
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Neurosci. , 11 January 2022
Sec. Neuropharmacology
Volume 15 - 2021 | https://doi.org/10.3389/fnins.2021.787512
This article is part of the Research Topic Complementary/Alternative Therapies for Epilepsy and Related Psychiatric Comorbidities View all 7 articles
Introduction: Millions all over the world live with epilepsy, and they may require long-term drug treatment. The use and interest in complementary and alternative medicine (CAM) have grown over the previous years. Coadministration of herbal products with medicines may result in adverse drug reactions (ADRs) and/or unfavorable interactions. The aims of this study were to determine the prevalence of CAM use among patients with epilepsy, to compare the results to those of the patients with diabetes mellitus (DM), to reveal factors that may drive the use of CAM, and to measure outcomes and adherence. It was also our intent to have state-of-the-art information on CAM use in our region among patients with the two diseases above.
Materials and Methods: We conducted a non-interventional study using a self-developed questionnaire. It was distributed among adult patients with either epilepsy or DM who also suffered from cardiovascular consequences. A database was compiled from the anonymous questionnaires filled in voluntarily by the patients. Basic statistics were used to analyze this database.
Results: A total of 227 questionnaires were filled in by 127 patients (55.9%) with epilepsy and 100 patients (44.1%) with DM. Mean age was 54.54 ± 17.33 years. Of the patients, 50.2% were male. Average body weight was 80.3 ± 17.3 kg. Of the patients, 22 (9.7%) used CAM because they believed in CAM. Two of them reported ADRs. Among the patients with epilepsy, the ratio was only 7.9% compared to 12% among those with DM. While the number of CAM users was higher among younger patients with epilepsy, it was the elderly patients with DM who tended to use CAM.
Conclusion: Attention should be paid to reliance on CAM during the follow-up. Our finding that health-conscious patients tend to use CAM more often (than the general population) may indicate it is necessary to discuss CAM usage sincerely. CAMs modulating cytochrome P450 (CYP) enzymes were the most common, leading to interactions with medication used and resulting in ADRs. This shows the importance of educating patients and treating team including clinical pharmacists in this field.
Millions all over the world suffer from epilepsy. It affects both sexes and all ages with slightly higher prevalence and incidence in men. There are dozens of risk factors closely associated with the onset of epilepsy (Walsh et al., 2017). Greater frequency of stroke, neurodegenerative diseases, and tumors are the most common etiological factors among the elderly (Beghi, 2020). In most cases, epilepsy requires lifelong treatment and/or follow-up in different life stages, posing a challenge for the patient, family, and attending epileptologist alike.
Despite patients having access to a high variety of new antiepileptic drugs (AEDs) in many parts of the world, the old ones are still essential for different reasons (Vajda and Eadie, 2014).
Diabetes mellitus (DM) is a burden of health care, and it leads to several complications, e.g., macrovascular complications (Amutha and Mohan, 2016). DM also requires lifelong treatment with the importance of lifestyle changes.
In a population-based study of epilepsy incidence in association with type 2 diabetes mellitus (T2DM) and severe hypoglycemia based on Taiwan’s National Health insurance claims incorporating 751,792 people with T2DM and 824,253 matched controls identified in 2002–2003 with follow-up until incidence of epilepsy in 2011 (Chin-Li et al., 2018), it was found that T2DM may increase the risk of epilepsy independently of severe hypoglycemia. That study showed 50% elevated hazard of epilepsy in T2DM patients consistently observed for all gender and age stratifications. Moreover, both epilepsy and T2DM patients are commonly prescribed many medicines simultaneously for long periods, and they may seek complementary and alternative medicine (CAM) therapies, which makes it useful to compare patients’ adherence to their prescribed medications, CAM use, and the impact on patients’ satisfaction.
Both diseases are common and have standardized treatment protocols. These diseases have only one pharmacological class as a treatment group; on the contrary, hypertension has more. Similar to DM, epilepsy rarely has severe consequences such as malignant arrhythmia, prolongation of QT, and sudden unexpected death in epilepsy (SUDEP) (Devinsky et al., 2016).
According to the National Center for Complementary and Alternative Medicine (NCCAM) and National Institutes of Health (NIH), “complementary and alternative medicine or modalities (CAM)” was defined as health care approaches with a history of use or origins outside of mainstream medicine (Kramlich, 2014). The use and interest in CAM have grown over the previous years, which emphasizes the importance of considering various contributing factors. Studies have reported a significant increase in CAM use among patients living with chronic diseases (Bücker et al., 2008). The main motivation for CAM use is to avoid polytherapy and disappointment due to the results after conventional therapies. Media, family, and friends are the main sources of information for CAM. Among the elderly, increasing CAM use could be detected, including using herbal supplements concomitantly with conventional medications. This might be due to various concerns like polypharmacy, decreased organ functions, and increased sensitivity to some medications (Elmer et al., 2007). Patients tend to use CAM thinking that CAM therapy is safe and being unaware of the potential adverse effects. Unfortunately, these expose them to an increased risk for admission to an intensive care unit (Bello et al., 2012). CAM therapies may be considered as a choice when conventional care turns out to be unsuccessful or when therapy escalation is deemed to be too risky or no longer beneficial for severely ill patients (Gilmour et al., 2011).
Complementary and alternative medicine therapies, especially herbal medication, can modify the effect of AEDs and antidiabetics, even decrease them and result in ineffectiveness; moreover, they can cause toxic adverse drug reactions (ADRs). CAM may have unfavorable interactions with antidiabetics. Herbals, as an example of CAM, can affect clinical safety and efficacy via additive/synergistic or antagonistic interactions among the herbal components and drug molecules. While negative or harmful interactions tend to receive more attention due to safety considerations, additive/synergistic effects induced by herbal drug interactions may result in an enhancement of desired pharmacological effects (Gupta et al., 2017).
Gymnema sylvestre is an example of an unfavorable herbal interaction with antidiabetics. In an animal study using a chemically induced diabetic rat model, a decrease in plasma metformin concentration and increase in blood glucose levels were seen in animals treated with the combination of gymnema tea and metformin when compared to those receiving metformin alone, suggesting an antagonistic interaction between metformin and gymnema (Raja et al., 2013).
Traditional medicines for the treatment of DM are probably based mainly on treatment of its obvious symptoms of pronounced thirst and polyuria (Marles and Farnsworth, 1995). Nature is an outstanding source of antidiabetic medicines (Chang et al., 2013), and plants are valuable dietary supplements to improve blood glucose control and prevent long-term complications in T2DM (Gallagher et al., 2013). Folklore remedies are used for managing DM due to their non-toxic nature, cost-effectiveness, easy availability, and long-lasting effects over their synthetic counterparts (Bhagour et al., 2016). Certain fruits and vegetables are functional foods, and their consumption reduces the incidence of T2DM. Hypoglycemic effects of fruits and vegetables may be due to their inducing nature on pancreatic β-cells for insulin secretion or bioactive compounds such as flavonoids, alkaloids, and anthocyanins, which act as insulin-like molecules or insulin secretagogues (Beidokhti and Jager, 2017). Examples of used antidiabetic CAM therapies are avocado, Musa paradisiaca, Aloe vera, barberry, caraway, olive oil, and dill.
Focusing on patients living with epilepsy in countries that apply a western-based medical system, CAM is used not only to enhance general health but also to prevent seizures or to alleviate symptoms of comorbidities or the ADRs of antiepileptic medications (Ekstein and Schachter, 2010). The most frequently taken products include ginseng, Ginkgo biloba, St. John’s wort, echinacea, garlic, and soy. Interestingly, the aforementioned products have not been reported to have either beneficial or detrimental effects on seizures, though their presumed activity on the P450 system could potentially lead to interactions with AEDs metabolized by the liver. Melatonin, kava kava, and valerian were reported to have sleep-inducing and anticonvulsant effects; however, melatonin and kava kava were also associated with the aggravation of epilepsy. Ephedra and caffeine have been linked to proconvulsive effects. Some infrequently used products have shown beneficial effects on seizures, epilepsy comorbidities, or complications of epilepsy including skullcap, grapefruit juice, hops, and omega-3 fatty acids. Other countries have widely practiced forms of traditional medicine, among which Ayurveda and traditional Chinese medicine (TCM) are the best known. In Ayurveda, epileptic patients are prescribed mixtures of natural products, containing herbal extracts (like Acacia arabica, Acoruscalamus, and Bacoppamonnieri), as well as animal ghee, honey, and milk. Likewise, TCM involves mixtures of different herbal extracts (each containing many active compounds) either to treat the seizure disorder directly or to maintain the general wellbeing of a patient.
In many instances, herbal drugs are used simultaneously with modern drugs. Generally, all drugs with a narrow therapeutic index may either have increased adverse effects or be less effective when used in conjunction with herbal products (Mehmood et al., 2019). Efficacy and clinical safety can be affected via additive/synergistic or antagonistic interactions among the herbal components and drug molecules (Gupta et al., 2017).
Diabetes mellitus is one of the chronic diseases reported to increase the probability of CAM use (Bishop and Lewith, 2010). Many interactions between CAM and AEDs through different mechanisms like intrinsic proconvulsant properties have their effect on the cytochrome P450 (CYP) enzymes and P-glycoproteins or changing disposition of AEDs (Samuels et al., 2008). Besides the important ADRs caused by hepatic enzymes, it is very important to mention the role of P-glycoprotein in therapy-refractory epilepsies. It is a well-known fact that some AEDs may induce P-glycoproteins. These interactions are difficult to predict; moreover, most patients do not inform their physicians that they are taking such medicine.
The main goals of this study were as follows: to determine the prevalence of CAM use among our patients with epilepsy and to compare the results to those of the patients with DM, to reveal factors that may drive the use of CAM, and to measure the outcome and adherence in order to improve care. We were also interested whether there was any difference between the prevalence of CAM use in two different chronic disease groups, i.e., epilepsy and DM. Since in our region CAM prevalence had not been studied yet, our intent was to have state-of-the-art information about CAM use.
Two self-developed questionnaires (including open-ended and closed-ended questions) were used in order to investigate the use of CAM and outcomes among adult patients with epilepsy (127 patients) and DM (100 patients). In both groups, the patients were diagnosed at least 1 year prior to filling in the questionnaire. Patients previously diagnosed with epilepsy at the Department of Neurology by two senior epileptologists (IF and KF) took part in regular checkups in a tertiary university hospital. Patients with DM were treated at the departments of cardiology and neurology and already had vascular consequences (e.g., stroke or myocardial infarction) of the disease. As a result, they were hospitalized in a tertiary university hospital and had undergone percutaneous coronary intervention (angioplasty with stent or thrombolysis). Similarly to epilepsy, DM may require lifelong treatment and attention from the family and attending physician in different stages of life. Nevertheless, social acceptance is better in DM; it is not a stigmatizing disease, that is why this population was chosen for comparison.
The following inclusion criteria were used in both groups: (a) only adult patients were involved in this study and (b) patient willingness to participate on a voluntary basis.
Inclusion criteria in the epilepsy group: patient was diagnosed with epilepsy according to International League Against Epilepsy (ILAE) classification (Fisher et al., 2014) and took part in regular checkups prior to be involved in the survey.
Inclusion criteria in the diabetes mellitus group: patients with a primary diagnosis of DM and subsequent vascular consequences; and during the study period: the patient was hospitalized to treat a vascular event.
Exclusion criteria were as follows: (a) dependent patient, (b) serious medical condition, and (c) other major comorbidities.
Patients filled in the questionnaire anonymously and voluntarily between December 2018 and September 2019. It contained questions that were about patients’ demographics, lifestyle activities, seizure freedom, prescribed AEDs, adherence, satisfaction, reported adverse effects, CAM therapy, and quality of life. A patient was a “smoker” if he smoked actively, a “non-smoker” if he had never smoked, and “stopped smoking” if he quitted the habit at least 1 year before filling in the questionnaire. The self-developed questionnaire for DM sufferers had some questions identical with the one used in the other study on epileptic patients. In addition, patients living with DM had specific questions concerning diabetic diet, owning a glucometer at home, frequency of measuring blood glucose levels, family history of DM, last measured fasting blood glucose, last measured HbA1c%, prescribed antidiabetics, adherence, and general satisfaction with therapy.
Both aforementioned surveys were developed through the authors’ collaborative work. Surveys were reviewed and discussed by many clinicians and experts to obtain face validation.
Data were entered in a database for further evaluation. In order to reduce the risk of error, data were first entered into spreadsheet by one of the authors; later, two authors collaborated in reviewing the merged database.
Controlled disease as a good outcome was considered in case of patients living with epilepsy if they declared themselves as seizure-free, in case of patients with DM if fasting glucose level was <7.0 mmol/L and/or HbA1c < 6.5%.
Adherence to prescribed medication was classified into three categories:
1 – Good (as long as patients had taken at least 90% of their prescribed medicines or maximum 3 days of drug holiday a month).
2 – Less often (as long as they had taken at least 50%–90% of their prescribed medicines).
3 – Poor (as long as they had taken at least < 50% of their prescribed medicines).
Physical activity was defined as any physical activity lasting at least 30 min a day.
The five-point Likert scale was used to measure a patient’s overall quality of life, where one meant wellbeing.
Statistical analysis was carried out using the SPSS for Windows 19.0 (SPSS Inc., Chicago, IL, United States) and Microsoft Office Excel 2016.
Two-sample T test and F test were used to analyze our patients’ data. Categorical variables were assessed using Pearson χ2 test and Fisher’s exact test.
As per standard pharmacovigilance practices, the values of the odds ratio (OR) were computed using 2 × 2 contingency table.
Significant differences were considered if p < 0.05.
Two hundred twenty-seven patients completed the questionnaire (127 patients with epilepsy and 100 patients with DM). Of them, 114 (50.2%) were male and 112 (49.3%) were female, while a participant did not state gender preference. Mean age was 54.54 ± 17.33 years; people with epilepsy were significantly younger.
Patients with epilepsy listed the following CAM used parallel to the anticonvulsant therapy: lemon balm (Melissa officinalis) (three patients), pomegranate (Punica granatum), valerian (Valeriana officinalis), rose hip (Rosa canina), aloe vera, Sedacur [contains lemon balm, valerian, hop (Humulus lupulus)], linden (Tilia), mint (Mentha), thymus, and senna.
Patients with DM reported the following CAM used parallel to the antidiabetic therapy: cinnamon (Cinnamomum cassia), dill (Anethum graveolens), European blueberry (Vaccinium myrtillus), nettle (Urtica dioica), gurmar (Gymnema sylvestre), Tea “György” (fantasy name; contains dandelion-Taraxacum officinale, nettle-Urticadioica, perforate St. John’s wort-Hypericum perforatum, European blueberry-Vaccinium myrtillus, and chicory-Cichorium intybus) by three patients.
Patients have not used homeopathic remedies.
Reported CAM therapies alongside their ADRs and potential drug–herb interactions are listed in Table 1. Among ADRs are gastrointestinal (GI) and central nervous system (CNS) symptoms and dermatological conditions. In drug–herb interactions, a wide range of medicines are affected due to the modulation of CYP enzymes such as CYP3A4 and CYP2C9, which are responsible for the metabolism of many medicines.
Basic patients’ characteristics were assessed in Table 2. Comparing patients with epilepsy and DM, statistically significant differences were revealed in body weight, smoking status, and alcohol consumption.
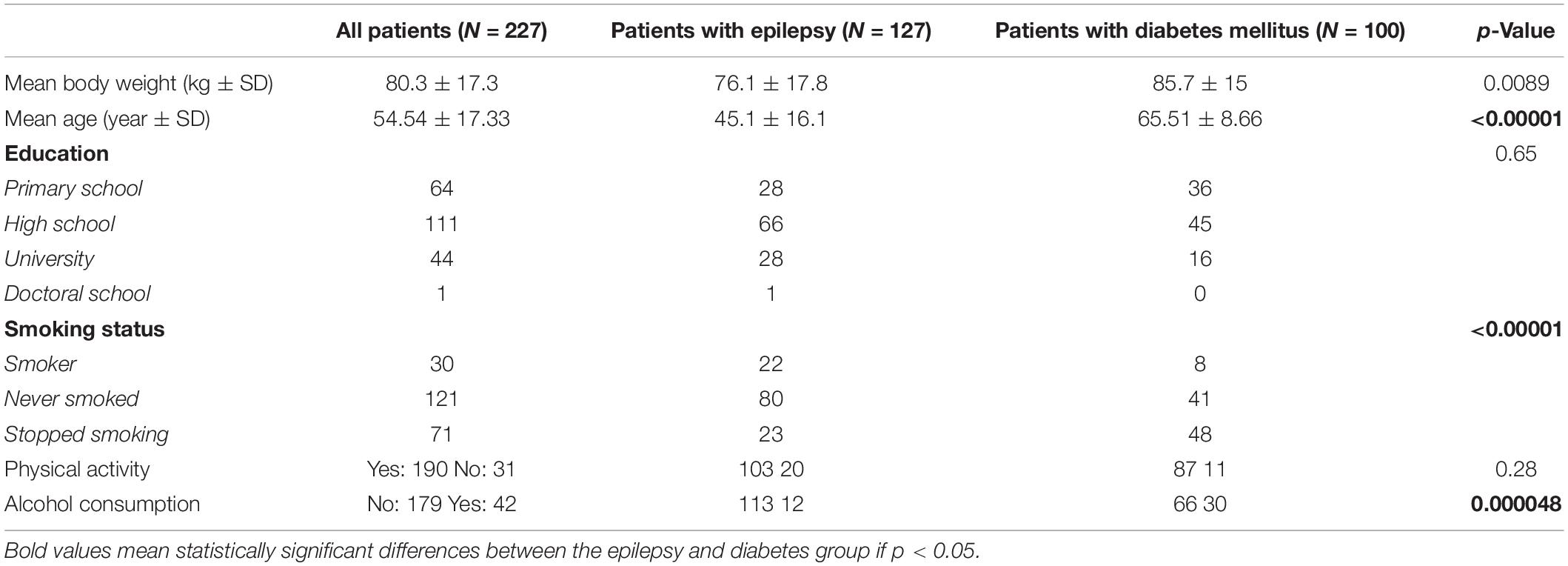
Table 2. Basic characteristics of overall study population, patients living with epilepsy, and patients living with diabetes mellitus.
Patients living with epilepsy had better disease control (73.2% vs. 28%), but CAM was not significant among the groups despite that 7% of the patients with epilepsy had controlled disease using CAM vs. 1% of patients with DM. Adherence rate was higher among patients with epilepsy (Table 3).
The ratio of CAM use was 9.7% in the overall study population. Among patients with epilepsy, it was less than that among diabetic patients; 7.9 and 12%, respectively. Interestingly, among patients living with epilepsy, CAM users were mainly younger, while the CAM-using DM patients were members of the elderly population. Only two patients (9.1% of CAM users–prevalence of CAM ADR) reported ADR from the used CAM (Table 4).
Nevertheless, we failed to find a significant association between CAM use and age, gender, adherence to prescribed medicine (although it should be noted that at 90% CI, it is significant), ADR due to prescribed medicine, control of disease, smoking, satisfaction with prescribed medicines, and education. A patient reporting alcohol consumption and physical activity had 4.56 and 9.1 times odds of using CAM, respectively, compared to a person who did not (Table 5).
In this study, we have included two chronic diseases requiring lifelong treatment. They should be given special attention not only by the patients but by the relatives as well. Epileptic seizure is unforeseen and, in many cases, dramatic. Besides this, epilepsy is stigmatizing and enigmatic, which may motivate patients to use uncommon treatments. These might even give the patient an illusion that they can rule their whimsical disease by themselves. It is a well-known fact that the consequences of DM can be reduced with diet and changes in lifestyle, and this depends on the patients. The increased interest in CAM and potential source of ADR or interaction play a pivotal role in having information on the use of CAM in order to find the best-tailored therapy for each patient.
A systematic review revealed the prevalence of use of any CAM between 9.8 and 76% in the general population and concluded that periodic surveys were important to monitor CAM use at the population level (Harris et al., 2012).
In our study, the overall prevalence was similar to the findings of the above systematic review, but fewer users (7.9%) could be detected among patients with epilepsy. By comparing two groups with a similar cultural background, we could decrease the bias and assume that the characteristics of the diseases contribute to differences in findings (Barner et al., 2010). If compared to the population with DM, treatment characteristics differed in some points such as adherence, which could be attributed to the unexpectedness and dramatic nature of epilepsy; in contrast, in DM, high blood sugar level is “just” a laboratory finding. A seizure is an unpleasant feedback that might affect the quality of life (e.g., job, driving license, and relationships), and most patients try to avoid it. Epileptologists believe that continuous care and a good physician–patient relationship improve trust and sincerity.
Unlike in the article by Nahin et al. (2007), smoking and obesity were not confirmed as risk factors related to CAM use in the current study population. Physical activity was linked to CAM use, which may suggest health-conscious people may want to rule their disease by using additional treatment and make every effort for their wellbeing. This hypothesis is supported by other findings in this study, for instance, CAM use was independent of the control of disease, satisfaction with prescribed medicines, and quality of life assessment.
Surprisingly, alcohol consumption elevated reliance on using CAM by more than four times. But patients have not reported heavy drinking; only one patient confessed daily drinking of two bottles of beer. In a Norwegian study among women, similarly to our findings, more frequent natural medicine use was detected among frequent alcohol drinkers (Sivertsen et al., 2018).
Complementary and alternative medicine users were younger in the “epileptic” group, but it can be attributed to their younger mean age than patients with DM.
In Table 1, most CAMs are used for both epilepsy and DM patients. Despite the small size of the study population, 9.1% of CAM users reported ADRs. These CAMs typically modulated CYP enzymes. The CAMs used by our patients might have had interactions with AEDs, partially on CYP enzymes, partially that absorption could have been disturbed. Being aware of their use, it might prevent an unnecessary change in AED therapy. This finding emphasizes the importance of history taking, which should include CAM use and, in addition to patients’ education, the knowledge of different CAM effects by the treating team including clinical pharmacists.
It must be mentioned that although patients using CAM are only a minority in the studied population, attention should be paid to reliance on CAM during the follow-up. Our finding that health-conscious patients tend to use CAM more often (than the general population) may indicate it is necessary to discuss CAM usage honestly. CAMs capable of modulating CYP enzymes were the most popular among users, but they led to interactions with medication and resulted in ADRs. This shows that educating patients and treating them by a team including a clinical pharmacist in this field are essential.
The authors are aware that the study has several/certain limitations. Nevertheless, the strength of the study is the cross-sectional view and honest responses due to anonymously collected questionnaires. Although the size of the sample was limited, only patients willing to participate were included in the study, which resulted in a selection bias. The facts mentioned above might imply that the study was underpowered to identify all clinically important predictors of CAM use.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving human participants were reviewed and approved by Institutional Ethics Committee of the University of Debrecen (Registration Number: DE RKEB/IKEB 5098-2018 and 5099-2018). Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
All authors listed have made a substantial, direct, and intellectual contribution to the work, and approved it for publication.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Agus, H. H., Tekin, P., Bayav, M., Semiz, A., and Sen, A. (2009). Drug interaction potential of the seed extract of Urtica urens L. (dwarf nettle). Phytother. Res. 23, 1763–1770. doi: 10.1002/ptr.2848
Alamgeer, Muhammad, N. M., Muhammad, R., Muhammad, N. H. M., Taseer, A., Abdul Qayyum, K., et al. (2013). Evaluation of hypoglycemic activity of thymus serpyllum linn in glucose treated mice. IJBMSP 3, 33–36.
Amutha, A., and Mohan, V. (2016). Diabetes complications in childhood and adolescent onset type 2 diabetes-a review. J. Diabetes Complications 30, 951–957.
Andrade, C. (2014). Potentially significant versus clinically significant drug interactions: pomegranate juice as a case in point. J. Clin. Psychiatry 75, e292–e293. doi: 10.4088/JCP.14f09112
Arash, A., Mohammad, M., Jamal, M., Mohammad, T., and Azam, A. (2013). Effects of the aqueous extract of anethum graveolens leaves on seizure induced by pentylenetetrazole in mice. MJMS 20, 23–30.
Arokiyaraj, S., Balamurugan, R., and Augustian, P. (2011). Antihyperglycemic effect of hypericum perforatum ethyl acetate extract on streptozotocin-induced diabetic rats. Asian Pac. J. Trop. Biomed. 1, 386–390. doi: 10.1016/S2221-1691(11)60085-3
Banihani, S., Swedan, S., and Alguraan, Z. (2013). Pomegranate and type 2 diabetes. Nutr. Res. 33, 341–348. doi: 10.1016/j.nutres.2013.03.003
Barner, J., Bohman, T., Brown, C., and Richards, K. (2010). Use of complementary and alternative medicine for treatment among African-Americans: a multivariate analysis. Res. Soc. Adm. Pharm. 6, 196–208. doi: 10.1016/j.sapharm.2009.08.001
Basch, E., Ulbricht, C., Hammerness, P., Bevins, A., and Sollars, D. (2004). Thyme (Thymus vulgaris L.), thymol. J. Herb. Pharmacother. 4, 49–67. doi: 10.1300/j157v04n01_07
Bayani, M., Ahmadi-hamedani, M., and Jebelli Javan, A. (2017). Study of hypoglycemic, hypocholesterolemic and antioxidant activities of iranian mentha spicata leaves aqueous extract in diabetic rats. Iran. J. Pharm. Res. 16, 75–82.
Beidokhti, M., and Jager, A. (2017). Review of antidiabetic fruits, vegetables, beverages, oils and spices commonly consumed in the diet. J. Ethnopharmacol. 201, 26–41. doi: 10.1016/j.jep.2017.02.031
Belemkar, S., Kumar, A., and Pata, M. K. (2013). Pharmacological screening of herbal extract of Piper nigrum (Maricha) and cinnamomum zeylanicum for anticonvulsant activity. Inventi Rapid. 2013, 1–5.
Bello, N., Winit-Watjana, W., Baqir, W., and McGarry, K. (2012). Disclosure and adverse effects of complementary and alternative medicine used by hospitalized patients in the North East of England. Pharm. Pract. 10, 125–135. doi: 10.4321/s1886-36552012000300002
Bhagour, K., Arya, D., and Gupta, R. S. (2016). A review: antihyperglycemic plant medicines in management of diabetes. Acupunct. Relat. Ther. 4, 7–16. doi: 10.1016/j.arthe.2016.11.001
Bishop, F., and Lewith, G. (2010). Who uses CAM? A narrative review of demographic characteristics and health factors associated with CAM use. Evidence based complement. Alternat. Med. 7, 11–28. doi: 10.1093/ecam/nen023
Boudreau, M., and Beland, F. (2006). An evaluation of the biological and toxicological properties of Aloe barbadensis (miller), Aloe vera. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 24, 103–154. doi: 10.1080/10590500600614303
Bücker, B., Groenewold, M., Schoefer, Y., and Schäfer, T. (2008). The use of complementary alternative medicine (CAM) in 1 001 German adults: results of a population-based telephone survey. Gesundheitswesen 70, e29–e36. doi: 10.1055/s-2008-1081505
Cameron, A., Bansal, A., Dua, T., Hill, S., Moshe, S., Mantel-Teeuwisse, A., et al. (2012). Mapping the availability, price, and affordability of antiepileptic drugs in 46 countries. Epilepsia 53, 962–969. doi: 10.1111/j.1528-1167.2012.03446.x
Cárdenas, R. N., González, T. M. E., Aguirre, H. E., Ruíz, G. M., Sampieri, A., Coballase, U. E., et al. (2014). Anticonvulsant and antioxidant effects of Tilia americana var. mexicana and flavonoids constituents in the pentylenetetrazole-induced seizures. Oxid. Med. Cell. Longev. 2014:329172. doi: 10.1155/2014/329172
Cerny, A., and Schmid, K. (1999). Tolerability and efficacy of valerian/lemon balm in healthy volunteers (a double-blind, placebo-controlled, multicentre study). Fitoterapia 70, 221–228.
Chang, C., Lin, Y., Bartolome, A., Chen, Y., Chiu, S., and Yang, W. (2013). Herbal therapies for type 2 diabetes mellitus: chemistry, biology, and potential application of selected plants and compounds. Evid Based Complement Alternat. Med. 2013:378657. doi: 10.1155/2013/378657
Chin-Li, L., Ya-Hui, C., Yu, S., and Chung-Yi, L. (2018). A population-based study of epilepsy incidence in association with type 2 diabetes and severe hypoglycaemia. Diabetes Res. Clin. Pract. 140, 97–106. doi: 10.1016/j.diabres.2018.03.020
Christensen, R., Bartels, E., Altman, R., Astrup, A., and Bliddal, H. (2008). Does the hip powder of Rosa canina (rosehip) reduce pain in osteoarthritis patients?–a meta-analysis of randomized controlled trials. Osteoarthr. Cartil. 16, 965–972. doi: 10.1016/j.joca.2008.03.001
Choudhary, M., Kochhar, A., and Sangha, J. (2011). Hypoglycemic and hypolipidemic effect of Aloe vera L. in non-insulin dependent diabetics. J Food Sci Technol. 51, 90–96. doi: 10.1007/s13197-011-0459-0
Chung, M., Cho, S., Bhuiyan, M., Kim, K., and Lee, S. (2010). Anti-diabetic effects of lemon balm (Melissa officinalis) essential oil on glucose- and lipid-regulating enzymes in type 2 diabetic mice. Br. J. Nutr. 104, 180–188. doi: 10.1017/S0007114510001765
Collins, J., Evason, K., and Kornfeld, K. (2006). Pharmacology of delayed aging and extended lifespan of Caenorhabditis elegans. Exp. Gerontol. 41, 1032–1039. doi: 10.1016/j.exger.2006.06.038
Cramer, K., Charrois, T. L., and Vohra, S. (2006). Valerian: practical management of adverse effects and drug interactions. Can. Pharm. J. 139, 39–41.
De Smet, P. A. G. M., Chandler, R. F., Keller, K., and Hänsel, R. (1993). Adverse Effects of Herbal Drugs. Berlin: Springer.
Devinsky, O., Hesdorffer, D., Thurman, D., Lhatoo, S., and Richerson, G. (2016). Sudden unexpected death in epilepsy: epidemiology, mechanisms, and prevention. Lancet. Neurol. 15, 1075–1088. doi: 10.1016/S1474-4422(16)30158-2
Draz, S. N., Abo-Zid, M. M., Ally, A. F., and El-Debas, A. A. (2010). Hypoglycemic and hypolipidemic effect of, chicory (Cichorium intybus L.) herb in, diabetic rats. Minufiya J. Agric. Res. 35, 1201–1208.
Dwivedi, D., and Gupta, P. (2017). Estimation of heavy metal ions in the leaf extracts of study sample prepared from azadirachta indica, gymnema sylvestre and ocimum sanctum. J. Chem. Cheml. Sci. 7, 781–784.
Ekstein, D., and Schachter, S. (2010). Natural products in epilepsy-the present situation and perspectives for the future. Pharmaceuticals 3, 1426–1445. doi: 10.3390/ph3051426
Elmer, G., Lafferty, W., Tyree, P., and Lind, B. (2007). Potential interactions between complementary/alternative products and conventional medicines in a Medicare population. Ann. Pharmacother. 41, 1617–1624. doi: 10.1345/aph.1K221
Espiritu, M., Chen, J., Yadav, J., Larkin, M., Pelletier, R., Chan, J., et al. (2020). Mechanisms of herb-drug interactions involving cinnamon and CYP2A6: focus on time-dependent inhibition by cinnamaldehyde and 2-methoxycinnamaldehyde. Drug Metab. Dispos. 48, 1028–1043. doi: 10.1124/dmd.120.000087
Fisher, R., Acevedo, C., Arzimanoglou, A., Bogacz, A., Cross, J., Elger, C., et al. (2014). ILAE official report: a practical clinical definition of epilepsy. Epilepsia 55, 475–482. doi: 10.1111/epi.12550
Gallagher, A., Flatt, P., Duffy, G., and Abdel-Wahab, Y. (2013). The effects of traditional antidiabetic plants on in vitro glucose diffusion. Nutr. Res. 2003, 413–424. doi: 10.1016/s0271-5317(02)00533-x
Ghayour, M., Behnam-Rassouli, M., Ghayour, N., Tehranipour, M., and Kamyabi-Abkooh, A. (2012). Investigating the anti-epileptic and sedative effects of hydroalcoholic extract of melissa officinalis (Lemon Balm) leaf on pentylenetetrazol induced epileptiform seizures in wistar rat. J. Med. Plants 11, 64–73.
Gilmour, J., Harrison, C., Asadi, L., Cohen, M. H., Aung, S., and Vohra, S. (2011). Considering complementary and alternative medicine alternatives in cases of life-threatening illness: applying the best-interests test. Pediatrics 128, 175–180. doi: 10.1542/peds.2010-2720F
Gollapalle, L. V., Marikunte, V. V., Nunna, B. L. P., and Godavarthi, A. (2021). Evaluation of Anti-epileptic activity of Leaf extracts of Punica granatum on experimental models of epilepsy in mice. J. Intercult. Ethnopharmacol. 5, 415–421. doi: 10.5455/jice.20160904102857
Goodarzi, M., Khodadadi, I., Tavilani, H., and Abbasi, O. E. (2016). The role of anethum graveolens L. (Dill) in the management of diabetes. J. Trop. Med. 2016:1098916. doi: 10.1155/2016/1098916
Grace, M., Ribnicky, D., Kuhn, P., Poulev, A., Logendra, S., Yousef, G., et al. (2009). Hypoglycemic activity of a novel anthocyanin-rich formulation from lowbush blueberry, Vaccinium angustifolium Aiton. Phytomedicine 16, 406–415. doi: 10.1016/j.phymed.2009.02.018
Guo, X., and Mei, N. (2016). Aloe vera: A review of toxicity and adverse clinical effects. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 34, 77–96. doi: 10.1080/10590501.2016.1166826
Gupta, R., Chang, D., Nammi, S., Bensoussan, A., Bilinski, K., and Roufogalis, B. (2017). Interactions between antidiabetic drugs and herbs: an overview of mechanisms of action and clinical implications. Diabetol. Metab. Syndr. 9:59. doi: 10.1186/s13098-017-0254-9
Hajimonfarednejad, M., Ostovar, M., Raee, M., Hashempur, M., Mayer, J., and Heydari, M. (2019). Cinnamon: a systematic review of adverse events. Clin. Nutr. 38, 594–602. doi: 10.1016/j.clnu.2018.03.013
Harris, P., Cooper, K., Relton, C., and Thomas, K. (2012). Prevalence of complementary and alternative medicine (CAM) use by the general population: a systematic review and update. Int. J. Clin. Pract. 66, 924–939. doi: 10.1111/j.1742-1241.2012.02945.x
Henderson, L., Yue, Q., Bergquist, C., Gerden, B., and Arlett, P. (2002). St John’s wort (Hypericum perforatum): drug interactions and clinical outcomes. Br. J. Clin. Pharmacol. 54, 349–356. doi: 10.1046/j.1365-2125.2002.01683.x
Holcomb, S. S. (2021). Common herb-drug interactions: what you should know: the nurse practitioner. Nurse Pract. 34, 21–29. doi: 10.1097/01.NPR.0000350567.61353.2b
Hosseinzadeh, H., Karimi, G., and Rakhshanizadeh, M. (2005). Anticonvulsant effect of Hypericum perforatum: role of nitric oxide. J. Ethnopharmacol. 98, 207–208. doi: 10.1016/j.jep.2004.12.007
Kavalali, G., Tuncel, H., Göksel, S., and Hatemi, H. (2003). Hypoglycemic activity of urtica pilulifera in streptozotocin-diabetic rats. J. Ethnopharmacol. 84, 241–245. doi: 10.1016/s0378-8741(02)00315-x
Keifer, D., Ulbricht, C., Abrams, T. R., Basch, E., Giese, N., Giles, M., et al. (2008). Peppermint (Mentha Xpiperita) an evidence-based systematic review by the natural standard research collaboration. J. Herb. Pharmacother. 7, 91–143. doi: 10.1300/j157v07n02_07
Kikuchi, H., Kogure, S., Arai, R., Saino, K., Ohkubo, A., Tsuda, T., et al. (2017). Rosehip inhibits xanthine oxidase activity and reduces serum urate levels in a mouse model of hyperuricemia. Biomed. Rep. 6, 539–544. doi: 10.3892/br.2017.888
Kramlich, D. (2014). Introduction to complementary, alternative, and traditional therapies. Crit. Care Nurse 34, 50–6;quiz57. doi: 10.4037/ccn2014807
Kyrou, I., Christou, A., Panagiotakos, D., Stefanaki, C., Skenderi, K., Katsana, K., et al. (2017). Effects of a hops (Humulus lupulus L.) dry extract supplement on self-reported depression, anxiety and stress levels in apparently healthy young adults: a randomized, placebo-controlled, double-blind, crossover pilot study. Hormones 16, 171–180. doi: 10.14310/horm.2002.1738
Lee, K.-M., Jung, J.-S., Song, D.-K., and Kim, Y.-H. (1993). Neuropharmacological activity of Humulus lupulus extracts. Korean J. Pharmacogn. 24, 231–234.
Li, J., Luo, J., Chai, Y., Guo, Y., Tianzhi, Y., and Bao, Y. (2021). Hypoglycemic effect of Taraxacum officinale root extract and its synergism with Radix Astragali extract. Food Sci. Nutr. 9, 2075–2085. doi: 10.1002/fsn3.2176
Loshali, A., Joshi, B. C., Sundriyal, A., and Uniyal, S. (2021). Antiepileptic effects of antioxidant potent extract from Urtica dioica Linn. root on pentylenetetrazole and maximal electroshock induced seizure models. Heliyon 7:e06195. doi: 10.1016/j.heliyon.2021.e06195
Lu, Z., Jia, Q., Wang, R., Wu, X., Wu, Y., Huang, C., et al. (2011). Hypoglycemic activities of A- and B-type procyanidin oligomer-rich extracts from different Cinnamon barks. Phytomedicine 18, 298–302. doi: 10.1016/j.phymed.2010.08.008
Marles, R., and Farnsworth, N. (1995). Antidiabetic plants and their active constituents. Phytomedicine 2, 137–189. doi: 10.1016/S0944-7113(11)80059-0
Mascolo, N., Capasso, R., and Capasso, F. (1998). Senna. A safe and effective drug–Mascolo–1998–phytotherapy research–wiley online library. Phytother. Res. 12, S143–S145.
Mehmood, Z., Khan, M. S., Qais, F. A., and Ahmad, I. (2019). “Chapter 18–herb and modern drug interactions: efficacy, quality, and safety aspects,” in New Look to Phytomedicine, eds M. S. Ahmad Khan, I. Ahmad, and D. Chattopadhyay (Cambridge, MA: Academic Press), 503–520.
Mohammadi, A., Mansoori, B., Aghapour, M., Shirjang, S., Nami, S., and Baradaran, B. (2016). The Urtica dioica extract enhances sensitivity of paclitaxel drug to MDA-MB-468 breast cancer cells. Biomed. Pharmacother. 83, 835–842. doi: 10.1016/j.biopha.2016.07.056
Murakami, N., Murakami, T., Kadoya, M., Matsuda, H., Yamahara, J., and Yoshikawa, M. (1996). New hypoglycemic constituents in “gymnemic acid” from Gymnema sylvestre. Chem. Pharm. Bull. 44, 469–471. doi: 10.1248/cpb.44.469
Nahin, R., Dahlhamer, J., Taylor, B., Barnes, P., Stussman, B., Simile, C., et al. (2007). Health behaviors and risk factors in those who use complementary and alternative medicine. BMC Public Health 7:217. doi: 10.1186/1471-2458-7-217
Patel, S. (2017). Rose hip as an underutilized functional food: evidence-based review. Trends Food Sci. Technol. 63, 29–38. doi: 10.1016/j.tifs.2017.03.001
Pavel, D., Marcel, K., and Lukáš, J. (2017). Hop phytochemicals and their potential role in metabolic syndrome prevention and therapy. Molecules 22:1761. doi: 10.3390/molecules22101761
Perović, J., Tumbas, ŠV., Kojić, J., Krulj, J., Moreno, D., García-Viguera, C., et al. (2021). Chicory (Cichorium intybus L.) as a food ingredient–Nutritional composition, bioactivity, safety, and health claims: a review. Food Chem. 336:127676. doi: 10.1016/j.foodchem.2020.127676
Pilerood, S. A., and Prakash, J. (2013). Nutritional and medicinal properties of valerian (Valeriana Officinalis) herb: a review. Int. J. Food Nutr. Diet. 1, 25–32.
Posadzki, P., Watson, L., and Ernst, E. (2013). Herb-drug interactions: an overview of systematic reviews. Br. J. Clin. Pharmacol. 75, 603–618. doi: 10.1111/j.1365-2125.2012.04350.x
Prokop, J., Lnĕničková, K., Cibiček, N., Kosina, P., Tománková, V., Jourová, L., et al. (2019). Effect of bilberry extract (Vaccinium myrtillus L.) on drug-metabolizing enzymes in rats. Food Chem. Toxicol. 129, 382–390.
Raja, P., Thejaswini, J., Gurupadayya, B., Mruthyunjaya, K., and Latha, S. C. (2013). Evaluation of influence of gymnema tea on antidiabetic activity of metformin in diabetic rats. Indo. Am. J. Pharm. Res. 3:12.
Rasmussen, M., Zamaratskaia, G., and Ekstrand, B. (2011). In vivo effect of dried chicory root (Cichorium intybus L.) on xenobiotica metabolising cytochrome P450 enzymes in porcine liver. Toxicol. Lett. 200, 88–91. doi: 10.1016/j.toxlet.2010.10.018
Rathor, N., Arora, T., Manocha, S., Patil, A., Mediratta, P., and Sharma, K. (2014). Anticonvulsant activity of Aloe vera leaf extract in acute and chronic models of epilepsy in mice. J. Pharm. Pharmacol. 66, 477–485. doi: 10.1111/jphp.12181
Samuels, N., Finkelstein, Y., Singer, S., and Oberbaum, M. (2008). Herbal medicine and epilepsy: proconvulsive effects and interactions with antiepileptic drugs. Epilepsia 49, 373–380. doi: 10.1111/j.1528-1167.2007.01379.x
Setty, A., and Sigal, L. (2005). Herbal medications commonly used in the practice of rheumatology: mechanisms of action, efficacy, and side effects. Semin. Arthritis Rheum. 34, 773–784. doi: 10.1016/j.semarthrit.2005.01.011
Sivertsen, K., Lukic, M., and Kristoffersen, A. (2018). Gender specific association between the use of complementary and alternative medicine (CAM) and alcohol consumption and injuries caused by drinking in the sixth Tromsø study. BMC Complement. Altern. Med. 18:239. doi: 10.1186/s12906-018-2301-y
Skalicka-Woźniak, K., Walasek, M., Aljarba, T., Stapleton, P., Gibbons, S., Xiao, J., et al. (2018). The anticonvulsant and anti-plasmid conjugation potential of Thymus vulgaris chemistry: an in vivo murine and in vitro study. Food Chem. Toxicol. 120, 472–478. doi: 10.1016/j.fct.2018.07.045
Spirling, L. I., and Daniels, I. R. (2001). Botanical perspectives on health Peppermint: more than just an after-dinner mint. J. R. Soc. Promot. Health 121, 62–63. doi: 10.1177/146642400112100113
Sweeney, B., Vora, M., Ulbricht, C., and Basch, E. (2005). Evidence-based systematic review of dandelion (Taraxacum officinale) by natural standard research collaboration. J. Herb. Pharmacother. 5, 79–93. doi: 10.1300/j157v05n01_09
Talebi, F., Malchi, F., Abedi, P., and Jahanfar, S. (2020). Effect of dill (Anethum Graveolens Linn) seed on the duration of labor: a systematic review. Complement. Ther. Clin. Pract. 41:101251. doi: 10.1016/j.ctcp.2020.101251
Tochukwu, O. C., Abigail, O., Alexander, E. M., and Ugbedeojo, S. P. (2021). The phytochemical constituents, hypoglycemic, and antioxidant activities of Senna occidentalis (L.) ethanolic leaf extract in high sucrose diet fed drosophila melanogaster. J. Adv. Biol. Biotechnol. 24, 48–63. doi: 10.9734/jabb/2021/v24i330206
Tolstikova, T. G., Pavlova, A. V., Dolgikh, M. P., Ilyina, I. V., Ardashov, O. V., Volcho, K. P., et al. (2009). (4S,5R,6R)-para-mentha-1,8-dien-5,6-diol is a new highly effective anticonvulsant agent. Dokl. Biol. Sci. 429, 494–496. doi: 10.1134/s0012496609060039
Ulbricht, C., Abrams, T., Basch, E., Davies-Heerema, T., Foppa, I., Hammerness, P., et al. (2011). An evidence-based systematic review of gymnema (Gymnema sylvestre R. Br.) by the natural standard research collaboration. J. Diet. Suppl. 8, 311–330. doi: 10.3109/19390211.2011.597977
Ulbricht, C., Basch, E., Basch, S., Bent, S., Boon, H., Burke, D., et al. (2009). An evidence-based systematic review of bilberry (Vaccinium myrtillus) by the natural standard research collaboration. J. Diet. Suppl. 6, 162–200. doi: 10.1080/19390210902861858
Vajda, F., and Eadie, M. (2014). The clinical pharmacology of traditional antiepileptic drugs. Epileptic Disord. 16, 395–408. doi: 10.1684/epd.2014.0704
Viladomiu, M., Hontecillas, R., Lu, P., and Bassaganya-Riera, J. (2013). Preventive and prophylactic mechanisms of action of pomegranate bioactive constituents. Evidence Based Complement. Alternat. Med. 2013:789764. doi: 10.1155/2013/789764
Walsh, S., Donnan, J., Fortin, Y., Sikora, L., Morrissey, A., Collins, K., et al. (2017). A systematic review of the risks factors associated with the onset and natural progression of epilepsy. Neurotoxicology 61, 64–77. doi: 10.1016/j.neuro.2016.03.011
Wang, H. X., and Ng, T. B. (1999). Natural products with hypoglycemic, hypotensive, hypocholesterolemic, antiatherosclerotic and antithrombotic activities. Life Sci. 65, 2663–2677. doi: 10.1016/s0024-3205(99)00253-2
Keywords: complementary and alternative medicines, epilepsy, diabetes mellitus, outcome, adherence
Citation: Girgis MMF, Fekete K, Homoródi N, Márton S, Fekete I and Horváth L (2022) Use of Complementary and Alternative Medicine Among Patients With Epilepsy and Diabetes Mellitus, Focusing on the Outcome of Treatment. Front. Neurosci. 15:787512. doi: 10.3389/fnins.2021.787512
Received: 30 September 2021; Accepted: 06 December 2021;
Published: 11 January 2022.
Edited by:
Zhenghao Xu, Zhejiang Chinese Medical University, ChinaReviewed by:
Carmen Rubio, Manuel Velasco Suárez Instituto Nacional de Neurología y Neurocirugía, MexicoCopyright © 2022 Girgis, Fekete, Homoródi, Márton, Fekete and Horváth. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: László Horváth, bGhvcnZhdGhAbWVkLnVuaWRlYi5odQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.