
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Neurosci., 10 November 2021
Sec. Neurodevelopment
Volume 15 - 2021 | https://doi.org/10.3389/fnins.2021.771557
This article is part of the Research TopicUnderstanding the Causal Link Between Inflammation and Neurodevelopmental DisordersView all 8 articles
A correction has been applied to this article in:
Corrigendum: Intrauterine Viral Infections: Impact of Inflammation on Fetal Neurodevelopment
Intrauterine viral infections during pregnancy by pathogens such as Zika virus, Cytomegalovirus, Rubella and Herpes Simplex virus can lead to prenatal as well as postnatal neurodevelopmental disorders. Although maternal viral infections are common during pregnancy, viruses rarely penetrate the trophoblast. When they do cross, viruses can cause adverse congenital health conditions for the fetus. In this context, maternal inflammatory responses to these neurotropic pathogens play a significant role in negatively affecting neurodevelopment. For instance, intrauterine inflammation poses an increased risk of neurodevelopmental disorders such as microcephaly, schizophrenia, autism spectrum disorder, cerebral palsy and epilepsy. Severe inflammatory responses have been linked to stillbirths, preterm births, abortions and microcephaly. In this review, we discuss the mechanistic basis of how immune system shapes the landscape of the brain and how different neurotropic viral pathogens evoke inflammatory responses. Finally, we list the consequences of neuroinflammation on fetal brain development and discuss directions for future research and intervention strategies.
Neurodevelopment is a complex developmental process that begins during the third week of gestation and continues postnatally until adulthood (Stiles and Jernigan, 2010). In the fetal brain, about 86 billion neurons must be generated in a spatiotemporally coordinated manner, with any deviations resulting in developmental defects and cognitive impairments (Azevedo et al., 2009; Herculano-Houzel, 2009; Kang et al., 2011). The formation of neuroectoderm, which gives rise to the neural tube, is the first step in brain development. Following that, forebrain, midbrain, and hindbrain are generated in a controlled and systematic manner, developing into a functional brain (Chan et al., 2017; Elshazzly and Caban, 2019). Symmetric division of neuroepithelial cells (stem cells) that line the ventricles of the neuroectoderm results in the generation of additional neural stem cells, whereas asymmetrical division gives rise to radial glia and later differentiated cells such as neurons and various glial cells (Götz and Huttner, 2005; Subramanian et al., 2017). Newly formed neurons migrate to the cortical plate and populate distinct layers, culminating in the six-layered neocortex, which accounts for the vast majority of brain volume (Nadarajah and Parnavelas, 2002; Ayala et al., 2007). Therefore, the balance between symmetric and asymmetric cell divisions is critical in determining the brain volume and cognitive capacity (Lu et al., 2000; Huttner and Kosodo, 2005; Knoblich, 2008; Lazutkin et al., 2019). After neurons complete their migration, their projections- axons and dendrites- form synapses with appropriate synaptic partners. The earliest synaptic connections thus formed in the preplate, as early as week 5, aid in the establishment of neuronal connections from thalamus and brainstem. These early synapses contribute to the formation of more stable connections later in development (Tau and Peterson, 2010). Genetic defects, environmental perturbations or pathogens can promote mitotic defects in progenitor cells or apoptosis resulting in hypocellularity leading to disruption of the stem cell balance, which often manifests as neurodevelopmental disorders such as microcephaly and lissencephaly (Chavali et al., 2014; Heffernan and Hare, 2018). In contrast to these catastrophic failures of cell death and fate specification, changes in the neural circuit formation and functions can result in other neurodevelopmental disorders such as autism spectrum disorders (ASD) and schizophrenia.
Proliferation and programmed cell death are both vital in sculpting the landscape of a developing brain. Programmed cell death is required for (i) neural tube closure and neuroepithelial modeling (Yamaguchi et al., 2011) and (ii) to reduce replication errors by terminating cells with unfavorable genomic changes (Rehen et al., 2001; Peterson et al., 2012; Bushman and Chun, 2013). Furthermore, programmed cell death also regulates neuronal identity and patterning by eradicating population of neurons with improper projections, which might affect axonal targeting (Clarke et al., 1998; Yamaguchi and Miura, 2015). Failure to do so and eliminate surplus neurons can result in detrimental neurodevelopmental consequences (Kanold, 2009). For instance, individuals with ASD have an enlarged brain phenotype (macrocephaly) in their early years, characterized by an aberrant expansion of neocortical excitatory neurons, skewing the balance between excitatory and inhibitory circuits (Courchesne et al., 2011; Fang et al., 2014; Wei et al., 2014). On the other hand, uncontrolled apoptosis of progenitors can cause microcephaly or schizophrenia, which is characterized by localized non-lethal apoptosis, that can cause neurite and synaptic loss (Oppenheim, 1991; Chambers et al., 2004; Jarskog et al., 2005).
In this regard, the role of immune system in brain development is paramount (Zengeler and Lukens, 2021). In the sections that follow, we provide insights into how the proper functioning of the immune system is critical for brain development, and how prolonged inflammation caused by the maternal immune system in response to viral infections might jeopardize fetal neurodevelopment.
The central nervous system (CNS) and the immune system are complex organ systems which are intricately linked. Indeed, there is an evolutionary correlation in the emergence of acquired immunity and the highly developed myelin sheath in neurons (Stassart et al., 2018). The immune system of the brain comprises physical barriers such as blood-brain barrier (BBB), blood-CSF barrier (perivascular BCSFB) and an innate immune system composed of specialized non-neuronal cells known as microglia (Fenstermacher, 1980; Ballabh et al., 2004; Daneman and Prat, 2015) (Figure 1). Furthermore, in response to environmental insults, immune cells from the periphery can infiltrate via the lymphatic drainage portal (glial-lymphatic pathway) (Iliff et al., 2012). The presence of various physical barriers such as BBB and BCSFB indicates an evolutionarily conserved strategy for brain protection. The BBB forms as early as gestational week 8, beginning with telencephalon vascularization and progressing through coordinated cell-cell communications between CNS and the neurovascular unit during the subsequent stages (Saunders et al., 2012). The adult BBB is made up of endothelial cells, pericytes, astrocytes, microglia, and neurons. The embryonic BBB, on the other hand, lacks neurons and astrocytes but is nonetheless functional (Pardridge, 2005; Abbott et al., 2010; Daneman and Prat, 2015). The influx transporters of the BBB transport glucose, minerals, vitamins, hormones and other essential substances, through diffusion, receptor mediated transcytosis or solute carrier transporters to meet the nutrient needs of the developing brain (Roberts et al., 2008). Thus, infections or other neurotoxins breaking the BBB during pregnancy would have a detrimental impact on fetal neurodevelopment. The principal purpose of BCSFB, which is made up of cuboidal epithelium from the choroid plexus, is to secrete cerebrospinal fluid (CSF) into the brain ventricles, where it bathes neurogenic niches in nutrients (Saunders et al., 2012; Liddelow, 2015).
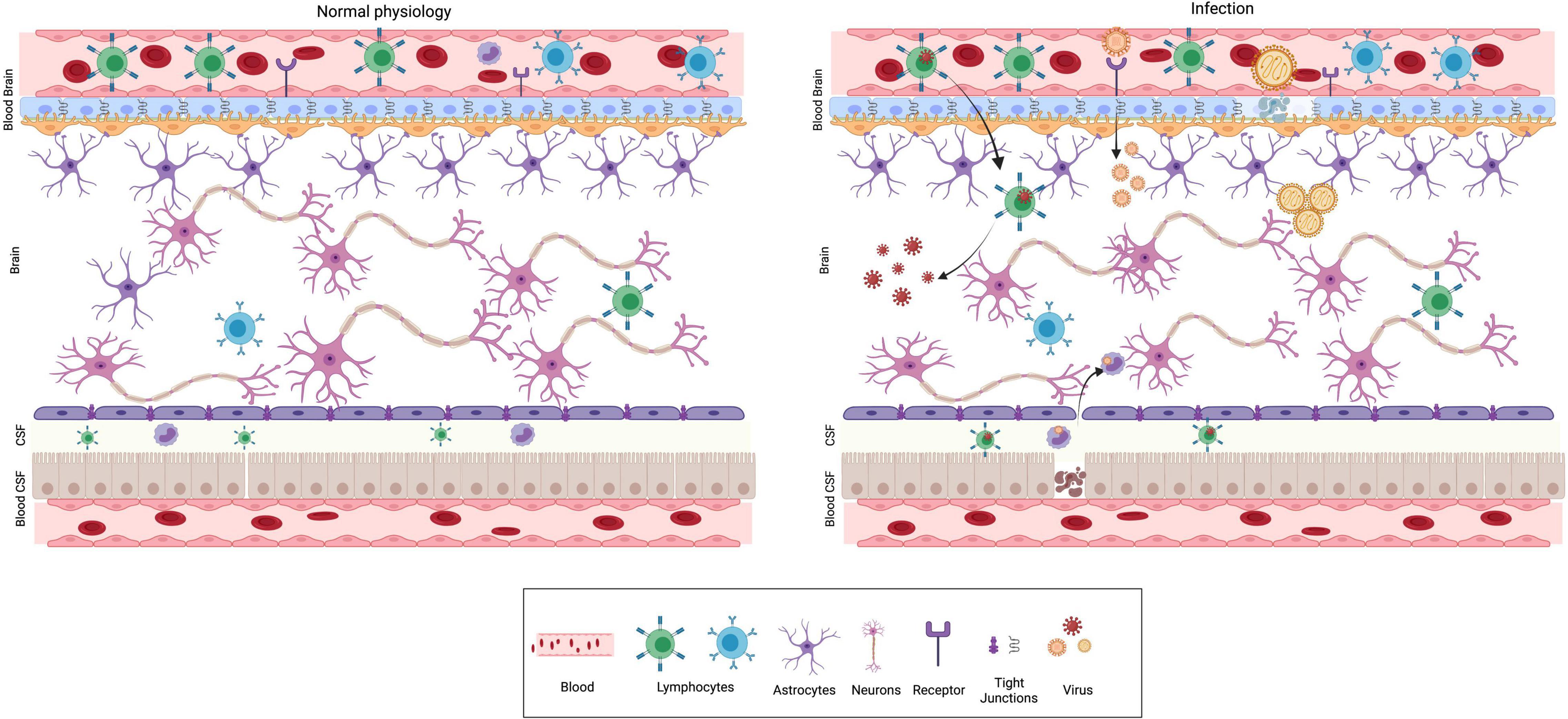
Figure 1. Physical barriers of the brain in normal physiology and infection. The major physical barriers to gain entry into the brain are: Blood Brain Barrier (BBB) and Blood CSF (BCSF) Barrier. BBB is composed of endothelial cells, pericytes and glial cells. The endothelial cells are held together by tight junctions, which prevent permeability and acts as a barrier. The BCSFB is composed of epithelial cells of the choroid plexus and ependymal cells. Under normal physiological conditions (left panel), pathogens, toxins, several immune modulatory molecules are barred from entering the brain by the presence of these barriers. Neurotropic viruses can breach the BBB using different mechanisms as depicted from left to right. (i) Trojan horse mechanism—virus infects peripheral immune cells, followed by the infected cell crossing BBB ultimately resulting in CNS infection. (ii) Receptor mediated transcytosis—several viruses can directly bind to receptors and cross the BBB by transcytosis. (iii) Some viruses are capable of dissolving tight junctions and compromise BBB’s protective function.
In addition to these barriers, the innate immune cells of brain play a significant role in neurodevelopment. The various roles of the brain’s major innate immune cells, namely microglia, in neurodevelopment are well established. Microglia are brain tissue resident macrophages that arise from hematopoietic progenitors and populate the developing brain before BBB formation (Nayak et al., 2014; Colonna and Butovsky, 2017). Microglia provide tropic support to the other major cells of brain such as astrocytes and neurons and facilitate response to signals received from the surrounding neural environment (Nayak et al., 2014; Prinz et al., 2019) (Figure 2). For example, microglia maintain the homeostasis of a healthy brain by synaptic pruning, by constant monitoring of synaptic function which allows for maturation or elimination based on special complement and/or chemokine receptors found exclusively in microglia (Harrison et al., 1998; Paolicelli et al., 2011). Upon activation of the complement cascade, the effector complement C3 tags the appropriate synapse. Microglia express specific C3 receptors (C3R) which bind to the C3 fragment and phagocytose the synapse, resulting in pruning (Figure 2) (Stevens et al., 2007; Magdalon et al., 2020). Furthermore, microglia promotes (i) angiogenesis and vascularization by clearing excess vessels (Dudiki et al., 2020), (ii) proliferation and migration of neurons and glia (Aarum et al., 2003), (iii) programmed cell death of neural stem cells and neurons (Sierra et al., 2010), (iv) myelination (Traiffort et al., 2020), (v) establishment of neuronal circuits (Miyamoto et al., 2016), and (vi) the abundance of neural stem cells (Cunningham et al., 2013).
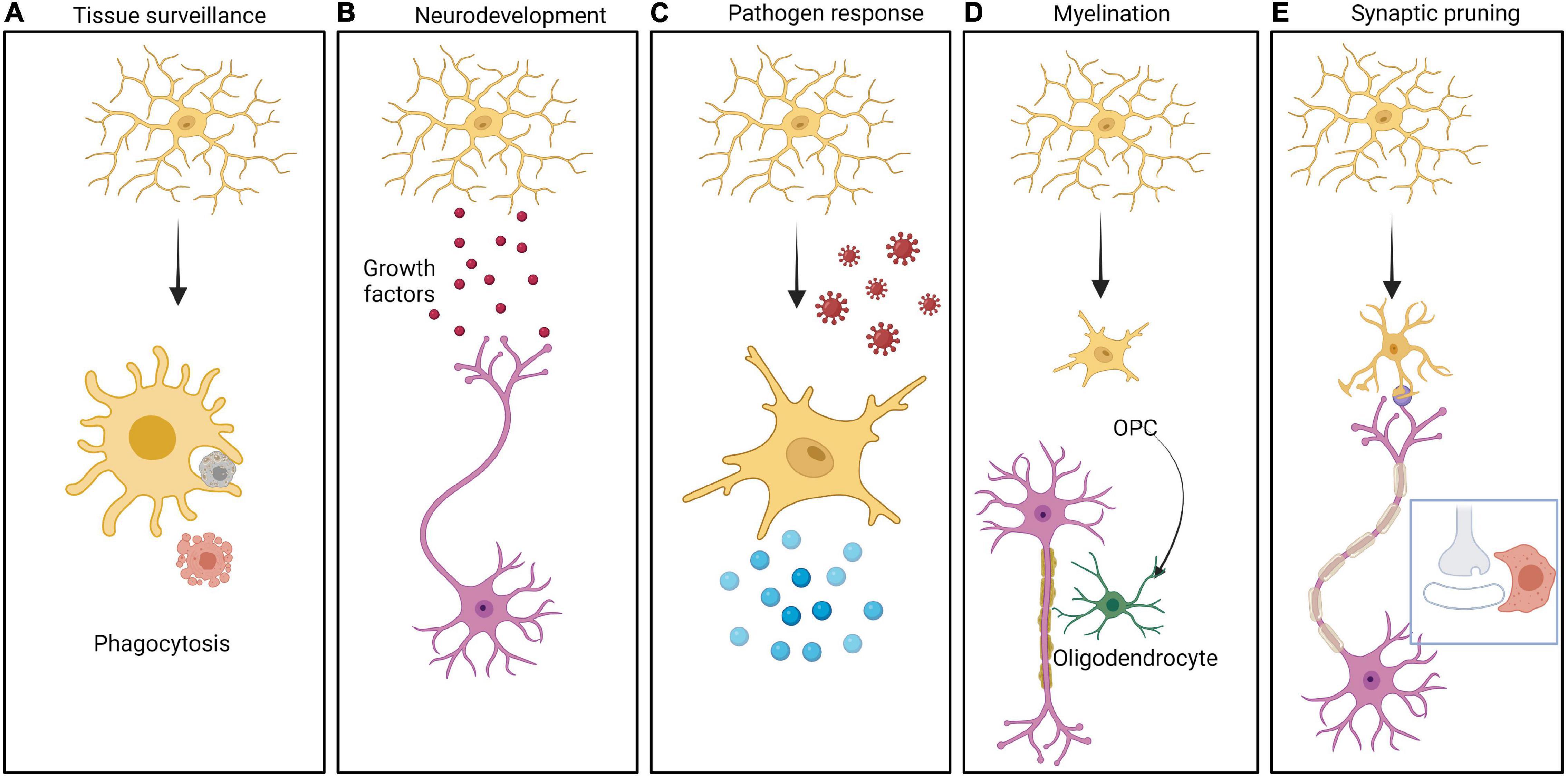
Figure 2. Different roles of Microglia in brain function. The CNS resident innate immune cells, namely microglia can be resting or activated in response to different stimuli. Multiple roles of microglia are depicted in the panels from left to right as follows: (A) Cells undergoing apoptosis in the CNS are cleared by microglia by phagocytosis. (B) Microglia secrete a large variety of growth factors including brain derived neurotrophic factor (BDNF), insulin like growth factor 1 (IGF-1), arginase-1 (Arg-1), nerve growth factor (NGF) etc. These molecules help in neurodevelopment as well as in CNS homeostasis. (C) Microglia serves as a defense against pathogens, by getting activated followed by secretion of pro-inflammatory cytokines generating an inflammatory response. (D) Microglia play an important role in myelinating and demyelinating neurons mediated by oligodendrocytes. (E) Microglia takes active part in synaptic pruning by engulfment of synapses in a complement/chemokine mediated manner.
Primary adaptive immune cells (T and B cells) are less common in the brain parenchyma than microglia and are mostly found in the choroid plexus and meninges. T cells, despite their small number, play an important role in spatial learning, memory, and stress response (Kipnis, 2016; Korn and Kallies, 2017). Interestingly, Morris-water-maze test on mice showed that a distinct population of antigen positive CD4+ T cells specifically at the meninges promote learning and memory (Radjavi et al., 2014). While B cells might not be required for these functions, they do accumulate in the neonatal brain and decline with age (Tanabe and Yamashita, 2018). B cells (B1a) promote the proliferation of Oligodendrocyte precursor cell via IgM-Fcα-μR signaling and contribute to oligodendrogenesis (Tanabe and Yamashita, 2018). Thus immune cells not only defend against invading microbes and fight infection, but they also contribute to the brain development through the secretion of unique molecules known as cytokines.
What is the source of cytokines in the fetal brain? Basal cytokine production begins in the fetal brain as early as the fifth week of gestation and is critical for normal neurodevelopment (Mousa et al., 1999; Deverman and Patterson, 2009). These cytokines are primarily secreted by microglia and astrocytes, while the role of neurons in inflammatory response is becoming more evident (Freidin et al., 1992; Hanisch, 2002; Galic et al., 2012; Stolp, 2013). Additionally, maternal cytokines can enter the fetal brain from periphery and/or via the placenta. Maternal cytokines can enter the brain by (i) passive diffusion bypassing the BBB where they bind to endothelial cells and induce secondary messenger signaling, (ii) endocytosis or (iii) by direct secretion at the nerve terminals (Watkins et al., 1995). Based on their role during immune response, the secreted cytokines can be broadly classified as pro- or anti-inflammatory cytokines. The major anti-inflammatory cytokines viz., IL4, IL10, IL1RA, IL11 and IL13, belong to the Interleukin (IL) family. Among the most important pro-inflammatory cytokines are the tumor necrosis factor α (TNFα), interferon γ (IFNγ), IL1 and IL18. However, cytokines can be pro- or anti-inflammatory depending on the context, as exemplified by IL6 and transforming growth factor beta (TGFβ) (Sanjabi et al., 2009; Tanaka et al., 2014). TGFβ is an important mediator of oligodendrocyte differentiation and polarizes microglia to sites of injury near pericytes (O’Keefe et al., 1999). The role of pro-inflammatory cytokines in normal neurodevelopment has come to the forefront particularly with the revelation that enhancement in the levels TNFα and IL1β coincide with major neurodevelopmental events and their roles in neuronal connectivity (Garay and McAllister, 2010; Ratnayake et al., 2013). Additionally, IFNγ in combination with TNFβ and IL1 induces the expression of the major histocompatibility complex 1 (MHC1), which is a glycoprotein expressed by all nucleated cells in the body (Raval et al., 1998). MHCs are broadly classified into two classes: Class I (ubiquitously expressed) and Class II (Antigen presenting cells). Activated MHC1 negatively regulates axon outgrowth, dendritic branching and synaptic density particularly in the hippocampal region of the brain, which is required for learning and memory (Cebrián et al., 2014). As seen with schizophrenia, any condition that triggers maternal MHC1 signaling has the potential to downregulate neuronal synapses and connectivity (Gaser et al., 2004).
Accumulating evidence suggests that a special class of cytokines called chemokines, initially thought only to attract immune cells to the site of inflammation, play a variety of roles in CNS development and in adult brain functioning (Miller et al., 2008; Trettel et al., 2020). For example, chemokine CXCR4 which evolved prior to the emergence of immune system has an important role in neural stem cell migration during injuries. CXCL12-CXCR4 influence the migration of neuronal precursors, axon guidance/pathfinding and maintenance of neural progenitor cells (NPCs) (Dziembowska et al., 2005; Li and Ransohoff, 2008). Chemokine signaling via CX3CL1 and CX3CR1 is neuroprotective and mediates communication between neurons and microglia, thus affecting synaptic pruning (Biber et al., 2007; Pawelec et al., 2020).
The balance between different cytokines in immune system also helps to preserve the barrier integrity. Interferons (Type1), such as IFNβ and IFNα that signal through IFNαR, are produced in response to pathogen-associated molecular patterns (PAMPs)(Zanin et al., 2021). These IFNs promote tight junction formation and preserve its integrity via the cytoskeletal GTPase Rac1, that promotes endothelial barrier function (Al-Sadi et al., 2009; Meunier and Broz, 2016). In contrast, IFNγ–CXCL10 signaling enhances BBB permeability due to internalization and downregulation of tight junction proteins (Daniels and Klein, 2015). Therefore, external stimuli such as viral infections that elicit different maternal cytokine secretion could have a long-standing consequence on the fetal neurodevelopment.
How do different viruses trigger immune response and reprogram neurodevelopment? The link between maternal infection status and fetal brain development garnered attention in late 1960s and early 1970s, when an increased infant mortality and/or high incidences of neurosensory deficiencies in newborns during the rubella epidemic was reported (Stern et al., 1969; Chess, 1971; Hutton, 2016). Children born during the rubella epidemic had delayed development and cognitive impairment. Similarly, maternal influenza infection has been linked to an increased risk of schizophrenia in adult life (Mednick et al., 1988; Kendell and Kemp, 1989). Subsequently, a number of clinical epidemiological studies revealed that a plethora of viral infections in pregnant mothers can result in neurological abnormalities ranging from encephalitis in the developing fetus to neuroprogressive and neurodegenerative diseases postnatally (Scola and Duong, 2017; Table 1).
Herpes Simplex Virus (HSV-1) is one of the most common DNA viruses that affects almost a quarter of pregnant women worldwide during different stages of pregnancy. HSV-1 has been recognized as one of the significant causes of neurodevelopmental disabilities in children who are exposed prenatally (Straface et al., 2012). Primary infection of HSV-1 is usually asymptomatic and the risk of neonatal infection rises as the pregnancy progresses. On the other hand, HSV-2 infection is relatively uncommon but has been implicated in ASD (Straface et al., 2012). The other large DNA virus also belonging to the Herpesviridae, namely Cytomegalovirus (CMV) is a major public health issue leading to lifelong latent infection (Cheeran et al., 2009; Jackson et al., 2011). The vertical transmission risk of CMV is 30–40% of which infection during the first trimester of pregnancy is a prognostic for sensorineural loss and neuropsychological disorders such as schizophrenia in infants (Adler et al., 2007). The Human Immunodeficiency Virus (HIV) is also known to cause neurodevelopmental and neuropsychiatric disorders (Ghafouri et al., 2006; Van Rie et al., 2008).
RNA viruses, primarily of the Flaviviridae family are known to be neurotropic pathogens (Pierson and Diamond, 2020). The most recent outbreak was caused by Zika virus (ZKV), which was mildly febrile in adults but induced severe developmental defects like microcephaly in progeny (Krauer et al., 2017). Other related flaviviruses such as West Nile Virus (WNV), Japanese Encephalitis Virus (JEV) and Dengue virus (DENV) have much lower rates of vertical transmission, but are neurotropic (Li et al., 2017). Once they cross the placental barrier, viruses can breach fetal brain barriers through diverse mechanisms (Ayala-Nunez and Gaudin, 2020) (Figure 1). Polio and measles viruses (Oglesbee and Niewiesk, 2011; Ohka et al., 2012) can directly infect and lyse the endothelial and epithelial cells of the BBB and enter the brain parenchyma directly. The most common mechanism appears to be the downregulation of different tight junction proteins such as occludin and claudin that constitute the barrier. Neurotropic viruses such as WNV, HSV1 and ZKV, affect the barrier integrity by the downregulation of tight junction proteins, mediated by secretory cytokines such as TNFα (Verma et al., 2009; Chiu et al., 2020; He et al., 2020). JEV and DENV can damage the BBB by downregulating tight junction proteins by disrupting the endothelial glycocalyx (Puerta-Guardo et al., 2016). Cell free viruses such as HIV and ZKV enter the BBB via transcytotic or paracellular pathways, without compromising the membrane integrity (Agrawal et al., 2013; Calderón-Peláez et al., 2019). Another prevalent mode involves trojan horse like mechanism, where the virus infects lymphocytes or monocytes in the peripheral parts of the body that can migrate into the brain by paracellular or transcellular pathway to cross the BBB (Kim et al., 2003). For instance, HIV1 infected CD4+T cells, HTLV1 infected CD8+T cells and ZKV infected monocytes can all infiltrate the brain parenchyma. In the case of HSV1, the virions can also directly enter CNS via anterograde transport to reach the axonal shaft and tip in the neuronal periphery, where they are released (Kim et al., 2003; Ayala-Nunez et al., 2019). The more recent outbreak of SARS CoV2, though rarely transmitted to the fetus, can break the BSCF barrier by infecting epithelial cells expressing ACE2 receptor (Pellegrini et al., 2020). Compared to the BBB, the tight junctions in BCSFB are more prone to microbial penetration due to the nature of epithelial cells (Redzic, 2011; Dando et al., 2014). The “leaky barriers” of the brain not only allow the invasion of viruses, but also are amenable for invasion by the peripheral immune components and secretory proteins, which can lead to an altered redox state in the brain. The dysfunctional BBB has been implicated in a number of neurodevelopmental disorders such as seizures, epilepsy, and schizophrenia (Kealy et al., 2020). Viruses such as HSV1 and CMV which are latent upon entry in fetuses, can reactivate postnatally and compromise the permeability of BBB by activating microglia and mounting inflammatory response, resulting in massive influx of other signaling molecules and peripheral immune components into the brain. This initiates a string of pernicious events ultimately leading to decline of brain functions such as cognition and memory. Thus, it would be interesting to compare and contrast different viral neuroinflammatory signatures caused by minor changes in barrier composition vs. total barrier breakdown. Identifying mechanisms as to how the BBB can repair itself will also have implications in designing effective intervention strategies.
How do distinct pathogens manifest different pathological outcomes in terms of neurological disorders? Small motifs called Pathogen associated molecular patterns (PAMPs) unique to each pathogen is sensed by pattern receptors (PRR) such as Toll like receptor (TLRs), which trigger different inflammatory reactions. These in turn translate into different developmental abnormalities (Figure 3).
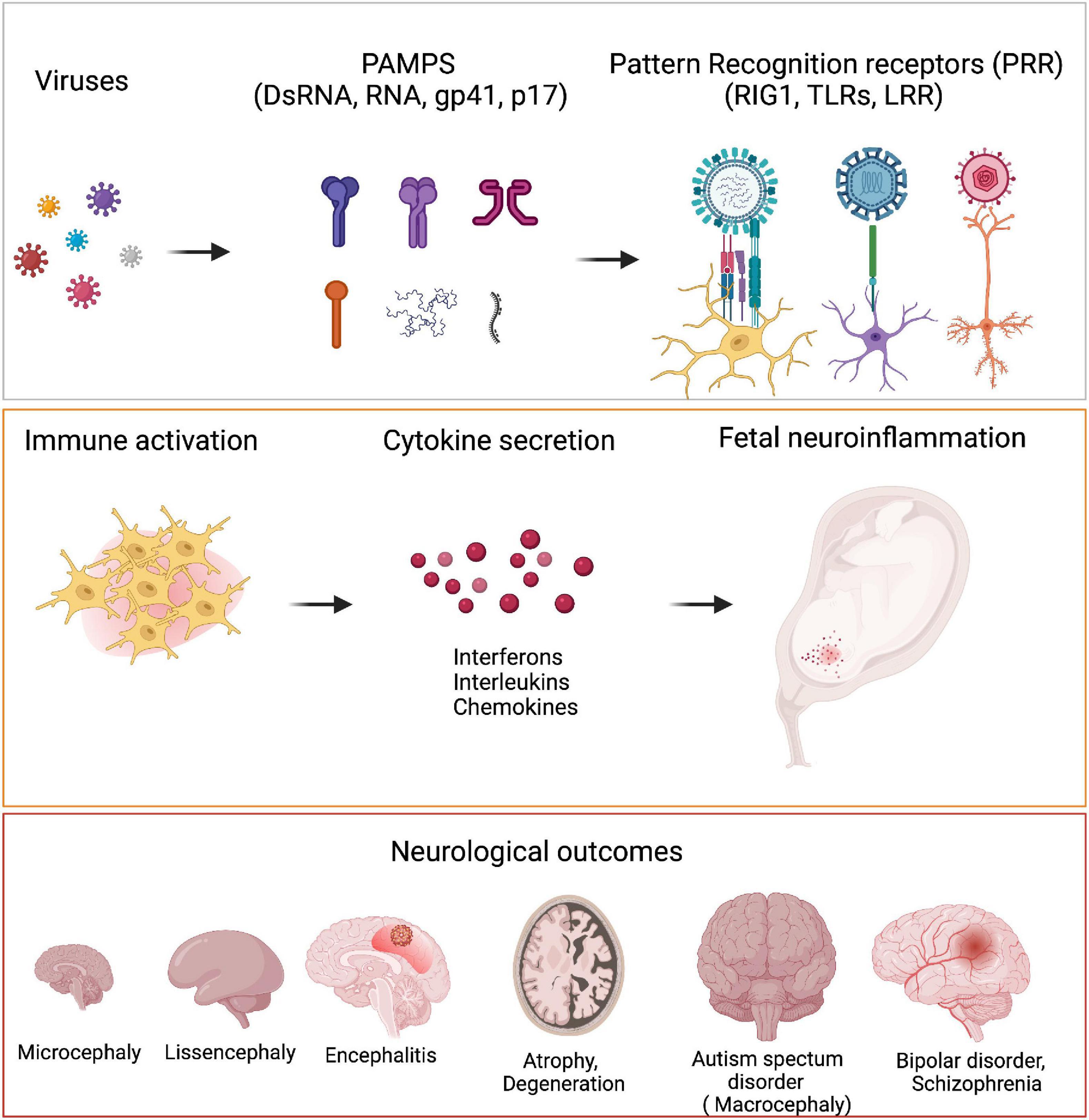
Figure 3. Viruses and neurological outcomes. Viruses have different Pathogen associated molecular pattern (PAMPs) which are sensed by the host cells through their Pattern Recognition Receptors (PRRs). Binding of PAMPs to PRRs leads to host cells secreting different cytokines like IFNα, IL6, IL4, etc., to generate an inflammatory response. PRRs are present on microglia, neurons and astrocytes. During neurodevelopment, these cells secrete several cytokines when they are triggered by viral PAMPs, that bring about detrimental consequences in terms of neurodevelopment. Fetal exposure to pro-inflammatory cytokines in high levels can manifest as neurodevelopmental disorders like microcephaly, lissencephaly, encephalitis, ASD as well as several neuropsychiatric disorders like bipolar disorder and schizophrenia.
When there is a productive infection, the maternal immune system is activated, resulting in systemic inflammation, mediated by cytokine release. While the inflammatory cytokines can cross the placental and BBB barriers and are thought to be protective, they can also have detrimental effects (Banks et al., 1995). Proinflammatory cytokine signatures are frequently linked to a variety of neurological disorders including microcephaly, cerebral palsy, schizophrenia and ASD (Ratnayake et al., 2013). Direct injection of pro-inflammatory cytokines such as IL1β and TNFα into the brains of mice, for example, resulted in social withdrawal, altered cognition and diurnal rhythm. In contrast, Insulin Growth factor 1 or IL10 injections attenuated the sickness behavior (Bluthé et al., 2000) alluding to the role of balance in pro- and anti-inflammatory cytokines in orchestration of behavior (Munshi et al., 2019). Such association of cytokines with sickness behavior has also been observed in humans, especially in individuals diagnosed with depression, schizophrenia and autism. Individuals with these neurological conditions exhibit elevated serum anti-inflammatory cytokines such as IL6, IL2R, IFN, IL13, TGF, and IL1 (Knuesel et al., 2014; Xu et al., 2015; Jiang et al., 2018). IL6, the most commonly secreted cytokine during infection, has the potential to alter the cognitive behavior of the progeny to an extent that there is a negative correlation between maternal IL6 and the memory of young children (Smith et al., 2007; Wu et al., 2015; Rudolph et al., 2018). Similarly, elevated levels of CXCL8 (IL8), and TNFα as well as the acute phase protein C-reactive protein (CRP) in maternal serum are linked to an increased risk of schizophrenia in offspring (Buka et al., 2001; Brown et al., 2004). Notably, the interplay between pro-inflammatory cytokines such as TLR4, IL1, IL6 and TNFα released upon maternal infection during pregnancy and preterm births is well established (Pandey et al., 2017). Increased secretion of cytokines such as IL8, IL1β and CRP have been linked with microcephaly, ventriculomegaly and low intelligence quotient (Dammann and O’Shea, 2008; Leviton et al., 2016). Thus, the neurological consequences of many viral infections may be related to the cytokines released rather than the viruses themselves.
In fact, the timing of maternal infection is crucial to the neurodevelopmental outcomes (Solek et al., 2018). Mimicking viral infection by the use of poly I:C at gestation day (GD) 9 or GD17 (early vs. mid stage gestation) in mice showed that while GD 9 infected mice off-springs showed defective spatial exploration, GD17 infected offsprings displayed preservative behavior reflecting autism (Meyer et al., 2006). Similarly, immune challenge in early-mid gestational period leads to increased activation of TNFα and IL10 which is associated with increased postnatal apoptosis (Meyer et al., 2006). Thus immunological challenges at different times of prenatal development may have adverse but variable neurodevelopmental manifestations (Meyer et al., 2007). Therefore, the local immune response in the brain has to be stringently modulated to prevent a huge cytolytic damage, since majority of the cells in CNS (neurons) are non-renewable and terminally differentiated.
Do all viral infections elicit similar cytokine profiles? Interestingly, all viruses tend to elicit similar pro-inflammatory cytokines. However, each virus elicits different responses in different cell types based on the type of receptors (Table 1). Upon infection, maternal immune activation produces significant amounts of pro-inflammatory cytokines, many of which including IL6, TNFα, IL10 and IL1β can be detected in the fetal brain (Meyer et al., 2009). This occurs due to the response of different CNS cell types such as neurons, astrocytes and glial cells to the cytokines and infections. Most of these cells have specialized surface receptors such as TLRs (Okun et al., 2011), retinoic acid inducible gene I (RIG) like receptors (Loo and Gale, 2011), mitochondrial antiviral signaling (MAVS) (Nair and Diamond, 2016) and cytokine receptors (Perry et al., 2010). However, differences in the pathogen spread and persistence varies based on the expression levels of each of these immune receptors, expression of innate immune genes and the extent of IFN1 response (Cho et al., 2013). For instance, early cortical neurons which have lower levels of RIG1 are more permissive to WNV and ZKV while in hippocampal neurons high basal expression of type I IFN can restrict varicella virus (Cho et al., 2013; Kennedy et al., 2015). Although HSV1 and ZKV deplete neural progenitor pools and cause a similar phenotype, namely microcephaly, they engage different molecular mechanisms. HSV1 perturbs neuroepithelial polarity and is more severe, while ZKV affects neural progenitor cells without altering the polarity. Furthermore, the sensitivity of ZKV and HSV1 to IFN1 significantly varies, with HSV1 being able to neutralise IFNb unlike ZKV (Krenn et al., 2021).
The predominant host response mechanism that is triggered by several viral infections is the activation of microglia. Fetal microglia differ from adult microglia in their morphology and gene expression profiles (Ginhoux et al., 2013; Kracht et al., 2020). Maternal immune activation with poly I:C in mice revealed that the offspring had an early push toward a more mature microglial developmental state, with a number of autism susceptibility genes differentially expressed (Ozaki et al., 2020). Thus, when maternal immune activation occurs at early gestation, the changes can be sustained in microglia for a longer duration resulting in rewired neural circuits. This rewiring is linked to behavioral defects seen postnatally. While an increased number of activated microglia is essential to stave off infectious agents, a prolonged activation is detrimental leading to neurodevelopmental disorders (Czeh et al., 2011). This is not due to an increase in microglia, but because their immune response is skewed toward a pro-inflammatory state, thereby exposing the fetal and postnatal brain to neuronal loss (Y. S. Kim and Joh, 2006). Once activated, microglia can secrete complement components, the uncontrolled secretion of which could result in abnormal synaptic pruning. This is well exemplified by the fact that the injection of the mouse with poly I:C triggered sustained complement subcomponent C1q secretion in the prefrontal cortex of offspring which often coats the synapse to be eliminated (Han et al., 2017). Notably, mice defective for C1q and CX3CR1 exhibit enhanced excitatory synaptic connectivity similar to those observed in subsets of ASD patients (Chu et al., 2010; Paolicelli et al., 2011; Fagan et al., 2017). The sequestration or inactivation of the complement cascade employed by viruses as an evasion strategy could thus play an indirect role in manifestation of neurodevelopmental disorders (Stoermer and Morrison, 2011).
The source of cytokines in brain is not limited to the resident immune cells but includes immune cells that migrate to the brain guided by chemokine secretion. These peripheral immune cells additionally contribute to an increased production of cytokines in the brain. For example, CXCL10 is overexpressed during a flaviviral infections such ZKV, DENV and WNV and attracts CD-8+ T-cells as a protective mechanism (Klein et al., 2005). However, too much of CXCL10 causes an increase in intracellular calcium levels and triggers apoptosis (Sui et al., 2006). Other chemokines such as CCL4, CCL5 are also significantly upregulated while CCL2 and CXCL8 are significantly downregulated. Interestingly, latent infections can also produce these chemokines which explains the immune infiltration seen in certain cases (Melchjorsen et al., 2003). While the interaction of peripheral immune cell signaling with microglia during viral infections confers protection, it can indirectly damage CNS through synaptic stripping and neuronal death by stimulating neurons to produce CCL2, which acts as a receptor for microglia to phagocytose them (Di Liberto et al., 2018; Moseman et al., 2020).
Transcriptome profiles of host immune cells post-viral infections reveal the upregulation of interferon-stimulated gene family (ISGs) and cell type specific pro-inflammatory genes. For example, CMV-infected pericytes show an upregulation of RANTES, IL6, IL7, IL11, and cyclooxygenase 2 (COX-2) (Cheeran et al., 2001; Zhu et al., 2001). Infected astrocytes predominantly express CCL2 and show TGFβ activation while microglia produce TNFα, IL6 alongside CXCL10, CCL2, CCL3, and CCL5. In such instances, production of anti-inflammatory cytokines such as IL10 is paramount to negate the detrimental consequences of neuroinflammation. This is reflected by the association of genetic variation in IL10 gene with altered HCMV disease occurrence following allogeneic stem cell transplantation and HIV co-infection (Sezgin et al., 2010).
The common presumption that increased production and release of pro-inflammatory cytokines into the fetus can cause brain damage has now been refined. A slender shift in the excess pro- or anti-inflammatory cytokines during an infectious response is sufficient to disrupt normal brain development (Deverman and Patterson, 2009). Contrarily, a uniform change in the expression of pro and anti-inflammatory cytokines such as IL6 and IL10, do not alter post-natal abnormalities, as observed in mice (Meyer et al., 2009). Importantly, viral genomes constantly and rapidly evolve to evade host immune surveillance, resulting in viral proteins mimicking and/or degrading critical immune modulatory signaling pathways. As a case in point, during the viral lytic cycle, CMV produces a functional ortholog of IL10 (UL111A, vIL10) that can suppress a number of innate and adaptive host immune responses including pro-inflammatory cytokine secretion (Jenkins et al., 2004). In the case of ZKV, the RdRP NS5 protein binds to and degrades STAT2 which is essential for IFN1 response (Kumar et al., 2016). HSV1 on the other hand uses the Infected Cell Protein 0 to engage with the host proteasome pathway to degrade Interferon-Stimulated Gene (ISG) products (Van Sant et al., 2001). Additionally, HSV1 prevents the phosphorylation of eukaryotic initiation factor 2, required for translation, by blocking Protein kinase R and recruiting protein phosphatase 1a by the viral protein ICP34.5 (Li et al., 2011). DNA viruses, specifically Herpesviruses and Poxviruses, circumvent interferon response by making their own soluble viroceptors/virokines, which can intercept the activities of host cytokines by sequestering them (Smith and Kotwal, 2001). This is exemplified by the binding of the poxvirus protein B8R to IFNγ which attenuates the inflammatory response (Johnston and McFadden, 2003). Emulating this, IFNγ peptide mimetics have been engineered which can circumvent the binding by B8R and be used as an antiviral therapeutic (Ahmed et al., 2005).
Gender differences in the severity and prevalence of different viral infections are another confounding factor in determining the outcomes of inflammatory responses (Klein and Flanagan, 2016; Mallard et al., 2019). Studies suggest that female fetuses are more resistant to intra-uterine stress and that the male offspring’s immunological homeostasis is particularly affected by maternal immune activation (Goldenberg et al., 2006; Zager et al., 2013). This sexual dimorphism adds complexity to neurological outcomes (Nelson and Lenz, 2017). Males have been shown to be more susceptible to ASD, ADHD, schizophrenia, and bipolar disorder than females (Werling and Geschwind, 2013; Werling, 2016). Males have a faster microglial maturation pathway, which is connected with differential gene expression of immune-related genes (Hanamsagar et al., 2017). While the inherent differences between the male and the female immune systems such as elevated Type I IFN response, T cell numbers, TLR3 response could play a role (Chavez-Valdez et al., 2019), it remains unclear if the transmission risks are the same between the male and the female progeny. Furthermore, differences in immunological regulation between males and females due to glucocorticoid-stimulated cytokine release may also contribute to the sexual dichotomy of the neuroendocrine axis (Bailey et al., 2003; Silverman et al., 2005; Duma et al., 2010). A sustained increase in cytokine production can cause the pituitary to secrete adrenocorticotropic hormone (ACTH), which causes the adrenal gland to release cortisol. Cortisol levels above a certain threshold can harm the hippocampus, affecting learning and memory and promote atrophy in the HPA, hippocampus, and amygdala (Sroykham and Wongsawat, 2019).
Understanding the role of inflammation in neurodevelopmental disorders has now opened up avenues for targeting the immune system of patients with neuropsychiatric disorders such as bipolar disorder, ASD and schizophrenia. For example, the two drugs risperidone and aripiprazole now approved for treatment to improve irritability in ASD and schizophrenia have shown to possess anti-inflammatory effects (Juncal-Ruiz et al., 2018). Likewise use of anti-inflammatory drugs such as minocycline and pioglitazone are being explored to improve irritability and depressive behaviors (Rosenblat, 2019). Thus, a systematic approach to characterizing the molecular and cellular effects of these antipsychotic drugs on inflammation, as well as correlating their clinical response, will pave the way for effective combinatorial therapies, in case of neurodevelopmental disorders with immune dysregulation. Likewise, specific non-steroidal anti-inflammatory drugs (NSAID) such as naproxen have been shown to be effective against ZKV entry (Pan et al., 2018), Influenza virus (Dilly et al., 2018) and reactivation of HSV-1 at least in cellular models. However, this use needs to be exercised with caution since NSAIDs can have diverse effects in addition to their modulation of microglial activation (Ajmone-Cat et al., 2010).
Prenatal exposure to maternal immune activation has been recognized as a risk factor for adverse neurological outcomes by a number of preclinical and epidemiological studies. However, an in depth molecular understanding of how the prenatal exposure to immune activation results in offspring’s neuronal and cognitive impairments in different stages of gestation is lacking. Targeting multiple inflammatory indicators at various times during pregnancy after viral infections will be needed for the development of successful neurodevelopmental disorder therapies. For example, most of the viral infections or neurological diseases show elevated levels of TNFα. Thus intuitively neutralizing this cytokine could ameliorate or prevent the disorder. However, studies thus far have reported discordant findings in this regard. While in murine CMV model and WNV infection model, TNFα antagonist indeed rectified cerebellar abnormalities and developmental gene expression, it has been found to augment Multiple Sclerosis phenotype in humans by increasing peripheral and CNS autoimmunity (Sicotte and Voskuhl, 2001; Shrestha et al., 2008). Although acute infection can be cleared by drug treatment, the extant immune responses, even after pathogen clearance, could cause long-term psychiatric and neurocognitive issues in survivors (Klein et al., 2017). Since many neurodevelopmental disorders have intermediate phenotypes and are subjective, the adoption of multiple complementary methods such as neurochemical investigations and cytokine profiles for accurate diagnosis is necessary. In this regard, use of a powerful platform such as 3D brain organoids with an integrated neuroendocrine and neuro-immune axis should aid to unravel how different viral infections can induce diverse immune gene expression programs in a spatiotemporal manner (Krenn et al., 2021).
Despite the fact that a link between intrauterine viral infections and neurodevelopmental disorders has been established, our ability to prevent or correct such abnormalities using pharmaceutical interventions has been limited. Several studies have shown that regardless of the virus, maternal immune activation and subsequent inflammatory response may be a key determinant of neurological outcomes. Therefore, elucidating the gestation stage specific and sex specific effects of viral infection, as well as inflammatory response codes for various viruses would allow for the development of early diagnosis and intervention strategies for neurodevelopmental disorders.
PLC designed the structure and contents of the review. SG and PLC prepared the figures and wrote the manuscript. Both authors contributed to the article and approved the submitted version.
PLC is supported by Wellcome Trust Indian alliance intermediate grant (IA/I/19/1/504280) and SERB Women Excellence Award (WEA/2020/000026). SG is supported by CSIR Senior Research Fellowship.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The authors would like to thank Sreenivas Chavali, IISER Tirupati for critical inputs during manuscript preparation. Figures were created with BioRender.com.
ASD, autism spectrum disorder; BBB, Blood Brain Barrier; BCSFB, Blood CSF Barrier; CMV, Cytomegalovirus; CNS, central nervous system; CSF, cerebrospinal fluid; HSV, Herpes Simplex Virus; HIV, human immunodeficiency virus; HTLV-1, human T-lymphotropic virus 1; IFN, interferon; IL, interleukin; JEV, Japanese encephalitis virus; WNV, West Nile virus; ZKV, Zika virus.
Aarum, J., Sandberg, K., Haeberlein, S. L. B., and Persson, M. A. A. (2003). Migration and differentiation of neural precursor cells can be directed by microglia. Proc. Natl. Acad. Sci. U.S.A. 100, 15983–15988. doi: 10.1073/pnas.2237050100
Abbott, N. J., Patabendige, A. A. K., Dolman, D. E. M., Yusof, S. R., and Begley, D. J. (2010). Structure and function of the blood-brain barrier. Neurobiol. Dis. 37, 13–25. doi: 10.1016/j.nbd.2009.07.030
Adler, S. P., Nigro, G., and Pereira, L. (2007). Recent advances in the prevention and treatment of congenital cytomegalovirus infections. Semin. Perinatol. 31, 10–18. doi: 10.1053/j.semperi.2007.01.002
Agrawal, T., Sharvani, V., Nair, D., and Medigeshi, G. R. (2013). Japanese encephalitis virus disrupts cell-cell junctions and affects the epithelial permeability barrier functions. PLoS One 8:e69465. doi: 10.1371/journal.pone.0069465
Ahmed, C. M. I., Burkhart, M. A., Subramaniam, P. S., Mujtaba, M. G., and Johnson, H. M. (2005). Peptide mimetics of gamma interferon possess antiviral properties against vaccinia virus and other viruses in the presence of poxvirus B8R protein. J. Virol. 79, 5632–5639. doi: 10.1128/JVI.79.9.5632-5639.2005
Ajmone-Cat, M. A., Bernardo, A., Greco, A., and Minghetti, L. (2010). Non-steroidal anti-inflammatory drugs and brain inflammation: effects on microglial functions. Pharmaceuticals (Basel) 3, 1949–1965. doi: 10.3390/ph3061949
Al-Sadi, R., Boivin, M., and Ma, T. (2009). Mechanism of cytokine modulation of epithelial tight junction barrier. Front. Biosci. (Landmark Ed.) 14:2765–2778. doi: 10.2741/3413
Ayala, R., Shu, T., and Tsai, L. H. (2007). Trekking across the brain: the journey of neuronal migration. Cell 128, 29–43. doi: 10.1016/j.cell.2006.12.021
Ayala-Nunez, N. V., Follain, G., Delalande, F., Hirschler, A., Partiot, E., Hale, G. L., et al. (2019). Zika virus enhances monocyte adhesion and transmigration favoring viral dissemination to neural cells. Nat. Commun. 10:4430. doi: 10.1038/s41467-019-12408-x
Ayala-Nunez, N. V., and Gaudin, R. (2020). A viral journey to the brain: current considerations and future developments. PLoS Pathog. 16:e1008434. doi: 10.1371/journal.ppat.1008434
Azevedo, F. A. C., Carvalho, L. R. B., Grinberg, L. T., Farfel, J. M., Ferretti, R. E. L., Leite, R. E. P., et al. (2009). Equal numbers of neuronal and nonneuronal cells make the human brain an isometrically scaled-up primate brain. J. Comp. Neurol. 513, 532–541. doi: 10.1002/cne.21974
Bailey, M., Engler, H., Hunzeker, J., and Sheridan, J. F. (2003). The hypothalamic-pituitary-adrenal axis and viral infection. Viral Immunol. 16, 141–157. doi: 10.1089/088282403322017884
Ballabh, P., Braun, A., and Nedergaard, M. (2004). The blood-brain barrier: an overview: structure, regulation, and clinical implications. Neurobiol. Dis. 16, 1–13. doi: 10.1016/j.nbd.2003.12.016
Banks, W. A., Kastin, A. J., and Broadwell, R. D. (1995). Passage of cytokines across the blood-brain barrier. Neuroimmunomodulation 2, 241–248. doi: 10.1159/000097202
Biber, K., Neumann, H., Inoue, K., and Boddeke, H. W. G. M. (2007). Neuronal “On” and “Off” signals control microglia. Trends Neurosci. 30, 596–602. doi: 10.1016/j.tins.2007.08.007
Bluthé, R. M., Layé, S., Michaud, B., Combe, C., Dantzer, R., and Parnet, P. (2000). Role of interleukin-1beta and tumour necrosis factor-alpha in lipopolysaccharide-induced sickness behaviour: a study with interleukin-1 type I receptor-deficient mice. Eur. J. Neurosci. 12, 4447–4456.
Brown, A. S., Hooton, J., Schaefer, C. A., Zhang, H., Petkova, E., Babulas, V., et al. (2004). Elevated maternal interleukin-8 levels and risk of schizophrenia in adult offspring. Am. J. Psychiatry 161, 889–895. doi: 10.1176/appi.ajp.161.5.889
Buka, S. L., Tsuang, M. T., Torrey, E. F., Klebanoff, M. A., Wagner, R. L., and Yolken, R. H. (2001). Maternal cytokine levels during pregnancy and adult psychosis. Brain Behav. Immun. 15, 411–420. doi: 10.1006/brbi.2001.0644
Bushman, D. M., and Chun, J. (2013). The genomically mosaic brain: aneuploidy and more in neural diversity and disease. Semin. Cell Dev. Biol. 24, 357–369. doi: 10.1016/j.semcdb.2013.02.003
Calderón-Peláez, M.-A., Velandia-Romero, M. L., Bastidas-Legarda, L. Y., Beltrán, E. O., Camacho-Ortega, S. J., and Castellanos, J. E. (2019). Dengue virus infection of blood–brain barrier cells: consequences of severe disease. Front. Microbiol. 10:1435. doi: 10.3389/fmicb.2019.01435
Cebrián, C., Loike, J. D., and Sulzer, D. (2014). Neuronal MHC-I expression and its implications in synaptic function, axonal regeneration and Parkinson’s and other brain diseases. Front. Neuroanat. 8:114. doi: 10.3389/fnana.2014.00114
Chambers, R. A., Potenza, M. N., Hoffman, R. E., and Miranker, W. (2004). Simulated apoptosis/neurogenesis regulates learning and memory capabilities of adaptive neural networks. Neuropsychopharmacology 29, 747–758. doi: 10.1038/sj.npp.1300358
Chan, C. J., Heisenberg, C. P., and Hiiragi, T. (2017). Coordination of morphogenesis and cell-fate specification in development. Curr. Biol. 27, R1024–R1035. doi: 10.1016/j.cub.2017.07.010
Chavali, P. L., Pütz, M., and Gergely, F. (2014). Small organelle, big responsibility: the role of centrosomes in development and disease. Philos. Trans. R. Soc. Lond. B Biol. Sci. 369:20130468. doi: 10.1098/rstb.2013.0468
Chavez-Valdez, R., Mottahedin, A., Stridh, L., Yellowhair, T. R., Jantzie, L. L., Northington, F. J., et al. (2019). Evidence for sexual dimorphism in the response to TLR3 activation in the developing neonatal mouse brain: a pilot study. Front. Physiol. 10:306. doi: 10.3389/fphys.2019.00306
Cheeran, M. C., Hu, S., Yager, S. L., Gekker, G., Peterson, P. K., and Lokensgard, J. R. (2001). Cytomegalovirus induces cytokine and chemokine production differentially in microglia and astrocytes: antiviral implications. J. Neurovirol. 7, 135–147. doi: 10.1080/13550280152058799
Cheeran, M. C.-J., Lokensgard, J. R., and Schleiss, M. R. (2009). Neuropathogenesis of congenital cytomegalovirus infection: disease mechanisms and prospects for intervention. Clin. Microbiol. Rev. 22, 99–126. doi: 10.1128/CMR.00023-08
Chess, S. (1971). Autism in children with congenital rubella. J. Autism Child. Schizophr. 1, 33–47. doi: 10.1007/BF01537741
Chiu, C.-F., Chu, L.-W., Liao, I.-C., Simanjuntak, Y., Lin, Y.-L., Juan, C.-C., et al. (2020). The mechanism of the Zika virus crossing the placental barrier and the blood-brain barrier. Front. Microbiol. 11:214. doi: 10.3389/fmicb.2020.00214
Cho, H., Proll, S. C., Szretter, K. J., Katze, M. G., Gale, M. J., and Diamond, M. S. (2013). Differential innate immune response programs in neuronal subtypes determine susceptibility to infection in the brain by positive-stranded RNA viruses. Nat. Med. 19, 458–464. doi: 10.1038/nm.3108
Chu, Y., Jin, X., Parada, I., Pesic, A., Stevens, B., Barres, B., et al. (2010). Enhanced synaptic connectivity and epilepsy in C1q knockout mice. Proc. Natl. Acad. Sci. U.S.A. 107, 7975–7980. doi: 10.1073/pnas.0913449107
Clarke, P. G., Posada, A., Primi, M. P., and Castagné, V. (1998). Neuronal death in the central nervous system during development. Biomed. Pharmacother. 52, 356–362. doi: 10.1016/s0753-3322(99)80002-x
Colonna, M., and Butovsky, O. (2017). Microglia function in the central nervous system during health and neurodegeneration. Annu. Rev. Immunol. 35, 441–468. doi: 10.1146/annurev-immunol-051116-052358
Courchesne, E., Mouton, P. R., Calhoun, M. E., Semendeferi, K., Ahrens-Barbeau, C., Hallet, M. J., et al. (2011). Neuron number and size in prefrontal cortex of children with autism. JAMA 306, 2001–2010. doi: 10.1001/jama.2011.1638
Cunningham, C. L., Martínez-Cerdeño, V., and Noctor, S. C. (2013). Microglia regulate the number of neural precursor cells in the developing cerebral cortex. J. Neurosci. 33, 4216–4233. doi: 10.1523/JNEUROSCI.3441-12.2013
Czeh, M., Gressens, P., and Kaindl, A. M. (2011). The Yin and Yang of microglia. Dev. Neurosci. 33, 199–209. doi: 10.1159/000328989
Dammann, O., and O’Shea, T. M. (2008). Cytokines and perinatal brain damage. Clin. Perinatol. 35, 643–663,v. doi: 10.1016/j.clp.2008.07.011
Dando, S. J., Mackay-Sim, A., Norton, R., Currie, B. J., St John, J. A., Ekberg, J. A. K., et al. (2014). Pathogens penetrating the central nervous system: infection pathways and the cellular and molecular mechanisms of invasion. Clin. Microbiol. Rev. 27, 691–726. doi: 10.1128/CMR.00118-13
Daneman, R., and Prat, A. (2015). The blood-brain barrier. Cold Spring Harb. Perspect. Biol. 7:a020412. doi: 10.1101/cshperspect.a020412
Daniels, B. P., and Klein, R. S. (2015). Knocking on closed doors: host interferons dynamically regulate blood-brain barrier function during viral infections of the central nervous system. PLoS Pathog. 11:e1005096. doi: 10.1371/journal.ppat.1005096
Deverman, B. E., and Patterson, P. H. (2009). Cytokines and CNS development. Neuron 64, 61–78. doi: 10.1016/j.neuron.2009.09.002
Di Liberto, G., Pantelyushin, S., Kreutzfeldt, M., Page, N., Musardo, S., Coras, R., et al. (2018). Neurons under T cell attack coordinate phagocyte-mediated synaptic stripping. Cell 175, 458–471.e19. doi: 10.1016/j.cell.2018.07.049
Dilly, S., Fotso Fotso, A., Lejal, N., Zedda, G., Chebbo, M., Rahman, F., et al. (2018). From naproxen repurposing to naproxen analogues and their antiviral activity against influenza A virus. J. Med. Chem. 61, 7202–7217. doi: 10.1021/acs.jmedchem.8b00557
Dudiki, T., Meller, J., Mahajan, G., Liu, H., Zhevlakova, I., Stefl, S., et al. (2020). Microglia control vascular architecture via a TGFβ1 dependent paracrine mechanism linked to tissue mechanics. Nat. Commun. 11:986. doi: 10.1038/s41467-020-14787-y
Duma, D., Collins, J. B., Chou, J. W., and Cidlowski, J. A. (2010). Sexually dimorphic actions of glucocorticoids provide a link to inflammatory diseases with gender differences in prevalence. Sci. Signal. 3:ra74. doi: 10.1126/scisignal.2001077
Dziembowska, M., Tham, T. N., Lau, P., Vitry, S., Lazarini, F., and Dubois-Dalcq, M. (2005). A role for CXCR4 signaling in survival and migration of neural and oligodendrocyte precursors. Glia 50, 258–269. doi: 10.1002/glia.20170
Elshazzly, M., and Caban, O. (2019). “Embryology, central nervous system,” in StatPearls, (Treasure Island, FL: StatPearls Publishing).
Fagan, K., Crider, A., Ahmed, A. O., and Pillai, A. (2017). Complement C3 expression is decreased in autism spectrum disorder subjects and contributes to behavioral deficits in rodents. Mol. Neuropsychiatry 3, 19–27. doi: 10.1159/000465523
Fang, W.-Q., Chen, W.-W., Jiang, L., Liu, K., Yung, W.-H., Fu, A. K. Y., et al. (2014). Overproduction of upper-layer neurons in the neocortex leads to autism-like features in mice. Cell Rep. 9, 1635–1643. doi: 10.1016/j.celrep.2014.11.003
Freidin, M., Bennett, M. V., and Kessler, J. A. (1992). Cultured sympathetic neurons synthesize and release the cytokine interleukin 1 beta. Proc. Natl Acad. Sci. U.S.A. 89, 10440–10443. doi: 10.1073/pnas.89.21.10440
Galic, M. A., Riazi, K., and Pittman, Q. J. (2012). Cytokines and brain excitability. Front. Neuroendocrinol. 33:116–125. doi: 10.1016/j.yfrne.2011.12.002
Garay, P. A., and McAllister, A. K. (2010). Novel roles for immune molecules in neural development: implications for neurodevelopmental disorders. Front. Synaptic Neurosci. 2:136. doi: 10.3389/fnsyn.2010.00136
Gaser, C., Nenadic, I., Buchsbaum, B. R., Hazlett, E. A., and Buchsbaum, M. S. (2004). Ventricular enlargement in schizophrenia related to volume reduction of the thalamus, striatum, and superior temporal cortex. Am. J. Psychiatry 161, 154–156. doi: 10.1176/appi.ajp.161.1.154
Ghafouri, M., Amini, S., Khalili, K., and Sawaya, B. E. (2006). HIV-1 associated dementia: symptoms and causes. Retrovirology 3:28. doi: 10.1186/1742-4690-3-28
Ginhoux, F., Lim, S., Hoeffel, G., Low, D., and Huber, T. (2013). Origin and differentiation of microglia. Front. Cell. Neurosci. 7:45. doi: 10.3389/fncel.2013.00045
Goldenberg, R. L., Andrews, W. W., Faye-Petersen, O., Cliver, S., Goepfert, A. R., and Hauth, J. C. (2006). The alabama preterm birth project: placental histology in recurrent spontaneous and indicated preterm birth. Am. J. Obstet. Gynecol. 195, 792–796. doi: 10.1016/j.ajog.2006.05.050
Götz, M., and Huttner, W. B. (2005). The cell biology of neurogenesis. Nat. Rev. Mol. Cell Biol. 6, 777–788. doi: 10.1038/nrm1739
Han, M., Zhang, J.-C., and Hashimoto, K. (2017). Increased levels of C1q in the prefrontal cortex of adult offspring after maternal immune activation: prevention by 7,8-dihydroxyflavone. Clin. Psychopharmacol. Neurosci. 15, 64–67. doi: 10.9758/cpn.2017.15.1.64
Hanamsagar, R., Alter, M. D., Block, C. S., Sullivan, H., Bolton, J. L., and Bilbo, S. D. (2017). Generation of a microglial developmental index in mice and in humans reveals a sex difference in maturation and immune reactivity. Glia 65, 1504–1520. doi: 10.1002/glia.23176
Hanisch, U.-K. (2002). Microglia as a source and target of cytokines. Glia 40, 140–155. doi: 10.1002/glia.10161
Harrison, J. K., Jiang, Y., Chen, S., Xia, Y., Maciejewski, D., McNamara, R. K., et al. (1998). Role for neuronally derived fractalkine in mediating interactions between neurons and CX3CR1-expressing microglia. Proc. Natl. Acad. Sci. U.S.A. 95, 10896–10901. doi: 10.1073/pnas.95.18.10896
He, Q., Liu, H., Huang, C., Wang, R., Luo, M., and Lu, W. (2020). Herpes simplex virus 1-induced blood-brain barrier damage involves apoptosis associated with GM130-mediated golgi stress. Front. Mol. Neurosci. 13:2. doi: 10.3389/fnmol.2020.00002
Heffernan, A. L., and Hare, D. J. (2018). Tracing environmental exposure from neurodevelopment to neurodegeneration. Trends Neurosci. 41, 496–501. doi: 10.1016/j.tins.2018.04.005
Herculano-Houzel, S. (2009). The human brain in numbers: a linearly scaled-up primate brain. Front. Hum. Neurosci. 3:31. doi: 10.3389/neuro.09.031.2009
Huttner, W. B., and Kosodo, Y. (2005). Symmetric versus asymmetric cell division during neurogenesis in the developing vertebrate central nervous system. Curr. Opin. Cell Biol. 17, 648–657. doi: 10.1016/j.ceb.2005.10.005
Hutton, J. (2016). Does rubella cause autism: a 2015 reappraisal? Front. Hum. Neurosci. 10:25. doi: 10.3389/fnhum.2016.00025
Iliff, J. J., Wang, M., Liao, Y., Plogg, B. A., Peng, W., Gundersen, G. A., et al. (2012). A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci. Transl. Med. 4:147ra111. doi: 10.1126/scitranslmed.3003748
Jackson, S. E., Mason, G. M., and Wills, M. R. (2011). Human cytomegalovirus immunity and immune evasion. Virus Res. 157, 151–160. doi: 10.1016/j.virusres.2010.10.031
Jarskog, L. F., Glantz, L. A., Gilmore, J. H., and Lieberman, J. A. (2005). Apoptotic mechanisms in the pathophysiology of schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 29, 846–858. doi: 10.1016/j.pnpbp.2005.03.010
Jenkins, C., Abendroth, A., and Slobedman, B. (2004). A novel viral transcript with homology to human interleukin-10 is expressed during latent human cytomegalovirus infection. J. Virol. 78, 1440–1447. doi: 10.1128/jvi.78.3.1440-1447.2004
Jiang, N. M., Cowan, M., Moonah, S. N., and Petri, W. A. J. (2018). The impact of systemic inflammation on neurodevelopment. Trends Mol. Med. 24, 794–804. doi: 10.1016/j.molmed.2018.06.008
Johnston, J. B., and McFadden, G. (2003). Poxvirus immunomodulatory strategies: current perspectives. J. Virol. 77, 6093–6100. doi: 10.1128/jvi.77.11.6093-6100.2003
Juncal-Ruiz, M., Riesco-Dávila, L., Ortiz-García de la Foz, V., Martínez-Garcia, O., Ramírez-Bonilla, M., Ocejo-Viñals, J. G., et al. (2018). Comparison of the anti-inflammatory effect of aripiprazole and risperidone in 75 drug-naïve first episode psychosis individuals: a 3 months randomized study. Schizophr. Res. 202, 226–233. doi: 10.1016/j.schres.2018.06.039
Kang, H. J., Kawasawa, Y. I., Cheng, F., Zhu, Y., Xu, X., Li, M., et al. (2011). Spatio-temporal transcriptome of the human brain. Nature 478, 483–489. doi: 10.1038/nature10523
Kanold, P. O. (2009). Subplate neurons: crucial regulators of cortical development and plasticity. Front. Neuroanat. 3:16. doi: 10.3389/neuro.05.016.2009
Kealy, J., Greene, C., and Campbell, M. (2020). Blood-brain barrier regulation in psychiatric disorders. Neurosci. Lett. 726:133664. doi: 10.1016/j.neulet.2018.06.033
Kendell, R. E., and Kemp, I. W. (1989). Maternal influenza in the etiology of schizophrenia. Arch. Gen. Psychiatry 46, 878–882. doi: 10.1001/archpsyc.1989.01810100020004
Kennedy, P. G. E., Rovnak, J., Badani, H., and Cohrs, R. J. (2015). A comparison of herpes simplex virus type 1 and varicella-zoster virus latency and reactivation. J. Gen. Virol. 96(Pt 7), 1581–1602. doi: 10.1099/vir.0.000128
Kim, W.-K., Corey, S., Alvarez, X., and Williams, K. (2003). Monocyte/macrophage traffic in HIV and SIV encephalitis. J. Leukoc. Biol. 74, 650–656. doi: 10.1189/jlb.0503207
Kim, Y. S., and Joh, T. H. (2006). Microglia, major player in the brain inflammation: their roles in the pathogenesis of Parkinson’s disease. Exp. Mol. Med. 38, 333–347.
Kipnis, J. (2016). Multifaceted interactions between adaptive immunity and the central nervous system. Science (New York, N.Y.) 353, 766–771. doi: 10.1126/science.aag2638
Klein, R. S., Garber, C., and Howard, N. (2017). Infectious immunity in the central nervous system and brain function. Nat. Immunol. 18, 132–141. doi: 10.1038/ni.3656
Klein, R. S., Lin, E., Zhang, B., Luster, A. D., Tollett, J., Samuel, M. A., et al. (2005). Neuronal CXCL10 directs CD8+ T-cell recruitment and control of West Nile virus encephalitis. J. Virol. 79, 11457–11466. doi: 10.1128/JVI.79.17.11457-11466.2005
Klein, S. L., and Flanagan, K. L. (2016). Sex differences in immune responses. Nat. Rev. Immunol. 16, 626–638. doi: 10.1038/nri.2016.90
Knoblich, J. A. (2008). Mechanisms of asymmetric stem cell division. Cell 132, 583–597. doi: 10.1016/j.cell.2008.02.007
Knuesel, I., Chicha, L., Britschgi, M., Schobel, S. A., Bodmer, M., Hellings, J. A., et al. (2014). Maternal immune activation and abnormal brain development across CNS disorders. Nat. Rev. Neurol. 10, 643–660. doi: 10.1038/nrneurol.2014.187
Korn, T., and Kallies, A. (2017). T cell responses in the central nervous system. Nat. Rev. Immunol. 17, 179–194. doi: 10.1038/nri.2016.144
Kracht, L., Borggrewe, M., Eskandar, S., Brouwer, N., Chuva de Sousa Lopes, S. M., Laman, J. D., et al. (2020). Human fetal microglia acquire homeostatic immune-sensing properties early in development. Science 369, 530–537. doi: 10.1126/science.aba5906
Krauer, F., Riesen, M., Reveiz, L., Oladapo, O. T., Porgo, V., Haefliger, A., et al. (2017). Zika virus infection as a cause of congenital brain abnormalities and guillain – barre syndrome: systematic review. PLoS Med. 14:e1002203. doi: 10.1371/journal.pmed.1002203
Krenn, V., Bosone, C., Burkard, T. R., Garcez, P. P., Mirazimi, A., Knoblich, A., et al. (2021). Organoid modeling of Zika and herpes simplex virus 1 infections reveals virus-specific responses leading to microcephaly. Cell Stem Cell 28, 1362–1379.e7. doi: 10.1016/j.stem.2021.03.004
Kumar, A., Hou, S., Airo, A. M., Limonta, D., Mancinelli, V., Branton, W., et al. (2016). Zika virus inhibits type-I interferon production and downstream signaling. EMBO Rep. 17, 1766–1775. doi: 10.15252/embr.201642627
Lazutkin, A., Podgorny, O., and Enikolopov, G. (2019). Modes of division and differentiation of neural stem cells. Behav. Brain Res. 374:112118. doi: 10.1016/j.bbr.2019.112118
Leviton, A., Allred, E. N., Fichorova, R. N., Kuban, K. C. K., Michael, O., Shea, T., et al. (2016). Systemic inflammation on postnatal days 21 and 28 and indicators of brain dysfunction 2years later among children born before the 28th week of gestation. Early Hum. Dev. 93, 25–32. doi: 10.1016/j.earlhumdev.2015.11.004
Li, G., Ning, Z., Liu, Y., and Li, X. (2017). Neurological manifestations of dengue infection. Front. Cell. Infect. Microbiol. 7:449. doi: 10.3389/fcimb.2017.00449
Li, M., and Ransohoff, R. M. (2008). Multiple roles of chemokine CXCL12 in the central nervous system: a migration from immunology to neurobiology. Prog. Neurobiol. 84, 116–131. doi: 10.1016/j.pneurobio.2007.11.003
Li, Y., Zhang, C., Chen, X., Yu, J., Wang, Y., Yang, Y., et al. (2011). ICP34.5 protein of herpes simplex virus facilitates the initiation of protein translation by bridging eukaryotic initiation factor 2alpha (eIF2alpha) and protein phosphatase 1. J. Biol. Chem. 286, 24785–24792. doi: 10.1074/jbc.M111.232439
Liddelow, S. A. (2015). Development of the choroid plexus and blood-CSF barrier. Front. Neurosci. 9:32. doi: 10.3389/fnins.2015.00032
Loo, Y.-M., and Gale, M. (2011). Immune signaling by RIG-I-like receptors. Immunity 34, 680–692. doi: 10.1016/j.immuni.2011.05.003
Lu, B., Jan, L., and Jan, Y. N. (2000). Control of cell divisions in the nervous system: symmetry and asymmetry. Annu. Rev. Neurosci. 23, 531–556. doi: 10.1146/annurev.neuro.23.1.531
Magdalon, J., Mansur, F., Teles E Silva, A. L., de Goes, V. A., Reiner, O., and Sertié, A. L. (2020). Complement system in brain architecture and neurodevelopmental disorders. Front. Neurosci. 14:23. doi: 10.3389/fnins.2020.00023
Mallard, C., Tremblay, M.-E., and Vexler, Z. S. (2019). Microglia and neonatal brain injury. Neuroscience 405, 68–76. doi: 10.1016/j.neuroscience.2018.01.023
Mednick, S. A., Machon, R. A., Huttunen, M. O., and Bonett, D. (1988). Adult schizophrenia following prenatal exposure to an influenza epidemic. Arch. Gen. Psychiatry 45, 189–192. doi: 10.1001/archpsyc.1988.01800260109013
Melchjorsen, J., Sørensen, L. N., and Paludan, S. R. (2003). Expression and function of chemokines during viral infections: from molecular mechanisms to in vivo function. J. Leukoc. Biol. 74, 331–343. doi: 10.1189/jlb.1102577
Meunier, E., and Broz, P. (2016). Interferon-inducible GTPases in cell autonomous and innate immunity. Cell. Microbiol. 18, 168–180. doi: 10.1111/cmi.12546
Meyer, U., Feldon, J., and Yee, B. K. (2009). A review of the fetal brain cytokine imbalance hypothesis of schizophrenia. Schizophr. Bull. 35, 959–972. doi: 10.1093/schbul/sbn022
Meyer, U., Nyffeler, M., Engler, A., Urwyler, A., Schedlowski, M., Knuesel, I., et al. (2006). The time of prenatal immune challenge determines the specificity of inflammation-mediated brain and behavioral pathology. J. Neurosci. 26, 4752–4762. doi: 10.1523/JNEUROSCI.0099-06.2006
Meyer, U., Yee, B. K., and Feldon, J. (2007). The neurodevelopmental impact of prenatal infections at different times of pregnancy: the earlier the worse? Neuroscientist 13, 241–256. doi: 10.1177/1073858406296401
Miller, R. J., Rostene, W., Apartis, E., Banisadr, G., Biber, K., Milligan, E. D., et al. (2008). Chemokine action in the nervous system. J. Neurosci. 28, 11792–11795. doi: 10.1523/JNEUROSCI.3588-08.2008
Miyamoto, A., Wake, H., Ishikawa, A. W., Eto, K., Shibata, K., Murakoshi, H., et al. (2016). Microglia contact induces synapse formation in developing somatosensory cortex. Nat. Commun. 7:12540. doi: 10.1038/ncomms12540
Moseman, E. A., Blanchard, A. C., Nayak, D., and McGavern, D. B. (2020). T cell engagement of cross-presenting microglia protects the brain from a nasal virus infection. Sci. Immunol. 5:eabb1817. doi: 10.1126/sciimmunol.abb1817
Mousa, A., SeigerA, Kjaeldgaard, A., and Bakhiet, M. (1999). Human first trimester forebrain cells express genes for inflammatory and anti-inflammatory cytokines. Cytokine 11, 55–60. doi: 10.1006/cyto.1998.0381
Munshi, S., Parrilli, V., and Rosenkranz, J. A. (2019). Peripheral anti-inflammatory cytokine Interleukin-10 treatment mitigates interleukin-1β–induced anxiety and sickness behaviors in adult male rats. Behav. Brain Res. 372:112024. doi: 10.1016/j.bbr.2019.112024
Nadarajah, B., and Parnavelas, J. G. (2002). Modes of neuronal migration in the developing cerebral cortex. Nat. Rev. Neurosci. 3, 423–432. doi: 10.1038/nrn845
Nair, S., and Diamond, M. S. (2016). Innate immune interactions within the central nervous system modulate pathogenesis of viral infections. Curr. Opin. Immunol. 36, 47–53. doi: 10.1016/j.coi.2015.06.011
Nayak, D., Roth, T. L., and McGavern, D. B. (2014). Microglia development and function. Annu. Rev. Immunol. 32, 367–402. doi: 10.1146/annurev-immunol-032713-120240
Nelson, L. H., and Lenz, K. M. (2017). The immune system as a novel regulator of sex differences in brain and behavioral development. J. Neurosci. Res. 95, 447–461. doi: 10.1002/jnr.23821
Oglesbee, M., and Niewiesk, S. (2011). Measles virus neurovirulence and host immunity. Future Virol. 6, 85–99. doi: 10.2217/fvl.10.70
Ohka, S., Nihei, C., Yamazaki, M., and Nomoto, A. (2012). Poliovirus trafficking toward central nervous system via human poliovirus receptor-dependent and -independent pathway. Front. Microbiol. 3:147. doi: 10.3389/fmicb.2012.00147
O’Keefe, G. M., Nguyen, V. T., and Benveniste, E. N. (1999). Class II transactivator and class II MHC gene expression in microglia: modulation by the cytokines TGF-beta, IL-4, IL-13 and IL-10. Eur. J. Immunol. 29, 1275–1285. doi: 10.1002/(SICI)1521-4141(199904)29:04<1275::AID-IMMU1275<3.0.CO;2-T
Okun, E., Griffioen, K. J., and Mattson, M. P. (2011). Toll-like receptor signaling in neural plasticity and disease. Trends Neurosci. 34, 269–281. doi: 10.1016/j.tins.2011.02.005
Oppenheim, R. W. (1991). Cell death during development of the nervous system. Annu. Rev. Neurosci. 14, 453–501. doi: 10.1146/annurev.ne.14.030191.002321
Ozaki, K., Kato, D., Ikegami, A., Hashimoto, A., Sugio, S., Guo, Z., et al(2020). Maternal immune activation induces sustained changes in fetal microglia motility. Sci. Rep. 10:21378. doi: 10.1038/s41598-020-78294-2
Pan, T., Peng, Z., Tan, L., Zou, F., Zhou, N., Liu, B., et al. (2018). Nonsteroidal anti-inflammatory drugs potently inhibit the replication of zika viruses by inducing the degradation of AXL. J. Virol. 92:e01018-18. doi: 10.1128/JVI.01018-18
Pandey, M., Chauhan, M., and Awasthi, S. (2017). Interplay of cytokines in preterm birth. Indian J. Med. Res. 146, 316–327. doi: 10.4103/ijmr.IJMR_1624_14
Paolicelli, R. C., Bolasco, G., Pagani, F., Maggi, L., Scianni, M., Panzanelli, P., et al. (2011). Synaptic pruning by microglia is necessary for normal brain development. Science (New York, N.Y.) 333, 1456–1458. doi: 10.1126/science.1202529
Pawelec, P., Ziemka-Nalecz, M., Sypecka, J., and Zalewska, T. (2020). The impact of the CX3CL1/CX3CR1 axis in neurological disorders. Cells 9:2277. doi: 10.3390/cells9102277
Pellegrini, L., Albecka, A., Mallery, D. L., Kellner, M. J., Paul, D., Carter, A. P., et al. (2020). SARS-CoV-2 infects the brain choroid plexus and disrupts the blood-CSF barrier in human brain organoids. Cell Stem Cell 27, 951–961.e5. doi: 10.1016/j.stem.2020.10.001
Perry, V. H., Nicoll, J. A. R., and Holmes, C. (2010). Microglia in neurodegenerative disease. Nat. Rev. Neurol. 6, 193–201. doi: 10.1038/nrneurol.2010.17
Peterson, S. E., Yang, A. H., Bushman, D. M., Westra, J. W., Yung, Y. C., Barral, S., et al. (2012). Aneuploid cells are differentially susceptible to caspase-mediated death during embryonic cerebral cortical development. J. Neurosci. 32, 16213–16222. doi: 10.1523/JNEUROSCI.3706-12.2012
Pierson, T. C., and Diamond, M. S. (2020). The continued threat of emerging flaviviruses. Nat. Microbiol. 5, 796–812. doi: 10.1038/s41564-020-0714-0
Prinz, M., Jung, S., and Priller, J. (2019). Microglia biology: one century of evolving concepts. Cell 179, 292–311. doi: 10.1016/j.cell.2019.08.053
Puerta-Guardo, H., Glasner, D. R., and Harris, E. (2016). Dengue virus NS1 disrupts the endothelial glycocalyx, leading to hyperpermeability. PLoS Pathog. 12:e1005738. doi: 10.1371/journal.ppat.1005738
Radjavi, A., Smirnov, I., and Kipnis, J. (2014). Brain antigen-reactive CD4+ T cells are sufficient to support learning behavior in mice with limited T cell repertoire. Brain Behav. Immun. 35, 58–63. doi: 10.1016/j.bbi.2013.08.013
Ratnayake, U., Quinn, T., Walker, D. W., and Dickinson, H. (2013). Cytokines and the neurodevelopmental basis of mental illness. Front. Neurosci. 7:180. doi: 10.3389/fnins.2013.00180
Raval, A., Puri, N., Rath, P. C., and Saxena, R. K. (1998). Cytokine regulation of expression of class I MHC antigens cytokines which modulate expression of MHC class I molecules. Exp. Mol. Med. 30, 1–13.
Redzic, Z. (2011). Molecular biology of the blood-brain and the blood-cerebrospinal fluid barriers: similarities and differences. Fluids Barriers CNS 8:3. doi: 10.1186/2045-8118-8-3
Rehen, S. K., McConnell, M. J., Kaushal, D., Kingsbury, M. A., Yang, A. H., and Chun, J. (2001). Chromosomal variation in neurons of the developing and adult mammalian nervous system. Proc. Natl. Acad. Sci. U.S.A. 98, 13361–13366. doi: 10.1073/pnas.231487398
Roberts, L. M., Black, D. S., Raman, C., Woodford, K., Zhou, M., Haggerty, J. E., et al. (2008). Subcellular localization of transporters along the rat blood-brain barrier and blood-cerebral-spinal fluid barrier by in vivo biotinylation. Neuroscience 155, 423–438. doi: 10.1016/j.neuroscience.2008.06.015
Rosenblat, J. D. (2019). Targeting the immune system in the treatment of bipolar disorder. Psychopharmacology 236, 2909–2921. doi: 10.1007/s00213-019-5175-x
Rudolph, M. D., Graham, A. M., Feczko, E., Miranda-Dominguez, O., Rasmussen, J. M., Nardos, R., et al. (2018). Maternal IL-6 during pregnancy can be estimated from newborn brain connectivity and predicts future working memory in offspring. Nat. Neurosci. 21, 765–772. doi: 10.1038/s41593-018-0128-y
Sanjabi, S., Zenewicz, L. A., Kamanaka, M., and Flavell, R. A. (2009). Anti-inflammatory and pro-inflammatory roles of TGF-beta, IL-10, and IL-22 in immunity and autoimmunity. Curr. Opin. Pharmacol. 9, 447–453. doi: 10.1016/j.coph.2009.04.008
Saunders, N. R., Liddelow, S. A., and Dziegielewska, K. M. (2012). Barrier mechanisms in the developing brain. Front. Pharmacol. 3:46. doi: 10.3389/fphar.2012.00046
Scola, G., and Duong, A. (2017). Prenatal maternal immune activation and brain development with relevance to psychiatric disorders. Neuroscience 346, 403–408. doi: 10.1016/j.neuroscience.2017.01.033
Sezgin, E., Jabs, D. A., Hendrickson, S. L., Van Natta, M., Zdanov, A., Lewis, A., et al. (2010). Effect of host genetics on the development of cytomegalovirus retinitis in patients with AIDS. J. Infect. Dis. 202, 606–613. doi: 10.1086/654814
Shrestha, B., Zhang, B., Purtha, W. E., Klein, R. S., and Diamond, M. S. (2008). Tumor necrosis factor alpha protects against lethal West Nile virus infection by promoting trafficking of mononuclear leukocytes into the central nervous system. J. Virol. 82, 8956–8964. doi: 10.1128/JVI.01118-08
Sicotte, N. L., and Voskuhl, R. R. (2001). Onset of multiple sclerosis associated with anti-TNF therapy. Neurology 57, 1885–1888. doi: 10.1212/wnl.57.10.1885
Sierra, A., Encinas, J. M., Deudero, J. J. P., Chancey, J. H., Enikolopov, G., Overstreet-Wadiche, L. S., et al. (2010). Microglia shape adult hippocampal neurogenesis through apoptosis-coupled phagocytosis. Cell Stem Cell 7, 483–495. doi: 10.1016/j.stem.2010.08.014
Silverman, M. N., Pearce, B. D., Biron, C. A., and Miller, A. H. (2005). Immune modulation of the hypothalamic-pituitary-adrenal (HPA) axis during viral infection. Viral Immunol. 18, 41–78. doi: 10.1089/vim.2005.18.41
Smith, S. A., and Kotwal, G. J. (2001). Virokines: novel immunomodulatory agents. Expert Opin. Biol. Ther. 1, 343–357. doi: 10.1517/14712598.1.3.343
Smith, S. E. P., Li, J., Garbett, K., Mirnics, K., and Patterson, P. H. (2007). Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 27, 10695–10702. doi: 10.1523/JNEUROSCI.2178-07.2007
Solek, C. M., Farooqi, N., Verly, M., Lim, T. K., and Ruthazer, E. S. (2018). Maternal immune activation in neurodevelopmental disorders. Dev. Dyn. 247, 588–619. doi: 10.1002/dvdy.24612
Sroykham, W., and Wongsawat, Y. (2019). Effects of brain activity, morning salivary cortisol, and emotion regulation on cognitive impairment in elderly people. Medicine 98:e16114. doi: 10.1097/MD.0000000000016114
Stassart, R. M., Möbius, W., Nave, K.-A., and Edgar, J. M. (2018). The axon-myelin unit in development and degenerative disease. Front. Neurosci. 12:467. doi: 10.3389/fnins.2018.00467
Stern, H., Booth, J. C., Elek, S. D., and Fleck, D. G. (1969). Microbial causes of mental retardation. The role of prenatal infections with cytomegalovirus, rubella virus, and toxoplasma. Lancet 2, 443–448. doi: 10.1016/s0140-6736(69)90162-7
Stevens, B., Allen, N. J., Vazquez, L. E., Howell, G. R., Christopherson, K. S., Nouri, N., et al. (2007). The classical complement cascade mediates CNS synapse elimination. Cell 131, 1164–1178. doi: 10.1016/j.cell.2007.10.036
Stiles, J., and Jernigan, T. L. (2010). The basics of brain development. Neuropsychol. Rev. 20, 327–348. doi: 10.1007/s11065-010-9148-4
Stoermer, K. A., and Morrison, T. E. (2011). Complement and viral pathogenesis. Virology 411, 362–373. doi: 10.1016/j.virol.2010.12.045
Stolp, H. B. (2013). Neuropoietic cytokines in normal brain development and neurodevelopmental disorders. Mol. Cell. Neurosci. 53, 63–68. doi: 10.1016/j.mcn.2012.08.009
Straface, G., Selmin, A., Zanardo, V., De Santis, M., Ercoli, A., and Scambia, G. (2012). Herpes simplex virus infection in pregnancy. Infect. Dis. Obstet. Gynecol. 2012:385697. doi: 10.1155/2012/385697
Subramanian, L., Bershteyn, M., Paredes, M. F., and Kriegstein, A. R. (2017). Dynamic behaviour of human neuroepithelial cells in the developing forebrain. Nat. Commun. 8:14167. doi: 10.1038/ncomms14167
Sui, Y., Stehno-Bittel, L., Li, S., Loganathan, R., Dhillon, N. K., Pinson, D., et al. (2006). CXCL10-induced cell death in neurons: role of calcium dysregulation. Eur. J. Neurosci. 23, 957–964. doi: 10.1111/j.1460-9568.2006.04631.x
Tanabe, S., and Yamashita, T. (2018). B-1a lymphocytes promote oligodendrogenesis during brain development. Nat. Neurosci. 21, 506–516. doi: 10.1038/s41593-018-0106-4
Tanaka, T., Narazaki, M., and Kishimoto, T. (2014). IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 6:a016295. doi: 10.1101/cshperspect.a016295
Tau, G. Z., and Peterson, B. S. (2010). Normal development of brain circuits. Neuropsychopharmacology. 35, 147–168. doi: 10.1038/npp.2009.115
Traiffort, E., Kassoussi, A., Zahaf, A., and Laouarem, Y. (2020). Astrocytes and microglia as major players of myelin production in normal and pathological conditions. Front. Cell. Neurosci. 14:79. doi: 10.3389/fncel.2020.00079
Trettel, F., Di Castro, M. A., and Limatola, C. (2020). Chemokines: key molecules that orchestrate communication among neurons, microglia and astrocytes to preserve brain function. Neuroscience 439, 230–240. doi: 10.1016/j.neuroscience.2019.07.035
Van Rie, A., Mupuala, A., and Dow, A. (2008). Impact of the HIV/AIDS epidemic on the neurodevelopment of preschool-aged children in Kinshasa, Democratic Republic of the Congo. Pediatrics 122, e123–e128. doi: 10.1542/peds.2007-2558
Van Sant, C., Hagglund, R., Lopez, P., and Roizman, B. (2001). The infected cell protein 0 of herpes simplex virus 1 dynamically interacts with proteasomes, binds and activates the cdc34 E2 ubiquitin-conjugating enzyme, and possesses in vitro E3 ubiquitin ligase activity. Proc. Natl. Acad. Sci. U.S.A. 98, 8815–8820. doi: 10.1073/pnas.161283098
Verma, S., Lo, Y., Chapagain, M., Lum, S., Kumar, M., Gurjav, U., et al. (2009). West Nile virus infection modulates human brain microvascular endothelial cells tight junction proteins and cell adhesion molecules: transmigration across the in vitro blood-brain barrier. Virology 385, 425–433. doi: 10.1016/j.virol.2008.11.047
Watkins, L. R., Maier, S. F., and Goehler, L. E. (1995). Cytokine-to-brain communication: a review & analysis of alternative mechanisms. Life Sci. 57, 1011–1026. doi: 10.1016/0024-3205(95)02047-m
Wei, H., Alberts, I., and Li, X. (2014). The apoptotic perspective of autism. Int. J. Dev. Neurosci. 36, 13–18. doi: 10.1016/j.ijdevneu.2014.04.004
Werling, D. M. (2016). The role of sex-differential biology in risk for autism spectrum disorder. Biol. Sex Differ. 7:58. doi: 10.1186/s13293-016-0112-8
Werling, D. M., and Geschwind, D. H. (2013). Sex differences in autism spectrum disorders. Curr. Opin. Neurol. 26, 146–153. doi: 10.1097/WCO.0b013e32835ee548
Wu, W., Dietze, K. K., Gibbert, K., Lang, K. S., Trilling, M., Yan, H., et al. (2015). TLR ligand induced IL-6 counter-regulates the anti-viral CD8(+) T cell response during an acute retrovirus infection. Sci. Rep. 5:10501. doi: 10.1038/srep10501
Xu, N., Li, X., and Zhong, Y. (2015). Inflammatory cytokines: potential biomarkers of immunologic dysfunction in autism spectrum disorders. Mediators Inflamm. 2015:531518. doi: 10.1155/2015/531518
Yamaguchi, Y., and Miura, M. (2015). Programmed cell death in neurodevelopment. Dev. Cell 32, 478–490. doi: 10.1016/j.devcel.2015.01.019
Yamaguchi, Y., Shinotsuka, N., Nonomura, K., Takemoto, K., Kuida, K., Yosida, H., et al. (2011). Live imaging of apoptosis in a novel transgenic mouse highlights its role in neural tube closure. J. Cell Biol. 195, 1047–1060. doi: 10.1083/jcb.201104057
Zager, A., Pinheiro, M. L., Ferraz-de-Paula, V., Ribeiro, A., and Palermo-Neto, J. (2013). Increased cell-mediated immunity in male mice offspring exposed to maternal immune activation during late gestation. Int. Immunopharmacol. 17, 633–637. doi: 10.1016/j.intimp.2013.08.007
Zanin, N., Viaris de Lesegno, C., Lamaze, C., and Blouin, C. M. (2021). Interferon receptor trafficking and signaling: journey to the cross roads. Front. Immunol. 11:3512. doi: 10.3389/fimmu.2020.615603
Zengeler, K. E., and Lukens, J. R. (2021). Innate immunity at the crossroads of healthy brain maturation and neurodevelopmental disorders. Nat. Rev. Immunol. 21, 454–468. doi: 10.1038/s41577-020-00487-7
Keywords: autism spectrum disorder (ASD), blood brain Barrier (BBB), blood CSF barrier (BCSFB), microcephaly, neurodevelopment, inflammation, cytokines
Citation: Ganguli S and Chavali PL (2021) Intrauterine Viral Infections: Impact of Inflammation on Fetal Neurodevelopment. Front. Neurosci. 15:771557. doi: 10.3389/fnins.2021.771557
Received: 06 September 2021; Accepted: 18 October 2021;
Published: 10 November 2021.
Edited by:
Yasir Ahmed Syed, Cardiff University, United KingdomReviewed by:
Sara A. Abdulla, Qatar Biomedical Research Institute, QatarCopyright © 2021 Ganguli and Chavali. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Pavithra L. Chavali, cGxjaGF2YWxpQGNjbWIucmVzLmlu
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.