
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Med., 14 December 2021
Sec. Pulmonary Medicine
Volume 8 - 2021 | https://doi.org/10.3389/fmed.2021.794599
This article is part of the Research TopicCOVID-19 Related Acute Vascular Distress Syndrome: from Physiopathology to TreatmentView all 10 articles
Background and Objective: Infection by the novel coronavirus disease 2019 (COVID-19) has been associated with different types of thrombotic complications same as portal vein thrombosis (PVT). However, by emerging vaccines of COVID, the thrombosis did not seem to be concerning anymore. Until new findings showed that, the vaccine of COVID itself can cause PVT.
Method: We performed an electronic search in PubMed, Scopus, and Web of Sciences to evaluate the possibility of occurring PVT due to infection and vaccination of COVID-19. The results were reported in a narrative method and categorized into tables.
Result: Overall, 40 cases of PVT from 34 studies were reviewed in this article. The prevalence of PVT following COVID-19 was more remarkable in males. However, it was more common in females after vaccinations of COVID-19 in the reviewed cases. Regardless of etiology, 20 of PVT cases reviewed in this article had at least one comorbidity. The most common clinical presentation was abdominal pain (AP). After anticoagulant therapies, most of the patients improved or discharged.
Conclusion: As long as the laboratory findings are not appropriate enough to predict PVT, the diagnosis of this complication with whatever underlying reason is challengeable, while rapid diagnosis and treatment of that are vital. Therefore, by providing available data in an organized way, we aimed to prepare the information of infected patients for better and easier future diagnosis of PVT in new cases.
- On the basis of studies we reviewed, PVT following COVID-19 was reported more in males, however; this complication was more mentioned in the females after vaccination of COVID-19.
- Patients with comorbidities were more likely to develop portal vein thrombosis with both underlying reasons.
- Liver CT scan beside laboratory findings were useful solutions in diagnosing this complication.
- We suggest examining more about the underlying mechanism of PVT after vaccination because there was a case mentioned in our study with thrombocytosis so vaccine-induced thrombosis might not be the only mechanism leading to PVT.
The new member of the Coronaviridae family has started the novel coronavirus disease 2019 (COVID-19) pandemic, which was first reported in Wuhan, China, appearing as a worldwide health crisis. To date, about 239 million people from 223 different countries in the world have experienced some form of this disease. This dreadful pandemic led to over 4.8 million deaths totally and still, the number keeps on increasing (1–3).
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), as it was named by the WHO, is a single-stranded RNA virus. Different proteins play roles in the structure and function of this virus. One of the most important ones is the spike glycoprotein, the extrinsic crown-shaped construction of the virus, which is the key connector for the fusion of SARS-CoV-2 and human being cells (4).
The clinical presentations of COVID-19 vary depending upon the immune system, gender, and age of the patients. General symptoms including fever, cough, and fatigue are common in many patients, but several complications such as thrombosis, severe respiratory symptoms, heart, kidney, and multiorgan failure are less prevalent (4–6). The incidence rate of some symptoms was determined by a meta-analysis in 50,466 patients, as followed; fever 89.1, cough 72.2, fatigue 42.5, and abnormal CT in 96.6% of the cases (7). COVID-19 is associated with different types of coagulation abnormalities and is shown to be associated with an increased risk of arterial thrombosis and venous thromboembolism (VTE). It can mainly cause, disseminated intravascular coagulation (DIC), VTE, deep vein thrombosis (DVT), portal vein thrombosis (PVT), and other coagulopathies (8).
Portal vein thrombosis is an abnormal rare condition associated with malignancy, liver cirrhosis, and acute abdominal inflammation (9). PVT cases due to non-cirrhotic reasons are scarce. After cirrhosis, myeloproliferative neoplasms, surgery, and inflammatory conditions are three major triggers leading to obstruction in the portal vein (10). This disorder usually occurs when thrombus blocks the portal vein partially or completely. This obstruction of the portal vein is categorized in different ways. Due to Baveno VI criteria, PVT may happen whether because of extra hepatic portal vein obstruction or the intrahepatic one. However, splenic or super mesenteric veins are not involved. Another categorization demonstrates that in the chronic form of PVT, patients generally develop symptoms such as varicose veins and hypersplenism that are associated with portal hypertension. Nevertheless, in the acute form, local and systemic prothrombotic factors are the main reasons for this complication (9, 11). The clinical presentations of acute PVT, show a wide spectrum of asymptomatic indications to severe intestinal ischemia and infarction (12). Several surveys indicated that activation of the coagulation pathway by COVID-19 infection might be due to the inflammatory response of cytokines to virus invasion (13). For example, IL-6 can increase the expression of tissue factor (TF) from mononuclear cells, which may lead to clot formation. Besides this, other inflammatory cytokines such as tumor necrosis factor alpha- α (TNF-α) and IL-1 can play roles in anticoagulant pathway inhibition (8).
After introducing different vaccines of COVID-19 to the world, in addition to PVT following the COVID-19, several people have encountered with PVT after being vaccinated against this virus all around the world.
Since PVT may happen due to several reasons including infectious disease, by reviewing the available data from other articles, we tried to study the PVT, which is either caused by COVID-19 itself, or vaccination while narrating the differences and similarities.
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) for developing and reporting this article (14).
All the case report studies that stated PVT on patients with COVID-19 were included in this study. Every study that reported consequences related to PVT following COVID-19 or all the types of vaccines of COVID-19 was included. Ultrasonography and contrast-enhanced CT are two gold-standard investigations of PVT. All the articles using each of these two diagnostic methods besides the studies which had not mentioned the diagnostic method but been approved and published as a PVT case due to infection of COVID-19 or vaccination were included. All the studies without available English full-text were excluded. Studies that report incomplete data or irrelevant subjects were also excluded.
PICOD:
Population: All patients with COVID-19 or vaccinated people against COVID-19.
Intervention: Not applicable.
Comparison: Not applicable.
Outcome: Portal vein thrombosis.
Design: Case-report studies.
We did an electronic search of PubMed, Scopus, and Web of Sciences to September 7, 2021, without language restrictions. The whole data extracted with these search term combinations “2019 nCoV” or 2019 nCoV or “2019 novel coronavirus” or COVID-19 or “new coronavirus” or “novel coronavirus” or “SARS-CoV-2” or (Wuhan and coronavirus) or “SARS-CoV” or “2019-nCoV” or “SARS-CoV-2” and (“portal vein thrombosis” or “portal venous thrombosis”).
Besides this, these mesh terms were also searched COVID Vaccine and Neurology, AstraZeneca COVID vaccine, ChAdOx1 nCoV-19 COVID vaccine, AZD1222 COVID vaccine, Janssen COVID vaccine, Johnson & Johnson COVID vaccine, Ad26.COV2 COVID vaccine and “portal vein thrombosis” or “portal venous thrombosis” (Appendix 1).
Then, we merged them in Endnote V.8. All the reference lists from the included studies and relevant systematic reviews were hand-searched for additional studies.
The duplicate studies were removed and the title, abstract, and full-text of records were screened by two independent reviewers based on pre-mentioned inclusion and exclusion criteria. A third reviewer reviewed the record in case of discrepancy, and disagreement was resolved by consultation.
Two independent reviewers extracted and tabulated all the relevant data using a researcher-made checklist. Disagreement was resolved by consensus between all the authors. The data extraction checklist includes items such as author name and year of publication, demographic data, clinical presentation, COVID-19 diagnosis test, clinical manifestations related to PVT (same as fever, APs, etc.), PVT location, treatment, and outcome of the therapy (Table 1). Besides all these, the data of laboratory experiments of the patients were categorized in a separate table (Table 2). As long as not all of these parameters were crucial enough to extract the suitable data from the articles with COVID-19 vaccinated cases, we designed another table with the extra following subheadings: Type of vaccine and diagnostic tests, COVID-19 infection test, number of days until the start of symptoms, and abnormal parameters in laboratory examinations.
A third reviewer rechecked the extracted data.
All the studies were checked in terms of quality by two independent reviewers using an eight-item Joanna Briggs Institute (JBI) checklist for case report studies. The potential disagreement was resolved by consultation with a third reviewer. This checklist includes eight questions and four-rating score (Yes, No, Unclear, and Not applicable). Each question was scored 1 point for yes, 0 points for unclear and no. Then, studies were categorized as having a high risk of bias if the summary score was 0 to <3, moderate risk of bias if the summary score was between 3 and <6 points, and low risk of bias if the summary score was 6 or higher.
Due to potential heterogeneity between studies, we reported the results in a narrative method and categorized them into several items available in Tables 1–3.
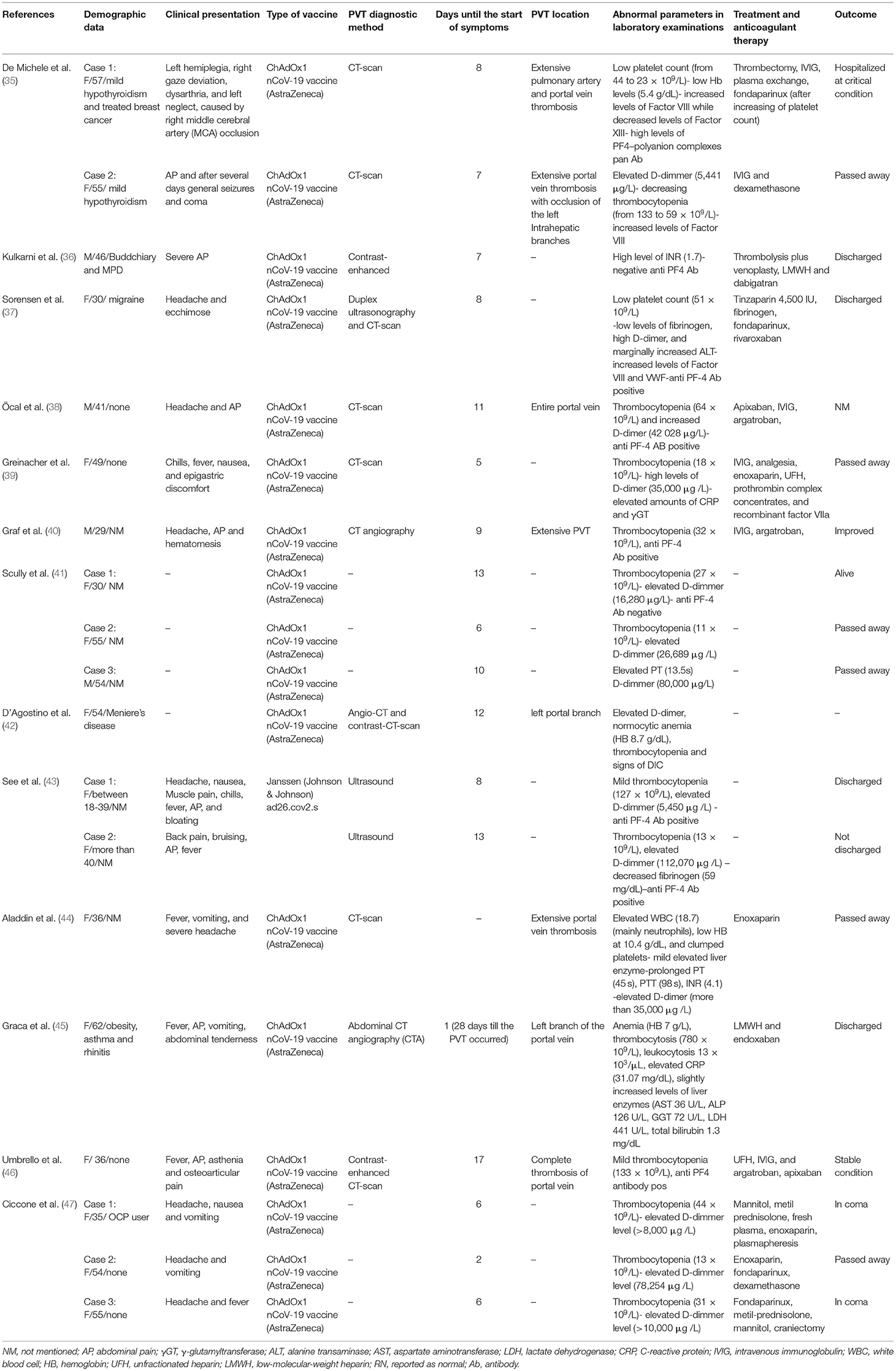
Table 3. Data extraction table of cases suffering from PVT as a side effect of vaccination of COVID-19.
The initial search produced 200 articles from three main databases. After removing duplicates, 123 articles remained. These 123 articles were evaluated based on title and abstract and eventually, 53 articles were selected. The full text of these 53 articles was assessed for eligibility criteria. Finally, 34 studies with 40 cases reported the incidence of PVT resulting from COVID-19 (21 cases) or vaccination of COVID-19 (19 cases) due to our inclusion criteria and 19 studies were excluded due to incomplete data, irrelevant subject, or non-availability of full text (Figure 1).
The characteristics of the included studies are summarized in the three tables. All of the 34 studies were case reports. The data about patients with the PVT following COVID demonstrates a wide age range of 3–79 years. About 66.7% of 21 cases were male. Thirteen of twenty-one cases (61.9%) had at least one comorbidity. There were five cases with liver disorders (four cases with alcohol consumption and one case with controlled hepatitis) three cases with brain injuries, two cases with asthma, two cases of thrombosis, and one case with pregnancy.
However, these amounts differed in PVT following vaccination. The median age of these cases was about 45 years (range 29–62 years) (the age of two cases were reported as ranges, so they were ignored in calculating median age). Only four cases were male and about 79% of the cases were female. Seven of nineteen cases had the previous history of the disease. Hypothyroidism was reported in two cases, Budd-Chiari syndrome and myeloproliferative disorder (MPD) were reported in one case at the same time. Other previous diseases such as migraine, asthma, Meniere's disease, and oral contraceptive pill (OCP) use were each reported in one separate case. Five had no known comorbidities and in seven cases, it was not mentioned. All of the cases of this complication had received the AstraZeneca vaccine except two of whom were injected with Johnson & Johnson.
The JBI tool for quality assessment of included studies yielded scores ranging from 4 to 7. Mean methodological quality was 6.06 out of 8. A total of 31 studies were classified as low risk of bias (77.5%) and 9 studies were with moderate risk of bias (22.5%). Details of the answers to the 8 questions of the tools are given in Appendix 2.
As was expected, almost different presentations were observed in PVT following COVID and vaccines of COVID. After infecting with COVID-19, 14 cases presented the APs probably because of their PVT. Ten cases presented fever, which is one of the most common symptoms of both the virus and PVT. Seven cases had respiratory problems. Nausea and vomiting were observed in five cases. Four cases complained from cough and the same number of cases were involved with mental problems. Other presentations same as diarrhea, jaundice, and hypertension were presented in fewer cases.
As the same, the AP was a prevailing manifestation in most cases (eight cases) of PVT following vaccination, especially at the right upper quarter (RUQ), where the liver is located. Then, headache (eight cases) and fever (seven cases) were the most prevalent ones, respectively. Nausea and vomiting were presented in the eight cases. Ecchymosis, chills, and muscle pains were less common.
The most remarkable point about laboratory tests in COVID-19 infected patients was the high level of CRP in all the cases that this index was measured (it was measured in 11 cases).
Only two out of twenty-one patients were associated with abnormal hemoglobin (Hb) levels. Variable platelet counts (PLT) were reported in cases (1 case with decreased, 1 case with increased, and 11 cases with normal counts of PLTs). White blood cell (WBC) counts never dropped under the normal range. Among the cases manifesting PVT following COVID-19, nearly half of the patients (11 cases) appeared with leukocytosis. Liver enzymes were normal (5 cases) to elevated (9 cases) as was expected in PVT disease. Bilirubin was elevated only in three cases. The level of D-dimer was elevated remarkably more than normal in nine patients. Coagulation tests were not performed for most of the patients and did not show a significant increase in performed cases (PT, PTT, and INR were normal in 7, 6, and 5 cases, respectively, and were only elevated in 2 cases).
The results of laboratory experiments in PVT following vaccination showed different algorithms. Thrombocytopenia was a prevalent finding in most of the cases. Fifteen out of nineteen (79%) had experienced low-platelet counts. However, one case was reported with thrombocytosis. The D-dimer level was elevated in 14 cases as a sign of thrombosis. Anti-PF4 antibody was positive in seven cases and negative in two cases. Coagulation tests were abnormal in nine cases. Other complementary data are given in Table 3 (Only abnormal indices are reported).
To treat PVT following COVID-19, different drugs were prescribed depending upon the condition of the patient. Enoxaparin was the most commonly utilized treatment and fortunately, most cases improved after treating it. Six cases used heparin. Moreover, the same number of cases (3) were treated with warfarin or Apixaban. Although in several cases, heparin or enoxaparin were prescribed as prophylaxis therapy before the occurrence of PVT and after the diagnosis COVID-19, PVT developed in some cases. In 1 case, thrombectomy was performed, but finally the patient passed away. Most of these drugs led to improving or discharging the patient. There were only two cases with not clear outcomes and two cases who were passed away.
Nevertheless, when PVT was presented after vaccination the most utilized drugs were intravenous immunoglobulin (IVIG) and low molecular weight heparin (LMWH). Argatroban, Apixaban, unfractionated heparin, thrombectomy, and thrombolysis were the other more prevalent treatments used, respectively. From the postvaccination PVT, six cases passed away, five cases were reported as still in charge, and six cases were discharged (the outcomes of two patients were not mentioned).
In this study, we review 40 cases with PVT because of new etiologies, the COVID and the vaccines of COVID-19. In this study, the median age was 41 years with the preference sex of males in cases of COVID. However, in PVT following vaccination, the median age of cases was 45 and 79% of them were females. The average number of days until onset of the symptoms was 8.3 days. As long as this is a systematic review of the cases, it is only possible to narrate the existed data. For comparisons, a cohort study is suggested on two homogenate groups. The underlying mechanism of PVT following COVID-19 is not precisely clear but Marjot et al. declared that the attachment of the virus might occur through ACE-2 receptors on the surface of cholangiocytes so the local presentation of COVID-19 in the liver, makes the body to produce different cytokines against it, which leads to the liver injury (Figure 2) (48). As portal vein is a vital part of the liver, the thrombosis may be formed because of the same reason. In addition, there is another theory provided by Mohseni Afshar et al. suggesting a mechanism for vaccine-induced thrombosis (VIT). They recommended that thrombi are formed in the vessels dependent or independent of heparin. As long as most of the vaccinated cases were non-heparin users, this heparin-induced thrombocytopenia (HIT) may be happened in a spontaneous or autoimmune way (called aHIT). In aHIT, it is not necessary for heparin to be present and other reasons same as free DNA do job of heparin. The free DNA attaches to PF4, and then platelets to make thrombus (49). The positive anti-PF4 antibody in seven out of nine reviewed cases somehow confirmed these findings. Almost in every type of HIT, the decreased counts of platelets are supposed to observe (Figure 3). However, in a 62-year-old woman with PVT that was reported by Graca (mentioned in Table 1), the opposite happened. The case was an asthmatic patient with the thrombocytosis of 780 × 103 per milliliter (45). Therefore, still further studies are needed to figure out other underlying mechanisms of the PVT following vaccination and solve this paradox. A recent cohort study stated the incidence of PVT following COVID-19 was 392.3 per million people, which was significantly higher than in PVT following vaccination (AstraZeneca and Jansen vaccines were excluded from this study) (49, 50).

Figure 2. Possible underlying mechanism of PVT by COVID-19 infection. Cholangiocyte is a kind of liver cell which has ACE II receptors presented on the surface more than hepatocytes and endothelium. By direct fusing of COVID-19 to these cells, first the direct injury of the liver happens because of the accumulation of bile acids. Then several inflammatory cytokines (IL6, TNF-alpha) are secreted and they play as inflammation and thrombosis triggers in the liver. Clot formation in the portal vein might be because of the increased expression of tissue factor (TF) from mononuclear cells, possibly done by IL-6. Beside this, other inflammatory cytokines such as tumor necrosis factor alpha (TNF-alpha) and IL-1 can play roles in anticoagulant pathway inhibition.

Figure 3. Similar to HIT, vaccine induced thrombocytopenia and thrombosis (VITI) occurs because of free DNA available in COVID-19 vaccines. The free DNA stimulates the production of PF-4 molecules from platelets. The PF4-free DNA attaches to FCRYll on the surface of platelets and the platelet clot shapes.
Chronic PVT is a persistent obstruction of the portal vein often more than 6 months from the onset of the presentation. Therefore, the mentioned data suggest that PVT following infection of COVID-19 is of acute form. In PVT, the clinical presentation of the patients are as followed: AP (61%), hepatomegaly (67%), and ascites (83%) while about 20% of the patients have no symptoms (10). Data presented in this study demonstrated that in PVT following both etiologies, the AP was the most prevalent presentation whether after COVID infection or vaccination (66% after COVID-19 and 42% after vaccination).
Examinations of available data demonstrated that laboratory indices are not proper assistances in confirmed diagnosis or prognosis of PVT, and ultrasonography or contrast-enhanced CT scan of the hepatic portal vein is a better way for a valid diagnosis. The Baveno VI criteria suggest that Doppler ultrasound, CT scan and MRI are the best ways to diagnose both the presence and extension of PVT. However, some laboratory indices will be helpful. The elevation of D-dimer was expected, same as PVT cases due to non-COVID-19 etiologies (51). The liver function tests were normal to elevated because in PVT, the liver retrieves the condition by enhancing hepatic arterial flow (52, 53). In accordance with our result, another study suggested that mild elevation of ALT and AST enzymes are estimated in 29–39% and 38–63 % of patients of COVID, respectively (48). There was also a study, which mentioned that in cases of PVT, evaluation of mean platelet volume (MPV) would help as a diagnostic index. They declared that the larger the platelets, the higher the thrombotic conditions (54). However, the MPV amount was not mentioned in any of the studies we examined. As long as PVT is an uncommon complication without pathognomonic clinical manifestations, the mentioned laboratory indices beside general clinical presentations such as fever and AP are helpful in diagnosis of PVT in COVID-19 infected patients. Moreover, these parameters shift to AP and headache beside thrombocytopenia, D-dimer levels, coagulation tests, and anti-PF4 antibody detection in the cases who are suspected to PVT following vaccination.
Up to now, anticoagulants are the suggested treatments for PVT. Due to presence of hypercoagulable states, the treatment can be considered long term or short term. Heparin and low-molecular-weight heparin (such as enoxaparin) was prescribed in more than half of the cases. Nearly all of them were improved except 2 with uncertain feedbacks. While most of the studies suggest these 2 drugs for solving the thrombosis problem in COVID-19 infected patients, there is one case report that advises platelet count monitoring due the probable risk of HIT. In addition, because the similar mechanism (aHIT) underlies the PVT related vaccination of COVID-19, IVIG, LMWH, and fondaparinux were the most utilized drugs for patients with this trouble. Two of COVID-19 infected and six of vaccinated cases were passed away. Due to heterogeneity of the age, sex, and comorbidities of the patients it is not possible to report which condition is associated with less mortality but due to other studies it is clear that vaccine is safe (55) and the mortality rate of thrombosis is much lower than the infection itself. However, in general, the thrombosis morbidity rate is less after vaccination and even when it is occurred, the condition is manageable. Nevertheless, as long as HIT-like mechanisms are suggested as the main responsible of PVT occurrence after vaccination, the use of heparin is with more caution and the wide-spectrum of the utilized drugs somehow confirms this challengeable condition.
However, we were confronted with several limitations same as small numbers of studies and lack of strong evidence, so further examinations are needed in future studies for more information.
In this systematic review, we have tried to prepare data available on two new etiologies of acute PVT. Even if the patient is receiving anticoagulants as prophylaxis therapy of PVT, this complication might happen after infection or vaccination of COVID-19. Therefore, it is recommended that upon observing the clinical symptoms mentioned (the most important one is ap), provide a liver CT scan for the patient for checking whether the thrombosis involved this vein or not.
Same as infection of COVID-19, the morbidity rate was higher in male PVT cases after infection with the virus. Although the reviewed cases suggested if the PVT was presented due to vaccination, it is more prevalent in females. As it was the most common comorbidity in the presented cases, liver disorders, might had been deteriorated through drug-induced injury, inflammation or anoxia that resulted from COVID-19 (56).
Further studies are needed to exactly clarify that how the virus and the vaccination can lead to thrombosis of portal vein. Moreover, a cohort study is suggested to compare the data and results in two homogenate groups of patients with PVT following COVID-19 and vaccination of COVID-19.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
SK and AR contributed in design, acquisition of data, and drafting the manuscript. MA-Z contributed in the interpretation of data and drafting the manuscript. GS was the study supervisor and contributed to all the aspects of the study. All authors contributed to the article and approved the submitted version.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2021.794599/full#supplementary-material
1. Singhal T. A review of coronavirus disease-2019 (COVID-19). Ind J Pediatr. (2020) 87:281–6. doi: 10.1007/s12098-020-03263-6
2. Sonzogni A, Previtali G, Seghezzi M, Grazia Alessio M, Gianatti A, Licini L, et al. Liver histopathology in severe COVID 19 respiratory failure is suggestive of vascular alterations. Liver Int. (2020) 40:2110–6. doi: 10.1111/liv.14601
3. COVID-19 Coronavirus Pandemic,. (2021). Available online at: https://www.worldometers.info/coronavirus/ (accessed October 13, 2021).
4. Samudrala PK, Kumar P, Choudhary K, Thakur N, Wadekar GS, Dayaramani R, et al. Virology, pathogenesis, diagnosis and in-line treatment of COVID-19. Eur J Pharmacol. (2020) 883:173375. doi: 10.1016/j.ejphar.2020.173375
5. Zhao B, Ni C, Gao R, Wang Y, Yang L, Wei J, et al. Recapitulation of SARS-CoV-2 infection and cholangiocyte damage with human liver ductal organoids. Protein Cell. (2020) 11:771–5. doi: 10.1007/s13238-020-00718-6
6. Baratella E, Ruaro B, Marrocchio C, Starvaggi N, Salton F, Giudici F, et al. Interstitial lung disease at high resolution CT after SARS-CoV-2-related acute respiratory distress syndrome according to pulmonary segmental anatomy. J Clin Med. (2021) 10:3985. doi: 10.3390/jcm10173985
7. Sun P, Qie S, Liu Z, Ren J, Li K, Xi J. Clinical characteristics of hospitalized patients with SARS-CoV-2 infection: a single arm meta-analysis. J Med Virol. (2020) 92:612–7. doi: 10.1002/jmv.25735
8. Levi M, Thachil J, Iba T, Levy JH. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. (2020) 7:e438. doi: 10.1016/S2352-3026(20)30145-9
9. Acuna-Villaorduna A, Tran V, Gonzalez-Lugo JD, Azimi-Nekoo E, Billett HH. Natural history and clinical outcomes in patients with portal vein thrombosis by etiology: a retrospective cohort study. Thromb Res. (2019) 174:137–40. doi: 10.1016/j.thromres.2018.12.019
10. Northup PG, Garcia-Pagan JC, Garcia-Tsao G, Intagliata NM, Superina RA, Roberts LN, et al. Vascular liver disorders, portal vein thrombosis, and procedural bleeding in patients with liver disease: 2020 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. (2021) 73:366–413. doi: 10.1002/hep.31646
11. Rokkam VRP, Kutti Sridharan G, Vegunta R, Vegunta R, Boregowda U, Mohan BP. Clostridium difficile and COVID-19: novel risk factors for acute portal vein thrombosis. Case Rep Vasc Med. (2021) 2021:8832638. doi: 10.1155/2021/8832638
12. Parikh S, Shah R, Kapoor P. Portal vein thrombosis. Am J Med. (2010) 123:111–9. doi: 10.1016/j.amjmed.2009.05.023
13. Al-Ani F, Chehade S, Lazo-Langner A. Thrombosis risk associated with COVID-19 infection. A scoping review. Thromb Res. (2020) 192:152–60. doi: 10.1016/j.thromres.2020.05.039
14. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann T, Mulrow CD, et al. Mapping of reporting guidance for systematic reviews and meta-analyses generated a comprehensive item bank for future reporting guidelines. J Clin Epidemiol. (2020) 118:60–8. doi: 10.1016/j.jclinepi.2019.11.010
15. Borazjani R, Seraj SR, Fallahi MJ, Rahmanian Z. Acute portal vein thrombosis secondary to COVID-19: a case report. BMC Gastroenterol. (2020) 20:386. doi: 10.1186/s12876-020-01518-2
16. de Barry O, Mekki A, Diffre C, Seror M, El Hajjam M, Carlier R-Y. Arterial and venous abdominal thrombosis in a 79-year-old woman with COVID-19 pneumonia. Radiol Case Rep. (2020) 15:1054–7. doi: 10.1016/j.radcr.2020.04.055
17. Franco-Moreno A, Piniella-Ruiz E, Montoya-Adarraga J, Ballano-Franco C, Alvarez-Miguel F, Peinado-Martinez C, et al. Portal vein thrombosis in a patient with COVID-19. Thromb Res. (2020) 194:150–2. doi: 10.1016/j.thromres.2020.06.019
18. Jafari SH, Naseri R, Khalili N, Haseli S, Bahmani M. Portal vein thrombosis associated with COVID-19: points to consider. BJR Case Rep. (2020) 6:20200089. doi: 10.1259/bjrcr.20200089
19. La Mura V, Artoni A, Martinelli I, Rossio R, Gualtierotti R, Ghigliazza G, et al. Acute portal vein thrombosis in SARS-COV-2 infection: a case report. Am J Gastroenterol. (2020) 115:1140–2. doi: 10.14309/ajg.0000000000000711
20. Low SW, Swanson KL, McCain JD, Sen A, Kawashima A, Pasha SF. Gastric ischemia and portal vein thrombosis in a COVID-19-infected patient. Endoscopy. (2020) 52:E465–e6. doi: 10.1055/a-1230-3357
21. Malik ZR, Razaq Z, Siff M, Sheikh S. COVID-19 presenting as Banti's syndrome. Cureus. (2020) 12:9096. doi: 10.7759/cureus.9096
22. Ofosu A, Ramai D, Novikov A, Sushma V. Portal vein thrombosis in a patient with COVID-19. Am J Gastroenterol. (2020) 115:1545–6. doi: 10.14309/ajg.0000000000000781
23. Abeysekera KW, Karteszi H, Clark A, Gordon FH. Spontaneous portomesenteric thrombosis in a non-cirrhotic patient with SARS-CoV-2 infection. BMJ Case Rep. (2020) 13:238906. doi: 10.1136/bcr-2020-238906
24. Kolli S, Oza VM. SARS-CoV-2 and portal vein thrombosis: a rare gastrointestinal manifestation of COVID-19. Cureus. (2021) 13:14340. doi: 10.7759/cureus.14340
25. Petters LM, Vogel TP, Munoz FM, Hernandez JA, Koohmaraie S, Nowicki MJ, et al. Multisystem inflammatory syndrome in children associated with SARS-CoV-2 in a solid organ transplant recipient. Am J Transplant. (2021) 21:2596–9. doi: 10.1111/ajt.16572
26. Sinz S, Glaser-Gallion F, Steffen T. Portal vein thrombosis in COVID-19 infection. Surgical Case Rep. (2021) 7:87. doi: 10.1186/s40792-021-01173-z
27. Miyazato Y, Ishikane M, Inada M, Ohmagari N. Acute portal vein thrombosis with COVID-19 and cirrhosis. IDCases. (2021) 24:e01094. doi: 10.1016/j.idcr.2021.e01094
28. Sharma N, Shukla R, Kumar K, Arora S, Warrier R, Philip S. Portal vein thrombosis-a rare complication of SARS-CoV-2 infection. SN Compr Clin Med. (2021) 2021:1–4. doi: 10.1007/s42399-021-00877-5
29. Rehman A, Thoppil AJ, Wallach SL. Portal vein thrombosis and splenic infarction in a COVID-19 patient. Cureus. (2021) 13:16843. doi: 10.7759/cureus.16843
30. Agarwal L, Agarwal A, Advani S, Katiyar V, Chaturvedi A, Madhusudhan KS. The eyes see what the mind seeks: a systematic review of abdominal imaging findings in patients with COVID-19. Br J Radiol. (2021) 94:20201220. doi: 10.1259/bjr.20201220
31. Jeilani M, Hill R, Riad M, Abdulaal Y. Superior mesenteric vein and portal vein thrombosis in a patient with COVID-19: a rare case. BMJ Case Rep. (2021) 14:244049. doi: 10.1136/bcr-2021-244049
32. Randhawa J, Kaur J, Randhawa HS, Kaur S, Singh H. Thrombosis of the portal vein and superior mesenteric vein in a patient with subclinical COVID-19 infection. Cureus. (2021) 13:14366. doi: 10.7759/cureus.14366
33. Rivera-Alonso D, Rivera-Alonso I, Burneo-Esteves M, Martinez-Ruiz C, Rojo-Abecia M, Moreno-Sanguino C. Acute portal vein thrombosis in mild cholecystitis. A consequence of coronavirus disease 2019 infection? Cirugia Y Cirujanos. (2021) 89:399–402. doi: 10.24875/CIRU.20001044
34. Lari E, Lari A, AlQinai S, Abdulrasoul M, AlSafran S, Ameer A, et al. Severe ischemic complications in Covid-19—A case series. Int J Surg Case Rep. (2020) 75:131–5. doi: 10.1016/j.ijscr.2020.09.009
35. De Michele M, Iacobucci M, Chistolini A, Nicolini E, Pulcinelli F, Cerbelli B, et al. Malignant cerebral infarction after ChAdOx1 nCov-19 vaccination: a catastrophic variant of vaccine-induced immune thrombotic thrombocytopenia. Nat Commun. (2021) 12:4663. doi: 10.1038/s41467-021-25010-x
36. Kulkarni AV, Reddy J, Singh JR, Sreekanth V, Reddy A, Sharma M, et al. Exercise caution with ChAdOx1 COVID-19 vaccination in Chronic Budd-Chiari syndrome with a thrombophilic genetic predisposition. J Clin Exp Hepatol. (2021). doi: 10.1016/j.jceh.2021.07.009. [Epub ahead of print].
37. Sorensen ALT, Rolland M, Hartmann J, Harboe ZB, Roed C, Jensen TO, et al. A case of thrombocytopenia and multiple thromboses after vaccination with ChAdOx1 nCoV-19 against SARS-CoV-2. Blood Adv. (2021) 5:2569–74. doi: 10.1182/bloodadvances.2021004904
38. Öcal O, Stecher SS, Wildgruber M. Portal vein thrombosis associated with ChAdOx1 nCov-19 vaccination. Lancet Gastroenterol Hepatol. (2021) 6:676. doi: 10.1016/S2468-1253(21)00197-7
39. Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic thrombocytopenia after CHADOX1 NCoV-19 vaccination. New Engl J Med. (2021) 384:2092–101. doi: 10.1056/NEJMoa2104840
40. Graf T, Thiele T, Klingebiel R, Greinacher A, Schabitz WR, Greeve I. Immediate high-dose intravenous immunoglobulins followed by direct thrombin-inhibitor treatment is crucial for survival in Sars-Covid-19-adenoviral vector vaccine-induced immune thrombotic thrombocytopenia VITT with cerebral sinus venous and portal vein thrombosis. J Neurol. (2021) 268:4483–5. doi: 10.1007/s00415-021-10599-2
41. Scully M, Singh D, Lown R, Poles A, Solomon T, Levi M, et al. Pathologic antibodies to platelet factor 4 after ChAdOx1 nCoV-19 vaccination. New Engl J Med. (2021) 384:2202–11. doi: 10.1056/NEJMoa2105385
42. D'Agostino V, Caranci F, Negro A, Piscitelli V, Tuccillo B, Fasano F, et al. A rare case of cerebral venous thrombosis and disseminated intravascular coagulation temporally associated to the COVID-19 vaccine administration. J Pers Med. (2021) 11:285. doi: 10.3390/jpm11040285
43. See I, Su JR, Lale A, Woo EJ, Guh AY, Shimabukuro TT, et al. US case reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26. COV2. S vaccination, March 2 to April 21, 2021. JAMA. (2021) 325:2448–56. doi: 10.1001/jama.2021.7517
44. Aladdin Y, Algahtani H, Shirah B. Vaccine-induced immune thrombotic thrombocytopenia with disseminated intravascular coagulation and death following the ChAdOx1 nCoV-19 vaccine. J Stroke Cerebrovasc Dis. (2021) 30:105938. doi: 10.1016/j.jstrokecerebrovasdis.2021.105938
45. Graça LL, Amaral MJ, Serôdio M, Costa B. Extensive thrombosis after COVID-19 vaccine: cause or coincidence? BMJ Case Rep. (2021) 14:e244878. doi: 10.1136/bcr-2021-244878
46. Umbrello M, Brena N, Vercelli R, Foa RA, Femia M, Rossi U, et al. Successful treatment of acute spleno-porto-mesenteric vein thrombosis after ChAdOx1 nCoV-19 vaccine. A case report. J Crit Care. (2021) 65:72–5. doi: 10.1016/j.jcrc.2021.05.021
47. Ciccone A, Zanotti B. The importance of recognizing cerebral venous thrombosis following anti-COVID-19 vaccination. Eur J Intern Med. (2021) 89:115–7. doi: 10.1016/j.ejim.2021.05.006
48. Marjot T, Webb GJ, Barritt AS, Moon AM, Stamataki Z, Wong VW, et al. COVID-19 and liver disease: mechanistic and clinical perspectives. Nat Rev Gastroenterol Hepatol. (2021) 18:348–64. doi: 10.1038/s41575-021-00426-4
49. Mohseni Afshar Z, Babazadeh A, Janbakhsh A, Afsharian M, Saleki K, Barary M, et al. Vaccine-induced immune thrombotic thrombocytopenia after vaccination against Covid-19: A clinical dilemma for clinicians and patients. Rev Med Virol. (2021) 2021:e2273. doi: 10.1002/rmv.2273
50. Sharifian-Dorche M, Bahmanyar M, Sharifian-Dorche A, Mohammadi P, Nomovi M, Mowla A. Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis post COVID-19 vaccination; a systematic review. J Neurol Sci. (2021) 2021:117607. doi: 10.1016/j.jns.2021.117607
51. Liu K, Chen J, Zhang K, Wang S, Li X, A. diagnostic prediction model of acute symptomatic portal vein thrombosis. Ann Vasc Surg. (2019) 61:394–9. doi: 10.1016/j.avsg.2019.04.037
52. Mantaka A, Augoustaki A, Kouroumalis EA, Samonakis DN. Portal vein thrombosis in cirrhosis: diagnosis, natural history, and therapeutic challenges. Ann Gastroenterol. (2018) 31:315. doi: 10.20524/aog.2018.0245
53. Intagliata NM, Caldwell SH, Tripodi A. Diagnosis, development, and treatment of portal vein thrombosis in patients with and without cirrhosis. Gastroenterology. (2019) 156:1582–99.e1. doi: 10.1053/j.gastro.2019.01.265
54. Lin W-y, Lu X, Fan F-j, Hu Y. Predictive effect of mean platelet volume in patients with portal vein thrombosis: a meta-analysis of case-control studies. Curr Med Sci. (2018) 38:575–81. doi: 10.1007/s11596-018-1916-z
55. COVID-19 Vaccine Safety Update. (2021). Available online at: https://www.ema.europa.eu/en/documents/covid-19-vaccine-safety-update/covid-19-vaccine-safety-update-comirnaty-11-august-2021_en.pdf (accessed October 13, 2021).
Keywords: COVID-19, vaccines, liver diseases, portal vein, venous thrombosis, case report
Citation: Kheyrandish S, Rastgar A, Arab-Zozani M and Sarab GA (2021) Portal Vein Thrombosis Might Develop by COVID-19 Infection or Vaccination: A Systematic Review of Case-Report Studies. Front. Med. 8:794599. doi: 10.3389/fmed.2021.794599
Received: 13 October 2021; Accepted: 17 November 2021;
Published: 14 December 2021.
Edited by:
Yazine Mahjoub, University Hospital Center (CHU) of Amiens, FranceReviewed by:
Soedarsono Soedarsono, Airlangga University, IndonesiaCopyright © 2021 Kheyrandish, Rastgar, Arab-Zozani and Sarab. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gholamreza Anani Sarab, Z2hhbnNhQHlhaG9vLmNvbQ==
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.