- 1Laboratory of Forest Protection, Graduate School of Bioagricultural Sciences, Nagoya University, Nagoya, Japan
- 2Department of Forest Microbiology, Forestry and Forest Products Research Institute (FFPRI), Tsukuba, Japan
Ficus carica plantations in Japan were first reported to be infested by an ambrosia beetle species, identified as Euwallacea interjectus, in 1996. The purpose of this study was to determine the symbiotic fungi of female adults of E. interjectus emerging from F. carica trees infected with fig wilt disease (FWD). Dispersal adults (51 females) of E. interjectus, which were collected from logs of an infested fig tree in Hiroshima Prefecture, Western Japan, were separated into three respective body parts (head, thorax, and abdomen) and used for fungal isolation. Isolated fungi were identified based on the morphological characteristics and DNA sequence data. Over 13 species of associated fungi were detected, of which a specific fungus, Fusarium kuroshium, was dominant in female head (including oral mycangia). The plant-pathogenic fungus of FWD, Ceratocystis ficicola, was not observed within any body parts of E. interjectus. We further discussed the relationship among E. interjectus and its associated fungi in fig tree.
Introduction
Ambrosia beetles are wood-inhabiting insects which cultivate fungi as a source of nutrition typically in dead, but occasionally also in healthy, woody hosts (Hulcr and Stelinski, 2017). Most of the ambrosia beetles store and transport their fungal associates to and from their natal galleries in specialized transporting organs called mycangia (singular form: mycangium) (Batra, 1963; Hulcr and Cognato, 2010; Joseph and Keyhani, 2021) or mycetangia (singular form: mycetangium) (Vega and Biedermann, 2020). Once released from the mycangia, the fungi grow in galleries to provide nutrition to the beetles and their offspring (Batra, 1963; Mueller et al., 2005; Bentz and Six, 2006). Ambrosia beetle-associated fungi play a key role in the wood by competing with the decaying fungi of the woody tissues that they infest and thereby reducing their rate of decay (Skelton et al., 2019). Many ambrosia beetles and their associated fungi are ecologically constrained to the dying or dead trees and usually remain harmless even after establishment in non-native regions (Cognato et al., 2015). Nevertheless, some invasive symbioses have been found to shift from non-pathogenic saprotrophy in native ranges to prolific tree killing in invaded ranges and cause significant damage (Hulcr and Dunn, 2011; Ranger et al., 2015; Hulcr et al., 2017; Carrillo et al., 2019; Joseph and Keyhani, 2021).
Ambrosia beetles are successful invaders (Cognato et al., 2015; Li et al., 2015; Stouthamer et al., 2017; Dzurenko et al., 2021) which exist in part due to their haplodiploid mating system, wide host range, and association with the primary ambrosia fungi, such as Fusarium species, which can act as both the nutritional symbionts and weak phytopathogens (Kasson et al., 2013; O’Donnell et al., 2015). There were many differences in fungal symbionts, geographic range, host preference, and potential for symbiont switching in natural populations of these beetles (Carrillo et al., 2019, 2020). Fusarium symbionts of ambrosia beetles belong to the genus Euwallacea from the monophyletic group within clade 3 of the Fusarium solani species complex (FSSC). It is also known as the ambrosia Fusarium clade (AFC; Kasson et al., 2013). Recently, some species in the FSSC, including the AFC species, were reassigned to the genus Neocosmospora based on gene sequences and morphology (Sandoval-Denis and Crous, 2018; Sandoval-Denis et al., 2019; Veloso et al., 2021). Previous studies have identified the phenotypic characteristics that can be used to distinguish the AFC species (Aoki et al., 2018; Lynn et al., 2021) and elucidate their host range and pathogenic potential (Eskalen et al., 2013; Kuroda et al., 2017). This provided a basis for the development, management, monitoring, and eradication strategies. However, the specific mechanism of symbiosis between the Euwallacea beetle and the AFC has not yet been explored. To date, only eight of the 19 AFC species in the world have been described (Table 1). It has been argued for a relatively long time that the mycangial symbionts of ambrosia beetles are strictly asexual (Mayers et al., 2017). Obligate ambrosia beetle–fungus mutualism has been represented as a one-on-one relationship (Batra, 1963). Noticeably, it has been shown in recent discoveries that the relationship between Euwallacea beetle and its AFC symbiosis is more likely promiscuous in native areas as opposed to strictly obligate to a specific combination of fungi which is observed in the invaded areas (Carrillo et al., 2019).
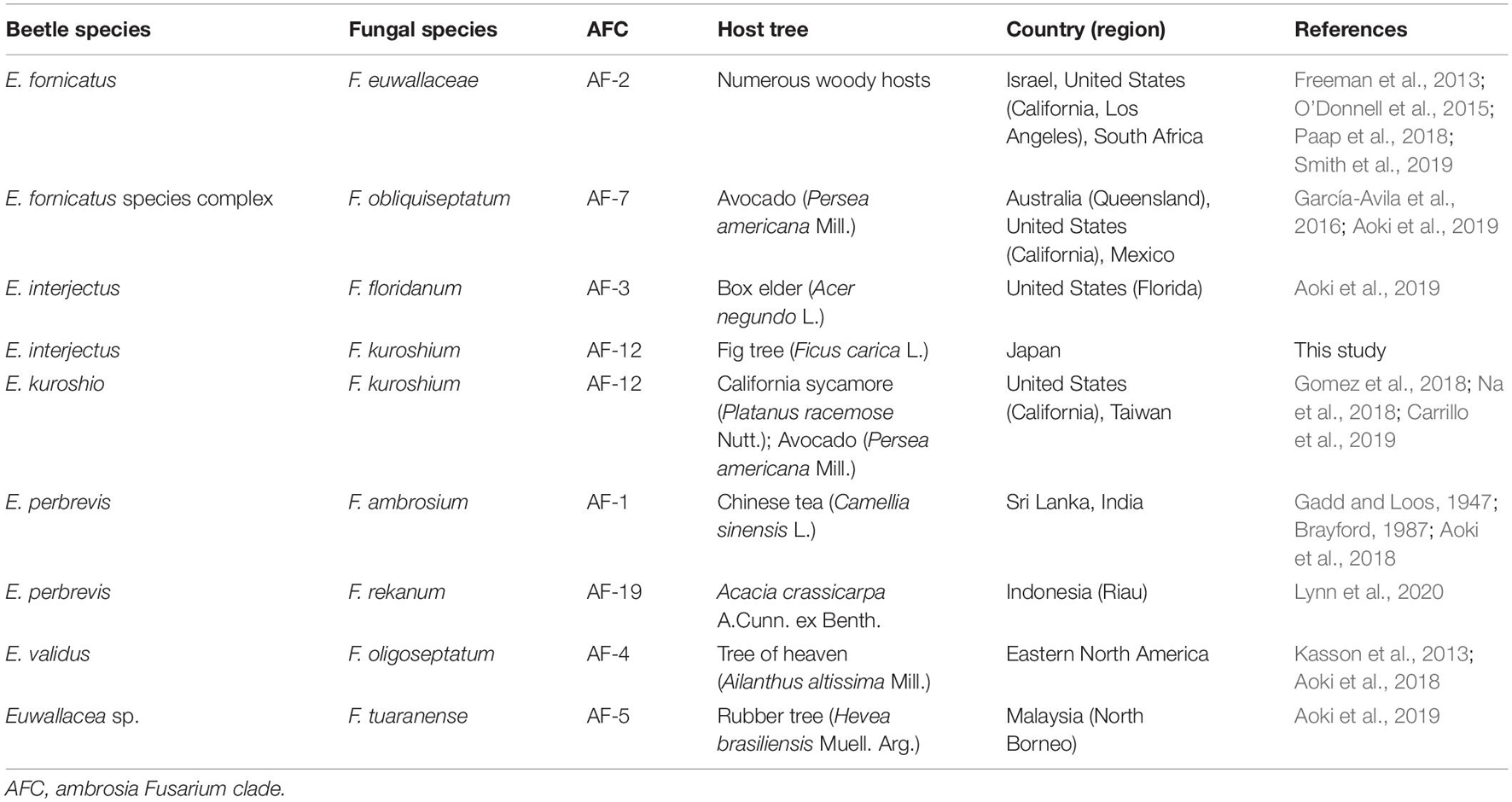
Table 1. A worldwide summary of Euwallacea beetle–Fusarium fungus symbiosis in relation to tree damage.
In Japan, the known causal agent of wilt disease on fig trees (Ficus carica L.) is Ceratocystis ficicola (Kajitani and Masuya, 2011). Fig wilt disease (FWD) has been found to be caused by the infestation of ambrosia beetle, Euwallacea interjectus (Blandford) (Coleoptera: Curculionidae: Scolytinae) (Kajitani, 1996, 1999; Morita et al., 2012; Kajii et al., 2013). It is a wood-boring pest of many tree species, such as poplar trees in Argentina and China and box elder (Acer negundo L.) in the United States (Samuelson, 1981; Aoki et al., 2019; Landi et al., 2019; Wang et al., 2020). Kajitani (1996) found that E. interjectus probably carried C. ficicola because C. ficicola was isolated from the fig twig into which its elytron (upper wing) was inoculated (Kajitani, 1999). In contrast, a few recent studies have discovered the presence of mycangia in the oral region (head) of female adults rather than in elytra (Li et al., 2018; Jiang et al., 2019; Figure 1). However, the role of E. interjectus in FWD as a suspected vector for C. ficicola is still unclear in Japan. It has been hypothesized that the oral mycangia of female adults might carry the fungal symbiont and/or plant pathogen to damage the host trees. Therefore, further investigation of fungal associates within the oral mycangia of E. interjectus would help in understanding the ecology and tree damage of the beetle species.

Figure 1. Adult Euwallacea interjectus. (A) Female and (B) male in lateral view. (C) Cross-sections of oral mycangia (colored in yellow) with compacted fungal inoculum (colored in red) in female from red dotted line in panel (A) by micro-computed tomography scan in Jiang et al. (2019) (unpublished image).
The purpose of this study was to investigate the beetle–fungi relationship via the identification of the recovered fungal species from the ambrosia beetle (E. interjectus). This was carried out by using morphology and multi-gene phylogeny, which helped in determining whether these fungal associates are pathogens of the host (fig tree). Of all the fungal associates, our aim was to isolate the fungal species present among the body parts (head, thorax, and abdomen). These were further examined for the identification of fungal species in the wild E. interjectus population from the diseased fig tree. This would help in clarifying the role of E. interjectus in spreading FWD in Japan. This study contributes to the better understanding of insect–fungus symbiosis in microbial ecology and infection by insects with fungal symbiont in applied microbiology.
Materials and Methods
Insect Preparation
Many pinholes made by insects were found on fig trees (variety Houraishi) infected with FWD in a fig orchard at Fukuyama, Hiroshima Prefecture, Western Japan. These insects were assumed to be E. interjectus based on the preliminary observation. All the insect specimens were identified by comparing the observed morphological characteristics with the previously published data (Cognato et al., 2015). This was done using an OLYMPUS SZ6045-TRPT stereomicroscope (Olympus Optical Co., Ltd., Tokyo, Japan) equipped with an OLYMPUS DP12 high-resolution microscope camera and Microsystems Digital Imaging software. Two branches, A (diameter = 11.5 cm; length = 31.5 cm) and B (diameter = 12.8 cm; length = 39.5 cm), were cut from different sections of the same infected fig tree on December 12, 2017. The infected fig tree in this study had typical FWD symptoms, showing wilting and discoloration of leaves. These branches had brown-discolored sapwood with no decay on the crosscut areas. They were stored in a cage present in the greenhouse at the Nagoya University Higashiyama Campus on December 15, 2017. During the emergence of insects from the branches, newly emerging beetle adults were sampled in the cage every 1 to 2 days. All specimens were directly kept into 1.5-ml sterile microcentrifuge tubes using disinfected forceps and transferred to the laboratory.
Fungal Isolation and Culturing
Potato dextrose agar (PDA: 4 g potato starch, 20 g dextrose, 15 g agar, and distilled water up to 1 L) supplemented with streptomycin sulfate (100 mg/L) and synthetic low-nutrient agar (SNA: 1 g KH2PO4, 1 g KNO3, 0.5 g MgSO4⋅7H2O, 0.5 g KCl, 0.2 g glucose, 20 g agar, and 1 L distilled water) were autoclaved at 121°C for 15 min and were used in all the experiments. Sterile petri dishes (INA-OPTIKA Co., Ltd., Osaka, Japan) were prepared with 10 ml of PDA or SNA culture medium and kept in a sterile laminar flow chamber under UV light until the solidification of the culture medium. Fungal cultures on the PDA were used to characterize the colony and odor, whereas those on SNA were employed for the examination of microscopic characteristics.
Whole beetles were surface-washed by vortexing for 15 s in 1 ml sterile distilled water and one small drop of Tween 20 (<10 μl). A second wash was performed using sterile distilled water only. Each washed beetle was separated into body parts, viz., head, thorax, and abdomen, via two sterilized pins under an OLYMPUS SZ6045-TRPT stereomicroscope (Olympus Optical Co., Ltd., Tokyo, Japan). Afterward, the three respective body parts were inoculated individually on PDA plates. The fungi were allowed to grow (25°C, dark) for 5 to 10 days before sub-identification. The total number of colonies formed on each plate was recorded, and five colonies from each sample were selected and streaked for purification. Representative subcultures of the dominant morphotype recovered from the three respective body parts were stored on PDA slants at room temperature.
Microscopic Observation
The specimens were initially characterized based on morphology and were grouped based on the similarities in the morphotype (such as growth speed, color, thickness, transparency, and texture). The spores were observed and photographed using a phase-contrast microscope Olympus BX41 (Olympus Optical Co., Ltd., Tokyo, Japan) and an Olympus FX380 3CCD digital camera system connected to the FLvFs software (Flovel Image Filling System version 2.30.03, Tokyo, Japan).
Fungal Identification
At least one culture from different groups was selected for DNA extraction, including a representative from each morphological group. Fungal identification was performed based on the morphological characteristics. This data was further supported by the sequencing of the internal transcribed spacer (ITS) rDNA and ambrosia Fusarium sequence data of three genes, viz., translational elongation factor 1-α (TEF1) and DNA-directed RNA polymerase II largest (RPB1) and second largest subunit (RPB2).
The mycelium of each isolate was harvested from the 2-week-old plates, and their DNAs were extracted by PrepMan® Ultra Sample preparation reagent (Applied BiosystemsTM) as per the instructions of the manufacturer. Polymerase chain reaction (PCR) amplification was conducted by using these extracted DNAs and primers ITS5/ITS4 (White et al., 1990) for ITS rDNA, EF1/EF2 (O’Donnell et al., 1998) for TEF1, newly designed primers, AF-RPB1F (5′-TTCCTCACCAAGGAGCAGAT-3′)/AF-RPB1R (5′-TCGCCAATAACATGGTCAAA-3′) for RPB1 and AF-RPB2F (5′-ACGATCCATGGAGTTCCTCA-3′)/AF-RPB2R (5′-CGTTGTACATGACCTCGAAA-3′) for RPB2. Then, 20 μl of PCR mixture consisted of 10 μl of GoTaq master mix (Promega Co., Ltd.), 1 μl of DNA template, 0.5 μl of each primer (10 mM), and 8 μl of sterile distilled water. The PCR conditions included initial denaturation at 95°C for 4 min, 40 cycles of 30 s at 94°C, 30 s at 53°C (annealing temperature), and 50 s at 72°C, with a final elongation at 72°C for 8 min for ITS regions. For the other sequence regions, appropriate annealing temperatures were used as 55°C for EF1/EF2 and AF-RPB2 and 48°C for AF-RPB1. The amplicons were confirmed by running the PCR product on 1% agarose gel with GelRedTM Nucleic Acid Gel Stain (Biotium, Hayward, CA, United States). Furthermore, the PCR products were purified by ExoSAP-IT PCR Product Cleanup reagent (Applied BiosystemsTM) following the instructions of the manufacturer and sequenced in both directions by using the BigDye Terminator v. 3.1 ready reaction mixture (Perkin-Elmer, Warrington, United Kingdom). Sequence data was obtained on ABI PRISMTM 3100 genetic analyzer (Applied Biosystems, Foster City, CA, United States). They were assembled using CAP3 (Huang and Madan, 1999), combined in species, edited on AliView (Larsson, 2014), and used for phylogenetic analysis.
Phylogenetic Analyses
Multigene phylogenetic analyses of Fusarium spp. were conducted in this study using concatenated DNA sequences of the ITS rDNA, TEF1-α, RPB1, and RPB2 gene regions. The sequences were obtained from the NCBI database for 64 isolates previously used in the AFC phylogenetic analyses (Kasson et al., 2013; Carrillo et al., 2019; Sandoval-Denis et al., 2019; Lynn et al., 2020) (the accession numbers are included in Supplementary Table 2). It was aligned using MUSCLE algorithm (Edgar, 2004) and adjusted manually. The maximum-likelihood tree with the ultrafast bootstrap of 1,000 replicates (Hoang et al., 2018) was inferred using the IQ-TREE program (Nguyen et al., 2015) with the Model Finder option (Kalyaanamoorthy et al., 2017). Fusarium pseudensiforme (NRRL 46517) was used as the outgroup.
Data Analyses
The relative dominance (RD, %) and frequency of occurrence (FO, %) of fungal species isolated from the head, thorax, and abdomen of female adults of E. interjectus were calculated using the equations listed below:
Results
Beetle Specimens
In total, 453 beetle specimens (♀: 439; ♂: 14) were collected from fig branches during March 2, 2018–August 20, 2018. The dispersal peak of females and males was observed in April and March, respectively. Based on morphological characterization, all the beetles were identified as E. interjectus (Figure 1). In this study, the sex ratio of adults emerging from the galleries was about 31:1 (♀: ♂). This value was much higher than that of Xyleborini ambrosia beetle which has an average ratio of 13:1 (Kirkendall, 1993; Cooperband et al., 2016; Castro et al., 2019; Cognato et al., 2020). A total of 51 adult specimens (female and alive) were randomly selected and used for fungal isolation during March 2, 2018–April 6, 2018 (Supplementary Table 1).
Fungal Flora
A total of 96 isolates were selected and purified. Of these, 34 isolates were from the head, 31 were from the thorax, and 31 were from the abdomen (Table 2). After morphological categorization, 25 selected isolates were sequenced. Finally, 13 filamentous fungi (Fusarium kuroshium, Arthrinium arundinis, Cladosporium cladosporioides, Acremonium sp., Fusarium decemcellulare, Xylariales sp., Pithomyces chartarum, Roussoella sp., Phialophora sp., Stachybotrys longispora, Paecilomyces formosus, Sarocladium implicatum, and Bionectria pityrodes) were identified, and two unknown isolates could also be detected (Table 2).
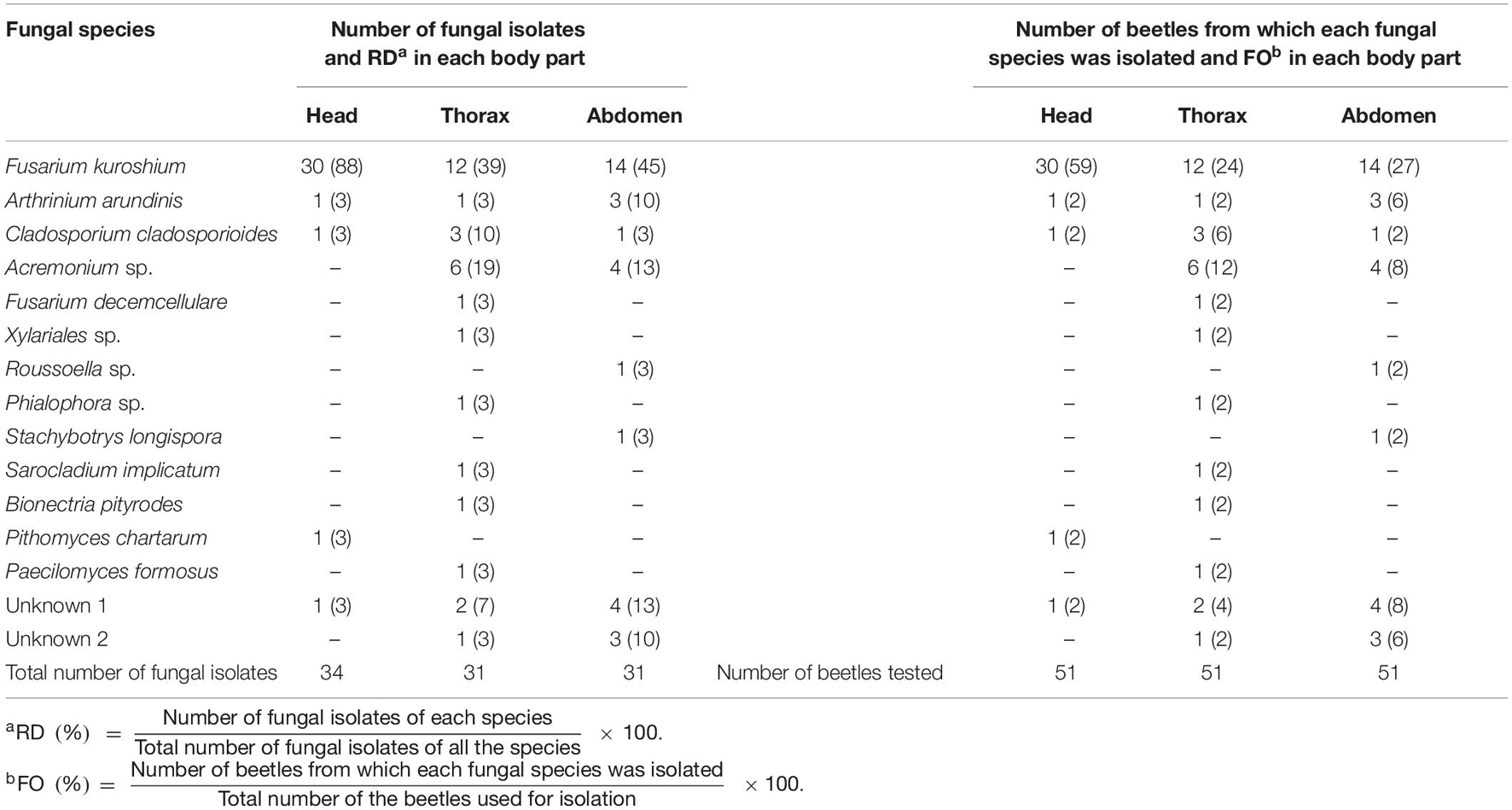
Table 2. Relative dominance (RD, %) and frequency of occurrence (FO, %) of fungal species isolated from the head, thorax, and abdomen of female adults of Euwallacea interjectus in this study.
Phylogenetic Analysis
We obtained each sequence data for FSSC in this study (427 bp for ITS, 380 bp for TEF1, 1,400 bp for RPB1, and 658 bp for RPB2). The sequences of ITS were homologous to those of F. euwallaceae and F. kuroshium. The sequences of EF1-α genes were homologous to those of F. kuroshium (KX262220 and KX262216). The RPB1 sequences were similar to those of F. kuroshium and F. floridanum, but there were 3-bp differences among them. The RPB2 sequences were identical to those of Fusarium spp. (AF13 and AF17) and F. kuroshium. In the phylogenetic tree constructed by IQTree, F. kuroshium was placed in the distinct clade among the ambrosia fusaria but closely related to Fusarium spp. (AF13 and AF14). The number of parsimony informative sites were 9, 13, 69, and 40 bp for ITS, EF1-α, RPB1, and RPB2, respectively.
Fusarium kuroshium was identified based on morphology (Figure 2) and phylogenetic placement (Figure 3). It was characterized by the greenish conidial masses formed on the sporodochia and dolphin-shaped macroconidia, which are typical of ambrosia fusaria associated with Euwallacea beetles (Figure 2). It was phylogenetically from the clade of ambrosia Fusarium but closely related to F. kuroshium (Figure 3).
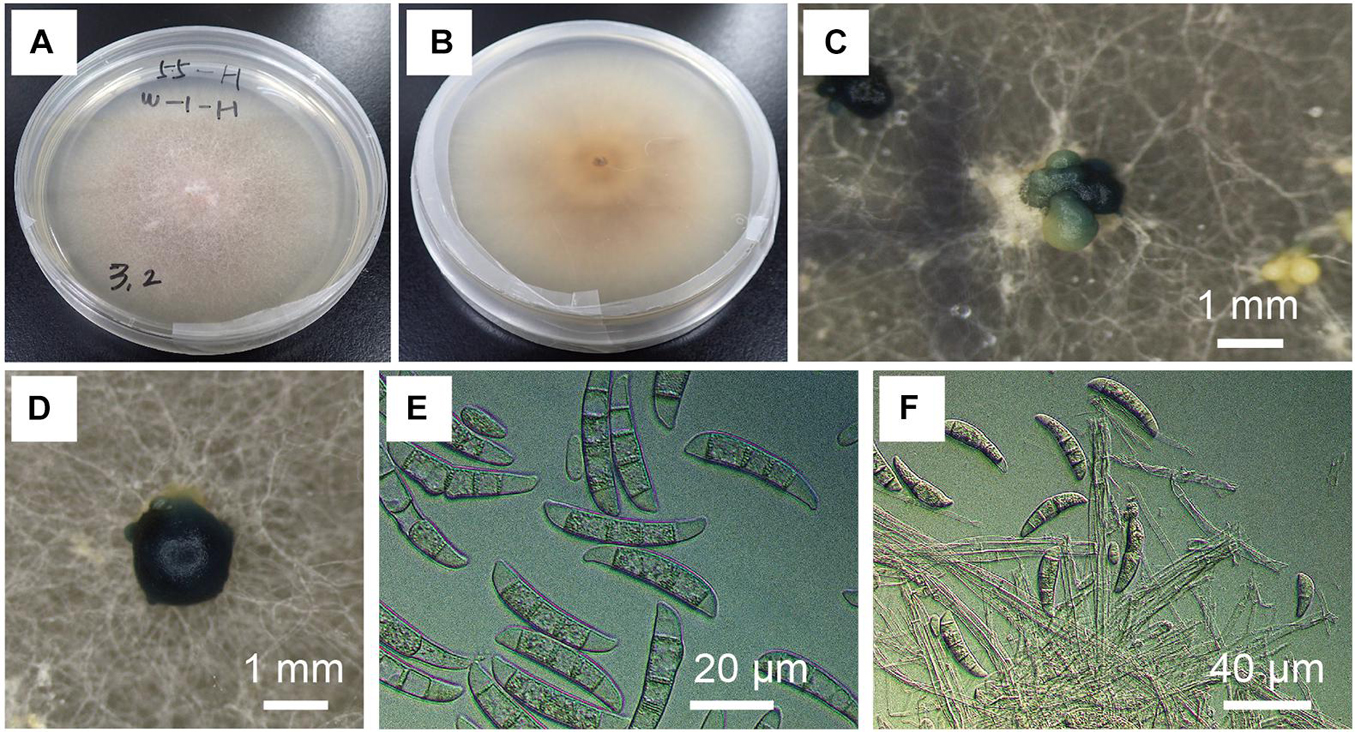
Figure 2. Morphological characteristics of Fusarium kuroshium. (A,B) Colony on potato dextrose agar (PDA) at 25°C 10 days after inoculation. (C,D) Greenish conidial masses formed on sporodochia in culture on PDA (25°C, dark, 2 months). (E) Conidial spores in the colony on PDA (25°C, dark, 2 months). (F) Conidial spores in the colony on synthetic low-nutrient agar (25°C, dark, 1 month).

Figure 3. Multilocus phylogenetic analysis of ambrosia Fusarium clade conducted with four genes: ribosomal internal transcribed spacer, elongation factor 1-α, DNA-directed RNA polymerase II largest subunit, and DNA-directed RNA polymerase II second largest subunit. The diagram was constructed using IQ-TREE maximum likelihood method bootstrapped with 1,000 replications. The red box indicates the phylogenetic placement of the Fusarium species associated with Euwallacea interjectus distributed in Hiroshima Prefecture, Western Japan.
Fusarium kuroshium is problematic in the taxonomy of ambrosia fusaria. It may contain cryptic species or hybrids because their placements were not congruent in the phylogenetic trees constructed by different methods. Species concept of F. kuroshium should be re-evaluated using a population genetics approach on ambrosia fusaria.
Relative Dominance and Frequency of Occurrence
The relative dominance varies from 3 to 88% (Table 2). The vast majority of isolates from the head was F. kuroshium with a RD of 88%. The dominance of F. kuroshium from the thorax and abdomen was 39 and 45%, respectively (Table 2). The frequency of occurrence of F. kuroshium isolated from the head was higher (59%) than that of other fungi and from other body parts (Table 2).
Discussion
In this study, “symbiotic” relationship is considered as a type of close and long-term biological interaction between ambrosia beetle and its fungal associates. The fungal associates should be stored in its mycangia before release into the galleries and thus significantly dominant among fungal isolates from the mycangia. Our findings showed a novel symbiotic relationship of E. interjectus–F. kuroshium in fig tree of Japan. To the best of our knowledge, this is the first time to identify this combination in the world. Fusarium kuroshium has been found to be closely associated with the oral mycangia (head) of female adults in E. interjectus population (Figure 1), suggesting a mycangial fungus possibly with pathogenicity for the fig tree.
This study provided a detailed survey of the different fungal communities associated with the female adults of an ambrosia beetle, E. interjectus, among different body parts. We identified fewer fungal species from the head as compared to the thorax and abdomen. Therefore, it could be inferred that the dominance of F. kuroshium in the head was higher than that in other parts (Table 2). The previous study showed that the mycangia of E. interjectus was located within their oral mycangia as observed by micro-CT scans (Jiang et al., 2019; Figure 1). This fact could provide an explanation for the higher presence of F. kuroshium in the head than the thorax and the abdomen because mycangia act as reservoirs to house the fungi during transport (Joseph and Keyhani, 2021).
The Euwallacea beetles–Fusarium fungi symbiosis has been found in many other countries and regions such as Israel (Mendel et al., 2012), United States (O’Donnell et al., 2015, 2016; Swain et al., 2017), Panama, Costa Rica (Kirkendall and Odegaard, 2007), Taiwan (Carrillo et al., 2019), and has been found to cause a severe disease to some tree species (Aoki et al., 2018, 2019; Table 1). The extensive native range of species in the Euwallacea spp. and the results of phylogenetic analyses showed that certain Euwallacea species recovered from Florida, United States, and Taiwan were associated with multiple Fusarium species from the AFC (O’Donnell et al., 2015; Carrillo et al., 2019). This further suggested that there was a substantial biological variation from native populations of Euwallacea spp. and their Fusarium spp. associates (Gomez et al., 2018). Some recent studies showed that Fusarium floridanum was farmed by E. interjectus in box elder (Acer negundo L.) in Florida, United States (Aoki et al., 2019). Fusarium kuroshium was found to be the most abundant fungal species from macerated heads of Euwallacea kuroshio (Kuroshio shot hole borer) which was recovered from the California sycamore (Platanus racemosa Nutt.) and avocado (Persea americana Mill.) in California, United States (Gomez et al., 2018; Na et al., 2018; Table 1). Our results indicated that E. interjectus has an association with an original species of the genus F. kuroshium in Japan and may switch to a new fungal symbiont to adapt to newly infested areas. Consistent with a recent study by Carrillo et al. (2019), we hypothesized that a strict relationship exists between the Euwallacea spp. and Fusarium spp. in the native areas of these organisms. This is in contrast to the promiscuous symbiosis observed in non-native areas (O’Donnell et al., 2015; Stouthamer et al., 2017). Given that several exotic pest insects have switched or gained fungal associates after they were introduced into non-indigenous areas (Carrillo et al., 2014, 2019, 2020), co-phylogenetic analyses were conducted to assess symbiont fidelity within the symbiosis (O’Donnell et al., 2015). Recent studies have also shown that Euwallacea fornicatus (Polyphagous Shot Hole Borer) and E. kuroshio can survive and reproduce on each other’s symbiotic fungi in their invasive range on artificial media (Carrillo et al., 2020). However, the ability of E. interjectus to switch its fungal symbiont is still an unresolved question.
Researchers have devoted over 40 years to resolve the dispersal process of FWD, and only one fungal species, C. ficicola, that causes wilt symptom has been discovered in fig orchards in Japan (Kato et al., 1982; Kajitani and Masuya, 2011; Kajii et al., 2013; Morita et al., 2016; Yakushiji et al., 2019). Our initial hypothesis was that C. ficicola could be isolated from the wild E. interjectus beetle, which emerged from fig trees with FWD. However, the results obtained after fungal isolation indicated another direction since unexpected fungi were collected rather than C. ficicola. These results suggested that C. ficicola is not infested via the oral mycangia of E. interjectus. Under certain conditions, the surface structure of elytra might accidentally trap fungal spores, which are transported by chance to fig trees. E. interjectus has numerous pits and setae on its exoskeleton, both of which appear suitable for transporting spores of fungus (Jiang et al., 2019). Our study also displayed that F. kuroshium, A. arundinis, and C. cladosporioides were common in all the three body parts (Table 2). It has been reported in few studies that F. kuroshium caused Fusarium dieback on woody host species in California, United States (Na et al., 2018). It has been found that A. arundinis was a fungal pathogen strain of Korean ginseng root rot in Korea (Durairaj et al., 2018), and C. cladosporioides was the causal agent of Cladosporium rot in grapes in Chile (Briceño and Latorre, 2008; Mengal et al., 2020). These findings indicated that the unexpected fungi, particularly F. kuroshium, possibly take part in FWD in Japan. Inoculation experiments of unexpected fungi on fig trees or saplings have to be conducted to answer this question.
Ambrosia beetles depend upon their fungal symbionts for development and reproduction (Saucedo et al., 2018). Here the E. interjectus–F. kuroshium symbiotic association seems to exhibit strict specificity based on the dominance of fungal isolates. Thus, F. kuroshium, as a potential food source of E. interjectus, can play a nutritional role (primary or auxiliary) in this symbiotic system. If so, E. interjectus probably avoids the colonization of a branch of stem sections of the fig which are already occupied by the pathogen. Further extensive studies are needed to obtain the evidence which could support the actual function and specificity of this novel symbiotic association.
Data Availability Statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
Author Contributions
HK and Z-RJ conceived the study. Z-RJ collected the beetle samples, isolated all examined fungi, analyzed the data, and wrote an early version of the manuscript. HM sequenced all the examined fungi and constructed the phylogenetic tree. HK supervised the study and reviewed the manuscript. All authors contributed to the study and approved the submitted version.
Funding
This work was supported by the Japan Society for the Promotion of Science KAKENHI Grant (Grants-in-Aid for Scientific Research numbers 17H03831, 18KK0180, 19H02994, and 20H03026).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We thank Takeshige Morita and Shota Jikumaru of the Agricultural Technology Research Center of Hiroshima Prefectural Technology Research Institute, who helped with the sample collection of the ambrosia beetle-attacking trees in the fig orchard. We also thank Naoki Hijii, Wataru Toki, and other members of the Forest Protection Lab of Nagoya University for their suggestions to improve our work.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2021.725210/full#supplementary-material
References
Aoki, T., Kasson, M. T., Berger, M. C., Freeman, S., Geiser, D. M., and O’Donnell, K. (2018). Fusarium oligoseptatum sp. nov., a mycosymbiont of the ambrosia beetle Euwallacea validus in the Eastern US and typification of F. ambrosium. Fungal Syst. Evol. 1, 23–39. doi: 10.3114/fuse.2018.01.03
Aoki, T., Smith, J. A., Kasson, M. T., Freeman, S., Geiser, D. M., Geering, A. D., et al. (2019). Three novel Ambrosia Fusarium Clade species producing clavate macroconidia known (F. floridanum and F. obliquiseptatum) or predicted (F. tuaranense) to be farmed by Euwallacea spp. (Coleoptera: Scolytinae) on woody hosts. Mycologia 111, 919–935. doi: 10.1080/00275514.2019.1647074
Batra, L. R. (1963). Ecology of ambrosia fungi and their dissemination by beetles. Trans. Kans. Acad. Sci. 66, 213–236. doi: 10.2307/3626562
Bentz, B. J., and Six, D. L. (2006). Ergosterol content of fungi associated with Dendroctonus ponderosae and Dendroctonus rufipennis (Coleoptera: Curculionidae, Scolytinae). Ann. Entomol. Soc. Am. 99, 189–194. doi: 10.1603/0013-8746(2006)099[0189:ECOFAW]2.0.CO;2
Brayford, D. (1987). Fusarium bugnicourtii sp. nov., and its relationship to F. tumidum and F. tumidum var. coeruleum. Trans. Br. Mycol. Soc. 89, 347–351.
Briceño, E. X., and Latorre, B. A. (2008). Characterization of Cladosporium rot in grapevines, a problem of growing importance in Chile. Plant Dis. 92, 1635–1642. doi: 10.1094/PDIS-92-12-1635
Carrillo, D., Duncan, R. E., Ploetz, J. N., Campbell, A. F., Ploetz, R. C., and Peña, J. E. (2014). Lateral transfer of a phytopathogenic symbiont among native and exotic ambrosia beetles. Plant Pathol. 63, 54–62. doi: 10.1111/ppa.12073
Carrillo, J. D., Dodge, C., Stouthamer, R., and Eskalen, A. (2020). Fungal symbionts of the polyphagous and Kuroshio shot hole borers (Coleoptera: Scolytinae, Euwallacea spp.) in California can support both ambrosia beetle systems on artificial media. Symbiosis 80, 155–168. doi: 10.1007/s13199-019-00652-0
Carrillo, J. D., Rugman-Jones, P. F., Husein, D., Stajich, J. E., Kasson, M. T., Carrillo, D., et al. (2019). Members of the Euwallacea fornicatus species complex exhibit promiscuous mutualism with ambrosia fungi in Taiwan. Fungal Genet. Biol. 133:103269. doi: 10.1016/j.fgb.2019.103269
Castro, J., Smith, S. M., Cognato, A. I., Lanfranco, D., Martinez, M., and Guachambala, M. (2019). Life cycle and development of Coptoborus ochromactonus (Coleoptera: Curculionidae: Scolytinae), a pest of balsa. J. Econ. Entomol. 112, 729–735. doi: 10.1093/jee/toy403
Cognato, A. I., Hoebeke, E. R., Kajimura, H., and Smith, S. M. (2015). History of the exotic ambrosia beetles Euwallacea interjectus and Euwallacea validus (Coleoptera: Curculionidae: Xyleborini) in the United States. J. Econ. Entomol. 108, 1129–1135. doi: 10.1093/jee/tov073
Cognato, A. I., Sari, G., Smith, S. M., Beaver, R. A., Li, Y., Hulcr, J., et al. (2020). The essential role of taxonomic expertise in the creation of DNA databases for the identification and delimitation of Southeast Asian ambrosia beetle species (Curculionidae: Scolytinae: Xyleborini). Front. Ecol. Evol. 8:27. doi: 10.3389/fevo.2020.00027
Cooperband, M. F., Stouthamer, R., Carillo, D., Eskalen, A., Thibault, T., Cossé, A. A., et al. (2016). Biology of two members of the Euwallacea fornicatus species complex (Coleoptera: Curculionidae: Scolytinae), recently invasive in the U.S.A., reared on an ambrosia beetle artificial diet. Agric. For. Entomol. 18, 223–237. doi: 10.1111/afe.12155
Durairaj, K., Velmurugan, P., Vedhanayakisri, A. K., Chang, W. S., Senthilkumar, P., Choi, K. M., et al. (2018). Molecular and phenotypic characterization of pathogenic fungal strains isolated from ginseng root rot. Physiol. Mol. Plant Pathol. 104, 141–146. doi: 10.1016/j.pmpp.2018.09.006
Dzurenko, M., Ranger, C. M., Hulcr, J., Galko, J., and Kaòuch, P. (2021). Origin of non-native Xylosandrus germanus, an invasive pest ambrosia beetle in Europe and North America. J. Pest Sci. 94, 553–562. doi: 10.1007/s10340-020-01283-x
Edgar, R. C. (2004). MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797. doi: 10.1093/nar/gkh340
Eskalen, A., Stouthamer, R., Lynch, S. C., Rugman-Jones, P. F., Twizeyimana, M., Gonzalez, A., et al. (2013). Host range of Fusarium dieback and its ambrosia beetle (Coleoptera: Scolytinae) vector in southern California. Plant Dis. 97, 938–951. doi: 10.1094/PDIS-11-12-1026-RE
Freeman, S., Sharon, M., Maymon, M., Mendel, Z., Protasov, A., Aoki, T., et al. (2013). Fusarium euwallaceae sp. nov. – a symbiotic fungus of Euwallacea sp., an invasive ambrosia beetle in Israel and California. Mycologia 105, 1595–1606. doi: 10.3852/13-066
Gadd, C. H., and Loos, C. A. (1947). The ambrosia fungus of Xyleborus fornicatus Eich. Br. Mycol. Soc. 31, 1–2. doi: 10.1016/S0007-1536(47)80003-8
García-Avila, C. D. J., Trujillo-Arriaga, F. J., López-Buenfil, J. A., González-Gómez, R., Carrillo, D., Cruz, L. F., et al. (2016). First report of Euwallacea nr. fornicatus (Coleoptera: Curculionidae) in Mexico. Fla. Entomol. 99, 555–556. doi: 10.1653/024.099.0335
Gomez, D. F., Skelton, J., Steininger, M. S., Stouthamer, R., Rugman-Jones, P., Sittichaya, W., et al. (2018). Species delineation within the Euwallacea fornicatus (Coleoptera: Curculionidae) complex revealed by morphometric and phylogenetic analyses. Insect Syst. Evol. 2, 1–11. doi: 10.1093/isd/ixy018
Hoang, D. T., Chernomor, O., Von Haeseler, A., Minh, B. Q., and Vinh, S. L. (2018). UFBoot2: improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 35, 518–522. doi: 10.1093/molbev/msx281
Huang, X., and Madan, A. (1999). CAP3: a DNA sequence assembly program. Genome Res. 9, 868–877. doi: 10.1101/gr.9.9.868
Hulcr, J., and Cognato, A. I. (2010). Repeated evolution of crop theft in fungus-farming ambrosia beetles. Evolution 64, 3205–3212. doi: 10.1111/j.1558-5646.2010.01055.x
Hulcr, J., and Dunn, R. R. (2011). The sudden emergence of pathogenicity in insect-fungus symbioses threatens naive forest ecosystems. Proc. R. Soc. B 278, 2866–2873. doi: 10.1098/rspb.2011.1130
Hulcr, J., and Stelinski, L. L. (2017). The ambrosia symbiosis: from evolutionary ecology to practical management. Annu. Rev. Entomol. 62, 285–303. doi: 10.1146/annurev-ento-031616-035105
Hulcr, J., Black, A., Prior, K., Chen, C. Y., and Li, H. F. (2017). Studies of ambrosia beetles (Coleoptera: Curculionidae) in their native ranges help predict invasion impact. Fla. Entomol. 100, 257–261. doi: 10.1653/024.100.0219
Jiang, Z.-R., Kinoshita, S. I., Sasaki, O., Cognato, A. I., and Kajimura, H. (2019). Non-destructive observation of the mycangia of Euwallacea interjectus (Blandford) (Coleoptera: Curculionidae: Scolytinae) using X-ray computed tomography. Entomol. Sci. 22, 173–181. doi: 10.1111/ens.12353
Joseph, R., and Keyhani, N. O. (2021). Fungal mutualisms and pathosystems: life and death in the ambrosia beetle mycangia. Appl. Microbiol. Biotechnol. 105, 3393–3410. doi: 10.1007/s00253-021-11268-0
Kajii, C., Morita, T., Jikumaru, S., Kajimura, H., Yamaoka, Y., and Kuroda, K. (2013). Xylem dysfunction in Ficus carica infected with wilt fungus Ceratocystis ficicola and the role of the vector beetle Euwallacea interjectus. IAWA. 34, 301–312. doi: 10.1163/22941932-00000025
Kajitani, Y. (1996). The possibility of transmission of fig Ceratocystis canker disease by an ambrosia beetle (Xyleborus interjectus Eichhoff). Ann. Phytopathol. Soc. Jpn. 62:275. [In Japanese]
Kajitani, Y. (1999). The dispersal period of the Xyleborus interjectus (Coleoptera, Scolytidae), a vector of the fig Ceratocystis canker, and the organ carrying the causal fungus. Ann. Phytopathol. Soc. Jpn. 65:377. [In Japanese]
Kajitani, Y., and Masuya, H. (2011). Ceratocystis ficicola sp. nov., a causal fungus of fig canker in Japan. Mycoscience 52, 349–353. doi: 10.1007/S10267-011-0116-5
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K. F., Von Haeseler, A., and Jermiin, L. S. (2017). ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Methods. 14, 587–589. doi: 10.1038/nmeth.4285
Kasson, M. T., O’Donnell, K., Rooney, A. P., Sink, S., Ploetz, R. C., Ploetz, J. N., et al. (2013). An inordinate fondness for Fusarium: phylogenetic diversity of fusaria cultivated by ambrosia beetles in the genus Euwallacea on avocado and other plant hosts. Fungal Genet. Biol. 56, 147–157. doi: 10.1016/j.fgb.2013.04.004
Kato, K., Yokota, K., and Miyagawa, T. (1982). A new disease, Ceratocystis canker of fig caused by Ceratocystis fimbriata Ellis et Halsted. Plant Prot. 36, 55–59. [In Japanese]
Kirkendall, L. R. (1993). “Ecology and evolution of biased sex ratios in bark and ambrosia beetles,” in Evolution and Diversity of Sex Ratios in Insects and Mites, eds D. L. Wrensch and M. A. Ebbert (New York, NY: Chapman and Hall), 235–345.
Kirkendall, L. R., and Odegaard, F. (2007). Ongoing invasions of old-growth tropical forests: establishment of three incestuous beetle species in southern Central America (Curculionidae: Scolytinae). Zootaxa 1588, 53–62. doi: 10.11646/zootaxa.1588.1.3
Kuroda, K., Chuma, I., Kihara, T., Murakami, T., Takashina, K., Hiraoka, D., et al. (2017). First report of Fusarium solani species complex as a causal agent of Erythrina variegata decline and death after gall formation by Quadrastichus erythrinae on Okinawa Island, Japan. J. Gen. Plant Pathol. 83, 344–357. doi: 10.1007/s10327-017-0738-3
Landi, L., Braccini, C. L., Knížek, M., Pereyra, V. A., and Marvaldi, A. E. (2019). A newly detected exotic ambrosia beetle in Argentina: Euwallacea interjectus (Coleoptera: Curculionidae: Scolytinae). Fla. Entomol. 102, 240–242. doi: 10.1653/024.102.0141
Larsson, A. (2014). AliView: a fast and lightweight alignment viewer and editor for large data sets. Bioinformatics 30, 3276–3278. doi: 10.1093/bioinformatics/btu531
Li, Y., Ruan, Y., Kasson, M. T., Stanley, E. L., Gillett, C. P. D. T., Johnson, A. J., et al. (2018). Structure of the ambrosia beetle (Coleoptera: Curculionidae) mycangia revealed through micro-computed tomography. J. Insect Sci. 18:5. doi: 10.1093/jisesa/iey096
Li, Y., Simmons, D. R., Bateman, C. C., Short, D. P. G., Kasson, M. T., Rabaglia, R. J., et al. (2015). New fungus-insect symbiosis: culturing, molecular, and histological methods determine saprophytic Polyporales mutualists of Ambrosiodmus ambrosia beetles. PloS One 10:e0137689. doi: 10.1371/journal.pone.0137689
Lynn, K. M. T., Wingfield, M. J., Durán, A., Marincowitz, S., Oliveira, L. S., De Beer, Z. W., et al. (2020). Euwallacea perbrevis (Coleoptera: Curculionidae: Scolytinae), a confirmed pest on Acacia crassicarpa in Riau, Indonesia, and a new fungal symbiont; Fusarium rekanum sp. nov. Antonie van Leeuwenhoek 113, 803–823. doi: 10.1007/s10482-020-01392-8
Lynn, K. M. T., Wingfield, M. J., Durán, A., Oliveira, L. S., De Beer, Z. W., and Barnes, I. (2021). Novel Fusarium mutualists of two Euwallacea species infesting Acacia crassicarpa in Indonesia. Mycologia 113, 536–558. doi: 10.1080/00275514.2021.1875708
Mayers, C. G., Harrington, T. C., and Ranger, C. M. (2017). First report of a sexual state in an ambrosia fungus: Ambrosiella cleistominuta sp. nov. associated with the ambrosia beetle Anisandrus maiche. Botany 95, 503–512. doi: 10.1139/cjb-2016-0297
Mendel, Z., Protasov, A., Sharon, M., Zveibil, A., Ben Yehuda, S., O’Donnell, K., et al. (2012). An Asian ambrosia beetle Euwallacea fornicatus and its novel symbiotic fungus Fusarium sp. pose a serious threat to the Israeli avocado industry. Phytoparasitica 40, 235–238. doi: 10.1007/s12600-012-0223-7
Mengal, H. S., Abro, M. A., Jatoi, G. H., Nawab, L., Poussio, G. B., Ahmed, N., et al. (2020). Efficacy of different fungicides, botanical extracts and bio-control agents against Cladosporium cladosporioides, the causal agent of Cladosporium rot in grapes. Acta Ecol. Sin. 40, 300–305. doi: 10.1016/j.chnaes.2019.08.002
Morita, T., Hara, H., Mise, D., and Jikumaru, S. (2012). A case study of Ceratocystis canker epidemic in relation with Euwallacea interjectus infestation. Ann. Rept. Kansai Pl. Prot. 54, 29–34. [In Japanese with English abstract]
Morita, T., Jikumaru, S., and Kuroda, K. (2016). Disease development in Ficus carica plants after inoculation with Ceratocystis ficicola. (1) Relation between xylem dysfunction and wilt symptoms. Jpn. J. Phytopathol. 82, 301–309. [In Japanese with English abstract]. doi: 10.3186/jjphytopath.82.301
Mueller, U. G., Gerardo, N. M., Aanen, D. K., Six, D. L., and Schultz, T. R. (2005). The evolution of agriculture in insects. Annu. Rev. Ecol. Evol. Syst. 36, 563–595. doi: 10.1146/annurev.ecolsys.36.102003.152626
Na, F., Carrillo, J. D., Mayorquin, J. S., Ndinga-Muniania, C., Stajich, J. E., Stouthamer, R., et al. (2018). Two novel fungal symbionts Fusarium kuroshium sp. nov. and Graphium kuroshium sp. nov. of Kuroshio shot hole borer (Euwallacea sp. nr. fornicatus) cause Fusarium dieback on woody host species in California. Plant Dis. 102, 1154–1164. doi: 10.1094/PDIS-07-17-1042-RE
Nguyen, L. T., Schmidt, H. A., Von Haeseler, A., and Minh, B. Q. (2015). IQ-TREE: a fast and effective stochastic algorithm for estimating maximum likelihood phylogenies. Mol. Biol. Evol. 32, 268–274. doi: 10.1093/molbev/msu300
O’Donnell, K., Kistler, H. C., Cigelnik, E., and Ploetz, R. C. (1998). Multiple evolutionary origins of the fungus causing Panama disease of banana: concordant evidence from nuclear and mitochondrial gene genealogies. Proc. Natl. Acad. Sci. U.S.A. 95, 2044–2049. doi: 10.1073/pnas.95.5.2044
O’Donnell, K., Libeskind-Hadas, R., Hulcr, J., Bateman, C., Kasson, M. T., Ploetz, R. C., et al. (2016). Invasive Asian Fusarium–Euwallacea ambrosia beetle mutualists pose a serious threat to forests, urban landscapes and the avocado industry. Phytoparasitica 44, 435–442. doi: 10.1007/s12600-016-0543-0
O’Donnell, K., Sink, S., Libeskind-Hadas, R., Hulcr, J., Kasson, M. T., Ploetz, R. C., et al. (2015). Discordant phylogenies suggest repeated host shifts in the Fusarium–Euwallacea ambrosia beetle mutualism. Fungal Genet. Biol. 82, 277–290. doi: 10.1016/j.fgb.2014.10.014
Paap, T., De Beer, Z. W., Migliorini, D., Nel, W. J., and Wingfield, M. J. (2018). The polyphagous shot hole borer (PSHB) and its fungal symbiont Fusarium euwallaceae: a new invasion in South Africa. Australas. Plant Pathol. 47, 231–237. doi: 10.1007/s13313-018-0545-0
Ranger, C. M., Schultz, P. B., Frank, S. D., Chong, J. H., and Reding, M. E. (2015). Non-native ambrosia beetles as opportunistic exploiters of living but weakened trees. PLoS One 10:e0131496. doi: 10.1371/journal.pone.0131496
Samuelson, G. A. (1981). A synopsis of Hawaiian Xyleborini (Coleoptera: Scolytidae). Pac. Insects 23, 20–92.
Sandoval-Denis, M., and Crous, P. W. (2018). Removing chaos from confusion: assigning names to common human and animal pathogens in Neocosmospora. Persoonia 41, 109–129. doi: 10.3767/persoonia.2018.41.06
Sandoval-Denis, M., Lombard, L., and Crous, P. W. (2019). Back to the roots: a reappraisal of Neocosmospora. Persoonia 43, 90–185. doi: 10.3767/persoonia.2019.43.04
Saucedo, J. R., Ploetz, R. C., Konkol, J. L., Ángel, M., Mantilla, J., Menocal, O., et al. (2018). Nutritional symbionts of a putative vector, Xyleborus bispinatus, of the laurel wilt pathogen of avocado, Raffaelea lauricola. Symbiosis 75, 29–38. doi: 10.1007/s13199-017-0514-3
Skelton, J., Jusino, M. A., Carlson, P. S., Smith, K., Banik, M. T., Lindner, D. L., et al. (2019). Relationships among wood-boring beetles, fungi, and the decomposition of forest biomass. Mol. Ecol. 28, 4971–4986. doi: 10.1111/mec.15263
Smith, S. M., Gomez, D. F., Beaver, R. A., Hulcr, J., and Cognato, A. I. (2019). Reassessment of the species in the Euwallacea fornicatus (Coleoptera: Curculionidae: Scolytinae) complex after the rediscovery of the “lost” type specimen. Insects 10:9. doi: 10.3390/insects10090261
Stouthamer, R., Rugman-Jones, P., Thu, P. Q., Eskalen, A., Thibault, T., Hulcr, J., et al. (2017). Tracing the origin of a cryptic invader: phylogeography of the Euwallacea fornicatus (Coleoptera: Curculionidae: Scolytinae) species complex. Agric. For. Entomol. 19, 366–375. doi: 10.1111/afe.12215
Swain, S., Eskalen, A., Lynch, S., and Latham, S. (2017). Scolytid beetles and associated fungal symbionts threaten California hardwoods. Western Arborist. 43, 54–60.
Vega, F. E., and Biedermann, P. H. W. (2020). On interactions, associations, mycetangia, mutualists and symbiotes in insect-fungus symbioses. Fungal Ecol. 44:100909. doi: 10.1016/j.funeco.2019.100909
Veloso, J. S., Câmara, M. P., and Souza, R. M. (2021). Guava decline: updating its etiology from ‘Fusarium solani’ to Neocosmospora falciformis. Eur. J. Plant Pathol. 159, 455–460. doi: 10.1007/s10658-020-02161-z
Wang, Z., Li, Y., Ernstsons, A. S., Sun, R., Hulcr, J., and Gao, L. (2020). The infestation and habitat of the ambrosia beetle Euwallacea interjectus (Coleoptera: Curculionidae: Scolytinae) in the riparian zone of Shanghai, China. Agric. For. Entomol. 23, 104–109. doi: 10.1111/afe.12405
White, T. J., Bruns, T., Lee, S., and Taylor, J. W. (1990). “Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics,” in PCR Protocols: A Guide to Methods and Applications, eds M. A. Innis, D. H. Gelfand, J. J. Sninsky, and T. J. White (New York, NY: Academic Press), 315–322.
Keywords: ambrosia Fusarium clade, Euwallacea interjectus, fig wilt disease, Fusarium kuroshium, Fusarium solani species complex, multi-gene phylogeny, mycangia
Citation: Jiang Z-R, Masuya H and Kajimura H (2021) Novel Symbiotic Association Between Euwallacea Ambrosia Beetle and Fusarium Fungus on Fig Trees in Japan. Front. Microbiol. 12:725210. doi: 10.3389/fmicb.2021.725210
Received: 15 June 2021; Accepted: 17 August 2021;
Published: 28 September 2021.
Edited by:
Hassan Salem, Max Planck Institute for Developmental Biology, Max Planck Society (MPG), GermanyReviewed by:
Zvi Mendel, Agricultural Research Organization (ARO), IsraelKin-Ming (Clement) Tsui, Weill Cornell Medicine - Qatar, Qatar
Copyright © 2021 Jiang, Masuya and Kajimura. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hisashi Kajimura, a2FqaW11cmFAYWdyLm5hZ295YS11LmFjLmpw
 Zi-Ru Jiang
Zi-Ru Jiang Hayato Masuya2
Hayato Masuya2 Hisashi Kajimura
Hisashi Kajimura