
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
BRIEF RESEARCH REPORT article
Front. Med. , 12 October 2022
Sec. Dermatology
Volume 9 - 2022 | https://doi.org/10.3389/fmed.2022.997183
This article is part of the Research Topic Psychosocial Aspects of Skin Conditions and Diseases View all 15 articles
Itch and pain are common after burns. Neuropathic mechanisms may underlie both modalities but remain not well-understood. This study aims to prospectively document neuropathic pain symptoms and to identify potential itch symptom profiles that differ regarding duration and co-occurrence with neuropathic pain which may inform underlying pathophysiological mechanisms and respond to different treatments. Adult burn survivors (n = 192) self-reported itch and neuropathic pain at 2 weeks post-discharge, 3, 6, 12, and 18 months post-burn. Based on the presence of itch and pain symptoms over time, participants were allocated to one itch profile: transient itch/pain, chronic itch, or chronic itch & pain. Profiles were compared on itch intensity over time using General Linear Modeling. Age, gender, burn severity, posttraumatic stress (PTS) symptoms and baseline itch intensity were examined as potential predictors of the profiles in a Multi-nominal regression analysis. Neuropathic pain occurred in 54% after discharge which decreased to 24% 18 months later. Itch intensity was highest in the chronic itch & pain profile. Compared to the transient itch profile, the chronic itch & pain profile was associated with higher burn severity and more PTS symptoms. Compared to the chronic itch profile, the chronic itch & pain profile was associated with more PTS symptoms. Findings suggest that biological and psycho-dermatological processes underlie both chronic neuropathic pain and itch processes in burn scars. Further research should elucidate the mechanisms underlying the different itch profiles, with specific focus on skin innervation and psychological factors.
Over the past decade, studies have shown that prevalence rates of pain and itch after the acute phase of burn injury continue to be high. During hospitalization, most patients suffer from pain and itch (1, 2). Although the vast majority of studies shows a subsequent symptom decrease along with scar maturation processes (3, 4), a subgroup develops chronic itch and pain (1, 5) that seems localized within the scars (3, 6). Typically, prevalence rates of itch exceed those of pain [e.g. (7, 8)], indicating itch profiles co-occurring with and without pain. Because pain and itch intensity are highly correlated (9) and share common predictors, severity of both pain and itch may be linked. Examples are e.g., burn severity, particularly related to depth of the wound (1, 3, 10) and posttraumatic stress (PTS) symptoms (10–12). There is convincing evidence for an entangled relationship between chronic pain and PTS symptoms across many patient groups (13). Evidence for a connection between chronic itch and PTS symptoms has also been described (14), but far less studies are currently available compared to pain.
A neuropathic mechanism is assumed to underlie both pain and itch after burns. Neuropathic pain symptoms such as pins and needles, shooting, and burning pain have been described (15, 16), qualifying as spontaneous pain sensations (stimulus-independent) or paresthesia (e.g., burning pain, electric shocks) (17–19). Also itch is assumed neuropathic, particularly after the acute phase when the role of histamine and substance P have abated (20), subscribing that chronic itch seems mostly non-histaminergic (21). Both neuropathic pain and itch can develop after a lesion of the somatosensory system, with involvement of both peripheral and central processes (17, 22). Although peripheral nerve fibers may regenerate after burns, abnormal nerve fiber density in scars has been reported (23). In general, it is assumed that itch is predominantly peripherally activated because, as yet, central sensitization could not be established (6, 24).
Within a neuropathic pathology, also after burns, itch and pain temporally and spatially concur (9). But the underlying neuronal pathways are not fully understood. Current theories, e.g., the labeled line, selectivity, and pattern theory, propose that itch can result from various neuronal pathways, amongst which itch-specialized primary afferent neurons (pruriceptors) and nociceptors are involved (25, 26). Pruriceptors are assumed to transduce itch when being activated by specific molecular markers (e.g., IL-31) (26, 27). Nociceptors may respond to both algogens and pruritogens and are supposed to be differentially activated based on spatial (e.g., focal nociceptive input will produce itch) and temporal aspects of the peripheral input; hence the experienced pain or itch results from the combination of activated fibers (26, 28). Additionally, via inhibitory spinal interneurons, pain signals may inhibit itch transmission. However, in neuropathic itch, it has been put forward that the co-occurrence of itch and pain may result from impaired spinal inhibition, despite current inconclusive evidence (25, 28). Based on the current theories, one may argue that chronic itch and pain after burns may be related and explained by those various mechanisms. Therefore, identifying sensory profiles of itch and pain symptom and biological and psychological differences across the profiles may further elucidate underlying mechanisms.
This 18-months multi-center longitudinal study aims to document neuropathic pain prevalence as well as potential symptom profiles of itch and neuropathic pain that may present after burns, and to explore potential predictors, such as burn severity, age, gender and PTS symptoms potentially related to the symptom profiles.
This study was part of a larger longitudinal multi-center project examining pain after burn injuries. Previous papers about this project described pain measured with the Brief Pain Inventory (2, 12, 29) but did not focus on itch and neuropathic pain. Patients were included in the study between April 2010 and December 2012 from five burn centers in the Netherlands and in Belgium. Adult patients admitted to the burn centers for >24 h were eligible for inclusion in the study. Exclusion criteria included poor Dutch proficiency, acute or chronic cognitive problems, or when the injury was deliberate. Patients requiring mechanical ventilation were invited to participate as soon as they were able to provide informed consent. During the study period, 340 patients met the inclusion criteria of which 84 declined participation and 40 were missed. A group of 216 patients signed informed consent (64%). They did not differ from the 124 patients not included in the study in terms of age, gender, and affected body area [see also (12)].
Neuropathic pain and itch were measured using an adapted version of the self-report Leeds Assessment of Neuropathic Symptoms and Signs (LANSS) Pain Scale (30), a validated screening questionnaire for neuropathic pain (31). The original scale measures the presence (present yes/no) of five symptoms: unpleasant sensations (pricking, tingling, pin-pricks), color differences (motted and looking more red), abnormally sensitive to touch, pain comes suddenly and in bursts (electric shocks), perceived skin temperature (hot or burning). We added to the original version: (1) as part of the item perceived skin temperature, cold sensations were added (original scale only includes hot or burning), (2) a sixth item measuring itch, and (3) in the case that the participant scored “yes,” the intensity of the symptom was scored on a 7-point Likert scale ranging from 0 (not troublesome) to 6 (severely troublesome) which allows to measure the intensity of the symptom, and (4) items were adapted in order to measure pain related to the scars. e.g., “Does the pain feel like a strange and unpleasant sensation in your scars?,” and “Does the pain cause the scar to look different to normal skin or to that of scars that are not painful?.” Unlike the original LANSS, physical assessments of allodynia and altered pin-prick thresholds were not tested in this study because patients self-reported their symptoms. The scale was translated into Dutch by two researchers and back-translated by a native English speaker.
Posttraumatic stress (PTS) symptoms were measured using the Impact of Event Scale-Revised (IES-R) (32). The IES-R measures intrusive, avoidant and hyperarousal symptoms associated with a traumatic event. The original 15 items of the IES (33) and the seven hyperarousal items of the IES-R were used and scored with a 4-point scale (0-1-3-5). The construct validity and reliability of the Dutch version of the IES-R was acceptable (34). Cronbach's alpha was high (0.96). In this study, the 3-month measurement indicative of PTS symptoms rather than acute traumatic stress symptoms was used as a predictor.
Demographic characteristics (i.e., gender and age) and burn severity (i.e., percentage total body surface area (TBSA) burned as well as skin graft procedures) were recorded from the medical file. TBSA burned is the estimated percentage body surface area affected by partial and full-thickness burns.
The study was approved by an ethics committee in the Netherlands (METC Noord-Holland NL27996.094.09) and Belgium (Ghent University B670201112923) and by local institutional review boards of the participating hospitals, and was conducted in accordance with the Helsinki Declaration. Eligible patients were identified by local researchers during admission to the hospital. Oral and written information was provided. Written informed consent was obtained from each patient. Patients completed printed questionnaires in-hospital (e.g., psychological questionnaires), 2 weeks after discharge (T1), 3 months (T2), 6 months (T3), 12 months (T4), and 18 months (T5) after the burn event.
First, descriptive analyses were performed and patients with complete follow-up were compared with patients who had incomplete follow-up on burn characteristics and demographics using student t-tests. Second, itch and neuropathic pain profiles were examined. Two persons (NVL and AvL) independently categorized patients according to the duration of itch and pain [based on literature (3), we used 6 months as cutoff point for chronic itch post-burn which is associated with scar maturation, in contrast to 6 weeks akin the definition of chronic itch resulting from other causes (35)], and potential co-occurrence of neuropathic pain into the following groups: (1) patients reporting itch and/or pain that disappeared after 6 months (transient itch/pain); (2) patients reporting itch but never reported pain after 3 months postburn (because we can not exclude that patients may have had small wounds in the postacute phase) (chronic itch); (3) patients reporting itch and pain at least 2 out of 5 measurements of which at least once after 6 months (chronic itch & pain). Beyond the scope of this paper, other profiles included: (4) patients reporting only pain (chronic pain); (5) patients reporting no pain or itch (no pain/itch). Discrepancies in the categorization of patients were resolved by discussion.
Third, to examine potential differences in the course of itch intensity for the three itch profiles (independent variable), General Linear Modeling (GLM) for repeated measures was conducted with SPSS Statistics for Windows (Version 27.0. Armonk, NY: IBM Corp) with itch intensity over time (T1–T5) as the within-subjects dependent variable and the three itch profiles as independent variable. To investigate potential predictors of the three itch profiles, multi-nominal logistic regression analysis, which uses maximum likelihood estimation to evaluate the probability of categorical membership (of the three itch profiles), was used. Established predictors of both itch and neuropathic pain after burns (age, gender, TBSA burned, surgeries and PTS symptoms) controlling for T1 itch intensity were examined.
Informed consent was provided by 216 patients, but 24 did not complete any of the measurements leaving a final sample size of 192. At discharge (T1), 177 assessments (92%) were available, 166 (86%) at 3 months (T2), 155 (81%) at 6 months (T3), 152 (79%) at 12 months (T4) and 146 (76%) at 18 months (T5). The 146 patients who completed T5 were older [t(213) = −4.585, p < 0.001], and had higher TBSA burned [t(213) = −2.779, p = 0.006] and more surgeries [t(213) = −2.415, p = 0.017] compared to 46 patients lost to follow-up between T2 and T5.
Of the participants, 129 (67%) were male and 63 (33%) were female. Participants were on average 41.56 years old (SD = 15.58). TBSA burned ranged from 1 to 75% (M = 9.34, SD = 8.85). Ninety participants (46.9%) did not require surgery, 102 (53.1%) needed one or more skin graft procedures. The mean score indexing PTS symptoms was 21.46 (SD = 23.89) at 3 months post-burn.
The percentage of patients reporting itch decreased over time from 78% (T1) to 43% (T5). For pain, this was 54% (T1) and 24% (T5). Figure 1A presents the prevalence rates of itch and neuropathic pain symptoms (percentage reporting the symptom was present) at the respective time points for the total sample. Whereas itch prevalence rates decreased steadily, pain symptom prevalence remained relatively stable. Figure 1B presents the intensity of itch and pain symptoms measured using a 7-point Likert scale. The blue bars show that itch intensity in the total sample decreased over time, mainly due to the increasing number of patients in which itch disappeared. The orange bars show that itch intensity in the subgroup that also experienced neuropathic pain symptoms was higher on average and more stable. The varying number of patients for every symptom over time can be found in Supplementary Table 1, also presenting additional descriptive details such as mean, standard deviation and median.
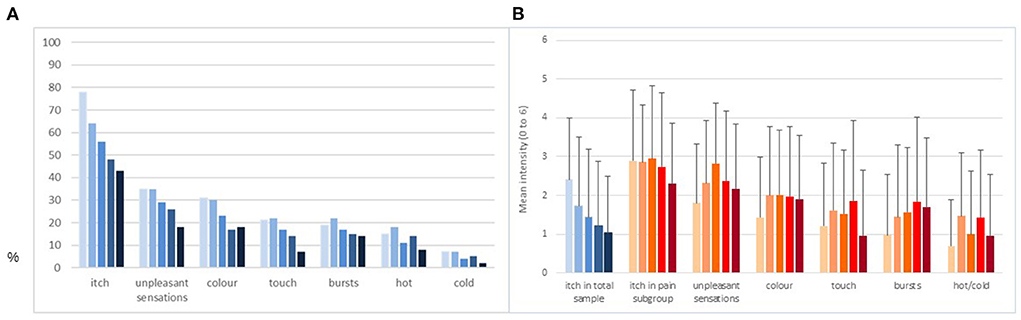
Figure 1. Prevalence rates and observed means of itch intensity and neuropathic pain symptom intensity in complete cohort. (A) Percentage of patients indicating the symptom was present. (B) Observed means of intensity and standard deviations of the symptoms scored on a 7-point Likert scale. Bars from left to right represent the five time points from 2 weeks post discharge (T1, n = 177), 3 (T2, n = 166), 6 (T3, n = 156), 12 (T4, n = 155), and 18 (T5, n = 146) months post-burn. Blue bars relate to the total sample. Orange bars relate to the subsample experiencing neuropathic pain symptoms.
Categorization into the different itch profiles was as follows: 51 patients (26.6%) reported transient itch/pain, 46 patients (24.0%) reported chronic itch, 41 patients (21.4%) reported chronic itch & pain, 7 patients (3.6%) reported chronic pain, 11 patients (5.7%) never reported pain or itch. Twenty-seven patients (14.0%) had no measurements after 6 months, or the symptom pattern could not be attributed to the aforementioned groups (n = 9; 4.6%). Table 1 presents the means and standard deviations of the predictor variables for the different profiles. The chronic itch & pain profile included most patients that needed surgery, had more PTS symptom and comprised more women.
GLM was used to study possible differences in itch intensity over time across the three itch profiles. Figure 2 shows that itch intensity was highest in the chronic itch & pain profile. The main effect of the profiles was significant, [F(2, 93) = 44.80, p < 0.001], as was the main linear effect of time, [F(1, 93) = 36.925, p < 0.001]. This suggests that both the three itch profiles and time explain variation in itch intensity and therefore are relevant to consider. The interaction of these two factors (i.e., the itch profiles and time) was also significant, [F(2, 93) = 3.409, p = 0.037]. This indicates that the profiles show different patterns of itch intensity over time. Figure 2 shows that patients reporting transient itch/pain showed, unsurprisingly, an early steep decline in itch intensity ultimately resulting in complete itch alleviation. Of more interest is the difference between the two chronic profiles, where in both profiles, itch intensity slightly decreased, but remained substantial, with higher itch intensity in the chronic itch & pain profile than in the chronic itch profile.
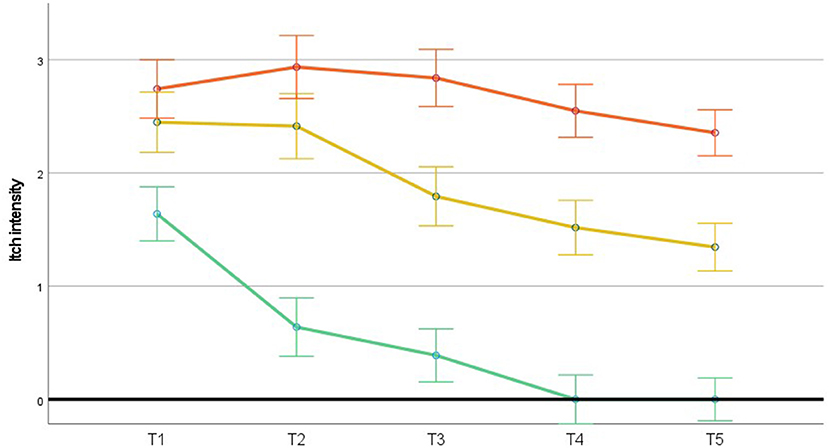
Figure 2. Time course of estimated means of itch intensity (ranging from 0 to 6) in the three itch profiles. N = 96 (full cases). Upper line (orange) = chronic itch & pain (n = 31), middle line (yellow) = chronic itch (n = 29), lower line (green) = transient itch/pain (n = 36). Error bars represent 1 standard deviation. T1 = 2 weeks post-discharge, T2 = 3 months post-burn, T3 = 6 months post-burn, T4 = 12 months post-burn and T5 = 18 months post-burn.
Using multi-nominal logistic regression analysis, we tested whether the three itch profiles were associated with differences regarding gender, burn severity (TBSA burned and needing surgery) age, PTS symptoms and itch intensity measured post-discharge. The fit between the model containing only the intercept and data improved with the inclusion of the predictor variables [χ2(12, n = 112) = 43.09, p < 0.001, Nagelkerke R2 = 0.279]. This indicates that inclusion of the predictors is meaningful and explains variance across the profiles. In the upper part of Table 2A the transient itch/pain profile was the reference group which means that the outcomes of the two chronic itch profiles were compared to the transient itch/pain profile. The results revealed that compared to transient itch/pain profile, younger patients (p = 0.045) and those with a larger TBSA burned (p = 0.034), and needing surgery (p = 0.060) were more likely to be assigned to the chronic itch profile. Needing surgery (p = 0.004) and higher levels of PTS symptoms (p = 0.024) increased the likelihood to be assigned to the chronic itch & pain profile. In the lower part of Table 2B, the chronic itch & pain profile was the reference category which allowed to investigate differences across the two chronic profiles. The results showed one statistically significant difference: PTS symptom levels were higher in the chronic itch & pain profile (p = 0.019).
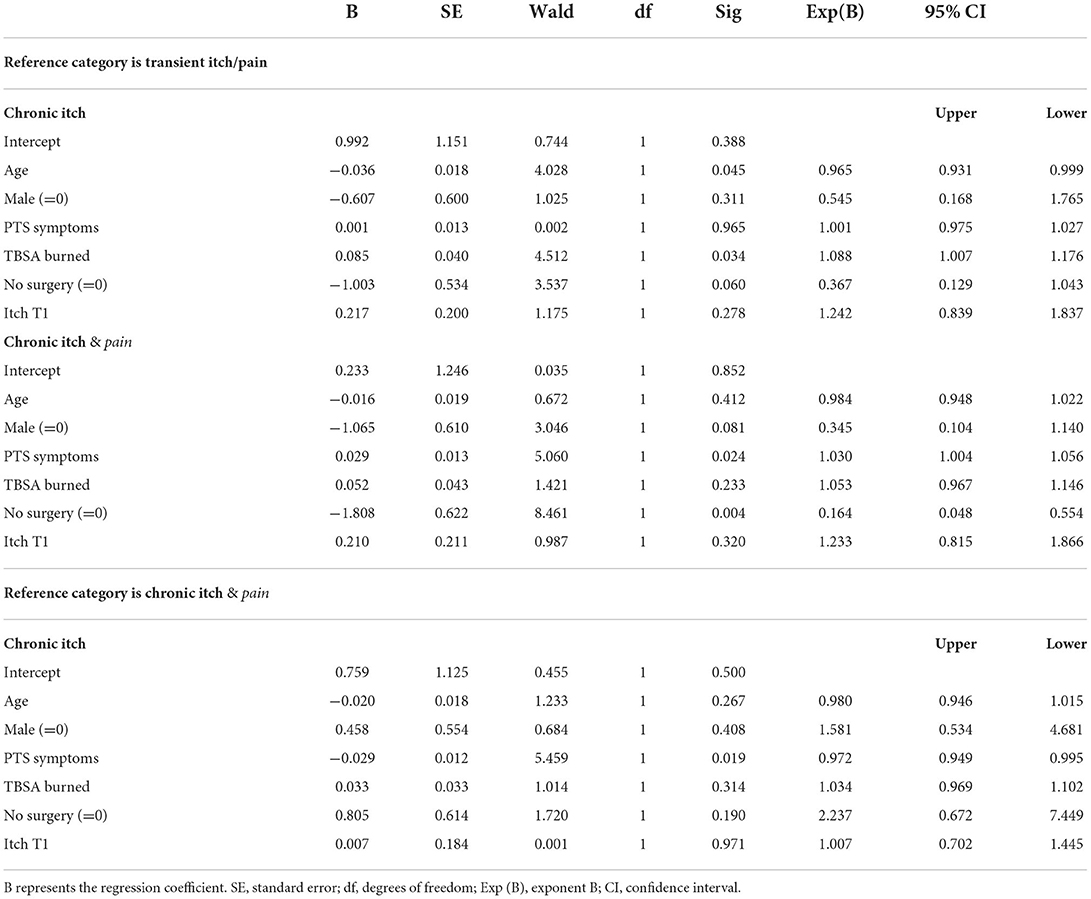
Table 2. Burn characteristics and demographics tested with multi-nominal regression analysis to predict classification into one of the three itch profiles.
This study prospectively documents itch and neuropathic pain symptom development and investigated itch profiles regarding duration and co-occurrence with neuropathic pain in adult burn survivors. Neuropathic pain symptoms were reported by 54% of the participants at 2 weeks post-discharge which declined to 24% at 18 months postburn. For itch, prevalence rates were 78% at 2 weeks postdischarge and 43% at 18 months postburn. Itch intensity was most severe in the chronic itch & pain profile. Compared to the transient itch/pain profile, both chronic profiles were associated with more severe burns, and the chronic itch & pain profile was associated with more PTS symptoms.
Patients' symptom profiles differ regarding co-occurrence with neuropathic pain, including transient itch/pain, chronic itch and chronic itch & pain. Itch intensity in the transient itch/pain profile showed a rapid decrease and the patients had less severe burns. This corroborates earlier findings that partial thickness burns more likely produce temporal itch (3) and may be predominantly histaminergic evoked in the early phase of wound healing streching out to the early remodeling phase in which antihistamines provide relief in a subgroup of patients (20).
Significantly higher itch intensities were perceived by the patients within the chronic itch & pain profile compared to those in the chronic itch profile. Provisional support for an association of mixed sensations and symptom severity may come from a study in which patients reporting both neuropathic pain and itch more likey needed both gabapentin and pregabalin compared to patients reporting itch only who received gabapentin to achieve symptom relief (36). Both chronic profiles were associated with more severe burns than the transient itch/pain profile. Particularly wounds that needed surgery, i.e., full thickness burns, may have affected skin innervation patterns. The newly regenerating nerve branches may evoke itch and/or pain due to spatial arrangement and spontaneous activity in regenerating sprouts and/or local inflammation (28). When only few epidermal nociceptors are focally activated and many are not, those may produce itch which is described as a “mismatch signal” (25, 37). Possibly, itch-specific pathways are involved in which mediators such as IL-31, pruriceptive neurons, and spinal neurons expressing gastrin-releasing peptide (GRP) play a role, although the latter may also apply to nociceptors (26). Increased levels of IL-31 have been identified in hypertrophic burn scars (38) but the involvement of GRP has not been investigated to our knowledge. What remains unclear is why the co-occurrence of pain and itch produces higher itch intensity. Although speculative, reduced descending inhibition may play a role. Future research may focus on different neuronal pathways in burn scars that may explain variation in itch intensity as well as temporal and spatial co-occurrence of itch and pain.
Higher PTS symptom levels were particularly associated with the chronic itch & pain profile. This is in line with studies showing a link between PTS symptoms and higher itch intensity (10, 39). Possibly, PTS symptoms affect central processing, potentially decreasing the threshold for pain, and perhaps also for itch. As shown in a study using electroencephalography (EEG) oscillatory activity, itch and pain seem processed differently in burn survivors with PTS symptoms compared to those without PTS symptoms (24). We could speculate that PTS symptoms may influence top-down sensory predictions, which play an important role in symptom perception (40). Due to the repeated peripheral somatosensory input (bottom-up), the brain has learnt to predict upcoming somatosensory sensations. This can result in the actual neurobiological perception of symptoms in the brain becoming aligned with the prediction via active interoceptive inference. Especially when sensory input is imprecise and in case of chronic symptoms, predictive processes are supposed to significantly modulate perception (40, 41). In this light, the threat resulting from a traumatic burn event and associated pain may form strong perceptual priors with a high probability, modifying later sensory perceptions, including pain and itch, corroborating that PTS symptoms amplify predictive coding processes (42). Another explanation may relate to increased production of peripheral inflammatory mediators. Elevated corticosteroids and alterations in cytokines related to psychological stress have been associated with slower wound healing (43) and an association between PTS symptoms and lower oxytocin levels in burn wounds have been found (44). This suggest that PTS symptoms can also exert an effect at skin-level through increased production of excitatory skin mediators, one of the mechanisms explaining neuropathic itch (28) and calls for more attention to identify and treat PTS symptoms.
A small effect of younger age was found associated with the categorization to the chronic itch profile, which corroborates earlier findings (1, 45) of which the authors explained the effect of age by neurological and vascular aging of the skin.
The neuropathic pain symptoms prevalence rate of 54% 2 weeks post-discharge was high compared to a study that reported 28% pain at 6 weeks post-burn (9), but 6 to 18 month prevalence rates were within the same range, be it 24% in the current study vs. 21% in the study of Mauck et al. However, it is substantially higher than the 6% prevalence rate (113/1,880 patients) reported in a retrospective chart review study (46). Likely, the prospective and systematic examination of pain symptoms explains the higher prevalence rates. In line with other studies, unpleasant sensations such as pin-pricks was the most frequently reported neuropathic pain symptom, e.g. (15). But also other symptoms such as bursts, sensitive touch, and burning pain were reported which overall indicates that pain symptoms in scars are of neuropathic origin.
This study has clinical and research implications. First, the itch profiles may inform clinical practice and future research into treatments. It is recommended to screen patients for sensory symptoms, specifically focused at the co-occurrence of itch and neuropathic pain, and other risk factors to tailor prescription of for instance gabapentin or pregabalin in an earlier stage (36). Second, more clinical attention to detect and treat PTS symptoms in an early phase is recommended as it may also improve pain and itch outcomes. Third, results call for further exploration of the involvement of different neuronal pathways and contribution of central sensitization processes in the various itch profiles, that may pave the way to conduct targeted medication clinical trials. For example, psychophysically assessing itch and pain modulation may predict course of symptoms and therapeutic (e.g., post-operative) outcomes for both chronic itch and pain (47, 48).
This study also has limitations. First, although the literature is positive about using self-report questionnaires to assess spontaneous pain-related sensations (31), allodynia, and loss of sensory function were not measured because clinical examination is required (49). Loss of sensory function or numbness has been documented in burn scars, indicating its relevance (50). Clinical assessments that document stimulus-evoked sensory sensations and temporal summation may inform underlying nerve damage and consequently, therapy (49). Second, the LANSS was modified, including the addition of a 7-point Likert scale, and warrants further validation along with clinical tests to establish its reliability and validity in burn scars. Third, the sample size of the different profiles was small which limits statistical power. Consequently, replication research is warranted. Additionally, the small sample size was deemed too small to use more sophisticated statistical analyses to explore latent classes (read: itch profiles), which could replace the classification of participants based on the duration of the complaints and itch-pain co-occurrence.
In conclusion, the current study shows that the co-occurrence of chronic itch and chronic neuropathic pain is associated with higher itch intensity compared to chronic itch only. This suggests different underlying mechanisms, perhaps related to different neuronal pathways or differences in modulation systems, but this should be considered as hypothesis generating. The role of PTS symptoms may point to altered central processing, which may be another pathway explaining higher itch intensity. Future research focussing on peripheral and central processing of itch from a bio-psychological perspective is warranted. This may ultimately inform pathophysiological and pharmacological mechanisms in future studies—and hence lead to better treatment and improved quality of life of individuals after burn injury.
The raw data supporting the conclusions of this article will be made available by the authors, upon reasonable request.
The studies involving human participants were reviewed and approved by METC Noord-Holland, Netherlands & Ghent University, Belgium. The patients/participants provided their written informed consent to participate in this study.
Conception and/or design of the study: NVL, AdJ, and HH. Interpretation of the data and drafted the work: NVL and AvL. Revision the paper: AdJ and HH. All authors approved the final version and agree to be accountable for all aspects of the work.
Funding was provided by Fonds NutsOhra Grant Numbers 0901-057 and 1101-035.
This study represents the collaborative efforts of the Dutch-Belgian psychosocial research group, consisting of M. Bremer, G. Bakker, A. Boekelaar, A. van de Steenoven, A.S. Goemanne, H. Hendrickx, and E. Vandermeulen, representing the burn centers in Groningen, Beverwijk, Rotterdam, Ghent, and Brussels.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2022.997183/full#supplementary-material
1. Carrougher GJ, Martinez EM, McMullen KS, Fauerbach JA, Holavanahalli RK, Herndon DN, et al. Pruritus in adult burn survivors: postburn prevalence and risk factors associated with increased intensity. J Burn Care Res. (2013) 34:94–101. doi: 10.1097/BCR.0b013e3182644c25
2. de Jong AE, Bremer M, Hofland HW, Schuurmans MJ, Middelkoop E, van Loey NE. The visual analogue thermometer and the graphic numeric rating scale: a comparison of self-report instruments for pain measurement in adults with burns. Burns. (2015) 41:333–40. doi: 10.1016/j.burns.2014.07.002
3. Kuipers HC, Bremer M, Braem L, Goemanne AS, Middelkoop E, van Loey NE. Itch in burn areas after skin transplantation: patient characteristics, influencing factors and therapy. Acta Derm Venereol. (2015) 95:451–6. doi: 10.2340/00015555-1960
4. van der Wal MB, Vloemans JF, Tuinebreijer WE, van de Ven P, van Unen E, van Zuijlen PP, et al. Outcome after burns: an observational study on burn scar maturation and predictors for severe scarring. Wound Repair Regen. (2012) 20:676–87. doi: 10.1111/j.1524-475X.2012.00820.x
6. van Laarhoven AI, Ulrich DJ, Wilder-Smith OH, van Loey NE, Nieuwenhuis M, van der Wee NJ, et al. psychophysiological processing of itch in patients with chronic post-burn itch: an exploratory study. Acta Derm Venereol. (2016) 96:613–8. doi: 10.2340/00015555-2323
7. Tracy LM, Edgar DW, Schrale R, Cleland H, Gabbe BJ, sites BAL-TOPPp, et al. Predictors of itch and pain in the 12 months following burn injury: results from the Burns Registry of Australia and New Zealand (BRANZ) Long-Term Outcomes Project. Burns Trauma. (2020) 8:tkz004. doi: 10.1093/burnst/tkz004
8. Mauck MC, Smith J, Shupp JW, Weaver MA, Liu A, Bortsov AV, et al. Pain and itch outcome trajectories differ among European American and African American survivors of major thermal burn injury. Pain. (2017) 158:2268–76. doi: 10.1097/j.pain.0000000000001029
9. Mauck MC, Smith J, Liu AY, Jones SW, Shupp JW, Villard MA, et al. chronic pain and itch are common, morbid sequelae among individuals who receive tissue autograft after major thermal burn injury. Clin J Pain. (2017) 33:627–34. doi: 10.1097/AJP.0000000000000446
10. Kwa KAA, Pijpe A, Rashaan ZM, Tuinebreijer WE, Breederveld RS, van Loey NE. Course and predictors of pruritus following burns: a multilevel analysis. Acta Derm Venereol. (2018) 98:636–40. doi: 10.2340/00015555-2935
11. Prasad A, Thode HC, Jr., Sandoval S, Singer AJ. The association of patient and burn characteristics with itching and pain severity. Burns. (2019) 45:348–53. doi: 10.1016/j.burns.2018.06.011
12. Van Loey NE, Klein-Konig I, de Jong AEE, Hofland HWC, Vandermeulen E, Engelhard IM. Catastrophizing, pain and traumatic stress symptoms following burns: A prospective study. Eur J Pain. (2018) 22:1151–9. doi: 10.1002/ejp.1203
13. Ravn SL, Hartvigsen J, Hansen M, Sterling M, Andersen TE. Do post-traumatic pain and post-traumatic stress symptomatology mutually maintain each other? A systematic review of cross-lagged studies. Pain. (2018) 159:2159–69. doi: 10.1097/j.pain.0000000000001331
14. Gupta MA. Somatization disorders in dermatology. Int Rev Psychiatry. (2006) 18:41–7. doi: 10.1080/09540260500466832
15. Schneider JC, Harris NL, El Shami A, Sheridan RL, Schulz JT IIIrd, Bilodeau ML, et al. A descriptive review of neuropathic-like pain after burn injury. J Burn Care Res. (2006) 27:524–8. doi: 10.1097/01.BCR.0000226019.76946.5D
16. Kwak IS, Park SY, Choi YH, Cho SI, Yang YS, Cho YS, et al. Clinical and histopathological features of post burn pruritus. J Burn Care Res. (2016) 37:343–9. doi: 10.1097/BCR.0000000000000392
17. Stumpf A, Stander S. Neuropathic itch: diagnosis and management. Dermatol Ther. (2013) 26:104–9. doi: 10.1111/dth.12028
18. Truini A, Garcia-Larrea L, Cruccu G. Reappraising neuropathic pain in humans–how symptoms help disclose mechanisms. Nat Rev Neurol. (2013) 9:572–82. doi: 10.1038/nrneurol.2013.180
19. Attal N. Neuropathic pain: mechanisms, therapeutic approach, and interpretation of clinical trials. Continuum. (2012) 18:161–75. doi: 10.1212/01.CON.0000411564.41709.2d
20. Goutos I. Neuropathic mechanisms in the pathophysiology of burns pruritus: redefining directions for therapy and research. J Burn Care Res. (2013) 34:82–93. doi: 10.1097/BCR.0b013e3182644c44
21. Yosipovitch G, Rosen JD, Hashimoto T. Itch: From mechanism to (novel) therapeutic approaches. J Allergy Clin Immunol. (2018) 142:1375–90. doi: 10.1016/j.jaci.2018.09.005
22. Colloca L, Ludman T, Bouhassira D, Baron R, Dickenson AH, Yarnitsky D, et al. Neuropathic pain. Nat Rev Dis Primers. (2017) 3:17002. doi: 10.1038/nrdp.2017.2
23. Anderson JR, Zorbas JS, Phillips JK, Harrison JL, Dawson LF, Bolt SE, et al. Systemic decreases in cutaneous innervation after burn injury. J Invest Dermatol. (2010) 130:1948–51. doi: 10.1038/jid.2010.47
24. Millard SK, Bokelmann K, Schalbroek R, van der Wee NJA, Van Loey NEE, van Laarhoven AIM. No indications for altered EEG oscillatory activity in patients with chronic post-burn itch compared to healthy controls. Sci Rep. (2022) 12:5184. doi: 10.1038/s41598-022-08742-8
25. Steinhoff M, Oaklander AL, Szabo IL, Stander S, Schmelz M. Neuropathic itch. Pain. (2019) 160 Suppl 1:S11–S6. doi: 10.1097/j.pain.0000000000001551
26. Carstens E, Carstens MI, Follansbee T. Coding of itch and pain: neurophysiological parallels and differences. In:Yosipovitch G, Andersen HH, Arendt-Nielsen L, , editors. Itch and Pain: Similarities, Interactions, and Differences. Philadelphia, PA: Wolters Kluwer (2020). p. 32–44.
27. Kunimura K, Fukui Y. The molecular basis for IL-31 production and IL-31-mediated itch transmission: from biology to drug development. Int Immunol. (2021) 33:731–6. doi: 10.1093/intimm/dxab065
29. de Vries V, de Jong AEE, Hofland HWC, Van Loey NE. Pain and posttraumatic stress symptom clusters: a cross-lagged study. Front Psychol. (2021) 12:669231. doi: 10.3389/fpsyg.2021.669231
30. Bennett M. The LANSS Pain Scale: the Leeds assessment of neuropathic symptoms and signs. Pain. (2001) 92:147–57. doi: 10.1016/S0304-3959(00)00482-6
31. Attal N, Bouhassira D, Baron R. Diagnosis and assessment of neuropathic pain through questionnaires. Lancet Neurol. (2018) 17:456–66. doi: 10.1016/S1474-4422(18)30071-1
32. Weiss DS, Marmar CR. The impact of event scale—Revised. In:Wilson JP, Keane TM, , editors. Assessing Psychological Trauma and PTSD. New York, NY: Guilford Press (1997). p. 399–411.
33. Horowitz M, Wilner N, Alvarez W. Impact of event scale: a measure of subjective stress. Psychosom Med. (1979) 41:209–18. doi: 10.1097/00006842-197905000-00004
34. Olde E, Kleber R, van der Hart O, Pop V. Childbirth and posttraumatic stress responses: a validation study of the Dutch impact of event scale – revised. European J Psychol Assess. (2006) 22:259–67. doi: 10.1027/1015-5759.22.4.259
36. Kaul I, Amin A, Rosenberg M, Rosenberg L, Meyer WJ, 3rd. Use of gabapentin and pregabalin for pruritus and neuropathic pain associated with major burn injury: a retrospective chart review. Burns. (2018). 44:414–22. doi: 10.1016/j.burns.2017.07.018
37. Schmelz M. Itch and pain. Neurosci Biobehav Rev. (2010) 34:171–6. doi: 10.1016/j.neubiorev.2008.12.004
38. Lee MY, Shin E, Kim H, Kwak IS, Choi Y. Interleukin-31, interleukin-31RA, and OSMR expression levels in post-burn hypertrophic scars. J Pathol Transl Med. (2018) 52:307–13. doi: 10.4132/jptm.2018.08.03
39. Van Loey NE, Bremer M, Faber AW, Middelkoop E, Nieuwenhuis MK. Itching following burns: epidemiology and predictors. Br J Dermatol. (2008) 158:95–100. doi: 10.1111/j.1365-2133.2007.08278.x
40. Buchel C, Geuter S, Sprenger C, Eippert F. Placebo analgesia: a predictive coding perspective. Neuron. (2014) 81:1223–39. doi: 10.1016/j.neuron.2014.02.042
41. Kaptchuk TJ, Hemond CC, Miller FG. Placebos in chronic pain: evidence, theory, ethics, and use in clinical practice. BMJ. (2020) 370:m1668. doi: 10.1136/bmj.m1668
42. Kube T, Berg M, Kleim B, Herzog P. Rethinking post-traumatic stress disorder - A predictive processing perspective. Neurosci Biobehav Rev. (2020) 113:448–60. doi: 10.1016/j.neubiorev.2020.04.014
43. Gouin JP, Kiecolt-Glaser JK. The impact of psychological stress on wound healing: methods and mechanisms. Immunol Allergy Clin North Am. (2011) 31:81–93. doi: 10.1016/j.iac.2010.09.010
44. Van Loey NE, Hofland HWC, Vlig M, Vandermeulen E, Rose T, Beelen RHJ, et al. Associations between traumatic stress symptoms, pain and bio-active components in burn wounds. Psychoneuroendocrinology. (2018) 96:1–5. doi: 10.1016/j.psyneuen.2018.05.037
45. Casaer M, Kums V, Wouters PJ, Van den kerckhove E, Van den Berghe G. Pruritus in patients with small burn injuries. Burns. (2008) 34:185–91. doi: 10.1016/j.burns.2007.03.004
46. Klifto KM, Dellon AL, Hultman CS. Prevalence and associated predictors for patients developing chronic neuropathic pain following burns. Burns Trauma. (2020) 8:tkaa011. doi: 10.1093/burnst/tkaa011
47. Petersen KK, McPhee ME, Hoegh MS, Graven-Nielsen T. Assessment of conditioned pain modulation in healthy participants and patients with chronic pain: manifestations and implications for pain progression. Curr Opin Support Palliat Care. (2019) 13:99–106. doi: 10.1097/SPC.0000000000000419
48. Ramon M, Yakov S, Kuperman P, Granot M. The role of itch and pain modulation in the prediction of phototherapy outcomes: a prospective cohort study. J Dermatolog Treat. (2020) 33:1037–41. doi: 10.1080/09546634.2020.1800572
49. Baron R, Forster M, Binder A. Subgrouping of patients with neuropathic pain according to pain-related sensory abnormalities: a first step to a stratified treatment approach. Lancet Neurol. (2012) 11:999–1005. doi: 10.1016/S1474-4422(12)70189-8
Keywords: pruritus, neuropathic pain, scars, posttraumatic stress symptoms, burns
Citation: Van Loey NEE, de Jong AEE, Hofland HWC and van Laarhoven AIM (2022) Role of burn severity and posttraumatic stress symptoms in the co-occurrence of itch and neuropathic pain after burns: A longitudinal study. Front. Med. 9:997183. doi: 10.3389/fmed.2022.997183
Received: 18 July 2022; Accepted: 20 September 2022;
Published: 12 October 2022.
Edited by:
Manuel Pedro Pereira, University Hospital Münster, GermanyReviewed by:
Emilie Brenaut, Centre Hospitalier Regional Universitaire (CHU) de Brest, FranceCopyright © 2022 Van Loey, de Jong, Hofland and van Laarhoven. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: N. E. E. Van Loey, TG9leU4yQG1hYXNzdGFkemlla2VuaHVpcy5ubA==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.