- Department of Ophthalmology, Jiangxi Province Ocular Disease Clinical Research Center, The First Affiliated Hospital of Nanchang University, Nanchang, China
In recent years, the impact of various chronic eye diseases on quality of life has become increasingly apparent. Therefore, it is particularly important to control the progress of chronic diseases at an early stage. Many studies have used neuroimaging methods to explore the effects of chronic eye diseases on the brain, and to identify changes in brain function that may act as markers for early diagnosis and treatment. This article reviews the clinical application of different techniques of functional magnetic resonance imaging in chronic eye diseases.
Application of ALFF in Chronic Eye DiseaseS
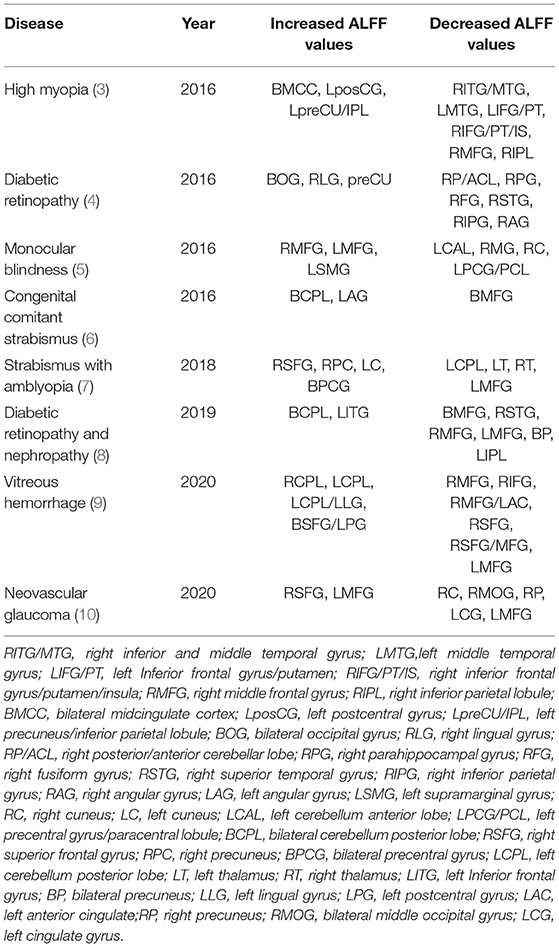
As a resting-state functional magnetic resonance imaging (MRI) analysis tool, the amplitude of low-frequency fluctuation (ALFF) has high accuracy and repeatability and does not require definition of regions of interest (ROI) in advance. By calculating the root mean square of the blood oxygen level dependent (BOLD) signal power spectrum at low frequencies (0.01HZ−0.08HZ), the neuronal activity in different brain regions is expressed and is an indicator of spontaneous neuronal activity (1). The value of ALFF technology in detecting brain neuron activity has been confirmed in previous experiments (2). Changes in ALFF value reflect disease progression, in part and are widely used in the diagnosis and monitoring of eye diseases. ALFF markers for chronic eye disease are shown in Table 1, where the middle frontal gyrus (MFG) features prominently. However, changes in ALFF values at the MFG have implications that vary with disease. For example, in patients with monocular blindness (5) and neovascular glaucoma (10), ALFF changes at the MFG are related to visual perception impairment or compensation, while similar changes in diabetic vitreous hemorrhage (9) indicate visual motor disorder, and in strabismic amblyopia (7), diabetic retinopathy and nephropathy (8) they may indicate a tendency toward anxiety and depression.
Application of DC in Chronic Eye Diseases
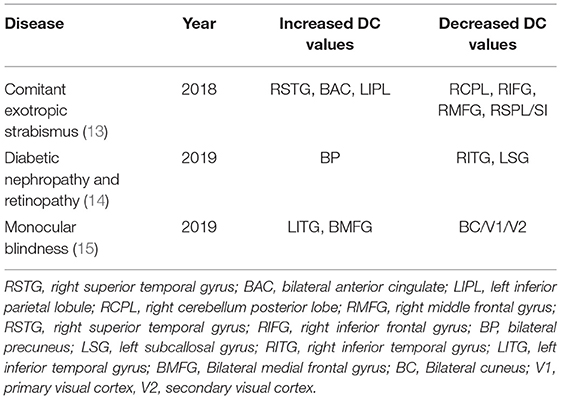
Voxel-wise degree centrality (DC) is another commonly used technique in resting state functional MRI technology. It assesses functional connectivity within the brain by measuring the topological structure of brain functional connectors at voxel level, and provides the correlation between different nodes and the importance of each node in the network structure (11). The degree of direct functional connection between two nodes can be expressed by DC values. A high DC value indicates a higher degree of direct connection between the node and other nodes. A change in DC values indicates a change in connectivity between the node and the network and clearly shows the state of each node (12). This method can be used to find any changes in brain functional connectivity in chronic ocular disease, and the changemay be an important marker for disease detection. Chronic diseases and their corresponding DC value markers are shown in Table 2.
Application of REHO in Chronic Eye Diseases
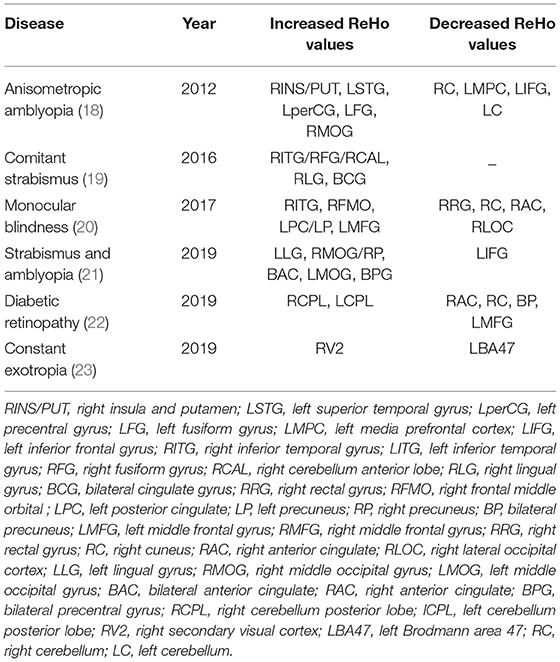
Regional homogeneity (ReHo) is a widely used analytical method in resting state MRI, and plays an important role in exploring changes in local synchronization of voxels in brain regions (16). The analysis of ReHo is based on the measurement of voxels, and is used to measure the synchronization of adjacent regions based on the similarity between the time series of a given voxel and its nearest adjacent time series (calculated by the Kendall consistency coefficient of BOLD time series) (17). The coherence and centrality of regional brain activity are closely related to the ReHo values. ReHo is usually calculated in the low frequency range (0.01Hz–0.1Hz). Like ALFF, ReHo does not need a priori definition of ROI and can provide information about the activity of the whole brain. ReHo has been widely used in various studies to explore the local synchronization of spontaneous fMRI signals. In addition, the ReHo method is used in many studies of chronic eye diseases. Altered ReHo values can be used as a marker to monitor progress of various diseases, as shown by Table 3. ReHo measures at the inferior temporal gyrus (ITG) and the right cuneus (RC) are common markersfor chronic eye disease. The significantly increased ReHo values of the ITG in concomitant exotropia (23) and monocular blindness (20) indicated the compensatory mechanism of visual function. In monocular blindness, the ReHo values of RC decrease significantly, indicating the interruption of synchronous neural activity. The RC also showed a decreased ReHo value in diabetic retinopathy (22), reflecting vision-related dysfunction in this area. In patients with anisometropic amblyopia (18), a clear increase in RC was related to the compensatory mechanism of eye movement.
Application of FC in Chronic Eye Diseases
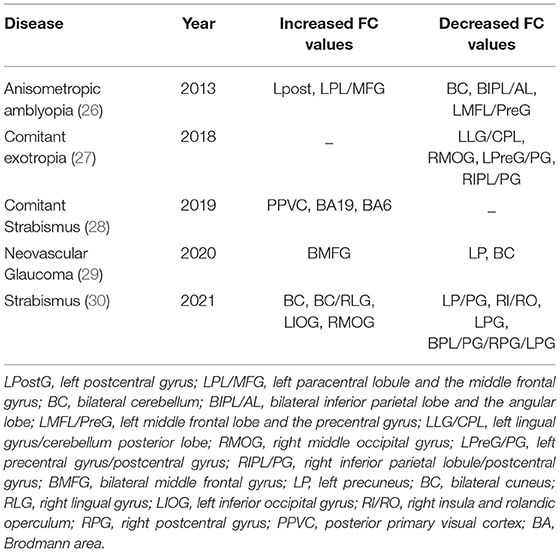
Functional connectivity (FC) is a seed-based or ROI-based functional connection, in which areas functionally related to activities in the seed area may be found (24). In seed-based analysis, cross-correlation is calculated between the time series of the seed and the rest of the brain to assess the activity of the selected brain region. Functional connections may be considered as brain areas which have been activated similarly, indicating that they have a similar role in brain functional activity. Physiologically, the relevant brain regions may not be directly connected by nerve fibers, but the connectivity matrix shows connection strength and range including indirect connections. Flexibility and sensitivity of this technique has resulted in it being widely used in the study of various brain functional diseases (25). The FC value indicates the intensity of activity and may be used to mark changes in brain functional activity caused by disease. Application of the FC method as a marker in brain functional activity of chronic ophthalmopathy is shown in Table 4.
Application of VBM in Chronic Eye Diseases
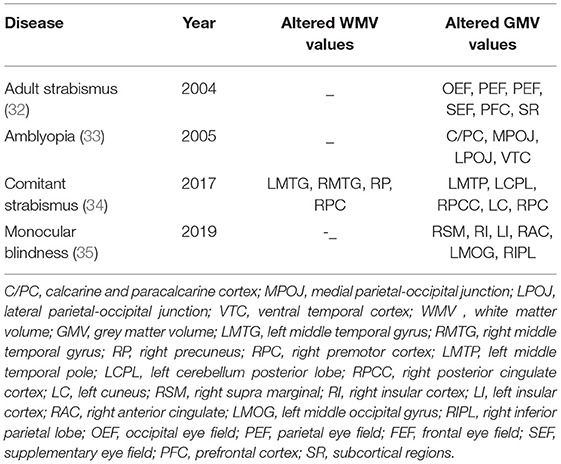
Voxel-based morphometry (VBM) is an MRI whole-brain analysis technique for measuring density and volume changes of gray and white matter at the voxel level, and is used to enhance understanding of the anatomical structure of the brain (30). In contrast with some other resting MRI techniques, VBM has no preset region of interest, it detects changes in neural activity across all parts of the brain, and is an objective measure so is minimally influenced by subjective factors. The VBM approach filters the white and gray matter areas with statistically significant activity by comparing the processed MRI images (31), and can be used to detect pathological changes of brain function caused by disease. Changes of this kind have been found to accompany progression of many ophthalmic diseases, as shown in Table 5.
Application of VMHC in Chronic Eye Diseases
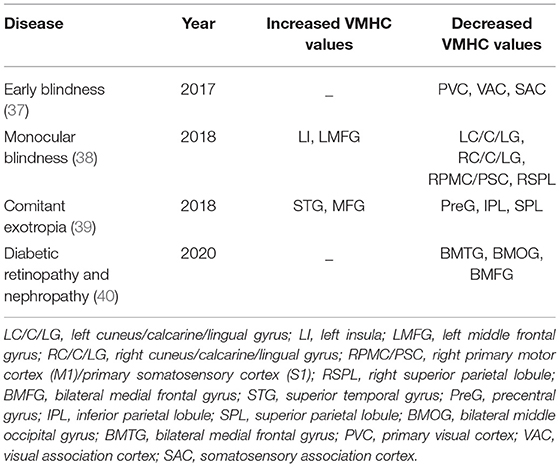
Voxel-mirrored homotopic connectivity (VMHC) is a new resting-state FC analysis method to measure the functional connection between hemispheres (36). This method can detect abnormal functional activity in local brain areas and changes of functional connection and synchronization of neural activity between corresponding regions in bilateral cerebral hemispheres at rest, which shows that the degree of separation of cerebral hemispheres is its outstanding advantage. The normal human brain generally has the characteristic of high synchronization of spontaneous nerve activity in homotopic regions between hemispheres. Many studies have confirmed that this characteristic may be generally destroyed in patients with chronic eye diseases, suggesting that hemispheric dysfunction plays an important role in brain dysfunction in patients with chronic eye diseases. Using VMHC, this loss of synchrony has been demonstrated in patients with diseases of this kind, as shown in Table 6.
Application of Other Techniques in Chronic Eye Diseases
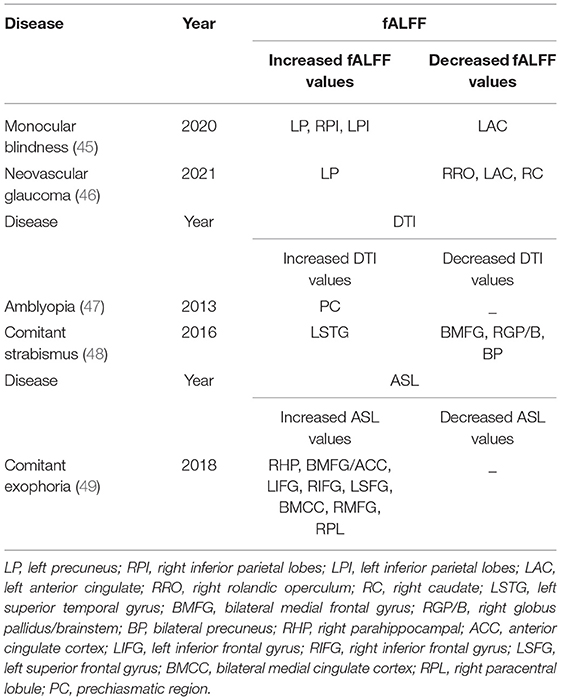
The fractional amplitude of low-frequency fluctuation (fALFF) provides a measure of inherent spontaneous brain activity (41). Measurement of fALFF values need to be carried out within a specified frequency range, and spontaneous brain activity can be expressed by the measurement of cerebral blood flow at the amplitude of low frequency oscillation (0.01–0.08 Hz) (42). The fALFF technique has the advantages of high sensitivity and specificity and is non-invasive, so it is widely used in brain functional activity imaging. Diffusion tensor imaging (DTI) is a widely used MRI method, which describes the diffusion direction of water as average diffusion coefficient (MD; diffusion within voxels) and fractional anisotropy (FA) (43). The overall extent of water diffusion may also be displayed. On the basis of eigenvalues (λ1, λ2, λ3) of diffusion tensor, scalar values ranging from 0 to 1 can be obtained. These are the FA values, which measure the overall directionality of water diffusion and the complexity of cytoskeleton structure, of great significance to the movement of water inside and outside of cells (44). Changes in direction of water diffusion help understand the pathological changes of myelin and other related brain tissues. On this basis, some studies have explored the application of DTI in eye diseases, and the value of DTI as a marker in the diagnosis of diseases. Arterial spin labeling (ASL) is a new technology developed on the basis of magnetic resonance perfusion imaging, which has high accuracy and is non-invasive. It can detect blood flow changes reflecting pathological changes in various regions of the brain. The ASL method has been successfully applied to trace the changes of local blood flow in eye diseases and is beneficial to disease diagnosis. Table 7 shows the application of fALFF, DTI and ASL methods in chronic eye diseases.
Summary and Future Prospects
In summary, each magnetic resonance imaging technique has its own characteristics. To summarize the above, both ALFF and fALFF show regional differences in the brain, with high accuracy and repeatability, and do not need to pre-define regions of interest (ROI) (1), while fALFF makes improvements in noise reduction on the basis of ALFF (50); DC is more sensitive to showing the changes of connectivity in the brain network structure and the state changes of each node, so as to show the correlation of each network structure (11, 12); Both ReHo and FC can show the temporal distribution of voxels in brain functional regions (17, 25). ReHo focuses on describing the consistency within regions, while FC focuses on describing the synchronization between regions, but neither of them directly describes the intensity of brain activity in a certain region, that is, activity detection cannot be carried out. VMHC, as a new static FC analysis method, is more sensitive to the changes of functional synchronization between the two hemispheres of the brain (36). VBM focuses on exploring the changes of brain anatomy (51); DTI has irreplaceable advantages in understanding the complex cytoskeleton structure and other fine structures of the brain (43); ASL can track the changes of local blood flow in eye diseases and improve the accuracy of diagnosis.
In recent years, MRI has been increasingly widely used to explore disease-related changes in brain activity and functional connections. It provides a useful imaging index for understanding the mechanism and monitoring the progress of pathological changes in disease. Its role as a disease marker has been confirmed in many studies. Most chronic eye diseases are characterized by occult and chronic progression, which easily leads to missed diagnosis and misdiagnosis. Using MRI, changes in spontaneous brain activity which occur with eye diseases may be detected at an early stage and monitored, and then accurately locate the brain region where lesions occur, and combine clinical symptoms based on the consistent physiological functions of different brain regions to improve the accuracy of diagnosis, aiding both early diagnosis and treatment of chronic eye diseases. However, the application of magnetic resonance imaging as a marker in chronic eye diseases has some limitations, since physiological and hardware-related factors may affect the experimental results. In addition, due to overlapping functions of different brain regions, it may not be possible to accurately locate the affected areas of the diseased brain. Despite these limitations, MRI technology has great potential and scope to provide indicators of onset and progression of chronic eye diseases. With the continuous progress of technology, MRI technology will usher in a broader range of applications, increasing the scope for exploration of chronic eye diseases.
Author Contributions
C-YY was responsible for the writing of the manuscript. RH was responsible for data analysis and the later submission. S-QL was responsible for collecting and screening the data. YS was responsible for the revision of the paper. All authors contributed to the article and approved the submitted version.
Funding
The Central Government Guides Local Science and Technology Development Foundation (20211ZDG02003); Key Research Foundation of Jiangxi Province (No: 20181BBG70004); Excellent Talents Development Project of Jiangxi Province (20192BCBL23020); Natural Science Foundation of Jiangxi Province (20181BAB205034); Grassroots Health Appropriate Technology Spark Promotion Plan Project of Jiangxi Province (No: 20188003); Health Development Planning Commission Science Foundation of Jiangxi Province (No: 20201032); Health Development Planning Commission Science TCM Foundation of Jiangxi Province (No: 2018A060).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Zuo XN, Di Martino A, Kelly C, Shehzad ZE, Gee DG, Klein DF, et al. The oscillating brain: complex and reliable. Neuroimage. (2010) 49:1432–45. doi: 10.1016/j.neuroimage.2009.09.037
2. Logothetis NK, Pauls J, Augath M, Trinath T, Oeltermann A. Neurophysiological investigation of the basis of the fMRI signal. Nature. (2001) 412:150–7. doi: 10.1038/35084005
3. Huang X, Zhou FQ, Hu YX, Xu XX, Zhou X, Zhong YL, et al. Altered spontaneous brain activity pattern in patients with high myopia using amplitude of low-frequency fluctuation: a resting-state fMRI study. Neuropsychiatr Dis Treat. (2016) 12:2949–56. doi: 10.2147/NDT.S118326
4. Wang ZL, Zou L, Lu ZW, Xie XQ, Jia ZZ, Pan CJ, et al. Abnormal spontaneous brain activity in type 2 diabetic retinopathy revealed by amplitude of low-frequency fluctuations: a resting-state fMRI study. Clin Radiol. (2017) 72:340.e1–7. doi: 10.1016/j.crad.2016.11.012
5. Li Q, Huang X, Ye L, Wei R, Zhang Y, Zhong YL, et al. Altered spontaneous brain activity pattern in patients with late monocular blindness in middle-age using amplitude of low-frequency fluctuation: a resting-state functional MRI study. Clin Interv Aging. (2016) 11:1773–80. doi: 10.2147/CIA.S117292
6. Tan G, Huang X, Zhang Y, Wu AH, Zhong YL, Wu K, et al. A functional MRI study of altered spontaneous brain activity pattern in patients with congenital comitant strabismus using amplitude of low-frequency fluctuation. Neuropsychiatr Dis Treat. (2016) 12:1243–50. doi: 10.2147/NDT.S104756
7. Min YL, Su T, Shu YQ, Liu WF, Chen LL, Shi WQ, et al. Altered spontaneous brain activity patterns in strabismus with amblyopia patients using amplitude of low-frequency fluctuation: a resting-state fMRI study. Neuropsychiatr Dis Treat. (2018) 14:2351–9. doi: 10.2147/NDT.S171462
8. Wang Y, Shao Y, Shi WQ, Jiang L, Wang XY, Zhu PW, et al. The predictive potential of altered spontaneous brain activity patterns in diabetic retinopathy and nephropathy. EPMA J. (2019) 10:249–59. doi: 10.1007/s13167-019-00171-4
9. Shi WQ, Tang LY, Lin Q, Li B, Jiang N, Zhu PW, et al. Altered spontaneous brain activity patterns in diabetic patients with vitreous hemorrhage using amplitude of lowfrequency fluctuation: a restingstate fMRI study. Mol Med Rep. (2020) 22:2291–9. doi: 10.3892/mmr.2020.11294
10. Peng ZY, Liu YX Li B, Ge QM, Liang RB Li QY, et al. Altered spontaneous brain activity patterns in patients with neovascular glaucoma using amplitude of low-frequency fluctuations: a functional magnetic resonance imaging study. Brain Behav. (2021) 1:e02018. doi: 10.1002/brb3.2018
11. Wu GR, Stramaglia S, Chen H, Liao W, Marinazzo D. Mapping the voxel-wise effective connectome in resting state FMRI. PLoS ONE. (2013) 8:e73670. doi: 10.1371/journal.pone.0073670
12. Xu QH Li QY, Yu K, Ge QM, Shi WQ Li B, et al. Altered brain network centrality in patients with diabetic optic neuropathy: a resting-state fmri study. Endocr Pract. (2020). doi: 10.4158/EP-2020-0045
13. Tan G, Dan ZR, Zhang Y, Huang X, Zhong YL, Ye LH, et al. Altered brain network centrality in patients with adult comitant exotropia strabismus: A resting-state fMRI study. J Int Med Res. (2018) 46:392–402. doi: 10.1177/0300060517715340
14. Wang Y, Jiang L, Wang XY, Chen W, Shao Y, Chen QK, et al. Evidence of altered brain network centrality in patients with diabetic nephropathy and retinopathy: an fMRI study using a voxel-wise degree centrality approach. Ther Adv Endocrinol Metab. (2019) 10:2042018819865723. doi: 10.1177/2042018819865723
15. Huang X, Li HJ, Peng DC, Ye L, Yang QC, Zhong YL, et al. Altered brain network centrality in patients with late monocular blindness: a resting-state fMRI study. Arch Med Sci. (2019) 15:1301–7. doi: 10.5114/aoms.2019.87133
16. Zang Y, Jiang T, Lu Y, He Y, Tian L. Regional homogeneity approach to fMRI data analysis. Neuroimage. (2004) 22:394–400. doi: 10.1016/j.neuroimage.2003.12.030
17. Tononi G, McIntosh AR, Russell DP, Edelman GM. Functional clustering: identifying strongly interactive brain regions in neuroimaging data. Neuroimage. (1998) 7:133–49. doi: 10.1006/nimg.1997.0313
18. Lin X, Ding K, Liu Y, Yan X, Song S, Jiang T. Altered spontaneous activity in anisometropic amblyopia subjects: revealed by resting-state FMRI. PLoS ONE. (2012) 7:e43373. doi: 10.1371/journal.pone.0043373
19. Huang X, Li SH, Zhou FQ, Zhang Y, Zhong YL, Cai FQ, et al. Altered intrinsic regional brain spontaneous activity in patients with comitant strabismus: a resting-state functional MRI study. Neuropsychiatr Dis Treat. (2016) 12:1303–8. doi: 10.2147/NDT.S105478
20. Huang X, Ye CL, Zhong YL, Ye L, Yang QC Li HJ, et al. Altered regional homogeneity in patients with late monocular blindness: a resting-state functional MRI study. Neuroreport. (2017) 28:1085–91. doi: 10.1097/WNR.0000000000000855
21. Shao Y, Li QH Li B, Lin Q, Su T, Shi WQ, et al. Altered brain activity in patients with strabismus and amblyopia detected by analysis of regional homogeneity: a restingstate functional magnetic resonance imaging study. Mol Med Rep. (2019) 19:4832–40. doi: 10.3892/mmr.2019.10147
22. Liao XL, Yuan Q, Shi WQ Li B, Su T, Lin Q, et al. Altered brain activity in patients with diabetic retinopathy using regional homogeneity: a resting-state fmri study. Endocr Pract. (2019) 25:320–7. doi: 10.4158/EP-2018-0517
23. Shi H, Wang Y, Liu X, Xia L, Chen Y, Lu Q, et al. Cortical alterations by the abnormal visual experience beyond the critical period: a resting-state fMRI study on constant exotropia. Curr Eye Res. (2019) 44:1386–92. doi: 10.1080/02713683.2019.1639767
24. Maldjian JA, Davenport EM, Whitlow CT Graph theoretical analysis of resting-state MEG data: Identifying interhemispheric connectivity and the default mode. Neuroimage. (2014). 96:88–94. doi: 10.1016/j.neuroimage.2014.03.065
25. Su T, Yuan Q, Liao XL, Shi WQ, Zhou XZ, Lin Q, et al. Altered intrinsic functional connectivity of the primary visual cortex in patients with retinal vein occlusion: a resting-state fMRI study. Quant Imaging Med Surg. (2020) 10:958–69. doi: 10.21037/qims.2020.03.24
26. Ding K, Liu Y, Yan X, Lin X, Jiang T. Altered functional connectivity of the primary visual cortex in subjects with amblyopia. Neural Plast. (2013) 2013:612086. doi: 10.1155/2013/612086
27. Zhu PW, Huang X, Ye L, Jiang N, Zhong YL, Yuan Q, et al. Altered intrinsic functional connectivity of the primary visual cortex in youth patients with comitant exotropia: a resting state fMRI study. Int J Ophthalmol. (2018) 11:668–73.
28. Yan X, Wang Y, Xu L, Liu Y, Song S, Ding K, et al. Altered functional connectivity of the primary visual cortex in adult comitant strabismus: a resting-state functional MRI study. Curr Eye Res. (2019) 44:316–23. doi: 10.1080/02713683.2018.1540642
29. Wu YY, Wang SF, Zhu PW, Yuan Q, Shi WQ, Lin Q, et al. Altered intrinsic functional connectivity of the primary visual cortex in patients with neovascular glaucoma: a resting-state functional magnetic resonance imaging study. Neuropsychiatr Dis Treat. (2020) 16:25–33. doi: 10.2147/NDT.S228606
30. Yu K, Lin Q, Ge QM Yu CY, Li QY, Pan YC, et al. Measuring functional connectivity in patients with strabismus using stationary functional magnetic resonance imaging: a resting-state network study. Acta Radiol. (2021) 10:284185120983978. doi: 10.1177/0284185120983978
31. Ashburner J, Friston KJ. Voxel-based morphometry–the methods. Neuroimage. (2000) 11:805–21. doi: 10.1006/nimg.2000.0582
32. Chan ST, Tang KW, Lam KC, Chan LK, Mendola JD, Kwong KK. Neuroanatomy of adult strabismus: a voxel-based morphometric analysis of magnetic resonance structural scans. Neuroimage. (2004) 22:986–94. doi: 10.1016/j.neuroimage.2004.02.021
33. Mendola JD, Conner IP, Roy A, Chan ST, Schwartz TL, Odom JV, et al. Voxel-based analysis of MRI detects abnormal visual cortex in children and adults with amblyopia. Hum Brain Mapp. (2005) 25:222–36. doi: 10.1002/hbm.20109
34. Ouyang J, Yang L, Huang X, Zhong YL, Hu PH, Zhang Y, et al. The atrophy of white and gray matter volume in patients with comitant strabismus: evidence from a voxel-based morphometry study. Mol Med Rep. (2017) 16:3276–82. doi: 10.3892/mmr.2017.7006
35. Shi WQ, He Y, Li QH, Tang LY Li B, Lin Q, et al. Central network changes in patients with advanced monocular blindness: a voxel-based morphometric study. Brain Behav. (2019) 9:e01421. doi: 10.1002/brb3.1421
36. Zuo XN, Kelly C, Di Martino A, Mennes M, Margulies DS, Bangaru S, et al. Growing together and growing apart: regional and sex differences in the lifespan developmental trajectories of functional homotopy. J Neurosci. (2010) 30:15034–43. doi: 10.1523/JNEUROSCI.2612-10.2010
37. Hou F, Liu X, Zhou Z, Zhou J, Li H. Reduction of interhemispheric functional brain connectivity in early blindness: a resting-state fMRI study. Biomed Res Int. (2017) 2017:6756927. doi: 10.1155/2017/6756927
38. Shao Y, Bao J, Huang X, Zhou FQ, Ye L, Min YL, et al. Comparative study of interhemispheric functional connectivity in left eye monocular blindness versus right eye monocular blindness: a resting-state functional MRI study. Oncotarget. (2018) 9:14285–95. doi: 10.18632/oncotarget.24487
39. Zhang Y, Zhu PW, Huang X, Ma MY, Shi WQ, Tao Q. Alternations of interhemispheric functional connectivity in patients with comitant exotropia: a resting state fMRI study. Int J Clin Exp Med. (2018) 10:10966–73. Available online at: http://210.35.251.113/s/us/e-century/G.https/web/journal_search.php?journal=ijcem&q=Alternations%20of%20interhemispheric%20functional%20connectivity%20in%20patients%20with%20comitant%20exotropia%3A%20a%20resting%20state%20fMRI%20study
40. Wang Y, Wang X, Chen W, Shao Y, Zhou J, Chen Q, et al. Brain function alterations in patients with diabetic nephropathy complicated by retinopathy under resting state conditions assessed by voxel-mirrored homotopic connectivity. Endocr Pract. (2020) 26:291–8. doi: 10.4158/EP-2019-0355
41. Guo Z, Liu X, Li J, Wei F, Hou H, Chen X, et al. Fractional amplitude of low-frequency fluctuations is disrupted in Alzheimer's disease with depression. Clin Neurophysiol. (2017) 128:1344–9. doi: 10.1016/j.clinph.2017.05.003
42. Zou QH, Zhu CZ, Yang Y, Zuo XN, Long XY, Cao QJ, et al. An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF. J Neurosci Methods. (2008) 172:137–41. doi: 10.1016/j.jneumeth.2008.04.012
43. Beaulieu C. The basis of anisotropic water diffusion in the nervous system - a technical review. NMR Biomed. (2002) 15:435–55. doi: 10.1002/nbm.782
44. Pierpaoli C, Basser PJ. Toward a quantitative assessment of diffusion anisotropy. Magn Reson Med. (1996) 36:893–906. doi: 10.1002/mrm.1910360612
45. Fang JW Yu YJ, Tang LY, Chen SY, Zhang MY, Sun T, et al. Abnormal fractional amplitude of low-frequency fluctuation changes in patients with monocular blindness: a functional magnetic resonance imaging (MRI) study. Med Sci Monit. (2020) 26:e926224. doi: 10.12659/MSM.926224
46. Zhang YQ, Peng MY, Wu SN, Yu CY, Chen SY, Tan SW, et al. Fractional amplitude of low-frequency fluctuation in patients with neovascular glaucoma: a resting-state functional magnetic resonance imaging study. Quant Imaging Med Surg. (2021) 10:855. doi: 10.21037/qims-20-855
47. Gümüstas S, Altintas Ö, Anik Y, Kaya A, Altintas L, Inan N, et al. Anterior visual pathways in amblyopia: quantitative assessment with diffusion tensor imaging. J Pediatr Ophthalmol Strabismus. (2013) 50:369–74. doi: 10.3928/01913913-20131125-04
48. Huang X, Li HJ, Zhang Y, Peng DC, Hu PH, Zhong YL, et al. Microstructural changes of the whole brain in patients with comitant strabismus:evidence from a diffusion tensor imaging study. Neuropsychiatr Dis Treat. (2016) 12:2007–14. doi: 10.2147/NDT.S108834
49. Huang X, Zhou S, Su T, Ye L, Zhu PW, Shi WQ, et al. Resting cerebral blood flow alterations specific to the comitant exophoria patients revealed by arterial spin labeling perfusion magnetic resonance imaging. Microvasc Res. (2018) 120:67–73. doi: 10.1016/j.mvr.2018.06.007
50. Wallis I, Ellis L, Suh K, Pfenninger KH, et al. Immunolocalization of a neuronal growth-dependent membrane glycoprotein. J Cell Biol. (1985) 101:1990–8. doi: 10.1083/jcb.101.5.1990
Keywords: magnetic resonance imaging, chronic eye diseases, markers, neurology, application
Citation: Yu C-Y, Huang R, Li S-Q and Shao Y (2022) Neuroimaging Markers of Chronic Eye Diseases and Their Application Values. Front. Neurol. 13:854605. doi: 10.3389/fneur.2022.854605
Received: 14 January 2022; Accepted: 11 May 2022;
Published: 14 June 2022.
Edited by:
Yuzhen Xu, Tongji University, ChinaReviewed by:
Benito de Celis Alonso, Meritorious Autonomous University of Puebla, MexicoHe Wang, Xuzhou Medical University, China
Yuan Liu, University of Miami Health System, United States
Copyright © 2022 Yu, Huang, Li and Shao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yi Shao, ZnJlZWJlZTk5QDE2My5jb20=
†These authors have contributed equally to this work
 Chen-Yu Yu†
Chen-Yu Yu† Rong Huang
Rong Huang Yi Shao
Yi Shao