
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Oncol. , 21 July 2021
Sec. Gynecological Oncology
Volume 11 - 2021 | https://doi.org/10.3389/fonc.2021.716467
This article is part of the Research Topic Molecular Genetics and Emerging Therapies for Epithelial Ovarian Cancer: Basic Research and Clinical Perspectives View all 29 articles
 Vincenza Conteduca1*
Vincenza Conteduca1* Emanuela Scarpi2
Emanuela Scarpi2 Alberto Farolfi1
Alberto Farolfi1 Nicole Brighi1
Nicole Brighi1 Lorena Rossi1
Lorena Rossi1 Giorgia Gurioli3
Giorgia Gurioli3 Cristian Lolli1
Cristian Lolli1 Giuseppe Schepisi1
Giuseppe Schepisi1 Sara Bleve1
Sara Bleve1 Caterina Gianni1
Caterina Gianni1 Alessandra Virga3
Alessandra Virga3 Amelia Altavilla1
Amelia Altavilla1 Salvatore Luca Burgio1
Salvatore Luca Burgio1 Cecilia Menna1
Cecilia Menna1 Ugo De Giorgi1
Ugo De Giorgi1Introduction: Melphalan, as a bifunctional alkylating agent has been shown to be selectively efficient in BRCA-deficient case reports of epithelial ovarian cancer (EOC). The clinical benefit of melphalan on unselected platinum-resistant EOC population and stratified by BRCA status has not been clearly elucidated. We aimed to determine the response to melphalan in patients with recurrent EOC after platinum-based therapy.
Material and Methods: This retrospective observational study included patients with recurrent EOC treated with melphalan between February 2007 to July 2020. Eligibility criteria included having a histological confirmation of EOC, previous treatment with carboplatin plus paclitaxel regimens, and disease recurrence during treatment with or within 6 months of the end of the platinum-based chemotherapy.
Results: A total of 75 platinum-resistant EOC patients were enrolled. Median age was 69 years (range 41-82). Median of previous therapies before melphalan was 4 (range 1-7). We observed a median follow-up of 32 months (range 1-62), progression-free survival (PFS) and overall survival (OS) of 3.6 months (range 2.9-4.7) and 9.5 months (range 8.0-14.1), respectively. In the whole population, 1 complete response, 6 partial responses and 37 stable diseases were registered with an overall clinical benefit rate of 58.7%. In BRCA1/2 mutant patients, we showed a significant longer PFS compared to BRCA1/2 wild type patients (6.2 versus 2.6 months; hazard ratio (HR) 0.25, 95% confidence interval (CI) 0.10-0.61; p=0.002). Moreover, a trend was seen for BRCA1/2 mutants to have a better OS (25.9 versus 8.0 months; HR 0.38; 95% CI 0.12-1.19; p=0.097).
Conclusions: Our study represents the largest cohort of heavily-pretreated EOC patients receiving melphalan treatment. Here, we report a considerable clinical activity of melphalan chemotherapy, more evident in a subset of BRCA1/2 mutated patients. Prospective studies to validate these findings are warranted.
Ovarian cancer is a leading cause of death from gynaecologic cancers worldwide (1). Despite optimal debulking surgery, appropriate first-line chemotherapy based on taxane-platinum doublets and combination/maintenance therapy with bevacizumab or poly(adenosine diphosphate-ribose) polymerase inhibitors (PARPi), approximately 60-70% of patients eventually relapse (2, 3). For recurrent patients, a rate of response more than 60% is reported in platinum-sensitive patients (occurring at least 6 months after last treatment completion) receiving platinum-based combination chemotherapy, whilst the response rate dramatically drops to less than 20% (4) in platinum-resistant women (recurring within 6 months after the last therapy) who can receive several drugs characterized by different mechanisms of action and, in general, a modest activity, such as topotecan (5), gemcitabine (6), liposomal doxorubicin (7), oral etoposide (8), ifosfamide (9) and oxaliplatin plus leucovorin and 5-fluorouracil (FOLFOX-4) (10). Thus, there is an urgent need to identify agents active in this group of EOC platinum-resistant patients.
Melphalan is a nitrogen mustard-like alkylating agent, administered orally or parenterally and mainly used for the treatment of multiple myeloma. Very little information is available regarding the use of melphalan for the treatment of epithelial ovarian cancer (EOC), thus providing controversial results to date (11, 12). Additionally, melphalan, considered as a bifunctional alkylating agent that induces inter- and intra-strand DNA cross-links, has been shown to be selectively efficient in BRCA-defıcient case reports of EOC (13, 14) (Figure 1).
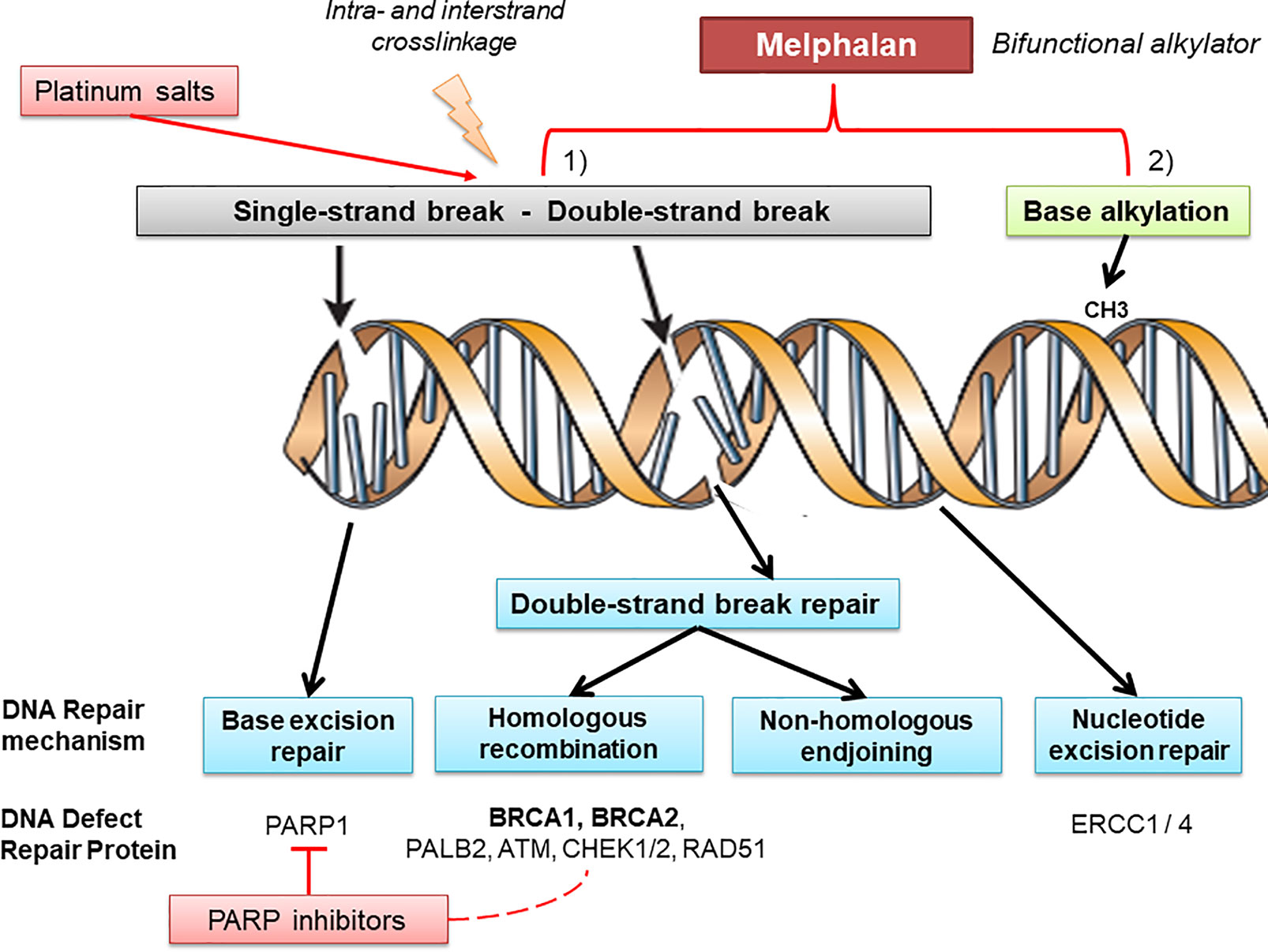
Figure 1 Melphalan and DNA repair mechanisms. DNA is continually exposed to a series of insults that cause a range of lesions, from single-strand breaks to base alkylation events. Several mechanisms of DNA repair (such as base excision repair, homologous recombination, non-homologous endjoining, and nucleotide excision repair) can be involved recruiting different key proteins which belong to pathways used in the therapeutic strategy in ovarian cancer. Alkylating agents, platinum salts, and PARP inhibitors are particularly effective in DNA defect repair deficient tumors, albeit through different molecular mechanisms. Melphalan is a bifunctional alkylating agent that produces intra- and inter-strand cross-links in double-strand DNA and provides base alkylation, whereas platinum mainly generates intra-strand crosslinks through platinum coordinated complexes and PARP1 inhibitors block base excision repair leading to single-strand breaks. As a different spectrum of DNA damage is produced by each drug, it is possible that the DNA damage produced by melphalan may be more reliant on BRCA protein products for repair.
Here we evaluated the efficacy and toxicity of melphalan in heavily-pre-treated platinum-resistant EOC. In addition, through an exploratory analysis, we aimed to show an increased sensitivity to melphalan in patients harbouring BRCA1/2-defıciency.
This is a retrospective single-centre case series of patients with ovarian cancer receiving melphalan from February 2007 to July 2020. Eligibility criteria were: histological confirmation of epithelial ovarian cancer, previous treatment with carboplatin plus paclitaxel regimen, and disease recurrence during treatment within 6 months from the end of the platinum-based chemotherapy. Additional eligibility criteria included: Eastern Cooperative Oncology Group (ECOG) performance status 0–2, and adequate cardiac, renal, hepatic and bone marrow function. Metastatic disease was documented by bone scan, computed tomography or magnetic resonance imaging. For each patient, we extracted clinical-pathologic features, treatment history and outcomes with follow-up data from medical records. Moreover, we collected molecular data including somatic and/or germ line BRCA1/2 status from each subject, when available. The study was approved by the Institutional Review Board of IRCCS Istituto Romagnolo per lo Studio dei Tumori (IRST) “Dino Amadori”, Meldola, Italy.
Melphalan (2 mg/tablet, Alkeran™; Excella GmbH; Feucht, Germany) was orally administered at dosage of 0,20 mg/Kg daily for 5 consecutive days monthly until evidence of either progressive disease (PD) or unacceptable toxicity.
Patients were evaluated monthly for safety and dosing compliance. Renal, liver and bone marrow function were assessed at every cycle, while cancer antigen 125 (CA-125) and radiographic evaluation were left to the discretion of the treating physician, but were usually performed after at least every three months during treatment.
Tumour response was usually evaluated every three cycles by repeating baseline assessments using imaging studies (computed tomography and/or magnetic resonance imaging) according to the Response Evaluation Criteria in Solid Tumours (RECIST) for patients with measurable disease (15). CA-125 was evaluated in recurrent disease using CA-125 response criteria developed by the Gynaecologic Cancer InterGroup (16). Toxicity was graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), version 4 (17).
All data were analyzed by descriptive statistics. Relationships between patients’ characteristics were tested using the Chi-square test for categorical variables and the median test for continuous variables. The Kaplan-Meier method was used to estimate progression-free survival (PFS) and overall survival (OS), with two-sided 95% confidence intervals (95% CI). PFS was defined as the time from the start of melphalan until disease progression or last tumour evaluation or death from any cause. OS was defined as the time from the start of melphalan until death from any cause or last follow-up. Survival curves were compared using the log-rank test. All statistical analyses were carried out with SAS statistical software, version 9.4 (SAS Institute, Cary, NC, USA). A two-sided p-value < 0.05 was deemed statistically significant for all the analyses.
Between February 2007 and July 2020, a total of 75 patients were eligible. Median age was 69 years (range 41-82). Median of previous therapies before melphalan was 4 (range 1-7). At the time of study entry, all patients were defined as resistant to the last platinum treatment. We excluded EOC patients receiving concurrent use of other anticancer agents or treatments. The majority of the patients (~90%) had high-grade serous type. Forty-three patients (57.3%) had initial International Federation of Gynecology and Obstetrics (FIGO) stage III, and 66 (88.0%) underwent primary or interval debulking surgery. Patient characteristics are summarized in Table 1.
The median follow-up was 32 months (range 1-62). All patients had measurable disease. In the whole population, 1 complete response (CR), 6 partial responses (PR) and 37 stable diseases (SD) were registered with an overall clinical benefit rate (CR+PR+SD) of 58.7%. A CA-125 response was observed in 20 (26.7%) melphalan-treated patients and it was not associated with objective radiological response (CR or PR).
Median PFS and OS were of 3.6 months (range 2.9-4.7) and 9.5 months (range 8.0-14.1), respectively (Table 1). BRCA status was associated with outcomes at univariate analysis. On the other hand, univariate analyses did not identify any other significant factors (including age, debulking surgery, FIGO stage, ECOG performance status, pre-treatment CA-125, hemoglobin, neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) predicting PFS and OS (Table 2).
We studied the association of BRCA1/2 status with melphalan treatment. In EOC patients with available molecular data, when comparing baseline characteristics of BRCA1/2 mutant to BRCA1/2 wild type patients, no differences were observed (Supplementary Table 1).
Based on BRCA mutational status, we reported in 11 BRCA1/2 mutated patients an overall response rate (ORR) (CR+PR) of 18.2%, SD and PD in 36.4% and 45.4% of cases, respectively; whereas 25 BRCA1/2 wild type patients had ORR of 4%, SD and PD in 36% and 60% of cases, respectively.
In BRCA1/2 mutant EOC receiving melphalan, we observed a significant longer PFS compared to BRCA1/2 wild type patients (median, 6.2 versus 2.6 months; hazard ratio (HR) 0.25, 95% confidence interval (CI) 0.10-0.61; p = 0.002) (Figure 2A). Moreover, a trend was seen for BRCA1/2 mutants to have a better OS (median, 25.9 versus 8.0 months; HR 0.38; 95% CI 0.12-1.19; p = 0.097) (Figure 2B).
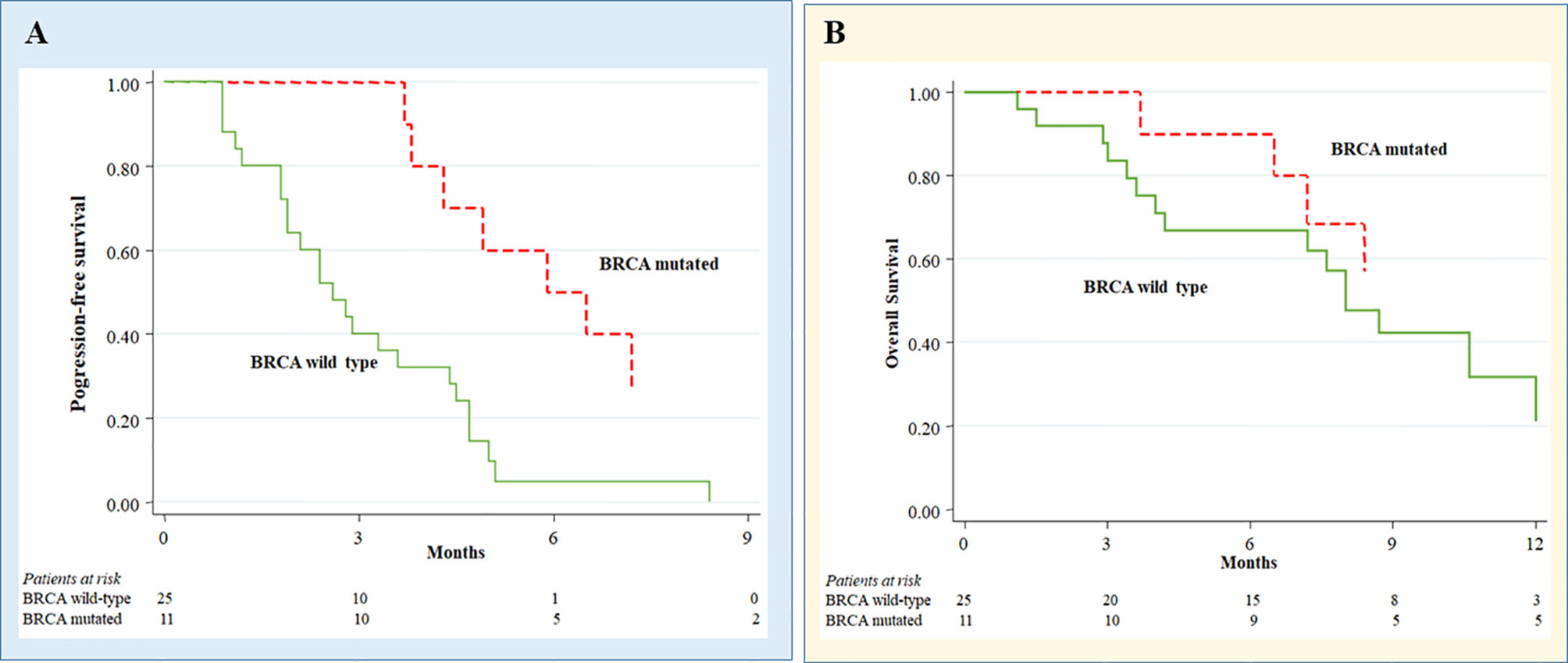
Figure 2 Melphalan treatment outcomes in ovarian cancer patients according to BRCA status. Progression-free survival (A) and Overall survival (B) in melphalan-treated ovarian cancer patients according to BRCA status.
Thirty-two patients (42.7%) were treated with initial standard melphalan regimen. In 21 patients (28%), the dose of melphalan was reduced by at least 75%, and in more than 80% of cases, reduction was required because of hematologic toxicity. However, only 4 (5.3%) patients receiving melphalan discontinued treatment because of unacceptable toxicity (two grade 3 thrombocytopenia, one grade 2 anemia, and one grade 4 neutropenia).
The hematological and non-hematological adverse events occurred in our series are reported in Table 3. As expected, myelotoxicity was the prevalent toxicity with 17.3% of patient experiencing grade 3-4 thrombocytopenia, 13.4% and 6.7% of patients reporting grade 3-4 neutropenia and anemia, respectively. Nausea and vomiting and fatigue were the most frequent non-hematologic events (grade 3: 2.7% and 4% of patients, respectively). Neither cardiac and renal toxicities nor treatment-related deaths were reported.
There was no difference of hematological toxicity related to melphalan treatment between BRCA mutants versus wild-type patients.
Our study represents the largest cohort of EOC patients treated with melphalan reported to date. Currently, the management of platinum-resistant EOC represents one of the most important unmet medical issues. Despite great research endeavors over the last decades, standard treatments have often inadequate clinical benefit. Here, we report a considerable clinical activity of melphalan chemotherapy in this difficult-to-treat patients group. For exploratory purposes, we also investigated a subset of BRCA1/2 mutated patients showing improved outcomes and enhanced sensitivity to melphalan chemotherapy compared to BRCA1/2 wild type women.
An overall clinical benefit of 58.7% in a population who has received a median of four previous chemotherapy lines appears as a notable result. In heavily-pre-treated platinum-resistant EOC patients with a median of four prior therapeutic lines, the overall clinical benefit observed in this work was slightly higher than that observed in our previous retrospective experience of women receiving FOLFOX-4 and topotecan as salvage chemotherapy lines (30.8% and 48.3%, respectively) (10). A multicenter retrospective study showed that weekly paclitaxel monochemotherapy had similar clinical benefit rate of 36%, with a median PFS of 21 weeks (18), which is consistent with the PFS of 4.7–5.3 months in the SaPPrOC trial (19). Both these studies suggested a therapeutic role of weekly paclitaxel in patients with EOC regardless of BRCA1/2 status. Similar evidence was reported in the prospective MITO-15 phase II trial (20) in which trabectedin showed a very similar clinical benefit (54.2%) to melphalan, and no differences in treatment outcomes were observed according to BRCA1/2 status (20).
According to these findings, the outcomes of melphalan treatment were quite similar to those reported with other drugs for heavily pre-treated platinum-resistant EOC patients. However, there is increasing evidence to consider BRCA mutation status when selecting not only PARPi agents but also chemotherapy regimens, such as melphalan treatment. In support of this conjecture, melphalan was shown to be selectively toxic to BRCA2-deficient breast cancer cell lines and to produce a longer relapse-free survival in mice than platinum or olaparib (21).
Overall, we recognize some limitations of our study such as the relatively modest sample size, influencing the statistical significance especially for OS, the clinical and histological heterogeneity of the patients’ cohort and its retrospective, non-randomized design. In addition, several studies showed that BRCA mutant patients have, in general, better prognosis, likely due to the high response rate to platinum-based chemotherapy. This aspect could be a confounding factor in the interpretation of our survival data in melphalan-treated patients according to BRCA status; however, the exact effect of BRCA1/2 mutations on EOC prognosis is still controversial (22–24). Lastly, we have considered only BRCA1/2 mutations and not other alterations in DNA defect repair genes and no patient did prior therapy with PARPi. Nevertheless, our preliminary results suggest that BRCA status is associated with sensitivity to melphalan therapy. Thus, since BRCA-related EOC represents a distinct entity within the ovarian cancer spectrum, developing a subtype-specific treatment tailored to the unique cancer biology of ‘BRCA-pathway’ ovarian tumours (arising from germ-line or somatic BRCA mutations) may lead to an improved disease management. Some preclinical studies demonstrated a decreased likelihood of response to subsequent chemotherapy following olaparib treatment due to the development of cross-resistance between PARPi and platinum-based regimens through the acquisition of secondary mutations restoring BRCA1/2 protein expression (25, 26). In this context, the availability of other active DNA alkylating agents could constitute an additional therapeutic option for resistant or recurrent EOC BRCA1/2 mutant patients.
In heavily pre-treated EOC patients, melphalan chemotherapy is an effective and well-tolerated treatment. Discovering the underlying molecular mechanism of chemo-responsiveness could lead to subtype-specific treatment selection. This study supports the notion that the knowledge on BRCA status may improve clinical decision-making in choosing between different therapies for platinum-resistant EOC. Prospective trials including overall BRCA/Homologous recombination deficiency assessment are warranted.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving human participants were reviewed and approved by COMITATO ETICO della Romagna. The patients/participants provided their written informed consent to participate in this study.
VC was involved in the conception of the study, acquisition and analysis of the data, and wrote the first draft of the manuscript. AF, NB, LR, GG, CL, GS, SB, CG, AV, AA, SLB, and CM were involved in the acquisition of the data. VC and UDG were involved in the conception and design of the study. VC, ES, and UDG contributed to data analysis and interpretation of data. VC, ES, AA, NB, and UDG critically revised the manuscript for important intellectual content. VC and ES participated in analyzing the results and drafting the manuscript. All authors contributed to the article and approved the submitted version.
VC has served as consultant/advisory board member for Janssen, Astellas, Merck, AstraZeneca, Bayer; has received speaker honoraria or travel support from Astellas, Janssen, Ipsen, Bayer and Sanofi. UDG reports research support from AstraZeneca, Roche, and Sanofi; and consultancy fees from Astellas, Bayer, Bristol Myers Squibb, Ipsen, Janssen, Merck, Pfizer, and Sanofi.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors would like to thank Alessia Filograna at IRCCS Istituto Scientifico Romagnolo per lo Studio dei Tumori (IRST) “Dino Amadori” for her nursing support.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2021.716467/full#supplementary-material
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin (2021) 71:209–49. doi: 10.3322/caac.21660
2. Pignata S, Cecere S, Du Bois A, Harter P, Heitz F. Treatment of Recurrent Ovarian Cancer. Ann Oncol (2017) 28:viii51–6. doi: 10.1093/annonc/mdx441
3. Conteduca V, Kopf B, Burgio SL, Bianchi E, Amadori D, De Giorgi U. The Emerging Role of Anti-Angiogenic Therapy in Ovarian Cancer (Review). Int J Oncol (2014) 44:1417–24. doi: 10.3892/ijo.2014.2334
4. Hanker LC, Loibl S, Burchardi N, Pfisterer J, Meier W, Pujade-Lauraine E, et al. The Impact of Second to Sixth Line Therapy on Survival of Relapsed Ovarian Cancer After Primary Taxane/Platinum-Based Therapy. Ann Oncol (2012) 23:2605–12. doi: 10.1093/annonc/mds203
5. Bookman MA, Malmström H, Bolis G, Gordon A, Lissoni A, Krebs JB, et al. Topotecan for the Treatment of Advanced Epithelial Ovarian Cancer: An Open-Label Phase II Study in Patients Treated After Prior Chemotherapy That Contained Cisplatin or Carboplatin and Paclitaxel. J Clin Oncol (1998) 16:3345–452. doi: 10.1200/JCO.1998.16.10.3345
6. Takei Y, Takahashi Y, Machida S, Taneichi A, Takahashi S, Nagashima T, et al. Response to and Toxicity of Gemcitabine for Recurrent Ovarian Cancer According to Number of Previous Chemotherapy Regimens. J Obstet Gynaecol Res (2017) 43:358–64. doi: 10.1111/jog.13203
7. Markman M, Kennedy A, Webster K, Peterson G, Kulp B, Belinson J. Phase 2 Trial of Liposomal Doxorubicin (40 mg/m(2)) in Platinum/Paclitaxel-Refractory Ovarian and Fallopian Tube Cancers and Primary Carcinoma of the Peritoneum. Gynecol Oncol (2000) 78:369–72. doi: 10.1006/gyno.2000.5921
8. Kucukoner M, Isikdogan A, Yaman S, Gumusay O, Unal O, Ulas A, et al. Oral Etoposide for Platinum-Resistant and Recurrent Epithelial Ovarian Cancer: A Study by the Anatolian Society of Medical Oncology. Asian Pac J Cancer Prev (2012) 13:3973–6. doi: 10.7314/APJCP.2012.13.8.3973
9. Markman M, Kennedy A, Sutton G, Hurteau J, Webster K, Peterson G, et al. Phase 2 Trial of Single Agent Ifosfamide/Mesna in Patients With Platinum/Paclitaxel Refractory Ovarian Cancer Who Have Not Previously Been Treated With an Alkylating Agent. Gynecol Oncol (1998) 70:272–4. doi: 10.1006/gyno.1998.5065
10. Conteduca V, Gurioli G, Rossi L, Scarpi E, Lolli C, Schepisi G, et al. Oxaliplatin Plus Leucovorin and 5-Fluorouracil (FOLFOX-4) as a Salvage Chemotherapy in Heavily-Pretreated Platinum-Resistant Ovarian Cancer. BMC Cancer (2018) 18:1267. doi: 10.1186/s12885-018-5180-1
11. Hasan J, Jayson GC. Oral Melphalan as a Treatment for Platinum-Resistant Ovarian Cancer. Br J Cancer (2003) 88:1828–30. doi: 10.1038/sj.bjc.6601044
12. Davis-Perry S, Hernandez E, Houck KL, Shank R. Melphalan for the Treatment of Patients With Recurrent Epithelial Ovarian Cancer. Am J Clin Oncol (2003) 26:429–33. doi: 10.1097/01.COC.0000027269.06091.E9
13. Osher DJ, Kushner YB, Arseneau J, Foulkes WD. Melphalan as a Treatment for BRCA-Related Ovarian Carcinoma: Can You Teach an Old Drug New Tricks? J Clin Pathol (2011) 64:924–6. doi: 10.1136/jcp.2010.086405
14. Fan FS, Yang CF. Complete Response to Orally Administered Melphalan in Malignant Pleural Effusion From an Occult Female Genital Organ Primary Neoplasm With BRCA1/2 Mutations: A Case Report. J Med Case Rep (2018) 12:122. doi: 10.1186/s13256-018-1674-3
15. Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New Response Evaluation Criteria in Solid Tumours: Revised RECIST Guideline (Version 1.1). Eur J Cancer (2009) 452:228–47. doi: 10.1016/j.ejca.2008.10.026
16. Rustin GJ, Vergote I, Eisenhauer E, Pujade-Lauraine E, Quinn M, Thigpen T, et al. Definitions for Response and Progression in Ovarian Cancer Clinical Trials Incorporating RECIST 1.1 and CA 125 Agreed by the Gynecological Cancer Intergroup (GCIG). Int J Gynecol Cancer (2011) 21:419–23. doi: 10.1097/IGC.0b013e3182070f17
17. Cancer Therapy Evaluation Program. Common Terminology Criteria for Adverse Events, Version 3.0. Bethesda: National Cancer Institute (2003). Available at: http://ctep.cancer.gov/forms/CTCAEv4.pdf.
18. Tan DS, Yap TA, Hutka M, Roxburgh P, Ang J, Banerjee S, et al. Implications of BRCA1 and BRCA2 Mutations for the Efficacy of Paclitaxel Monotherapy in Advanced Ovarian Cancer. Eur J Cancer (2013) 49:1246–53. doi: 10.1016/j.ejca.2012.11.016
19. McNeish IA, Ledermann JA, Webber L, James L, Kaye SB, Hall M, et al. A Randomised, Placebo-Controlled Trial of Weekly Paclitaxel and Saracatinib (AZD0530) in Platinum-Resistant Ovarian, Fallopian Tube or Primary Peritoneal Cancer. Ann Oncol (2014) 25:1988–95. doi: 10.1093/annonc/mdu363
20. Lorusso D, Scambia G, Pignata S, Sorio R, Amadio G, Lepori S, et al. Prospective Phase II Trial of Trabectedin in BRCA-Mutated and/or BRCAness Phenotype Recurrent Ovarian Cancer Patients: The MITO 15 Trial. Ann Oncol (2016) 27:487–93. doi: 10.1093/annonc/mdv608
21. Evers B, Schut E, van der Burg E, Braumuller TM, Egan DA, Holstege H, et al. A High-Throughput Pharmaceutical Screen Identifies Compounds With Specific Toxicity Against BRCA2-deficient Tumors. Clin Cancer Res (2010) 16:99–108. doi: 10.1158/1078-0432.CCR-09-2434
22. McLaughlin JR, Rosen B, Moody J, Pal T, Fan I, Shaw PA, et al. Long-Term Ovarian Cancer Survival Associated With Mutation in BRCA1 or BRCA2. J Natl Cancer Inst (2013) 105:141–8. doi: 10.1093/jnci/djs494
23. Hyman DM, Zhou Q, Iasonos A, Grisham RN, Arnold AG, Phillips MF, et al. Improved Survival for BRCA2-Associated Serous Ovarian Cancer Compared With Both BRCA-Negative and BRCA1-associated Serous Ovarian Cancer. Cancer (2012) 118:3703–9. doi: 10.1002/cncr.26655
24. Yang D, Khan S, Sun Y, Hess K, Shmulevich I, Sood AK, et al. Association of BRCA1 and BRCA2 Mutations With Survival, Chemotherapy Sensitivity, and Gene Mutator Phenotype in Patients With Ovarian Cancer. JAMA (2011) 306:1557–65. doi: 10.1001/jama.2011.1456
25. Norquist B, Wurz KA, Pennil CC, Garcia R, Gross J, Sakai W, et al. Secondary Somatic Mutations Restoring BRCA1/2 Predict Chemotherapy Resistance in Hereditary Ovarian Carcinomas. J Clin Oncol (2011) 29:3008–15. doi: 10.1200/JCO.2010.34.2980
Keywords: melphalan, ovarian cancer, BRCA, platinum resistance, survival
Citation: Conteduca V, Scarpi E, Farolfi A, Brighi N, Rossi L, Gurioli G, Lolli C, Schepisi G, Bleve S, Gianni C, Virga A, Altavilla A, Burgio SL, Menna C and De Giorgi U (2021) Melphalan as a Promising Treatment for BRCA-Related Ovarian Carcinoma. Front. Oncol. 11:716467. doi: 10.3389/fonc.2021.716467
Received: 28 May 2021; Accepted: 06 July 2021;
Published: 21 July 2021.
Edited by:
Nicholas Pavlidis, University of Ioannina, GreeceReviewed by:
Sarah Taylor, University of Pittsburgh, United StatesCopyright © 2021 Conteduca, Scarpi, Farolfi, Brighi, Rossi, Gurioli, Lolli, Schepisi, Bleve, Gianni, Virga, Altavilla, Burgio, Menna and De Giorgi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Vincenza Conteduca, dmluY2VuemEuY29udGVkdWNhQGlyc3QuZW1yLml0
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.