- 1Department of Neurology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences, Beijing, China
- 2Department of Respiratory and Critical Care, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences, Beijing, China
Background: Neurological immune-related adverse events (nirAEs) are rare toxicities of immune-checkpoint inhibitors (ICI). With the increase use of ICIs, incidence of nirAEs is growing, among which ICI related MG (irMG) is causing high fatality rate. Given the limited evidence, data from a large cohort of patients with irMG is needed to aid in recognition and management of this fatal complication.
Objective: This study aimed to summarize clinical characteristics of irMG and explore predictors of irMG clinical outcome.
Methods: We summarized our institution's patients who were diagnosed as irMG between Sep 2019 and Oct 2021. We systematically reviewed the literature through Oct 2021 to identify all similar reported patients who met inclusion criteria. As the control group, patients with idiopathic MG were used. We collected data on clinical features, management, and outcomes of both irMG and idioMG cases. Further statistical analysis was conducted.
Results: Sixty three irMG patients and 380 idioMG patients were included in the final analysis. For irMG patients, six were from our institution while the rest 57 were from reported cases. The average age of irMG patients is 70.16 years old. Forty three were male. Average time from first ICI injection to symptom onset was 5.500 weeks. Eleven patients had a past history of MG. Higher MGFA classification and higher QMGS rates were observed in irMG patients compared to idioMG patients. For complication, more irMG patients had myositis or myocarditis overlapping compared to idioMG patients. The most commonly used treatment was corticosteroids for both idioMG and irMG. Twenty one patients (35%) with irMG had unfavorable disease outcome. Single variate and multivariate binary logistic regression proved that association with myocarditis, high MGFA classification or QMGS rates at first visit were negatively related to disease outcome in irMG patients.
Conclusion: irMG is a life-threatening adverse event. irMG has unique clinical manifestations and clinical outcome compared to idioMG. When suspicious, early evaluation of MGFA classification, QMGS rates and myositis/myocarditis evaluation are recommended.
Introduction
Immune checkpoint inhibitors (ICIs) are regarded as effective treatments for different types of advanced cancers (1). Despite impressive benefits observed from using ICIs, these treatments may be associated with serious immune-related adverse events (irAEs) caused by the induction of off-target inflammatory and autoimmune responses (1, 2).
ICI-related neurological adverse events are relatively infrequent; however, pooled analyses have shown that they are associated with increased morbidity and mortality (3–5). Myasthenia gravis (MG) is an autoimmune disorder mediated by autoantibodies, including anti-acetylcholine receptor (AChR) or anti-muscle associated receptor tyrosine kinase (MUSK) antibodies that target the neuromuscular junction (6). MG induced by ICI treatment or ICI-induced relapse of pre-existing MG is known as immune-related MG (irMG) (7). The incidence rate of irMG is 0.1%−0.2% according to the current literature (2, 8). Because of the low incidence rate and limited number of described cases, characterization of clinical features and prediction of disease outcome for irMG is difficult based on a patient's clinical manifestations. In this study, we described the clinical features of 63 patients with irMG and aimed to identify possible factors that may be useful for predicting irMG prognosis.
Materials and Methods
Patients
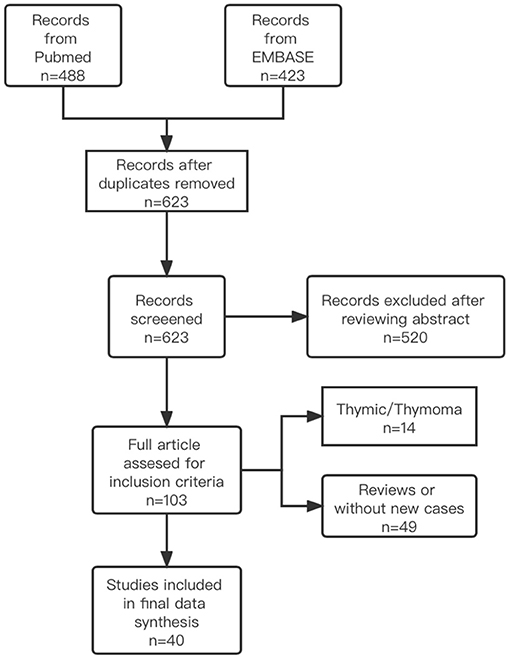
Patients who were diagnosed with irMG at PUMCH between September 2019 and October 2021 were included in the study cohort. We also searched the PubMed and EMBASE databases through October 2021 for case reports, case series, and observational studies that described patients with cancer and MG who received ICI treatment. The database searches did not include language or study design restrictions. Titles and abstracts were screened by two independent investigators to identify potentially relevant articles. Then, the full text of each selected article was retrieved and reviewed. A detailed clinical description of each patient was generated. The keywords included in the search were (“immune checkpoint inhibitors” OR “nivolumab” OR “ipilimumab” OR “pebrolizumab” OR “avelumab” OR “durvalumab” OR “atezolizumab” OR “anti-PD-1” OR “anti-PD-L1” OR “anti-CTLA-4”) AND (“immune related MG” OR “irMG” OR “MG” OR “Myasthenia Gravis” OR “ocular Myasthenia Gravis”). An inclusion diagram of patients is shown in Figure 1. The quality appraisal of the reported cases from the literature is shown in Supplementary Table 3. The inclusion criteria for both the PUMCH patients and cases identified in the literature included: (1) diagnosed with cancer; (2) any type of ICI used before MG onset or relapse; (3) definite or probable diagnosis of new-onset MG or deterioration of symptoms of well-controlled MG; and (4) a detailed description of the patient's clinical course was available. A definite diagnosis of MG was based on the presentation of ocular and/or systemic muscle weakness and at least one of the following criteria: (1) elevated titers of anti-AChR or anti-MUSK antibodies, (2) findings suggestive of MG in electrodiagnostic tests, (3) positive edrophonium test, or (4) positive ice pack test. A probable diagnosis of MG was made based on high clinical suspicion by the neurologist's report that confirmed the diagnosis of MG. We excluded patients with thymoma as an indicator for ICI treatment.
Controls
We also studied patients with idiopathic MG (idioMG), who were diagnosed at the PUMCH Neurology Department and registered at PUMCH MG registry as the control group. We have excluded idioMG patients with thymoma.
Methods
For both the PUMCH and literature identified patients, we extracted variables for patient demographics and baseline characteristics, including age, sex, type of ICI, indication of ICI, cancer staging, time between disease onset and first and last ICI injection, and severity of irAEs. We assessed the clinical severity of irMG using the Myasthenia Gravis Foundation of America (MGFA) classification system. For PUMCH patients, the quantitative myasthenia gravis score (QMGS) was determined by a trained neurologist at each patient's first visit. The QMGS was also collected for literature-identified patients if available. Data on clinical manifestations of MG (ptosis, diplopia, dyspnea, limb weakness and dysphagia); titer results for anti-AchR, anti-MUSK, and anti-titin antibodies; and overlap with myositis, myocarditis, or other system irAEs were also collected if available. Myositis was defined as elevated creatine kinase (CK) levels after disease onset. Myocarditis was defined as elevated cardiac troponin I levels, dynamic changes in electrocardiogram data, or symptoms of acute coronary artery syndrome. An unfavorable outcome was defined as tracheotomy, endotracheal intubation, or death directly caused by ICI-related MG.
Statistical Analysis
Baseline characteristics of irMG group were evaluated using frequencies and percentages for categorical data, while median and range were used to describe continuous data. Comparisons of categorical variables between control group and patient group were tested for significance using the x2 test. Continuous variables were compared using the Mann-Whitney U test. We performed single variate binary logistic regression analyses to determine the odds ratios (ORs) for associations between certain clinical or demographic factors and risk of unfavorable outcomes for irMG. Factors that were significantly associated with an unfavorable outcome were analyzed together in a multivariate binary logistic regression model. This analysis was performed with the maximal level of adjustment. All tests were 2-sided, and Bonferroni correction was applied to the α level to adjust for multiple comparisons. Bonferroni-adjusted p values are reported in the tables. Statistical analyses were carried out using the SPSS 24.0 statistical package (SPSS; Chicago, IL, USA). The study was approved by the local ethics committee.
Results
For irMG group, six patients from PUMCH were diagnosed with irMG. Of 623 unique articles from the literature, 40 publications describing 57 patients met the inclusion criteria (3, 6, 9–48). Therefore, a total of 63 patients were included in our final analysis. For idioMG group, we included 380 patients from PUMCH MG registry during the same period.
irMG Patient Demographic and Baseline Characteristics
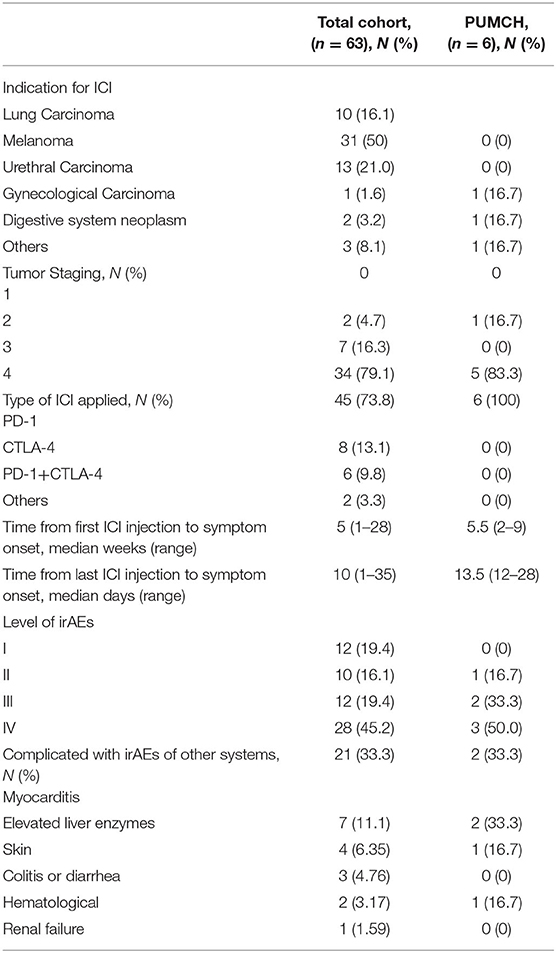
irMG patients' characteristics are shown in detail in Supplementary Table 1. A summary of demographic and baseline characteristics is shown in Table 1. The most common indication for ICIs was melanoma followed by urethral and lung carcinoma. The majority of patients had progressive tumor staging, and inhibitors of programmed cell death 1 (PD-1) were the most commonly applied therapeutics in the total cohort. The median time from ICI injection to symptom onset was 5 weeks, while the median time from the last ICI injection to symptom onset was 10 days. The severity of irAEs in the majority of patients in our cohort were classified at level IV. More than 60% of the patients had irAEs involving other systems. The most commonly involved system was cardiovascular system, followed by digestive system. Skin and hematological system irAEs were also observed.
irMG Characteristics and Comparison With idioMG Group
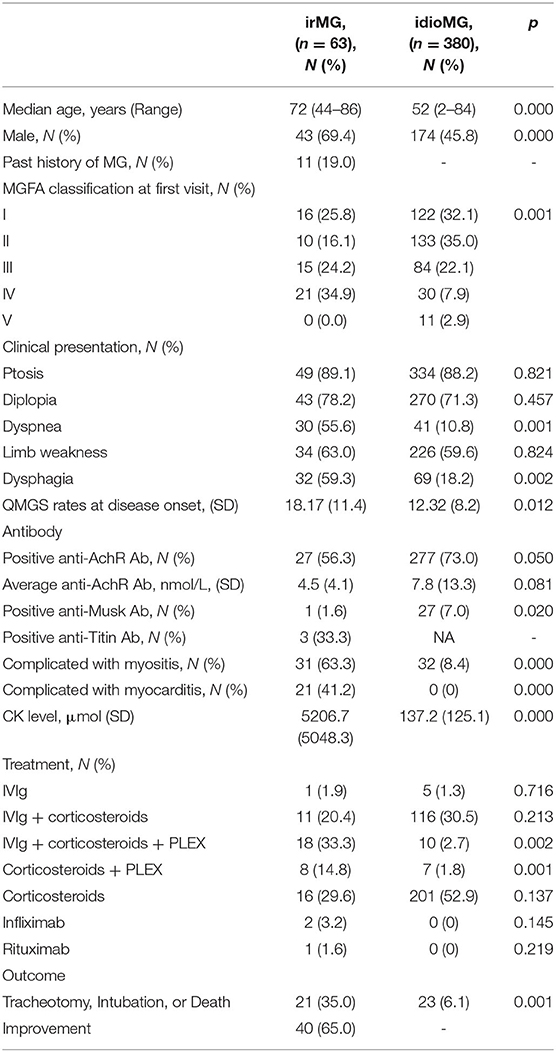
The patients' irMG characteristics, treatments, and outcomes are shown in detail in Supplementary Table 2. A comparison of irMG and idioMG characteristics are shown in Table 2. Among the 63 patients identified with irMG, 11 had a past history of well-managed MG and presented with a flare-up of MG after ICI initiation. The MGFA classification and QMGS rates clearly demonstrated that the disease was more severe in patients with irMG than in patients with idioMG. For clinical manifestations, bulbar symptoms and dyspnea were seen more frequently in patients with irMG. Serologic tests revealed that the frequency rates of anti-AchR antibody and anti-Musk antibody were significantly higher in idioMG group. Besides, the titer of anti-AChR antibodies was relatively low in patients with irMG compared to patients with idioMG. In irMG group, three patients were positive for anti-titin antibodies among nine patients tested (33.3%), which was not commonly seen in idioMG group. In irMG group, markedly elevated CK levels were observed with an average level of 5206.7 IU/L, which was scarcely found in idioMG group. In irMG group, 21 patients were diagnosed with myocarditis, while no patient had cardiac muscle involvement in idioMG group. Sixty one patients (96.8%) from irMG group required hospitalization after disease onset. Corticosteroids were used in more than 90% of patients for management for both irMG and idioMG. IVIg and PLEX were most commonly added to reduce the rapid progression of symptoms in irMG patients while infliximab and rituximab were used in 2 and 1 patients, respectively (33). For idioMG, PLEX was not commonly conducted in our center and we have no experience of using infliximab or rituximab. Unfavorable outcomes including death, intubation or tracheotomy was observed in 21 patients (35%) in irMG group, among which 14 patients (66.7%) died. Reasons of unfavorable outcomes include onset of a myasthenic crisis (13, 62%), infection or other complication (3, 14%) and cardiac incidence (5, 24%). Compared to irMG group, the incidence rate of unfavorable outcomes in idioMG group is relatively low. Discontinuation or withholding of ICI was recommended for 61 patients (97%) in our cohort, while 2 patients continued ICI treatments with well-controlled MG symptoms (41).
Associations Between Demographic and Clinical Factors and irMG Outcome
Results of single variate binary logistic regression are shown in Figure 2. Application of cytotoxic T-lymphocyte associated protein 4 (CTLA-4) and PD-1 inhibitors together was negatively related to an unfavorable outcome for ICI-related MG (OR = 12.142, p = 0.050). Evaluation parameters for MG severity, which included QMGS and MGFA classification at the first clinical visit, were indicative of disease outcome. A QMGS > 18.167 (OR = 6.667, p = 0.035) and MGFA classification IV (OR = 1.036, p = 0.000) were both related to unfavorable disease outcome. An overlap of myocarditis with irAEs in other systems was significantly associated with unfavorable irMG outcome. Although associated with myositis was not relevant to disease outcome, creatine kinase levels > 5,000 U/L were negatively related to disease outcome (OR = 6.667, p = 0.023). For treatment, we found that, compared to using steroids alone, administering IVIg, steroids plus PLEX, or IVIg plus steroids may be protective factors for irMG outcome. From the factors that were analyzed in the single variate binary logistic model, we included the type of ICI applied, MGFA classification, QMGS, overlap of myocarditis with other system irAEs, and treatments in the multivariate binary logistic regression model. The multivariate analysis (Figure 3) showed that, associated with myocarditis, QMGS ≥ 18.167 and MGFA classification IV were negatively related to the outcome of irMG. Compared with corticosteroids alone, utilization of IVIg and PLEX may be a positive prognostic factor for irMG.
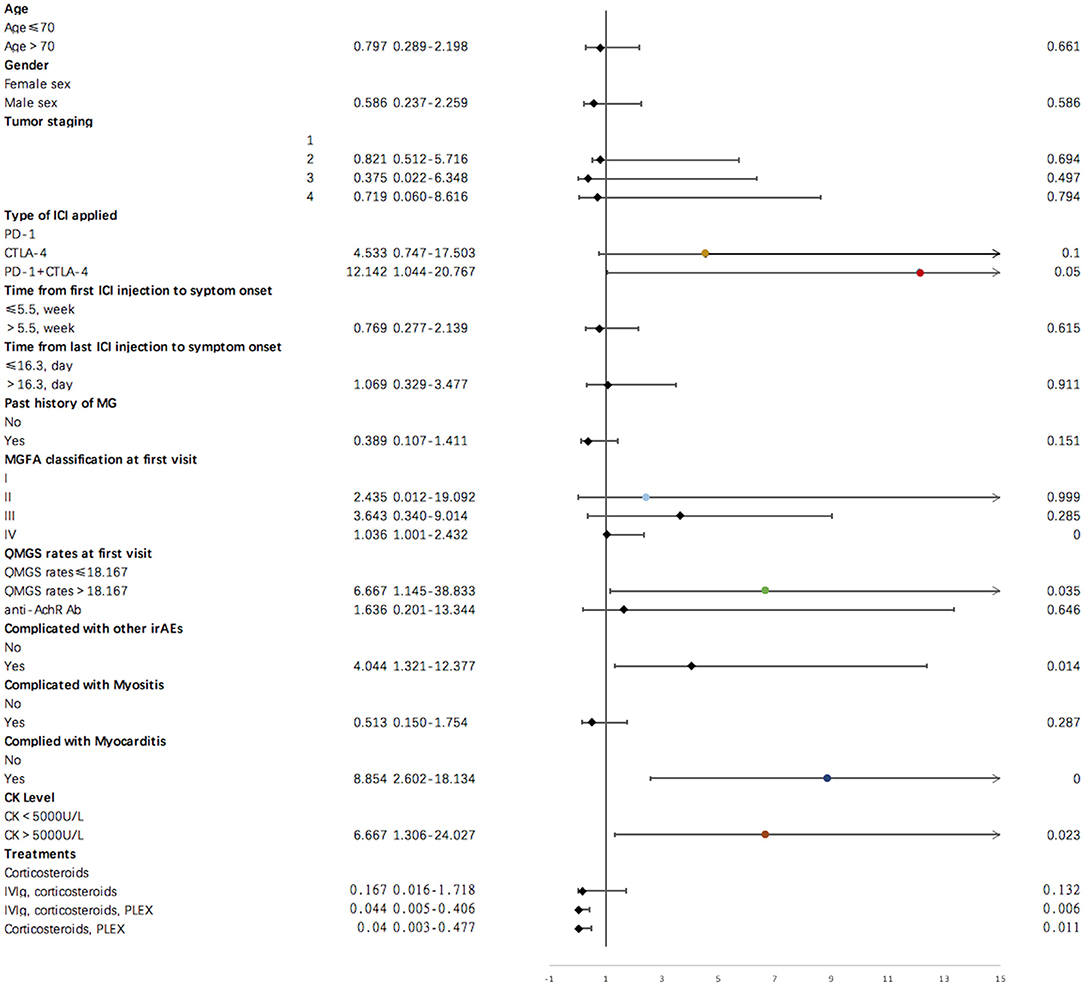
Figure 2. Results of single variate binary logistic regression for demographic and clinical factors and irMG outcome.
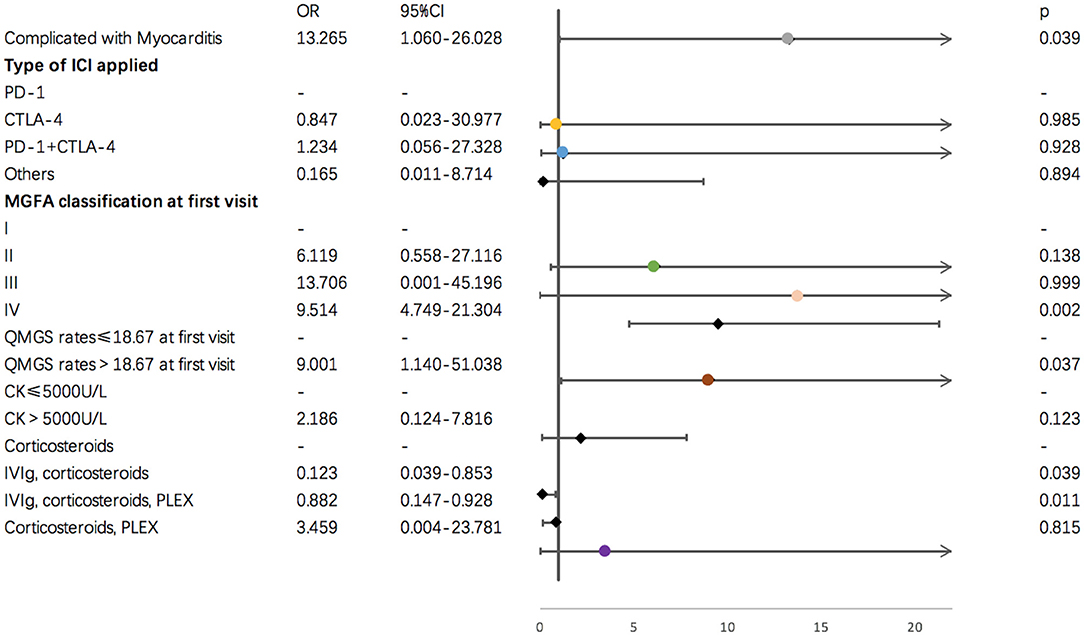
Figure 3. Results of multi-variate binary logistic regression for demographic and clinical factors and irMG outcome.
Discussion
In this study, we report an extensive case series of ICI-related MG with detailed clinical features, treatments, and disease outcome. Our study innovatively identified several clinical factors that may be useful for predicting irMG prognosis.
Our findings support that irMG has several different clinical features compared with idioMG, which has also been proved by previous studies (10, 11). Demographically, the age at diagnosis of irMG was significantly greater than that of patients with idioMG. For clinical severity, the majority of idioMG patients fall within the MGFA classes I and II at the time of diagnosis and present with a slow clinical deterioration course (47), while the majority of irMG patients were categorized in MGFA classes III and IV at the first visit with a high QMGS rates. Serologically, the positive rate of anti-AchR antibody in idioMG patients has been reported to be around 70–80% (49–51), which is statistically higher than the positive rate of irMG group. For anti-MUSK antibody, positive rate in idioMG patients is ~5–10% (51), while in irMG group, positive rate was only 1.6%. This finding shows that the prevalence of seronegative patients in irMG was higher than that in classical MG, which has been previously proved by other studies (7, 41, 52). The demographic and serological differences could be caused by the bias due to that case reports and case series tend to report irMG patients with more severe clinical manifestations, still the differences that we observed indicated that irMG and idioMG are clinically distinct disease entity.
We also observed that irMG were more likely to be associated with myositis or myocarditis, which has been described in only 0.9% of patients with idioMG (6, 7, 53). Some researchers believe that the elevation of serum CK in patients with irMG reflects inflammatory involvement of skeletal muscles rather than rhabdomyolysis (7). Other investigators have hypothesized that myositis is the main clinical manifestation of irMG patients, whereas a positive antibody result is a marker of activated autoimmunity (23, 54). High association rate of myocarditis in irMG group is noticeable. Although the mechanism of this phenomenon is now still not well-established, molecular mimicry and the critical role of PD-1 signaling pathways in regulating autoimmune responses myocardium might be responsible (19).
It is important for physicians to identify factors that might be indicative of disease outcome. We found that higher MGFA classification and a higher QMGS at the first visit were predictive for an unfavorable disease outcome. Both MGFA classification and QMGS are parameters for prognosis prediction and severity evaluation in idioMG patients, and our study supported that the utilization of these measures is valid in irMG patients. It has been demonstrated that associated with myositis may increase muscle weakness in patients with irMG (7, 51, 53), suggesting that a substantial proportion of an irMG patient's clinical symptoms is associated with the accompanying myositis. Although associating with myositis was not related to disease outcome in our analysis, still in single variate analysis, we have found that CK>5,000 U/L was a negative prognosis factor. Thus, awareness of early recognition of muscle involvement in possible irMG patients is important. In this regard, serum CK tests before and after treatment with irMG is required. Association with myocarditis has been reported to be negatively related to irAEs outcome (22, 47, 54). Because it is not uncommon for irMG patients to have myocarditis [39.7% in our cohort, 20%−40% in published series cases (4, 55, 56)], we believe that particular attention including ECG, echocardiography and serum troponin tests should be conducted for myocarditis identification to allow timely multidisciplinary management.
Our data suggest that patients who received IVIg or PLEX experienced improved irMG outcomes compared with those who received steroids alone. Although corticosteroids are recommended as a first-line treatment for irMG (2, 57), the use of steroids as a sole first-line therapy may not be ideal because steroid use itself can cause an acute exacerbation of MG symptoms (58). Although the worsening in symptoms has been described as transient (59), the use of steroids alone in irMG may be associated with a poorer prognosis because these patients may not be able to survive a transient worsening of symptoms considering their older age and advanced stage of malignancy (41). Apart from this, the role of steroids in controlling immune dysregulation in irMG patients might be limited by the constant presence of the circulating ICIs as the original trigger of irAEs. Since IVIg and PLEX could accelerate the process of ICIs mAbs elimination, they could mediate a faster improvement of symptoms (41). From our clinical experience and analysis, the use of IVIg and PLEX together with corticosteroids has led to favorable outcomes in irMG patients, which has been demonstrated in other studies as well (3, 19, 41, 60). However, for irMG initial treatment, no consistent conclusion could be drawn from the big variety of published reports. Given the small number of patients and the retrospective nature of our study, we think further researches for irMG treatment is highly required. Besides, physicians should be aware of early treatments of vital organ dysfunction. Timely intubation for respiratory failure, pacemaker implantation for fatal arrhythmia, and vasopressor and even extracorporeal membrane oxygenation for cardiogenic shock should be considered if clinically needed and available.
Our study has some limitations. Although it is the first study that identifies possible factors responsible for irMG outcome, the relatively small sample size and retrospective nature of the study design limit the reliability of our study results. Because of the variability in the data available from case reports or case series, there were missing data regarding clinical features, hospital course, and outcomes of some patients, which subjected our results to reporting bias. Besides, the information obtained from the collected case reports represents only a small fraction of the actual number of cases worldwide. Nevertheless, our study enhances the understanding of irMG clinical manifestations and factors involved in irMG prognosis.
With the boost of ICIs utilization and awareness of the disease, we believe that the number of patients with irMG is poised to rapid increase. Additional therapeutic studies concerning irMG in the future are needed to decrease the irAE-related mortality and increase the safety of immune therapy.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics Statement
The studies involving human participants were reviewed and approved by Peking Union Medical College Hospital. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
JS and YG contributed to the conception and design of the study. JS, YT, YH, KL, and JY collected the clinical data. JS and YH contributed to the potential article identifications and article quality evaluation. JS wrote the manuscript. YG and LZ edited the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by the National Clinical Cohort Study of Rare Diseases for Key R&D Program (Grant Number: 2016YFC091501) and Beijing Municipal Science & Technology Commission Capital Characteristic Clinic Project Z181100001718145.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fneur.2022.858628/full#supplementary-material
References
1. Sharma P, Allison JP. The future of immune checkpoint therapy. Science (New York, NY). (2015) 348:56–61. doi: 10.1126/science.aaa8172
2. Shi J, Niu J, Shen D, Liu M, Tan Y, Li Y, et al. Clinical diagnosis and treatment recommendations for immune checkpoint inhibitor-related adverse reactions in the nervous system. Thorac Cancer. (2020) 11:481–7. doi: 10.1111/1759-7714.13266
3. Diamanti L, Picca A, Bini P, Gastaldi M, Alfonsi E, Pichiecchio A, et al. Characterization and management of neurological adverse events during immune-checkpoint inhibitors treatment: an Italian multicentric experience. Neurol Sci. (2021) 43:2031–41. doi: 10.1007/s10072-021-05561-z
4. Johansen A, Christensen SJ, Scheie D, Højgaard JLS, Kondziella D. Neuromuscular adverse events associated with anti-PD-1 monoclonal antibodies: systematic review. Neurology. (2019) 92:663–74. doi: 10.1212/WNL.0000000000007235
5. Bolz S, Ramakrishnan T, Fleischer M, Livingstone E, Stolte B, Thimm A, et al. Detect it so you can treat it: a case series and proposed checklist to detect neurotoxicity in checkpoint therapy. eNeurologicalSci. (2021) 22:100324. doi: 10.1016/j.ensci.2021.100324
6. Drachman DB. Myasthenia gravis. N Engl J Med. (1994) 330:1797–810. doi: 10.1056/NEJM199406233302507
7. Suzuki S, Ishikawa N, Konoeda F, Seki N, Fukushima S, Takahashi K, et al. Nivolumab-related myasthenia gravis with myositis and myocarditis in Japan. Neurology. (2017) 89:1127–34. doi: 10.1212/WNL.0000000000004359
8. Makarious D, Horwood K, Coward JIG. Myasthenia gravis: an emerging toxicity of immune checkpoint inhibitors. Eur J Cancer (Oxford, England: 1990). (2017) 82:128–36. doi: 10.1016/j.ejca.2017.05.041
9. Khokher W, Bhuta S, Kesireddy N, Abuhelwa Z, Mhanna M, Iftikhar S, et al. Refractory nivolumab-induced myasthenia gravis treated with abatacept. Am J Ther. (2021). doi: 10.1097/mjt.0000000000001405. [Epub ahead of print].
10. Alnahhas I, Wong J. A case of new-onset antibody-positive myasthenia gravis in a patient treated with pembrolizumab for melanoma. Muscle Nerve. (2017) 55:E25–e6. doi: 10.1002/mus.25496
11. Algaeed M, Mukharesh L, Heinzelmann M, Kaminski HJ. Pearls & Oy-sters: pembrolizumab-induced myasthenia gravis. Neurology. (2018) 91:e1365–e7. doi: 10.1212/WNL.0000000000006278
12. Fukasawa Y, Sasaki K, Natsume M, Nakashima M, Ota S, Watanabe K, et al. Nivolumab-induced myocarditis concomitant with myasthenia gravis. Case Rep Oncol. (2017) 10:809–12. doi: 10.1159/000479958
13. Johnson DB, Saranga-Perry V, Lavin PJ, Burnette WB, Clark SW, Uskavitch DR, et al. Myasthenia gravis induced by ipilimumab in patients with metastatic melanoma. J Clin Oncol. (2015) 33:e122–4. doi: 10.1200/JCO.2013.51.1683
14. Liu Q, Ayyappan S, Broad A, Narita A. Pembrolizumab-associated ocular myasthenia gravis. Clin Experiment Ophthalmol. (2019) 47:796–8. doi: 10.1111/ceo.13499
15. Liao B, Shroff S, Kamiya-Matsuoka C, Tummala S. Atypical neurological complications of ipilimumab therapy in patients with metastatic melanoma. Neuro Oncol. (2014) 16:589–93. doi: 10.1093/neuonc/nou001
16. Mathews EP, Romito JW. Management of immune checkpoint inhibitor-related acute hypoxic neuromuscular respiratory failure using high-flow nasal cannula. Proceedings (Baylor University Medical Center). (2020) 33:407–8. doi: 10.1080/08998280.2020.1744793
17. Montes V, Sousa S, Pita F, Guerreiro R, Carmona C. Myasthenia gravis induced by ipilimumab in a patient with metastatic melanoma. Front Neurol. (2018) 9:150. doi: 10.3389/fneur.2018.00150
18. Mehta JJ, Maloney E, Srinivasan S, Seitz P, Cannon M. Myasthenia gravis induced by nivolumab: a case report. Cureus. (2017) 9:e1702. doi: 10.7759/cureus.1702
19. Pathak R, Katel A, Massarelli E, Villaflor VM, Sun V, Salgia R. Immune checkpoint inhibitor-induced myocarditis with myositis/myasthenia gravis overlap syndrome: a systematic review of cases. Oncologist. (2021) 26:1052–61. doi: 10.1002/onco.13931
20. Onda A, Miyagawa S, Takahashi N, Gochi M, Takagi M, Nishino I, et al. Pembrolizumab-induced ocular myasthenia gravis with anti-titin antibody and necrotizing myopathy. Intern Med (Tokyo, Japan). (2019) 58:1635–8. doi: 10.2169/internalmedicine.1956-18
21. Phadke SD, Ghabour R, Swick BL, Swenson A, Milhem M, Zakharia Y. Pembrolizumab therapy triggering an exacerbation of preexisting autoimmune disease: a report of 2 patient cases. J Investig Med High Impact Case Rep. (2016) 4:2324709616674316. doi: 10.1177/2324709616674316
22. Salim A, Tapia Rico G, Shaikh A, Brown MP. A systematic review of immune checkpoint inhibitor-related neurological adverse events and association with anti-neuronal autoantibodies. Expert Opin Biol Ther. (2021) 21:1237–51. doi: 10.1080/14712598.2021.1897101
23. Shelly S, Triplett JD, Pinto MV, Milone M, Diehn FE, Zekeridou A, et al. Immune checkpoint inhibitor-associated myopathy: a clinicoseropathologically distinct myopathy. Brain Commun. (2020) 2:fcaa181. doi: 10.1093/braincomms/fcaa181
24. Takai M, Kato D, Iinuma K, Maekawa YM, Nakane K, Tsuchiya T, et al. Simultaneous pembrolizumab-induced myasthenia gravis and myocarditis in a patient with metastatic bladder cancer: a case report. Urol Case Rep. (2020) 31:101145. doi: 10.1016/j.eucr.2020.101145
25. Takizawa T, Kojima M, Suzuki S, Osada T, Kitagawa S, Nakahara J, et al. New onset of myasthenia gravis after intravesical Bacillus Calmette-Guerin: a case report and literature review. Medicine. (2017) 96:e8757. doi: 10.1097/MD.0000000000008757
26. Todo M, Kaneko G, Shirotake S, Shimada Y, Nakano S, Okabe T, et al. Pembrolizumab-induced myasthenia gravis with myositis and presumable myocarditis in a patient with bladder cancer. IJU Case Rep. (2020) 3:17–20. doi: 10.1002/iju5.12128
27. Tozuka T, Sugano T, Noro R, Takano N, Hisakane K, Takahashi S, et al. Pembrolizumab-induced agranulocytosis in a pulmonary pleomorphic carcinoma patient who developed interstitial lung disease and ocular myasthenia gravis. Oxford Med Case Rep. (2018) 2018:omy094. doi: 10.1093/omcr/omy094
28. Tomisaki I. Editorial comment to pembrolizumab-induced myasthenia gravis with myositis and presumable myocarditis in a patient with bladder cancer. IJU Case Rep. (2020) 3:20. doi: 10.1002/iju5.12132
29. Wilson R, Menassa DA, Davies AJ, Michael S, Hester J, Kuker W, et al. Seronegative antibody-mediated neurology after immune checkpoint inhibitors. Ann Clin Transl Neurol. (2018) 5:640–5. doi: 10.1002/acn3.547
30. Zhu J, Li Y. Myasthenia gravis exacerbation associated with pembrolizumab. Muscle Nerve. (2016) 54:506–7. doi: 10.1002/mus.25055
31. Dang T, Macwan S, Dasanu CA. Late-onset double-seronegative myasthenia gravis syndrome and myasthenic crisis due to nivolumab use for Hodgkin's lymphoma. J Oncol Pharm Pract. (2020) 2020:1078155220976797. doi: 10.1177/1078155220976797
32. March KL, Samarin MJ, Sodhi A, Owens RE. Pembrolizumab-induced myasthenia gravis: a fatal case report. J Oncol Pharm Pract. (2018) 24:146–9. doi: 10.1177/1078155216687389
33. Rajendram P, Torbic H, Duggal A, Campbell J, Hovden M, Dhawan V, et al. Critically ill patients with severe immune checkpoint inhibitor related neurotoxicity: a multi-center case series. J Crit Care. (2021) 65:126–32. doi: 10.1016/j.jcrc.2021.05.020
34. Puwanant A, Isfort M, Lacomis D, Živković SA. Clinical spectrum of neuromuscular complications after immune checkpoint inhibition. Neuromuscul Disord. (2019) 29:127–33. doi: 10.1016/j.nmd.2018.11.012
35. Shirai T, Kiniwa Y, Sato R, Sano T, Nakamura K, Mikoshiba Y, et al. Presence of antibodies to striated muscle and acetylcholine receptor in association with occurrence of myasthenia gravis with myositis and myocarditis in a patient with melanoma treated with an anti-programmed death 1 antibody. Eur J Cancer (Oxford, England: 1990). (2019) 106:193–5. doi: 10.1016/j.ejca.2018.10.025
36. Yang Y, Xu L, Wang D, Hui B, Li X, Zhou Y, et al. Anti-PD-1 and regorafenib induce severe multisystem adverse events in microsatellite stability metastatic colorectal cancer: a case report. Immunotherapy. (2021) 13:1317–23. doi: 10.2217/imt-2020-0327
37. Ziobro AS, LaPlante RL, DeMari SR, Clark LM, Kingsley DJ, Smith AJ. Myasthenia gravis associated with Programmed Death-1 (PD-1) receptor inhibitor pembrolizumab: a 40-day case report. J Pharm Pract. (2021) 34:166–70. doi: 10.1177/0897190020970750
38. Jeyakumar N, Etchegaray M, Henry J, Lelenwa L, Zhao B, Segura A, et al. The terrible triad of checkpoint inhibition: a case report of myasthenia gravis, myocarditis, and myositis induced by cemiplimab in a patient with metastatic cutaneous squamous cell carcinoma. Case Rep Immunol. (2020) 2020:5126717. doi: 10.1155/2020/5126717
39. Hayakawa N, Kikuchi E, Suzuki S, Oya M. Myasthenia gravis with myositis induced by pembrolizumab therapy in a patient with metastatic urothelial carcinoma. Int Cancer Conf J. (2020) 9:123–6. doi: 10.1007/s13691-020-00408-4
40. Botta C, Agostino RM, Dattola V, Cianci V, Calandruccio ND, Bianco G, et al. Myositis/myasthenia after pembrolizumab in a bladder cancer patient with an autoimmunity-associated HLA: immune-biological evaluation and case report. Int J Mol Sci. (2021) 22:6246. doi: 10.3390/ijms22126246
41. Safa H, Johnson DH, Trinh VA, Rodgers TE, Lin H, Suarez-Almazor ME, et al. Immune checkpoint inhibitor related myasthenia gravis: single center experience and systematic review of the literature. J Immunother Cancer. (2019) 7:319. doi: 10.1186/s40425-019-0774-y
42. Hibino M, Maeda K, Horiuchi S, Fukuda M, Kondo T. Pembrolizumab-induced myasthenia gravis with myositis in a patient with lung cancer. Respirol Case Rep. (2018) 6:e00355. doi: 10.1002/rcr2.355
43. Lau KH, Kumar A, Yang IH, Nowak RJ. Exacerbation of myasthenia gravis in a patient with melanoma treated with pembrolizumab. Muscle Nerve. (2016) 54:157–61. doi: 10.1002/mus.25141
44. Nguyen BH, Kuo J, Budiman A, Christie H, Ali S. Two cases of clinical myasthenia gravis associated with pembrolizumab use in responding melanoma patients. Melanoma Res. (2017) 27:152–4. doi: 10.1097/CMR.0000000000000310
45. Hajihossainlou B, Vasileva A, Manthri S, Chakraborty K. Myasthenia gravis induced or exacerbated by immune checkpoint inhibitors: a rising concern. BMJ Case Rep. (2021) 14:e243764. doi: 10.1136/bcr-2021-243764
46. Giglio D, Berntsson H, Fred Å, Ny L. Immune Checkpoint Inhibitor-Induced Polymyositis and Myasthenia Gravis with Fatal Outcome. Case Rep Oncol. (2020) 13:1252–7. doi: 10.1159/000510740
47. Melzer N, Ruck T, Fuhr P, Gold R, Hohlfeld R, Marx A, et al. Clinical features, pathogenesis, and treatment of myasthenia gravis: a supplement to the Guidelines of the German Neurological Society. J Neurol. (2016) 263:1473–94. doi: 10.1007/s00415-016-8045-z
48. Derle E, Benli S. Ipilimumab treatment associated with myasthenic crises and unfavorable disease course. Neurol Sci. (2018) 39:1773–4. doi: 10.1007/s10072-018-3471-6
49. Dresser L, Wlodarski R, Rezania K, Soliven B. Myasthenia gravis: epidemiology, pathophysiology and clinical manifestations. J Clin Med. (2021) 10:2235. doi: 10.3390/jcm10112235
50. Gilhus NE, Verschuuren JJ. Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol. (2015) 14:1023–36. doi: 10.1016/S1474-4422(15)00145-3
51. Andersen JB, Gilhus NE, Sanders DB. Factors affecting outcome in myasthenia gravis. Muscle Nerve. (2016) 54:1041–9. doi: 10.1002/mus.25205
52. Huang YT, Chen YP, Lin WC, Su WC, Sun YT. Immune checkpoint inhibitor-induced myasthenia gravis. Front Neurol. (2020) 11:634. doi: 10.3389/fneur.2020.00634
53. Grob D, Brunner N, Namba T, Pagala M. Lifetime course of myasthenia gravis. Muscle Nerve. (2008) 37:141–9. doi: 10.1002/mus.20950
54. Hamada N, Maeda A, Takase-Minegishi K, Kirino Y, Sugiyama Y, Namkoong H, et al. Incidence and distinct features of immune checkpoint inhibitor-related myositis from idiopathic inflammatory myositis: a single-center experience with systematic literature review and meta-analysis. Front Immunol. (2021) 12:803410. doi: 10.3389/fimmu.2021.803410
55. Moslehi JJ, Salem JE, Sosman JA, Lebrun-Vignes B, Johnson DB. Increased reporting of fatal immune checkpoint inhibitor-associated myocarditis. Lancet (London, England). (2018) 391:933. doi: 10.1016/S0140-6736(18)30533-6
56. Touat M, Maisonobe T, Knauss S, Ben Hadj Salem O, Hervier B, Auré K, et al. Immune checkpoint inhibitor-related myositis and myocarditis in patients with cancer. Neurology. (2018) 91:e985–e94. doi: 10.1212/WNL.0000000000006124
57. Puzanov I, Diab A, Abdallah K, Bingham CO. 3rd, Brogdon C, Dadu R, et al. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. (2017) 5:95. doi: 10.1186/s40425-017-0300-z
58. Miller RG, Milner-Brown HS, Mirka A. Prednisone-induced worsening of neuromuscular function in myasthenia gravis. Neurology. (1986) 36:729–32. doi: 10.1212/WNL.36.5.729
59. Bae JS, Go SM, Kim BJ. Clinical predictors of steroid-induced exacerbation in myasthenia gravis. J Clin Neurosci. (2006) 13:1006–10. doi: 10.1016/j.jocn.2005.12.041
Keywords: myasthenia gravis, MGFA, QMG, immune-related adverse effects, immune checkpoint inhibitors
Citation: Shi J, Tan Y, Huang Y, Li K, Yan J, Guan Y and Zhang L (2022) Association Between Clinical Factors and Result of Immune Checkpoint Inhibitor Related Myasthenia Gravis: A Single Center Experience and Systematic Review. Front. Neurol. 13:858628. doi: 10.3389/fneur.2022.858628
Received: 20 January 2022; Accepted: 15 March 2022;
Published: 07 April 2022.
Edited by:
Nils Erik Gilhus, University of Bergen, NorwayReviewed by:
Piotr Szczudlik, Medical University of Warsaw, PolandTimothy John Day, Royal Melbourne Hospital, Australia
Copyright © 2022 Shi, Tan, Huang, Li, Yan, Guan and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yuzhou Guan, Z3Vhbnl6MDAxQDE2My5jb20=
 Jiayu Shi
Jiayu Shi Ying Tan
Ying Tan Yangyu Huang
Yangyu Huang Ke Li
Ke Li Jingwen Yan1
Jingwen Yan1 Yuzhou Guan
Yuzhou Guan