
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
BRIEF RESEARCH REPORT article
Front. Neurosci., 25 January 2021
Sec. Sleep and Circadian Rhythms
Volume 15 - 2021 | https://doi.org/10.3389/fnins.2021.638122
This article is part of the Research TopicDevelopment of Circadian Clock FunctionsView all 11 articles
Senescent cells, which show the permanent growth arrest in response to various forms of stress, accumulate in the body with the progression of age, and are associated with aging and age-associated diseases. Although the senescent cells are growth arrested, they still demonstrate high metabolic rate and altered gene expressions, indicating that senescent cells are still active. We recently showed that the circadian clock properties, namely phase and period of the cells, are altered with the establishment of replicative senescence. However, whether cellular senescence triggers the alteration of circadian clock properties in the cells is still unknown. In this study we show that the oxidative stress-induced premature senescence induces the alterations of the circadian clock, similar to the phenotypes of the replicative senescent cells. We found that the oxidative stress-induced premature senescent cells display the prolonged period and delayed phases. In addition, the magnitude of these changes intensified over time, indicating that cellular senescence changes the circadian clock properties. Our current results corroborate with our previous findings and further confirm that cellular senescence induces altered circadian clock properties, irrespective of the replicative senescence or the stress-induced premature senescence.
Cellular senescence is the state of permanent growth arrest of cells. The senescent cells have been found to be accumulated in the body with aging, and have been associated with various age-related diseases, for example, atherosclerosis (Wang and Bennett, 2012; Childs et al., 2018; Cho et al., 2020), osteoarthritis (Jeon et al., 2017, 2019; Xu et al., 2017), alveolar lung diseases (Hashimoto et al., 2016; Schafer et al., 2017; Houssaini et al., 2018), and cancer (Parrinello et al., 2005; Bavik et al., 2006; Liu and Hornsby, 2007; Bhatia et al., 2008; Campisi et al., 2011; Castro-Vega et al., 2015; Ortiz-Montero et al., 2017). Removal of the senescent cells from the body, either using the pharmacologic interventions (Chang et al., 2016; Yosef et al., 2016; Baar et al., 2017; Lehmann et al., 2017; Schafer et al., 2017; Bussian et al., 2018; Zhang et al., 2019) or genetic ablations (Baker et al., 2011, 2016; Childs et al., 2016; Hashimoto et al., 2016; Bussian et al., 2018), have recently been reported to lead to the extended healthspan of prematurely and naturally aged mice and also attenuated the already existing diseases in mouse models of disease. Various forms of stress such as excessive cell proliferation, oncogenic stress and extreme DNA damage induce cellular senescence. These different forms of stress lead to the cells having the different types of the cellular senescence, such as the replicative senescence, oncogene-induced senescence and the stress-induced premature senescence. Despite the fact that the various types of the senescent cells are permanently growth arrested, they still have their individual differential transcriptome signatures, and secretory phenotype (Maciel-Baron et al., 2016; Hernandez-Segura et al., 2017; Nakao et al., 2020). Hence it can be postulated that the presence of the replicative senescent cells, oncogene-induced senescent cells, and stress-induced premature senescent cells may affect the physiological systems differentially. In vivo it is currently impossible to distinguish between the different types of the senescent cells and the effects they exert (Hernandez-Segura et al., 2017). Recently, we found that circadian clock properties are altered with replicative senescence. However, whether the alteration of the circadian clock is specific for the replicative senescent cells or is also observed in the other types of senescence programs is still largely known.
The circadian clock, which is an intrinsic time-keeping system of almost all living systems on earth, possesses robust and flexible mechanisms against environmental light/dark condition (Partch et al., 2014; Bass and Lazar, 2016; Takahashi, 2017; Honma, 2018). However, it has been found that the circadian clock becomes less robust and flexible with aging, both at the animal level (Valentinuzzi et al., 1997) and also at the tissue levels (Nakamura et al., 2015). Also, at the cellular level, we recently found that the circadian clock is altered with the establishment of replicative senescence; the circadian period becomes longer, and the peak phases are delayed compared with the proliferative cells (Ahmed et al., 2019). We assume that cellular senescence affects the circadian clock mechanism, but not vice versa, since we have reported that the fibroblast cells derived from Bmal1 knockout mice embryo in which circadian clock is completely disrupted, show the normal senescence process (Nakahata et al., 2018). Although in our previous paper, we showed that the circadian clock is altered with the establishment of replicative senescence, till date, no evidence has directly demonstrated that cellular senescence per se affects the circadian clock mechanism. Hence, in this study, we induce oxidative stress-induced premature senescence of human primary fibroblasts, to investigate whether other types of senescence affect the circadian clock and therefore, confirm that cellular senescence affects the circadian clock, irrespective of the type of senescence.
Primary human lung fetal fibroblasts (TIG-3) of Japanese origin were kindly provided by Drs. T. Takumi and T. Akagi. The cells were cultured in DMEM-4.5 g/L glucose (Nacalai Tesque, Japan) supplemented with 10% FBS (Sigma) and antibiotics (100 units/mL penicillin, 100 μg/mL streptomycin, Nacalai Tesque, Japan) at 37°C and 5% CO2 in a humidified incubator. The proliferative cells used in this study were established in our previous report (Ahmed et al., 2019) which consisted of cells in the passage range of P25-29.
For the induction of oxidative stress-induced premature senescence H2O2 was used as the stressor. The proliferative cells were plated on 6-well plates at the seeding density of 8.5 × 104 cells/well on Day-0 (Figure 1A). On Day-1, (i.e., 24 h after plating) cells were incubated with various concentrations of H2O2 (Wako, Japan) as indicated, for 2 h, then rinsed with DMEM twice and incubated for 22 h. Cells treated with equivalent volumes of dH2O as H2O2 were considered as controls. This process was repeated on Day-2 and Day-3. Then cells were cultured until Day-9, splitting on Day-4 and Day-7, each time with the seeding density at 8.5 × 104 cells/well.
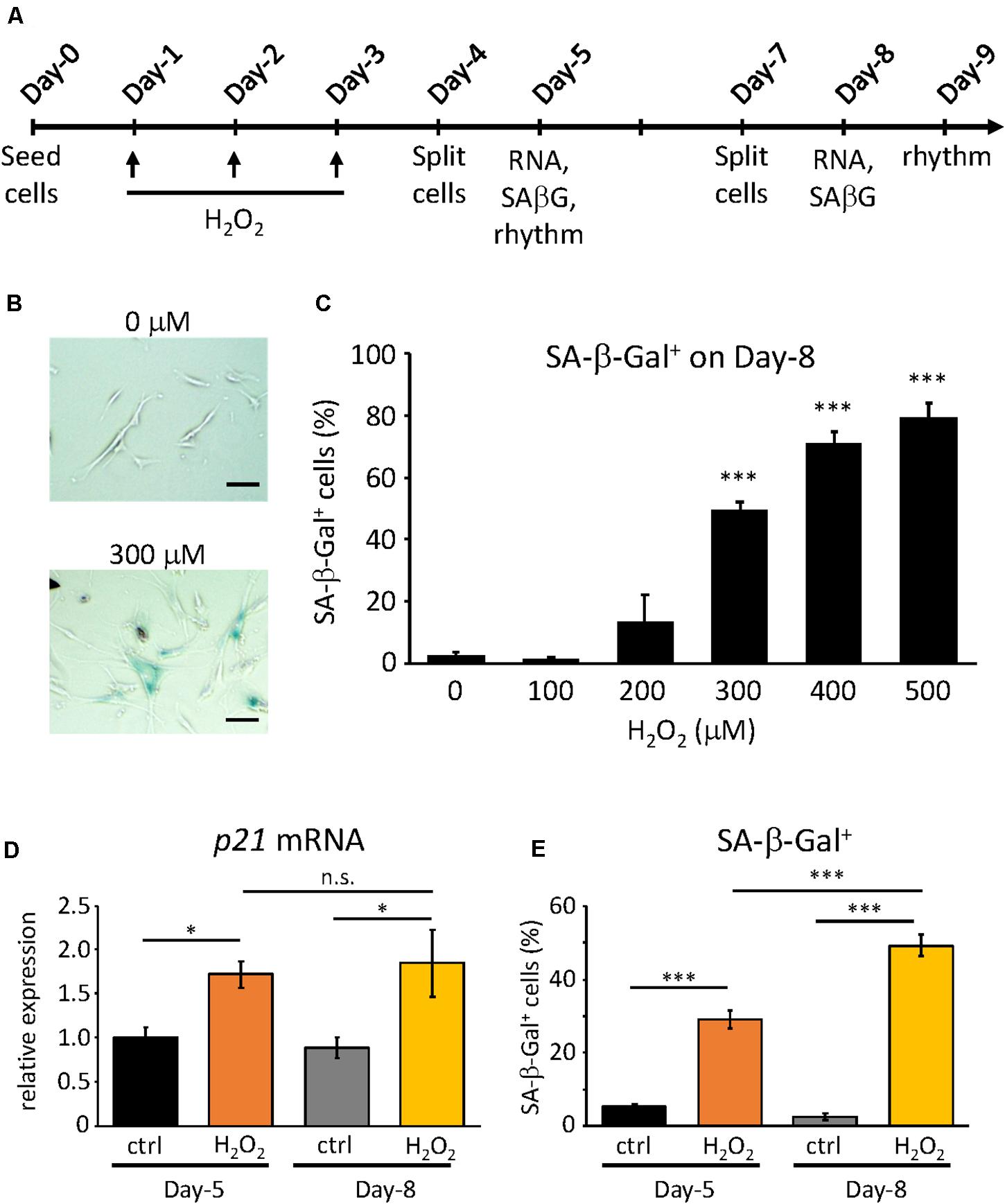
Figure 1. Confirmation of H2O2-induced premature senescence. (A) Scheme of this study. RNA, SA-β-Gal and rhythm mean RNA extraction, SA-β-Gal assay and real-time luciferase monitoring assay, respectively (B) Representative pictures of SA-β-Gal positive cells (blue) at Day-9 under control (upper) and 300 μM H2O2-treated conditions. Scale bars represent 100 μm (micrometer). (C) The percentage of SA-β-Gal positive cells were quantified at different concentrations. (D) p21CIP1 gene expressions under control or 300 μM H2O2-treated conditions at indicated days were analyzed by qPCR. The expression level of control condition at Day-5 was set to 1. (E) The percentage of SA-β-Gal positive cells under control or 300 μM H2O2-treated conditions at indicated days were quantified. n.s., not significant, ∗p < 0.05, ∗∗∗p < 0.001, by Student’s two-tailed t test.
Lentivirus production was performed as described previously (Ahmed et al., 2019). For infection of the target TIG-3 cells, cells in the Passage range of 25–29 in the previous study (Ahmed et al., 2019) were used. The culture medium was replaced with the lentivirus suspension supplemented with 8 μg/ml protamine sulfate (Nacalai Tesque, Japan). 24 h later the cells were washed with PBS once and cultured 2 more days with fresh medium. Infected cells were kept in liquid nitrogen until cells were subjected to experiments.
These methods were described preciously (Ahmed et al., 2019).
Values are reported as mean ± SEM. Statistical differences were determined by a Student’s two-tailed t test. Statistical significance is displayed as ∗p < 0.05, ∗∗p < 0.01, or ∗∗∗p < 0.001.
In our previous study, we obtained the proliferative and replicative senescent TIG-3 cells by serial passaging and found that senescent TIG-3 cells possess altered circadian clock properties with prolonged period and delayed phase (Ahmed et al., 2019). To address whether the senescence process triggers the alteration of circadian clock properties, we induced the oxidative stress-induced premature senescence using the proliferative cells, which consisted of cells in the Passage range of 25–29 in the previous study (Ahmed et al., 2019). In order to induce oxidative stress-induced premature senescence, we chose H2O2, as it is one of the most widely used stressors and also because it is thought of as a natural inducer of oxidative stress (Toussaint et al., 2000). To optimize the concentration of H2O2, we first exposed the cells to varying concentrations of H2O2 for 2 h, performing 3 consecutive H2O2 treatments every 24 h (Figure 1A). Senescent cells are known to exhibit a plethora of features such as enlarged, flattened morphology, increased senescence-associated β-galactosidase (SA-β-Gal) activity (Debacq-Chainiaux et al., 2009; Khaidizar et al., 2017), and increased expressions of cell cycle inhibitors (p16INK4a, p19ARF, and p21CIP1) (Stein et al., 1999; Krishnamurthy et al., 2004; Khaidizar et al., 2017). Hence on Day-8, the cells were checked for some of the aforementioned features. Starting at 300 μM, the cells appeared to be larger in size and flattened and showed significantly higher percentage of the SA-β-Gal-positive cells compared to the control cells (Figures 1B,C). Higher concentrations also gave correspondingly higher percentage of SA-β-Gal-positive cells, however, increasing number of cell deaths also occurred. As such, we determined that the optimum concentration that could induce significant SA-β-Gal activity was 300 μM (49.3% ± 2.9), and this percentage of SA-β-Gal-positive cells was in the same range to that found in the replicative senescent cells, as reported previously (Ahmed et al., 2019).
Next, we sought to characterize the process of the oxidative stress-induced premature senescence after the exposure of TIG-3 cells to H2O2. To this end, we checked the two senescence features, the cell cycle inhibitor p21 mRNA expression level and SA-β-Gal activity, at two time points i.e., on Day-5 and Day-8 at 300 μM of H2O2. On Day-5, both p21 mRNA expression (p = 0.02) and SA-β-Gal positive cells (p = 4.8 × 10–8) in the H2O2-treated cells were increased compared to the control cells (Figures 1D,E). As expected, on Day-8 both senescence features were significantly higher in H2O2-treated cells (p = 0.01 and 5.5 × 10–5 for p21 mRNA expression and SA-β-Gal positive cells, respectively). Intriguingly, p21 expression levels were comparable in H2O2-treated cells between Day-5 and Day-8 (p = 0.77, Figure 1D), whereas SA-β-Gal positive cells on Day-8 was significantly higher than that on Day-5 (p = 1.6 × 10–12, Figure 1E). These results indicate that the cells start ceasing proliferation almost immediately after exposure to the stressor H2O2, however, the development of the oxidative stress-induced premature senescence process is gradual, the intensity of which increases with time.
Next we assessed the changes in the circadian clock properties of the cells, both at Day-5 and Day-9, compared to the control cells. For this purpose, we used the TIG-3 cells lentivirally infected with the bmal1 promoter-driven luciferase gene (Brown et al., 2005). The infected cells were synchronized with dexamethasone and were subjected to real-time luciferase assay. As shown in Figure 2A, the circadian oscillation patterns of the control cells both on Day-5 and Day-9 were very close to each other (see Supplementary Figure 1 for raw data of oscillation patterns). Intriguingly, the oscillation pattern of H2O2-treated cells on Day-5, which have already shown senescent features (Figures 1D,E), was similar to those of control cells, suggesting that circadian clock is intact in Day-5 senescent cells. On the contrary, the oscillation pattern of the H2O2-treated cells on Day-9 was shifted to the right (Figure 2A), suggesting the alteration of the circadian clock i.e., a delay in their clock timings. In order to more precisely check the timings of the cells, the trough times of the cells on Day-5 and Day-9 were extracted. For the cells on Day-5, the 1st trough times were 28.87 ± 0.35 h and 29.57 ± 0.39 h for the control cells and H2O2-treated cells, respectively, with no statistically significant difference (p = 0.19, Figure 2B). For the cells on Day-9, the 1st trough times were 29.65 ± 0.17 h and 32.34 ± 0.11 h for the control cells and H2O2-treated cells, respectively, with statistically significant difference (p = 1.5 × 10–11). For the 2nd trough times, the control cells at Day-5 showed 53.15 ± 0.37 h while the H2O2-treated cells showed 54.78 ± 0.48 h, with statistically significant difference (p = 0.01). For the Day-9 cells, the 2nd trough times were 53.94 ± 0.23 h and 59.09 ± 0.60 h, for the control cells and H2O2-treated cells, respectively, with statistically significant difference (p = 4.4 × 10–8). We then compared intra-group trough times of the cells between Day-5 and Day-9. As expected, there were no differences of 1st and 2nd trough times in the control cells. On the other hand, for 1st trough times in H2O2-treated cells, the cells at Day-5 showed 29.57 ± 0.39 h while the cells at Day-9 showed 32.34 ± 0.11 h, with statistically significant difference (p = 1.9 × 10–6). Also, for 2nd trough times in H2O2-treated cells, the cells at Day-5 showed 54.78 ± 0.48 h and the cells at Day-9 showed 59.09 ± 0.60 h, with statistically significant difference (p = 1.0 × 10–5). These results indicate that the H2O2-treated cells on Day-9, with the higher level of the senescent features, consistently displayed the delayed trough timings, which is in accordance with the replicative senescent cells reported in our previous study (Ahmed et al., 2019). Meanwhile, the trough timings of H2O2-treated cells on Day-5, with the milder level of the senescent features, were similar to those of control cells, which suggests that the alteration of circadian clock by H2O2 on Day-5 is much milder than that on Day-9.
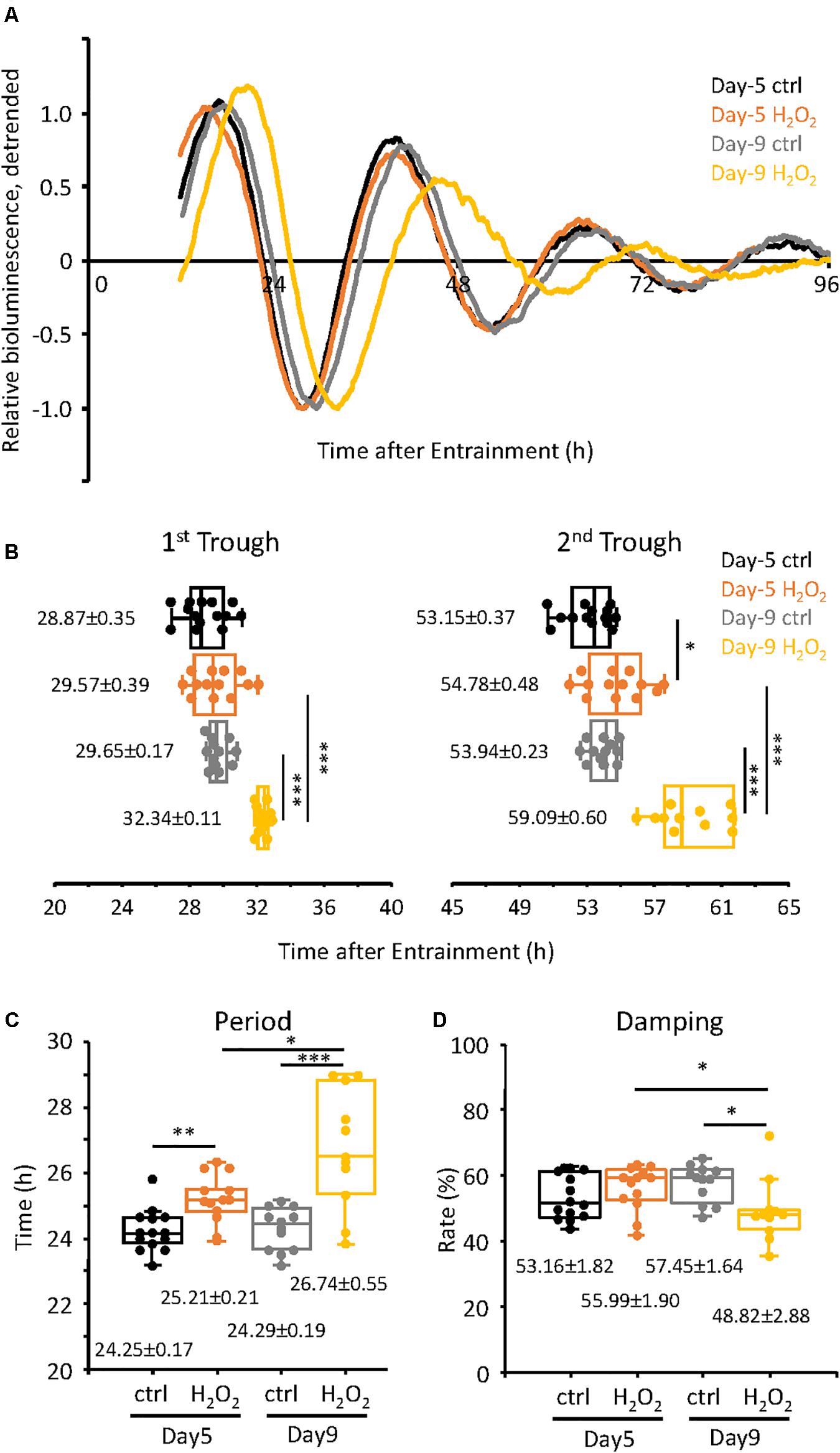
Figure 2. Alteration of circadian clock in H2O2-induced premature senescent cells at Day-9 was observed. (A) Relative oscillation patterns of luciferase of control and 300 μM H2O2-treated cells at Day-5 and -9 were monitored by using a real-time luciferase monitoring system. Lowest intensity of each sample was set to –1. (B) Box-whisker plots of trough-times are displayed. Values are mean ± SEM. (C,D) Box-whisker plots of period lengths (C) and damping ratio (D) in cells with control and H2O2-treated cells at Day-5 and -9 are displayed. Values are mean ± SEM. ∗p < 0.05, ∗∗p < 0.01, and ∗∗∗p < 0.001, by Student’s two-tailed t test.
We further checked the period and damping rate of the cells on Day-5 and Day-9 (Figures 2C,D). Period was calculated as time difference between 1st and 2nd trough times. For the cells on Day-5, the period length of the control cells was 24.25 ± 0.17 h and that of the H2O2-treated cells was 25.21 ± 0.21 h, p = 0.002, with a period extension of 0.96 h in the H2O2-treated cells. For the cells at Day-9, the period of the control cells was 24.29 ± 0.19 h while that of the H2O2-treated cells was 26.74 ± 0.55 h, p = 2.8 × 10–4, with a period extension of 2.45 h. Furthermore, the period of the H2O2-treated cells on Day-9 was 1.53 h longer than that on Day-5, with statistically significant different (p = 0.011). In case of the damping rate of the circadian oscillation patterns of the cells, Day-5 cells did not show any significant difference in their damping rates, for both the control and H2O2-treated cells (Figure 2D). For the cells of Day-9, the oscillation pattern of the H2O2-treated cells damped down more than the control cells, p = 0.015. Also, the damping of the H2O2-treated cells on Day-9 damped down more than that on Day-5, p = 0.044. Collectively, the period changes and damping rates suggest that the H2O2-treated cells on Day-9 display the higher intensity alterations of the circadian clock properties, although period changes start with the initiation of the process of oxidative stress-induced premature senescence.
To confirm the above results, we also analyzed the data of Figure 2 mathematically using the Cosinor software (Supplementary Figure 2). For the period, control cells on Day-5 had the period of 23.70 ± 0.21 h while the H2O2-treated cells had period of 25.99 ± 0.14 h, p = 2.6 × 10–9; for Day-9 cells, the period of the control cells was 25.03 ± 0.16 h while that of the H2O2-treated cells was 26.85 ± 0.19 h, p = 2.5 × 10–7 (Supplementary Figure 2A), both of which are consistent with the manual extraction of the period data (Figure 2). Again, the period of H2O2-treated cells on Day-9 was significantly longer than that on Day-5, p = 0.001. In case of the acrophase, on Day-5, the control cells had an acrophase of −320.00 ± 5.76 while the H2O2-treated cells had an acrophase of −317.69 ± 5.00, with no statistically significant difference (p = 0.77, Supplementary Figure 2B), indicating that there is no phase delay between the cells at the beginning of the oxidative senescence development. On Day-9, the control cells had an acrophase of −322.25 ± 2.36, which was comparable to those of Day-5, while the H2O2-treated cells had an acrophase of −356.64 ± 1.06, p = 2.0 × 10–11 (Supplementary Figure 2B), indicating phase delay on Day-9 in accordance with Figure 2B. Also, the acrophase of H2O2-treated cells on Day-9 was significantly different from that on Day-5, p = 0.001.
3xH2O2 treatment induced the initiation of cellular senescence easily, however, it altered all the circadian clock properties gradually. Therefore, we conclude that the circadian changes observed in the H2O2-treated cells are a result of the oxidative stress-induced premature senescence of the cells, not simply an effect of the H2O2 on the cells per se.
Finally, endogenous circadian gene expressions in H2O2-induced senescent cells on Day-9 were analyzed. Similar to our previous results in replicative senescent cells (Ahmed et al., 2019), PER1, PER2, and CRY1 mRNAs were downregulated in senescent cells, however, CRY2, REV-ERBa, and BMAL1 mRNAs were comparable to control non-senescent cells (Figure 3). These results suggest that not only circadian phenotypes, but also molecular regulations for circadian clock are similar, irrespective of the type of cellular senescence.
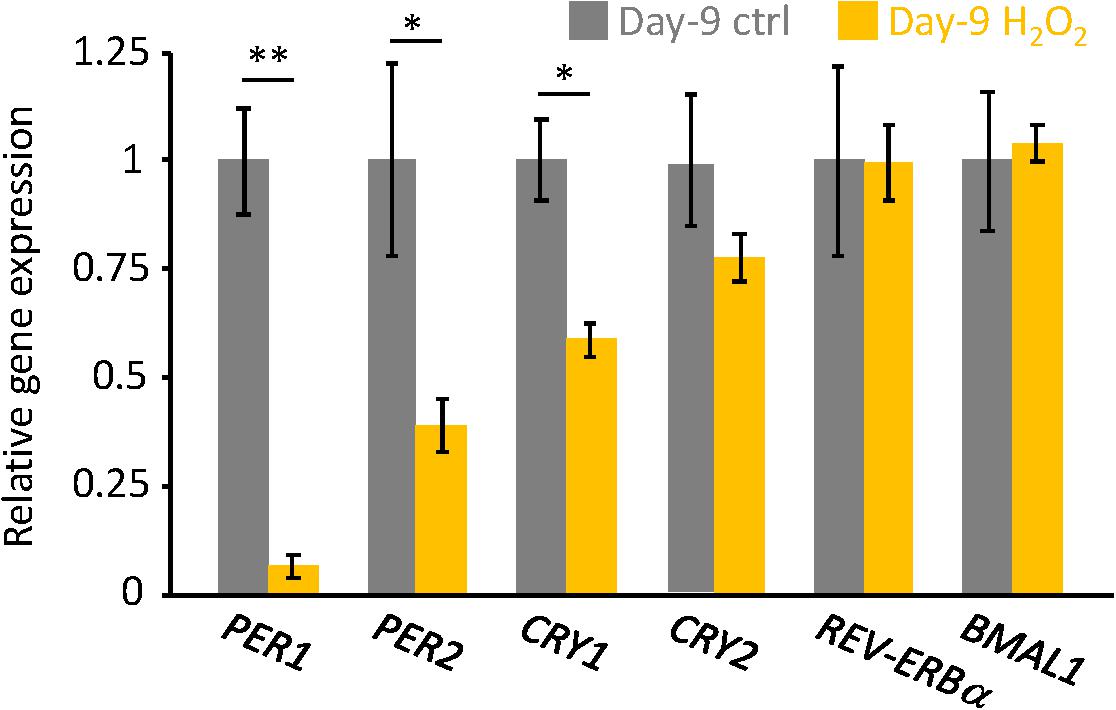
Figure 3. The endogenous circadian clock genes expression level was downregulated in the senescent cells. PER1, PER2, CRY1, CRY2, REV-ERBa, and BMAL1 mRNAs in unsynchronized cells were analyzed by qPCR. Each sample was normalized by 18S rRNA. Expression levels of each gene in control cells were set to 1. ∗p < 0.05, ∗∗∗p < 0.01, by Student’s two-tailed t test.
In this study, we revealed that the oxidative stress-induced premature senescence triggers the alteration of circadian clock properties, that is, the delayed phase and period extension. Also, we have recently reported that the period and phase of circadian clock in the replicative senescent cells was prolonged and delayed compared to the proliferative cells, respectively (Ahmed et al., 2019). Based on our findings, we propose that cellular senescence induces the period extension and delayed phase of circadian clock properties by similar molecular mechanisms, irrespective of the replicative senescence or the oxidative stress-induced premature senescence.
In aged organisms, in addition to the replicative senescent cells, the stress-induced premature senescent cells occupy a major portion of the senescent cells (Campisi, 2005; Khapre et al., 2011). Oxidative stress is one of the strongest contributors of stress-induced premature senescence and is likely one of the major mediators of stress-induced premature senescence in vivo (Chen et al., 1995; Campisi, 2005; Khapre et al., 2011). Interestingly, several studies from model animals and humans have demonstrated that aging can also lead to alteration of the circadian clock (Pittendrigh and Daan, 1974; Witting et al., 1994; Valentinuzzi et al., 1997; Van Someren, 2000; Davidson et al., 2008; Sellix et al., 2012; Mattis and Sehgal, 2016). These evidence suggest that the attenuation of circadian clock functions with aging is in accordance with the accumulation of the senescent cells in vivo. Senolytic drugs (Chang et al., 2016; Yosef et al., 2016; Lehmann et al., 2017) which selectively eliminate senescent cells, or transgenic mice, such as INK-ATTAC (Baker et al., 2011) and p16-3MR mice (Demaria et al., 2014), in which senescent cells can be selectively eliminated in an inducible fashion, will be good strategies to address this hypothesis.
As already discussed in our previous study (Ahmed et al., 2019), the altered circadian clock properties have also been reported by Nakamura et al. (2015) using ex vivo SCN tissue of old mice. Compared to the consistent results from cellular and tissue levels, results at the organismal level have been controversial, some reports demonstrate prolonged period (Valentinuzzi et al., 1997), but others show shortened period (Pittendrigh and Daan, 1974; Witting et al., 1994). Aging phenotype is the result of complex intra- and inter-organ communications and individual contributions of different factors to total aging phenotype are still unknown. This is probably the reason for the controversial reports at the organismal level. Further investigations to unravel individual factors affecting total aging phenotype will be required.
We concluded in this study that 3×H2O2-treated cells on Day-5 have already entered the senescent phase, because of the high expression and level of p21 mRNA and SA-b-Gal activity, respectively (Figures 1D,E), and H2O2-treated cells on Day-9 were more maturated. Meanwhile the alteration of circadian clock properties in H2O2-treated cells on Day-5 occurred only in terms of the period prolongation, and on Day-9 the period was much longer than that on Day-5. Intriguingly, phase and damping rate were altered only on Day-9, suggesting that molecular mechanisms of the period prolongation and delayed-phase/damping are independent. These results also suggest that the molecular mechanisms in circadian period regulations are vulnerable to cellular senescence, while the molecular mechanisms in circadian phase regulations are more robust than those in period regulations. Compared to our 3xH2O2 treatment, acute single H2O2 treatment with high dose has been reported to alter circadian clock properties; H2O2 treatment resets circadian clock mediated by the dimerization of BMAL1 and HSF1 (Tamaru et al., 2013), induces phase changes of circadian clock in mouse embryonic fibroblast (MEF) cells and mouse peripheral tissues (Tahara et al., 2016), increases the amplitude of circadian clock by activating NRF2 following Cry1 expression in stable Per2:Luc reporter MEF cells (Wible et al., 2018), elicits phase-dependent PER2 degradation and circadian phase shifts in mouse fibroblasts (Putker et al., 2018), and shortens the circadian period by downregulating Rev-erva/b mRNAs via the activation of PRX2/STAT3 pathway in stable Bmal1:dLuc reporter NIH3T3 cells (Ji et al., 2019). These circadian phenotypes triggered by acute H2O2 are different from our current results, thereby indicating that the circadian phenotypes observed in our study are a result of the oxidative stress-induced premature senescence of the cells, not simply an effect of the H2O2 on circadian clock per se.
Li et al. (2020) have recently reported that increased non-genetic variation in gene expression predominantly drives circadian period prolongation in clonal cell lines (Li et al., 2020). Our studies demonstrated that variations in trough times and periods are larger in replicative/stress-induced premature senescent cells, compared to those in proliferative/control cells. Meanwhile, senescent cells are not homogeneous, they are heterogenous mixture of cells, for example, the percentage of SA-b-Gal positive cells was not 100% (Figure 1D). These data support that variation in circadian gene expression among senescent cells is greater. Furthermore, aging has been associated with increased stochastic transcriptional noise (Bahar et al., 2006; Enge et al., 2017; Martinez-Jimenez et al., 2017; Tang et al., 2019), therefore, increased transcriptional noise in senescent cells might be one of the causes to induce prolonged circadian period. Analyses of circadian period in single cells and single-cell RNA-sequence will provide an answer for this possibility.
Senescent cells are metabolically active, and increase in the AMP/ATP ratio and decrease in NAD+ amount have been reported during senescence (James et al., 2015; Khaidizar et al., 2017). Increase in the AMP/ATP ratio promotes AMP-activated protein kinase (AMPK), which acts as a sensor of the reduced energetic state and further activates catabolic pathways while inhibiting anabolic ones (Hardie, 2003; Garcia and Shaw, 2017). Meanwhile it has been reported that mTOR, which is an intracellular nutrient sensor for high cellular energy state and associated with autophagy, is also upregulated during senescence (Herranz et al., 2015; Laberge et al., 2015; Nacarelli and Sell, 2017). Decrease in NAD+ amount attenuates enzymatic activities of NAD+-dependent enzymes, such as sirtuin family deacetylase (SIRT1-7) and poly (ADP-ribose) polymerases (PARPs) (Imai and Guarente, 2014, 2016; Schultz and Sinclair, 2016). Many of aforementioned signaling molecules are reported to regulate circadian clock properties. AMPK is a rhythmically expressed kinase and phosphorylates CK1ε, resulting in enhanced phosphorylation and degradation of PER2 (Um et al., 2007; Sahar and Sassone-Corsi, 2012) and CRY1 (Lamia et al., 2009; Sahar and Sassone-Corsi, 2012; Jordan and Lamia, 2013). AMPK activation by AMPK agonist, AICAR, or glucose deprivation, increased the circadian period and decreased the amplitude (Lamia et al., 2009), which are consistent with our finding in senescent cells, although another AMPK agonist metformin shortened circadian period (Um et al., 2007). mTOR perturbation, such as RNAi knockdown or mTOR inhibitors, lengthened circadian period in fibroblast, SCN, and animal behaviors (Zhang et al., 2009; Ramanathan et al., 2018), these reports show opposite effects to our findings. NAD+ shows rhythmic 24 h oscillation and post-translationally modifies histone H3, BMAL1, PER2 and CLOCK by SIRT1 and PARP1 (Nakahata et al., 2008; Ramsey et al., 2009; Asher et al., 2010). Decrease in NAD+ by FK866 treatment amplified E-box-regulated circadian genes, such as per2 and dbp mRNAs (Nakahata et al., 2009). Current study demonstrated that the amplitude of circadian oscillation driven by bmal1-promoter was damped more in senescent cells (Figure 2D), which is probably due to the increase in E-box-regulated circadian gene, rev-erb, the repressor for bmal1 gene regulation. Intriguingly, it has been demonstrated that H2O2 decreases intracellular NAD+ in some primary cells, suggesting that senescent cells in our study also possesses low NAD+ (Furukawa et al., 2007; Han et al., 2016). Evidences mentioned here imply that altered signaling pathways during senescence affects circadian clock properties, however, as far as we know, molecular connections between cellular senescence and circadian clock remain largely uncovered. Therefore, further investigations addressing this will be required to understand, maintain and cure the circadian clock mechanisms in the elderly.
In summary, our results indicate that cellular senescence alters the circadian clock, irrespective of the type of cellular senescence. In aged individuals, disruption of the circadian clock functions has been associated with many age-related diseases, however, the underlying cause of this disruption of the circadian clock was largely unknown. Our novel findings, therefore, open up new avenues to investigate the underlying mechanisms that lead to the disruption of the circadian clock function in aged organisms.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
RA performed experiments and drafted the manuscript. YN performed experiments, designed the overall approach, coordinated the study, and wrote the manuscript. KS contributed to coordination of the study. YB contributed to design and coordination of the study. All authors contributed to the article and approved the submitted version.
This work was supported by JSPS KAKENHI Grant Number 17K08569 (YN).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fnins.2021.638122/full#supplementary-material
Ahmed, R., Ashimori, A., Iwamoto, S., Matsui, T., Nakahata, Y., and Bessho, Y. (2019). Replicative senescent human cells possess altered circadian clocks with a prolonged period and delayed peak-time. Aging (Albany Ny) 11, 950–973. doi: 10.18632/aging.101794
Asher, G., Reinke, H., Altmeyer, M., Gutierrez-Arcelus, M., Hottiger, M. O., and Schibler, U. (2010). Poly(ADP-ribose) polymerase 1 participates in the phase entrainment of circadian clocks to feeding. Cell 142, 943–953. doi: 10.1016/j.cell.2010.08.016
Baar, M. P., Brandt, R. M., Putavet, D. A., Klein, J. D., Derks, K. W., Bourgeois, B. R., et al. (2017). Targeted Apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 169, 132–147.e116.
Bahar, R., Hartmann, C. H., Rodriguez, K. A., Denny, A. D., Busuttil, R. A., Dolle, M. E., et al. (2006). Increased cell-to-cell variation in gene expression in ageing mouse heart. Nature 441, 1011–1014. doi: 10.1038/nature04844
Baker, D. J., Childs, B. G., Durik, M., Wijers, M. E., Sieben, C. J., Zhong, J., et al. (2016). Naturally occurring p16(Ink4a)-positive cells shorten healthy lifespan. Nature 530, 184–189. doi: 10.1038/nature16932
Baker, D. J., Wijshake, T., Tchkonia, T., Lebrasseur, N. K., Childs, B. G., Van De Sluis, B., et al. (2011). Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479, 232–236. doi: 10.1038/nature10600
Bass, J., and Lazar, M. A. (2016). Circadian time signatures of fitness and disease. Science 354, 994–998. doi: 10.1126/science.aah4965
Bavik, C., Coleman, I., Dean, J. P., Knudsen, B., Plymate, S., and Nelson, P. S. (2006). The gene expression program of prostate fibroblast senescence modulates neoplastic epithelial cell proliferation through paracrine mechanisms. Cancer Res. 66, 794–802. doi: 10.1158/0008-5472.can-05-1716
Bhatia, B., Multani, A. S., Patrawala, L., Chen, X., Calhoun-Davis, T., Zhou, J., et al. (2008). Evidence that senescent human prostate epithelial cells enhance tumorigenicity: cell fusion as a potential mechanism and inhibition by p16INK4a and hTERT. Int. J. Cancer 122, 1483–1495. doi: 10.1002/ijc.23222
Brown, S. A., Fleury-Olela, F., Nagoshi, E., Hauser, C., Juge, C., Meier, C. A., et al. (2005). The period length of fibroblast circadian gene expression varies widely among human individuals. PLoS Biol. 3:e338. doi: 10.1371/journal.pbio.0030338
Bussian, T. J., Aziz, A., Meyer, C. F., Swenson, B. L., Van Deursen, J. M., and Baker, D. J. (2018). Clearance of senescent glial cells prevents tau-dependent pathology and cognitive decline. Nature 562, 578–582. doi: 10.1038/s41586-018-0543-y
Campisi, J. (2005). Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120, 513–522. doi: 10.1016/j.cell.2005.02.003
Campisi, J., Andersen, J. K., Kapahi, P., and Melov, S. (2011). Cellular senescence: a link between cancer and age-related degenerative disease? Semin. Cancer Biol. 21, 354–359.
Castro-Vega, L. J., Jouravleva, K., Ortiz-Montero, P., Liu, W. Y., Galeano, J. L., Romero, M., et al. (2015). The senescent microenvironment promotes the emergence of heterogeneous cancer stem-like cells. Carcinogenesis 36, 1180–1192. doi: 10.1093/carcin/bgv101
Chang, J., Wang, Y., Shao, L., Laberge, R. M., Demaria, M., Campisi, J., et al. (2016). Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nat. Med. 22, 78–83. doi: 10.1038/nm.4010
Chen, Q., Fischer, A., Reagan, J. D., Yan, L. J., and Ames, B. N. (1995). Oxidative DNA damage and senescence of human diploid fibroblast cells. Proc. Natl. Acad. Sci. U.S.A. 92, 4337–4341. doi: 10.1073/pnas.92.10.4337
Childs, B. G., Baker, D. J., Wijshake, T., Conover, C. A., Campisi, J., and Van Deursen, J. M. (2016). Senescent intimal foam cells are deleterious at all stages of atherosclerosis. Science 354, 472–477. doi: 10.1126/science.aaf6659
Childs, B. G., Li, H., and Van Deursen, J. M. (2018). Senescent cells: a therapeutic target for cardiovascular disease. J. Clin. Invest. 128, 1217–1228. doi: 10.1172/jci95146
Cho, J. H., Kim, E. C., Son, Y., Lee, D. W., Park, Y. S., Choi, J. H., et al. (2020). CD9 induces cellular senescence and aggravates atherosclerotic plaque formation. Cell Death Differ. 27, 2681–2696. doi: 10.1038/s41418-020-0537-9
Davidson, A. J., Yamazaki, S., Arble, D. M., Menaker, M., and Block, G. D. (2008). Resetting of central and peripheral circadian oscillators in aged rats. Neurobiol. Aging 29, 471–477. doi: 10.1016/j.neurobiolaging.2006.10.018
Debacq-Chainiaux, F., Erusalimsky, J. D., Campisi, J., and Toussaint, O. (2009). Protocols to detect senescence-associated beta-galactosidase (SA-betagal) activity, a biomarker of senescent cells in culture and in vivo. Nat. Protoc. 4, 1798–1806. doi: 10.1038/nprot.2009.191
Demaria, M., Ohtani, N., Youssef, S. A., Rodier, F., Toussaint, W., Mitchell, J. R., et al. (2014). An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev. Cell 31, 722–733. doi: 10.1016/j.devcel.2014.11.012
Enge, M., Arda, H. E., Mignardi, M., Beausang, J., Bottino, R., Kim, S. K., et al. (2017). Single-cell analysis of human pancreas reveals transcriptional signatures of aging and somatic mutation patterns. Cell 171:e314.
Furukawa, A., Tada-Oikawa, S., Kawanishi, S., and Oikawa, S. (2007). H2O2 accelerates cellular senescence by accumulation of acetylated p53 via decrease in the function of SIRT1 by NAD+ depletion. Cell Physiol. Biochem. 20, 45–54.
Garcia, D., and Shaw, R. J. (2017). AMPK: mechanisms of cellular energy sensing and restoration of metabolic balance. Mol. Cell 66, 789–800. doi: 10.1016/j.molcel.2017.05.032
Han, X., Tai, H., Wang, X., Wang, Z., Zhou, J., Wei, X., et al. (2016). AMPK activation protects cells from oxidative stress-induced senescence via autophagic flux restoration and intracellular NAD(+) elevation. Aging Cell 15, 416–427. doi: 10.1111/acel.12446
Hardie, D. G. (2003). Minireview: the AMP-activated protein kinase cascade: the key sensor of cellular energy status. Endocrinology 144, 5179–5183. doi: 10.1210/en.2003-0982
Hashimoto, M., Asai, A., Kawagishi, H., Mikawa, R., Iwashita, Y., Kanayama, K., et al. (2016). Elimination of p19ARF-expressing cells enhances pulmonary function in mice. JCI Insight 1:e87732.
Hernandez-Segura, A., De Jong, T. V., Melov, S., Guryev, V., Campisi, J., and Demaria, M. (2017). Unmasking transcriptional heterogeneity in senescent cells. Curr. Biol. 27, 2652–2660.e2654.
Herranz, N., Gallage, S., Mellone, M., Wuestefeld, T., Klotz, S., Hanley, C. J., et al. (2015). mTOR regulates MAPKAPK2 translation to control the senescence-associated secretory phenotype. Nat. Cell Biol. 17, 1205–1217. doi: 10.1038/ncb3225
Honma, S. (2018). The mammalian circadian system: a hierarchical multi-oscillator structure for generating circadian rhythm. J. Physiol. Sci. 68, 207–219. doi: 10.1007/s12576-018-0597-5
Houssaini, A., Breau, M., Kebe, K., Abid, S., Marcos, E., Lipskaia, L., et al. (2018). mTOR pathway activation drives lung cell senescence and emphysema. JCI Insight 3:e93203.
Imai, S., and Guarente, L. (2014). NAD+ and sirtuins in aging and disease. Trends Cell Biol. 24, 464–471. doi: 10.1016/j.tcb.2014.04.002
Imai, S., and Guarente, L. (2016). It takes two to tango: NAD+ and sirtuins in aging/longevity control. npj Aging Mechanisms Dis. 2:16017.
James, E. L., Michalek, R. D., Pitiyage, G. N., De Castro, A. M., Vignola, K. S., Jones, J., et al. (2015). Senescent human fibroblasts show increased glycolysis and redox homeostasis with extracellular metabolomes that overlap with those of irreparable DNA damage, aging, and disease. J. Proteome Res. 14, 1854–1871. doi: 10.1021/pr501221g
Jeon, O. H., Kim, C., Laberge, R. M., Demaria, M., Rathod, S., Vasserot, A. P., et al. (2017). Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nat. Med. 23, 775–781. doi: 10.1038/nm.4324
Jeon, O. H., Wilson, D. R., Clement, C. C., Rathod, S., Cherry, C., Powell, B., et al. (2019). Senescence cell-associated extracellular vesicles serve as osteoarthritis disease and therapeutic markers. JCI Insight 4:e125019.
Ji, G., Lv, K., Chen, H., Wang, Y., Zhang, Y., Li, Y., et al. (2019). Hydrogen peroxide modulates clock gene expression via PRX2-STAT3-REV-ERBalpha/beta pathway. Free Radic. Biol. Med. 145, 312–320. doi: 10.1016/j.freeradbiomed.2019.09.036
Jordan, S. D., and Lamia, K. A. (2013). AMPK at the crossroads of circadian clocks and metabolism. Mol. Cell Endocrinol. 366, 163–169. doi: 10.1016/j.mce.2012.06.017
Khaidizar, F. D., Nakahata, Y., Kume, A., Sumizawa, K., Kohno, K., Matsui, T., et al. (2017). Nicotinamide phosphoribosyltransferase delays cellular senescence by upregulating SIRT1 activity and antioxidant gene expression in mouse cells. Genes Cells 22, 982–992. doi: 10.1111/gtc.12542
Khapre, R. V., Kondratova, A. A., Susova, O., and Kondratov, R. V. (2011). Circadian clock protein BMAL1 regulates cellular senescence in vivo. Cell Cycle 10, 4162–4169. doi: 10.4161/cc.10.23.18381
Krishnamurthy, J., Torrice, C., Ramsey, M. R., Kovalev, G. I., Al-Regaiey, K., Su, L., et al. (2004). Ink4a/Arf expression is a biomarker of aging. J. Clin. Invest. 114, 1299–1307. doi: 10.1172/jci22475
Laberge, R. M., Sun, Y., Orjalo, A. V., Patil, C. K., Freund, A., Zhou, L., et al. (2015). MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat. Cell Biol. 17, 1049–1061. doi: 10.1038/ncb3195
Lamia, K. A., Sachdeva, U. M., Ditacchio, L., Williams, E. C., Alvarez, J. G., Egan, D. F., et al. (2009). AMPK regulates the circadian clock by cryptochrome phosphorylation and degradation. Science 326:5951.
Lehmann, M., Korfei, M., Mutze, K., Klee, S., Skronska-Wasek, W., Alsafadi, H. N., et al. (2017). Senolytic drugs target alveolar epithelial cell function and attenuate experimental lung fibrosis ex vivo. Eur. Respir. J. 50:1602367. doi: 10.1183/13993003.02367-2016
Li, Y., Shan, Y., Desai, R. V., Cox, K. H., Weinberger, L. S., and Takahashi, J. S. (2020). Noise-driven cellular heterogeneity in circadian periodicity. Proc. Natl. Acad. Sci. U.S.A. 117, 10350–10356. doi: 10.1073/pnas.1922388117
Liu, D., and Hornsby, P. J. (2007). Senescent human fibroblasts increase the early growth of xenograft tumors via matrix metalloproteinase secretion. Cancer Res. 67, 3117–3126. doi: 10.1158/0008-5472.can-06-3452
Maciel-Baron, L. A., Morales-Rosales, S. L., Aquino-Cruz, A. A., Triana-Martinez, F., Galvan-Arzate, S., Luna-Lopez, A., et al. (2016). Senescence associated secretory phenotype profile from primary lung mice fibroblasts depends on the senescence induction stimuli. Age (Dordr) 38:26.
Martinez-Jimenez, C. P., Eling, N., Chen, H. C., Vallejos, C. A., Kolodziejczyk, A. A., Connor, F., et al. (2017). Aging increases cell-to-cell transcriptional variability upon immune stimulation. Science 355, 1433–1436. doi: 10.1126/science.aah4115
Mattis, J., and Sehgal, A. (2016). Circadian rhythms, sleep, and disorders of aging. Trends Endocrinol. Metab. 27, 192–203. doi: 10.1016/j.tem.2016.02.003
Nacarelli, T., and Sell, C. (2017). Targeting metabolism in cellular senescence, a role for intervention. Mol. Cell Endocrinol. 455, 83–92. doi: 10.1016/j.mce.2016.08.049
Nakahata, Y., Kaluzova, M., Grimaldi, B., Sahar, S., Hirayama, J., Chen, D., et al. (2008). The NAD+-dependent deacetylase SIRT1 modulates CLOCK-mediated chromatin remodeling and circadian control. Cell 134, 329–340. doi: 10.1016/j.cell.2008.07.002
Nakahata, Y., Sahar, S., Astarita, G., Kaluzova, M., and Sassone-Corsi, P. (2009). Circadian control of the NAD+ salvage pathway by CLOCK-SIRT1. Science 324, 654–657. doi: 10.1126/science.1170803
Nakahata, Y., Yasukawa, S., Khaidizar, F. D., Shimba, S., Matsui, T., and Bessho, Y. (2018). Bmal1-deficient mouse fibroblast cells do not provide premature cellular senescence in vitro. Chronobiol. Int. 35, 730–738. doi: 10.1080/07420528.2018.1430038
Nakamura, T. J., Nakamura, W., Tokuda, I. T., Ishikawa, T., Kudo, T., Colwell, C. S., et al. (2015). Age-related changes in the circadian system unmasked by constant conditions. eNeuro 2:ENEURO.0064-15.2015.
Nakao, M., Tanaka, H., and Koga, T. (2020). Cellular senescence variation by metabolic and epigenomic remodeling. Trends Cell Biol. 30, 919–922. doi: 10.1016/j.tcb.2020.08.009
Ortiz-Montero, P., Londono-Vallejo, A., and Vernot, J. P. (2017). Senescence-associated IL-6 and IL-8 cytokines induce a self- and cross-reinforced senescence/inflammatory milieu strengthening tumorigenic capabilities in the MCF-7 breast cancer cell line. Cell Commun. Signal. 15:17.
Parrinello, S., Coppe, J. P., Krtolica, A., and Campisi, J. (2005). Stromal-epithelial interactions in aging and cancer: senescent fibroblasts alter epithelial cell differentiation. J. Cell Sci. 118, 485–496. doi: 10.1242/jcs.01635
Partch, C. L., Green, C. B., and Takahashi, J. S. (2014). Molecular architecture of the mammalian circadian clock. Trends Cell Biol. 24, 90–99. doi: 10.1016/j.tcb.2013.07.002
Pittendrigh, C. S., and Daan, S. (1974). Circadian oscillations in rodents: a systematic increase of their frequency with age. Science 186, 548–550. doi: 10.1126/science.186.4163.548
Putker, M., Crosby, P., Feeney, K. A., Hoyle, N. P., Costa, A. S. H., Gaude, E., et al. (2018). Mammalian circadian period, but not phase and amplitude, is robust against redox and metabolic perturbations. Antioxid. Redox Signal. 28, 507–520. doi: 10.1089/ars.2016.6911
Ramanathan, C., Kathale, N. D., Liu, D., Lee, C., Freeman, D. A., Hogenesch, J. B., et al. (2018). mTOR signaling regulates central and peripheral circadian clock function. PLoS Genet. 14:e1007369. doi: 10.1371/journal.pgen.1007369
Ramsey, K. M., Yoshino, J., Brace, C. S., Abrassart, D., Kobayashi, Y., Marcheva, B., et al. (2009). Circadian clock feedback cycle through NAMPT-mediated NAD+ biosynthesis. Science 324, 651–654. doi: 10.1126/science.1171641
Sahar, S., and Sassone-Corsi, P. (2012). Regulation of metabolism: the circadian clock dictates the time. Trends Endocrinol. Metab. 23, 1–8. doi: 10.1016/j.tem.2011.10.005
Schafer, M. J., White, T. A., Iijima, K., Haak, A. J., Ligresti, G., Atkinson, E. J., et al. (2017). Cellular senescence mediates fibrotic pulmonary disease. Nat. Commun. 8:14532.
Schultz, M. B., and Sinclair, D. A. (2016). Why NAD+ declines during aging: it’s destroyed. Cell Metab. 23, 965–966. doi: 10.1016/j.cmet.2016.05.022
Sellix, M. T., Evans, J. A., Leise, T. L., Castanon-Cervantes, O., Hill, D. D., Delisser, P., et al. (2012). Aging differentially affects the re-entrainment response of central and peripheral circadian oscillators. J. Neurosci. 32, 16193–16202. doi: 10.1523/jneurosci.3559-12.2012
Stein, G. H., Drullinger, L. F., Soulard, A., and Dulić, V. (1999). Differential roles for cyclin-dependent kinase inhibitors p21 and p16 in the mechanisms of senescence and differentiation in human fibroblasts. Mol. Cell Biol. 19, 2109–2117. doi: 10.1128/mcb.19.3.2109
Tahara, Y., Yokota, A., Shiraishi, T., Yamada, S., Haraguchi, A., Shinozaki, A., et al. (2016). In vitro and in vivo phase changes of the mouse circadian clock by oxidative stress. J. Circadian Rhythms 14, 4.
Takahashi, J. S. (2017). Transcriptional architecture of the mammalian circadian clock. Nat. Rev. Genet. 18, 164–179. doi: 10.1038/nrg.2016.150
Tamaru, T., Hattori, M., Ninomiya, Y., Kawamura, G., Vares, G., Honda, K., et al. (2013). ROS stress resets circadian clocks to coordinate pro-survival signals. PLoS One 8:e82006. doi: 10.1371/journal.pone.0082006
Tang, H., Geng, A., Zhang, T., Wang, C., Jiang, Y., and Mao, Z. (2019). Single senescent cell sequencing reveals heterogeneity in senescent cells induced by telomere erosion. Protein Cell 10, 370–375. doi: 10.1007/s13238-018-0591-y
Toussaint, O., Dumont, P., Dierick, J. F., Pascal, T., Frippiat, C., Chainiaux, F., et al. (2000). Stress-induced premature senescence. Essence of life, evolution, stress, and aging. Ann. N. Y. Acad. Sci. 908, 85–98. doi: 10.1111/j.1749-6632.2000.tb06638.x
Um, J. H., Yang, S., Yamazaki, S., Kang, H., Viollet, B., Foretz, M., et al. (2007). Activation of 5′-AMP-activated kinase with diabetes drug metformin induces casein kinase Iepsilon (CKIepsilon)-dependent degradation of clock protein mPer2. J. Biol. Chem. 282, 20794–20798. doi: 10.1074/jbc.c700070200
Valentinuzzi, V. S., Scarbrough, K., Takahashi, J. S., and Turek, F. W. (1997). Effects of aging on the circadian rhythm of wheel-running activity in C57BL/6 mice. Am. J. Physiol. 273, R1957–R1964.
Van Someren, E. J. W. (2000). Circadian and sleep disturbances in the elderly. Exp. Gerontol. 35, 1229–1237. doi: 10.1016/s0531-5565(00)00191-1
Wang, J. C., and Bennett, M. (2012). Aging and atherosclerosis: mechanisms, functional consequences, and potential therapeutics for cellular senescence. Circ. Res. 111, 245–259. doi: 10.1161/circresaha.111.261388
Wible, R. S., Ramanathan, C., Sutter, C. H., Olesen, K. M., Kensler, T. W., Liu, A. C., et al. (2018). NRF2 regulates core and stabilizing circadian clock loops, coupling redox and timekeeping in Mus musculus. eLife 7:e31656.
Witting, W., Mirmiran, M., Bos, N. P., and Swaab, D. F. (1994). The effect of old age on the free-running period of circadian rhythms in rat. Chronobiol. Int. 11, 103–112. doi: 10.3109/07420529409055896
Xu, M., Bradley, E. W., Weivoda, M. M., Hwang, S. M., Pirtskhalava, T., Decklever, T., et al. (2017). Transplanted senescent cells induce an osteoarthritis-like condition in mice. J. Gerontol. A Biol. Sci. Med. Sci. 72, 780–785.
Yosef, R., Pilpel, N., Tokarsky-Amiel, R., Biran, A., Ovadya, Y., Cohen, S., et al. (2016). Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL. Nat. Commun. 7:11190.
Zhang, E. E., Liu, A. C., Hirota, T., Miraglia, L. J., Welch, G., Pongsawakul, P. Y., et al. (2009). A genome-wide RNAi screen for modifiers of the circadian clock in human cells. Cell 139, 199–210. doi: 10.1016/j.cell.2009.08.031
Zhang, P., Kishimoto, Y., Grammatikakis, I., Gottimukkala, K., Cutler, R. G., Zhang, S., et al. (2019). Senolytic therapy alleviates Abeta-associated oligodendrocyte progenitor cell senescence and cognitive deficits in an Alzheimer’s disease model. Nat. Neurosci. 22, 719–728. doi: 10.1038/s41593-019-0372-9
Keywords: circadian, clock, senescence, aging, oxidative stress
Citation: Ahmed R, Nakahata Y, Shinohara K and Bessho Y (2021) Cellular Senescence Triggers Altered Circadian Clocks With a Prolonged Period and Delayed Phases. Front. Neurosci. 15:638122. doi: 10.3389/fnins.2021.638122
Received: 05 December 2020; Accepted: 04 January 2021;
Published: 25 January 2021.
Edited by:
Takahiro J. Nakamura, Meiji University, JapanCopyright © 2021 Ahmed, Nakahata, Shinohara and Bessho. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yasukazu Nakahata, eWFzdS1uYWthaGF0YUBuYWdhc2FraS11LmFjLmpw
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.