- Department of Rheumatology, Affiliated Hospital of Nantong University, Nantong, China
The pandemic of coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2 has caused a large number of deaths, and there is still no effective treatment. COVID-19 can induce a systemic inflammatory response, and its clinical manifestations are diverse. Recently, it has been reported that COVID-19 patients may develop myositis and interstitial pulmonary disease similar to dermatomyositis (DM). This condition is similar to the rapidly progressive interstitial lung disease associated with MDA5+ DM that has a poor prognosis and high mortality, and this poses a challenge for an early identification. Suppression of the immune system can protect COVID-19 patients by preventing the production of inflammatory cytokines. This article attempts to explore the possibility of a relationship between COVID-19 and DM in terms of the potential pathogenesis and clinical features and to analyze the therapeutic effect of the immunosuppressive drugs that are commonly used for the treatment of both DM and COVID-19.
Introduction
In December 2019, a novel infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was reported (1). Compared with SARS-CoV and MERS-CoV, SARS-CoV-2 is more infectious; therefore, coronavirus disease 2019 (COVID-19) has become a global epidemic. The most common symptoms of COVID-19 are fever, cough, dyspnea, myalgia, and fatigue (2), and pulmonary involvement is significantly associated with a poor prognosis and a high mortality. Viruses can cause a variety of autoimmune diseases, and patients with autoimmune diseases are more susceptible to infection due to the pathogenesis of the autoimmune disease and/or the use of immunosuppressants. Among the potential autoimmune diseases that may be related to COVID-19, the most intriguing is idiopathic inflammatory myopathy (IIM), a heterogeneous disease that primarily affects the skeletal muscles and can be divided into polymyositis, dermatomyositis (DM), immune-mediated necrotizing myopathy, anti-synthase syndrome, and inclusion body myositis (3). During the COVID-19 outbreak, several studies have recognized a striking similarity between COVID-19 and DM due to the lung and muscle involvement and the presence of a rash. DM typically presents with characteristic skin manifestations that accompany or precede muscle weakness, and interstitial lung disease (ILD) is a common complication of DM. The pathogenesis of DM is still unclear, although it is likely associated with inappropriate complement activation and interferon (IFN) response (4, 5), which results in the production of myositis-specific autoantibodies that may be related to unique clinical features. Obviously, COVID-19 is closely related to DM.
COVID-19 and Autoimmunity
For many infections, the immune system provides an appropriate response that mediates resistance to the invading microorganism. Appropriate innate and adaptive responses promote the coordinated production of proinflammatory cytokines that can control pathogens. Several environmental factors have been suspected to trigger or even exacerbate existing autoimmune conditions in genetically susceptible patients, and these factors include viral, bacterial, and parasitic infections (6). Viruses can induce autoimmunity in genetically susceptible individuals through multiple mechanisms, including molecular mimicry, epitope spreading, bystander activation, and the immortalization of infected B cells. Chronic relapsing/reactivated infections caused by Epstein-Barr virus (EBV) are associated with the occurrence or onset of various autoimmune diseases, including DM (7). Similar to SARS-CoV, SARS-CoV-2 uses the membranous angiotensin-converting enzyme 2 (ACE2) receptors to enter human cells (8). Then, SARS-CoV-2 can trigger innate and adaptive immune responses, known as cytokine storms. This abnormal elevation of inflammatory cytokines, such as interleukin (IL)-1, IL-6, IL-10, interferon γ (IFN-γ), monocyte chemotactic protein-1 (MCP-1), and granulocyte-macrophage colony-stimulating factor, can interact with the complement and coagulation systems, leading to acute respiratory distress syndrome, disseminated intravascular coagulation, and even multiple organ failure (9). Li et al. demonstrated significant expression of ACE2 receptors on alveolar epithelial cells, which may explain why pulmonary involvement is a hallmark of COVID-19 (10). A retrospective study revealed that SARS-CoV-2 might act primarily on lymphocytes, especially T lymphocytes. Moreover, patients with severe COVID-19 had higher neutrophils and fewer lymphocytes than patients with mild COVID-19, indicating that an increased neutrophil-to-lymphocyte ratio (NLR) in patients with severe COVID-19 and the use of lymphocyte subset monitoring are helpful for early screening, diagnosis, and treatment of severe COVID-19 (11).
Pathogenesis and Clinical Characteristics of DM
DM is a rare disease characterized by distinct cutaneous manifestations and a clinically heterogeneous systemic presentation. Pathognomonic skin manifestations such as Gottron’s papules and heliotrope rash are conducive to the early diagnosis of DM. Therefore, DM can be difficult to diagnose in the absence of the characteristic dermatologic features. DM has been suggested to occur more frequently in women and African Americans (12), and the age range of the patients at the time of the diagnosis of DM is bimodal, with the peak frequencies occurring in 5- to 14-year-old children and in 40- to 60-year-old adults (13). Epidemiological data has demonstrated that subsets of DM patients that were diagnosed by the presence of different myositis-specific autoantibodies tend to be diagnosed at specific times of the year (14). The pathogeneses of the different types and subtypes of myositis are incompletely understood. Predisposition to IIM are attributable to genetic and environmental factors, and viral or bacterial infections may induce the occurrence of the disease. The seasonal clustering of symptoms suggests that viral or bacterial infections could be common environmental triggers. A case-control study showed that a higher number EBV genomes are present and that anti-Epstein-Barr nuclear antigen 1 antibodies were detected at a higher frequency in DM patients than in healthy control counterparts (15). Autoimmune mechanisms are important in the pathogenesis of IIM, and myositis-specific or associated autoantibodies can be observed in patients with IIM, which is often associated with particular clinical features (16). With the further study of DM, many novel autoantibodies against myositis have been found, including anti-melanoma differentiation-associated gene 5 (MDA5), anti-transcriptional intermediary factor 1 γ, anti-nuclear matrix protein 2, anti-Ku and anti-Mi-2 (17). Anti-MDA5+ DM is typically associated with rapidly progressive interstitial lung disease (RP-ILD), which has a high early mortality (18). It is still controversial whether the immune activation seen in DM is antibody-dependent or is triggered by a classical complement cascade (4). The IFN pathway is activated in the different clinical subtypes of myositis, and type 1 IFN1 is most upregulated in DM patients (5). The expression level of IFN-induced genes was correlated with the indicators of DM disease activity (19). Three different ligand families activate the IFN pathway by binding to cell surface receptors and by stimulating the expression of IFN-inducible genes via the Janus kinase (JAK)/signal transducer and the activator of transcription (STAT) signaling pathway (20). A persistent IFN response promotes antigen presentation and lymphocyte responses and induces chemokine expression, and the resulting T and B cell activation may also be responsible for the production of autoantibodies (20).
Approximately 80% of DM patients have myopathy, which usually presents as proximal muscle weakness (4). Extramuscular involvement is common in IIM; the skin, lungs, joints, heart, and gastrointestinal tract can be affected in IIM; and the degree of involvement varies according to the different subtypes of IIM (21). ILD is a common complication of DM, and the severity of the pulmonary symptoms varies among patients with different subtypes (22). Patients with mild ILD are stable and respond well to treatment, while some patients with RP-ILD have more severe disease and have a poor prognosis (23, 24). In addition, the extent of muscle involvement can also be fatal for DM patients because weakness of the intercostal muscles may lead to respiratory disorders and acute respiratory distress syndrome (25).
Correlation on COVID-19 and DM
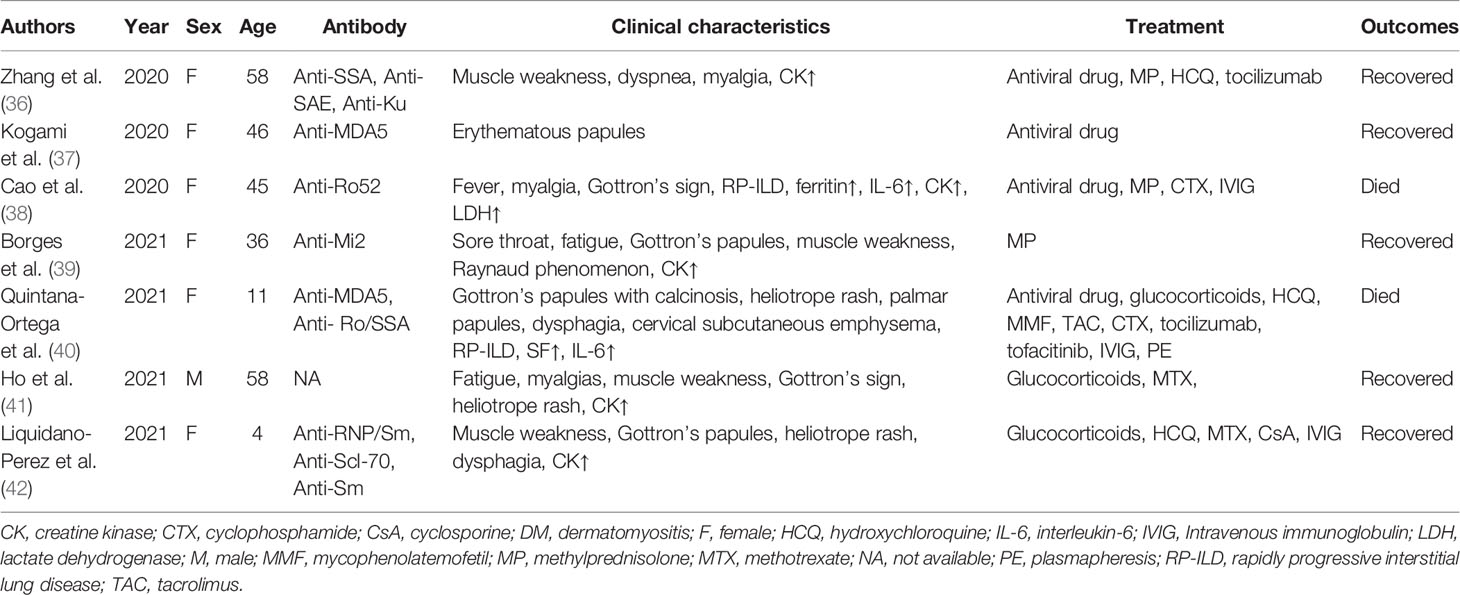
The similarity of COVID-19 and DM implies a common underlying mechanism (Figure 1). MDA5, a myositis-specific autoantibody, is an intracellular sensor of the intermediates or byproducts of double-stranded RNA viral replication that trigger the innate response and subsequent production of cytokines that activate macrophages and helper T cells (26). Increased levels of several cytokines, such as IFN-γ, IL-8, and MCP-1 (which are similar to the cytokine storms seen in severe COVID-19 patients), were found in patients with anti-MDA5+ DM (27). IFN1, which plays a major role in the muscle fiber damage in DM patients, is involved in the organ damage seen in COVID-19 (28). Megremis et al. proposed that DM patients have three linear epitopes of immunogenicity that have a high sequence identity with the SARS-CoV-2 protein; thus, a potential exposure to a virus within the coronavirus family may lead to the development of DM (29). An Italian patient study reported a higher prevalence of COVID-19 in patients with autoimmune diseases than in the general population (30). Movahedi et al. noticed a sudden outbreak of new cases of juvenile dermatomyositis during the COVID-19 pandemic, with a higher incidence among women (31). Notably, 76.5% of COVID-19 patients are women (30), and the ratio of males to females in DM is 1:2 (4). Autoimmune diseases are generally more prevalent in women than men, with the strongest sex biases seen in Sjogren’s syndrome, systemic lupus erythematosus, autoimmune thyroid disease, and scleroderma (32). Estrogen, especially 17-β estradiol and prolactin, can act as humoral immune enhancers, which causes women to be more susceptible to autoimmune diseases (33). In addition, because of incomplete X chromosome inactivation caused by random silencing in females during the early stages of embryogenesis, several immune-related genes may be upregulated, and their overexpression may influence the immune response (34, 35). Several cases of DM associated with COVID-19 have been summarized (Table 1). Of the seven patients that were reported to have both DM and COVID-19, 85.7% were female, and two of these patients were anti-MDA5+. Most patients had distinct cutaneous manifestations, with elevated CK. In addition, the female mortality was 33.3%, even after receiving aggressive treatment. Based on our summary table, we should raise awareness of the fact that women are more susceptible to COVID-19 combined with DM and experience more severe conditions, and effective treatment interventions would help to improve survival. It is increasingly recognized that the features of severe COVID-19 are similar to those of DM, especially anti-MDA5+ DM because these conditions have similar involvement of the lung and are associated with the development of rash, fatigue, and myalgia. In addition, the imaging findings of COVID-19 are comparable to those of the ILD seen in anti-MDA5+ DM because diffuse ground-glass opacifications are often present in both of these diseases, and the presence of the ground-glass opacifications suggest peribronchovascular consolidation (26). Compared with other DM subtypes, the RP-ILD seen in anti-MDA5+ DM usually develops rapidly, rarely relapses, and responds poorly to glucocorticoids and immunosuppressants, which seems to be similar to a viral infection (27). In recent case reports, autoantibodies against myositis, such as anti-MDA5, anti-SAE, anti-Mi2, anti-Ku, and anti-Ro52, have been detected in COVID-19 patients with DM. Viral infections seem to be associated with certain subtypes of DM.
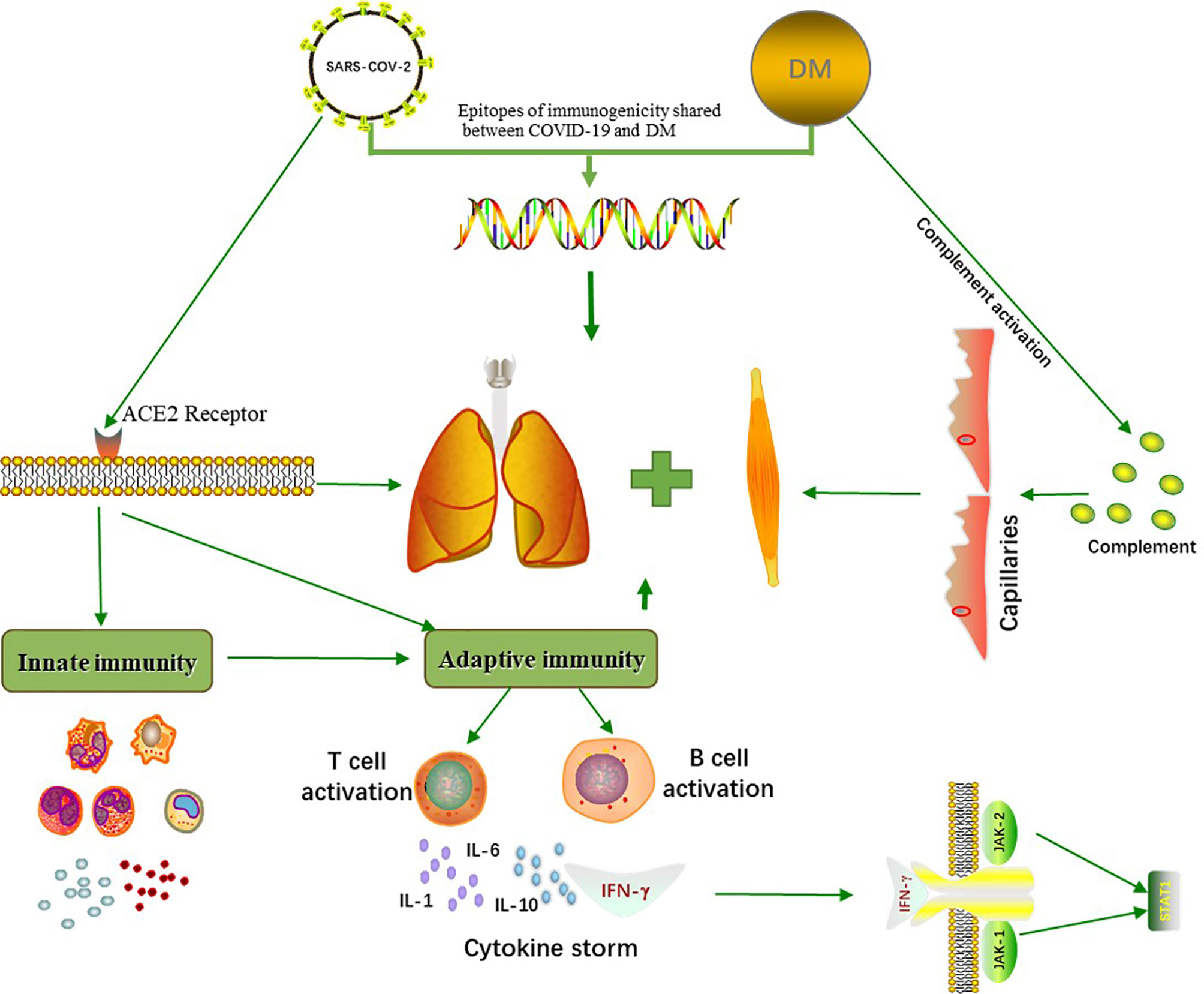
Figure 1 Common pathogenetic mechanisms between COVID-19 and DM. COVID-19 and DM share three immunogenic linear epitopes with high sequence consistency. SARS-CoV-2 enters human cells through the membranous angiotensin-converting enzyme 2 (ACE2) receptors, triggering an innate and adaptive immune response. This initiates the production of cytokines such as IL-1, IL-6, IL-10 and IFN-γ, which can induce lung and muscle damage. The activation of complement in DM patients results in capillary destruction, which further affects multiple organs, and cytokine storms are associated with the occurrence of certain subtypes of DM.
More than 10% of COVID-19 patients were reported to have muscular symptoms and elevated CK levels (43). Muscle biopsies from patients with COVID-19 show abnormal presence of Myxovirus resistance protein A in muscle fibers and capillaries. Myxovirus resistance protein A is one of the IFN1-inducible proteins, is overexpressed in biopsy muscle specimens from DM patients and may be a more sensitive marker of DM (44, 45), suggesting that autoimmune myositis may be caused by COVID-19. The SARS-CoV-2 receptor ACE2 is expressed in a variety of human tissues, including the skeletal muscles, where ACE2 expression is the lowest (10). The expression and distribution of ACE2 remind us that SARS-CoV-2 may cause muscle injury through direct or indirect mechanisms. Beydon et al. reported a case of this type of myositis diagnosed by MRI (46). However, SARS-CoV, which has the same receptor as SARS-CoV-2, was not found in the muscle tissue of the patient at autopsy (47, 48). In addition, the muscle manifestations of COVID-19 patients can be contributed to rhabdomyolysis caused by SARS-CoV-2 (49). Rhabdomyolysis is a life-threatening disease that requires aggressive hydration in order to avoid acute renal failure, which can worsen the oxygenation status in COVID-19 patients (49, 50). Therefore, when patients have focal muscle pain and fatigue, the possibility of rhabdomyolysis should be taken seriously (50). Symptoms of myopathy in severe systemic viral diseases are multifactorial, and further muscle biopsies and antibody screening are required.
Treatment Options
Since hyperinflammation underlies COVID-19 and is associated with the disease severity, anti-inflammatory therapy may be beneficial to patients. Immune impairment may inhibit viral-induced cytokine storm syndrome (11). Immunosuppressive agents have a variety of mechanisms targeting the cellular and humoral immune responses, so the use of immunosuppressive agents may have a protective effect for COVID-19 patients. Along with the increased understanding of COVID-19, several drugs commonly used to treat DM have potential therapeutic effects for COVID-19 patients.
Glucocorticoids
Glucocorticoids inhibit inflammatory cytokines and reduce the proliferation and differentiation of lymphocytes and macrophages, and they have immunosuppressive effects on the body and may increase susceptibility to COVID-19 (51). There is no evidence that patients infected with COVID-19 benefit from glucocorticoids, and COVID-19 patients may be more prone to the side effects of glucocorticoid treatment (52). However, retrospective analyses have shown that the use of glucocorticoids reduces hospital mortality in patients with COVID-19 cytokine storms (53). Dexamethasone has been shown to reduce the mortality in patients with COVID-19 who receive invasive mechanical ventilation (54). Strangfeld et al. reported a higher mortality rate in patients receiving higher dosages of glucocorticoids (>10 mg/day prednisolone equivalent dose) than in those who did not receive glucocorticoids (55). The efficacy of glucocorticoids in COVID-19 is controversial, especially in terms of doses and risk of side effects.
Conventional Synthetic Disease-Modifying Antirheumatic Drugs
Some conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), such as methotrexate, hydroxychloroquine (HCQ), and azathioprine, suppress the cytokine storm associated with COVID-19 (11). Methotrexate, a dihydrofolate reductase inhibitor, is widely used in autoimmune diseases because of its pleiotropic therapeutic effects on various immune cells and mediators and its inhibition of the body’s overall inflammatory response (56). In patients with DM, the use of HCQ can effectively improve the skin lesions. Chloroquine (CQ) blocks the fusion of the virus to the host cell by interfering with the terminal glycosylation of the cell receptor, ACE2, and by inhibiting sialic acid biosynthesis, which is used as a receptor by the viruses through inhibiting quinine reductase-2 (57). On the other hand, CQ can also play an antiviral role by reducing IFN1 (51). Wang et al. found that CQ played an important role in multiple stages of COVID-19 in vitro, and it had immunomodulatory effects that synergically enhanced its antiviral effects (58). However, the mortality of COVID-19 patients treated with HCQ increased, and there was no benefit from CQ (59). In addition, adverse drug reactions associated with CQ and HCQ increased dramatically during the COVID-19 pandemic (60). Mycophenolate mofetil, calcineurin inhibitors, and cyclophosphamide as immunosuppressants have also shown a good therapeutic effect in the treatment of myositis. Mycophenolate mofetil is an inhibitor of purine synthesis that inhibits inosine monophosphate dehydrogenase, reduces lymphocyte proliferation, and has antifibrosis properties (61). Mycophenolate mofetil showed anti-SARS-COV-2 activity and it could be worth considering used for clinical treatment of COVID-19 (62). In addition, tacrolimus and cyclosporine produced dose-dependent inhibitory effects on NK cell degranulation and IFN-γ in vitro (63), and tacrolimus also reduced the ability of dendritic cells to stimulate T cells, resulting in decreased production of CXCL-10 and IL-12 (64). Tacrolimus has a positive effect on survival in liver transplant patients with symptoms of COVID-19 compared to other immunosuppressants, including cyclosporine and mycophenolate mofetil (65). Nevertheless, there was no improvement in severe COVID-19 patients who received a combination of methylprednisolone pulses and tacrolimus (66). A prospective study demonstrated that a combination of high-dose glucocorticoids, tacrolimus and intravenous cyclophosphamide improved survival in anti-MDA5+ DM patients complicated with ILD. This regimen also has an increased risk of opportunistic infections, leading to an exacerbation of ILD (67). Some csDMARDs, such as methotrexate, mycophenolate mofetil, and tacrolimus may provide a therapeutic strategy for COVID-19. The efficacy and safety of CQ and HCQ remain controversial and need further research.
Interleukin Receptor Antagonists
Considering the mechanism of COVID-19, cytokine-targeting biologicals and signaling molecule inhibitors are also promising therapeutic approaches (11). Anakinra is a recombinant IL-1 receptor antagonist that can be used to treat autoinflammatory diseases (51). Zong et al. demonstrated that patients with myositis may respond to anakinra, especially DM patients with skin rash (68). Currently, a study is underway that combines anakinra with anti-IFN-γ antibodies for the treatment of patients with COVID-19 (51). IL-6 is a pleiotropic cytokine that plays a key role in the cytokine storm. The IL-6 receptor antagonist tocilizumab is used for the treatment of refractory juvenile DM (69). Tocilizumab may be a salvage therapy for anti-MDA5+ DM associated with RP-ILD patients refractory to an intensive immunosuppressive regimen (70). A retrospective study of patients with severe COVID-19 showed that tocilizumab improved the clinical symptoms and that the early use of tocilizumab effectively controlled the progression of the symptoms (71). Another study also demonstrated that corticosteroids combined with tocilizumab or anakinra reduced mortality in hospitalized patients with COVID-19 and that corticosteroids combined with tocilizumab had better survival outcomes (53). Interleukin receptor antagonists anakinra and tocilizumab may be alternative drugs for COVID-19 patients, but given the intrinsic limitations of retrospective studies, randomized clinical trials are still warranted.
JAK Inhibitors
JAK inhibitors are novel target synthetic immunoregulatory agents that not only inhibit tyrosine kinases, which are involved in intracellular viral transport and epithelial endocytosis, but also inhibit intracellular signal transduction of various inflammatory cytokines (51). A patient with refractory JDM was described as having clinical improvement after treatment with baricitinib. The JAK inhibitor baricitinib binds to cyclin G-associated kinases, a regulator of endocytosis, to block the entry of SARS-CoV-2 into cells and the inflammatory cytokine storm (72). A randomized controlled trial demonstrated that baricitinib combined with remdesivir reduced the recovery time in COVID-19 patients, especially in patients receiving high-flow oxygen or noninvasive ventilation, and it also accelerated the clinical improvement of patients and was associated fewer serious adverse events (73). In the early phase of COVID-19, baricitinib may prevent an excessive inflammatory response and the rapid progression of respiratory failure (74). Kurasawa et al. proposed that tofacitinib combined with conventional treatment might control refractory MDA5+ DM complicated with ILD (75). Furthermore, tofacitinib improved the survival 6 months after the onset of anti-MDA5+ amyopathic DM-associated ILD (76). In a randomized controlled trial, tofacitinib reduced the incidences of mortality and respiratory failure in patients with COVID-19 (77). Baricitinib and tofacitinib can cause thrombosis, and patients with COVID-19 are in hypercoagulable states; therefore, careful monitoring for the development of thrombosis during treatment with JAK inhibitors is necessary (78). JAK inhibitors, such as baricitinib and tofacitinib could be available treatments for COVID-19, thrombosis should be paid extensive attention to.
Intravenous Immunoglobulin and Plasmapheresis
Intravenous immunoglobulin (IVIG) is effective in the treatment of refractory DM (79). IVIG improves patient strength, reduces mortality, and promotes recovery. There is a potential for patients with COVID-19 to respond to early immunotherapy, especially IVIG, which may also provide a variety of potential protective antibodies and anti-cytokine effects (80). Elevated cytokine levels are found in the sera of DM patients with ILD, and plasmapheresis is an option to remove the elevated cytokines as an additional supportive treatment (27, 67). Immunoglobulin and plasmapheresis might be considered in patients affected with SARS-CoV-2. A retrospective study demonstrated that COVID-19 patients who were not treated with DMARDs had a higher mortality rate than those who received methotrexate monotherapy, while leflunomide, antimalarials, TNF inhibitors, abatacept, belimumab, and IL-6 inhibitors were not associated with an increased risk of death (55). The efficacy of high-dose corticosteroids, IVIG, JAK inhibitors, and T-cell modulators for COVID-19 has been reported in a small series of cases, and clinical trials are currently under investigation (81).
Conclusion and Perspectives
During the recent pandemic, several cases of COVID-19 were reported to be related to DM. In this review article, we discussed the possible relationship between COVID-19 and DM based on the similar potential pathogenic mechanisms and clinical manifestations. More importantly, we provide a direction for the treatment of COVID-19 through an analysis of immunosuppressive agents that are commonly used for DM. We have highlighted that the manifestations of DM, such as ILD and myositis, can also be observed in patients with COVID-19. Therefore, these manifestations of DM are difficult to distinguish in the early clinical stages, and even certain subtypes of DM my involve the same immune response that is caused by COVID-19. It is noteworthy that women have been more susceptible to DM during the COVID-19 pandemic and have more severe clinical symptoms, which means that extensive clinical attention should be given to the possibility of autoimmune diseases in female patients with COVID-19 and that the treatment regimens should be more aggressive in these patients.
In conclusion, infection with SARS-CoV-2 can induce the occurrence of DM. COVID-19 and DM potentially have a common pathogenesis, such as IFN1, which not only participates in organ damage of COVID-19 but also mediates muscle fiber damage in patients with DM. Although there have been some studies supporting the relationship between COVID-19 and DM, the exact mechanism is largely unknown, and more evidence is needed to confirm this association. Routinely available drugs for DM have provided an alternative therapeutic strategy for COVID-19. Further studies should be done to assess the efficacy and safety of the regimen to tackle the inflammatory stages of COVID-19. We recommend that all newly diagnosed DM cases should be tested for COVID-19 during the pandemic, especially for certain subtypes of DM patients, such as anti-MDA5+, and early differential diagnosis is helpful to improve the survival of these patients.
Author Contributions
JQ: study design, data analysis, and manuscript revision. HX: literature search, data collection, and article writing. All authors contributed to the article and approved the submitted version.
Funding
This project was sponsored by grants from China International Medical Foundation (Z-2018-40), Nantong Science and Technology Bureau Project (MSZ18108 and JCZ20058), and the Special Clinical Basic Research Key Project of Nantong University (2019JZ001).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A Pneumonia Outbreak Associated With a New Coronavirus of Probable Bat Origin. Nat (2020) 579:270–3. doi: 10.1038/s41586-020-2012-7
2. Meo SA, Alhowikan AM, Al-Khlaiwi T, Meo IM, Halepoto DM, Iqbal M, et al. Novel Coronavirus 2019-Ncov: Prevalence, Biological and Clinical Characteristics Comparison With SARS-CoV and MERS-CoV. Eur Rev Med Pharmacol Sci (2020) 24:2012–9. doi: 10.26355/eurrev_202002_20379
3. Vencovský J, Alexanderson H, Lundberg IE. Idiopathic Inflammatory Myopathies. Rheum Dis Clin North Am (2019) 45:569–81. doi: 10.1016/j.rdc.2019.07.006
4. DeWane ME, Waldman R, Lu J. Dermatomyositis: Clinical Features and Pathogenesis. J Am Acad Dermatol (2020) 82:267–81. doi: 10.1016/j.jaad.2019.06.1309
5. Pinal-Fernandez I, Casal-Dominguez M, Derfoul A, Pak K, Plotz P, Miller FW, et al. Identification of Distinctive Interferon Gene Signatures in Different Types of Myositis. Neurol (2019) 93:e1193–204. doi: 10.1212/wnl.0000000000008128
6. Bax CE, Maddukuri S, Ravishankar A, Pappas-Taffer L, Werth VP. Environmental Triggers of Dermatomyositis: A Narrative Review. Ann Transl Med (2021) 9:434. doi: 10.21037/atm-20-3719
7. Houen G, Trier NH. Epstein-Barr Virus and Systemic Autoimmune Diseases. Front Immunol (2021) 11:587380. doi: 10.3389/fimmu.2020.587380
8. Zhang H, Penninger JM, Li Y, Zhong N, Slutsky AS. Angiotensin-Converting Enzyme 2 (ACE2) as a SARS-CoV-2 Receptor: Molecular Mechanisms and Potential Therapeutic Target. Intensive Care Med (2020) 46:586–90. doi: 10.1007/s00134-020-05985-9
9. Mangalmurti N, Hunter CA. Cytokine Storms: Understanding COVID-19. Immun (2020) 53:19–25. doi: 10.1016/j.immuni.2020.06.017
10. Li MY, Li L, Zhang Y, Wang XS. Expression of the SARS-CoV-2 Cell Receptor Gene ACE2 in a Wide Variety of Human Tissues. Infect Dis Poverty (2020) 9:45. doi: 10.1186/s40249-020-00662-x
11. Lu C, Li S, Liu Y. Role of Immunosuppressive Therapy in Rheumatic Diseases Concurrent With COVID-19. Ann Rheum Dis (2020) 79:737–9. doi: 10.1136/annrheumdis-2020-217460
12. Findlay AR, Goyal NA, Mozaffar T. An Overview of Polymyositis and Dermatomyositis. Muscle Nerve (2015) 51:638–56. doi: 10.1002/mus.24566
13. Aussy A, Boyer O, Cordel N. Dermatomyositis and Immune-Mediated Necrotizing Myopathies: A Window on Autoimmunity and Cancer. Front Immunol (2017) 8:992. doi: 10.3389/fimmu.2017.00992
14. Nishina N, Sato S, Masui K, Gono T, Kuwana M. Seasonal and Residential Clustering at Disease Onset of Anti-MDA5-Associated Interstitial Lung Disease. RMD Open (2020) 6:e001202. doi: 10.1136/rmdopen-2020-001202
15. Chen DY, Chen YM, Lan JL, Chen HH, Hsieh CW, Wey SJ, et al. Polymyositis/dermatomyositis and Nasopharyngeal Carcinoma: The Epstein-Barr Virus Connection? J Clin Virol (2010) 49:290–5. doi: 10.1016/j.jcv.2010.08.015
16. Betteridge Z, Tansley S, Shaddick G, Chinoy H, Cooper RG, New RP, et al. Frequency, Mutual Exclusivity and Clinical Associations of Myositis Autoantibodies in a Combined European Cohort of Idiopathic Inflammatory Myopathy Patients. J Autoimmun (2019) 101:48–55. doi: 10.1016/j.jaut.2019.04.001
17. Betteridge Z, McHugh N. Myositis-Specific Autoantibodies: An Important Tool to Support Diagnosis of Myositis. J Intern Med (2016) 280:8–23. doi: 10.1111/joim.12451
18. Gono T, Sato S, Kawaguchi Y, Kuwana M, Hanaoka M, Katsumata Y, et al. Anti-MDA5 Antibody, Ferritin and IL-18 are Useful for the Evaluation of Response to Treatment in Interstitial Lung Disease With Anti-MDA5 Antibody-Positive Dermatomyositis. Rheumatol (Oxford) (2012) 51:1563–70. doi: 10.1093/rheumatology/kes102
19. Walsh RJ, Kong SW, Yao Y, Jallal B, Kiener PA, Pinkus JL, et al. Type I Interferon-Inducible Gene Expression in Blood Is Present and Reflects Disease Activity in Dermatomyositis and Polymyositis. Arthritis Rheumatol (2007) 56:3784–92. doi: 10.1002/art.22928
20. Ivashkiv LB, Donlin LT. Regulation of Type I Interferon Responses. Nat Rev Immunol (2014) 14:36–49. doi: 10.1038/nri3581
21. Dalakas MC, Hohlfeld R. Polymyositis and Dermatomyositis. Lancet (2003) 362:971–82. doi: 10.1016/S0140-6736(03)14368-1
22. Fathi M, Lundberg IE, Tornling G. Pulmonary Complications of Polymyositis and Dermatomyositis. Semin Respir Crit Care Med (2007) 28:451–8. doi: 10.1055/s-2007-985666
23. Selva-O'Callaghan A, Pinal-Fernandez I, Trallero-Araguás E, Milisenda JC, Grau-Junyent JM, Mammen AL. Classification and Management of Adult Inflammatory Myopathies. Lancet Neurol (2018) 17:816–28. doi: 10.1016/S1474-4422(18)30254-0
24. Sato S, Kuwana M. Clinically Amyopathic Dermatomyositis. Curr Opin Rheumatol (2010) 22:639–43. doi: 10.1097/BOR.0b013e32833f1987
25. Selva-O'Callaghan A, Sanchez-Sitjes L, Muñoz-Gall X, Mijares-Boeckh-Behrens T, Solans-Laque R, Angel Bosch-Gil J, et al. Respiratory Failure Due to Muscle Weakness in Inflammatory Myopathies: Maintenance Therapy With Home Mechanical Ventilation. Rheumatol (Oxford) (2000) 39:914–6. doi: 10.1093/rheumatology/39.8.914
26. Mehta P, Machado PM, Gupta L. Understanding and Managing Anti-MDA 5 Dermatomyositis, Including Potential COVID-19 Mimicry. Rheumatol Int (2021) 41:1021–36. doi: 10.1007/s00296-021-04819-1
27. Xu L, Wang L, Lv C, Tan W. Anti-MDA-5-Positive Dermatomyositis Associated Rapidly Progressive Interstitial Lung Disease, a Virus-Triggered Autoimmune-Like Symptom? Rheumatol (Oxford) (2021) 60:4428–9. doi: 10.1093/rheumatology/keab224
28. Tanboon J, Nishino I. COVID-19-Associated Myositis may be Dermatomyositis. Muscle Nerve (2021) 63:E9–e10. doi: 10.1002/mus.27105
29. Megremis S, Walker TDJ, He X, Ollier WER, Chinoy H, Hampson L, et al. Antibodies Against Immunogenic Epitopes With High Sequence Identity to SARS-CoV-2 in Patients With Autoimmune Dermatomyositis. Ann Rheum Dis (2020) 79:1383–6. doi: 10.1136/annrheumdis-2020-217522
30. Ferri C, Giuggioli D, Raimondo V, L'Andolina M, Tavoni A, Cecchetti R, et al. COVID-19 and Rheumatic Autoimmune Systemic Diseases: Report of a Large Italian Patients Series. Clin Rheumatol (2020) 39:3195–204. doi: 10.1007/s10067-020-05334-7
31. Movahedi N, Ziaee V. COVID-19 and Myositis; True Dermatomyositis or Prolonged Post Viral Myositis? Pediatr Rheumatol Online J (2021) 19:86. doi: 10.1186/s12969-021-00570-w
32. Ortona E, Pierdominici M, Maselli A, Veroni C, Aloisi F, Shoenfeld Y. Sex-Based Differences in Autoimmune Diseases. Ann Ist Super Sanita (2016) 52:205–12. doi: 10.4415/ANN_16_02_12
33. Zandman-Goddard G, Peeva E, Shoenfeld Y. Gender and Autoimmunity. Autoimmun Rev (2007) 6:366–72. doi: 10.1016/j.autrev.2006.10.001
34. Selmi C, Brunetta E, Raimondo MG, Meroni PL. The X Chromosome and the Sex Ratio of Autoimmunity. Autoimmun Rev (2012) 11:A531–7. doi: 10.1016/j.autrev.2011.11.024
35. Rubtsova K, Marrack P, Rubtsov AV. Sexual Dimorphism in Autoimmunity. J Clin Invest (2015) 125:2187–93. doi: 10.1172/JCI78082
36. Zhang H, Charmchi Z, Seidman RJ, Anziska Y, Velayudhan V, Perk J. COVID-19-Associated Myositis With Severe Proximal and Bulbar Weakness. Muscle Nerve (2020) 62:E57–60. doi: 10.1002/mus.27003
37. Kogami M, Suzuki S, Nanjo Y, Ikeda K, Tamura N, Sasaki S, et al. Complication of Coronavirus Disease 2019 During Remission Induction Therapy Against Anti-MDA5 Antibody-Positive Dermatomyositis. Rheumatol Adv Pract (2020) 4:rkaa068. doi: 10.1093/rap/rkaa068
38. Cao M, Zhang S, Chu D, Xiao M, Liu X, Yu L, et al. COVID-19 or Clinical Amyopathic Dermatomyositis Associated Rapidly Progressive Interstitial Lung Disease? A Case Report. BMC Pulm Med (2020) 20:304. doi: 10.1186/s12890-020-01335-z
39. Borges NH, Godoy TM, Kahlow BS. Onset of Dermatomyositis in Close Association With COVID-19 - a First Case Reported. Rheumatol (Oxford) (2021) 60:SI96. doi: 10.1093/rheumatology/keab290
40. Quintana-Ortega C, Remesal A, Ruiz de Valbuena M, de la Serna O, Laplaza-González M, Álvarez-Rojas E, et al. Fatal Outcome of Anti-MDA5 Juvenile Dermatomyositis in a Paediatric COVID-19 Patient: A Case Report. Mod Rheumatol Case Rep (2021) 5:101–7. doi: 10.1080/24725625.2020.1832755
41. Ho BVK, Seger EW, Kollmann K, Rajpara A. Dermatomyositis in a COVID-19 Positive Patient. JAAD Case Rep (2021) 13:97–9. doi: 10.1016/j.jdcr.2021.04.036
42. Liquidano-Perez E, García-Romero MT, Yamazaki-Nakashimada M, Maza-Morales M, Rivas-Calderón MK, Bayardo-Gutierrez B, et al. Juvenile Dermatomyositis Triggered by SARS-CoV-2. Pediatr Neurol (2021) 121:26–7. doi: 10.1016/j.pediatrneurol.2021.05.011
43. Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, et al. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol (2020) 77:683–90. doi: 10.1001/jamaneurol.2020.1127
44. Uruha A, Nishikawa A, Tsuburaya RS, Hamanaka K, Kuwana M, Watanabe Y, et al. Sarcoplasmic MxA Expression: A Valuable Marker of Dermatomyositis. Neurol (2017) 88:493–500. doi: 10.1212/WNL.0000000000003568
45. Manzano GS, Woods JK, Amato AA. Covid-19-Associated Myopathy Caused by Type I Interferonopathy. N Engl J Med (2020) 383:2389–90. doi: 10.1056/NEJMc2031085
46. Beydon M, Chevalier K, Al Tabaa O, Hamroun S, Delettre AS, Thomas M, et al. Myositis as a Manifestation of SARS-CoV-2. Ann Rheum Dis (2020). doi: 10.1136/annrheumdis-2020-217573
47. Ding Y, He L, Zhang Q, Huang Z, Che X, Hou J, et al. Organ Distribution of Severe Acute Respiratory Syndrome (SARS) Associated Coronavirus (SARS-CoV) in SARS Patients: Implications for Pathogenesis and Virus Transmission Pathways. J Pathol (2004) 203:622–30. doi: 10.1002/path.1560
48. Li W, Moore MJ, Vasilieva N, Sui J, Wong SK, Berne MA, et al. Angiotensin-Converting Enzyme 2 is a Functional Receptor for the SARS Coronavirus. Nat (2003) 426:450–4. doi: 10.1038/nature02145
49. Jin M, Tong Q. Rhabdomyolysis as Potential Late Complication Associated With COVID-19. Emerg Infect Dis (2020) 26:1618–20. doi: 10.3201/eid2607.200445
50. Parekh R, Care DA, Tainter CR. Rhabdomyolysis: Advances in Diagnosis and Treatment. Emerg Med Pract (2012) 14:1–15.
51. Lakota K, Perdan-Pirkmajer K, Hočevar A, Sodin-Semrl S, Rotar Ž, Čučnik S, et al. COVID-19 in Association With Development, Course, and Treatment of Systemic Autoimmune Rheumatic Diseases. Front Immunol (2020) 11:611318. doi: 10.3389/fimmu.2020.611318
52. Russell CD, Millar JE, Baillie JK. Clinical Evidence Does Not Support Corticosteroid Treatment for 2019-Ncov Lung Injury. Lancet (2020) 395:473–5. doi: 10.1016/S0140-6736(20)30317-2
53. Narain S, Stefanov DG, Chau AS, Weber AG, Marder G, Kaplan B, et al. Comparative Survival Analysis of Immunomodulatory Therapy for Coronavirus Disease 2019 Cytokine Storm. Chest (2021) 159:933–48. doi: 10.1016/j.chest.2020.09.275
54. Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, et al. Dexamethasone in Hospitalized Patients With Covid-19. N Engl J Med (2021) 384:693–704. doi: 10.1056/NEJMoa2021436
55. Strangfeld A, Schäfer M, Gianfrancesco MA, Lawson-Tovey S, Liew JW, Ljung L, et al. Factors Associated With COVID-19-Related Death in People With Rheumatic Diseases: Results From the COVID-19 Global Rheumatology Alliance Physician-Reported Registry. Ann Rheum Dis (2021) 80:930–42. doi: 10.1136/annrheumdis-2020-219498
56. Brown PM, Pratt AG, Isaacs JD. Mechanism of Action of Methotrexate in Rheumatoid Arthritis, and the Search for Biomarkers. Nat Rev Rheumatol (2016) 12:731–42. doi: 10.1038/nrrheum.2016.175
57. Tripathy S, Dassarma B, Roy S, Chabalala H. Matsabisa MG. A Review on Possible Modes of Action of Chloroquine/Hydroxychloroquine: Repurposing Against SAR-CoV-2 (COVID-19) Pandemic. Int J Antimicrob Agents (2020) 56:106028. doi: 10.1016/j.ijantimicag.2020.106028
58. Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, et al. Remdesivir and Chloroquine Effectively Inhibit the Recently Emerged Novel Coronavirus (2019-Ncov) In Vitro. Cell Res (2020) 30:269–71. doi: 10.1038/s41422-020-0282-0
59. Axfors C, Schmitt AM, Janiaud P, Van't Hooft J, Abd-Elsalam S, Abdo EF, et al. Mortality Outcomes With Hydroxychloroquine and Chloroquine in COVID-19 From an International Collaborative Meta-Analysis of Randomized Trials. Nat Commun (2021) 12:2349. doi: 10.1038/s41467-021-22446-z
60. Perez J, Roustit M, Lepelley M, Revol B, Cracowski JL, Khouri C. Reported Adverse Drug Reactions Associated With the Use of Hydroxychloroquine and Chloroquine During the COVID-19 Pandemic. Ann Intern Med (2021) 174:878–80. doi: 10.7326/m20-7918
61. Broen JCA, van Laar JM. Mycophenolate Mofetil, Azathioprine and Tacrolimus: Mechanisms in Rheumatology. Nat Rev Rheumatol (2020) 16:167–78. doi: 10.1038/s41584-020-0374-8
62. Kato F, Matsuyama S, Kawase M, Hishiki T, Katoh H, Takeda M. Antiviral Activities of Mycophenolic Acid and IMD-0354 Against SARS-CoV-2. Microbiol Immunol (2020) 64:635–9. doi: 10.1111/1348-0421.12828
63. Morteau O, Blundell S, Chakera A, Bennett S, Christou CM, Mason PD, et al. Renal Transplant Immunosuppression Impairs Natural Killer Cell Function In Vitro and In Vivo. PloS One (2010) 5:e13294. doi: 10.1371/journal.pone.0013294
64. Tiefenthaler M, Hofer S, Ebner S, Ivarsson L, Neyer S, Herold M, et al. In Vitro Treatment of Dendritic Cells With Tacrolimus: Impaired T-Cell Activation and IP-10 Expression. Nephrol Dial Transplant (2004) 19:553–60. doi: 10.1093/ndt/gfg594
65. Belli LS, Fondevila C, Cortesi PA, Conti S, Karam V, Adam R, et al. Protective Role of Tacrolimus, Deleterious Role of Age and Comorbidities in Liver Transplant Recipients With Covid-19: Results From the ELITA/ELTR Multi-Center European Study. Gastroenterol (2021) 160:1151–63.e3. doi: 10.1053/j.gastro.2020.11.045
66. Solanich X, Antolí A, Rocamora-Blanch G, Padullés N, Fanlo-Maresma M, Iriarte A, et al. Methylprednisolone Pulses Plus Tacrolimus in Addition to Standard of Care vs. Standard of Care Alone in Patients With Severe COVID-19. A Randomized Controlled Trial. Front Med (Lausanne) (2021) 8:691712. doi: 10.3389/fmed.2021.691712
67. Tsuji H, Nakashima R, Hosono Y, Imura Y, Yagita M, Yoshifuji H, et al. Multicenter Prospective Study of the Efficacy and Safety of Combined Immunosuppressive Therapy With High-Dose Glucocorticoid, Tacrolimus, and Cyclophosphamide in Interstitial Lung Diseases Accompanied by Anti-Melanoma Differentiation-Associated Gene 5-Positive Dermatomyositis. Arthritis Rheumatol (2020) 72:488–98. doi: 10.1002/art.41105
68. Zong M, Dorph C, Dastmalchi M, Alexanderson H, Pieper J, Amoudruz P, et al. Anakinra Treatment in Patients With Refractory Inflammatory Myopathies and Possible Predictive Response Biomarkers: A Mechanistic Study With 12 Months Follow-Up. Ann Rheum Dis (2014) 73:913–20. doi: 10.1136/annrheumdis-2012-202857
69. Spencer CH, Rouster-Stevens K, Gewanter H, Syverson G, Modica R, Schmidt K, et al. Biologic Therapies for Refractory Juvenile Dermatomyositis: Five Years of Experience of the Childhood Arthritis and Rheumatology Research Alliance in North America. Pediatr Rheumatol Online J (2017) 15:50. doi: 10.1186/s12969-017-0174-0
70. Zhang X, Zhou S, Wu C, Li M, Wang Q, Zhao Y, et al. Tocilizumab for Refractory Rapidly Progressive Interstitial Lung Disease Related to Anti-MDA5-Positive Dermatomyositis. Rheumatol (Oxford) (2021) 60:e227–8. doi: 10.1093/rheumatology/keaa906
71. Xu X, Han M, Li T, Sun W, Wang D, Fu B, et al. Effective Treatment of Severe COVID-19 Patients With Tocilizumab. Proc Natl Acad Sci USA (2020) 117:10970–5. doi: 10.1073/pnas.2005615117
72. Richardson P, Griffin I, Tucker C, Smith D, Oechsle O, Phelan A, et al. Baricitinib as Potential Treatment for 2019-Ncov Acute Respiratory Disease. Lancet (2020) 395:e30–1. doi: 10.1016/s0140-6736(20)30304-4
73. Kalil AC, Patterson TF, Mehta AK, Tomashek KM, Wolfe CR, Ghazaryan V, et al. Baricitinib Plus Remdesivir for Hospitalized Adults With Covid-19. N Engl J Med (2021) 384:795–807. doi: 10.1056/NEJMoa2031994
74. Puxeddu I, Ferro F, Bartoloni E, Elefante E, Baldini C, Scirè C, et al. COVID-19: The New Challenge for Rheumatologists. One Year Later. Clin Exp Rheumatol (2021) 39:203–13.
75. Kurasawa K, Arai S, Namiki Y, Tanaka A, Takamura Y, Owada T, et al. Tofacitinib for Refractory Interstitial Lung Diseases in Anti-Melanoma Differentiation-Associated 5 Gene Antibody-Positive Dermatomyositis. Rheumatol (Oxford) (2018) 57:2114–9. doi: 10.1093/rheumatology/key188
76. Chen Z, Wang X, Ye S. Tofacitinib in Amyopathic Dermatomyositis-Associated Interstitial Lung Disease. N Engl J Med (2019) 381:291–3. doi: 10.1056/NEJMc1900045
77. Guimarães PO, Quirk D, Furtado RH, Maia LN, Saraiva JF, Antunes MO, et al. Tofacitinib in Patients Hospitalized With Covid-19 Pneumonia. N Engl J Med (2021) 385:406–15. doi: 10.1056/NEJMoa2101643
78. Mehta P, Ciurtin C, Scully M, Levi M, Chambers RC. JAK Inhibitors in COVID-19: The Need for Vigilance Regarding Increased Inherent Thrombotic Risk. Eur Respir J (2020) 56:2001919. doi: 10.1183/13993003.01919-2020
79. Dalakas MC, Illa I, Dambrosia JM, Soueidan SA, Stein DP, Otero C, et al. A Controlled Trial of High-Dose Intravenous Immune Globulin Infusions as Treatment for Dermatomyositis. N Engl J Med (1993) 329:1993–2000. doi: 10.1056/NEJM199312303292704
80. Dalakas MC. Guillain-Barré Syndrome: The First Documented COVID-19-Triggered Autoimmune Neurologic Disease: More to Come With Myositis in the Offing. Neurol Neuroimmunol Neuroinflamm (2020) 7:e781. doi: 10.1212/NXI.0000000000000781
Keywords: SARS-CoV-2, COVID-19, dermatomyositis, pathogenesis, treatment
Citation: Qian J and Xu H (2022) COVID-19 Disease and Dermatomyositis: A Mini-Review. Front. Immunol. 12:747116. doi: 10.3389/fimmu.2021.747116
Received: 25 July 2021; Accepted: 24 December 2021;
Published: 13 January 2022.
Edited by:
Michele Maria Luchetti Gentiloni, Marche Polytechnic University, ItalyCopyright © 2022 Qian and Xu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jie Qian, ZHJxaWFuamllQDEyNi5jb20=
 Jie Qian
Jie Qian Hui Xu
Hui Xu