
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Immunol. , 17 January 2023
Sec. Microbial Immunology
Volume 13 - 2022 | https://doi.org/10.3389/fimmu.2022.1085551
 Alex Kayongo1,2,3,4
Alex Kayongo1,2,3,4 Nicole M. Robertson5
Nicole M. Robertson5 Trishul Siddharthan6
Trishul Siddharthan6 Moses Levi Ntayi1,2,3
Moses Levi Ntayi1,2,3 Josephine Caren Ndawula1
Josephine Caren Ndawula1 Obondo J. Sande3
Obondo J. Sande3 Bernard S. Bagaya3
Bernard S. Bagaya3 Bruce Kirenga1
Bruce Kirenga1 Harriet Mayanja-Kizza2
Harriet Mayanja-Kizza2 Moses L. Joloba3
Moses L. Joloba3 Sofia K. Forslund7,8,9,10,11*
Sofia K. Forslund7,8,9,10,11*Chronic Obstructive Pulmonary Disease (COPD) has significantly contributed to global mortality, with three million deaths reported annually. This impact is expected to increase over the next 40 years, with approximately 5 million people predicted to succumb to COPD-related deaths annually. Immune mechanisms driving disease progression have not been fully elucidated. Airway microbiota have been implicated. However, it is still unclear how changes in the airway microbiome drive persistent immune activation and consequent lung damage. Mechanisms mediating microbiome-immune crosstalk in the airways remain unclear. In this review, we examine how dysbiosis mediates airway inflammation in COPD. We give a detailed account of how airway commensal bacteria interact with the mucosal innate and adaptive immune system to regulate immune responses in healthy or diseased airways. Immune-phenotyping airway microbiota could advance COPD immunotherapeutics and identify key open questions that future research must address to further such translation.
Chronic Obstructive Pulmonary Disease (COPD) has been characterized by persistent respiratory symptoms and airflow limitation due to distal airway abnormalities (1). This is usually caused by exposure to noxious particles or gases and is influenced by several host factors (1). Chronic airway inflammation drives small airway changes and destruction of lung parenchymal tissue (2–5). Individuals diagnosed with COPD have varying degrees of chronic bronchitis, distal airway disease, and parenchymal destruction (6). COPD is the third leading cause of death worldwide, with several epidemiologic studies reporting a global prevalence of approximately 11.7% (95% CI: 8.4-15%) (1). According to recent projections, the prevalence is expected to rise over the next 40 years, with approximately 5 million people succumbing to COPD-related death annually (1). The prevalence in Africa has been reported as overall similar to other regions (1, 7).
The mechanisms driving COPD progression have yet to become fully known. The airway microbiome has been implicated in several respiratory diseases, such as COPD, bronchiectasis, and asthma (8). However, it is still unclear how changes in airway microbiota drive persistent immune activation and consequent lung damage in COPD. Whereas N. subflava has been recently demonstrated to drive bronchiectasis, only indirect inference is presently possible for COPD (9, 10). Because of the existence of a well-documented overlap between COPD and bronchiectasis (11–15), it is plausible that N. subflava could contribute to COPD pathogenesis. This, however, needs further investigation. Several studies published elsewhere have also described the airway microbiome in COPD and health (16–30). Dysbiosis (defined as an unhealthy microbial compositional state) in the airways among COPD patients has been associated with disease progression and poor outcomes (23, 29, 31–37). Immune activation of the airways drives COPD progression (38–46). However, it is still unknown how dysbiosis fuels such persistent airway immune activation in COPD. In this review, we examine how dysbiosis may mediate COPD-associated airway inflammation. Although the mechanisms of airway microbiome-immune crosstalk have not been thoroughly investigated, we give a detailed account of how airway commensal bacteria interact with the mucosal innate and adaptive immune system to regulate immune responses in diseased airways. Furthermore, borrowing a page from gut microbiome-immune interactions published elsewhere (47–53), we suggest possible mechanisms worth investigating that could be contributing to COPD disease.
Upon microbial interaction with mucosal immune cells, metabolic and epigenetic changes occur (54–66), inducing immunologic tolerance aimed at minimizing damage potentially arising from responses against invading bacteria (67–69). Several researchers have highlighted the salience of microbiome-mediated immune regulation (50, 70–78). One of the earliest pieces of evidence for this immune regulation was the observation of gene reprogramming following colonization with a bacterial commensal in germ-free mice (79). Such changes are mediated via the activity of bacterial metabolites, discussed in detail in a separate section in this review. Remarkably, most of them (62, 80) attenuate pro-inflammatory responses via epigenetic changes in immune cells, inducing a switch from transcriptionally active to silent chromatin states (67). For instance, butyrate, a short-chain fatty acid, suppresses the activity of NF-kB, consequently inhibiting the production of pro-inflammatory cytokines (81, 82). Similarly, ethionine suppresses the activity of NF-kB and TNFα following stimulation with lipopolysaccharide (LPS) (83), while lactate promotes histone acetylation at the IL10 promoter, enhancing IL10 transcription in macrophages (84). The metabolite deoxycholate alters H3K4me3 and H3K27me3 in bone marrow granulocyte progenitor cells, leading to neutrophil proliferation (85). In germ-free and antibiotic-treated mice, dendritic cell activation and the consequent production of type 1 interferons is impaired, a response driven by reduced levels of H3K4me3 on transcriptional start sites of pro-inflammatory response factors irf-3 and -7 (86). Immune reprogramming has also been noted among adaptive immune cells. Treg cells are induced via the activity of HDAC at the locus of the Foxp3 transcription factor (87–89). In contrast, Th17 differentiation is inhibited (90–92). Comparative analysis of metabolites shows a differing degree of HDAC inhibition, epigenetics, and immune functional consequences (80).
Epigenetic modifications in germ-free mice have been characterized by methylation patterns on inflammatory genes such as those encoding Toll-like receptors, chemokines, and cytokines (93). In neonatal mice, the microbiome induces decreased methylation of the chemokine-encoding gene Cxcl16, which is critical in recruiting iNKT cells into the mucosa (94). Consequently, this change ameliorates inflammation in the gut and the airway mucosa (94). In another scenario, comparative epigenetic analysis of myeloid cells, derived from microbiome-colonized germ-free mice, shows a trimethylation pattern of histone H3 at lysine 4 of the loci of pro-inflammatory genes such as the genes encoding type 1 interferons, which as a result inhibits pro-inflammatory signals (95). Indeed, the role of epigenetics in mucosal immunity has been confirmed by a resultant loss in barrier integrity following the deletion of the histone deacetylase-3 gene from epithelial cells (96). In a nutshell, these findings support the role of microbiota in reprogramming mucosal immune cells, as illustrated in Figures 1, 2. Although the gut microbiome in these studies provides reprogramming signals, available studies further implicate airway microbiome initiating and mediating immune reprogramming locally at the mucosa. As supporting evidence, intranasal administration of a bacterial lysate abrogates experimental allergic asthma by targeting dendritic cells, epithelial cells, and type 2 ILCs (97). Whether similar findings occur in the setting of COPD remains to be investigated.
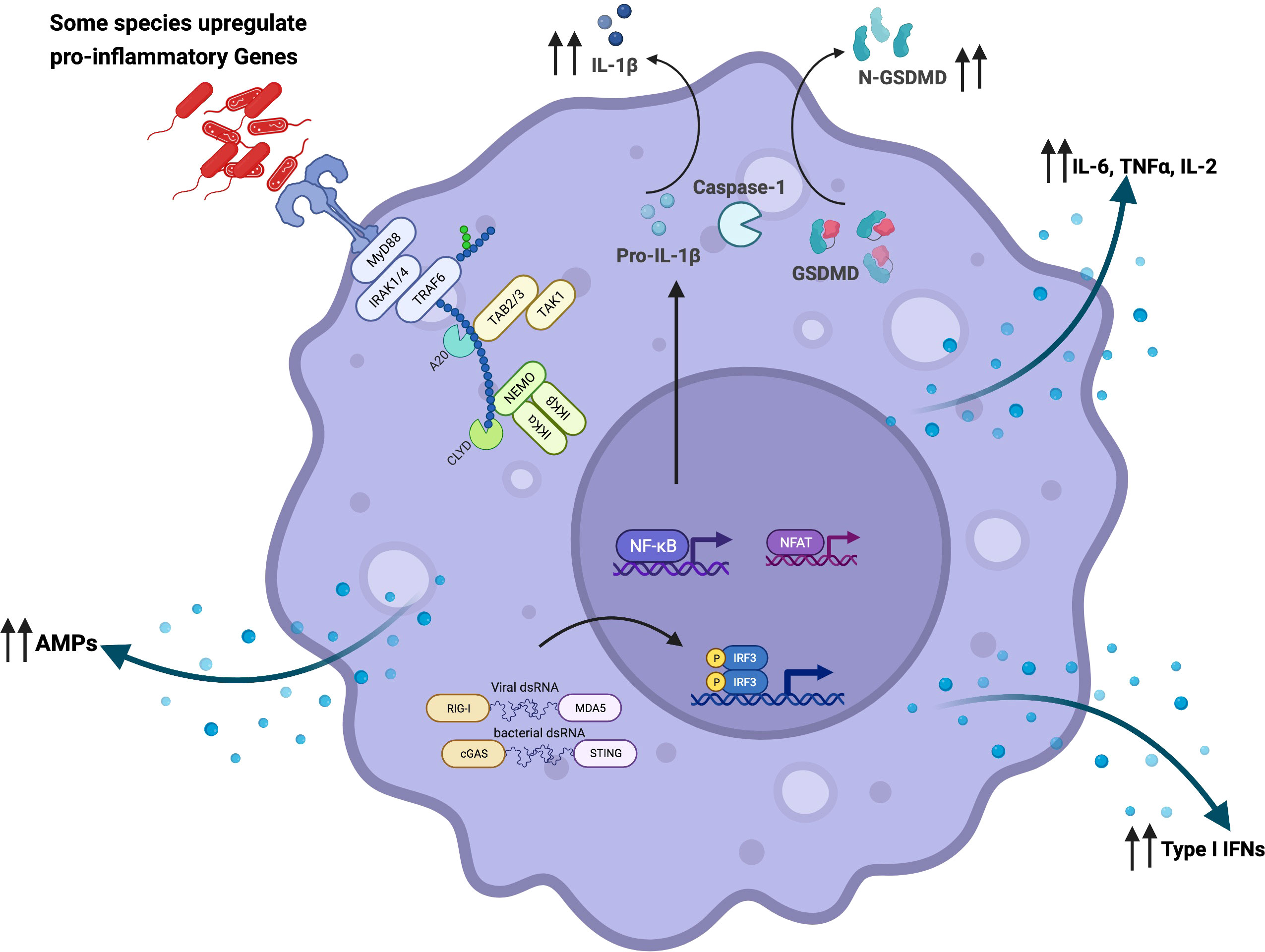
Figure 1 Microbial interaction with the innate immune system activates and reprograms immune cells: Airway microbiome-derived products such as lipopolysaccharide are detected by surface and intracellular immune sensors such as Toll-like receptors (TLRs). In a MyD88-dependent manner, airway epithelial cells, alveolar macrophages, and dendritic cells become activated and upregulate the expression of pro-inflammatory cytokines such as IL-1β, IL-18, TNFα, IL-6, and IL-2. These activated cells orchestrate immune inflammation. Microbicidal activity of alveolar macrophages is also potentiated through increased gene expression of antimicrobial peptides and other lytic proteins. The microbiome has also been shown to potentiate macrophage-bacterial killing and clearance via GM-CSF signaling. Furthermore, depletion of commensal bacteria has been shown to reprogram alveolar macrophages from classical (M1) to alternative (M2) phenotype characterized by the expression of higher levels of Arg1, CCL24, IL-13, IL-10, IL-6, and IL-1β. In addition to MyD88-dependent activation, intracellular sensing of microbial-derived products by NOD1, NOD2, and NLRP6 in the epithelial cells set the pace for a pro-inflammatory signal along the mucosa. This restricts mucosal colonization by pathogenic bacteria via the secretion of IL-1β and IL-18. Microbiome-driven-type-I-IFN responses dependent on cGAS-STING activation have also been described. Created with BioRender.com.
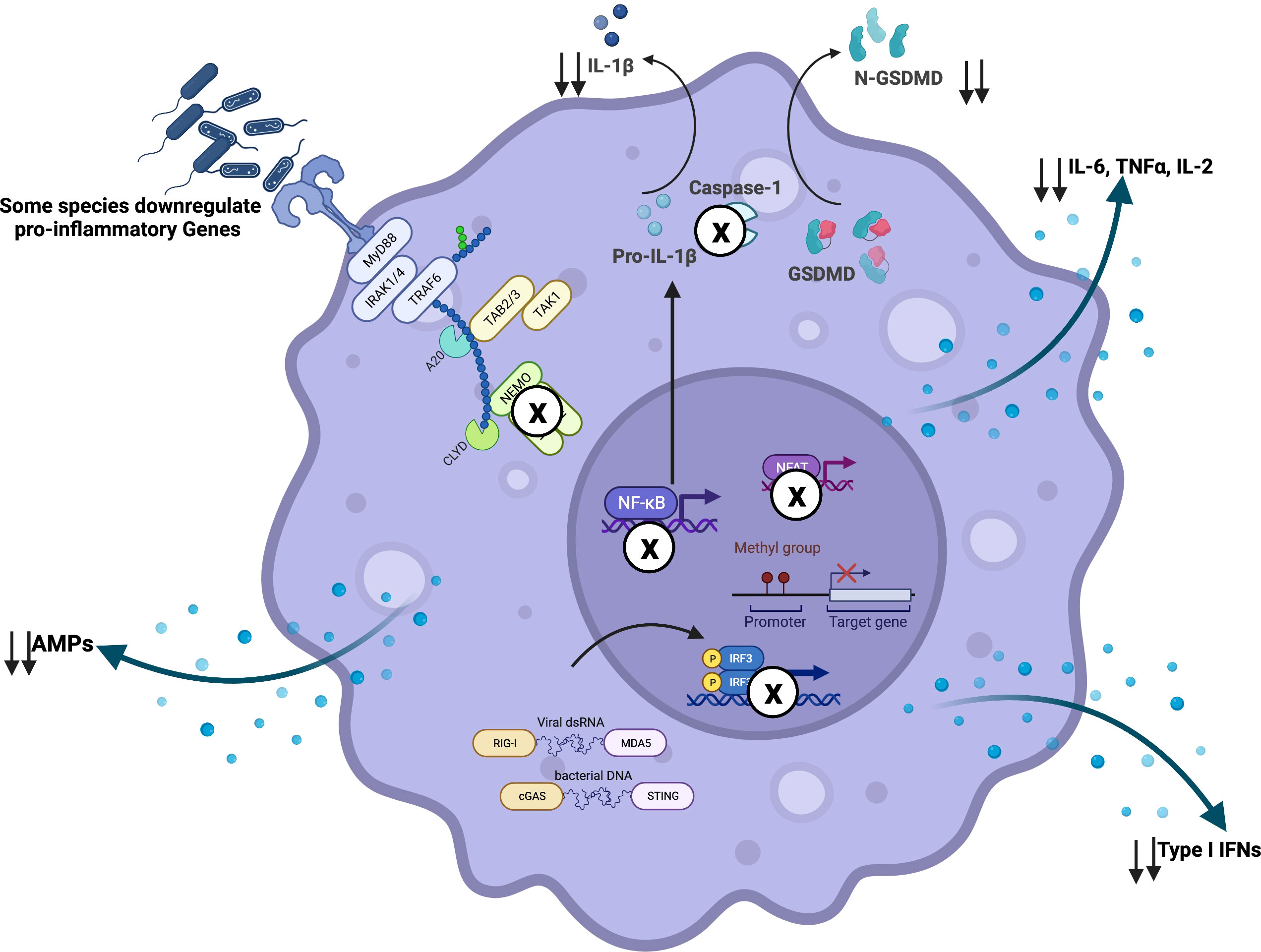
Figure 2 Airway microbiome-driven immunomodulation: Airway microbiome downregulates pro-inflammatory signals via interference with immune signaling machinery. For instance, R. mucilaginosa has been shown to inhibit immune activation via NF-κB dependent mechanisms. As an immune evasion strategy, pathobionts have been described to inhibit TRIF and NFAT signaling, phagolysosomal fusion and acidification, as well as inflammasome activation. In other scenarios, gene methylation may promote transcriptional repression of pro-inflammatory genes. Created with BioRender.com.
Mechanistically, microbiome-derived signals such as lipopolysaccharide (LPS) and bacterial DNA stimulate alveolar macrophages and dendritic cells via surface and intracellular sensors, producing several activating signals (chemokines and cytokines) (Figure 3). In the next section, we elaborate on such responses and describe how they activate other immune cellular players, i.e., innate lymphoid cells, γδ-T cells, neutrophils, monocytes, and lymphocytes (Figure 4). A well-coordinated immune response from this cellular network maintains a robust immune barrier at the respiratory mucosa, which preserves the microbial ecology (98, 99). In diseased airways, however, repeated insults such as respiratory infections, cigarette smoke, and particulate matter alter microbiome composition, reducing microbial diversity (100). Consequently, the ensuing dysbiosis triggers immune activation, potentially propagating inflammation and tissue damage observed in COPD (Figure 5) (101, 102). Several observations highlighting this concept have been published. They are summarized here as follows. Infection of distal airways during early childhood with pathogenic bacteria has been reported to cause severe lung damage associated with impaired lung growth and a consequent reduction in lung function (FEV1/FVC) in adult life (103–105). Although these studies do not establish specific bacterial species orchestrating this damage, several studies report organisms such as S. pneumoniae, H. influenzae, M. catarrhalis, S. aureus, and K. pneumoniae as culprits (106–110). Besides bacterial-induced early childhood-lung injuries and associated COPD in adult life, an interesting hypothesis termed the vicious circle was described, stating that once bacterial pathogens have successfully colonized the airways following impaired mucociliary clearance secondary to a primary insult, they persist, inducing chronic airway inflammation (111). Following the publication of early reports in 2010 describing the lung microbiome (112–115), authors revised the vicious circle to suggest that insults such as tobacco smoke exposure, which impair airway mucosal defenses mediate dysbiosis, leading to dysregulated immune response, further impairment of mucosal defense and ensuing dysbiosis, inducing inflammation and further dysbiosis (99). In distal airways, this inflammation contributes to tissue damage and progressive obstruction, as is the case for P. aeruginosa and N. subflava (9, 116, 117). These effects are mediated via the release of bacterial outer membrane vesicles harboring extracellular products, such as LPS, a potent inflammatory stimulus demonstrated by earlier studies to induce lung emphysema in hamsters (118). Several COPD human studies have supported this hypothesis, showing that bacterial colonization of the airways induces inflammation (111, 115–117, 119–121) and impairs lung function (FEV1) (122).

Figure 3 Airway microbiome immune crosstalk in alveoli: Following successful colonization of the respiratory tract, bacterial species replicate rapidly and establish dense biological communities (1). Some bacteria inhibit other species via the secretion of antimicrobial peptides and lytic enzymes(1). Alveolar macrophages engulf bacteria(2) and become activated via MyD88-dependent and -independent mechanisms(3). Airway microbiome sensing potentiates bacterial killing(3). Some intracellular pathobionts infect alveolar macrophages and access the lung interstitium (4). Activated macrophages secrete chemoattractants and promote the migration of other cellular players, such as monocytes, neutrophils, and adaptive immune response cells, into the airways to clear pathobionts (5). Dendritic cells (DCs) continuously sample airway bacteria that attach and colonize the mucosa via protruding dendrites(6). Airway microbiome sensing by bronchial epithelial cells, alveolar macrophages, and DCs activate innate lymphoid cells (ILCs) (7), which modulate other immune cells’ activity. Depending on the type and degree of microbial exposure, DCs induce a wide range of immune responses, from immune tolerance induced by plasmacytoid DCs (pDCs) to inflammation induced by conventional DCs (cDCs)(8). Continuous microbial sampling and trafficking by activated DCs, and alveolar macrophages deliver processed microbial antigens to naïve CD4+T and CD8+T cells within mucosa-associated lymphoid tissue (MALT) and draining lymph nodes (8). Created with BioRender.com.
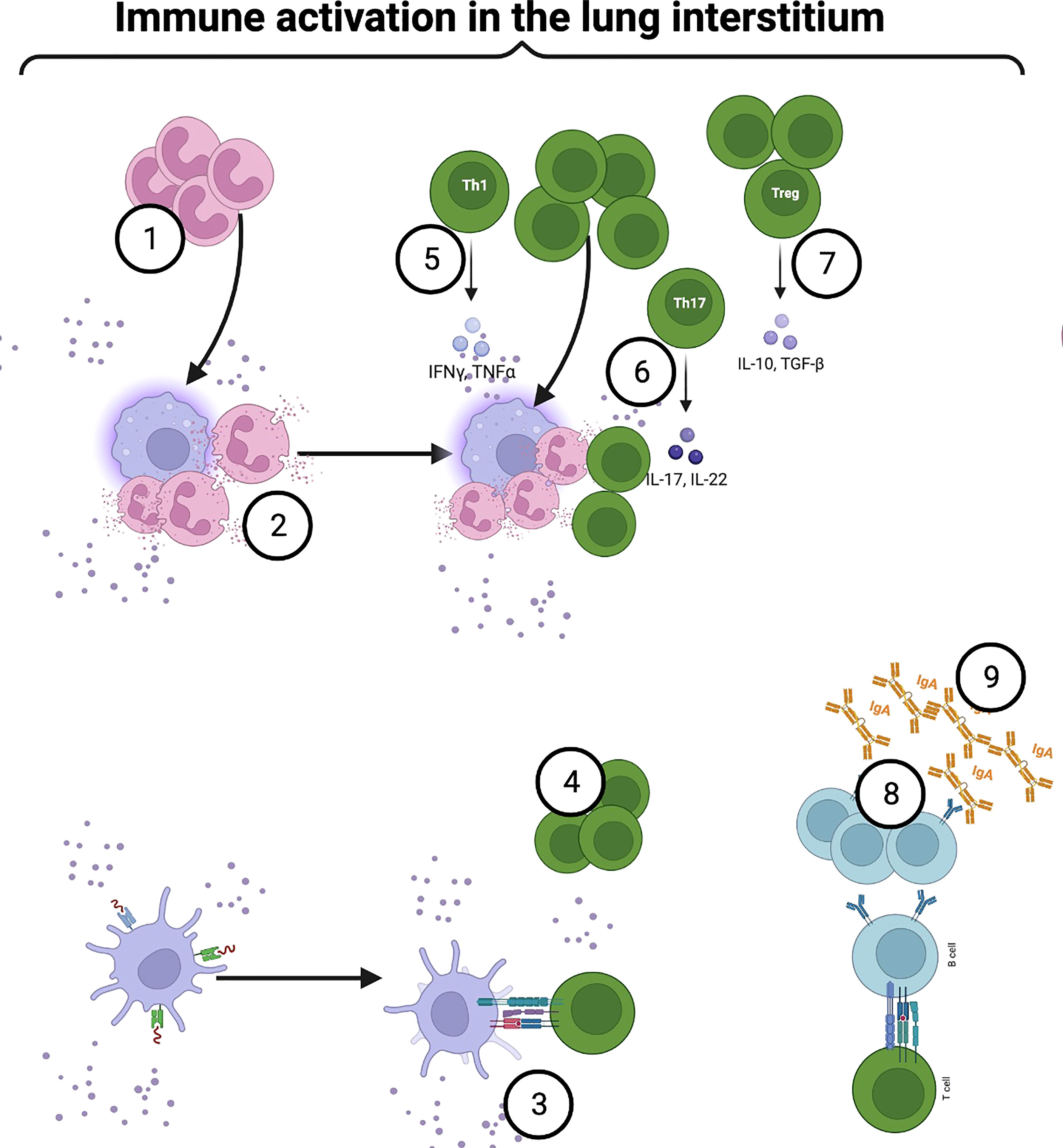
Figure 4 Airway microbiome immune crosstalk in the lung interstitium: Some intracellular pathobionts infect alveolar macrophages and access the interstitium. Activated macrophages secrete chemoattractants and promote neutrophilic infiltration into the airways to clear pathobionts (1). Upon bacterial sensing, neutrophils become activated, degranulate and release NETs (2). Continuous sampling and trafficking by activated DCs, and alveolar macrophages deliver processed microbial antigens to naïve CD4+T within mucosa-associated lymphoid tissue (MALT) and draining lymph nodes (4). Under a steady state, mature lung cDCs are preferentially programmed to induce a Th2 immune response. However, following immune sensing and activation, the production of IL-12, IL-23, IL-27, and notch ligand by airway DCs, alveolar macrophages, and epithelial cells induce a Th1 response (5). Among CD4+T cell phenotypes, microbiome-mediated mucosal inflammation has been strongly linked to aberrant Th17 (6). Besides the Th17 phenotype, microbial interaction with mucosal CD4+T cells induces immune tolerance (7). MyD88-dependent TLR2 activation by capsular polysaccharide A induces the expansion of Foxp3+T cells within the mucosa (7). Foxp3+T cells drive IL-10 production, facilitating mucosal immune tolerance (7). In another mechanism, microbial-induced Tregs promote mucosal memory B or plasma cells’ IgA secretion (8). Created with BioRender.com.
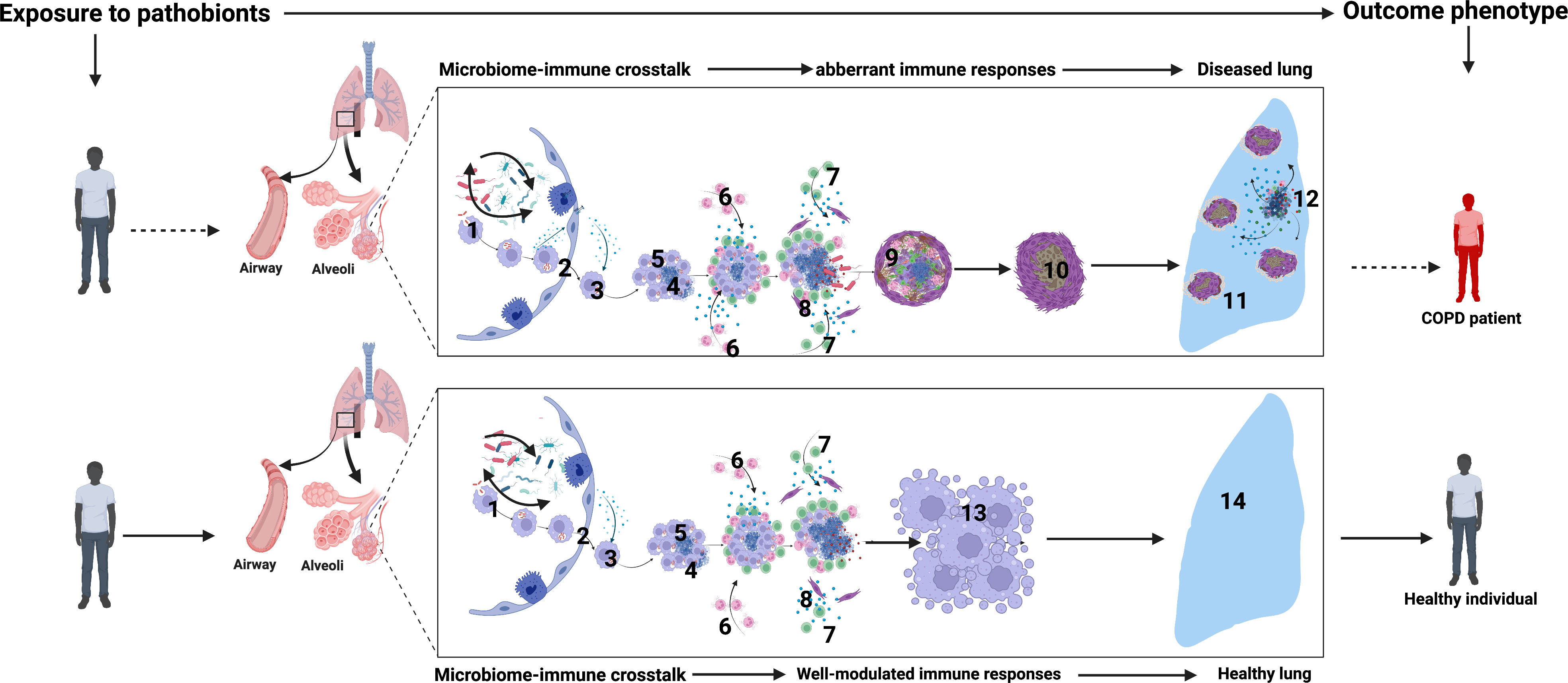
Figure 5 Airway microbiome-immune crosstalk in healthy versus diseased lungs (COPD): 1. Initial crosstalk between airway microbial communities and the mucosal innate and adaptive immune cell activation sets the tone of airway mucosal immune responses. 2. Infected macrophages from the distal airways and alveolar spaces migrate into the interstitium in an IL-1R-dependent manner. 3. Intracellular bacteria replicate within macrophages. 4. Some intracellular pathobionts induce infected macrophage apoptosis and expression of host lytic proteins in an ESX-1-dependent manner. 5. Newly recruited macrophages engulf infected cell debris. 6. Neutrophils infiltrate the airways to orchestrate inflammation, engulf dying infected cells, and kill bacteria through NETosis and the release of lytic enzymes. 7. Microbial-specific T cells arrive in airways and produce cytokines such as IFN-γ, which enhance the microbicidal activity of alveolar macrophages, monocytes, and DCs 8. Macrophage and neutrophil necroptosis lead to the release of lytic proteins such as MMPs, defensins, and cathepsin G into the extracellular space. Subsequent induction of lytic proteins causes extensive tissue damage. 9. Over time, with attempted healing and subsequent inflammation, extensive lung damage, fibrosis, and reduced lung function ensue. 10-11. Extensive fibrosis further reduces lung compliance and worsens lung function. 12. Sustained dysbiosis driven by periodic insults such as bacterial, viral, and fungal infections and air pollution or smoking orchestrate more damage and susceptibility. 13. In contrast, in healthy airways, robust innate and adaptive immune reprogramming promotes a highly modulated immune microenvironment with balanced lethal and resolving inflammatory states. This is optimal to contain airway commensals and pathobionts hence maintaining healthy lungs seen in 14. Created with BioRender.com.
Airway mucosa houses niche-specific bacterial communities varying in biomass density and diversity as one transition from the upper to lower respiratory tract (123). A physiological gradient primarily drives this difference as a consequence of a gradual increase in the relative humidity (124) and temperature (125), a decrease in the partial pressure of oxygen (pO2), an increase in the partial pressure of carbon dioxide (pCO2), and a gradually increasing pH along the respiratory tract (126, 127). pO2/pCO2 gradient is further determined by airway ventilation and the environmental air quality (126, 127). These physiological parameters determine niche-specific selective growth conditions that ultimately shape the microbial communities along the respiratory tract. Therefore, changes in airway physiological conditions directly impact airway microbial composition. During inhalation, bacteria-containing particles are deposited into the respiratory tract. Particles larger than 10μm in diameter remain in the upper respiratory tract, whereas particles smaller than 1μm in diameter reach the alveolar spaces (128). Pili and fimbriae facilitate the attachment of deposited bacteria on the respiratory mucosa (129). Most bacteria fail to attach to the mucosa and are cleared via mucociliary action of the respiratory tract (130); some become neutralized by mucosal surface IgA (131, 132) while others are broken down by mucosal antimicrobial peptides (133). The implication of these immune protective mechanisms in COPD is further elaborated in separate sections. Successfully attached bacteria colonize the airway mucosa, replicating rapidly to establish dense biological niches along the respiratory tract (134) (Figure 3). These bacteria thrive in well-balanced ecosystems, reaching saturation above which competition for space and nutrients limits their growth (135–142). Under physiological conditions, an ecosystem characterized by high bacterial numbers and diversity promotes the utilization of a versatile pool of metabolites. It favors competition for nutrients (135–142), making it difficult for pathogenic bacteria to invade and colonize these niches. In diseased airways, deranged physiological conditions promote the overgrowth of specific bacterial species, decreasing community complexity and increasing preference for the consumption of particular metabolites (129). As a result, reduced competition for nutrients makes it easier for pathogenic bacteria to survive and colonize the respiratory mucosa (143, 144).
Similarly, the depletion of specific bacterial species, for instance, following prolonged antibiotic use, reduces pressure on the ecosystem, creating space and nutrients for other bacterial species to invade and colonize the mucosal field (145). Successful colonization and subsequent mucosal invasion by pathogenic micro-organisms trigger immune activation, inflammation, tissue damage, and further dysbiosis, creating a positive feedback loop that sustains airway inflammation. Evidence from several human airway microbiome studies summarized in a recently published multi-omics meta-analysis supports dysbiosis in inducing immune activation, inflammation, and tissue damage. Although the role of airway microbiome in COPD causation cannot be directly deduced, these findings show a difference in the bacterial communities in healthy and diseased states (24). During the early stages of COPD, airways are predominantly colonized with H. influenzae, M. catarrhalis, and S. pneumoniae. These bacterial species drive inflammation and tissue damage as previously described (111, 115–117, 119–121). Mechanisms of tissue damage induced by these bacteria are discussed further in the following sections. In advanced disease, impaired mucosal defense and reduced O2 tension in the airways favor the growth of P. aeruginosa, which promotes further inflammation and a vicious circle of tissue damage (146). Exposure and successful airway colonization by new strains of these bacteria trigger inflammation associated with exacerbation (108, 110, 146, 147) and impaired lung function (122).
Cell-to-cell communication between microbes also influences composition along the respiratory mucosa (134) (Figure 3). Interaction between different bacterial species along the airway mucosa determines microbial diversity with implications for COPD (108). Interspecies interactions have been observed among COPD patients colonized with bacterial species such as H. influenzae, M. catarrhalis, S. pneumoniae, and P. aeruginosa (110). Their composition significantly varies as individuals transition from stable disease to COPD exacerbation (147). In stable COPD, S. pneumoniae enrichment positively correlates with H. influenzae enrichment, while P. aeruginosa enrichment negatively correlates with H. influenzae and M. catarrhalis and positively correlates with S. pneumoniae enrichment (108). In the exacerbation state, the relationship between S. pneumoniae and H. influenzae enrichment disappears with the persistence of a correlation between P. aeruginosa, H. influenzae, and M. catarrhalis (108). These observations reveal extensive bacterial interactions among many unculturable bacterial species colonizing the airways. Most likely, this complex interaction is driven by underlying bacterial cell-to-cell communication as individual species establish their niche in the airways. Studies describing the molecular basis of this interaction in the airways are limited. A few illustrate bacterial interactions between selected species following nasopharyngeal colonization (110, 148–151). As described below, extensive studies performed on the gut microbiome illuminate these molecular mechanisms. We believe these pathways are shared. Gut microbiome studies have described bacterial species which directly inhibit other species within the same ecological niche through the secretion of antimicrobial peptides and lytic enzymes. Mechanistically, these factors impede the growth of adjacent bacterial cells via inhibition of peptidoglycan wall synthesis, pore formation, bacterial lysis, and nucleic acid degradation (152). A group of AMPs termed bacteriocins such as microcin, thuricin, and lantibiotics inhibit gram-negative bacteria such as E.coli, S. enterica, C. difficile, and E. faecalis (153–156). Other bacterial species deploy Type VI secretion system (T6SS) through quorum sensing to transport toxic cargo into their micro-environment or other bacterial cells (157–159). Consequently, this kills potentially pathogenic bacteria, maintaining a balanced ecosystem in the respiratory mucosa. For instance, the uptake of nucleic acids from commensal Neisseria species induces cell death of pathogenic N. gonorrhea (160). This is caused by the misrecognition of the DNA’s methylation pattern in N. gonorrhea (160). Similarly, the injection of a defective enzyme into a nearby bacterial cell by P. aeruginosa depletes the target cell’s ATP resulting in microbial cell death (161). B. fragilis secretes a ubiquitin-like protein via T6SS with potent inhibitory activity against coresident strains of B. fragilis (162). Besides secretion systems, bacteria utilize nanotubes to transport plasmids from bacterium to bacterium and extract nutrients such as iron from mammalian host cells (163, 164). As previously stated, a few airway microbiome studies have reported such complex bacterial ecosystem behavior. We hope that with the advent of enriched microbiology culture techniques such as mucin-enriched media, detailed investigations focusing on cell-to-cell interactions between airway bacterial species can be pursued. Such interactions occur in the respiratory mucosa shaping the airway microbiome. As elaborated previously, in diseased airways, the depletion of a single commensal species promotes the replication of another potentially pathogenic species. Disruption of such a balance in the ecosystem consequently triggers and sustains mucosal inflammation and bystander tissue damage (147) (Figures 3–5), as detailed in the following sections. This phenomenon has been described in COPD when the distal airway is colonized with bacterial species such as P. aeruginosa, depleting species such as H. influenzae and M. catarrhalis, and enriching other bacteria such as S. pneumoniae. We believe that the differential changes in the abundance of unculturable bacterial species inhabiting the airway following colonization with new strains or species occur, driving dysbiosis, which triggers acute inflammation associated with exacerbation (108, 110, 146, 147). Other possibly interacting bacteria that induce exacerbations, including Chlamydia spp, L. pneumophilia, Mycoplasma spp, and Non-typable H. influenzae, have been described elsewhere to drive immune inflammation (165)
A recent focus and comprehensive investigation of the microbiome metabolites, geared towards illuminating microbiome cell-to-cell communication and microbe-host cell immune crosstalk, has drastically improved our understanding of the mechanisms of microbiome mediated immune-modulation in health or dysregulation in disease. Microbial metabolites and their receptors have been described in detail elsewhere (80, 166, 167). Worth mentioning are metabolites of carbohydrates, amino acids, lipids, bile acids, and nucleic acids. In-depth characterization of microbial species based on their ability to produce specific metabolites shows that members of the Bacteroidetes phylum are good producers of acetate (C2) and propionate (C3). In contrast, Firmicutes are efficient producers of butyrate (C4). Other molecules, such as lactate and succinate, are converted to propionate (C3) by several species (168–173). C1-C4 molecules comprise the classical short-chain fatty acids (SCFAs). They can be transported systemically from their production site to other sites, for instance, from the gut to the liver, muscles, lungs, and brain, acting in an endocrine manner (80). These metabolites act on cognate G protein-coupled receptors (GPR43, GPR41, GPR109A, and Olfr78) (174, 175), widely distributed on several immune cells including but not limited to epithelial cells, neutrophils, macrophages, and dendritic cells to induce immune responses (176–180). Signaling via these GPRs generates pro-inflammatory responses via activation of MAP/PI3K kinases and mTOR (181). Anti-inflammatory responses are also generated when intracellular β-arrestins are engaged, inhibiting NF-kB (182–184). Both GPR41 and 43 promote proinflammatory responses, whereas only GPR43 can alternatively generate an anti-inflammatory signal (182–184). It is yet to become known which pathways are preferentially selected by SCFAs. To achieve an efficient immune modulatory program, phased engagement of both receptors would be expected to occur under physiological conditions. Failure of this to happen most likely, results in immune dysregulation, as could be the case for airway dysbiosis. As previously described, SCFAs induce epigenetic changes reprogramming mucosal immune cells to tolerate commensal bacteria.
In dysbiosis, SCFAs induce several defense mechanisms aimed at maintaining mucosal integrity, such as goblet cell activation, mucus hypersecretion (185, 186), replenishment of mucosal surface IgA levels (187–191), production of RegIIIγ (192) and NLRP3 inflammasome activation with the secretion of pro-inflammatory cytokines (193). Whereas these mechanisms have been demonstrated in the gut, parallel mechanisms could exist in the airways. Due to limited data supporting airway accumulation of SCFAs (194), the direct role of SCFAs on airway mucosal immune response cannot be deduced. Several investigators argued that peripheral immune cell activation and subsequent recruitment into the airways drove immune crosstalk (194). Indeed, several studies confirmed this hypothesis. Using animal models, authors demonstrated that the systemic effects of SCFAs were mediated via the bone marrow in a mechanism similar to trained immunity (195–199). In an allergic airway inflammation model, systemic acetate and propionate induced the production of macrophage and dendritic cell progenitor cells (MDPs) as well as common dendritic cell progenitor cells (CDPs) in the bone marrow, which subsequently migrated to the airways to form mature DCs (200–205). In another study, it was demonstrated that although the effects of SCFAs on bone marrow were context-dependent, they ultimately primed myeloid cell proliferation and subsequent migration into the airways, modulating immune responses in an airway influenza infection model (200, 206–208). The relevance of this crosstalk in the context of COPD pathogenesis is supported by evidence of the high prevalence of pulmonary impairment among individuals with chronic inflammatory bowel disease (50%) and individuals with irritable bowel syndrome (33%). Additionally, individuals with COPD are 2 to 3 times more likely to be diagnosed with irritable bowel disease and increased intestinal permeability (209–211). These observations warrant further investigation to underpin mechanisms of lung damage secondary to SCFAs.
Besides carbohydrate metabolism, other genera such as Clostridium, Bacillus, Lactobacillus, and Streptococcus metabolize amino acids including but not limited to glycine, lysine, arginine, leucine, isoleucine, and valine (212). Their biogenic derivatives, such as ammonia, monoamines, polyamines, histamines, agmatine, and cadaverine, induce immune responses via G protein-coupled receptors (213). Furthermore, the metabolism of branched-chain amino acids yields branched-short SCFAs such as isobutyrate, valerate, and isovalerate, which like C4 molecules, induce histone deacetylation, consequently regulating immune responses, as previously elaborated (214, 215). Of critical importance to immune activation and function is the molecule indole produced from tryptophan metabolism (216). Its derivatives, kynurenine, indole-3-acetic acid, and tryptamine, activate the aryl hydrocarbon receptor (AhR) (217), inducing ILC3 and Th17 immune responses (213, 218). A plethora of microbial species metabolizes bile acids as well. Bile acids induce immune cell responses via their receptors (FXR, VDR, PXR, and TGR5) (219, 220). Similarly, these metabolites induce several immune responses to preserve mucosal integrity (221–224) (73, 80, 200). In dysbiosis, metabolites are significantly altered, dysregulating immune activation networks locally and systemically with persistent inflammation. In COPD, this persistent inflammation drives tissue damage with reduced lung function and enhanced respiratory symptoms. Recently, several metabolites such as polyamines, xanthine, glycosphingolipids, and glycerophospholipids have been correlated with enhanced respiratory symptoms and reduced lung function (225). In another recent study, a microbiome-derived metabolic COPD signature comprising 46% lipid, 20% xenobiotic, and 20% amino acid-related metabolites has been reported (29). With recent advances in omics technology, there is a need to further characterize microbiome-derived metabolites as mediators of airway inflammation in COPD.
From the previous section, we can deduce that diverse metabolites’ activity drives the microbiome to impact immune responses locally and systemically. Several authors have referred to this crosstalk as the gut-lung axis to highlight the existence of communication between the gut microbiome and airway immune mucosal system as previously described (194, 226–230). Sufficient data indeed supports the role played by the gut microbiome in driving airway inflammation to cause COPD, as previously described (227, 231, 232). However, the mechanistic underpinnings of the gut-lung axis are yet to be fully deciphered in the context of COPD. Furthermore, whether a bidirectional lung-gut axis exists remains to be answered. Chronic airway diseases such as COPD, asthma, and cystic fibrosis show airway dysbiosis and induce gastrointestinal disturbances such as irritable bowel disease (227). However, this is most likely a result of oral intake of exogenous molecules such as antibiotics and other therapies for chronic respiratory diseases that ultimately cause gut dysbiosis and associated conditions. Mechanistic research utilizing parabiosis animal models is needed to decipher the gut-lung axis fully.
Besides mucosal immune modulation via microbiome derived-metabolites, airway microbiome pathogen-associated molecular patterns (PAMPs) are sensed by the host pattern recognition receptors (PPRs) upon successful attachment to the mucosa (233) (Figure 1, 2). Toll-like receptor TLR2, in conjunction with TLR1/6, recognizes bacterial lipoproteins, whereas TLR4 recognizes bacterial lipopolysaccharide (LPS) (234, 235). Via MyD88-dependent TLR signaling, mucosal epithelial cells secrete AMPs which inhibit bacteria by targeting cytoplasmic and cell-wall components (192, 236) (Figures 1, 2). The significance of such a response has been demonstrated in mouse models where microbial depletion via antibiotic exposure significantly diminishes the mucosal secretion of AMPs (237), with a consequent reduction in the clearance of pathogenic bacteria (237). This is reversed upon mucosal replenishment with a bacterial cocktail, which activates epithelial cells to produce more AMPs (237). In addition to MyD88-dependent activation, sensing of microbial-derived products by nucleotide-binding oligomerization domain-containing receptors such as NOD1, NOD2, and NOD, LRR- and pyrin domain-containing 6 (NLRP6) inflammasome in the epithelial cells sets the pace for a pro-inflammatory signal along the mucosa (238–240). This response restricts mucosal colonization by pathogenic bacteria via secretion of IL-1β and IL-18 cytokines, which are processed from their precursors upon NLRP6 inflammasome activation (238) (Figures 1, 2). In an animal model, authors showed by using mice deficient in IL-18 that activation of the NLRP6 inflammasome induced IL-18 secretion by epithelial cells and prevented gut colonization by pathobionts (238). A recent study has described a microbiome-mediated type I interferon immune response, dependent on cGAS-STING activation irrespective of MyD88-dependent signaling or direct host-bacteria interactions (241) (Figures 1, 2). This study demonstrated that bacterial-derived outer membrane vesicles (OMVs) delivered into distal host cells activated the cGAS-STING-IFN-I axis, promoting clearance of both DNA and RNA viruses (241). Whether such findings can be applied to respiratory mucosa remains to be answered. Given similarities in embryonic origin, structure, and the immune responses mounted in the gut and airway mucosa (227, 242), we believe that a similar response is orchestrated along the airway to maintain mucosal integrity. Indeed, in one animal study, authors demonstrated the induction of a robust and broad innate immune protection of the airways following S. pneumoniae infection, effective against Gram-positive and Gram-negative bacteria and the fungus A. fumigatus (243). The response was characterized by activation of NF-κB, Type I/II IFNs, and Card9-Bcl-Malt pathways associated with upregulated expression of antimicrobial peptides (243). Amidst dysbiosis, sustained immune signaling results in aberrant immune response and inflammation along the mucosa, with bystander tissue damage. Earlier studies demonstrated the tissue-damaging effects of bacterial infection with non-typable H. influenzae using models of nasal turbinate epithelium (244). Similar observations have been recently reported for N. subflava (9). In dysbiosis, bacterial components such as lipoproteins, lipopolysaccharide, and peptidoglycans are released, inducing innate immune responses. These sustain inflammation and tissue damage associated with progressive obstruction observed in COPD.
The airway microbiome maintains the integrity of the mucus layer along the respiratory mucosa, preventing massive contact between microbes and the epithelial cells (134). Mucus along the respiratory airway is a major innate immune barrier that, to a significant extent, traps bacteria and clears the airway of these bacteria via mucociliary mechanisms (130). Goblet cells produce a variety of glycosylated mucin proteins along the mucosa (245). Mucins are secreted from the submucosal glandular and goblet cells lining the epithelium (246). Under homeostasis, the airway epithelial lining contains a few goblet cells and a moderate number of submucosal glands (247). Upon activation by bacterial products, environmental toxins, and proinflammatory cytokines, TNF-α, IL-1β, IFN-γ, IL-17A, IL-4, IL-9, and IL-13, goblet cells expand drastically, and submucosal glands increase in frequency, secreting several mucins (248). MUC5AC and 5B are the most predominant among mucin glycoproteins so far described, with MUC2 being the pioneer member characterized (248, 249). Further studies revealed that the gene encoding MUC2 is located next to the MUC5A on chromosome 11p15.5 (250), and its expression occurs early during goblet cell hyperplasia preceding that of the MUC5AC gene (248). Early studies demonstrated that bacterial-derived products such as LPS induced MUC2 expression in vitro using intestinal models via the Ras-MEK1/2-ERK1/2 signaling (251–253). Furthermore, the involvement of TLRs and NF-kB in mucin gene expression has been demonstrated (246). H. influenzae lysates upregulate MUC2 expression via the NF-kB activation (246). Other mechanisms reported to date include signaling via the TGF-β/Smad pathway (246).
Predominantly, mucin-2 is secreted in copious amounts on the mucosa following TLR2/4 and NOD1/2 activation (134). Together with other mucin proteins, mucin 2 forms an impervious layer that prevents massive contact between microbes and the epithelial cells. This barrier prevents potentially exaggerated immune responses. In the well-studied gut microbiome, bacterial species such as B. thetaiotaomicron and F. prausnitzii induce mucin gene expression, protein glycosylation, and mucus-secreting goblet cell differentiation (254). In addition to mucus production, F. prausnitzii maintains mucosal integrity by strengthening epithelial cell tight junctions in an IL-10-dependent manner (255, 256). Therefore, such bacterial species’ depletion reduces mucosal integrity and increases susceptibility to colonization by pathogenic bacteria (Figure 5). Once pathogenic bacteria successfully establish infection in the airways, mucus hypersecretion is induced as part of the acute inflammatory response (257–259). Early studies demonstrated some species, such as H. influenzae, S. pneumoniae, and P. aeruginosa, as potent mucin inducers (260). These studies further described P. aeruginosa as a proteolytic mucin-inducer in contrast to H. influenzae and S. pneumoniae as non-proteolytic mucin-inducers (260). Therefore, it is plausible that in the setting of dysbiosis predominated by known culprits (H. influenzae, M. catarrhalis, S. pneumoniae, and P. aeruginosa), sustained inflammation and mucin gene expression drives mucus production, compounded by impaired mucociliary clearance, distal airway occlusion, and reduced peak expiratory flow, typically observed in patients with chronic bronchitis (257–259). Mucin hypersecretion in COPD patients has been well-described in several studies (246, 261–270). Its accumulation in the airways of COPD patients has been reported as a predictor of mortality, especially among those with a lower baseline FEV1 (271). Mucus hypersecretion further reduces diffusion and exchange of gases across narrowed airways, worsening an already existing ventilation-perfusion mismatch. Furthermore, in dysbiosis, airway mucociliary activity is severely impaired, further complicating mucus clearance (111, 272–275). Mechanistically, bacterial pathogens induce direct epithelial cell injury, significantly disrupt mucociliary mechanics, and promote hypersecretion of mucus, which creates a conducive environment for pathogens to thrive (111, 272, 276–278).
As previously described, one of the outcomes of airway microbiome-immune crosstalk is the secretion of diverse cationic amphipathic peptides, known as antimicrobial peptides (279–281). These molecules kill invading bacterial pathogens in the mucosa, preserving microbial ecology (282, 283). In this review, we discuss two major classes of human-derived antimicrobial peptides: cathelicidins and beta-defensins (284, 285), and further highlight studies implicating such molecules in COPD pathogenesis (286, 287). We acknowledge the existence and role played by other lytic proteins, such as lysozymes (288–291), lactoferrin (292–294), secretory leucocyte protease inhibitor (295–298), cathepsins (299–303), granzymes (304–309), and S100 proteins (310–317). These molecules have been extensively discussed in the referenced articles.
The CAMP gene transcribes cathelicidin (LL-37) in mucosal epithelial cells and other cells of the immune system (318, 319). As elaborated elsewhere, LL-37 induces: (i) microbial killing via cell wall disruption (320–322), (ii) production of pro-inflammatory cytokines such as IL-1β, IL-6, and IL-17 (323–327), and anti-inflammatory cytokine IL-10, which suppresses IL-6, IL-12, TNF, HLA-DR, CD80, CD83, CD86, and CCR7 expression (326, 328, 329). Furthermore, LL-37 induces chemokines CCL2 and CCL7 in IFNγ-dependent manner (323) and polarizes macrophage differentiation towards classical (M1) phenotype and DCs towards cDC1s (330, 331). It also activates the inflammasome in macrophages and monocytes via the P2X7 receptor (332) and induces neutrophilic and eosinophilic migration (319). Other effects, such as angiogenesis, wound healing, and apoptosis, have also been reported (279). It is worth mentioning that, of all cells expressing LL-37, epithelial cells express the molecule only upon activation by inflammatory signals such as infection (318). Vitamin D3 has been demonstrated to regulate the expression of LL-37 via several vitamin D3 response elements located in the CAMP gene promoter region (333). Other than epithelial cells, Vitamin D3 regulates LL-37 expression in monocytes, keratinocytes, and neutrophils in an LPS-synergistic manner via TLR-1/2 in response to bacterial infection (319). Mechanistically, LL-37 binds and activates several extracellular and intracellular receptors (334), inducing pro-inflammatory genes via the activation of transcription factors, NF-kB, CREB1, HIF1α, AP-1/2, and EGR-1 (326, 334, 335).
Besides the known protective effects of LL-37 against invading pathogens at the mucosa, several studies have implicated this peptide in COPD (289, 315, 336, 337, 337, 338, 338, 339, 339, 340, 340, 341, 341, 342, 342, 343, 343, 344, 344, 345, 345, 346, 346–351). Investigators have demonstrated its association with poor lung function (348, 349). In a setting of cigarette-smoke exposure, low levels of LL-37 have been independently associated with lower FEV1. This decrease in FEV1 is most significant among individuals with more deficient vitamin D3 (350) and is sustained at 6 and 18 months post-recruitment (351). Whereas low LL-37 was associated with a history of bacterial pneumonia, the inclusion of pneumonia in the adjusted model did not change the findings (350), implying a direct role of LL-37 in affecting lung function. It is still unclear whether increased susceptibility to bacterial infections sets the pace for inflammation and ensuing tissue damage in low LL-37 or whether the peptide directly impacts lung function. However, a recent study favoring the former demonstrates that inhibition of LL-37 promotes bacterial infection, dysbiosis, and inflammation (352). In contrast, earlier in-vitro studies tend to the latter, showing that endogenous cathelicidin (mCRAMP), a mouse ortholog, reduces emphysema severity, probably relating to its anti-inflammatory and healing features (353). Further investigation into the molecular mechanism of LL-37-mediated-COPD pathogenesis is urgently needed. In the setting of well-established COPD, the profile of LL-37 completely reverses, with several studies reporting elevated levels in COPD compared to healthy controls (349, 354–360). This peptide is further elevated during acute COPD exacerbations compared to stable disease due to inflammation secondary to bacterial infection (296), supported by elevated pro-inflammatory cytokines, IL-6, and IL-8 (296). Moreover, in stable COPD, high levels of LL-37 predict acute exacerbation (296, 361). As expected, COPD patients with vitamin D deficiency have significantly lower LL-37 (362–365). This association is even more evident among individuals with acute exacerbation (296, 361). Complex changes in antimicrobial peptides have been reported in dysbiosis among individuals with. For instance, following infection of the airways with a new bacterial strain, LL-37 increases, while lysozyme and SLPI decrease (289). These changes in LL-37 and SLPI are more significant in exacerbation compared to the stable disease (289). Within COPD stages, LL-37 significantly varies from one study to another. Several investigators have reported conflicting results of LL-37 levels based on COPD severity stages (366). Most studies to date have been limited by small sample sizes, heterogeneous sampling (induced versus expectorated sputum versus BAL fluid), and COPD heterogeneity (smoking versus non-smoke-related COPD). This warrants the careful design of adequately powered studies investigating LL-37 as a COPD severity biomarker. Nevertheless, a few studies on COPD stratification based on exacerbation risk shows that the LL-37 is lower in high-risk exacerbators compared to low-risk counterparts (361, 366, 367). Similarly, COPD stratification based on severity shows that LL-37 is significantly reduced in severe disease compared to mild/moderate disease (296, 348). Several authors argue that this observation is most likely driven by a high background of NF-kB activation in severe disease, which has been reported to inhibit LL-37 (366).
A second family of antimicrobial peptides, called defensins, exists and contributes to immune protection against pathogenic bacteria, as described elsewhere (283). Two classes, α- and β-defensins, exist based on the length of their peptides (368, 369). These molecules contribute to granulocyte antimicrobial activity, intestinal mucosal cell defense, and cutaneous host defenses. Six members of the α-defensin include neutrophil peptides (type1-4 α-defensins) secreted by neutrophils, monocytes, lymphocytes, and NK cells. At the same time, types 5 and 6 are expressed and secreted by Paneth cells of the small intestines, epithelial cells of the airways, and gastrointestinal and female reproductive tracts (370). In contrast, β-defensins are widely distributed and expressed in epithelial cells, monocytes, macrophages, and DCs (370). Whereas type 1 β-defensin is constitutively expressed in epithelial cells, type 2 β-defensin is inducible by NF-kB activation (370). Bacteria in the mucosa stimulate the production of both type 3 and 4 β-defensins (371, 372). TNF has also been reported to stimulate the production of type 3 β-defensins. Like LL-37, the secretion of β-defensins produces both pro- and anti-inflammatory signals. For instance, upon activation, neutrophils secrete α-defensins which increase the expression of TNF and IFNγ, with a consequent increase in the expression of FcγRIIB and FcγRI (373). These changes ultimately enhance macrophage phagocytosis. On the contrary, activation of necroptotic neutrophils induces high levels of α-defensins which antagonize the release of nitric oxide and pro-inflammatory cytokines from macrophages, toning down the pro-inflammatory signals (374). Other effects described elsewhere include chemotaxis, angiogenesis, wound healing, antitumor, antifungal, antiviral, and less potent antimicrobial activity (279). β-defensins have been implicated in COPD pathogenesis and disease progression (297, 315, 375–381). Reduced lung function has been associated with increased levels of defensins (380, 382–384). Earlier studies investigating mechanisms of tissue damage in COPD by neutrophils demonstrated that upon neutrophilic infiltration of the airways and activation, neutrophils release defensins which not only play a role in antimicrobial defense but also induce lung tissue injury in addition to other molecules such as serine elastases, cathepsin G and proteinase 3 (375). In in vitro models, these molecules decreased the integrity of the epithelial cell layer and frequency of ciliary beat, as well as inducing mucus hypersecretion and mediating immune modulation (375, 385). The activity of defensins is known to be regulated by the α1-proteinase inhibitor. This molecule competitively blocks defensin’s cytotoxic and stimulatory activity toward epithelial cells (375).
Airway microbiota has been demonstrated to mobilize and reprogram alveolar macrophages, as illustrated in Figures 1, 2. In antibiotic-treated mice, depletion of commensal bacteria induced lower frequencies and numbers of alveolar macrophages. Specifically, these macrophages were reprogrammed to express higher levels of Arg1, CCL24, IL-13, IL-10, IL-6, and IL-1β, consistent with the M2 phenotype (386). These macrophages returned to normal levels following the administration of a consortium of commensal bacteria in the respiratory mucosa of animals previously treated with antibiotics (386). In addition, the microbiome has been shown to potentiate macrophage-bacterial killing and clearance via granulocyte-macrophage colony-stimulating factor (GM-CSF) signaling, reported to be driven by the IL-17/22 axis (387). Models of commensal colonization in antibiotic-treated and germ-free mice have shown that potent NLR-stimulating bacteria in the upper airways promote resistance to airway mucosal colonization by pathogenic bacteria through NOD2 and GM-CSF signaling (387). Antibiotic treatment has also been associated with impaired alveolar macrophage metabolism and defective bactericidal activity (388). Whole-genome mapping of alveolar macrophages revealed upregulation of metabolic pathways in dysbiosis (389), correlating with reduced cellular responsiveness (389). Compared with controls, alveolar macrophages obtained from microbiome-depleted mice showed a diminished capacity to phagocytose bacteria (389). In dysbiosis, pathobionts directly infect alveolar macrophages and use them as vehicles to access the lung interstitium (Figures 3, 4), attracting neutrophils, monocytes, and other inflammatory cells to orchestrate inflammation and lung damage (9, 390). In dysbiosis, the airway microenvironment drives the activation of alveolar macrophages, resulting in mixed phenotypes observed in COPD (391). Colonizing the distal airways with pathogenic bacteria sets an inflammatory tone characterized by increased levels of pro-inflammatory cytokines such as TNF-α and IL-6 (391). The induction of M1/M2 macrophage phenotypes results in a mixed phenotype of airway inflammation and tissue damage driven by classical (i.e., M1) phenotype and airway remodeling and fibrosis driven by alternative (i.e., M2) phenotype. Similar to smoke-induced COPD, other studies report a gradual transition from upregulation of M1 genes to downregulation of the same genes followed by upregulation of M2-related genes (9, 392), which is usually a typical pattern followed from acute inflammation to resolution. Chronic inflammation driven by dysbiosis with a resultant mixed M1/M2 phenotype is the most likely occurrence. M1-driven lung damage is mediated via matrix metalloproteinase (MMP) activity, oxidative stress caused by reactive oxygen and nitrogen species, and many other lytic proteins on parenchymal tissue as elaborated elsewhere (2, 4, 393–396).
Whereas airway-based human studies demonstrating the direct impact of the microbiome on airway neutrophilic responses are currently scarce, much evidence is inferred from data obtained from intestinal mouse models, which have been extensively reviewed and well-elaborated elsewhere (397). Among highlighted studies, the microbiome has been demonstrated to modulate the neutrophil function and aging via TLR- and MyD88-mediated signaling pathways (398). In a germ-free mouse model, the inflammatory challenge was associated with a reduction in neutrophil recruitment and cytokine production (399), which reversed in the setting of lipopolysaccharide pre-treatment. Notably, this arrest in neutrophilic migration and function was IL-10 dependent (399). In another study, depletion of the microbiome significantly reduced the quantity of circulating aged neutrophils, thus considerably reducing neutrophil-mediated tissue damage (398).
Furthermore, the microbiome has been reported to influence the development and function of neutrophils in several micro-environments (75). Following the development, immature neutrophils demonstrate limited functional quality characterized by reduced proinflammatory activity. However, after microbial sensing via MyD88-dependent pathways, neutrophils become better at phagocytosis and execution (Figure 4) (400, 401). In germ-free and antibiotic-treated mouse models, neutropoiesis in bone marrow is severely impaired, consequently delaying systemic bacterial clearance (401). Similarly, microbiome-derived metabolites such as short-chain fatty acids (SCFAs) drive neutropoiesis via similar mechanisms as previously described (200). In another study, the same metabolites and cell-wall components were demonstrated to induce IL-17 cytokine production from type 3 innate lymphoid cells (ILC3s) and IL-17-producing memory mucosal CD4+T cells, consequently ramping up neutropoiesis via G-CSF production (402). A neutrophilic response at the airway mucosa is critical to eliminate pathogenic micro-organisms, which overcome colonization resistance and successfully invade the mucosal barrier. Airway dysbiosis activates neutrophils via the MyD88-dependent pathway, resulting in NF-KB and MAPK signaling in airway epithelial cells (403–406). Through NETosis (407–409), activated neutrophils release serine-proteinases such as elastases, cathepsin G, and proteinase-3 from primary azurophilic granules. These proteinases cleave elastin, causing lung tissue damage observed in COPD (395, 410–412). Furthermore, secondary and tertiary granules release two metalloproteinases, i.e., collagenases (MMP8) and gelatinases (MMP9), which degrade lung extracellular matrix (395). Persistent neutrophil activation and NETosis orchestrate bystander tissue damage (407–409). (Figure 4, 5). In dysbiosis, LPS and TNF-α induce the production of neutrophilic chemoattractants by activated epithelial cells such as CXCL1 and CXCL8 (413), which promote neutrophilic infiltration into the airways, further driving neutrophilic inflammation and tissue damage (413). As discussed in the proceeding sections, Th17 activation further augments neutrophilic inflammation and tissue damage (414, 415).
The innate lymphoid cell family is comprised of cytotoxic cells (the natural killer cells) and non-cytotoxic subsets (referred to as ILC1, ILC2, and ILC3, based on similar nomenclature as T helper cellular sub-phenotypes) (75). Whereas studies investigating the microbiome’s influence on ILCs within the airway mucosa are few, the effect of the microbiome on ILC2 and ILC3 cells has been highlighted (416). In one study, intranasal administration of OM-85 bacterial lysate abrogated experimental allergic asthma by targeting IL-33/ILC2 axis (97). In the next section, we focus on findings from the mouse gut to infer mechanisms influencing ILC-specific immune responses along the airway mucosa. Studies examining the microbiome’s influence on ILCs have concentrated mainly on ILC3s. Recently published reports show that the depletion of ILC3s in mouse models significantly abrogates IL-22 production, with consequent loss of bacterial control within the mucosa, resulting in disease (417). In another study, authors characterized the effect of the gut microbiome on ILC regulatory landscape using antibiotic intervention and germ-free mouse models at the single-cell level. ILCs differentially integrate signals from the microbial microenvironment and generate phenotypic and functional plasticity (418). Furthermore, via ILC3 activation, the microbiome modulates the activity of other immune cells within the mucosa. For instance, (i) microbiome induces commensal-specific CD4+T cells to maintain tolerance at the mucosa (419, 420); (ii) Microbial sensing, inflammasome activation, and the production of IL-1β by macrophages drives GM-CSF secretion by ILC3s, required for macrophage function and induction of tolerance (421); (iii) the production of TNF-β by activated ILC3s drives IgA production at the mucosa (422). (iv) Finally, microbial induction of IL-22 production by ILC3s induces the expression of antimicrobial peptides from epithelial cells required for mucosal host defense (423). It is, therefore, plausible that dysbiosis at the airway mucosa disrupts ILC3-mediated immune regulation, consequently driving aberrant Th17-mediated damage (424) (Figure 5). Although the literature on ILCs in COPD is still minimal (425, 426), the role of ILC3s in COPD warrants investigation since IL-17, its principal cytokine, has already been described as a known driver of neutrophilic inflammation, and individuals with COPD have increased levels of IL-17 (426, 427). Furthermore, IL-17, IL-22, and IL-23-expressing immune cells have been reported in bronchial biopsies from COPD patients (428). Gene expression analysis in lung tissue from COPD patients further provides evidence for the role of ILC3 in COPD (429).
Dendritic cells (DCs) are strategically positioned within the airway mucosa, residing in mucosa-associated lymphoid tissue (MALT), such as the Welder’s ring in the upper airways, bronchial lymph nodes in the distal airways, as well as multiple satellites within the lamina propria along the airway mucosal tract (430, 431). Using their dendrites, DCs continuously sample airway bacteria that attach and colonize the mucosa (432–435) or gain access to the MALT via the epithelial tight junction barrier (431, 436–438). Extensive work reviewed elsewhere (431, 438, 439) suggests that depending on the type and degree of microbial exposure, airway DCs induce a wide range of immune responses from immune tolerance induced by plasmacytoid DCs to inflammation induced by conventional DCs (cDCs) (439). Phenotypic characterization of the microbiome based on DC immune responses is still a work in progress. In a recent study investigating the effect of airway microbiome-derived bacterial strains on DC activation, CD4+T cell priming, and cytokine response (440), P. aeruginosa induced high levels of TNF-α, IL−12, and IL-6 from DCs and primed CD4+T cells to secrete IFN-γ and IL-22 compared to S. pneumoniae and R. mucilaginosa (440). In another study, it has been confirmed that R. mucilaginosa inhibits airway immune pro-inflammatory responses via NF-κB-dependent mechanisms (38). This evidence illustrates the impact of dysbiosis on mucosal DC activation and eventual T-cell licensing required to orchestrate inflammation and tissue damage. Studies describing how activated DCs induced by dysbiosis drive COPD pathogenesis are minimal or still a work in progress. The role of activated DCs in driving COPD pathology has been described in the context of cigarette smoke-induced COPD (441–445), which is outside the scope of this review. As described above, bacterial species such as P. aeruginosa, known to predominate in advanced COPD, activate DCs, inducing high levels of TNF-α, IL−12, and IL-6, priming CD4+T cells to secrete IFN-γ and IL-22 (440). Whereas activated DCs secondary to cigarette smoke exposure have been reported to skew adaptive immune response towards Th1 and cytotoxic T cell lymphocyte (CTL) responses (known hallmarks of chronic inflammation in COPD) (442), currently, it’s not known how dysbiosis induces DCs into either type 1 or 2 DCs, which ultimately dictates CD4+T cell phenotypes.
As previously described, continuous microbial sampling and trafficking by activated DCs, and alveolar macrophages deliver processed microbial antigens to naïve CD4+T and CD8+T cells within MALT and draining lymph nodes (439) (Figures 4, 5). Following microbial-driven immune sensing and activation, the production of IL-12, IL-23, IL-27, and notch ligand by airway DCs, alveolar macrophages, and epithelial cells induces a Th1 response that regulates mucosal colonization by potentially pathogenic bacteria (439). The microbiome is required for optimal mucosal T-cell development, function, and memory. This role has been demonstrated in intestinal mouse models where germ-free mice have been observed to have developmental defects in lymphoid tissues (446, 447). Specifically, these animals display reduced frequencies of mucosal CD4+ and CD8+T cells and decreased numbers of TCR-expressing intraepithelial lymphocytes. Among CD4+T cell phenotypes, microbiome-mediated mucosal inflammation has been strongly linked to aberrant Th17 and suppressed Treg responses in COPD. For instance, enrichment of the lung microbiome with oral taxa is associated with Th17 lung inflammation in COPD (39). Similarly, in bleomycin-induced mouse interstitial pulmonary fibrosis (IPF), airway dysbiosis induces IL-17 cytokine, which ameliorates following either specific airway bacterial depletion or IL-17 blockade. Three commensal bacteria belonging to the genera Bacteroides and Prevotella promote fibrotic pathogenesis through MyD88-dependent IL-17R signaling via bacterial exosomes (448). These findings have been replicated in human studies where Prevotella and Veillonella spp have been associated with enhanced Th17 inflammatory response in COPD (449). In another animal-based study, S. mitis, V. parvula, and P. melaninogenica induced dysbiosis-mediated inflammasome and Th1/Th17 activation with reduced susceptibility to S. pneumoniae. These data imply an immunoprotective role of specific bacterial species in the airways (40). However, chronic Th17 inflammation in persistent dysbiosis promotes lung tissue damage (448). Besides Th17, microbial interaction with mucosal CD4+T cells induces immune tolerance. This process involves several mechanisms extensively described elsewhere (450). We highlight some of these mechanisms here. MyD88-dependent TLR2 activation by microbial-derived PAMPs, such as capsular polysaccharide A from B. fragilis, has been shown to induce the expansion of Foxp3+Tcells within the mucosa (451). Foxp3+T cells drive IL-10 production, facilitating mucosal immune tolerance. B. fragilis have also been shown to deliver antigenic products through bacterial exosomes, which upon phagocytosis by host immune cells via a non-canonical autophagy pathway, induce IL-10 expressing Foxp3+T cells (450, 452). In another mechanism, microbial-induced Tregs promote mucosal memory B or plasma cells’ IgA secretion, epithelial cells’ tight-junction protein expression, and goblet cells’ mucus production in an IL-10-dependent manner (453, 454). This maintains microbial biomass and diversity and facilitates Treg expansion through a symbiotic regulatory loop, preventing overt inflammation (450). Short-chain fatty acids potentiate Foxp3+ cell differentiation and immunomodulatory activity in the gut mucosa as previously discussed (87–89, 455). This evidence implies a tight regulatory role played by mucosal commensal bacteria in maintaining a robust mucosal Treg response. In dysbiosis, however, a heightened Th17 immune response increases TGF-β production with consequent Treg downregulation (456, 457).
A recently published article describes the role of the microbiome in shaping B-cell immune responses (63). The authors discuss microbiome-driven B cell immune activation, antibody class switching from IgM to IgA, and maintenance of memory B or plasma cells at the mucosal surface (63, 458–460). Although studies referenced in this article are biased toward the gut mucosa, emerging evidence from airway studies confirms the engagement of similar immune responses (461). Upon antigenic encounter at the mucosa, naïve B cells, via antigen-specific B cell receptors, engage the antigen, inducing an activating signal. B cells proliferate and undergo clonal selection, affinity maturation, and class switching within mucosal lymphoid tissue and some in lymph nodes, producing high-quality IgA antibodies (462). This process largely depends on T follicular helper cells (461). A recent study revealed increased expression of the cytokine IL-21 among patients with COPD, primarily in CD4+T cells. IL-21 promotes B cell maturation, antibody affinity maturation, and differentiation into plasma cells. Without co-stimulation, B cells exposed to IL-21 undergo apoptosis which controls bystander B cell immune activation (463). In addition to T cell-dependent help, evidence shows that bacterial products such as LPS can induce human IgM+ B cells to directly class switch to IgA-secreting plasma cells (461). T-independent induction of IgA has been shown to occur in isolated lymphoid follicles and the mucosal lamina propria, generating polyreactive IgA with low affinity for commensal bacteria (462). Upon production by plasma cells, IgA is transported across the mucosal epithelial cells via transcytosis and secreted onto the mucosal surface in a process that requires the binding of dimeric IgA to the polymeric IgA receptor, at least in intestinal models (464). Mucosal surface IgA preserves microbial ecology at the mucosa in several ways. It binds and enchains dividing bacteria, limiting their association with the epithelial cells (465). Selective binding of IgA to some bacterial species also inhibits bacterial cell growth. Studies have demonstrated the induction of sustained commensal-specific IgA memory responses, which become attenuated when a new bacterial strain or species invades the mucosa (458). This evidence implies a continuous IgA repertoire modification to match the dynamic changes in the mucosal microbial communities. This is possible via re-entry and further affinity maturation by somatic hypermutation of existing B cell memory clones in the mucosal lymphoid tissue (466). Bacterial coating with IgM and IgG has also been demonstrated in human intestinal models. Specifically, IgM-secreting plasma cells recognize mucus-dwelling commensals (467). Without IgA, a compensatory IgM response ensues to contain the bacteria (467). It is worth mentioning that microbiome-specific IgA, IgM, and IgG antibodies produced locally can act systemically in similar mechanisms as already described in the gut-lung axis (468). Microbiome-derived metabolites also modulate B cell immune function via induction of epigenetic changes via HDAC inhibitory activity, which induces histone acetylation, enhancing gene expression necessary for B cell differentiation into IgA/G secreting plasma cells (189, 469).
Following the proposition that autoimmunity significantly contributes to COPD pathogenesis, investigators have concentrated their efforts on underpinning the role of B cell immune responses in COPD (470–475). Earlier studies demonstrated the presence of lung lymphoid follicles and elastin-specific antibodies among patients with advanced emphysema (476, 477). Furthermore, unbiased gene expression analysis among emphysema patients revealed a strong link between airway B cells and emphysema (478). This preliminary data provided evidence to investigate further the mechanistic role of B cells in COPD pathogenesis. Studies have additionally demonstrated B cells’ critical role in promoting COPD immunopathology (479–491). For instance, enhanced B cell differentiation into IgA-producing plasma cells has been demonstrated in the airways of COPD patients, where the bronchial epithelial cells primarily provide B cell differentiating signals via the IL-6/IL-6 receptor and BAFF-APRIL/TACI pathways (492). IgA-producing B cells are increased in the distal airways of COPD patients compared to healthy controls and positively correlate with COPD severity scores (461). Results are similar in animal models following infection with P. aeruginosa, a known pathogen in COPD (461). Mechanistically, the direct role of mucosal IgA in inducing lung damage has not yet been fully elucidated (493). The non-inflammatory nature of IgA highly suggests that this molecule is less likely to contribute to COPD pathogenesis directly. However, evidence from known pathological consequences of IgA, such as IgA nephropathy (494), warrants further investigation into the direct role of IgA in COPD pathology. In airway dysbiosis, increased mucosal surface IgA is associated with airway inflammation in COPD (495–499).
This review highlights numerous airway microbiome-mediated immune pathways, mostly in animal models, that drive COPD pathogenesis. Such responses are characterized by alveolar macrophage, dendritic cell, neutrophil, monocyte, innate lymphoid cell, and Th1/Th17 cell activation phenotypes whose interaction with airway epithelial cells culminates into sustained lung inflammation and tissue damage. Although a few human-based COPD studies have also been highlighted, more research is needed to test and validate these findings in human COPD cohorts. Immunophenotyping microbiome habitats has the potential to advance microbiome-based therapeutics. Microbiome-resulting local immunophenotypes are, however, thus far poorly characterized. Accordingly, deep immune phenotyping of the airway host-microbiome interface, possible through multi-omics approaches, may meaningfully inform far more precise interventions in COPD (500).
AK: Conceptualization, methodology, original draft writing, editing, and funding acquisition. NMR, MN, JN: Draft review and editing. TS, MJ, HM-K, BB, OJS, BK, and SF: Mentorship, funding acquisition and drafting the manuscript for important intellectual content. All authors contributed to the article and approved the submitted version.
This review article was funded by grants awarded to AK from the European Union Developing Countries Clinical Trials Partnership (EDCTP) under Award number TMA2020CDF3194 and Fogarty International Center of the National Institutes of Health (NIH) under Award Number R21TW012354. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the supporting offices.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Venkatesan P. GOLD report: 2022 update. Lancet Respir Med (2022) 10(2):e20. doi: 10.1016/S2213-2600(21)00561-0
2. Chung K, Adcock I. Multifaceted mechanisms in COPD: inflammation, immunity, and tissue repair and destruction. Eur Respir J (2008) 31(6):1334–56. doi: 10.1183/09031936.00018908
3. Barbu C, Iordache M, Man M. Inflammation in COPD: pathogenesis, local and systemic effects. Rom J Morphol Embryol (2011) 52(1):21–7.
4. Barnes P, Burney P, Silverman EK, Celi BR, Vestbo J, Wedzicha JA, et al. Chronic obstructive pulmonary disease. Nat Rev Dis Primers (2015) 1:15076. doi: 10.1038/nrdp.2015.76
5. Fabbri L, Luppi F, Beghé B, Rabe K. Complex chronic comorbidities of COPD. Eur Respir J (2008) 31(1):204–12. doi: 10.1183/09031936.00114307
6. Saetta M, Turato G, Maestrelli P, Mapp CE, Fabbri LM. Cellular and structural bases of chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2001) 163(6):1304–9. doi: 10.1164/ajrccm.163.6.2009116
7. Adeloye D, Chua S, Lee C, Basquill C, Papana A, Theodoratou E, et al. Global and regional estimates of COPD prevalence: Systematic review and meta–analysis. J Global Health (2015) 5(2). doi: 10.7189/jogh.05.020415
8. Natalini JG, Singh S, Segal LN. The dynamic lung microbiome in health and disease. Nat Rev Microbiol (2022), 1–14. doi: 10.1038/s41579-022-00821-x
9. Li L, Mac Aogáin M, Xu T, Jaggi TK, Chan LL, Qu J, et al. Neisseria species as pathobionts in bronchiectasis. Cell Host Microbe (2022) 30(9):1311–27.e8. doi: 10.1016/j.chom.2022.08.005
10. Singh S, Segal LN. A lung pathobiont story: Thinking outside the koch’s postulate box. Cell Host Microbe (2022) 30(9):1196–8. doi: 10.1016/j.chom.2022.08.012
11. O'Donnell AE. Bronchiectasis in patients with COPD: a distinct COPD phenotype? Chest (2011) 140(5):1107–8. doi: 10.1378/chest.11-1484
12. Martínez-García MÁ, Soler-Cataluña JJ, Sanz YD, Serra PC, Lerma MA, Vicente JB, et al. Factors associated with bronchiectasis in patients with COPD. Chest (2011) 140(5):1130–7. doi: 10.1378/chest.10-1758
13. Patel IS, Vlahos I, Wilkinson TM, Lloyd-Owen SJ, Donaldson GC, Wilks M, et al. Bronchiectasis, exacerbation indices, and inflammation in chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2004) 170(4):400–7. doi: 10.1164/rccm.200305-648OC
14. Agusti A, Calverley P, Celli B, Coxson HO, Edwards LD, Lomas DA, et al. Characterisation of COPD heterogeneity in the ECLIPSE cohort. Respir Res (2010) 11(1):1–14. doi: 10.1186/1465-9921-11-122
15. Wedzicha JA, Hurst JR. Structural and functional co-conspirators in chronic obstructive pulmonary disease exacerbations. Proc Am Thorac Soc (2007) 4(8):602–5. doi: 10.1513/pats.200707-106TH
16. Wang L, Hao K, Yang T, Wang C. Role of the lung microbiome in the pathogenesis of chronic obstructive pulmonary disease. Chin Med J (2016) 130(17):2107–11. doi: 10.4103/0366-6999.211452
17. Tiew PY, Jaggi TK, Chan LL, Chotirmall SH. The airway microbiome in COPD, bronchiectasis and bronchiectasis-COPD overlap. Clin Respir J (2021) 15(2):123–33. doi: 10.1111/crj.13294
18. Ding K, Chen J, Zhan W, Zhang S, Chen Y, Long S, et al. Microbiome links cigarette smoke-induced chronic obstructive pulmonary disease and dietary fiber via the gut-lung axis: A narrative review. COPD: J Chronic Obstructive Pulm Dis (2022) 19(1):10–7. doi: 10.1080/15412555.2021.2019208
19. Dy R, Sethi S. The lung microbiome and exacerbations of COPD. Curr Opin pulm Med (2016) 22(3):196–202. doi: 10.1097/MCP.0000000000000268
20. Dickson RP, Erb-Downward JR, Martinez FJ, Huffnagle GB. The microbiome and the respiratory tract. Annu Rev Physiol (2016) 78:481. doi: 10.1146/annurev-physiol-021115-105238
21. Ditz B, Christenson S, Rossen J, Brightling C, Kerstjens HA, van den Berge M, et al. Sputum microbiome profiling in COPD: beyond singular pathogen detection. Thorax (2020) 75(4):338–44. doi: 10.1136/thoraxjnl-2019-214168
22. Wang Z, Bafadhel M, Haldar K, Spivak A, Mayhew D, Miller BE, et al. Lung microbiome dynamics in COPD exacerbations. Eur Respir J (2016) 47(4):1082–92. doi: 10.1183/13993003.01406-2015
23. Engel M, Endesfelder D, Schloter-Hai B, Kublik S, Granitsiotis MS, Boschetto P, et al. Influence of lung CT changes in chronic obstructive pulmonary disease (COPD) on the human lung microbiome. PloS One (2017) 12(7):e0180859. doi: 10.1371/journal.pone.0180859
24. Wang Z, Yang Y, Yan Z, Liu H, Chen B, Liang Z, et al. Multi-omic meta-analysis identifies functional signatures of airway microbiome in chronic obstructive pulmonary disease. ISME J (2020) 14(11):2748–65. doi: 10.1038/s41396-020-0727-y
25. Yatera K, Noguchi S, Mukae H. The microbiome in the lower respiratory tract. Respir Invest (2018) 56(6):432–9. doi: 10.1016/j.resinv.2018.08.003
26. Richardson H, Dicker AJ, Barclay H, Chalmers JD. The microbiome in bronchiectasis. Eur Respir Rev. (2019) 28(153). doi: 10.1183/16000617.0048-2019
27. Budden KF, Shukla SD, Rehman SF, Bowerman KL, Keely S, Hugenholtz P, et al. Functional effects of the microbiota in chronic respiratory disease. Lancet Respir Med (2019) 7(10):907–20. doi: 10.1016/S2213-2600(18)30510-1
28. Huang YJ, Erb-Downward JR, Dickson RP, Curtis JL, Huffnagle GB, Han MK. Understanding the role of the microbiome in chronic obstructive pulmonary disease: principles, challenges, and future directions. Trans Res (2017) 179:71–83. doi: 10.1016/j.trsl.2016.06.007
29. Bowerman KL, Rehman SF, Vaughan A, Lachner N, Budden KF, Kim RY, et al. Disease-associated gut microbiome and metabolome changes in patients with chronic obstructive pulmonary disease. Nat Commun (2020) 11(1):1–15. doi: 10.1038/s41467-020-19701-0
30. Russo C, Colaianni V, Ielo G, Valle MS, Spicuzza L, Malaguarnera L. Impact of lung microbiota on COPD. Biomedicines (2022) 10(6):1337. doi: 10.3390/biomedicines10061337
31. Wang Z, Singh R, Miller BE, Tal-Singer R, Van Horn S, Tomsho L, et al. Sputum microbiome temporal variability and dysbiosis in chronic obstructive pulmonary disease exacerbations: an analysis of the COPDMAP study. Thorax (2018) 73(4):331–8. doi: 10.1136/thoraxjnl-2017-210741
32. Wang YJ, Wu SS, Chu J, Kong XY. Lung microbiome mediates the progression from chronic obstructive pulmonary disease to lung cancer through inflammation. Yi Chuan= Hereditas (2021) 43(1):30–9. doi: 10.16288/j.yczz.20-315
33. Leitao Filho FS, Alotaibi NM, Ngan D, Tam S, Yang J, Hollander Z, et al. Sputum microbiome is associated with 1-year mortality after chronic obstructive pulmonary disease hospitalizations. Am J Respir Crit Care Med (2019) 199(10):1205–13. doi: 10.1164/rccm.201806-1135OC
34. Dicker AJ, Huang JT, Lonergan M, Keir HR, Fong CJ, Tan B, et al. The sputum microbiome, airway inflammation, and mortality in chronic obstructive pulmonary disease. J Allergy Clin Immunol (2021) 147(1):158–67. doi: 10.1016/j.jaci.2020.02.040
35. Diao W, Shen N, Du Y, Erb-Downward JR, Sun X, Guo C, et al. Symptom-related sputum microbiota in stable chronic obstructive pulmonary disease. Int J chronic obstructive pulm dis (2018) 13:2289. doi: 10.2147/COPD.S167618
36. Rogers GB, Zain NMM, Bruce KD, Burr LD, Chen AC, Rivett DW, et al. A novel microbiota stratification system predicts future exacerbations in bronchiectasis. Ann Am Thorac Soc (2014) 11(4):496–503. doi: 10.1513/AnnalsATS.201310-335OC
37. Lee SH, Lee Y, Park JS, Cho Y-J, Yoon HI, Lee C-T, et al. Characterization of microbiota in bronchiectasis patients with different disease severities. J Clin Med (2018) 7(11):429. doi: 10.3390/jcm7110429
38. Rigauts C, Aizawa J, Taylor SL, Rogers GB, Govaerts M, Cos P, et al. Rothia mucilaginosa is an anti-inflammatory bacterium in the respiratory tract of patients with chronic lung disease. Eur Respir J (2022) 59(5). doi: 10.1183/13993003.01293-2021
39. Segal LN, Clemente JC, Tsay J-CJ, Koralov SB, Keller BC, Wu BG, et al. Enrichment of the lung microbiome with oral taxa is associated with lung inflammation of a Th17 phenotype. Nat Microbiol (2016) 1(5):1–11. doi: 10.1038/nmicrobiol.2016.31
40. Wu BG, Sulaiman I, Tsay J-CJ, Perez L, Franca B, Li Y, et al. Episodic aspiration with oral commensals induces a MyD88-dependent, pulmonary T-helper cell type 17 response that mitigates susceptibility to streptococcus pneumoniae. Am J Respir Crit Care Med (2021) 203(9):1099–111. doi: 10.1164/rccm.202005-1596OC
41. Shukla SD, Budden KF, Neal R, Hansbro PM. Microbiome effects on immunity, health and disease in the lung. Clin Trans Immunol (2017) 6(3):e133. doi: 10.1038/cti.2017.6
42. Paudel KR, Dharwal V, Patel VK, Galvao I, Wadhwa R, Malyla V, et al. Role of lung microbiome in innate immune response associated with chronic lung diseases. Front Med (2020) 7:554. doi: 10.3389/fmed.2020.00554
43. D’Anna SE, Balbi B, Cappello F, Carone M, Di Stefano A. Bacterial–viral load and the immune response in stable and exacerbated COPD: significance and therapeutic prospects. Int J Chronic Obstructive Pulm Dis (2016) 11:445. doi: 10.2147/COPD.S93398
44. Han MK, Huang YJ, LiPuma JJ, Boushey HA, Boucher RC, Cookson WO, et al. Significance of the microbiome in obstructive lung disease. Thorax (2012) 67(5):456–63. doi: 10.1136/thoraxjnl-2011-201183
45. Sauler M, McDonough JE, Adams TS, Kothapalli N, Barnthaler T, Werder RB, et al. Characterization of the COPD alveolar niche using single-cell RNA sequencing. Nat Commun (2022) 13(1):1–17. doi: 10.1038/s41467-022-28062-9
46. Boyton R, Reynolds C, Quigley K, Altmann D. Immune mechanisms and the impact of the disrupted lung microbiome in chronic bacterial lung infection and bronchiectasis. Clin Exp Immunol (2013) 171(2):117–23. doi: 10.1111/cei.12003
47. Ivanov II, Littman DR. Modulation of immune homeostasis by commensal bacteria. Curr Opin Microbiol (2011) 14(1):106–14. doi: 10.1016/j.mib.2010.12.003
48. Wang Q, Xu R. Data-driven multiple-level analysis of gut-microbiome-immune-joint interactions in rheumatoid arthritis. BMC Genomics (2019) 20(1):1–10. doi: 10.1186/s12864-019-5510-y
49. Alexander M, Turnbaugh PJ. Deconstructing mechanisms of diet-microbiome-immune interactions. Immunity (2020) 53(2):264–76. doi: 10.1016/j.immuni.2020.07.015
50. Ichinohe T, Miyama T, Kawase T, Honjo Y, Kitaura K, Sato H, et al. Next-generation immune repertoire sequencing as a clue to elucidate the landscape of immune modulation by host–gut microbiome interactions. Front Immunol (2018) 9:668. doi: 10.3389/fimmu.2018.00668
51. Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res (2020) 30(6):492–506. doi: 10.1038/s41422-020-0332-7
52. Gunaswetha K, Sujatha E, Bramhachari PV. Understanding the interplay between the host immune–microbiome interactions: A state of the art review. Microbiom Hum Health Dis (2021), 123–41. doi: 10.1007/978-981-16-3156-6_8
53. Trujillo J, Lunjani N, Ryan D, O’Mahony L. Microbiome-immune interactions and relationship to asthma severity. J Allergy Clin Immunol (2022) 149(2):533–4. doi: 10.1016/j.jaci.2021.12.774
54. Kelly B, O'neill LA. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res (2015) 25(7):771–84. doi: 10.1038/cr.2015.68
55. Yoon BR, Oh Y-J, Kang SW, Lee EB, Lee W-W. Role of SLC7A5 in metabolic reprogramming of human monocyte/macrophage immune responses. Front Immunol (2018) 9:53. doi: 10.3389/fimmu.2018.00053
56. Basit F, Mathan T, Sancho D, De Vries IJM. Human dendritic cell subsets undergo distinct metabolic reprogramming for immune response. Front Immunol (2018) 9:2489. doi: 10.3389/fimmu.2018.02489
57. Michaeloudes C, Bhavsar PK, Mumby S, Xu B, Hui CKM, Chung KF, et al. Role of metabolic reprogramming in pulmonary innate immunity and its impact on lung diseases. J Innate Immun (2020) 12(1):31–46. doi: 10.1159/000504344
58. Krautkramer KA, Kreznar JH, Romano KA, Vivas EI, Barrett-Wilt GA, Rabaglia ME, et al. Diet-microbiota interactions mediate global epigenetic programming in multiple host tissues. Mol Cell (2016) 64(5):982–92. doi: 10.1016/j.molcel.2016.10.025
59. Peery RC, Pammi M, Claud E, Shen L. Epigenome–a mediator for host-microbiome crosstalk. Semin Perinatol (2021) 45. doi: 10.1016/j.semperi.2021.151455
60. Dominguez-Andres J, Netea MG. Long-term reprogramming of the innate immune system. J leukoc Biol (2019) 105(2):329–38. doi: 10.1002/JLB.MR0318-104R
61. Shenderov BA. The human microbiota and chronic disease: dysbiosis as a cause of human pathology. Human Microbiota Chronic Dis: Dysbiosis Cause Human Pathol (2016) 17:179. doi: 10.1002/9781118982907.ch11
62. Amatullah H, Jeffrey KL. Epigenome-metabolome-microbiome axis in health and IBD. Curr Opin Microbiol (2020) 56:97–108. doi: 10.1016/j.mib.2020.08.005
63. McCoy KD, Burkhard R, Geuking MB. The microbiome and immune memory formation. Immunol Cell Biol (2019) 97(7):625–35. doi: 10.1111/imcb.12273
64. Saeed S, Quintin J, Kerstens HH, Rao NA, Aghajanirefah A, Matarese F, et al. Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. science (2014) 345(6204):1251086. doi: 10.1126/science.1251086
65. Cheng S-C, Quintin J, Cramer RA, Shepardson KM, Saeed S, Kumar V, et al. mTOR-and HIF-1α–mediated aerobic glycolysis as metabolic basis for trained immunity. Science (2014) 345(6204):1250684. doi: 10.1126/science.1250684
66. Zhou L, Lin Q, Sonnenberg GF. Metabolic control of innate lymphoid cells in health and disease. Nat Metab (2022) 4:1650–9. doi: 10.1038/s42255-022-00685-8
67. Bandyopadhaya A, Tsurumi A, Maura D, Jeffrey KL, Rahme LG. A quorum-sensing signal promotes host tolerance training through HDAC1-mediated epigenetic reprogramming. Nat Microbiol (2016) 1(12):1–9. doi: 10.1038/nmicrobiol.2016.174
68. Ayres JS, Schneider DS. Tolerance of infections. Annu Rev Immunol (2012) 30:271–94. doi: 10.1146/annurev-immunol-020711-075030
69. Soares MP, Gozzelino R, Weis S. Tissue damage control in disease tolerance. Trends Immunol (2014) 35(10):483–94. doi: 10.1016/j.it.2014.08.001
70. Butler MI, Cryan JF, Dinan TG. Man and the microbiome: a new theory of everything? Annu Rev Clin Psychol (2019) 15:371–98. doi: 10.1146/annurev-clinpsy-050718-095432
71. Fischbach MA. Microbiome: focus on causation and mechanism. Cell (2018) 174(4):785–90. doi: 10.1016/j.cell.2018.07.038
72. Lozupone CA. Unraveling interactions between the microbiome and the host immune system to decipher mechanisms of disease. Msystems (2018) 3(2):e00183–17. doi: 10.1128/mSystems.00183-17
73. Levy M, Blacher E, Elinav E. Microbiome, metabolites and host immunity. Curr Opin Microbiol (2017) 35:8–15. doi: 10.1016/j.mib.2016.10.003
74. Levast B, Li Z, Madrenas J. The role of IL-10 in microbiome-associated immune modulation and disease tolerance. Cytokine (2015) 75(2):291–301. doi: 10.1016/j.cyto.2014.11.027
75. Thaiss CA, Zmora N, Levy M, Elinav E. The microbiome and innate immunity. Nature (2016) 535(7610):65–74. doi: 10.1038/nature18847
76. Lin T-L, Shu C-C, Chen Y-M, Lu J-J, Wu T-S, Lai W-F, et al. Like cures like: pharmacological activity of anti-inflammatory lipopolysaccharides from gut microbiome. Front Pharmacol (2020) 11:554. doi: 10.3389/fphar.2020.00554
77. Kuipers F, de Boer JF, Staels B. Microbiome modulation of the host adaptive immunity through bile acid modification. Cell Metab (2020) 31(3):445–7. doi: 10.1016/j.cmet.2020.02.006
78. Shinde T, Vemuri R, Shastri S, Perera AP, Gondalia SV, Beale DJ, et al. Modulating the microbiome and immune responses using whole plant fibre in synbiotic combination with fibre-digesting probiotic attenuates chronic colonic inflammation in spontaneous colitic mice model of IBD. Nutrients (2020) 12(8):2380. doi: 10.3390/nu12082380
79. Hooper LV, Wong MH, Thelin A, Hansson L, Falk PG, Gordon JI. Molecular analysis of commensal host-microbial relationships in the intestine. Science (2001) 291(5505):881–4. doi: 10.1126/science.291.5505.881
80. Kim CH. Immune regulation by microbiome metabolites. Immunology (2018) 154(2):220–9. doi: 10.1111/imm.12930
81. Chang PV, Hao L, Offermanns S, Medzhitov R. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Proc Natl Acad Sci (2014) 111(6):2247–52. doi: 10.1073/pnas.1322269111
82. Park JW, Kim HY, Kim MG, Jeong S, Yun C-H, Han SH. Short-chain fatty acids inhibit staphylococcal lipoprotein-induced nitric oxide production in murine macrophages. Immune netw (2019) 19(2). doi: 10.4110/in.2019.19.e9
83. Röth D, Chiang AJ, Hu W, Gugiu GB, Morra CN, Versalovic J, et al. Two-carbon folate cycle of commensal lactobacillus reuteri 6475 gives rise to immunomodulatory ethionine, a source for histone ethylation. FASEB J (2019) 33(3):3536–48. doi: 10.1096/fj.201801848R
84. Heim CE, Bosch ME, Yamada KJ, Aldrich AL, Chaudhari SS, Klinkebiel D, et al. Lactate production by staphylococcus aureus biofilm inhibits HDAC11 to reprogramme the host immune response during persistent infection. Nat Microbiol (2020) 5(10):1271–84. doi: 10.1038/s41564-020-0756-3
85. Burgess SL, Leslie JL, Uddin J, Oakland DN, Gilchrist C, Moreau GB, et al. Gut microbiome communication with bone marrow regulates susceptibility to amebiasis. J Clin Invest (2020) 130(8):4019–24. doi: 10.1172/JCI133605
86. Schaupp L, Muth S, Rogell L, Kofoed-Branzk M, Melchior F, Lienenklaus S, et al. Microbiota-induced type I interferons instruct a poised basal state of dendritic cells. Cell (2020) 181(5):1080–96.e19. doi: 10.1016/j.cell.2020.04.022
87. Furusawa Y, Obata Y, Fukuda S, Endo TA, Nakato G, Takahashi D, et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature (2013) 504(7480):446–50. doi: 10.1038/nature12721
88. Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, Deroos P, et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature (2013) 504(7480):451–5. doi: 10.1038/nature12726
89. Smith PM, Howitt MR, Panikov N, Michaud M, Gallini CA, Bohlooly-y M, et al. The microbial metabolites, short-chain fatty acids, regulate colonic treg cell homeostasis. Science (2013) 341(6145):569–73. doi: 10.1126/science.1241165
90. Chen L, Sun M, Wu W, Yang W, Huang X, Xiao Y, et al. Microbiota metabolite butyrate differentially regulates Th1 and Th17 cells’ differentiation and function in induction of colitis. Inflamm bowel dis (2019) 25(9):1450–61. doi: 10.1093/ibd/izz046
91. Zhang M, Zhou L, Wang Y, Dorfman RG, Tang D, Xu L, et al. Faecalibacterium prausnitzii produces butyrate to decrease c-myc-related metabolism and Th17 differentiation by inhibiting histone deacetylase 3. Int Immunol (2019) 31(8):499–514. doi: 10.1093/intimm/dxz022
92. Zhou L, Zhang M, Wang Y, Dorfman RG, Liu H, Yu T, et al. Faecalibacterium prausnitzii produces butyrate to maintain Th17/Treg balance and to ameliorate colorectal colitis by inhibiting histone deacetylase 1. Inflamm bowel dis (2018) 24(9):1926–40. doi: 10.1093/ibd/izy182
93. Takahashi K, Sugi Y, Nakano K, Tsuda M, Kurihara K, Hosono A, et al. Epigenetic control of the host gene by commensal bacteria in large intestinal epithelial cells. J Biol Chem (2011) 286(41):35755–62. doi: 10.1074/jbc.M111.271007
94. Olszak T, An D, Zeissig S, Vera MP, Richter J, Franke A, et al. Microbial exposure during early life has persistent effects on natural killer T cell function. Science (2012) 336(6080):489–93. doi: 10.1126/science.1219328
95. Ganal SC, Sanos SL, Kallfass C, Oberle K, Johner C, Kirschning C, et al. Priming of natural killer cells by nonmucosal mononuclear phagocytes requires instructive signals from commensal microbiota. Immunity (2012) 37(1):171–86. doi: 10.1016/j.immuni.2012.05.020
96. Alenghat T, Osborne LC, Saenz SA, Kobuley D, Ziegler CG, Mullican SE, et al. Histone deacetylase 3 coordinates commensal-bacteria-dependent intestinal homeostasis. Nature (2013) 504(7478):153–7. doi: 10.1038/nature12687
97. Pivniouk V, Gimenes-Junior JA, Ezeh P, Michael A, Pivniouk O, Hahn S, et al. Airway administration of OM-85, a bacterial lysate, blocks experimental asthma by targeting dendritic cells and the epithelium/IL-33/ILC2 axis. J Allergy Clin Immunol (2022) 149(3):943–56. doi: 10.1016/j.jaci.2021.09.013
98. Zhang N, He Q-S. Commensal microbiome promotes resistance to local and systemic infections. Chin Med J (2015) 128(16):2250–5. doi: 10.4103/0366-6999.162502
99. Mammen MJ, Sethi S. COPD and the microbiome. Respirology (2016) 21(4):590–9. doi: 10.1111/resp.12732
100. Dickson RP, Martinez FJ, Huffnagle GB. The role of the microbiome in exacerbations of chronic lung diseases. Lancet (2014) 384(9944):691–702. doi: 10.1016/S0140-6736(14)61136-3
101. Hussell T, Lui S, Jagger C, Morgan D, Brand O. The consequence of matrix dysfunction on lung immunity and the microbiome in COPD. Eur Respir Rev. (2018) 27(148). doi: 10.1183/16000617.0032-2018
102. Reibman J, Hsu Y, Chen LC, Bleck B, Gordon T. Airway epithelial cells release MIP-3α/CCL20 in response to cytokines and ambient particulate matter. Am J Respir Cell Mol Biol (2003) 28(6):648–54. doi: 10.1165/rcmb.2002-0095OC
103. Barker DJ, Godfrey K, Fall C, Osmond C, Winter P, Shaheen S. Relation of birth weight and childhood respiratory infection to adult lung function and death from chronic obstructive airways disease. Br Med J (1991) 303(6804):671–5. doi: 10.1136/bmj.303.6804.671
104. Shaheen SO, Barker D, Shiell AW, Crocker FJ, Wield GA, Holgate ST. The relationship between pneumonia in early childhood and impaired lung function in late adult life. Am J Respir Crit Care Med (1994) 149(3):616–9. doi: 10.1164/ajrccm.149.3.8118627
105. Johnston ID, Strachan DP, Anderson HR. Effect of pneumonia and whooping cough in childhood on adult lung function. New Engl J Med (1998) 338(9):581–7. doi: 10.1056/NEJM199802263380904
106. Vuori E, Peltola H, Kallio MJ, Leinonen M, Hedman K, group S-Ts. Etiology of pneumonia and other common childhood infections requiring hospitalization and parenteral antimicrobial therapy. Clin Infect dis (1998) 27(3):566–72. doi: 10.1086/514697
107. Rudan I, Boschi-Pinto C, Biloglav Z, Mulholland K, Campbell H. Epidemiology and etiology of childhood pneumonia. Bull World Health Organ (2008) 86:408–16B. doi: 10.2471/BLT.07.048769
108. Jacobs DM, Ochs-Balcom HM, Zhao J, Murphy TF, Sethi S. Lower airway bacterial colonization patterns and species-specific interactions in chronic obstructive pulmonary disease. J Clin Microbiol (2018) 56(10):e00330–18. doi: 10.1128/JCM.00330-18
109. Patel I, Seemungal T, Wilks M, Lloyd-Owen S, Donaldson G, Wedzicha J. Relationship between bacterial colonisation and the frequency, character, and severity of COPD exacerbations. Thorax (2002) 57(9):759–64. doi: 10.1136/thorax.57.9.759
110. Sethi S, Murphy TF. Infection in the pathogenesis and course of chronic obstructive pulmonary disease. New Engl J Med (2008) 359(22):2355–65. doi: 10.1056/NEJMra0800353
111. Sethi S. Bacterial infection and the pathogenesis of COPD. Chest (2000) 117(5):286S–91S. doi: 10.1378/chest.117.5_suppl_1.286S
112. Hilty M, Burke C, Pedro H, Cardenas P, Bush A, Bossley C, et al. Disordered microbial communities in asthmatic airways. PloS One (2010) 5(1):e8578. doi: 10.1371/journal.pone.0008578
113. Charlson ES, Bittinger K, Haas AR, Fitzgerald AS, Frank I, Yadav A, et al. Topographical continuity of bacterial populations in the healthy human respiratory tract. Am J Respir Crit Care Med (2011) 184(8):957–63. doi: 10.1164/rccm.201104-0655OC
114. Erb-Downward JR, Thompson DL, Han MK, Freeman CM, McCloskey L, Schmidt LA, et al. Analysis of the lung microbiome in the “healthy” smoker and in COPD. PloS One (2011) 6(2):e16384. doi: 10.1371/journal.pone.0016384
115. Sethi S, Maloney J, Grove L, Wrona C, Berenson CS. Airway inflammation and bronchial bacterial colonization in chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2006) 173(9):991–8. doi: 10.1164/rccm.200509-1525OC
116. Garcia-Vidal C, Almagro P, Romaní V, Rodriguez-Carballeira M, Cuchi E, Canales L, et al. Pseudomonas aeruginosa in patients hospitalised for COPD exacerbation: a prospective study. Eur Respir J (2009) 34(5):1072–8. doi: 10.1183/09031936.00003309
117. Martinez-García MA, Rigau D, Barrecheguren M, García-Ortega A, Nuñez A, Yepez GO, et al. Long-term risk of mortality associated with isolation of pseudomonas aeruginosa in COPD: A systematic review and meta-analysis. Int J Chronic Obstructive Pulm Dis (2022) 17:371. doi: 10.2147/COPD.S346294
118. Stolk J, Rudolphus A, Davies P, Osinga D, Dijkman JH, Agarwal L, et al. Induction of emphysema and bronchial mucus cell hyperplasia by intratracheal instillation of lipopolysaccharide in the hamster. J pathol (1992) 167(3):349–56. doi: 10.1002/path.1711670314
119. Desai H, Eschberger K, Wrona C, Grove L, Agrawal A, Grant B, et al. Bacterial colonization increases daily symptoms in patients with chronic obstructive pulmonary disease. Ann Am Thorac Soc (2014) 11(3):303–9. doi: 10.1513/AnnalsATS.201310-350OC
120. Tumkaya M, Atis S, Ozge C, Delialioglu N, Polat G, Kanik A. Relationship between airway colonization, inflammation and exacerbation frequency in COPD. Respir Med (2007) 101(4):729–37. doi: 10.1016/j.rmed.2006.08.020
121. Zhang M, Li Q, Zhang X-Y, Ding X, Zhu D, Zhou X. Relevance of lower airway bacterial colonization, airway inflammation, and pulmonary function in the stable stage of chronic obstructive pulmonary disease. Eur J Clin Microbiol Infect dis (2010) 29(12):1487–93. doi: 10.1007/s10096-010-1027-7
122. Wilkinson TM, Patel IS, Wilks M, Donaldson GC, Wedzicha JA. Airway bacterial load and FEV1 decline in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2003) 167(8):1090–5. doi: 10.1164/rccm.200210-1179OC
123. Man WH, de Steenhuijsen Piters WA, Bogaert D. The microbiota of the respiratory tract: gatekeeper to respiratory health. Nat Rev Microbiol (2017) 15(5):259–70. doi: 10.1038/nrmicro.2017.14
124. Keck T, Lindemann J. Numerical simulation and nasal air-conditioning. GMS Curr topics otorhinolaryngol Head Neck surg (2010) 9. doi: 10.1097/00005537-200406000-00015
125. Ingenito E, Solway J, McFadden E, Pichurko B, Bowman H, Michaels D, et al. Indirect assessment of mucosal surface temperatures in the airways: theory and tests. J Appl Physiol (1987) 63(5):2075–83. doi: 10.1152/jappl.1987.63.5.2075
126. West JB. Regional differences in the lung. Chest (1978) 74(4):426–37. doi: 10.1016/S0012-3692(15)37392-X
127. Brunworth JD, Garg R, Mahboubi H, Johnson B, Djalilian HR. Detecting nasopharyngeal reflux: a novel pH probe technique. Ann Otol Rhinol Laryngol (2012) 121(7):427–30. doi: 10.1177/000348941212100701
128. Brown JH, Cook K, Ney FG, Hatch T. Influence of particle size upon the retention of particulate matter in the human lung. Am J Public Health Nations Health (1950) 40(4):450–80. doi: 10.2105/AJPH.40.4.450
129. Kline KA, Fälker S, Dahlberg S, Normark S, Henriques-Normark B. Bacterial adhesins in host-microbe interactions. Cell Host Microbe (2009) 5(6):580–92. doi: 10.1016/j.chom.2009.05.011
130. Wanner A, Salathé M, O'Riordan TG. Mucociliary clearance in the airways. Am J Respir Crit Care Med (1996) 154(6):1868–902. doi: 10.1164/ajrccm.154.6.8970383
131. Oh JE, Song E, Moriyama M, Wong P, Zhang S, Jiang R, et al. Intranasal priming induces local lung-resident b cell populations that secrete protective mucosal antiviral IgA. Sci Immunol (2021) 6(66):eabj5129. doi: 10.1126/sciimmunol.abj5129
132. Lamm ME, Nedrud JG, Kaetzel CS, Mazanec MB. IgA and mucosal defense. Apmis (1995) 103(1-6):241–6. doi: 10.1111/j.1699-0463.1995.tb01101.x
133. Bevins CL. (2007). Antimicrobial peptides: antimicrobial peptides: Ciba foundation symposium 186. In: Novartis Foundation Symposia. Wiley Online Library. doi: 10.1002/9780470514658.ch15
134. Leshem A, Liwinski T, Elinav E. Immune-microbiota interplay and colonization resistance in infection. Mol Cell (2020) 78(4):597–613. doi: 10.1016/j.molcel.2020.03.001
135. Momose Y, Hirayama K, Itoh K. Competition for proline between indigenous escherichia coli and e. coli O157: H7 in gnotobiotic mice associated with infant intestinal microbiota and its contribution to the colonization resistance against e. coli O157: H7. Antonie Van Leeuwenhoek (2008) 94(2):165–71. doi: 10.1007/s10482-008-9222-6
136. Curtis MM, Hu Z, Klimko C, Narayanan S, Deberardinis R, Sperandio V. The gut commensal bacteroides thetaiotaomicron exacerbates enteric infection through modification of the metabolic landscape. Cell Host Microbe (2014) 16(6):759–69. doi: 10.1016/j.chom.2014.11.005
137. Leatham MP, Banerjee S, Autieri SM, Mercado-Lubo R, Conway T, Cohen PS. Precolonized human commensal escherichia coli strains serve as a barrier to e. coli O157: H7 growth in the streptomycin-treated mouse intestine. Infect immun (2009) 77(7):2876–86. doi: 10.1128/IAI.00059-09
138. Cassat JE, Skaar EP. Iron in infection and immunity. Cell Host Microbe (2013) 13(5):509–19. doi: 10.1016/j.chom.2013.04.010
139. Deriu E, Liu JZ, Pezeshki M, Edwards RA, Ochoa RJ, Contreras H, et al. Probiotic bacteria reduce salmonella typhimurium intestinal colonization by competing for iron. Cell Host Microbe (2013) 14(1):26–37. doi: 10.1016/j.chom.2013.06.007
140. Cerasi M, Ammendola S, Battistoni A. Competition for zinc binding in the host-pathogen interaction. Front Cell infect Microbiol (2013) 3:108. doi: 10.3389/fcimb.2013.00108
141. Gielda LM, DiRita VJ. Zinc competition among the intestinal microbiota. MBio (2012) 3(4):e00171–12. doi: 10.1128/mBio.00171-12
142. Litvak Y, Mon KK, Nguyen H, Chanthavixay G, Liou M, Velazquez EM, et al. Commensal enterobacteriaceae protect against salmonella colonization through oxygen competition. Cell Host Microbe (2019) 25(1):128–39.e5. doi: 10.1016/j.chom.2018.12.003
143. Fonseca D, Hand T, Han S-J, Gerner M, Zaretsky A, Byrd A, et al. Microbiota-dependent sequelae of acute infection compromise tissue-specific immunity. Cell (2015) 163(2):354–66. doi: 10.1016/j.cell.2015.08.030
144. Zachar Z, Savage DC. Microbial interference and colonization of the murine gastrointestinal tract by listeria monocytogenes. Infect immun (1979) 23(1):168–74. doi: 10.1128/iai.23.1.168-174.1979
145. Van der Waaij D, Berghuis-de Vries J, Lekkerkerk-Van der Wees J. Colonization resistance of the digestive tract in conventional and antibiotic-treated mice. Epidemiol Infect (1971) 69(3):405–11. doi: 10.1017/S0022172400021653
146. Murphy TF, Brauer AL, Eschberger K, Lobbins P, Grove L, Cai X, et al. Pseudomonas aeruginosa in chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2008) 177(8):853–60. doi: 10.1164/rccm.200709-1413OC
147. Sethi S, Evans N, Grant BJ, Murphy TF. New strains of bacteria and exacerbations of chronic obstructive pulmonary disease. New Engl J Med (2002) 347(7):465–71. doi: 10.1056/NEJMoa012561
148. Jacoby P, Watson K, Bowman J, Taylor A, Riley TV, Smith DW, et al. Modelling the co-occurrence of streptococcus pneumoniae with other bacterial and viral pathogens in the upper respiratory tract. Vaccine (2007) 25(13):2458–64. doi: 10.1016/j.vaccine.2006.09.020
149. Madhi SA, Adrian P, Kuwanda L, Cutland C, Albrich WC, Klugman KP. Long-term effect of pneumococcal conjugate vaccine on nasopharyngeal colonization by streptococcus pneumoniae–and associated interactions with staphylococcus aureus and haemophilus influenzae colonization–in HIV-infected and HIV-uninfected children. J Infect dis (2007) 196(11):1662–6. doi: 10.1086/522164
150. Regev-Yochay G, Dagan R, Raz M, Carmeli Y, Shainberg B, Derazne E, et al. Association between carriage of streptococcus pneumoniae and staphylococcus aureus in children. Jama (2004) 292(6):716–20. doi: 10.1001/jama.292.6.716
151. Chien Y-W, Vidal JE, Grijalva CG, Bozio C, Edwards KM, Williams JV, et al. Density interactions between streptococcus pneumoniae, haemophilus influenzae and staphylococcus aureus in the nasopharynx of young Peruvian children. Pediatr Infect Dis J (2013) 32(1):72. doi: 10.1097/INF.0b013e318270d850
152. Kommineni S, Bretl DJ, Lam V, Chakraborty R, Hayward M, Simpson P, et al. Bacteriocin production augments niche competition by enterococci in the mammalian gastrointestinal tract. Nature (2015) 526(7575):719–22. doi: 10.1038/nature15524
153. Schamberger GP, Diez-Gonzalez F. Selection of recently isolated colicinogenic escherichia coli strains inhibitory to escherichia coli O157: H7. J Food protect (2002) 65(9):1381–7. doi: 10.4315/0362-028X-65.9.1381
154. Sassone-Corsi M, Nuccio S-P, Liu H, Hernandez D, Vu CT, Takahashi AA, et al. Microcins mediate competition among enterobacteriaceae in the inflamed gut. Nature (2016) 540(7632):280–3. doi: 10.1038/nature20557
155. Rea MC, Sit CS, Clayton E, O'Connor PM, Whittal RM, Zheng J, et al. A posttranslationally modified bacteriocin with a narrow spectrum of activity against clostridium difficile. Proc Natl Acad Sci (2010) 107(20):9352–7. doi: 10.1073/pnas.0913554107
156. Kim SG, Becattini S, Moody TU, Shliaha PV, Littmann ER, Seok R, et al. Microbiota-derived lantibiotic restores resistance against vancomycin-resistant enterococcus. Nature (2019) 572(7771):665–9. doi: 10.1038/s41586-019-1501-z
157. Zhao W, Caro F, Robins W, Mekalanos JJ. Antagonism toward the intestinal microbiota and its effect on vibrio cholerae virulence. Science (2018) 359(6372):210–3. doi: 10.1126/science.aap8775
158. Basler M, Mekalanos J. Type 6 secretion dynamics within and between bacterial cells. Science (2012) 337(6096):815. doi: 10.1126/science.1222901
159. Ross BD, Verster AJ, Radey MC, Schmidtke DT, Pope CE, Hoffman LR, et al. Human gut bacteria contain acquired interbacterial defence systems. Nature (2019) 575(7781):224–8. doi: 10.1038/s41586-019-1708-z
160. Kim WJ, Higashi D, Goytia M, Rendón MA, Pilligua-Lucas M, Bronnimann M, et al. Commensal neisseria kill neisseria gonorrhoeae through a DNA-dependent mechanism. Cell Host Microbe (2019) 26(2):228–39.e8. doi: 10.1016/j.chom.2019.07.003
161. Ahmad S, Wang B, Walker MD, Tran H-KR, Stogios PJ, Savchenko A, et al. An interbacterial toxin inhibits target cell growth by synthesizing (p) ppApp. Nature (2019) 575(7784):674–8. doi: 10.1038/s41586-019-1735-9
162. Chatzidaki-Livanis M, Geva-Zatorsky N, Comstock LE. Bacteroides fragilis type VI secretion systems use novel effector and immunity proteins to antagonize human gut bacteroidales species. Proc Natl Acad Sci (2016) 113(13):3627–32. doi: 10.1073/pnas.1522510113
163. Bhattacharya S, Baidya AK, Pal RR, Mamou G, Gatt YE, Margalit H, et al. A ubiquitous platform for bacterial nanotube biogenesis. Cell Rep (2019) 27(2):334–42.e10. doi: 10.1016/j.celrep.2019.02.055
164. Pal RR, Baidya AK, Mamou G, Bhattacharya S, Socol Y, Kobi S, et al. Pathogenic e. coli extracts nutrients from infected host cells utilizing injectisome components. Cell (2019) 177(3):683–96.e18. doi: 10.1016/j.cell.2019.02.022
165. Su Y-C, Jalalvand F, Thegerström J, Riesbeck K. The interplay between immune response and bacterial infection in COPD: focus upon non-typeable haemophilus influenzae. Front Immunol (2018) 9:2530. doi: 10.3389/fimmu.2018.02530
166. Garrett WS. Immune recognition of microbial metabolites. Nat Rev Immunol (2020) 20(2):91–2. doi: 10.1038/s41577-019-0252-2
167. Skelly AN, Sato Y, Kearney S, Honda K. Mining the microbiota for microbial and metabolite-based immunotherapies. Nat Rev Immunol (2019) 19(5):305–23. doi: 10.1038/s41577-019-0144-5
168. Macfarlane S, Macfarlane GT. Regulation of short-chain fatty acid production. Proc Nutr Soc (2003) 62(1):67–72. doi: 10.1079/PNS2002207
169. Derrien M, Vaughan EE, Plugge CM, de Vos WM. Akkermansia muciniphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int J syst evol Microbiol (2004) 54(5):1469–76. doi: 10.1099/ijs.0.02873-0
170. Louis P, Hold GL, Flint HJ. The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol (2014) 12(10):661–72. doi: 10.1038/nrmicro3344
171. Louis P, Young P, Holtrop G, Flint HJ. Diversity of human colonic butyrate-producing bacteria revealed by analysis of the butyryl-CoA: acetate CoA-transferase gene. Environ Microbiol (2010) 12(2):304–14. doi: 10.1111/j.1462-2920.2009.02066.x
172. Morrison DJ, Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes (2016) 7(3):189–200. doi: 10.1080/19490976.2015.1134082
173. Kasubuchi M, Hasegawa S, Hiramatsu T, Ichimura A, Kimura I. Dietary gut microbial metabolites, short-chain fatty acids, and host metabolic regulation. Nutrients (2015) 7(4):2839–49. doi: 10.3390/nu7042839
174. Colosimo DA, Kohn JA, Luo PM, Piscotta FJ, Han SM, Pickard AJ, et al. Mapping interactions of microbial metabolites with human G-protein-coupled receptors. Cell Host Microbe (2019) 26(2):273–82.e7. doi: 10.1016/j.chom.2019.07.002
175. Chen H, Nwe P-K, Yang Y, Rosen CE, Bielecka AA, Kuchroo M, et al. A forward chemical genetic screen reveals gut microbiota metabolites that modulate host physiology. Cell (2019) 177(5):1217–31.e18. doi: 10.1016/j.cell.2019.03.036
176. Brown AJ, Goldsworthy SM, Barnes AA, Eilert MM, Tcheang L, Daniels D, et al. The orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J Biol Chem (2003) 278(13):11312–9. doi: 10.1074/jbc.M211609200
177. Nilsson NE, Kotarsky K, Owman C, Olde B. Identification of a free fatty acid receptor, FFA2R, expressed on leukocytes and activated by short-chain fatty acids. Biochem Biophys Res Commun (2003) 303(4):1047–52. doi: 10.1016/S0006-291X(03)00488-1
178. Thangaraju M, Cresci GA, Liu K, Ananth S, Gnanaprakasam JP, Browning DD, et al. GPR109A is a g-protein–coupled receptor for the bacterial fermentation product butyrate and functions as a tumor suppressor in colon. Cancer Res (2009) 69(7):2826–32. doi: 10.1158/0008-5472.CAN-08-4466
179. Husted AS, Trauelsen M, Rudenko O, Hjorth SA, Schwartz TW. GPCR-mediated signaling of metabolites. Cell Metab (2017) 25(4):777–96. doi: 10.1016/j.cmet.2017.03.008
180. Morita N, Umemoto E, Fujita S, Hayashi A, Kikuta J, Kimura I, et al. GPR31-dependent dendrite protrusion of intestinal CX3CR1+ cells by bacterial metabolites. Nature (2019) 566(7742):110–4. doi: 10.1038/s41586-019-0884-1
181. Thorburn AN, Macia L, Mackay CR. Diet, metabolites, and “western-lifestyle” inflammatory diseases. Immunity (2014) 40(6):833–42. doi: 10.1016/j.immuni.2014.05.014
182. Li M, van Esch BC, Wagenaar GT, Garssen J, Folkerts G, Henricks PA. Pro-and anti-inflammatory effects of short chain fatty acids on immune and endothelial cells. Eur J Pharmacol (2018) 831:52–9. doi: 10.1016/j.ejphar.2018.05.003
183. Gao H, Sun Y, Wu Y, Luan B, Wang Y, Qu B, et al. Identification of β-arrestin2 as a G protein-coupled receptor-stimulated regulator of NF-κB pathways. Mol Cell (2004) 14(3):303–17. doi: 10.1016/S1097-2765(04)00216-3
184. Kim MH, Kang SG, Park JH, Yanagisawa M, Kim CH. Short-chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice. Gastroenterology (2013) 145(2):396–406. e10. doi: 10.1053/j.gastro.2013.04.056
185. Gaudier E, Jarry A, Blottiere H, De Coppet P, Buisine M, Aubert J, et al. Butyrate specifically modulates MUC gene expression in intestinal epithelial goblet cells deprived of glucose. Am J Physiology-Gastrointest Liver Physiol (2004) 287(6):G1168–G74. doi: 10.1152/ajpgi.00219.2004
186. Willemsen L, Koetsier M, Van Deventer S, Van Tol E. Short chain fatty acids stimulate epithelial mucin 2 expression through differential effects on prostaglandin E1 and E2 production by intestinal myofibroblasts. Gut (2003) 52(10):1442–7. doi: 10.1136/gut.52.10.1442
187. Hooper LV, Macpherson AJ. Immune adaptations that maintain homeostasis with the intestinal microbiota. Nat Rev Immunol (2010) 10(3):159–69. doi: 10.1038/nri2710
188. McGuckin MA, Lindén SK, Sutton P, Florin TH. Mucin dynamics and enteric pathogens. Nat Rev Microbiol (2011) 9(4):265–78. doi: 10.1038/nrmicro2538
189. Kim M, Qie Y, Park J, Kim CH. Gut microbial metabolites fuel host antibody responses. Cell Host Microbe (2016) 20(2):202–14. doi: 10.1016/j.chom.2016.07.001
190. Wu W, Sun M, Chen F, Cao AT, Liu H, Zhao Y, et al. Microbiota metabolite short-chain fatty acid acetate promotes intestinal IgA response to microbiota which is mediated by GPR43. Mucosal Immunol (2017) 10(4):946–56. doi: 10.1038/mi.2016.114
191. Takeuchi T, Miyauchi E, Kanaya T, Kato T, Nakanishi Y, Watanabe T, et al. Acetate differentially regulates IgA reactivity to commensal bacteria. Nature (2021) 595(7868):560–4. doi: 10.1038/s41586-021-03727-5
192. Natividad JM, Hayes CL, Motta J-P, Jury J, Galipeau HJ, Philip V, et al. Differential induction of antimicrobial REGIII by the intestinal microbiota and bifidobacterium breve NCC2950. Appl Environ Microbiol (2013) 79(24):7745–54. doi: 10.1128/AEM.02470-13
193. Macia L, Tan J, Vieira AT, Leach K, Stanley D, Luong S, et al. Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat Commun (2015) 6(1):1–15. doi: 10.1038/ncomms7734
194. Dang AT, Marsland BJ. Microbes, metabolites, and the gut–lung axis. Mucosal Immunol (2019) 12(4):843–50. doi: 10.1038/s41385-019-0160-6
195. Netea MG, van der Meer JW. Trained immunity: an ancient way of remembering. Cell Host Microbe (2017) 21(3):297–300. doi: 10.1016/j.chom.2017.02.003
196. Netea MG, Domínguez-Andrés J, Barreiro LB, Chavakis T, Divangahi M, Fuchs E, et al. Defining trained immunity and its role in health and disease. Nat Rev Immunol (2020) 20(6):375–88. doi: 10.1038/s41577-020-0285-6
197. Schulthess J, Pandey S, Capitani M, Rue-Albrecht KC, Arnold I, Franchini F, et al. The short chain fatty acid butyrate imprints an antimicrobial program in macrophages. Immunity (2019) 50(2):432–45.e7. doi: 10.1016/j.immuni.2018.12.018
198. Watt R, Parkin K, Martino D. The potential effects of short-chain fatty acids on the epigenetic regulation of innate immune memory. Challenges (2020) 11(2):25. doi: 10.3390/challe11020025
199. Bekkering S, Domínguez-Andrés J, Joosten LA, Riksen NP, Netea MG. Trained immunity: reprogramming innate immunity in health and disease. Annu Rev Immunol (2021) 39:667–93. doi: 10.1146/annurev-immunol-102119-073855
200. Trompette A, Gollwitzer ES, Yadava K, Sichelstiel AK, Sprenger N, Ngom-Bru C, et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat Med (2014) 20(2):159–66. doi: 10.1038/nm.3444
201. Landsman L, Jung S. Lung macrophages serve as obligatory intermediate between blood monocytes and alveolar macrophages. J Immunol (2007) 179(6):3488–94. doi: 10.4049/jimmunol.179.6.3488
202. Landsman L, Varol C, Jung S. Distinct differentiation potential of blood monocyte subsets in the lung. J Immunol (2007) 178(4):2000–7. doi: 10.4049/jimmunol.178.4.2000
203. Geissmann F, Manz MG, Jung S, Sieweke MH, Merad M, Ley K. Development of monocytes, macrophages, and dendritic cells. Science (2010) 327(5966):656–61. doi: 10.1126/science.1178331
204. Fogg DK, Sibon C, Miled C, Jung S, Aucouturier P, Littman DR, et al. A clonogenic bone marrow progenitor specific for macrophages and dendritic cells. Science (2006) 311(5757):83–7. doi: 10.1126/science.1117729
205. Liu K, Victora GD, Schwickert TA, Guermonprez P, Meredith MM, Yao K, et al. In vivo analysis of dendritic cell development and homeostasis. Science (2009) 324(5925):392–7. doi: 10.1126/science.1170540
206. Trompette A, Gollwitzer ES, Pattaroni C, Lopez-Mejia IC, Riva E, Pernot J, et al. Dietary fiber confers protection against flu by shaping Ly6c– patrolling monocyte hematopoiesis and CD8+ T cell metabolism. Immunity (2018) 48(5):992–1005.e8. doi: 10.1016/j.immuni.2018.04.022
207. Labonte AC, Tosello-Trampont A-C, Hahn YS. The role of macrophage polarization in infectious and inflammatory diseases. Mol Cells (2014) 37(4):275. doi: 10.14348/molcells.2014.2374
208. Shirey KA, Pletneva LM, Puche AC, Keegan AD, Prince GA, Blanco JC, et al. Control of RSV-induced lung injury by alternatively activated macrophages is IL-4Rα-, TLR4-, and IFN-β-dependent. Mucosal Immunol (2010) 3(3):291–300. doi: 10.1038/mi.2010.6
209. Yazar A, Atis S, Konca K, Pata C, Akbay E, Calikoglu M, et al. Respiratory symptoms and pulmonary functional changes in patients with irritable bowel syndrome. Am J gastroenterol (2001) 96(5):1511–6. doi: 10.1111/j.1572-0241.2001.03748.x
210. Keely S, Talley NJ, Hansbro PM. Pulmonary-intestinal cross-talk in mucosal inflammatory disease. Mucosal Immunol (2012) 5(1):7–18. doi: 10.1038/mi.2011.55
211. Rutten EP, Lenaerts K, Buurman WA, Wouters EF. Disturbed intestinal integrity in patients with COPD: effects of activities of daily living. Chest (2014) 145(2):245–52. doi: 10.1378/chest.13-0584
212. Dai Z-L, Wu G, Zhu W-Y. Amino acid metabolism in intestinal bacteria: links between gut ecology and host health. Front Biosci-Landmark (2011) 16(5):1768–86. doi: 10.2741/3820
213. Zelante T, Iannitti RG, Cunha C, De Luca A, Giovannini G, Pieraccini G, et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity (2013) 39(2):372–85. doi: 10.1016/j.immuni.2013.08.003
214. Vinolo MA, Rodrigues HG, Hatanaka E, Sato FT, Sampaio SC, Curi R. Suppressive effect of short-chain fatty acids on production of proinflammatory mediators by neutrophils. J Nutr Biochem (2011) 22(9):849–55. doi: 10.1016/j.jnutbio.2010.07.009
215. Usami M, Kishimoto K, Ohata A, Miyoshi M, Aoyama M, Fueda Y, et al. Butyrate and trichostatin a attenuate nuclear factor κB activation and tumor necrosis factor α secretion and increase prostaglandin E2 secretion in human peripheral blood mononuclear cells. Nutr Res (2008) 28(5):321–8. doi: 10.1016/j.nutres.2008.02.012
216. Venkatesh M, Mukherjee S, Wang H, Li H, Sun K, Benechet AP, et al. Symbiotic bacterial metabolites regulate gastrointestinal barrier function via the xenobiotic sensor PXR and toll-like receptor 4. Immunity (2014) 41(2):296–310. doi: 10.1016/j.immuni.2014.06.014
217. Miller CA. Expression of the human aryl hydrocarbon receptor complex in yeast: activation of transcription by indole compounds. J Biol Chem (1997) 272(52):32824–9. doi: 10.1074/jbc.272.52.32824
218. Qiu J, Heller JJ, Guo X, Zong-ming EC, Fish K, Fu Y-X, et al. The aryl hydrocarbon receptor regulates gut immunity through modulation of innate lymphoid cells. Immunity (2012) 36(1):92–104. doi: 10.1016/j.immuni.2011.11.011
219. Kawamata Y, Fujii R, Hosoya M, Harada M, Yoshida H, Miwa M, et al. AG protein-coupled receptor responsive to bile acids. J Biol Chem (2003) 278(11):9435–40. doi: 10.1074/jbc.M209706200
220. Maruyama T, Miyamoto Y, Nakamura T, Tamai Y, Okada H, Sugiyama E, et al. Identification of membrane-type receptor for bile acids (M-BAR). Biochem Biophys Res Commun (2002) 298(5):714–9. doi: 10.1016/S0006-291X(02)02550-0
221. Zhang Z, Li J, Zheng W, Zhao G, Zhang H, Wang X, et al. Peripheral lymphoid volume expansion and maintenance are controlled by gut microbiota via RALDH+ dendritic cells. Immunity (2016) 44(2):330–42. doi: 10.1016/j.immuni.2016.01.004
222. Kiss EA, Vonarbourg C, Kopfmann S, Hobeika E, Finke D, Esser C, et al. Natural aryl hydrocarbon receptor ligands control organogenesis of intestinal lymphoid follicles. Science (2011) 334(6062):1561–5. doi: 10.1126/science.1214914
223. Li Y, Innocentin S, Withers DR, Roberts NA, Gallagher AR, Grigorieva EF, et al. Exogenous stimuli maintain intraepithelial lymphocytes via aryl hydrocarbon receptor activation. Cell (2011) 147(3):629–40. doi: 10.1016/j.cell.2011.09.025
224. Lee JS, Cella M, McDonald KG, Garlanda C, Kennedy GD, Nukaya M, et al. AHR drives the development of gut ILC22 cells and postnatal lymphoid tissues via pathways dependent on and independent of notch. Nat Immunol (2012) 13(2):144–51. doi: 10.1038/ni.2187
225. Madapoosi SS, Cruickshank-Quinn C, Opron K, Erb-Downward JR, Begley LA, Li G, et al. Lung microbiota and metabolites collectively associate with clinical outcomes in milder stage chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2022) 206(4):427–39. doi: 10.1164/rccm.202110-2241OC
226. Marsland BJ, Trompette A, Gollwitzer ES. The gut–lung axis in respiratory disease. Ann Am Thorac Soc (2015) 12(Supplement 2):S150–S6. doi: 10.1513/AnnalsATS.201503-133AW
227. Budden KF, Gellatly SL, Wood DL, Cooper MA, Morrison M, Hugenholtz P, et al. Emerging pathogenic links between microbiota and the gut–lung axis. Nat Rev Microbiol (2017) 15(1):55–63. doi: 10.1038/nrmicro.2016.142
228. Enaud R, Prevel R, Ciarlo E, Beaufils F, Wieërs G, Guery B, et al. The gut-lung axis in health and respiratory diseases: a place for inter-organ and inter-kingdom crosstalks. Front Cell Infect Microbiol (2020) 10:9. doi: 10.3389/fcimb.2020.00009
229. Dumas A, Bernard L, Poquet Y, Lugo-Villarino G, Neyrolles O. The role of the lung microbiota and the gut–lung axis in respiratory infectious diseases. Cell Microbiol (2018) 20(12):e12966. doi: 10.1111/cmi.12966
230. Bradley CP, Teng F, Felix KM, Sano T, Naskar D, Block KE, et al. Segmented filamentous bacteria provoke lung autoimmunity by inducing gut-lung axis Th17 cells expressing dual TCRs. Cell Host Microbe (2017) 22(5):697–704.e4. doi: 10.1016/j.chom.2017.10.007
231. Vaughan A, Frazer ZA, Hansbro PM, Yang IA. COPD and the gut-lung axis: the therapeutic potential of fibre. J Thorac dis (2019) 11(Suppl 17):S2173. doi: 10.21037/jtd.2019.10.40
232. He Y, Wen Q, Yao F, Xu D, Huang Y, Wang J. Gut–lung axis: the microbial contributions and clinical implications. Crit Rev Microbiol (2017) 43(1):81–95. doi: 10.1080/1040841X.2016.1176988
233. Baccala R, Gonzalez-Quintial R, Lawson BR, Stern ME, Kono DH, Beutler B, et al. Sensors of the innate immune system: their mode of action. Nat Rev Rheumatol (2009) 5(8):448–56. doi: 10.1038/nrrheum.2009.136
234. Sandor F, Buc M. Toll-like receptors. i. structure, function and their ligands. Folia biol (2005) 51(5):148.
235. Cook DN, Pisetsky DS, Schwartz DA. Toll-like receptors in the pathogenesis of human disease. Nat Immunol (2004) 5(10):975–9. doi: 10.1038/ni1116
236. Nguyen LT, Haney EF, Vogel HJ. The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol (2011) 29(9):464–72. doi: 10.1016/j.tibtech.2011.05.001
237. Caballero S, Kim S, Carter RA, Leiner IM, Sušac B, Miller L, et al. Cooperating commensals restore colonization resistance to vancomycin-resistant enterococcus faecium. Cell Host Microbe (2017) 21(5):592–602.e4. doi: 10.1016/j.chom.2017.04.002
238. Papatriantafyllou M. Inflammasome shapes the microbiota. Nat Rev Immunol (2011) 11(7):439. doi: 10.1038/nri3012
239. Hara H, Seregin SS, Yang D, Fukase K, Chamaillard M, Alnemri ES, et al. The NLRP6 inflammasome recognizes lipoteichoic acid and regulates gram-positive pathogen infection. Cell (2018) 175(6):1651–64.e14. doi: 10.1016/j.cell.2018.09.047
240. Gonçalves P, Di Santo JP. An intestinal inflammasome–the ILC3–cytokine tango. Trends Mol Med (2016) 22(4):269–71. doi: 10.1016/j.molmed.2016.02.008
241. Erttmann SF, Swacha P, Aung KM, Brindefalk B, Jiang H, Härtlova A, et al. The gut microbiota prime systemic antiviral immunity via the cGAS-STING-IFN-I axis. Immunity (2022) 55(5):847–61.e10. doi: 10.1016/j.immuni.2022.04.006
242. Kunisawa J, Nochi T, Kiyono H. Immunological commonalities and distinctions between airway and digestive immunity. Trends Immunol (2008) 29(11):505–13. doi: 10.1016/j.it.2008.07.008
243. Evans SE, Scott BL, Clement CG, Larson DT, Kontoyiannis D, Lewis RE, et al. Stimulated innate resistance of lung epithelium protects mice broadly against bacteria and fungi. Am J Respir Cell Mol Biol (2010) 42(1):40–50. doi: 10.1165/rcmb.2008-0260OC
244. Read RC, Wilson R, Rutman A, Lund V, Todd HC, Brain AP, et al. Interaction of nontypable haemophilus influenzae with human respiratory mucosa in vitro. J Infect Dis (1991) 163(3):549–58. doi: 10.1093/infdis/163.3.549
245. Gum J. Mucin genes and the proteins they encode: structure, diversity, and regulation. Am J Respir Cell Mol Biol (1992) 7:557. doi: 10.1165/ajrcmb/7.6.557
246. Thai P, Loukoianov A, Wachi S, Wu R. Regulation of airway mucin gene expression. Annu Rev Physiol (2008) 70:405. doi: 10.1146/annurev.physiol.70.113006.100441
247. Bergeron C, Boulet L-P. Structural changes in airway diseases: characteristics, mechanisms, consequences, and pharmacologic modulation. Chest (2006) 129(4):1068–87. doi: 10.1378/chest.129.4.1068
248. Rose MC, Voynow JA. Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiol Rev (2006) 86(1):245–78. doi: 10.1152/physrev.00010.2005
249. Thornton DJ, Gray T, Nettesheim P, Howard M, Koo JS, Sheehan JK. Characterization of mucins from cultured normal human tracheobronchial epithelial cells. Am J physiol-Lung Cell Mol Physiol (2000) 278(6):L1118–L28. doi: 10.1152/ajplung.2000.278.6.L1118
250. Rousseau K, Byrne C, Kim YS, Gum JR, Swallow DM, Toribara NW. The complete genomic organization of the human MUC6 and MUC2 mucin genes. Genomics (2004) 83(5):936–9. doi: 10.1016/j.ygeno.2003.11.003
251. Li J-D, Feng W, Gallup M, Kim J-H, Gum J, Kim Y, et al. Activation of NF-κB via a src-dependent ras-MAPK-pp90rsk pathway is required for pseudomonas aeruginosa-induced mucin overproduction in epithelial cells. Proc Natl Acad Sci (1998) 95(10):5718–23. doi: 10.1073/pnas.95.10.5718
252. Li JD, Feng W, Gallup M, Kim JH, Gum J, Kim Y, et al. Activation of NF-kappaB via a src-dependent ras-MAPK-pp90rsk pathway is required for pseudomonas aeruginosa-induced mucin overproduction in epithelial cells. Proc Natl Acad Sci U S A. (1998) 95(10):5718–23. doi: 10.1073/pnas.95.10.5718
253. Lemjabbar H, Basbaum C. Platelet-activating factor receptor and ADAM10 mediate responses to staphylococcus aureus in epithelial cells. Nat Med (2002) 8(1):41–6. doi: 10.1038/nm0102-41
254. Wrzosek L, Miquel S, Noordine M-L, Bouet S, Chevalier-Curt MJ, Robert V, et al. Bacteroides thetaiotaomicron and faecalibacterium prausnitziiinfluence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol (2013) 11(1):1–13. doi: 10.1186/1741-7007-11-61
255. Kelly CJ, Zheng L, Campbell EL, Saeedi B, Scholz CC, Bayless AJ, et al. Crosstalk between microbiota-derived short-chain fatty acids and intestinal epithelial HIF augments tissue barrier function. Cell Host Microbe (2015) 17(5):662–71. doi: 10.1016/j.chom.2015.03.005
256. Zheng L, Kelly CJ, Battista KD, Schaefer R, Lanis JM, Alexeev EE, et al. Microbial-derived butyrate promotes epithelial barrier function through IL-10 receptor–dependent repression of claudin-2. J Immunol (2017) 199(8):2976–84. doi: 10.4049/jimmunol.1700105
257. Ramos FL, Krahnke JS, Kim V. Clinical issues of mucus accumulation in COPD. Int J chronic obstructive pulm dis (2014) 9:139. doi: 10.2147/COPD.S38938
258. Burgel P, Martin C. Mucus hypersecretion in COPD: should we only rely on symptoms? Eur Respir Rev (2010) 19(116):94–6. doi: 10.1183/09059180.00004410
259. Kim V, Han MK, Vance GB, Make BJ, Newell JD, Hokanson JE, et al. The chronic bronchitic phenotype of COPD: an analysis of the COPDGene study. Chest (2011) 140(3):626–33. doi: 10.1378/chest.10-2948
260. Adler KB, Hendley DD, Davis GS. Bacteria associated with obstructive pulmonary disease elaborate extracellular products that stimulate mucin secretion by explants of guinea pig airways. Am J pathol (1986) 125(3):501.
261. Leikauf GD, Borchers MT, Prows DR, Simpson LG. Mucin apoprotein expression in COPD. Chest (2002) 121(5):166S–82S. doi: 10.1378/chest.121.5_suppl.166S
262. Turner J, Jones CE. Regulation of mucin expression in respiratory diseases. Biochem Soc Trans (2009) 37(4):877–81. doi: 10.1042/BST0370877
263. Caramori G, Di Gregorio C, Carlstedt I, Casolari P, Guzzinati I, Adcock I, et al. Mucin expression in peripheral airways of patients with chronic obstructive pulmonary disease. Histopathology (2004) 45(5):477–84. doi: 10.1111/j.1365-2559.2004.01952.x
264. Lu W, Zheng J. The function of mucins in the COPD airway. Curr Respir Care Rep (2013) 2(3):155–66. doi: 10.1007/s13665-013-0051-3
265. Caramori G, Casolari P, Di Gregorio C, Saetta M, Baraldo S, Boschetto P, et al. MUC5AC expression is increased in bronchial submucosal glands of stable COPD patients. Histopathology (2009) 55(3):321–31. doi: 10.1111/j.1365-2559.2009.03377.x
266. Lo Bello F, Ieni A, Hansbro PM, Ruggeri P, Di Stefano A, Nucera F, et al. Role of the mucins in pathogenesis of COPD: implications for therapy. Expert Rev Respir Med (2020) 14(5):465–83. doi: 10.1080/17476348.2020.1739525
267. Ma J, Rubin BK, Voynow JA. Mucins, mucus, and goblet cells. Chest (2018) 154(1):169–76. doi: 10.1016/j.chest.2017.11.008
268. Zheng Z, Qi Y, Xu X, Jiang H, Li Z, Yang Q, et al. Sputum mucin 1 is increased during the acute phase of chronic obstructive pulmonary disease exacerbation. J Thorac dis (2017) 9(7):1873. doi: 10.21037/jtd.2017.06.63
269. Voynow JA, Gendler SJ, Rose MC. Regulation of mucin genes in chronic inflammatory airway diseases. Am J Respir Cell Mol Biol (2006) 34(6):661–5. doi: 10.1165/rcmb.2006-0035SF
270. Radicioni G, Ceppe A, Ford AA, Alexis NE, Barr RG, Bleecker ER, et al. Airway mucin MUC5AC and MUC5B concentrations and the initiation and progression of chronic obstructive pulmonary disease: an analysis of the SPIROMICS cohort. Lancet Respir Med (2021) 9(11):1241–54. doi: 10.1016/S2213-2600(21)00079-5
271. Lange P, Nyboe J, Appleyard M, Jensen G, Schnohr P. Relation of ventilatory impairment and of chronic mucus hypersecretion to mortality from obstructive lung disease and from all causes. Thorax (1990) 45(8):579–85. doi: 10.1136/thx.45.8.579
272. Rogers DF. Mucociliary dysfunction in COPD: effect of current pharmacotherapeutic options. Pulm Pharmacol Ther (2005) 18(1):1–8. doi: 10.1016/j.pupt.2004.08.001
273. Ito JT, Ramos D, Lima FF, Rodrigues FM, Gomes PR, Moreira GL, et al. Nasal mucociliary clearance in subjects with COPD after smoking cessation. Respir Care (2015) 60(3):399–405. doi: 10.4187/respcare.03266
274. Bennett WD, Henderson AG, Ceppe A, Zeman KL, Wu J, Gladman C, et al. Effect of hypertonic saline on mucociliary clearance and clinical outcomes in chronic bronchitis. ERJ Open Res (2020) 6(3). doi: 10.1183/23120541.00269-2020
275. Smaldone GC, Foster WM, O'Riordan TG, Messina MS, Perry RJ, Langenback EG. Regional impairment of mucociliary clearance in chronic obstructive pulmonary disease. Chest (1993) 103(5):1390–6. doi: 10.1378/chest.103.5.1390
276. Bhowmik A, Chahal K, Austin G, Chakravorty I. Improving mucociliary clearance in chronic obstructive pulmonary disease. Respir Med (2009) 103(4):496–502. doi: 10.1016/j.rmed.2008.10.014
277. Nawroth JC, van der Does AM, Ryan A, Kanso E. Multiscale mechanics of mucociliary clearance in the lung. Philos Trans R Soc B (2020) 375(1792):20190160. doi: 10.1098/rstb.2019.0160
278. Whitsett JA. Airway epithelial differentiation and mucociliary clearance. Ann Am Thorac Soc (2018) 15(Supplement 3):S143–S8. doi: 10.1513/AnnalsATS.201802-128AW
279. Hancock RE, Haney EF, Gill EE. The immunology of host defence peptides: beyond antimicrobial activity. Nat Rev Immunol (2016) 16(5):321–34. doi: 10.1038/nri.2016.29
280. Hancock RE, Sahl H-G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol (2006) 24(12):1551–7. doi: 10.1038/nbt1267
281. Fjell CD, Hiss JA, Hancock RE, Schneider G. Designing antimicrobial peptides: form follows function. Nat Rev Drug disc (2012) 11(1):37–51. doi: 10.1038/nrd3591
282. Brogden KA. Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat Rev Microbiol (2005) 3(3):238–50. doi: 10.1038/nrmicro1098
283. Ganz T. Defensins: antimicrobial peptides of innate immunity. Nat Rev Immunol (2003) 3(9):710–20. doi: 10.1038/nri1180
284. Fabisiak A, Murawska N, Fichna J. LL-37: Cathelicidin-related antimicrobial peptide with pleiotropic activity. Pharmacol Rep (2016) 68(4):802–8. doi: 10.1016/j.pharep.2016.03.015
285. Sun E, Belanger CR, Haney EF, Hancock RE. Host defense (antimicrobial) peptides. Pept Appl biomed Biotechnol bioeng (2018), 253–85. doi: 10.1016/B978-0-08-100736-5.00010-7
286. Zhang Y, Jiang Y, Sun C, Wang Q, Yang Z, Pan X, et al. The human cathelicidin LL-37 enhances airway mucus production in chronic obstructive pulmonary disease. Biochem Biophys Res Commun (2014) 443(1):103–9. doi: 10.1016/j.bbrc.2013.11.074
287. Zuyderduyn S, Ninaber DK, Hiemstra PS, Rabe KF. The antimicrobial peptide LL-37 enhances IL-8 release by human airway smooth muscle cells. J Allergy Clin Immunol (2006) 117(6):1328–35. doi: 10.1016/j.jaci.2006.03.022
288. Cantor JO, Shteyngart B, Cerreta JM, Turino GM. The effect of lysozyme on elastase-mediated injury. Exp Biol Med (2002) 227(2):108–13. doi: 10.1177/153537020222700205
289. Parameswaran GI, Sethi S, Murphy TF. Effects of bacterial infection on airway antimicrobial peptides and proteins in COPD. Chest (2011) 140(3):611–7. doi: 10.1378/chest.10-2760
290. Ohbayashi H, Setoguchi Y, Fukuchi Y, Shibata K, Sakata Y, Arai T. Pharmacological effect of lysozyme for COPD and bronchial asthma with sputum: A randomized, placebo-controlled, cross-over study. Eur Respir J (2014) 44(Suppl 58).
291. Cohen AB, Chenoweth DE, Hugli TE. The release of elastase, myeloperoxidase, and lysozyme from human alveolar macrophages. Am Rev Respir Dis (1982) 126(2):241–7.
292. Parameswaran G, Sethi S, Murphy T. Antimicrobial polypeptides and moraxella catarrhalis in COPD. B50 LUNG HOST DEFENSES TO INFECTION AND HOST CELL PATHOGEN INTERACTION. Am Thorac Soc (2009), A3225. doi: 10.1164/ajrccm-conference.2009.179.1_MeetingAbstracts.A3225
293. Rytilä P, Rehn T, Ilumets H, Rouhos A, Sovijärvi A, Myllärniemi M, et al. Increased oxidative stress in asymptomatic current chronic smokers and GOLD stage 0 COPD. Respir Res (2006) 7(1):1–10. doi: 10.1186/1465-9921-7-69
294. Culpitt SV, de Matos C, Russell RE, Donnelly LE, Rogers DF, Barnes PJ. Effect of theophylline on induced sputum inflammatory indices and neutrophil chemotaxis in chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2002) 165(10):1371–6. doi: 10.1164/rccm.2105106
295. Gompertz S, Bayley D, Hill S, Stockley R. Relationship between airway inflammation and the frequency of exacerbations in patients with smoking related COPD. Thorax (2001) 56(1):36–41. doi: 10.1136/thorax.56.1.36
296. Persson LJ, Aanerud M, Hardie JA, Nilsen RM, Bakke PS, Eagan TM, et al. Antimicrobial peptide levels are linked to airway inflammation, bacterial colonisation and exacerbations in chronic obstructive pulmonary disease. Eur Respir J (2017) 49(3). doi: 10.1183/13993003.01328-2016
297. Cane J, Tregidgo L, Thulborn S, Finch D, Bafadhel M. Antimicrobial peptides SLPI and beta defensin-1 in sputum are negatively correlated with FEV1. Int J Chronic Obstructive Pulm Dis (2021) 16:1437. doi: 10.2147/COPD.S301622
298. Taggart CC, Lowe GJ, Greene CM, Mulgrew AT, O'Neill SJ, Levine RL, et al. Cathepsin b, l, and s cleave and inactivate secretory leucoprotease inhibitor. J Biol Chem (2001) 276(36):33345–52. doi: 10.1074/jbc.M103220200
299. Zheng T, Zhu Z, Wang Z, Homer RJ, Ma B, Riese RJ, et al. Inducible targeting of IL-13 to the adult lung causes matrix metalloproteinase–and cathepsin-dependent emphysema. J Clin Invest (2000) 106(9):1081–93. doi: 10.1172/JCI10458
300. Zheng T, Kang MJ, Crothers K, Zhu Z, Liu W, Lee CG, et al. Role of cathepsin s-dependent epithelial cell apoptosis in IFN-γ-induced alveolar remodeling and pulmonary emphysema. J Immunol (2005) 174(12):8106–15. doi: 10.4049/jimmunol.174.12.8106
301. Barnes PJ, Shapiro SD, Pauwels R. Chronic obstructive pulmonary disease: molecular and cellularmechanisms. Eur Respir J (2003) 22(4):672–88. doi: 10.1183/09031936.03.00040703
302. Nakajima T, Nakamura H, Owen CA, Yoshida S, Tsuduki K, Chubachi S, et al. Plasma cathepsin s and cathepsin s/cystatin c ratios are potential biomarkers for COPD. Dis Markers (2016) 2016. doi: 10.1155/2016/4093870
303. Andrault P-M, Schamberger AC, Chazeirat T, Sizaret D, Renault J, Staab-Weijnitz CA, et al. Cigarette smoke induces overexpression of active human cathepsin s in lungs from current smokers with or without COPD. Am J Physiol-Lung Cell Mol Physiol (2019) 317(5):L625–L38. doi: 10.1152/ajplung.00061.2019
304. Ngan DA, Vickerman SV, Granville DJ, Man SP, Sin DD. The possible role of granzyme b in the pathogenesis of chronic obstructive pulmonary disease. Ther Adv Respir Dis (2009) 3(3):113–29. doi: 10.1177/1753465809341965
305. Hodge S, Hodge G, Nairn J, Holmes M, Reynolds PN. Increased airway granzyme b and perforin in current and ex-smoking COPD subjects. COPD: J Chronic Obstructive Pulm Dis (2006) 3(4):179–87. doi: 10.1080/15412550600976868
306. Vernooy JH, Moüller GM, van Suylen RJ, van Spijk MP, Cloots RH, Hoet PH, et al. Increased granzyme a expression in type II pneumocytes of patients with severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2007) 175(5):464–72. doi: 10.1164/rccm.200602-169OC
307. Golec M, Reichel C, Mackiewicz B, Skórska C, Curzytek K, Lemieszek M, et al. Cathelicidin LL-37, granzymes, TGF-beta1 and cytokines levels in induced sputum from farmers with and without COPD. Ann Agric Environ Med (2009) 16(2):289–97.
308. Morissette MC, Parent J, Milot J. Perforin, granzyme b, and FasL expression by peripheral blood T lymphocytes in emphysema. Respir Res (2007) 8(1):1–9. doi: 10.1186/1465-9921-8-62
309. Kim WD, Chi HS, Choe KH, Oh YM, Lee SD, Kim KR, et al. A possible role for CD 8+ and non-CD 8+ cell granzyme b in early small airway wall remodelling in centrilobular emphysema. Respirology (2013) 18(4):688–96. doi: 10.1111/resp.12069
310. Sattar Z, Lora A, Jundi B, Railwah C, Geraghty P. The S100 protein family as players and therapeutic targets in pulmonary diseases. Pulm Med (2021) 2021. doi: 10.1155/2021/5488591
311. Lin C-R, Bahmed K, Criner GJ, Marchetti N, Tuder RM, Kelsen S, et al. S100A8 protects human primary alveolar type II cells against injury and emphysema. Am J Respir Cell Mol Biol (2019) 60(3):299–307. doi: 10.1165/rcmb.2018-0144OC
312. Kim RY, Sunkara KP, Bracke KR, Jarnicki AG, Donovan C, Hsu AC, et al. A microRNA-21–mediated SATB1/S100A9/NF-κB axis promotes chronic obstructive pulmonary disease pathogenesis. Sci Trans Med (2021) 13(621):eaav7223. doi: 10.1126/scitranslmed.aav7223
313. Lorenz E, Muhlebach MS, Tessier PA, Alexis NE, Hite RD, Seeds MC, et al. Different expression ratio of S100A8/A9 and S100A12 in acute and chronic lung diseases. Respir Med (2008) 102(4):567–73. doi: 10.1016/j.rmed.2007.11.011
314. Railwah C, Lora A, Zahid K, Goldenberg H, Campos M, Wyman A, et al. Cigarette smoke induction of S100A9 contributes to chronic obstructive pulmonary disease. Am J Physiol-Lung Cell Mol Physiol (2020) 319(6):L1021–L35. doi: 10.1152/ajplung.00207.2020
315. Pouwels SD, Heijink IH, ten Hacken NH, Vandenabeele P, Krysko DV, Nawijn MC, et al. DAMPs activating innate and adaptive immune responses in COPD. Mucosal Immunol (2014) 7(2):215–26. doi: 10.1038/mi.2013.77
316. Skronska-Wasek W, Durlanik S, Le HQ, Schroeder V, Kitt K, Garnett JP, et al. The antimicrobial peptide S100A8/A9 produced by airway epithelium functions as a potent and direct regulator of macrophage phenotype and function. Eur Respir J (2022) 59(4). doi: 10.1183/13993003.02732-2020
317. Andresen E, Lange C, Strodthoff D, Goldmann T, Fischer N, Sahly H, et al. S100A7/psoriasin expression in the human lung: unchanged in patients with COPD, but upregulated upon positive s. aureusdetection. BMC pulm Med (2011) 11(1):1–10. doi: 10.1186/1471-2466-11-10
318. Vandamme D, Landuyt B, Luyten W, Schoofs L. A comprehensive summary of LL-37, the factotum human cathelicidin peptide. Cell Immunol (2012) 280(1):22–35. doi: 10.1016/j.cellimm.2012.11.009
319. Nijnik A, Hancock RE. The roles of cathelicidin LL-37 in immune defences and novel clinical applications. Curr Opin hematol (2009) 16(1):41–7. doi: 10.1097/MOH.0b013e32831ac517
320. Xhindoli D, Pacor S, Benincasa M, Scocchi M, Gennaro R, Tossi A. The human cathelicidin LL-37–a pore-forming antibacterial peptide and host-cell modulator. Biochim Biophys Acta (BBA)-Biomem (2016) 1858(3):546–66. doi: 10.1016/j.bbamem.2015.11.003
321. Schneider VA, Coorens M, Ordonez SR, Tjeerdsma-van Bokhoven JL, Posthuma G, van Dijk A, et al. Imaging the antimicrobial mechanism (s) of cathelicidin-2. Sci Rep (2016) 6(1):1–11. doi: 10.1038/srep32948
322. Ramanathan B, Davis EG, Ross CR, Blecha F. Cathelicidins: microbicidal activity, mechanisms of action, and roles in innate immunity. Microbes infec (2002) 4(3):361–72. doi: 10.1016/S1286-4579(02)01549-6
323. Yu J, Mookherjee N, Wee K, Bowdish DM, Pistolic J, Li Y, et al. Host defense peptide LL-37, in synergy with inflammatory mediator IL-1β, augments immune responses by multiple pathways. J Immunol (2007) 179(11):7684–91. doi: 10.4049/jimmunol.179.11.7684
324. Peric M, Koglin S, Kim S-M, Morizane S, Besch R, Prinz JC, et al. IL-17A enhances vitamin D3-induced expression of cathelicidin antimicrobial peptide in human keratinocytes. J Immunol (2008) 181(12):8504–12. doi: 10.4049/jimmunol.181.12.8504
325. Minns D, Smith KJ, Alessandrini V, Hardisty G, Melrose L, Jackson-Jones L, et al. The neutrophil antimicrobial peptide cathelicidin promotes Th17 differentiation. Nat Commun (2021) 12(1):1–16. doi: 10.1038/s41467-021-21533-5
326. Mookherjee N, Hamill P, Gardy J, Blimkie D, Falsafi R, Chikatamarla A, et al. Systems biology evaluation of immune responses induced by human host defence peptide LL-37 in mononuclear cells. Mol BioSyst (2009) 5(5):483–96. doi: 10.1039/b813787k
327. Shaykhiev R, Sierigk J, Herr C, Krasteva G, Kummer W, Bals R. The antimicrobial peptide cathelicidin enhances activation of lung epithelial cells by LPS. FASEB J (2010) 24(12):4756–66. doi: 10.1096/fj.09.151332
328. Kandler K, Shaykhiev R, Kleemann P, Klescz F, Lohoff M, Vogelmeier C, et al. The anti-microbial peptide LL-37 inhibits the activation of dendritic cells by TLR ligands. Int Immunol (2006) 18(12):1729–36. doi: 10.1093/intimm/dxl107
329. Khoo A-L, Chai LY, Koenen HJ, Oosting M, Steinmeyer A, Zuegel U, et al. Vitamin D3 down-regulates proinflammatory cytokine response to mycobacterium tuberculosis through pattern recognition receptors while inducing protective cathelicidin production. Cytokine (2011) 55(2):294–300. doi: 10.1016/j.cyto.2011.04.016
330. van der Does AM, Beekhuizen H, Ravensbergen B, Vos T, Ottenhoff TH, van Dissel JT, et al. LL-37 directs macrophage differentiation toward macrophages with a proinflammatory signature. J Immunol (2010) 185(3):1442–9. doi: 10.4049/jimmunol.1000376
331. Davidson DJ, Currie AJ, Reid GS, Bowdish DM, MacDonald KL, Ma RC, et al. The cationic antimicrobial peptide LL-37 modulates dendritic cell differentiation and dendritic cell-induced T cell polarization. J Immunol (2004) 172(2):1146–56. doi: 10.4049/jimmunol.172.2.1146
332. Elssner A, Duncan M, Gavrilin M, Wewers MD. A novel P2X7 receptor activator, the human cathelicidin-derived peptide LL37, induces IL-1β processing and release. J Immunol (2004) 172(8):4987–94. doi: 10.4049/jimmunol.172.8.4987
333. Wang T-T, Nestel FP, Bourdeau V, Nagai Y, Wang Q, Liao J, et al. Cutting edge: 1, 25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression. J Immunol (2004) 173(5):2909–12. doi: 10.4049/jimmunol.173.5.2909
334. Hilchie AL, Wuerth K, Hancock RE. Immune modulation by multifaceted cationic host defense (antimicrobial) peptides. Nat Chem Biol (2013) 9(12):761–8. doi: 10.1038/nchembio.1393
335. Peyssonnaux C, Boutin AT, Zinkernagel AS, Datta V, Nizet V, Johnson RS. Critical role of HIF-1α in keratinocyte defense against bacterial infection. J Invest Dermatol (2008) 128(8):1964–8. doi: 10.1038/jid.2008.27
336. Knudsen KS, Lehmann S, Nielsen R, Tangedal S, Haaland I, Hiemstra PS, et al. The lower airways microbiome and antimicrobial peptides in idiopathic pulmonary fibrosis differ from chronic obstructive pulmonary disease. PloS One (2022) 17(1):e0262082. doi: 10.1371/journal.pone.0262082
337. Cane J, Thulborn S, Piper S, Finch D, Bafadhel M. Antimicrobial peptides in inflammatory phenotypes of COPD. Thorax (2017). doi: 10.1136/thoraxjnl-2017-210983.255
338. Persson L, Aanerud M, Hardie JA, Bakke PS, Eagan TM, Hiemstra PS. Associations between haemophilus influenzae and levels of sputum antimicrobial peptides in COPD. Eur Respir Soc (2016) 48(Suppl_2). doi: 10.1183/13993003.congress-2016.OA4994
339. Seiler F, Bals R, Beisswenger C. Function of antimicrobial peptides in lung innate immunity. Antimicrobial Peptides (2016), 33–52. doi: 10.1007/978-3-319-24199-9_3
340. Neumann A, Berends ET, Nerlich A, Molhoek EM, Gallo RL, Meerloo T, et al. The antimicrobial peptide LL-37 facilitates the formation of neutrophil extracellular traps. Biochem J (2014) 464(1):3–11. doi: 10.1042/BJ20140778
341. Tjabringa GS, Ninaber DK, Drijfhout JW, Rabe KF, Hiemstra PS. Human cathelicidin LL-37 is a chemoattractant for eosinophils and neutrophils that acts via formyl-peptide receptors. Int Arch Allergy Immunol (2006) 140(2):103–12. doi: 10.1159/000092305
342. van der Does AM, Hiemstra PS, Mookherjee N. Antimicrobial host defence peptides: immunomodulatory functions and translational prospects. Antimicrobial Peptides (2019), 149–71. doi: 10.1007/978-981-13-3588-4_10
343. Huang JT-J, Cant E, Keir HR, Barton AK, Kuzmanova E, Shuttleworth M, et al. Endotyping COPD, bronchiectasis and the ‘COPD-bronchiectasis association’. Am J Respir Crit Care Med (2022) 206(4). doi: 10.1164/rccm.202108-1943OC
344. van Wetering S, Tjabringa GS, Hiemstra PS. Interactions between neutrophil-derived antimicrobial peptides and airway epithelial cells. J leukoc Biol (2005) 77(4):444–50. doi: 10.1189/jlb.0604367
345. Herr C, AlAlwani S, Eschmann R, Beißwenger C, Bals R. The antimicrobial peptide CRAMP protects against emphysema. Eur Respir Soc; (2012) 40(56).
346. Zhu M, Wang T, Wang C, Ji Y. The association between vitamin d and COPD risk, severity, and exacerbation: an updated systematic review and meta-analysis. Int J chronic obstructive pulm dis (2016) 11:2597. doi: 10.2147/COPD.S101382
347. Sibila O, Perea L, Cantó E, Shoemark A, Cassidy D, Smith AH, et al. Antimicrobial peptides, disease severity and exacerbations in bronchiectasis. Thorax (2019) 74(9):835–42. doi: 10.1136/thoraxjnl-2018-212895
348. Burkes RM, Ceppe AS, Couper DJ, Comellas AP, Wells JM, Peters SP, et al. Plasma cathelicidin is independently associated with reduced lung function in COPD: analysis of the subpopulations and intermediate outcome measures in COPD study cohort. Chronic Obstructive Pulm Dis: J COPD Found (2020) 7(4):370. doi: 10.15326/jcopdf.7.4.2020.0142
349. Golec M, Lemieszek MK, Dutkiewicz J, Milanowski J, Barteit S. A scoping analysis of cathelicidin in response to organic dust exposure and related chronic lung illnesses. Int J Mol Sci (2022) 23(16):8847. doi: 10.3390/ijms23168847
350. Lambert AA, Kirk GD, Astemborski J, Neptune ER, Mehta SH, Wise RA, et al. A cross sectional analysis of the role of the antimicrobial peptide cathelicidin in lung function impairment within the ALIVE cohort. PloS One (2014) 9(4):e95099. doi: 10.1371/journal.pone.0095099
351. Burkes RM, Astemborski J, Lambert AA, Brown TT, Wise RA, Kirk GD, et al. Plasma cathelicidin and longitudinal lung function in current and former smokers. PloS One (2019) 14(2):e0212628. doi: 10.1371/journal.pone.0212628
352. Singanayagam A, Glanville N, Cuthbertson L, Bartlett NW, Finney LJ, Turek E, et al. Inhaled corticosteroid suppression of cathelicidin drives dysbiosis and bacterial infection in chronic obstructive pulmonary disease. Sci Trans Med (2019) 11(507):eaav3879. doi: 10.1126/scitranslmed.aav3879
353. Alalwani MS. Cathelicidin has a protective role against pulmonary emphysema. Eur Respir J (2014) 44(Suppl 58).
354. Golec M, Reichel C, Lemieszek M, Mackiewicz B, Buczkowski J, Sitkowska J, et al. Cathelicidin LL-37 in bronchoalveolar lavage and epithelial lining fluids from COPD patients and healthy individuals. J Biol regulators homeostatic agents (2012) 26(4):617–25.
355. Xiao W, Hsu Y-P, Ishizaka A, Kirikae T, Moss RB. Sputum cathelicidin, urokinase plasminogen activation system components, and cytokines discriminate cystic fibrosis, COPD, and asthma inflammation. Chest (2005) 128(4):2316–26. doi: 10.1378/chest.128.4.2316
356. Keir HR, Richardson H, Gilmour A, De Lima DA, Veluchamy A, Hennayake C, et al. The cathelicidin LL-37 and microbial dysbiosis in COPD patients receiving inhaled corticosteroids. Eur Respir Soc (2022) 8(8). doi: 10.1183/23120541.LSC-2022.185
357. Burkes R, Ceppe A, Donohue J, Doerschuk C, Drummond M. Plasma cathelicidin measurements during COPD exacerbations and outcomes. C32 COPD: TRANSLATIONAL AND MECHANISTIC STUDIES. Am Thorac Soc (2019), A4530–A. doi: 10.1164/ajrccm-conference.2019.199.1_MeetingAbstracts.A4530
359. Jiang Y-Y, Xiao W, Zhu M-X, Yang Z-H, Pan X-J, Zhang Y, et al. The effect of human antibacterial peptide LL-37 in the pathogenesis of chronic obstructive pulmonary disease. Respir Med (2012) 106(12):1680–9. doi: 10.1016/j.rmed.2012.08.018
360. Tatsuta M, Kan-o K, Ishii Y, Yamamoto N, Ogawa T, Fukuyama S, et al. Effects of cigarette smoke on barrier function and tight junction proteins in the bronchial epithelium: protective role of cathelicidin LL-37. Respir Res (2019) 20(1):1–14. doi: 10.1186/s12931-019-1226-4
361. Yang Y-M, Guo Y-F, Zhang H-S, Sun T-Y. Antimicrobial peptide LL-37 circulating levels in chronic obstructive pulmonary disease patients with high risk of frequent exacerbations. J Thorac dis (2015) 7(4):740. doi: 10.3978/j.issn.2072-1439.2015.04.33
362. Lehouck A, Mathieu C, Carremans C, Baeke F, Verhaegen J, Van Eldere J, et al. High doses of vitamin d to reduce exacerbations in chronic obstructive pulmonary disease: a randomized trial. Ann Internal Med (2012) 156(2):105–14. doi: 10.7326/0003-4819-156-2-201201170-00004
363. Mathyssen C, Aelbrecht C, Serré J, Everaerts S, Maes K, Gayan-Ramirez G, et al. Local expression profiles of vitamin d-related genes in airways of COPD patients. Respir Res (2020) 21(1):1–10. doi: 10.1186/s12931-020-01405-0
364. Kokturk N, Baha A, Oh YM, Young Ju J, Jones PW. Vitamin d deficiency: What does it mean for chronic obstructive pulmonary disease (COPD)? a compherensive review for pulmonologists. Clin Respir J (2018) 12(2):382–97. doi: 10.1111/crj.12588
365. Soeroto AY, Setiawan D, Asriputri NN, Darmawan G, Laurus G, Santoso P. Association between vitamin d levels and FEV1, number of exacerbations, and CAT score in stable COPD patients in Indonesia. Int J Gen Med (2021) 14:7293. doi: 10.2147/IJGM.S333039
366. Uysal P, Simsek G, Durmus S, SÖZER V, Aksan H, Yurt S, et al. Evaluation of plasma antimicrobial peptide LL-37 and nuclear factor-kappaB levels in stable chronic obstructive pulmonary disease. Int J Chronic Obstructive Pulm Dis (2019) 14. doi: 10.2147/COPD.S185602
367. Burkes R, Ceppe A, O'Neal W, Han M, Curtis J, Comellas A, et al. Low plasma cathelicidin levels are associated with reduced lung function and prior COPD exacerbations: An analysis of the SPIROMICS cohort. Am J Resp Med (AJRCCM) (2018) 197:A7442–A.
368. Hazlett L, Wu M. Defensins in innate immunity. Cell Tissue Res (2011) 343(1):175–88. doi: 10.1007/s00441-010-1022-4
369. Tang Y-Q, Selsted M. Characterization of the disulfide motif in BNBD-12, an antimicrobial beta-defensin peptide from bovine neutrophils. J Biol Chem (1993) 268(9):6649–53. doi: 10.1016/S0021-9258(18)53299-3
370. Semple F, Dorin JR. β-defensins: multifunctional modulators of infection, inflammation and more? J innate Immun (2012) 4(4):337–48. doi: 10.1159/000336619
371. Harder Jr, Bartels J, Christophers E, Schroüder J-M. Isolation and characterization of human μ-Defensin-3, a novel human inducible peptide antibiotic. J Biol Chem (2001) 276(8):5707–13. doi: 10.1074/jbc.M008557200
372. García J-RC, Krause A, Schulz S, Rodríguez-Jiménez F-J, Klüver E, Adermann K, et al. Human β-defensin 4: a novel inducible peptide with a specific salt-sensitive spectrum of antimicrobial activity. FASEB J (2001) 15(10):1819–21. doi: 10.1096/fj.00-0865fje
373. Soehnlein O, Kai-Larsen Y, Frithiof R, Sorensen OE, Kenne E, Scharffetter-Kochanek K, et al. Neutrophil primary granule proteins HBP and HNP1–3 boost bacterial phagocytosis by human and murine macrophages. J Clin Invest (2008) 118(10):3491–502. doi: 10.1172/JCI35740
374. Miles K, Clarke DJ, Lu W, Sibinska Z, Beaumont PE, Davidson DJ, et al. Dying and necrotic neutrophils are anti-inflammatory secondary to the release of α-defensins. J Immunol (2009) 183(3):2122–32. doi: 10.4049/jimmunol.0804187
375. Hiemstra P, Van Wetering S, Stolk J. Neutrophil serine proteinases and defensins in chronic obstructive pulmonary disease: effects on pulmonary epithelium. Eur Respir J (1998) 12(5):1200–8. doi: 10.1183/09031936.98.12051200
376. Matsushita I, Hasegawa K, Nakata K, Yasuda K, Tokunaga K, Keicho N. Genetic variants of human β-defensin-1 and chronic obstructive pulmonary disease. Biochem Biophys Res Commun (2002) 291(1):17–22. doi: 10.1006/bbrc.2002.6395
377. Pace E, Ferraro M, Minervini MI, Vitulo P, Pipitone L, Chiappara G, et al. Beta defensin-2 is reduced in central but not in distal airways of smoker COPD patients. PloS One (2012) 7(3):e33601. doi: 10.1371/journal.pone.0033601
378. Andresen E, Günther G, Bullwinkel J, Lange C, Heine H. Increased expression of beta-defensin 1 (DEFB1) in chronic obstructive pulmonary disease. PloS One (2011) 6(7):e21898. doi: 10.1371/journal.pone.0021898
379. Liao Z, Dong J, Hu X, Wang T, Wan C, Li L, et al. Enhanced expression of human β-defensin 2 in peripheral lungs of patients with chronic obstructive pulmonary disease. Peptides (2012) 38(2):350–6. doi: 10.1016/j.peptides.2012.09.013
380. Wallace AM, He J-Q, Burkett KM, Ruan J, Connett JE, Anthonisen NR, et al. Contribution of alpha-and beta-defensins to lung function decline and infection in smokers: an association study. Respir Res (2006) 7(1):1–8. doi: 10.1186/1465-9921-7-76
381. Pierson T, Learmonth-Pierson S, Pinto D, van Hoek ML. Cigarette smoke extract induces differential expression levels of beta-defensin peptides in human alveolar epithelial cells. Tobacco induced dis (2013) 11(1):1–8. doi: 10.1186/1617-9625-11-10
382. Schutte BC, McCray PB Jr. (Beta)-defensins in lung host defense. Annu Rev Physiol (2002) 64:709. doi: 10.1146/annurev.physiol.64.081501.134340
383. Chen CI-U, Schaller-Bals S, Paul KP, Wahn U, Bals R. β-defensins and LL-37 in bronchoalveolar lavage fluid of patients with cystic fibrosis. J Cystic Fibrosis (2004) 3(1):45–50. doi: 10.1016/j.jcf.2003.12.008
384. Hiratsuka T, Mukae H, Iiboshi H, Ashitani J, Nabeshima K, Minematsu T, et al. Increased concentrations of human β-defensins in plasma and bronchoalveolar lavage fluid of patients with diffuse panbronchiolitis. Thorax (2003) 58(5):425–30. doi: 10.1136/thorax.58.5.425
385. Bdeir K, Higazi AA-R, Kulikovskaya I, Christofidou-Solomidou M, Vinogradov SA, Allen TC, et al. Neutrophil α-defensins cause lung injury by disrupting the capillary–epithelial barrier. Am J Respir Crit Care Med (2010) 181(9):935–46. doi: 10.1164/rccm.200907-1128OC
386. Cheng M, Chen Y, Wang L, Chen W, Yang L, Shen G, et al. Commensal microbiota maintains alveolar macrophages with a low level of CCL24 production to generate anti-metastatic tumor activity. Sci Rep (2017) 7(1):1–12. doi: 10.1038/s41598-017-08264-8
387. Brown RL, Sequeira RP, Clarke TB. The microbiota protects against respiratory infection via GM-CSF signaling. Nat Commun (2017) 8(1):1–11. doi: 10.1038/s41467-017-01803-x
388. Khan N, Mendonca L, Dhariwal A, Fontes G, Menzies D, Xia J, et al. Intestinal dysbiosis compromises alveolar macrophage immunity to mycobacterium tuberculosis. Mucosal Immunol (2019) 12(3):772–83. doi: 10.1038/s41385-019-0147-3
389. Schuijt TJ, Lankelma JM, Scicluna BP, e Melo F, Roelofs JJ, de Boer JD, et al. The gut microbiota plays a protective role in the host defence against pneumococcal pneumonia. Gut (2016) 65(4):575–83. doi: 10.1136/gutjnl-2015-309728
390. Cohen SB, Gern BH, Delahaye JL, Adams KN, Plumlee CR, Winkler JK, et al. Alveolar macrophages provide an early mycobacterium tuberculosis niche and initiate dissemination. Cell Host Microbe (2018) 24(3):439–46.e4. doi: 10.1016/j.chom.2018.08.001
391. Gutierrez P, Closa D, Piñer R, Bulbena O, Menéndez R, Torres A. Macrophage activation in exacerbated COPD with and without community-acquired pneumonia. Eur Respir J (2010) 36(2):285–91. doi: 10.1183/09031936.00118909
392. Shaykhiev R, Krause A, Salit J, Strulovici-Barel Y, Harvey B-G, O'Connor TP, et al. Smoking-dependent reprogramming of alveolar macrophage polarization: implication for pathogenesis of chronic obstructive pulmonary disease. J Immunol (2009) 183(4):2867–83. doi: 10.4049/jimmunol.0900473
393. Gharib SA, Manicone AM, Parks WC. Matrix metalloproteinases in emphysema. Matrix Biol (2018) 73:34–51. doi: 10.1016/j.matbio.2018.01.018
394. Churg A, Zhou S, Wright JL. Matrix metalloproteinases in COPD. Eur Respir J (2012) 39(1):197–209. doi: 10.1183/09031936.00121611
395. Tetley TD. Macrophages and the pathogenesis of COPD. Chest (2002) 121(5):156S–9S. doi: 10.1378/chest.121.5_suppl.156S
397. Zhang D, Frenette PS. Cross talk between neutrophils and the microbiota. Blood J Am Soc Hematol (2019) 133(20):2168–77. doi: 10.1182/blood-2018-11-844555
398. Zhang D, Chen G, Manwani D, Mortha A, Xu C, Faith JJ, et al. Neutrophil ageing is regulated by the microbiome. Nature (2015) 525(7570):528–32. doi: 10.1038/nature15367
399. Fagundes CT, Amaral FA, Vieira AT, Soares AC, Pinho V, Nicoli JR, et al. Transient TLR activation restores inflammatory response and ability to control pulmonary bacterial infection in germfree mice. J Immunol (2012) 188(3):1411–20. doi: 10.4049/jimmunol.1101682
400. Karmarkar D, Rock KL. Microbiota signalling through MyD88 is necessary for a systemic neutrophilic inflammatory response. Immunology (2013) 140(4):483–92. doi: 10.1111/imm.12159
401. Abi Abdallah DS, Egan CE, Butcher BA, Denkers EY. Mouse neutrophils are professional antigen-presenting cells programmed to instruct Th1 and Th17 T-cell differentiation. Int Immunol (2011) 23(5):317–26. doi: 10.1093/intimm/dxr007
402. Deshmukh HS, Liu Y, Menkiti OR, Mei J, Dai N, O'leary CE, et al. The microbiota regulates neutrophil homeostasis and host resistance to escherichia coli K1 sepsis in neonatal mice. Nat Med (2014) 20(5):524–30. doi: 10.1038/nm.3542
403. Amatngalim GD, Schrumpf JA, Henic A, Dronkers E, Verhoosel RM, Ordonez SR, et al. Antibacterial defense of human airway epithelial cells from chronic obstructive pulmonary disease patients induced by acute exposure to nontypeable haemophilus influenzae: modulation by cigarette smoke. J innate immun (2017) 9(4):359–74. doi: 10.1159/000455193
404. Gómez MI, Prince A. Airway epithelial cell signaling in response to bacterial pathogens. Pediatr pulmonol (2008) 43(1):11–9. doi: 10.1002/ppul.20735
405. Liu X, Yin S, Chen Y, Wu Y, Zheng W, Dong H, et al. LPS−induced proinflammatory cytokine expression in human airway epithelial cells and macrophages via NF−κB, STAT3 or AP−1 activation. Mol Med Rep (2018) 17(4):5484–91. doi: 10.3892/mmr.2018.8542
406. Bertelsen A, Elborn JS, Schock BC. Microbial interaction: Prevotella spp. reduce p. aeruginosa induced inflammation in cystic fibrosis bronchial epithelial cells. J Cystic Fibrosis (2021) 20(4):682–91. doi: 10.1016/j.jcf.2021.04.012
407. Obermayer A, Stoiber W, Krautgartner W-D, Klappacher M, Kofler B, Steinbacher P, et al. New aspects on the structure of neutrophil extracellular traps from chronic obstructive pulmonary disease and in vitro generation. PloS One (2014) 9(5):e97784. doi: 10.1371/journal.pone.0097784
408. Grabcanovic-Musija F, Obermayer A, Stoiber W, Krautgartner W-D, Steinbacher P, Winterberg N, et al. Neutrophil extracellular trap (NET) formation characterises stable and exacerbated COPD and correlates with airflow limitation. Respir Res (2015) 16(1):1–12. doi: 10.1186/s12931-015-0221-7
409. Pedersen F, Waschki B, Marwitz S, Goldmann T, Kirsten A, Malmgren A, et al. Neutrophil extracellular trap formation is regulated by CXCR2 in COPD neutrophils. Eur Respir J (2018) 51(4). doi: 10.1183/13993003.00970-2017
410. Pandey KC, De S, Mishra PK. Role of proteases in chronic obstructive pulmonary disease. Front Pharmacol (2017) 8:512. doi: 10.3389/fphar.2017.00512
411. Stockley RA. Neutrophils and the pathogenesis of COPD. Chest (2002) 121(5):151S–5S. doi: 10.1378/chest.121.5_suppl.151S
412. Churg A, Wright JL. Proteases and emphysema. Curr Opin pulm Med (2005) 11(2):153–9. doi: 10.1097/01.mcp.0000149592.51761.e3
413. Inui T, Watanabe M, Nakamoto K, Sada M, Hirata A, Nakamura M, et al. Bronchial epithelial cells produce CXCL1 in response to LPS and TNFα: A potential role in the pathogenesis of COPD. Exp Lung Res (2018) 44(7):323–31. doi: 10.1080/01902148.2018.1520936
414. Barnes PJ. Immunology of asthma and chronic obstructive pulmonary disease. Nat Rev Immunol (2008) 8(3):183–92. doi: 10.1038/nri2254
415. Traves SL, Donnelly LE. Th17 cells in airway diseases. Curr Mol Med (2008) 8(5):416–26. doi: 10.2174/156652408785160998
416. Seo G-Y, Giles DA, Kronenberg M. The role of innate lymphoid cells in response to microbes at mucosal surfaces. Mucosal Immunol (2020) 13(3):399–412. doi: 10.1038/s41385-020-0265-y
417. Sonnenberg GF, Monticelli LA, Alenghat T, Fung TC, Hutnick NA, Kunisawa J, et al. Innate lymphoid cells promote anatomical containment of lymphoid-resident commensal bacteria. Science (2012) 336(6086):1321–5. doi: 10.1126/science.1222551
418. Gury-BenAri M, Thaiss CA, Serafini N, Winter DR, Giladi A, Lara-Astiaso D, et al. The spectrum and regulatory landscape of intestinal innate lymphoid cells are shaped by the microbiome. Cell (2016) 166(5):1231–46.e13. doi: 10.1016/j.cell.2016.07.043
419. Hepworth MR, Monticelli LA, Fung TC, Ziegler CG, Grunberg S, Sinha R, et al. Innate lymphoid cells regulate CD4+ T-cell responses to intestinal commensal bacteria. Nature (2013) 498(7452):113–7. doi: 10.1038/nature12240
420. Hepworth MR, Fung TC, Masur SH, Kelsen JR, McConnell FM, Dubrot J, et al. Group 3 innate lymphoid cells mediate intestinal selection of commensal bacteria–specific CD4+ T cells. Science (2015) 348(6238):1031–5. doi: 10.1126/science.aaa4812
421. Mortha A, Chudnovskiy A, Hashimoto D, Bogunovic M, Spencer SP, Belkaid Y, et al. Microbiota-dependent crosstalk between macrophages and ILC3 promotes intestinal homeostasis. Science (2014) 343(6178):1249288. doi: 10.1126/science.1249288
422. Kruglov AA, Grivennikov SI, Kuprash DV, Winsauer C, Prepens S, Seleznik GM, et al. Nonredundant function of soluble LTα3 produced by innate lymphoid cells in intestinal homeostasis. Science (2013) 342(6163):1243–6. doi: 10.1126/science.1243364
423. Goto Y, Obata T, Kunisawa J, Sato S, Ivanov II, Lamichhane A, et al. Innate lymphoid cells regulate intestinal epithelial cell glycosylation. Science (2014) 345(6202):1254009. doi: 10.1126/science.1254009
424. Le Rouzic O, Pichavant M, Frealle E, Guillon A, Si-Tahar M, Gosset P. Th17 cytokines: novel potential therapeutic targets for COPD pathogenesis and exacerbations. Eur Respir J (2017) 50(4). doi: 10.1183/13993003.02434-2016
425. Ebbo M, Crinier A, Vély F, Vivier E. Innate lymphoid cells: major players in inflammatory diseases. Nat Rev Immunol (2017) 17(11):665–78. doi: 10.1038/nri.2017.86
426. Ardain A, Porterfield JZ, Kløverpris HN, Leslie A. Type 3 ILCs in lung disease. Front Immunol (2019) 10:92. doi: 10.3389/fimmu.2019.00092
427. Linden A, Hoshino H, Laan M. Airway neutrophils and interleukin-17. Eur Respir J (2000) 15(5):973–7. doi: 10.1034/j.1399-3003.2000.15e28.x
428. Di Stefano A, Caramori G, Gnemmi I, Contoli M, Vicari C, Capelli A, et al. T Helper type 17-related cytokine expression is increased in the bronchial mucosa of stable chronic obstructive pulmonary disease patients. Clin Exp Immunol (2009) 157(2):316–24. doi: 10.1111/j.1365-2249.2009.03965.x
429. Suzuki M, Sze MA, Campbell JD, Brothers JF, Lenburg ME, McDonough JE, et al. The cellular and molecular determinants of emphysematous destruction in COPD. Sci Rep (2017) 7(1):1–9. doi: 10.1038/s41598-017-10126-2
430. von Garnier C, Filgueira L, Wikstrom M, Smith M, Thomas JA, Strickland DH, et al. Anatomical location determines the distribution and function of dendritic cells and other APCs in the respiratory tract. J Immunol (2005) 175(3):1609–18. doi: 10.4049/jimmunol.175.3.1609
431. Iwasaki A. Mucosal dendritic cells. Annu Rev Immunol (2007) 25:381–418. doi: 10.1146/annurev.immunol.25.022106.141634
432. Jahnsen FL, Strickland DH, Thomas JA, Tobagus IT, Napoli S, Zosky GR, et al. Accelerated antigen sampling and transport by airway mucosal dendritic cells following inhalation of a bacterial stimulus. J Immunol (2006) 177(9):5861–7. doi: 10.4049/jimmunol.177.9.5861
433. Veres TZ, Voedisch S, Spies E, Tschernig T, Braun A. Spatiotemporal and functional behavior of airway dendritic cells visualized by two-photon microscopy. Am J pathol (2011) 179(2):603–9. doi: 10.1016/j.ajpath.2011.04.039
434. Rate A, Upham JW, Bosco A, McKenna KL, Holt PG. Airway epithelial cells regulate the functional phenotype of locally differentiating dendritic cells: implications for the pathogenesis of infectious and allergic airway disease. J Immunol (2009) 182(1):72–83. doi: 10.4049/jimmunol.182.1.72
435. Kim D-Y, Sato A, Fukuyama S, Sagara H, Nagatake T, Kong IG, et al. The airway antigen sampling system: respiratory m cells as an alternative gateway for inhaled antigens. J Immunol (2011) 186(7):4253–62. doi: 10.4049/jimmunol.0903794
436. Shaykhiev R, Bals R. Interactions between epithelial cells and leukocytes in immunity and tissue homeostasis. J leukoc Biol (2007) 82(1):1–15. doi: 10.1189/jlb.0207096
437. Soloff AC, Barratt-Boyes SM. Enemy at the gates: dendritic cells and immunity to mucosal pathogens. Cell Res (2010) 20(8):872–85. doi: 10.1038/cr.2010.94
438. Holt PG, Strickland DH, Wikström ME, Jahnsen FL. Regulation of immunological homeostasis in the respiratory tract. Nat Rev Immunol (2008) 8(2):142–52. doi: 10.1038/nri2236
439. Hammad H, Lambrecht BN. Recent progress in the biology of airway dendritic cells and implications for understanding the regulation of asthmatic inflammation. J Allergy Clin Immunol (2006) 118(2):331–6. doi: 10.1016/j.jaci.2006.03.041
440. Whiteson K, Agrawal S, Agrawal A. Differential responses of human dendritic cells to metabolites from the oral/airway microbiome. Clin Exp Immunol (2017) 188(3):371–9. doi: 10.1111/cei.12943
441. Ezzati Givi M, Redegeld F A, Folkerts G, Mortaz E. Dendritic cells in pathogenesis of COPD. Curr Pharm design (2012) 18(16):2329–35. doi: 10.2174/138161212800166068
442. Tsoumakidou M, Demedts IK, Brusselle GG, Jeffery PK. Dendritic cells in chronic obstructive pulmonary disease: new players in an old game. Am J Respir Crit Care Med (2008) 177(11):1180–6. doi: 10.1164/rccm.200711-1727PP
443. Van Pottelberge GR, Bracke KR, Joos GF, Brusselle GG. The role of dendritic cells in the pathogenesis of COPD: liaison officers in the front line. COPD: J Chronic Obstructive Pulm Dis (2009) 6(4):284–90. doi: 10.1080/15412550903049124
444. Vassallo R, Walters PR, Lamont J, Kottom TJ, Yi ES, Limper AH. Cigarette smoke promotes dendritic cell accumulation in COPD; a lung tissue research consortium study. Respir Res (2010) 11(1):1–13. doi: 10.1186/1465-9921-11-45
445. Van Pottelberge GR, Bracke KR, Demedts IK, De Rijck K, Reinartz SM, Van Drunen CM, et al. Selective accumulation of langerhans-type dendritic cells in small airways of patients with COPD. Respir Res (2010) 11(1):1–21. doi: 10.1186/1465-9921-11-35
446. Obata T, Goto Y, Kunisawa J, Sato S, Sakamoto M, Setoyama H, et al. Indigenous opportunistic bacteria inhabit mammalian gut-associated lymphoid tissues and share a mucosal antibody-mediated symbiosis. Proc Natl Acad Sci (2010) 107(16):7419–24. doi: 10.1073/pnas.1001061107
447. Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TK, et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature (2010) 464(7293):1367–70. doi: 10.1038/nature08900
448. Yang D, Chen X, Wang J, Lou Q, Lou Y, Li L, et al. Dysregulated lung commensal bacteria drive interleukin-17B production to promote pulmonary fibrosis through their outer membrane vesicles. Immunity (2019) 50(3):692–706.e7. doi: 10.1016/j.immuni.2019.02.001
449. Segal LN, Clemente JC, Tsay J-CJ, Koralov SB, Keller BC, Wu BG, et al. Enrichment of the lung microbiome with oral taxa is associated with lung inflammation of a Th17 phenotype. Nat Microbiol (2016) 1(5):16031. doi: 10.1038/nmicrobiol.2016.31
450. Pandiyan P, Bhaskaran N, Zou M, Schneider E, Jayaraman S, Huehn J. Microbiome dependent regulation of tregs and Th17 cells in mucosa. Front Immunol (2019) 10:426. doi: 10.3389/fimmu.2019.00426
451. Round JL, Lee SM, Li J, Tran G, Jabri B, Chatila TA, et al. The toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science (2011) 332(6032):974–7. doi: 10.1126/science.1206095
452. Wang S, Charbonnier L-M, Rivas MN, Georgiev P, Li N, Gerber G, et al. MyD88 adaptor-dependent microbial sensing by regulatory T cells promotes mucosal tolerance and enforces commensalism. Immunity (2015) 43(2):289–303. doi: 10.1016/j.immuni.2015.06.014
453. Tsuji M, Komatsu N, Kawamoto S, Suzuki K, Kanagawa O, Honjo T, et al. Preferential generation of follicular b helper T cells from Foxp3+ T cells in gut peyer's patches. Science (2009) 323(5920):1488–92. doi: 10.1126/science.1169152
454. Kawamoto S, Maruya M, Kato LM, Suda W, Atarashi K, Doi Y, et al. Foxp3+ T cells regulate immunoglobulin a selection and facilitate diversification of bacterial species responsible for immune homeostasis. Immunity (2014) 41(1):152–65. doi: 10.1016/j.immuni.2014.05.016
455. Zhang M, Zhou Q, Dorfman RG, Huang X, Fan T, Zhang H, et al. Butyrate inhibits interleukin-17 and generates tregs to ameliorate colorectal colitis in rats. BMC gastroenterol (2016) 16(1):1–9. doi: 10.1186/s12876-016-0500-x
456. Li MO, Wan YY, Flavell RA. T Cell-produced transforming growth factor-β1 controls T cell tolerance and regulates Th1-and Th17-cell differentiation. Immunity (2007) 26(5):579–91. doi: 10.1016/j.immuni.2007.03.014
457. Konkel JE, Zhang D, Zanvit P, Chia C, Zangarle-Murray T, Jin W, et al. Transforming growth factor-β signaling in regulatory T cells controls T helper-17 cells and tissue-specific immune responses. Immunity (2017) 46(4):660–74. doi: 10.1016/j.immuni.2017.03.015
458. Hapfelmeier S, Lawson MA, Slack E, Kirundi JK, Stoel M, Heikenwalder M, et al. Reversible microbial colonization of germ-free mice reveals the dynamics of IgA immune responses. Science (2010) 328(5986):1705–9. doi: 10.1126/science.1188454
459. Bunker JJ, Flynn TM, Koval JC, Shaw DG, Meisel M, McDonald BD, et al. Innate and adaptive humoral responses coat distinct commensal bacteria with immunoglobulin a. Immunity (2015) 43(3):541–53. doi: 10.1016/j.immuni.2015.08.007
460. Macpherson AJ, Gatto D, Sainsbury E, Harriman GR, Hengartner H, Zinkernagel RM. A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science (2000) 288(5474):2222–6. doi: 10.1126/science.288.5474.2222
461. Ladjemi MZ, Martin C, Lecocq M, Detry B, Nana FA, Moulin C, et al. Increased IgA expression in lung lymphoid follicles in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2019) 199(5):592–602. doi: 10.1164/rccm.201802-0352OC
462. Bunker JJ, Bendelac A. IgA responses to microbiota. Immunity (2018) 49(2):211–24. doi: 10.1016/j.immuni.2018.08.011
463. Good KL, Bryant VL, Tangye SG. Kinetics of human b cell behavior and amplification of proliferative responses following stimulation with IL-21. J Immunol (2006) 177(8):5236–47. doi: 10.4049/jimmunol.177.8.5236
464. Kaetzel CS, Robinson JK, Chintalacharuvu KR, Vaerman J-P, Lamm ME. The polymeric immunoglobulin receptor (secretory component) mediates transport of immune complexes across epithelial cells: a local defense function for IgA. Proc Natl Acad Sci (1991) 88(19):8796–800. doi: 10.1073/pnas.88.19.8796
465. Moor K, Diard M, Sellin ME, Felmy B, Wotzka SY, Toska A, et al. High-avidity IgA protects the intestine by enchaining growing bacteria. Nature (2017) 544(7651):498–502. doi: 10.1038/nature22058
466. Lindner C, Thomsen I, Wahl B, Ugur M, Sethi MK, Friedrichsen M, et al. Diversification of memory b cells drives the continuous adaptation of secretory antibodies to gut microbiota. Nat Immunol (2015) 16(8):880–8. doi: 10.1038/ni.3213
467. Magri G, Comerma L, Pybus M, Sintes J, Lligé D, Segura-Garzón D, et al. Human secretory IgM emerges from plasma cells clonally related to gut memory b cells and targets highly diverse commensals. Immunity (2017) 47(1):118–34.e8. doi: 10.1016/j.immuni.2017.06.013
468. Haas A, Zimmermann K, Graw F, Slack E, Rusert P, Ledergerber B, et al. Systemic antibody responses to gut commensal bacteria during chronic HIV-1 infection. Gut (2011) 60(11):1506–19. doi: 10.1136/gut.2010.224774
469. Okamura T, Gohda E, Kohge T, Yamamoto I. An increase in histone acetylation and IL-2 antagonizing the immunoinhibitory effect are necessary for augmentation by butyrate of in vitro anti-TNP antibody production. Biol Pharm Bullet (1999) 22(12):1288–92. doi: 10.1248/bpb.22.1288
470. Aoshiba K, Nagai A. Senescence hypothesis for the pathogenetic mechanism of chronic obstructive pulmonary disease. Proc Am Thorac Soc (2009) 6(7):596–601. doi: 10.1513/pats.200904-017RM
471. Huertas A, Palange P. COPD: a multifactorial systemic disease. Ther Adv Respir dis (2011) 5(3):217–24. doi: 10.1177/1753465811400490
472. Packard TA, Li QZ, Cosgrove GP, Bowler RP, Cambier JC. COPD is associated with production of autoantibodies to a broad spectrum of self-antigens, correlative with disease phenotype. Immunol Res (2013) 55(1):48–57. doi: 10.1007/s12026-012-8347-x
473. Feghali-Bostwick CA, Gadgil AS, Otterbein LE, Pilewski JM, Stoner MW, Csizmadia E, et al. Autoantibodies in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2008) 177(2):156–63. doi: 10.1164/rccm.200701-014OC
474. Cosio MG, Guerassimov A. Chronic obstructive pulmonary disease: inflammation of small airways and lung parenchyma. Am J Respir Crit Care Med (1999) 160(supplement_1):S21–S5. doi: 10.1164/ajrccm.160.supplement_1.7
475. Richmond BW, Mansouri S, Serezani A, Novitskiy S, Blackburn JB, Du RH, et al. Monocyte-derived dendritic cells link localized secretory IgA deficiency to adaptive immune activation in COPD. Mucosal Immunol (2021) 14(2):431–42. doi: 10.1038/s41385-020-00344-9
476. Hogg JC, Chu F, Utokaparch S, Woods R, Elliott WM, Buzatu L, et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N Engl J Med (2004) 350(26):2645–53. doi: 10.1056/NEJMoa032158
477. Lee SH, Goswami S, Grudo A, Song LZ, Bandi V, Goodnight-White S, et al. Antielastin autoimmunity in tobacco smoking-induced emphysema. Nat Med (2007) 13(5):567–9. doi: 10.1038/nm1583
478. Faner R, Cruz T, Casserras T, López-Giraldo A, Noell G, Coca I, et al. Network analysis of lung transcriptomics reveals a distinct b-cell signature in emphysema. Am J Respir Crit Care Med (2016) 193(11):1242–53. doi: 10.1164/rccm.201507-1311OC
479. Polverino F, Seys LJ, Bracke KR, Owen CA. B cells in chronic obstructive pulmonary disease: moving to center stage. Am J Physiol-Lung Cell Mol Physiol (2016) 311(4):L687–L95. doi: 10.1152/ajplung.00304.2016
480. Gosman MM, Willemse BW, Jansen DF, Lapperre TS, van Schadewijk A, Hiemstra PS, et al. Increased number of b-cells in bronchial biopsies in COPD. Eur Respir J (2006) 27(1):60–4. doi: 10.1183/09031936.06.00007005
481. Litsiou E, Semitekolou M, Galani IE, Morianos I, Tsoutsa A, Kara P, et al. CXCL13 production in b cells via toll-like receptor/lymphotoxin receptor signaling is involved in lymphoid neogenesis in chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2013) 187(11):1194–202. doi: 10.1164/rccm.201208-1543OC
482. Brusselle G, Demoor T, Bracke K, Brandsma C, Timens W. Lymphoid follicles in (very) severe COPD: beneficial or harmful? Eur Respir J (2009) 34(1):219–30. doi: 10.1183/09031936.00150208
483. Seys LJ, Verhamme FM, Schinwald A, Hammad H, Cunoosamy DM, Bantsimba-Malanda C, et al. Role of b cell–activating factor in chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2015) 192(6):706–18. doi: 10.1164/rccm.201501-0103OC
484. Polverino F, Cosio BG, Pons J, Laucho-Contreras M, Tejera P, Iglesias A, et al. B cell–activating factor. an orchestrator of lymphoid follicles in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2015) 192(6):695–705. doi: 10.1164/rccm.201501-0107OC
485. Sullivan J-L, Bagevalu B, Glass C, Sholl L, Kraft M, Martinez FD, et al. B cell–adaptive immune profile in emphysema-predominant chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2019) 200(11):1434–9. doi: 10.1164/rccm.201903-0632LE
486. Lin S, Ma Z, Huang Y, Sun Y, Yi H. Chronic obstructive pulmonary disease is characterized by reduced levels and defective suppressive function of regulatory b cells in peripheral blood. Mol Immunol (2022) 141:87–93. doi: 10.1016/j.molimm.2021.11.007
487. Brandsma C-A, Kerstjens HA, van Geffen WH, Geerlings M, Postma DS, Hylkema MN, et al. Differential switching to IgG and IgA in active smoking COPD patients and healthy controls. Eur Respir J (2012) 40(2):313–21. doi: 10.1183/09031936.00011211
488. Jacobs M, Verschraegen S, Salhi B, Anckaert J, Mestdagh P, Brusselle GG, et al. IL-10 producing regulatory b cells are decreased in blood from smokers and COPD patients. Respir Res (2022) 23(1):1–12. doi: 10.1186/s12931-022-02208-1
489. Lanzilli G, Traggiai E, Braido F, Garelli V, Folli C, Chiappori A, et al. Administration of a polyvalent mechanical bacterial lysate to elderly patients with COPD: effects on circulating T, b and NK cells. Immunol Lett (2013) 149(1-2):62–7. doi: 10.1016/j.imlet.2012.11.009
490. Chen F, Wu W, Jin L, Millman A, Palma M, El-Naccache DW, et al. B cells produce the tissue-protective protein RELMα during helminth infection, which inhibits IL-17 expression and limits emphysema. Cell Rep (2018) 25(10):2775–83.e3. doi: 10.1016/j.celrep.2018.11.038
491. Curtis JL. B cells caught in the act: class switching to IgA in lung lymphoid follicles in chronic obstructive pulmonary disease. Am Thorac Soc (2019) 199(5):548–50. doi: 10.1164/rccm.201810-1907ED
492. Ladjemi MZ, Lecocq M, Weynand B, Bowen H, Gould HJ, Van Snick J, et al. Increased IgA production by b-cells in COPD via lung epithelial interleukin-6 and TACI pathways. Eur Respir J (2015) 45(4):980–93. doi: 10.1183/09031936.00063914
493. Curtis JL. B cells caught in the act: Class switching to IgA in lung lymphoid follicles in chronic obstructive pulmonary disease. Am J Respir Crit Care Med (2019) 199(5):548–50. doi: 10.1164/rccm.201810-1907ED
494. Roberts IS. Pathology of IgA nephropathy. Nat Rev Nephrol (2014) 10(8):445–54. doi: 10.1038/nrneph.2014.92
495. Staples KJ, Taylor S, Thomas S, Leung S, Cox K, Pascal TG, et al. Relationships between mucosal antibodies, non-typeable haemophilus influenzae (NTHi) infection and airway inflammation in COPD. PloS One (2016) 11(11):e0167250. doi: 10.1371/journal.pone.0167250
496. Veeramachaneni SB, Sethi S. Pathogenesis of bacterial exacerbations of COPD. Copd (2006) 3(2):109–15. doi: 10.1080/15412550600651347
497. Bengoechea JA. Secretory IgA and COPD: a new kid on the block? Am J Respir Crit Care Med (2011) 184(3):285–7. doi: 10.1164/rccm.201105-0821ED
498. Millares L, Martí S, Ardanuy C, Liñares J, Santos S, Dorca J, et al. Specific IgA against pseudomonas aeruginosa in severe COPD. Int J Chron Obstruct Pulmon Dis (2017) 12:2807–11. doi: 10.2147/COPD.S141701
499. Murphy TF, Brauer AL, Aebi C, Sethi S. Antigenic specificity of the mucosal antibody response to moraxella catarrhalis in chronic obstructive pulmonary disease. Infect Immun (2005) 73(12):8161–6. doi: 10.1128/IAI.73.12.8161-8166.2005
Keywords: COPD, lung microbiome, mucosal immunity, inflammation, innate immunity, adaptive immunity
Citation: Kayongo A, Robertson NM, Siddharthan T, Ntayi ML, Ndawula JC, Sande OJ, Bagaya BS, Kirenga B, Mayanja-Kizza H, Joloba ML and Forslund SK (2023) Airway microbiome-immune crosstalk in chronic obstructive pulmonary disease. Front. Immunol. 13:1085551. doi: 10.3389/fimmu.2022.1085551
Received: 31 October 2022; Accepted: 28 December 2022;
Published: 17 January 2023.
Edited by:
Martin James Holland, University of London, United KingdomReviewed by:
Charles Pilette, Université Catholique de Louvain, BelgiumCopyright © 2023 Kayongo, Robertson, Siddharthan, Ntayi, Ndawula, Sande, Bagaya, Kirenga, Mayanja-Kizza, Joloba and Forslund. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sofia K. Forslund, sofia.forslund@mdc-berlin.de
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.