
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Med., 15 November 2022
Sec. Nuclear Medicine
Volume 9 - 2022 | https://doi.org/10.3389/fmed.2022.1030094
This article is part of the Research TopicTargeted Alpha Particle Therapy in OncologyView all 17 articles
Osteosarcoma is a high-grade sarcoma characterized by osteoid formation, nearly universal expression of IGF1R and with a subset expressing HER-2. These qualities provide opportunities for the use of the alpha particle-emitting isotopes to provide targeted radiation therapy via alpha particles precisely to bone-forming tumors in addition to IFG1R or Her-2 expressing metastases. This review will detail experience using the alpha emitter radium-223 (223Ra, tradename Xofigo), that targets bone formation, in osteosarcoma, specifically related to patient selection, use of gemcitabine for radio-sensitization, and using denosumab to increasing the osteoblastic phenotype of these cancers. A case of an inoperable left upper lobe vertebral-paraspinal-mediastinal osteoblastic lesion treated successfully with 223Ra combined with gemcitabine is described. Because not all areas of osteosarcoma lesions are osteoblastic, but nearly all osteosarcoma cells overexpress IGF1R, and some subsets expressing Her-2, the anti-IGF1R antibody FPI-1434 linked to actinium-225 (225Ac) or the Her-2 antibody linked to thorium-227 (227Th) may become other means to provide targeted alpha particle therapy against osteosarcoma (NCT03746431 and NCT04147819).
Pathologic diagnosis of osteosarcoma requires the demonstration of bone formation in the form of osteoid production (1). Despite accurate pathologic diagnosis, genomic instability has resulted in osteosarcomas having heterogeneous molecular signatures, with a relative paucity of actionable molecular targets. Many osteosarcoma tumors and metastases harbor p53 mutations or other mechanisms (e.g., MDM2 amplification) that interfere with apoptosis after damage from standard chemotherapy, newer agents such as tyrosine kinase inhibitors (TKI) of vascular endothelial growth factor (VEGF) (2, 3), and/or radiation therapy (4, 5).
Although osteosarcoma has long been considered relatively radio-resistant (6), this assessment was in the pre-chemotherapy era; radiotherapy has been shown to be more effective against osteosarcoma when given in combination with chemotherapy (5, 7–10) or using proton radiotherapy (11). Another approach that is more biologically effective for bone metastases than conventional low dose fractionated radiation to enhance radiation effectiveness is stereotactic body radiotherapy (SBRT) which delivers precise high dose fractions (12–17). The high Linear Energy Transfer (LET) of alpha particles emitted by 223Ra, 225Ac, or 227Th causes hard to repair double strand breaks, providing another way to potentially overcome the intrinsic biologic resistance of osteosarcoma to radiotherapy (18–20).
The importance of local control measures, especially surgery was shown in a series by Jaffe (21). Current osteosarcoma protocols use variations of the 3-drug (Methotrexate Adriamycin, Platinum, MAP) or 5-drug (MAP + Ifosfamide/etoposide, MAPIE) chemotherapy similar to that reported by the Euramos-1 study (22, 23). The addition of Mifamurtide may also improve outcomes (24–27). Metastatic disease, age > 18 (28) and poor response to neoadjuvant chemotherapy are associated with worse prognosis that to date we have not been able to effectively overcome (29, 30).
Ifosfamide is clearly an active drug in osteosarcoma as shown by its effectiveness against bone metastases and responses in patients not responding to MAP (31). Ifosfamide/mesna can be given with reduced toxicity and improved quality of life when given as an outpatient (32–37). If surgery is not possible or would have an unacceptable effect on the quality of life after response to ifosfamide/mesna, then use of not only radiotherapy with radio-sensitizers (10), but also alpha emitting radiopharmaceuticals such as 223Ra can provide options for local and systemic control (12, 17).
An osteoblastic phenotype is often suspected when calcified osteosarcoma metastases are seen on scans. However, active bone formation for the metastases >1 cm should be demonstrated using 99mTcMDP bone scan or 18FNa bone PET-CT before contemplating use of 223Ra in osteosarcoma (12, 17). Better images are obtained when planar images are combined with CT (SPECT-CT). 18FNa bone PET-CT has increased sensitivity toward osteoblastic metastases and, because a standard uptake value can be obtained on individual metastases, 18FNa bone PET-CT also provides a semi-quantitative assessment of disease burden that can be followed to measure the treatment response (38–46). Radiation is excellent if delivered precisely to tumors avoiding normal tissue. Thus, if there is avid 99mTc-MDP (47) and/or 18FNa uptake in osteosarcoma metastases or a local recurrence, then the patient is excellent candidate for the use of 223Ra to deliver alpha particle radiation to osteoblastic osteosarcoma tumors and minimal radiation to the surrounding normal tissues, be it adjacent lung, spine, or limb salvage hardware from prior surgeries. If little or no bone formation is seen on these imaging modalities, then the patient is not a good candidate for 223Ra.
We have given 223Ra in osteosarcoma using the standard dose and monthly infusion schedule of 1.49 microCi/kg intravenously monthly (12) and at 50, 75, and 100 kBq/kg in a dose escalation study (48). From the perspective of the patient, getting 223Ra is relatively simple: there is a discussion of the minimal radiation safety requirements (wash hands, flush toilet 2 × because unbound 223Ra comes out in the stool), and in our Nuclear Medicine Departments getting 223Ra is similar to getting a bone scan injection and takes approximately 10 min. Our current practice is to use the standard 223Ra dose on a Wednesday or Thursday to allow gemcitabine to be given as a radio-sensitizer the following day. We also use 223Ra in combination with other agents such as denosumab and local control measures in an attempt to both improve the efficacy of 223Ra and also to treat areas of metastases that do not have 223Ra deposition as illustrated in Figure 1.
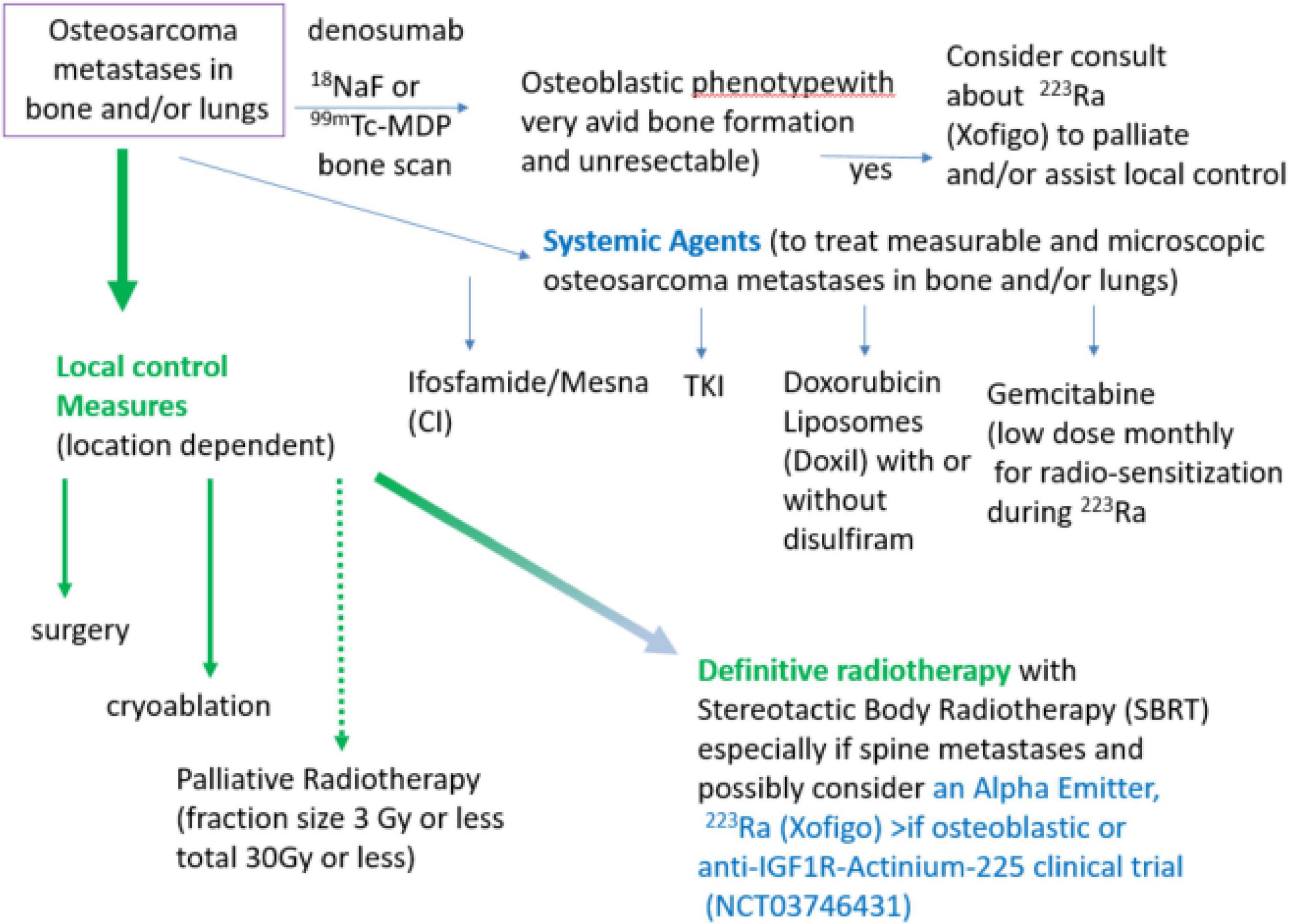
Figure 1. Algorithm of treatment of metastatic, recurrent osteosarcoma with systemic agents, local control measures, and with alpha-emitters 223Ra or Anti-IGF1R-Actinium-225. Note if unresectable, referral to a center with expertise in administration of alpha emitters in combination with radiotherapy will be needed.
Denosumab is a fully humanized anti-RANKL antibody that improves bone density. It is used to treat osteoporosis, reduce skeletal complications of bone metastases, and treat giant cell tumor of bone (49–53). We have made the observation that some osteosarcomas increase the amount of bone formation after denosumab. Thus, monthly denosumab injections during 223Ra therapy can increase the amount of 223Ra deposited in osteoblastic metastases in osteosarcoma (12).
Gemcitabine is an excellent radio-sensitizer (10, 54–59). The toxicity of gemcitabine is dependent on not only schedule and dose, but also infusion duration. Shorter infusions (30 min) are associated with less hematologic toxicity than 90 min infusions. Gemcitabine is given daily 5 × had unacceptable mucosal toxicity. Weekly or day 1 and 8 of 3-week cycles are better tolerated. Since gemcitabine must be taken up and phosphorylated to act on the cancer cell, longer infusion times are associated with more hematologic toxicity (60, 61). Giving gemcitabine 600 mg/m2 intravenously (iv) once over 30 min 1 day after 223Ra is deposited in osteoblastic tumors is a convenient monthly strategy that allows gemcitabine to increase effects within osteoblastic metastases with minimal hematologic toxicity.
The following case (Figure 2) illustrates the successful use of 223Ra and gemcitabine (62). A 27-year-old patient presented with a large osteosarcoma tumor involving T2-4 extending into both the spinal canal and the left upper lobe. Because of giant cell features, he was initially given denosumab, but when molecular testing revealed FGFR mutation and pathology was reviewed, the diagnosis of osteosarcoma was made. He received 2 cycles of MAP chemotherapy and then because of minimal response was switched to ifosfamide + etoposide. Because the tumor was deemed unresectable, 50.4 Gy over 28 fractions with concurrent ifosfamide + etoposide was given during cycles 5 and 6. He received 2 more cycles of ifosfamide + etoposide then had radiographic progression and clinical worsening (weakness of both lower extremities, some tingling, and need to use a cane). Cardiothoracic, orthopedic, and spine surgeons reviewed his case at the sarcoma conference at the Cleveland Clinic and also deemed the tumor to be unresectable because of the combination of vertebral, spinal canal, and mediastinal involvement. Bone scan with Spect-CT showed avid 99mTc-MDP uptake and he was given 6 monthly cycles of Denosumab, 223Ra, followed by gemcitabine. Cytopenias were modest, no transfusions were needed. The patient experienced a clinical response as characterized by increased strength in his legs, no longer requiring a cane to ambulate and resolution of paresthesia. Uptake of 18FDG as well and 99mTc-MDP was decreased on repeat imaging. After the response to 223Ra monthly 6 ×, he was given oral cyclophosphamide for 6 months (Figure 2). He is now over 9 months off therapy without evidence of recurrence. His activity level has increased and he is able to skateboard (even able to do tricks such as a “treflip,” insert top on Figure 2; Supplementary Video 1), rock climbs often, and has gone skydiving six times.
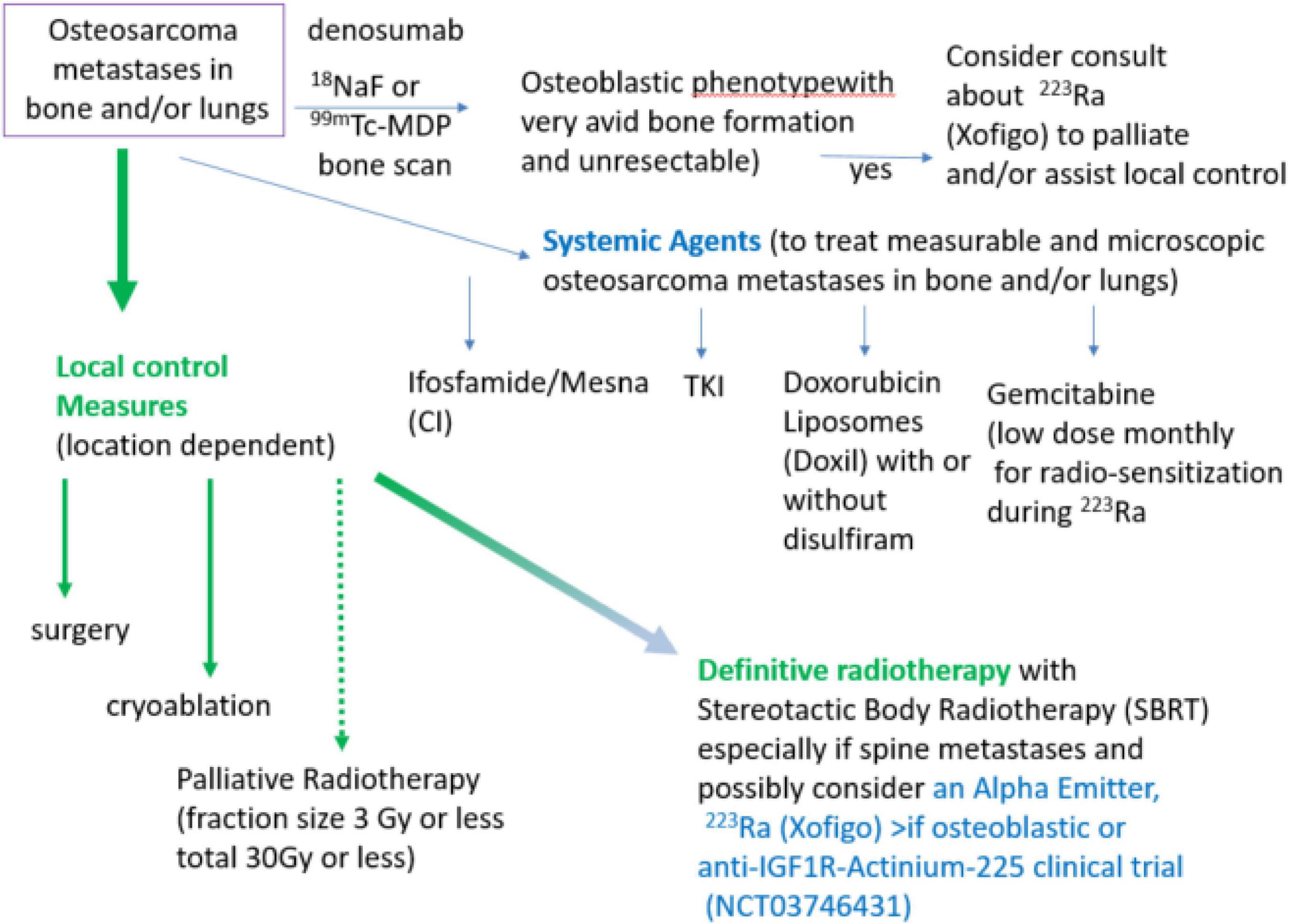
Figure 2. Top shows 99mTc-MDP screening bone scan with avid uptake indicating suitability for alpha particle therapy with the bone-seeking radiopharmaceutical, 223RaCl2 (Xofigo). 223Ra (blue), then gemcitabine (green) next day + monthly denosumab 6 × monthly cycles resulted in improvement, then resolution of symptoms of the unresectable mediastinal and vertebral body osteosarcoma. Currently, the patient is on no therapy and enjoys active lifestyles, such as skateboarding, rock climbing, and skydiving (62).
Local and systemic therapy is often needed before the logistics of evaluating osteoblastic phenotype and obtaining 223Ra for osteosarcoma treatment can be solved. Some active agents in the relapsed metastatic osteosarcoma setting are illustrated in Figure 1 (ifosfamide, TKI, and doxorubicin liposomes). Since many patients have had MAP initially without ifosfamide, ifosfamide with or without etoposide is often the 2nd line therapy of choice (31–34). When ifosfamide is given with mesna as a continuous infusion, thrombocytopenia, encephalopathy, and renal toxicity are seen less often (33–37, 63). We have also demonstrated that outpatient continuous infusion of ifosfamide + mesna was associated with fewer transfusions and episodes of fever and neutropenia (63). If continuous infusion of ifosfamide + mesna is to be used with 223Ra, we would recommend starting 1 day after 223Ra administration using a dose of 1 gm/m2/d × 1 week. Administration of PEG–GCSF after completion of the ifosfamide infusion is also recommended. This regimen can be repeated every 4 weeks to allow for the combination of the cytotoxic effects when the bone-seeking radiopharmaceutical is most active in bone-forming lesions and to allow for hematologic recovery as 223Ra decays.
Tyrosine kinase inhibitors including regorafenib (2, 64) and cabozantinib (3) have efficacy against osteosarcoma. Although a dose adjustment of TKI is sometimes needed to limit skin or GI toxicity, we have found the use of glutamine-disaccharide (Healios) can be helpful in ameliorating GI side effects and helping in eating and nutrition while on these agents (35).
Liposomal doxorubicin (tradenames Doxil or Caelyx) has very low heart toxicity (65, 66). This preparation can be given monthly and has modest hematologic toxicity and is not associated with alopecia when given at 40 mg/m2. Thus liposomal doxorubicin has high patient acceptance among relapsed osteosarcoma patients. Cold packs on hands, feet, and the use of glutamine + disaccharide (Healios) can be used to limit hand/foot erythroderma and mucositis/esophagitis, respectively (35). Liposomal doxorubicin is probably most suitable in relapsed osteosarcoma patients who had an initial excellent response to MAP chemotherapy. There is also a clinical trial using liposomal doxorubicin in combination with disulfiram to try to target slowly repopulating cancer stem cells high in aldehyde dehydrogenase (67, 68) (NCT05210374 M. Trucco, PI).
As illustrated in Figure 1, surgery, cryoablation, and/or radiation can provide local control of osteosarcoma metastases. Location and number of metastases (“oligometastatic” is <10) may determine whether to do surgery, cryoablation, or to definitively treat with radiation (e.g., 3 Gy × 20 fractions RT or SBRT 8 Gy × 5 fractions = 40 Gy) or whether palliative radiation (e.g., 3 Gy × 10 fractions) is most appropriate. Reasons to use local control include treatment or prevention of pain as well as reduction of tumor burden, particularly where tumor growth may cause complications (e.g., spine or sacral metastases, and hilar or mediastinal metastases. Unfortunately, for the most common pattern of end-stage metastases (numerous lung metastases) neither whole lung radiation nor 223Ra will provide effective doses. Clinical trials such as anti-IGF1R–Actinium 225 (NCT03746431) or Doxil + disulfiram (NCT05210374) would be appropriate in these situations.
Sarcomas, particularly Ewing sarcoma and osteosarcoma have overexpression of IGF1R (69). Although cold antibody was only modestly effective in Ewing sarcoma and not in osteosarcoma (69), chelation of the alpha emitter 225Ac can arm the anti-IGF1R antibody to become a potent alpha emitter (70, 71). Table 1 compares 223Ra, which targets areas of bone turnover, with anti-IGF1R-Actinium-225. Currently, the clinical trial NCT0374631 is open at MD Anderson Cancer Center, City of Hope, Memorial Sloan Kettering, University of Minnesota, Dana Farber Cancer Institute, University of Pennsylvania, Juravinski/Hamilton Health, CHU-Montreal, Princess Margaret (Toronto), and CHU Quebec. We expect patients <18 years old to be able to be enrolled when the recommended phase 2 dose is achieved. Thus, the anti-IGF1R-Actinium-225 strategy may be another way to treat osteosarcoma metastases that are not osteoblastic and with alpha-particle radiation that effectively acts at short distances in a powerful manner. Nevertheless, the expression of IGF-1R in normal tissue and/or non-specific binding of antibodies may limit the effectiveness of this approach.
Her-2 is expressed in a subset of osteosarcomas. Earlier attempts to target this Her2 expression using trastuzumab were unsuccessful. However, clinical trials using Her2-targeted CAR T-cells suggest that Her2-targeted therapy could be active in osteosarcoma (72). Moreover, better-designed novel antibody drug conjugates like Trastuzumab-Deruxtecan (T-DXd) is showing activity in low Her-2 expressing breast cancers, are also being explored in osteosarcoma. HER2-thorium-227 targeted conjugate (TTC) has recently entered clinical trials in Europe and the USA. “A First in Human Study of BAY2701439 to Look at Safety, How the Body Absorbs, Distributes, and Excretes the Drug, and How Well the Drug Works in Participants With Advanced Cancer Expressing the HER2 Protein” (NCT04147819) is a combination of the alpha-emitting radionuclide thorium-227, an antibody targeting HER2, and a chelator molecule that strongly attaches the thorium-227 to the antibody. This technology harnesses the antibody’s ability to target HER2 by using it to transport the alpha particle emitting thorium-227 to the tumor. Both radium-223 and thorium-227 decay produce alpha particle radiation (Table 1) that causes highly lethal double strand DNA damage in tumor cells, but also useful emission for gamma scintigraphy (73). Although the first in human trial is open for breast and gastric only, the expansion part of the study will include patients with a range of tumor indications with HER2 expression which occurs on osteosarcoma. Only in the context of a clinical trial will it be possible to determine whether benefits for the binding to HER-2 on osteosarcoma outweigh potential toxicity from expression on normal cells and/or non-specific binding of the alpha emitter.
Alpha emitters have some potent biological advantages that may eventually prove useful for the treatment of osteosarcoma. However, the rarity of this sarcoma and specific situations to test efficacy in randomized clinical trials will be very difficult. Perhaps the use of patients as their own controls with benefit as improved quality of life and/or clinical course better than expected—especially compared to historical controls (74) is possibly the best we can do currently.
PA: writing of the manuscript and experience with 223Ra combination therapy. VS: editing of manuscript and experience with 223Ra, anti-IGF1R-Ac225 antibody, and Her-2 TTC. MT: editing of manuscript and current treatment of relapsed osteosarcoma including doxorubicin liposomes with or without disulfiram. All authors contributed to the article and approved the submitted version.
This work was funded by the Cleveland Clinic Sarcoma program. VS was an Andrew Sabin Family Foundation Fellow at The University of Texas MD Anderson Cancer Center. VS acknowledged the support of the Jacquelyn A. Brady Fund. VS was supported by the US National Institutes of Health (NIH) (Grant nos. R01CA242845 and R01CA273168), the MD Anderson Cancer Center Department of Investigational Cancer Therapeutics is supported by the Cancer Prevention and Research Institute of Texas (Grant no. RP1100584), the Sheikh Khalifa Bin Zayed Al Nahyan Institute for Personalized Cancer Therapy (Grant no. 1U01 CA180964), the NCATS (Center for Clinical and Translational Sciences) (Grant no. UL1 TR000371), and the MD Anderson Cancer Center Support (Grant no. P30 CA016672).
The efforts of physicians and physicists in the Departments of Nuclear Medicine at MD Anderson and Cleveland Clinic area acknowledged. The efforts of the C level and Clinic Pediatric Hematology/Oncology infusion nurses and care coordination by Shauna Sartoski, RN for 223RaCl2 (Xofigo) and chemotherapy infusions have been much appreciated.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2022.1030094/full#supplementary-material
Supplementary video 1 | Excellent return of lower extremity function after 223-Ra treatment of thoracic paraspinal osteosarcoma as demonstrated by skateboarding.
1. Rivera-Valentin RK, Zhu L, Hughes DP. Bone sarcomas in pediatrics: progress in our understanding of tumor biology and implications for therapy. Paediatr Drugs. (2015) 17:257–71. doi: 10.1007/s40272-015-0134-4
2. Duffaud F, Mir O, Boudou-Rouquette P, Piperno-Neumann S, Penel N, Bompas E, et al. Efficacy and safety of regorafenib in adult patients with metastatic osteosarcoma: a non-comparative, randomised, double-blind, placebo-controlled, phase 2 study. Lancet Oncol. (2019) 20:120–33. doi: 10.1016/S1470-2045(18)30742-3
3. Italiano A, Mir O, Mathoulin-Pelissier S, Penel N, Piperno-Neumann S, Bompas E, et al. Cabozantinib in patients with advanced Ewing sarcoma or osteosarcoma (CABONE): a multicentre, single-arm, phase 2 trial. Lancet Oncol. (2020) 21:446–55. doi: 10.1016/S1470-2045(19)30825-3
4. Prudowsky ZD, Yustein JT. Recent insights into therapy resistance in osteosarcoma. Cancers. (2020) 13:83. doi: 10.3390/cancers13010083
5. Hernberg MM, Kivioja AH, Böhling TO, Janes RJ, Wiklund TA. Chemoradiotherapy in the treatment of inoperable high-grade osteosarcoma. Med Oncol. (2011) 28:1475–80. doi: 10.1007/s12032-010-9592-2
6. Dahlin DC. Prognostic factors in osteosarcoma. Int J Radiat Oncol Biol Phys. (1980) 6:1755. doi: 10.1016/0360-3016(80)90266-7
7. Machak GN, Tkachev SI, Solovyev YN, Sinyukov PA, Ivanov SM, Kochergina NV, et al. Neoadjuvant chemotherapy and local radiotherapy for high-grade osteosarcoma of the extremities. Mayo Clin Proc. (2003) 78:147–55. doi: 10.4065/78.2.147
8. Anderson PM. Effectiveness of radiotherapy for osteosarcoma that responds to chemotherapy. Mayo Clin Proc. (2003) 78:145–6. doi: 10.4065/78.2.145
9. Mahajan A, Woo SY, Kornguth DG, Hughes D, Huh W, Chang EL, et al. Multimodality treatment of osteosarcoma: radiation in a high-risk cohort. Pediatr Blood Cancer. (2008) 50:976–82. doi: 10.1002/pbc.21451
10. Anderson P, Aguilera D, Pearson M, Woo S. Outpatient chemotherapy plus radiotherapy in sarcomas: improving cancer control with radiosensitizing agents. Cancer Control. (2008) 15:38–46. doi: 10.1177/107327480801500105
11. Patel S, DeLaney TF. Advanced-technology radiation therapy for bone sarcomas. Cancer Control. (2008) 15:21–37. doi: 10.1177/107327480801500104
12. Anderson PM, Scott J, Parsai S, Zahler S, Worley S, Shrikanthan S, et al. 223-Radium for metastatic osteosarcoma: combination therapy with other agents and external beam radiotherapy. ESMO Open. (2020) 5:e000635. doi: 10.1136/esmoopen-2019-000635
13. Baumann BC, Bernstein KA, DeLaney TF, Simone CB II, Kolker JD, Choy E, et al. Multi-institutional analysis of stereotactic body radiotherapy for sarcoma pulmonary metastases: High rates of local control with favorable toxicity. J Surg Oncol. (2020) 122:877–83. doi: 10.1002/jso.26078
14. Baumann BC, Nagda SN, Kolker JD, Levin WP, Weber KL, Berman AT, et al. Efficacy and safety of stereotactic body radiation therapy for the treatment of pulmonary metastases from sarcoma: A potential alternative to resection. J Surg Oncol. (2016) 114:65–9. doi: 10.1002/jso.24268
15. Park C, Papiez L, Zhang S, Story M, Timmerman RD. Universal survival curve and single fraction equivalent dose: useful tools in understanding potency of ablative radiotherapy. Int J Radiat Oncol Biol Phys. (2008) 70:847–52. doi: 10.1016/j.ijrobp.2007.10.059
16. Anderson PM, Subbiah V, Rohren E. Bone-seeking radiopharmaceuticals as targeted agents of osteosarcoma: samarium-153-EDTMP and radium-223. Adv Exp Med Biol. (2014) 804:291–304. doi: 10.1007/978-3-319-04843-7_16
17. Anderson PM. Radiopharmaceuticals for Treatment of Osteosarcoma. Adv Exp Med Biol. (2020) 1257:45–53. doi: 10.1007/978-3-030-43032-0_4
18. Bruland OS, Nilsson S, Fisher DR, Larsen RH. High-linear energy transfer irradiation targeted to skeletal metastases by the alpha-emitter 223Ra: adjuvant or alternative to conventional modalities? Clin Cancer Res. (2006) 12(20 Pt. 2):6250s–7s. doi: 10.1158/1078-0432.CCR-06-0841
19. Haro KJ, Scott AC, Scheinberg DA. Mechanisms of resistance to high and low linear energy transfer radiation in myeloid leukemia cells. Blood. (2012) 120:2087–97. doi: 10.1182/blood-2012-01-404509
20. Melville G, Melville P. A theoretical model for the production of Ac-225 for cancer therapy by neutron capture transmutation of Ra-226. Appl Radiat Isot. (2013) 72:152–7. doi: 10.1016/j.apradiso.2012.09.019
21. Jaffe N, Carrasco H, Raymond K, Ayala A, Eftekhari F. Can cure in patients with osteosarcoma be achieved exclusively with chemotherapy and abrogation of surgery? Cancer. (2002) 95:2202–10. doi: 10.1002/cncr.10944
22. Bielack SS, Carrasco H, Raymond K, Ayala A, Eftekhari F. Methotrexate, Doxorubicin, and Cisplatin (MAP) Plus Maintenance Pegylated Interferon Alfa-2b Versus MAP alone in patients with resectable high-grade osteosarcoma and good histologic response to preoperative MAP: first results of the EURAMOS-1 good response randomized controlled trial. J Clin Oncol. (2015) 33:2279–87.
23. Marina NM, Smeland S, Bielack SS, Bernstein M, Jovic G, Krailo MD, et al. Comparison of MAPIE versus MAP in patients with a poor response to preoperative chemotherapy for newly diagnosed high-grade osteosarcoma (EURAMOS-1): an open-label, international, randomised controlled trial. Lancet Oncol. (2016) 17:1396–408. doi: 10.1016/S1470-2045(16)30214-5
24. Meyers PA, Schwartz CL, Krailo MD, Healey JH, Bernstein ML, Betcher D, et al. Osteosarcoma: the addition of muramyl tripeptide to chemotherapy improves overall survival–a report from the Children’s Oncology Group. J Clin Oncol. (2008) 26:633–8. doi: 10.1200/JCO.2008.14.0095
25. Chou AJ, Kleinerman ES, Krailo MD, Chen Z, Betcher DL, Healey JH, et al. Addition of muramyl tripeptide to chemotherapy for patients with newly diagnosed metastatic osteosarcoma: a report from the Children’s Oncology Group. Cancer. (2009) 115:5339–48. doi: 10.1002/cncr.24566
26. Anderson PM, Meyers P, Kleinerman E, Venkatakrishnan K, Hughes DP, Herzog C, et al. Mifamurtide in metastatic and recurrent osteosarcoma: a patient access study with pharmacokinetic, pharmacodynamic, and safety assessments. Pediatr Blood Cancer. (2014) 61:238–44. doi: 10.1002/pbc.24686
27. Anderson PM, Tomaras M, McConnell K. Mifamurtide in osteosarcoma–a practical review. Drugs Today. (2010) 46:327–37. doi: 10.1358/dot.2010.46.5.1500076
28. Janeway KA, Barkauskas DA, Krailo MD, Meyers PA, Schwartz CL, Ebb DH, et al. Outcome for adolescent and young adult patients with osteosarcoma: a report from the Children’s Oncology Group. Cancer. (2012) 118:4597–605. doi: 10.1002/cncr.27414
29. Bacci G, Mercuri M, Longhi A, Ferrari S, Bertoni F, Versari M, et al. Grade of chemotherapy-induced necrosis as a predictor of local and systemic control in 881 patients with non-metastatic osteosarcoma of the extremities treated with neoadjuvant chemotherapy in a single institution. Eur J Cancer. (2005) 41:2079–85. doi: 10.1016/j.ejca.2005.03.036
30. Musta E, van Geloven N, Anninga J, Gelderblom H, Fiocco M. Short-term and long-term prognostic value of histological response and intensified chemotherapy in osteosarcoma: a retrospective reanalysis of the BO06 trial. BMJ Open. (2022) 12:e052941. doi: 10.1136/bmjopen-2021-052941
31. Goorin AM, Harris MB, Bernstein M, Ferguson W, Devidas M, Siegal GP, et al. Phase II/III trial of etoposide and high-dose ifosfamide in newly diagnosed metastatic osteosarcoma: a pediatric oncology group trial. J Clin Oncol. (2002) 20:426–33. doi: 10.1200/JCO.2002.20.2.426
32. Meazza C, Bisogno G, Casanova M, Zanetti I, Carli M, Ferrari A. Full-dose ifosfamide can be safely administered to outpatients. Pediatr Blood Cancer. (2008) 50:375–8. doi: 10.1002/pbc.20958
33. Meazza C, Casanova M, Luksch R, Podda M, Favini F, Cefalo G, et al. Prolonged 14-day continuous infusion of high-dose ifosfamide with an external portable pump: feasibility and efficacy in refractory pediatric sarcoma. Pediatr Blood Cancer. (2010) 55:617–20. doi: 10.1002/pbc.22596
34. Anderson P. Continuously improving ifosfamide/mesna: a winning combination. Pediatr Blood Cancer. (2010) 55:599–600. doi: 10.1002/pbc.22652
35. Anderson PM, Thomas SM, Sartoski S, Scott JG, Sobilo K, Bewley S, et al. Strategies to mitigate chemotherapy and radiation toxicities that affect eating. Nutrients. (2021) 13:4397. doi: 10.3390/nu13124397
36. Martin-Liberal J, Alam S, Constantinidou A, Fisher C, Khabra K, Messiou C, et al. Clinical activity and tolerability of a 14-day infusional Ifosfamide schedule in soft-tissue sarcoma. Sarcoma. (2013) 2013:868973. doi: 10.1155/2013/868973
37. Zhang Y, Kawedia JD, Myers AL, McIntyre CM, Anderson PM, Kramer MA, et al. Physical and chemical stability of high-dose ifosfamide and mesna for prolonged 14-day continuous infusion. J Oncol Pharm Pract. (2014) 20:51–7. doi: 10.1177/1078155213478284
38. Etchebehere E, Brito AE, Kairemo K, Rohren E, Araujo J, Macapinlac H. Is interim (18)F-fluoride PET/CT a predictor of outcomes after radium-223 therapy? Radiol Bras. (2019) 52:33–40. doi: 10.1590/0100-3984.2017.0178
39. Etchebehere EC, Araujo JC, Fox PS, Swanston NM, Macapinlac HA, Rohren EM. Prognostic Factors in Patients Treated with 223Ra: the role of skeletal tumor burden on baseline 18F-fluoride PET/CT in predicting overall survival. J Nucl Med. (2015) 56:1177–84. doi: 10.2967/jnumed.115.158626
40. Etchebehere EC, Araujo JC, Milton DR, Erwin WD, Wendt RE III, Swanston NM, et al. Skeletal tumor burden on baseline 18F-Fluoride PET/CT predicts bone marrow failure after 223Ra therapy. Clin Nucl Med. (2016) 41:268–73. doi: 10.1097/RLU.0000000000001118
41. Harmon SA, Bergvall E, Mena E, Shih JH, Adler S, McKinney Y, et al. A Prospective Comparison of (18)F-Sodium Fluoride PET/CT and PSMA-Targeted (18)F-DCFBC PET/CT in Metastatic Prostate Cancer. J Nucl Med. (2018) 59:1665–71. doi: 10.2967/jnumed.117.207373
42. Harmon SA, Mena E, Shih JH, Adler S, McKinney Y, Bergvall E, et al. A comparison of prostate cancer bone metastases on (18)F-sodium fluoride and prostate specific membrane antigen ((18)F-PSMA) PET/CT: discordant uptake in the same lesion. Oncotarget. (2018) 9:37676–88. doi: 10.18632/oncotarget.26481
43. Harmon SA, Perk T, Lin C, Eickhoff J, Choyke PL, Dahut WL, et al. Quantitative assessment of early [(18)F]Sodium Fluoride positron emission tomography/computed tomography response to treatment in men with metastatic prostate cancer to bone. J Clin Oncol. (2017) 35:2829–37. doi: 10.1200/JCO.2017.72.2348
44. Kairemo K, Milton DR, Etchebehere E, Rohren EM, Macapinlac HA. Final Outcome of 223Ra-therapy and the Role of 18F-fluoride-PET in response evaluation in metastatic castration-resistant prostate cancer-A single institution experience. Curr Radiopharm. (2018) 11:147–52. doi: 10.2174/1874471011666180629145030
45. Kairemo K, Rohren EM, Anderson PM, Ravizzini G, Rao A, Macapinlac HA, et al. Development of sodium fluoride PET response criteria for solid tumours (NAFCIST) in a clincal trial of radium-223 in osteosarcoma: from RECIST to PERCIST to NAFCIST. EMSO Open. (2019) 4:e000439. doi: 10.1136/esmoopen-2018-000439
46. Rohren EM, Etchebehere EC, Araujo JC, Hobbs BP, Swanston NM, Everding M, et al. Determination of skeletal tumor burden on 18F-Fluoride PET/CT. J Nucl Med. (2015) 56:1507–12. doi: 10.2967/jnumed.115.156026
47. Nilsson S, Larsen RH, Fosså SD, Balteskard L, Borch KW, Westlin JE, et al. First clinical experience with alpha-emitting radium-223 in the treatment of skeletal metastases. Clin Cancer Res. (2005) 11:4451–9. doi: 10.1158/1078-0432.CCR-04-2244
48. Subbiah V, Anderson PM, Kairemo K, Hess K, Huh WW, Ravi V, et al. Alpha particle Radium 223 dichloride in high-risk osteosarcoma: a phase I dose escalation trial. Clin Cancer Res. (2019) 25:3802–10. doi: 10.1158/1078-0432.CCR-18-3964
49. Branstetter DG, Nelson SD, Manivel JC, Blay JY, Chawla S, Thomas DM, et al. Denosumab induces tumor reduction and bone formation in patients with giant-cell tumor of bone. Clin Cancer Res. (2012) 18:4415–24. doi: 10.1158/1078-0432.CCR-12-0578
50. Chawla S, Henshaw R, Seeger L, Choy E, Blay JY, Ferrari S, et al. Safety and efficacy of denosumab for adults and skeletally mature adolescents with giant cell tumour of bone: interim analysis of an open-label, parallel-group, phase 2 study. Lancet Oncol. (2013) 14:901–8. doi: 10.1016/S1470-2045(13)70277-8
51. Seeman E, Delmas PD, Hanley DA, Sellmeyer D, Cheung AM, Shane E, et al. Microarchitectural deterioration of cortical and trabecular bone: differing effects of denosumab and alendronate. J Bone Miner Res. (2010) 25:1886–94. doi: 10.1002/jbmr.81
52. Thomas D, Henshaw R, Skubitz K, Chawla S, Staddon A, Blay JY, et al. Denosumab in patients with giant-cell tumour of bone: an open-label, phase 2 study. Lancet Oncol. (2010) 11:275–80. doi: 10.1016/S1470-2045(10)70010-3
53. Zebaze RM, Libanati C, Austin M, Ghasem-Zadeh A, Hanley DA, Zanchetta JR, et al. Differing effects of denosumab and alendronate on cortical and trabecular bone. Bone. (2014) 59:173–9. doi: 10.1016/j.bone.2013.11.016
54. Shewach DS, Lawrence TS. Radiosensitization of human tumor cells by gemcitabine in vitro. Semin Oncol. (1995) 22(4 Suppl. 11):68–71.
55. Shewach DS, Lawrence TS. Radiosensitization of human solid tumor cell lines with gemcitabine. Semin Oncol. (1996) 23(5 Suppl. 10):65–71.
56. Shewach DS, Lawrence TS. Gemcitabine and radiosensitization in human tumor cells. Invest New Drugs. (1996) 14:257–63. doi: 10.1007/BF00194528
57. Lawrence TS, Chang EY, Hahn TM, Shewach DS. Delayed radiosensitization of human colon carcinoma cells after a brief exposure to 2’,2’-difluoro-2’-deoxycytidine (Gemcitabine). Clin Cancer Res. (1997) 3:777–82.
58. Lawrence TS, Eisbruch A, McGinn CJ, Fields MT, Shewach DS. Radiosensitization by gemcitabine. Oncology. (1999) 13(10 Suppl 5):55–60.
59. Lawrence TS, Eisbruch A, Shewach DS. Gemcitabine-mediated radiosensitization. Semin Oncol. (1997) 24(2 Suppl 7):S7–24.
60. Brand R, Capadano M, Tempero M. A phase I trial of weekly gemcitabine administered as a prolonged infusion in patients with pancreatic cancer and other solid tumors. Invest New Drugs. (1997) 15:331–41. doi: 10.1023/A:1005981317532
61. Tempero M, Plunkett W, Ruiz Van Haperen V, Hainsworth J, Hochster H, Lenzi R. Randomized phase II comparison of dose-intense gemcitabine: thirty-minute infusion and fixed dose rate infusion in patients with pancreatic adenocarcinoma. J Clin Oncol. (2003) 21:3402–8. doi: 10.1200/JCO.2003.09.140
62. Anderson PM. Radium Knocks out Tumor cells in Case of Unresectable Paraspinalosteosarcoma. New York, NY: Consult QD (2022).
63. Plutt AA, Stanton MP, Zembillas AS, Pierson CE, Zahler S, Anderson PM, et al. Excellent Tolerability of Ifosfamide and Mesna Via Continuous Infusion in a Pediatric Patient Population. J Pediatr Hematol Oncol. (2021) [Epub ahead of print]. doi: 10.1097/MPH.0000000000002361
64. Daudigeos-Dubus E, Le Dret L, Lanvers-Kaminsky C, Bawa O, Opolon P, Vievard A, et al. Regorafenib: antitumor activity upon mono and combination therapy in preclinical pediatric malignancy models. PLoS One. (2015) 10:e0142612. doi: 10.1371/journal.pone.0142612
65. Blank N, Laskov I, Kessous R, Kogan L, Lau S, Sebag IA, et al. Absence of cardiotoxicity with prolonged treatment and large accumulating doses of pegylated liposomal doxorubicin. Cancer Chemother Pharmacol. (2017) 80:737–43. doi: 10.1007/s00280-017-3412-8
66. Skubitz KM, Blaes AH, Konety SH, Francis GS. Cardiac safety profile of patients receiving high cumulative doses of pegylated-liposomal doxorubicin: use of left ventricular ejection fraction is of unproven value. Cancer Chemother Pharmacol. (2017) 80:787–98. doi: 10.1007/s00280-017-3420-8
67. Tomita H, Tanaka K, Tanaka T, Hara A. Aldehyde dehydrogenase 1A1 in stem cells and cancer. Oncotarget. (2016) 7:11018–32. doi: 10.18632/oncotarget.6920
68. Kang JH, Lee SH, Hong D, Lee JS, Ahn HS, Ahn JH, et al. Aldehyde dehydrogenase is used by cancer cells for energy metabolism. Exp Mol Med. (2016) 48:e272. doi: 10.1038/emm.2016.103
69. Anderson PM, Bielack SS, Gorlick RG, Skubitz K, Daw NC, Herzog CE, et al. A phase II study of clinical activity of SCH 717454 (robatumumab) in patients with relapsed osteosarcoma and Ewing sarcoma. Pediatr Blood Cancer. (2016) 63:1761–70. doi: 10.1002/pbc.26087
70. Thiele NA, Wilson JJ. Actinium-225 for targeted alpha therapy: coordination chemistry and current chelation approaches. Cancer Biother Radiopharm. (2018) 33:336–48. doi: 10.1089/cbr.2018.2494
71. Muller C, van der Meulen NP, Benešová M, Schibli R. Therapeutic Radiometals Beyond (177)Lu and (90)Y: production and application of promising alpha-particle, beta(-)-particle, and auger electron emitters. J Nucl Med. (2017) 58(Suppl 2):91S–6S. doi: 10.2967/jnumed.116.186825
72. Ahmed N, Brawley VS, Hegde M, Robertson C, Ghazi A, Gerken C, et al. Human Epidermal Growth Factor Receptor 2 (HER2) -Specific Chimeric Antigen Receptor-Modified T Cells for the Immunotherapy of HER2-Positive Sarcoma. J Clin Oncol. (2015) 33:1688–96. doi: 10.1200/JCO.2014.58.0225
73. Murray I, Rojas B, Gear J, Callister R, Cleton A, Flux GD. Quantitative dual-isotope planar imaging of thorium-227 and Radium-223 using defined energy windows. Cancer Biother Radiopharm. (2020) 35:530–9. doi: 10.1089/cbr.2019.3554
74. Lagmay JP, Krailo MD, Dang H, Kim A, Hawkins DS, Beaty O III, et al. Outcome of patients with recurrent osteosarcoma enrolled in seven phase II Trials through children’s cancer group, pediatric oncology group, and children’s oncology group: learning from the past to move forward. J Clin Oncol. (2016) 34:3031–8. doi: 10.1200/JCO.2015.65.5381
Keywords: osteosarcoma, osteoblastic metastases, bone metastases, lung metastases, radiosensitization, denosumab, IGF1R antibody FPI-1434-225Ac
Citation: Anderson PM, Subbiah V and Trucco MM (2022) Current and future targeted alpha particle therapies for osteosarcoma: Radium-223, actinium-225, and thorium-227. Front. Med. 9:1030094. doi: 10.3389/fmed.2022.1030094
Received: 28 August 2022; Accepted: 07 October 2022;
Published: 15 November 2022.
Edited by:
Asta Juzeniene, Oslo University Hospital, NorwayReviewed by:
Jacek Capala, National Cancer Institute (NIH), United StatesCopyright © 2022 Anderson, Subbiah and Trucco. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Peter M. Anderson, YW5kZXJzcEBjY2Yub3Jn
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.