
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Immunol. , 27 February 2025
Sec. Autoimmune and Autoinflammatory Disorders : Autoimmune Disorders
Volume 16 - 2025 | https://doi.org/10.3389/fimmu.2025.1514306
This article is part of the Research Topic Imaging in the Diagnosis and Treatment of Eye Diseases View all 17 articles
Purpose: To report an uncommon case of Vogt-Koyanagi-Harada (VKH) disease in an elderly patient with unilateral choroidal detachment and describe its multimodal imaging features and prognosis.
Method: Case report and literature review of clinical features in VKH with choroidal detachment.
Results: A 76-year-old woman presented with bilateral blurred vision and headache 6 months prior to visiting our hospital. She was diagnosed with iridocyclitis at another hospital and received local anti-inflammatory treatment without improvement. Slit-lamp examination showed bilateral mutton-fat and dust-like keratic precipitates, anterior chamber and vitreous cells, and posterior synechiae in the right eye. Fundus examination revealed bilateral optic disc swelling and choroidal detachment in the left eye. Fluorescein angiography revealed bilateral optic disc leakage, punctate hyperfluorescence in the posterior pole, and elevated fluorescence leakage in the left eye’s temporal area. Indocyanine Green Angiography showed multiple of choroidal hypoperfusion areas in the left eye, with an elevated fluorescence blockage on the temporal side. Optical coherence tomography showed subretinal fluid, wavy retinal pigment epithelium, and choroidal thickening in both eyes. Based on ocular and neurological findings, the patient was diagnosed with bilateral VKH. After ruling out infectious factors, she received high-dose systemic corticosteroids and immunosuppressants. The choroidal detachment and serous retinal detachment gradually resolved.
Conclusion: This case is the first report of unilateral choroidal detachment associated with VKH in an elderly patient. VKH patients with choroidal detachment reported in previous studies were predominantly elderly and Asian, characterized by optic disc hyperfluorescence and choroidal detachment. Multimodal imaging can help clinicians better diagnose and manage atypical types of VKH.
Vogt-Koyanagi-Harada (VKH) disease is an autoimmune disorder characterized by chronic granulomatous uveitis in both eyes, often accompanied by changes in neurological system, inner ear, and skin. It is more common in certain pigmented races, particularly Asian, Middle Eastern, Hispanic, and Native American (1–3). The most common age for onset of VKH is between 20 and 50 years (2, 4), but children (5) and elderly individuals (6) can also be affected. Women are more likely to develop the disease than men (7, 8). Immunopathological studies indicated that VKH is associated with autoimmune inflammation mediated by CD4+ T cells targeting melanocytes (9). Tyrosinase peptides probably acts as the autoantigen in VKH, with T cells T cells mounting an immune response by targeting the tyrosinase peptides produced by melanocytes (10). In addition, the human leukocyte antigen (HLA) genotype (DRB1*0405) is closely associated with genetic susceptibility to VKH (11, 12). The ocular features of VKH mainly include pan-uveitis with serous retinal detachment, and in the late stage, sunset glow fundus may appear (3). Systemic manifestations in the prodromal phase include headache, fever, neck stiffness, vertigo, tinnitus, and hearing loss. In later stages of the disease, vitiligo and poliosis may occur (7). Here, we report a case of an elderly VKH patient with unilateral choroidal detachment and bilateral serous retinal detachment. Furthermore, we summarize and review the clinical features of previously reported cases of choroidal detachment in VKH.
A 76-year-old woman presented with progressive bilateral blurred vision for 6 months. Prior to the significant vision loss, she sought consultation at a local hospital’ s neurology department due to headache, dizziness, and neck stiffness. The local ophthalmologist diagnosed her with bilateral iridocyclitis and treated her with non-steroidal anti-inflammatory and steroid eye drops. However, her symptoms did not improve, and vision progressively deteriorated. Her medial and ocular history were unremarkable and the patient denied oral ulcers, joint pain, or skin rash. At the first visit to our hospital, her visual acuity was 0.1 in the right eye and counting fingers (with no improvement upon correction) in the left. The intraocular pressure was 14 mmHg in the right eye and 12 mmHg in the left. The axial length was 23.61 mm in the right eye and 23.37 mm in the left. The scleral thickness was 0.48mm in the right eye and 0.46mm in the left. Slit-lamp examination revealed bilateral ciliary injection, a slightly shallow anterior chamber, a large number of mutton-fat and dust-like keratic precipitates, anterior chamber cells (+++), Tyndall effect (+++), and lens opacity (Figures 1A–D). The right eye also showed iris posterior synechiae. Apparent vitreous opacity with a large number of inflammatory cells were detected. Fundus examination showed bilateral mild optic disc swelling and blurred disc margins. The cup-to-disc ratio was small, with retinal folds and scattered yellow-white lesions visible in the posterior pole. Choroidal detachment was observed in the temporal region of the left eye (Figures 1E, F). Fundus autofluorescence examination showed low autofluorescence in the vitreous of both eyes, with a hypoautofluorescent elevated area visible in the temporal region of the left eye (Figures 1G, H). Optical coherence tomography (OCT) revealed wavy retinal pigment epithelium (RPE), subretinal fluid, and significant choroidal thickening in both eyes. The subretinal fluid in the left eye appeared compartmentalized (Figures 1I, J). Fundus fluorescein angiography (FFA) in the venous phase showed retinal vein staining of both eyes, with blurred disc margins and hyperfluorescence of the optic disc (Figures 2A–D). Punctate and patchy hyperfluorescence was observed in the posterior pole of the right eye. The left eye showed numerous punctate and linear areas of hypofluorescence, with retinal elevation in the temporal and inferior peripheral regions accompanied by vascular wall staining. Severe hyperfluorescent leakage of the optic disc was observed in the late stage. Indocyanine green angiography showed numerous punctate hypofluorescent areas in all quadrants of the retina in both eyes during both early and late phases (Figures 2E, F). Linear hypofluorescence was observed in the left eye, with fluorescent blockage in the temporal peripheral region. Ocular ultrasonography revealed weak punctate and continuous band-like echoes in the vitreous of both eyes, and diffuse thickening of the ocular wall echo in the right eye. In the left eye, one end of the echo was attached to the peripheral ocular wall, while the other end was connected to the equatorial ocular wall, with an anechoic area beneath. Color Doppler Flow Imaging detected blood flow signals along the band-like echoes in both eyes. Retinal detachment was observed in both eyes, along with choroidal detachment in the left eye. No associated scleral thickening, masses, or “T” sign was noted (Figure 2G). Ultrasound biomicroscopy (UBM) revealed abnormal anterior segment echoes in both eyes, with anterior synechiae of the iris and suprachoroidal effusion in the left eye (Figure 2H). The results of the complete blood count, C-reactive protein, erythrocyte sedimentation rate, rheumatoid factor, serology for syphilis, HIV antibody test, hepatitis B virus antigen, hepatitis C virus antigen, serum angiotensin converting enzyme, chest CT, and T-spot test were all within normal limits. The patient’s HLA typing was negative. The patient was diagnosed with bilateral complete VKH based on her ocular abnormalities and extra-ocular symptoms (13). The patient initially received a posterior subtenon injection of 20 mg triamcinolone acetonide in both eyes. After ruling out infectious causes, the patient was started on oral prednisone (1.0 mg/kg) with gradual tapering. Concurrently, the patient began treatment with oral methotrexate, and topical treatments with prednisolone acetate, pranoprofen, and compound tropicamide eye drops. Two weeks later, the choroidal detachment in the left eye and retinal detachment in the right eye completely resolved. The serous retinal detachment in the left eye significantly subsided, and the choroidal thickness in both eyes markedly decreased (Figures 3A–F). Ocular ultrasonography showed resolution of the choroidal detachment in the left eye (Figure 2I), and UBM revealed abnormal anterior segment echoes in both eyes and the resolution of suprachoroidal effusion in the left eye (Figure 2J). At the one and a half months follow-up, the patient’s condition remained stable, with visual acuity of 0.8 in the right eye and 0.6 in the left eye. OCT showed resolution of the serous retinal detachment in both eyes, with choroidal thickness returning to normal. In the right eye, the ellipsoid zone in the macular area appeared thinned, while the left eye showed disruption and loss of the ellipsoid zone, accompanied by high-reflective signals (Figures 3G, H).
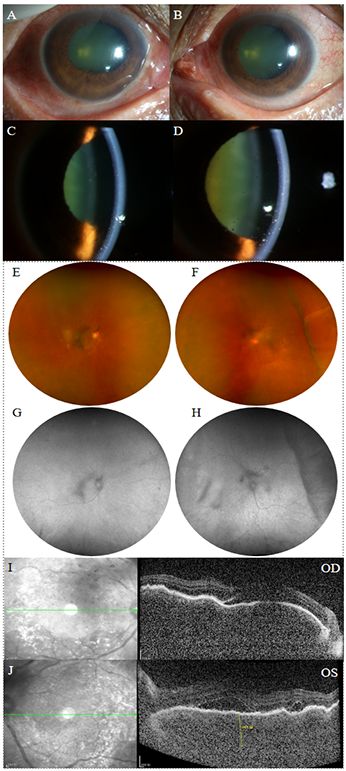
Figure 1. Anterior segment photography, fundus photography, fundus autofluorescence, and OCT images of the patient at her initial visit to our hospital. (A-D) Both eyes showed ciliary injection, a slightly shallow anterior chamber, a large amount of mutton-fat and dust-like keratic precipitates and anterior chamber cells (+++). The right eye showed iris posterior synechiae. (E, F) Fundus photography showed bilateral mild optic disc swelling and blurred disc margins, with retinal folds and scattered yellow-white lesions in the posterior pole. Choroidal detachment was observed in the temporal region of the left eye. (G, H) Fundus autofluorescence showed low autofluorescence in the vitreous of both eyes, with a hypoautofluorescent elevated area in the temporal region of the left eye. (I, J) OCT revealed wavy RPE, subretinal fluid, and significant choroidal thickening in both eyes. The subretinal fluid in the left eye appeared compartmentalized.
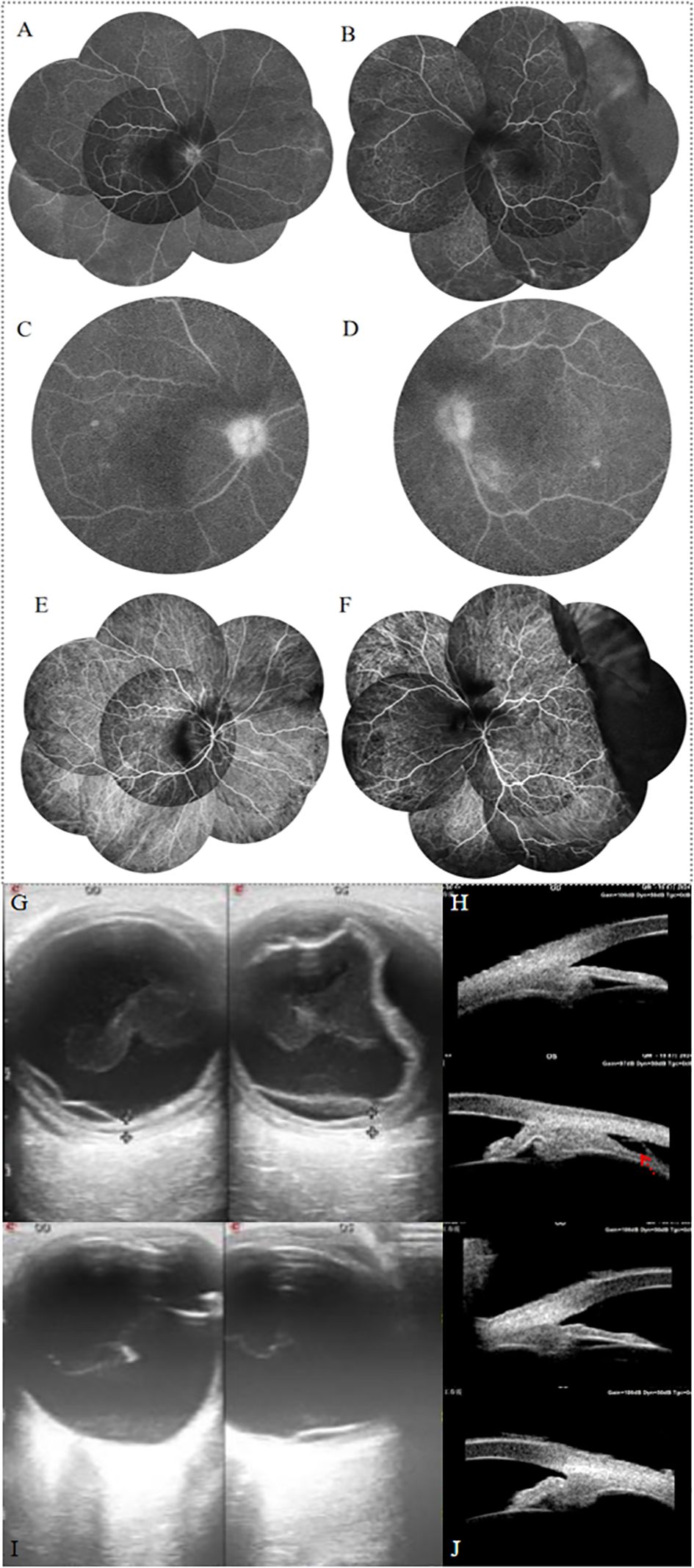
Figure 2. (A, B) FFA revealed peripheral retinal vessel wall staining of both eyes, with blurred disc margins and hyperfluorescence of the optic disc. The left eye showed numerous punctate and linear areas of hypofluorescence, with retinal elevation in the temporal and inferior peripheral regions accompanied by vascular wall staining. (C, D) Late-phase FFA showed optic disc leakage in both eyes, with punctate-patchy hypofluorescence in the posterior pole. (E, F) Indocyanine green angiography showed numerous punctate and linear hypofluorescence in both eyes. In the left eye, multiple areas of choroidal hypoperfusion are observed, with an elevated fluorescence blockage on the temporal side. (G) Ocular ultrasonography revealed weak punctate and continuous band-like echoes in the vitreous of both eyes, and diffuse thickening of the ocular wall echo in the right eye. In the left eye, one end of the echo was attached to the peripheral ocular wall, while the other end was connected to the equatorial ocular wall, with an anechoic area beneath. Color Doppler Flow Imaging detected blood flow signals along the band-like echoes in both eyes. Retinal detachment was observed in both eyes, along with choroidal detachment in the left eye. No associated scleral thickening, masses, or “T” sign was noted. (H) UBM revealed abnormal anterior segment echoes in both eyes, with anterior synechiae of the iris and suprachoroidal effusion in the left eye (red arrow). After 2 weeks of treatment, Ocular ultrasonography showed significant recovery of the choroidal detachment in the left eye (I), and UBM revealed abnormal anterior segment echoes in both eyes and the resolution of suprachoroidal effusion in the left eye (J).
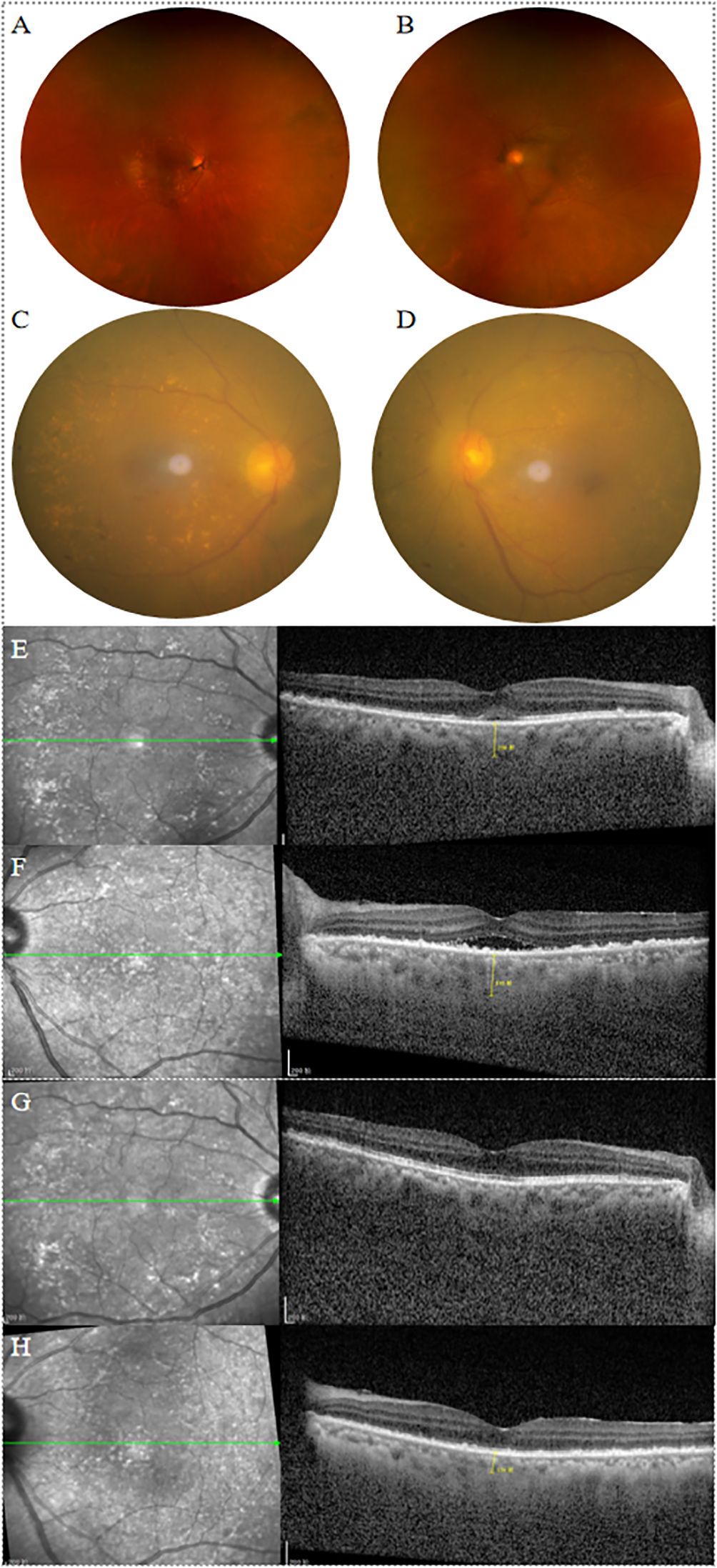
Figure 3. (A-D) After 2 weeks of treatment, fundus images showed vitreous opacity, a reddish optic disc with clear margins, dilated veins, and flat retina in both eyes. Numerous yellow-white lesions were observed in the posterior pole of both eyes, and choroidal detachment in the left eye had resolved. (E, F) After 2 weeks of treatment, OCT showed significant absorption of subretinal fluid in both eyes, with a marked decrease in choroidal thickness. (G, H) After one and a half months of treatment, OCT showed resolution of the serous retinal detachment in both eyes, with choroidal thickness returning to normal. In the right eye, the ellipsoid zone in the macular area appeared thinned, while the left eye showed disruption and loss of the ellipsoid zone, accompanied by high-reflective signals.
A search of the PubMed database was conducted for published cases of VKH-associated choroidal detachment. The keywords used were Vogt-Koyanagi-Harada disease/Vogt-Koyanagi-Harada Syndrome/Vogt-Koyanagi-Harada’s disease/choroidal detachment. Studies published in languages other than English were excluded. We reviewed the clinical features, multimodal imaging findings, treatments and prognosis of previously reported cases of choroidal detachment in VKH.
VKH is a multisystem autoimmune disease characterized by bilateral chronic granulomatous uveitis. Extraocular manifestations include neurological symptoms, auditory disturbances, as well as skin and hair abnormalities. Choroidal detachment (14), optic disc swelling type (15), and unilateral serous retinal detachment (16) are three uncommon clinical manifestations of VKH. This case is the first report of unilateral choroidal detachment in an elderly VKH patient, with multimodal imaging playing a crucial role in facilitating early diagnosis. FFA revealed severe optic disc leakage and peripheral retinal vessel wall staining, likely due to inflammation-induced vascular edema and leakage. The linear hypofluorescent areas in both eyes were attributed to the presence of choroidal folds, which were likely caused by choroidal thickening. When the patient first presented to our hospital, OCT revealed significant choroidal thickening of nearly 600 μm. The thickening was primarily associated with inflammatory infiltration and increased exudation (17). Elevated fluorescein leakage was observed in the temporal region of the left eye, indicating the presence of choroidal detachment. Severe choroidal inflammation was likely responsible for the choroidal detachment in this case. Inflammation can increase the permeability of the choroidal capillaries, resulting in vascular leakage (14, 18). The unilateral choroidal detachment in this patient occurred in the eye with more severe inflammation, and the rapid improvement following corticosteroid anti-inflammatory treatment further supports this cause. Jae et al. reported that compression of the vortex vein may lead to increased extravascular fluid retention and choroidal detachment (14). However, when our patient first presented to our hospital, she had a large number of inflammatory cells in the anterior chamber, posterior synechiae of the iris, significant cataracts, and vitreous opacities. Furthermore, due to the limitations of our equipment and imaging technique, both the 10-minute FFA and late-stage ICGA results were unclear, and we were unable to visualize the vortex vein. We recommend that future studies use ultra-widefield ICGA to further explore the mechanisms underlying choroidal detachment. Choroidal detachment is an uncommon manifestation of VKH, with only five cases of VKH-related choroidal detachment have been reported in English literature to date (14, 18–21). Table 1 summarizes the clinical characteristics of these five cases. There were three males and two females, with an age of onset ranging from 59 to 68 years. The onset age of our patient was 76 years, suggesting a higher incidence of choroidal detachment in elderly VKH patients (22). Three of the patients were Japanese, while the other two were Caucasian and Korean, respectively, suggesting a higher prevalence of VKH-related choroidal detachment among Asians. All patients presented with bilateral choroidal detachment, with one patient additionally diagnosed with synovitis-acne-pustulosis-hyperostosis-osteitis syndrome (spondylitis and palmoplantar pustulosis). In four patients, choroidal detachment resolved following treatment with systemic corticosteroids alone or combined with immunosuppressants. The time required for treatment ranged from 4 to 95 days.
Uveal effusion syndrome (UES) with peripheral choroidal detachment and exudative retinal detachment is an important differential diagnosis in this case. UES is characterized by ciliochoroidal detachment, exudative retinal detachment, and secondary RPE changes (23). UES can be classified into three types: Type 1 is characterized by nanophthalmos with scleral abnormalities, Type 2 involves normal axial length with scleral abnormalities, and Type 3 is idiopathic UES, presenting with both normal axial length and normal sclera (24). UES typically occurs in both eyes but usually presents without signs of anterior segment inflammation. Unlike the rapid onset of VKH, UES usually has a chronic onset and appears to be unresponsive to corticosteroid treatment (25). Our patient had normal axial length in both eyes, and ocular ultrasonography showed no evidence of scleral thickening. However, the rapid onset, presence of anterior segment inflammation, and good response to corticosteroid treatment help distinguish this case from idiopathic UES. Posterior scleritis is another important differential diagnosis in this case. It is an ocular inflammatory disease that typically presents with pain, redness, photophobia, and decreased vision. Around 40% of posterior scleritis cases are associated with systemic conditions, such as rheumatoid polyarthritis, systemic lupus erythematosus, and pANCA(+) systemic vasculitis (26). Ocular ultrasonography in posterior scleritis shows thickening of the choroid and sclera. A characteristic “T sign” can be observed when retrobulbar edema surrounds the optic nerve (27, 28). Posterior scleritis is typically unilateral, with only about one third of cases involving both eyes (29, 30). Our patient had no eye pain or the aforementioned ocular ultrasonography findings, and both of her eyes were affected simultaneously. Additionally, our case can be differentiated from choroidal metastasis. In later stages of choroidal metastasis, patients can exhibit pain, diplopia, uveitis, and secondary glaucoma due to metastasis to the ciliary body or iris. B-scan ultrasonography typically reveals an elevated subretinal mass and secondary exudative retinal detachment (31). OCT findings are characterized by the “lumpy bumpy appearance” (32). Our case did not show these imaging findings and can be differentiated through the patient’s medical history and systemic examination.
VKH presents with a wide range of clinical manifestations. In addition to choroidal detachment, the optic disc swelling type and unilateral exudative retinal detachment are also uncommon manifestations. The optic disc swelling type of VKH is primarily characterized by significant optic disc edema, with OCT detecting no or only minimal serous retinal detachment. Patients with optic disc swelling type VKH tend to be predominantly female and have an older age of onset. These patients usually have milder visual impairment prior to treatment; however, treatment is often delayed, increasing the risk of progressing to a chronic form of the disease (15). Nakao et al. suggested that the incidence of disc swelling is associated with disc morphology, with the cup-to-disc ratio in eyes with optic disc swelling in VKH was smaller (33). The main finding on FFA in this case was optic disc swelling. The patient’s age and optic disc morphology are consistent with the typical characteristics of the optic disc swelling type, but the presence of serous retinal detachment indicates a more severe inflammatory response in the left eye. If the patient presents with only optic disc swelling, differentiation from optic neuritis is necessary. EDI-OCT showed that the initial choroidal thickness and the changes in choroidal thickness before and after treatment are greater in the optic disc swelling type of VKH than in optic neuritis (34). The diagnostic criteria for VKH require involvement of both eyes (8). Although there are rare reports of unilateral VKH (16, 35–37), it is inherently a bilateral disease. EDI-OCT and ICGA can detect subclinical inflammation in the contralateral eye. Even with predominantly unilateral presentation, prompt diagnosis and systemic corticosteroid treatment are essential. Recognizing these atypical types of VKH is essential to help clinicians avoid missed diagnoses, misdiagnoses and treatment delay.
We report a case of an elderly female with VKH, presenting with unilateral choroidal detachment and bilateral serous retinal detachment, and describe her multimodal imaging findings. This case, along with previously reported cases, indicates that choroidal detachment is an uncommon manifestation of VKH. VKH-related choroidal detachment is more commonly seen in patients over 60 years old, typically presenting with optic disc swelling. Systemic corticosteroid therapy has proven to be effective in its treatment. Furthermore, multimodal imaging enables clinicians to make early and precise diagnoses of atypical VKH, leading to earlier detection, timely treatment, and improved outcomes.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
The studies involving humans were approved by Beijing Tongren Hospital, Capital Medical University, Beijing, China. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
CP: Writing – original draft, Writing – review & editing. YJ: Writing – review & editing. CC: Writing – original draft, Writing – review & editing.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported by the National Natural Science Foundation of China (No. 82101147).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Rao NA, Gupta A, Dustin L, Chee SP, Okada AA, Khairallah M, et al. Frequency of distinguishing clinical features in vogt-koyanagi-harada disease. Ophthalmology. (2010) 117:591–9:9.e1. doi: 10.1016/j.ophtha.2009.08.030
2. O’Keefe GA, Rao NA. Vogt-koyanagi-harada disease. Surv Ophthalmol. (2017) 62:1–25. doi: 10.1016/j.survophthal.2016.05.002
3. Joye A, Suhler E. Vogt-koyanagi-harada disease. Curr Opin Ophthalmol. (2021) 32:574–82. doi: 10.1097/icu.0000000000000809
4. Damico FM, Kiss S, Young LH. Vogt-koyanagi-harada disease. Semin Ophthalmol. (2005) 20:183–90. doi: 10.1080/08820530500232126
5. Hamade IH, Al Shamsi HN, Al Dhibi H, Chacra CB, Abu-El-Asrar AM, Tabbara KF. Uveitis survey in children. Br J Ophthalmol. (2009) 93:569–72. doi: 10.1136/bjo.2008.148015
6. Ikeda N, Hayasaka S, Hayasaka Y. Uveitis and pseudouveitis presenting for the first time in Japanese elderly patients. Ophthalmologica. (2005) 219:263–6. doi: 10.1159/000086108
7. Tayal A, Daigavane S, Gupta N. Vogt-koyanagi-harada disease: A narrative review. Cureus. (2024) 16:e58867. doi: 10.7759/cureus.58867
8. Group SoUNSW. Classification criteria for vogt-koyanagi-harada disease. Am J Ophthalmol. (2021) 228:205–11. doi: 10.1016/j.ajo.2021.03.036
9. Norose K, Yano A. Melanoma specific th1 cytotoxic T lymphocyte lines in vogt-koyanagi-harada disease. Br J Ophthalmol. (1996) 80:1002–8. doi: 10.1136/bjo.80.11.1002
10. Kobayashi H, Kokubo T, Takahashi M, Sato K, Miyokawa N, Kimura S, et al. Tyrosinase epitope recognized by an hla-dr-restricted T-cell line from a vogt-koyanagi-harada disease patient. Immunogenetics. (1998) 47:398–403. doi: 10.1007/s002510050375
11. Shindo Y, Inoko H, Yamamoto T, Ohno S. Hla-drb1 typing of vogt-koyanagi-harada’s disease by pcr-rflp and the strong association with drb1*0405 and drb1*0410. Br J Ophthalmol. (1994) 78:223–6. doi: 10.1136/bjo.78.3.223
12. Kim MH, Seong MC, Kwak NH, Yoo JS, Huh W, Kim TG, et al. Association of hla with vogt-koyanagi-harada syndrome in koreans. Am J Ophthalmol. (2000) 129:173–7. doi: 10.1016/s0002-9394(99)00434-1
13. Read RW, Holland GN, Rao NA, Tabbara KF, Ohno S, Arellanes-Garcia L, et al. Revised diagnostic criteria for vogt-koyanagi-harada disease: report of an international committee on nomenclature. Am J Ophthalmol. (2001) 131:647–52. doi: 10.1016/s0002-9394(01)00925-4
14. Kwak JH, Baek J, Ra H. Multimodal imaging of annular choroidal detachment in a patient with vogt-koyanagi-harada disease. Ocul Immunol Inflammation. (2021) 29:911–4. doi: 10.1080/09273948.2019.1704023
15. Okunuki Y, Tsubota K, Kezuka T, Goto H. Differences in the clinical features of two types of vogt-koyanagi-harada disease: serous retinal detachment and optic disc swelling. Jpn J Ophthalmol. (2015) 59:103–8. doi: 10.1007/s10384-014-0367-8
16. Forster DJ, Green RL, Rao NA. Unilateral manifestation of the vogt-koyanagi-harada syndrome in a 7-year-old child. Am J Ophthalmol. (1991) 111:380–2. doi: 10.1016/s0002-9394(14)72334-7
17. Maruko I, Iida T, Sugano Y, Oyamada H, Sekiryu T, Fujiwara T, et al. Subfoveal choroidal thickness after treatment of vogt-koyanagi-harada disease. Retina. (2011) 31:510–7. doi: 10.1097/IAE.0b013e3181eef053
18. Yamamoto N, Naito K. Annular choroidal detachment in a patient with vogt-koyanagi-harada disease. Graefes Arch Clin Exp Ophthalmol. (2004) 242:355–8. doi: 10.1007/s00417-003-0832-4
19. Kishi A, Nao-i N, Sawada A. Ultrasound biomicroscopic findings of acute angle-closure glaucoma in vogt-koyanagi-harada syndrome. Am J Ophthalmol. (1996) 122:735–7. doi: 10.1016/s0002-9394(14)70498-2
20. Elaraoud I, Andreatta W, Jiang L, Damer K, Al-Ibrahim J. A mystery of bilateral annular choroidal and exudative retinal detachment with no systemic involvement: is it part of vogt-koyanagi-harada disease spectrum or a new entity? Case Rep Ophthalmol. (2017) 8:1–6. doi: 10.1159/000453543
21. Hasegawa M, Fukutomi A, Jinno A, Fujita K, Kamei M. Unusual case of vogt-koyanagi-harada disease associated with sapho syndrome: A case report. Case Rep Ophthalmol. (2018) 9:202–8. doi: 10.1159/000487226
22. Kiyomoto C, Imaizumi M, Kimoto K, Abe H, Nakano S, Nakatsuka K. Vogt-koyanagi-harada disease in elderly Japanese patients. Int Ophthalmol. (2007) 27:149–53. doi: 10.1007/s10792-007-9067-4
23. Elagouz M, Stanescu-Segall D, Jackson TL. Uveal effusion syndrome. Surv Ophthalmol. (2010) 55:134–45. doi: 10.1016/j.survophthal.2009.05.003
24. Hodges B, Omoruyi F, Allison K. Uveal effusion syndrome: A case report. J Med Case Rep. (2024) 18:169. doi: 10.1186/s13256-024-04496-1
25. Gao Y, Wu Q, Xu J, Tian Q, Xie X, Du X, et al. Management and follow-up of uveal effusion syndrome: A case report. BMC Ophthalmol. (2023) 23:355. doi: 10.1186/s12886-023-03078-x
26. Lavric A, Gonzalez-Lopez JJ, Majumder PD, Bansal N, Biswas J, Pavesio C, et al. Posterior scleritis: analysis of epidemiology, clinical factors, and risk of recurrence in a cohort of 114 patients. Ocul Immunol Inflammation. (2016) 24:6–15. doi: 10.3109/09273948.2015.1005240
27. Robertson R, Ansari FA, Gafoor S, Idolor ON, Amakye D. Posterior scleritis: A case report and literature review of the management of the condition. Cureus. (2024) 16:e61537. doi: 10.7759/cureus.61537
28. Benson WE. Posterior scleritis. Surv Ophthalmol. (1988) 32:297–316. doi: 10.1016/0039-6257(88)90093-8
29. Wen C, Duan H. Bilateral posterior scleritis presenting as acute primary angle closure: A case report. World J Clin cases. (2021) 9:3779–86. doi: 10.12998/wjcc.v9.i15.3779
30. Gonzalez-Gonzalez LA, Molina-Prat N, Doctor P, Tauber J, Sainz de la Maza M, Foster CS. Clinical features and presentation of posterior scleritis: A report of 31 cases. Ocul Immunol Inflammation. (2014) 22:203–7. doi: 10.3109/09273948.2013.840385
31. Kongwattananon W, Pothikamjorn T, Somkijrungroj T. Posterior segment manifestations of ocular metastasis. Curr Opin Ophthalmol. (2023) 34:522–8. doi: 10.1097/icu.0000000000000988
32. Arevalo JF, Fernandez CF, Garcia RA. Optical coherence tomography characteristics of choroidal metastasis. Ophthalmology. (2005) 112:1612–9. doi: 10.1016/j.ophtha.2005.03.030
33. Nakao K, Abematsu N, Mizushima Y, Sakamoto T. Optic disc swelling in vogt-koyanagi-harada disease. Invest Ophthalmol Vis Sci. (2012) 53:1917–22. doi: 10.1167/iovs.11-8984
34. Maruko I, Iida T, Sugano Y, Go S, Sekiryu T. Subfoveal choroidal thickness in papillitis type of vogt-koyanagi-harada disease and idiopathic optic neuritis. Retina. (2016) 36:992–9. doi: 10.1097/iae.0000000000000816
35. Usui Y, Goto H, Sakai J, Takeuchi M, Usui M, Rao NA. Presumed vogt-koyanagi-harada disease with unilateral ocular involvement: report of three cases. Graefes Arch Clin Exp Ophthalmol. (2009) 247:1127–32. doi: 10.1007/s00417-009-1068-8
36. Agrawal A, Biswas J. Unilateral vogt-koyanagi-harada disease: report of two cases. Middle East Afr J Ophthalmol. (2011) 18:82–4. doi: 10.4103/0974-9233.75898
Keywords: Vogt-Koyanagi-Harada disease, choroidal detachment, serous retinal detachment, multimodal imaging, VKH disease
Citation: Peng C, Jiao Y and Chen C (2025) Unilateral choroidal detachment in an elderly patient with Vogt-Koyanagi-Harada disease: a case report and literature review. Front. Immunol. 16:1514306. doi: 10.3389/fimmu.2025.1514306
Received: 20 October 2024; Accepted: 11 February 2025;
Published: 27 February 2025.
Edited by:
Weihua Yang, Southern Medical University, ChinaReviewed by:
Marc De Smet, MIOS - Retina and Inflammation, SwitzerlandCopyright © 2025 Peng, Jiao and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chunli Chen, Y2hlbmNodW5saTEyNUAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.