
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Immunol., 03 February 2022
Sec. Inflammation
Volume 13 - 2022 | https://doi.org/10.3389/fimmu.2022.814319
This article is part of the Research TopicThe Mechanism of Metabolic Immune Microenvironment, Inflammation and Blood Milk Barrier in MastitisView all 5 articles
Formononetin (FOR), a natural flavonoid derived from Radix Astragali, has been reported to have anti-inflammatory and anti-oxidative effects. However, its protective mechanism against mastitis is still unknown. Nuclear factor kappa-B (NF-κB) signaling pathway plays an important role in inflammation, especially mastitis. Aryl hydrocarbon receptor (AhR) is involved in inflammatory regulation and defense against diseases. We investigated the protective effect of FOR on LPS-induced mastitis in mice and the effect of Ahr and NF-κB signaling pathways on the development of mastitis. In this study, mastitis model was induced by LPS injection through the nipple duct. Protective effect of FOR on LPS-induced mastitis was assessed by FOR pretreatment. The protective mechanism of FOR against mastitis was further investigated using LPS stimulation on mouse mammary epithelial cells EpH4-Ev. The results showed that LPS-induced mammary histological injury was inhibited by FOR. FOR significantly inhibited LPS-induced MPO activity. FOR administration enhanced the integrity of blood-milk barrier. In vitro and in vivo experiments showed that FOR inhibited LPS-induced NF-κB signaling pathway activation and the production of inflammatory factors TNF-α and IL-1ß. Moreover, FOR increased the expression of tight junction protein and enhanced blood-milk barrier integrity. LPS activated AhR and Src expression. But FOR induced significant increase in AhR inhibited Src phosphorylation to exert anti-inflammatory effects. In addition, AhR antagonist CH223191 reversed the inhibition of FOR on Src expression. And the inhibition of FOR on NF-κB activation and inflammatory cytokine production were reversed by AhR antagonist CH223191. In conclusion, FOR had protective effects against LPS-induced mastitis via suppressing inflammation and enhancing blood-milk barrier integrity via AhR-induced Src inactivation.
Mastitis is an inflammatory response of mammary tissues caused by various reasons (1). Among these factors, bacteria have been known as the major cause for mastitis (2). Bacteria can reach the mammary gland and cause the inflammatory response in the mammary tissues (3). Lipopolysaccharide (LPS), a chemical component unique to the outer membrane of Gram-negative bacteria, could trigger an inflammatory response through increasing the production of inflammatory cytokines (4). Elevated inflammatory cytokines were observed in patients of mastitis and LPS-induced mouse mastitis model (5). LPS also induces the production of oxidative mediator and studies showed that both inflammatory response and oxidative stress were participated in the progression of mastitis (6, 7).
Medicinal herbs have been used to treat mastitis for a long time (8). Studies showed that a large body of herbal medicines had anti-inflammatory and anti-oxidative effects (9, 10). Formononetin (FOR), a flavonoid isolated from Radix Astragali, has been described to have anti-inflammatory and anti-oxidative activities (11). It has been reported FOR could protect rats against myocardial ischemia/reperfusion injury (12). Previous evidence suggested that FOR had protective effects against LPS-induced acute lung injury in mice (13). FOR also protected mice against concanavalin-A-induced autoimmune hepatitis through inhibiting inflammatory response (14). FOR could inhibit airway inflammation in mouse allergic asthma. In vitro, FOR significantly attenuated the expression of LPS-induced inflammatory cytokine in RAW264.7 cells (15). In addition, FOR could inhibit IL-1-induced inflammatory mediator in rat chondrocytes (16). In this study, we detected the effect of FOR on the protection against LPS-induced mastitis in mice. Our findings demonstrated that FOR protected mice against mastitis through inhibiting inflammation and enhancing blood-milk barrier integrity.
FOR (Catalog No. 94334), FITC-albumin (Catalog No. A9771), DAPI (Catalog No. D9542) and LPS (Escherichia coli serotype 055: B5, Catalog No. L2880) were obtained from Sigma (St. Louis, MO, USA). The phospho-IκBα (Tyr305, Catalog No. AF3239), NF-κB p65 (Ser311, Catalog No. AF3389), AhR (Ser36, Catalog No. AF3278) and Src (Tyr419, Catalog No. AF3162) antibodies were purchased from Affinity Biosciences (Changzhou, Jiangsu, China). ELISA kits for TNF-α (Catalog No. 430907) and IL-1β (Catalog No. 432601) were acquired from BioLegend (San Diego, CA, USA). EpH4-Evs were obtained from the American Type Culture Collection (ATCC, ATCC® CRL-3063™).
Thirty female lactating C57BL/6 mice (7-9 weeks old) were purchased from Liaoning changsheng Biotechnology Co., Ltd. (Benxi, Liaoning, China). The mice were housed in individually ventilated cages and given enough diet and water at 25 ± 1°C. Approval for this experiment was obtained from the institutional animal care and use committee of Jilin University. LPS (10 mg/kg) was dissolved in 50 μL phosphate-buffered saline (PBS) and injected into the breast ducts of L4 (on the left) and R4 (on the right) for 24 h. FOR (10, 20, 30 mg/kg) were dissolved in normal saline and administered 1 h prior LPS treatment.
The collected mammary glands were fixed in 4% paraformaldehyde. Hematoxylin and eosin (H&E) staining was used, followed by observation of morphological changes in breast tissues under a microscope and digital camera (Olympus, Tokyo, Japan).
24 h after LPS treatment, the mammary glands were collected and the myeloperoxidase (MPO) level in mammary tissues was determined using the detection kit (Catalog No. A044-1-1, Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s guidelines.
The mammary glands were collected in the same way as before. ELISA kits (BioLegend, CA, USA) were utilized to detect the production of TNF-α and IL-1ß in the mammary tissues.
The protein extraction kit (Catalog No. P0013M, Beyotime Biotechnology, Shanghai, China) was used for the extraction of total protein from cells. The concentration of protein was measured with the BCA kit (Catalog No. P0012S Beyotime Biotechnology, Shanghai, China). 30 μg protein was loaded by 12% SDS-PAGE and transferred to the PVDF membranes. Then, the membranes were blocked with 5% BSA and incubated with NF-κB and AhR signaling pathway antibodies. Finally, the membranes were incubated with secondary antibodies and detected using the ECL chemiluminescence reagent.
The collected mammary glands were fixed in 4% paraformaldehyde, embedded in paraffin and tissue sections were made. The sections were then dewaxed and antigenically repaired. 5% goat serum was used to block nonspecific interactions, overnight at 4°C. 50 mg/mL FITC-albumin mother solution was prepared with double-distilled water and diluted 40-fold with 5% goat serum. The sections were washed with PBS and incubated with FITC-albumin at 4°C overnight. The sections were washed with PBS containing 0.1% Triton and stained with DAPI to avoid light for 5-8 minutes. Finally, the sections were sealed with glycerin and observed as soon as possible.
EpH4-Ev cells were incubated in DMEM supplemented with 10% fetal bovine serum (FBS) at 37°C. The effect of FOR on cell viability was measured by MTT assay. Then, the cells were pre-treated with FOR (10, 20, and 30 μM) 1 h before LPS treatment. The cytokines were measured by ELISA 24 h after LPS treatment (1μg/mL). The key protein levels involved in the NF-κB signaling pathway were detected 30 min after LPS treatment by western blot. For AhR inhibitory experiment, cells were pretreated with FOR (30 μM) and CH223191 (10 μM) for 1 h, and then stimulated with LPS for indicated times.
All the results were expressed as means ± SEM. The data were analyzed using one way analysis of variance (ANOVA) followed by the Tukey’s post hoc test. Value of P < 0.05 was considered to be significant.
After LPS treatment, typical inflammatory manifestations such as redness, swelling, heat, and pain began to appear in the mammary tissue. Compared to normal breast tissue (Figure 1A), H&E staining showed serious mammary histopathological changes, such as inflammatory cell infiltration, edema, and hyperemia (Figure 1B). It suggested that LPS was capable of inducing mastitis in mice. However, LPS-induced mammary histopathological changes were inhibited by FOR. The histopathological changes in the mammary glands of mice gradually diminished as the increasing concentration of FOR (Figures 1C–E).
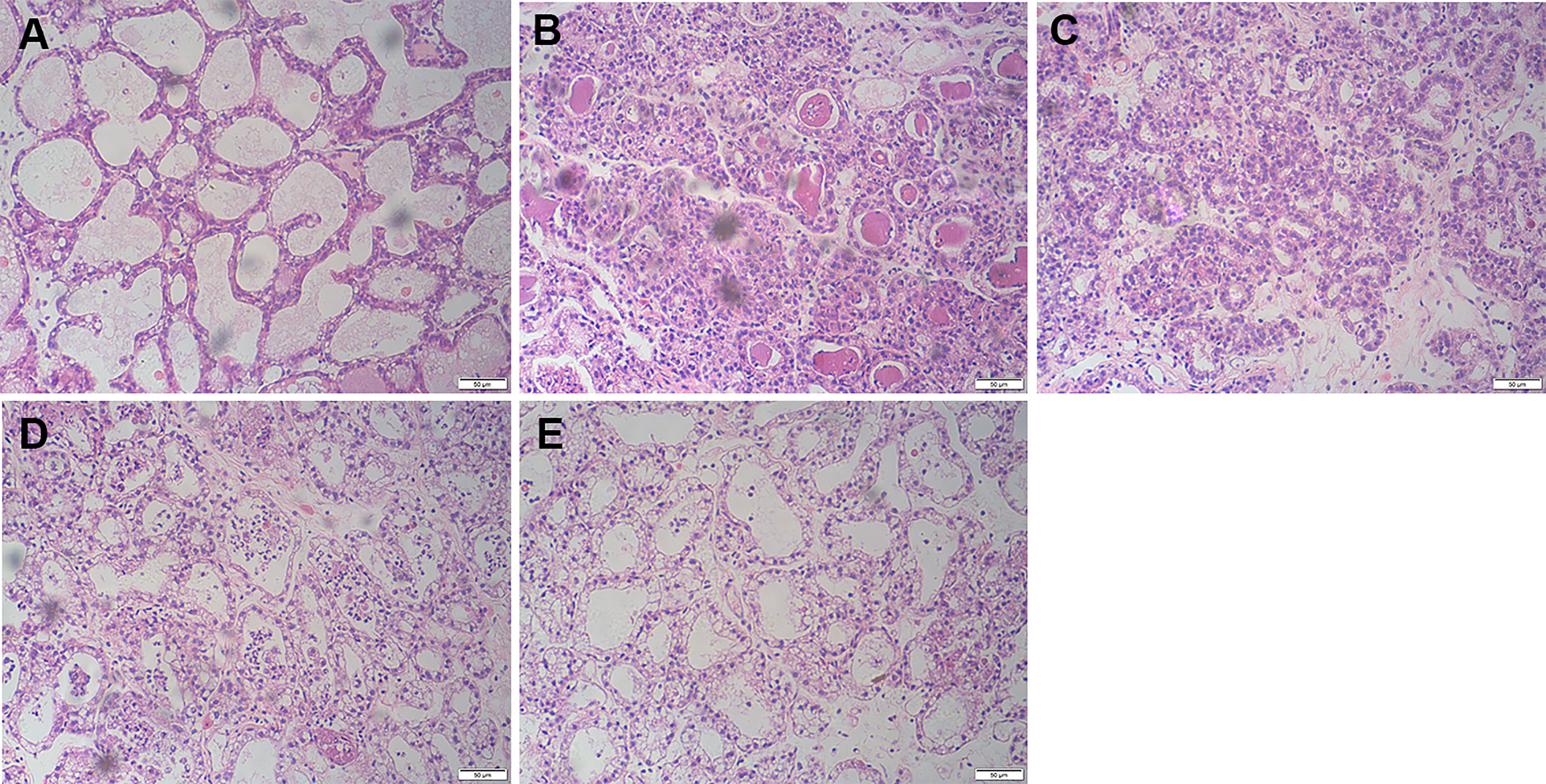
Figure 1 Effects of FOR on LPS-induced histopathological changes in mice with mammary (magnification 200×). (A) Control group: normal mammary gland tissue structure. (B) LPS group: severe inflammatory cell infiltration. (C–E) LPS + FOR (10, 20, 30 mg/kg) groups: gradually decreasing inflammatory cell infiltration after administration of FOR.
The MPO activity increased significantly in the LPS group in comparison to the controls. Conversely, the MPO activity in the LPS+FOR group was much lower when compared to the LPS group (Figure 2). This result suggested FOR could inhibit the infiltration of neutrophils into lung tissues.
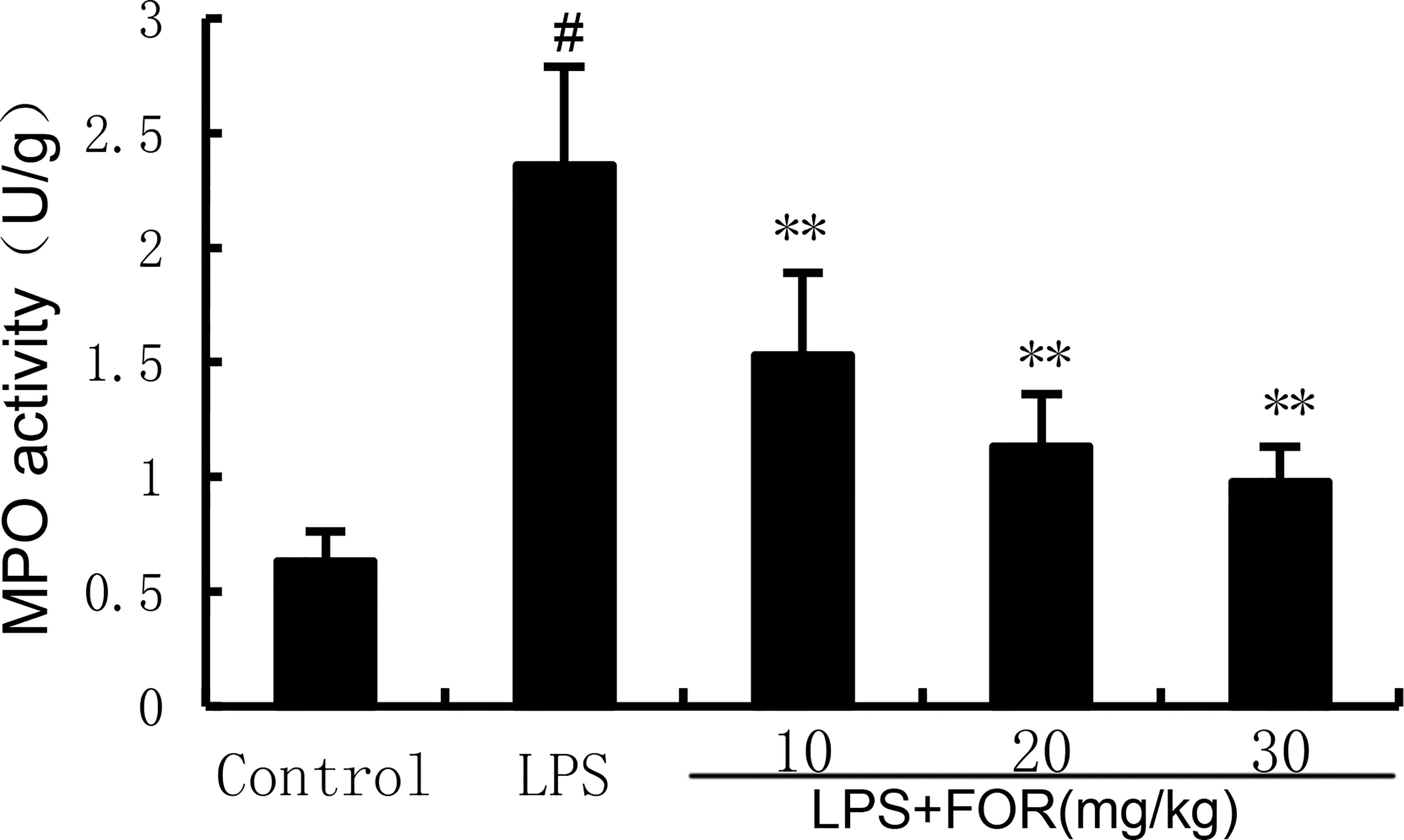
Figure 2 FOR suppresses LPS-induced MPO activity. The results are expressed as the mean ± SEM of triple parallel measurements. #(P < 0.01) is significantly different from control group; **(P < 0.01) is significantly different from LPS group.
TNF-α, and IL-1β levels in mammary tissues were significantly elevated as compared to the control group in the LPS group. In contrast, TNF-α and IL-1β levels of mammary tissues were much lower in the LPS+FOR group comparing to the LPS group (Figure 3). In vitro, treatment of FOR (10, 20, and 30 μM) also inhibited LPS-induced TNF-α and IL-1β production in EpH4-Ev cells (Figure 4). These results suggested FOR could inhibit LPS-induced inflammatory response.
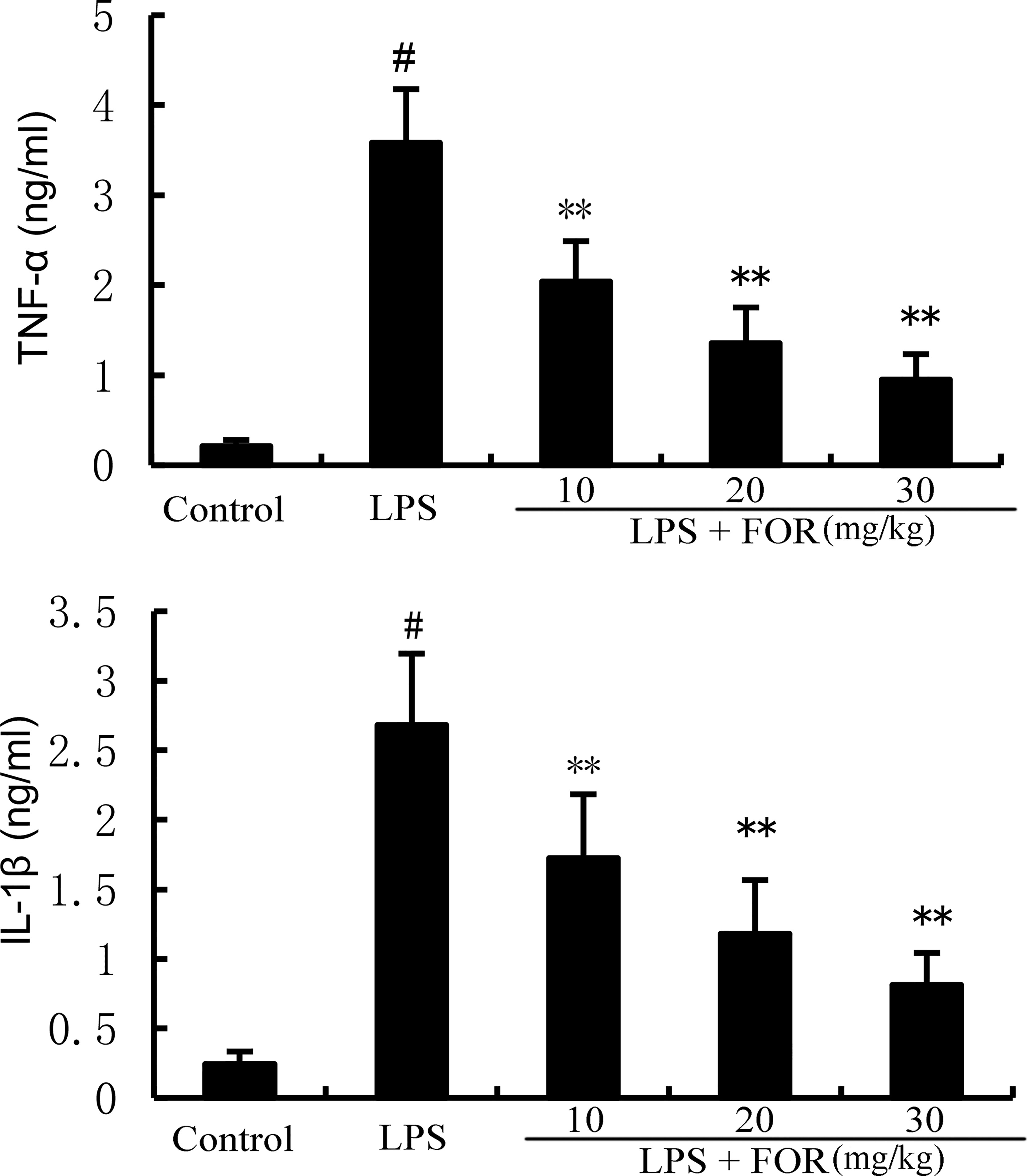
Figure 3 FOR decreases LPS-induced generation of TNF-α and IL-1β in breast tissue. The data herein are expressed as mean ± SEM obtained from triple parallel measurements. #(P < 0.01) is significantly different from control group; **(P < 0.01) is significantly different from LPS group.
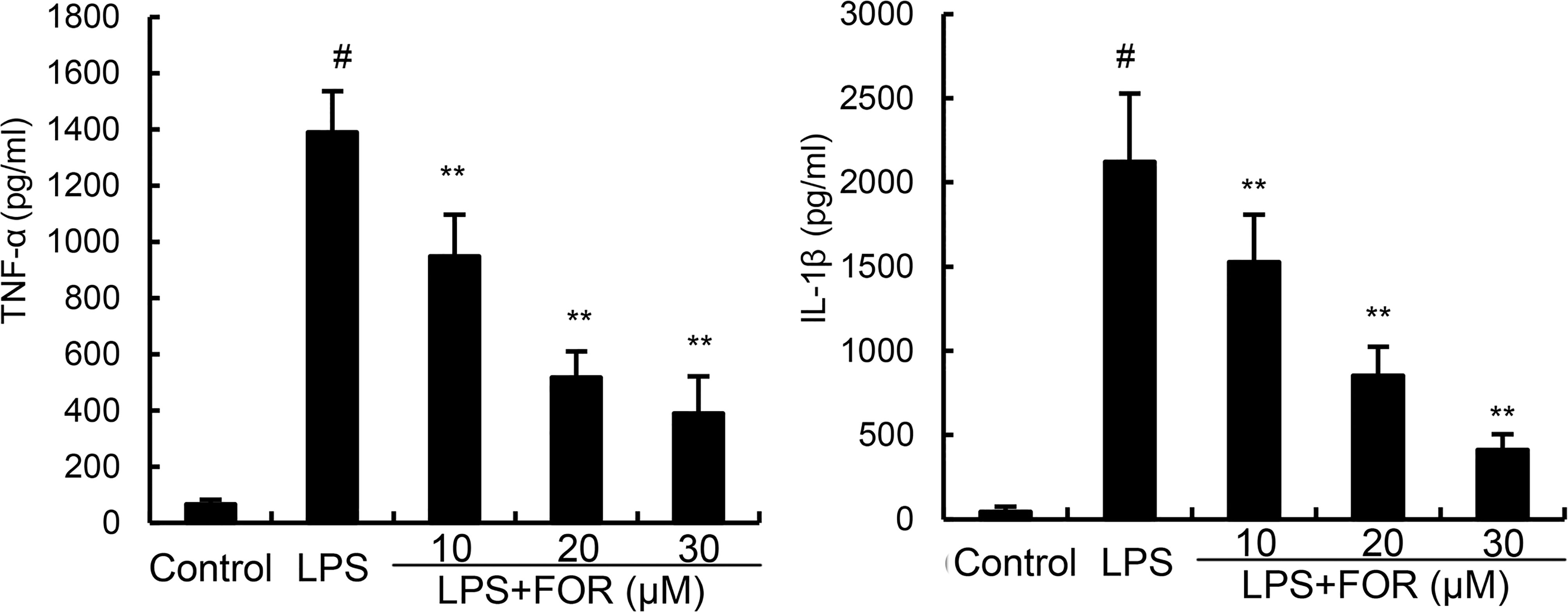
Figure 4 FOR reduces LPS-induced secretion of TNF-α and IL-1β in EpH4-Ev cells. The results herein are expressed as mean ± SEM of triple parallel measurements. #(P < 0.01) is significantly different from control group; **(P < 0.01) is significantly different from LPS group.
The effects of FOR on blood-milk barrier integrity were assessed by detecting the expression of tight junction protein claudin-3, occludin, and ZO-1. The results showed that claudin-3, occludin, and ZO-1 expression decreased significantly in the LPS group in comparison with controls. In contrast, claudin-3, occludin, and ZO-1 expression were increased by FOR compared to LPS group (Figure 5).
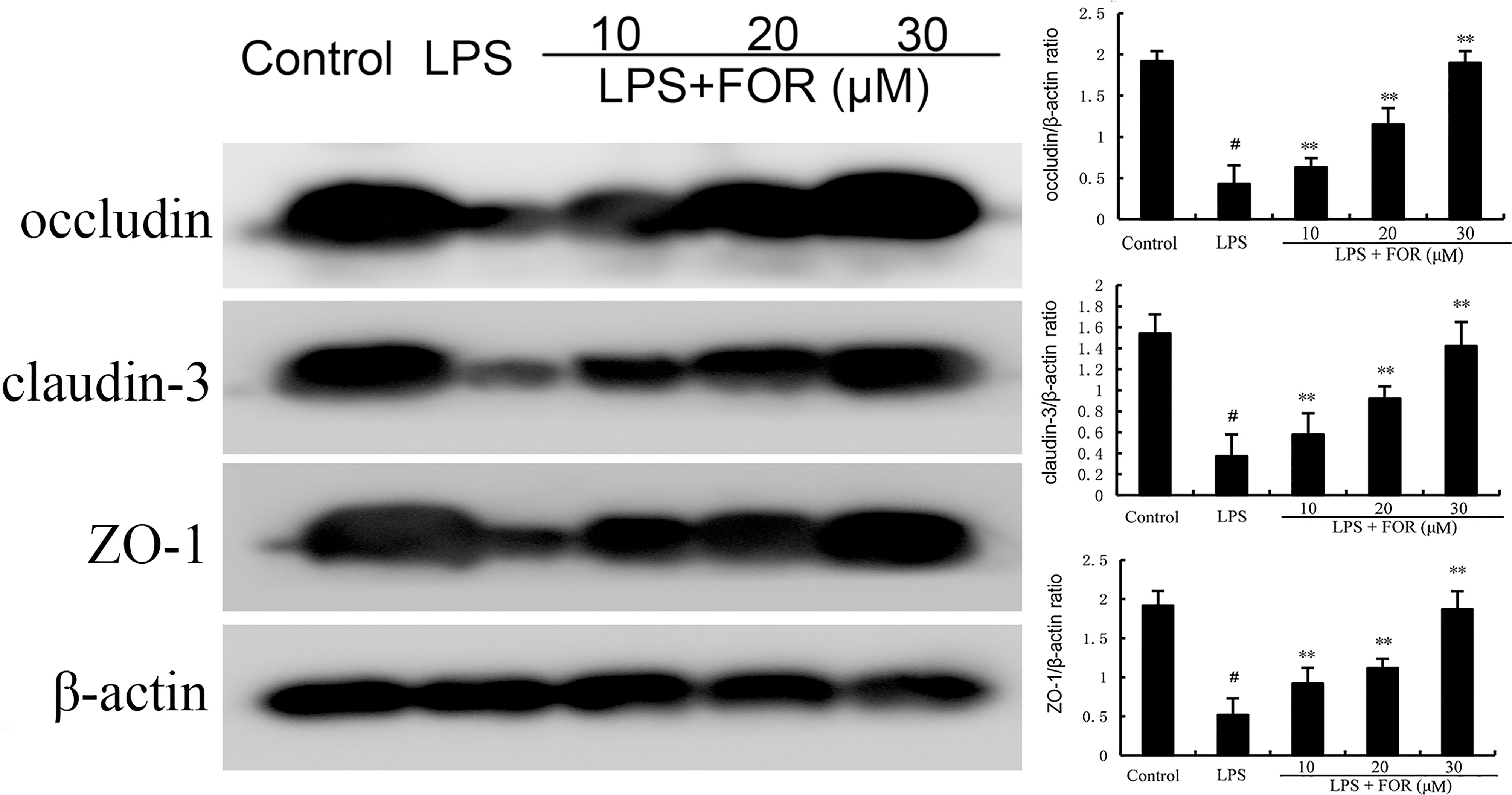
Figure 5 FOR promotes the expression of tight junction-related proteins. The present results are displayed as the mean ± SEM of triple parallel measurements. #(P < 0.01) is significantly different from control group; **(P < 0.01) is significantly different from LPS group.
The localization of FITC-albumin was observed by immunofluorescence detection. As shown in Figure 6, green and blue show FITC-albumin and nuclei (DAPI), respectively. The infiltration of large amounts of FITC-albumin into the lumen of the glandular vesicles indicated that LPS was capable of causing blood-milk barrier disruption (Figure 6B). However, FOR significantly inhibited LPS-induced increase of blood-milk barrier permeability (Figures 6C–E).
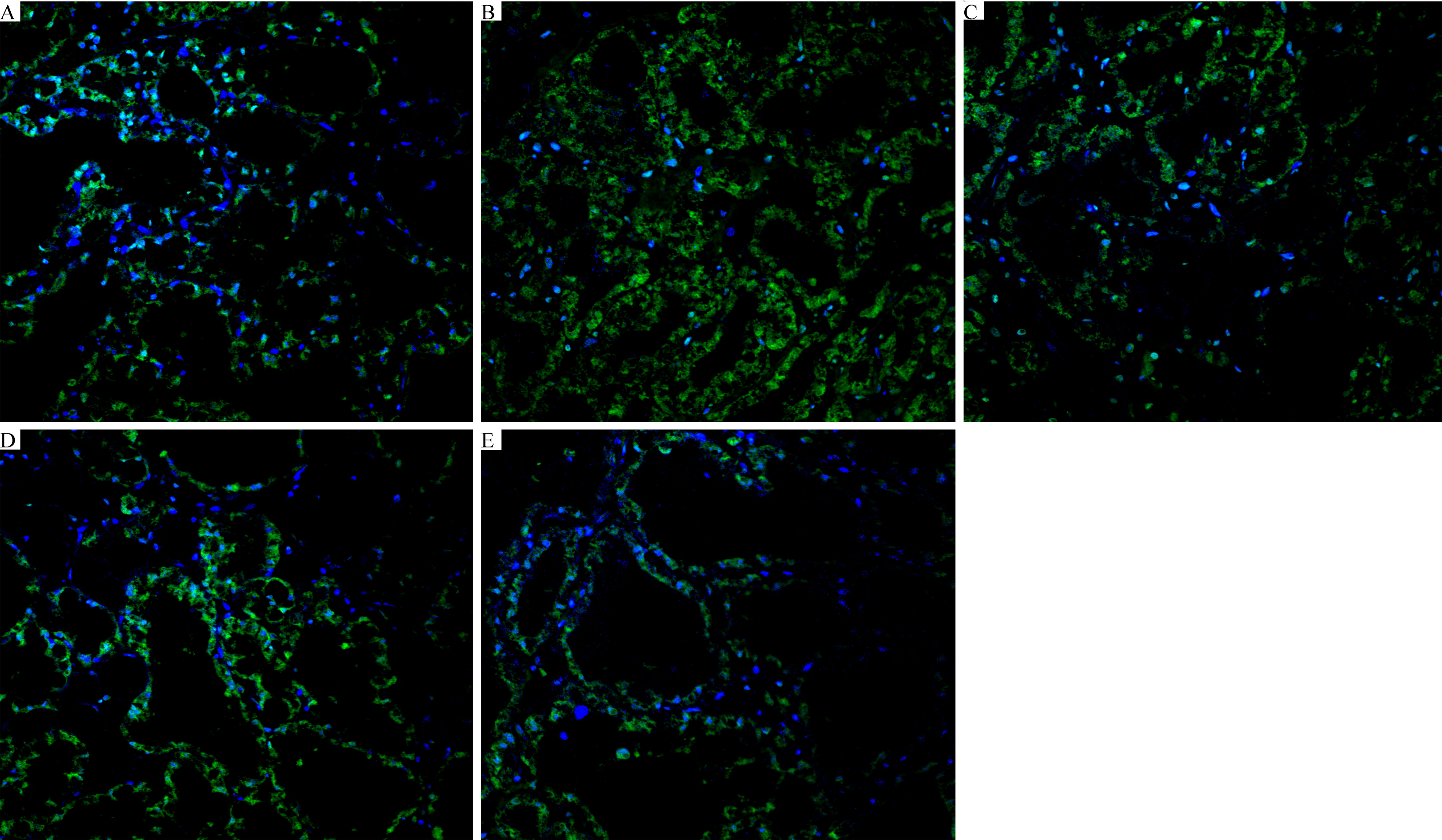
Figure 6 FOR suppresses the LPS-induced blood-milk barrier disruption (magnification 400×). (A) Control group: normal mammary gland tissue structure. (B) LPS group: severe FITC-albumin infiltration and exfoliation of epithelial cells. (C–E) LPS + FOR (10, 20, 30 mg/kg) groups: gradually intact blood-milk barrier after administration of FOR. Blue spots represented the nuclei; green spots were albumin.
NF-κB is a crucial transcription factor that regulates the production of inflammatory mediators. As the results shown in Figure 7, the phosphorylation levels of NF-κB and IκBα increased significantly in the LPS group in comparison to the controls. Conversely, the phosphorylation levels of NF-κB and IκBα in the LPS+FOR groups were much lower when compared to the LPS group (Figure 7). These findings suggested FOR suppressed LPS-induced inflammatory response via inhibition of the NF-κB signal pathway.
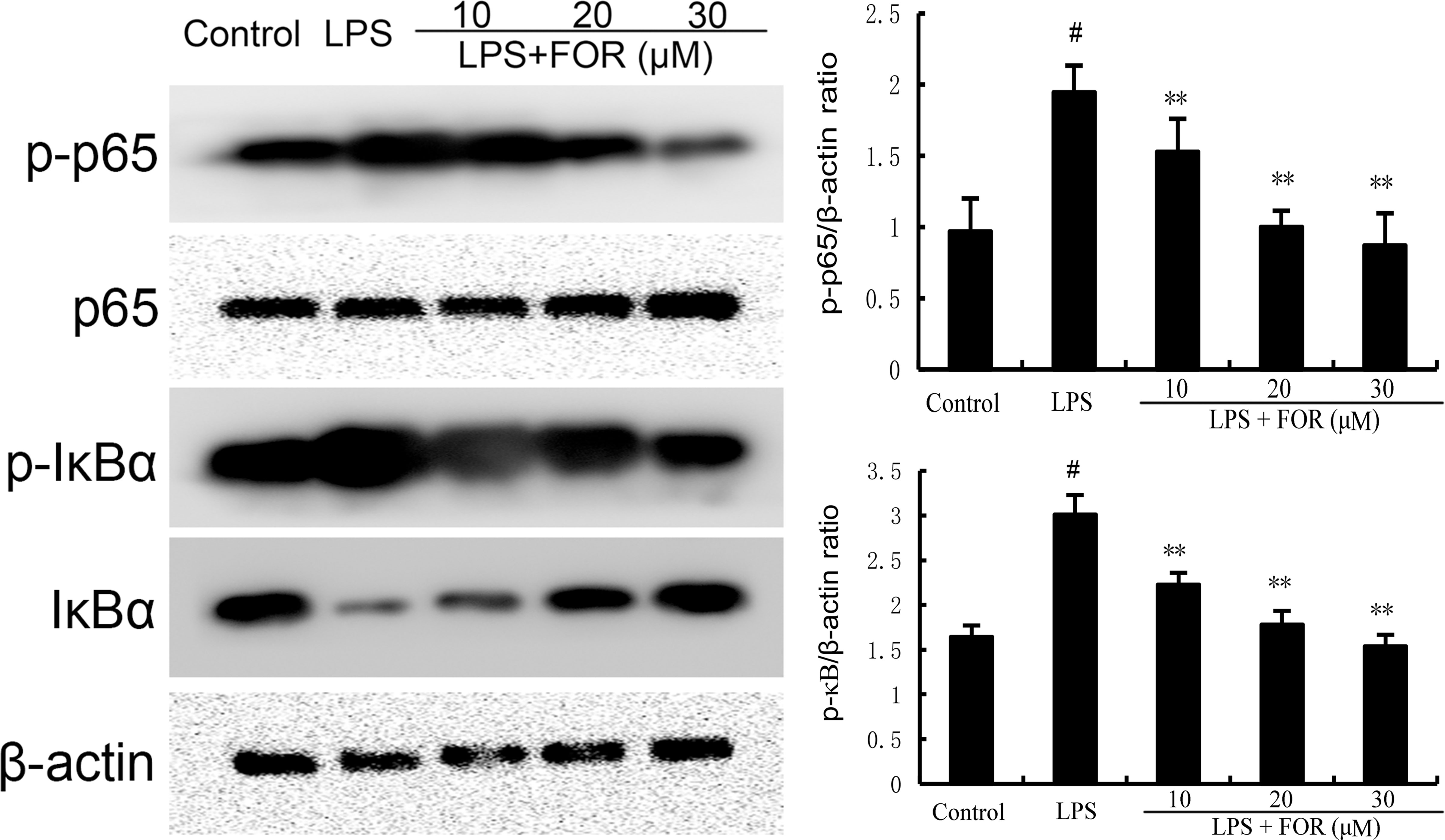
Figure 7 FOR inhibits LPS-induced NF-κB activation. The present results are displayed as the mean ± SEM of triple parallel measurements. #(P < 0.01) is significantly different from control group; **(P < 0.01) is significantly different from LPS group.
AhR/Src signaling pathway participated in the regulation of inflammation. As shown in Figure 8, FOR increased the expression of AhR in a concentration-dependent manner. Meanwhile, LPS increased the phosphorylation level of Src and the increase was inhibited by the treatment of FOR (Figure 8).
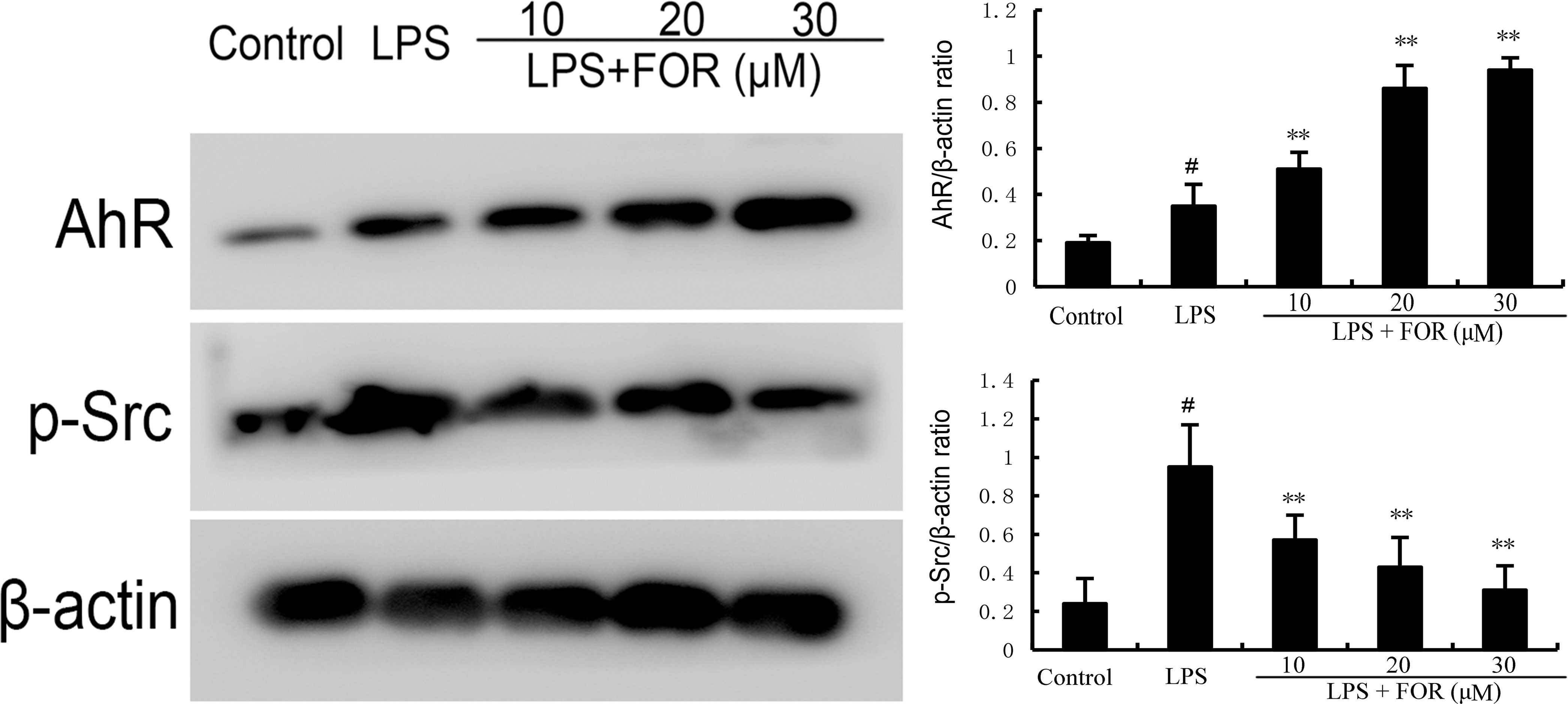
Figure 8 FOR increases AhR expression and inhibits the phosphorylation level of Src. The present results are displayed as the mean ± SEM of triple parallel measurements. #(P < 0.01) is significantly different from control group; **(P < 0.01) is significantly different from LPS group.
LPS was used to stimulate EpH4-Evs cells, and cell viability was measured by MTT assay. When there was a significant decrease in cell viability and a significant increase in inflammatory factors, it suggested that LPS had a significant inflammatory effect on EpH4-Evs cells. The optimal concentration and time of LPS stimulation on EpH4-Evs cells were determined by pre-experiments. To further clarify the anti-inflammatory mechanism of FOR, AhR antagonist CH223191 was used in this study. The results showed that the inhibitory effects of FOR to NF-κB activation (Figure 9B) and inflammatory cytokine generation (Figure 9A) were reversed by AhR antagonist CH223191. Furthermore, the enhanced blood-milk barrier integrity induced by FOR was also inhibited by AhR antagonist CH223191 (Figure 9C). These results indicated FOR inhibited LPS-induced inflammation and enhanced blood-milk barrier integrity via AhR-induced Src inactivation.
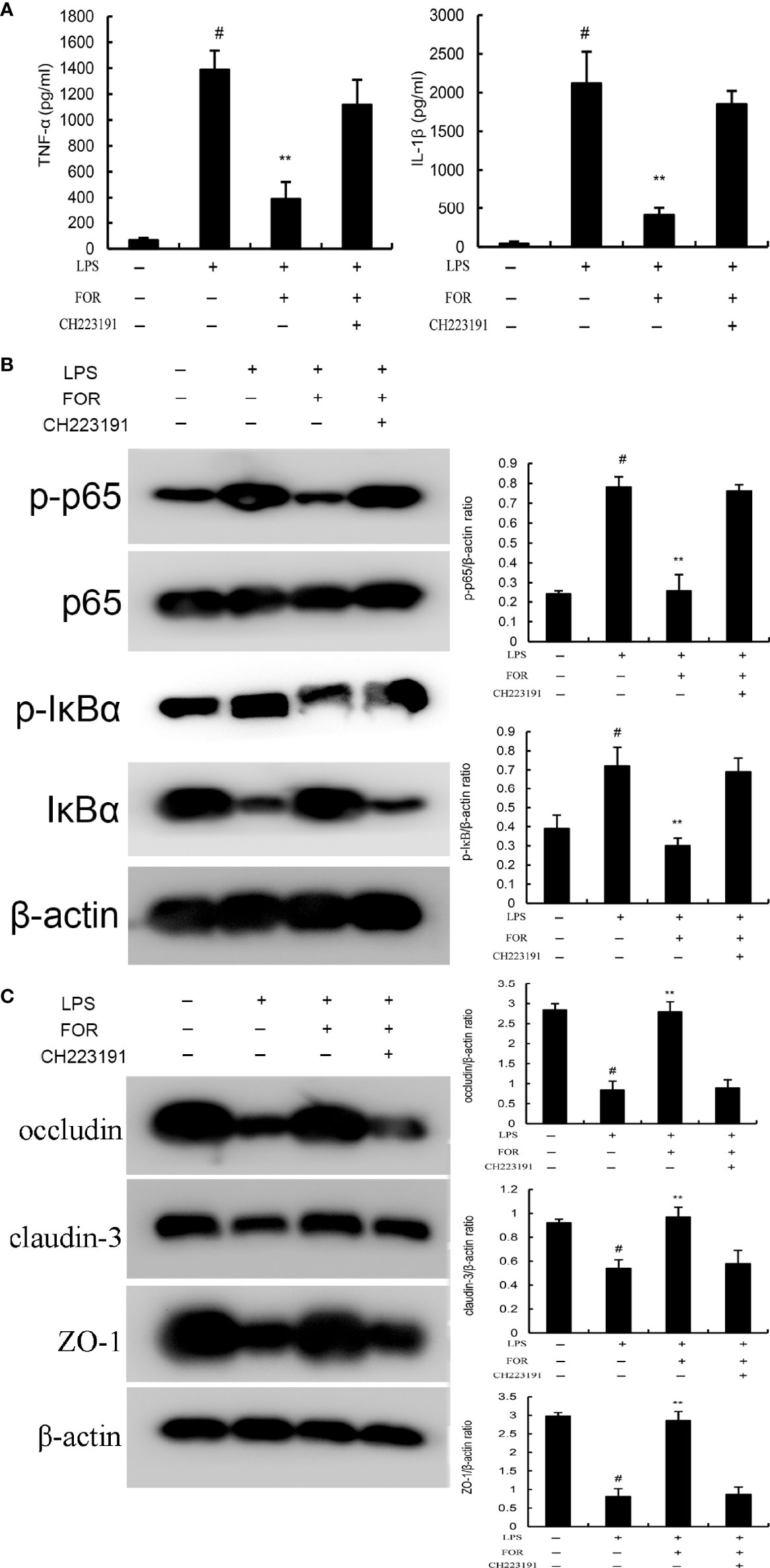
Figure 9 FOR inhibits LPS-induced inflammation via AhR-induced Src inactivation. (A) The levels of proinflammatory cytokines TNF-α and IL-1β in EpH4-Evs cells. (B) The expression of NF-κB signaling pathway. (C) The expression of tight junction proteins occludin, claudin-3, and ZO-1 in EpH4-Evs cells. The present results are displayed as the mean ± SEM of triple parallel measurements. #(P < 0.01) is significantly different from control group; **(P < 0.01) is significantly different from LPS group.
In this study, we evaluated the protection of FOR against LPS-induced mastitis. The principal findings are as follows FOR significantly inhibited infiltration of neutrophils, inflammatory cytokine production, and MPO activity in LPS-induced mastitis in mice. Its mechanism was by suppressing inflammation and enhancing blood-milk barrier integrity.
Inflammation was involved in the development of mastitis (17). The invading bacteria could activate the innate immune system and lead to the production of inflammatory and oxidative mediators (18). These increased inflammatory mediators such as TNF-α, IL-1ß and IL-6 contributed to the pathogenesis of mastitis (19). They could cause damage to breast tissue and cells (20). In the present study, it was demonstrated that FOR remarkably suppressed the generation of LPS-induced inflammatory cytokine. In combined with the mammary histopathological results, the results suggested FOR suppressed LPS-induced mastitis through attenuating inflammatory response.
The integrity of blood-milk barrier is the premise to ensure the normal lactation function of mammary tissue (21). It has been known that tight junction is the key structure to maintain the integrity of blood-milk barrier (22). It was found that the integrity of blood-milk barrier was destroyed when mastitis occurred (23). Blood-milk barrier dysfunction could lead to excessive neutrophils entering the mammary tissue and causing damage (24). Therefore, maintaining the integrity of blood-milk barrier is of great significance to treat mastitis. In this study, our results showed that FOR could increase the expression of tight junction proteins in the mammary epithelium and enhance the integrity of the blood-milk barrier.
NF-κB is an important molecule that regulates the transcription of inflammatory cytokines (25). It was involved in the regulation of inflammatory cytokine production and employed as a target for the treatment of many inflammatory diseases (26). In recent years, many natural herbal medicines have been found to protect mice against mastitis through inhibiting NF-κB activation (27, 28). And we found that FOR was able to significantly inhibit LPS-induced NF-κB activation. AhR is a ligand activated transcription factor (29). Previous research has demonstrated the role of AhR in the metabolism of poisons and chemicals in the environment, regulating biological rhythm, reproduction and oxidative stress (30). Recently, it has been found that AhR has important functions in innate and adaptive immune response, autoimmune diseases, infection and inflammation (31, 32). AHR may function as a pattern recognition receptor in host resistance to pathogenic microorganisms (32). LPS can activate AHR, which decreases the expression of pro-inflammatory cytokines and regulates long-term systemic inflammation (33). In addition, AHR is a component of a retinoic acid-induced signalsome involved in neutrophilic granulocyte differentiation (34). AHR and NF-κB signaling pathways interact with each other. LPS-induced activation of NF-κB in the mouse thymus involves RelA-dependent AhR expression and enhanced activity of AhR-regulated genes (35). Src is a nonreceptor protein tyrosine kinase that is capable of forming a complex with AhR (36). AhR deficiency results in enhanced phosphorylation of Src (37). Recent studies demonstrated that activation of AhR could lead to the inhibition of Src, thus increasing the expression of anti-inflammatory cytokines and enhancing intestinal barrier function to alleviate inflammation (36, 38). In the present work, it was shown that LPS activated AhR and Src expression and correlated closely with NF-κB signaling pathway activation. However, FOR could concentration-dependently increase the expression of AhR and inhibit Src phosphorylation to exert anti-inflammatory effects. Furthermore, AhR antagonist reversed the inhibitory effects of FOR on NF-κB activation and inflammatory factor production. The enhanced blood-milk barrier integrity induced by FOR was also inhibited by AhR antagonist.
In conclusion, the results indicated the protective effect of FOR on LPS-induced mastitis through suppressing inflammation and enhancing blood-milk barrier integrity. FOR can be a potential therapeutic agent for mastitis.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding authors.
The animal study was reviewed and approved by Institutional Animal Care and Use Committee of Jilin University.
YF and BL designed the experiment. KX, PS, ZG, and ZL did the experiment. XH and PS revised the manuscript. YF analyzed the data. KX and BL wrote the paper. All authors contributed to the article and approved the submitted version.
The work was supported by the National Natural Science Foundation of China (Nos. 32122087).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Hu X, Guo J, Zhao C, Jiang P, Maimai T, Yanyi L, et al. The Gut Microbiota Contributes to the Development of Staphylococcus Aureus-Induced Mastitis in Mice. Isme J (2020) 14:1897–910. doi: 10.1038/s41396-020-0651-1
2. Klaas IC, Zadoks RN. An Update on Environmental Mastitis: Challenging Perceptions. Transbound Emerg Dis (2018) 65(Suppl 1):166–85. doi: 10.1111/tbed.12704
3. Wellnitz O, Bruckmaier RM. The Innate Immune Response of the Bovine Mammary Gland to Bacterial Infection. Vet J (London Engl 1997) (2012) 192:148–52. doi: 10.1016/j.tvjl.2011.09.013
4. Purba FY, Ueda J, Nii T, Yoshimura Y, Isobe N. Effects of Intrauterine Infusion of Bacterial Lipopolysaccharides on the Mammary Gland Inflammatory Response in Goats. Vet Immunol Immunopathol (2020) 219:109972. doi: 10.1016/j.vetimm.2019.109972
5. Guo W, Liu B, Hu G, Kan X, Li Y, Gong Q, et al. Vanillin Protects the Blood-Milk Barrier and Inhibits the Inflammatory Response in LPS-Induced Mastitis in Mice. Toxicol Appl Pharmacol (2019) 365:9–18. doi: 10.1016/j.taap.2018.12.022
6. Turk R, Piras C, Kovačić M, Samardžija M, Ahmed H, De Canio M, et al. Proteomics of Inflammatory and Oxidative Stress Response in Cows With Subclinical and Clinical Mastitis. J Proteomics (2012) 75:4412–28. doi: 10.1016/j.jprot.2012.05.021
7. Turk R. The Role of Oxidative Stress and Inflammatory Response in the Pathogenesis of Mastitis in Dairy Cows. Mljekarstvo (2017) 67:91–101. doi: 10.15567/mljekarstvo.2017.0201
8. Mushtaq S, Shah AM, Shah A, Lone SA, Hussain A, Hassan QP, et al. Bovine Mastitis: An Appraisal of Its Alternative Herbal Cure. Microb Pathog (2018) 114:357–61. doi: 10.1016/j.micpath.2017.12.024
9. Cao R, Fu K, Lv X, Li W, Zhang N. Protective Effects of Kaempferol on Lipopolysaccharide-Induced Mastitis in Mice. Inflammation (2014) 37:1453–8. doi: 10.1007/s10753-014-9870-9
10. Ghasemian M, Owlia S, Owlia MB. Review of Anti-Inflammatory Herbal Medicines. Adv Pharmacol Sci (2016) 2016:9130979. doi: 10.1155/2016/9130979
11. Sugimoto M, Ko R, Goshima H, Koike A, Shibano M, Fujimori K. Formononetin Attenuates H(2)O(2)-Induced Cell Death Through Decreasing ROS Level by PI3K/Akt-Nrf2-Activated Antioxidant Gene Expression and Suppressing MAPK-Regulated Apoptosis in Neuronal SH-SY5Y Cells. Neurotoxicology (2021) 85:186–200. doi: 10.1016/j.neuro.2021.05.014
12. Wang DS, Yan LY, Yang DZ, Lyu Y, Fang LH, Wang SB, et al. Formononetin Ameliorates Myocardial Ischemia/Reperfusion Injury in Rats by Suppressing the ROS-TXNIP-NLRP3 Pathway. Biochem Biophys Res Commun (2020) 525:759–66. doi: 10.1016/j.bbrc.2020.02.147
13. Ma Z, Ji W, Fu Q, Ma S. Formononetin Inhibited the Inflammation of LPS-Induced Acute Lung Injury in Mice Associated With Induction of PPAR Gamma Expression. Inflammation (2013) 36:1560–6. doi: 10.1007/s10753-013-9700-5
14. Liu G, Zhao W, Bai J, Cui J, Liang H, Lu B. Formononetin Protects Against Concanavalin-A-Induced Autoimmune Hepatitis in Mice Through Its Anti-Apoptotic and Anti-Inflammatory Properties. Biochem Cell Biol (2021) 99:231–40. doi: 10.1139/bcb-2020-0197
15. Hwang JS, Kang ES, Han SG, Lim DS, Paek KS, Lee CH, et al. Formononetin Inhibits Lipopolysaccharide-Induced Release of High Mobility Group Box 1 by Upregulating SIRT1 in a Pparδ-Dependent Manner. PeerJ (2018) 6:e4208. doi: 10.7717/peerj.4208.
16. Cho IA, Kim TH, Lim H, Park JH, Kang KR, Lee SY, et al. Formononetin Antagonizes the Interleukin-1β-Induced Catabolic Effects Through Suppressing Inflammation in Primary Rat Chondrocytes. Inflammation (2019) 42:1426–40. doi: 10.1007/s10753-019-01005-1
17. Zhao C, Hu X, Bao L, Wu K, Feng L, Qiu M, et al. Aryl Hydrocarbon Receptor Activation by Lactobacillus Reuteri Tryptophan Metabolism Alleviates Escherichia Coli-Induced Mastitis in Mice. PLoS Pathog (2021) 17:e1009774. doi: 10.1371/journal.ppat.1009774
18. Wan Z, Lan R, Zhou Y, Xu Y, Wang Z, Luo Z, et al. Taurine-Mediated IDOL Contributes to Resolution of Streptococcus Uberis Infection. Infect Immun (2021) 89:e00788–20. doi: 10.1128/IAI.00788-20
19. Chen H, Mo X, Yu J, Huang Z. Alpinetin Attenuates Inflammatory Responses by Interfering Toll-Like Receptor 4/Nuclear Factor Kappa B Signaling Pathway in Lipopolysaccharide-Induced Mastitis in Mice. Int Immunopharmacol (2013) 17:26–32. doi: 10.1016/j.intimp.2013.04.030
20. Ingman WV, Glynn DJ, Hutchinson MR. Inflammatory Mediators in Mastitis and Lactation Insufficiency. J Mammary Gland Biol Neoplasia (2014) 19:161–7. doi: 10.1007/s10911-014-9325-9
21. Hu X, Li S, Fu Y, Zhang N. Targeting Gut Microbiota as a Possible Therapy for Mastitis. Eur J Clin Microbiol Infect Dis (2019) 38:1409–23. doi: 10.1007/s10096-019-03549-4
22. Jiang A, Zhang Y, Zhang X, Wu D, Liu Z, Li S, et al. Morin Alleviates LPS-Induced Mastitis by Inhibiting the PI3K/AKT, MAPK, NF-κb and NLRP3 Signaling Pathway and Protecting the Integrity of Blood-Milk Barrier. Int Immunopharmacol (2020) 78:105972. doi: 10.1016/j.intimp.2019.105972
23. Wang JJ, Wei ZK, Zhang X, Wang YN, Fu YH, Yang ZT. Butyrate Protects Against Disruption of the Blood-Milk Barrier and Moderates Inflammatory Responses in a Model of Mastitis Induced by Lipopolysaccharide. Br J Pharmacol (2017) 174:3811–22. doi: 10.1111/bph.13976
24. Wellnitz O, Bruckmaier RM. Invited Review: The Role of the Blood-Milk Barrier and Its Manipulation for the Efficacy of the Mammary Immune Response and Milk Production. J Dairy Sci (2021) 104:6376–88. doi: 10.3168/jds.2020-20029
25. Jimi E, Fei H, Nakatomi C. NF-κb Signaling Regulates Physiological and Pathological Chondrogenesis. Int J Mol Sci (2019) 20:6275. doi: 10.3390/ijms20246275
26. Lawrence T, Gilroy DW, Colville-Nash PR, Willoughby DA. Possible New Role for NF-kappaB in the Resolution of Inflammation. Nat Med (2001) 7:1291–7. doi: 10.1038/nm1201-1291
27. Wang J, Guo C, Wei Z, He X, Kou J, Zhou E, et al. Morin Suppresses Inflammatory Cytokine Expression by Downregulation of Nuclear Factor-κb and Mitogen-Activated Protein Kinase (MAPK) Signaling Pathways in Lipopolysaccharide-Stimulated Primary Bovine Mammary Epithelial Cells. J Dairy Sci (2016) 99:3016–22. doi: 10.3168/jds.2015-10330
28. Wang Y, Zhang X, Wei Z, Wang J, Zhang Y, Shi M, et al. Platycodin D Suppressed LPS-Induced Inflammatory Response by Activating Lxrα in LPS-Stimulated Primary Bovine Mammary Epithelial Cells. Eur J Pharmacol (2017) 814:138–43. doi: 10.1016/j.ejphar.2017.07.037
29. Gabriely G, Wheeler MA, Takenaka MC, Quintana FJ. Role of AHR and HIF-1α in Glioblastoma Metabolism. Trends Endocrinol Metab (2017) 28:428–36. doi: 10.1016/j.tem.2017.02.009
30. Ichihara S, Li P, Mise N, Suzuki Y, Izuoka K, Nakajima T, et al. Ablation of Aryl Hydrocarbon Receptor Promotes Angiotensin II-Induced Cardiac Fibrosis Through Enhanced C-Jun/HIF-1α Signaling. Arch Toxicol (2019) 93:1543–53. doi: 10.1007/s00204-019-02446-1
31. Beamer CA, Shepherd DM. Role of the Aryl Hydrocarbon Receptor (AhR) in Lung Inflammation. Semin Immunopathol (2013) 35:693–704. doi: 10.1007/s00281-013-0391-7
32. Bock KW. Aryl Hydrocarbon Receptor (AHR)-Mediated Inflammation and Resolution: Non-Genomic and Genomic Signaling. Biochem Pharmacol (2020) 182:114220. doi: 10.1016/j.bcp.2020.114220
33. Neavin DR, Liu D, Ray B, Weinshilboum RM. The Role of the Aryl Hydrocarbon Receptor (AHR) in Immune and Inflammatory Diseases. Int J Mol Sci (2018) 19:3851. doi: 10.3390/ijms19123851
34. Bunaciu RP, Jensen HA, MacDonald RJ, LaTocha DH, Varner JD, Yen A. 6-Formylindolo(3,2-B)Carbazole (FICZ) Modulates the Signalsome Responsible for RA-Induced Differentiation of HL-60 Myeloblastic Leukemia Cells. PLoS One (2015) 10:e0135668. doi: 10.1371/journal.pone.0135668
35. Vogel CF, Khan EM, Leung PS, Gershwin ME, Chang WL, Wu D, et al. Cross-Talk Between Aryl Hydrocarbon Receptor and the Inflammatory Response: A Role for Nuclear Factor-κb. J Biol Chem (2014) 289:1866–75. doi: 10.1074/jbc.M113.505578
36. Zhu J, Luo L, Tian L, Yin S, Ma X, Cheng S, et al. Aryl Hydrocarbon Receptor Promotes IL-10 Expression in Inflammatory Macrophages Through Src-STAT3 Signaling Pathway. Front Immunol (2018) 9:2033. doi: 10.3389/fimmu.2018.02033
37. Matoba H, Takamoto M, Fujii C, Kawakubo M, Kasuga E, Matsumura T, et al. Cecal Tumorigenesis in Aryl Hydrocarbon Receptor-Deficient Mice Depends on Cecum-Specific Mitogen-Activated Protein Kinase Pathway Activation and Inflammation. Am J Pathol (2020) 190:453–68. doi: 10.1016/j.ajpath.2019.10.005
Keywords: formononetin, LPS, mastitis, AhR, NF-κB
Citation: Xiang K, Shen P, Gao Z, Liu Z, Hu X, Liu B and Fu Y (2022) Formononetin Protects LPS-Induced Mastitis Through Suppressing Inflammation and Enhancing Blood-Milk Barrier Integrity via AhR-Induced Src Inactivation. Front. Immunol. 13:814319. doi: 10.3389/fimmu.2022.814319
Received: 13 November 2021; Accepted: 10 January 2022;
Published: 03 February 2022.
Edited by:
Yi Zhu, Baylor College of Medicine, United StatesReviewed by:
Bo Liu, Inner Mongolia Agricultural University, ChinaCopyright © 2022 Xiang, Shen, Gao, Liu, Hu, Liu and Fu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bin Liu, bGl1YmluMTE3OUAxNjMuY29t; Yunhe Fu, ZnV5dW5oZXNreUAxNjMuY29t
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.