
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Immunol. , 20 March 2025
Sec. Cancer Immunity and Immunotherapy
Volume 16 - 2025 | https://doi.org/10.3389/fimmu.2025.1541675
This article is part of the Research Topic Immune Predictive and Prognostic Biomarkers in Immuno-Oncology: Refining the Immunological Landscape of Cancer View all 11 articles
Background: The Oxidative Balance Score (OBS) represents a novel metric for assessing systemic oxidative stress, where elevated scores reflect increased antioxidant exposure. This study aims to explore the association between OBS and all-cause and cancer-specific mortality among cancer survivors.
Methods: An observational cohort study was conducted involving 4099 cancer survivors, utilizing data obtained from the National Health and Nutrition Examination Survey (NHANES) covering the years 1999 to 2018. The endpoints were established by cross-referencing data with the National Death Index (NDI). The OBS was developed based on dietary and lifestyle factors. Cox proportional hazards regression models were employed to examine the relationship between OBS and mortality risks. Restricted cubic spline was utilized to evaluate whether OBS exhibited a nonlinear association with the risk of death. Furthermore, Kaplan-Meier survival curves were generated to assess cumulative survival differences across various OBS outcomes.
Results: Over an average follow-up of 84.00 months, 1481 (26.29%) participants died, including 484 (8.9%) who died from cancer. In the fully adjusted model, multivariable Cox regression revealed that each unit increase in OBS was linked to a 1.8% decrease in all-cause mortality risk (HR 0.982, 95%CI 0.972-0.991) and a 2.6% decrease in cancer-specific mortality risk (HR 0.974, 95%CI 0.958-0.991). In the context of all-cause mortality, the risk of death was found to be significantly lower in quartiles Q2, Q3 and Q4 when compared to the OBS in quartile Q1. The hazard ratios (HRs) and 95% confidence intervals (CIs) for Q2, Q3 and Q4 were as follows: Q2 (HR 0.833, 95%CI 0.707-0.981), Q3 (HR 0.789, 95%CI 0.650-0.958) and Q4 (HR 0.699, 95%CI 0.579-0.844). Regarding cancer-specific mortality, the HRs and 95%CIs for Q2, Q3 and Q4 in comparison to Q1 were as follows: Q2 (HR 0.663, 95%CI 0.505-0.869), Q3 (HR 0.688, 95%CI 0.488-0.969) and Q4 (HR 0.595, 95%CI 0.435-0.815). Similar associations were noted when the dietary and lifestyle components of the OBS were analyzed separately.
Conclusion: The findings indicate that higher levels of OBS are associated with a decrease in all-cause and cancer-specific mortality among cancer survivors. Our findings may contribute to the refinement of lifestyle intervention recommendations for this population.
Cancer remains one of the most pressing public health challenges globally, marked by rising incidence and mortality rates. According to the latest estimates from the International Agency for Research on Cancer (IARC), nearly 20 million new cases of cancer (including non-melanoma skin cancer [NMSC]) were reported in 2022, resulting in approximately 9.7 million cancer-related deaths (including NMSC). It is estimated that one in five individuals, regardless of gender, will get cancer during their lifetime, with approximately one in nine men and one in twelve women succumbing to the disease (1). Research has demonstrated that dietary and lifestyle factors play a significant role in the occurrence and progression of cancer. In recent years, numerous studies have focused on the positive effects of modifiable dietary and lifestyle factors in cancer prevention and the improvement of cancer prognosis (2–4).
The emergence of various pathological and physiological conditions, including cancer, is frequently linked to excessive cellular oxidative stress. Cells maintain redox homeostasis to prevent oxidative damage to DNA, proteins, and lipids (5). The disruption of the equilibrium between pro-oxidants and antioxidants, commonly referred to as oxidative stress, has been recognized as a significant pathogenic and pathophysiological factor in numerous chronic diseases, which are leading contributors to mortality (6, 7). The Oxidative Balance Score (OBS) is a metric designed to quantify the overall exposure to oxidants and antioxidants within an individual’s diet and lifestyle. A higher OBS indicates a predominant exposure to an excess of antioxidants (8). In contrast to research that concentrates on individual nutrients, the OBS offers a substantial advantage by integrating multiple oxidants and antioxidants, thereby providing a more comprehensive assessment of overall oxidative stress. Empirical evidence suggests that the OBS is an effective tool for evaluating oxidative stress status (9, 10).
To date, there have been limited epidemiological studies investigating the association between OBS and cancer prognosis (11). Consequently, the present study seeks to examine the relationship between OBS and the prognosis of cancer survivors, utilizing data from the National Health and Nutrition Examination Survey.
The National Center for Health Statistics (NCHS), a component of the Centers for Disease Control and Prevention (CDC), initiated multiple cycles of the National Health and Nutrition Examination Survey (NHANES) in the United States beginning in 1999. This study utilized a retrospective observational cohort design, analyzing data obtained from NHANES spanning the years 1999 to 2018. NHANES employs a complex, stratified, multi-stage probability cluster design to conduct a nationally representative survey of the health and nutritional status of the non-institutionalized civilian population in the United States. The survey provides comprehensive information regarding its methodologies and analytical guidelines (12). Data concerning nutrition and health status are collected through a series of household interviews, physical examinations, and laboratory measurements.
Furthermore, the National Center for Health Statistics (NCHS) has established connections between numerous demographic surveys and death certificate data obtained from the National Death Index (NDI). The data have been meticulously processed to reduce the risk of subject identification, and a publicly accessible version of the follow-up data concerning mortality rates for adult participants, spanning from the date of survey participation until December 31, 2019, has been made available.
Initially, a total of 96,811 participants were included in the study. However, we excluded participants under the age of 20 whose data were not publicly available (N=37,607) and those who did not meet the mortality follow-up criteria (N=140). We also excluded individuals without information from the malignant tumor questionnaire (N=4,718) and based on the cancer questionnaire, we excluded those who denied having cancer or malignancies (N=49,723). Additionally, individuals lacking dietary and lifestyle data necessary for calculating the oxidative balance score and any specified study covariates (N=1,064) were also excluded. The final cohort included 4,099 cancer survivors (see Figure 1).
The calculation of the Oxidative Balance Score (OBS) encompasses 16 dietary nutrients and 4 lifestyle factors, which consist of 15 antioxidant components and 5 pro-oxidant components (13). A majority of components previously utilized for the calculation of the Oxidative Balance Score(OBS) have been identified, while six additional components have been newly selected based on their availability in the data and their correlation with oxidative stress (OS). These components include riboflavin, niacin, vitamin B6, vitamin B12, magnesium, and copper (8). These 16 nutrients are obtained from the initial dietary recall interview. The 4 lifestyle factors considered are physical activity, body mass index (BMI), alcohol consumption, and smoking, with smoking levels assessed through cotinine measurements. The 5 pro-oxidant components identified are total fat, iron, BMI, alcohol consumption, and cotinine, while the remaining factors are categorized as antioxidant components. The components of oxidative balance score (OBS) are categorized into four distinct groups: (1) dietary antioxidants, which include fiber, β-carotene, riboflavin, niacin, vitamin B6, total folate, vitamin B12, vitamin C, vitamin E, calcium, magnesium, zinc, copper, and selenium; (2) dietary pro-oxidants, comprising total fat and iron; (3) lifestyle antioxidants, represented by physical activity; and (4) lifestyle pro-oxidants, which encompass alcohol, smoking, and BMI.
According to the existing literature (13), alcohol consumption is classified into three categories: non-drinkers, light to moderate drinkers (defined as women consuming 0-15 grams per day and men consuming 0-30 grams per day), and heavy drinkers (defined as women consuming 15 grams or more per day and men consuming 30 grams or more per day). These categories are assigned scores of 2, 1, and 0, respectively (13). The levels of physical activity (PA) were assessed utilizing the NHANES Physical Activity Questionnaire (PAQ).In the process of calculating the PA score, it is essential to first gather pertinent data from the Physical Activity Questionnaire (PAQ) (14). Subsequently, the PAQ data will be converted into Metabolic Equivalent (MET) using the following formula: Physical Activity (PA) in MET-minutes per week (MET-min/wk) = MET × weekly frequency of each physical activity (PA) × duration. Due to the variations in the NHANES Physical Activity Questionnaire (PAQ) across different cycles, the physical activity calculations were modified to align with the respective survey year. From 1999 to 2006, PA was classified into categories such as walking or bicycling, home or yard tasks, muscle-strengthening activities, and screen time (e.g., watching television or using a computer). In contrast, from 2007 to 2018, PA was organized into two overarching categories: work-related activities, which were further subdivided into moderate-intensity and vigorous-intensity tasks, and leisure activities, encompassing walking, bicycling, and both vigorous- and moderate-intensity recreational activities. In accordance with NHANES recommendations, the metabolic equivalent (MET) score coefficients for both vigorous work-related and leisure time activities were set at 8.0, while the MET score coefficients for moderate work-related and leisure time activities, as well as for walking or bicycling as a mode of transportation, were established at 4.0. We evaluated physical activity levels utilizing MET scores in accordance with the NHANES guidelines. Participants were classified into three distinct categories: 0 points for those engaging in less than 400 MET minutes per week, 1 point for those accumulating between 400 and 1,000 MET minutes per week, and 2 points for individuals exceeding 1,000 MET minutes per week. Other components are evaluated based on quartiles, stratified by gender. For antioxidant components, scores range from 0 to 2, corresponding to the lowest and highest quartiles, respectively. Conversely, the scoring for pro-oxidant components is inverted, with the highest quartile receiving 0 points and the lowest quartile receiving 2 points. Detailed scoring criteria for each OBS component are provided in Supplementary Table 1. The overall Oxidative Balance Score (OBS) is computed by aggregating the scores of each component, resulting in a score range of 3 to 36, where a higher score signifies greater exposure to antioxidants. Furthermore, we categorized the OBS into dietary and lifestyle components based on their sources to investigate their combined and independent effects on the mortality risk among cancer survivors.
Participants who responded affirmatively to the inquiry, “Have you ever been told by a doctor or other health professional that you had cancer or a malignancy of any kind?” (variable MCQ220)?”. Individuals who answer “yes” were categorized as cancer survivors and were subsequently asked What kind of cancer was it?” and “How old were you when this cancer was first diagnosed?. The variable “years since first cancer diagnosis” was calculated by subtracting the age at which the participant was first diagnosed with cancer from their current age.
The follow-up period extends from the date of the interview to the last follow-up date, which is December 31, 2019, or to the date of death, whichever occurs first. The National Death Index (NDI) records furnish information regarding the causes of death for the participants included in the study. The mortality outcomes are classified in accordance with the International Classification of Diseases and Related Health Problems, 10th Revision (ICD-10) codes, which are documented as the primary cause of death. The study endpoints encompass all-cause mortality as well as cancer-related mortality, specifically codes C00 to C97.
During the household interviews, demographic data were collected, including age, gender, race and ethnicity (categorized as Hispanic American, non-Hispanic Black, non-Hispanic White, or other), education level (less than high school, high school or equivalent, or college or above), marital status (single or non-single), poverty income ratio (PIR), and medical history concerning diabetes, hypertension, and cardiovascular disease (CVD). The poverty income ratio serves as an index of poverty status, calculated by dividing the total household income by the poverty threshold. In accordance with the analysis guidelines, this ratio is classified into three categories: ≤1.3, 1.3-3.5, and >3.5 (12). The definition of diabetes is based on an affirmative response to any of the following questions:: “Have you ever been told by a doctor that you have diabetes?” or “Are you now taking insulin?” or “Are you now taking diabetes pills to lower your blood sugar?”. Hypertension is defined as a self-reported diagnosis, the use of antihypertensive medication, or meeting any of the following criteria: systolic/diastolic blood pressure of ≥140/90 mmHg. Cardiovascular disease (CVD) is defined as a self-reported diagnosis of any of the following conditions: stroke, angina pectoris, myocardial infarction, coronary heart disease, or heart failure.
In our analysis, the complex sampling design of the National Health and Nutrition Examination Survey (NHANES) incorporated sample weights, clustering, and stratification, which are essential for the appropriate analysis of NHANES data (15). Baseline characteristics are represented by the quartiles of the observed variable (OBS). Normally distributed data are presented as mean ± standard error (SE), while non-normally distributed data are expressed as median (interquartile range), and categorical variables are reported as counts (percentages). The Kaplan-Meier method was employed to plot survival curves associated with OBS. Multivariable Cox regression models (Model 1, Model 2, and Model 3) were utilized to evaluate the relationship between OBS and both all-cause mortality and cancer mortality. Model 1 was not adjusted for any covariates. Model 2 was adjusted for baseline demographic variables, which included age (measured in continuous years), gender, race, and ethnicity (categorized as Hispanic American, non-Hispanic Black, non-Hispanic White, or other). Furthermore, Model 3 included additional adjustments for social factors, such as education level, marital status, and poverty income ratio, as well as for chronic diseases, which encompassed diabetes, hypertension, and cardiovascular diseases(CVD). The log-rank test and Kaplan-Meier (K-M) survival analysis were employed to investigate differences in survival probabilities. To visualize the association between OBS and both all-cause mortality and cancer mortality among cancer survivors, a restricted cubic spline (RCS) with three knots was constructed based on the fully adjusted Cox model. Additionally, we conducted a series of sensitivity analyses to evaluate the robustness of our findings. Initially, we excluded participants who succumbed to cancer within 2 years of diagnosis to mitigate the potential for reverse causality. Subsequently, we incorporated an additional adjustment covariate: the year of diagnosis. Finally, we performed independent stratified analyses within specific subgroups categorized by age, sex, race, education, poverty-to-income ratio, hypertension, diabetes, and CVD to ascertain whether the associations between observational variables and all-cause mortality, as well as cancer mortality, varied across these subgroups for each covariate category.
All statistical analyses were performed utilizing R version 4.4.1. A two-sided p-value of less than 0.05 was deemed statistically significant.
Table 1 presents the baseline characteristics of participants categorized by OBS quartiles. The study comprised a total of 4,099 cancer survivors. The mean age of participants was 62.34 years (± 0.36), with non-Hispanic whites constituting the predominant demographic within the study population. No statistically significant differences were observed in age and gender across the groups (P all >.05). In comparison to participants in the lowest OBS quartile (Q1), those in the highest OBS quartile (Q4) were more likely to be non-Hispanic white, not single, possess a higher poverty income level, and have no prior history of diabetes, hypertension, or cardiovascular disease. Additional data on cancer survivors classified by cancer type and OBS quartiles can be found in the Supplementary Materials, specifically in Supplementary Table 2.
The median follow-up duration was 84.00 months (range: 45.00 to 140.00 months), during which a total of 1,481 individuals (26.29%) succumbed, including 484 individuals who died from cancer. Table 2 illustrates the association between oxidative Balance Score (OBS), encompassing both dietary and lifestyle factors, and all-cause as well as cancer-specific mortality among cancer survivors. In the unadjusted model (Model 1), an increase in OBS was significantly correlated with a decrease in the risk of all-cause mortality (hazard ratio [HR]0.972, 95% confidence interval [CI] 0.963–0.982, p < 0.0001). When comparing the lowest quartile of OBS (Q1) to the highest quartile (Q4), the latter was associated with a 45.3% reduction in the risk of all-cause mortality (HR 0.547, 95% CI 0.452-0.663, p for trend <0.0001). In Model 2, after adjusting for gender, age, and race, each unit increase in OBS corresponded to a 3.3% reduction in the risk of all-cause mortality (HR 0.967, 95% CI 0.958–0.975, p < 0.0001). In this adjusted model, participants in the OBS Q4 group exhibited a 46.5% lower risk of all-cause mortality compared to the Q1 group (HR 0.535, 95% CI 0.450–0.636, p for trend <0.0001). In the fully adjusted model (Model 3), each unit increase in OBS was associated with a 1.8% reduction in the risk of all-cause mortality (HR 0.982, 95% CI 0.972–0.991, p < 0.0001). When comparing the Q4 group to the Q1 group, the risk of all-cause mortality was reduced by 30.1% (HR 0.699, 95% CI 0.579–0.844, p for trend <0.001). Furthermore, in Model 3, both dietary OBS and lifestyle OBS demonstrated a negative correlation with the risk of all-cause mortality among cancer survivors (p = 0.006 and p < 0.0001, respectively). In this model, compared to Q1, the dietary OBS and lifestyle OBS in Q4 were associated with a 24.5% reduction in all-cause mortality risk (HR 0.755, 95% CI 0.637–0.896, p for trend <0.001) and a 41.7% reduction in all-cause mortality risk (HR 0.583, 95% CI 0.477–0.713, p for trend <0.0001), respectively.
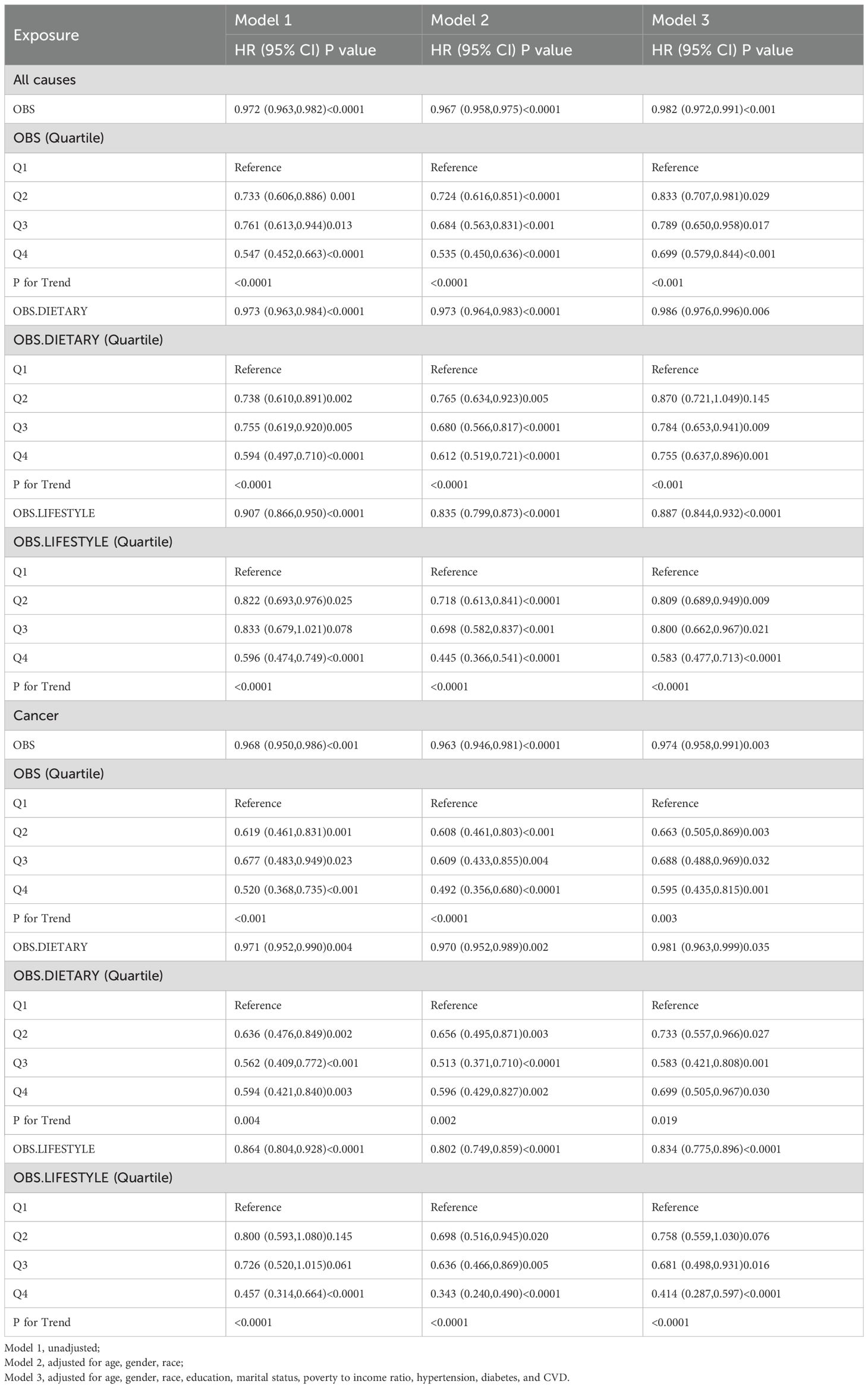
Table 2. Multivariable Cox regression models analysis of the relationship between OBS and mortality among cancer survivors in NHANES 1999–2018.
Table 2 presents the association between OBS and cancer mortality. In the unadjusted model (Model 1), a significant reduction in cancer mortality risk was observed with increasing OBS (HR 0.968, 95% CI 0.950, 0.986, p < 0.001). When comparing the OBS quartile 1 (Q1) group to the quartile 4 (Q4) group, the cancer mortality risk decreased by 48.0% (HR 0.520, 95% CI 0.368, 0.735, p for trend < 0.001). In Model 2, after adjusting for gender, age, and race, each unit increase in OBS was associated with a 6.7% reduction in cancer mortality risk (HR 0.963, 95% CI 0.946, 0.981, p < 0.0001). Participants in the Q4 group exhibited a 50.8% lower cancer mortality risk compared to the Q1 group (HR 0.492, 95% CI 0.356, 0.680, p for trend < 0.0001). In the fully adjusted model (Model 3), each unit increase in OBS corresponded to a 2.6% decrease in cancer mortality risk (HR 0.974, 95% CI 0.958, 0.991, p = 0.003). The cancer mortality risk in the OBS Q4 group was reduced by 40.5% relative to the Q1 group (HR 0.595, 95% CI 0.435, 0.815, p for trend = 0.003). Furthermore, in Model 3, both dietary OBS and lifestyle OBS demonstrated a negative correlation with cancer mortality risk among cancer survivors (p = 0.035 and p < 0.0001, respectively). Specifically, compared to participants in the Q1 group, those in the Q4 group exhibited a 30.1% lower mortality risk associated with dietary OBS (HR 0.699, 95% CI 0.505, 0.967, p for trend = 0.019) and a 58.6% lower cancer mortality risk associated with lifestyle OBS (HR 0.414, 95% CI 0.287, 0.597, p for trend < 0.0001).
Kaplan-Meier (K-M) survival analysis, as illustrated in Figure 2, indicated significant differences in all-cause mortality and cancer mortality across the four groups (all P < 0.05). Participants in the highest quartile (Q4) of the observational variable (OBS) exhibited the lowest mortality rate, whereas those in the lowest quartile (Q1) demonstrated the highest mortality rate.
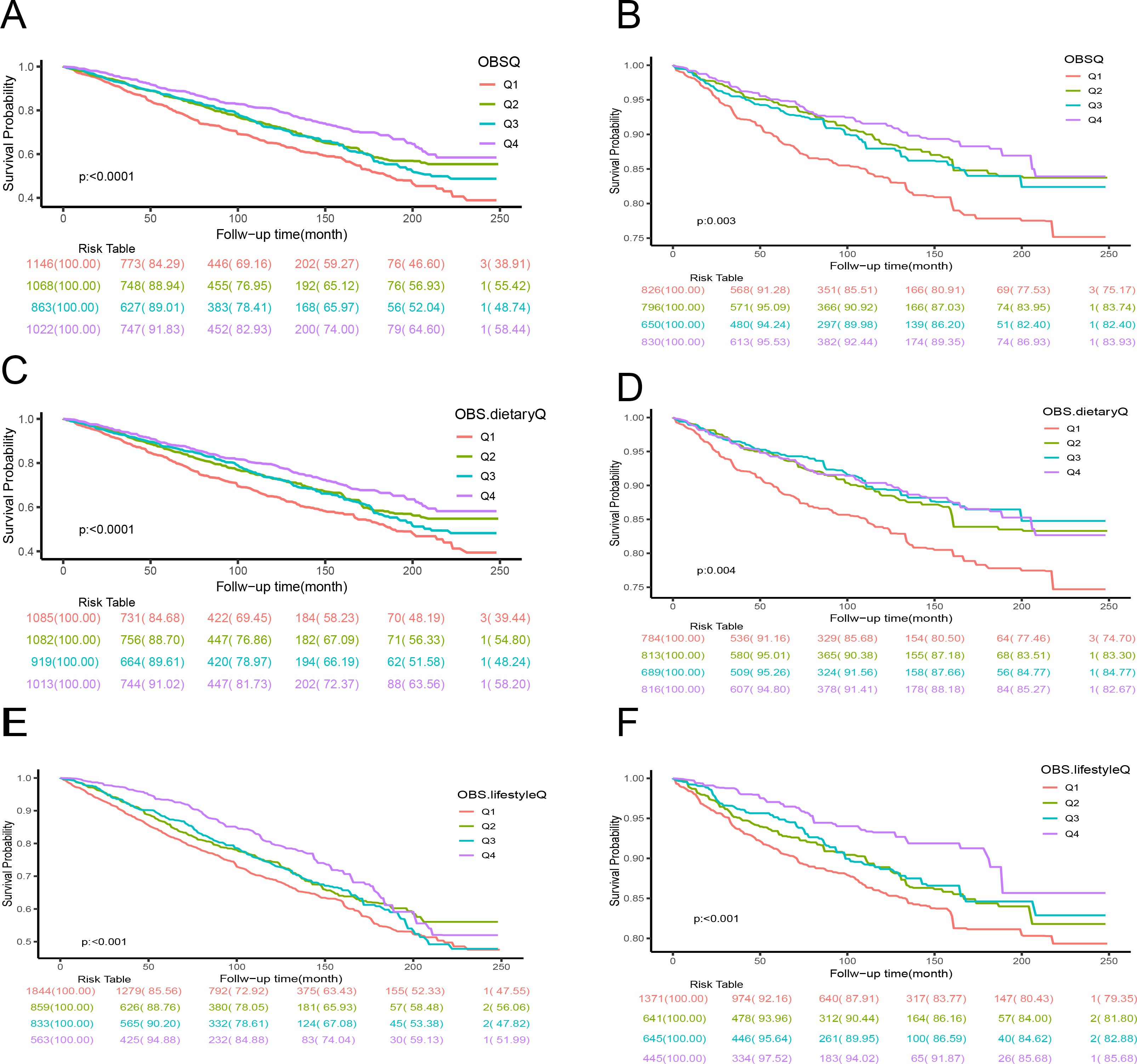
Figure 2. The Kaplan-Meier analyses for mortality across the four groups are presented as follows: (A) Kaplan-Meier curve illustrating the relationship between observational (OBS) data and all-cause mortality; (B) Kaplan-Meier curve depicting the association between OBS data and cancer-specific mortality; (C) Kaplan-Meier curve representing the correlation between dietary OBS and all-cause mortality rate; (D) Kaplan-Meier curve showing the link between dietary OBS and cancer-specific mortality; (E) Kaplan-Meier curve for lifestyle OBS and all-cause mortality rate; (F) Kaplan-Meier curve for lifestyle OBS and cancer-specific mortality.
We also performed a restricted cubic spline (RCS) analysis utilizing the fully adjusted Cox regression model (Model 3). The results indicated a negative correlation between OBS and both all-cause mortality and cancer mortality among cancer survivors (see Figure 3).
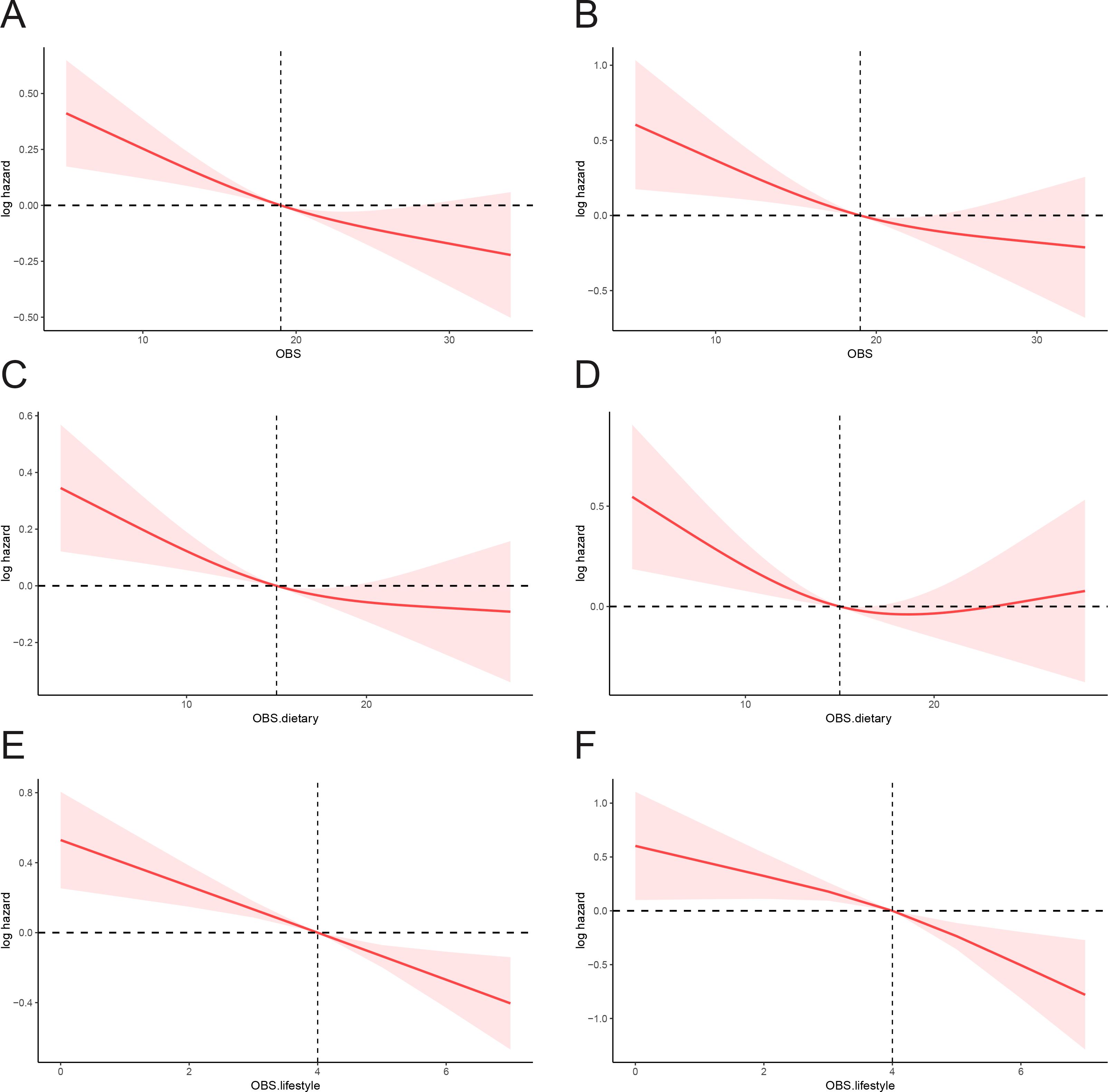
Figure 3. RCS analyses for mortality: (A) OBS and all-cause mortality; (B) OBS and cancer-specific mortality; (C) Dietary OBS and all-cause mortality; (D) Dietary OBS and cancer-specific mortality; (E) Lifestyle OBS and all-cause mortality; (F) Lifestyle OBS and cancer-specific mortality.
A stratified analysis was performed to evaluate the robustness of the regression results concerning the relationship between OBS and mortality among various subgroups of cancer survivors (see Table 3). The findings suggest that, in the majority of subgroups, the relationship between OBS and mortality is consistent, exhibiting a negative correlation that is statistically significant. Furthermore, no significant interactions were identified between age, gender, race, education level, marital status, poverty income ratio (PIR), hypertension, diabetes, cardiovascular disease (CVD) history, and OBS (P for interaction>0.05). To mitigate the potential for reverse causality, we excluded participants who succumbed to cancer within two years of their diagnosis. The results remained statistically significant (Supplementary Table 3). Additionally, further adjustments for the number of years since diagnosis did not yield a significant alteration in the results (Supplementary Table 4).
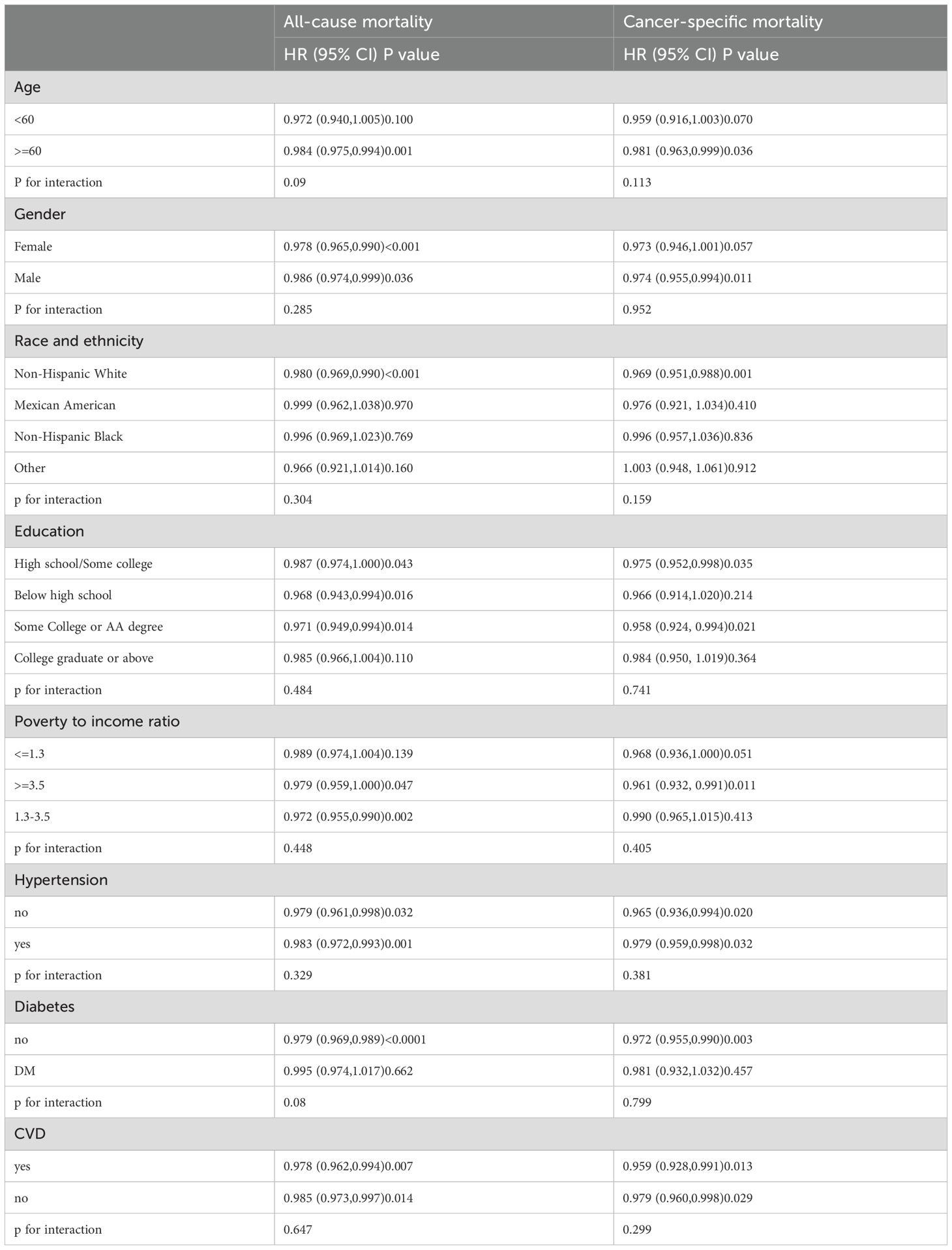
Table 3. Stratified analysis of the relationship between OBS and mortality among cancer survivors in NHANES 1999–2018.
A hierarchical analysis was performed using Model 3. In each instance, the model did not include the hierarchical variables themselves.
This study represents a pioneering effort to elucidate the influence of modifiable dietary and lifestyle factors, specifically pro-antioxidants and antioxidants on the survival prognosis of cancer survivors. The research employs a comprehensive measure of oxidative stress known as the Oxidative Balance Score (OBS). Through the analysis of nationally representative data from the United States collected over the past two decades, we found that elevated OBS scores correlated with a reduced risk of all-cause mortality and cancer-specific mortality. Furthermore, similar associations were observed for the dietary and lifestyle components of the OBS when examined independently. The findings presented above suggest that diets and lifestyles characterized by elevated levels of antioxidants, such as those abundant in antioxidant fiber,β-carotene, riboflavin, niacin, vitamin B6, total folate, vitamin B12, vitamin C, vitamin E, calcium, magnesium, zinc, copper, and selenium, while concurrently low in pro-oxidant fats and iron, combined with adherence to a physical activity regimen, reduced alcohol consumption, and avoidance of smoking, are associated with a decreased risk of all-cause mortality among cancer survivors, as well as a reduced risk of cancer-specific mortality.
In recent years, the relationship between oxidative stress and cancer has attracted considerable interest within the scientific community. Oxidative stress is intricately linked to tumor development and can affect the behavior of tumor cells through complex regulatory networks, including mitochondrial stress, endoplasmic reticulum stress, and ferritin deposition (16). ROS has the capacity to directly damage DNA, resulting in various forms of DNA damage, such as strand breaks. This damage can alter the expression levels of essential genes associated with proto-oncogenes, oncogenes, and DNA damage repair mechanisms, thereby facilitating tumorigenesis (17, 18). Furthermore, ROS can induce mutagenic break repair and activate the SOS response by damaging DNA bases, which interrupts replication and triggers a critical transition from high-fidelity to error-prone DNA polymerases, ultimately leading to an increase in carcinogenic mutations (19, 20). Research indicates that ROS can influence tumorigenesis and cellular transformation by oxidizing cysteine residues, which subsequently activate the three most prevalent oncogenic switch genes in human cancers: HRAS, NRAS, and KRAS (21).
A growing body of evidence indicates that elevated levels of ROS play a critical role in promoting and maintaining the malignant biological characteristics of cancer cells, particularly their aggressive metastatic phenotype (22). In the context of malignant transformation, early-stage tumor cells frequently invade adjacent stromal cells through the process of epithelial-mesenchymal transition (EMT) (23, 24). During this transition, ROS facilitate tumor metastasis by inducing Rho family GTPase-dependent cytoskeletal rearrangements, promoting the degradation of extracellular matrix proteins via matrix metalloproteinases, and enhancing angiogenesis through hypoxia-inducible factor activation (25). Additionally, various redox-regulated factors, including heat shock factor 1 (HSF1) (26), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) (27), and matrix metalloproteinases (MMPs) (28), also contribute to the promotion of metastasis. For instance, elevated ROS levels in tumor cells activate NF-κB, which in turn stimulates the expression of the transcription factor Snail, downregulates epithelial cadherin (E-cadherin), and increases the expression of neurocalcin and vimentin. This cascade of events disrupts intercellular connections and initiates the EMT process, thereby enhancing the metastatic potential of tumor cells (29).
Recently, a prospective study (30) conducted by Gu et al. involving 98,395 adults in the United States found that a higher OBS is associated with a reduced risk of colorectal cancer in women, however, this association was not observed in men. This finding suggests that adherence to an antioxidant-rich diet and lifestyle may contribute to the prevention of colorectal cancer. Additionally, research in a biracial US cohort (11) demonstrated that, after controlling for various confounding factors, a higher OBS reflects a predominant exposure to antioxidants, which is significantly correlated with decreased cancer mortality. Sensitivity analyses indicated that participants in the highest quartile of OBS were more likely to be non-smokers and non-drinkers. This observation suggests that individuals with higher OBS may possess greater health awareness and improved living conditions; however, the study did not account for confounding variables such as education level and household income, which may undermine the credibility of the findings. Furthermore, a recent multinational cohort study (31) examined the relationship between five lifestyle factors—body mass index (BMI), smoking, alcohol consumption, dietary habits (as measured by the 2015 Healthy Eating Index, HEI-2015), and physical activity—and mortality among cancer survivors. The results indicated that each healthy lifestyle factor—specifically, non-smoking, moderate alcohol consumption, adequate physical exercise, a nutritious diet, and optimal BMI—was independently associated with prolonged survival in cancer survivors. These findings are consistent with our own results, which reveal a negative correlation between overall OBS and the risk of mortality in cancer survivors. Similar effects were noted for independent dietary OBS and lifestyle OBS, underscoring the potential protective role of an antioxidant-rich diet and lifestyle for cancer survivors.
In summary, OBS evaluates the extent of oxidative stress by considering the interplay between various pro-oxidants and antioxidants. Oxidative stress is a critical factor in tumorigenesis, progression, and metastasis, operating through several mechanisms, such as the induction of DNA damage, the activation of oncogenic signaling pathways, the promotion of EMT, and the regulation of redox-sensitive factors. Elevated levels of ROS not only facilitate the malignant transformation of tumors but also sustain their invasive and metastatic capabilities. A high OBS is typically correlated with a more favorable prognosis in cancer patients.
Our research presents several advantages. Firstly, by employing a comprehensive scoring system (OBS) that simultaneously evaluates dietary and lifestyle factors, we conducted an extensive assessment of both pro-oxidative and anti-oxidative exposures, rather than relying on a singular indicator. This methodology facilitates a more nuanced understanding of the relationship between oxidative stress and mortality among cancer survivors. Secondly, the use of a nationally representative, high-quality sample enhances the generalizability of our findings. However, this study is not without its limitations. First, it is important to note that the cancer data utilized from the NHANES database predominantly relied on participants’ self-reported information and 24-hour dietary recall, which may have heightened the risk of diagnostic bias and potential recall bias. Second, although adjustments were made for tumor-related factors, such as since first cancer diagnosis, NHANES database lacks comprehensive data regarding the staging and treatment of specific cancers. Third, the calculation of the Oxidative Balance Score is predicated on the assumption that all oxidants and antioxidants exhibit a linear correlation with the level of oxidative stress, failing to consider the potential threshold effect of antioxidants. It is plausible that high doses of antioxidants may induce toxic pro-oxidant activity due to this threshold effect. Finally, despite our efforts to account for several confounding variables, the possibility of unrecognized confounders in our analyses and subgroup analyses cannot be entirely ruled out.
Future research plans should prioritize large-scale, multicenter randomized controlled trials to rigorously assess the effectiveness of interventions aimed at improving oxidative balance scores on survival outcomes for cancer survivors. Additionally, there is an urgent need for more comprehensive in vitro mechanistic studies to validate the potential mechanisms of various antioxidant therapies in this population. These efforts will provide stronger guidance for clinical practice and advance our understanding of cancer management.
In conclusion, the present study showed a significant negative correlation between OBS and both all-cause mortality and cancer-specific mortality among cancer survivors, utilizing data from NHANES. Specifically, higher OBS scores, indicating increased exposure to antioxidant factors relative to pro-oxidant factors in diet and lifestyle, were linked to lower risks of all-cause and cancer-specific mortality. Similar associations were noted when the dietary and lifestyle components of the OBS were analyzed separately. These findings suggest that the OBS may offer further insights into the prognostic impact of lifestyle interventions on survival rates among cancer survivors.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
The National Health and Nutrition Examination Survey (NHANES) is a publicly accessible database approved by the Institutional Review Board (IRB) of the National Center for Health Statistics. All participants provided written informed consent during their participation in the national survey. As secondary analysis does not require additional IRB approval, this study was exempt from further ethical review and approval. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants’ legal guardians/next of kin in accordance with the national legislation and institutional requirements.
QG: Conceptualization, Data curation, Investigation, Methodology, Validation, Writing – original draft. XZ: Conceptualization, Investigation, Methodology, Writing – original draft, Formal analysis, Supervision. MC: Conceptualization, Formal analysis, Investigation, Methodology, Project administration, Writing – original draft. RX: Funding acquisition, Supervision, Writing – review & editing. QZ: Supervision, Writing – review & editing, Formal analysis, Resources.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the National Natural Science Foundation of China (82070197 and 82370232).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1541675/full#supplementary-material
1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. (2024) 74:229–63. doi: 10.3322/caac.21834
2. Narimatsu H, Yaguchi YT. The role of diet and nutrition in cancer: prevention, treatment, and survival. Nutrients. (2022) 14:3329. doi: 10.3390/nu14163329
3. Mishra A, Giuliani G, Longo VD. Nutrition and dietary restrictions in cancer prevention. Biochim Biophys Acta Rev Cancer. (2024) 1879:189063. doi: 10.1016/j.bbcan.2023.189063
4. Castro-Espin C, Agudo A. The role of diet in prognosis among cancer survivors: A systematic review and meta-analysis of dietary patterns and diet interventions. Nutrients. (2022) 14:348. doi: 10.3390/nu14020348
5. Azmanova M, Pitto-Barry A. Oxidative stress in cancer therapy: friend or enemy? Chembiochem. (2022) 23:e202100641. doi: 10.1002/cbic.202100641
6. Son DH, Lee HS, Seol SY, Lee YJ, Lee JH. Association between the oxidative balance score and incident chronic kidney disease in adults. Antioxid (Basel). (2023) 12:235. doi: 10.3390/antiox12020335
7. Stanner SA, Hughes J, Kelly CN, Buttriss J. A review of the epidemiological evidence for the ‘antioxidant hypothesis’. Public Health Nutr. (2004) 7:407–22. doi: 10.1079/PHN2003543
8. Goodman M, Bostick RM, Dash C, Terry P, Flanders WD, Mandel J. A summary measure of pro- and anti-oxidant exposures and risk of incident, sporadic, colorectal adenomas. Cancer Causes Control. (2008) 19:1051–64. doi: 10.1007/s10552-008-9169-y
9. Hernandez-Ruiz A, Garcia-Villanova B, Guerra-Hernandez E, Amiano P, Ruiz-Canela M, Molina-Montes EA. Review of A priori defined oxidative balance scores relative to their components and impact on health outcomes. Nutrients. (2019) 11:774. doi: 10.3390/nu11040774
10. Yun B, King M, Draz MS, Kline T, Rodriguez-Palacios A. Oxidative reactivity across kingdoms in the gut: Host immunity, stressed microbiota and oxidized foods. Free Radic Biol Med. (2022) 178:97–110. doi: 10.1016/j.freeradbiomed.2021.11.009
11. Kong SY, Goodman M, Judd S, Bostick RM, Flanders WD, McClellan W. Oxidative balance score as predictor of all-cause, cancer, and noncancer mortality in a biracial US cohort. Ann Epidemiol. (2015) 25:256–262 e1. doi: 10.1016/j.annepidem.2015.01.004
12. Johnson CL, Paulose-Ram R, Ogden CL, Carroll MD, Kruszon-Moran D, Dohrmann SM, et al. National health and nutrition examination survey: analytic guidelines, 1999-2010. Vital Health Stat 2. (2013) 161):1–24.
13. Zhang W, Peng SF, Chen L, Chen HM, Cheng XE, Tang YH. Association between the oxidative balance score and telomere length from the national health and nutrition examination survey 1999-2002. Oxid Med Cell Longev. (2022) 2022:1345071. doi: 10.1155/2022/1345071
14. Liang J, Huang S, Jiang N, Kakaer A, Chen Y, Liu M, et al. Association between joint physical activity and dietary quality and lower risk of depression symptoms in US adults: cross-sectional NHANES study. JMIR Public Health Surveill. (2023) 9:e45776. doi: 10.2196/45776
15. Hicks CW, Wang D, Matsushita K, Windham BG, Selvin E. Peripheral neuropathy and all-cause and cardiovascular mortality in U.S. Adults: A prospective cohort study. Ann Intern Med. (2021) 174:167–74. doi: 10.7326/M20-1340
16. Li K, Deng Z, Lei C, Ding X, Li J, Wang C. The role of oxidative stress in tumorigenesis and progression. Cells. (2024) 13:441. doi: 10.3390/cells13050441
17. Garg M, Braunstein G, Koeffler HP. LAMC2 as a therapeutic target for cancers. Expert Opin Ther Targets. (2014) 18:979–82. doi: 10.1517/14728222.2014.934814
18. Bishayee A, Sethi G. Bioactive natural products in cancer prevention and therapy: Progress and promise. Semin Cancer Biol. (2016) 40-41:1–3. doi: 10.1016/j.semcancer.2016.08.006
19. Moore JM, Correa R, Rosenberg SM, Hastings PJ. Persistent damaged bases in DNA allow mutagenic break repair in Escherichia coli. PloS Genet. (2017) 13:e1006733. doi: 10.1371/journal.pgen.1006733
20. Russo M, Crisafulli G, Sogari A, Reilly NM, Arena S, Lamba S, et al. Adaptive mutability of colorectal cancers in response to targeted therapies. Science. (2019) 366:1473–80. doi: 10.1126/science.aav4474
21. Messina S, De Simone G, Ascenzi P. Cysteine-based regulation of redox-sensitive Ras small GTPases. Redox Biol. (2019) 26:101282. doi: 10.1016/j.redox.2019.101282
22. Piskounova E, Agathocleous M, Murphy MM, Hu Z, Huddlestun SE, Zhao Z, et al. Oxidative stress inhibits distant metastasis by human melanoma cells. Nature. (2015) 527:186–91. doi: 10.1038/nature15726
23. Nieto MA, Huang RY, Jackson RA, Thiery JP. Emt: 2016. Cell. (2016) 166:21–45. doi: 10.1016/j.cell.2016.06.028
24. Kim YM, Muthuramalingam K, Cho M. Redox regulation of NOX isoforms on FAK((Y397))/SRC((Y416)) phosphorylation driven epithelial-to-mesenchymal transition in Malignant cervical epithelial cells. Cells. (2020) 9:1555. doi: 10.3390/cells9061555
25. Pani G, Galeotti T, Chiarugi P. Metastasis: cancer cell’s escape from oxidative stress. Cancer Metastasis Rev. (2010) 29:351–78. doi: 10.1007/s10555-010-9225-4
26. Xi C, Hu Y, Buckhaults P, Moskophidis D, Mivechi NF. Heat shock factor Hsf1 cooperates with ErbB2 (Her2/Neu) protein to promote mammary tumorigenesis and metastasis. J Biol Chem. (2012) 287:35646–57. doi: 10.1074/jbc.M112.377481
27. Li W, Cao L, Han L, Xu Q, Ma Q. Superoxide dismutase promotes the epithelial-mesenchymal transition of pancreatic cancer cells via activation of the H2O2/ERK/NF-kappaB axis. Int J Oncol. (2015) 46:2613–20. doi: 10.3892/ijo.2015.2938
28. Zhang Q, Han Q, Yang Z, Ni Y, Agbana YL, Bai H, et al. G6PD facilitates clear cell renal cell carcinoma invasion by enhancing MMP2 expression through ROS−MAPK axis pathway. Int J Oncol. (2020) 57:197–212. doi: 10.3892/ijo.2020.5041
29. Jiang J, Wang K, Chen Y, Chen H, Nice EC, Huang C. Redox regulation in tumor cell epithelial-mesenchymal transition: molecular basis and therapeutic strategy. Signal Transduct Target Ther. (2017) 2:17036. doi: 10.1038/sigtrans.2017.36
30. Gu H, Li B, Xiang L, Xu Z, Tang Y, Zhu Z, et al. Association between oxidative stress exposure and colorectal cancer risk in 98,395 participants: results from a prospective study. Front Nutr. (2023) 10:1284066. doi: 10.3389/fnut.2023.1284066
Keywords: oxidative balance score, all-cause mortality, cancer mortality, cancer survivors, diet, lifestyle
Citation: Gao Q, Zhu X, Chen M, Xia R and Zhang Q (2025) Association between oxidative balance score and all-cause and cancer-specific mortality among cancer survivors. Front. Immunol. 16:1541675. doi: 10.3389/fimmu.2025.1541675
Received: 08 December 2024; Accepted: 03 March 2025;
Published: 20 March 2025.
Edited by:
Anand Rotte, Arcellx Inc, United StatesReviewed by:
Elizabeth Ann Salerno, Washington University in St. Louis, United StatesCopyright © 2025 Gao, Zhu, Chen, Xia and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Rong Xia, eGlhcm9uZ2NuQDE2My5jb20=; Qi Zhang, ZnJpZGF5MDQ1MUAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.