
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Microbiol. , 02 March 2021
Sec. Terrestrial Microbiology
Volume 12 - 2021 | https://doi.org/10.3389/fmicb.2021.570908
CO2 fixation by autotrophic microbes has a significant effect on the carbon cycle in temperate grasslands. Nitrogen (N) deposition in soil has been steadily increasing for decades, which has consequences for soil microorganisms. However, the impact of this deposition on the diversity and abundance of CO2-fixing soil microorganisms remains unclear in temperate grasslands. In the present study, the cbbL gene, a key gene in the Calvin–Benson–Bassham cycle that encodes the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase, was used to study CO2-fixing microbes under different rates of N addition (0, 15, 30, 50, 100, and 150 kg N ha–1 yr–1) in a 9-year field experiment in a temperate grassland. The results showed that N addition led to significant reductions in cbbL gene abundance and genetic diversity and altered cbbL gene community composition. High N addition enhanced the relative abundances of Acidiferrobacterales and Rhizobiales but reduced those of Burkholderiales and Rhodobacterales. Structural equation modeling further revealed that N addition primarily reduced cbbL genetic diversity by increasing the soil NO3-N content and decreasing the soil pH. N addition indirectly reduced cbbL gene abundance, possibly by increasing the soil N/phosphorus (P) ratio and decreasing the soil pH. These findings suggest that N addition increases the soil available N and causes soil acidification, which may inhibit growth of CO2-fixing microbes to some extent.
During the past few decades, the amount of nitrogen (N) deposited from the global atmosphere due to human activities has significantly increased, and developing countries experiencing the most rapid increases (Gruber and Galloway, 2008). Researchers predict that global N deposition will reach 195 Tg N yr–1 by 2050 (Galloway et al., 2004). At present, concerns regarding global atmospheric N deposition are primarily focused on Western Europe, North America, and East Asia (mainly China) (Mo et al., 2008). From 1961 to 2008, the proportion of N deposition increased by 59% in China (Lu and Tian, 2014) and reached 10–18 kg N ha–1 in the northern grasslands of the country (Zhang et al., 2008; Liu et al., 2011). The rate of atmospheric N deposition is increasing annually and is expected to accelerate in the future (Neff et al., 2002). N deposition changes the physicochemical properties of soil, including with respect to soil acidification and the N–P balance (Hong et al., 2019), altering soil microbial diversity (Wang C. et al., 2018), and affecting the global ecosystem (Penuelas et al., 2013).
The terrestrial biosphere currently absorbs approximately 30% of anthropogenic CO2 emissions (Arora and Melton, 2018). In the past, it was generally believed that carbon fixation was primarily dependent on plant photosynthesis and that soil microorganisms contributed to the carbon cycle by participating in the degradation of organic matter rather than by CO2 fixation. The role of microorganisms in carbon sequestration may also be underestimated (Hart et al., 2013). Microbial phototrophic CO2 fixation accounts for a substantial proportion of global primary productivity (Guzman et al., 2019). However, the contributions of microbial biomass to soil organic matter appear to be much higher than the 1–5% reported by other researchers (Simpson et al., 2007). Lipids, carbohydrates, and proteins have been observed to be produced directly from the CO2 taken up by microorganisms (Hart et al., 2013). CO2-fixing microorganisms are a group of microorganisms that, like plants, transform atmospheric CO2 into organic matter. Soil autotrophic bacteria are important for sequestrating atmospheric CO2 (Lynn et al., 2017) and affect the renewal and circulation of organic matter. The CO2-fixing capacity of soil microbes has received widespread attention. However, the factors affecting CO2-fixing microbes are not well known.
The autotrophic microbial community was previously characterized by targeting the large subunit (encoded by the cbbL gene) of form I ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) (Zhao et al., 2018). The cbbL gene is an established biomarker that is useful for studying autotrophic bacteria in various soil ecosystems owing to its widespread geographic distribution, functional significance, the increasing number of published sequences of this gene from chemoautotrophs, and its utility in assessing autotrophic microbial diversity in various habitats (Ge et al., 2016; Yue et al., 2019).
The cbbL-containing bacteria has gene have been linked to soil available N (Zhou et al., 2019), soil pH (Li M. Q. et al., 2020), and soil organic carbon (SOC) (Xiao et al., 2014; Li et al., 2018). First, increased soil available N may affect cbbL-containing bacterial abundance by altering the balance of C/N or N/P ratios (Dong et al., 2019). Second, variations in soil pH are likely to regulate the responses of cbbL containing bacteria to environmental changes (Zhao et al., 2018; Li P. P. et al., 2020). Finally, alterations in SOC levels can lead to changes in the abundances of cbbL-containing bacteria, particularly upon significant changes in SOC levels (Huang et al., 2018). The relative contribution of these drivers may vary between ecosystems. N deposition is expected to have positive or negative effects on cbbL gene-containing CO2-fixing microorganisms, which may depend on soil available nutrients and the intensity of N addition, potentially leading to changes in the composition of the cbbL gene community. In addition, N deposition can enhance the soil N content and change the soil pH, which in turn may influence the cbbL gene-containing CO2-fixing microorganisms (Zhou et al., 2019).
Arid and semiarid ecosystems account for approximately 41% of the global land area (Ferrenberg et al., 2015). The vegetative growth of these ecosystems is restricted by many environmental factors, including soil moisture, highlighting the importance of soil CO2-fixing microbes (Guo et al., 2015). The Stipa baicalensis steppe represents one of the most widely distributed temperate grassland communities in Eurasia and is primarily located on the eastern Mongolian Plateau and in most of the Songliao Plain of China. Although CO2-fixing microorganisms make important contributions to ecosystem functions and processes (Zhao et al., 2018), we are only beginning to understand how their communities are shaped by N deposition. In the present study, we conducted field-simulated N deposition experiments to assess the response of soil CO2-fixing microbes to different levels of N addition in temperate grassland to address the following two questions: (1) how do the abundance and diversity of cbbL-containing bacteria respond to N addition, and (2) what soil environmental factors mediate the response of cbbL-containing bacteria to N addition? We hypothesized that N addition affects cbbL-containing bacteria by altering soil physicochemical factors (Xiao et al., 2014; Zhou et al., 2019). The results of the present study provide a theoretical basis for the investigation of soil microbial carbon sequestration potential and soil carbon cycles in temperate grasslands. Research on this topic is important for predicting the possible changes in soil carbon sequestration in grassland ecosystems under changes in N deposition.
The field survey was conducted on the Stipa baicalensis steppe, which is located in the Hulun Buir grassland (48°30′N, 119°42′E; 765 m) of the Inner Mongolia Autonomous Region, China (Qin et al., 2020). The experimental area has a typical temperate continental monsoon climate with warm summers, cold winters, an annual precipitation of 396 mm and an annual mean temperature of –0.7°C. Most precipitation (66%) in this region occurs in the summer months, and the soil type is primarily a Haplic Calcisol (according to the Food and Agriculture Organization classification). The native vegetation in the study area consists of grasses dominated by species such as Stipa baicalensis and Leymus chinensis. Common species include Cleistogenes squarrosa, Carex pediformis, Filifolium sibiricum, Achnatherum sibiricum, Thalictrum petaloideum, Serratula centauroides, Melissitus ruthenica, and Carex duriuscula.
The experimental simulation of N deposition began in 2010, and a randomized block design with four replicates was adopted. Six experimental treatments were performed to simulate current and future N deposition levels such that 24 plots (each 8 m by 8 m) were established. The natural level of N deposition in the Inner Mongolian grassland is approximately 18.1 kg Nha–1 yr–1 (Zhang et al., 2008). N was added to the six experimental treatments (Stevens et al., 2004; Liu et al., 2011; Lu and Tian, 2014) at 0, 15, 30, 50, 100, and 150 kg N ha–1 yr–1 (designated as N0, N15, N30, N50, N100, and N150, respectively). N was added twice a year (mid-June and mid-July) by spraying plots with an aqueous solution of NH4NO3.
Soil sampling was conducted on 10 August 2018 in each of the 24 plots. Before sampling, the litter layer (litter, roots, and stones) was carefully removed. Soil samples were collected from 10 random points across each plot using a soil corer (0–15 cm deep with a 2 cm inner diameter) and mixed to obtain one representative composite sample. The soil samples were placed in self-sealing bags for storage at 4°C. The samples were immediately transported to the laboratory and divided into three subsamples. Subsamples for ammonium N (NH4-N) and nitrate N (NO3-N) concentration analyses were stored at 4°C for no longer than 1 week, and the subsamples used for soil pH, SOC, and total P (TP) analyses were air dried. The soil physicochemical factors differed under different levels of N addition (Supplementary Table 1). The subsamples used for gene abundance and high-throughput sequencing analyses were stored at –80°C.
Soil pH was measured using a soil to water ratio of 1:2.5 using a Delta 320 pH meter (Mettler Toledo Instruments, Shanghai, China). The SOC content was determined with a macro elemental analyzer (Vario MAX C/N; Elementar Analysensysteme, Hanau, Germany). The total N content was determined by Kjeldahl digestion, and the total P concentration was measured using the ammonium molybdate method following H2SO4-H2O2-HF digestion. The soil available N (the sum of NH4-N and NO3-N) was measured with an FIA Star 5000 flow-injection autoanalyzer (Foss Tecator, Höganäs, Sweden).
Genomic DNA was extracted from each soil sample using a PowerSoil DNA Isolation kit (MoBio Laboratories, Carlsbad, CA, United States) according to the manufacturer’s protocol. The integrity and yield of the genomic DNA were assessed by 0.8% agarose gel electrophoresis.
The copy number of the target gene in the DNA samples was determined using the absolute quantitative method. The abundance of the cbbL gene was determined by real-time PCR (Applied Biosystems 7900, United States) with the primers K2f (5′-ACCAYCAAGCCSAAGCTSGG-3′) and V2r (5′-GCCTTCSAGCTTG CCSACCRC-3′). Each DNA sample was diluted 10 times, and 2 μl of the diluted DNA (approximately 150 ng of DNA) was then taken as the reaction volume. The 18 μl reaction mixture contained 10 μl of 2 × Taq MasterMix (Takara Bio Inc., Shiga, Japan), 0.5 μl each of the specific forward and reverse PCR primers (Invitrogen, Shanghai, China), and 7 μl of H2O. The cycling parameters involved predenaturation at 95°C for 5 min, followed by 30 cycles of 94°C for 30 s, 55°C for 30 s, and 72°C for 30 s, which was followed by a final incubation at 72°C for 10 min. The assays were performed using three technical replicates per sample. A 10-fold dilution series (101105) of plasmid DNA harboring the cbbL gene was used to generate a PCR standard curve. At the end of the PCR amplification, the melting curve was analyzed, and a single melting curve peak was observed for each sample. The cbbL copies were calculated according to the parameter threshold cycle (Ct) obtained using the 7500 software (version 1.0.6).
Using TruSeq v1/v2 kits (Illumina, San Diego, CA, United States), adaptors A and B, the former of which harbored an 8-nucleotide barcode sequence, were added to the forward and reverse primer sequences, respectively. DNA was detected by 1% agarose gel electrophoresis after genomic DNA extraction. Specific barcoded primers or fusion primers with misplaced bases were synthesized according to the specified sequencing region. The same primer set (K2f [5′-ACCAYCAAGCCSAAGCTSGG-3′] and V2r [5′-GCCTTCSAGCTTG CCSACCRC-3′]) and a thermal profile for real-time PCR were used for cbbL gene amplification with Illumina MiSeq sequencing. The PCR products of the same sample were mixed and detected by 2% agarose gel electrophoresis. Then, the PCR products were recovered via gel extraction using an AxyPrep DNA gel recovery kit (Axygen Inc., Union City, CA, United States) and a Tris-HCl elution and were detected by 2% agarose electrophoresis. All the PCR steps were performed with a Mastercycler Gradient (Eppendorf, Hamburg, Germany). The resulting purified amplicons were pooled in equimolar concentrations and paired-end sequenced on an Illumina MiSeq PE300 platform (Illumina, San Diego, CA, United States) by Allwegene Technology Co., Ltd. (Beijing, China). The raw sequence data were submitted to the National Center for Biotechnology Information Sequence Reads Archive under accession No. PRJNA633225.
The cbbL gene sequences were checked for close relatives to known cbbL sequences in GenBank (the National Center for Biotechnology Information database) using the BLAST program1. Trimmomatic (version 0.36) and PEAR (version 0.96) were used to manipulate the FASTQ data. The sliding-window strategy was adopted using a window size set to 50 bp, an average mass value of 20, and a minimum reserved sequence length of 120. FLASH (version 1.20) and PEAR were used to merge the two end sequences according to the overlap relationship of the PE. The minimum overlap was set to 10 bp, and the mismatch rate was 0.1 to obtain the FASTA sequence. The raw data were screened, and sequences were removed from consideration if they were shorter than 200 bp, had a low-quality score (≤20), contained ambiguous bases, or did not exactly match primer sequences and barcode tags. Clean tags were clustered (or denoised) to generate operational taxonomic units (OTUs) using the UPARSE method (version 9.2) (Edgar, 2013) and the UNOISE method (Rognes et al., 2016). The sequences were clustered into OTUs at a similarity level of 97% (Edgar, 2013), resulting in the identification of 1542 OTUs, from which 1528 OTUs were extracted. A rarefaction curve was constructed by the random sampling of sequences and the number of OTUs that they represented (Amato et al., 2013). Sample rarefaction curves can be used to check the rationality of the data (Supplementary Figure 1). If the curve tends to be flat, more data will only generate a small number of new OTUs, indicating that the amount of sequencing data used is reasonable. The Shannon–Wiener curve (Edgar, 2013) is an index that reflects the diversity of microorganisms in a sample and is constructed using the microbial diversity index of each sample at different sequencing depths to reflect the microbial diversity of each sample with different sequencing quantities (Supplementary Figure 2). If the curve tends to be flat, the amount of sequencing data is large enough to reflect most of the microbial information in the sample. The alpha diversity indices of the cbbL-harboring bacteria were calculated with MOTHUR software at a 97% similarity based on the OTU clustering results.
ANOVA was performed to test the effects of the N addition gradient on cbbL gene abundance, cbbL OTUs, Shannon index values, bacterial phospholipid-derived fatty acids (PLFAs) and dominant cbbL-containing bacteria using IBM SPSS 20.0. Fisher’s least significant difference (LSD) multiple range test was used to determine the significance of differences among the N addition treatments.
Correlation analysis was used to assess how cbbL gene abundance and cbbL OTUs were related to the Shannon index using IBM SPSS 20.0.
We estimated the strength of the relationships between N addition and NO3-N and NH4-N contents, the N/P ratio, pH, cbbL gene abundance and cbbL diversity by structural equation modeling (SEM) using IBM AMOS 21.0. In this model, we hypothesized that N addition may directly alter the abundance and diversity of the cbbL gene and indirectly change the NO3-N and NH4-N contents, N/P ratio and pH value. We used the chi-square test (χ2), goodness-of-fit index (GFI), root mean square error of approximation (RMSEA) and Akaike information criterion (AIC) to assess the degree of fit of this model.
The overlap of the number of OTU groups under different N addition treatments is represented by a Venn diagram. Principal coordinate analysis (pCoA) was used to visualize the differences in cbbL gene communities among the different N addition treatments using the R software environment (version 3.6.1). To determine if N addition altered cbbL gene community composition, a permutational analysis of variance (PERMANOVA) analysis was conducted with the Bray–Curtis similarity index using the R software environment (version 3.6.1). The biomarkers with significant differences in abundance between groups under the different N addition treatments were identified by linear discriminant analysis effect size (LEfSe; score = 3).
The results showed that compared to the N0 treatment, the N150 treatment reduced the cbbL gene abundance, with an observed decrease of 8% (Figure 1A and Supplementary Table 2), whereas no differences were observed among the N0-N100 treatments. The OTUs decreased with increased N addition. Compared to the N0 treatment, the N150 treatment significantly reduced the OTUs by 9% (Figure 1B and Supplementary Table 2), whereas no significant differences were observed among the N0-N100 treatments. N addition significantly affected the Shannon index (P < 0.001; Figure 1C and Supplementary Table 2). Compared to that of the N0 treatment, the Shannon index decreased by 10.0 and 22.69% under the N100 and N150 treatments, respectively, whereas no differences were observed among the N0–N50 treatments. No significant differences were observed in the other cbbL genetic diversity indices (Chao1, observed species, and PD whole tree) with different N addition levels (P > 0.05; Supplementary Table 2). The results showed that compared to the N0 treatment, the N100 treatment enhanced the bacterial PLFAs, with an increase of 75% (Supplementary Figure 3 and Supplementary Table 2).
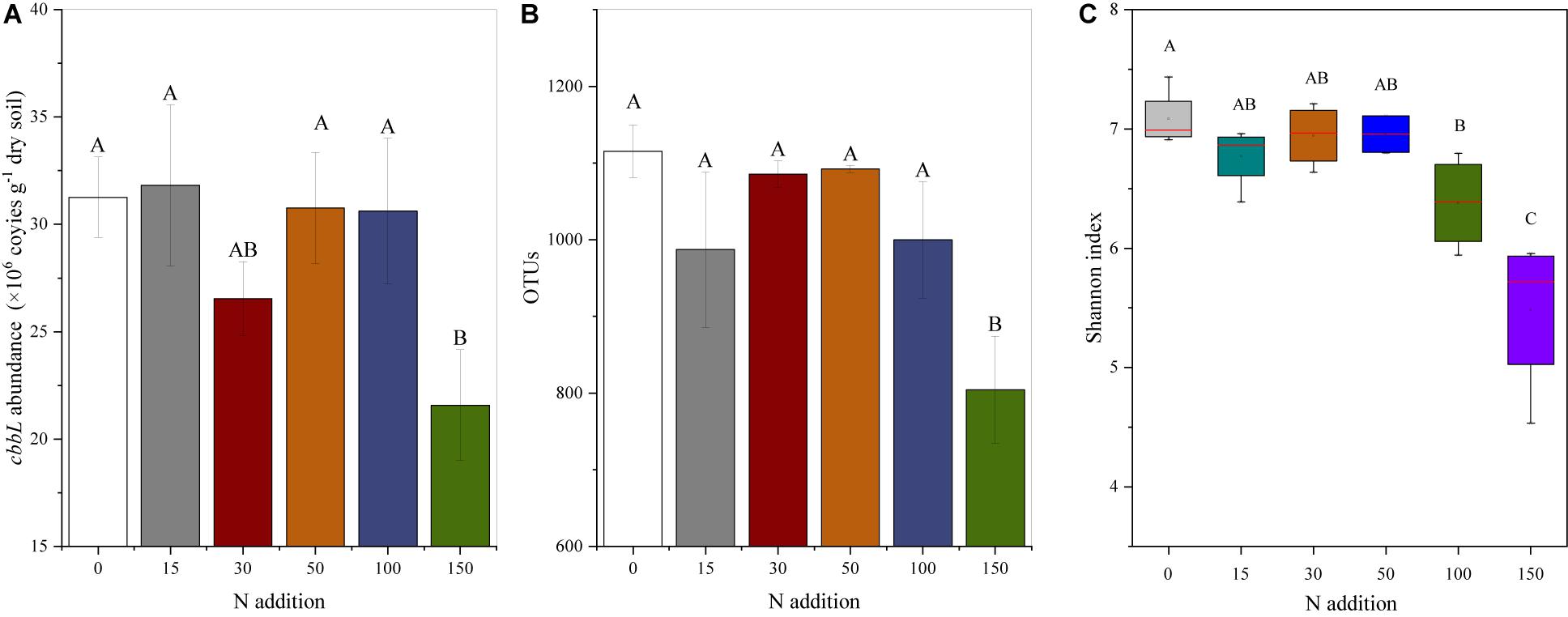
Figure 1. Effects of N addition on (A) cbbL gene abundance, (B) OTU abundance, and (C) diversity. The error bars show one standard error of the mean (A,B). The central mark in each box indicates the average value, the central line indicates the median, and the bottom and top edges of the box indicate the 25th and 75th percentiles, respectively. The whiskers extend to the most extreme data points that are not considered outliers, and the outliers are labeled with the ‘+’ symbol (C). Different letters above the bars indicate significant differences based on the LSD multiple range test (P < 0.05).
Under the different levels of N addition, 1061 common OTUs were observed, representing 69.7% of the total OTUs. The N0, N15, N100, and N150 treatments had 18, 13, 19, and 13 specific OTUs, respectively (Figure 2A). The PCoA results showed that the cbbL gene communities clustered strongly based on different N addition levels (Figure 2B). The PERMANOVA results for the cbbL gene communities was consistent with those of the PCoA and could explain 46.99% of the variation in the cbbL-containing bacterial communities under different N addition levels. Specifically, the first coordinate (PCoA1) separated the N100 and N150 treatments from the other N treatments (N0, N15, N30, and N50), while the second coordinate (PCoA2) explained the remaining 8.95% of the dissimilarity.
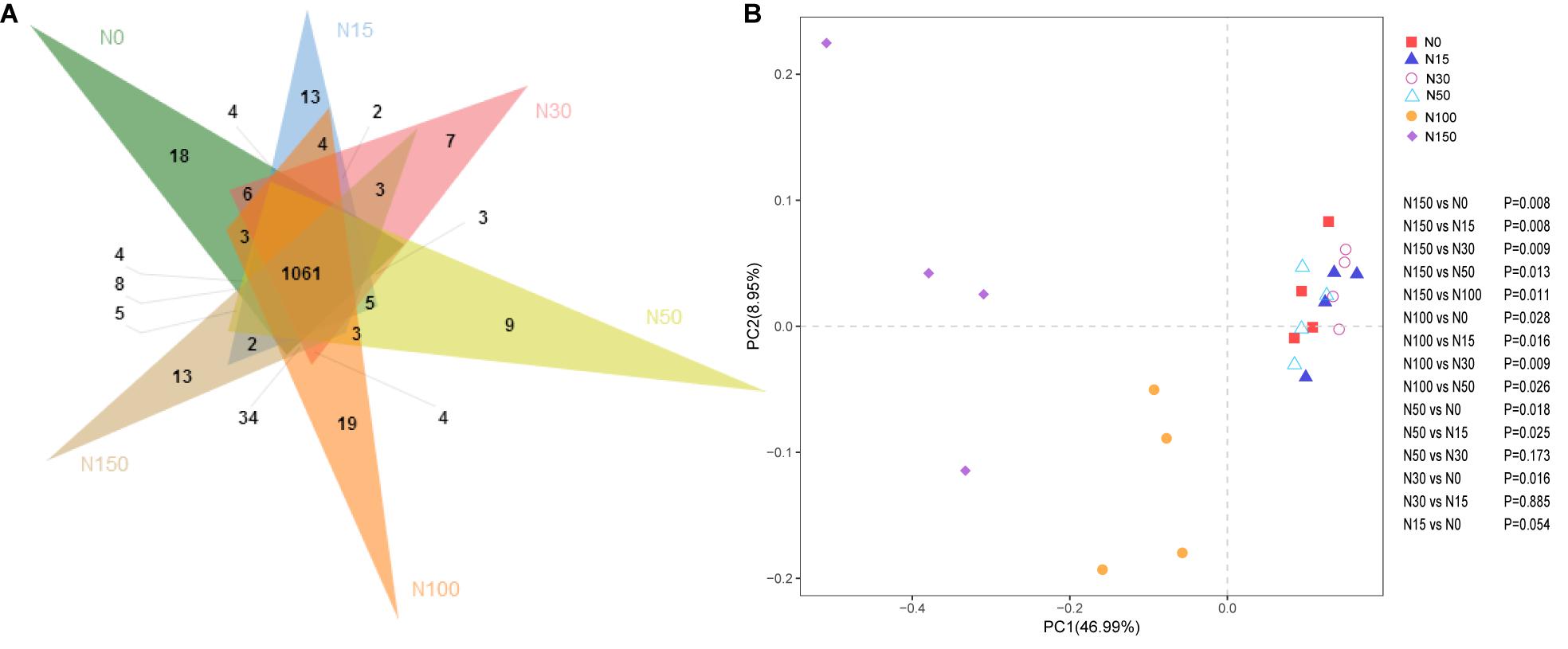
Figure 2. Effects of N addition on cbbL gene community composition. (A) Venn diagram of different N addition treatments. Different colors represent different N addition rates. The overlapping areas of the different colors represent the intersections (i.e., the common OTUs occur in areas where the color overlap, whereas the non-overlapping areas show unique OTUs). (B) Principal coordinate analysis (weighted PCoA) of cbbL gene communities based on Bray–Curtis dissimilarity matrices across different N addition treatments. Different shapes represent different N addition treatments. The percent variation explained by the plotted principal coordinates is indicated on the axes. Pairwise PERMANOVA results are displayed to the right of the PCoA. The cbbL-containing microbiota from the N100 (orange circles) and N150 (purple diamond) treatments clustered separately from cbbL-containing microbiota of the N0–N50 treatments (P < 0.05 by PERMANOVA).
At the phylum level, members of the phylum Proteobacteria were the dominant cbbL-containing bacteria (relative abundance > 80%), the relative abundance of which increased with increasing N addition (P < 0.001; Figure 3A and Supplementary Table 2). The results showed that compared to the N0 treatment, the N150 treatment enhanced the relative abundance of Proteobacteria, with an observed increase of 11% (Supplementary Table 3), whereas no differences were observed among the N0–N50 treatments.
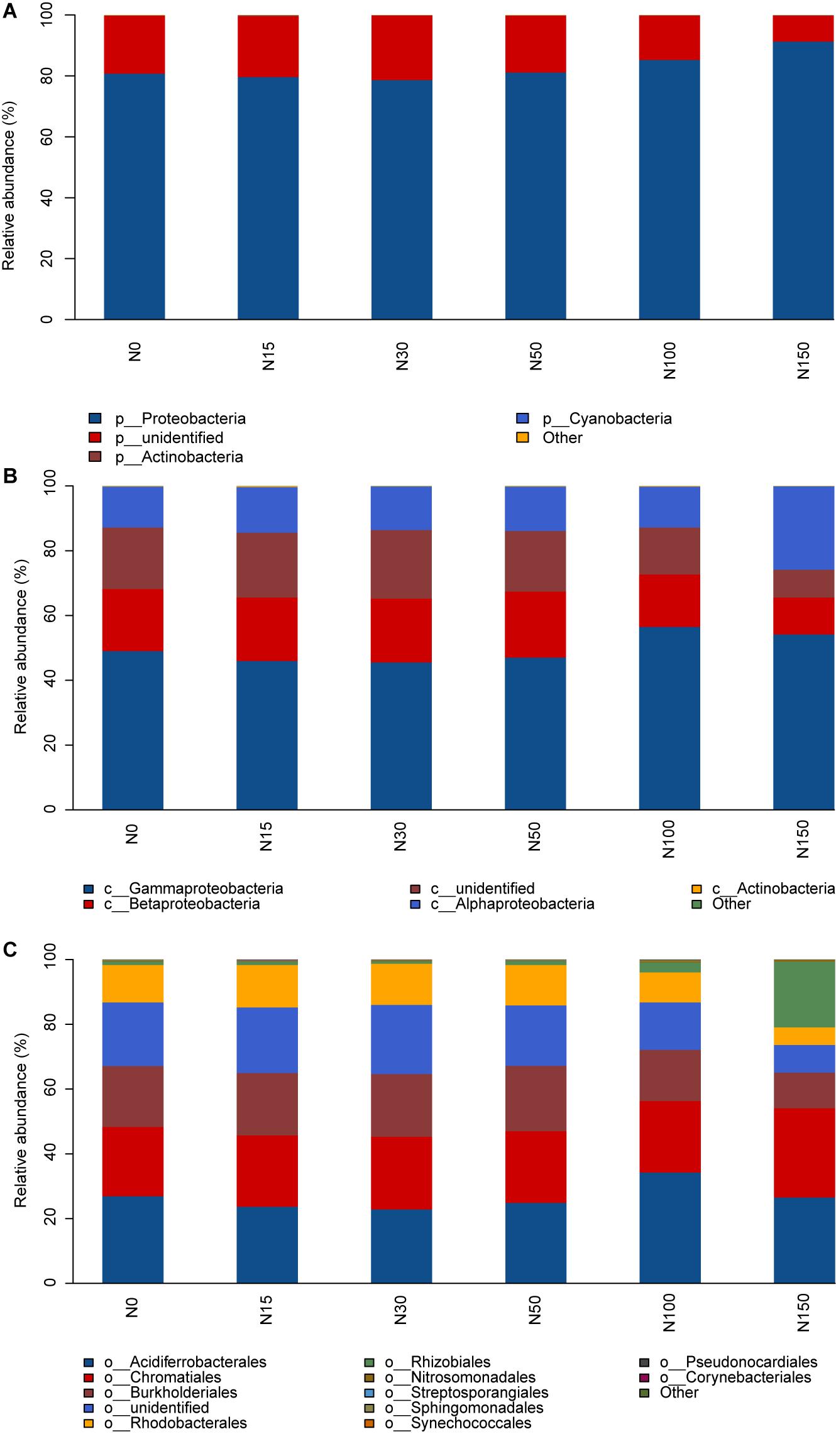
Figure 3. Relative abundances of the dominant phyla (A), classes (B), and orders (C) under different levels of N addition.
At the class level, Gammaproteobacteria (45.4–56.2% relative abundance), Betaproteobacteria (11.25–20.25% relative abundance), and Alphaproteobacteria (12.75–25.75% relative abundance) were the dominant cbbL-containing bacteria (Figure 3B). The relative abundances of Gammaproteobacteria and Alphaproteobacteria were enhanced with increasing N addition (P < 0.01; Figure 3B and Supplementary Tables 2, 3). Compared to the N15 treatment, the N150 treatment significantly increased the relative abundance of Gammaproteobacteria by 8%. Compared to the N0 treatment, the N150 treatment significantly increased the relative abundance of Alphaproteobacteria by 13%. The relative abundance of Betaproteobacteria was significantly decreased under the N100 and N150 treatments, whereas no differences were observed among the N0–N50 treatments (P < 0.001; Supplementary Table 3).
At the order level, Acidiferrobacterales (23.00–34.25% relative abundance), Chromatiales (21.5–27.5% relative abundance), Burkholderiales (11.00–20.00% relative abundance), Rhizobiales (1.0–20.00% relative abundance), and Rhodobacterales (5.50–13.00% relative abundance) were the dominant cbbL-containing bacteria (Figure 3C). The N100 treatment significantly increased the relative abundance of Acidiferrobacterales, whereas no differences were observed among the N0–N50 and N150 treatments. Compared to the N0 treatment, the N150 treatment significantly increased the relative abundance of Rhizobiales by 19%, whereas no differences were observed among the N0–N100 treatments. The relative abundance of Burkholderiales and Rhodobacterales was significantly decreased in the N100 and N150 additions, whereas no differences were observed among the N0–N50 treatments (P < 0.001; Figure 3C and Supplementary Tables 2, 3).
The structure of the cbbL -containing gene community changed under N addition in 10 families (Supplementary Figure 4). Under the N100 treatment, significant differences in cbbL-containing microbes occurred in the families Acidiferrobacteraceae (order Acidiferrobacterales) and Sphingomonadaceae (order Sphingomonadales). Under the N150 treatment, a significant difference in cbbL-containing microbes occurred in the family Bradyrhizobiaceae (order Rhizobiales).
The correlation analysis results showed that the Shannon index was positively correlated with cbbL gene abundance (r2 = 0.464, P = 0.022; Supplementary Figure 5). The number of OTUs was positively correlated with cbbL gene abundance (r2 = 0.620, P = 0.001; Supplementary Table 4). The abundance of cbbL-containing microbes was significantly correlated with soil properties. The cbbL gene abundance (r2 = 0.473, P = 0.002; Supplementary Table 5) and the Shannon index were negatively correlated with total N (r2 = 0.541, P = 0.006; Supplementary Table 4), while the Shannon index was negatively correlated with NO3-N (r2 = 0.596, P = 0.002; Supplementary Table 5) and NH4-N (r2 = 0.521, P = 0.009; Supplementary Table 5). A positive correlation was observed between pH and the number of OTUs (r2 = 0.443, P = 0.03; Supplementary Table 5), cbbL gene abundance (r2 = 0.442, P = 0.031; Supplementary Table 5) and the Shannon index (r2 = 0.634, P = 0.001; Supplementary Table 5). In addition, the cbbL gene abundance (r2 = 0.460, P = 0.024; Supplementary Table 5) and the Shannon index (r2 = 0.598, P = 0.002, Supplementary Table 4) were negatively correlated with the N/P ratio. There was a positive correlation between C/N and the number of OTUs (r2 = 0.438, P = 0.032; Supplementary Table 5), cbbL gene abundance (r2 = 0.552, P = 0.005; Supplementary Table 5) and the Shannon index (r2 = 0.606, P = 0.002; Supplementary Table 5). No correlations were observed between cbbL-containing microbes and SOC levels (Supplementary Table 5).
The SEM results explained 70% of the variation in cbbL gene abundance (Figure 4A and Supplementary Table 6) and showed that N addition indirectly affected the cbbL gene abundance by altering the soil N/P ratio and soil pH. However, the contribution of available N was highly limited. The SEM explained 88% of the variation in the diversity of cbbL-containing microbial OTUs. In addition, the SEM results (Figure 4B and Supplementary Table 6) showed that N addition indirectly affected the diversity of cbbL-containing microbes by altering the soil NO3-N content and soil pH.
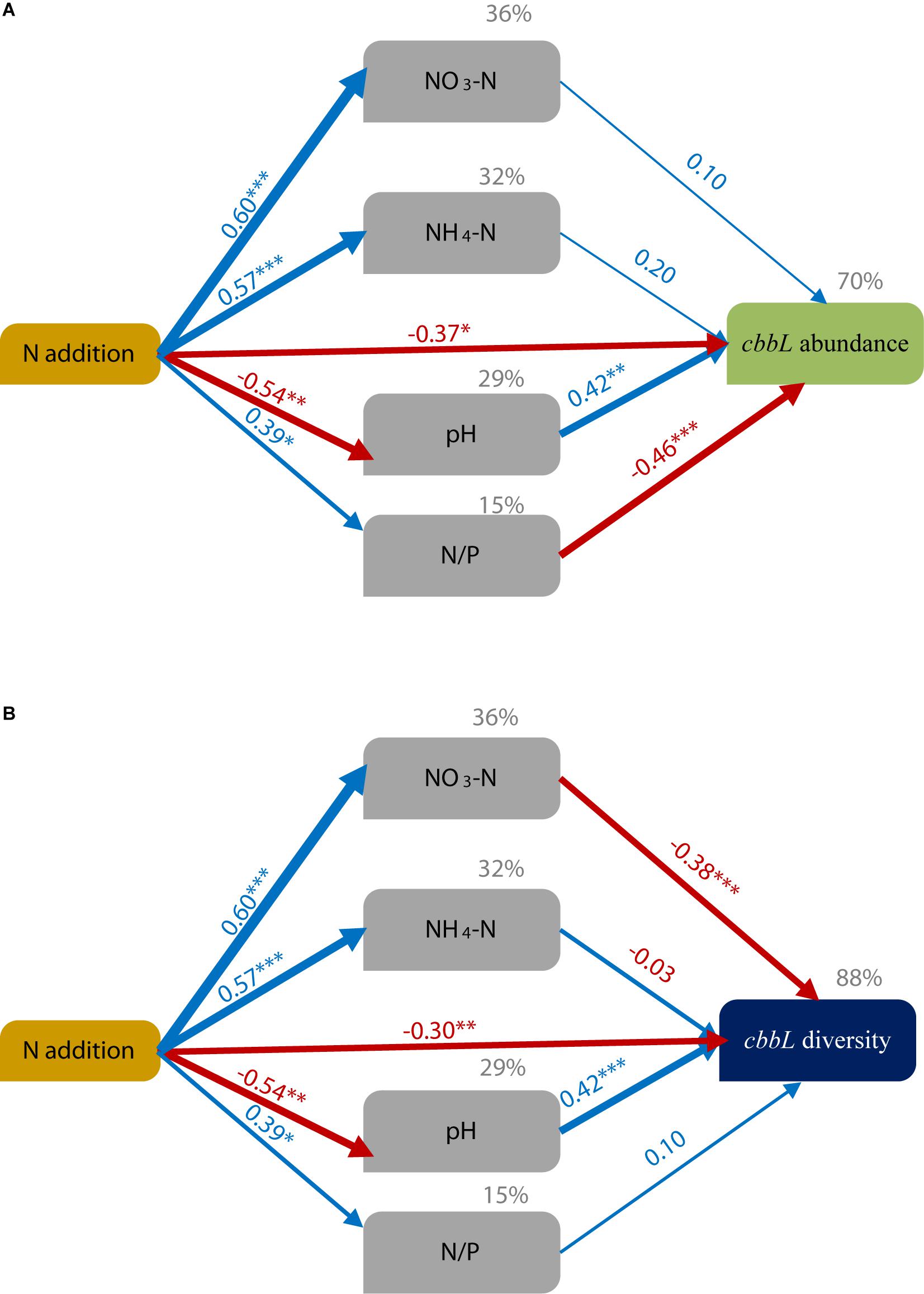
Figure 4. Structural equation modeling (SEM) of the effects of N addition on soil physicochemical factors (pH, NO3-N, NH4-N, and the N/P ratio) and CO2-fixing microbes [cbbL gene abundance (A) and cbbL diversity (B)]. The final model resulted in a good fit to the data. ***P ≤ 0.001, **P ≤ 0.01, *P ≤ 0.05. For cbbL gene abundance: χ2 = 6.575, df = 6, P = 0.362; RMSEA = 0.065; GFI = 0.919 and AIC = 36.575. For cbbL diversity: χ2 = 6.575, df = 6, P = 0.362; RMSEA = 0.065; GFI = 0.919; and AIC = 36.575. The number above the arrow indicates the standardized path coefficient, and the width of the arrow indicates the effect size of the relationship. The percent (gray) variance explained (R2) is shown above each variable. The blue arrows indicate a significant positive relationship (P < 0.05), while the red arrows indicate a negative relationship (P < 0.05).
In the present study, we investigated the responses of CO2-fixing microbes to N additions through a 9-year controlled experiment conducted in temperate grassland. Three primary results emerged: (1) N addition reduced the cbbL gene abundance and the diversity of cbbL-containing microbes; (2) the decrease in cbbL-containing microbial diversity was associated with an increase in NO3-N content and pH; and (3) N deposition reduced cbbL gene abundance, possibly by altering the soil pH and N/P ratio.
Treseder (2008) previously observed that microbial biomass decreased by 15% under N deposition, indicating that atmospheric N deposition would inhibit the growth and reproduction of soil microorganisms in 28 regions worldwide. Our results demonstrated that high N addition reduced both cbbL gene abundance and diversity. Research findings on the effects of N addition on microbial diversity have been inconsistent. The results of a previous meta-analysis revealed that N addition, particularly high N addition (above 100 kg N ha–1 yr–1), decreased soil microbial diversity, although the effects may vary among different ecosystems (Wang C. et al., 2018). In contrast, Zhou et al. (2019) observed that cbbL gene diversity was lowest under 0N addition conditions and that there was a positive effect of nutrient addition on soil cbbL gene diversity after 26 years of fertilization. Under long-term field fertilization, an increase in soil available nutrients has been shown to promote cbbL gene abundance and diversity (Yuan et al., 2012). Our results showed that high N addition (150 kg N ha–1 yr–1) decreased the cbbL gene abundance and diversity in temperate grasslands after 9 years of N addition. Thus, the different response patterns among various plant community compositions and long-term fertilization may depend on the ecosystem environment.
The decreased cbbL gene abundance and diversity were observed to be linked to changes in soil properties, particularly the concentration of NO3-N, the soil pH and the N/P ratio, associated with N addition. As N addition increased, the NO3-N content and the N/P ratio increased significantly, whereas the soil pH decreased significantly (Supplementary Table 1). The soil pH decreased with the increased addition of NO3-N, which is consistent with the results of previous studies (Zeng et al., 2016; Wang Q. et al., 2018; Hong et al., 2019). Zhao et al. (2018) also observed that cbbL gene abundance was significantly correlated with soil pH. N addition has been shown to result in an imbalance in the soil N/P ratio in terrestrial ecosystems (Vitousek et al., 2010; Li et al., 2016). The soil N/P ratio appeared to have a negative effect on cbbL gene abundance, with the N/P ratio being the key influencing factor. The soil available N content was previously shown to be significantly correlated with the gene abundance of CO2-fixing microorganisms (Huang et al., 2018; Du et al., 2019). However, the contributions of NH4-N and NO3-N contents to the cbbL gene abundance were limited under N addition conditions in our present study.
Nutrient availability can alter microbial diversity (Zhang et al., 2012). Our results showed that N addition primarily reduced the diversity of cbbL-containing microbes by altering the soil NO3-N concentration and pH. Zhao et al. (2018) previously reported that high concentrations of available N increase the diversity of CO2-fixing microorganisms. These results contrast with our findings, which indicated that high NO3-N concentrations suppress the diversity of CO2-fixing microorganisms. Soil microorganisms are becoming increasingly vulnerable due to increasing N deposition in grasslands (Wang C. et al., 2018).
N addition consistently altered cbbL gene community composition. The order Acidiferrobacterales was previously identified as the dominant group of CO2-fixing of microbes in temperate grassland, while Rhizobiales became the dominant order under high N addition. Zhou et al. (2019) observed that Proteobacteria was the dominant known soil cbbL-containing bacterial phylum with the highest abundance after 26 years of fertilization. Consistent with their results, we observed that the relative abundance of Proteobacteria was greater than 80% and increased with N addition. Compared to low and intermediate rates of N addition, the CO2-fixing microbial community characteristics were different under the N100 and N150 treatments. The relative abundances of Acidiferrobacterales and Rhizobiales were higher in the N100 and N150 treatments than those observed in the other treatments. Consistent with our results, Zhang et al. (2017) observed that the relative abundance of Acidobacteria increased at a high N level. The response of Acidiferrobacterales is primarily caused by N addition and pH changes (Ramirez et al., 2012). Our results showed that members of the order Rhizobiales became the dominant CO2-fixing microbes under the N150 treatment (from a 1% relative abundance under N0 addition to a 20.25% relative abundance under N150), which might play a significant role in microbial CO2 fixation. Research on grassland and forest ecosystems has also demonstrated that Rhizobium is the dominant bacterium among carbon-fixing microorganisms (Guo et al., 2015; Li et al., 2018; Zhao et al., 2018). The dominant populations of CO2-fixing microorganisms in different research areas vary greatly and are ultimately determined by soil properties and genetic characteristics (Tolli and King, 2005).
Soil organic carbon is one of the most significant factors influencing the abundance and diversity of cbbL-containing bacteria (Yuan et al., 2012; Xiao et al., 2014; Li et al., 2018), but our results indicated that SOC contributed little to the changes in CO2-fixing microbes under N addition. Research shows that the SOC concentration, particularly significant changes in SOC concentration, has a significant relationship with the diversity and abundance of cbbL-containing bacterial communities (Osborn et al., 2000; Yuan et al., 2012). In our present study, N addition did not alter the SOC concentration (Supplementary Table 1). Another N deposition study also reported no impact of N addition on SOC (Schleuss et al., 2019). Such patterns suggest that the contribution of SOC to CO2-fixing microbes is highly limited under the N addition conditions studied. This inconsistency in the results suggests that other important factors besides SOC may trigger changes in CO2-fixing microbes. We also considered other factors that could potentially impact CO2-fixing microbes in response to N addition, such as the N/P ratio. Several previous studies have reported that the N/P ratio, particularly under P deficiency, is crucial under N addition conditions and impacts CO2-fixing microbes (Dong et al., 2019). Consistent with their results, we observed significant negative correlations between the N/P ratio and the abundance of CO2-fixing microbes.
In the present study, the coregulatory mechanism of soil available N and pH is the most likely factor responsible for the observed loss of the abundance and diversity of CO2-fixing microbes under high N addition. First, N deposition can influence the soil available N concentration (Liu et al., 2013), where high NO3-N levels may inhibit CO2-fixing microbial activity. The increased concentration of NH4-N did not significantly influence CO2-fixing microbial diversity. Since effects of NH4-N and NO3-N on soil CO2-fixing microbial diversity have been observed, with the increasing proportion of NO3-N in N deposition, the impact of N deposition on soil CO2-fixing microbiota may become more severe (Liu et al., 2013). However, observations on the effects of soil NH4-N were from one grassland ecosystem, and whether the same phenomenon will happen in other systems should be further studied. Second, the significant positive correlation between the changes in the abundance and diversity of CO2-fixing microbes and the changes in soil pH and SEM results both indicated a link between CO2-fixing microbes and soil pH. Soil pH changes in soil co-occur with interactions among soil available nutrition (e.g., available N) (Lammel et al., 2018), masking many indirect effects of pH on soil CO2-fixing microbes. High levels of N addition can drive soil acidification both directly and indirectly (Guo et al., 2010). The consequence of soil acidification is the destruction of microecological balance, which leads to the loss of a stable and healthy soil environment (Wan et al., 2020) and is unfavorable for CO2-fixing microbial growth.
To the best of our knowledge, this is the first study to report the negative effects of N addition on the CO2-fixing microbes present in temperate grassland. We showed that N addition primarily reduced the abundance and diversity of CO2-fixing microbes by altering soil available N and soil pH. These findings suggest that N addition can alter CO2-fixing microbes and indicate the importance of the coregulation of soil available N and pH under N addition. The contribution of SOC to alterations in the various CO2-fixing microbes under increased N addition was very limited. As different effects of NH4-N and NO3-N on soil CO2-fixing microbial diversity were observed in the present study, we further speculate that with the increasing proportion of NO3-N in N deposition, the impact of N deposition on soil CO2-fixing microbiota may become more severe.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
DY conceived and designed the experiments. ML, HZ, JZ, and HL performed the experiments. JQ analyzed the data. JQ and DY wrote the manuscript. All authors approved the final manuscript.
This work was supported by the National Natural Science Foundation of China (41877343 and 42007046), the Cooperative Innovation Project of Agricultural Science and Technology Innovation Program of CAAS (CAAS-XTCX2016015), and the National Special Transgenic Project (2016ZX08012005-005).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2021.570908/full#supplementary-material
Supplementary Figure 1 | Sample rarefaction curves. F, Fo, S, and T represent the experimental replicates.
Supplementary Figure 2 | Shannon–Wiener curves. F, Fo, S, and T represent the experimental replicates.
Supplementary Figure 3 | Effects of N addition on the bacterial Plfas. The different letters above bars indicate significant differences based on the Lsd multiple range test (P < 0.05). The central mark in each box indicates the average value, the central line indicates the median, and the bottom and top edges of the box indicate the 25th and 75th percentiles, respectively. The whiskers extend to the most extreme data points that are not considered outliers, and the outliers are labeled with the ‘+’ symbol.
Supplementary Figure 4 | Cladogram showing carbon dioxide-fixing microbial community compositions under different N addition treatments (LefSe; score = 3). The six rings of the cladogram represent the domain (innermost) phylum, class, order, family, and genus. The enlarged circles in dark green, blue, and red are differentially abundant taxa identified as taxonomic biomarkers under the different treatments. Lineages with linear discriminant analysis scores with a threshold value of 3.0 were used to identify the degree of differentiation between treatments.
Supplementary Figure 5 | Relationship between the cbbL Shannon index values and abundance. The solid red line indicates a significant relationship, and the shaded area shows the 95% confidence interval of the fit.
Supplementary Table 1 | Soil physicochemical factors under different N addition levels.
Supplementary Table 2 | Results (F values) of an Anova of the effects of N addition on bacterial Plfas, cbbL gene abundance, Otus, the Shannon index, and the relative abundance of dominant cbbL-containing microbes.
Supplementary Table 3 | Soil cbbL-containing microbes under different N addition levels.
Supplementary Table 4 | Correlation analysis between the bacterial Plfas and cbbL Otus, abundance, and Shannon index values.
Supplementary Table 5 | Correlation analyses between soil properties and the cbbL Otus, abundance, and Shannon index values.
Supplementary Table 6 | Structural equation modeling of the effect of N addition on the cbbL diversity (Otus) and abundance through all plausible interaction pathways. The tables show the unstandardized path coefficients (estimates), standard error of regression weights (S.E.), critical values for the regression weights (C.R.), and levels of significance of the regression weights (P). ∗∗∗ Indicates P ≤ 0.001, ∗∗ indicates P ≤ 0.01, and ∗ indicates P ≤ 0.05.
Amato, K. R., Yeoman, C. J., Kent, A., Righini, N., Carbonero, F., Estrada, A., et al. (2013). Habitat degradation impacts black howler monkey (Alouatta pigra) gastrointestinal microbiomes. Isme J. 7, 1344–1353. doi: 10.1038/ismej.2013.16
Arora, V. K., and Melton, J. R. (2018). Reduction in global area burned and wildfire emissions since 1930s enhances carbon uptake by land. Nat. Commun. 9:1326.
Dong, C. C., Wang, W., Liu, H. Y., Xu, X. T., and Zeng, H. (2019). Temperate grassland shifted from nitrogen to phosphorus limitation induced by degradation and nitrogen deposition: evidence from soil extracellular enzyme stoichiometry. Ecol. Indicat. 101, 453–464. doi: 10.1016/j.ecolind.2019.01.046
Du, Y., Wang, T. Y., Anane, P. S., Li, Q., Liu, S. X., and Wang, C. Y. (2019). Effects of different types of biochar on basic properties and bacterial communities of black soil. Appl. Ecol. Environ. Res. 17, 5305–5319. doi: 10.15666/aeer/1702_53055319
Edgar, R. C. (2013). UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 10, 996–998. doi: 10.1038/Nmeth.2604
Ferrenberg, S., Reed, S. C., and Belnap, J. (2015). Climate change and physical disturbance cause similar community shifts in biological soil crusts. Proc. Natl. Acad. Sci. U.S.A. 112, 12116–12121. doi: 10.1073/pnas.1509150112
Galloway, J. N., Dentener, F. J., Capone, D. G., Boyer, E. W., Howarth, R. W., Seitzinger, S. P., et al. (2004). Nitrogen cycles: past, present, and future. Biogeochemistry 70, 153–226. doi: 10.1007/s10533-004-0370-0
Ge, T. D., Wu, X. H., Liu, Q., Zhu, Z. K., Yuan, H. Z., Wang, W., et al. (2016). Effect of simulated tillage on microbial autotrophic CO2 fixation in paddy and upland soils. Sci. Rep. 6:19784. doi: 10.1038/srep19784
Gruber, N., and Galloway, J. N. (2008). An Earth-system perspective of the global nitrogen cycle. Nature 451, 293–296. doi: 10.1038/nature06592
Guo, G. X., Kong, W. D., Liu, J. B., Zhao, J. X., Du, H. D., Zhang, X. Z., et al. (2015). Diversity and distribution of autotrophic microbial community along environmental gradients in grassland soils on the Tibetan Plateau. Appl. Microbiol. Biotechnol. 99, 8765–8776. doi: 10.1007/s00253-015-6723-x
Guo, J. H., Liu, X. J., Zhang, Y., Shen, J. L., Han, W. X., Zhang, W. F., et al. (2010). Significant acidification in major Chinese croplands. Science 327, 1008–1010. doi: 10.1126/science.1182570
Guzman, M. S., Rengasamy, K., Binkley, M. M., Jones, C., Ranaivoarisoa, T. O., Singh, R., et al. (2019). Phototrophic extracellular electron uptake is linked to carbon dioxide fixation in the bacterium Rhodopseudomonas palustris. Nat. Commun. 10:1355.
Hart, K. M., Kulakova, A. N., Allen, C. C. R., Simpson, A. J., Oppenheimer, S. F., Masoom, H., et al. (2013). Tracking the fate of microbially sequestered carbon dioxide in soil organic matter. Environ. Sci. Technol. 47, 5128–5137. doi: 10.1021/es3050696
Hong, S. B., Gan, P., and Chen, A. P. (2019). Environmental controls on soil pH in planted forest and its response to nitrogen deposition. Environ. Res. 172, 159–165. doi: 10.1016/j.envres.2019.02.020
Huang, X. Z., Wang, C., Liu, Q., Zhu, Z. K., Lynn, T. M., Shen, J. L., et al. (2018). Abundance of microbial CO2-fixing genes during the late rice season in a long-term management paddy field amended with straw and straw-derived biochar. Canad. J. Soil Sci. 98, 306–316. doi: 10.1139/cjss-2017-0098
Lammel, D. R., Barth, G., Ovaskainen, O., Cruz, L. M., Zanatta, J. A., Ryo, M., et al. (2018). Direct and indirect effects of a pH gradient bring insights into the mechanisms driving prokaryotic community structures. Microbiome 6:1186. doi: 10.1186/s40168-018-0482-8
Li, M. Q., Xu, J. Q., Jiang, Z. W., and Li, Q. L. (2020). Molecular understanding of autotrophic CO2-fixing bacterial communities in composting based on RuBisCO genes analysis. J. Biotechnol. 320, 36–43. doi: 10.1016/j.jbiotec.2020.06.010
Li, P. P., Chen, W. J., Han, Y. L., Wang, D. C., Zhang, Y. T., and Wu, C. F. (2020). Effects of straw and its biochar applications on the abundance and community structure of CO2-fixing bacteria in a sandy agricultural soil. J. Soils Sedim. 20, 2225–2235. doi: 10.1007/s11368-020-02584-5
Li, Y., Niu, S. L., and Yu, G. R. (2016). Aggravated phosphorus limitation on biomass production under increasing nitrogen loading: a meta-analysis. Glob. Change Biol. 22, 934–943. doi: 10.1111/gcb.13125
Li, Y. C., Liang, X., Tang, C. X., Li, Y. F., Chen, Z. H., Chang, S. X., et al. (2018). Moso bamboo invasion into broadleaf forests is associated with greater abundance and activity of soil autotrophic bacteria. Plant Soil 428, 163–177. doi: 10.1007/s11104-018-3648-z
Liu, X. J., Duan, L., Mo, J. M., Du, E. Z., Shen, J. L., Lu, X. K., et al. (2011). Nitrogen deposition and its ecological impact in China: an overview. Environ. Pollut. 159, 2251–2264. doi: 10.1016/j.envpol.2010.08.002
Liu, X. J., Zhang, Y., Han, W. X., Tang, A. H., Shen, J. L., Cui, Z. L., et al. (2013). Enhanced nitrogen deposition over China. Nature 494, 459–462. doi: 10.1038/nature11917
Lu, C. Q., and Tian, H. Q. (2014). Half-century nitrogen deposition increase across China: a gridded time-series data set for regional environmental assessments. Atmosph. Environ. 97, 68–74. doi: 10.1016/j.atmosenv.2014.07.061
Lynn, T. M., Ge, T. D., Yuan, H. Z., Wei, X. M., Wu, X. H., Xiao, K. Q., et al. (2017). Soil Carbon-fixation rates and associated bacterial diversity and abundance in three natural ecosystems. Microb. Ecol. 73, 645–657. doi: 10.1007/s00248-016-0890-x
Mo, J., Zhang, W., Zhu, W., Gundersen, P., Fang, Y., Li, D., et al. (2008). Nitrogen addition reduces soil respiration in a mature tropical forest in southern China. Glob. Change Biol. 14, 403–412. doi: 10.1111/j.1365-2486.2007.01503.x
Neff, J. C., Townsend, A. R., Gleixner, G., Lehman, S. J., Turnbull, J., and Bowman, W. D. (2002). Variable effects of nitrogen additions on the stability and turnover of soil carbon. Nature 419, 915–917. doi: 10.1038/nature01136
Osborn, A. M., Moore, E. R. B., and Timmis, K. N. (2000). An evaluation of terminal-restriction fragment length polymorphism (T-RFLP) analysis for the study of microbial community structure and dynamics. Environ. Microbiol. 2, 39–50. doi: 10.1046/j.1462-2920.2000.00081.x
Penuelas, J., Poulter, B., Sardans, J., Ciais, P., van der Velde, M., Bopp, L., et al. (2013). Human-induced nitrogen-phosphorus imbalances alter natural and managed ecosystems across the globe. Nat. Commun. 4:2934. doi: 10.1038/ncomms3934
Qin, J., Liu, H., Zhao, J., Wang, H., Zhang, H., Yang, D., et al. (2020). The roles of bacteria in soil organic carbon accumulation under nitrogen deposition in stipa baicalensis steppe. Microorganisms 8:326. doi: 10.3390/microorganisms8030326
Ramirez, K. S., Craine, J. M., and Fierer, N. (2012). Consistent effects of nitrogen amendments on soil microbial communities and processes across biomes. Glob. Change Biol. 18, 1918–1927. doi: 10.1111/j.1365-2486.2012.02639.x
Rognes, T., Flouri, T., Nichols, B., Quince, C., and Mahe, F. (2016). VSEARCH: a versatile open source tool for metagenomics. Peerj 4:e2584. doi: 10.7717/peerj.2584
Schleuss, P. M., Widdig, M., Heintz-Buschart, A., Guhr, A., Martin, S., Kirkman, K., et al. (2019). Stoichiometric controls of soil carbon and nitrogen cycling after long-term nitrogen and phosphorus addition in a mesic grassland in South Africa. Soil Biol. Biochem. 135, 294–303. doi: 10.1016/j.soilbio.2019.05.018
Simpson, A. J., Simpson, M. J., Smith, E., and Kelleher, B. P. (2007). Microbially derived inputs to soil organic matter: are current estimates too low? Environ. Sci. Technol. 41, 8070–8076. doi: 10.1021/es071217x
Stevens, C. J., Dise, N. B., Mountford, J. O., and Gowing, D. J. (2004). Impact of nitrogen deposition on the species richness of grasslands. Science 303, 1876–1879. doi: 10.1126/science.1094678
Tolli, J., and King, G. M. (2005). Diversity and structure of bacterial chemolithotrophic communities in pine forest and agroecosystem soils. Appl. Environ. Microbiol. 71, 8411–8418. doi: 10.1128/Aem.71.12.8411-8418.2005
Treseder, K. K. (2008). Nitrogen additions and microbial biomass: a meta-analysis of ecosystem studies. Ecol. Lett. 11, 1111–1120. doi: 10.1111/j.1461-0248.2008.01230.x
Vitousek, P. M., Porder, S., Houlton, B. Z., and Chadwick, O. A. (2010). Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen-phosphorus interactions. Ecol. Appl. 20, 5–15. doi: 10.1890/08-0127.1
Wan, W. J., Tan, J. D., Wang, Y., Qin, Y., He, H. M., Wu, H. Q., et al. (2020). Responses of the rhizosphere bacterial community in acidic crop soil to pH: changes in diversity, composition, interaction, and function. Sci. Total Environ. 700:134418.
Wang, C., Liu, D. W., and Bai, E. (2018). Decreasing soil microbial diversity is associated with decreasing microbial biomass under nitrogen addition. Soil Biol. Biochem. 120, 126–133. doi: 10.1016/j.soilbio.2018.02.003
Wang, Q., Wang, C., Yu, W. W., Turak, A., Chen, D. W., Huang, Y., et al. (2018). Effects of nitrogen and phosphorus inputs on soil bacterial abundance, diversity, and community composition in chinese fir plantations. Front. Microbiol. 9:1543. doi: 10.3389/fmicb.2018.01543
Xiao, K. Q., Bao, P., Bao, Q. L., Jia, Y., Huang, F. Y., Su, J. Q., et al. (2014). Quantitative analyses of ribulose-1,5-bisphosphate carboxylase/oxygenase (RubisCO) large-subunit genes (cbbL) in typical paddy soils. FEMS Microbiol. Ecol. 87, 89–101. doi: 10.1111/1574-6941.12193
Yuan, H., Ge, T., Wu, X., Liu, S., Tong, C., Qin, H., et al. (2012). Long-term field fertilization alters the diversity of autotrophic bacteria based on the ribulose-1,5-biphosphate carboxylase/oxygenase (RubisCO) large-subunit genes in paddy soil. Appl. Microbiol. Biotechnol. 95, 1061–1071. doi: 10.1007/s00253-011-3760-y
Yue, L. Y., Kong, W. D., Ji, M. K., Liu, J. B., and Morgan-Kiss, R. M. (2019). Community response of microbial primary producers to salinity is primarily driven by nutrients in lakes. Sci. Total Environ. 696:134001.
Zeng, J., Liu, X. J., Song, L., Lin, X. G., Zhang, H. Y., Shen, C. C., et al. (2016). Nitrogen fertilization directly affects soil bacterial diversity and indirectly affects bacterial community composition. Soil Biol. Biochem. 92, 41–49. doi: 10.1016/j.soilbio.2015.09.018
Zhang, H. F., Liu, H. M., Zhao, J. N., Wang, L. L., Li, G., Huangfu, C. H., et al. (2017). Elevated precipitation modifies the relationship between plant diversity and soil bacterial diversity under nitrogen deposition in Stipa baicalensis steppe. Appl. Soil Ecol. 119, 345–353. doi: 10.1016/j.apsoil.2017.06.005
Zhang, L. M., Hu, H. W., Shen, J. P., and He, J. Z. (2012). Ammonia-oxidizing archaea have more important role than ammonia-oxidizing bacteria in ammonia oxidation of strongly acidic soils. Isme J. 6, 1032–1045. doi: 10.1038/ismej.2011.168
Zhang, Y., Zheng, L. X., Liu, X. J., Jickells, T., Cape, J. N., Goulding, K., et al. (2008). Evidence for organic N deposition and its anthropogenic sources in China. Atmospher. Environ. 42, 1035–1041. doi: 10.1016/j.atmosenv.2007.12.015
Zhao, K., Kong, W. D., Wang, F., Long, X. E., Guo, C. Y., Yue, L. Y., et al. (2018). Desert and steppe soils exhibit lower autotrophic microbial abundance but higher atmospheric CO2 fixation capacity than meadow soils. Soil Biol. Biochem. 127, 230–238. doi: 10.1016/j.soilbio.2018.09.034
Zhou, Z. F., Wei, W. L., Shi, X. J., Li, Y. M., Liu, X. H., and Wang, M. X. (2019). Twenty-six years of chemical fertilization decreased soil RubisCO activity and changed the ecological characteristics of soil cbbL-carrying bacteria in an entisol. Appl. Soil Ecol. 141, 1–9. doi: 10.1016/j.apsoil.2019.05.005
Keywords: nitrogen deposition, grassland, diversity, cbbL gene, CO2-fixing microbes
Citation: Qin J, Li M, Zhang H, Liu H, Zhao J and Yang D (2021) Nitrogen Deposition Reduces the Diversity and Abundance of cbbL Gene-Containing CO2-Fixing Microorganisms in the Soil of the Stipa baicalensis Steppe. Front. Microbiol. 12:570908. doi: 10.3389/fmicb.2021.570908
Received: 09 June 2020; Accepted: 10 February 2021;
Published: 02 March 2021.
Edited by:
Marc Gregory Dumont, University of Southampton, United KingdomReviewed by:
Kai Xue, University of Chinese Academy of Sciences, ChinaCopyright © 2021 Qin, Li, Zhang, Liu, Zhao and Yang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jianning Zhao, emhhb2ppYW5uaW5nQGNhYXMuY24=; Dianlin Yang, eWFuZ2RpYW5saW5AY2Fhcy5jbg==; Jie Qin, cWluamllMzMxMzEzQDE2My5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.