
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Microbiol. , 27 February 2023
Sec. Antimicrobials, Resistance and Chemotherapy
Volume 14 - 2023 | https://doi.org/10.3389/fmicb.2023.1128497
This article is part of the Research Topic Common Themes in Drug Resistance: from Viruses to Human View all 7 articles
 Raluca Ioana Dascălu1
Raluca Ioana Dascălu1 Alexandra Bolocan2,3
Alexandra Bolocan2,3 Dan Nicolae Păduaru2,3*
Dan Nicolae Păduaru2,3* Alexandru Constantinescu2,3
Alexandru Constantinescu2,3 Magda Mihaela Mitache4
Magda Mihaela Mitache4 Anca Daniela Stoica5,6
Anca Daniela Stoica5,6 Octavian Andronic2,3,7
Octavian Andronic2,3,7Helicobacter pylori (Hp), a well-known human pathogen, causes one of the most common chronic bacterial infections and plays an important role in the emergence of chronic progressive gastric inflammation and a variety of gastrointestinal diseases. The prevalence of Hp infection varies worldwide and is indirectly proportional to socio-economic status, especially during childhood. The response to the eradication therapy significantly depends on the antibiotic resistance specific to each geographical region; thus, currently, given the increasing prevalence of antimicrobial resistance (especially to clarithromycin, metronidazole, and levofloxacin), successful treatment for Hp eradication has become a real challenge and a critical issue. The most incriminated factors associated with multidrug resistance (MDR) in Hp proved to be the overuse or the improper use of antibiotics, poor medication adherence, and bacterial-related factors including efflux pumps, mutations, and biofilms. Up to 30% of first-line therapy fails due to poor patient compliance, high gastric acidity, or high bacteremia levels. Hence, it is of great importance to consider new eradication regimens such as vonoprazan-containing triple therapies, quintuple therapies, high-dose dual therapies, and standard triple therapies with probiotics, requiring further studies and thorough assessment. Strain susceptibility testing is also necessary for an optimal approach.
Helicobacter pylori (Hp) represents a slow-growth Gram-negative helical- or spiral-shaped flagellated bacterium, a urease producer (Bacteriology and Epidemiology of Helicobacter pylori Infection–UpToDate, 2022). Infection with Hp is one of the most common chronic bacterial infections, playing an important role in the emergence of chronic progressive gastric inflammation and a variety of gastrointestinal diseases, such as gastric or duodenal ulcers, gastric cancer, or MALT lymphoma (Singh et al., 2017). This pathogen proves strict tropism and is well adapted to the gastric milieu, with specific adhesion to the gastric epithelial cells. Hp colonizes the mucus layer in the gastric antrum or areas of gastric metaplasia in the duodenum, and it requires at least three key characteristics in order to induce the infection: the production of an active urease, the presence of flagella, and the presence of adhesins (Boyanova et al., 2019). Deterioration of the gastric parietal cell is mostly caused by the release of enzymes and the induction of apoptosis by binding to the molecules of the class II major histocompatibility complex (MHC II) (Kusters et al., 2006). The production of bacterial urease allows the conversion of urea into ammonia and chloride, with a direct cytotoxic effect. The passage through the mucus layer to the gastric surface epithelium is made easier by its spiral shape, flagella, and the mucolytic enzymes (Boyanova et al., 2019). Hp attaches through some adhesion molecules, including BabA, which, by binding to the Lewis antigen, expressed on the surface of gastric mucosa cells, leads to gastritis among subjects who are infected (Chang et al., 2018). Furthermore, ulcers and gastric cancer appear most frequently when the infectious strain expresses CagA (cytotoxic-associated protein) and VacA (vacuolating toxin) genes, secondary to an important inflammatory and immune response, mainly associated with the synthesis of interleukin 8 (IL-8), a significant mediator of gastric inflammation (Tshibangu-Kabamba and Yamaoka, 2021). Nevertheless, genetic background is also involved. Thus, even if adhesion is dependent upon the binding of bacterial surface adhesins to specific epithelial cell receptors, host factors could modulate this process (Podolsky et al., 2015). For instance, certain individuals might express specific surface receptors or greater numbers of receptors, making them more susceptible to Hp attachment and colonization. Polymorphisms leading to increased IL-1β levels are associated with atrophic gastritis and cancer (Boyanova et al., 2019).
Epidemiologically, Hp affects over 4.4 billion individuals worldwide, with a global incidence of infection reaching up to 50% (Lee, 2019). The prevalence of infection varies between countries, within them, and within the subpopulations of the same country, and it seems that in developing countries, the prevalence is much higher (80–90% of the population) when compared to developed countries (20–50% of the population) (Eusebi et al., 2014; Khoder et al., 2019). It has been demonstrated that the risk of acquiring Hp infection is strongly associated with socio-economic status and hygiene conditions early in life (such as the density of housing, overcrowding, and lack of running water). The highest rate of infection is shown in groups with low socio-economic status and during childhood (Borka Balas et al., 2022). Thereby, those aged between 10 and 19 years from a high socio-economic class had a 20% frequency of Hp infection, while individuals of the same age from a low socio-economic class had a 60% frequency (Hooi, 2017; Talebi and Abadi, 2017). Serologic evidence of Hp is rarely found before age 10 but increases to 10% in those aged between 18 and 30 years and to 50% in those older than 60 years (Lee, 2019). The incidence increases with age, probably due to acquisition during childhood, a period when hygiene is poorer (cohort effect) (Mégraud and Lehours, 2007). Moreover, recent literature highlighted an increased possibility of persistent infection with Hp infection associated with the consumption of salted food (Bacteriology and Epidemiology of Helicobacter pylori Infection–UpToDate, 2022).
Currently available guidelines recommend that all patients with gastric or duodenal ulcers benefit from eradication therapy if Hp is present (Chey et al., 2017). The selection of treatment regimen should be based on local antibiotic resistance patterns (if known), previous exposure and allergies to specific antibiotics, cost, side effects, and ease of administration (Treatment Regimens for Helicobacter pylori in Adults—UpToDate, 2022). Good compliance with treatment is highly necessary. In general, eradication regimens consist of two antibiotics administered together with a double dose of proton pump inhibitors (PPIs), to strongly suppress gastric acid secretion (Boyanova et al., 2019). Metronidazole, clarithromycin, amoxicillin, tetracycline, and bismuth represent the pylon agents of the treatment and first-line standard regimens consist of triple therapy (including PPI, clarithromycin, and amoxicillin or metronidazole) for 14 days, concomitant therapy/non-bismuth quadruple therapy (including PPI, clarithromycin, amoxicillin, and a nitroimidazole–tinidazole or metronidazole) for 10 to 14 days, hybrid therapy, as an alternative to clarithromycin triple therapy—consists of PPI and amoxicillin for 7 days followed by PPI, amoxicillin, clarithromycin, and a nitroimidazole for 7 days, and sequential therapy—the 10-day clarithromycin-containing sequential therapy regimen is based on PPI and amoxicillin for 5 days, followed by PPI, clarithromycin, and nitroimidazole (metronidazole) for 5 days (Feldman et al., 2015) (Treatment Regimens for Helicobacter pylori in Adults—UpToDate, 2022).
However, the efficacy of Hp eradication treatment has decreased dramatically, and up to 30% of first-line therapy fails (Chey et al., 2017). Several factors, such as poor patient compliance and resistance of Hp strain to several commonly prescribed antibiotics (including vancomycin, trimethoprim, and sulfonamides) or inadequate acid suppression are associated with eradication failure, but the most significant factor incriminated seems to be the increasing regional antibiotic resistance to drugs (Boyanova et al., 2019; Kuo, 2021). It needs to be mentioned that there is a high incidence of resistance to metronidazole and clarithromycin, especially in certain populations, and resistance to clarithromycin has doubled in Europe in the last decade (Boyanova et al., 2019; Megraud, 2021; Treatment Regimens for Helicobacter pylori in Adults—UpToDate, 2022). Resistance to amoxicillin, tetracycline, and rifabutin is generally low (< 5%), except in countries where they are available without medical prescription, and resistance could exceed 50% (Treatment Regimens for Helicobacter pylori in Adults—UpToDate, 2022). Hence, in patients who have failed standard therapy, salvage regimens should be considered: bismuth quadruple therapy (including PPI, tetracycline, metronidazole, and bismuth subsalicylate), levofloxacin-based therapy triple (including PPI, levofloxacin, and amoxicillin), or quadruple (including PPI, levofloxacin, nitazoxanide, and doxycycline)—other levofloxacin-based quadruple therapies include PBLA (PPI, bismuth, levofloxacin, and amoxicillin), PBLT (PPI, bismuth, levofloxacin, and tetracycline), and PBLM (PPI, bismuth, levofloxacin, and metronidazole) (Shah et al., 2021), high-dose dual therapy (including PPI and amoxicillin—at least 2 g divided three or four times per day to avoid low trough levels) for 14 days—particularly in patients in whom dual metronidazole/clarithromycin resistance or levofloxacin resistance is suspected, rifabutin triple therapy (including PPI two times daily, rifabutin and amoxicillin) for 14 days, and clarithromycin-based therapy (including PPI, bismuth, clarithromycin, and tetracycline) (Chey et al., 2017; Treatment Regimens for Helicobacter pylori in Adults—UpToDate, 2022). It should be highlighted that levofloxacin should be used only if the Hp strain is known to be sensitive to it or if the population levofloxacin resistance rates are <15%, taking into account that levofloxacin resistance decreases the eradication success rate of levofloxacin-containing regimens by 20–40% (Karamanolis et al., 2014; Lee, 2019; Lee et al., 2019). Regarding clarithromycin-based therapy, it could only be used as a salvage regimen in patients with no risk factors for macrolide resistance (no prior macrolide exposure and local clarithromycin resistance known to be <15%) (Talebi and Abadi, 2017; Lee, 2019). Moreover, rifabutin-based triple therapy is not only expensive but also could lead to reversible myelotoxicity and could increase the prevalence of rifabutin-resistant mycobacteria (Lee et al., 2019). According to The Maastricht V/Florence consensus, the first-line treatment regimen is given considering the resistance to clarithromycin, determined by antibiogram cultures or molecular tests, with a threshold of 15% (Malfertheiner et al., 2017). In regions with increased resistance to clarithromycin, bismuth quadruple therapy is recommended as the first-line therapy, and if not available, sequential therapy or quadruple therapy is recommended. Furthermore, to amplify the efficiency of both standard therapy and salvage regimens, the latest Maastricht V consensus recommends increasing the duration of treatment administration from 7 to 14 days and the usage of a double dose of PPI compared to the last recommendation, acquiring higher eradication rates with 5% and 8–12%, respectively (Malfertheiner et al., 2017).
Worldwide, an alarming and substantial concern arises from multidrug resistance (MDR) in Hp, leading to therapeutic regimen failures and low eradication rates. MDR is defined as resistance to ≥3 antibiotics of different classes and it depends on the geographical area, study period, and patients' characteristics (Boyanova et al., 2019; Sukri, 2022) (Figure 1).
Taking into consideration the widespread and improper use of antibiotics, one crucial factor influencing MDR in Hp is national outpatient antibiotic consumption; thus, one study assessing the primary Hp resistance in 18 European countries revealed that outpatient antibiotic consumption influenced both macrolide and fluoroquinolone resistance (Megraud, 2013). Furthermore, Hp efflux pumps and biofilms (multidimensional matrix-enclosed bacterial communities linked to chronic infections and reduced antibiotic susceptibility) seem to be involved in MDR (Singh et al., 2017). Not only were enhanced expression of efflux pump (hp1165 and hefA) genes involved in tetracycline and MDR resistance noticed in Hp biofilms but also the upregulation of genes four transmembrane ABC transporters and other (hp0656, hp0946) efflux proteins were detected (Attaran et al., 2017; Kazakos et al., 2017). In contrast, resistance could be undetected when low-density inoculum is used for susceptibility testing, in case of mixed infections which could carry both susceptible and resistant isolates to an antibiotic (Mansour, 2016; Lee, 2019). Last but not least, poor adherence to current guidelines for the management of Hp infection could lead to the occurrence of MDR strains and treatment failure.
Clarithromycin is a macrolide antibiotic whose action is based on the interaction with the peptidyl transferase loop of the V domain of the 23S ribosomal RNA molecule, which could inhibit bacterial protein synthesis (Talebi and Abadi, 2017). Hence, point mutations in the V domain of the 23S ribosomal RNA might inhibit the affinity between clarithromycin and the peptidyl transferase loop, leading to clarithromycin resistance (Stone, 1996; Talebi and Abadi, 2017). Among responsible mutations, the 23S ribosomal RNA A2143G, A2142G, and A2142C were reported to be the most frequent, accounting for up to 90% (Mégraud and Lehours, 2007). Another potential mechanism incriminated in clarithromycin resistance of Hp is represented by the efflux pump system, and the currently available data suggest that the existence of efflux pumps in Hp strains could have a synergic effect to induce antibiotic resistance in parallel with 23S rRNA mutations (Talebi and Abadi, 2017). In contrast, one study suggested that the outer membrane protein (OMP) alterations might be involved in the Hp resistance to clarithromycin. By using comparative proteomic analyses of clarithromycin-susceptible and -resistant Hp strains to identify Hp OMPs, Smiley et al. reported that iron-regulated membrane protein, urease B, elongation factor thermo unstable, and putative OMP were downregulated, whereas HopT (BabB) transmembrane protein, HofC, and OMP31 were upregulated in clarithromycin-resistant Hp (Smiley et al., 2013).
Metronidazole is a synthetic nitroimidazole, representing one of the mainstay drugs for the treatment of anaerobic infections. Concerning the mechanisms responsible for resistance to metronidazole in Hp, data highlighted mutations of rdxA, a gene that encodes an oxygen-insensitive NADPH nitroreductase, as being the main cause (Kim, 2009; Lee, 2018). Furthermore, mutations in other redox genes such as frxA (encoding the NADPH flavin oxidoreductase) and fdxB (encoding the ferredoxin-like protein) might also induce Hp resistance to metronidazole (Saranathan, 2020; Metronidazole: An overview–UpToDate, 2022). In contrast, in one study, Mehrabadi et al. suggested that the resistance nodulation cell division (RND) family of efflux pumps might be involved in the metronidazole resistance of Hp clinical isolates and they reported that excess amounts of metronidazole increased the gene expression levels of the outer membrane protein (TolC) homologs of RND pumps (Mehrabadi et al., 2010; Lee, 2018).
Levofloxacin represents a fluoroquinolone drug that exerts its antibacterial effect through the interaction with DNA gyrase, encoded by gyrA and gyrB. Thus, point mutations in the quinolone resistance-determining regions of gyrA might restrict this process, leading to fluoroquinolone resistance of Hp (Tankovic et al., 2003). Rimbara et al. indicated that a gyrB mutation at position 463 might also induce Hp resistance to fluoroquinolone (Rimbara et al., 2012). The literature shows that mutations in 87, 88, 91, and 97 positions of gyrA are the most common (Rimbara et al., 2012; Shetty et al., 2019; Keikha et al., 2022).
Amoxicillin, one of the most commonly used antibiotics in the primary care setting, is a beta-lactamase antibiotic that interacts with penicillin-binding proteins (PBPs) and inhibits the synthesis of the cell wall, resulting in bacterial dissolution (Akhavan et al., 2022). Evidence highlighted that high levels of amoxicillin resistance are associated with the production of beta-lactamase in Hp (Sukri, 2022). Decreased membrane permeability to amoxicillin or the alteration of the efflux pump might also be involved. In addition, it seems that the most common mechanism that contributes to low or moderate levels of amoxicillin resistance is represented by point mutations in the pbp 1A gene, but mutations in the pbp 2, pbp 3, hefC, hopC, and hofH were also mentioned in the literature (Okamoto, 2002; Qureshi et al., 2014).
Tetracycline is a macrolide antibiotic that inhibits protein synthesis by blocking the attachment of charged tRNA at the P site peptide chain. Tetracycline binds to the 30S and 50S subunits of microbial ribosomes and bacteria usually acquire resistance from the horizontal transfer of a gene that either encodes an efflux pump or a ribosomal protection protein (Grossman, 2016). Data reported that single and double-base-pair mutations were only responsible for low levels of tetracycline resistance, while triple-base-pair mutations 16S rDNA AGA (926–928) were associated with high levels of resistance (Gerrits et al., 2003). Moreover, in one study, Anoushiravani et al. suggested that proton motive force-dependent efflux mechanisms might be involved in the resistance of Hp clinical isolates to tetracycline (Anoushiravani et al., 2009).
As mentioned earlier, MDR is defined as resistance to ≥3 antibiotics of different classes, and the increasing presence of Hp strains with an MDR profile represents a serious threat globally (Boyanova et al., 2019). Despite various mutations that simultaneously induce resistance to separate drug families conferring a cumulative MDR profile, there are additional mechanisms responsible for MDR in Hp, but data are still limited (Tuan, 2019). For instance, Hp could turn into quiescent cells named coccoid forms for which substantially increased minimum inhibitory concentrations of different antibiotics are required to achieve bactericidal effects (Kadkhodaei et al., 2020). Hence, considering subsequent ultrastructural modifications in the cell membrane and metabolic pathways that reduce drug target exposure and drug penetration, the coccoid formation could be a leading cause of MDR (Kadkhodaei et al., 2020). In addition, biofilm formation could play an important role in antibiotic resistance, yet the mechanism is not completely understood (Hathroubi et al., 2018). Furthermore, even though the information is limited, studies reported that the upregulation of efflux pump systems against a corresponding group of substrates and restricted drug uptake owing to the downregulation of expression of outer membrane proteins or lipopolysaccharides could also be involved in acquiring MDR in Hp (Bos et al., 2004; Hirata, 2010; Ge, 2018) (Table 1).
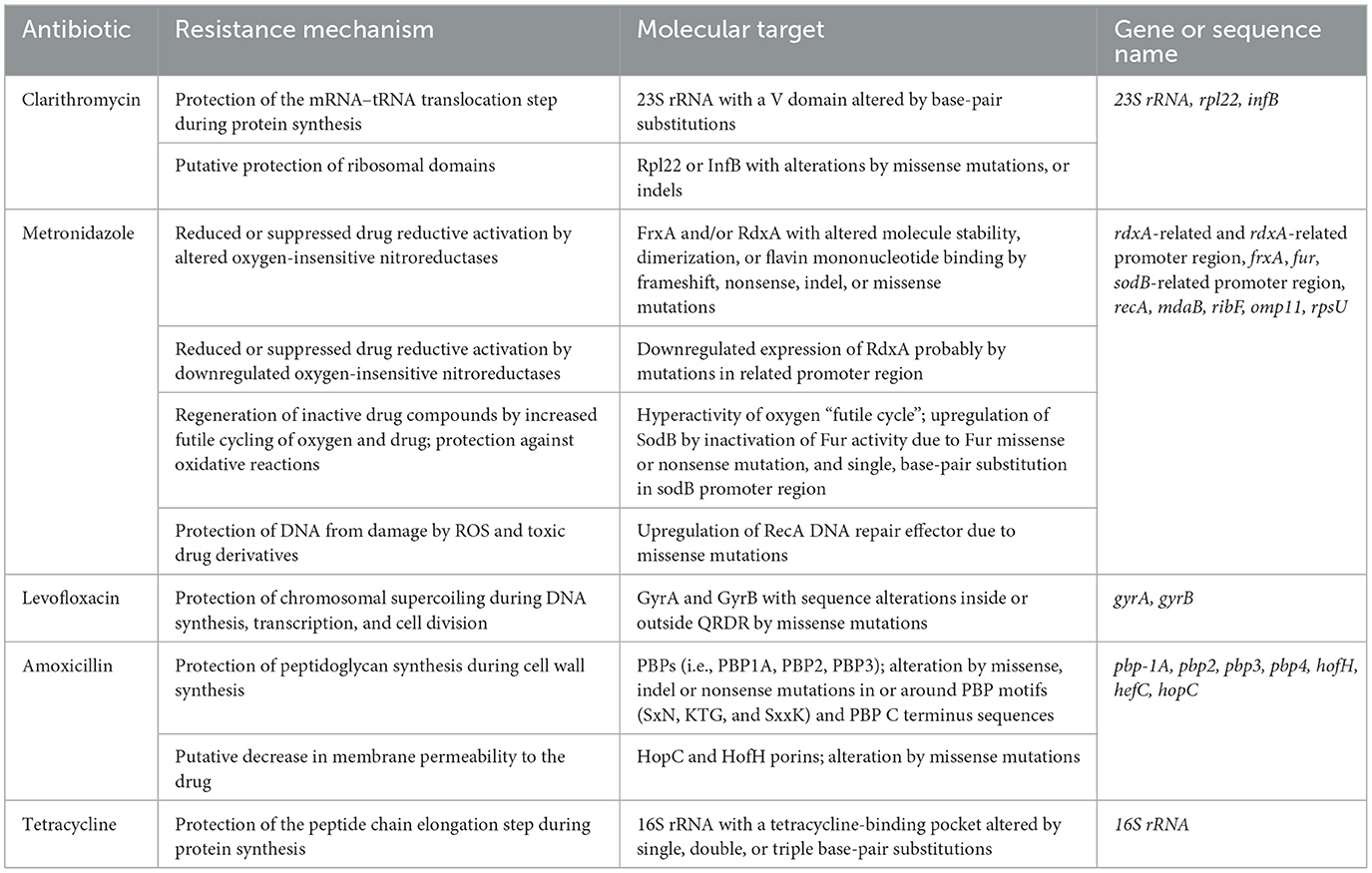
Table 1. Biological mechanisms, molecular target, and gene or sequence name of drug resistance in Helicobacter pylori species reported following standard recommendations in molecular diagnostics from the Human Genome Variation Society (Den Dunnen and Antonarakis, 2001; Kusters et al., 2006; Ogino, 2007; Chang et al., 2018).
Heteroresistance consists of the presence of a heterogeneous population of bacteria with one subpopulation or several subpopulations that exhibit increased levels of antibiotic resistance compared to the main population and could be considered a proclaimer of single-drug resistance or MDR (Andersson et al., 2019). Monoclonal or polyclonal differentiation of bacterial population represents the main mechanisms responsible for this phenomenon (Ailloud, 2019; Andersson et al., 2019). It seems that anatomical and physiological differences between the antral and oxyntic gastric mucosa compose an evolutionary force that transduces intragastric migrations of bacteria from the same clone and rapid adaptation to microniches within the host; therefore, the population structure of bacteria could be partitioned through evolutionary bottlenecks in subgroups (Ailloud, 2019). Concerning Hp, it is more probable that heteroresistance is determined by the same strain with and without resistance attributes rather than a co-infection with different strains (Ailloud, 2019). Studies suggest that multiple gastric biopsy specimens or multiple bacterial colonies from the same sample should be obtained for drug susceptibility testing so as to counteract this phenomenon (Kao, 2014; Andersson et al., 2019).
The prevalence of MDR is characterized by a regional variation and appears to be changing over time. Literature reported lower rates of primary MDR, ranging from ≤10% in Austria, Portugal, Argentina, France, and Bulgaria to >20% in India and >40% in Peru, while in treated patients, MDR rates were >16%, reaching 31.6% in Vietnam (Almeida, 2014; Phan, 2015; Mansour, 2016; Zollner-Schwetz, 2016; Boehnke, 2017; Boyanova, 2017). In one meta-analysis evaluating the primary antibiotic resistance in the Asia-Pacific region between 2006 and 2015, Kuo et al. reported a primary resistance to clarithromycin, metronidazole, and levofloxacin of 20%, 47%, and 21%, respectively (Kuo, 2017; Savoldi et al., 2018). Both tetracycline and amoxicillin primary resistance rates were 3%, and the amoxicillin primary resistance rate was relatively low, up to 5% in the majority of countries (Lee et al., 2019). The prevalence of primary phenotypic resistance to levofloxacin was >10% in France (17.2%), Belgium (16.8%), Japan (15%), Hong Kong (11.5%), and Korea (10.4%), but <10% in Taiwan (5.8%) (Kuo, 2017). Concerning secondary resistance rates, one study reported secondary resistance rates to amoxicillin, clarithromycin, metronidazole, tetracycline, and levofloxacin of 13.1, 92.5, 87.7, 14.3, and 70.1%, respectively, in patients who had received these antibiotics in their prior therapies in Taiwan (Liou, 2013). In addition, one recent study conducted in Korea reported secondary resistance rates to amoxicillin, clarithromycin, metronidazole, tetracycline, and levofloxacin of 17.1, 78.0, 51.2, 12.2, and 70.7%, respectively (Lee et al., 2019). There are also variations in MDR rates among regions in the same country (Savoldi et al., 2018). For instance, in Korea, MDR prevalence was <20% in Seoul and Chungcheong and >30% in Cholla and Gyeonggi (Lee, 2019).
Most frequently, MDR in Hp is triple or quadruple, though several countries, such as China or Bulgaria, reported quintuple resistance (Zhang et al., 2015; Boyanova, 2017). For instance, in Spain, triple resistance altered from 3.3% in 2013 to 1.8% in 2015 and 2.4% in 2017 (Cosme, 2019). In Bulgaria, the quintuple Hp resistance proved to be to amoxicillin, metronidazole, clarithromycin, tetracycline, and levofloxacin; while in China, the quintuple resistance proved to be to clarithromycin, metronidazole, levofloxacin, tetracycline, and rifampicin or amoxicillin, clarithromycin, metronidazole, levofloxacin, and rifampicin or amoxicillin, clarithromycin, metronidazole, levofloxacin, and tetracycline; in Chinese patients, triple, quadruple, and quintuple resistance rates were 24.9, 7.3, and 2.3%, respectively (Zhang et al., 2015; Boyanova, 2017). Moreover, in China, while the resistance rates to amoxicillin and tetracycline have remained relatively stable and low when it comes to clarithromycin, metronidazole, and levofloxacin, the resistance rates have increased considerably; thus, the eradication rate of standard triple therapies has gradually decreased from 88.54% (pre-2004) to 77.66% (2005–2009) and 71.13% (2010–2013) (Wang, 2014; Zhang et al., 2015). Consistently, in Japan, studies reported high rates of clarithromycin and levofloxacin resistance (16.4–81.1% and 42.3–43.2%, respectively), and low rates of amoxicillin resistance (~0.03%) (Nishizawa et al., 2015; Sugimoto, 2017). Moreover, an increasing trend of resistance to clarithromycin, levofloxacin, and metronidazole was observed in the USA, as well. Hence, in a group of veteran patients, Shiota et al. reported resistance rates of 31.3, 20.3, and 16.4% to levofloxacin, metronidazole, and clarithromycin, respectively, with an increase in clarithromycin resistance from 9.1% on 2009–2010 to 24.2% during 2011–2013 (Shiota et al., 2015). Multiple single-center studies conducted in Poland, Greece, and Germany showed high clarithromycin resistance in Poland and Greece and increased metronidazole resistance in Poland and Germany (Karamanolis et al., 2014; Karpiński et al., 2015; Regnath et al., 2017). In Turkey, Kocazeybek et al. reported resistance rates of 24.86, 33.75, 23.77, 3.51, and 0.97% for clarithromycin, metronidazole, levofloxacin, tetracycline and amoxicillin, respectively (Kocazeybek and Tokman, 2016). In contrast to the rising tendency of Hp multi-resistance to antibiotics observed in most European countries, Mourad-Baars et al. reported remarkably low rates of Hp antibiotic resistance in the Netherlands, particularly for clarithromycin and metronidazole (Mourad-Baars et al., 2015).
Unfortunately, for now, there are not sufficiently large epidemiological studies in Romania to evaluate the level of resistance to clarithromycin; however, metronidazole has shown an increased resistance rate in vitro, over 90%, most probably due to overuse for other infections.
The main clinical implication of bacterial resistance in vitro is a substantial decrease in the efficacy of the Hp treatment—an outcome that has been previously outlined as well. The theoretical consequence is an associated increase in clinical complications such as gastric cancer or peptic ulcers due to the longer duration of infections (Tshibangu-Kabamba and Yamaoka, 2021). The treatment efficacy is majorly impacted primarily by pretreatment antibiotic resistance according to studies that mainly assessed the effect of single-drug resistance to clarithromycin, metronidazole, and levofloxacin (Kasahun et al., 2020; Zou, 2020). Dramatic reductions in treatment efficacy have been found in the case of single-drug resistance to clarithromycin during triple and quadruple therapies containing clarithromycin (Dore et al., 2000; Zou, 2020). Lower decreases in the treatment success rate have been found for metronidazole resistance with triple and non-bismuth quadruple therapies. Nevertheless, even in the presence of bacterial resistance, a very high therapeutic efficacy was reported with bismuth quadruple therapies (Fischbach and Evans, 2007; Zou, 2020). Further evidence is needed to evaluate the effect of single-drug resistance to levofloxacin (Zou, 2020). Overall, an increasing pattern of single-drug resistance in Hp has been noted worldwide which could be associated with an increase in the rate of treatment failure (Kasahun et al., 2020).
The clinical implications become even more challenging in the case of MDR and heteroresistance in Hp when compared to that of single-drug resistance due to the multiple drug molecules that are affected simultaneously in the former case (Andersson et al., 2019). However, despite the clinical challenges, there is evidence of Hp eradication being attainable in patients (Fischbach and Evans, 2007). The effectiveness of the eradication therapy was suggested to also depend on factors related to the host rather than only on considerations of the pathogen's biology (Chey et al., 2017). For instance, several factors that dictate the extent to which bacterial resistance affects the efficacy of a given therapy are represented by the dose of antimicrobial agents, the duration of therapy, and the components used in therapy (Graham et al., 2014; Smith et al., 2014). Moreover, the interplay between the drug resistance of Hp and other microbial species is also of high concern for human health. It has been demonstrated that, despite inducing gastrointestinal dysbiosis, Hp infection and the associated eradication therapy could significantly increase MDR and single-drug resistance mechanisms in other microbial species through its drug resistance and eradication failure (Chen, 2018; Iino, 2018; Wu et al., 2019; Guo, 2020). Based on these findings, the successful eradication of Hp translates into long-term benefits for other microbial drug resistance mechanisms.
The changing profile of Hp antibiotic resistance has reached alarming levels worldwide, with a great impact on the efficacy of empirical therapies. Despite the appropriate eradication regimens, it seems that about 10% of patients might have a persistent infection; hence, further development and discovery of novel regimens and approaches against Hp infection are needed (Siddique et al., 2018).
In an era of increasing MDR, classic bismuth-containing quadruple therapies (BQTs) consisting of bismuth, a PPI, metronidazole, and tetracycline remain the central pylon and are recommended as first-line treatments for Hp infections. In a review focused on the role of bismuth in improving Hp eradication with triple therapies, Graham and Dore suggested that bismuth in addition to a 14-day triple therapy might improve eradication rates with almost 40%, even in a high prevalence of antimicrobial resistance (Graham and Dore, 2016). In addition, taking into account that most eradication regimens are not efficient for treating MDR infections, currently, bismuth-containing quadruple therapy is strongly recommended in regions with high Hp resistance to both clarithromycin and metronidazole, according to Maastricht V consensus (Malfertheiner et al., 2017). A combination of the three-in-one capsules (metronidazole 125 mg, tetracycline 125 mg, and bismuth subcitrate 140 mg) named Pylera has been approved by the United States Food and Drug Administration (FDA) (Treatment Regimens for Helicobacter pylori in Adults—UpToDate, 2022). Pylera could be used in combination with double-dose PPIs and the literature highlighted that eradication success of PPI/Pylera therapy was ≥90% in infections with metronidazole-resistant strains as well as second-line therapy following clarithromycin-based regimens, regardless of PPI dose or type (Nyssen et al., 2019).
Furthermore, maintaining the gastric microenvironment at a pH > 6 represents a key factor in Hp eradication, and the main role of PPIs in the treatment of Hp infection is to elevate the gastric pH. Gene polymorphisms of the principal enzyme implicated in the metabolism of PPIs, CYP2C19, have a major implication in the efficacy of Hp eradication, and literature showed that in CYP2C19 extensive metabolizers, second-generation PPIs such as esomeprazole and rabeprazole provided better eradication rates than first-generation PPIs including omeprazole, pantoprazole, and lansoprazole (McNicholl et al., 2012; Padol et al., 2012). In one study comparing the acid-inhibitory effects of four PPIs (omeprazole, lansoprazole, esomeprazole, and rabeprazole), Sahara et al. reported median pH levels of 5.0, 4.7, 5.4, and 4.8 after treatment with omeprazole, lansoprazole, esomeprazole, and rabeprazole, respectively. The study showed that 20 mg of esomeprazole dosed two times daily provided the strongest inhibition in rapid CYP2C19 metabolizers (Sahara, 2013). In addition, evaluating the effect of CYP2C19 polymorphisms on the efficiency of Hp eradication, literature indicated esomeprazole or rabeprazole as being the less influenced PPIs by CYP2C19 polymorphisms recommended in Hp eradication regimens, particularly in areas with a high proportion of rapid metabolizers, such as Europe and North America (Malfertheiner et al., 2017).
A novel potassium-competitive acid blocker, vonoprazan, is reported to provide better efficacy, safety profile, and prolonged activity when compared to other PPIs, due to the capacity to accumulate in high concentrations and to be slowly cleared from gastric glands, increasing gastric pH levels and producing more potent and sustained acid-inhibitory effects (Graham, 2017). For instance, in Japan, vonoprazan proved higher eradication rates, approximately 90%, in triple therapy with amoxicillin and clarithromycin or metronidazole (Tanabe, 2017; Furuta, 2020). Moreover, in a randomized, double-blind, multicenter, parallel-group study, Murakami et al. reported an eradication rate of Hp of 92.6 vs. 75.9% with lansoprazole (Murakami et al., 2016). In the clarithromycin-susceptible strain subpopulation, a vonoprazan triple therapy was not superior to a lansoprazole triple therapy, with eradication rates being 97.6 vs. 97.3%; whereas in the subpopulation with clarithromycin-resistant strains, it was noticed a higher eradication rate with the vonoprazan triple therapy than the lansoprazole triple therapy, 82.0 vs. 40.0%, both the first-line and second-line vonoprazan triple therapies being well-tolerated. However, a cure rate of 82% is still low; hence, further studies concerning the efficacy of vonoprazan-based therapies with higher doses and longer durations are required (Murakami et al., 2016; Graham, 2017).
New antibacterial agents such as delafloxacin (a newer fluoroquinolone) or flavodoxin inhibitors (7-nitrobenzoxadiazole derivatives) are also mentioned in the literature, but further research regarding their effectiveness and safety is needed.
Antibiotic adjuvants such as probiotics (Pbs) have great potential, considering the ability to raise antibiotic activity and inhibit several resistance mechanisms by immune modulation, producing antioxidants and antimicrobial substances, altering local pH, or affecting Hp colonization and adherence to gastric cells (Ruggiero, 2014; González-Bello, 2017). Numerous meta-analyses highlighted the significant role of Pbs, mainly Lactobacillus or Saccharomyces boulardii or Bacillus clausii, in improving the eradication rate of Hp. In one study, McFarland et al. reported high eradication rates, >90%, using four probiotic mixtures (L. acidophilus/B. animalis, L. helveticus/L. rhamnosus, L. acidophilus/B. longum/E. faecalis and the eight-strain mixture) as adjuvants for Hp eradication, for long duration (3–5 weeks) and at high doses (McFarland et al., 2016). Another meta-analysis found that patients treated with Pbs had a higher eradication rate than those not treated with Pbs (80.3 vs. 72.2%) (Lv, 2015). It seems that Pbs might significantly reduce the adverse effects of the treatment regimens, and the administration before or after the eradication treatment for >2 weeks could improve the eradication success (Lv, 2015).
Recently, the new concept of the anti-biofilm approach has gained interest, but information about the molecular mechanisms which lead to Hp biofilm formation is still deficient. However, two synthetic anti-biofilm peptides, IDR-1018 and DJK-5, were discovered, which are active only against the bacteria without affecting the planktonic H. pylori and affect different biofilm formation stages (Windham et al., 2018). Moreover, in one study, adding a glycolipid biosurfactant, rhamnolipid, raised the anti-biofilm activity of the treatment and the combination of rhamnolipid, PPI, and amoxicillin conducted to 95% biofilm eradication (Chen et al., 2019).
Another potential perspective consists in anti-Hp vaccines, which could be prophylactic, by preventing initial Hp colonization, or therapeutic, as a possible alternative of or adjunct to the eradication therapy (Talebi and Abadi, 2016). One randomized placebo-controlled trial in phase III reported high rates of vaccination success (71.8%) with an anti-Hp oral recombinant vaccine in Chinese children within the first year (Zeng et al., 2015). The development of effective vaccines could represent a powerful strategy to reduce the prevalence of Hp infection and for eradication failure, but it requires epitope mapping, choice of antigen determinants, genomic approach, and safety assessment (Mirzaei et al., 2017).
Last but not least, it is of great importance to perform strain susceptibility testing. One study reported successful eradication of 94% and fewer adverse effects (15%) when using antibiotic susceptibility guided regimens vs. eradication success of 87% with empirical concomitant therapy with more frequent adverse effects (31%), supporting the major impact of culture-guided treatment (Cosme et al., 2016).
The alarming increase of MDR in Hp infection, leading to eradication failure, represents a serious challenge, and addressing this global problem implies not only novel antimicrobial drugs and treatment strategies but also improved diagnostic tools to guide clinicians in further optimizing currently available regimens. Hence, strict adherence to current guidelines and a complex multidisciplinary approach for improving the standard treatment regimens or developing novel strategies in the battle against Hp MDR is highly necessary.
RD, MM, AS, and OA contributed to conception and design of the study. OA organized the database. DP and AC wrote the first draft of the manuscript. RD, AC, AS, and OA wrote sections of the manuscript. All authors have contributed equally in writing, reading, and approving the final manuscript.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The handling editor MCC declared a past co-authorship with the author AB.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Ailloud, F, Didelot, X, Woltemate, S, Pfaffinger, G, Overmann, J, and Bader, R. C. (2019). Within-host evolution of Helicobacter pylori shaped by niche-specific adaptation, intragastric migrations and selective sweeps. Nat. Commun. 10, 1–13. doi: 10.1038/s41467-019-10050-1
Akhavan, B. J., Khanna, N. R., and Vijhani, P. (2022). Amoxicillin Helicobacter Pylori. Treasure Island, FL: StatPearls Publishing (2022). p. 387–396
Almeida, N, Romãozinho, J. M, Donato, M. M, Luxo, C, Cardoso, O, and Cipriano, M. A. (2014). Helicobacter pylori antimicrobial resistance rates in the central region of Portugal. Clin. Microbiol. Infect. 20, 1127–1133. doi: 10.1111/1469-0691.12701
Andersson, D. I., Nicoloff, H., and Hjort, K. (2019). Mechanisms and clinical relevance of bacterial heteroresistance. Nat. Rev. Microbiol. 17, 479–496. doi: 10.1038/s41579-019-0218-1
Anoushiravani, M., Falsafi, T., and Niknam, V. (2009). Proton motive force-dependent efflux of tetracycline in clinical isolates of Helicobacter pylori. J. Med. Microbiol. 58, 1309–1313. doi: 10.1099/jmm.0.010876-0
Attaran, B., Falsafi, T., and Ghorbanmehr, N. (2017). Effect of biofilm formation by clinical isolates of Helicobacter pylori on the efflux-mediated resistance to commonly used antibiotics basic study. World J. Gastroenterol. 23, 1163–1170. doi: 10.3748/wjg.v23.i7.1163
Bacteriology and Epidemiology of Helicobacter pylori Infection–UpToDate. (2022). Available online at: https://www.uptodate.com/contents/bacteriology-and-epidemiology-of-helicobacter-pylori-infection?search=Helicobacterpylori&source$=$search_result&selectedTitle$=$4$\sim$150&usage_type$=$default&display_rank$=$3 (accessed November 21, 2022).
Boehnke, K. F, Valdivieso, M, Bussalleu, A, Sexton, R, Thompson, K. C, and Osorio, S. (2017). Antibiotic resistance among Helicobacter pylori clinical isolates in Lima, Peru. Infect. Drug Resist. 10, 85–90. doi: 10.2147/IDR.S123798
Borka Balas, R., Meliţ, L. E, and Mărginean, C. O. (2022). Worldwide prevalence and risk factors of Helicobacter pylori infection in children. Children. 9, 9. doi: 10.3390/children9091359
Bos, M. P., Tefsen, B., Geurtsen, J., and Tommassen, J. (2004). Identification of an outer membrane protein required for the transport of lipopolysaccharide to the bacterial cell surface. Proc. Natl. Acad. Sci. USA. 101, 9417–9422. doi: 10.1073/pnas.0402340101
Boyanova, L, Gergova, G, Evstatiev, I, Spassova, Z, Kandilarov, N, and Yaneva, P. (2017). Helicobacter pylori resistance to six antibiotics by two breakpoint systems and resistance evolution in Bulgaria. Infect Dis. (Lond). 48, 56–62. doi: 10.3109/23744235.2015.1082035
Boyanova, L., Hadzhiyski, P., Kandilarov, N., Markovska, R., and Mitov, I. (2019). Multidrug resistance in Helicobacter pylori: current state and future directions. Expert Rev. Clin. Pharmacol. 12(9):909–915. doi: 10.1080/17512433.2019.1654858
Chang, W. L., Yeh, Y. C., and Sheu, B. S. (2018). The impacts of H. pylori virulence factors on the development of gastroduodenal diseases. J Biomed Sci. 25, 1–9. doi: 10.1186/s12929-018-0466-9
Chen, L, Xu, W, Lee, A, He, J, Huang, B, and Zheng, W. (2018). The impact of Helicobacter pylori infection, eradication therapy and probiotic supplementation on gut microenvironment homeostasis: an open-label, randomized clinical trial. EBioMedicine. 35:87–96. doi: 10.1016/j.ebiom.2018.08.028
Chen, X., Li, P., Shen, Y., Zou, Y., Yuan, G., and Hu, H. (2019). Rhamnolipid-involved antibiotics combinations improve the eradication of Helicobacter pylori biofilm in vitro: a comparison with conventional triple therapy. Microb. Pathog. 131, 112–119. doi: 10.1016/j.micpath.2019.04.001
Chey, W. D., Leontiadis, G. I., Howden, C. W., and Moss, S. F. (2017). ACG clinical guideline: treatment of Helicobacter pylori Infection. Am. J. Gastroenterol. 112(2):212–238. doi: 10.1038/ajg.2016.563
Cosme, A, Iranzo, S. T, Ros, M. M, Silvestre, M. F-R, Galán, H. A, and Lizasoain, J. (2019). Helicobacter pylori antimicrobial resistance during a 5-year period (2013–2017) in northern Spain and its relationship with the eradication therapies. Helicobacter. 24, e12557. doi: 10.1111/hel.12557
Cosme, A., Lizasoan, J., Montes, M., et al. (2016). Antimicrobial susceptibility-guided therapy versus empirical concomitant therapy for eradication of Helicobacter pylori in a region with high rate of clarithromycin resistance. Helicobacter. 21, 29–34. doi: 10.1111/hel.12231
Den Dunnen, J. T., and Antonarakis, E. (2001). Nomenclature for the description of human sequence variations. Hum. Genet. 109, 121–124. doi: 10.1007/s004390100505
Dore, M. P., Leandro, G., Realdi, G., Sepulveda, A. R., and Graham, D. Y. (2000). Effect of pretreatment antibiotic resistance to metronidazole and clarithromycin on outcome of Helicobacter pylori therapy: a meta-analytical approach. Dig. Dis. Sci. 45, 68–76. doi: 10.1023/a:1005457226341
Eusebi, L. H., Zagari, R. M., and Bazzoli, F. (2014). Epidemiology of Helicobacter pylori Infection. Helicobacter. 19, 1–5. doi: 10.1111/hel.12165
Feldman, M., Friedman, L. S., and Brandt, L. J. (2015). Sleisenger Gastrointestinal and Liver Disease: E-Book: Pathophysiology, Diagnosis, Management. Elsevier Health Sciences.
Fischbach, L., and Evans, E. L. (2007). Meta-analysis: the effect of antibiotic resistance status on the efficacy of triple and quadruple first-line therapies for Helicobacter pylori. Aliment. Pharmacol. Ther. 26, 343–357. doi: 10.1111/j.1365-2036.2007.03386.x
Furuta, T, Yamade, M, Kagami, T, Uotani, T, Suzuki, T, and Higuchi, T. (2020). Dual therapy with vonoprazan and amoxicillin is as effective as triple therapy with vonoprazan, amoxicillin and clarithromycin for eradication of Helicobacter pylori. Digestion. 101, 743–751. doi: 10.1159/000502287
Ge, X, Cai, Y, Chen, Z, Gao, S, Geng, X, and Li, Y. (2018). Bifunctional enzyme SpoT is involved in biofilm formation of Helicobacter pylori with multidrug resistance by upregulating efflux pump Hp1174 (gluP). Antimicrob. Agents Chemother. 62, 11. doi: 10.1128/AAC.00957-18
Gerrits, M. M., Berning, M., Van Vliet, A. H. M., Kuipers, E. J., and Kusters, J. G. (2003). Effects of 16S rRNA gene mutations on tetracycline resistance in Helicobacter pylori. Antimicrob. Agents Chemother. 47, 2984. doi: 10.1128/AAC.47.9.2984-2986.2003
González-Bello, C. (2017). Antibiotic adjuvants – A strategy to unlock bacterial resistance to antibiotics. Bioorg. Med. Chem. Lett. 27, 4221–4228. doi: 10.1016/j.bmcl.2017.08.027
Graham, D. Y. (2017). Vonoprazan Helicobacter pylori eradication therapy: ethical and interpretation issues. Gut. 66, 384–386. doi: 10.1136/gutjnl-2016-311796
Graham, D. Y., and Dore, M. P. (2016). Helicobacter pylori therapy: a paradigm shift. Expert Rev. Anti. Infect. Ther. 14, 577–585. doi: 10.1080/14787210.2016.1178065
Graham, D. Y., Lee, Y. C., and Wu, M. S. (2014). Rational Helicobacter pylori therapy: evidence-based medicine rather than medicine-based evidence. Clin. Gastroenterol Hepatol. 12(2):177-186.e3. doi: 10.1016/j.cgh.2013.05.028
Grossman, T. H. (2016). Tetracycline Antibiotics and Resistance. Cold Spring Harb. Perspect. Med. 6, a025387. doi: 10.1101/cshperspect.a025387
Guo Y, Zhang, Y, Gerhard, M, Gao, J-J, Mejias-Luque, R, Zhang, L. (2020). Effect of Helicobacter pylori on gastrointestinal microbiota: a population-based study in Linqu, a high-risk area of gastric cancer. Gut. 69, 1598–1607. doi: 10.1136/gutjnl-2019-319696
Hathroubi, S., Servetas, S. L., Windham, I., Merrell, D. S., and Ottemann, K. M. (2018). Helicobacter pylori biofilm formation and its potential role in pathogenesis. Microbiol. Mol. Biol. Rev. 82, 2. doi: 10.1128/MMBR.00001-18
Hirata K, Suzuki, H, Nishizawa, T, Tsugawa, H, Muraoka, H, Saito, Y. (2010). Contribution of efflux pumps to clarithromycin resistance in Helicobacter pylori. J. Gastroenterol Hepatol. 25, 1. doi: 10.1111/j.1440-1746.2009.06220.x
Hooi J. K. Y, Lai, W. Y, Ng, W. K, Suen, M. M. Y, Underwood, F. E, Tanyingoh, D. (2017). Global prevalence of Helicobacter pylori infection: systematic review and meta-analysis. Gastroenterology. 153, 420–429. doi: 10.1053/j.gastro.2017.04.022
Iino C, Shimoyama, T, Chinda, D, Arai, T, Chiba, D, Nakaji, S. (2018). Infection of Helicobacter pylori and atrophic gastritis influence Lactobacillus in gut microbiota in a Japanese population. Front. Immunol. 9, 712. doi: 10.3389/fimmu.2018.00712
Kadkhodaei, S., Siavoshi, F., and Akbari Noghabi, K. (2020). Mucoid and coccoid Helicobacter pylori with fast growth and antibiotic resistance. Helicobacter. 25, 2. doi: 10.1111/hel.12678
Kao C-Y, Lee, A-Y, Huang, A-H, Song, P-Y, Yang, Y-J, Sheu, S-M. (2014). Heteroresistance of Helicobacter pylori from the same patient prior to antibiotic treatment. Infect. Genet. Evol. 23, 196–202. doi: 10.1016/j.meegid.2014.02.009
Karamanolis, G. P., Daikos, G. L., Xouris, D., Goukos, D., Delladetsima, I., and Ladas, S. D. (2014). The evolution of Helicobacter pylori antibiotics resistance over 10 years in Greece. Digestion. 90, 229–231. doi: 10.1159/000369898
Karpiński, T. M., Andrzejewska, E., Eder, P., Linke, K., and Szkaradkiewicz, A. (2015). Evaluation of antimicrobial resistance of Helicobacter pylori in the last 15 years in west Poland. Acta Microbiol. Immunol. Hung. 62, 287–293. doi: 10.1556/030.62.2015.3.6
Kasahun, G. G., Demoz, G. T., and Desta, D. M. (2020). Primary resistance pattern of Helicobacter pylori to antibiotics in adult population: a systematic review. Infect. Drug Resist. 13, 1567. doi: 10.2147/IDR.S250200
Kazakos, E. I., Dorrell, N., Polyzos, S. A., Deretzi, G., and Kountouras, J. (2017). Comment on “Effect of biofilm formation by clinical isolates of Helicobacter pylori on the efflux-mediated resistance to commonly used antibiotics.” World J. Gastroenterol. 23, 6194–6196. doi: 10.3748/wjg.v23.i33.6194
Keikha, M., Askari, P., Ghazvini, K., and Karbalaei, M. (2022). Levofloxacin-based therapy as an efficient alternative for eradicating Helicobacter pylori infection in Iran: a systematic review and meta-analysis. J. Glob. Antimicrob. Resist. 29, 420–429. doi: 10.1016/j.jgar.2021.10.019
Khoder, G., Sualeh Muhammad, J., Mahmoud, I., Soliman, S. S. M., and Burucoa, C. (2019). Prevalence of Helicobacter pylori and its associated factors among healthy asymptomatic residents in the United Arab Emirates. Pathogens. 8, 2. doi: 10.3390/pathogens8020044
Kim S. Y, Joo, Y. M, Lee, H. S, Chung, I-S, Yoo, Y-J, Merrell, D. S. (2009). Genetic analysis of Helicobacter pylori clinical isolates suggests resistance to metronidazole can occur without the loss of functional rdxA. J. Antibiot. 62, 43–50. doi: 10.1038/ja.2008.6
Kocazeybek, B., and Tokman, H. B. (2016). Prevalence of primary antimicrobial resistance of H. pylori in Turkey: a systematic review. Helicobacter. 21, 251–260. doi: 10.1111/hel.12272
Kuo C-J, Lee, C-H, Chang, M-L, Lin, C-Y, Lin, W-R, Su, M-Y. (2021). Multidrug resistance: the clinical dilemma of refractory Helicobacter pylori infection. J. Microbiol. Immunol. Infect. 54(6):1184–1187. doi: 10.1016/j.jmii.2021.03.006
Kuo Y-T, Liou, J-M, El-Omar, E. M, Wu, J-Y, Leow, A. H. R, Goh, K. L. (2017). Primary antibiotic resistance in Helicobacter pylori in the Asia-Pacific region: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2, 707–715. doi: 10.1016/S2468-1253(17)30219-4
Kusters, J. G., Van Vliet, A. H. M., and Kuipers, E. J. (2006). Pathogenesis of Helicobacter pylori infection. Clin. Microbiol. Rev. 19, 449. doi: 10.1128/CMR.00054-05
Lee J. H, Ahn, J. Y, Choi, K. D, Jung, H-Y, Kim, J. M, Baik, G. H. (2019). Nationwide antibiotic resistance mapping of Helicobacter pylori in Korea: a prospective multicenter study. Helicobacter. 24, e12592. doi: 10.1111/hel.12592
Lee, J. Y., Kim, N., Nam, R. H., In Choi, S., Lee, J. W., and Lee, D. H. (2019). Primary and secondary antibiotic resistance of Helicobacter pylori in Korea from 2003 to 2018. Helicobacter. 24, e12660. doi: 10.1111/hel.12660
Lee S. M, Kim, N, Kwon, Y. H, Nam, R. H, Kim, J. M, Park, J. Y. (2018). rdxA, frxA, and efflux pump in metronidazole-resistant Helicobacter pylori: their relation to clinical outcomes. J. Gastroenterol. Hepatol. 33, 681–688. doi: 10.1111/jgh.13906
Liou J-M, Chen, C-C, Chang, C-Y, Chen, M-J, Fang, Y-J, Lee, J-Y. (2013). Efficacy of genotypic resistance-guided sequential therapy in the third-line treatment of refractory Helicobacter pylori infection: a multicentre clinical trial. J. Antimicrob. Chemother. 68, 450–456.
Lv Z, Wang, B, Zhou, X, Wang, F, Xie, Y, Zheng, H. (2015). Efficacy and safety of probiotics as adjuvant agents for Helicobacter pylori infection: a meta-analysis. Exp. Ther. Med. 9, 707. doi: 10.3892/etm.2015.2174
Malfertheiner, P., Megraud, F., O'Morain, C., et al. (2017). Management of Helicobacter pylori infection—the maastricht v/florence consensus report. Gut. 66, 6–30. doi: 10.1136/gutjnl-2016-312288
Mansour K. B, Fendri, C, Battikh, H, Garnier, M, Zribi, M, Jlizi, A. (2016). Multiple and mixed Helicobacter pylori infections: Comparison of two epidemiological situations in Tunisia and France. Infect. Genet. Evol. 37, 43–48. doi: 10.1016/j.meegid.2015.10.028
McFarland, L. V., Huang, Y., Wang, L., and Malfertheiner, P. (2016). Systematic review and meta-analysis: multi-strain probiotics as adjunct therapy for Helicobacter pylori eradication and prevention of adverse events. United Eur Gastroenterol J. 4, 546–561. doi: 10.1177/2050640615617358
McNicholl, A. G., Linares, P. M., Nyssen, O. P., Calvet, X., and Gisbert, J. P. (2012). Meta-analysis: esomeprazole or rabeprazole vs. first-generation pump inhibitors in the treatment of Helicobacter pylori infection. Aliment Pharmacol. Ther. 36, 414–425. doi: 10.1111/j.1365-2036.2012.05211.x
Megraud F, Bruyndonckx, R, Coenen, S, Wittkop, L, Huang, T-D, Hoebeke, M. (2021). Helicobacter pylori resistance to antibiotics in Europe in 2018 and its relationship to antibiotic consumption in the community. Gut. 70, 1815–1822. doi: 10.1136/gutjnl-2021-324032
Megraud F, Coenen, S, Versporten, A, Kist, M, Lopez-Brea, M, Hirschl, A. M. (2013). Helicobacter pylori resistance to antibiotics in Europe and its relationship to antibiotic consumption. Gut. 62, 34–42. doi: 10.1136/gutjnl-2012-302254
Mégraud, F., and Lehours, P. (2007). Helicobacter pylori detection and antimicrobial susceptibility testing. Clin. Microbiol. Rev. 20, 280–322. doi: 10.1128/CMR.00033-06
Mehrabadi, J. F., Sirous, M., Daryani, N. E., Eshraghi, S., Akbari, B., and Shirazi, M. H. (2010). Assessing the role of the RND efflux pump in metronidazole resistance of Helicobacter pylori by RT-PCR assay. J. Infect. Dev. Ctries. 5, 88–93. doi: 10.3855/jidc.1187
Metronidazole: An overview–UpToDate. (2022). Available online at: https://www.uptodate.com/contents/metronidazole-an-overview?source=mostViewed_widget#H5 (accessed December 2, 2022).
Mirzaei, N., Poursina, F., Moghim, S., Rashidi, N., and Ghasemian Safaei, H. (2017). The study of H. pylori putative candidate factors for single- and multi-component vaccine development. Crit. Rev. Microbiol. 43, 631–650. doi: 10.1080/1040841X.2017.1291578
Mourad-Baars, P. E. C., Wunderink, H. F., Luisa Mearin, M., Veenendaal, R. A., Wit, J. M., and Veldkamp, K. E. (2015). Low antibiotic resistance of Helicobacter pylori in the netherlands. Helicobacter. 20, 69–70. doi: 10.1111/hel.12175
Murakami, K., Sakurai, Y., Shiino, M., Funao, N., Nishimura, A., and Asaka, M. (2016). Vonoprazan, a novel potassium-competitive acid blocker, as a component of first-line and second-line triple therapy for Helicobacter pylori eradication: a phase III, randomised, double-blind study. Gut. 65, 1439–1446. doi: 10.1136/gutjnl-2015-311304
Nishizawa, T., Maekawa, T., Watanabe, N., et al. (2015). Clarithromycin versus metronidazole as first-line Helicobacter pylori eradication. J. Clin. Gastroenterol. 49, 468–471. doi: 10.1097/MCG.0000000000000165
Nyssen, O. P., McNicholl, A. G., and Gisbert, J. P. (2019). Meta-analysis of three-in-one single capsule bismuth-containing quadruple therapy for the eradication of Helicobacter pylori. Helicobacter. 24, e12570. doi: 10.1111/hel.12570
Ogino S, Gulley, M. L, den Dunnen, J. T, Wilson, R. B, Committtee, A. f. M. P. T. a. E. (2007). Standard mutation nomeclature in molecular diagnostics: practical and educational challenges. J Mol Diagnostics. 9, 1–6. doi: 10.2353/jmoldx.2007.060081
Okamoto T, Yoshiyama, H, Nakazawa, T, Park, I-D, Chang, M-W, Yanai, H. (2002). A change in PBP1 is involved in amoxicillin resistance of clinical isolates of Helicobacter pylori. J. Antimicrob. Chemother. 50, 849–856. doi: 10.1093/jac/dkf140
Padol, S., Yuan, Y., Thabane, M., Padol, I. T., and Hunt, R. H. (2012). The Effect of CYP2C19 Polymorphisms on H. pylori Eradication. Am. J. Gastroenterol. 101, 1467–1475. doi: 10.1111/j.1572-0241.2006.00717.x
Phan T. N, Santona, A, Tran, V. H, Tran, T. N. H, Le, V. A, Cappuccinelli, P. (2015). High rate of levofloxacin resistance in a background of clarithromycin- and metronidazole-resistant Helicobacter pylori in Vietnam. Int. J. Antimicrob. Agents. 45, 244–248. doi: 10.1016/j.ijantimicag.2014.10.019
Podolsky, D. K., Camilleri, M., Fitz, J. G., Kalloo, A. N., Shanahan, F., and Wang, T. C. (2015). Yamada's Textbook of Gastroenterology. New York, NY: John Wiley & Sons. doi: 10.1002/9781118512074
Qureshi, N. N., Gallaher, B., and Schiller, N. L. (2014). Evolution of amoxicillin resistance of Helicobacter pylori in Vitro: Characterization of resistance mechanisms. Microb. Drug Resist. 20, 509–516. doi: 10.1089/mdr.2014.0019
Regnath, T., Raecke, O., Enninger, A., and Ignatius, R. (2017). Increasing metronidazole and rifampicin resistance of Helicobacter pylori isolates obtained from children and adolescents between 2002 and 2015 in southwest Germany. Helicobacter. 22, e12327. doi: 10.1111/hel.12327
Rimbara, E., Noguchi, N., Kawai, T., and Sasatsu, M. (2012). Fluoroquinolone resistance in Helicobacter pylori: role of mutations at position 87 and 91 of GyrA on the level of resistance and identification of a resistance conferring mutation in GyrB. Helicobacter. 17, 36–42. doi: 10.1111/j.1523-5378.2011.00912.x
Ruggiero, P. (2014). Use of probiotics in the fight against Helicobacter pylori. World J. Gastrointest. Pathophysiol. 5, 384. doi: 10.4291/wjgp.v5.i4.384
Sahara S, Sugimoto, M, Uotani, T, Ichikawa, H, Yamade, M, Iwaizumi, M. (2013). Twice-daily dosing of esomeprazole effectively inhibits acid secretion in CYP2C19 rapid metabolisers compared with twice-daily omeprazole, rabeprazole or lansoprazole. Aliment. Pharmacol. Ther. 38, 1129–1137. doi: 10.1111/apt.12492
Saranathan R, Levi, M. H, Wattam, A. R, Malek, A, Asare, E, Behin, D. S. (2020). Helicobacter pylori infections in the bronx, New York: surveying antibiotic susceptibility and strain lineage by whole-genome sequencing. J. Clin. Microbiol. 58, 3. doi: 10.1128/JCM.01591-19
Savoldi, A., Carrara, E., Graham, D. Y., Conti, M., and Tacconelli, E. (2018). Prevalence of Antibiotic Resistance in Helicobacter pylori: a systematic review and meta-analysis in world health organization regions. Gastroenterology. 155, 1372–1382.e17. doi: 10.1053/j.gastro.2018.07.007
Shah, S. C., Iyer, P. G., and Moss, S. F. (2021). AGA clinical practice update on the management of refractory Helicobacter pylori infection: expert review. Gastroenterology. 160, 1831. doi: 10.1053/j.gastro.2020.11.059
Shetty, V., Lamichhane, B., Yen Tay, C., Pai, G., Lingadaika, R., Balaraju, G., et al. (2019). High primary resistance to metronidazole and levofloxacin, and a moderate resistance to clarithromycin in Helicobacter pylori isolated from Karnataka patients. BMC. 11, 21. doi: 10.1186/s13099-019-0305-x
Shiota, S., Reddy, R., Alsarraj, A., El-Serag, H. B., and Graham, D. Y. (2015). Antibiotic Resistance of Helicobacter pylori Among Male United States Veterans. Clin. Gastroenterol. Hepatol. 13(9):1616. doi: 10.1016/j.cgh.2015.02.005
Siddique, O., Ovalle, A., Siddique, A. S., and Moss, S. F. (2018). Helicobacter pylori Infection: An update for the internist in the age of increasing global antibiotic resistance. Am. J. Med. 131, 473–479. doi: 10.1016/j.amjmed.2017.12.024
Singh, S., Singh, S. K., Chowdhury, I., and Singh, R. (2017). Understanding the mechanism of bacterial biofilms resistance to antimicrobial agents. Open Microbiol. J. 11, 53. doi: 10.2174/1874285801711010053
Smiley, R., Bailey, J., Sethuraman, M., Posecion, N., and Showkat Ali, M. (2013). Comparative proteomics analysis of sarcosine insoluble outer membrane proteins from clarithromycin resistant and sensitive strains of Helicobacter pylori §. J. Microbiol. 51, 612–618. doi: 10.1007/s12275-013-3029-5
Smith, S. M., O'Morain, C., and McNamara, D. (2014). Antimicrobial susceptibility testing for Helicobacter pylori in times of increasing antibiotic resistance. World J. Gastroenterol. 20(29):9912–9921. doi: 10.3748/wjg.v20.i29.9912
Stone G. G, Shortridge, D, Flamm, R. K, Versalovic, J, Beyer, J, Idler, K. (1996). Identification of a 23S rRNA gene mutation in clarithromycin-resistant Helicobacter pylori. Helicobacter. 1, 227–228. doi: 10.1111/j.1523-5378.1996.tb00043.x
Sugimoto M, Sahara, S, Ichikawa, H, Kagami, T, Ban, H, Otsuka, T. (2017). Four-times-daily dosing of rabeprazole with sitafloxacin, high-dose amoxicillin, or both for metronidazole-resistant infection with Helicobacter pylori in Japan. Helicobacter. 22, e12319. doi: 10.1111/hel.12319
Sukri A, Hanafiah, A, Yusoff, H, Nizam, N. A. S, Nameyrra, Z, Wong, Z. (2022). Multidrug-resistant Helicobacter pylori strains: a five-year surveillance study and its genome characteristics. Antibiotics. 11, 1–12. doi: 10.3390/antibiotics11101391
Talebi, A., and Abadi, B. (2016). Inevitable approach. World J. Gastroenterol. 22, 3150–3157. doi: 10.3748/wjg.v22.i11.3150
Talebi, A., and Abadi, B. (2017). Resistance to clarithromycin and gastroenterologist's persistence roles in nomination for Helicobacter pylori as high priority pathogen by World Health Organization. World J. Gastroenterol. 23, 6379–6384. doi: 10.3748/wjg.v23.i35.6379
Tanabe H, Ando, K, Sato, K, Ito, T, Goto, M, Sato, T. (2017). Efficacy of vonoprazan-based triple therapy for Helicobacter pylori eradication: a multicenter study and a review of the literature. Dig Dis Sci. 62, 3069–3076. doi: 10.1007/s10620-017-4664-1
Tankovic, J., Lascols, C., Sculo, Q., Petit, J. C., and Soussy, C. J. (2003). Single and double mutations in gyra but not in gyrb are associated with low- and high-level fluoroquinolone resistance in Helicobacter pylori. Antimicrob. Agents Chemother. 47, 3942. doi: 10.1128/AAC.47.12.3942-3944.2003
Treatment Regimens for Helicobacter pylori in Adults—UpToDate. (2022). Available online at: https://www.uptodate.com/contents/treatment-regimens-for-helicobacter-pylori-in-adults (accessed November 23, 2022).
Tshibangu-Kabamba, E., and Yamaoka, Y. (2021). Helicobacter pylori infection and antibiotic resistance–from biology to clinical implications. Nat. Rev. Gastroenterol. Hepatol. 18, 613–629. doi: 10.1038/s41575-021-00449-x
Tuan V. P, Narith, D, Tshibangu-Kabamba, E, Dung, H. D. Q, Viet, P. T, Sokomoth, S. (2019). A next-generation sequencing-based approach to identify genetic determinants of antibiotic resistance in cambodian Helicobacter pylori clinical isolates. J Clin Med. 8, 858. doi: 10.3390/jcm8060858
Wang B, Lv, Z-F, Wang, Y-H, Wang, H, Liu, X-Q, Xie, Y. (2014). Standard triple therapy for Helicobacter pylori infection in China: a meta-analysis. World J. Gastroenterol. 20(40):14973–14985. doi: 10.3748/wjg.v20.i40.14973
Windham, I. H., Servetas, S. L., Whitmire, J. M., Pletzer, D., Hancock, R. E. W., and Merrell, D. S. (2018). Helicobacter pylori biofilm formation is differentially affected by common culture conditions, and proteins play a central role in the biofilm matrix. Appl. Environ. Microbiol. 84, e00391–18. doi: 10.1128/AEM.00391-18
Wu, L., Wang, Z., Sun, G., et al. (2019). Effects of anti-H. pylori triple therapy and a probiotic complex on intestinal microbiota in duodenal ulcer. Sci. Rep. 9, 1. doi: 10.1038/s41598-019-49415-3
Zeng, M., Mao, X. H., Li, J. X., et al. (2015). Efficacy, safety, and immunogenicity of an oral recombinant Helicobacter pylori vaccine in children in China: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 386, 1457–1464. doi: 10.1016/S0140-6736(15)60310-5
Zhang, Y. X., Zhou, L. Y., Song, Z. Q., Ding, Y., Zhang, J. Z., and He, L. H. (2015). Primary antibiotic resistance of Helicobacter pylori strains isolated from patients with dyspeptic symptoms in Beijing: a prospective serial study Prospective Study. World J. Gastroenterol. 21, 2786–2792. doi: 10.3748/wjg.v21.i9.2786
Zollner-Schwetz I, Leitner, E, Plieschnegger, W, Semlitsch, G, Stepan, V, Reiter, L. (2016). Primary resistance of Helicobacter pylori is still low in Southern Austria. Int. J. Med. Microbiol. 306, 206–211. doi: 10.1016/j.ijmm.2016.04.003
Keywords: Helicobacter pylori, infection, multidrug resistance, antibiotic, eradication
Citation: Dascălu RI, Bolocan A, Păduaru DN, Constantinescu A, Mitache MM, Stoica AD and Andronic O (2023) Multidrug resistance in Helicobacter pylori infection. Front. Microbiol. 14:1128497. doi: 10.3389/fmicb.2023.1128497
Received: 20 December 2022; Accepted: 30 January 2023;
Published: 27 February 2023.
Edited by:
Mariana Carmen Chifiriuc, University of Bucharest, RomaniaReviewed by:
Luminita Marutescu, University of Bucharest, RomaniaCopyright © 2023 Dascălu, Bolocan, Păduaru, Constantinescu, Mitache, Stoica and Andronic. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dan Nicolae Păduaru,  ZGFuLnBhZHVyYXJ1QHVtZmNkLnJv
ZGFuLnBhZHVyYXJ1QHVtZmNkLnJv
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.