
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Neurosci. , 02 April 2025
Sec. Neural Technology
Volume 19 - 2025 | https://doi.org/10.3389/fnins.2025.1554490
This article is part of the Research Topic Neuroengineering for health and disease: a multi-scale approach View all 3 articles
Objective: Typical Ramsay Hunt syndrome (RHS) is a rare peripheral facial neuropathy associated with reactivation of latent varicella-zoster virus in the geniculate ganglion. Atypical RHS is characterized by the involvement of multiple cranial nerves and cervical roots, leading to more complex manifestations. The primary goal of treatment is to reduce the occurrence of late complications, especially in patients with devastating postherpetic neuralgia (PHN). To date, there is no definitive effective treatment. We present a case series of patients with typical and atypical RHS and severe PHN, who were successfully treated with transcutaneous facial nerve stimulation (FNS).
Materials and methods: This is a retrospective case series including two atypical RHS cases and one typical RHS case. The first patient with atypical RHS suffered from persistent otalgia with severe paroxysmal radiation to the dermatome of fifth cranial nerve (CN V) and IX lesion. The second atypical RHS patient with CN V and VII lesions had persistent frontotemporal neuralgia and otalgia, with severe paroxysmal radiation to the CN V and IX dermatome. The third typical patient had persistent otalgia with severe paroxysmal exacerbations. An FNS in the stylomastoid foramen was successfully performed under ultrasound guidance in combination with DSA. Pain assessment was performed during treatment and follow-up, including the type of pain (persistent pain, breakthrough pain, and tactile allodynia) and pain distribution. Pain intensity was assessed using the Number Rate Scale (NRS) and the Verbal Rating Scale (VRS). The therapeutic effect was assessed using the Pain Relief Scale (PRS). In addition, the Pain Relief Ratio (PRR) was calculated as (NRSPre-T - NRSPost-T)/NRSPre-T × 100%, and the treatment was considered effective if the PRR was greater than 50%.
Results: The t-FNS showed excellent pain relief, particularly for breakthrough pain. The breakthrough pain completely ceased before the FNS was turned off, and the persistent pain decreased from moderate to mild intensity before the patients were discharged. The mild persistent pain of the first patient on the 3-month follow-up and the third patient on the 2-month follow-up had completely disappeared, but the mild persistent pain of the second patient was still felt in the temporal region for 1 year.
Conclusion: For the first time, transcutaneous FNS was successfully used to treat intractable PHN in patients with typical and atypical RHS. However, further research is needed to determine the optimal procedure and specific stimulation parameters.
Ramsay Hunt syndrome (RHS), with an incidence of approximately 5 per 100,000 people per year (Murakami et al., 1997; Tiemstra and Khatkhate, 2007), is caused by the reactivation of the herpes zoster virus in the geniculate ganglion. Therefore, the prophylactic use of a vaccine against the herpes zoster virus in high-risk patients can reduce the incidence of RHS. Typical RHS is characterized by a combination of three symptoms: painful vesicles, otalgia, and ipsilateral facial palsy. However, there are some unusual variants associated with atypical RHS (Nishizawa et al., 2021; Zhang and Wei, 2020; Lee et al., 2014). Although herpes zoster is inherently a self-limiting disease, herpetic neuralgia is the first symptom that can appear without facial paralysis or rash (Coulson et al., 2011). Moreover, postherpetic neuralgia, a devastating complication of RHS, lasts longer than the healing of the rash (Choo et al., 1997). Patients often report a decreased quality of life and difficulties with daily activities, which can affect their physical, psychological, and social well-being. Therefore, the primary goal of treatment is to reduce the incidence of postherpetic neuralgia and to achieve better pain relief.
Currently, there are individual case reports on the treatment of RHS. However, early diagnosis and intervention within 72 h of symptom onset are crucial. Effective pain relief often requires a multimodal approach that includes medication (NSAIDs, opioids, tricyclic antidepressants, and anticonvulsants) (Liao et al., 2021), nerve block (Jacques et al., 2019; Liao et al., 2021), acupuncture (Giralt et al., 2020; Liao et al., 2021), and physical therapy (linear polarized near-infrared light irradiation) (Liao et al., 2021). Jeon and Lee (2018) recommended using a combination of antiviral agents (e.g., acyclovir) and corticosteroids to manage RHS-related neuralgia, particularly when administered early to mitigate nerve damage and improve outcomes (Jeon and Lee, 2018). Jacques et al. (2019) also presented a case in which blockade of the terminal branches of the nervus intermedius successfully reduced neuralgia symptoms for 3 months in a patient with severe pain due to Ramsay Hunt syndrome (Jacques et al., 2019). Subdermal injections of BoNT have been shown to be potentially beneficial in cases of RHS with severe ear lobes (Soumekh, 2024), and pulsed radiofrequency (PRF) applied to the greater auricular nerve has provided significant pain relief in refractory otalgia following RHS (Kim et al., 2021). Liao et al. (2021) implemented a multimodal approach, combining oral gabapentin, pulsed radiofrequency (PRF) application to the Gasserian ganglion for pain in the trigeminal nerve region, linear-polarized near-infrared light irradiation for pain in the facial nerve region, and 2% lidocaine spray for pain in the glossopharyngeal nerve region. This method improved pain management and quality of life in a 78-year-old patient with a 3-month history of PHN secondary to RHS with polycranial nerve (V, VII, VIII, and IX) involvement (Liao et al., 2021). Tympanic nerve neurectomy resulted in significant pain relief and improvement in quality of life in a 45-year-old patient with chronic pain in the right-sided facial, ear, and jaw that persisted for 9 years after RHS (Sarathy et al., 2023). However, these techniques have only proven their effectiveness in individual cases.
In this study, we present two patients with atypical RHS who experienced severe neuralgia in unusually extensive facial and cervical dermatomes without facial paralysis. Additionally, we included a typical RHS patient with severe otalgia and ipsilateral facial paralysis after recovery from herpes. The intractable neuralgia was successfully treated with transcutaneous facial nerve stimulation (t-FNS). This study aims to evaluate the safety and efficacy of ultrasound-and DSA-guided t-FNS in patients with refractory RHS neuralgia, addressing the limitations of existing treatments and exploring its potential as an alternative neuromodulation therapy.
Patients were selected from the Affiliated Hospital of Guilin Medical University based on a diagnosis of refractory RHS neuralgia. The inclusion criteria included (1) confirmed diagnosis of RHS neuralgia with or without other cranial nerves involved, (2) no response to conventional treatments, and (3) eligibility for neurostimulation therapy. The exclusion criteria included (1) contraindications to neurostimulation and (2) severe comorbidities that could affect the treatment response. The retrospective analysis involved three patients with RHS who suffered from severe PHN and were successfully treated with FNS. Before implanting the t-FNS lead, a Nadbath–Rehman block was performed at the stylomastoid foramen with 1 mL of 1% lidocaine immediately after breakthrough pain recurred, and the pain stopped immediately. FNS at the stylomastoid foramen was then recommended. We discussed the risks of the procedure with the patient, which included bleeding, nerve injury, and infection, along with the potential benefits and alternatives. Once all questions were answered and the patient understood and accepted the procedure, informed consent was obtained.
As no technical procedure was available, a pilot protocol for t-FNS was developed, and the procedure was performed under ultrasound guidance in combination with DSA (Figure 1).
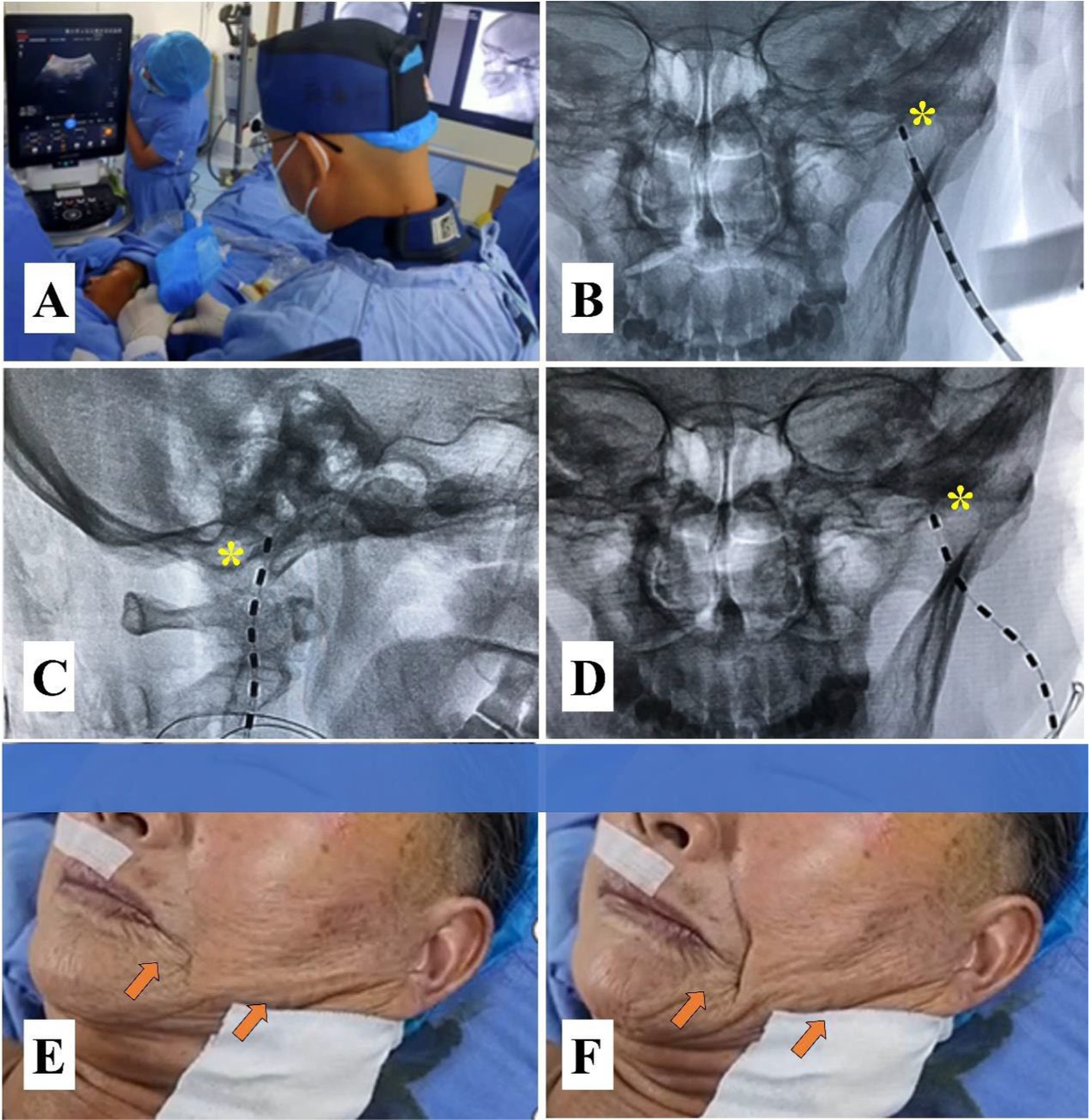
Figure 1. A pilot protocol for transcutaneous facial nerve stimulation (t-FNS). (A) Ultrasound-guided injection of 0.5% lidocaine for regional anesthesia and widening of the puncture route to avoid intravascular injection. (B) The lead was implanted medial to the MP using a manual 15° arcuate hollow needle in the anteroposterior view of the DSA. (C) The lead was again placed anterior to the MP in the lateral view of the DSA. (D) The anteroposterior view of the DSA confirmed that the lead was placed medial to the MP. (E) A slight contraction of the facial muscle was elicited by a test stimulation voltage of 0.3 V. (F) A significant contraction of the facial muscle was elicited by a stimulation voltage of 1 V. The yellow asterisk indicates the MP, and the orange arrow represents the contraction of the facial muscle.
The patient was positioned supine on a DSA table with the head tilted to the opposite side. Blood pressure, heart rate, oxygen saturation, and electrocardiographic waveforms were continuously monitored. She received oxygen at 3 L/min and a continuous intravenous infusion of 0.9% saline. Midazolam (Jiangsu Enhua Pharmaceutical Co. Ltd.) at a dosage of 1 mg and fentanyl (Jiangsu Enhua Pharmaceutical Co. Ltd.) at 50 μg were injected intravenously for sedation. For regional anesthesia, 10 mL of 0.5% lidocaine was injected into the puncture site tissue under ultrasound guidance using an 8–3 MHz transducer (Sonosite Inc., Bothell, USA).
A manual 15° arcuate hollow needle (16 G) connected to a 5-ml syringe with continuous negative pressure was inserted into the skin and slowly advanced toward the foramen stylomastoideum. The procedure was guided by ultrasound to avoid vascular injury, and DSA confirmed the target at the medial and anterior parts of the mastoid process. As soon as the patient complained of discomfort or blood flowed into the negative pressure syringe, the puncture procedure had to be stopped. After the direction was adjusted, the procedure was continued until the needle reached the target. A trial lead (3,086, Abbott Medical, Plano, USA) was implanted through the insertion needle until it touched the bone. Then, the introducer needle was withdrawn, and the placement of the lead was reconfirmed by DSA at the medial and anterior parts of the mastoid process (MP).
The placement of the lead was confirmed by a stimulation test after the lead was connected to the external stimulator (3,510, St. Jude Medical, Inc. Fullerton, CA). The lead was placed close to the facial nerve when the contraction of the facial muscle was evoked by a voltage of less than 0.5 V. The lead was anchored into the skin using sterile 3.0 sutures (Vicryl, Ethicon, Peterborough, ON), and a neck collar was worn to prevent displacement of the lead.
The contact polarity was set as follows: Contact point 1 (−) and contact point 2 (+); the stimulation frequency was 40 Hz, and the pulse width was 200 μs. The amplitude adjustment from 0.5 V to 1 V allowed contraction of the facial muscles that did not disturb the patient’s sleep, and the amplitude adjustment from 1 V to 3 V resulted in a stronger contraction of the facial muscles up to the patient’s upper tolerance limit during the non-sleep period.
Depending on the type of pain, including persistent pain (tingling, burning, and itching), breakthrough pain (stabbing and shooting), and tactile allodynia, the patient reported the pain distribution as “yes” or “no” and rated the pain intensity using the NRS (‘0’ for no pain and ‘10’ for unbearable pain) and the Verbal Rating Scale (VRS) (0 = no pain; 1 = mild pain; 2 = moderate pain; and 3 = severe pain or 4 = intense pain). The patient also rated the efficacy using the Pain Relief Scale (PRS) (0 = none, 1 = mild, 2 = moderate, 3 = severe, and 4 = total). In addition, the Pain Relief Ratio (PRR) was calculated as (NRSPre-T - NRSPost-T)/NRSPre-T × 100%, and treatment was considered effective if the PRR was greater than 50%.
The FNS showed excellent pain relief in all three cases, especially for breakthrough pain. The details are described in detail below.
A 71-year-old female patient suffering from a rash in the left external auditory canal was diagnosed with herpes zoster 15 days ago and received intravenous acyclovir (0.25 g three times daily) for 7 days. She also reported pain in the area of the rash and was given oral gabapentin (initial dose 300 mg three times daily and up to 600 mg three times daily) and tramadol extended-release capsules (100 mg daily). However, her pain worsened over time, and she was referred to our hospital for further treatment.
The patient complained of a persistent type of pain with paroxysmal exacerbations in intensity. The persistent pain was “incessant tingling, burning, and itching” in the external auditory canal and auricle, with an intensity of 5 on a numerical rating scale (NRS) of 0–10. She also reported a severe, stabbing, and shooting otalgia that occurred almost half an hour apart, lasted for 3–5 min, and radiated to the frontotemporal, mandibular, pharyngeal, upper cervical, and occipital regions with an intensity of 10 on an NRS (Figure 2). The patient was unable to fall asleep and expressed exaggerated fear, anxiety, and depression. Physical examination revealed that tactile allodynia can be triggered when the external auditory canal is lightly touched with a cotton swab, and the cervico-occipital region needs to be cooled when the breakthrough pain occurs.
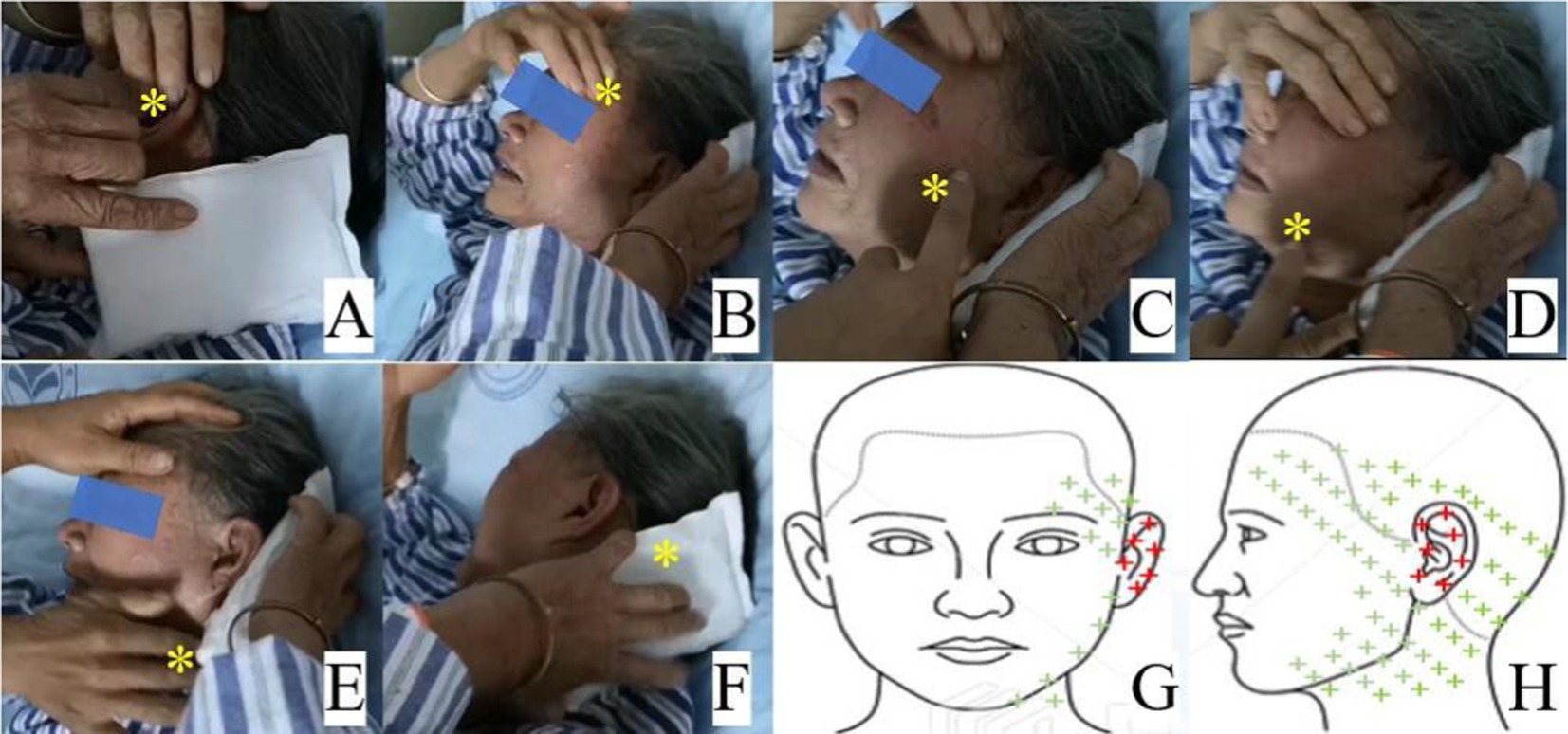
Figure 2. Pain regions of the breakthrough pain attack (A–F) and the pain distribution in the schematic representation (G,H). A: external auditory canal and auricle; (B) frontotemporal region; (C,D) mandibular region; (E) pharyngeal and upper cervical region; (F) occipital region; (G,H): schematic diagram showing pain distribution of persistent pain in red plus and breakthrough pain in red and green plus. The yellow asterisk indicates the region of breakthrough pain.
After ruling out the conditions, such as tumors, hemorrhage, swelling, infection, or inflammation through magnetic resonance imaging of the brain and cervical spine, a diagnostic nerve blockade of the supraorbital nerve, glossopharyngeal nerve, greater occipital nerve, lesser occipital nerve, and cervical 2 (C2) nerve with 1% lidocaine was performed based on the affected painful dermatome; however, it was unsuccessful. When the breakthrough pain recurred, a Nadbath–Rehman block was performed at the stylomastoid foramen using 1 mL of 1% lidocaine, and the pain stopped immediately. Following the result, she was diagnosed with atypical RHS with severe otalgia radiating to the dermatome of the trigeminal, glossopharyngeal, and occipital nerves, with no facial paralysis. The patient was then recommended pulsed radiofrequency (PRF) at the stylomastoid foramen. After obtaining informed consent, PRF of the facial nerve (90 V for 10 min each, twice) was performed under the guidance of digital subtraction angiography (DSA) (Figure 3). However, the patient only experienced less than 20% pain relief for approximately 4 h. Then, t-FNS at the stylomastoid foramen was recommended to the patient based on our successful experience treating postherpetic neuralgia with spinal cord and trigeminal nerve stimulation.
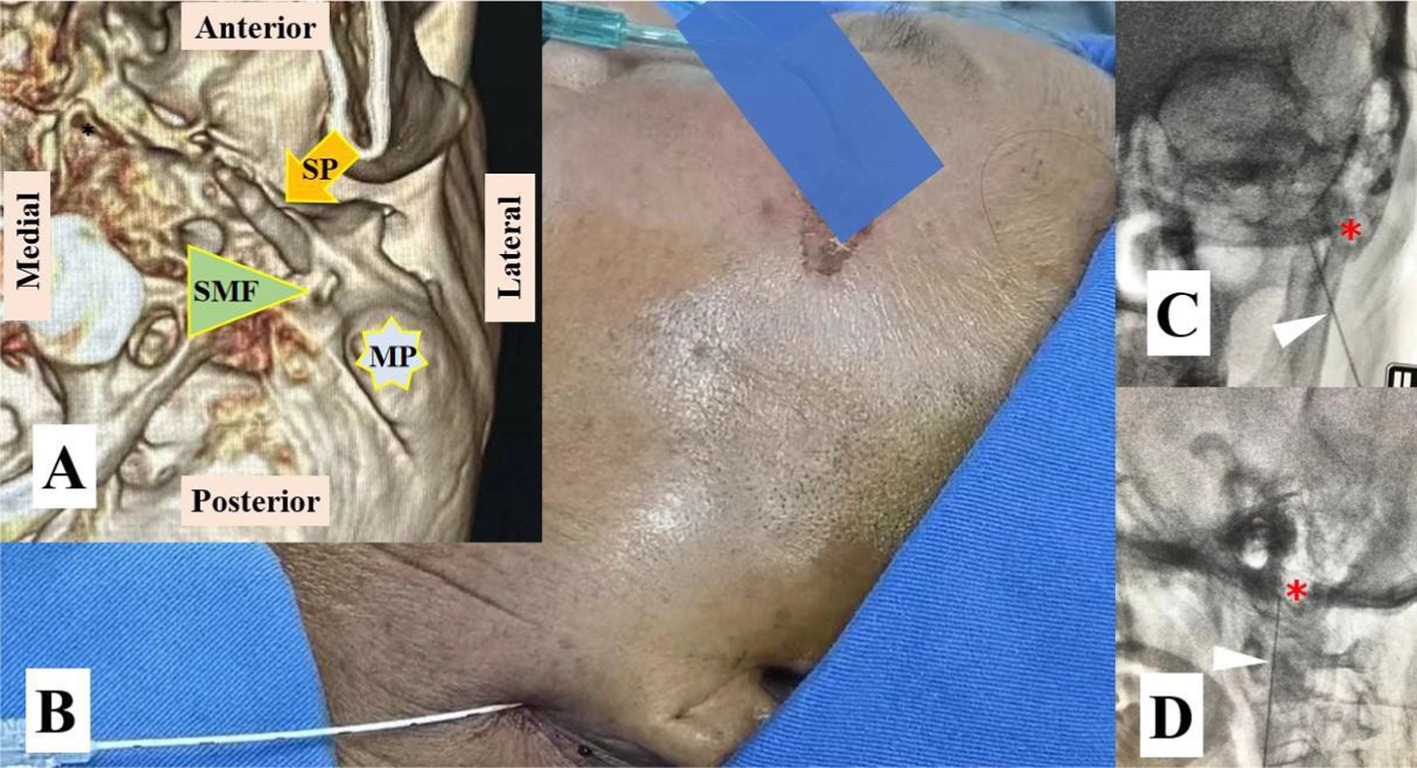
Figure 3. Pulsed radiofrequency (PRF) at the stylomastoid foramen of the facial nerve. (A) VR 3D image showing the stylomastoid foramen (SMF) between the mastoid process (MP) and the styloid process (SP). (B) PRF was performed with a modified procedure based on the Nadbath– Rehman block method; (C) The RF needle is located medial to the MP in the anteroposterior view of digital subtraction angiography (DSA); (D) The RF needle is located anterior to the MP in the lateral view of DSA. The red asterisk represents the MP and the white triangle represents the RF needle.
The patient herself evaluated the clinical efficacy before and after treatment. Persistent pain was rated as moderate before treatment. The pain decreased by 60% on the fifth day of treatment and had disappeared after 3 months. The range of persistent pain was reduced from the external auditory canal and auricle before treatment to the external auditory canal on the fourth day of treatment until it disappeared during the 3-month follow-up. FNS showed an excellent effect on breakthrough pain. Her breakthrough pain decreased by more than 50% on day one and disappeared completely on day five. The frequency of breakthrough pain decreased dramatically from more than 24 times daily before treatment to four times on the first day, two times on the second day, one time on the third day, and two times on the fourth day during treatment, and it disappeared on the fifth day. The duration of each seizure decreased from 3–5 min before treatment to 0.5–1 min on the first day, 30 s on the second day, 20 s on the third day, and 5 s on the fourth day during treatment. The radiating pain to the frontal and temporal regions on the first day, to the mandibular and pharyngeal regions on the second day, and the occipital region and auricle on the third day had completely disappeared, and the breakthrough pain occurred only in the external auditory meatus (EAM) on the fourth day. Tactile allodynia disappeared during the 3-month follow-up. The patient reported stable efficacy when the t-FNS was switched off on day 7, with no relapses (Table 1).
A 55-year-old male patient presented to our pain clinic with severe neuralgia in the left head and face. He had a rash in the external auditory canal and frontotemporal region 1 month before his admission to our clinic. He was given intravenous acyclovir (0.25 g three times daily) for herpes simplex virus and oral gabapentin (initially 300 mg three times daily and up to 600 mg three times daily) and tramadol extended-release capsules (100 mg daily) for pain relief. However, his aggravated pain made him unbearable after the rash improved; in particular, he was overwhelmed by the breakout pain.
The patient also reported a persistent type of pain with paroxysmal exacerbations in intensity. His persistent pain was “incessant tingling, burning, and itching” in the external auditory canal, auricle, and frontal-temporal regions with an intensity of 5/10 on an NRS. His severe stabbing and shooting otalgia and frontal-temporal neuralgia radiated to the infraorbital, mandibular, pharyngeal, upper cervical, and occipital regions less than an hour apart, and the intensity was 10/10 on an NRS and lasted approximately 5–6 min during the pain attack (Figures 4,B). The patient could not fall asleep and expressed exaggerated fear, anxiety, and depression. Physical examination revealed pigmentation and hypoesthesia of the skin in the supraorbital and frontal regions and exhibited tactile allodynia of the skin in the supraorbital frontal-temporal region and external auditory canal.
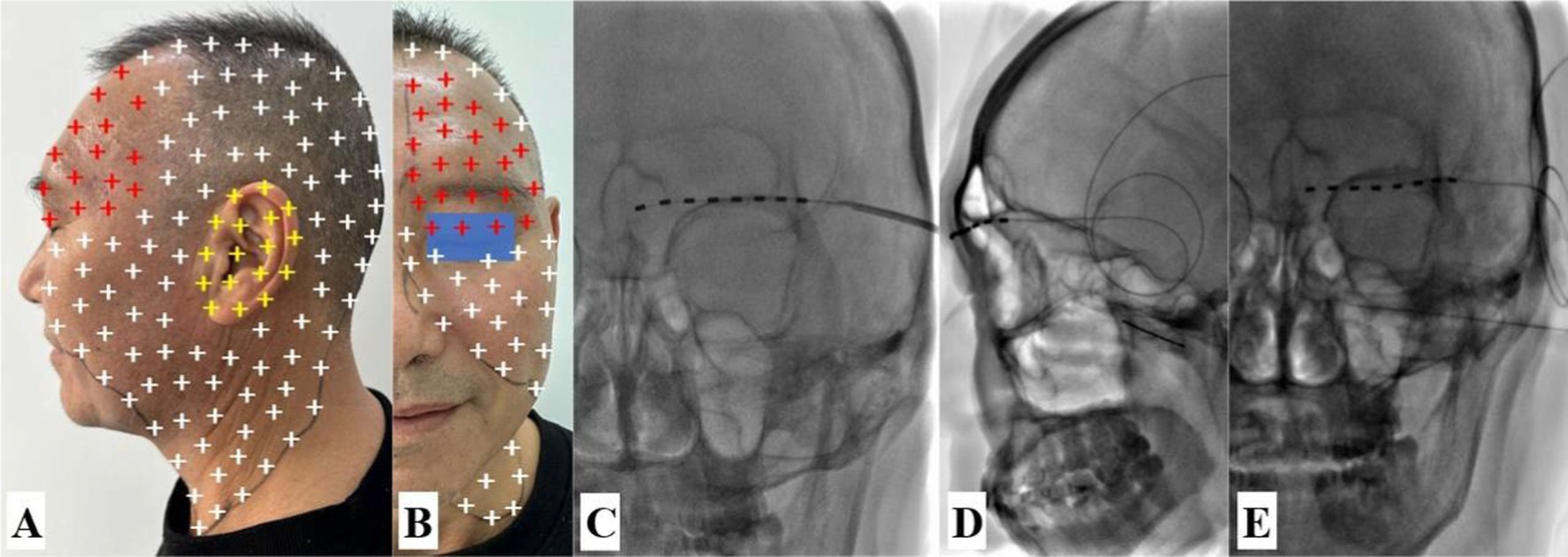
Figure 4. Schematic representation of pain distribution, transcutaneous supraorbital nerve stimulation (t-SNS), and diagnostic maxillary nerve block. (A,B) Persistent pain region (red and yellow plus) and eruptive pain region (red, yellow and white plus). (C) The t-SNS was implanted under DSA guidance. (D,E) DSA-guided diagnostic maxillary nerve block via the pterygopalatine fossa was confirmed in the lateral and anteroposterior views. The white arrow points to the needle.
Based on the affected painful dermatome, the pain in the frontal and temporal areas decreased significantly after the blockade of the supraorbital nerve with 1% lidocaine. Transcutaneous supraorbital nerve stimulation was performed after informed consent was obtained, and his persistent pain and breakthrough pain in the frontal and temporal regions also decreased, but there was no benefit in other regions (Figure 4C). Diagnostic blockade of the maxillary nerve was completely ineffective (Figures 4D,E), but his severe paroxysmal pain stopped immediately after diagnostic facial nerve block at the stylomastoid foramen with 1 mL of 1% lidocaine. Based on the symptoms and physical signs in conjunction with the results of the diagnostic nerve block, the patient was diagnosed with atypical RHS involving the fifth cranial nerve (CN V). The t-FNS was recommended based on our previous successful treatment of case 1, and the risks, benefits, and alternatives of the procedure were discussed with the patient. After obtaining informed consent, the procedure was successfully performed according to the pilot protocol (Figure 5).
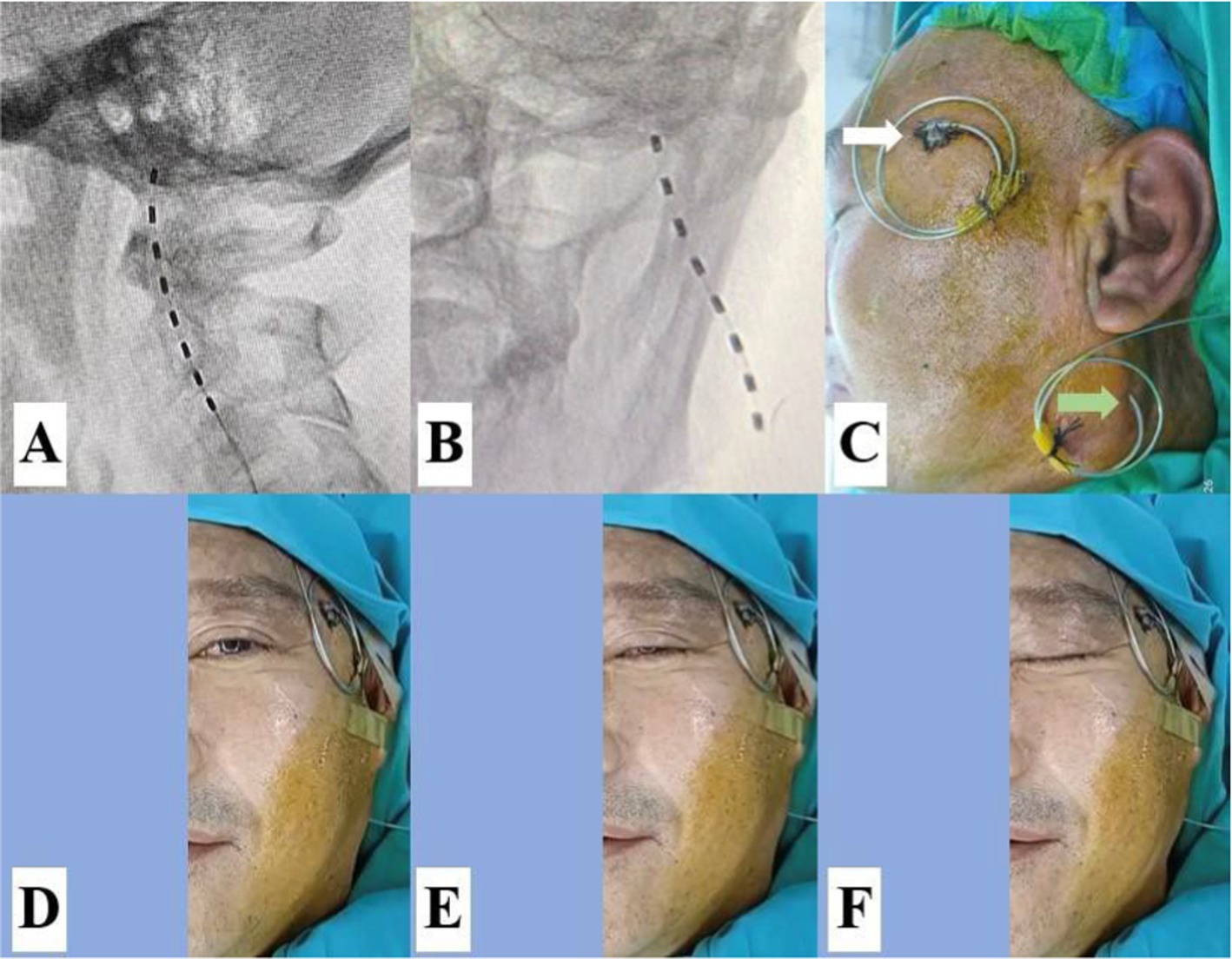
Figure 5. It was confirmed that the lead was placed close to the facial nerve at the stylomastoid foramen. (A,B) The lead tip was located anterior and medial to the MP. (C) The lead for transcutaneous supraorbital nerve stimulation (t-SNS, white arrow) and the lead for transcutaneous facial nerve stimulation (t-FNS, green arrow) were attached separately to the skin. (D) Slight ptosis was observed before the stimulation test. (E,F) The slight and distinct contractions of the facial muscles were elicited by the stimulation test at 0.3 V and 0.5 V.
t-FNS combined with t-SNS showed excellent pain relief in atypical RHS with CN V involvement. The moderate intensity of persistent pain decreased by more than 50% on day 9. On day 11, persistent pain in the forehead region and auricle had completely disappeared, and on day 15, after treatment, the pain in the external auditory canal completely disappeared. However, his mild, persistent pain in the temporal region persisted for a year. Before the treatment, the patient suffered from severe paroxysmal pain. The treatment resulted in excellent pain relief, including intensity, frequency, duration, and distribution of pain during paroxysmal exacerbations. The intensity, frequency, and duration of the pain attacks were already reduced by 50% on the first day and ceased completely on day 15 after treatment. In addition, the treatment stopped the onset in the pharyngeal and upper cervical regions on the third day, in the infraorbital and mandibular regions on the fifth day, in the frontal and occipital regions on the seventh day, and in the auricle on the thirteenth day. Finally, the idiopathic otalgia completely disappeared on day 15. However, his mild tactile allodynia in the temporal region was still present for 1 year (Table 2).

Table 2. The efficacy of transcutaneous facial nerve stimulation (t-FNS) and supraorbital nerve stimulation (t-SNS).
The patient was a 67-year-old woman with a history of painful right EAM and ipsilateral facial paralysis without hearing loss and vertigo. She was diagnosed with typical RHS and was administered intravenous prednisolone (1 mg/kg per day), acyclovir (15 mg/kg per day), oral pregabalin (initial dosage of 75 mg three times a day and up to 150 mg three times a day), and mecobalamin (0.5 mg three times a day). Although the vesicular rash had disappeared 10 days later, the paroxysmal otalgia progressively worsened. Therefore, additional tramadol extended-release capsules (100 mg every 6 h) were administered orally, but this was ineffective. The patient was admitted to our pain clinic after 1 month for further treatment.
She reported persistent pain with paroxysmal exacerbations of intensity. Her persistent pain was an “incessant tingling and itching” in the EAM with an intensity of 4/10 on the NRS, but her sharp and shooting otalgia was 8/10 on an NRS, lasted approximately 3–5 min, and occurred more than 10 times a day with indefinite interval time. Physical examination revealed a cutaneous eschar in the EAM and facial paralysis on the right side (Figures 6A,B). Routine blood and urine tests, magnetic resonance imaging of the brain, computed tomography, and electrocardiography findings were normal. She expressed heightened fear and anxiety about the possibility of experiencing next attack.
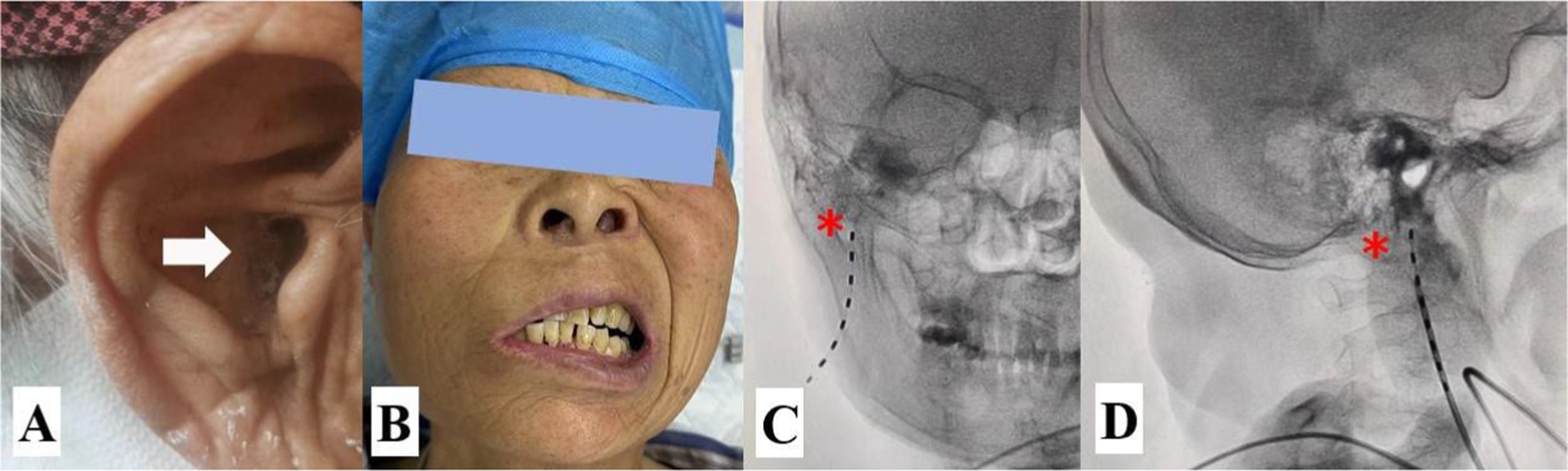
Figure 6. Clinical features and lead placement. (A) Cutaneous eschar in the EAM; (B) Facial palsy; (C) The lead tip was located medial to the MP in the anteroposterior view of the DSA. (D) The tip of the lead was located anterior to the MP in the lateral view of the DSA. The white arrow indicates the cutaneous eschar and pain location in the EAM, and the yellow asterisk indicates the MP.
According to her medical history of symptoms and physical signs, she was diagnosed with typical RHS with severe otalgia. The FNS was recommended, and the risks, benefits, and alternatives of the procedure were discussed with the patient. After obtaining informed consent, the procedure was performed in accordance the pilot protocol (Figures 6C,D).
The t-FNS significantly relieved both the persistent pain and the breakthrough pain in EAM, especially the breakthrough pain. The patient’s persistent otalgia decreased significantly from moderate to mild after treatment for 5 consecutive days, and the mild persistent pain completely resolved at the 2-month follow-up. The severe pain attack decreased to moderate pain immediately after the start of treatment and disappeared, and the breakthrough pain had completely disappeared for 5 consecutive days after treatment. The frequency of seizures decreased from more than 10 times a day before treatment to 4 times on the first day, twice on the third and fifth day, and finally disappeared. The duration of the individual seizures also decreased from 3 to 5 min before treatment to 1 min, 20 s, and 5 s on the first, third, and fifth day, respectively (Table 3).
Among the three cases, two atypical cases of RHS with severe neuralgia in unusually extensive dermatomes without facial paralysis and one typical case of RHS with severe otalgia and facial paralysis. Typical RHS is known to cause otalgia through its effects on the facial nerve (CN VII). However, atypical RHS affects multiple cranial nerves, particularly V, VII, VIII, and XII, as well as the cervical nerves, and often causes severe neuralgia in unusually widespread facial and cervical dermatomes (Sun et al., 2011; Xanthopoulos et al., 2002). Although the first case experienced unusually widespread pain, it disappeared immediately after the facial nerve was blocked. Therefore, her widespread pain in the frontal-temporal, mandibular, pharyngeal, upper cervical, and occipital regions was caused by the breakthrough of the facial nerve postherpetic neuralgia. The second case also experienced unusually widespread pain, but he suffered from a rash in the external auditory canal and frontal-temporal region, and his pain in the frontal-temporal region decreased significantly after blockage of the supraorbital nerve. His widespread pain in the frontal-temporal, infraorbital, mandibular, pharyngeal, upper cervical, and occipital regions also disappeared immediately after blocking the facial nerve. Thus, case 2 was diagnosed as atypical RHS with CN V involvement. The third case was diagnosed as a typical RHS with severe otalgia based on her clinical features.
The mechanism of RHS neuralgia is primarily related to polycranial neuritis, in which both sensory and motor nerves are significantly damaged. Some patients, especially those with underlying conditions such as diabetes, severe infections that further exacerbate the nerve damage and increase the neuralgic pain, making them resistant to conventional treatments (Sun et al., 2011). The pathophysiology underlying the intractable pain appears to stem from viral-induced nerve inflammation and subsequent neuronal degeneration. This neuronal damage disrupts normal sensory signal transmission, resulting in persistent and severe pain sensations even after the primary infection subsides (Alicandri-Ciufelli et al., 2012). The interplay between neuronal inflammation, degeneration, and persistent sensitization of pain pathways contributes to the chronicity and intractability of neuralgia associated with RHS (Sun et al., 2011).
Although there are various treatments for postherpetic neuralgia (PHN), spinal cord stimulation (SCS) is an established treatment option for patients with pharmacologically resistant PHN (Isagulyan et al., 2023). Recently, some studies on peripheral nerve stimulation (PNS) have actively investigated and increasingly used in clinical practice. This therapeutic approach uses electrical stimulation of the peripheral nerves to relieve pain and has shown promise in the treatment of PHN. Yakovlev and Peterson (2007) detailed the PNS in the right subscapular and right paraspinal areas of the upper thoracic region in a case with intractable PHN in which conventional treatment failed to provide improvement, highlighting success in the treatment of PHN (Yakovlev and Peterson, 2007). Several case studies have further illustrated the successful outcomes of PNS for the treatment of herpetic trigeminal neuralgia (Wan and Song, 2021; Liu et al., 2021; Han et al., 2020). In the current report, the first patient experienced about 20% pain relief for approximately 4 h after PRF of the facial nerve was performed. Therefore, t-FNS was recommended to relieve her PHN. Our results align with prior research on neuromodulation for neuropathic pain, which demonstrated varying degrees of pain relief in refractory cases. Compared to transcutaneous electrical nerve stimulation (TENS) and repetitive transcranial magnetic stimulation (rTMS), t-FNS has proven to be a reliable method for RHS neuralgia with fewer procedural risks.
In terms of efficacy, t-FNS showed good therapeutic results for different pain patterns. At the end of treatment, the paroxysmal pain in three patients and persistent pain in two patients completely disappeared. Only one patient experienced long-term mild pain in the distribution area of a branch of the trigeminal nerve, which continued to decrease over time. In terms of safety, this technique is performed under the combined guidance of ultrasound and DSA. Ultrasound monitoring can prevent vascular damage, whereas DSA guidance enables precise implantation of the electrodes in the target area. Anesthesia is provided through intravenous sedation and local anesthesia. Intravenous sedation not only alleviates the patient’s anxiety during the procedure but also allows for timely reporting of complaints. This proactive communication can help prevent the occurrence of complications such as nerve and vascular injuries. A local infiltration of 10 mL of 0.5% lidocaine is injected into the tissue from the skin to the stylomastoid foramen, which not only provides effective pain relief but also dilates the puncture channel. Thus, the risk of nerve and vascular injury was further reduced during the procedure. No procedure-related adverse events were observed during follow-up. All patients tolerated the intervention well, with no reports of infection, bleeding, or unintended nerve stimulation effects. Although no complications were observed in this study, potential risks such as transient discomfort at the stimulation site, device-related irritation, or inadequate response should be considered in future research.
Despite its promising results, this study has several limitations. First, as a retrospective case series with a small sample size, the findings may not be generalized to a broader patient population. Selection bias may also be present because only patients who received t-FNS were included. Additionally, the lack of a control group prevents direct comparisons with other treatment modalities. Future research should focus on optimizing stimulation parameters (e.g., frequency, intensity, and duration) to maximize clinical efficacy. Finally, prospective trials with longer follow-up periods are essential to assess the durability of treatment effects and to identify predictors of long-term success.
t-FNS provides immediate and long-term pain relief for patients suffering RHS neuralgia refractory to conventional therapy. The procedure was performed under ultrasound guidance in combination with DSA, effectively avoiding procedure-related complications. These findings suggest that t-FNS may serve as a promising minimally invasive neuromodulation option for patients who have exhausted conventional treatments. However, given the limitations of this case series, additional well-designed controlled trials are necessary to confirm its efficacy, establish optimal patient selection criteria, and standardize treatment protocols for broader clinical application.
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
The studies involving humans were approved by The Affiliated Hospital of Guilin Medical University. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Y-BD: Methodology, Investigation, Writing – original draft. YZ: Investigation, Methodology, Project administration, Writing – review & editing. Y-XL: Investigation, Writing – original draft. CL: Investigation, Writing – original draft. P-PJ: Data curation, Writing – original draft. S-YW: Data curation, Writing – original draft. W-YG: Conceptualization, Formal analysis, Funding acquisition, Methodology, Supervision, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the National Natural Science Foundation of China (82060221).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Alicandri-Ciufelli, M., Aggazzotti-Cavazza, E., Genovese, E., Monzani, D., and Presutti, L. (2012). Herpes zoster oticus: a clinical model for a transynaptic, reflex pathways, viraltransmission hypotheses. Neurosci. Res. 74, 7–9. doi: 10.1016/j.neures.2012.06.001
Choo, P. W., Galil, K., Donahue, J. G., Walker, A. M., Spiegelman, D., and Platt, R. (1997). Risk factors for postherpetic neuralgia. Arch. Intern. Med. 157, 1217–1224.9183233. doi: 10.1001/archinte.1997.00440320117011
Coulson, S., Croxson, G. R., Adams, R., and Oey, V. (2011). Prognostic factors in herpes zoster oticus (ramsay hunt syndrome). Otol. Neurotol. 32, 1025–1030. doi: 10.1097/MAO.0b013e3182255727
Giralt, S. I., Carvajal, G., García-Janeras, A., Fabà, A., and Nishishinya, A. M. B. (2020). A severe case of Ramsay hunt syndrome treated with acupuncture and related techniques. Complement Ther. Clin. Pract. 39:101119. doi: 10.1016/j.ctcp.2020.101119
Han, R., Guo, G., Ni, Y., Wang, Z., Yang, L., Zhang, J., et al. (2020). Clinical efficacy of short-term peripheral nerve stimulation in Management of Facial Pain Associated with Herpes Zoster Ophthalmicus. Front. Neurosci. 14:574713. doi: 10.3389/fnins.2020.574713
Isagulyan, E., Tkachenko, V., Semenov, D., Asriyants, S., Dorokhov, E., Makashova, E., et al. (2023). The effectiveness of various types of electrical stimulation of the spinal cord for chronic pain in patients with Postherpetic neuralgia: a literature review. Pain Res. Manag. 2023, 6015680–6015688. doi: 10.1155/2023/6015680
Jacques, N., Karoutsos, S., Aubry, K., and Nathan-Denizot, N. (2019). Peripheral nerve block efficacy on refractory neuralgia complicating Ramsay hunt syndrome: a case report. A A Pract. 13, 215–217. doi: 10.1213/XAA.0000000000001035
Jeon, Y., and Lee, H. (2018). Ramsay hunt syndrome. J. Dent. Anesth. Pain Med. 18, 333–337. doi: 10.17245/jdapm.2018.18.6.333
Kim, Y. S., Son, J. S., Lee, H., and Doo, A. R. (2021). A case report of refractory otalgia after Ramsay hunt syndrome successfully treated by applying pulsed radiofrequency to the great auricular nerve: a CARE-compliant article. Medicine 100:e27285. doi: 10.1097/MD.0000000000027285
Lee, H. H., Huang, L. K., Hu, C. J., and Chen, C. C. (2014). Atypical Ramsay hunt syndrome. Acta Neurol. Taiwanica 23, 80–81
Liao, Y. M., Lu, H. F., Xie, P., Zhao, Y., Han, Q., Zhang, Q. X., et al. (2021). Thermographic follow-up of postherpetic neuralgia (PHN) subsequent to Ramsay hunt syndrome with multicranial nerve (V, VII, VIII and IX) involvement: a case report. BMC Neurol. 21:39. doi: 10.1186/s12883-021-02071-5
Liu, D. Y., Chen, J. S., Lin, C. Y., Gong, Q. J., Zhao, Q., and Wan, L. (2021). Subcutaneous peripheral nerve stimulation for treatment of acute/subacute herpes zoster-related trigeminal neuralgia: a retrospective research. Clin. J. Pain 37, 867–871. doi: 10.1097/AJP.0000000000000981
Murakami, S., Hato, N., Horiuchi, J., Honda, N., Gyo, K., and Yanagihara, N. (1997). Treatment of Ramsay hunt syndrome with acyclovir-prednisone: significance of early diagnosis and treatment. Ann. Neurol. 41, 353–357. doi: 10.1002/ana.410410310
Nishizawa, T., Ishikawa, K., Matsuo, T., Higuchi, N., Ishiguro, K., and Mori, N. (2021). Atypical Ramsay hunt syndrome (zoster sine herpete) with otitis media. J. Gen. Fam. Med. 22, 344–346. doi: 10.1002/jgf2.433
Sarathy, A., An, C., Sajisevi, M., and Brundage, W. (2023). A case report of refractory postherpetic neuralgia after Ramsay hunt syndrome treated with tympanic nerve neurectomy. Ear Nose Throat J. 2023:1455613231189950. doi: 10.1177/01455613231189950
Soumekh, F. (2024). Botulinum toxin subdermal injection for the treatment of neuropathic pain in Ramsay hunt syndrome (P5-13.006). Neurology 102:7_supplement_1. doi: 10.1212/WNL.0000000000204966
Sun, W. L., Yan, J. L., and Chen, L. L. (2011). Ramsay hunt syndrome with unilateral polyneuropathy involving cranial nerves V, VII, VIII, and XII in a diabetic patient. Quintessence Int. 42, 873–877
Tiemstra, J. D., and Khatkhate, N. (2007). Bell’s palsy: diagnosis and management. Am. Fam. Physician 76, 997–1002
Wan, C. F., and Song, T. (2021). Short-term peripheral nerve stimulation relieve pain for elder herpes zoster ophthalmicus patients: a retrospective study. Neuromodulation 24, 1121–1126. doi: 10.1111/ner.13288
Xanthopoulos, J., Noussios, G., Papaioannides, D., Exarchakos, G., and Assimakopoulos, D. (2002). Ramsay hunt syndrome presenting as a cranial polyneuropathy. Acta Otorhinolaryngol. Belg. 56, 319–323
Yakovlev, A. E., and Peterson, A. T. (2007). Peripheral nerve stimulation in treatment of intractable postherpetic neuralgia. Neuromodulation 10, 373–375. doi: 10.1111/j.1525-1403.2007.00126.x
Keywords: Ramsay Hunt syndrome, postherpetic neuralgia, otalgia, facial nerve stimulation, neuromodulation
Citation: Deng Y-B, Zhong Y, Lin Y-X, Lin C, Ji P-P, Wang S-Y and Gong W-Y (2025) Successful treatment of intractable neuralgia in patients with typical and atypical Ramsay Hunt syndrome by transcutaneous facial nerve stimulation: a case series study. Front. Neurosci. 19:1554490. doi: 10.3389/fnins.2025.1554490
Received: 02 January 2025; Accepted: 10 March 2025;
Published: 02 April 2025.
Edited by:
Michela Chiappalone, University of Genoa, ItalyReviewed by:
Svetlana Valjarevic, University of Belgrade, SerbiaCopyright © 2025 Deng, Zhong, Lin, Lin, Ji, Wang and Gong. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wei-Yi Gong, OTQyMjYxNTk3QHFxLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.