
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
GENERAL COMMENTARY article
Front. Immunol. , 25 March 2025
Sec. Cancer Immunity and Immunotherapy
Volume 16 - 2025 | https://doi.org/10.3389/fimmu.2025.1575090
This article is part of the Research Topic The Tumor Microenvironment and Immunotherapy for Head and Neck Tumors View all 16 articles
This article is a commentary on:
Disulfidptosis-Related Gene Signatures as Prognostic Biomarkers and Predictors of Immunotherapy Response in HNSCC
A Commentary on
Disulfidptosis-related gene signatures as prognostic biomarkers and predictors of immunotherapy response in HNSCC
by Qin H, Xu J, Yue Y, Chen M, Zhang Z, Xu P, Zheng Y, Zeng H, Weng J, Yang J and Yu F (2025) Front. Immunol. 15:1456649. doi: 10.3389/fimmu.2024.1456649
We read with great interest the research “Disulfidptosis-related gene signatures as prognostic biomarkers and predictors of immunotherapy response in HNSCC” by Qin et al. (1), which was recently published on Jan 17, 2025, in the Journal of Frontiers in Immunology. This article elucidated the potential significance of disulfidptosis-related genes (DRGs) in head and neck squamous cell carcinoma (HNSCC), as demonstrated through rigorous bioinformatics analysis and experimental validation. Through in-depth mechanistic investigations, they revealed the mechanisms by which these genes regulate tumor cell death and influence the functionality of immune cells within the tumor microenvironment. Additionally, the efficacy of these genes in forecasting patients’ responsiveness to immune checkpoint inhibitors was assessed, offering valuable insights for the advancement of novel therapeutic strategies. Although acknowledging the significant contribution of that study, Qin et al. had misunderstood Figure 11B in the section titled “TMB, MSI, mRNAsi, and Drug Sensitivity Analysis”—they believed that in high-risk HNSCC, the sensitivity of belinostat, SB52334, and CAL101 was significantly higher than in the low-risk group, while Dasatinib, Pazopanib, and Docetaxel showed higher sensitivity in low-risk HNSCC (1).
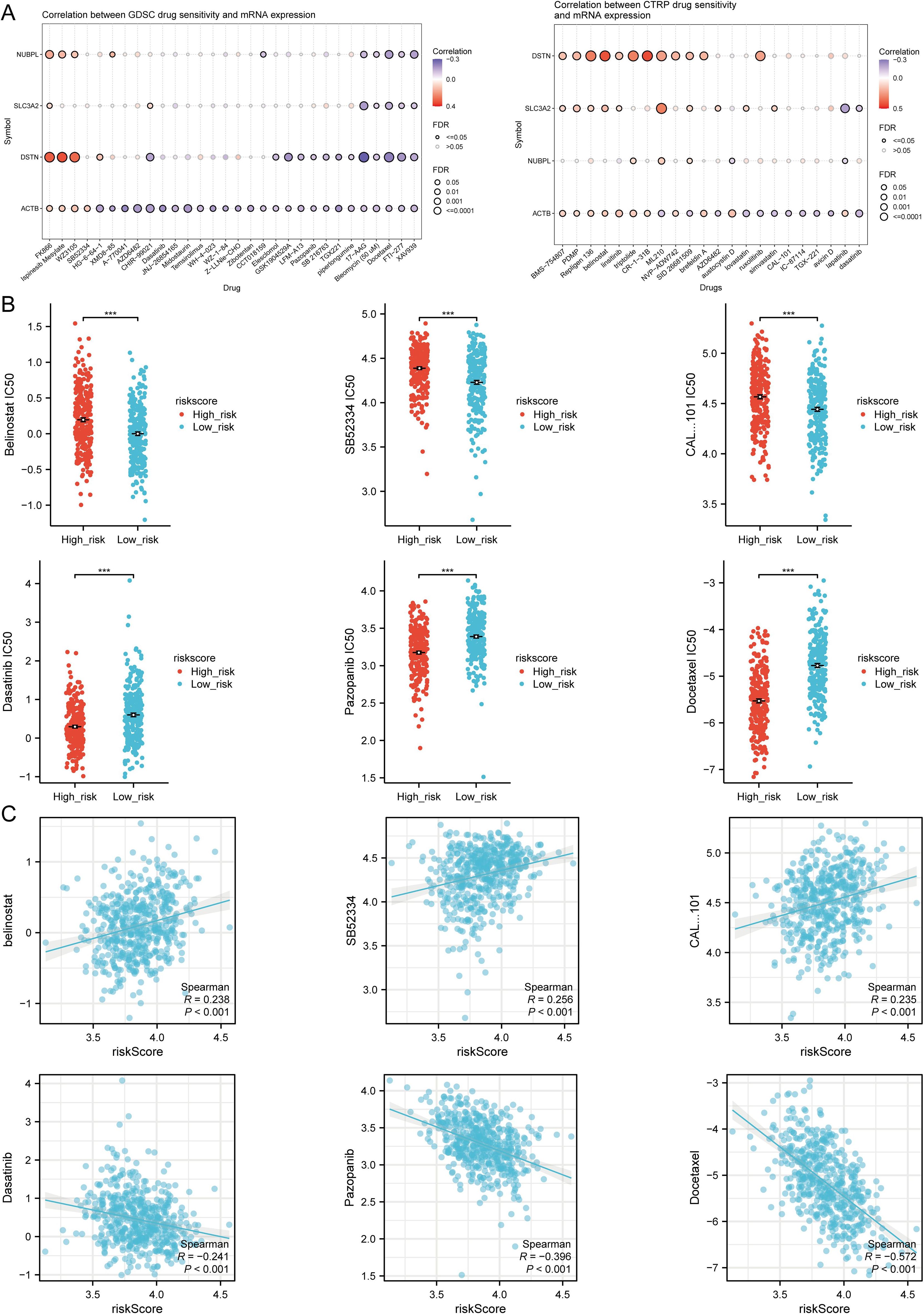
Figure 11. Drug sensitivity analysis. (A) Predictive antitumor drugs based on the three prognostic DRGs expression in HNSCC from the GDSC and CTRP datasets. (B) The distribution of IC50 scores in the high and low risk groups. (C) Spearson correlation analysis of IC50 score and riskscore. ***p<0.001. (Image source: Qin et al., Disulfidptosis-related gene signatures as prognostic biomarkers and predictors of immunotherapy response in HNSCC).
IC50, defined as the half maximal inhibitory concentration, signifies the concentration at which a drug or inhibitor diminishes the activity of a biological process (such as an enzyme, receptor, or cell) to half of its maximum level under specific experimental conditions. A lower IC50 value indicates that the drug can achieve a 50% inhibitory effect at a lower concentration, suggesting a higher potency and sensitivity of the drug (2, 3). Therefore, the correct interpretation of Figure 11B in the original text of Qin et al. (1) is that in HNSCC patients, the IC50 values for belinostat, SB52334, and CAL-101 are significantly higher in the high-risk group compared to the low-risk group, suggesting lower sensitivity of the high-risk group to these drugs. Conversely, the IC50 values for Dasatinib, Pazopanib, and Docetaxel are significantly lower in the high-risk group, indicative of higher sensitivity of this group to these drugs relative to the low-risk group.
In summary, readers should exercise caution when reading the appropriate chapters to ensure accurate comprehension.
JX: Writing – original draft, Writing – review & editing. CX: Writing – original draft. QY: Writing – original draft. LC: Writing – review & editing. MW: Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the Natural Science Foundation of Southwest Medical University, Grant (No. 2023QN008 and 2023QN103). This work was further supported by the National Natural Science Foundation project (No. 82372825).
The authors declare that the research was conducted in the absence of any commercial or financial 57 relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Qin H, Xu J, Yue Y, Chen M, Zhang Z, Xu P, et al. Disulfidptosis-related gene signatures as prognostic biomarkers and predictors of immunotherapy response in hnscc. Front Immunol. (2024) 15:1456649. doi: 10.3389/fimmu.2024.1456649
2. Cer RZ, Mudunuri U, Stephens R, Lebeda FJ. Ic50-to-ki: A web-based tool for converting ic50 to ki values for inhibitors of enzyme activity and ligand binding. Nucleic Acids Res. (2009) 37:W441–5. doi: 10.1093/nar/gkp253
Keywords: half maximal inhibitory concentration (IC50), medication, disulfidptosis-related genes (DRGs), head and neck squamous cell carcinoma (HNSCC), immunotherapy
Citation: Xue J, Xue C, Yin Q, Chen L and Wang M (2025) Commentary: Disulfidptosis-related gene signatures as prognostic biomarkers and predictors of immunotherapy response in HNSCC. Front. Immunol. 16:1575090. doi: 10.3389/fimmu.2025.1575090
Received: 11 February 2025; Accepted: 04 March 2025;
Published: 25 March 2025.
Edited by:
Lei Tao, Fudan University, ChinaReviewed by:
Lukasz Chlewicki, Eli Lilly, United StatesCopyright © 2025 Xue, Xue, Yin, Chen and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ligang Chen, Y2hlbmdsaWdhbmcuY29vbEAxNjMuY29t; Ming Wang, c3dtdXdtbUAxNjMuY29t
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.