- 1Parkview Cancer Institute, Fort Wayne, IN, United States
- 2Division of Medical Oncology, University of Kansas Medical Center, Fairway, KS, United States
- 3Subspecialty Medicine, Division of Hematology & Medical Oncology, Kansas City VA Medical Center, Kansas City, MO, United States
Recent application of immunotherapy in clinical oncology revolutionized our management of advanced human cancers. Check point inhibitors targeting CTLA4 and PD-1/PD-L1 axis are immunotherapeutic agents currently available to treat a variety of cancers. However, a novel therapeutic approach is needed to further improve patient outcome with these agents. Indoleamine 2,3-dioxygenase 1 (IDO1) is a rate-limiting enzyme in the metabolism of essential amino acid tryptophan in the peripheral tissue. IDO1 is overexpressed in human cancer cells and suppresses effector T cell function and promotes regulatory T cells (Tregs). Overexpression of IDO1 is associated with poor patient survival in several types of human cancer. These findings indicate that IDO1 is a promising target that can improve the treatment outcome in the field of Immuno-oncology. Several orally available IDO1 inhibitors including Epacadostat have entered human clinical trials over the last few years without a major safety concern. Although there is no objective response in single-agent trials, combination regimens with PD-1 inhibitors appear to exceed the activity of PD-1 inhibitors alone. Recent phase III ECHO 301 trial testing the combination of Epacadostat with Pembrolizumab in melanoma did not show superior outcome compared to Pembrolizumab alone. This lead to halting of other phase III trials using IDO1 inhibitors. In this minireview, we will discuss the recent clinical development of Epacadostat and other IDO1 inhibitors.
Introduction
Cancer remains one of the leading causes of mortality worldwide, affecting 8.8 million people annually (1). Immune checkpoint inhibitors (CPI) enhance the T-cell mediated anti-tumor effect by disrupting the interaction between the PD-1 receptor and PD-L1 ligand. They were recently approved for a wide variety of cancer types and changed the landscape of therapy in cancer (2). These include nivolumab (N)/pembrolizumab (P) (anti-PD-1) and atezolizumab/durvalumab (anti-PD-L1) (3–6). Ipilimumab is the only clinically available anti-CTLA4 inhibitor with FDA approval limited to melanoma (7). As more immune targets are identified, many novel immune therapies to enhance T-cell function entered clinical trials.
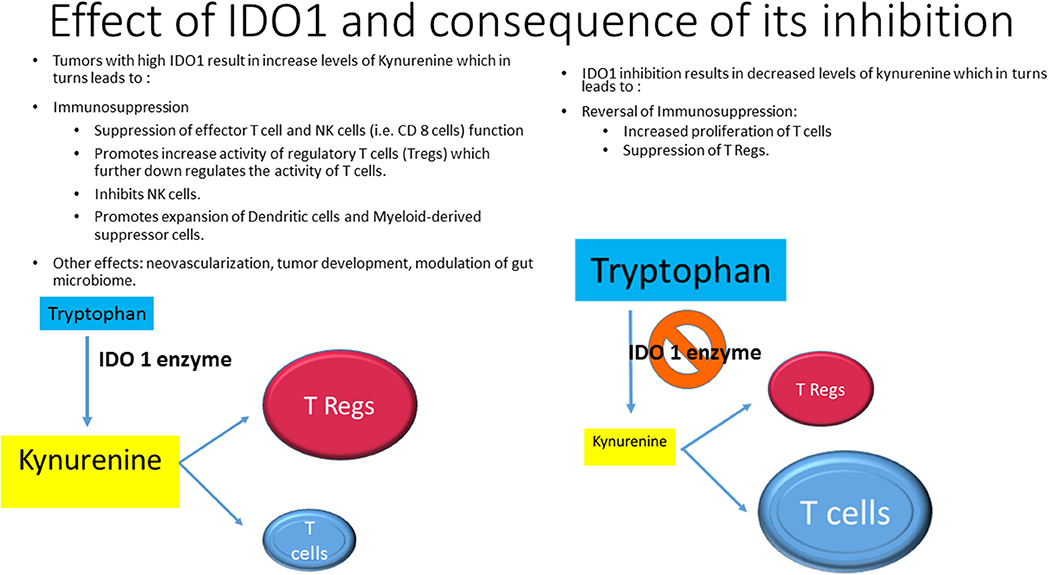
Indoleamine 2,3-dioxygenase1 (IDO1), IDO2, and are rate-limiting enzymes in the metabolism of essential amino acid tryptophan (Try), converting it to kynurenine (Kyn), in the peripheral tissue. Tryptophan 2,3-dioxynase (TDO) is in hepatic tissue. The increased activity of IDO1 leads to suppression of effector T cell (i.e., CD 8 cells) function and increase the activity of regulatory T cells (Tregs) which further downregulates the activity of T cells. In addition, IDO1 inhibits NK cell function and promotes the expansion and activation of dendritic cells (DC) and myeloid-derived suppressor cells (MDSC). Other effects include neovascularization, tumor development and modulation of gut microbiome (8) (Figure 1). Several IDO1 inhibitors are currently under preclinical or clinical investigation (9). This minireview will primarily focus on recent clinical research of Epacadostat (E) and other IDO1 inhibitors and discuss available data and future developments.
The Role of IDO1 in the Regulation of Cancer Immunity and Preclinical Development of IDO1 Inhibitors
Oncogenesis is regulated by the interaction between cancer and various immune cells (9). Effector T cells such as CD8+ cells play a dominant role in negatively controlling cancer development, whereas T regulatory cells (Tregs) suppress immunogenic response and allow immune escape of cancer cells. Increased IDO1 activity leads to accumulation of Kyn which suppresses NK cell/effector T cell function and activates Tregs (9). Its overexpression has been observed in many types of human cancers and it is associated with poor survival (10–13). IDO1 has been described as a novel therapeutic immune target in recent years (14).
Researchers at NewLink Genetics developed a tryptophan mimetic 1-methyl-tryptophan (1MT) which exists in two stereoisomers with different biological properties (15). The L isomer (1-L-MT) is the more potent inhibitor of IDO1, whereas the D isomer (1-D-MT, Indoximod) exhibited significantly higher anti-tumor activity by reversing T-cell function and synergizing with chemotherapy agents (16) but is a more selective IDO2 inhibitor (17) which metabolizes tryptophan much less efficiently than IDO1 (18, 19). Recent studies showed that Idoximod is an indirect inhibitor of IDO1 pathway by reversing mTORC1 inhibition induced by tryptophan depletion (20, 21).
Epacadostat (INCB024360) is an orally available reversible competitive potent IDO1 inhibitor and has entered clinical studies (22). It showed a potent anti-IDO1 activity with in vitro enzymatic and cell-based assay with an average IC50 value of 71.8 and 10 nM, respectively. It has a >1,000-fold selectivity for IDO1 compared to IDO2 and TDO. E increased T/NK cell proliferation, expanded CD86 high DCs, increased IFN-gamma production and suppressed Tregs in vitro. In vivo, E also suppressed tumor growth in immunocompetent but not in immunodeficient mice, although plasma Kyn/Try levels were significantly suppressed in both models.
Other IDO1 inhibitors in clinical development include Navoximod (NLG-919) which is an oral IDO1 and TDO inhibitor; BMS-986205 which is an irreversible IDO1 inhibitor; PF-06840003 and BGS-5777 which are IDO1 inhibitors that can cross blood-brain barrier (8). IOM2983, RG70099, NLG802, HTI-1090, KHK2455 are still in early development (21, 23–26).
Results in Clinical Investigation
Single-Agent Studies
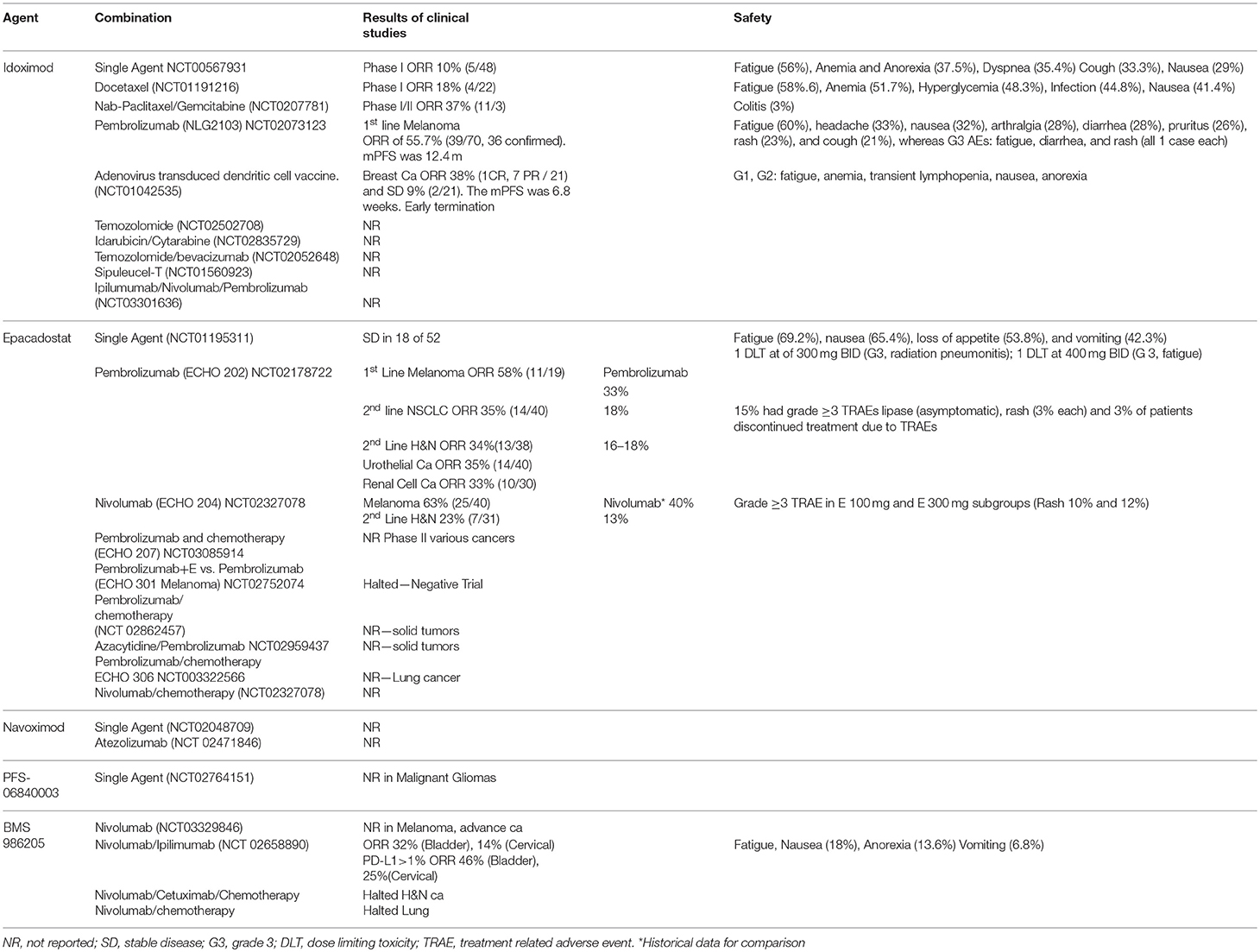
A phase I study in 48 patients with advanced solid tumor tested oral Indoximod at a dose ranging from 200 mg once daily to 2,000 mg twice daily (16). Idoximod was well tolerated and no patients discontinued treatment due to toxicity. Maximum tolerated dose (MTD) was not reached. Of note, three patients in the lowest dose level developed grade (G) 2 hypophysitis were previously treated with CPI (2 with Ipilimumab, 1 with anti-CD40 inhibitor). Efficacy analysis demonstrated five patients with stable disease (SD) of >6 months duration.
A dose escalation phase I study in 52 patients with advanced solid tumors tested E at a dose ranging from 50 mg once daily to 700 mg twice daily (BID) (27). E was well tolerated and MTD was not reached. The most common adverse events (AEs) was fatigue and others are described in Table 1. Seven patients discontinued treatment due to AEs, including 2 patients with dose-limiting toxicities (DLTs). PK studies showed a half-life of 2.4–3.9 h with modest variability between subjects and non-significant effect with ingestion of food with high-fat content. PD study demonstrated decreased in plasma Kyn level and Kyn\ Try ratios at all doses. The best response was SD in 18 of 52 patients lasting 16 weeks or longer seen in 7 patients (13.5%). As single drugs, these 2 agents were only able to produce SD with tolerable side effects.
Combination Studies With Indoximod (NLG-8189)
NLG2103 was a phase II study of Indoximod in combination with CPI in 94 patients with advanced melanoma (28). P was given at 2 mg/kg IV every 3 weeks with oral Indoximod at 1,200 mg BID in a 21-day cycle until intolerance or disease progression. Fatigue was the most common AE and other are listed in Table 1. ORR was 55.7% and median progression-free survival (PFS) was 12.4 m (29). A phase III trial NLG 2107 using Indoximod or Placebo plus P or N in patients with unresectable or metastatic melanoma is ongoing (30). Several studies are combining Idoximod with other systemic therapy such as cancer vaccines or cytotoxic chemotherapeutic agents. Based on the pre-clinical mouse model of breast cancer demonstrating the synergistic effect of Indoximod in combination with Paclitaxel on tumor regression (31), a phase I study of Indoximod in combination with Docetaxel was conducted in 27 patients with advanced solid tumors with ORR of 18%. The combination was well tolerated with no increased toxicity relative to single-agent docetaxel. Other ongoing studies combined Idoximod with gemcitabine/nab-paclitaxel, temozolomide, sipuleucel-T, or adenovirus-p53 transduced DC vaccine (32–35). The last study in breast cancer patients (NCT01042535) was terminated early and the data is not evaluable, making it difficult to assert the utility of this approach.
Combination Studies With Epacadostat
IDO1 inhibitors could enhance the effect of CPI by preventing immune escape through suppression of Kyn level and consequent suppression of Tregs and activation of T cells (Figure 1). Several studies were initiated to prove this concept.
ECHO202 was a multi-disease cohort, phase I/II study to define MTD and efficacy/safety in advanced solid tumor. Patients were treated with P at 2 mg/kg or 200 mg every 3 weeks in combination with oral E 25–300 mg BID until disease progression or intolerance. The results of the melanoma cohort showed an ORR of 58% (36). The efficacy data of other cohorts and AEs are described in Table 1. The response in different types of cancers, ranged from 33 to 58% and they were higher than historical single agent P studies (16 to 33%) (Table 1) (37–40). Safety analysis in 244 patients in the ECHO 202 study is also detailed in Table 1. Most were asymptomatic lipase elevation and there were no treatment-related deaths (41). Based on overall safety and efficacy, E 100 mg BID was selected as the recommended phase II dose.
ECHO204 was another multi-disease cohort, phase I/II study of E in combination with N with similar design and outcome of ECHO202. Preliminary results reported in 2017 also indicated that the combination has minimally increased toxicity and promising efficacy data compared to studies using single agent N (Table 1) (42). Overall, these early phase studies suggest that despite the lack of single agent anti-tumor activity, the addition of E to CPI are well-tolerated and produced relatively higher ORRs compared to studies using single agent CPI. The encouraging results of these 2 studies lead Incyte to launch several phases III registration trials to define the efficacy of E in combination with P and/or chemotherapy in solid tumors (ECHO 301–310) (43–51). Several of them are also adding E and CPI to chemotherapy (Table 1). Other investigator-initiated/sponsored trials using various combination partner drugs are also underway, including Sirolimus, Azacytidine, cancer vaccine, Itacinitib (52–55).
Recently, the enthusiasm with IDO1 inhibitors was dampened with the negative results of ECHO-301, a phase III trial in patients with unresectable or metastatic melanoma comparing E with P vs. P alone. The study did not meet its primary objective of improvement in PFS (56). The reasons for this negative trial are undefined. Further research regarding possible imbalance of patient groups, including pre-treatment use of prednisone which could decrease the response of P (57), differences in tumor mutation burden and patient's microbiota (58) could help to elucidate the reason for this negative trial. Other possibilities to explore are compliance with oral administration of E, level of inhibition of Kyn/Try ratio and development of inflammation as mechanism of resistance (59).
Other IDO1 Inhibitors in Clinical Development
BMS-986205, a novel, and more potent IDO1 selective inhibitor, is currently in development by Bristol-Myers Squibb. A phase I/II study of BMS-986205 alone or in combination with N was reported in 2017 (60). Patients with advanced cancers were treated at 25–200 mg once daily for a 2-week lead-in period, followed by the addition of N 240 mg IV every 2 weeks. Among the 42 evaluable patients, all treatment-related adverse events were G1-2 except three G3 toxicities. Serum Kyn was substantially reduced at all doses with >60% mean reduction at the 100 and 200 mg doses. Although efficacy result has not been reported, BMS-986205 showed a significant reduction in intratumoral Kyn in samples from 13 patients. Limited data of clinical efficacy were presented with a dramatic reduction in the size of the target lesion in 3 patients: head and neck cancer (63%), clear cell renal carcinoma (42%) and triple-negative breast cancer with 8 prior therapies (48%). A 3-arm phase I/II trial in combination with nivolumab at 2 doses or with Nivolumab plus Ipilimumab is underway.
Regrettably, 2 phase III trials of this agent in combination with Nivolumab/chemotherapy in H&N cancer and another in Lung cancer were halted after the press release of ECHO-301 negative results. Other new IDO1 inhibitors in clinical development do not have results to review (Table 1).
Novel Approaches to Test IDO1 Inhibitors in Clinical Trials
In addition to testing IDO1 inhibitors in advanced malignancies, researchers are investigating the role of IDO1 inhibitors in other clinical settings. A phase I trial combining E with external beam radiation therapy (XRT) and investigational TLR9 agonist SD-101 in patients with lymphoma (61) is based on the synergistic effect of XRT and CPI (62). In early-stage disease, a phase I/IIb trial is ongoing to assess the safety, toxicity, and PFS in patients who had undergone maximum debulking surgery for ovarian cancer using E (63) based on the improvement of recurrence-free survival using P/N as adjuvant therapy in resected melanoma (64, 65) and durvalumab in stage III lung cancer (6).
Similar to neoadjuvant or window-of-opportunity trials in early-stage disease (66, 67), IDO inhibitors are under evaluation in several diseases to primarily determine the pathologic effect in the tumor by collecting pre- and post-treatment tissue materials (68, 69). Future studies integrating peptide vaccine, anti-angiogenesis agents with IDO1 inhibitors are also of interest in view of its potential synergistic pathways (8).
Conclusion
IDO1 inhibitors have been in active clinical investigation and preliminary results suggest that they are well tolerated and produce additive efficacy when combined with CPI despite low levels of activity as a single agent. Epacadostat, a highly selective IDO1 inhibitor had increased response when combined with N or P in phase I/II trials and entered phase III trials. However, the negative results of the phase III trial ECHO 301 raised the questions about the efficacy of this combination strategy. Given the potential of IDO1 inhibitors to enhance immunologic function, it would be premature to abandon its development. Further research in combination with other synergistic agents and molecular characterization of responders and non-responders will be vital in the future of this agent in the field of immunotherapy.
Disclosure
The University of Kansas Research Institute has initiated a contract negotiation for an investigator-sponsored clinical trial sponsored by Incyte (51).
Author Contributions
TK and CH contributed conception and design of the study. TK wrote the first draft of the manuscript. All authors contributed to manuscript revision, read and approved the submitted version.
Conflict of Interest Statement
CH has received research funding from Bristol Myers Squibb, Merck, Bayer, Genentech, Lilly, Novartis and BPI; served as co-investigator in a clinical trial sponsored by Incyte; and received consulting fee from Ipsen.
The remaining author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer KM and handling Editor declared their shared affiliation.
Acknowledgments
Authors thank Ms. Kathleen Brumfield for administrative support.
References
1. World Health Organization. Media Center, Cancer, Key Facts. Available online at: http://www.who.int/mediacentre/factsheets/fs297/en/ (Accessed 20 March, 2018).
2. Tang J, Shalabi A, Hubbard-Lucey VM. Comprehensive analysis of the clinical immuno-oncology landscape. Ann Oncol. (2018) 29:84–91. doi: 10.1093/annonc/mdx755
3. Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WE, Poddubskaya E, et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. (2015) 373:123–35. doi: 10.1093/annonc/mdy04
4. Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csoszi T, Fülöp A, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. (2016) 375:1823–33. doi: 10.1056/NEJMoa1606774
5. Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet (2017) 389:255–65. doi: 10.1016/S0140-6736(16)32517-X
6. Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. (2017) 377:1919–29. doi: 10.1056/NEJMoa1709937
7. Highlights of Prescribing Information. OPDIVO (nivolumab) injection, for intravenous use. Initial U.S. Approval: 2014, Revised: 3/2018. Available online at: https://packageinserts.bms.com/pi/pi_opdivo.pdf. (Accessed March 20, 2018).
8. Liu M, Wang X, Wang L, Ma X, Gong Z, Zhang S, et al. Targeting the IDO1 pathway in cancer: from bench to bedside. J Hematol Oncol. (2018) 11:100. doi: 10.1186/s13045-018-0644-y
9. Hornyák L, Dobos N, Koncz G, Karányi Z, Páll D, Szabó Z et al. The role of indoleamine-2,3-dioxygenase in cancer development, diagnostics, and therapy. Front Immunol. (2018) 9:151. doi: 10.3389/fimmu.2018.00151
10. Uyttenhove C, Pilotte L, Theate I, Stroobant V, Colau D, Parmentier N, et al. Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat Med. (2003) 9:1269–74. doi: 10.1038/nm934
11. Pan K, Wang H, Chen MS, Zhang HK, Weng DS, Zhou J, et al. Expression and prognosis role of indoleamine 2,3-dioxygenase in hepatocellular carcinoma. J Cancer Res Clin Oncol. (2008) 134:1247–53. doi: 10.1007/s00432-008-0395-1
12. Ino K, Yamamoto E, Shibata K, Kajiyama H, Yoshida N, Terauchi M, et al. Inverse correlation between tumoral indoleamine 2,3-dioxygenase expression and tumor-infiltrating lymphocytes in endometrial cancer: its association with disease progression and survival. Clin Cancer Res. (2008). 14:2310–7. doi: 10.1158/1078-0432.CCR-07-4144
13. Astigiano S, Morandi B, Costa R, Mastracci L, D'Agostino A, Ratto GB, et al. Eosinophil granulocytes account for indoleamine 2,3-dioxygenase-mediated immune escape in human non-small cell lung cancer. Neoplasia (2005) 7:390–6. doi: 10.1593/neo.04658
14. Platten M, von Knebel DN, Oezen I, Wick W, Ochs K. Cancer immunotherapy by targeting IDO1/TDO and their downstream effectors. Front Immunol. (2015) 5:673. doi: 10.3389/fimmu.2014.00673
15. Cady SG, Sono M. 1-Methyl-DL-tryptophan, β-(3-benzofuranyl)-DL-alanine (the oxygen analog of tryptophan), and β- [3-benzo(b)thienyl]-DL-alanine (the sulfur analog of tryptophan) are competitive inhibitors for indoleamine 2,3-dioxygenase. Arch Biochem Biophys. (1991) 291:326–33. doi: 10.1016/0003-9861(91)90142-6
16. Soliman HH, Minton SE, Han HS, Ismail-Khan R, Neuger A, Khambati FA, et al. A phase I study of indoximod in patients with advanced Malignancies. Oncotarget (2016) 7:22928–38. doi: 10.18632/oncotarget.8216
17. Soliman H, Antonia S, Sullivan D, Vanahanian N, Link CJ. Overcoming tumor antigen anergy in human malignancies using the novel indeolamine 2,3-dioxygenase (IDO) enzyme inhibitor, 1-methyl-D-tryptophan (1MT). J Clin Oncol. (2009) 27(Suppl.):3004. doi: 10.1200/jco.2009.27.15_suppl.3004
18. Liu X, Shin N, Koblish HK, Yang G, Wang Q, Wang K, et al. Selective inhibition of IDO1 effectively regulates mediators of antitumor immunity. Blood (2010) 115:3520–30. doi: 10.1182/blood-2009-09-246124
19. Beatty GL, O'Dwyer PJ, Clark J, Shi JG, Newton RC, Schaub R, et al. Phase I study of the safety, pharmacokinetics (PK), and pharmacodynamics (PD) of the oral inhibitor of indoleamine 2,3-dioxygenase (IDO1) INCB024360 in patients (pts) with advanced malignancies. J Clin Oncol. (2013) 31(Suppl.). doi: 10.1200/jco.2013.31.15_suppl.3025
20. Metz R, Rust S, Duhadaway JB, Mautino MR, Munn DH, Vahanian NN, et al. IDO inhibits a tryptophan sufficiency signal that stimulates mTOR: a novel IDO effector pathway targeted by D-1-methyl-tryptophan. Oncoimmunology (2012) 1:1460–8. doi: 10.4161/onci.21716
21. Prendergast GC, Malachowski WP, DuHadaway JB, Muller AJ. Discovery of IDO1 inhibitors: from bench to bedside. Cancer Res. (2017) 77:6795–811. doi: 10.1158/0008-5472.CAN-17-2285
22. Jochems C, Fantini M, Fernando RI, Kwilas AR, Donahue RN, Lepone LM, et al. The IDO1 selective inhibitor epacadostat enhances dendritic cell immunogenicity and lytic ability of tumor antigen-specific T cells. Oncotarget (2016) 7:37762–72. doi: 10.18632/oncotarget.9326
23. Mautino MR, Kumar S, Zhuang H, Waldo J, Jaipuri F, Potturi H, et al. A novel prodrug of indoximod with enhanced pharmacokinetic properties. Cancer Res (2017) 77(Suppl.). doi: 10.1158/1538-7445.AM2017-4076
24. A Trial of HTI-1090 in Subjects With Advanced Solid Tumors. ClinicalTrials.gov Identifier: NCT03208959. Available online at: https://clinicaltrials.gov/ct2/show/NCT03208959 (Accessed March 20, 2018).
25. KHK2455 Alone and in Combination With Mogamulizumab in Subjects With Locally Advanced or Metastatic Solid Tumors. ClinicalTrials.gov Identifier: NCT02867007. Available online at: https://clinicaltrials.gov/ct2/show/NCT02867007?term=KHK2455&rank=1 (Accessed March 20, 2018).
26. First in Patient Study for PF-06840003 in Malignant Gliomas. ClinicalTrials.gov Identifier: NCT02764151. Available online at: https://clinicaltrials.gov/ct2/show/NCT02764151?term=PF-06840003&rank=1 (Accessed March 20, 2018).
27. Beatty GL, O'Dwyer PJ, Clark J, Shi JG, Bowman KJ, Scherle PA, et al. First-in-human phase I study of the oral inhibitor of indoleamine 2,3-dioxygenase-1 epacadostat (INCB024360) in patients with advanced solid malignancies. Clin Cancer Res. (2017) 23:3269–76. doi: 10.1158/1078-0432.CCR-16-2272
28. Zakharia Y, McWilliams R, Shaheen M, Grossman K, Drabick J, Milhem M, et al. Interim analysis of the Phase 2 clinical trial of the IDO pathway inhibitor indoximod in combination with pembrolizumab for patients with advanced melanoma. Cancer Res. (2017) 77(Suppl). doi: 10.1158/1538-7445
29. Zakharia Y, Rixe O, Ward JH, Drabick JJ, Shaheen MF, Milhem MM, et al. Phase 2 trial of the IDO pathway inhibitor indoximod plus checkpoint inhibition for the treatment of patients with advanced melanoma. J Clin Oncol. (2018) 36:9512. doi: 10.1200/JCO.2018.36.15_suppl.9512
30. A Study of Indoximod or Placebo Plus Pembrolizumab or Nivolumab for Subjects With Unresectable or Metastatic Melanoma (NLG2107). ClinicalTrials.gov Identifier NCT03301636. Available online at: https://clinicaltrials.gov/ct2/show/NCT03301636 (Accessed March 20, 2018).
31. Muller AJ, DuHadaway JB, Donover PS, Sutanto-Ward E, Prendergast GC. Inhibition of indoleamine 2,3-dioxygenase, an immunoregulatory target of the cancer suppression gene Bin1, potentiates cancer chemotherapy. Nat Med. (2005) 11:312–9. doi: 10.1038/nm1196
32. Study of IDO Inhibitor in Combination With Gemcitabine and Nab-Paclitaxel in Patients With Metastatic Pancreatic Cancer. ClinicalTrials.gov Identifier: NCT02077881. Available online at: https://clinicaltrials.gov/ct2/show/NCT02077881?intr=Indoximod&titles=gemcitabine&rank=1 (Accessed March 20, 2018).
33. Study of IDO Inhibitor and Temozolomide for Adult Patients With Primary Malignant Brain Tumors. ClinicalTrials.gov Identifier: NCT02052648. Available online at: https://clinicaltrials.gov/ct2/show/NCT02052648?intr=Indoximod&titles=temozolomide&rank=2 (Accessed March 20, 2018).
34. Phase II Study of Sipuleucel-T and Indoximod for Patients With Refractory Metastatic Prostate Cancer. ClinicalTrials.gov Identifier: NCT01560923 Available online at: https://clinicaltrials.gov/ct2/show/NCT01560923?intr=Indoximod&titles=sipuleucel-T&rank=1 (Accessed March 20, 2018).
35. Vaccine Therapy and 1-MT in Treating Patients With Metastatic Breast Cancer. ClinicalTrials.gov Identifier: NCT01042535. Available online at: https://clinicaltrials.gov/ct2/show/NCT01042535?intr=Indoximod&titles=vaccine&rank=1 (Accessed August 30, 2018).
36. Gangadhar TC, Hamid O, Smith DC, Bauer TM, Wasser JS, Olszanski AJ, et al. Epacadostat plus pembrolizumab in patients with advanced melanoma and select solid tumors: updated phase 1 results from ECHO-202/KEYNOTE-037. Ann Oncol. (2016) 27:379–400. doi: 10.1093/annonc/mdw379
37. Hamid O, Bauer TM, Spira AI, Olszanski AJ, Patel SP, Wasser JS, et al. Epacadostat plus pembrolizumab in patients with SCCHN: preliminary phase 1/2 results from ECHO-202/KEYNOTE-037. J Clin Oncol. (2017) 35(Suppl):6010. doi: 10.1200/JCO.2017.35.15_suppl.6010
38. Gangadhar TC, Schneider BJ, Bauer TM, Wasser JS, Spira AI, Patel SP, et al. Efficacy and safety of epacadostat plus pembrolizumab treatment of NSCLC: Preliminary phase I/II results of ECHO-202/KEYNOTE-037. J Clin Oncol. (2017) 35(Suppl.):9014. doi: 10.1200/JCO.2017.35.15_suppl.9014
39. Smith DC, Gajewski T, Hamid O, Wasser JS, Olszanski AJ, Patel SP, et al. Epacadostat plus pembrolizumab in patients with advanced urothelial carcinoma: preliminary phase I/II results of ECHO-202/KEYNOTE-037. J Clin Oncol. (2017) 35(Suppl.):4503. doi: 10.1093/annonc/mdw379.06
40. Lara P, Bauer TM, Hamid O, Smith DC, Gajewski T, Gangadhar TC, et al. Epacadostat plus pembrolizumab in patients with advanced RCC: preliminary phase I/II results from ECHO-202/KEYNOTE-037. J Clin Oncol. (2017) 35(Suppl.):4515. doi: 10.1200/JCO.2017.35.15_suppl.4515
41. Hamid O, Bauer TM, Spira AI, Smith DC, Olszanski AJ, Tarhini AA, et al. Safety of epacadostat 100 mg bid plus pembrolizumab 200 mg Q3W in advanced solid tumors: phase 2 data from ECHO-202/KEYNOTE-037. J Clin Oncol. (2017) 35(Suppl.):3012. doi: 10.1200/JCO.2017.35.15_suppl.3012
42. Perez RP, Riese MJ, Lewis KD, Saleh MN, Daud A, Berlin J, et al. Epacadostat plus nivolumab in patients with advanced solid tumors: preliminary phase I/II results of ECHO-204. J Clin Oncol. (2017) 35(Suppl.):3003. doi: 10.1200/JCO.2017.35.15_suppl.3003
43. A Phase 3 Study of Pembrolizumab + Epacadostat or Placebo in Subjects With Unresectable or Metastatic Melanoma (Keynote-252 / ECHO-301). ClinicalTrials.gov Identifier: NCT02752074. Available online at: https://clinicaltrials.gov/ct2/show/NCT02752074?intr=Epacadostat&titles=ECHO&rank=2 (Accessed March 20, 2018).
44. Pembrolizumab (MK-3475) Plus Epacadostat vs Standard of Care in mRCC. ClinicalTrials.gov Identifier: NCT03260894. Available online at: https://clinicaltrials.gov/ct2/show/NCT03260894?intr=Epacadostat&titles=ECHO&draw=2&rank=15 (Accessed March 20, 2018).
45. Pembrolizumab + Epacadostat vs Pembrolizumab + Placebo in Recurrent or Progressive Metastatic Urothelial Carcinoma. ClinicalTrials.gov Identifier: NCT03374488. Available online at: https://clinicaltrials.gov/ct2/show/NCT03374488?intr=Epacadostat&titles=ECHO&draw=3&rank=14 (Accessed March 20, 2018).
46. Pembrolizumab Plus Epacadostat Pembrolizumab Monotherapy and the EXTREME Regimen in Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma (KEYNOTE-669/ECHO-304). ClinicalTrials.gov Identifier: NCT03358472. Available online at: https://clinicaltrials.gov/ct2/show/NCT03358472?intr=Epacadostat&titles=ECHO&rank=10 (Accessed March 20, 2018).
47. Pembrolizumab Plus Epacadostat vs Pembrolizumab Plus Placebo in Metastatic Non-Small Cell Lung Cancer (KEYNOTE-654-01/ECHO-305). ClinicalTrials.gov Identifier: NCT03322540. Available online at: https://clinicaltrials.gov/ct2/show/NCT03322540?intr=Epacadostat&titles=ECHO&draw=2&rank=12 (Accessed March 20, 2018).
48. Pembrolizumab Plus Epacadostat Alone or With Platinum-based Chemotherapy Versus Pembrolizumab Plus Platinum-based Chemotherapy Plus Placebo in Metastatic Non-Small Cell Lung Cancer (KEYNOTE-715-02/ECHO-306). ClinicalTrials.gov Identifier: NCT03322566. Available online at: https://clinicaltrials.gov/ct2/show/NCT03322566?intr=Epacadostat&titles=ECHO&draw=3&rank=13 (Accessed March 20, 2018).
49. Pembrolizumab in Combination With Epacadostat or Placebo in Cisplatin-ineligible Urothelial Carcinoma (KEYNOTE-672/ECHO-307). ClinicalTrials.gov Identifier: NCT03361865. Available online at: https://clinicaltrials.gov/ct2/show/NCT03361865?intr=Epacadostat&titles=ECHO&draw=2&rank=3 (Accessed March 20, 2018).
50. Nivolumab and Epacadostat With Platinum Doublet Chemotherapy Versus Platinum Doublet Chemotherapy in Non-Small Cell Lung Cancer. ClinicalTrials.gov Identifier: NCT03348904. Available online at: https://clinicaltrials.gov/ct2/show/NCT03348904 (Accessed March 20, 2018).
51. Nivolumab Plus Epacadostat in Combination With Chemotherapy Versus the EXTREME Regimen in Squamous Cell Carcinoma of the Head and Neck (CheckMate 9NA/ECHO-310). ClinicalTrials.gov Identifier: NCT03342352. Available online at: https://clinicaltrials.gov/ct2/show/NCT03342352?intr=Epacadostat&titles=ECHO&rank=7 (Accessed March 20, 2018).
52. Epacadostat (INCB24360) in Combination With Sirolimus in Advanced Malignancy. ClinicalTrials.gov Identifier: NCT03217669 Available online at: https://clinicaltrials.gov/ct2/show/NCT03217669?intr=Epacadostat&titles=sirolimus&rank=1 (Accessed March 20, 2018).
53. Azacitidine Combined With Pembrolizumab and Epacadostat in Subjects With Advanced Solid Tumors (ECHO-206). ClinicalTrials.gov Identifier: NCT02959437. Available online at: https://clinicaltrials.gov/ct2/show/NCT02959437?intr=Azacitidine&titles=epacadostat&rank=2 (Accessed March 20, 2018).
54. Epacadostat and Vaccine Therapy in Treating Patients With Stage III-IV Melanoma. ClinicalTrials.gov Identifier: NCT01961115. Available online at: https://clinicaltrials.gov/ct2/show/NCT01961115?intr=epacadostat&titles=vaccine&rank=1 (Accessed March 20, 2018).
55. Itacitinib Combined With INCB024360 and/or Itacitinib Combined With INCB050465 in Advanced Solid Tumors. ClinicalTrials.gov Identifier: NCT02559492. Available online at: https://clinicaltrials.gov/ct2/show/NCT02559492?intr=Itacitinib&titles=epacadostat&rank=1
57. Arbour KC, Mezquita L, Long N, Auclin E, Ni A, Bernal GM, et al. Deleterious effect of baseline steroids on efficacy of PD-(L)1 blockade in patients with NSCLC. J Clin Oncol. (2018) 36(Suppl.):9003. doi: 10.1200/JCO.2018.36.15_suppl.9003
58. Routy B, Le Chatelier E, Derosa, L, Duong, CPM, Alou, MT, Daillère R, et al. Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science (2018) 359:91–7. doi: 10.1126/science.aan3706
59. Muller AJ, Sharma MD, Chandler PR, DuHadaway JB, Everhart ME, Johnson BA, et al. Chronic inflammation that facilitates tumor progression creates local immune suppression by inducing indoleamine 2, 3 dioxygenase. Proc Natl Acad Sci USA. (2008) 105:17073–8. doi: 10.1073/pnas.0806173105
60. Siu LL, Gelmon K, Chu Q, et al. BMS-986205, an optimized indoleamine 2,3-dioxygenase 1 (IDO1) inhibitor, is well tolerated with potent pharmacodynamic (PD) activity, alone and in combination with nivolumab (nivo) in advanced cancers in a phase 1/2a trial. Cancer Res. (2017) 77(Suppl.):CT116. doi: 10.1158/1538-7445.AM2017-CT116
61. Phase I/II Trial of Epacadostat Intralesional SD101 Radiotherapy in Patients With Lymphoma. ClinicalTrials.gov Identifier: NCT03322384. Available online at: https://clinicaltrials.gov/ct2/show/NCT03322384?intr=Epacadostat&titles=radiation&rank=1 (Accessed March 20, 2018).
62. Ahmed KA, Grass GD, Creelan B, Gray J, Kim S, Dilling TJ, et al. Tolerability and safety of thoracic radiation and immune checkpoint inhibitors among patients with lung cancer. Int J Radiat Oncol Biol Phys. 2017 98(Suppl.):224. doi: 10.1016/j.ijrobp.2017.01.063
63. DEC-205/NY-ESO-1 Fusion Protein CDX-1401 Poly ICLC and IDO1 Inhibitor INCB024360 in Treating Patients With Ovarian Fallopian Tube or Primary Peritoneal Cancer in Remission. ClinicalTrials.gov Identifier: NCT02166905. Available online at: https://clinicaltrials.gov/ct2/show/NCT02166905
64. Eggermont AM, Chiarion-Sileni V, Grob JJ, Dummer R, Wolchok JD, Schmidt H, et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. Lancet Oncol. (2015) 16:522–30. doi: 10.1016/S1470-2045(15)70122-1
65. Weber J, Mandala M, Del Vecchio M, Gogas, HJ, Arance, AM, Cowey, CL, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med. (2017) 377:1824–35. doi: 10.1056/NEJMoa1709030
66. Chaft JE, Forde PM, Smith KN, Anagnostou V, Cottrell T, Taube JM, et al. Neoadjuvant nivolumab in early-stage, resectable non-small cell lung cancers. J Clin Oncol. (2017) 35(Suppl.):8508. doi: 10.1200/JCO.2016.34.15_suppl.e20005
67. Jakub JW, Racz JM, Hieken TJ, Gonzalez AB, Kottschade LA, Markovic SN, et al. Neoadjuvant systemic therapy for regionally advanced melanoma. J Surg Oncol. (2017) 117:1164–9. doi: 10.1002/jso.24939
68. Neoadjuvant Pembrolizumab + Epacadostat Prior to Curative Surgical Care for Squamous Cell Carcinoma of the Head and Neck. ClinicalTrials.gov Identifier: NCT03325465. Available online at: https://clinicaltrials.gov/ct2/show/NCT03325465 (Accessed March 20, 2018).
69. Epacadostat Before Surgery in Treating Patients With Newly Diagnosed Stage III-IV Epithelial Ovarian Fallopian Tube or Primary Peritoneal Cancer. ClinicalTrials.gov Identifier: NCT02042430. Available online at: https://clinicaltrials.gov/ct2/show/NCT02042430 (Accessed March 20, 2018).
Keywords: immuno-oncology, IDO (indoleamine 2,3-dioxygenase), PD-1, tryptophan, clinical trials as topic, IDO1 inhibitor
Citation: Komiya T and Huang CH (2018) Updates in the Clinical Development of Epacadostat and Other Indoleamine 2,3-Dioxygenase 1 Inhibitors (IDO1) for Human Cancers. Front. Oncol. 8:423. doi: 10.3389/fonc.2018.00423
Received: 05 April 2018; Accepted: 11 September 2018;
Published: 04 October 2018.
Edited by:
Brion William Murray, Pfizer, United StatesCopyright © 2018 Komiya and Huang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chao H. Huang, Y2h1YW5nMkBrdW1jLmVkdQ==
 Takefumi Komiya
Takefumi Komiya Chao H. Huang
Chao H. Huang