
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Immunol., 13 October 2022
Sec. Microbial Immunology
Volume 13 - 2022 | https://doi.org/10.3389/fimmu.2022.1010572
This article is part of the Research TopicInnate Immunity and Cross-Talk with Microflora in the Regulation of Immune Recognition and Polarization During Immune-Related DiseasesView all 9 articles
Ankylosing spondylitis (AS), a chronic condition that commonly influences the spine and sacroiliac joints, usually progresses to stiffness and progressive functional limitation. Its fundamental etiology and pathogenesis are likely multifactorial and remain elusive. As environmental factors, gut microbiota performs critical functions in the pathogenesis of AS through various mechanisms, including interacting with genes, enhancing intestinal permeability, activating the gut mucosa immune system, and affecting the intestinal microbiota metabolites. This review provides an overview of recent advances in investigating gut microbiota in AS pathogenesis and discusses potential methods for future therapeutic intervention.
Ankylosing spondylitis (AS), a prototype of spondyloarthritis (SpA), affects the fibrous and synovial joints of the sacroiliac and spinal joints, with a prevalence estimated between 0.07% and 0.31% in the world population (1). Its sphere may incorporate extra-articular features, including anterior uveitis and inflammatory bowel diseases (IBD) such as Crohn’s disease (CD) and ulcerative colitis (UC). The etiological mechanism of AS is complex, developing via the convergence of genetic, microbial, environmental, and immunological factors (2–4). Notwithstanding, emerging evidence suggest the role of environmental factors such as the intestinal microbiome.
The microorganisms colonized in the human gastrointestinal tract mucosa are collectively called the gut microbiota or gut microbiome, which mainly includes symbiotic bacteria, fungi, and viruses. The gut commensal bacteria, also known as the “microbial organ”, are the most significant and complex component of the host immune system. This microbial organ is crucial for the mucosal immune system’s initiation, upkeep, and operation. In the early stages of the disease, the diversity, structure, and function of the gut microbiota differ from healthy individuals. The microbiota dysbiosis affects the immune system locally and throughout the body, thus predisposing humans to several pathologies, including AS, IBD, reactive arthritis, etc. (5–7). The objective of our review is to present the current comprehension of the role of the gut microbiome and its metabolites in the pathogenesis of AS and to further explore potential therapeutic avenues targeting the gut microbiome for the clinical management of AS.
Recent studies have shown that the occurrence and development of AS are related to changes in the diversity and composition of the gut microbiome (Table 1). Zhou et al. (5) found that Bacteroides coprophilus, Parabacteroides distasonis, Eubacterium siraeum, Acidaminococcus fermentans, Prevotella copri were enriched in AS, while Enterococcus faecium E980 and TX0133a01 were reduced by metagenomic shotgun sequencing. Liu et al. (11) performed 16S rRNA gene sequencing on stool samples from AS patients and healthy controls (HCs) and found that compared with HCs, relative abundance of Bacteroidetes was decreased in AS cases, but Firmicutes and Verrucobacterium were increased. Morever, specific gut bacteria were associated with disease activity of AS patients. Costello et al. (17) found increased abundances of Lachnospiriaceae, Ruminococcus, Rikenellaceae, Porphyromonadaceae and Bacteroidaceae, but decreased abundance of Veillonellaceae and Prevotellaceae in the terminal ileum of AS patients. These findings suggest that some AS-rich species may be triggers of autoimmunity. The Firmicutes/Bacteroidetes (F/B) ratio has been widely associated with maintaining normal intestinal homeostasis, and an increased abundance of specific Firmicutes species causes AS (11, 14). In addition, the researchers also observed changes in metabolites outside and inside the gut in AS patients (Table 2). The changes of intestinal metabolites are closely related to intestinal microbial metabolism, and the changes of extra-intestinal metabolites may also be partly related to the transfer of intestinal microbial metabolites.
AS patients with subclinical intestinal inflammation, up to 70% of whom 4-16% will progress to IBD (28). The reason for this phenomenon may be that the imbalanced distribution and metabolic disorders of gut microbiota increase the permeability of the gut mucosa, activates the gut immune system, and produces a variety of pro-inflammatory factors, leading to gut inflammation. A cross-sectional study revealed that inflammatory lesions in AS patients with standard and aberrant colonoscopy were independent of bowel symptomatology and treatments used to treat the underlying disease (29). Distinct fecal microbiome signatures associated with gut inflammatory markers such as calprotectin are present in AS patients (15). This suggests that gut inflammatory status is closely associated with the gut microbiota signature of AS patients. In addition, there is an apparent association between the progression of SpA and the severity of chronic intestinal inflammation (30). Some AS patients have suffered from subclinical intestinal inflammation or IBD before the onset of the disease, suggesting dysbiosis in preclinical AS patients. The relationship between intestinal dysbiosis, intestinal inflammation and AS is shown in Figure 1.
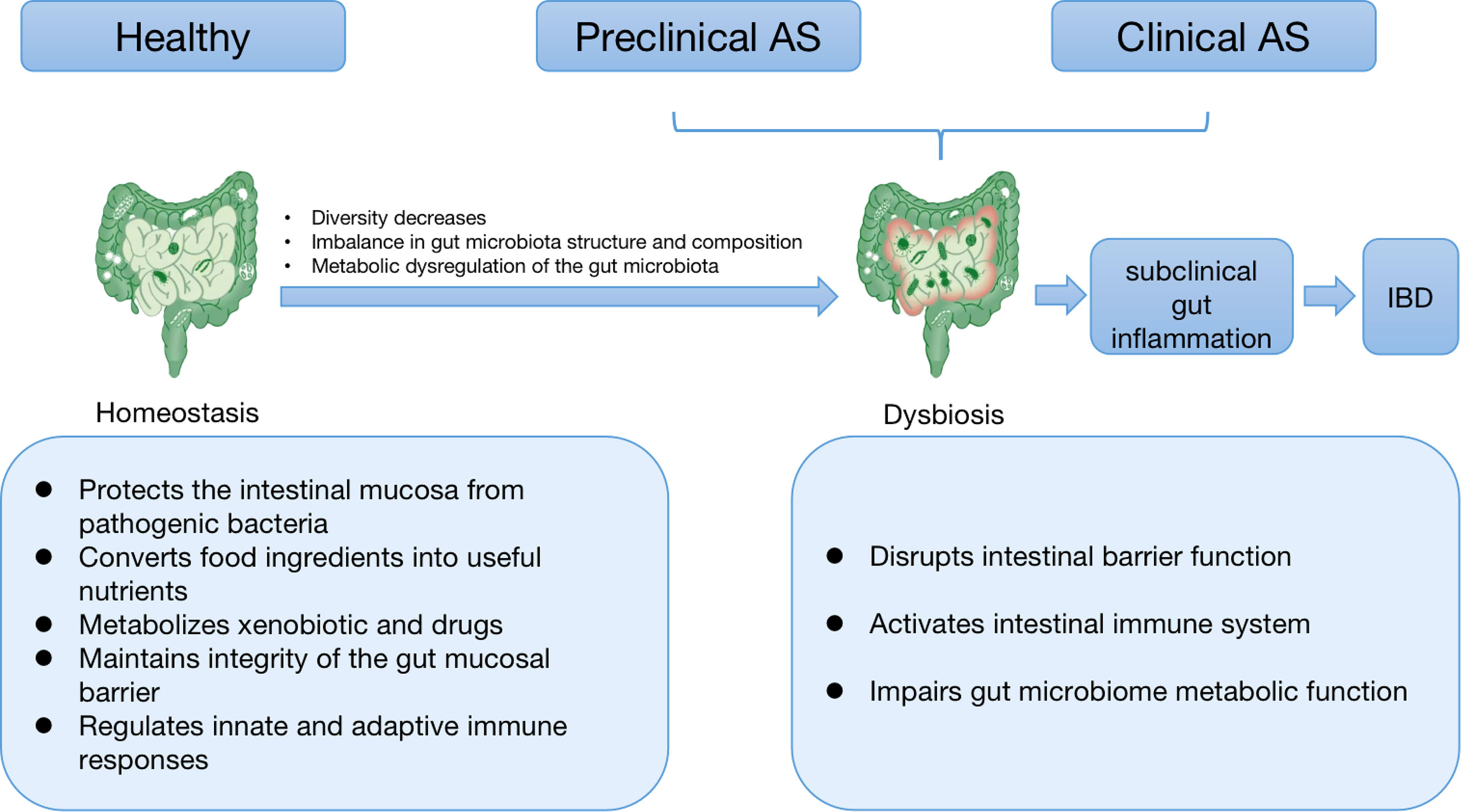
Figure 1 The relationship between intestinal dysbiosis, intestinal inflammation, and AS. Healthy gut microbiota is mainly responsible for human health. Gut microbial homeostasis imparts a specific function in protecting the intestinal mucosa from pathogenic bacteria, converting food components into valuable nutrients, metabolizing xenobiotics and drugs, and maintaining the integrity of the gut mucosal barrier and intestinal immune homeostasis. When species diversity begins to decline, the composition and function of intestinal microbiota change, and metabolism becomes dysregulated, we see a breakdown of the symbiosis, known as dysbiosis. Dysbiosis can disrupt intestinal barrier function, activate the intestinal immune system, and impair the metabolic process of the intestinal flora, thereby leading to subclinical intestinal inflammation. As the inflammation further intensifies, it develops into IBD with clinical manifestations. As is often accompanied by subclinical intestinal inflammation or IBD, some patients have suffered from dysbiosis in preclinical disease. This suggests that AS and IBD may have a common etiology–disturbed gut microbiota. In addition, there is a strong correlation between the severity of gut inflammation and the degree of spine inflammation in AS. Taken together, intestinal inflammation and immune responses in AS may be possible consequences of microbial dysbiosis. AS: ankylosing spondylitis; IBD: inflammatory bowel diseases.
Many studies have also revealed the critical roles of some bacteria in the pathogenesis of AS. Intestinal Klebsiella abundance was closely related to disease activity in AS (31). Klebsiella antibodies are closely associated with intestinal inflammation in axial AS patients (32). Wen et al.’s finding demonstrated that Actinobacteria are enriched in AS patients and might regulate the ubiquitination of IκB-α. This, in turn, activates NF-κB signaling and promotes the cumulation of pro-inflammatory cytokines in AS patients (16). In addition, fungi or cross-kingdom interactions between fungi and bacteria may contribute to the progression of AS. Elevated antibodies to the fungal cell wall element mannan have been observed in SpA (14). Functional polymorphisms have been identified in genes such as CARD9 and interleukin (IL)23R, which regulate the innate immune response to fungi (33).
Taken together, the gut microbiome and its abnormal metabolism are possible factors in the pathogenesis of AS. Therefore, we should focus on characterizing the species composition and metabolic pathways associated with AS, as well as the mechanisms by which the gut microbiome and its metabolites play a role in disease progression.
AS development is strongly associated with human leukocyte antigen (HLA)-B27, a major histocompatibility complex (MHC) allele. Several findings back up the concept that intestinal microbiota can increase the risk of AS by interacting with HLA-B27. The HLA-B27 transgenic rats spontaneously developed SpA-like disease, which relies on the gut microbiota to activate the IL-23/IL-17 pathway in the inflamed bowel and joints (34, 35). Interestingly, HLA-B27 transgenic rats cultured under aseptic conditions did not develop apparent inflammatory lesions in the intestine and joints (36). In contrast, normal luminal bacteria predictably and uniformly cause chronic colon, stomach, and joint inflammation in B27 transgenic rats (4), suggesting that the involvement of bacterial flora is necessary for the pathogenesis of HLA-B27.
There are two hypotheses regarding the interaction mechanisms between the microbiome and HLA-B27 in AS (Figure 2A). One of the hypotheses is the atherogenic peptide theory. This theory suggests that HLA-B27 presents bacteria or autoantigenic peptides to CD8+T or B cells, resulting in cross-reactivity with molecularly similar self-peptides (5, 37). Ebringer et al. (38) believed that there was molecular mimicry between HLA-B27 and some bacterial antigen components in the intestinal tract of patients with AS (such AS the polypeptide structure of Klebsiella and Shigella). In vitro, bacterial peptides from AS-enriched species triggered an increase of interferon (IFN)-γ producing cells, mimicking type II collagen. Several independent studies suggested that Klebsiella pneumonia may be the most likely etiopathogenetic trigger for AS because of molecular mimicry mechanisms and immune evidence, as well as the molecular similarity between Klebsiella and self-antigens (39, 40). Another theory is that HLA-B27 misfolds within the endoplasmic reticulum (ER) lead to ER stress followed by unfolded protein response (UPR) or autophagy, affecting downstream cellular functions that are hypothesized to contribute to the development of AS (41). This phenomenon has been demonstrated in HLA-B27 transgenic rats (42). However, no evidence of UPR transcriptional gene up-regulation was found in intestinal biopsies from AS patients, but autophagy was observed (43). Therefore, whether UPR is involved in human HLA-B27 and gut microbiome-driven inflammation remains unproven. Antoniou et al. found that misfolding of HLA-B27 and UPR cell conditions were associated with enhanced replication in Salmonella, which activates XBP-1 and ATF6. Their study provided evidence for the link between the interaction of gut microbiota with HLA-B27 and the pathogenesis of AS (44).
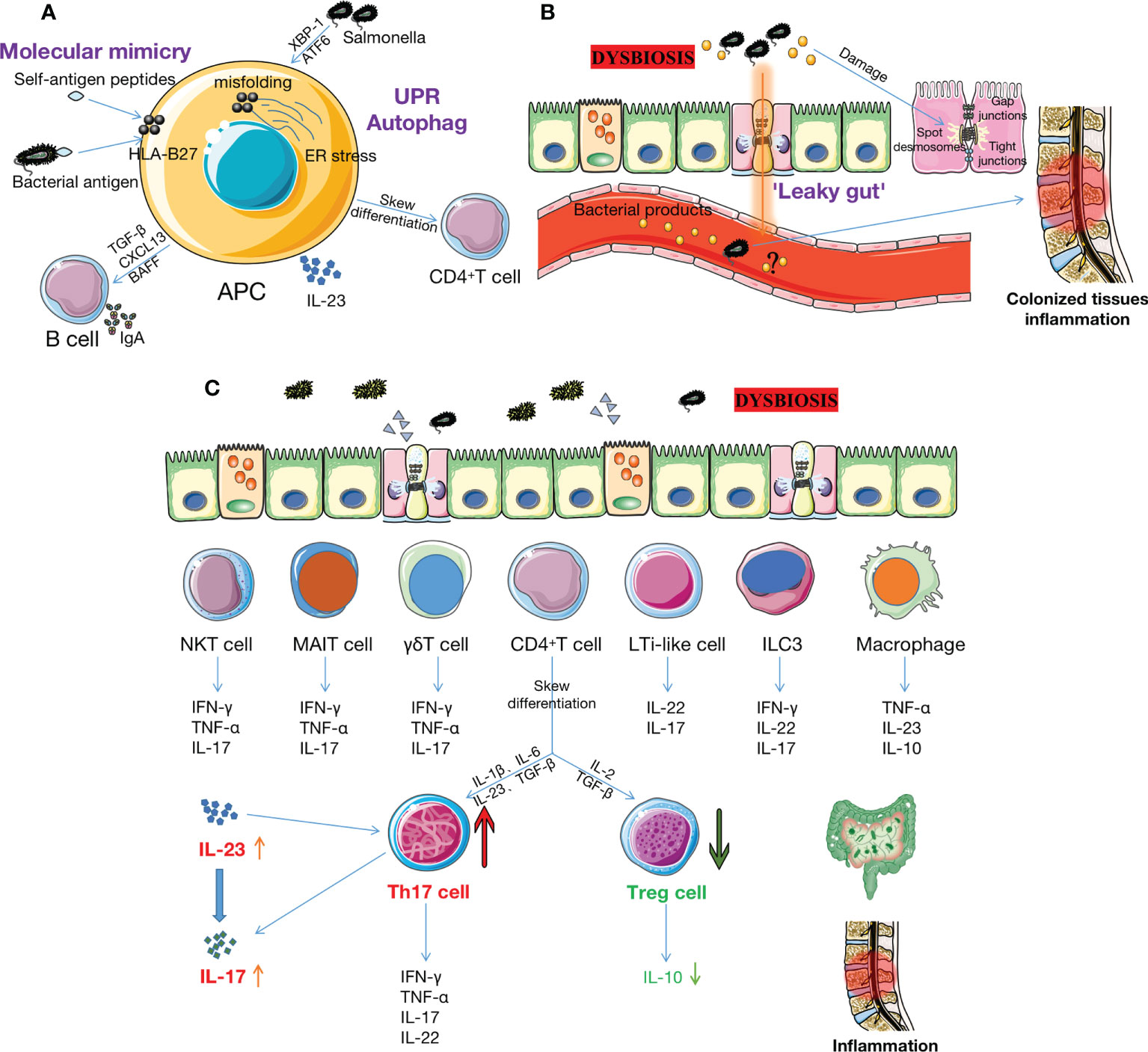
Figure 2 The intestinal microbiome and its metabolites may contribute to AS pathogenesis. (A) The interaction of gut microbiota and HLA-B27 may increase the risk of AS through two mechanisms. Firstly, HLA-B27 provides specific peptides, such as certain bacterial antigenic components (e.g.polypeptide structures of Klebsiella and Shigella), to APCs (e.g., CD8+ T or DC cells), resulting in cross-react on molecularly similar self-peptides. Secondly, misfolding of HLA-B27 within ER leads to ER stress, which triggers the UPR or autophagy, activating IL-23/IL-17 axis in the bowel and joints and simultaneously activating B cells to secrete IgA. However, the role of UPR in HLA-B27-driven inflammation has not been demonstrated in humans. (B) Dysbiosis leads to increased permeability between IECs, associated with reductions in cathelicidins and mucins, disruption of TJ proteins, impairment of GVBs, etc. Bacteria and bacterial products subsequently enter the bloodstream, inducing inflammation of the colonized tissue. However, it is uncertain whether there is bacterial translocation in AS patients. (C) Many immune cells (e.g., Th17, Treg, ILC3, MAIT, γδ T, NKT, LTi-like cells, DCs, macrophage et al.) reside in the intestinal mucosa. Dysbiosis triggers these cells to increase pro-inflammatory cytokines (such as IL-17, IL-23, TNF-α, IFN-γ, IL-7, IL-22, et al.) and decrease anti-inflammatory cytokines (such as IL-10). This activates the IL-23/IL-17 pathway and increases the Th17/Treg ratio, ultimately triggering or exacerbating inflammation in the gut and joints. AS: ankylosing spondylitis; HLA-B27: human leukocyte antigen B27; APCs: antigen-presenting cells; DC: dendritic cell; ER: endoplasmic reticulum; UPR: unfolded protein response; IECs: intestinal epithelial cells; TJ: tight junction; GVB: gut vascular barrier; Th17: T helper 17 cells; Treg: T regulatory cell; ILC3: innate lymphoid cell 3; MAIT: mucosal-associated invariant T cell; NKT: natural killer T cell; IL-17: interleukin 17; TNF-α: tumor necrosis factor-α; IFN-γ: interferon-γ; SCFAs: short chain fatty acids; Trp: tryptophan.
Several convincing evidences support gut microbiota’s role in maintaining the gastrointestinal tract’s structure and function. For instance, two soluble proteins, P40 and P75, produced by the Lactobacillus rhamnosus GG strain prevent cytokine-induced apoptosis of gut epithelial cells through epithelial growth factor receptor (EGFR) and protein kinase C (PKC)-dependent pathways (45). Bacteroidetes Thetaiotaomicron expresses the small proline-rich 2A proteins, which are indispensable for maintaining epithelial villous desmosomes structure (46). Intestinal microbiota participates in the development of intestinal microvessels by inducing the transcription factor angiopoietin-3, thereby promoting the structural evolution of intestinal mucosa (47). Lower gut surface area (48), thin villi (48), prolonged cell cycle time (49), and impaired peristalsis (50)were observed in germ-free (GF) mice. Therefore, gut microbiota is essential for maintaining normal gut structure and function. Tight junction (TJ) protein is the key to maintaining the integrity of the epithelial barrier. The abnormal function of TJ proteins leads to IBD even in the presence of normal underlying immunity (51). A mechanism for maintaining TJ is toll-like receptor (TLR)2-mediated signal transduction stimulated by microbial cell wall peptidoglycan. In vitro and ex vivo intestinal epithelial cell models suggest TLR2 stimulation promotes PI3K/Akt-mediated cell survival via MyD88, effectively preserving TJ-associated barrier assembly against stress-induced injury (52).
Increased permeability between intestinal epithelial cells (IECs) results in the development of AS (Figure 2B). Francesco et al. (53) performed ileum biopsy in 50 patients with HLA-B27+ AS patients and 20 controls. They found invasive and adherent bacteria, mainly gram-negative bacteria such as Escherichia coli and Prevotella, were present in the ileum of AS patients, which was related to reduced TJ protein and impaired gut vascular barrier (GVB). Another study observed increased gut permeability in AS patients and their relatives (54), though it is unclear whether pathobiont translocation is involved in AS (55). Endotoxin-induced uveitis associated with AS in mice may provide evidence (56). Epithelial and endothelial leakage often gives zonulin and bacterial products the opportunity to enter the bloodstream, which may activate innate and adaptive immune responses (53). However, we are not sure whether the loss of barrier integrity is a cause or a consequence of dysbiosis.
The gastrointestinal tract (GI) is regarded as the largest immune organ. Many immune cells reside in the intestinal mucosa, such as an innate lymphoid cell (ILC)3, mucosal-associated invariant T (MAIT), γδT, natural killer (NK)T, LTi-like cell, macrophage, neutrophil, dendritic cells (DCs), T cells, B cells et al. These cells play a crucial role in immune homeostasis regulation and suppression of inflammatory damage through innate and adaptive immune responses. Recent studies have shown that the gut microbiome’s composition affects the immune system’s development and modulates immune mediators. Dysbiosis may promote intestinal inflammation by affecting these cells, thereby aggravating the development of AS (57–61) (Figure 2C).
Alterations in the gut microbiome activate Paneth cells (PCs), which release the pro-inflammatory cytokines IL-23 and IL-7, causing lymphoid tissue inducer cells to cluster. Subsequently, ILC3s differentiate and produce IL-17 and IL-22. a4b7-expressing ILC3s migrate in the systemic circulation and accumulate in active inflammatory sites such as peripheral joints and bone marrow-rich peripheral tissues (57).
MAIT cells are a unique type of congenital antibacterial lymphocytes preferentially distributed in the intestine’s lamina propria (LP). Similar to IBD patients, AS patients have lower blood MAIT cell levels than HCs, possibly due to the recruitment of MAIT cells to inflammatory sites such as the gut and joints (62). MAIT cell activation is also associated with disease activity in AS patients. Metabolites produced by bacteria and fungi trigger rapid production of cytokines and chemokines responsible for host immune defense, such as IFN-γ and perforin, as well as production of pro-inflammatory cytokines responsible for the pathogenesis of AS, including IL-17 and tumor necrosis factor (TNF)-α (58, 63). In AS patients, IL-7 produced by intestinal PCs stimulates IL-17 production by MAIT cells (64). In summary, the presence and activation of MAIT cells in the intestines and joints of AS patients further supports the connection between the intestines and joints.
The gut microbiota also stimulates DCs to secrete CXCL13, TGF-β, and B-cell activating proteins, leading to immunoglobulin A (IgA) production and class switching by mucosal plasma cells. Although the underlying mechanism of this function remains unclear, it is speculated that this is mediated by MyD88 signaling in LP and follicular DC cells (65). In addition, reducing IL-10 produced by stimulation to autogenous Bacteroidetes cultures may be a mechanism for the onset and persistence of intestinal inflammation in AS (66).
The IL-23/IL-17 axis is an essential factor in the immunopathogenesis of AS. IL-23, which activated DCs and macrophages mainly secrete, stabilizes the phenotype of T helper 17 (Th17) cells and plays a decisive role in differentiating various subsets of IL-17-secreting cells (67, 68). Th17 cell was initially thought to be the primary source of IL-17. Recent studies found that many other lymphocytes, such as CD8+ cytotoxic T, Tc17, γδ T, MAIT, NK, and ILCs cells, could also secrete high levels of IL-17 (69–71). IL-17 promotes T cells activation and stimulates the production of pro-inflammatory cytokines and chemokines by fibroblasts, epithelial cells, endothelial and immune cells such as macrophages (72).
In HLA-B27/β2 microglobulin (β2m) transgenic rats, intestinal inflammation co-occurs with increased IL-23 and IL-17 expression in colon tissue (42). Interactions of cell surface ligands with specific microorganisms induce differentiation of IL-17 and IL-22 secreting cells, thereby programming their pathogenicity (73, 74). Dysbiosis characterized by an increased abundance of Prevotella species at mucosal sites may directly or indirectly affect type 17 immune responses through alterations in microbial metabolites or barrier function, thereby leading to a loss of immune tolerance and an increase in pro-inflammatory cytokines (such as IL-23) that trigger AS in susceptible populations. Prevotella predominantly activates TLR2, causing antigen-presenting cells to produce Th17-polarizing cytokines, including IL-1 and IL-23. Prevotella also stimulates the production of IL-6, IL-8, and CCL20 by epithelial cells, thereby promoting mucosal neutrophil recruitment. This phenomenon leads to the systemic dissemination of bacteria, bacterial products, and inflammatory mediators, thereby affecting systemic disease outcomes (75). All these findings suggest microbial dysbiosis triggers homeostasis changes in intestinal and joint inflammation via the IL-23/IL-17 pathway in AS.
Th17 and regulatory T (Treg) cells are differentiated from CD4+T cells. Th17 cells promote tissue inflammation and bone resorption, while Treg cells suppress autoimmunity and maintain immune homeostasis (76). Accumulating Tregs in inflamed joints contributes to the spontaneous resolution and remission process of arthritis in pSpA (77). The proportion of Tregs in peripheral blood monocytes and CD4+T cells was significantly reduced, and functional defects of Tregs promote the pathogenesis and progression of AS (78, 79). Several clinic studies observed an increased Th17/Treg ratio in AS (80–82). Reducing the Th17/Treg ratio helps promote the development of new bone formation in AS (83).
The gut microbiota composition modulates Th17/Treg balance in LP and thus may influence gut immunity, tolerance, and susceptibility to AS. Segmented filamentous bacteria (SFB) promotes SAA production in epithelial cells, which activates DCs to produce IL-6 and IL-23, resulting in the generation of Th17 cells (84). In the mouse gut, SFB triggers distorted T cell differentiation through TLR5 (85). Differentiation of Th17 cells also correlates with Cytophaga-Flavobacterium-Bacteroides (CFB) bacteria in the gut and is independent of TLRs, IL-21, or IL-23 signaling but requires proper TGF-β activation. The lack of Th17 cell induction in bacteria is accompanied by an increase in Foxp3+ Treg in LP (86). In mice, Tregs were most abundant in the colonic mucosa. Active Treg responses dominated by IL-10 production occurred in the gut of AS patients, and the number of Treg cells in LP of GF mice was significantly reduced (87). Still, it could be recovered by supplementation with specific bacteria such as Bacteroides fragilis, Clostridium consortium (especially Cluster IV and XIVa), and the “altered Scheidler flora” (a mixture of eight identified symbiotic bacteria) (88–90). Polysaccharide A (PSA), the immunomodulatory molecule of B. fragilis, promotes the transformation of CD4+T cells into Foxp3+Treg cells, which actively maintain mucosal tolerance by producing IL-10 during commensal colonization (91). We infer that their metabolic byproducts are sensed by cells of the immune system and affect the balance between pro-inflammatory and anti-inflammatory cells. For instance, C. Consortium and B. Fragilis have been reported to induce Treg cell differentiation by producing short-chain fatty acids (SCFAs) from dietary carbohydrates (92).
Microbial metabolites, such as SCFAs, tryptophan derivatives, vitamin B, etc., are thought to affect gut barrier function and exert immunomodulatory activities on immune cell subsets and non-immune cells (93). These crucial gut microbial metabolites and their roles in AS are summarized in Table 3. Asquit et al. analyzed the host and microbial metabolites of HLA-B27/β2m transgenic rats and found that various microbial and host metabolites were changed before and after the onset of the disease, affecting a variety of metabolisms way. These and other microbially derived biologically active mediators may provide a modality for early diagnosis and treatment of AS (106).
The gut microbiota produces SCFAs, mainly acetate, propionate, and butyrate, which are present in the intestinal mucosa and feces at a molar ratio of about 3:1:1 from food components (138). Phylum bacteroidetes primarily produce acetate and propionate as their predominant metabolic end products, while human colonic butyrate-producing bacteria are Firmicutes (139). SCFAs exert their functions mainly through two distinct pathways: Interaction with G protein-coupled receptors (GPR), GPR41 (FFAR3), GPR43 (FFAR2), and GPR109A (HM74b), and histone deacetylase (HDAC) inhibition. The biological functions of SCFAs in AS can be divided into effects on the intestinal barrier, immune cells, inflammation, and bone metabolism.
SCFAs improve mucosal barrier function. For example, Bifidobacteria enhances epithelial-mediated mucosal defenses by producing acetate, thereby inhibiting the transfer of EHEC Shiga toxin from the intestinal lumen to the bloodstream and protecting the host from fatal infections (97). Lectins, defensins, and cathelicidins play important roles in preventing harmful bacterial overgrowth and dysbiosis. In vitro studies have shown that SCFAs, especially butyrate, enhance cathelicidin LL-37 expression in colon cells. The interaction of SCFAs with the GPR43 receptor promotes the expression of the C-type lectin RegIIIγ and β-defensins 1, 3, and 4 in intestinal epithelial cells (94, 102). PCs function is altered in AS patients with subclinical intestinal inflammation, resulting in overexpression of PC-associated peptides, particularly human α-defensin 5 (HD-5) (140). In vitro, SCFAs can enhance the expression of Mucins (MUCs) in human goblet-like LS174T cells by inhibiting the corresponding histone acetylation and methylation in the promoter regions of HDAC and MUC2, and by AP-1 activation (103). MUCs promote the adhesion of Lactobacillus acidophilus and Bifidobacterium longum to cultured cells and inhibit the adhesion of Escherichia coli (141). Studies have shown that the production of MUCs, especially MUC1, in patients with AS is accompanied by an increase in the expression of IL-22 (142). IL-22 has protective effects on intestinal barrier integrity and immunity, and butyrate stimulates CD4+ T cells and ILCs to express IL-22 (98). Gene expression analysis of ileal samples in AS patients showed decreased intestinal TJ protein expression, leading to increased intestinal vascular barrier permeability, translocation of bacterial products in the blood, and subsequent intestinal and joint inflammation. This phenomenon is often accompanied by a marked upregulation of zonulin, which antagonizes TJ formation at the epithelial and endothelial levels (53). Butyrate is an essential enhancer of gut barrier function by upregulating TJ proteins (108–110).
SCFAs play an integral role in regulating energy homeostasis and affecting immune cell function in the host. SCFAs, especially butyrate, directly increased IFN-γ and granzyme B expression by cytotoxic T cells (CTLs) and differentiation of Tc17 cells to a CTL phenotype. This process relies on the strong inhibition of HDACs in CD8+ T cells rather than their interaction with specific SCFA-receptors GPR41 and GPR43. In addition, butyrate also increased the production of IFN-γ in CD8+ T cells by regulating mTOR activity and cellular metabolism (112). Gut IgA is vital in maintaining intestinal homeostasis and protecting the intestine from inflammation. Wu et al. reported that IgA is regulated by acetate through “metabolite sensing” GPR43. Mechanistically, acetate induces DC to express Aldh1a2, which converts vitamin A to retinoic acid (RA). However, blocking the RA signaling pathway can inhibit the inducible effect of acetate on the production of IgA in B cells (99). Butyrate has an immunomodulatory impact on intestinal macrophage function. Treatment of macrophages with butyrate downregulates LPS-induced pro-inflammatory mediators, including nitric oxide, IL-6, and IL-12. These effects have also been achieved through the inhibition of HDACs (113). After the Na+-coupled monocarboxylic acid transporter Slc5a8 transports butyrate and propionate into cells, butyrate and propionate reduce the expression of transcription factors PU.1 and RelB by inhibiting histone deacetylases, thereby blocking bone marrow stem cells differentiate into DCs (104).
The possible mechanism of the effect of SCFAs lies in the regulation of Th17/Treg balance, especially the strong upregulation of CD25+Foxp3+Treg in vivo and in vitro, which are mediated by inhibition of HDAC activity, resulting in the increase of anti-Th17 cytokines (such as IL-10 and IL-12) and the downregulation of pro-inflammatory cytokines (such as IL-23 and IL-17) levels in plasma and colonic mucosa (143, 144). Inhibition of T cell HDACs increases p70 S6 kinase acetylation and rS6 phosphorylation, modulating the mTOR pathways required for Th1, Th17, and IL-10+T cell generation (145). During Citrobacter rodentium infection, acetate enhances Th1 and Th17 cell induction but reduces the anti-CD3-induced inflammatory response in an IL-10-dependent manner. Butyrate promoted Th1 cell differentiation by increasing IFN-γ and T-bet expression while inhibited Th17 cell differentiation by decreasing IL-17, Rorαan d Rorγt expression. Interestingly, butyrate promoted IL-10 production in both cell conditions, thereby controlling the development of colitis (114). However, Asquith et al. demonstrated that oral propionate administration to HLA-B27/β2m transgenic rats significantly reduced HLA-B27-related intestinal inflammation and pro-inflammatory cytokines. Still, they did not find expression changes in Foxp3+ T cells, immunomodulatory cytokines IL-10 or IL-33, and TJ protein zonula occludens 1 (106). Sałkowska et al. investigated the effect of HDAC inhibition on Th17 differentiation. They found that treatment of Jurkat T cells and in vitro differentiated Th17 cells with 2 HDAC inhibitors (butyrate and apicidin) resulted in increased RORγT gene expression, which was associated with increased histone H4 acetylation near the proximal promoter of RORγT. In contrast, treatment of naive CD4+ cells differentiated into Th17 cells in vitro resulted in downregulation of RORγT expression when treated with the same inhibitor. This suggests a complex interaction with other mechanisms that sequentially modulate the expression of RORγT (146). A possible explanation for these studies is that alterations in SCFA content in AS patients lead to impaired Treg function, which results in activation of the IL23-IL17/22 axis and dysregulation of mucosal cells involved in type 3 immunity and regulated by RORγt (e.g., ILC3, invariant natural killer T (iNKT) cells, MAIT cells, and γδ-T cells) (147).
Dysbiosis is associated with the activation of NLRP3 inflammasome (148). Both autophagy and NLRP3 inflammasome activation can disrupt gut barrier integrity, and the two systems can activate each other (149). SCFAs have been shown to inhibit NLRP3 inflammasome activation in Caco-2 cells and LPS-induced autophagy. Acetate reduces interleukin-1β (IL-1β) production and neutrophil recruitment by inhibiting macrophages from activating inflammasomes and producing reactive oxygen species (ROS) (101). SCFAs also inhibited pro-inflammatory cytokines (e.g., iNOS, TNF-α, IL-1B, IL-17A, IL-6, and IFN-γ) and colonic inflammation while enhancing the expression of anti-inflammatory cytokines, IL-10 (106). Moreover, SCFAs, especially butyrate, diminished the production of CINC-2αβ, TNF-α, and NO by LPS-stimulated neutrophils (95) and have inhibitory effects on phagocytic and killing functions of neutrophils, thus playing an essential role in neutrophil inflammation in the gut of AS (100). NF-κB and ERK signaling pathways are at least partially involved in the anti-inflammatory activity of these SCFAs (96).
In AS, bone metabolism is simultaneously altered by increased bone resorption and juxtaposition of new bone (150). SCFAs are regulators of osteoclast metabolism and bone mass in vivo. In a model of experimental murine arthritis, SCFA appears to have an overall beneficial effect on bone - an increase in systemic bone density, a decrease in bone resorption, and a decrease in osteoclasts (107). Propionate and butyrate induce metabolic reprogramming of osteoclasts, enhancing glycolysis at the expense of oxidative phosphorylation, thereby down-regulating essential osteoclast genes such as TRAF6 and NFATc11 (107).
The host-absorbed tryptophan (Trp) is derived entirely from the diet for protein synthesis and other metabolic pathways. Bacteria metabolize Trp to uridine; They also metabolize dietary Trp to indole and other indole derivatives such as indole-3-propionic acid (IPA), indole-3-acetic acid (IAA), and indole-3-aldehyde (IAld) using tryptophanase. The host does not produce indole because tryptophanase is unique to bacteria (151). Indole and its derivatives have different effects on the host, from promoting inflammatory responses to regulating and eliminating inflammation, depending on specific metabolites, experimental models, cells, and receptors (152).
Indole-containing derivatives are absorbed by the host’s intestinal epithelium to indirectly shape the intestinal microbiome and modulate host responses, including barrier and immune functions, through either the aryl hydrocarbon receptor (AhR) or the pregnane X receptor (PXR) (118). This effect was later associated with the induction of Tregs (153). Additionally, the role of AhR in Th17 biology and the control of IL-22 production by T cells was also demonstrated (154, 155). IPA signals directly through epithelial cells within the intestine to maintain and repair the barrier, while IAA and I3Ald signal AhR on ILCs to increase IL-22 expression in the intestinal mucosa (119, 121, 122). Given the protective effect of IL-22 on gut barrier integrity and the expansion of IL-22-producing ILC3 in AS, this may suggest anti-inflammatory feedback. Venkatesh et al. (156) demonstrated that IPA, as a ligand of PXR, promotes intestinal barrier integrity by down-regulating epithelial TNF-α, inducing MDR1, and regulating epithelial junctional complexes. In addition, other Trp-derived metabolites, including Kynurenine, modulate epithelial IL-10R1 activity in an AhR-dependent manner (157). Such action was associated with protection in colitis and promoted epithelial wound healing (122). An assessment of bacterial metagenomics showed that the microbial community in HCs increased Trp synthesis, while the microbial community in axSpA increased Trp metabolism. The authors found that untreated AS patients had significantly lower Trp synthesis than treated AS patients and HCs (19). One study suggested that Trp metabolism to I3Ald and then IAA is one of the mechanisms by which the gut microbiota potentially affects axSpA development (21). Fecal metabolomics in pediatric SpA implicate decreased metabolic diversity, and changes in Trp metabolism are pathogenic factors (158).
Recent convincing results show amino acids (AAs) significantly affect host immune response. For instance, leucine, glutamine, and gamma-aminobutyric acid (GABA) play critical roles in mediating T cell function, including differentiation and activation of T cells, especially Th1 and Th17 cells (159–161). Mechanistically, this effect may largely depend on mTORC1 signaling since the differentiation of Th17 cells and the expression of IL-17 require AA-induced mTORC1 signaling (159). Spermidine spermine and putrescine, the primary polyamines (PAs) in human cells, originate from arginine metabolism. PAs are found in many foods but are also produced by gut microbiota, such as Bacteroides spp. and Fusobacterium spp (162). The study found that the expression of spermidine was up-regulated in HLA-B27/β2m transgenic rats compared to HCs (106). Liu et al. showed that adding putrescine to weaned piglet diets could reduce the incidence of diarrhea and improve intestinal integrity. Adding exogenous putrescine also decreased the mRNA levels of TNF-α, IL-6, and IL-8, and their upstream regulatory nuclear receptor kappa B p65 subunit in the jejunal mucosa of piglets improved anti-inflammatory function and inhibited inflammatory response (128). Furthermore, spermidine supplementation prevented IFN-α-induced overactivation of DCs and promoted Treg differentiation in vivo and in vitro (129).
The gut microbiota produces trimethylamine (TMA) from dietary phosphatidylcholine and quaternary amines such as choline, and L-carnitine, which are then converted to trimethylamine-n-oxide (TMAO) by heparin flavin monooxygenase(FMO), especially FMO1 and FMO3. TMAO is associated with systemic inflammatory disease activity (163). TMAO supplementation promotes Th1 and Th17 differentiation and increases the production of proinflammatory cytokines such as IL-17, IL-1β, and IL-6, which are mediated by polarized M1-like macrophages and require NLRP3 inflammasome activation (130). TMAO has been linked to the risk of cardiovascular disease (CVD), such as atherosclerosis, by inhibiting reverse cholesterol transport and increasing the expression of pro-atherosclerotic receptors on the surface of macrophages (164). CVD is a common comorbidity of AS and other SpA diseases, and TMPO may provide new insights into the underlying mechanisms and interventions associated with AS.
B vitamins such as vitamin B2 (riboflavin), vitamin B3 (niacin), and vitamin B9 (folate) are found in milk, eggs, liver, and green leafy vegetables. Nearly all Bacteroidetes, Fusobacteria, and Proteobacteria possess a Vitamin B biosynthesis pathway (165). The vitamin B metabolites produced by these microorganisms may delay the development of AS by promoting gut barrier function, inducing the proliferation of Treg cells, modulating the production of anti-inflammatory cytokines, and impacting the part of MAIT cells. Niacin is a receptor agonist of GPR109A. Studies have shown that the niacin-GPR109A interaction inhibits the production of IL-23 by colonic DCs, thereby impeding the proliferation of ILC3s in the colonic mucosa (132). There may be abnormal recycling of ILC3 from the gut to the joints, where they act close to the site of joint inflammation (166). Therefore, vitamin B3 produced by microbial metabolism may serve as a biological mediator of ILC3 activation in AS. The inosine (synthesized by the human microbiome, e.g., Lactobacillus reuteri) and histamine may inhibit inflammation by activating the adenosine A2A Receptor and histamine type 2 receptor (H2R), respectively. In addition, UroA (a major microbial metabolite derived from polyphenolics of berries and pomegranate fruits) and its potent synthetic analog (UAS03) up-regulate epithelial tight junction proteins by activating AhR- nuclear factor erythroid 2-related factor 2 (Nrf2)-dependent pathways.
The gut microbiota can be influenced by antibiotics, probiotics, prebiotics, medicines, mode of delivery and breastfeeding, diet, and other environmental factors. At present, resetting gut microbial dysbiosis through the methods mentioned above is emerging as a potential approach for the prevention and treatment of AS (Figure 3).
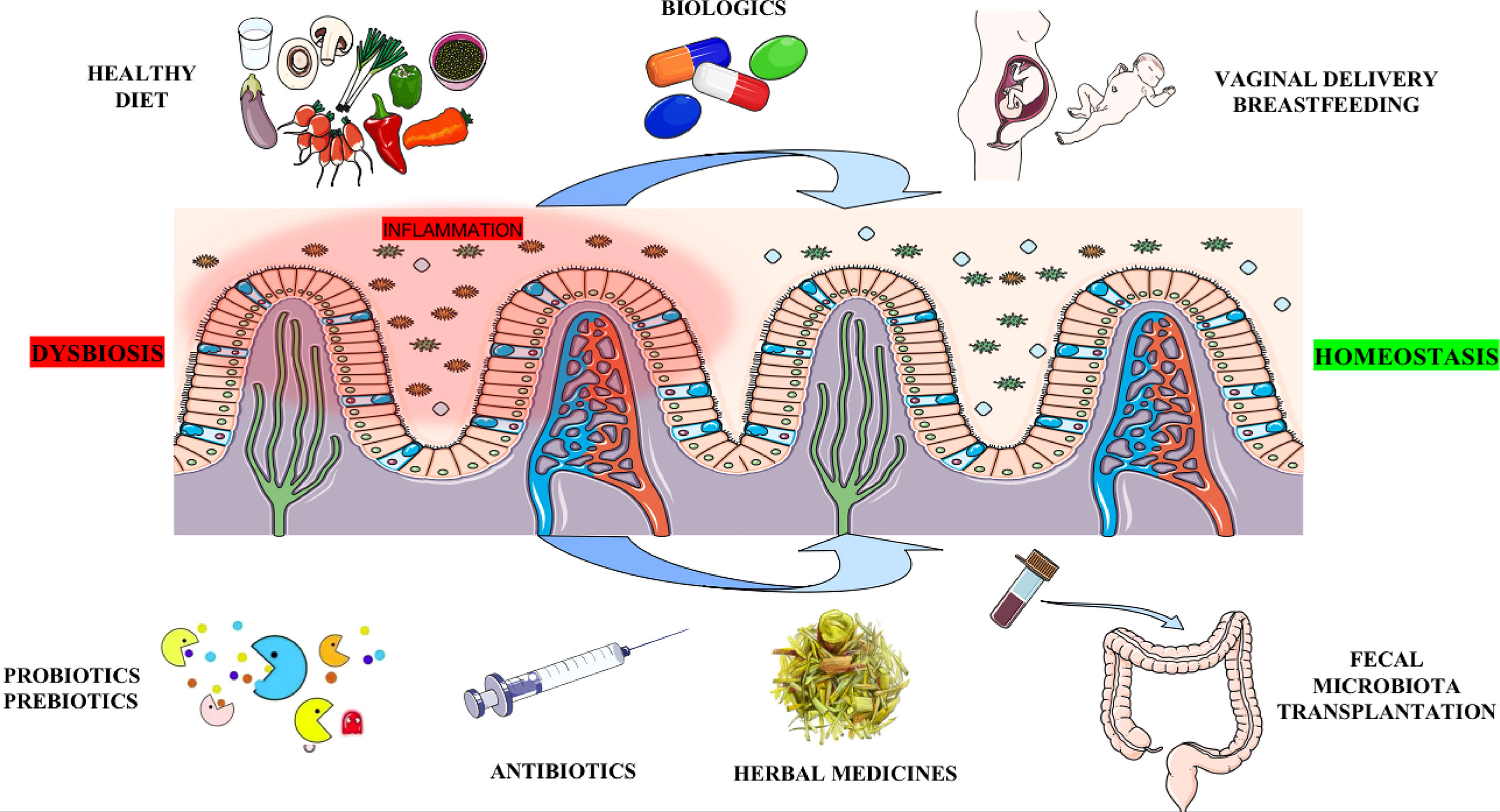
Figure 3 Potential methods for therapeutic intervention targeting the gut microbiota. Antibiotics may eliminate harmful bacteria in the gut. Probiotics, Prebiotics (e.g., SCFAs, Trp metabolites, Vitamin B, etc.), or FMT altering the gut micro-ecosystem serve as a critical strategy by increasing beneficial bacteria. Biologics and Chinese herbal medicine can treat AS by improving gut microecology. Vaginal delivery and breastfeeding participate in the original construction of healthy human gut microbiota. Finally, a healthy diet (such as low starch, high fiber, low fat, low sugar, etc.) fundamentally builds a healthy intestinal microbiota structure, thereby preventing the occurrence of AS. SCFAs: short chain fatty acids; Trp: tryptophan; FMT: fecal microbiota transplant.
AS is widely suspected as an infectious cause, which is thought to be associated with intestinal microbiota disorders. A variety of antibiotic drugs have been used to treat AS. For example, in a 12-week trial, moxifloxacin was well tolerated, safe, and improved inflammatory symptoms (167). Low dose sulfadiazine sulfapyridine (SSZ), a DMARD and sulfadiazine antibiotic, was influential in seronegative spondyloarthropathies patients with persistently active peripheral arthritis and prevented TNF-α-induced morphologically evident TJ rupture (168, 169). It has been suggested that rifaximin significantly reduced the severity of AS by changing gut microbiome composition with an elevated Bacteroidete/Firmicutes ratio and increasing some probiotics selectively, such as Lactobacillales (170). Rifaximin also prevented ileal histological changes, recovered gut barrier function, inhibited the activation of TLR-4/NF-κB signaling, and down-regulated inflammation factors (e.g., IL-17A, IL-6, IL-23, and TNF-α).
However, it has been reported that Extended-Spectrum β-Lactamase (ESBL) production in Klebsiella pneumoniae is closely related to resistance to ciprofloxacin (171). Co-resistance with other classes of antibiotics such as tetracycline, chloramphenicol, aminoglycosides, sulfonamides, and fluoroquinolones were also widespread in ESBL-producing strains (172). This reduces the value of AS in the treatment of Klebsiella pneumonia infection. In addition, antibiotics may inadvertently eliminate beneficial microbes, leading to an overgrowth of pathogenic and opportunistic pathogens such as Clostridium difficile in the gut (173). Since restoring microbiome composition is challenging, antibiotic use should be carefully decided.
Etanercept, an anti-TNF-α biologic, plays a crucial role in treating AS, attenuating the incidence and severity of arthritis. In mice, etanercept treatment restored intestinal microbiota composition similar to control mice (174). YIN et al. (19) and Maria et al. (175) found that TNFi treatment substantially restored the disturbed microbiome observed in untreated AS cases, including several critical bacterial species previously associated with AS and other related diseases, compared to HCs. In a study exploring responses to anti-TNF-α therapy, AS patients had a lower microbiota diversity at baseline than HCs at the same level but returned to normal after one month of treatment (12). Potential arthritic bacterial peptides were reduced in AS patients treated with TNFi compared with untreated patients. In addition, etanercept significantly restored TJ protein levels and disruption of ileum tissue, thereby restoring intestinal barrier function. IL-17i, another biologic, caused great changes in the abundance of specific taxa in AS patients, particularly Clostridiales and Candida albicans. As mentioned above, these data provide insight into biologic therapeutic strategies by modulating the gut microbiome in AS.
Probiotics are a kind of live nonpathogenic microorganisms that improve microbial balance in the gastrointestinal tract (176). The most used probiotic bacterial genera are lactic acid bacteria, mainly from the Lactobacillus genus. Bifidobacterium is also common, but other genera such as Enterococcus, Streptococcus, and Leuconostoc are also increasingly used. Also, probiotics may even fall into other fields, such as Saccharomyces. The actions of probiotics involve revealing different mechanisms, including production of bactericidal substances, competition with toxins and pathogens for adhesion to the gut epithelium, enhancement of innate immunity and modulation of inflammation through TLR-mediated signaling, promotion of gut epithelial cells survival, barrier function, and protective reactions regulating gut epithelial homeostasis through several signaling pathways (177). On this basis, altering the gut micro-ecosystem could serve as a key target for the prevention or treatment of AS.
Lactobacillus casei alleviates joint inflammation in mice with collagen-induced arthritis by reducing Th17 cell production by inhibiting IL-6, IL-17, and IL-23 (178). This effect was also mediated by reducing pro-inflammatory cytokines, inducing anti-inflammatory cytokine IL-10, and inhibiting cyclooxygenase-2 expression. Similar results were reported in an antigen-induced arthritis model study in Lewis rats using the Lactobacillus GG strain (179). Recent studies suggest that butyrate-producing microbes or Akkermansia-type bacteria may have the potential to enhance intestinal barrier function and alleviate inflammation, thereby relieving AS. Considering that SCFAs are largely free of side effects in vivo, these and other microbial-derived bioactive mediators may offer new therapeutic modalities for AS (106).
However, in a randomized controlled trial, despite the rationale for the therapy, the probiotic combination did not show a significant benefit compared to a placebo (180). In addition, bacteremia was reported in younger and immunocompromised patients, which was linked to probiotic treatment (181, 182). Therefore, further studies on the bioavailability and biosafety of probiotics in clinical applications are required.
Fecal microbiota transplantation (FMT) is the transplantation of functional flora in the feces of healthy people into the gastrointestinal tract of patients to rebuild new intestinal flora and treat intestinal and extra-intestinal diseases (183). FMT has been clinically practiced in Clostridium difficile infections, which cannot be cured with antibiotics alone (184). In a study of FMT combined with antibiotics in the treatment of UC, it was found that FMT, after antibiotic pretreatment, synergistically promoted the recovery of Bacteroides (185). However, more evidence is needed to assess whether FMT can be applied to AS.
Currently, the research on FMT is still in its infancy, and its efficacy and safety still need further consideration because this method is short-lived, requires frequently repeated transplantation, and may transmit pathogenic bacteria, prions, or cytomegalovirus (186). One case reported a young man with UC hospitalized with infectious symptoms, including abdominal cramps, diarrhea, and fever. Not long ago, he received a family FMT (186). In addition to the risk of infection, potential long-term threat to recipients should be noted in the clinical application of FMT. To select donors Carefully assessing donors clinically and not by indicators of gut microbiota composition could theoretically mitigate these risks.
The developing gut microbiota is susceptible to disturbance from external factors, such as parturition and early feeding patterns, maternal nutrition and health, and antibiotic use, especially during the perinatal and early infancy (187–189). Mode of delivery promotes exposure of neonates to specific species: vaginally delivered newborns carry microorganisms of the genera Bacteroides, Bifidobacterium, Parabacteroides, and Escherichia. The fecal microbiota of infants born by cesarean section contains Staphylococcus(e.g., Staphylococcus aureus, Veillonella, and Streptococcus) and Enterobacter. This colonization signature indicates that the bacteria originated from the mother’s oral mucosa, skin, and environment. The effect of delivery mode on intestinal microbiome composition was still detectable at age seven.
Breastfeeding was the most crucial factor regulating the intestinal microbiome after the neonatal period. One study showed that breastfeeding affects the microbiome and can prevent the development of AS (190). Breast milk components provide infants with nutrients and various bioactive compounds that affect immune system stimulation, growth, and regulation, protection from toxins and pathogens, cognitive development, and perhaps most notably, selective colonization and support of a protective microbiome (191–194). Breast milk contains indigestible polysaccharides such as human milk oligosaccharides (HMOs). Recently, studies have shown that only Bifidobacterium and Bacteroidetes could use HMOs as the sole nutrient and achieved enormous proliferation in the limited number of common intestinal bacteria tested, such as Clostridium, Lactobacillus, Escherichia coli, Enterococcus isolates, and Veillonella (195, 196). Interestingly, weaning immediately changes the baby’s microbiome (197).
Diet is the most direct link between the gastrointestinal tract and external conditions, which affects the composition and function of gut microbiota by affecting their gene expression and metabolites. ZHANG et al. (18) suggested that the gut microbiome alteration in AS was related to dietary factors. Lawrence et al. (198) showed that short-term macronutrient changes alter gut microbiota structure and submerge inter-individual distinction in microbial gene expression, suggesting that gut microbiota can respond rapidly to dietary changes, potentially facilitating the diversity of AS patients’ nutritional lifestyles. Given this scenario, there is growing concern that, as an easily modifiable environmental factor, a diet is an option for the prevention or treatment of AS (199). AS patients themselves consider diet to be more critical than medication (200). Recent studies have shown that the “Western diet” characterized by high starch and fat increases the risk of autoimmune disease by disrupting the gut barrier and affecting the structure and metabolites of microbes (201). Harmful bacteria in the gut depend on dietary starch for growth, so a “low-starch diet” that reduces the intake of “bread, cakes, and potatoes” may be beneficial for AS patients. The ‘low-starch diet’ reduced serum total IgA in HCs and patients, reducing inflammation and symptoms in patients with AS (200). Low fructose diets have also been associated with reductions in markers of inflammation, metabolic syndrome, and oxidative stress (202). Low fiber intake is also associated with an increased incidence of AS (203). Fibers, fermented in the colon, promote bacterial diversity, protect the mucosal barrier, and prompt production of SCFAs, positively maintaining gut microbial homeostasis and preventing inflammation (204–206). High-fiber feeding increases the production of SCFAs, especially acetate and butyrate. Host tolerance to food antigens depends on intestinal mucosal CD103+ DCs, which promote differentiation of Treg cells. SCFAs enhance intestinal mucosal immune tolerance and prevent food allergy by increasing retinal dehydrogenase activity in CD103+ DCs. This protective effect depends on the vitamin A in the food. This diet promoted IgA production and enhanced T follicular helper and mucosal germinal center responses. Mice lacking the SCFA receptors GPR43 or GPR109A exhibited aggravated food allergy and decreased CD103+ DCs. Therefore, a diet that includes fiber and vitamin A is necessary to prevent immune responses to food antigens and to protect the gastrointestinal mucosa (207). Drinking low-fat milk was associated with higher gut microbial diversity, whereas drinking high-fat dairy was associated with lower gut microbial diversity (208). The Mediterranean diet, which involves the intake of high-fiber whole grains, vegetables, and unsaturated fats such as olive oil, nuts, etc., is somewhat similar to an anti-inflammatory diet (209). Adherence to this diet was associated with reductions in inflammatory markers (210, 211). Because of the evidence, this diet seems promising as a possible strategy for addressing AS. In addition, α-Gal, a food allergen, is the leading natural antigen in mammalian red meat and may be involved in the pathogenesis of AS (212). On this account, allergenic foods such as beef, crab, and pork should be excluded from the daily diet of AS patients.
As an essential part of complementary and alternative medicines, Chinese herbal medicine has attracted more attention due to its solid curative effect and few side effects. However, the clinical application of Chinese herbal medicines has not been widely recognized due to its complex composition, unclear biological active compounds, and insufficient understanding of their underlying mechanisms. In recent years, herbal medicines have been increasingly used to treat AS patients and play an integral role in modulating gut microbiota during AS management (9). The ingredients of Herbs can modulate the diversity and composition of the gut microbiome (213); Herbal medicines can also affect the metabolism of gut microbiota, such as increasing the production of SCFAs (214); Furthermore, in the interaction of gut microbiota and herbs, herbal compounds can also exert their efficacy by converting into absorbable molecules with potent bioactivity (215). However, this therapeutic approach focuses primarily on relieving pain and delaying disease progression rather than completely controlling or treating AS. Further investigation of herbal medicines may help develop more effective treatments for AS.,,
There is increasing evidence that gut microbes are involved in the pathogenesis of AS. In-depth characterization of the gut microbiome can be achieved at high throughput and affordable cost through techniques independent of microbial cultures, such as 16s rDNA and metagenomic sequencing. These advanced techniques may provide clues to the intricate role of the gut microbiome in AS pathogenesis. In addition, technological advancements will facilitate the application of microbiome profiling in personalized therapy, providing more effective approaches to treating AS, including narrow-spectrum antibiotics or probiotic therapy, developing healthy eating habits, and introducing whole gut bacteria through FMT. Changes in the gut microbiome are dynamic. Consequently, certain microorganisms can be used as indicators to monitor disease activity and the effectiveness of treatment. In other words, rapid, comprehensive characterization of the gut microbiome may be required for doctors to diagnose and treat patients with AS correctly in the future.
All authors made a substantial contribution to discussion of the content and to review/editing of the manuscript before submission. Z-YS and DY drafted and wrote the manuscript. S-XZ revised the manuscript. All authors contributed to the article and approved the submitted version.
We acknowledge the National Natural Science Foundation of China (Grant No. 82001740) for invaluable funding.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Dean LE, Jones GT, MacDonald AG, Downham C, Sturrock RD, Macfarlane GJ. Global prevalence of ankylosing spondylitis. Rheumatol (Oxf) (2014) 53(4):650–7. doi: 10.1093/rheumatology/ket387
2. Voruganti A, Bowness P. New developments in our understanding of ankylosing spondylitis pathogenesis. Immunology (2020) 161(2):94–102. doi: 10.1111/imm.13242
3. Hwang MC, Ridley L, Reveille JD. Ankylosing spondylitis risk factors: A systematic literature review. Clin Rheumatol (2021) 40(8):3079–93. doi: 10.1007/s10067-021-05679-7
4. Rath HC, Herfarth HH, Ikeda JS, Grenther WB, Hamm TE Jr, Balish E, et al. Normal luminal bacteria, especially bacteroides species, mediate chronic colitis, gastritis, and arthritis in hla-B27/Human Beta2 microglobulin transgenic rats. J Clin Invest (1996) 98(4):945–53. doi: 10.1172/JCI118878
5. Zhou C, Zhao H, Xiao XY, Chen BD, Guo RJ, Wang Q, et al. Metagenomic profiling of the pro-inflammatory gut microbiota in ankylosing spondylitis. J Autoimmun (2020) 107:102360. doi: 10.1016/j.jaut.2019.102360
6. Lee M, Chang EB. Inflammatory bowel diseases (Ibd) and the microbiome-searching the crime scene for clues. Gastroenterology (2021) 160(2):524–37. doi: 10.1053/j.gastro.2020.09.056
7. Manasson J, Shen N, Garcia Ferrer HR, Ubeda C, Iraheta I, Heguy A, et al. Gut microbiota perturbations in reactive arthritis and postinfectious spondyloarthritis. Arthritis Rheumatol (2018) 70(2):242–54. doi: 10.1002/art.40359
8. Cardoneanu A, Cozma S, Rezus C, Petrariu F, Burlui AM, Rezus E. Characteristics of the intestinal microbiome in ankylosing spondylitis. Exp Ther Med (2021) 22(1):676. doi: 10.3892/etm.2021.10108
9. Huang R, Li F, Zhou Y, Zeng Z, He X, Fang L, et al. Metagenome-wide association study of the alterations in the intestinal microbiome composition of ankylosing spondylitis patients and the effect of traditional and herbal treatment. J Med Microbiol (2020) 69(6):797–805. doi: 10.1099/jmm.0.001107
10. Zhang F, Ma C, Zhang B, Bi L. Dynamic changes in gut microbiota under the influence of smoking and tnf-Alpha-Blocker in patients with ankylosing spondylitis. Clin Rheumatol (2020) 39(9):2653–61. doi: 10.1007/s10067-020-05032-4
11. Liu G, Hao Y, Yang Q, Deng S. The association of fecal microbiota in ankylosing spondylitis cases with c-reactive protein and erythrocyte sedimentation rate. Mediators Inflamm (2020) 2020:8884324. doi: 10.1155/2020/8884324
12. Zhang F, Ma C, Zhang B. Dynamic variations in gut microbiota in ankylosing spondylitis patients treated with anti-Tnf-Alpha for six months. Ann Clin Lab Sci (2020) 50(1):99–106.
13. Chen Z, Qi J, Wei Q, Zheng X, Wu X, Li X, et al. Variations in gut microbial profiles in ankylosing spondylitis: Disease phenotype-related dysbiosis. Ann Transl Med (2019) 7(20):571. doi: 10.21037/atm.2019.09.41
14. Li M, Dai B, Tang Y, Lei L, Li N, Liu C, et al. Altered bacterial-fungal interkingdom networks in the guts of ankylosing spondylitis patients. mSystems (2019) 4(2):e00176–18. doi: 10.1128/mSystems.00176-18
15. Klingberg E, Magnusson MK, Strid H, Deminger A, Stahl A, Sundin J, et al. A distinct gut microbiota composition in patients with ankylosing spondylitis is associated with increased levels of fecal calprotectin. Arthritis Res Ther (2019) 21(1):248. doi: 10.1186/s13075-019-2018-4
16. Wen C, Zheng Z, Shao T, Liu L, Xie Z, Le Chatelier E, et al. Quantitative metagenomics reveals unique gut microbiome biomarkers in ankylosing spondylitis. Genome Biol (2017) 18(1):142. doi: 10.1186/s13059-017-1271-6
17. Costello ME, Ciccia F, Willner D, Warrington N, Robinson PC, Gardiner B, et al. Brief report: Intestinal dysbiosis in ankylosing spondylitis. Arthritis Rheumatol (2015) 67(3):686–91. doi: 10.1002/art.38967
18. Zhang L, Han R, Zhang X, Fang G, Chen J, Li J, et al. Fecal microbiota in patients with ankylosing spondylitis: Correlation with dietary factors and disease activity. Clin Chim Acta (2019) 497:189–96. doi: 10.1016/j.cca.2019.07.038
19. Yin J, Sternes PR, Wang M, Song J, Morrison M, Li T, et al. Shotgun metagenomics reveals an enrichment of potentially cross-reactive bacterial epitopes in ankylosing spondylitis patients, as well as the effects of tnfi therapy upon microbiome composition. Ann Rheum Dis (2020) 79(1):132–40. doi: 10.1136/annrheumdis-2019-215763
20. Wang W, Yang GJ, Zhang J, Chen C, Jia ZY, Li J, et al. Plasma, urine and ligament tissue metabolite profiling reveals potential biomarkers of ankylosing spondylitis using nmr-based metabolic profiles. Arthritis Res Ther (2016) 18(1):244. doi: 10.1186/s13075-016-1139-2
21. Berlinberg AJ, Regner EH, Stahly A, Brar A, Reisz JA, Gerich ME, et al. Multi 'Omics analysis of intestinal tissue in ankylosing spondylitis identifies alterations in the tryptophan metabolism pathway. Front Immunol (2021) 12:587119. doi: 10.3389/fimmu.2021.587119
22. Chen R, Han S, Dong D, Wang Y, Liu Q, Xie W, et al. Serum fatty acid profiles and potential biomarkers of ankylosing spondylitis determined by gas chromatography-mass spectrometry and multivariate statistical analysis. BioMed Chromatogr (2015) 29(4):604–11. doi: 10.1002/bmc.3321
23. Gao P, Lu C, Zhang F, Sang P, Yang D, Li X, et al. Integrated gc-Ms and lc-Ms plasma metabonomics analysis of ankylosing spondylitis. Analyst (2008) 133(9):1214–20. doi: 10.1039/b807369d
24. Liu J, Zhu P, Peng J, Li K, Du J, Gu J, et al. Identification of disease-associated proteins by proteomic approach in ankylosing spondylitis. Biochem Biophys Res Commun (2007) 357(2):531–6. doi: 10.1016/j.bbrc.2007.03.179
25. Zhou Y, Zhang X, Chen R, Han S, Liu Y, Liu X, et al. Serum amino acid metabolic profiles of ankylosing spondylitis by targeted metabolomics analysis. Clin Rheumatol (2020) 39(8):2325–36. doi: 10.1007/s10067-020-04974-z
26. Gupta L, Guleria A, Rawat A, Kumar D, Aggarwal A. Nmr-based clinical metabolomics revealed distinctive serum metabolic profiles in patients with spondyloarthritis. Magn Reson Chem (2021) 59(2):85–98. doi: 10.1002/mrc.5083
27. Shao TJ, He ZX, Xie ZJ, Li HC, Wangn MJ, Wen CP, et al. Characterization of ankylosing spondylitis and rheumatoid arthritis using 1H NMR-based metabolomics of human fecal extracts. Metabolomics (2016) . 12:70. doi: 10.1007/s11306-016-1000-2
28. de Winter JJ, van Mens LJ, van der Heijde D, Landewe R, Baeten DL. Prevalence of peripheral and extra-articular disease in ankylosing spondylitis versus non-radiographic axial spondyloarthritis: A meta-analysis. Arthritis Res Ther (2016) 18:196. doi: 10.1186/s13075-016-1093-z
29. Maleh HC, Bica BE, Papi JA, de Azevedo MN, Carneiro AJ. [Colonoscopic evaluation in patients with ankylosing spondylitis]. Rev Bras Reumatol (2014) 54(5):342–8. doi: 10.1016/j.rbr.2014.03.020
30. Feng HY, Chan CH, Chu YC, Qu XM, Wang YH, Wei JC. Patients with ankylosing spondylitis have high risk of irritable bowel syndrome: A long-term nationwide population-based cohort study. Postgrad Med (2022) 134(3):290–6. doi: 10.1080/00325481.2022.2041338
31. Ebringer RW, Cawdell DR, Cowling P, Ebringer A. Sequential studies in ankylosing spondylitis. association of klebsiella pneumoniae with active disease. Ann Rheum Dis (1978) 37(2):146–51. doi: 10.1136/ard.37.2.146
32. Maki-Ikola O, Leirisalo-Repo M, Turunen U, Granfors K. Association of gut inflammation with increased serum iga class klebsiella antibody concentrations in patients with axial ankylosing spondylitis (as): Implication for different aetiopathogenetic mechanisms for axial and peripheral as? Ann Rheum Dis (1997) 56(3):180–3. doi: 10.1136/ard.56.3.180
33. Laurence M, Asquith M, Rosenbaum JT. Spondyloarthritis, acute anterior uveitis, and fungi: Updating the catterall-king hypothesis. Front Med (Lausanne) (2018) 5:80. doi: 10.3389/fmed.2018.00080
34. Hammer RE, Maika SD, Richardson JA, Tang JP, Taurog JD. Spontaneous inflammatory disease in transgenic rats expressing hla-B27 and human beta 2m: An animal model of hla-B27-Associated human disorders. Cell (1990) 63(5):1099–112. doi: 10.1016/0092-8674(90)90512-d
35. Glatigny S, Fert I, Blaton MA, Lories RJ, Araujo LM, Chiocchia G, et al. Proinflammatory Th17 cells are expanded and induced by dendritic cells in spondylarthritis-prone hla-B27-Transgenic rats. Arthritis Rheum (2012) 64(1):110–20. doi: 10.1002/art.33321
36. Taurog JD, Richardson JA, Croft JT, Simmons WA, Zhou M, Fernandez-Sueiro JL, et al. The germfree state prevents development of gut and joint inflammatory disease in hla-B27 transgenic rats. J Exp Med (1994) 180(6):2359–64. doi: 10.1084/jem.180.6.2359
37. Ewing C, Ebringer R, Tribbick G, Geysen HM. Antibody activity in ankylosing spondylitis sera to two sites on hla B27.1 at the mhc groove region (within sequence 65-85), and to a klebsiella pneumoniae nitrogenase reductase peptide (within sequence 181-199). J Exp Med (1990) 171(5):1635–47. doi: 10.1084/jem.171.5.1635
38. Ebringer A, Ghuloom M. Ankylosing spondylitis, hla-B27, and klebsiella: Cross reactivity and antibody studies. Ann Rheum Dis (1986) 45(8):703–4. doi: 10.1136/ard.45.8.703
39. Maki-Ikola O, Nissila M, Lehtinen K, Granfors K. Iga class serum antibodies against three different klebsiella serotypes in ankylosing spondylitis. Br J Rheumatol (1998) 37(12):1299–302. doi: 10.1093/rheumatology/37.12.1299
40. Ebringer A. The relationship between klebsiella infection and ankylosing spondylitis. Baillieres Clin Rheumatol (1989) 3(2):321–38. doi: 10.1016/s0950-3579(89)80024-x
41. Goodall JC, Wu C, Zhang Y, McNeill L, Ellis L, Saudek V, et al. Endoplasmic reticulum stress-induced transcription factor, chop, is crucial for dendritic cell il-23 expression. Proc Natl Acad Sci USA (2010) 107(41):17698–703. doi: 10.1073/pnas.1011736107
42. DeLay ML, Turner MJ, Klenk EI, Smith JA, Sowders DP, Colbert RA. Hla-B27 misfolding and the unfolded protein response augment interleukin-23 production and are associated with Th17 activation in transgenic rats. Arthritis Rheum (2009) 60(9):2633–43. doi: 10.1002/art.24763
43. Ciccia F, Accardo-Palumbo A, Rizzo A, Guggino G, Raimondo S, Giardina A, et al. Evidence that autophagy, but not the unfolded protein response, regulates the expression of il-23 in the gut of patients with ankylosing spondylitis and subclinical gut inflammation. Ann Rheum Dis (2014) 73(8):1566–74. doi: 10.1136/annrheumdis-2012-202925
44. Antoniou AN, Lenart I, Kriston-Vizi J, Iwawaki T, Turmaine M, McHugh K, et al. Salmonella exploits hla-B27 and host unfolded protein responses to promote intracellular replication. Ann Rheum Dis (2019) 78(1):74–82. doi: 10.1136/annrheumdis-2018-213532
45. Yan F, Cao H, Cover TL, Washington MK, Shi Y, Liu L, et al. Colon-specific delivery of a probiotic-derived soluble protein ameliorates intestinal inflammation in mice through an egfr-dependent mechanism. J Clin Invest (2011) 121(6):2242–53. doi: 10.1172/JCI44031
46. Lutgendorff F, Akkermans LM, Soderholm JD. The role of microbiota and probiotics in stress-induced gastro-intestinal damage. Curr Mol Med (2008) 8(4):282–98. doi: 10.2174/156652408784533779
47. Stappenbeck TS, Hooper LV, Gordon JI. Developmental regulation of intestinal angiogenesis by indigenous microbes Via paneth cells. Proc Natl Acad Sci USA (2002) 99(24):15451–5. doi: 10.1073/pnas.202604299
48. Banasaz M, Norin E, Holma R, Midtvedt T. Increased enterocyte production in gnotobiotic rats mono-associated with lactobacillus rhamnosus gg. Appl Environ Microbiol (2002) 68(6):3031–4. doi: 10.1128/AEM.68.6.3031-3034.2002
49. Alam M, Midtvedt T, Uribe A. Differential cell kinetics in the ileum and colon of germfree rats. Scand J Gastroenterol (1994) 29(5):445–51. doi: 10.3109/00365529409096836
50. Husebye E, Hellstrom PM, Midtvedt T. Intestinal microflora stimulates myoelectric activity of rat small intestine by promoting cyclic initiation and aboral propagation of migrating myoelectric complex. Dig Dis Sci (1994) 39(5):946–56. doi: 10.1007/BF02087542
51. Chelakkot C, Ghim J, Ryu SH. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp Mol Med (2018) 50(8):1–9. doi: 10.1038/s12276-018-0126-x
52. Cario E, Gerken G, Podolsky DK. Toll-like receptor 2 controls mucosal inflammation by regulating epithelial barrier function. Gastroenterology (2007) 132(4):1359–74. doi: 10.1053/j.gastro.2007.02.056
53. Ciccia F, Guggino G, Rizzo A, Alessandro R, Luchetti MM, Milling S, et al. Dysbiosis and zonulin upregulation alter gut epithelial and vascular barriers in patients with ankylosing spondylitis. Ann Rheum Dis (2017) 76(6):1123–32. doi: 10.1136/annrheumdis-2016-210000
54. Martinez-Gonzalez O, Cantero-Hinojosa J, Paule-Sastre P, Gomez-Magan JC, Salvatierra-Rios D. Intestinal permeability in patients with ankylosing spondylitis and their healthy relatives. Br J Rheumatol (1994) 33(7):644–7. doi: 10.1093/rheumatology/33.7.644
55. Fung TC, Bessman NJ, Hepworth MR, Kumar N, Shibata N, Kobuley D, et al. Lymphoid-Tissue-Resident commensal bacteria promote members of the il-10 cytokine family to establish mutualism. Immunity (2016) 44(3):634–46. doi: 10.1016/j.immuni.2016.02.019
56. Rosenbaum JT, McDevitt HO, Guss RB, Egbert PR. Endotoxin-induced uveitis in rats as a model for human disease. Nature (1980) 286(5773):611–3. doi: 10.1038/286611a0
57. Ciccia F, Rizzo A, Triolo G. Subclinical gut inflammation in ankylosing spondylitis. Curr Opin Rheumatol (2016) 28(1):89–96. doi: 10.1097/BOR.0000000000000239
58. Ciccia F, Guggino G, Rizzo A, Saieva L, Peralta S, Giardina A, et al. Type 3 innate lymphoid cells producing il-17 and il-22 are expanded in the gut, in the peripheral blood, synovial fluid and bone marrow of patients with ankylosing spondylitis. Ann Rheum Dis (2015) 74(9):1739–47. doi: 10.1136/annrheumdis-2014-206323
59. Boismenu R, Havran WL. Modulation of epithelial cell growth by intraepithelial gamma delta T cells. Science (1994) 266(5188):1253–5. doi: 10.1126/science.7973709
60. Kenna TJ, Davidson SI, Duan R, Bradbury LA, McFarlane J, Smith M, et al. Enrichment of circulating interleukin-17-Secreting interleukin-23 receptor-positive Gamma/Delta T cells in patients with active ankylosing spondylitis. Arthritis Rheum (2012) 64(5):1420–9. doi: 10.1002/art.33507
61. Jacques P, Venken K, Van Beneden K, Hammad H, Seeuws S, Drennan MB, et al. Invariant natural killer T cells are natural regulators of murine spondylarthritis. Arthritis Rheum (2010) 62(4):988–99. doi: 10.1002/art.27324
62. Koppejan H, Jansen D, Hameetman M, Thomas R, Toes REM, van Gaalen FA. Altered composition and phenotype of mucosal-associated invariant T cells in early untreated rheumatoid arthritis. Arthritis Res Ther (2019) 21(1):3. doi: 10.1186/s13075-018-1799-1
63. Chua WJ, Truscott SM, Eickhoff CS, Blazevic A, Hoft DF, Hansen TH. Polyclonal mucosa-associated invariant T cells have unique innate functions in bacterial infection. Infect Immun (2012) 80(9):3256–67. doi: 10.1128/IAI.00279-12
64. Cafforio P, Romito A, Grizzuti MA, Silvestris F. Methods for assessing programmed cell death. Recenti Prog Med (1996) 87(7-8):366–73.
65. Suzuki K, Maruya M, Kawamoto S, Sitnik K, Kitamura H, Agace WW, et al. The sensing of environmental stimuli by follicular dendritic cells promotes immunoglobulin a generation in the gut. Immunity (2010) 33(1):71–83. doi: 10.1016/j.immuni.2010.07.003
66. Stebbings SM, Taylor C, Tannock GW, Baird MA, Highton J. The immune response to autologous bacteroides in ankylosing spondylitis is characterized by reduced interleukin 10 production. J Rheumatol (2009) 36(4):797–800. doi: 10.3899/jrheum.080964
67. Schlosstein L, Terasaki PI, Bluestone R, Pearson CM. High association of an hl-a antigen, W27, with ankylosing spondylitis. N Engl J Med (1973) 288(14):704–6. doi: 10.1056/NEJM197304052881403
68. Taurog JD, Chhabra A, Colbert RA. Ankylosing spondylitis and axial spondyloarthritis. N Engl J Med (2016) 374(26):2563–74. doi: 10.1056/NEJMra1406182
69. Rudwaleit M, van der Heijde D, Landewe R, Listing J, Akkoc N, Brandt J, et al. The development of assessment of spondyloarthritis international society classification criteria for axial spondyloarthritis (Part ii): Validation and final selection. Ann Rheum Dis (2009) 68(6):777–83. doi: 10.1136/ard.2009.108233
70. Rudwaleit M, van der Heijde D, Landewe R, Akkoc N, Brandt J, Chou CT, et al. The assessment of spondyloarthritis international society classification criteria for peripheral spondyloarthritis and for spondyloarthritis in general. Ann Rheum Dis (2011) 70(1):25–31. doi: 10.1136/ard.2010.133645
71. Li J, Casanova JL, Puel A. Mucocutaneous il-17 immunity in mice and humans: Host defense vs. excessive inflammation. Mucosal Immunol (2018) 11(3):581–9. doi: 10.1038/mi.2017.97
72. Fragoulis GE, Liava C, Daoussis D, Akriviadis E, Garyfallos A, Dimitroulas T. Inflammatory bowel diseases and spondyloarthropathies: From pathogenesis to treatment. World J Gastroenterol (2019) 25(18):2162–76. doi: 10.3748/wjg.v25.i18.2162
73. Yang Y, Torchinsky MB, Gobert M, Xiong H, Xu M, Linehan JL, et al. Focused specificity of intestinal Th17 cells towards commensal bacterial antigens. Nature (2014) 510(7503):152–6. doi: 10.1038/nature13279
74. Zielinski CE, Mele F, Aschenbrenner D, Jarrossay D, Ronchi F, Gattorno M, et al. Pathogen-induced human Th17 cells produce ifn-gamma or il-10 and are regulated by il-1beta. Nature (2012) 484(7395):514–8. doi: 10.1038/nature10957
75. Larsen JM. The immune response to prevotella bacteria in chronic inflammatory disease. Immunology (2017) 151(4):363–74. doi: 10.1111/imm.12760
76. Josefowicz SZ, Lu LF, Rudensky AY. Regulatory T cells: Mechanisms of differentiation and function. Annu Rev Immunol (2012) 30:531–64. doi: 10.1146/annurev.immunol.25.022106.141623
77. Appel H, Wu P, Scheer R, Kedor C, Sawitzki B, Thiel A, et al. Synovial and peripheral blood Cd4+Foxp3+ T cells in spondyloarthritis. J Rheumatol (2011) 38(11):2445–51. doi: 10.3899/jrheum.110377
78. Miao J, Zhu P. Functional defects of treg cells: New targets in rheumatic diseases, including ankylosing spondylitis. Curr Rheumatol Rep (2018) 20(5):30. doi: 10.1007/s11926-018-0729-1
79. Li M, Zhou X, Zhou L, Yu Z, Fu L, Yang P. Meta-analysis of changes in the number and proportion of regulatory T cells in patients with ankylosing spondylitis. BioMed Res Int (2020) 2020:8709804. doi: 10.1155/2020/8709804
80. An H, Li X, Li F, Gao C, Li X, Luo J. The absolute counts of peripheral T lymphocyte subsets in patient with ankylosing spondylitis and the effect of low-dose interleukin-2. Med (Baltimore) (2019) 98(15):e15094. doi: 10.1097/MD.0000000000015094
81. Liu D, Liu B, Lin C, Gu J. Imbalance of peripheral lymphocyte subsets in patients with ankylosing spondylitis: A meta-analysis. Front Immunol (2021) 12:696973. doi: 10.3389/fimmu.2021.696973
82. Tan H, Huang S, Wang T. Clinical significance of peripheral blood Th1 and Th17 cell content and serum il-35 and il-17 expression in patients with ankylosing spondylitis. Evid Based Complement Alternat Med (2022) 2022:6540557. doi: 10.1155/2022/6540557
83. Xu F, Guanghao C, Liang Y, Jun W, Wei W, Baorong H. Treg-promoted new bone formation through suppressing Th17 by secreting interleukin-10 in ankylosing spondylitis. Spine (Phila Pa 1976) (2019) 44(23):E1349–55. doi: 10.1097/BRS.0000000000003169
84. Goto Y, Panea C, Nakato G, Cebula A, Lee C, Diez MG, et al. Segmented filamentous bacteria antigens presented by intestinal dendritic cells drive mucosal Th17 cell differentiation. Immunity (2014) 40(4):594–607. doi: 10.1016/j.immuni.2014.03.005
85. Rogier R, Koenders MI, Abdollahi-Roodsaz S. Toll-like receptor mediated modulation of T cell response by commensal intestinal microbiota as a trigger for autoimmune arthritis. J Immunol Res (2015) 2015:527696. doi: 10.1155/2015/527696
86. Ivanov II, Frutos Rde L, Manel N, Yoshinaga K, Rifkin DB, Sartor RB, et al. Specific microbiota direct the differentiation of il-17-Producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe (2008) 4(4):337–49. doi: 10.1016/j.chom.2008.09.009
87. Ciccia F, Accardo-Palumbo A, Giardina A, Di Maggio P, Principato A, Bombardieri M, et al. Expansion of intestinal Cd4+Cd25(High) treg cells in patients with ankylosing spondylitis: A putative role for interleukin-10 in preventing intestinal Th17 response. Arthritis Rheum (2010) 62(12):3625–34. doi: 10.1002/art.27699
88. Chambers RE, MacFarlane DG, Whicher JT, Dieppe PA. Serum amyloid-a protein concentration in rheumatoid arthritis and its role in monitoring disease activity. Ann Rheum Dis (1983) 42(6):665–7. doi: 10.1136/ard.42.6.665
89. Geuking MB, Cahenzli J, Lawson MA, Ng DC, Slack E, Hapfelmeier S, et al. Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunity (2011) 34(5):794–806. doi: 10.1016/j.immuni.2011.03.021
90. Round JL, Lee SM, Li J, Tran G, Jabri B, Chatila TA, et al. The toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science (2011) 332(6032):974–7. doi: 10.1126/science.1206095
91. Round JL, Mazmanian SK. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc Natl Acad Sci USA (2010) 107(27):12204–9. doi: 10.1073/pnas.0909122107
92. Smith PM, Howitt MR, Panikov N, Michaud M, Gallini CA, Bohlooly YM, et al. The microbial metabolites, short-chain fatty acids, regulate colonic treg cell homeostasis. Science (2013) 341(6145):569–73. doi: 10.1126/science.1241165
93. Scalise G, Ciancio A, Mauro D, Ciccia F. Intestinal microbial metabolites in ankylosing spondylitis. J Clin Med (2021) 10(15):3354. doi: 10.3390/jcm10153354
94. Zhao Y, Chen F, Wu W, Sun M, Bilotta AJ, Yao S, et al. Gpr43 mediates microbiota metabolite scfa regulation of antimicrobial peptide expression in intestinal epithelial cells Via activation of mtor and Stat3. Mucosal Immunol (2018) 11(3):752–62. doi: 10.1038/mi.2017.118
95. Vinolo MA, Rodrigues HG, Hatanaka E, Sato FT, Sampaio SC, Curi R. Suppressive effect of short-chain fatty acids on production of proinflammatory mediators by neutrophils. J Nutr Biochem (2011) 22(9):849–55. doi: 10.1016/j.jnutbio.2010.07.009
96. Park JS, Lee EJ, Lee JC, Kim WK, Kim HS. Anti-inflammatory effects of short chain fatty acids in ifn-Gamma-Stimulated raw 264.7 murine macrophage cells: Involvement of nf-kappab and erk signaling pathways. Int Immunopharmacol (2007) 7(1):70–7. doi: 10.1016/j.intimp.2006.08.015
97. Fukuda S, Toh H, Hase K, Oshima K, Nakanishi Y, Yoshimura K, et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature (2011) 469(7331):543–7. doi: 10.1038/nature09646
98. Yang W, Yu T, Huang X, Bilotta AJ, Xu L, Lu Y, et al. Intestinal microbiota-derived short-chain fatty acids regulation of immune cell il-22 production and gut immunity. Nat Commun (2020) 11(1):4457. doi: 10.1038/s41467-020-18262-6
99. Wu W, Sun M, Chen F, Cao AT, Liu H, Zhao Y, et al. Microbiota metabolite short-chain fatty acid acetate promotes intestinal iga response to microbiota which is mediated by Gpr43. Mucosal Immunol (2017) 10(4):946–56. doi: 10.1038/mi.2016.114
100. Vinolo MA, Hatanaka E, Lambertucci RH, Newsholme P, Curi R. Effects of short chain fatty acids on effector mechanisms of neutrophils. Cell Biochem Funct (2009) 27(1):48–55. doi: 10.1002/cbf.1533
101. Vieira AT, Macia L, Galvao I, Martins FS, Canesso MC, Amaral FA, et al. A role for gut microbiota and the metabolite-sensing receptor Gpr43 in a murine model of gout. Arthritis Rheumatol (2015) 67(6):1646–56. doi: 10.1002/art.39107
102. Schauber J, Svanholm C, Termen S, Iffland K, Menzel T, Scheppach W, et al. Expression of the cathelicidin ll-37 is modulated by short chain fatty acids in colonocytes: Relevance of signalling pathways. Gut (2003) 52(5):735–41. doi: 10.1136/gut.52.5.735
103. Burger-van Paassen N, Vincent A, Puiman PJ, van der Sluis M, Bouma J, Boehm G, et al. The regulation of intestinal mucin Muc2 expression by short-chain fatty acids: Implications for epithelial protection. Biochem J (2009) 420(2):211–9. doi: 10.1042/BJ20082222
104. Singh N, Thangaraju M, Prasad PD, Martin PM, Lambert NA, Boettger T, et al. Blockade of dendritic cell development by bacterial fermentation products butyrate and propionate through a transporter (Slc5a8)-dependent inhibition of histone deacetylases. J Biol Chem (2010) 285(36):27601–8. doi: 10.1074/jbc.M110.102947
105. Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature (2013) 504(7480):451–5. doi: 10.1038/nature12726
106. Asquith M, Davin S, Stauffer P, Michell C, Janowitz C, Lin P, et al. Intestinal metabolites are profoundly altered in the context of hla-B27 expression and functionally modulate disease in a rat model of spondyloarthritis. Arthritis Rheumatol (2017) 69(10):1984–95. doi: 10.1002/art.40183
107. Lucas S, Omata Y, Hofmann J, Bottcher M, Iljazovic A, Sarter K, et al. Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat Commun (2018) 9(1):55. doi: 10.1038/s41467-017-02490-4
108. Peng L, Li ZR, Green RS, Holzman IR, Lin J. Butyrate enhances the intestinal barrier by facilitating tight junction assembly Via activation of amp-activated protein kinase in caco-2 cell monolayers. J Nutr (2009) 139(9):1619–25. doi: 10.3945/jn.109.104638
109. Feng W, Wu Y, Chen G, Fu S, Li B, Huang B, et al. Sodium butyrate attenuates diarrhea in weaned piglets and promotes tight junction protein expression in colon in a Gpr109a-dependent manner. Cell Physiol Biochem (2018) 47(4):1617–29. doi: 10.1159/000490981
110. Zheng L, Kelly CJ, Battista KD, Schaefer R, Lanis JM, Alexeev EE, et al. Microbial-derived butyrate promotes epithelial barrier function through il-10 receptor-dependent repression of claudin-2. J Immunol (2017) 199(8):2976–84. doi: 10.4049/jimmunol.1700105
111. Gunzel D, Yu AS. Claudins and the modulation of tight junction permeability. Physiol Rev (2013) 93(2):525–69. doi: 10.1152/physrev.00019.2012
112. Luu M, Weigand K, Wedi F, Breidenbend C, Leister H, Pautz S, et al. Regulation of the effector function of Cd8(+) T cells by gut microbiota-derived metabolite butyrate. Sci Rep (2018) 8(1):14430. doi: 10.1038/s41598-018-32860-x
113. Chang PV, Hao L, Offermanns S, Medzhitov R. The microbial metabolite butyrate regulates intestinal macrophage function Via histone deacetylase inhibition. Proc Natl Acad Sci USA (2014) 111(6):2247–52. doi: 10.1073/pnas.1322269111
114. Chen L, Sun M, Wu W, Yang W, Huang X, Xiao Y, et al. Microbiota metabolite butyrate differentially regulates Th1 and Th17 cells' differentiation and function in induction of colitis. Inflamm Bowel Dis (2019) 25(9):1450–61. doi: 10.1093/ibd/izz046
115. Kechker P, Senderovich Y, Ken-Dror S, Laviad-Shitrit S, Arakawa E, Halpern M. Otitis media caused by v. cholerae O100: A case report and review of the literature. Front Microbiol (2017) 8:1619. doi: 10.3389/fmicb.2017.01619
116. Furusawa Y, Obata Y, Fukuda S, Endo TA, Nakato G, Takahashi D, et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature (2013) 504(7480):446–50. doi: 10.1038/nature12721
117. Singh N, Gurav A, Sivaprakasam S, Brady E, Padia R, Shi H, et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity (2014) 40(1):128–39. doi: 10.1016/j.immuni.2013.12.007
118. Korecka A, Dona A, Lahiri S, Tett AJ, Al-Asmakh M, Braniste V, et al. Bidirectional communication between the aryl hydrocarbon receptor (Ahr) and the microbiome tunes host metabolism. NPJ Biofilms Microbiomes (2016) 2:16014. doi: 10.1038/npjbiofilms.2016.14
119. Hubbard TD, Murray IA, Bisson WH, Lahoti TS, Gowda K, Amin SG, et al. Adaptation of the human aryl hydrocarbon receptor to sense microbiota-derived indoles. Sci Rep (2015) 5:12689. doi: 10.1038/srep12689
120. Shen J, Yang L, You K, Chen T, Su Z, Cui Z, et al. Indole-3-Acetic acid alters intestinal microbiota and alleviates ankylosing spondylitis in mice. Front Immunol (2022) 13:762580. doi: 10.3389/fimmu.2022.762580
121. Zelante T, Iannitti RG, Cunha C, De Luca A, Giovannini G, Pieraccini G, et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity Via interleukin-22. Immunity (2013) 39(2):372–85. doi: 10.1016/j.immuni.2013.08.003
122. Alexeev EE, Lanis JM, Kao DJ, Campbell EL, Kelly CJ, Battista KD, et al. Microbiota-derived indole metabolites promote human and murine intestinal homeostasis through regulation of interleukin-10 receptor. Am J Pathol (2018) 188(5):1183–94. doi: 10.1016/j.ajpath.2018.01.011
123. Ren W, Chen S, Yin J, Duan J, Li T, Liu G, et al. Dietary arginine supplementation of mice alters the microbial population and activates intestinal innate immunity. J Nutr (2014) 144(6):988–95. doi: 10.3945/jn.114.192120
124. Ren W, Yin J, Wu M, Liu G, Yang G, Xion Y, et al. Serum amino acids profile and the beneficial effects of l-arginine or l-glutamine supplementation in dextran sulfate sodium colitis. PloS One (2014) 9(2):e88335. doi: 10.1371/journal.pone.0088335
125. Ren W, Duan J, Yin J, Liu G, Cao Z, Xiong X, et al. Correction to: Dietary l-glutamine supplementation modulates microbial community and activates innate immunity in the mouse intestine. Amino Acids (2021) 53(2):319. doi: 10.1007/s00726-020-02916-2
126. Ma EH, Bantug G, Griss T, Condotta S, Johnson RM, Samborska B, et al. Serine is an essential metabolite for effector T cell expansion. Cell Metab (2017) 25(2):482. doi: 10.1016/j.cmet.2017.01.014
127. Liu G, Ma Y, Yang Q, Deng S. Modulation of inflammatory response and gut microbiota in ankylosing spondylitis mouse model by bioactive peptide iqw. J Appl Microbiol (2020) 128(6):1669–77. doi: 10.1111/jam.14588
128. Liu B, Jiang X, Cai L, Zhao X, Dai Z, Wu G, et al. Putrescine mitigates intestinal atrophy through suppressing inflammatory response in weanling piglets. J Anim Sci Biotechnol (2019) 10:69. doi: 10.1186/s40104-019-0379-9
129. Li G, Ding H, Yu X, Meng Y, Li J, Guo Q, et al. Spermidine suppresses inflammatory dc function by activating the Foxo3 pathway and counteracts autoimmunity. iScience (2020) 23(1):100807. doi: 10.1016/j.isci.2019.100807
130. Wu K, Yuan Y, Yu H, Dai X, Wang S, Sun Z, et al. The gut microbial metabolite trimethylamine n-oxide aggravates gvhd by inducing M1 macrophage polarization in mice. Blood (2020) 136(4):501–15. doi: 10.1182/blood.2019003990
131. Eckle SB, Corbett AJ, Keller AN, Chen Z, Godfrey DI, Liu L, et al. Recognition of vitamin b precursors and byproducts by mucosal associated invariant T cells. J Biol Chem (2015) 290(51):30204–11. doi: 10.1074/jbc.R115.685990
132. Kenna TJ, Hanson A, Costello ME, Brown MA. Functional genomics and its bench-to-Bedside translation pertaining to the identified susceptibility alleles and loci in ankylosing spondylitis. Curr Rheumatol Rep (2016) 18(10):63. doi: 10.1007/s11926-016-0612-x
133. Yamaguchi T, Hirota K, Nagahama K, Ohkawa K, Takahashi T, Nomura T, et al. Control of immune responses by antigen-specific regulatory T cells expressing the folate receptor. Immunity (2007) 27(1):145–59. doi: 10.1016/j.immuni.2007.04.017
134. Kunisawa J, Hashimoto E, Ishikawa I, Kiyono H. A pivotal role of vitamin B9 in the maintenance of regulatory T cells in vitro and in vivo. PloS One (2012) 7(2):e32094. doi: 10.1371/journal.pone.0032094
135. He B, Hoang TK, Wang T, Ferris M, Taylor CM, Tian X, et al. Resetting microbiota by lactobacillus reuteri inhibits T reg deficiency-induced autoimmunity Via adenosine A2a receptors. J Exp Med (2017) 214(1):107–23. doi: 10.1084/jem.20160961
136. Ganesh BP, Hall A, Ayyaswamy S, Nelson JW, Fultz R, Major A, et al. Diacylglycerol kinase synthesized by commensal lactobacillus reuteri diminishes protein kinase c phosphorylation and histamine-mediated signaling in the mammalian intestinal epithelium. Mucosal Immunol (2018) 11(2):380–93. doi: 10.1038/mi.2017.58
137. Singh R, Chandrashekharappa S, Bodduluri SR, Baby BV, Hegde B, Kotla NG, et al. Enhancement of the gut barrier integrity by a microbial metabolite through the Nrf2 pathway. Nat Commun (2019) 10(1):89. doi: 10.1038/s41467-018-07859-7
138. Cook SI, Sellin JH. Review article: Short chain fatty acids in health and disease. Aliment Pharmacol Ther (1998) 12(6):499–507. doi: 10.1046/j.1365-2036.1998.00337.x
139. Louis P, Flint HJ. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human Large intestine. FEMS Microbiol Lett (2009) 294(1):1–8. doi: 10.1111/j.1574-6968.2009.01514.x
140. Ciccia F, Bombardieri M, Rizzo A, Principato A, Giardina AR, Raiata F, et al. Over-expression of paneth cell-derived anti-microbial peptides in the gut of patients with ankylosing spondylitis and subclinical intestinal inflammation. Rheumatol (Oxford) (2010) 49(11):2076–83. doi: 10.1093/rheumatology/keq239
141. Jung TH, Park JH, Jeon WM, Han KS. Butyrate modulates bacterial adherence on Ls174t human colorectal cells by stimulating mucin secretion and mapk signaling pathway. Nutr Res Pract (2015) 9(4):343–9. doi: 10.4162/nrp.2015.9.4.343
142. Ciccia F, Accardo-Palumbo A, Alessandro R, Rizzo A, Principe S, Peralta S, et al. Interleukin-22 and interleukin-22-Producing Nkp44+ natural killer cells in subclinical gut inflammation in ankylosing spondylitis. Arthritis Rheum (2012) 64(6):1869–78. doi: 10.1002/art.34355
143. Zhang M, Zhou Q, Dorfman RG, Huang X, Fan T, Zhang H, et al. Butyrate inhibits interleukin-17 and generates tregs to ameliorate colorectal colitis in rats. BMC Gastroenterol (2016) 16(1):84. doi: 10.1186/s12876-016-0500-x
144. Asarat M, Apostolopoulos V, Vasiljevic T, Donkor O. Short-chain fatty acids regulate cytokines and Th17/Treg cells in human peripheral blood mononuclear cells in vitro. Immunol Invest (2016) 45(3):205–22. doi: 10.3109/08820139.2015.1122613
145. Park J, Kim M, Kang SG, Jannasch AH, Cooper B, Patterson J, et al. Short-chain fatty acids induce both effector and regulatory T cells by suppression of histone deacetylases and regulation of the mtor-S6k pathway. Mucosal Immunol (2015) 8(1):80–93. doi: 10.1038/mi.2014.44
146. Salkowska A, Karas K, Walczak-Drzewiecka A, Dastych J, Ratajewski M. Differentiation stage-specific effect of histone deacetylase inhibitors on the expression of rorgammat in human lymphocytes. J Leukoc Biol (2017) 102(6):1487–95. doi: 10.1189/jlb.6A0617-217R
147. Gracey E, Dumas E, Yerushalmi M, Qaiyum Z, Inman RD, Elewaut D. The ties that bind: Skin, gut and spondyloarthritis. Curr Opin Rheumatol (2019) 31(1):62–9. doi: 10.1097/BOR.0000000000000569
148. Guggino G, Mauro D, Rizzo A, Alessandro R, Raimondo S, Bergot AS, et al. Inflammasome activation in ankylosing spondylitis is associated with gut dysbiosis. Arthritis Rheumatol (2021) 73(7):1189–99. doi: 10.1002/art.41644
149. Feng Y, Wang Y, Wang P, Huang Y, Wang F. Short-chain fatty acids manifest stimulative and protective effects on intestinal barrier function through the inhibition of Nlrp3 inflammasome and autophagy. Cell Physiol Biochem (2018) 49(1):190–205. doi: 10.1159/000492853
150. Goldring SR. Osteoimmunology and bone homeostasis: Relevance to spondyloarthritis. Curr Rheumatol Rep (2013) 15(7):342. doi: 10.1007/s11926-013-0342-2
151. Lee JH, Lee J. Indole as an intercellular signal in microbial communities. FEMS Microbiol Rev (2010) 34(4):426–44. doi: 10.1111/j.1574-6976.2009.00204.x
152. Gutierrez-Vazquez C, Quintana FJ. Regulation of the immune response by the aryl hydrocarbon receptor. Immunity (2018) 48(1):19–33. doi: 10.1016/j.immuni.2017.12.012
153. Funatake CJ, Marshall NB, Steppan LB, Mourich DV, Kerkvliet NI. Cutting edge: Activation of the aryl hydrocarbon receptor by 2,3,7,8-Tetrachlorodibenzo-P-Dioxin generates a population of Cd4+ Cd25+ cells with characteristics of regulatory T cells. J Immunol (2005) 175(7):4184–8. doi: 10.4049/jimmunol.175.7.4184
154. Rutz S, Noubade R, Eidenschenk C, Ota N, Zeng W, Zheng Y, et al. Transcription factor c-maf mediates the tgf-Beta-Dependent suppression of il-22 production in T(H)17 cells. Nat Immunol (2011) 12(12):1238–45. doi: 10.1038/ni.2134
155. Yeste A, Mascanfroni ID, Nadeau M, Burns EJ, Tukpah AM, Santiago A, et al. Il-21 induces il-22 production in Cd4+ T cells. Nat Commun (2014) 5:3753. doi: 10.1038/ncomms4753
156. Venkatesh M, Mukherjee S, Wang H, Li H, Sun K, Benechet AP, et al. Symbiotic bacterial metabolites regulate gastrointestinal barrier function Via the xenobiotic sensor pxr and toll-like receptor 4. Immunity (2014) 41(2):296–310. doi: 10.1016/j.immuni.2014.06.014
157. Lanis JM, Alexeev EE, Curtis VF, Kitzenberg DA, Kao DJ, Battista KD, et al. Tryptophan metabolite activation of the aryl hydrocarbon receptor regulates il-10 receptor expression on intestinal epithelia. Mucosal Immunol (2017) 10(5):1133–44. doi: 10.1038/mi.2016.133
158. Stoll ML, Kumar R, Lefkowitz EJ, Cron RQ, Morrow CD, Barnes S. Fecal metabolomics in pediatric spondyloarthritis implicate decreased metabolic diversity and altered tryptophan metabolism as pathogenic factors. Genes Immun (2016) 17(7):400–5. doi: 10.1038/gene.2016.38
159. Ren W, Yin J, Duan J, Liu G, Tan B, Yang G, et al. Mtorc1 signaling and il-17 expression: Defining pathways and possible therapeutic targets. Eur J Immunol (2016) 46(2):291–9. doi: 10.1002/eji.201545886
160. Ren W, Liu G, Chen S, Yin J, Wang J, Tan B, et al. Melatonin signaling in T cells: Functions and applications. J Pineal Res (2017) 62(3):e12394. doi: 10.1111/jpi.12394
161. Ren W, Liu G, Yin J, Tan B, Wu G, Bazer FW, et al. Amino-acid transporters in T-cell activation and differentiation. Cell Death Dis (2017) 8(5):e2757. doi: 10.1038/cddis.2017.207
162. Tofalo R, Cocchi S, Suzzi G. Polyamines and gut microbiota. Front Nutr (2019) 6:16. doi: 10.3389/fnut.2019.00016
163. Coras R, Kavanaugh A, Boyd T, Huynh D, Lagerborg KA, Xu YJ, et al. Choline metabolite, trimethylamine n-oxide (Tmao), is associated with inflammation in psoriatic arthritis. Clin Exp Rheumatol (2019) 37(3):481–4.
164. Ierardi E, Sorrentino C, Principi M, Giorgio F, Losurdo G, Di Leo A. Intestinal microbial metabolism of phosphatidylcholine: A novel insight in the cardiovascular risk scenario. Hepatobiliary Surg Nutr (2015) 4(4):289–92. doi: 10.3978/j.issn.2304-3881.2015.02.01
165. Magnusdottir S, Ravcheev D, de Crecy-Lagard V, Thiele I. Systematic genome assessment of b-vitamin biosynthesis suggests Co-operation among gut microbes. Front Genet (2015) 6:148. doi: 10.3389/fgene.2015.00148
166. Fragoulis GE, Siebert S, McInnes IB. Therapeutic targeting of il-17 and il-23 cytokines in immune-mediated diseases. Annu Rev Med (2016) 67:337–53. doi: 10.1146/annurev-med-051914-021944
167. Ogrendik M. Treatment of ankylosing spondylitis with moxifloxacin. South Med J (2007) 100(4):366–70. doi: 10.1097/SMJ.0b013e31802fa2a8
168. Clegg DO, Reda DJ, Abdellatif M. Comparison of sulfasalazine and placebo for the treatment of axial and peripheral articular manifestations of the seronegative spondylarthropathies: A department of veterans affairs cooperative study. Arthritis Rheum (1999) 42(11):2325–9. doi: 10.1002/1529-0131(199911)42:11
169. Wang F, Graham WV, Wang Y, Witkowski ED, Schwarz BT, Turner JR. Interferon-gamma and tumor necrosis factor-alpha synergize to induce intestinal epithelial barrier dysfunction by up-regulating myosin light chain kinase expression. Am J Pathol (2005) 166(2):409–19. doi: 10.1016/s0002-9440(10)62264-x
170. Yang L, Liu B, Zheng J, Huang J, Zhao Q, Liu J, et al. Rifaximin alters intestinal microbiota and prevents progression of ankylosing spondylitis in mice. Front Cell Infect Microbiol (2019) 9:44. doi: 10.3389/fcimb.2019.00044
171. Paterson DL, Mulazimoglu L, Casellas JM, Ko WC, Goossens H, Von Gottberg A, et al. Epidemiology of ciprofloxacin resistance and its relationship to extended-spectrum beta-lactamase production in klebsiella pneumoniae isolates causing bacteremia. Clin Infect Dis (2000) 30(3):473–8. doi: 10.1086/313719
172. Nathisuwan S, Burgess DS, Lewis JS 2nd. Extended-spectrum beta-lactamases: Epidemiology, detection, and treatment. Pharmacotherapy (2001) 21(8):920–8. doi: 10.1592/phco.21.11.920.34529
173. Xu MQ, Cao HL, Wang WQ, Wang S, Cao XC, Yan F, et al. Fecal microbiota transplantation broadening its application beyond intestinal disorders. World J Gastroenterol (2015) 21(1):102–11. doi: 10.3748/wjg.v21.i1.102
174. Liu B, Yang L, Cui Z, Zheng J, Huang J, Zhao Q, et al. Anti-Tnf-Alpha therapy alters the gut microbiota in proteoglycan-induced ankylosing spondylitis in mice. Microbiologyopen (2019) 8(12):e927. doi: 10.1002/mbo3.927
175. Ditto MC, Parisi S, Landolfi G, Borrelli R, Realmuto C, Finucci A, et al. Intestinal microbiota changes induced by tnf-inhibitors in ibd-related spondyloarthritis. RMD Open (2021) 7(3):e001755. doi: 10.1136/rmdopen-2021-001755
176. Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, et al. Expert consensus document. the international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol (2014) 11(8):506–14. doi: 10.1038/nrgastro.2014.66
177. Vanderpool C, Yan F, Polk DB. Mechanisms of probiotic action: Implications for therapeutic applications in inflammatory bowel diseases. Inflamm Bowel Dis (2008) 14(11):1585–96. doi: 10.1002/ibd.20525
178. Amdekar S, Singh V, Singh R, Sharma P, Keshav P, Kumar A. Lactobacillus casei reduces the inflammatory joint damage associated with collagen-induced arthritis (Cia) by reducing the pro-inflammatory cytokines: Lactobacillus casei: Cox-2 inhibitor. J Clin Immunol (2011) 31(2):147–54. doi: 10.1007/s10875-010-9457-7
179. Hatakka K, Martio J, Korpela M, Herranen M, Poussa T, Laasanen T, et al. Effects of probiotic therapy on the activity and activation of mild rheumatoid arthritis–a pilot study. Scand J Rheumatol (2003) 32(4):211–5. doi: 10.1080/03009740310003695
180. Jenks K, Stebbings S, Burton J, Schultz M, Herbison P, Highton J. Probiotic therapy for the treatment of spondyloarthritis: A randomized controlled trial. J Rheumatol (2010) 37(10):2118–25. doi: 10.3899/jrheum.100193
181. Land MH, Rouster-Stevens K, Woods CR, Cannon ML, Cnota J, Shetty AK. Lactobacillus sepsis associated with probiotic therapy. Pediatrics (2005) 115(1):178–81. doi: 10.1542/peds.2004-2137
182. Apostolou E, Kirjavainen PV, Saxelin M, Rautelin H, Valtonen V, Salminen SJ, et al. Good adhesion properties of probiotics: A potential risk for bacteremia? FEMS Immunol Med Microbiol (2001) 31(1):35–9. doi: 10.1111/j.1574-695X.2001.tb01583.x
183. Fuentes S, de Vos WM. How to manipulate the microbiota: Fecal microbiota transplantation. Adv Exp Med Biol (2016) 902:143–53. doi: 10.1007/978-3-319-31248-4_10
184. Khoruts A, Sadowsky MJ. Understanding the mechanisms of faecal microbiota transplantation. Nat Rev Gastroenterol Hepatol (2016) 13(9):508–16. doi: 10.1038/nrgastro.2016.98
185. Ishikawa D, Sasaki T, Osada T, Kuwahara-Arai K, Haga K, Shibuya T, et al. Changes in intestinal microbiota following combination therapy with fecal microbial transplantation and antibiotics for ulcerative colitis. Inflammation Bowel Dis (2017) 23(1):116–25. doi: 10.1097/MIB.0000000000000975
186. Hohmann EL, Ananthakrishnan AN, Deshpande V. Case records of the Massachusetts general hospital Case 25-2014. a 37-Year-Old man with ulcerative colitis and bloody diarrhea. N Engl J Med (2014) 371(7):668–75. doi: 10.1056/NEJMcpc1400842
187. Rautava S, Luoto R, Salminen S, Isolauri E. Microbial contact during pregnancy, intestinal colonization and human disease. Nat Rev Gastroenterol Hepatol (2012) 9(10):565–76. doi: 10.1038/nrgastro.2012.144
188. Backhed F, Roswall J, Peng Y, Feng Q, Jia H, Kovatcheva-Datchary P, et al. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe (2015) 17(6):852. doi: 10.1016/j.chom.2015.05.012
189. Shin NR, Whon TW, Bae JW. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol (2015) 33(9):496–503. doi: 10.1016/j.tibtech.2015.06.011
190. Montoya J, Matta NB, Suchon P, Guzian MC, Lambert NC, Mattei JP, et al. Patients with ankylosing spondylitis have been breast fed less often than healthy controls: A case-control retrospective study. Ann Rheum Dis (2016) 75(5):879–82. doi: 10.1136/annrheumdis-2015-208187
191. Zivkovic AM, German JB, Lebrilla CB, Mills DA. Human milk glycobiome and its impact on the infant gastrointestinal microbiota. Proc Natl Acad Sci USA (2011) 108 Suppl 1:4653–8. doi: 10.1073/pnas.1000083107
192. Daniels MC, Adair LS. Breast-feeding influences cognitive development in Filipino children. J Nutr (2005) 135(11):2589–95. doi: 10.1093/jn/135.11.2589
193. German JB, Dillard CJ, Ward RE. Bioactive components in milk. Curr Opin Clin Nutr Metab Care (2002) 5(6):653–8. doi: 10.1097/00075197-200211000-00007
194. Harmsen HJ, Wildeboer-Veloo AC, Raangs GC, Wagendorp AA, Klijn N, Bindels JG, et al. Analysis of intestinal flora development in breast-fed and formula-fed infants by using molecular identification and detection methods. J Pediatr Gastroenterol Nutr (2000) 30(1):61–7. doi: 10.1097/00005176-200001000-00019
195. Marcobal A, Barboza M, Froehlich JW, Block DE, German JB, Lebrilla CB, et al. Consumption of human milk oligosaccharides by gut-related microbes. J Agric Food Chem (2010) 58(9):5334–40. doi: 10.1021/jf9044205
196. Ward RE, Ninonuevo M, Mills DA, Lebrilla CB, German JB. In vitro fermentability of human milk oligosaccharides by several strains of bifidobacteria. Mol Nutr Food Res (2007) 51(11):1398–405. doi: 10.1002/mnfr.200700150
197. Edwards CA, Parrett AM. Intestinal flora during the first months of life: New perspectives. Br J Nutr (2002) 88 Suppl 1:S11–8. doi: 10.1079/BJN2002625
198. David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE, et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature (2014) 505(7484):559–63. doi: 10.1038/nature12820
199. Brown K, DeCoffe D, Molcan E, Gibson DL. Diet-induced dysbiosis of the intestinal microbiota and the effects on immunity and disease. Nutrients (2012) 4(8):1095–119. doi: 10.3390/nu4081095
200. Ebringer A, Wilson C. The use of a low starch diet in the treatment of patients suffering from ankylosing spondylitis. Clin Rheumatol (1996) 15 Suppl 1:62–6. doi: 10.1007/BF03342649
201. Cao G, Wang Q, Huang W, Tong J, Ye D, He Y, et al. Long-term consumption of caffeine-free high sucrose cola beverages aggravates the pathogenesis of eae in mice. Cell Discov (2017) 3:17020. doi: 10.1038/celldisc.2017.20
202. Di Luccia B, Crescenzo R, Mazzoli A, Cigliano L, Venditti P, Walser JC, et al. Rescue of fructose-induced metabolic syndrome by antibiotics or faecal transplantation in a rat model of obesity. PloS One (2015) 10(8):e0134893. doi: 10.1371/journal.pone.0134893
203. Macfarlane TV, Abbood HM, Pathan E, Gordon K, Hinz J, Macfarlane GJ. Relationship between diet and ankylosing spondylitis: A systematic review. Eur J Rheumatol (2018) 5(1):45–52. doi: 10.5152/eurjrheum.2017.16103
204. Andoh A, Bamba T, Sasaki M. Physiological and anti-inflammatory roles of dietary fiber and butyrate in intestinal functions. JPEN J Parenter Enteral Nutr (1999) 23(5 Suppl):S70–3. doi: 10.1177/014860719902300518
205. Looijer-van Langen MA, Dieleman LA. Prebiotics in chronic intestinal inflammation. Inflamm Bowel Dis (2009) 15(3):454–62. doi: 10.1002/ibd.20737
206. Christl SU, Eisner HD, Dusel G, Kasper H, Scheppach W. Antagonistic effects of sulfide and butyrate on proliferation of colonic mucosa: A potential role for these agents in the pathogenesis of ulcerative colitis. Dig Dis Sci (1996) 41(12):2477–81. doi: 10.1007/BF02100146
207. Tan J, McKenzie C, Vuillermin PJ, Goverse G, Vinuesa CG, Mebius RE, et al. Dietary fiber and bacterial scfa enhance oral tolerance and protect against food allergy through diverse cellular pathways. Cell Rep (2016) 15(12):2809–24. doi: 10.1016/j.celrep.2016.05.047
208. Zhernakova A, Kurilshikov A, Bonder MJ, Tigchelaar EF, Schirmer M, Vatanen T, et al. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science (2016) 352(6285):565–9. doi: 10.1126/science.aad3369
209. Willett WC, Sacks F, Trichopoulou A, Drescher G, Ferro-Luzzi A, Helsing E, et al. Mediterranean Diet pyramid: A cultural model for healthy eating. Am J Clin Nutr (1995) 61(6 Suppl):1402S–6S. doi: 10.1093/ajcn/61.6.1402S
210. Sureda A, Bibiloni MDM, Julibert A, Bouzas C, Argelich E, Llompart I, et al. Adherence to the Mediterranean diet and inflammatory markers. Nutrients (2018) 10(1):62. doi: 10.3390/nu10010062
211. Dinu M, Pagliai G, Casini A, Sofi F. Mediterranean Diet and multiple health outcomes: An umbrella review of meta-analyses of observational studies and randomised trials. Eur J Clin Nutr (2018) 72(1):30–43. doi: 10.1038/ejcn.2017.58
212. Niu Q, Wei W, Huang Z, Zhang J, Yang B, Wang L. Association between food allergy and ankylosing spondylitis: An observational study. Med (Baltimore) (2019) 98(6):e14421. doi: 10.1097/MD.0000000000014421
213. Feng W, Ao H, Peng C. Gut microbiota, short-chain fatty acids, and herbal medicines. Front Pharmacol (2018) 9:1354. doi: 10.3389/fphar.2018.01354
214. Gao B, Wang R, Peng Y, Li X. Effects of a homogeneous polysaccharide from sijunzi decoction on human intestinal microbes and short chain fatty acids in vitro. J Ethnopharmacol (2018) 224:465–73. doi: 10.1016/j.jep.2018.06.006
Keywords: ankylosing spondylitis, HLA-B27, microbiota, microbiome, microbial metabolites
Citation: Song Z-Y, Yuan D and Zhang S-X (2022) Role of the microbiome and its metabolites in ankylosing spondylitis. Front. Immunol. 13:1010572. doi: 10.3389/fimmu.2022.1010572
Received: 03 August 2022; Accepted: 27 September 2022;
Published: 13 October 2022.
Edited by:
James Cheng-Chung Wei, Chung Shan Medical University Hospital, TaiwanReviewed by:
Tangyou Mao, Beijing University of Chinese Medicine, ChinaCopyright © 2022 Song, Yuan and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sheng-Xiao Zhang, c2hlbmd4aWFvX3poYW5nQDE2My5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.