
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Mar. Sci. , 03 August 2023
Sec. Marine Pollution
Volume 10 - 2023 | https://doi.org/10.3389/fmars.2023.1216299
This article is part of the Research Topic Toxicological Endpoints and Bioavailability of Emerging Contaminants and their Impacts on Marine Nutrient Cycles View all 6 articles
Seagrasses provide crucial ecosystem services of relevance for the marine environment. However, anthropogenic activities are causing global seagrass decline. Increasing microplastic (MP) concentrations have been recognized as a novel threat to many marine organisms, but their effects on marine plants remain underexplored. Here, we investigate the effects of microplastic (polyethylene (PE) and polypropylene (PP)) exposure on the photosynthesis and respiration of the seagrass Zostera marina L. and its associated epiphytes. Measurements were conducted on seagrass leaves with and without epiphyte cover, as well as on epiphytes scraped off the leaf surface. Net gas exchange and pH drift measurements were used to determine rates of photosynthesis and respiration, as well as the ability of leaves and epiphytes to utilize bicarbonate. In addition, variable chlorophyll fluorescence imaging was employed to quantify the photosynthetic capacity of seagrass leaves. Our results show a limited effect of short-term (14 days) microplastic exposure on seagrass leaves and their associated epiphytes, although the photosynthetic activity and respiration rates were gradually reduced for bare seagrass leaves with increasing microplastic concentrations (25-1000 mg MP L-1). A >50% reduction in dark respiration of bare leaves was found at the highest MP exposure, while respiration rates of leaves with epiphytes and separated epiphytes were reduced by maximally ~45 and 30% upon MP exposure, respectively. Short-term microplastic exposure did not alter i) the ability to utilize bicarbonate, ii) the maximum quantum yield of PSII (), nor iii) the light utilization efficiency of Z. marina leaves and associated epiphytes. The compensation irradiance decreased for all investigated specimens, and seagrass leaves (with and without epiphytes) were able to retain a positive net oxygen balance throughout all treatments. We speculate that the observed decrease in photosynthetic activity and respiration was caused by leachates from microplastics. Our findings thus indicate that seagrass Z. marina largely possess resilience toward microplastic pollution at its current level.
Seagrasses are the only true angiosperms in the ocean (Hemminga and Duarte, 2000), and they are considered important ecosystem engineers in marine environments because of their ability to modify abiotic factors (Jones et al., 1997). Seagrass meadows provide ecosystem services that support the overall functionality of the coastal environment (Mtwana Nordlund et al., 2016). This includes water purification by extracting nutrients from the water column (Short and Short, 1984) and coastal protection through current regulation and sediment stabilization (Gambi et al., 1990), providing protection against coastal erosion in many areas (Koch et al., 2009). Furthermore, current deceleration results in enhanced suspended particle sedimentation (Potouroglou et al., 2017), causing large amounts of sequestered carbon (Mcleod et al., 2011; Howard et al., 2017). In addition, seagrass meadows also provide habitats for a range of different marine organisms, ranging from invertebrates to iconic herbivores such as dugongs and sea turtles (Nordlund et al., 2018). Finally, seagrass meadows remain essential in retaining fish stocks, as they provide nurseries for several species, including the Atlantic Cod (Lilley and Unsworth, 2014; Mtwana Nordlund et al., 2016).
Seagrasses have evolved special adaptations to live in marine habitats, such as thin leaves, thin cuticles and large leaf surface area, as they lack stomata and thus rely on diffusion for gas exchange (Kuo and Hartog, 2006). Epiphytic communities residing on seagrass leaves, including algae and heterotrophic bacteria can affect the leaf microenvironment (Brush and Nixon, 2002; Noisette et al., 2020), where e.g., epiphyte cover reduces the light quantity and quality for the underlying leaf and increases the thickness of the diffusive boundary layer (DBL) (Brodersen et al., 2015a; Larkum et al., 2018). This DBL increase around leaves, limits oxygen supplies for respiration, increase levels of pH and lowers the availability for photosynthesis, resulting in reduced photosynthetic efficiency (i.e., O2 photon-1) and increased compensation irradiance (Brodersen et al., 2015a; Brodersen et al., 2020; Zhang et al., 2022). In addition, the internal ratio can decrease during the day, causing increased photorespiration and oxidative stress in the plant tissue (Maberly, 2014). Increased epiphyte cover has also been shown to decrease radial oxygen loss (ROL) from the below-ground tissues, leaving the plant more vulnerable to intrusion (Brodersen et al., 2015a). Ultimately, thick epiphyte cover can induce carbon limitation, as leaf epiphytes and seagrasses are competing for inorganic carbon (DIC) (Brodersen et al., 2020; Zhang et al., 2022). High photosynthetic activity can thus basify the phyllosphere (Brodersen et al., 2020), altering the carbon speciation equilibrium towards unfavorable bicarbonate () and carbonate () ions. The favored source of DIC for seagrasses is in the form of (i.e., can freely diffuse into the tissue), though can be utilized through energy-dependent, carbon concentrating mechanisms (CCM) often utilizing the enzyme carbonic anhydrase (Beer and Rehnberg, 1997). Epiphytic communities on seagrass leaves consist of several different genera of algae, including brown algae, green algae, and diatoms (Borum et al., 1984), and only some of these have effective CCMs (Tortell et al., 1997; Hepburn et al., 2011; Raven et al., 2011). However, seagrasses also host beneficial bacteria that rely on photosynthate excretions from seagrass leaves (Kirchman et al., 1984), and can play an important role in seagrass nutrient acquisition (Capone, 1977; Tarquinio et al., 2018); as well as, phagotrophic epiphytes (such as ectoprocts, sponges and tunicates) may supply CO2, ammonium and phosphate to the leaf microenvironment of aquatic plants (Wong and Vercaemer, 2012).
Anthropogenic activities and climate change threaten seagrass meadows worldwide (Orth and Duarte, 2006). Increasing ocean temperatures decrease the water oxygen content (Staehr and Borum, 2011; Staehr et al., 2018) and the dissolved CO2 (offset by increasing atmospheric CO2), and rising sea levels reduce the light availability for the seagrass leaf canopy (Saunders et al., 2013). Increased nutrient run-off in coastal waters has led to increased eutrophication (Short and Neckles, 1999), causing enhanced growth of epiphytic algae and bacteria on the surfaces of seagrass leaves (Richardson, 2006). In comparison, ocean plastic contamination is a more recently discovered and potentially increasing threat to many marine organisms (Avio et al., 2017). A considerable share of human waste is plastic, and ~10% of the annual global production ends up in the sea (Ryan et al., 2009), causing ~13 billion dollars’ worth of damage to marine ecosystems on a yearly basis (Avio et al., 2017). Breakdown of plastics eventually leads to the formation of microplastic (MP), which is defined as plastic particles under 5 mm in size (Barnes et al., 2009). Plastic production has increased by 43% from 2004 to 2015 (Hermabessiere et al., 2017), and it is expected that microplastic concentrations will increase as a consequence (Prata et al., 2019). Primary sources of MP are derived from the cosmetic industry, plastic pellets and other forms of MP released directly into the ocean (Ryan et al., 2009). Secondary MP results from the breakdown of larger plastic debris through mechanical and photo-oxidative damage (Cole et al., 2011). The effect of plastic debris on marine fauna through ingestion and entanglement has been documented (Wright et al., 2013; Ivar do Sul and Costa, 2014), and plastic leachates have been shown to inhibit the photosynthetic machinery of algal primary producers (Tetu et al., 2019; Dong et al., 2021). The effects of microplastic on seagrasses remains, however, largely unknown.
Seagrasses primarily inhabit coastal zones that are considered the most plastic polluted areas in the sea due to their proximity to the source (Shim et al., 2018). The increased sedimentation rate facilitated by seagrass meadows might also provide a novel ecosystem service, as microplastic can accumulate in the seagrass vegetated sediment (Huang et al., 2020; Jones et al., 2020) and microplastic particles have even been shown to adhere to seagrass leaves (Jones et al., 2020). Furthermore microplastic particles excrete chemical leachates, e.g., additives from the manufacturing process, during their degradation (Hahladakis et al., 2018). Microplastics can also act as vectors for persistent organic pollutants (POP), heavy metals and bacterial pathogens from the surrounding water (Teuten et al., 2009; Wang et al., 2016). It has been shown that the common plastic additive Bisphenol A (BPA), can markedly reduce the photosynthetic capacity in seagrasses (Adamakis et al., 2021), and increasing microplastic leachate concentration has been shown to impair photosynthesis and growth of the cyanobacterium Prochlorocccus (Tetu et al., 2019). Furthermore, microplastic exposure has been shown to impair growth of diazotrophic bacteria (Fernández-Juárez et al., 2021) that can associate with seagrass nutrient acquisition (Capone, 1977; Welsh, 2000). Altogether, microplastics thus might detrimentally influence epiphyte and seagrass photosynthesis and respiration, although such potential effects are still largely undetermined. The effects of microplastics on seagrasses are thus poorly understood, while microplastic concentrations in the ocean (Prata et al., 2019) as well as microplastic trapping in seagrass meadows are increasing (Huang et al., 2020).
In the present study, we investigated the effects of increasing microplastic (PE and PP) concentrations on the photosynthesis and respiration of eelgrass (Zostera marina) and its associated epiphytes. The treatments reflected environmentally relevant microplastic concentrations, but also included extreme events to elucidate potential future risks. Measurements were conducted on seagrass leaves with and without epiphytic biofilm, as well as, on separated epiphytes. This was done to assess the effects of microplastics on: (i) the balance between photosynthesis and respiration, (ii) the photosynthetic activity and efficiency (iii) CCM’s and thereby the utilization of bicarbonate as an inorganic carbon source, and (iv) effects on the photosynthetic capacity of Photosystem II (PSII) in seagrass leaves with and without epiphytes.
Four treatments with microplastic concentrations were set up: 0 (control), 25, 250 and 1000 mg MP L-1. Similar concentrations of MP have been used in other studies on the effects of microplastics on aquatic phototrophs (e.g., Sjollema et al., 2016). To produce microplastic fragments, we used the two most abundant plastic types found in European waters, polyethylene (PE) and polypropylene (PP) in a ratio of 6:4 (Jones et al., 2020). We used common household articles such as black PE garbage bags and PP hard plastic containers (Silvan A/S, Brabrand, Denmark). To make microplastic, garbage bags were cut with scissors, while the harder plastic containers were processed in a bullet blender. For each microplastic treatment, the microplastic particles were suspended in stirred and aerated seawater (16°C, salinity = 20) originating from the seagrass sampling site and were then incubated with a 14h:10h light-dark cycle (photon irradiance of ~350 µmol photons m-2 s-1; 400-700 nm) for three weeks prior to use in the MP treatments of seagrass. This MP pre-conditioning was done to allow for possible biofouling and adsorption of heavy metals and pollutants (Teuten et al., 2009), as well as possible desorption of leachates into the seawater, as seen in studies on microplastic aging in natural environments (Kedzierski et al., 2018). We estimated the mean size of the microplastic particles from three replicates of 50 samples for both PE and PP using an Olympus BX50 microscope connected to a computer with cellSens software (Olympus, Tokyo, Japan), while manually measuring larger pieces of microplastic occurring in the respective replicates by means of a caliper. The average particle size (mean ± SD) was and for PE and PP, respectively. These mean values were well below the upper size limit (5000 μm) in the definition of microplastic (Barnes et al., 2009).
Intact shoots with and without epiphytes, connected to a rhizome fragment with several root bundles from eelgrass (Zostera marina L.) were collected in mid-April 2022. The sampling site was at Julebæk in Northern Zealand, Denmark (56.058972863407355, 12.5759872939143) in shallow, brackish water (salinity of ~20) at one meter depth. The mean seawater temperature in April was ~10°C. The collected plant material was transported to the Marine Biology Section in Elsinore (University of Copenhagen, Denmark), where it was kept in a large, aerated aquarium with water temperatures of ~16°C and a photon irradiance of ~200 μmol photons m-2 s-1 (400-700nm) under a light/dark cycle of 14:10 h, as provided by metal-halide lamps (MEGACHROME, Giesesmann Aquaristic Gmbh, Nettetal, Germany). Seagrass specimens were mounted onto 1.8-mL glass vials filled with sand to keep them stationary and negatively buoyant. The plants were then quickly divided and transferred into the four different treatments with increasing MP concentrations (i.e., 0, 25, 250 and 1000 mg MP L-1) and were then cultured for two weeks prior to measurements under a photon irradiance of ~350 μmol photons m-2 s-1, as provided by tungsten halogen lamps (14h:10h light-dark cycle). The cultivation conditions mimicked the in situ irradiance and temperature conditions at our sampling site at the time of measurements (Staehr and Borum, 2011).
Respiration and net photosynthesis rates were measured as changes in O2 concentration in dark and light, respectively, measured inside small closed glass vials containing either seagrass leaf segments (with and without epiphytes) or separated epiphytes originating from seagrass leaves. The dark respiration was quantified through the net O2 exchange in darkness, and the net photosynthesis under defined photon irradiance levels of 50, 100, 300 and 600 μmol photons m-2 s-1. We used 4 cm segments of seagrass leaves with and without epiphytes, and separated epiphytes scraped off the leaf surface with a scalpel; all from the same shoot to conduct the gas exchange measurements. Each replicate was placed in a measuring chamber (1.8 mL) equipped with an oxygen sensor spot (OXSP5, PyroScience, GmbH, Aachen, Germany) and a small magnet inside the chamber (used for providing water circulation). To prevent mechanical interference from the magnets and damage to the leaf segments and especially separated epiphytes, small pieces of plankton nets (0.2 mm mask width) were placed above the magnets inside each measuring chamber. All four measuring chambers were positioned with the same distance to an adjustable fiber-optic tungsten halogen lamp (KL-2500LCD, Schott GmbH, Germany) with a trifurcated fiber bundle, each equipped with a collimating lens, to ensure that the PAR photon irradiance exposure was the same for all investigated specimens. Prior to measuring, pre-defined photon irradiance levels were established using a calibrated irradiance meter (ULM-500, Walz GmbH, Effeltrich, Germany) with a photon scalar irradiance sensor (3 mm diameter) to determine lamp settings.
The chambers were placed inside an aquarium containing deionized water to maintain a steady temperature inside the chambers during light measurements. The temperature in the aquarium ranged from 16-17°C during the measurements. Below the aquarium, two magnetic stirrers (IKA Magnetic Stirrers RCT basic, IKA®-Werke GmbH and Co. KG, Staufen, Germany) were placed, along with stirring magnets inside the measuring chambers they provided water circulation.
Each measuring chamber was connected to a 4-channel optical sensor system (FireSting-O2; FSO2-C4, PyroScience GmbH, Aachen, Germany) through an optical fiber that read out the sensor spot across the glass wall of the chamber. The sensor system was connected to a PC running the PyroScience Oxygen Logger software (PyroScience GmbH, Germany) that recorded O2 concentration versus time data, as well as the temperature in the aquarium containing the measuring chambers. Each day, prior to measurements, the four measuring chambers were calibrated by sensor readings in two solutions with known oxygen levels: 0 and 100% air saturation. We used sodium sulfite () for the 0% air saturation calibration and filtered (0.2 µm), air saturated seawater for the 100% air saturation calibration.
For each biological replicate, three leaf segments from one seagrass specimen were chosen: One segment without visible epiphytic biofilm (bare leaf, 40 mm length), one segment with visible epiphytic biofilm (leaf with epiphytes, 40 mm length; 100% areal epiphyte cover) and one segment wherefrom associated epiphytes were scraped off with a scalpel (separated epiphytes, originating from 40 mm leaf length with 100% areal epiphyte cover; Figure 1). The seagrass leaves selected for the experiment were visibly healthy (i.e., natural green appearance), excluding the youngest and oldest leaves, and all leaf segments were cut from the middle part of the leaf. This was done to minimize dissimilarities such as age differences between the samples (Brodersen et al., 2020).
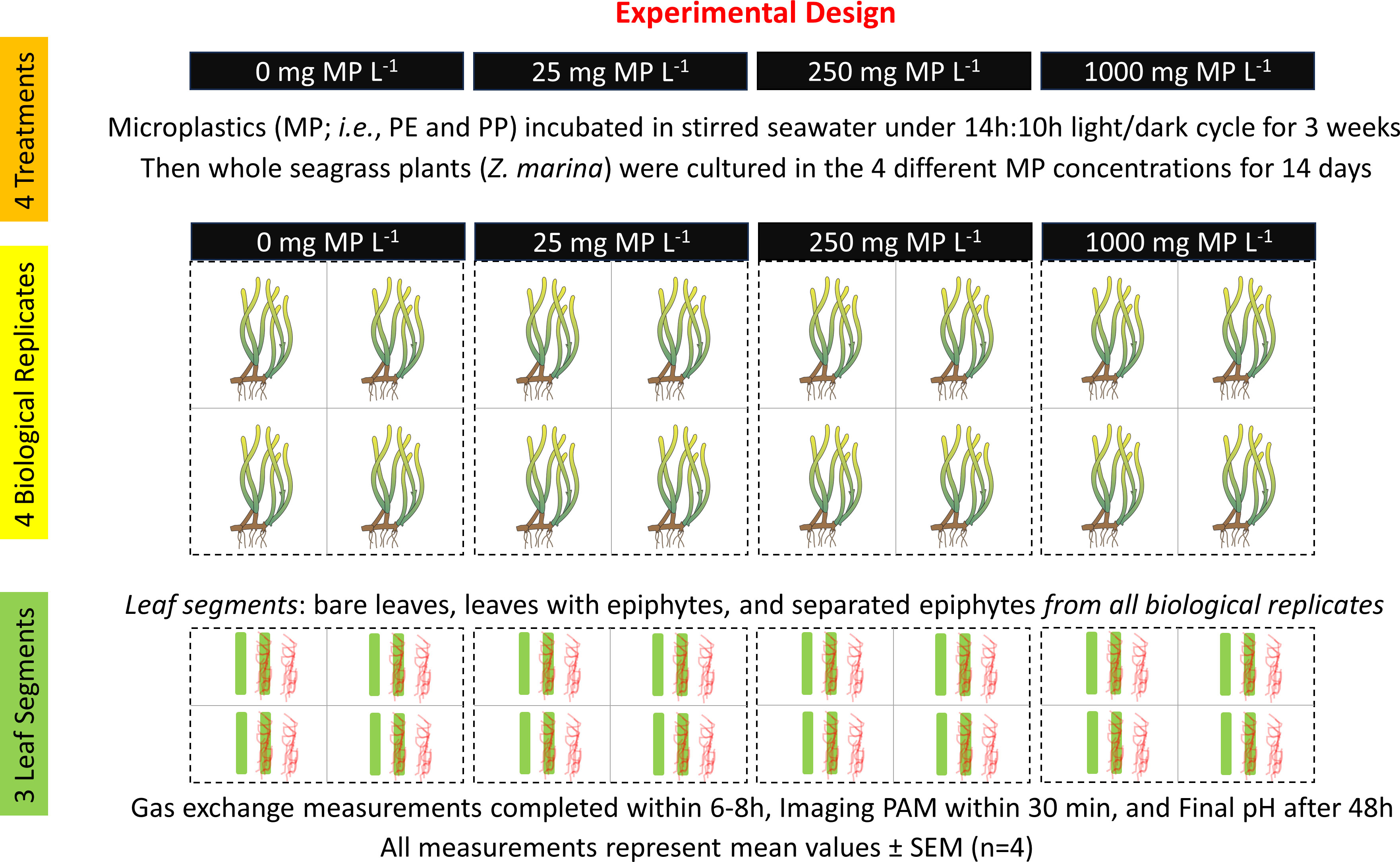
Figure 1 Experimental design. Seagrass illustrations originate from the IAN/UMCES symbol and image libraries (Diana Kleine, Marine Botany UQ (ian.umces.edu/media-library)).
The chosen seagrass leaf segments and separated epiphytes were placed inside the measuring chambers containing air saturated, double filtered seawater (0.2 μm filter size), with a salinity of 20. For each experimental run, there were samples from all leaf community types (i.e., bare leaf, leaf with epiphytes, and separated epiphytes), and a control (i.e., empty measuring chamber). For each MP treatment (i.e., 0, 25, 250 and 1000 mg MP L-1), four biological replicates were done for the gas exchange experiment (n = 4). The specimens were exposed to alternate dark/light cycles at increasing photon irradiances (50, 100, 300, and 600 μmol photons m-2 s-1). For each photon irradiance, the plants were exposed to a 10 min dark period and a 10 min light period. Each experimental run started and ended in darkness. Prior to measurements, the plants were acclimated to total darkness for 10 min to ensure the first measurements accurately represented the dark respiration. This initial dark period lowers the O2 concentration in the measuring chambers to ~70-80% air saturation, which furthermore ensures that the oxygen levels inside the chambers seldom exceeds 100% air saturation during the O2 flux measurements in the following 10 min light/dark periods and thus that the leaves are not likely carbon limited, assuming a photosynthetic quotient of 1 mol O2 produced per mol CO2 fixed.
After each experimental run, the seagrass leaf segments with and without epiphytes and separated epiphytes were transferred to plastic weighing trays, which were placed in a drying oven (60°C) for 24-48 hours prior to measurements. These weight data were used to weight-normalize data and obtain photosynthesis and respiration rates per gram dry weight (g DW-1). The control chambers did not show any change in O2 concentration during the experiments, and thus showed that no undesired production or consumption of O2 occurred within the measuring chambers.
To determine whether seagrass leaves and their associated epiphytes were able to utilize bicarbonate as an inorganic carbon source (DIC) during exposure to MP and MP leachates, final pH measurements were performed in accordance with Brodersen et al. (2020). A final pH of >9 indicates the ability to use bicarbonate as an inorganic carbon source (Borum et al., 2016). For each treatment (i.e., 0, 25, 250 and 1000 mg MP L-1), a total of four biological replicates were done (n=4). Within each biological replicate, three different leaf community types were measured, i.e., leaf without visible epiphyte cover (bare leaf, 60 mm segments), leaf with visible epiphyte cover (leaf with epiphytes, 60 mm segments), and epiphytes scraped from the leaf surface (separated epiphytes, derived from 60 mm segment). Following sampling, every replicate and leaf community type was transferred into separate Falcon tubes (50 mL) with 35 mL of filter-sterilized (0.2µm) seawater with a salinity of 20, leaving a small headspace to minimize possible photorespiration; in accordance with Borum et al. (2016). A separate control sample was run as well (n = 4), making a cluster containing a total of 16 Falcon tubes per treatment. Clusters of Falcon tubes were connected using duct tape, and incubated at saturating light levels (~500 μmol photons m-2 s-1) using tungsten halogen lamps similar to the ones used for the MP treatments. Light levels at the incubation site were determined using a photon scalar irradiance sensor connected to a universal light meter (ULM-500, Walz GmbH, Effeltrich, Germany). Incubations were initiated jointly with the equivalent gas exchange measurements; this was done to ensure the same MP exposure time (i.e., 14 d) and experimental time-period for each treatment. Each cluster of Falcon tubes was incubated for 48 h, before final pH measurements were performed. The combination of 48 h incubation and 6 cm fragments were chosen as a suitable time-period/fragment size, after testing several time/fragment combinations (i.e., 36 h/4 cm, 48 h/4 cm and 48 h/6 cm) for stable maximum pH values. pH levels were measured for each sample using a pH minisensor (BlueLine pH combination electrode, SI Analytics GmbH, Mainz, Germany) connected to a pH meter (PHM220 Lab pH Meter, Radiometer Analytical SAS, Lyon, France). Prior to measurements, the pH meter was calibrated using pH 7 and pH 10 buffers (VWR Chemicals, Pennsylvania, United States).
The maximal quantum yield of photosystem II () was measured on seagrass leaf segments as a proxy for their photosynthetic capacity (Murchie and Lawson, 2013). For this, we applied the saturation pulse method using a variable chlorophyll fluorescence imaging system (IMAGING-PAM M-series, MINI Version, Walz GmbH, Effeltrich, Germany; Ralph et al., 2005), which was connected to a PC running ImagingWin Software (Walz, GmbH, Effeltrich, Germany). For each biological replicate (n = 4) of any given treatment, two samples were imaged: a 4 cm leaf segment with epiphytes and one leaf segment without (bare leaves). The two samples were mounted with tape beside each other in a petri dish and kept in seawater (salinity of 20). The petri dish containing the two leaf community samples was then placed in the Imaging-PAM setup. Prior to imaging, the samples were dark-acclimated for 10 minutes (Beer and Björk, 2000; Brodersen et al., 2015b). The acquisition settings of the ImagingWin software were adjusted to avoid any actinic effect of the measuring light, giving a minimum fluorescence yield of ~0.1 in darkness and avoiding overexposure of the leaf segments and the associated epiphyte community during the saturation pulse measurement. After imaging, values were extracted from defined regions of interest using the ImagingWin software, utilizing nearly all the leaf tissue surface area to get the most accurate chlorophyll fluorescence readings.
Net photosynthesis was calculated by multiplying the linear slopes of the O2 vs time curve (nmol O2 L-1 h-1; extracted from approx. the 5-9 min time interval after exposure to the given light conditions, R2 values ≥ 0.95) for each light condition (i.e., 50, 100, 300 and 600 μmol photons m-2 s-1) with the volume of the measuring chamber (1.8 mL) and divided by the dry weight of the sample (g DW). Respiration rates were calculated in a similar way by selecting linear slopes on the O2 vs time curve (nmol O2 L-1 h-1) representing the initial dark-acclimation period (i.e., dark respiration) and for each post-illumination dark-period (i.e., here used as a proxy for light respiration rates at the investigated light conditions). Gross photosynthesis was calculated for each irradiance level by adding together the respective net photosynthesis rate and the absolute value of the respective respiration rate (i.e., as determined for the initial dark-period as well as each post illumination dark-period):
Photosynthesis vs. irradiance curves were fitted to exponential models using non-linear curve fitting in the software Origin Pro 2017 (Origin Lab Corporation, Northampton MA, United States). For the GP vs. irradiance curves, an exponential saturation model was fitted (Webb et al., 1974):
A similar model was used for the NP vs. irradiance curve fits, with an added term “R”, to account for respiration, in accordance with Spilling et al. (2010):
In the abovementioned saturation models, or NPMAX is the maximal photosynthetic activity at light saturation, is the light utilization efficiency (i.e., the initial slope of the photosynthesis versus irradiance curve) and R(E) is the respiration rate at the respective photon scalar irradiance. These photosynthetic parameters were all derived from the net photosynthesis (NP) saturation models for bare seagrass leaves, leaves with epiphytes and separated epiphytes. All fitted exponential saturation models provided values >0.9 and p ≤ 0.05 (i.e., statistically significant at the 5% significance level), and therefore all fits and their derived photosynthetic parameters were accepted for further analysis. For additional information on statistical analysis of all the exponential saturation models, please see the Supplementary Materials: General Notes and Tables S1-24.
To further evaluate potential effects of microplastics and microplastic leachates on seagrass and their associated epiphytes photosynthetic performance. , the photon scalar irradiance at the onset of light saturation, and , the compensation irradiance, i.e., the photon scalar irradiance at which a zero net production is obtained (NP = 0) were calculated for all three leaf sample types (i.e., bare leaf, leaf with epiphytes and separated epiphytes) and plotted against the microplastic concentration (mg MP L-1). was calculated as:
and was determined as:
To estimate the net daily oxygen production, DP, of seagrass leaves and epiphyte communities we assumed a light/dark exposure of 14/10 h, which is a realistic daily light cycle in mid-April (Staehr et al., 2018). The 14-h exposure was divided into 7 intervals lasting 2 h (i.e., 5-7, 7-9, 9-11, 11-13, 13-15, 15-17, 17-19). A mean photon irradiance was calculated for each time interval and used with the Spilling equation (3) to determine the net photosynthesis. The diel photon irradiance was derived from Staehr et al. (2018). We then calculated the net daily oxygen production (DP) of seagrass leaves and epiphyte communities as a function of increasing microplastic concentration, using as the net oxygen production at a given mean photon irradiance, the dark respiration rate multiplied by 10 hours, and the different predefined time intervals (Hansen et al., 2022):
To make final pH and curves, we calculated mean values using all four replicates of any given treatment. The means were plotted into Origin Pro 2017 to make vs. MP and final pH vs. MP concentration curves, respectively. For the final pH curves, means for leaves with and without epiphytes, as well as, separated epiphytes were used, whereas in the curves, only the means of leaf segments with and without epiphytes were used (as only these two sample types were utilized during the PAM imaging process).
We present first evidence that microplastic particles (PE and PP) have limited effects on the photosynthetic performance of Z. marina leaves and their associated epiphytic communities, and thus indicates relatively high resilience against short-term exposure to high concentrations of microplastics. However, some photosynthetic parameters such as the maximal net photosynthesis and especially the dark respiration rate were both negatively affected in bare seagrass leaves at very high microplastic concentrations.
The obtained values showed a difference between the bare seagrass leaves and the leaves with epiphyte cover, with bare leaves having a higher ratio (~0.73) compared to the leaves with epiphytic cover (~0.64) (Figure 2A). Such photosynthetic capacity of bare seagrass leaves is in line with other studies, that show a similar value (Yang et al., 2017). The lower of leaves with epiphytes is to be expected, as it represents a mixed signal between the seagrass leaf and the associated epiphytes. Generally, there were only minor differences in the obtained FV/FM values between the different microplastic treatments (Figure 2A). However, we found a decrease in the ratio for leaves with epiphytes incubated with 25 mg MP L-1 when compared to the control (Student’s t-test: p = 0.002; Figure 2A). In addition, there was an approximate increase of 3% in the ratio for bare leaves cultured at 250 and 1000 mg MP L-1 compared to the control (p = 0.03 and p = 0.103, respectively; Figure 2A). Although there were minor changes in ratios, these remained within normal fluorescence values for seagrass leaves (Prange and Dennison, 2000; Yang et al., 2017). Therefore, our results do not indicate a negative effect of short-term microplastic exposure on the capacity of photosystem II in seagrass leaves and associated epiphytes.
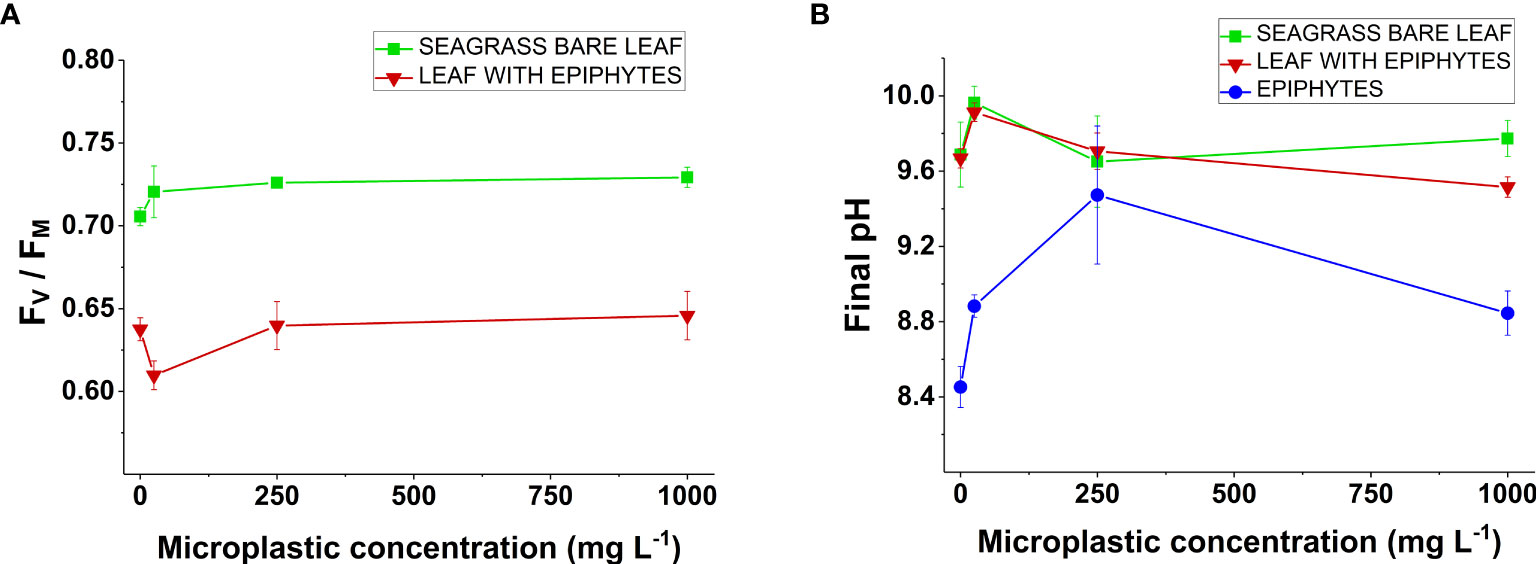
Figure 2 (A) Variable chlorophyll fluorescence of Z. marina. ratios as a function of increasing microplastic concentration (0, 25, 250, and 1000 mg MP L-1). Measurements were conducted on seagrass leaves with (red line) and without epiphytes (green line). Symbols with error-bars represent mean values ± SEM. Four biological replicates (n = 4). (B) Final pH values of Z. marina as a function of increasing microplastic concentration (0, 25, 250, and 1000 mg MP L-1). Measurements were conducted on seagrass leaves with (red line) and without epiphytes (green line), as well as on separated epiphytes (blue line). Symbols with error-bars represent mean values ± SEM. Four biological replicates (n = 4).
The common plastic additive Bisphenol A (BPA) has been found to decrease the effective quantum yield of photosystem II and increase the level of reactive oxygen species in seagrass leaves (Adamakis et al., 2021). However, we did not investigate the presence of BPA in the plastic polymers used for this study. In effect, plastic products containing high amounts of BPA might thus have given a different response. Furthermore, no microplastic particles were observed directly on the leaf samples used for variable chlorophyll fluorescence imaging, although this could be due to handling and processing, washing off any particles present. Other studies from Northern Europe found microplastic adhering to nearly every leaf examined (Jones et al., 2020). Hence, adherence is to be expected in situ, possibly altering the local ratio due to effects of shading and possible contamination.
A final pH above 9 was found in Z. marina leaves with and without epiphytes for all microplastic and control treatments (Figure 2B). In accordance with previous studies, this demonstrates that Z. marina can utilize as an inorganic carbon source (Beer and Rehnberg, 1997; Brodersen et al., 2020; Hansen et al., 2022). Additionally, our pH drift curves showed no profound changes in any of the microplastic treatments for leaves with and without epiphytes. Therefore, short-term microplastic exposure did not affect the ability to utilize bicarbonate via carbon concentrating meachanisms (CCM). CCM’s are energy-dependent processes (Larkum et al., 2017), which have been shown to be compromised in stress related scenarios, such as excessive heat exposure (Hansen et al., 2022). For separated epiphytes, the final pH was below 9 for nearly all treatments (Figure 2B). In general, this is in accordance with previous studies showing ineffective CCM’s in filamentous algae (i.e., similar to the seagrass microalgal epiphyte community, which largely consists of a mixture of brown algae, green algae, and diatoms, including several filamentous microalgae (Borum et al., 1984)) (Hepburn et al., 2011; Raven et al., 2011). Specimens cultured at 250 mg MP L-1 reached a final pH above 9, but these measurements showed a large variability (Figure 2B). This might be explained by the diversity of algal communities on seagrass leaves (Borum et al., 1984), and the varying ability to utilize among microalgal epiphytes (Tortell et al., 1997; Hepburn et al., 2011; Raven et al., 2011).
The gross photosynthesis (Figure 3A–L) increased with increasing photon scalar irradiance until saturation and was the highest for bare seagrass leaves exposed to 250 mg MP L-1 (Figure 3G), and lowest for separated epiphytes cultured at 25 mg MP L-1 (Figure 3F). There was a decrease in the respiration rates with increasing photon scalar irradiance for most specimens, except in separated epiphytes that remained relatively stable throughout all treatments. The net photosynthesis rate (Figures 3A–L) of all specimens also increased with increasing photon scalar irradiance, until saturation. The highest net photosynthesis rate was found in bare leaves exposed to 250 mg MP L-1 (Figure 3G) and the lowest was found in separated epiphytes exposed to 25 mg MP L-1 (Figure 3F). In general, all separated epiphyte samples exhibited low photosynthetic activity, which could indicate a high proportion of heterotrophic bacteria and dead or inactive algal biomass in the leaf epiphytic biofilm.
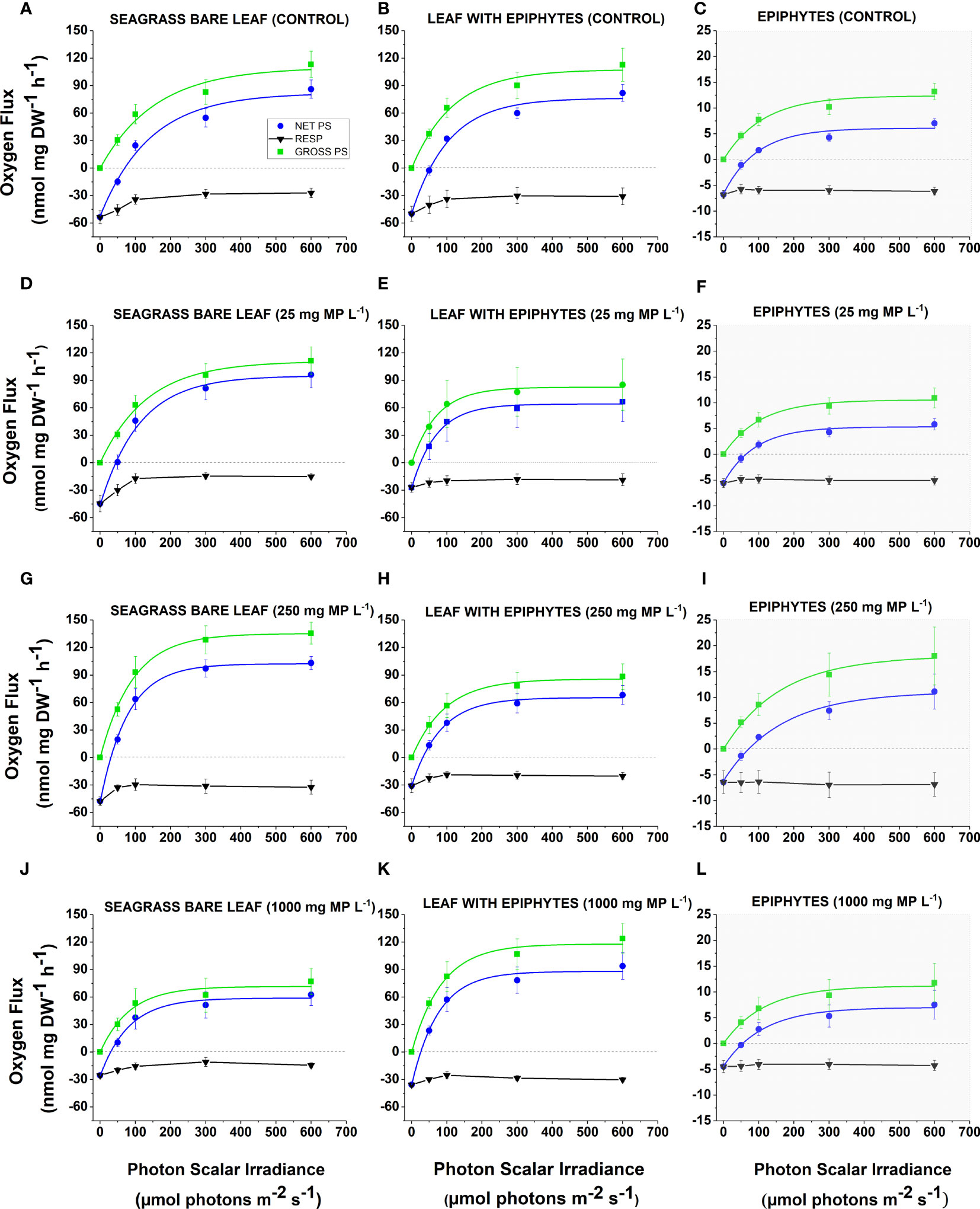
Figure 3 (A-L) Respiration, net and gross O2 production as a function of photon irradiance in Z. marina leaves with and without epiphytes, as well as separated epiphytes under different microplastic exposure. The different microplastic treatments were control (A–C), 25 mg MP L-1 (D–F), 250 mg MP L-1 (G, H, I) and 1000 mg MP L-1 (J–L). Black symbols and lines represent respiration. Blue symbols and lines represent net photosynthesis. Green symbols and lines represent gross photosynthesis. Grey graphs (C, F, I, L) show different y-axis scaling, as epiphyte values were markedly lower. Symbols with error bars show the mean values ± SEM. Four biological replicates (n = 4). The non-linear regression curve fits all had an R2 ≥ 0.90 and p ≤ 0.05 (see Supplementary Methods, Table S1-S24).
Maximal net photosynthesis rates (NPmax) were generally lower for leaves with epiphytes compared to the bare seagrass leaves except for specimens exposed to 1000 mg MP L-1 (Figure 4A). Previous studies have shown lower photosynthetic activity for leaves with epiphytic cover, due to a thicker diffusive boundary layer (DBL) limiting gas and nutrient exchange with the surrounding water-column (Brodersen et al., 2015a; Brodersen et al., 2020; Noisette et al., 2020). NPmax for bare seagrass leaves exposed to 1000 mg MP L-1 decreased ~30% compared to the control (Figure 4A). This is in line with studies showing that microplastics can inhibit the photosynthetic machinery of several algal species (Tetu et al., 2019; Dong et al., 2021).
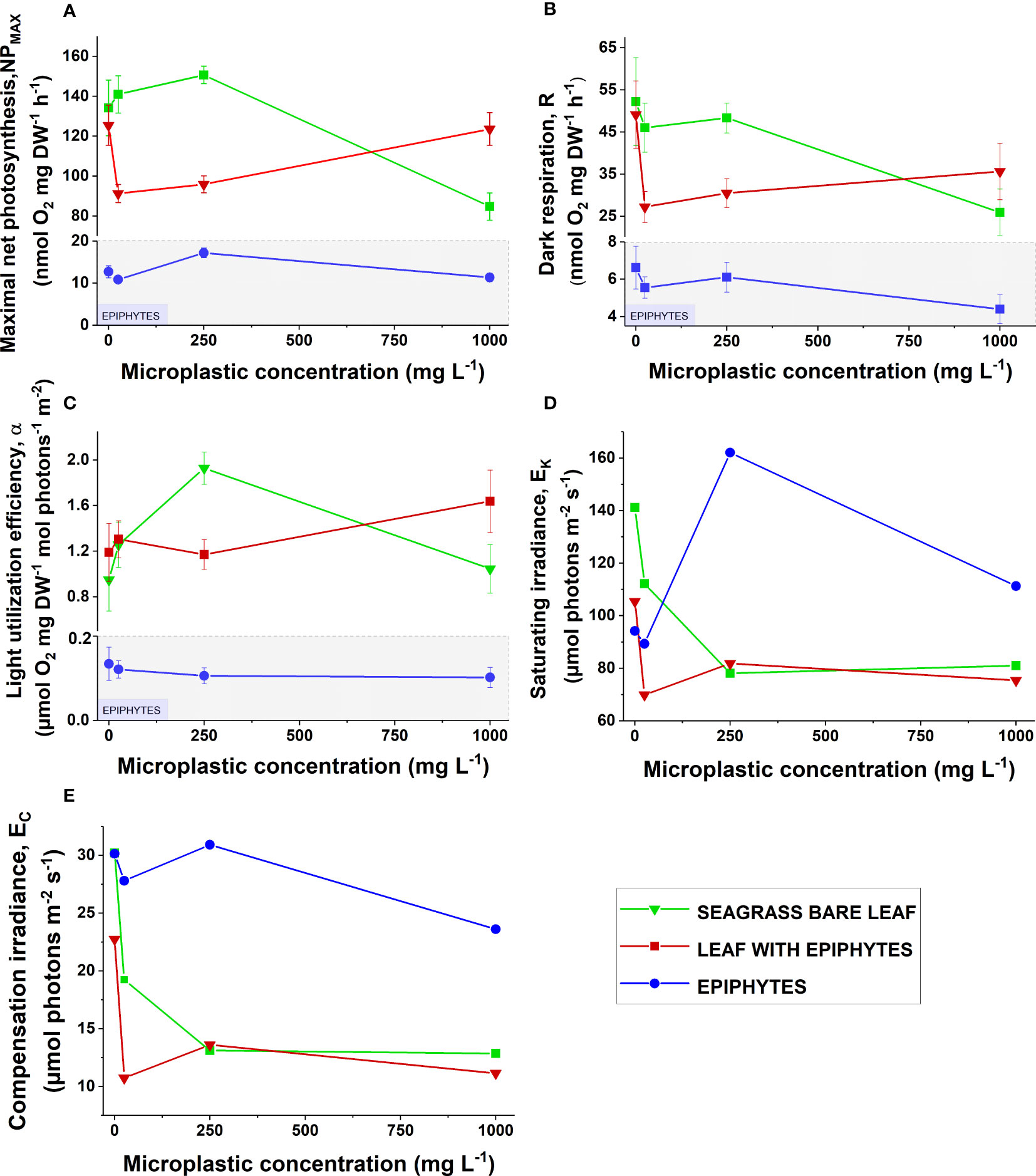
Figure 4 (A) Maximum photosynthetic rate (), (B) dark respiration (R), (C) light utilization efficiency (), (D) photon irradiance at the onset of photosynthesis saturation () and (E) compensation photon irradiance () as a function of increasing microplastic concentrations (0, 25, 250, and 1000 ). Green symbols and lines show bare seagrass leaves. Red symbols and lines show leaves with epiphytes. Blue symbols and lines show separated epiphytes. In subfigures (A-C), areas in gray show a different scale on the y-axis, as values for separate epiphytes were markedly lower. Symbols with error bars represent mean values ± SEM. Four biological replicates (n = 4).
Leachates from microplastics can inhibit photosynthesis and growth in cyanobacteria (Tetu et al., 2019), and in a study on the freshwater green alga Chlamydomonas reinhardtii, polystyrene microplastics caused photosynthetic inhibition, repressed RUBISCO activity and reduced respiration (Dong et al., 2021). Although these studies focused on microalgae, a similar response could occur in bare seagrass leaves at the highest microplastic concentration. An additional explanation to the determined decrease in NPmax for bare seagrass leaves exposed to 1000 mg MP L-1 could relate to heavy metals such as copper that might adhere to the microplastic surfaces. Copper has been shown to have acute toxic effects on other seagrass species, dramatically decreasing the photosynthetic capacity and increasing the amount of reactive oxygen species in the leaf tissue (Buapet et al., 2019). However, we found no significant negative effect of microplastics on the photosynthetic capacity (Fv/Fm) of Z. marina (Figure 2A). For seagrass leaves with epiphytes, there was a marked decrease in NPmax from the control to microplastic concentration exposures of 25 and 250 mg MP L-1, which ceased at 1000 mg MP L-1 where NPmax was similar to the control (Figure 4A). Thus, we found no clear evidence of microplastic-induced reduced photosynthesis in leaves with epiphytes, as the NPmax at the highest concentration of microplastic exposure was relatively unaffected.
It has been suggested that decreased epiphyte activity due to microplastic contamination could be beneficial to seagrasses (Gerstenbacher et al., 2022), as epiphytes are competing with seagrasses for nutrients and light (Brodersen et al., 2015; Brodersen et al., 2020; Brodersen and Kühl, 2022). In line with this, other studies on microalgae show inhibitory effects on growth and photosynthesis when exposed to high microplastic concentrations (Tetu et al., 2019; Dong et al., 2021). However, we found no clear evidence regarding NPmax and short-term microplastic exposure for separated epiphytes, and there was no apparent correlation between microplastic exposure and photosynthetic activity for leaves with epiphytes and separate epiphytes.
The dark respiration for bare seagrass leaves was reduced by ~50% in the 1000 mg MP L-1 treatment compared to the control (Figure 4B). Additionally, separated epiphytes showed a decrease in dark respiration from the control to 1000 mg MP L-1 of ~30% (Figure 4B). A pronounced decrease in dark respiration for leaves with epiphytes was observed, from the control to the 25 mg MP L-1 treatment of ~40% (Figure 4B). However, at a microplastic concentration of 1000 mg MP L-1, the dark respiration was approximately the same as in the control treatment. Microplastics have previously been shown to inhibit respiration in microalgae (Dong et al., 2021). The measured decrease in respiration rates might therefore be caused by respiratory inhibition in the seagrass leaf and/or its associated microbiome. For example, some strains of diazotrophic bacteria can be negatively affected by high concentrations of microplastics (Fernández-Juárez et al., 2021).
Seagrasses host diverse communities of epiphytic bacteria that are highly active (i.e., can display similar respiration rates as bare seagrass leaves and can produce high amounts of toxic nitric oxide within hypoxic/anoxia phyllospheres via incomplete denitrification; Larkum et al., 2018a; Brodersen and Kühl, 2022; Hansen et al., 2022) and a large proportion of the observed respiratory drop may thus be assigned to the impairment of leaf resident bacteria. Impaired photosynthesis in the seagrass leaf (i.e., NPmax; as seen in Figure 4A) might also decrease the amount of photosynthate excretions available for bacteria in the seagrass phyllosphere (see e.g., Moriarty et al., 1986; Pollard and Moriarty, 1991), resulting in reduced bacterial respiration. For seagrass photosynthesis, nitrogen is a limiting factor (as photosynthesis is a nitrogen demanding process, needing nitrogen for enzyme synthesis) and the ability to produce carbohydrates for mitochondrial respiration depends on it (Amthor, 1995). A decrease in NPmax, as shown for the highest microplastic concentration, and potential stimuli of microorganisms facilitating nitrogen uptake through the leaves, might therefore also to some extent help explain the observed decrease in respiration.
Short-term microplastic exposure showed no profound effect on the light utilization efficiency () expect for bare seagrass leaves exposed to 250 mg MP L-1, where the light utilization efficiency increased markedly compared to the control (Figure 4C). The light utilization efficiency was lowest for separated epiphytes (Figure 4C). In general, the light utilization efficiency of the leaf is expected to be higher for bare seagrass leaves compared to leaves with epiphytes, as epiphyte-cover reduces the light quality and quantity reaching the leaf surface (Brodersen et al., 2015a). However, this was not observed in our study, and might be explained by a relatively thin epiphyte-cover, as the sampled plants were collected early in the season for leaf epiphytic blooms (Borum et al., 1984). Finally, the light utilization efficiency of leaves with epiphytes was higher than that of the bare leaf in the 1000 mg MP L-1 treatment, possibly due to reduced competition with associated microalgal epiphytes, caused by leachate-related impairment (Tetu et al., 2019); in combination with a prior shade-induced optimization of the leaf photosynthetic apparatus for limiting light conditions (via the epiphyte cover), such as increased total chlorophyll and antenna chlorophyll to PSII in leaves (e.g., Ralph et al., 2007; Larkum et al., 2018b).
The photon irradiance at the onset of photosynthesis saturation () decreased for seagrass leaves with and without epiphytes with increasing microplastic concentrations, while the epiphyte increased (Figure 4D). The bare leaf decreased by ~50% in the 1000 mg MP L-1 treatment compared to control, and the leaf with epiphyte decreased by ~25%. The for separated epiphytes increased ~40% from the control to the 250 mg MP L-1 treatment, and about 25% in the 1000 mg MP L-1 treatment compared to the control. Effectively, this means that leaves with and without epiphytes in the 1000 mg MP L-1 treatment required less photon irradiance to reach the onset of photosynthesis saturation than that of the control, and the epiphytes required more light. The changes in saturation irradiance were most likely caused by fluctuations in the maximal net photosynthesis rates, NPmax (Figure 4A), as the light utilization efficiency in general did not change substantially (Figure 4C).
The compensation irradiance , i.e., the photon irradiance at which a net zero oxygen balance is obtained, generally decreased for all specimens with increasing microplastic concentration (Figure 4E). Bare leaf was affected the most, decreasing by ~50% compared to the control treatment, which fitted well with the comparable reduction in dark respiration. In the control treatment, bare leaves and separated epiphytes had similar compensation photon irradiances, in contrast to in leaves with epiphytes where compensation irradiances were markedly lower. Thus, a lower photon irradiance was generally needed for all specimens to retain a positive oxygen balance with increasing MP concentrations due to the reduced respiration rates. We expected seagrass leaves with epiphytes to have higher compensation irradiances than bare leaves, due to the impediment of light availability and gas exchange with the surrounding environment caused by the epiphytes (Brodersen et al., 2015). However, this was not the case in our study and might be explained by thin epiphytic cover on most of the sampled plants, as they were collected early in the season for epiphytic blooms (Borum et al., 1984). Altogether, bare seagrass leaves were affected the most by microplastic exposure as seen in the major decrease in both and , which was largely driven by the decrease in the photosynthetic activity (i.e., NPmax) and respiration rate. Lower consumption within the phyllosphere, possibly caused by respiratory inhibition of seagrasses and the associated epibiome (Dong et al., 2021), can thus reduce the irradiance level needed to attain a positive oxygen balance in microplastic exposed Z. marina plants.
The estimated net diel oxygen budget (Figure 5) provides insight into the balance of the bare seagrass leaves, leaves with epiphytes and separated epiphytes exposed to increasing microplastic concentrations, assuming a 14/10h light/dark-cycle. For all specimens, there was an increase in the net diel oxygen production of the seagrass leaves and epiphyte communities from the control to the microplastic treatments. Leaves with and without epiphyte cover had the highest net diel oxygen production and remained in surplus throughout all microplastic treatments (Figure 5). Separated epiphytes in the control and the 25 mg MP L-1 treatment, had a slightly negative net diel oxygen balance (Figure 5). However, at microplastic concentrations of 250 and 1000 mg MP L-1, there was an oxygen surplus (Figure 5). The net diel oxygen production of separated epiphytes was markedly lower than leaves with and without epiphytes. In other studies on seagrass photosynthetic rates, net diel oxygen production of epiphytes was dominant compared to seagrass leaves with and without epiphytes (Hansen et al., 2022). In this study, the lower performance of separated epiphytes was most likely caused by a dead/inactive leaf epiphytic biofilm with relatively high biomass of heterotrophic bacteria.
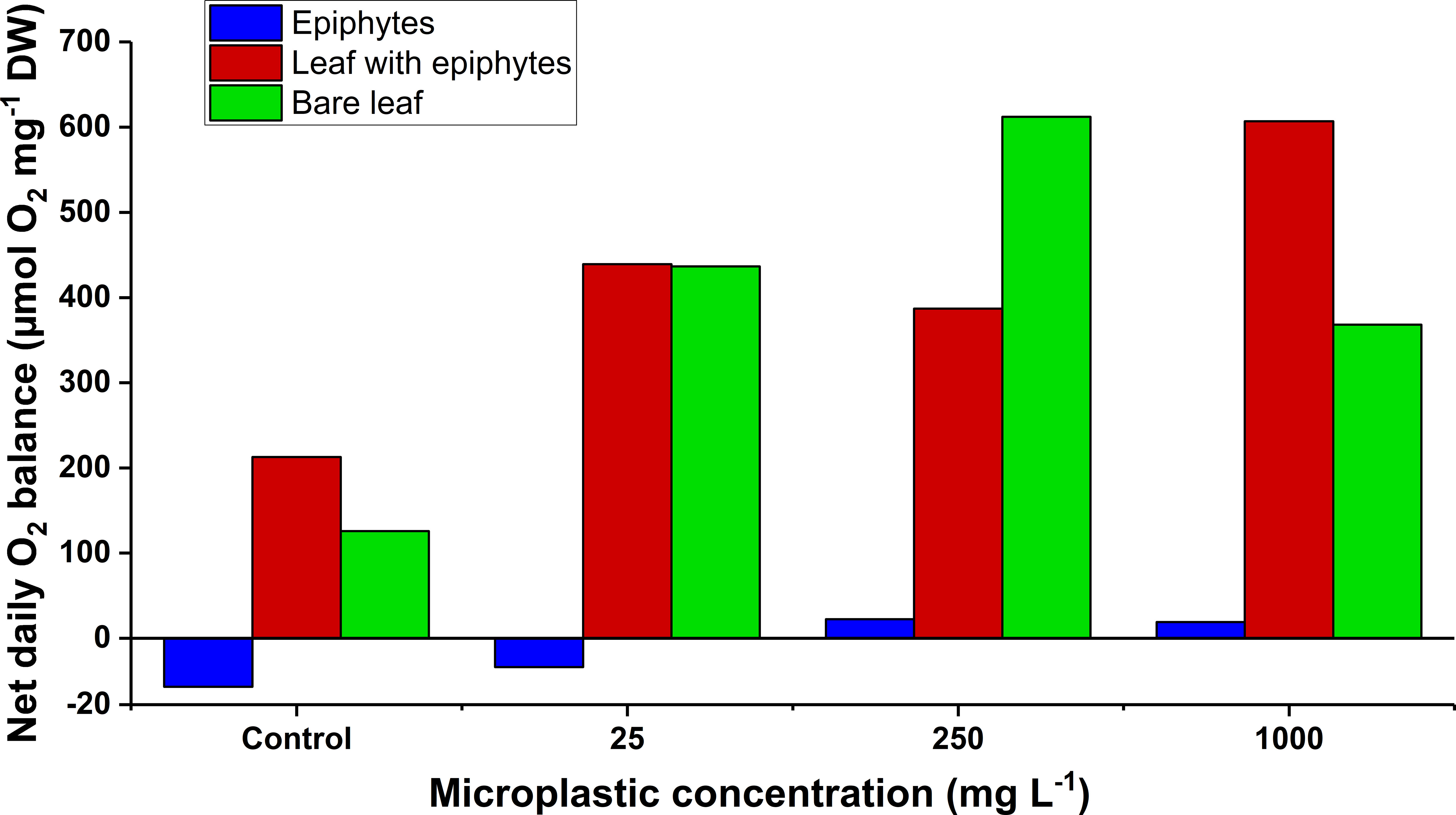
Figure 5 The net daily oxygen production as a function of increasing microplastic concentrations (control, 25, 250, and 1000 mg MP L-1) of the leaf and epiphyte communities. Blue bars represent epiphytes originating from Z. marina leaves. Red bars represent Z. marina leaves with epiphyte cover. Green bars represent bare Z. marina leaves. The net daily oxygen production was estimated from previously fitted exponential saturation models (n = 4) at increasing microplastic concentrations (Figure 3, all with ≥ 0.90 and p ≤ 0.05) using a14 h:10 h light/dark cycle measured in situ (Staehr et al., 2018). The black line at y = 0 demonstrates a neutral carbon balance. Slightly different scaling on the y-axis below y = 0, is due to markedly lower values for separate epiphytes.
The general increase of net diel oxygen production of leaves and epiphyte communities with microplastic exposure is in accordance with the observed drop in dark respiration, and . Lowered respiratory demands reduces the amount of produced oxygen needed to attain a net zero oxygen balance, and a lower compensation- and saturation irradiance reduces the amount of light needed to achieve this oxygen status. The increase in net diel oxygen production is therefore to a lesser extent due to increased photosynthetic activity or capacity (as seen in NPmax and ), but rather a result of the pronounced decrease in dark respiration rates. We speculate that this decrease in dark respiration could be related to a combination of: (i) respiratory inhibition of seagrass and associated epiphytes as previously shown in microalgae (Dong et al., 2021), and (ii) a decrease in the microbial respiration as microplastic previously has been shown to negatively affect bacteria that could be associated with seagrasses (Fernández-Juárez et al., 2021). Seagrasses rely on diffusion for gas exchange (Kuo and Hartog, 2006); therefore, the decreased respiration within the phyllosphere might improve the internal plant aeration and thus increase the radial oxygen loss (ROL) into the sediment and thereby the below-ground tissues oxidation capacity (Brodersen et al., 2017; Brodersen et al., 2018). However, such increase in DP might only benefit seagrass health and performance when living under environmental stress conditions increasing the risk of inadequate internal aeration (such as water deoxygenation during night-time). Furthermore, if part of the decrease in respiration can be attributed to decreased bacterial activity, of which some are important for seagrass nutrient acquisition (Capone, 1977; Larkum et al., 2018a), this might influence nutrient availability. As seagrasses can be limited by nitrogen (Amthor, 1995), this could reduce growth and performance in the long-term. In addition, it is important to emphasize that these measurements represent DPs of leaves and epiphyte communities, and thus do not account for the respiratory demand of non-photosynthetic seagrass tissues (such as rhizome and roots) that can account for 30-80% of the total plant biomass depending on species (Shafer and Kaldy, 2014).
Our results indicate resilience toward short-term, acute microplastic exposure on seagrass leaves and their associated epiphytic communities. To further evaluate microplastic effects on seagrasses, investigations on different microplastic types need to be performed. Long-term exposure of seagrass leaves to microplastic pollution should also be conducted, thus enabling leaf exposure to higher concentrations of leaching/vectored contaminants, as well as potential microplastic adhesion to leaf and epiphyte tissue surfaces. Although microplastic pollution is mostly fluctuating in the water-column owing to the largely positive buoyancy of particles, thus mainly exposing coastal marine organisms to microplastics for short time periods. However, within seagrass meadows prolonged exposure can also be expected owing to the pronounced sedimentation effect caused by the dense leaf canopy (Huang et al., 2020; Jones et al., 2020). Furthermore, it has been suggested that seagrass beds provide a novel ecosystem service by trapping microplastic (Huang et al., 2020). Microplastics can thus accumulate in the sediment of seagrass meadows (Huang et al., 2020; Jones et al., 2020), in which diazotrophs important for carbon and nutrient recycling reside (Welsh, 2000; Nielsen et al., 2001; Larkum et al., 2018a). Some strains of diazotrophic bacteria can be negatively affected by microplastics (Fernández-Juárez et al., 2021). Similar impairment of seagrass sediment diazotrophs might therefore upset the nutrient and carbon balance in situ, and adversely affect the performance of seagrass meadows. Therefore, future investigations on seagrass sediment and bacterial activity could be vital to understand the full extent of microplastic contamination in seagrass ecosystems. Recent studies have even suggested that microplastic trapping adds to the carbon sequestration pool of seagrass meadows (Huang et al., 2020). However, this novel ecosystem service of microplastic trapping might adversely affect organisms associated with seagrasses, such as sediment infauna. Seagrass beds are considered biodiversity hotspots that many organisms live- and feed on (Nordlund et al., 2018) and seagrasses have therefore been identified as potential vectors for microplastics entering the food web (Goss et al., 2018). Thus far, studies have found microplastic ingestion in many marine organisms (Wright et al., 2013; Ivar do Sul and Costa, 2014), but the long-term effects remain unclear. Therefore, elucidating the effects of microplastic on marine flora and fauna associated with seagrass beds, as well as the role of seagrasses in transferring microplastic into the food web, still needs to be studied in detail.
This study presents first experimental evidence that short-term exposure to microplastic (PE and PP) has limited effects on the seagrass Z. marina and its associated epiphytes. The photosynthetic capacity and efficiency of seagrass leaves and their associated epiphytes were not adversely affected at the concentrations and types of microplastics used here. Seagrasses in all treatments were able to utilize bicarbonate and CCMs were not affected by microplastics. The photosynthetic activity of bare seagrass leaves markedly decreased at very high microplastic concentrations, as opposed to leaves with epiphyte cover and epiphytes themselves possibly due to toxic leachates and heavy metal adsorption abilities of microplastic particles that have previously been shown to have detrimental effects on other photosynthetic organisms. Dark respiration decreased for nearly all specimens at high microplastic concentrations, and we speculate this decrease to be caused by leachate-related respiratory inhibition in seagrasses and/or leaf resident microbes. Respiration constitutes series of enzyme-driven reactions that allow seagrass to convert stored carbohydrate energy (e.g., in the rhizome) made by photosynthesis into energy they can utilize for important metabolic processes and growth, therefore such marked reductions in rates of respiration may affect seagrass growth and performance in the longer perspective. However, in this study where we used leaf segments the reduced respiration rates were rather related to reduced maintenance processes (e.g., protein synthesis, replacement of disassembled proteins after damage and/or recovery of leaked solutes), or at least in part to reduced energy (respiratory substrates) by e.g., routing through the alternate oxidase after mitochondrial redox complexes (Rasmusson et al., 2017). The decrease in dark respiration also caused a decrease in the compensation irradiance and increased the surplus of the net diel oxygen production. However, this may not benefit seagrass health and performance in the long term if important symbionts disappear from the leaf surface. Microplastic contamination is a novel threat to marine ecosystems, and our findings improve the still scarce knowledge base of seagrass-microplastic interactions. While Z. marina exhibited high resilience toward microplastic contamination, except at very high concentrations that remain unlikely in the natural environment, seagrass beds can trap microplastic particles with potential negative consequences for seagrass-associated organisms. But such negative effects of microplastic accumulation in seagrass meadows remain to be demonstrated.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
KB designed the research and supervised all experimental work. MK and PH provided infrastructure, additional supervision, and input to data analysis. JM and WG-A conducted the experiments, analyzed the data, and wrote the manuscript with editorial help from MK, PH and KB. All authors contributed to the article and approved the submitted version.
This study was funded by grants from the Villum Foundation (00028156; KB) and the Independent Research Fund Denmark (DFF-8021-00308B; MK).
We thank Peter A. Staehr for providing the light data used to calculate the diel O2 budgets.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2023.1216299/full#supplementary-material
Adamakis I.-D. S., Malea P., Sperdouli I., Panteris E., Kokkinidi D., Moustakas M. (2021). Evaluation of the spatiotemporal effects of bisphenol A on the leaves of the seagrass Cymodocea nodosa. J. Hazard. Mater. 404, 124001. doi: 10.1016/j.jhazmat.2020.124001
Amthor J. S. (1995). “Higher plant respiration and its relationships to photosynthesis,” in Ecophysiology of photosynthesis. Eds. Schulze E.-D., Caldwell M. M. (Berlin, Heidelberg: Springer), 71–101.
Avio C. G., Gorbi S., Regoli F. (2017). Plastics and microplastics in the oceans: From emerging pollutants to emerged threat. Mar. Environ. Res. 128, 2–11. doi: 10.1016/j.marenvres.2016.05.012
Barnes D. K. A., Galgani F., Thompson R. C., Barlaz M. (2009). Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B: Biol. Sci. 364 (1526), 1985–1998. doi: 10.1098/rstb.2008.0205
Beer S., Björk M. (2000). Measuring rates of photosynthesis of two tropical seagrasses by pulse amplitude modulated (PAM) fluorometry. Aquat. Bot. 66 (1), 69–76. doi: 10.1016/S0304-3770(99)00020-0
Beer S., Rehnberg J. (1997). The acquisition of inorganic carbon by the seagrass Zostera marina. Aquat. Bot. 56 (3), 277–283. doi: 10.1016/S0304-3770(96)01109-6
Borum J., Kaas H., Wium-Andersen S. (1984). Biomass variation and autotrophic production of an epiphyte-macrophyte community in a coastal Danish area: II. Epiphyte species composition, biomass and production. Ophelia 23, 165–179. doi: 10.1080/00785326.1984.10426612
Borum J., Pedersen O., Kotula L., Fraser M. W., Statton J., Colmer T. D., et al. (2016). Photosynthetic response to globally increasing CO2 of co-occurring temperate seagrass species. Plant Cell Environ. 39 (6), 1240–1250. doi: 10.1111/pce.12658
Brodersen K., Koren K., Moßhammer M., Ralph P., Kühl M., Santner J. (2017). Seagrass-mediated phosphorus and iron solubilization in tropical sediments. Environ. Sci. Technol. 51 (24), 14155–14163. doi: 10.1021/acs.est.7b03878
Brodersen K. E., Koren K., Revsbech N. P., Kühl M. (2020). Strong leaf surface basification and CO2 limitation of seagrass induced by epiphytic biofilm microenvironments. Plant Cell Environ. 43 (1), 174–187. doi: 10.1111/pce.13645
Brodersen K. E., Kühl M. (2022). Effects of epiphytes on the seagrass phyllosphere. Front. Mar. Sci. 9, 821614. doi: 10.3389/fmars.2022.821614
Brodersen K. E., Kühl M., Nielsen D. A., Pedersen O., Larkum A. W. D. (2018). “Rhizome, root/sediment interactions, aerenchyma and internal pressure changes in seagrasses,” in Seagrasses of Australia: Structure, Ecology and Conservation (Switzerland: Springer International Publishing. Springer Nature, Cham), 393–418.
Brodersen K., Lichtenberg M., Paz L.-C., Kühl M. (2015a). Epiphyte-cover on seagrass (Zostera marina L.) leaves impedes plant performance and radial O2 loss from the below-ground tissue. Front. Mar. Sci. 2. doi: 10.3389/fmars.2015.00058
Brodersen K. E., Nielsen D. A., Ralph P. J., Kühl M. (2015b). Oxic microshield and local pH enhancement protects Zostera muelleri from sediment derived hydrogen sulphide. New Phytol. 205 (3), 1264–1276. doi: 10.1111/nph.13124
Brush M., Nixon S. W. (2002). Direct measurements of light attenuation by epiphytes on eelgrass. Zostera marina. Mar. Ecol. Prog. Ser. 238, 73–79. doi: 10.3354/meps238073
Buapet P., Mohammadi N. S., Pernice M., Kumar M., Kuzhiumparambil U., Ralph P. J. (2019). Excess copper promotes photoinhibition and modulates the expression of antioxidant-related genes in Zostera muelleri. Aquat. Toxicol. 207, 91–100. doi: 10.1016/j.aquatox.2018.12.005
Capone D. T. B. (1977). Nitrogen fixation (acetylene reduction) in the phyllosphere of Thalassia testudinum. Mar. Biol. 40, 19–28. doi: 10.1007/BF00390623
Cole M., Lindeque P., Halsband C., Galloway T. S. (2011). Microplastics as contaminants in the marine environment: A review. Mar. Pollut. Bull. 62 (12), 2588–2597. doi: 10.1016/j.marpolbul.2011.09.025
Dong Y., Gao M., Qiu W., Song Z. (2021). Effects of microplastic on arsenic accumulation in Chlamydomonas reinhardtii in a freshwater environment. J. Hazard. Mater. 405, 124232. doi: 10.1016/j.jhazmat.2020.124232
Fernández-Juárez V., López-Alforja X., Frank-Comas A., Echeveste P., Bennasar-Figueras A., Ramis-Munar G., et al. (2021). “The good, the bad and the double-sword” Effects of microplastics and their organic additives in marine bacteria. Front. Microbiol. 11. doi: 10.3389/fmicb.2020.581118
Gambi M. C., Nowell A. R. M., Jumars P. A. (1990). Flume observations on flow dynamics in Zostera marina (eelgrass) beds. Mar. Ecol. Prog. Ser. 61 (1/2), 159–169. doi: 10.3354/meps061159
Gerstenbacher C. M., Finzi A. C., Rotjan R. D., Novak A. B. (2022). A review of microplastic impacts on seagrasses, epiphytes, and associated sediment communities. Environ. Pollut. 303, 119108. doi: 10.1016/j.envpol.2022.119108
Goss H., Jaskiel J., Rotjan R. (2018). Thalassia testudinum as a potential vector for incorporating microplastics into benthic marine food webs. Mar. Pollut. Bull. 135, 1085–1089. doi: 10.1016/j.marpolbul.2018.08.024
Hahladakis J. N., Velis C. A., Weber R., Iacovidou E., Purnell P. (2018). An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater. 344, 179–199. doi: 10.1016/j.jhazmat.2017.10.014
Hansen A. B., Pedersen A. S., Kühl M., Brodersen K. E. (2022). Temperature effects on leaf and epiphyte photosynthesis, bicarbonate use and diel O2 budgets of the seagrass zostera marina L. Front. Mar. Sci. 9. doi: 10.3389/fmars.2022.822485
Hepburn C. D., Pritchard D. W., Cornwall C. E., McLeod R. J., Beardall J., Raven J. A., et al. (2011). Diversity of carbon use strategies in a kelp forest community: implications for a high CO2 ocean. Global Change Biol. 17 (7), 2488–2497. doi: 10.1111/j.1365-2486.2011.02411.x
Hermabessiere L., Dehaut A., Paul-Pont I., Lacroix C., Jezequel R., Soudant P., et al. (2017). Occurrence and effects of plastic additives on marine environments and organisms: A review. Chemosphere 182, 781–793. doi: 10.1016/j.chemosphere.2017.05.096
Howard J., Sutton-Grier A., Herr D., Kleypas J., Landis E., Mcleod E., et al. (2017). Clarifying the role of coastal and marine systems in climate mitigation. Front. Ecol. Environ. 15 (1), 42–50. doi: 10.1002/fee.1451
Huang Y., Xiao X., Xu C., Perianen Y. D., Hu J., Holmer M. (2020). Seagrass beds acting as a trap of microplastics - Emerging hotspot in the coastal region? Environ. Pollut. 257, 113450. doi: 10.1016/j.envpol.2019.113450
Ivar do Sul J. A., Costa M. F. (2014). The present and future of microplastic pollution in the marine environment. Environ. Pollut. 185, 352–364. doi: 10.1016/j.envpol.2013.10.036
Jones K. L., Hartl M. G. J., Bell M. C., Capper A. (2020). Microplastic accumulation in a Zostera marina L. bed at Deerness Sound, Orkney, Scotland. Mar. Pollut. Bull. 152, 110883. doi: 10.1016/j.marpolbul.2020.110883
Jones C. G., Lawton J. H., Shachak M. (1997). Positive and negative effects of organisms as physical ecosystem engineers. Ecology 78 (7), 1946–1957. doi: 10.1890/0012-9658(1997)078[1946:PANEOO]2.0.CO;2
Kedzierski M., D'Almeida M., Magueresse A., Le Grand A., Duval H., César G., et al. (2018). Threat of plastic ageing in marine environment. Adsorption/desorption of micropollutants. Mar. Pollut. Bull. 127, 684–694. doi: 10.1016/j.marpolbul.2017.12.059
Kirchman D., Mazzella L., Alberte R. S., Mitchell R. (1984). Epiphytic bacterial production on Zostera marina. Mar. Ecol. Prog. Ser. 15, 117–123. doi: 10.3354/meps015117
Koch E. W., Barbier E. B., Silliman B. R., Reed D. J., Perillo G. M., Hacker S. D., et al. (2009). Non-linearity in ecosystem services: temporal and spatial variability in coastal protection. Front. Ecol. Environ. 7 (1), 29–37. doi: 10.1890/080126
Kuo J., Hartog C. d. (2006). Seagrass morphology, anatomy, and ultrastructure. Eds. Larkum A. W. D., Orth R. J., Duarte C. M. Seagrasses: biology, ecologyand conservation (Cham, Switzerland: Springer Nature), 51–87.
Larkum A. W. D., Davey P. A., Kuo J., Ralph P. J., Raven J. A. (2017). Carbon-concentrating mechanisms in seagrasses. J. Exp. Bot. 68 (14), 3773–3784. doi: 10.1093/jxb/erx206
Larkum A. W. D., Kendrick G. A., Ralph P. J. (2018a). Seagrasses of Australia: structurem ecology and conservation (Cham, Switzerland: Springer Nature), 348–350.
Larkum A. W., Pernice M., Schliep M., Davey P., Szabo M. (2018b). “Photosynthesis and Metabolism of Seagrasses,”. In Seagrasses of Australia (Springer), 315–342.
Lilley R. J., Unsworth R. K. F. (2014). Atlantic Cod (Gadus morhua) benefits from the availability of seagrass (Zostera marina) nursery habitat. Global Ecol. Conserv. 2, 367–377. doi: 10.1016/j.gecco.2014.10.002
Maberly S. C. (2014). The fitness of the environments of air and water for photosynthesis, growth, reproduction and dispersal of photoautotrophs: An evolutionary and biogeochemical perspective. Aquat. Bot. 118, 4–13.
Mcleod E., Chmura G. L., Bouillon S., Salm R., Björk M., Duarte C. M., et al. (2011). A blueprint for blue carbon: toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front. Ecol. Environ. 9 (10), 552–560. doi: 10.1890/110004
Moriarty D. J. W., Iverson R. L., Pollard P. C. (1986). Exudation of organic carbon by the seagrass Halodule wrightii Aschers and its effect on bacterial growth in the sediment. J. Exp. Mar. Bio. Ecol. 96 (2), 115–126.
Mtwana Nordlund L., Koch E. W., Barbier E. B., Creed J. C. (2016). Seagrass ecosystem services and their variability across genera and geographical regions. PloS One 11 (10), e0163091. doi: 10.1371/journal.pone.0163091
Murchie E. H., Lawson T. (2013). Chlorophyll fluorescence analysis: a guide to good practice and understanding some new applications. J. Exp. Bot. 64 (13), 3983–3998. doi: 10.1093/jxb/ert208
Nielsen L. B., Finster K., Welsh D. T., Donelly A., Herbert R. A., De Wit R., et al. (2001). Sulphate reduction and nitrogen fixation rates associated with roots, rhizomes and sediments from Zostera noltii and Spartina maritima meadows. Environ. Microbiol. 3 (1), 63–71. doi: 10.1046/j.1462-2920.2001.00160.x
Noisette F., Depetris A., Kühl M., Brodersen K. E. (2020). Flow and epiphyte growth effects on the thermal, optical and chemical microenvironment in the leaf phyllosphere of seagrass (Zostera marina). J. R. Soc. Interface 17 (171), 20200485. doi: 10.1098/rsif.2020.0485
Nordlund L. M., Jackson E. L., Nakaoka M., Samper-Villarreal J., Beca-Carretero P., Creed J. C. (2018). Seagrass ecosystem services – What's next? Mar. Pollut. Bull. 134, 145–151. doi: 10.1016/j.marpolbul.2017.09.014
Orth R. J., Duarte C. M. (2006). Seagrasses: biology, ecology and conservation (Netherlands: Springer, Dordrecht), 441–461.
Pollard P. C., Moriarty D. (1991). Organic carbon decomposition, primary and bacterial productivity, and sulphate reduction, in tropical seagrass beds of the Gulf of Carpentaria, Australia. Mar. Ecol. Prog. Ser. 69 (1), 149–159. doi: 10.3354/meps069149
Potouroglou M., Bull J., Krauss K., Kennedy H., Fusi M., Daffonchio D., et al. (2017). Measuring the role of seagrasses in regulating sediment surface elevation. Sci. Rep. 7 (1), 11917.
Prange J. A., Dennison W. C. (2000). Physiological responses of five seagrass species to trace metals. Mar. Pollut. Bull. 41 (7-12), 327–336. doi: 10.1016/S0025-326X(00)00126-0
Prata J. C., da Costa J. P., Lopes I., Duarte A. C., Rocha-Santos T. (2019). Effects of microplastics on microalgae populations: A critical review. Sci. Total Environ. 665, 400–405. doi: 10.1016/j.scitotenv.2019.02.132
Ralph P., Durako M.J., Enriquez S., Collier C., Doblin M. (2007). Impact of light limitation on seagrasses. J. Exp. Mar. Biol. Ecol. 350 (1-2), 176–193.
Ralph P. J., Schreiber U., Gademann R., Kühl M., Larkum A. W. D. (2005). Coral photobiology studied with a new imaging PAM fluorometer. J. Phycol. 41, 335–342. doi: 10.1111/j.1529-8817.2005.04034.x
Rasmusson L. M., Lauritano C., Procaccini G., Gullström M., Buapet P., Björk M. (2017). Respiratory oxygen consumption in the seagrass Zostera marina varies on a diel basis and is partly affected by light. Mar. Biol. 164 (6), 140. doi: 10.1007/s00227-017-3168-z
Raven J., Ball L., Beardall J., Giordano M., Maberly S. (2011). Algae lacking CO2 concentrating mechanisms. Can. J. Botany-Revue Can. Botanique 83, 879–890.
Richardson S. L. (2006). Response of epiphytic foraminiferal communities to natural eutrophication in seagrass habitats off Man O'War Cay, Belize. Mar. Ecol. 27 (4), 404–416. doi: 10.1111/j.1439-0485.2006.00096.x
Ryan P. G., Moore C. J., van Franeker J. A., Moloney C. L. (2009). Monitoring the abundance of plastic debris in the marine environment. Philos. Trans. R. Soc. B: Biol. Sci. 364 (1526), 1999–2012. doi: 10.1098/rstb.2008.0207
Saunders M. I., Leon J., Phinn S. R., Callaghan D. P., O'Brien K. R., Roelfsema C. M., et al. (2013). Coastal retreat and improved water quality mitigate losses of seagrass from sea level rise. Global Change Biol. 19 (8), 2569–2583. doi: 10.1111/gcb.12218
Shafer D. J., Kaldy J. E. (2014). Comparison of photosynthetic characteristics of the seagrass congeners Zostera marina and Zostera japonica. Aquat. Bot. 112, 91–97. doi: 10.1016/j.aquabot.2013.09.002
Shim W. J., Hong S. H., Eo S. (2018). “Chapter 1 - marine microplastics: abundance, distribution, and composition,” in Microplastic contamination in aquatic environments. Ed. Zeng E. Y. (Amsterdam, Netherlands: Elsevier), 1–26.
Short F. T., Neckles H. A. (1999). The effects of global climate change on seagrasses. Aquat. Bot. 63 (3), 169–196. doi: 10.1016/S0304-3770(98)00117-X
Short F. T., Short C. A. (1984). “The seagrass filter: purification of estuarine and coastal waters,” in The estuary as a filter. Ed. Kennedy V. S. (Amsterdam, Netherlands: Academic Press), 395–413.
Sjollema S. B., Redondo-Hasselerharm P., Leslie H. A., Kraak M. H. S., Vethaak A. D. (2016). Do plastic particles affect microalgal photosynthesis and growth? Aquat. Toxicol. 170, 259–261. doi: 10.1016/j.aquatox.2015.12.002
Spilling K., Titelman J., Greve T. M., Kühl M. (2010). Microsensor measurements of the external and internal microenvironment of fucus vesiculosus (PHAEOPHYCEAE)1. J. Phycol. 46 (6), 1350–1355. doi: 10.1111/j.1529-8817.2010.00894.x
Staehr P., Asmala E., Carstensen J., Krause-Jensen D., Reader H. (2018). Ecosystem metabolism of benthic and pelagic zones of a shallow productive estuary: Spatio-temporal variability. Mar. Ecol. Prog. Ser. 601, 15–32. doi: 10.3354/meps12697
Staehr P. A., Borum J. (2011). Seasonal acclimation in metabolism reduces light requirements of eelgrass (Zostera marina). J. Exp. Mar. Biol. Ecol. 407 (2), 139–146. doi: 10.1016/j.jembe.2011.05.031
Tarquinio F., Bourgoure J., Koenders A., Laverock B., Säwström C., Hyndes G. A. (2018). Microorganisms facilitate uptake of dissolved organic nitrogen by seagrass leaves. ISME J. 12 (11), 2796–2800. doi: 10.1038/s41396-018-0218-6
Tetu S. G., Sarker I., Schrameyer V., Pickford R., Elbourne L. D. H., Moore L. R., et al. (2019). Plastic leachates impair growth and oxygen production in Prochlorococcus, the ocean’s most abundant photosynthetic bacteria. Commun. Biol. 2 (1), 184. doi: 10.1038/s42003-019-0410-x
Teuten E., Saquing J., Knappe D., Barlaz M., Jonsson S., Björn A., et al. (2009). Transport and release of chemicals from plastic to the environment and to wildlife. Philos. Trans. R. Soc. London. Ser. B Biol. Sci. 364, 2027–2045. doi: 10.1098/rstb.2008.0284
Tortell P. D., Reinfelder J. R., Morel F. M. M. (1997). Active uptake of bicarbonate by diatoms. Nature 390 (6657), 243–244. doi: 10.1038/36765
Wang J., Tan Z., Peng J., Qiu Q., Li M. (2016). The behaviors of microplastics in the marine environment. Mar. Environ. Res. 113, 7–17. doi: 10.1016/j.marenvres.2015.10.014
Webb W. L., Newton M., Starr D. (1974). Carbon dioxide exchange of Alnus rubra. Oecologia 17 (4), 281–291. doi: 10.1007/BF00345747
Welsh D. T. (2000). Nitrogen fixation in seagrass meadows: Regulation, plant–bacteria interactions and significance to primary productivity. Ecol. Lett. 3 (1), 58–71. doi: 10.1046/j.1461-0248.2000.00111.x
Wong M. C., Vercaemer B. (2012). Effects of invasive colonial tunicates and a native sponge on the growth, survival, and light attenuation of eelgrass (Zostera marina). Aquat. Invasions 7, (3). doi: 10.3391/ai.2012.7.3.003
Wright S. L., Thompson R. C., Galloway T. S. (2013). The physical impacts of microplastics on marine organisms: A review. Environ. Pollut. 178, 483–492. doi: 10.1016/j.envpol.2013.02.031
Yang X. Q., Zhang Q. S., Zhang D., Sheng Z. T. (2017). Light intensity dependent photosynthetic electron transport in eelgrass (Zostera marina L.). Plant Physiol. Biochem. 113, 168–176. doi: 10.1016/j.plaphy.2017.02.011
Keywords: microplastics, pollution, epiphytes, photosynthesis, respiration, seagrass
Citation: Molin JM, Groth-Andersen WE, Hansen PJ, Kühl M and Brodersen KE (2023) Microplastic pollution associated with reduced respiration in seagrass (Zostera marina L.) and associated epiphytes. Front. Mar. Sci. 10:1216299. doi: 10.3389/fmars.2023.1216299
Received: 03 May 2023; Accepted: 06 July 2023;
Published: 03 August 2023.
Edited by:
James E. Kaldy, Pacific Ecological Systems Division - US EPA, United StatesReviewed by:
Cecelia Gerstenbacher, Boston University, United StatesCopyright © 2023 Molin, Groth-Andersen, Hansen, Kühl and Brodersen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kasper Elgetti Brodersen, ZWxnZXR0aUBydWMuZGs=
†Present address: Kasper Elgetti Brodersen, Environmental Dynamics, Department of Science and Environment, Roskilde University, Roskilde, Denmark
‡These authors share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.