- 1Department of Neurosurgery, Huashan Hospital, Shanghai Medical School, Fudan University, Shanghai, China
- 2Department of Neurosurgery, Hai'an People's Hospital, Nantong, China
- 3Department of Neurosurgery, Second Affiliated Hospital of Jiaxiang University, Jiaxing, China
- 4Department of Neurosurgery, Kashgar Prefecture Second People's Hospital, Kashgar, China
Intracranial pseudoaneurysms account for about 1% of intracranial aneurysms with a high mortality. The natural history of intracranial pseudoaneurysm is not well-understood, and its management remains controversial. This review provides an overview of the etiology, pathophysiology, clinical presentation, imaging, and management of intracranial pseudoaneurysms. Especially, this article emphasizes the factors that should be considered for the most appropriate management strategy based on the risks and benefits of each treatment option.
Introduction
Intracranial pseudoaneurysm is a rare entity and represents about 1% of all intracranial aneurysms, with an associated mortality of 20% or higher (1). The most common cause of pseudoaneurysm is trauma (2). Other causes are iatrogenic, infectious disease, radiation exposure, connective tissue disease, and sometimes they occur spontaneously (3–6). A pseudoaneurysm or false aneurysm is the product of damaging vessel wall resulting in an encapsulated hematoma in communication with the ruptured artery. Clinical presentations may vary depending on the rupture status, location, and size of the intracranial pseudoaneurysm (7). If untreated, the mortality rate for patients with intracranial pseudoaneurysm can reach high up to 50% due to delayed rupture and disastrous bleeding (1, 8, 9). Therefore, early diagnosis and efficient treatment are mandatory. In this review, we provide a comprehensive evaluation of the risks and benefits of different treatment options available for pseudoaneurysms, such as observation, microsurgical clipping, and endovascular embolization. Besides, the etiology, pathophysiology, clinical presentation, and imaging of intracranial pseudoaneurysms are also discussed.
Classification
Pseudoaneurysms account for about 1% of aneurysms in adults; however, the incidence rate in the pediatric group is more than 19% (9, 10). Anatomical anomalies, venous sinus thrombosis, multiple surgeries, and prior radiotherapy increase the incidence rate of the pseudoaneurysm. Intracranial pseudoaneurysms can be classified as traumatic, infectious, iatrogenic, and other types.
Traumatic Pseudoaneurysms
Head trauma is the most common cause of intracranial pseudoaneurysms. Closed or penetrating head trauma to cerebral blood vessels, which lead to pseudoaneurysm, could be classified as direct or indirect (1). A penetrating wound resulting from a variety of weapons and cutlery leads to direct vascular injury. Indirect vascular trauma is often encountered in seriously closed brain injuries, such as traffic accidents and bony prominences during major brain shifts.
Infectious Pseudoaneurysms
Infectious pseudoaneurysms can be caused by bacteria, tuberculous bacilli, or fungi (11–13). In comparison with other intracranial aneurysms, infectious aneurysms have a slight preference for younger people. Ruptured aneurysms have a higher rate of mortality. Most of the infectious pseudoaneurysms are located in the anterior circulation, and those aneurysms can be multiple in many cases. The definition of infectious pseudoaneurysms should be based on the angiographic features and demonstration of infection.
Iatrogenic Pseudoaneurysms
In addition to trauma and infection, iatrogenic vascular injury is another important cause of intracranial pseudoaneurysms. They generally involve the internal carotid artery (ICA) due to ICA injury after transsphenoidal or transcranial resection of sellar region tumors (6, 14–19). Iatrogenic pseudoaneurysm are less common in the anterior cerebral artery (12, 20–24), the basilar artery (18, 25–27), and the middle cerebral artery (22, 28–30). Pseudoaneurysms after mechanical thrombectomy or stent angioplasty is one of the potential complications associated with the endovascular procedure (31, 32). Although rare, we should raise the suspicion for this potentially lethal complication.
Other Types of Pseudoaneurysms
Other causes of intracranial pseudoaneurysms include Marfan's syndrome, fibromuscular dysplasia, vasculitis, rupture of true cerebral aneurysm or arteriovenous malformation, associated with moyamoya disease, and radiotherapy (4, 5, 33, 34). Dissecting pseudoaneurysms and blood blister-like aneurysms were out of the scope of the discussion.
Pathophysiology
Compared with the extracranial arteries, the intracranial arteries are thinner and stiffer. They have a thinner media and adventitia, absence of an external elastic lamina, and possess a thicker internal elastic lamina (35, 36). These features make the intracranial arteries more vulnerable to trauma.
Traumatic intracranial aneurysms can be histologically categorized as true or false. True aneurysms usually develop following a partial disruption of the arterial wall. The intima, internal elastic lamina, and media are damaged, whereas the adventitia is intact (1, 3, 8, 36, 37). False aneurysms or pseudoaneurysms result from disruption of the entire arterial wall (Figure 1). A contained hematoma forms outside the vessel, being restricted by perivascular connective tissues. However, it continues to communicate with the injured artery and is more likely to rebleed.
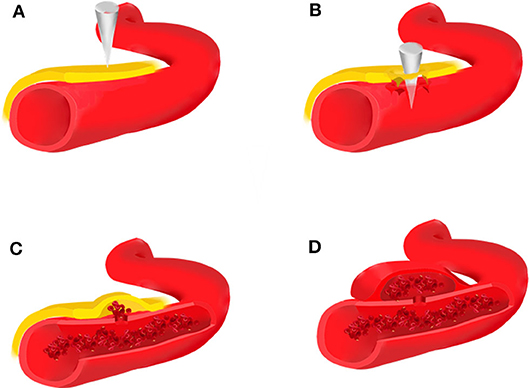
Figure 1. Brain trauma leads to rupture of the intima, media, and adventitia of the blood vessel (A,B), forming an organized hematoma cavity (C). When the hematoma forms outside the arterial wall, it continues to communicate with the injured vessel, thus predisposing it to re-bleeding (D).
Iatrogenic pseudoaneurysms can be classified as saccular and fusiform (36). Saccular pseudoaneurysms occur secondary to penetration or complete laceration of the arterial wall. They lack a true wall and are only contained by a fragile layer of connective tissue (7, 19). Fusiform pseudoaneurysm may result from thinning of the adventitia during surgical peeling of tumor from the adjacent vessel. In comparison to saccular pseudoaneurysms, they usually do not rupture.
A pseudoaneurysm can also form at the tip of the “true” aneurysm (34, 38). We speculated that it might be the thick hematoma around the aneurysm (38). The temporary disorder of cerebrospinal fluid circulation may also play a significant role in the formation of pseudoaneurysm, since the blood extravasating from the vessel could gather into a hematoma but not diffuse into the cerebrospinal fluid. Thus, a sufficient volume of the subarachnoid pool may result in the occurrence of the pseudoaneurysm (34).
Clinical Presentation
Intracranial pseudoaneurysms may present with intracranial hemorrhage, epistaxis, headaches, seizures, neurological deficits, and associated with other cerebrovascular diseases.
Intracranial hemorrhage is the most common presentation, manifesting as acute hemorrhage associated with the initial injury or in a delayed manner (39). Intraoperative arterial bleeding occurred in the majority of patients with iatrogenic vascular injury. Patients with no evidence of vascular injury during the operation may suffer postoperative or delayed hemorrhage, including intracerebral, intraventricular, and subarachnoid hemorrhage. Pseudoaneurysms of the middle meningeal artery typically are associated with epidural or subdural hematoma (40, 41).
Epistaxis is the common symptom of intracavernous ICA pseudoaneurysms. The intracavernous ICA is close to the sphenoid sinus, mostly bulging into the lateral sinus wall (37). A congenitally thin or even absent bony structure covering the cavernous ICA within the sphenoid sinus may provide less protection against bony erosion (42). Massive epistaxis may be caused by erosion of the lateral wall of the sphenoid sinus. We also cannot exclude the internal maxillary artery as another source of hemorrhage (9). Epistaxis may be delayed in 7 days to 8 months after trauma or iatrogenic intracavernous ICA injury. The initial episodes of epistaxis may be mild and not fatal. However, recurrent bleeding can lead to fatal blood loss. Thus, we should not neglect it, in order to prevent delayed diagnosis and treatment.
Focal neurological deficits are often associated with traumatic pseudoaneurysms of the ICA. Due to its proximity to other cavernous structures, including cranial nerves II, III, IV, V1, V2, and VI. Traumatic ICA pseudoaneurysms may present with cranial nerve deficits, unilateral blindness, or a carotid-cavernous fistula associated to skull base fractures (9, 11, 37, 43). Other symptoms include headache, seizures, neck rigidity, decreased mental state, paralysis, or reduced level of consciousness.
Imaging
Due to the high mortality related to pseudoaneurysm rupture, early diagnosis is mandatory. If intraoperative arterial bleeding occurs, iatrogenic vascular injury should be suspected. Angiography should be used for patients with perioperative hemorrhage or epistaxis. Digital subtraction angiography (DSA) is still the gold standard for the diagnosis of intracranial pseudoaneurysms since CTA and MRA have limited sensitivity for the detection of small aneurysms (44) DSA often demonstrates a globular shaped aneurysmal sac without a neck (5). Delayed filling and stagnation of contrast agents are the features of the pseudoaneurysm. If the initial imaging is negative, angiography shoud be repeated because pseudoaneurysms often develop days to weeks after injury. The optimal time interval between angiographies is still a matter of debate. Some studies reported negative initial angiograms within several hours or days of trauma. Follow-up angiograms showed an aneurysm weeks to months later. Therefore, initial angiography was suggested to be performed 1 or 2 weeks after vascular injury to avoid missed diagnoses (39, 44). However, another study recommends it 6 and 12 months postoperatively (15).
Management
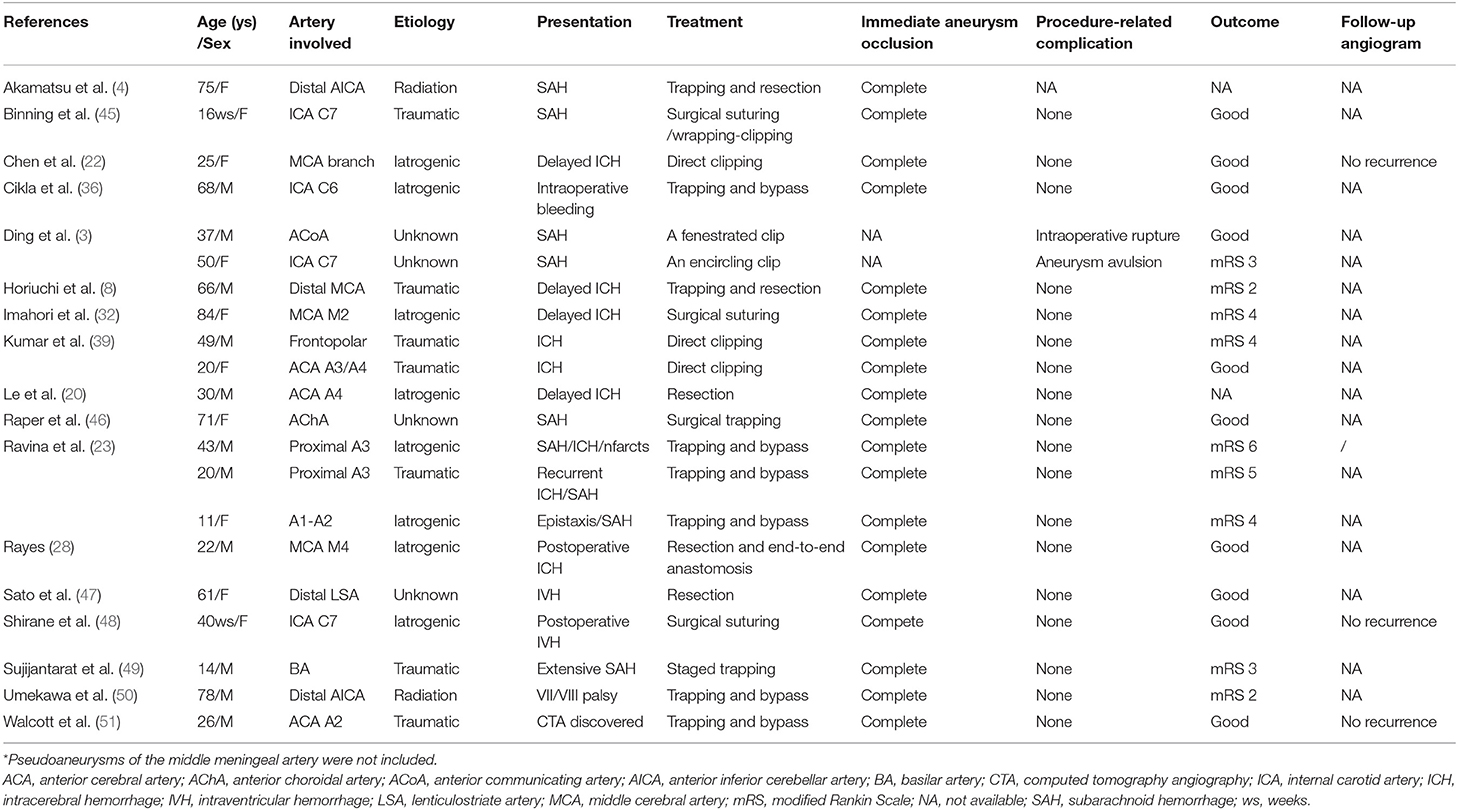
As the causes of pseudoaneurysms are different, treatment options are challenging. Management of intracranial pseudoaneurysms includes microsurgery, embolization, and conservative treatment (Tables 1, 2).
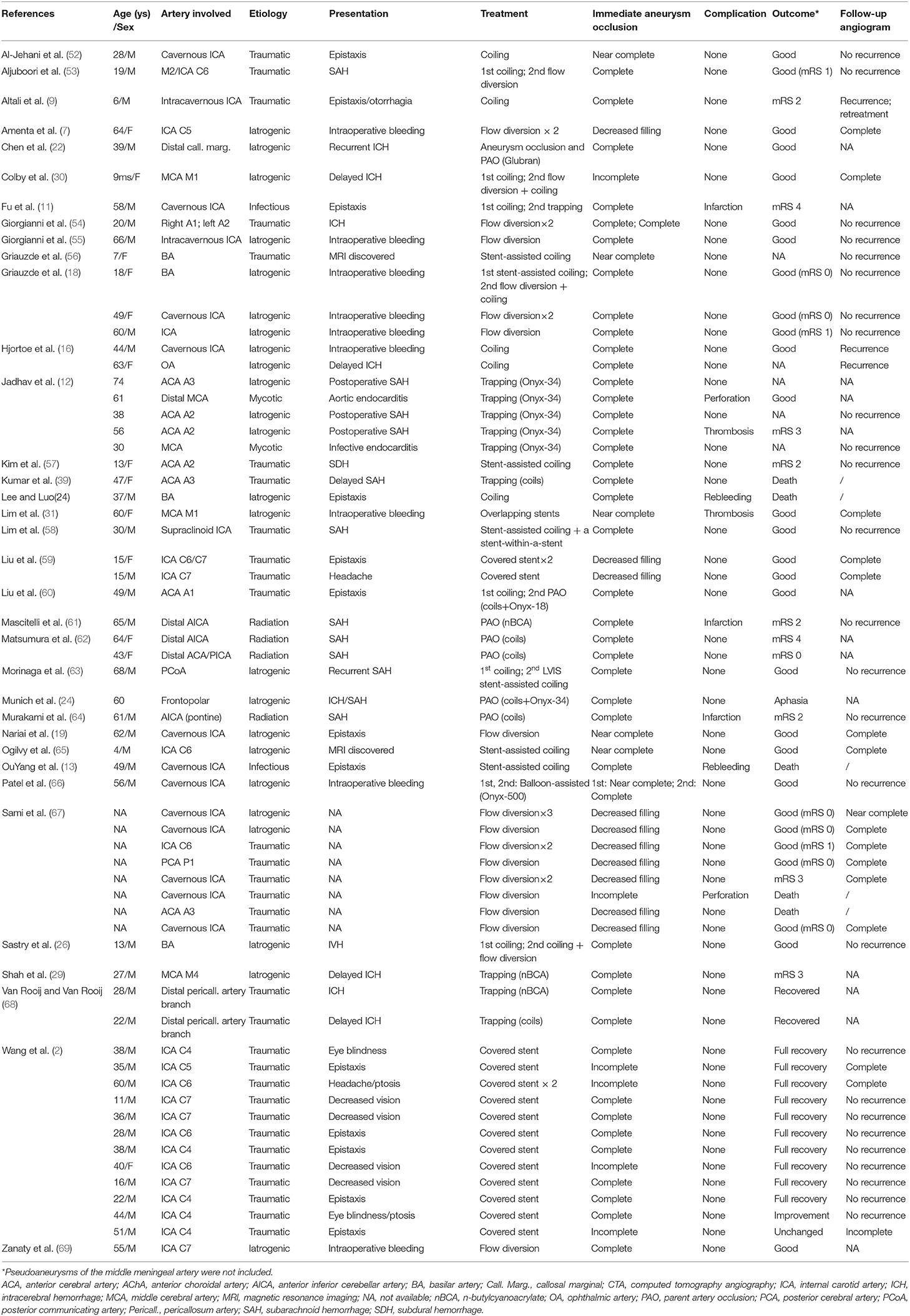
Table 2. Literature review of intracranial pseudoaneurysms treated with endovascular embolization from 2010*.
Microsurgery
Surgical intervention is typically reserved for the lesions of difficult catheterization or failed endovascular therapy, and presence of significant mass effect in ruptured pseudoaneurysms with acute hematoma, usually followed by clot evaculation and/or decompressive craniectomy. Direct surgery to treat pseudoaneurysms of the cavernous and petrous ICA is difficult. In the distal branch of intracranial arteries, such as the pericallosal artery, surgery still should be considered as an irreplaceable option. Surgical options include direct clipping, suturing, wrapping-clipping, ligation of the parent artery, and trapping with or without bypass (Table 1). However, different experts hold a variety of opinions on surgical strategies for pseudoaneurysms. Direct clipping of the aneurysmal neck may not be feasible because of the lack of a true vessel wall that makes clipping threatening and challenging (9, 45, 53). It often results in aneurysm avulsion and intraoperative bleeding due to the high fragility of the pseudowall (22). The orifice or defect can be repaired with direct microsurgical suturing (32, 45, 48). Subsequent wrapping-clipping supports the fragile wall and maintains the connectivity of the parent artery (45). Ligation of the parent artery can result in distal ischemic complications. Moreover, it may not prevent the rupture of pseudoaneurysms due to collateral retrograde flow into the lesion (37). Trapping, in which clips are placed on the parent vessel, both proximal and distal to the aneurysm, is the definitive treatment modality to eliminate the aneurysm. Trapping with or without bypass revascularization depends on the status of collateral supply. A low-flow bypass is often used to treat distal pseudoaneurysms (50), while a high-flow bypass is recommended for ICA pseudoaneurysms (36, 70). Resection of the aneurysm and end-to-end anastomosis is another possible treatment (28).
Endovascular Embolization
With the advances in techniques and materials, endovascular treatment has been an alternative to surgery for the treatment of intracranial pseudoaneurysms. Endovascular procedures include coiling, stent-assisted coiling, occlusion of the parent artery with or without aneurysm, and flow-diversion. The choice of endovascular technique is based on the location of the pseudoaneurysm, vascular anatomy, and clinical status of the patient.
Packing of the pseudoaneurysm with coils is available for those cases with a narrow neck pseudoaneurysm (52, 71). Because of the fragility of the pseudoaneurysm wall, it has the risk of microcatheter or coil perforation during the procedure. The advantage of selective pseudoaneurysm embolization is the preservation of the parent artery (Figure 2). However, pseudoaneurysm recurrence is still a major issue for patients treated with simple coiling (7, 9, 11, 14, 16, 26, 30, 53, 60, 63). Due to coil impaction into the thrombus (14), flow pulsatility may force into the interstices of the coil mass and lead to recurrent bleeding (27).
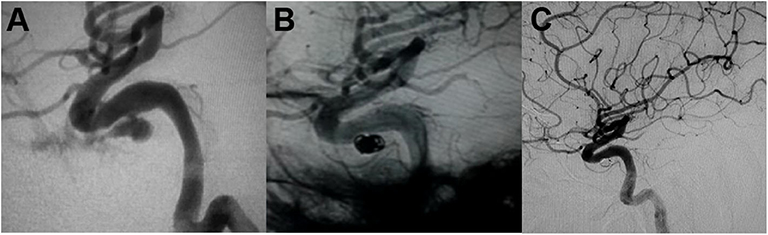
Figure 2. (A) Angiography demonstrated the intracavernous iatrogenic pseudoaneurysm of the left internal carotid artery (ICA). (B) The pseudoaneurysm was treated by endovascular coiling. (C) Angiogram at 4-month follow-up showed no evidence of aneurysmal filling [adapted from Lin et al. (72)].
Therefore, some studies suggested that occlusion of the parent artery and pseudoaneurysm may be the preferred therapy for distal pseudoaneurysms (12, 73). Because of having good collateral supply or retrograde flow from the distal to the trapped segment, occlusion of the parent artery may be safe in distal ACA (24, 68, 73). Coils (39, 73), or liquid embolization agents including glubran (22), n-butylcyanoacrylate (29, 68), and Onyx (12, 24, 74) can be used in parent artery occlusion. Onyx treatment is especially suitable for pseudoaneurysms with minuscule vessel wall as to avoid coiling. However, occlusion of the parent artery is not recommended for pseudoaneurysms of the ICA. Although having negative balloon occlusion test, 22% of patients develop ischemic complications following parent artery occlusion (Figure 3) (16, 52, 75).
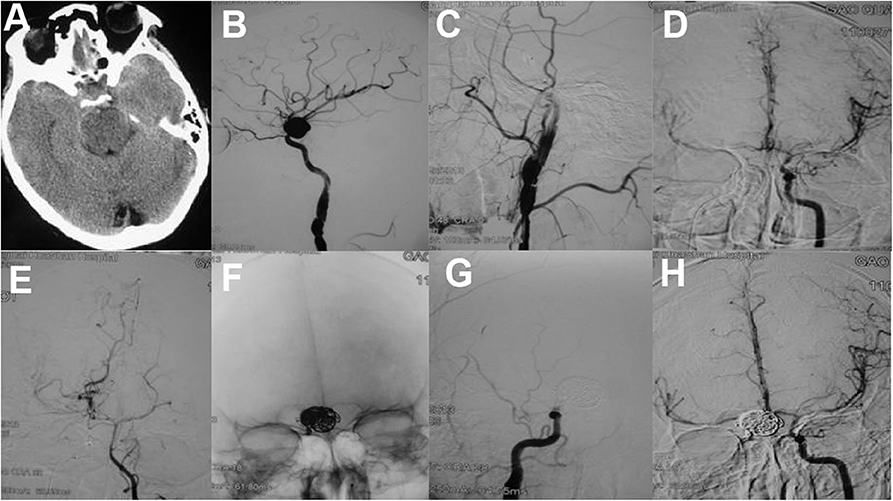
Figure 3. (A) An axial CT scan showed skull bone fracture and traumatic subarachonoid hemorrhage. (B) Two weeks later, the patient suffered rehemorrhage. A lateral cerebral angiogram of the right ICA demonstrated a large pseudoaneurysm at the C6 segment and a dissection of the C1 segment. Angiogram after balloon occlusion test (C) showed good compensation from the anterior communicating artery (D) and posterior communicating artery (E,F) The pseudoaneurysm and parent artery were trapped with six detachable coils. Postoperative right (G) and left (H) carotid angiograms showed exclusion of the pseudoaneurysm from the circulation [adapted from Lin et al. (72)].
Stent-assisted coiling is a promising treatment option for wide-necked pseudoaneurysms (56–58, 65). It allows for the preservation of the parent artery and avoidance of bypass surgery. However, aneurysm recanalization is not uncommon after single stent-assisted coiling (13, 18, 76). Stent-assisted coil embolization followed by stent-within-a-stent technique has been reported as an effective treatment for pseudoaneurysms (58). Overlapping stents with coils effectively prevent rebleeding and regrowth of the pseudoaneurysm. The overlapping stents may divert the flow away from the pseudoaneurysm, accelerate intraaneurysmal thrombosis, and reconstruct the parent artery by promoting neointima formation along the stent (31, 52, 58, 77).
Another reconstructive endovascular treatment modality is covered stent implantation (2, 59, 78). Endovascular deployment of covered stents can exclude blood flow through the stent as a physical barrier and keep the normal anatomic flow through the parent artery (Figure 4). Compared with the uncovered stents, covered stents decrease the incidence rate of neointimal proliferation and restenosis, at the same time, decrease embolization risk caused by thrombus debris during the process of stent deployment. However, the flexibility of covered stents and the stiffness of the delivery system are the main limitations for its usage in the tortuous ICA, which may result in dissection and vasospasm. Moreover, the occlusion of important perforators by the covered stent may occur when the pseudoaneurysm originates too close to the origin of the ophthalmic artery, the posterior communicating artery, or the anterior choroidal artery.
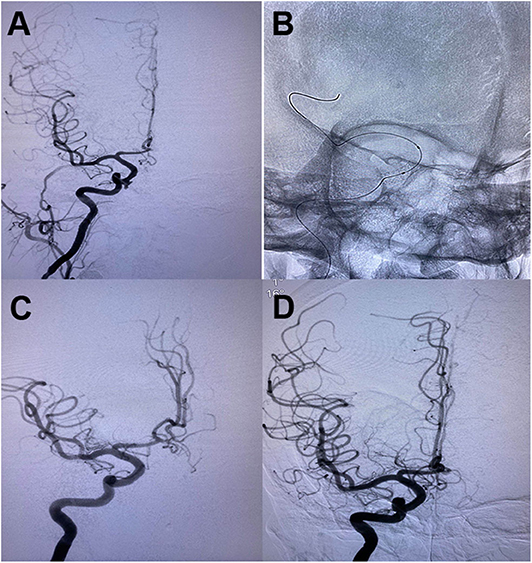
Figure 4. (A) Oblique cerebral angiogram showed a pseudoaneurysm in the cavernous segment of righ ICA following endoscopic transsphenoid surgery. (B) A 4*13 mm Willis covered stent was deployed across the pseudoaneurysm. (C) The control angiogram demonstrated complete obliteration of the pseudoaneurysm with preservation of carotid artery patency. (D) A follow-up angiogram showed no recanalization of the aneurysm and patentcy of the parent artery.
Recently, a flow-diverting strategy has been shown to be a promising treatment modality for patients with intracranial pseudoaneurysm. Flow-diverting stent reduces blood flow into the aneurysm, thus promoting thrombosis. It also provides a scaffold for endothelialization and reconstruction of the vessel wall. Previous studies have shown that pseudoaneurysms treated with flow-diverting stents have high complete occlusion rates and low complication rates (6, 7, 18, 19, 26, 30, 53–55, 67, 69, 79). However, the main limitation of flow-diverting stents is delayed aneurysm obliteration due to a lack of immediate thrombosis. It may take weeks for complete aneurysm occlusion, which leaves patients at risk for rebleeding during this time (6, 7, 19, 79). In addition, dural antiplatelet therapy after flow-diverting stent placement may increase the risk of postoperative intracranial hematomas. It should be used judiciously in the setting of ruptured pseudoaneurysms.
Conservative Treatment
Although high mortality rates of pseudoaneurysms were reported, these data are based on a review of the literature and a collection of only case reports. There is no large sample of pseudoaneurysms in single or multiple centers. Therefore, the true natural history of these pseudoaneurysms is unclear. Complete spontaneous resolution of pseudoaneurysms is considered to be an uncommon occurrence. Previous studies have demonstrated existence of spontaneous resolution in peripheral vessels or intracranial vessels, including the middle meningeal artery (80, 81), basilar artery (82–84), posterior cerebral artery (85) and pericallosal artery (39). The mechanism of spontaneous occlusion remains unclear. It may be due to vascular remodeling response to injury as well as spontaneous thrombus formation (84). Those studies suggest that some specific pseudoaneurysms may at least have a potentially benign course with conservative treatment. Observation might be considered in pseudoaneurysms with decreased size and flow in repeated conventional angiography compared with the initial images (84). However, a recent study reported an unusual course of a cerebral pseudoaneurysm (69). The pseudoaneurysm completely disappeared on the second angiogram, but was found to be enlarged on the third angiogram. Therefore, adequate follow-up is mandatory when conservation treatment is considered or even when the lesions have spontaneous obliteration.
An Illustrative Case
We present a case of 45-year-old man harboring an invasive pituitary adenoma, in whom an intracavernous carotid artery tear was caused by aggressive curettage of the left cavernous sinus portion of the lesion. Massive intraoperative bleeding was stopped by surgical packing. Subsequent emergent angiography demonstrated an elliptical shaped pseudoaneurysm located in the intracavernous portion of the left ICA (Figure 2A). The pseudoaneurysm was treated by endovascular coiling (Figure 2B). Complete occlusion of the pseudoaneurysm from the circulation with preservation of the parent artery was achieved with placement of six coils. Angiogram at 4-month follow-up showed no evidence of aneurysmal filling (Figure 2C).
Conclusion
Intracranial pseudoaneurysms are rare pathological entities, representing 1% of all intracranial aneurysms. Rupture of pseudoaneurysm is associated with high rates of morbidity and mortality. Early diagnosis and therapy is critical in those patients with clinical suspicion of a pseudoaneurysm, such as unexplained hemorrhages and epistaxis following a history of head trauma, surgery, or septicemia. Due to spontaneous occlusion of pseudoaneurysms occurred in a few patients, repeated imaging and prompt treatment should be necessary. Endovascular treatments, including coiling with or without stent, covered stent, flow diverting stent, and trapping, should be individualized to aneurysmal location, clinical condition, vascular anatomy, and assessment of the collateral circulation. Microsurgery may be a suitable alternative in cases not amenable to endovascular treatment, yielding favorable outcomes, especially in distal aneurysms or patients with huge hematoma.
Author Contributions
FX and JS: conception and design. YZ and FX drafted the article. ZL: data collection. All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
This study was supported by Grant No. (2019D01C093) from the Natural Science Foundation of Xinjiang Province; Grant No. (2018AD32016) from Jiaxing Science and Technology.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Larson PS, Reisner A, Morassutti DJ, Abdulhadi B, Harpring JE. Traumatic intracranial aneurysms. Neurosurg Focus. (2000) 8:e4. doi: 10.3171/foc.2000.8.1.1829
2. Wang W, Li MH, Li YD, Gu BX, Wang J, Zhang PL, et al. Treatment of traumatic internal carotid artery pseudoaneurysms with the willis covered stent: a prospective study. J Trauma. (2011) 70:816–22. doi: 10.1097/TA.0b013e3181f892af
3. Ding H, You C, Yin H. Nontraumatic and noninfectious pseudoaneurysms on the circle of willis: 2 case reports and review of the literature. Surg Neurol. (2008) 69:414–7. doi: 10.1016/j.surneu.2007.02.040
4. Akamatsu Y, Sugawara T, Mikawa S, Saito A, Ono S, Takayama K, et al. Ruptured pseudoaneurysm following gamma knife surgery for a vestibular schwannoma. J Neurosurg. (2009) 110:543–6. doi: 10.3171/2008.8.JNS08177
5. Brzozowski K, Frankowska E, Piasecki P, Ziecina P, Zukowski P, Bogusławska-Walecka R. The use of routine imaging data in diagnosis of cerebral pseudoaneurysm prior to angiography. Eur J Radiol. (2011) 80:e401–9. doi: 10.1016/j.ejrad.2010.12.019
6. Chen SH, McCarthy DJ, Sheinberg D, Hanel R, Sur S, Jabbour P, et al. Pipeline embolization device for the treatment of intracranial pesudoaneurysms. World Neurosurg. (2019) 127:e86–93. doi: 10.1016/j.wneu.2019.02.135
7. Amenta PS, Starke RM, Jabbour PM, Tjoumakaris SI, Gonzalez LF, Rosenwasser RH, et al. Successful treatment of a traumatic carotid pseudoaneurysm with the pipeline stent: case report and review of the literature. Surg Neurol Int. (2012) 3:160. doi: 10.4103/2152-7806.105099
8. Horiuchi T, Nakagawa F, Miyatake M, Iwashita T, Tanaka Y, Hongo K. Traumatic middle cerebral artery aneurysm: case report and review of the literature. Neurosurg Rev. (2007) 30:263–7. doi: 10.1007/s10143-007-0073-9
9. Altali K, Arruza L, López-Ibor L, Aleo E. Effective coil embolization of intracavernous carotid artery pseudoaneurysm with parental artery preservation following severe head trauma in a pediatric patient. Childs Nerv Syst. (2014) 30:967–70. doi: 10.1007/s00381-013-2312-4
10. Sakata N, Takebayashi S, Kojima M, Masawa N, Suzuki K, Takatama M. Pathology of a dissecting intracranial aneurysm. Neuropathology. (2000) 20:104–8. doi: 10.1046/j.1440-1789.2000.00275.x
11. Fu M, Patel T, Baehring JM, Bulsara KR. Cavernous carotid pseudoaneurysm following transsphenoidal surgery. J Neuroimaging. (2013) 23:319–25. doi: 10.1111/j.1552-6569.2011.00677.x
12. Jadhav AP, Pryor JC, Nogueira RG. Onyx embolization for the endovascular treatment of infectious and traumatic aneurysms involving the cranial and cerebral vasculature. J Neurointerv Surg. (2012) 5:562–5. doi: 10.1136/neurintsurg-2012-010460
13. OuYang M, Huang X, Wang Y. Endovascular treatment of infectious pseudoaneurysm of internal carotid artery. World Neurosurg. (2019) 125:42–3. doi: 10.1016/j.wneu.2019.01.147
14. Kadyrov NA, Friedman JA, Nichols DA, Cohen-Gadol AA, Link MJ, Piepgras DG. Endovascular treatment of an internal carotid artery pseudoaneurysm following transsphenoidal surgery. Case report J Neurosurg. (2002) 96:624–7. doi: 10.3171/jns.2002.96.3.0624
15. Ciceri EF, Regna-Gladin C, Erbetta A, Chiapparini L, Nappini S, Savoiardo M, et al. Iatrogenic intracranial pseudoaneurysms: neuroradiological and therapeutical considerations, including endovascular options. Neurol Sci. (2006) 27:317–22. doi: 10.1007/s10072-006-0703-y
16. Hjortoe S, Wagner A, Cortsen M. Endovascular embolization of intracranial iatrogenic pseudoaneurysms. A report of two cases and review of the literature. Neuradiol J. (2010) 23:479–83. doi: 10.1177/197140091002300420
17. Sylvester PT, Moran CJ, Derdeyn CP, Cross DT, Dacey RG, Zipfel GJ, et al. Endovascular management of internal carotid artery injuries secondary to endonasal surgery: case series and review of the literature. J Neurosurg. (2016) 125:1256–76. doi: 10.3171/2015.6.JNS142483
18. Griauzde J, Ravindra VM, Chaudhary N, Gemmete JJ, Mazur MD, Roark CD, et al. Use of the pipeline embolization device in the treatment of iatrogenic intracranial vascular injuries: a bi-institutional experience. Neurosurg Focus. (2017) 42:E9. doi: 10.3171/2017.3.FOCUS1735
19. Nariai Y, Kawamura Y, Takigawa T, Hyodo A, Suzuki K. Pipeline embolization for an iatrogenic intracranial internal carotid artery pseudoaneurysm after transspehenoidal pituitary tumor surgery: case report and review of the literature. Interv Neuroradiol. (2020) 26:74–82. doi: 10.1177/1591019919874943
20. Le H, Munshi I, Macdonald RL, Wollmann R, Frank J. Traumatic aneurysm resulting from insertion of an intracranial pressure monitor. Case illustration. J Neurosurg. (2001) 95:720. doi: 10.3171/jns.2001.95.4.0720
21. Horowitz M, Sharts M, Levy E, Albright AL, Pollack I. Endovascular management of ventricular catheter-induced anterior cerebral artery false aneurysm: technical case report. Neurosurgery. (2005) 57:E374. doi: 10.1227/01.NEU.0000168016.02106.0C
22. Chen Z, Zhang J, Miao H, Niu Y, Feng H, Zhu G. Delayed rupture of iatrogenic cerebral pseudoaneurysms after neurosurgical procedures: report of two cases. Clin Neurol Neurosurg. (2013) 115:1552–4. doi: 10.1016/j.clineuro.2012.12.024
23. Ravina K, Strickland BA, Rennert RC, Chien M, Mack WJ, Amar AP, et al. A3-A3 anastomosis in the management of complex anterior cerebral artery aneurysms: experience with in situ bypass and lessons learned from pseudoaneurysm cases. Oper Neurosurg. (2019) 17:247–60. doi: 10.1093/ons/opy334
24. Munich SA, Cress MC, Rangel-Castilla L, Krishna C, Siddiqui AH, Snyder KV. Importance of repeat angiography in the diagnosis of iatrogenic anterior cerebral artery territory pseudoaneurysm following endoscopic sinus surgery. J Neurointerv Surg. (2016) 8:e20. doi: 10.1136/neurintsurg-2015-011693.rep
25. Horowitz M, Albright AL, Jungreis C, Levy EI, Stevenson K. Endovascular management of a basilar artery false aneurysm secondary to endoscopic third ventriculostomy: case report. Neurosurgery. (2001) 49:1461–4. doi: 10.1097/00006123-200112000-00031
26. Sastry RA, Koch MJ, Grannan BL, Stapleton CJ, Butler WE, Patel AB. Flow diversion of a recurrent, iatrogenic basilar tip aneurysm in a pediatric patient: case report. J Neurosurg Pediatr. (2018) 21:90–3. doi: 10.3171/2017.7.PEDS17235
27. Lee CH, Luo CB. Pseudoaneurysm of the basilar artery presenting with epistaxis. Br J Neurosurg. (2018) 27:1–2. doi: 10.1080/02688697.2018.1445197
28. Rayes M, Bahgat DA, Kupsky WJ, Mittal S. Middle cerebral artery pseudoaneurysm formation following stereotactic biopsy. Can J Neurol Sci. (2008) 35:664–8. doi: 10.1017/S0317167100009513
29. Shah KJ, Jones AM, Arnold PM, Ebersole K. Intracranial pseudoaneurysm after intracranial pressure monitor placement. J Neurointerv Surg. (2016) 8:e3. doi: 10.1136/neurintsurg-2014-011410.rep
30. Colby GP, Jiang B, Bender MT, Beaty NB, Westbroek EM, Xu R, et al. Pipeline-assisted coil embolization of a large middle cerebral artery pseudoaneurysm in a 9-month-old infant: experience from the youngest flow diversion case. J Neurosurg Pediatr. (2018) 22:532–40. doi: 10.3171/2018.6.PEDS18165
31. Lim J, Suh SH, Lee KY, Hong CK, Park SW. Endovascular treatment of iatrogenic intracranial pseudoaneurysm following stent angioplasty. J Neuroimaging. (2012) 22:194–6. doi: 10.1111/j.1552-6569.2011.00591.x
32. Imahori T, Okamura Y, Sakata J, Shose H, Yamanishi S, Kohmura E. Delayed rebleeding from pseudoaneurysm after mechanical thrombectomy using a stent retriever due to small artery avulsion confirmed by open surgery: a case report. World Neurosurg. (2020) 133:150–4. doi: 10.1016/j.wneu.2019.09.141
33. Nubourgh Y, Bruninx G, Declour C, Vanderkelen B. Unusual occurrence of a pseudo-aneurysm of the middle cerebral artery in a patient with fibromuscular dysplasia. Acta Neurochir. (2000) 142:1311–4. doi: 10.1007/s007010070031
34. Nomura M, Mori K, Tamase A, Kamide T, Seki S, Iida Y, et al. Pseudoaneurysm formation due to rupture of intracranial aneurysms: case series and literature review. Neuroradiol J. (2017) 30:129–37. doi: 10.1177/1971400916684667
35. Gugliemi G. The interventional neuroradiological treatment of intracranial aneurysms. Adv Tech Stand Neurosurg. (1998) 24:215–60. doi: 10.1007/978-3-7091-6504-1_5
36. Cikla U, Li Y, Hernandez-Duran S, Kozan A, Baskaya MK. Treatment of supraclinoid internal carotid artery iatrogenic pseudoaneurysm with extracranial-to- intracranial bypass and trapping: demonstration of technique with video presentation. Turk Neurosurg. (2015) 25:305–9. doi: 10.5137/1019-5149.JTN.13039-14.1
37. Garg K, Gurjar HK, Singh PK, Singh M, Chandra PS, Sharma BS. Internal carotid artery aneurysms presenting with epistaxis-our experience and review of literature. Turk Neurosurg. (2016) 26:357–63. doi: 10.5137/1019-5149.JTN.12598-14.1
38. D'Urso P, Loumiotis I, Milligan BD, Cloft H, Lanzino G. “Real time” angiographic evidence of “pseudoaneurysm” formation after aneurysm rebleeding. Neurocrit Care. (2011) 14:459–62. doi: 10.1007/s12028-011-9522-y
39. Kumar A, Jakubovic R, Yang V, Dacosta L. Traumatic anterior cerebral artery aneurysms and management options in the endovascular era. J Clin Neurosci. (2016) 25:90–5. doi: 10.1016/j.jocn.2015.05.063
40. Gerosa A, Fanti A, Del Sette B, Bianco A, Cossandi C, Crobeddu E, et al. Posttraumatic middle meningeal artery pseudoaneurysm: case report and review of the literature. World Neurosurg. (2019) 128:225–9. doi: 10.1016/j.wneu.2019.05.030
41. Umana GE, Cristaudo C, Scalia G, Passanisi M, Corsale G, Tomarchio L, et al. Chronic epdidural hematoma caused by traumatic intracranial pseudoaneurysm of the middle meningeal artery: review of the literature with a focus on this unique entity. World Neurosurg. (2020) 136:198–204. doi: 10.1016/j.wneu.2019.12.179
42. Maldonado-Naranjo A, Kshettry VR, Toth G, Bain M. Non-traumatic superior hypophyseal aneurysm with associated pseudoaneurysm presenting with massive epistaxis. Clin Neurol Neurosurg. (2013) 115:2251–3. doi: 10.1016/j.clineuro.2013.07.003
43. Moon HT, Kim SH, Lee JW, Huh SK. Clinical analysis of traumatic cerebral pseudoaneurysms. Korean J Neurotrauma. (2015) 11:124–30. doi: 10.13004/kjnt.2015.11.2.124
44. Wewel J, Mangubat EZ, Muñoz L. Iatrogenic traumatic intracranial aneurysm after endoscopic sinus surgery. J Clin Neurosci. (2014) 21:2072–6. doi: 10.1016/j.jocn.2014.05.017
45. Binning MJ, Eskandari R, Couldwell WT. Direct surgical repair of carotid pseudoaneurysm in an infant. Childs Nerv Syst. (2010) 26:1151–3. doi: 10.1007/s00381-010-1225-8
46. Raper DMS, Rutledge WC, Winkler E, Abla AA. Spontaneous perforation of anterior choroidal artery with resultant pseudoaneurysm formation: unusual cause of subarachnoid hemorrhage. World Neurosurg. (2020) 134:141–4. doi: 10.1016/j.wneu.2019.10.174
47. Sato Y, Ando K, Kawaguchi M, Kakinuma K. Successful resection of a growing distal medial lenticulostriate artery pseudoaneurysm presenting with isolated intraventricular hemorrhage. J Stroke Cerebrovasc Dis. (2017) 26:e206–e209. doi: 10.1016/j.jstrokecerebrovasdis.2017.07.021
48. Shirane R, Kondo T, Yoshida YK, Furuta S, Yoshimoto T. Ruptured cerebral pseudoaneurysm caused by the removal of a ventricular catheter. J Neurosurg. (1999) 91:1031–3. doi: 10.3171/jns.1999.91.6.1031
49. Sujijantarat N, Pierson MJ, Kemp J, Coppens JR. Staged trapping of traumatic basilar trunk pseudoaneurysm: a case report and review of literature. World Neurosurg. (2017) 108:e7–12. doi: 10.1016/j.wneu.2017.08.144
50. Umekawa M, Hasegawa H, Shin M, Kawashima M, Nomura S, Nakatomi H, et al. Radiosurgery-induced anterior inferior cerebellar artery pseudoaneurysm treated with trapping and bypass. World Neurosurg. (2018) 116:209–13. doi: 10.1016/j.wneu.2018.04.161
51. Walcott BP, Nahed BV, Kahle KT, Sekhar LN, Ferreira MJ. Cerebrovascular bypass and aneurysm trapping for the treatment of an A2-segment anterior cerebral artery pseudoaneurysm and herniation through a skull base defect following trauma. J Clin Neurosci. (2012) 19:149–51. doi: 10.1016/j.jocn.2011.05.017
52. Al-Jehani HM, Alwadaani HA, Almolani FM. Traumatic intracranial internal carotid artery pseudoaneurysm presenting as epistaxis treated by endovascular coiling. Neurosciences. (2016) 21:60–3. doi: 10.17712/nsj.2016.1.20150514
53. Aljuboori Z, Meyer K, Ding D, James R. Endovascular treatment of a traumatic middle cerebral artery pseudoaneurysm with the pipeline flex embolization device. World Neurosurg. (2020) 133:201–4. doi: 10.1016/j.wneu.2019.10.008
54. Giorgianni A, Pellegrino C, Minotto R, Mercuri A, Frattini L, Baruzzi F, et al. Flow-diverter stenting of post-traumatic bilateral anterior cerebral artery pseudoaneurysm: a case report. Interv Neuroradiol. (2015) 21:23–8. doi: 10.1177/1591019915575441
55. Giorgianni A, Pozzi F, Pellegrino C, Padovan S, Karligkiotis A, Castelnuovo P, et al. An emergency placement of a flow diverter stent for an iatrogenic internal carotid artery injury during endoscopic pituitary surgery. World Neurosurg. (2019) 122:376–9. doi: 10.1016/j.wneu.2018.10.234
56. Griauzde J, Gemmete JJ, Chaudhary N, Pandey AS, Garton HJ. Basilar artery pseudoaneurysm presenting at 5-month follow-up after traumatic atlanto-occipital dislocation in a 7-year-old girl treated with intracranial stent placement and coiling. J Neurointerv Surg. (2014) 6:e8. doi: 10.1136/neurintsurg-2012-010573.rep
57. Kim SS, Kang DH, Park H, Lee CH, Hwang SH, Jung JM, et al. Short-term clinical and angiographic outcome in child with traumatic pseudoaneurysm in A2 segment of anterior cerebral artery after endovascular treatment: case report. Korean J Neurotrauma. (2014) 10:130–3. doi: 10.13004/kjnt.2014.10.2.130
58. Lim YC, Kang JK, Chung J. Reconstructive stent-buttressed coil embolization of a traumatic pseudoaneurysm of the supraclinoid internal carotid artery. Acta Neurochir. (2012) 154:477–80. doi: 10.1007/s00701-011-1251-7
59. Liu P, Yang P, Cai M, Qin J, Pan L. Treatment of pediatric traumatic intracranial pseudoaneurysm using endovascular covered stent: three case reports. World Neurosurg. (2016) 88:693.e1–6. doi: 10.1016/j.wneu.2015.12.037
60. Liu QL, Xue H, Qi CJ, Zhao P, Wang DH, Li G. Traumatic anterior cerebral artery pseudoaneurysm epistaxis. World Neurosurg. (2017) 100:e9–16. doi: 10.1016/j.wneu.2016.11.138
61. Mascitelli JR, McNeill IT, Mocco J, Berenstein A, DeMattia J, Fifi JT. Ruptured distal AICA pseudoaneurysm presenting years after vestibular schwannoma resection and radiation. J Neurointerv Surg. (2016) 8:e19. doi: 10.1136/neurintsurg-2015-011736.rep
62. Matsumura H, Kato N, Hosoo H, Fujiwara Y. Subarachonid hemorrhage due to anterior inferior cerebellar artery aneurysms associated with gamma knife surgery for vestibular schwannomas. Acta Neurochir. (2015) 157:1765–7. doi: 10.1007/s00701-015-2552-z
63. Morinaga Y, Nii K, Sakamoto K, Inoue R, Mitsutake T, Hanada H. Stent-assisted coil embolization for a ruptured posterior communicating artery pseudoaneurysm after endoscopic transsphenoidal surgery for pituitary adenoma. World Neurosurg. (2019) 123:301–5. doi: 10.1016/j.wneu.2018.12.047
64. Murakami M, Kawarabuki K, Inoue Y, Ohta T. Ruptured pseudoaneurysm after gamma knife surgery for vestibular schwannoma. Neurol Med Chir. (2016) 56:38–42. doi: 10.2176/nmc.cr.2015-0034
65. Ogilvy CS, Tawk RG, Mokin M, Yang X, Levy EI, Hopkins LN, et al. Stent-assisted coiling treatment of pediatric traumatic pseudoaneurysm resulting from tumor surgery. Pediatr Neurosurg. (2011) 47:442–8. doi: 10.1159/000339353
66. Patel AS, Horton TG, Kalapos P, Cockroft KM. Onyx-HD 500 embolization of a traumatic internal carotid artery pseudoaneurysm after transsphenoidal surgery. J Neuroimaging. (2015) 25:656–9. doi: 10.1111/jon.12221
67. Sami MT, Gattozzi DA, Soliman HM, Reeves AR, Moran CJ, Camarata PJ, et al. Use of PipelineTM embolization device for the treatment of traumatic intracranial pseudoaneurysms: case series and review of cases from literature. Clin Neurol Neurosurg. (2018) 169:154–60. doi: 10.1016/j.clineuro.2018.04.012
68. Van Rooij WJ, Van Rooij SB. Endovascular treatment of traumatic pericallosal artery aneurysms. A case report. Interv Neuroradiol. (2013) 19:56–9. doi: 10.1177/159101991301900108
69. Zanaty M, Chalouhi N, Jabbour P, Starke RM, Hasan D. The unusual angiographic course of intracranial pseudoaneurysms. Asian J Neurosurg. (2015) 10:327–30. doi: 10.4103/1793-5482.162721
70. Rangel-Castilla L, McDougall CG, Spetzler RF, Nakaji P. Urgent cerebral revascularization bypass surgery for iatrogenic skull base ICA injury. Neurosurgery. (2014) 4:640–8. doi: 10.1227/NEU.0000000000000529
71. Cappabianca P, Briganti F, Cavallo LM, de Divitiis E. Pseudoaneurysm of the intracavernous carotid artery following endoscopic endonasal transsphenoidal surgery, treated by endovascular approach. Acta Neurochir. (2001) 143:95–6. doi: 10.1007/s007010170144
72. Lin T, Chen D, Li J, Liao Y, Xu F, Leng B, et al. Diagnosis and treatment differences between iatrogenic and traumatic intracranial pseudoaneurysms. Chin J Neurosurg. (2018) 34:993–8.
73. Sim SY, Shin YS, Yoon SH. Endovascular internal trapping of traumatic pericallosal pseudoaneurysm with hydrogel-coated self-expandable coil in a child: a case report. Surg Neurol. (2008) 69:418–22. doi: 10.1016/j.surneu.2007.02.042
74. Medel R, Crowley RW, Hamilton DK, Dumont AS. Endovascular obliteration of an intracranial pseudoaneurysm: the utility of Onyx. J Neurosurg Pediatr. (2009) 4:445–8. doi: 10.3171/2009.6.PEDS09104
75. Elias AE, Chaudhary N, Pandery AS, Gemmete JJ. Intracranial endovascular balloon test occlusion: indications, methods, and predictive value. Neuroimaging Clin N Am. (2013) 23:695–702. doi: 10.1016/j.nic.2013.03.015
76. Cohen JE, Gomori JM, Segal R, Spivak A, Margolin E, Sviri G, et al. Results of endovascular treatment of traumatic intracranial aneurysms. Neurosurgery. (2008) 63:476–85. doi: 10.1227/01.NEU.0000324995.57376.79
77. Ruiz-Juretschke F, Castro E, Mateo Sierra O, Iza B, Manuel Garbizu J, Fortea F, et al. Massive epistaxis resulting from an intracavernous internal carotid artery traumatic pseudoaneurysm: complete resolution with overlapping uncovered stents. Acta Neurochir. (2009) 151:1681–4. doi: 10.1007/s00701-009-0294-5
78. Saatci I, Cekirge HS, Ozturk MH, Arat A, Ergungor F, Sekerci Z, et al. Treatment of internal carotid artery aneurysms with a covered stent: experience in 24 patients with mid-term follow-up results. Am J Neuroradiol. (2004) 25:1742–9.
79. Nerva JD, Morton RP, Levitt MR, Osbun JW, Ferreira MJ, Ghodke BV, et al. Pipeline embolization device as primary treatment for blister aneurysms and iatrogenic pseudoaneurysms of the internal carotid artery. J Neurointerv Surg. (2015) 7:210–6. doi: 10.1136/neurintsurg-2013-011047
80. Shah Q, Friedman J, Mamourian A. Spontaneous resolution of traumatic pseudoaneurysm of the middle meningeal artery. Am J Neuroradiol. (2005) 26:2530–2.
81. Srinivasan A, Lesiuk H, Goyal M. Spontaneous resolution of posttraumatic middle meningeal artery pseudoaneurysm. Am J Neuroradiol. (2006) 27:882–3.
82. Adrien C, Pierre-Henri L, Pieere T, Catherine C, Aapolline K, Klau M, et al. Spontaneous resolution of perforator aneurysms of the posterior circulation. J Neurosurg. (2014) 121:1107–11. doi: 10.3171/2014.7.JNS132411
83. Loevner LA, Ting TY, Hurst RW, Goldberg HI, Schut L. Spontaneous thrombosis of a basilar artery traumatic aneurysm in a child. Am J Neuroradiol. (1998) 19:386–8.
84. Turan N, Butler S, Larson TC 3rd, Marson A. Nontraumatic, posterior circulation pseudoaneurysm of the basilar artery summit with complete spontaneous resolution: case report and literature review. Surg Neurol Int. (2017) 8:50. doi: 10.4103/sni.sni_452_16
Keywords: intracranial pseudoaneurysms, trauma, iatrogenic, management, endovascular treatment
Citation: Zheng Y, Lu Z, Shen J and Xu F (2020) Intracranial Pseudoaneurysms: Evaluation and Management. Front. Neurol. 11:582. doi: 10.3389/fneur.2020.00582
Received: 22 February 2020; Accepted: 20 May 2020;
Published: 07 July 2020.
Edited by:
Thanh Nguyen, Boston University, United StatesReviewed by:
Jan Jack Gouda, Wright State University, United StatesMário de Barros Faria, Clinical Hospital of Porto Alegre, Brazil
Copyright © 2020 Zheng, Lu, Shen and Xu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jianguo Shen, c2hlbmppYW5ndW9AanhleS5jb20=; Feng Xu, ZmVuZ3h1LmRyQGdtYWlsLmNvbQ==
†These authors have contributed equally to this work
 Yongtao Zheng1†
Yongtao Zheng1† Feng Xu
Feng Xu