28, 58–79) were included in the present systematic review and meta-analysis.
More than 2,434,975 cases of breast cancer [two studies did not mention the number of primary breast cancer patients (70, 73)] were included in the study, of which 91,678 patients developed second primary cancers. The summary of the characteristics of the included studies is shown in Supplementary Table 1. The design of all studies, except the study of An et al. (74), was a retrospective cohort. The study of An et al. was considered as a retrospective case–controlled study design. In terms of data source, three studies used hospital-based data (61, 63, 74), and the others used population-based data. A total of 16 studies mentioned that the IARC definition was used for defining the second primary cancers (10, 12, 14–17, 63, 66–69, 71–73, 77, 79), but the remaining studies did not mention this. In general, the studies included in this systematic review cover data from 22 countries: Japan, South Korea, Taiwan, Singapore, Turkey, Israel, United Kingdom, Finland, Denmark, Sweden, Norway, Germany, France, Greece, Netherlands, Switzerland, Spain, Slovenia, Italy, Iceland, Canada, and Australia (Figure 2). Furthermore, most of the studies were conducted on women, except the studies of Hung et al. (75) and AIRTUM Working Group (14), which were conducted on both sexes, and the study of Hemminki et al. (66), which was conducted on men. The methodological quality of the included articles was evaluated as high, except for the articles that were prone to misclassification bias (61, 68) and/or selection bias (15, 17, 59, 61, 62, 70, 73, 74, 77).
The summary of pooled estimates based on sex, second primary cancer site, age at diagnosis of breast cancer, and time since breast cancer diagnosis is provided in Tables 1–4. The summarized SIR estimate for female patients (Figure 3A) and male patients (Figure 4A) was 1.27 (95% CI: 1.15, 1.39) and 1.28 (95% CI: 1.18, 1.38), respectively.
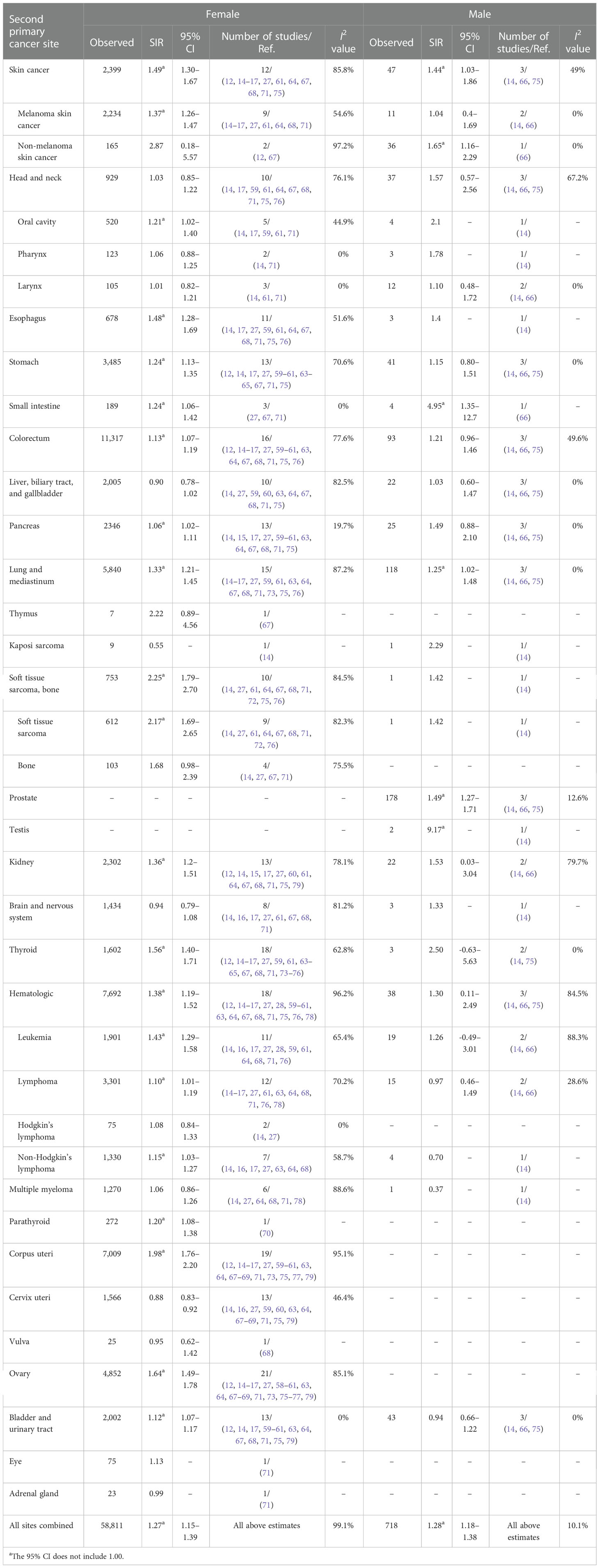
Table 1 Standardized incidence ratios (SIRs) of second cancers by second cancer site in female and male patients.
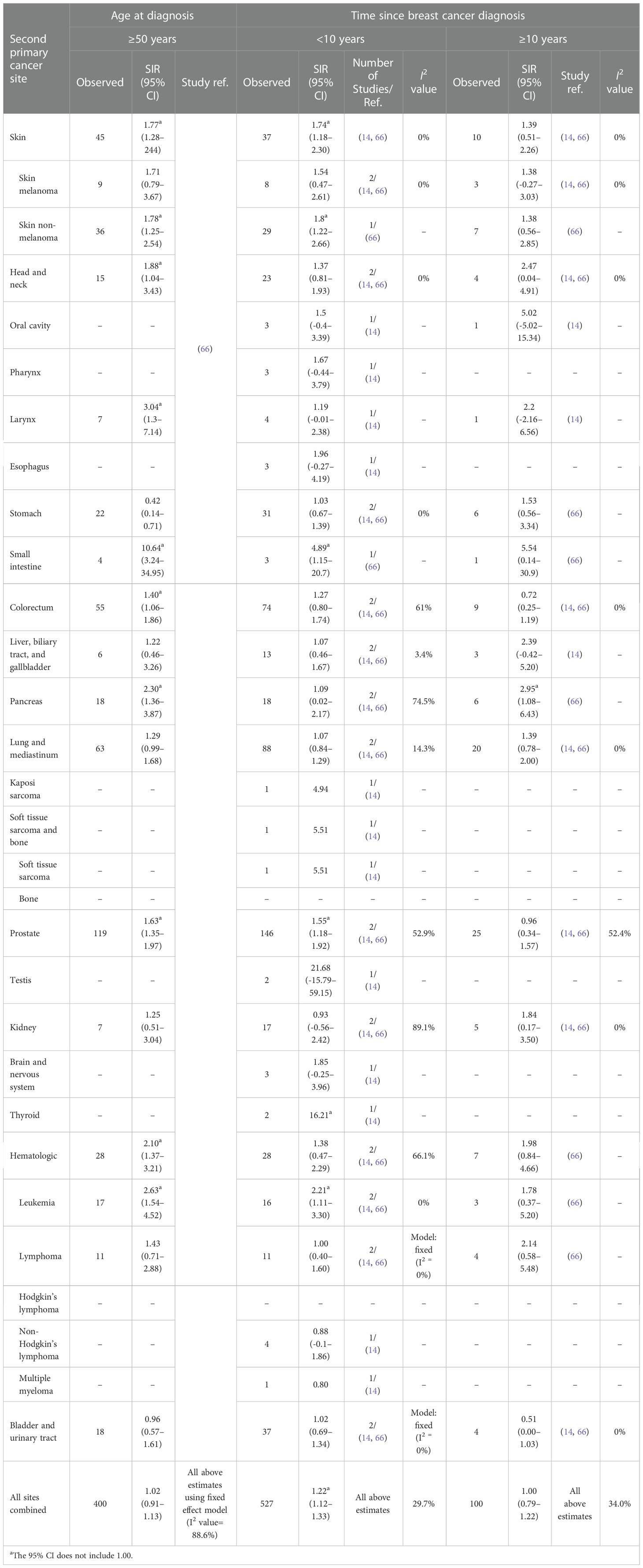
Table 2 Standardized incidence ratios (SIRs) of second cancers by second cancer site, age at diagnosis, and time since breast cancer diagnosis in male patients.
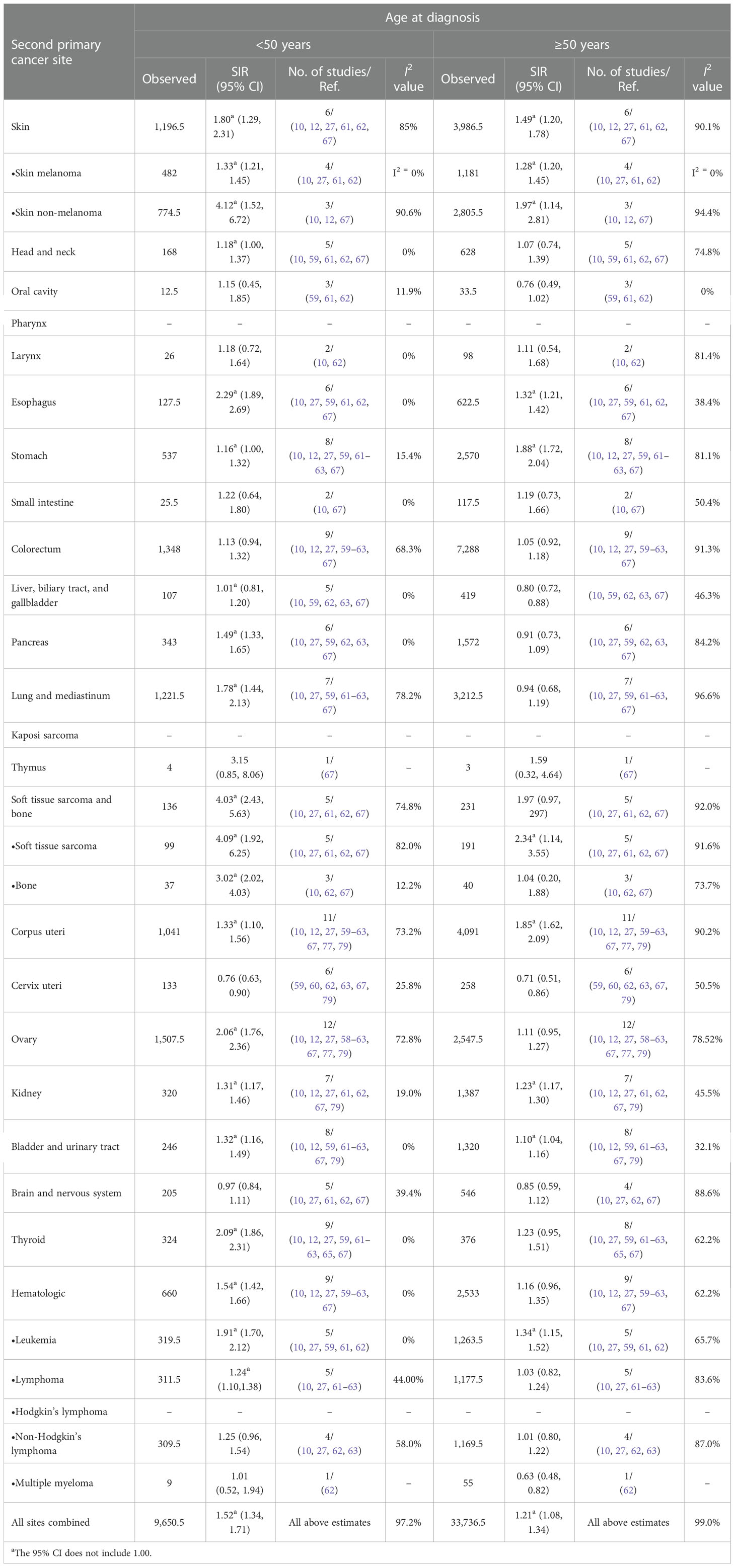
Table 3 Standardized incidence ratios (SIRs) of second cancers by site and age at diagnosis in female patients.
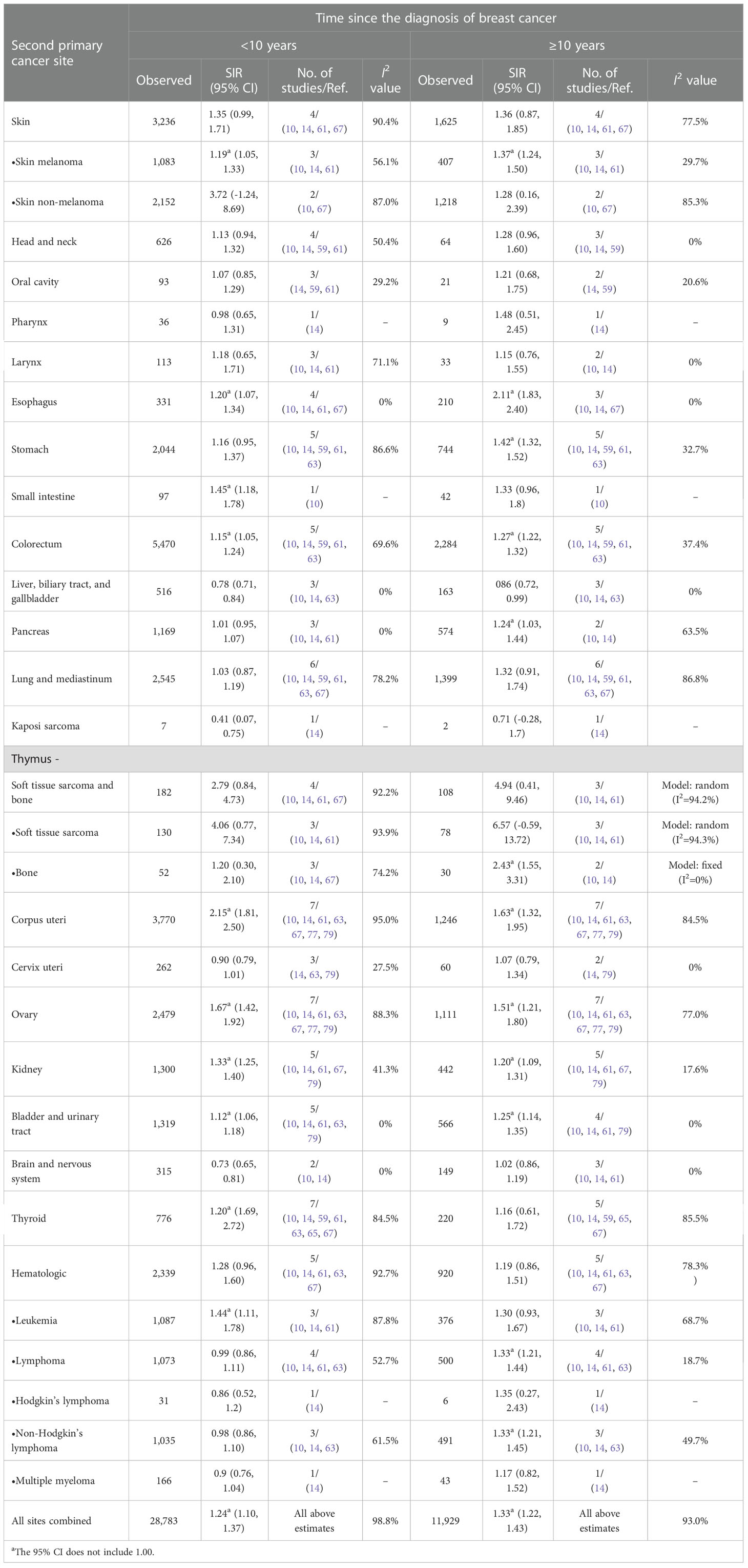
Table 4 Standardized incidence ratios (SIRs) of second cancers by second cancer size and time since the diagnosis of breast cancer in female patients.
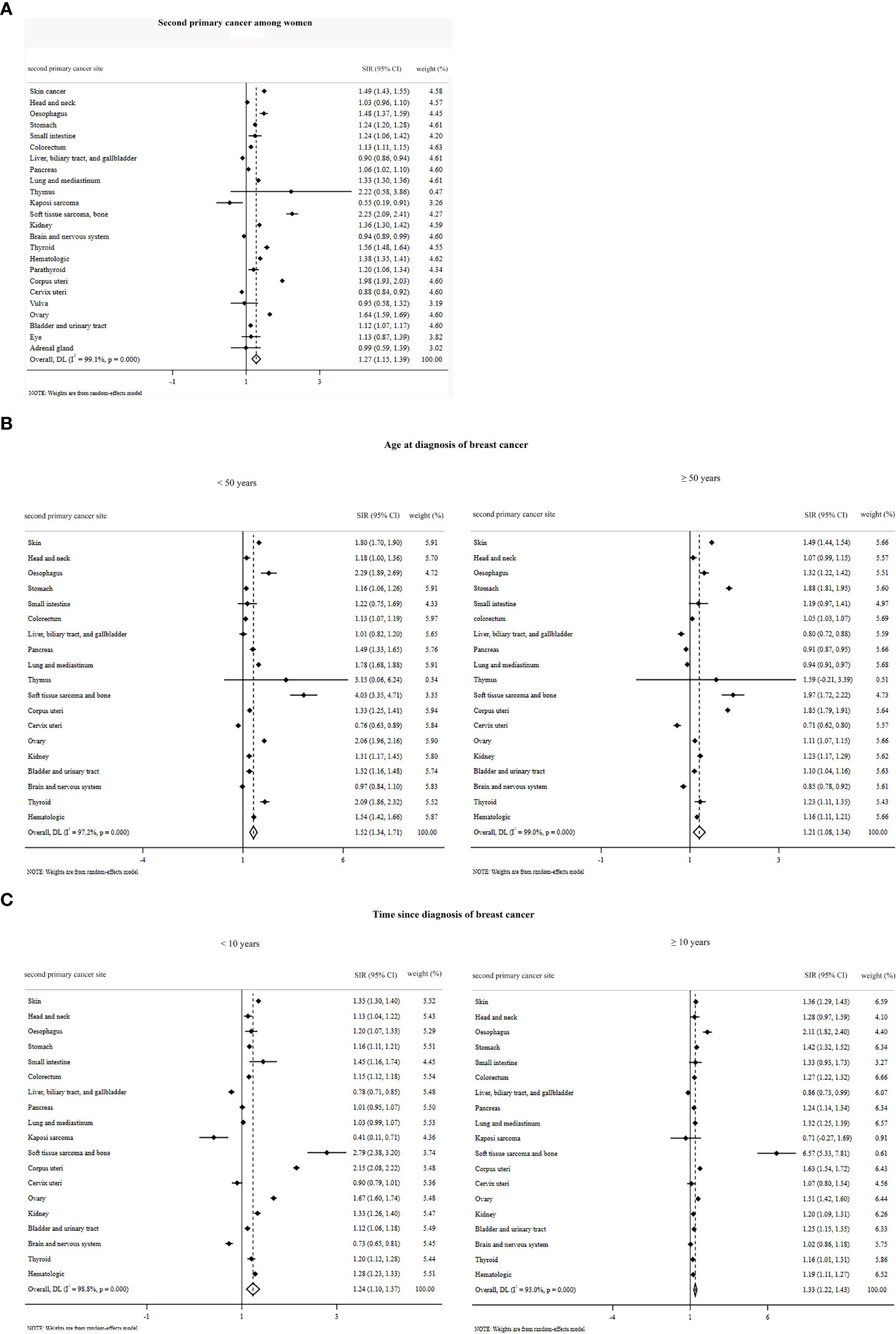
Figure 3 Overall standardized incidence ratios of second primary cancer among women (A) by age at diagnosis of breast cancer (B) and by time since the diagnosis of breast cancer (C).

Figure 4 Overall standardized incidence ratios of second primary cancer among men (A) by age at diagnosis of breast cancer (B) and by time since the diagnosis of breast cancer (C).
Where the age at diagnosis is concerned, the pooled estimate for men older than 50 years using the fixed-effect model (although the I2 value is higher than 50%, because only the estimates of one article (66) were pooled, the fixed model was used) was 1.02 (95% CI: 0.91, 1.13). Moreover, the SIR by time since diagnosis of breast cancer for male patients <10 years and ≥10 years was 1.22 (95% CI: 1.12, 1.33) and 1.00 (95% CI: 0.79, 1.22), respectively (Figures 4B, C). The overall SIR by age at diagnosis of breast cancer for female patients <50 years and ≥50 years was 1.52 (95% CI: 1.34, 1.71) and 1.21 (95% CI: 1.08, 1.34), respectively (Figure 3B). The overall SIR by time since diagnosis of breast cancer for female patients <10 years and ≥10 years was 1.24 (95% CI: 1.10, 1.37) and 1.33 (95% CI: 1.22, 1.43), respectively (Figure 3C).
Publication bias was seen in the following second primary cancers: among female patients based on second cancer sites—melanoma skin cancer [trim-and-fill method: 1.35 (95% CI: 1.23, 1.47)], soft tissue sarcoma and bone [trim-and-fill method: 1.87 (95% CI: 1.35, 2.39)], soft tissue sarcoma [trim-and-fill method: 2.16 (95% CI: 12.65, 2.66)], non-Hodgkin’s lymphoma [trim-and-fill method: 1.11 (95% CI: 0.99, 1.23)], corpus uteri [trim-and-fill method: 1.74 (95% CI: 1.54, 1.95)], and ovary [trim-and-fill method: 1.49 (95% CI: 1.33, 1.64)]; among female patients younger than 50 years—esophagus [trim-and-fill method: 2.27 (95% CI: 1.87, 2.66)]; and among female patients with second cancer diagnosis less than 10 years from breast cancer diagnosis—thyroid [trim-and-fill method: 2.20 (95% CI: 1.69, 2.72)] and kidney [trim-and-fill method: 1.32 (95% CI: 1.25, 1.40)].
The meta-regression analysis revealed that study quality only influences the risk estimate in the bladder and urinary tract cancer among female patients ≥50 years (regression coefficient = 0.2178597, P = 0.037).
In the sensitivity analysis, only the second primary cancer sites described below depended on a certain study. Based on the second cancer site among female patients, the SIRs of the liver and gallbladder and biliary tract were dependent on the study of Hung et al. (75). After omitting this study, the SIR and its association 95% CI were as follows: 0.85 (95% CI: 0.74, 0.96). Based on the second primary cancer site among female patients ≥50 years, the SIR for head and neck, soft tissue sarcoma and bone, thyroid, and hematologic cancers was dependent on the study of Evans et al. (62). After omitting this study, the SIR and its association 95% CI were as follows: head and neck—1.23 (95% CI: 1.13, 1.33), soft tissue sarcoma and bone—2.45 (95% CI: 1.04, 3.88), thyroid—1.30 (95% CI: 1.01, 1.59), and hematologic—1.23 (95% CI: 1.12, 1.34). Based on the second primary cancer site among female patients with a second cancer diagnosis less than 10 years after breast cancer diagnosis, the SIR of stomach cancer depended on the study of Tanaka et al. (63). After omitting this study, the SIR and its association 95% CI were 1.22 (95% CI: 1.01, 1.44). Furthermore, based on the second primary cancer site among female patients with a second primary cancer diagnosis ≥10 years after breast cancer diagnosis, the SIR of hematologic cancer depended on the study of Lee et al. (67). After omitting this study, the SIR and its association 95% CI were 1.31 (95% CI: 1.04, 1.59).
Our study demonstrated that breast cancer patients have a higher risk of having second primary cancer, and this risk varied across different cancer sites and were dependent on the time since and age at the diagnosis of primary breast cancer. The incidence of second cancer can be attributed not only to genetic and underlying causes but also to factors such as treatment complications, lifestyle (obesity, low exercise, and tobacco and alcohol use), immune function, hormonal imbalances, age, race, environmental factors, and shared etiologic factors (80, 81). Knowingly, all these factors—even the prognostic factors—among breast cancer patients contribute to susceptibility to a second primary cancer among these patients (82, 83). The risk of the second primary cancers differs for different cancer sites. Male breast cancer patients are at a higher risk of developing second primary colorectal, small intestine, pancreas, thyroid, kidney, and testis cancers (14, 66, 84). Female breast cancer patients are at a higher risk of developing second primary cancer of the endometrium, ovary, thyroid, lung, soft tissue sarcomas, leukemia, melanoma, stomach, and colorectum (9, 57, 61–64, 85–91). Many studies with the aim of trying to explain the susceptibility of breast cancer patients to these second primary cancers have been conducted. Some second primary cancers such as breast, lung, thyroid, esophagus, stomach, leukemia, and soft tissue sarcoma of the upper limbs and thorax are likely to develop due to being close to the radiotherapy site (10, 19, 92). The cytotoxic agents used in chemotherapy also increase the risk of leukemia after breast cancer. Genetic mutations such as mutations of BRCA, CDKN2A, PALB2, TP53, and CHEK2 genes are associated with second cancers of the ovary, breast, prostate, pancreas, melanoma, soft tissue sarcoma, osteosarcoma, leukemia, brain tumors, and others. A strong relationship between breast cancer and endometrial cancer has been proven, which can be attributed to breast cancer hormone therapy with tamoxifen and other factors, such as obesity and genetic mutations (10, 81, 93). The relationship between thyroid and breast cancer can be explained by predisposing factors such as genetics, obesity, hormonal factors, chemotherapy, low physical activity, alcohol, and radiotherapy (94). The etiological roles of obesity and alcohol and tobacco use in the development of second cancers are underestimated, while these factors play a greater role in the development of second primary cancers compared with genetics and treatment modalities. The most common cancers related to alcohol use include cancers of the oral cavity and pharynx, larynx, esophagus, liver, and breast. Moreover, obesity is a known risk factor for breast, colorectal, and kidney cancers (81, 93).
The present study is the first meta-analysis to investigate the risk of second cancers among women and men previously diagnosed with primary breast cancer by the site of second cancer, age at diagnosis of breast cancer, and time since diagnosis of breast cancer according to the IARC definition. Despite a large amount of research that has been done on the risk of developing second cancer among female breast cancer patients, few studies have been done on male breast cancer patients (45, 81, 95–98). According to the IARC definition and the inclusion criteria of this systematic review and meta-analysis, only three studies conducted among men (14, 66, 75) were eligible to be included in the study, which can reduce the reliability of the results among men. According to the results of the present study, breast cancer survivors among women and men have 27% and 28% extra risk of developing second primary cancers, respectively. This estimate is consistent with the reported risk in some previous studies among men (84) and women (10, 41, 59, 63). Based on our results and previous studies (7), premenopausal women (younger than 50 years) are at a higher risk of the occurrence of second cancers. Furthermore, the more time passes since breast cancer diagnosis, the more women are at risk of developing second primary cancer. However, based on the present results and the previous study (84), after 10 years from breast cancer diagnosis, men have a lower risk of developing second cancer compared with men diagnosed with breast cancer less than 10 years ago.
In the meta-analysis, in addition to high-quality studies, we also considered acceptable and low-quality studies. Meta-regression showed that the quality of the studies did not influence the estimated risk except in bladder and urinary tract cancer among female patients ≥50 years.
As a shortcoming of our study, researchers in this field are at risk of various types of bias due to differences among studies (such as populations, study designs, and settings) as well as unavoidable bias due to differences in the year the research was performed (due to advancement in breast cancer treatment and the possible change in the pattern of second cancers compared with previous decades). Therefore, we recommend that in the future, more research will be conducted that will also assess the mentioned factors.
Results indicate a higher risk of developing second primary non-breast cancers among male and female breast cancer patients. Therefore, this population might benefit from prevention and screening programs. To prevent and control second primary cancers, the results of our systematic review can help researchers to design studies on screening modalities and prevention programs concerning this issue.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
PP, AM, MM, and AB researched, screened, and drafted the manuscript. AK and SH analyzed the data. AM and NA-Y supervised the content. All authors contributed to the article and approved the submitted version.
The Meta-analysis internship program funded by the UOS Health Economics and Finance Research Group - University of Sharjah, Sharjah, United Arab Emirates lead to development and implementation of this research.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2022.1094136/full#supplementary-material
1. Smolarz B, Nowak AZ, Romanowicz H. Breast cancer–epidemiology, classification, pathogenesis and treatment (Review of literature). Cancers. (2022) 14(10):2569. doi: 10.3390/cancers14102569
2. Elemam O, Abdelkhalek S, Abdelmoety D, Baraka R, Yousef M. Multiple primary tumours, how frequent we can offer curative therapy? Asian Pacific J Cancer Care (2020) 5(2):71–8. doi: 10.31557/apjcc.2020.5.2.71-78
3. Ferlay J, Colombet M, Soerjomataram I, Parkin DM, Piñeros M, Znaor A, et al. Cancer statistics for the year 2020: An overview. Int J cancer. (2021) 149(4):778–89. doi: 10.1002/ijc.33588
4. Wu T-Y, Raghunathan V, Shi J, Hua W, Yu W, Deng A. Improving the outcomes of breast cancer in China: Physicians’ beliefs, recommendations, and practices for breast cancer screening. Asian Pacific J Cancer Care (2020) 5(4):251–8. doi: 10.31557/apjcc.2020.5.4.251-258
5. Richard TS, Ndopwang LC, Etet PFS, Kamdje AHN, Mohamadou H, Bruno TP, et al. Breast cancer awareness and detection of asymptomatic cases using breast palpation and fine-needle aspiration in bafoussam, Cameroon. Asian Pacific J Cancer Care (2020) 5(1):61–6. doi: 10.31557/apjcc.2020.5.1.61-66
6. Jemal A, Center MM, DeSantis C, Ward EM. Global patterns of cancer incidence and mortality rates and TrendsGlobal patterns of cancer. Cancer epidemiology Biomarkers Prev (2010) 19(8):1893–907. doi: 10.1158/1055-9965.EPI-10-0437
7. Molina-Montes E, Requena M, Sánchez-Cantalejo E, Fernández MF, Arroyo-Morales M, Espín J, et al. Risk of second cancers cancer after a first primary breast cancer: a systematic review and meta-analysis. Gynecologic Oncol (2015) 136(1):158–71. doi: 10.1016/j.ygyno.2014.10.029
8. Olayide A, Isiaka A, Ganiyu R, Samuel O, Halimat A, Julius O, et al. Demographic pattern, tumor size and stage of breast cancer in africa: A meta-analysis. Asian Pacific J Cancer Care (2021) 6(4):477–92. doi: 10.31557/apjcc.2021.6.4.477-492
9. Dores G, Schonfeld S, Chen J, Hodgson D, Fossa S, Storm H, et al. Long-term cause-specific mortality among 41,146 one-year survivors of Hodgkin lymphoma (HL). J Clin Oncol (2005) 23(16_suppl):6511–. doi: 10.1200/jco.2005.23.16_suppl.6511
10. Mellemkjær L, Friis S, Olsen JH, Scélo G, Hemminki K, Tracey E, et al. Risk of second cancer among women with breast cancer. Int J cancer. (2006) 118(9):2285–92. doi: 10.1002/ijc.21651
11. Sung H, Freedman RA, Siegel RL, Hyun N, DeSantis CE, Ruddy KJ, et al. Risks of subsequent primary cancers among breast cancer survivors according to hormone receptor status. Cancer. (2021) 127(18):3310–24. doi: 10.1002/cncr.33602
12. Molina-Montes E, Pollán M, Payer T, Molina E, Dávila-Arias C, Sánchez MJ. Risk of second primary cancer among women with breast cancer: A population-based study in Granada (Spain). Gynecologic Oncol (2013) 130(2):340–5. doi: 10.1016/j.ygyno.2013.04.057
13. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA: Cancer J Clin (2014) 64(1):9–29. doi: 10.3322/caac.21208
14. Group AW. Italian Cancer figures, report 2013: Multiple tumours. Epidemiologia e prevenzione (2013) 37(4-5 Suppl 1):1–152. doi: 10.1038/s41416-022-01940-1
15. Ricceri F, Fasanelli F, Giraudo MT, Sieri S, Tumino R, Mattiello A, et al. Risk of second primary malignancies in women with breast cancer: Results from the European prospective investigation into cancer and nutrition (EPIC). Int J cancer. (2015) 137(4):940–8. doi: 10.1002/ijc.29462
16. Silverman BG, Lipshitz I, Keinan-Boker L. Second primary cancers after primary breast cancer diagnosis in Israeli women, 1992 to 2006. J Global Oncol (2017) 3(2):135–42. doi: 10.1200/JGO.2016.003699
17. Bright CJ, Reulen RC, Winter DL, Stark DP, McCabe MG, Edgar AB, et al. Risk of subsequent primary neoplasms in survivors of adolescent and young adult cancer (Teenage and young adult cancer survivor study): a population-based, cohort study. Lancet Oncol (2019) 20(4):531–45. doi: 10.1016/S1470-2045(18)30903-3
18. Bao S, Jiang M, Wang X, Hua Y, Zeng T, Yang Y, et al. Nonmetastatic breast cancer patients subsequently developing second primary malignancy: A population-based study. Cancer Med (2021) 10(23):8662–72. doi: 10.1002/cam4.4351
19. Hoekstra N, Fleury E, Merino Lara TR, van der Baan P, Bahnerth A, Struik G, et al. Long-term risks of secondary cancer for various whole and partial breast irradiation techniques. Radiotherapy Oncol J Eur Soc Ther Radiol Oncol (2018) 128(3):428–33. doi: 10.1016/j.radonc.2018.05.032
20. Korde LA, Doody DR, Hsu L, Porter PL, Malone KE. Bisphosphonate use and risk of recurrence, second primary breast cancer, and breast cancer mortality in a population-based cohort of breast cancer patients. Cancer epidemiology Biomarkers Prev Publ Am Assoc Cancer Research cosponsored by Am Soc Prev Oncol (2018) 27(2):165–73. doi: 10.1158/1055-9965.EPI-17-0556
21. Kushner CJ, Hwang WT, Wang S, Solin LJ, Vapiwala N. Long-term risk of second malignancies in women after breast conservation therapy for ductal carcinoma in situ or early-stage breast cancer. Breast Cancer Res Treat (2018) 170(1):45–53. doi: 10.1007/s10549-018-4729-7
22. Xie LY, Lin C, Zhang H, Bao XH. Second malignancy in young early-stage breast cancer patients with modern radiotherapy: A long-term population-based study (A STROBE-compliant study). Medicine. (2018) 97(17):e0593. doi: 10.1097/MD.0000000000010593
23. Jabagi MJ, Vey N, Goncalves A, Le Tri T, Zureik M, Dray-Spira R. Risk of secondary hematologic malignancies associated with breast cancer chemotherapy and G-CSF support: A nationwide population-based cohort. Int J cancer. (2021) 148(2):375–84. doi: 10.1002/ijc.33216
24. van Seijen M, Lips EH, Fu L, Giardiello D, van Duijnhoven F, de Munck L, et al. Long-term risk of subsequent ipsilateral lesions after surgery with or without radiotherapy for ductal carcinoma in situ of the breast. Br J cancer. (2021) 125(10):1443–9. doi: 10.1038/s41416-021-01496-6
25. Schwartz AG, Ragheb NE, Swanson GM, Satariano WA. Racial and age differences in multiple primary cancers after breast cancer: A population-based analysis. Breast Cancer Res Treat (1989) 14(2):245–54. doi: 10.1007/BF01810741
26. Suris-Swartz PJ, Schildkraut JM, Vine MF, Hertz-Picciotto I. Age at diagnosis and multiple primary cancers of the breast and ovary. Breast Cancer Res Treat (1996) 41(1):21–9. doi: 10.1007/BF01807033
27. Prochazka M, Hall P, Granath F, Czene K. Family history of breast cancer and young age at diagnosis of breast cancer increase risk of second primary malignancies in women: A population-based cohort study. Br J cancer. (2006) 95(9):1291–5. doi: 10.1038/sj.bjc.6603404
28. Verkooijen HM, Fioretta G, Rapiti E, Vlastos G, Neyroud-Caspar I, Chappuis PO, et al. Family history of breast or ovarian cancer modifies the risk of secondary leukemia after breast cancer: results from a population-based study. Int J Cancer. (2008) 122(5):1114–7. doi: 10.1002/ijc.23212
29. Zheng G, Sundquist J, Sundquist K, Ji J. Family history of breast cancer as a second primary malignancy in relatives: a nationwide cohort study. BMC cancer. (2021) 21(1):1210. doi: 10.1186/s12885-021-08925-y
30. Brooke BS, Schwartz TA, Pawlik TM. MOOSE reporting guidelines for meta-analyses of observational studies. JAMA surgery. (2021) 156(8):787–8. doi: 10.1001/jamasurg.2021.0522
31. Cancer IAfRo, Organization WH. International association of cancer registries (IACR), European network of cancer registries (ENCR). In: International rules for multiple primary cancers (ICD-O-Third edition). Lyon: IARC (2000).
32. Megdal SP, Kroenke CH, Laden F, Pukkala E, Schernhammer ES. Night work and breast cancer risk: A systematic review and meta-analysis. Eur J cancer. (2005) 41(13):2023–32. doi: 10.1016/j.ejca.2005.05.010
34. Tang J-L, Liu JL. Misleading funnel plot for detection of bias in meta-analysis. J Clin Epidemiol (2000) 53(5):477–84. doi: 10.1016/S0895-4356(99)00204-8
35. Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. Bmj. (1997) 315(7109):629–34. doi: 10.1136/bmj.315.7109.629
36. Andersson M, Jensen MB, Engholm G, Henrik Storm H. Risk of second primary cancer among patients with early operable breast cancer registered or randomised in Danish breast cancer cooperative group (DBCG) protocols of the 77, 82 and 89 programmes during 1977-2001. Acta Oncol (Stockholm Sweden). (2008) 47(4):755–64. doi: 10.1080/02841860801978921
37. Beadle G, Baade P, Fritschi L. Acute myeloid leukemia after breast cancer: a population-based comparison with hematological malignancies and other cancers. Ann Oncol Off J Eur Soc Med Oncol (2009) 20(1):103–9. doi: 10.1093/annonc/mdn530
38. Brown LM, Chen BE, Pfeiffer RM, Schairer C, Hall P, Storm H, et al. Risk of second non-hematological malignancies among 376,825 breast cancer survivors. Breast Cancer Res Treat (2007) 106(3):439–51. doi: 10.1007/s10549-007-9509-8
39. Bryk S, Pukkala E, Färkkilä A, Heikinheimo M, Unkila-Kallio L, Riska A. Other primary malignancies among women with adult-type ovarian granulosa cell tumors. Int J gynecological Cancer Off J Int Gynecological Cancer Society. (2018) 28(8):1529–34. doi: 10.1097/IGC.0000000000001333
40. Buiatti E, Crocetti E, Acciai S, Gafà L, Falcini F, Milandri C, et al. Incidence of second primary cancers in three Italian population-based cancer registries. Eur J Cancer (Oxford Engl 1990). (1997) 33(11):1829–34. doi: 10.1016/S0959-8049(97)00173-1
41. Cluze C, Delafosse P, Seigneurin A, Colonna M. Incidence of second cancer within 5 years of diagnosis of a breast, prostate or colorectal cancer: a population-based study. Eur J Cancer Prev Off J Eur Cancer Prev Organisation (ECP). (2009) 18(5):343–8. doi: 10.1097/CEJ.0b013e32832abd76
42. Corso G, Veronesi P, Santomauro GI, Maisonneuve P, Morigi C, Peruzzotti G, et al. Multiple primary non-breast tumors in breast cancer survivors. J Cancer Res Clin Oncol (2018) 144(5):979–86. doi: 10.1007/s00432-018-2621-9
43. Cortesi L, De Matteis E, Rashid I, Cirilli C, Proietto M, Rivasi F, et al. Distribution of second primary malignancies suggests a bidirectional effect between breast and endometrial cancer a population-based study. Int J Gynecological Cancer. (2009) 19(8):1358–63. doi: 10.1111/IGC.0b013e3181b9f5d5
44. Crocetti E, Mattioli V, Buzzoni C, Franceschi S, Serraino D, Vaccarella S, et al. Risk of thyroid as a first or second primary cancer. a population-based study in Italy, 1998–2012. Cancer Med (2021) 10(19):6855–67. doi: 10.1002/cam4.4193
45. Dong C, Hemminki K. Second primary neoplasms in 633,964 cancer patients in Sweden, 1958–1996. Int J cancer. (2001) 93(2):155–61. doi: 10.1002/ijc.1317
46. Franceschi S, Levi F, Vecchia CLA, Randimbison L, Te VC. Second cancers following in situ carcinoma of the breast. Int J cancer. (1998) 77(3):392–5. doi: 10.1002/(SICI)1097-0215(19980729)77:3<392::AID-IJC14>3.0.CO;2-A
47. Howard RA, Gilbert ES, Chen BE, Hall P, Storm H, Pukkala E, et al. Leukemia following breast cancer: an international population-based study of 376,825 women. Breast Cancer Res Treat (2007) 105(3):359–68. doi: 10.1007/s10549-006-9460-0
48. Levi F, Randimbison L, Te VC, La Vecchia C. Second primary cancers in breast cancer patients in vaud, Switzerland. Cancer causes control CCC. (1998) 9(4):463–4. doi: 10.1023/a:1017159305119
49. Lin CY, Chen SH, Huang CC, Weng SF, Lee ST, Guo HR, et al. Risk of secondary cancers in women with breast cancer and the influence of radiotherapy: A national cohort study in Taiwan. Medicine. (2016) 95(49):e5556. doi: 10.1097/MD.0000000000005556
50. Lu YC, Lu CL, Chen YY, Chen PT, Lin MS, Chen W, et al. Trend of incidence of second primary malignancies following breast cancer in Taiwan: A 12-year nationwide cohort study. Breast J (2016) 22(3):360–2. doi: 10.1111/tbj.12582
51. Malmer B, Tavelin B, Henriksson R, Grönberg H. Primary brain tumours as second primary: a novel association between meningioma and colorectal cancer. Int J cancer. (2000) 85(1):78–81. doi: 10.1002/(SICI)1097-0215(20000101)85:1<78::AID-IJC14>3.0.CO;2-S
52. Marcheselli R, Marcheselli L, Cortesi L, Bari A, Cirilli C, Pozzi S, et al. Risk of second primary malignancy in breast cancer survivors: A nested population-based case-control study. J Breast Cancer. (2015) 18(4):378–85. doi: 10.4048/jbc.2015.18.4.378
53. Nielsen SF, Nordestgaard BG, Bojesen SE. Associations between first and second primary cancers: a population-based study. CMAJ. (2012) 184(1):E57–69. doi: 10.1503/cmaj.110167
54. Prochazka M, Granath F, Ekbom A, Shields PG, Hall P. Lung cancer risks in women with previous breast cancer. Eur J Cancer (Oxford Engl 1990). (2002) 38(11):1520–5. doi: 10.1016/S0959-8049(02)00089-8
55. Srinivasan R, Yang YX, Rubin SC, Morgan MA, Lewis JD. Women with a prior diagnosis of breast cancer are not at an increased risk for subsequent colorectal cancer. Am J gastroenterology. (2005) 100(12):2759–64. doi: 10.1111/j.1572-0241.2005.00316.x
56. Teppo L, Pukkala E, Saxén E. Multiple cancer–an epidemiologic exercise in Finland. J Natl Cancer Institute. (1985) 75(2):207–17. doi: 10.1093/jnci/75.2.207
57. Volk N, Pompe-Kirn V. Second primary cancers in breast cancer patients in Slovenia. Cancer causes control CCC. (1997) 8(5):764–70. doi: 10.1023/A:1018487506546
58. Prior P, Waterhouse JA. Multiple primary cancers of the breast and ovary. Br J cancer. (1981) 44(5):628–36. doi: 10.1038/bjc.1981.247
59. Murakami R, Hiyama T, Hanai A, Fujimoto I. Second primary cancers following female breast cancer in Osaka, Japan–a population-based cohort study. Japanese J Clin Oncol (1987) 17(4):293–302. doi: 10.1093/oxfordjournals.jjco.a039211
60. Brenner H, Siegle S, Stegmaier C, Ziegler H. Second primary neoplasms following breast cancer in saarland, Germany, 1968-1987. Eur J Cancer (Oxford Engl 1990) (1993) 29a(10):1410–4. doi: 10.1016/0959-8049(93)90013-6
61. Rubino C, de Vathaire F, Diallo I, Shamsaldin A, Lê MG. Increased risk of second cancers following breast cancer: role of the initial treatment. Breast Cancer Res Treat (2000) 61(3):183–95. doi: 10.1023/A:1006489918700
62. Evans HS, Lewis CM, Robinson D, Bell CM, Møller H, Hodgson SV. Incidence of multiple primary cancers in a cohort of women diagnosed with breast cancer in southeast England. Br J cancer. (2001) 84(3):435–40. doi: 10.1054/bjoc.2000.1603
63. Tanaka H, Tsukuma H, Koyama H, Kinoshita Y, Kinoshita N, Oshima A. Second primary cancers following breast cancer in the Japanese female population. Japanese J Cancer Res Gann. (2001) 92(1):1–8. doi: 10.1111/j.1349-7006.2001.tb01040.x
64. Levi F, Te VC, Randimbison L, La Vecchia C. Cancer risk in women with previous breast cancer. Ann Oncol Off J Eur Soc Med Oncol (2003) 14(1):71–3. doi: 10.1093/annonc/mdg028
65. Sadetzki S, Calderon-Margalit R, Peretz C, Novikov I, Barchana M, Papa MZ. Second primary breast and thyroid cancers (Israel). Cancer Causes Control. (2003) 14(4):367–75. doi: 10.1023/A:1023908509928
66. Hemminki K, Scélo G, Boffetta P, Mellemkjaer L, Tracey E, Andersen A, et al. Second primary malignancies in patients with male breast cancer. Br J cancer. (2005) 92(7):1288–92. doi: 10.1038/sj.bjc.6602505
67. Lee KD, Chen SC, Chan CH, Lu CH, Chen CG, Lin JT, et al. Increased risk for second primary malignancies in women with breast cancer diagnosed at young age: A population-based study in Taiwan. Cancer Epidemiol Biomarkers Prev (2008) 17(10):2647–55. doi: 10.1158/1055-9965.EPI-08-0109
68. Schaapveld M, Visser O, Louwman MJ, de Vries EG, Willemse PH, Otter R, et al. Risk of new primary nonbreast cancers after breast cancer treatment: A Dutch population-based study. J Clin Oncol Off J Am Soc Clin Oncol (2008) 26(8):1239–46. doi: 10.1200/JCO.2007.11.9081
69. Gulhan I, Eser S, Yakut C, Bige O, Ilhan E, Yildirim Y, et al. Second primary gynecologic cancers after breast cancer in Turkish women. Int J gynecological Cancer Off J Int Gynecological Cancer Society. (2009) 19(4):648–50. doi: 10.1111/IGC.0b013e3181a12e8b
70. Fallah M, Kharazmi E, Sundquist J, Hemminki K. Nonendocrine cancers associated with benign and malignant parathyroid tumors. J Clin Endocrinol Metab (2011) 96(7):E1108–14. doi: 10.1210/jc.2011-0099
71. Mellemkjær L, Christensen J, Frederiksen K, Pukkala E, Weiderpass E, Bray F, et al. Risk of primary non–breast cancer after female breast cancer by age at DiagnosisCancer after breast cancer. Cancer epidemiology Biomarkers Prev (2011) 20(8):1784–92. doi: 10.1158/1055-9965.EPI-11-0009
72. Levi F, Randimbison L, Maspoli-Conconi M, Blanc-Moya R, La Vecchia C. Incidence of second sarcomas: a cancer registry-based study. Cancer Causes Control. (2014) 25(4):473–7. doi: 10.1007/s10552-014-0349-7
73. Utada M, Ohno Y, Hori M, Soda M. Incidence of multiple primary cancers and interval between first and second primary cancers. Cancer Science. (2014) 105(7):890–6. doi: 10.1111/cas.12433
74. An JH, Hwangbo Y, Ahn HY, Keam B, Lee KE, Han W, et al. A possible association between thyroid cancer and breast cancer. Thyroid Off J Am Thyroid Assoc (2015) 25(12):1330–8. doi: 10.1089/thy.2014.0561
75. Hung MH, Liu CJ, Teng CJ, Hu YW, Yeh CM, Chen SC, et al. Risk of second non-breast primary cancer in Male and female breast cancer patients: A population-based cohort study. PloS One (2016) 11(2):e0148597. doi: 10.1371/journal.pone.0148597
76. Bazire L, De Rycke Y, Asselain B, Fourquet A, Kirova YM. Risks of second malignancies after breast cancer treatment: Long-term results. Cancer radiotherapie J la Societe francaise radiotherapie oncologique. (2017) 21(1):10–5. doi: 10.1016/j.canrad.2016.07.101
77. Chen MC, Lee KD, Lu CH, Wang TY, Huang SH, Chen CY. The bidirectional association among female hormone-related cancers: breast, ovary, and uterine corpus. Cancer Med (2018) 7(6):2299–306. doi: 10.1002/cam4.1473
78. Jabagi MJ, Vey N, Goncalves A, Le Tri T, Zureik M, Dray-Spira R. Evaluation of the incidence of hematologic malignant neoplasms among breast cancer survivors in France. JAMA network Open (2019) 2(1):e187147. doi: 10.1001/jamanetworkopen.2018.7147
79. Sundboll J, Farkas DK, Adelborg K, Schapira L, Tamang S, Norgaard M, et al. Risk of primary urological and genital cancers following incident breast cancer: a Danish population-based cohort study. Breast Cancer Res Treat (2020) 184(3):825–37. doi: 10.1007/s10549-020-05879-w
80. Hill BT. Etiology of cancer. Clin Ophthalmic Oncology: Springer; (2019) p:11–7. doi: 10.1007/978-3-030-04489-3_2
81. Sung H, Hyun N, Leach CR, Yabroff KR, Jemal A. Association of first primary cancer with risk of subsequent primary cancer among survivors of adult-onset cancers in the united states. Jama. (2020) 324(24):2521–35. doi: 10.1001/jama.2020.23130
82. Nicolini A, Ferrari P, Duffy MJ. Prognostic and predictive biomarkers in breast cancer: Past, present and future. In Seminars in cancer biology. (2018) 52:56–17. doi: 10.1016/j.semcancer.2017.08.010
83. Yadav S, Karam D, Bin Riaz I, Xie H, Durani U, Duma N, et al. Male Breast cancer in the united states: Treatment patterns and prognostic factors in the 21st century. Cancer. (2020) 126(1):26–36. doi: 10.1002/cncr.32472
84. Allen I, Hassan H, Sofianopoulou E, Eccles D, Turnbull C, Tischkowitz M, et al. Risk of developing a second primary cancer in male breast cancer survivors: A systematic review and meta-analysis. Br J Cancer (2022) 127:1660–9. doi: 10.1038/s41416-022-01940-1
85. Adami H-O, Bergkvist L, Krusemo U, Persson I. Breast cancer as a risk factor for other primary malignant diseases. a nationwide cohort study. J Natl Cancer Institute. (1984) 73(5):1049–55. doi: 10.1093/jnci/73.5.1049
86. Brenner H, Siegle S, Stegmaier C, Ziegler H. Second primary neoplasms following breast cancer in saarland, Germany, 1968–1987. Eur J Cancer. (1993) 29(10):1410–4. doi: 10.1016/0959-8049(93)90013-6
87. Curtis RE, Hankey BF, Myers MH, Young JL Jr. Risk of leukemia associated with the first course of cancer treatment: an analysis of the surveillance, epidemiology, and end results program experience. J Natl Cancer Institute. (1984) 72(3):531–44. doi: 10.1093/jnci/72.3.531
88. Ewertz M, Mouridsen HT. Second cancer following cancer of the female breast in Denmark, 1943-80. Natl Cancer Institute Monograph. (1985) 68:325–9.
89. Harvey EB, Brinton LA. Second cancer following cancer of the breast in Connecticut, 1935-82. Natl Cancer Institute Monograph. (1985) 68:99–112.
90. Murakami R, Hiyama T, Hanai A, Fujimoto I. Second primary cancers following female breast cancer in Osaka, Japan–a population-based cohort study. Japanese J Clin Oncol (1987) 17(4):293–302. doi: 10.1093/oxfordjournals.jjco.a039211
91. Teppo L, Pukkala E, Saxén E. Multiple cancer–an epidemiologic exercise in Finland. J Natl Cancer Institute. (1985) 75(2):207–17. doi: 10.1093/jnci/75.2.207
92. Dracham CB, Shankar A, Madan R. Radiation induced secondary malignancies: a review article. Radiat Oncol J (2018) 36(2):85. doi: 10.3857/roj.2018.00290
93. Zhai C, Cai Y, Lou F, Liu Z, Xie J, Zhou X, et al. Multiple primary malignant tumors-a clinical analysis of 15,321 patients with malignancies at a single center in China. J cancer. (2018) 9(16):2795. doi: 10.7150/jca.25482
94. Bolf EL, Sprague BL, Carr FE. A linkage between thyroid and breast cancer: A common etiology? Metachronous Thyroid Breast Cancer. Cancer Epidemiology Biomarkers Prev (2019) 28(4):643–9. doi: 10.1158/1055-9965.EPI-18-0877
95. Cancer IAfRo. International rules for multiple primary cancers (ICD-O third edition). Lyon: IARC (2004).
96. Chen T, Fallah M, Jansen L, Castro FA, Krilavicuite A, Katalinic A, et al. Distribution and risk of the second discordant primary cancers combined after a specific first primary cancer in German and Swedish cancer registries. Cancer letters. (2015) 369(1):152–66. doi: 10.1016/j.canlet.2015.08.014
97. Jégu J, Colonna M, Daubisse-Marliac L, Trétarre B, Ganry O, Guizard A-V, et al. The effect of patient characteristics on second primary cancer risk in France. BMC cancer. (2014) 14(1):1–14. doi: 10.1186/1471-2407-14-94
Keywords: breast cancer, multiple primary cancer, second primary cancer, incidence, risk
Citation: Parhizgar P, Bahadori Monfared A, Mohseny M, Keramatinia A, Hashemi Nazari SS, Rahman SA, Al Marzouqi A, Al-Yateem N and Mosavi Jarrahi A (2023) Risk of second primary cancer among breast cancer patients: A systematic review and meta-analysis. Front. Oncol. 12:1094136. doi: 10.3389/fonc.2022.1094136
Received: 09 November 2022; Accepted: 20 December 2022;
Published: 17 January 2023.
Edited by:
Nosheen Masood, Fatima Jinnah Women University, PakistanReviewed by:
Olivera Ivanov, University of Novi Sad, SerbiaCopyright © 2023 Parhizgar, Bahadori Monfared, Mohseny, Keramatinia, Hashemi Nazari, Rahman, Al Marzouqi, Al-Yateem and Mosavi Jarrahi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Nabeel Al-Yateem, bmFseWF0ZWVtQHNoYXJqYWguYWMuYWU=; Alireza Mosavi Jarrahi, cm1vc2F2aUB5YWhvby5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.