
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Immunol., 26 February 2021
Sec. Autoimmune and Autoinflammatory Disorders
Volume 12 - 2021 | https://doi.org/10.3389/fimmu.2021.632890
This article is part of the Research TopicAutoimmune Vasculitis: Advances in Pathogenesis and TherapiesView all 29 articles
A correction has been applied to this article in:
Corrigendum: Phenotype, Susceptibility, Autoimmunity, and Immunotherapy Between Kawasaki Disease and Coronavirus Disease-19 Associated Multisystem Inflammatory Syndrome in Children
 Ming-Ren Chen1,2†
Ming-Ren Chen1,2† Ho-Chang Kuo3†
Ho-Chang Kuo3† Yann-Jinn Lee1
Yann-Jinn Lee1 Hsin Chi1
Hsin Chi1 Sung Chou Li4
Sung Chou Li4 Hung-Chang Lee1
Hung-Chang Lee1 Kuender D. Yang1,5,6*
Kuender D. Yang1,5,6*Coronavirus disease-19 (COVID-19) in children is usually mild but some are susceptible to a Kawasaki disease (KD)-like multisystem inflammatory syndrome in children (MIS-C) in the convalescent stage, posing a need to differentiate the phenotype, susceptibility, autoimmunity, and immunotherapy between KD and MIS-C, particularly in the upcoming mass vaccination of COVID-19. Patients with MIS-C are prone to gastrointestinal symptoms, coagulopathy, and shock in addition to atypical KD syndrome with fever, mucocutaneous lesions, lymphadenopathy, and/or cardiovascular events. MIS-C manifests KD-like symptoms that alert physicians to early recognize and adopt the KD treatment regimen for patients with MIS-C. MIS-C linked to COVID-19 teaches us infection-associated autoimmune vasculitis and vice versa. Studies on genetic susceptibility have identified certain human leukocyte antigen (HLA) locus and toll-like receptor (TLR) associated with KD and/or COVID-19. Certain HLA subtypes, such as HLA-DRB1 and HLA-MICA A4 are associated with KD. HLA-B*46:01 is proposed to be the risk allele of severe COVID-19 infection, and blood group O type is a protective factor of COVID-19. The autoimmune vasculitis of KD, KD shock syndrome (KDSS), or MIS-C is mediated by a genetic variant of HLA, FcγR, and/or antibody-dependent enhancement (ADE) resulting in hyperinflammation with T helper 17 (Th17)/Treg imbalance with augmented Th17/Th1 mediators: interleukin-6 (IL-6), IL-10, inducible protein-10 (IP-10), Interferon (IFNγ), and IL-17A, and lower expression of Treg-signaling molecules, FoxP3, and transforming growth factor (TGF-β). There are certain similarities and differences in phenotypes, susceptibility, and pathogenesis of KD, KDSS, and MIS-C, by which a physician can make early protection, prevention, and precision treatment of the diseases. The evolution of immunotherapies for the diseases has shown that intravenous immunoglobulin (IVIG) alone or combined with corticosteroids is the standard treatment for KD, KDSS, and MIS-C. However, a certain portion of patients who revealed a treatment resistance to IVIG or IVIG plus corticosteroids, posing a need to early identify the immunopathogenesis, to protect hosts with genetic susceptibility, and to combat Th17/Treg imbalance by anti-cytokine or pro-Treg for reversal of the hyperinflammation and IVIG resistance. Based on physiological and pathological immunity of the diseases under genetic susceptibility and host milieu conditions, a series of sequential regimens are provided to develop a so-called “Know thyself, enemy (pathogen), and ever-victorious” strategy for the prevention and immunotherapy of KD and/or MIS-C.
Coronavirus disease-19 (COVID-19) is usually mild in children (1–3). However, 3–6 weeks after the disease or exposure to persons with COVID-19, some children are affected by multisystem inflammatory syndrome in children (MIS-C) (4–9). Those with MIS-C frequently have gastrointestinal symptoms, coagulopathy, and shock in addition to atypical Kawasaki disease (KD) symptoms with intractable fever, mucocutaneous lesions, lymphadenopathy, and/or cardiovascular events (4–8), which alert physicians to early recognition and adopt the KD treatment regimen for them (4–9). MIS-C occurring 3–6 weeks after contracting COVID-19 suggests that MIS-C is an infection-associated autoimmunity. The life-threatening infection-associated hyperinflammatory syndrome does not completely respond to intravenous immunoglobulin (IVIG) therapy, which is a standard treatment for KD. IVIG plus corticosteroids (4–8) or/and blockade of interleukin-1 (IL-1) or IL-6 action (9) have been used to treat patients with MIS-C. Now the questions remains, who are susceptible, what is (are) the trigger(s), how to predict and differentiate between KD and MIS-C, and which regimen is the optimal therapy for KD or MIS-C based on mechanistic infection immunity?
Kawasaki disease (KD) is a hyperinflammatory febrile vasculitis in children below 5 years of age, with at least four of the five clinical symptoms/signs: skin rashes (>90%), bilateral conjunctival injection (>90%), oral mucosal changes (>90%), peripheral extremity changes, and cervical lymphadenopathy (at least 1.5 cm in diameter), which might develop weeks after a mild respiratory or gastrointestinal symptom (10–12). Those with <4 criteria for KD are classified as incomplete or atypical KD. Children in the extremes of the age spectrum (≤6 months old, or ≥5 years old) tend to have atypical KD associated with delayed diagnosis and treatment (13, 14). Atypical presentation of KD in children may be associated with a higher risk of coronary arteritis because of a delayed diagnosis and treatment (14, 15). KD is previously called mucocutaneous lymph node syndrome by Tamisaku Kawasaki in 1974 (10), in regard to vasculitis including coronary arteritis and aneurysm (10–15). The hyperinflammatory response of KD is related to infection, autoimmunity, and/or genetic susceptibility (10–12). KD is more prevalent in East Asia, such as Japan, China, Korea, and Taiwan (10–12, 16–18). The incidence of KD varies from country to country, e.g., 4.5 per 100,000 children younger than 5 years of age in India, 25 per 100,000 in the USA, 56 per 100,000 in Taiwan, and over 250 per 100,000 people in Japan (17, 18).
Recently, a surge in the prevalence of KD-like illness in children has been found with the COVID-19 outbreak in the USA, UK, France, Spain, and Italy (4–9, 19). COVID-19 can cause acute respiratory distress syndrome (ARDS), carditis, thrombosis, and/or shock in adults, but generally, induce mild symptoms in infants and children (1–3). The COVID-19-associated MIS-C occurs in older children and tends to manifest with gastrointestinal symptoms, coagulopathy, and shock in addition to the KD symptoms. Patients with KD usually have thrombocytosis (10–15), but patients with MIS-C have high, normal, or low platelets (4–9), which may be related to coagulopathy or microangiopathy. The MIS-C is similar to KD shock syndrome (KDSS) occurring in relatively older children with atypical KD, showing shock, thrombosis, and IVIG resistance (20–22). The immune response in MIS-C is different from that in COVID-19. COVID-19 is contagious but MIS-C is not. MIS-C is due to post-infection autoimmunity because it occurs 3–6 weeks after the exposure to COVID-19 or persons with COVID-19. Patients with MIS-C have a unique serology with anti-S antibodies (IgG, IgM, and IgA) but not anti-N antibodies, in contrast, patients with COVID-19 have both anti-S and anti-N antibodies (23). The skin, gastrointestinal, and shock symptoms in MIS-C are sometimes undifferentiated from those in toxic shock syndrome (TSS), but the medical history is different because TSS is related to superantigens of bacteria which are usually associated with bacterial infections, surgical wound, or usage of tampons (24).
The COVID-19-related MIS-C, representing atypical KD syndrome in older children at a median age of 8 years (6, 9), is prone to IVIG resistance and life-threatening cardiovascular events, such as myocardial infarction, thrombosis, and/or shock (4–9, 19), which is the most life-threatening morbidity in children during the COVID-19 pandemic.
There are some overlapped and different symptoms and signs among KD, KDSS, and MIS-C in regard to age, sex, race, severity, and treatment responses (Table 1). Patients with KD usually have vasculitis in mucocutaneous regions (>90% of eyes, lips, and/or skin symptoms) and coronary arteritis, but few patients have myocarditis (~5%) and shock (~7%) (10–12). In contrast, KDSS frequently shows myocarditis, thrombosis, and shock (20–22). The KDSS is associated with a higher rate of IVIG treatment resistance, with older age and more serious hypotension, skin rash, leukocytosis, neutrophilia, and hypoalbuminemia, especially frequently found in Hispanics (20, 22). Patients with MIS-C more frequently have a shock, myocarditis, thrombosis, and gastrointestinal symptoms (4–9). Both patients with KDSS and MIS-C more frequently require intensive care supports, such as inotropic agents, ventilation support, anti-thrombotic therapy, and additional anti-inflammatory therapies (4–9, 19–22). KD is prevalent in Eastern Asia (10–12) but MIS-C is more frequently found in Western countries, especially Afro-Caribbean (4–9, 19). The mean age of patients with KD is 2.5 years, and that of patients with KDSS is 3.7 years (22). In the largest cohort report of 186 Afro-Caribbean patients with MIS-C, the median age is 8.3 years (9), showing prominent gastrointestinal symptoms and thrombosis with variable platelet counts (high, normal, or low).
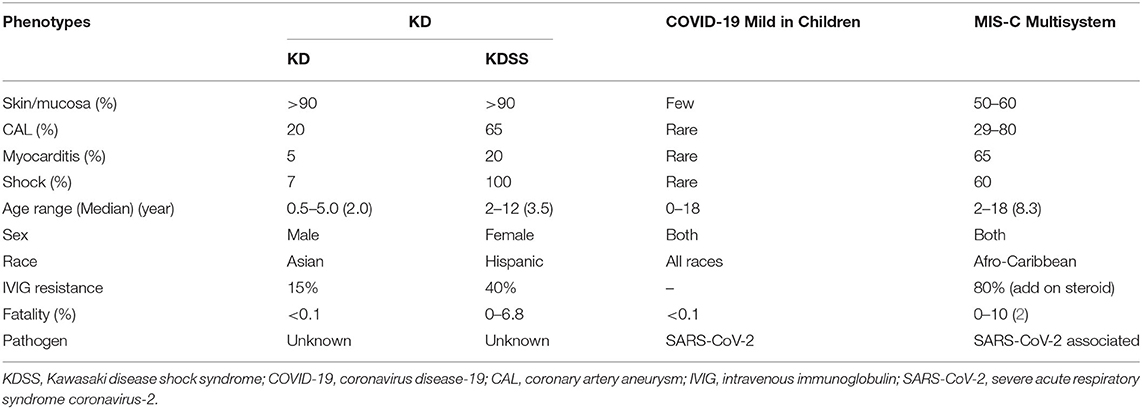
Table 1. Demographic data and phenotypes of Kawasaki disease (KD) and multisystem inflammatory syndrome in children (MIS-C).
In fact, KDSS is a severe form of KD with hypotension, coagulopathy, more cardiovascular involvement, and IVIG resistance, initially recognized in 2009 (20). In a retrospective analysis of 103 patients with KDSS, abnormalities in the coronary arteries were 65% and the mortality rate was 6.8% (22). Before the institution of IVIG therapy for KD, the KD mortality and cardiac morbidity were 2 and 20%, respectively. After the institution of the IVIG treatment for KD, the mortality decreased below 0.1% and coronary artery aneurysm downed to <4%. The MIS-C mortality ranges between 0 and 10% (average, 2%). KD is not contagious although many infections, such as Staphylococcus aureus, streptococci, rhinovirus, coronavirus, enterovirus, chlamydia, or Epstein-Barr (EB) virus had been associated with KD. About 40% of patients with KD have reactive skin erythema and/or scaling at the Bacillus Calmette–Guérin (BCG) inoculation site (11), suggesting the autoreactive antigen for KD may cross-react with the antigen of BCG, or the BCG reactivation is a bystander of hyperinflammatory reaction of KD. It is highly suspicious that the MIS-C is a severe KD-like vasculitis mediated by a COVID-19-induced autoimmune reaction. This is not the first time that human coronavirus (HCoV) is correlated to KD. In 2005, Esper et al. (25) reported that a novel human coronavirus called human coronavirus New Haven (HCoV-NH) was associated with an outbreak of KD in New Haven, showing RT-PCR detection of the positivity at 8/11 vs. 1/22 in a case-control study. This association of KD with HCoV was not replicated in a study at Taiwan using 53 consecutive KD samples in which no detectable HCoV-NH or HCoV-NL63 was observed in nasopharyngeal secretions (26). They, however, did not measure the serum antibodies against HCoV-NH or HCoV-NL63. These studies suggest that children in Western countries are susceptible to coronavirus-related KD-like vasculitis and children in Asian countries are susceptible to non-coronavirus-related KD vasculitis.
In laboratory data as shown in Table 2, patients with KD tend to have thrombocytosis, and patients with KDSS or MIS-C tend to have varied platelet counts. C-reactive protein (CRP) and procalcitonin levels are much higher in patients with KDSS or MIS-C. The ferritin levels are also higher in patients with KDSS or MIS-C. Lymphopenia is often prominent in patients with COVID-19 or MIS-C (1–9, 19), but not in patients with KD or KDSS (10–12, 20–22). Coagulopathy is also more often in KDSS or MIS-C (9, 19–22, 27, 28). Ferritin (>500–1,000 ng/ml) and D-dimer (>1,000–4,000 ng/ml) levels are much higher in children with KDSS and MIS-C (9, 19–22, 27, 28). Cytokine storm in the blood is quite similar between KD and MIS-C, showing augmented hypercytokinemia in IL-6, IL-10, IL-17, inducible protein-10 (IP-10) (CXCL10), and MCP-1 (CCL2), especially higher IL-6 and IL-10 levels in KDSS (27), and IL-10 and TNF-α levels in MIS-C (28). Over 80% of patients with MIS-C have detectable anti-S antibody against spike (S) antigen of severe acute respiratory syndrome coronavirus-2 (SARS-Cov-2), but less than one-third have detectable RNA of the virus (4–9). Apparently, MIS-C is mediated by a skewed immune response toward T helper 17 (Th17) reaction in the convalescent stage of COVID-19.
A patient with a viral infection usually has normal or lower leukocyte counts, and low CRP and procalcitonin levels unless he/she also has superimposed bacterial infections. However, regardless of whether COVID-19 is contagious or MIS-C is not contagious, lymphopenia and elevated CRP are found in both conditions in children. Apparently, SARS-CoV-2 induces a proinflammatory reaction in the acute stage of COVID-19 and a hyperinflammatory reaction of vasculitis (4–9, 19) with augmented levels of Th17 and Th1 mediators in MIS-C (28). There are many unsolved issues on the immunopathogenesis of KD and MIS-C. It is debatable whether MIS-C and KD are post-infectious hyperinflammation, autoinflammatory, or autoimmune disorders (29–34). Current autoimmune concepts have limitations to explain the pathogenesis of variant systemic vasculitis syndrome, which is not contagious but infection-associated hyperinflammation in the convalescent stage (29). Inflammation-inducing substances, not only those originating from pathogens, including toxins and pathogen-associated molecular patterns (PAMPs), but also those originating from injured or infected-host cells including pathogenic proteins, pathogenic peptides, and damage-associated molecular patterns (DAMPs), especially in intracellular pathogen infections, such as virus, chlamydia, BCG, and SARS-CoV-2, may alter the immune responses based on “the protein-homeostasis-system hypothesis” (30). Given the fact that marked different incidences in KD and MIS-C across the populations may be explained by colonization states of pathogens (31), and an imbalance of regulatory and cytotoxic SARS-CoV-2-reactive CD4 T cells in COVID-19 (32), we focused on the imbalanced Th17/Treg regulation for explanations of the same and different manifestations among KD, KDSS, COVID-19, and MIS-C in this perspective article.
We have studied the immune responses of KD for over two decades and look into those related to COVID-19 in the literature in order to explore the link and implication for effective prediction, prevention, and treatment of KD and MIS-C. Literature and clinical experience of KD management prompt early recognition of the KD-like vasculitis in MIS-C for IVIG immunotherapy and beyond, vice versa, information accumulated in MIS-C suggest that KD-like vasculitis is an infection-associated autoimmunity (32–34). Another possibility is an antibody-dependent enhancement (ADE) of FcγR-mediated autoimmunity that has been reported in SARS-CoV-2 infection (35) and also reported in SARS-CoV-1 infection (36). In a genetic association study of FcγRIIA polymorphisms with the severity of SARS-CoV-1 infections, Yuan et al. (37) found that variant FcγRIIA-R/R131 in the intensive care unit (ICU) subgroup of patients with SARS was significantly more frequent than in normal controls. We have previously studied immunopathogenesis of SARS-CoV-1 infection and found that SARS-CoV-1 infection caused an early innate augmentation with adaptive immunosuppression and then induced a late exacerbation (38, 39). To address the hyperinflammatory reaction of KD, we first demonstrate that overexpression of inducible nitric oxide (NO) synthase associated with elevated blood NO levels is present in patients with KD before IVIG therapy (40), and that the T-cell activation marker CD40L is prominently expressed on T cells and platelets in children with KD, which is reversed after IVIG therapy (41). This is comparative to the dynamic time course of immune responses of KD validated by a kinetic immunopathology in a series of autopsy classifications of early necrotizing vasculitis with innate phagocyte activation followed by a remodeling of adaptive immunity with lymphocyte infiltration in the convalescent stage (42).
Before the era of IVIG therapy, the rate of coronary artery aneurysm was 25% and mortality was 1–2% (10, 42–44) in patients with KD, after the institution of IVIG therapy the rate of coronary artery aneurysm downs to 3–4% and mortality to <0.1% (42, 43). Now the challenge for the KD treatment is IVIG resistance in 15–20% of KD patients, which requires additional immunotherapy. Similarly, KDSS and MIS-C have even higher IVIG resistance rates and more frequently require combined therapy with IVIG and steroid pulse therapy (19–22, 28). Interestingly, corticosteroids treatment before the IVIG institution era showed an exacerbated morbidity of coronary artery aneurysms in 64.7 vs. 20% treated with antibiotic alone or 11% treated with aspirin alone (44). Parameters in patients with IVIG resistance are persistent fever and elevated IL-6 levels (45). Furthermore, skewed T-cell polarization toward Th2 response favors the outcomes of IVIG therapy. A higher eosinophil count associated with a higher IL-5 level is a favorable marker for the success of IVIG treatment. In contrast, lower initial eosinophil counts and lower IL-4 and IL-5 levels are associated with IVIG-resistance (46). Patients with KD have prominent Th17 immune responses with augmented IL-6, IL-10, G-CSF, and IL-17A levels, and lower Treg pathway transcription factor FoxP3 expression before IVIG treatment (47). The augmented cytokine storm declines and the Treg cell increases after IVIG treatment.
The Th17 polarization with elevated IL-6, IL-17A, and G-CSF levels is correlated to a higher neutrophils vs. lymphocytes (N/L) ratio in KD patients complicated with IVIG resistance and coronary arteritis (48), which is similar to the severity of COVID-19 associated with an increase in neutrophils and decrease in lymphocytes and elevated INF-γ, IL-6, and IL-8 levels (49, 50). The cytokine profile in MIS-C is different from that in severe COVID-19 in higher IL-10 and TNF-α levels (28). It is shown that IL-6 together with TGF-β induces Th17 differentiation from naïve T cells (51), whereas IL-6 inhibits TGF-β-induced Treg differentiation via degradation of FoxP3 (52), suggesting that higher IL-6, IL-10, and IL-17A but lower FoxP3 and TGF-β expression in patients with KD or MIS-C is involved in the Th17/Treg imbalance. This is further supported by our finding that DNA polymorphisms of TGF-β-signaling pathway genes, e.g., TGF-β2 and SMAD3, were associated with the susceptibility of KD (53), and the Th17/Treg imbalance could also be mediated by epigenetic regulation of DNA methylation and/or micro RNAs (miRNAs) on innate and adaptive immune genes as a biomarker of KD (54–59). In HumanMethylation450 BeadChips assay, we have found that DNA hypomethylation on the promoter CG sites of many immune activation genes in leukocytes of patients with KD before IVIG treatment (54–57). The hypomethylated genes were associated with augmented gene (mRNA) expression, particularly the toll-like receptors (TLRs). The TLR1, 2, 4, 5, 8, and 9 receptor genes were significantly hypomethylated and associated with augmented mRNA expression (55). Similarly, other innate immunity genes, e.g., FcγR2A, IL-10, and S100A8 were also hypomethylated before IVIG treatment (54–57). Importantly, we found that the CpG site methylation changes >20% in the acute stage of KD were mainly hypomethylated (97%) genes but only 3% hypermethylated genes (56). After IVIG treatment, the hypomethylated genes and augmented mRNA expression reversed (54–57).
Moreover, it is found that miRNA expression is also a good biomarker of KD, which differentiated KD from other febrile diseases by a set of 4 miRNA expression at CT (miR-1246)-CT (miR-4436b-5p) and CT (miR-197-3p)-CT (miR-671-5p) (58). The miRNA control of Treg expression in patients with KD has been characterized before and after IVIG treatment (59). The epigenetic control of Treg development and maintenance has been defined predominantly via FoxP3 expression (60). These epigenetic profiles and functional markers of different Treg population (tTreg, iTreg, and pTreg) tend to have a promising role as specific mechanistic biomarkers for the prediction and prevention of Th17-mediated autoimmunity (61–63). The study model can be applied to study the epigenetic biomarkers and therapeutic targets of MIS-C and KD with and without shock syndrome by potential immunotherapy of cytokine inhibitor, DNA methylation, and/or miRNA expression in addition to IVIG with and without corticosteroids. The immunopathogenesis of KD and MIS-C probably progresses from an early Th17 response, followed by a later T-regulatory response. In the early Th17 response before IVIG treatment, the Treg pathway signals are depressed, and the reciprocal Th17/Treg imbalance reverses after IVIG treatment (47, 54–59). This is supported by the fact that the corticosteroids treatment alone in the acute stage was useless and even harmful in the 1970s (10, 44); instead, the combination of IVIG with corticosteroids showed a better response than the IVIG therapy alone in the 2010s, especially in the patients with IVIG resistance (64). Based on the rationales described above, we postulate that there are two phases during the development of KD or MIS-C syndrome; the early Th17 reaction and late Treg resolution stage have different immunopathogenic processes with individual biomarkers and require different immunotherapies.
A population-based surveillance system called COVID-19–associated hospitalization surveillance network (COVID-NET) analyzed 576 hospitalized COVID-19 pediatric patients and showed the prevalence of COVID-19 in children increased from 0.1 per 100,000 to 8 per 100,000 with the progress of COVID-19 pandemic, in which a race disparity in hospitalization deviated to Hispanic children, and nine (10.8%) of 83 admitted children had MIS-C (65), suggesting MIS-C may attribute to one-tenth of the admitted severe COVID-19 in children. MIS-C is rare or sporadic in adults (66, 67). COVID-19 deserves further studies on the autoimmunity under endogenous or exogenous milieu because it might directly trigger autoinflammatory conditions by molecular mimicry or cause autoimmunity in predisposed individuals in other environmental conditions (68). The algorithm for diagnosis and treatment of complete and incomplete KD in children has been proposed to diagnose and treat KD in adults (67, 68), and MIS-C in adults (67, 68).
Some adults have been diagnosed with atypical or incomplete KD, contemporarily or retrospectively (69, 70), and occasionally caused sudden death (71) or sequelae of the KD from Children (72). MIS-C has also been demonstrated in certain adults (67, 68). The autopsy of patients with COVID-19 showed a severe endothelial injury associated with the detectable intracellular virus, disrupted cell membranes, and widespread thrombosis with microangiopathy in the lungs (73, 74). The alveolar microthrombi were nine times more in patients with COVID-19 than in patients with influenza (75), and viral particles were detected in epithelial cells and endothelial cells of the lungs (73, 74). Micro-embolism and thrombosis indicating vasculitis and coagulopathy are similar between fatal patients with COVID-19 with ARDS and fatal patients with SARS (75, 76). Pathological findings of autopsy in KD are vasculitis with leukocyte infiltration, called periarteritis nodosa-like arteritis, coronary thrombosis with macrophage and lymphocyte infiltration, and aneurysm, but not pulmonary vasculitis (42, 77, 78). Autopsy features in MIS-C have not been characterized yet. We anticipate that abnormal proinflammatory insults with skewed Th17/Treg imbalance in MIS-C will be seen in the lesions of the cardiovascular system but not in the lesions of pulmonary vessels. The pathology in the COVID-19-induced ARDS showing pulmonary involvement with detectable viral RNA and thrombo-emboli in autopsy is different from the pathological finding in KD showing sterile vasculitis, leukocyte infiltration, and aneurysm, indicating immunity to infections in the former vs. autoimmunity in the latter, which could occur in both children and adults.
No specific genes have been linked to the susceptibility of MIS-C. The siblings of patients with KD are 10 times more likely to have KD. Several susceptibility genes (e.g., ITPKC, CASP3, CD40, and ORAI1) have been linked to KD (79), and KD is also associated with the human leukocyte antigen (HLA)-BW22J2 subtype, which is found specifically in Japanese and not in Caucasians (80). In patients with KD with coronary artery lesions (CAL), the frequency of HLA-DRB1*11 is significantly increased and that of HLA-DRB1*09 is decreased (81); In fact, HLA subtypes linked to KD are different between children in Asia and those in Western countries (82). We have previously found that HLA-DRB1 was associated with KD susceptibility (83). To search for the risk allele(s) of major histocompatibility complex (MHC) class 1, HLA-MICA (MHC class I chain-related gene A) locus, we found that the HLA-MICA A4 allele was significantly associated with the coronary artery aneurysm in patients with KD (84), and it has been validated in a genome-wide association case-control study in a Taiwanese population (85). We also found that DNA polymorphisms of TGF-β2 and SMAD3 are associated with the susceptibility of KD (53), and a dominant T allele of rs2243250 in the IL-4 gene conferred a great protective effect against the development of CAL in patients with KD (p = 0.006) (86). Taken together, this suggests that a specific HLA subtype could present a viral antigen peptide to T cells and induce a skewed Th17-Th1/Th2-Treg development involved in the altered hyperinflammation in KD. Although there is no any genetic association with MIS-C in the literature, the HLA-B*46:01 has been proposed to be associated with the severity of COVID-19 in a computational simulation by using SARS-CoV-2 whole-genome peptides for simulating their binding to 145 MHC class I HLA-A, -B, and -C genotypes, in which HLA-B*15:03 shows the greatest capacity to present highly conserved SARS-CoV-2 peptide which is shared among common human coronaviruses, suggesting that it could enable cross-protective T-cell-based immunity (87). Deletion or mutation of TLR7, which is a single-stranded RNA virus sensor in endosomes for induction of interferons (IFNs), contributes to the severity of COVID-19 in young adults (88). In contrast, imiquimod, a TLR agonist is proposed to enhance the defense against COVID-19 (89). Taken together, we would propose to clarify whether an HLA subtype, such as HLA-B*46:01 together with a TLR7 variant induces an augmented proinflammatory reaction under conditional milieu and alters the epigenetic control of FoxP3 expression resulting in the Th17/Treg imbalance in KD and/or MIS-C.
Interestingly, different blood group subtypes have also been shown to be associated with the susceptibility of COVID-19 (90–92). Initially, a report from Wuhan, China described blood group A subjects were more susceptible to COVID-19, and the presence of anti-A antibody was probably protective (90). Furthermore, another report from China showed that females but not males, with blood group A are susceptible to COVID-19 (91). In contrast, later in the USA, the other report described that B and AB blood groups were susceptible to the infection but not severity (92). The reproducible result among the studies is that the O blood group population is less susceptible to COVID-19 (90–92), but it remains controversial whether blood groups A and/or B population are susceptible to and/or vulnerable to severity. Perhaps, the differences are related to different studies in different races.
We are currently studying the association of MHC genotypes, haplotypes, and antigen presenting pocket prediction with KDSS and/or MIS-C via a consortium in Taiwan. Hopefully, the more the cases of MIS-C identified the more the opportunity to identify the association of KDSS or MIS-C with HLA subtypes in the COVID-19 outbreak or the COVID-19 mass vaccination. We could also study whether different H2 subtypes interacting with viral antigen under host situations lead to altered immunity contributing to KDSS or MIS-C in a mouse model using vaccine antigens with and without adjuvant. This experimental study would test whether the HCoV associated KD-like hyperinflammation is related to different races with varied HLAs by which different antigens induce altered immune responses because of different HLA subtypes and environments.
Since MIS-C reveals KD-like syndrome fulfilling complete or incomplete criteria, a physician could rapidly recognize and adopt the treatment regimen of KD for MIS-C, and mitigate the life-threatening disease. The treatment of KD in the acute febrile stage has evolved from corticosteroids, IVIG, and aspirin to a combination of IVIG, aspirin, and steroids through the past 50 years (10, 44, 64). Although long-term aspirin, whether it is high (anti-inflammatory) or low (antiplatelet) dose, does not appear to lower the frequency of coronary abnormalities (42), a low dose aspirin and/or antithrombotic treatment with low molecular weight heparin or warfarin is prescribed according to the progress of coronary aneurysm in the convalescent stage (42, 93). A combination of IVIG and corticosteroids significantly reduced the risk for coronary artery lesions compared with IVIG alone (7.6 vs. 18.9%; OR: 0.3; 95% CI 0.20–0.46) in a meta-analysis (64). Different dosing of IVIG for KD has been clarified (94), and different dosing of corticosteroids in the clinical trials at different countries explained the overall varied benefits on the outcomes of IVIG and corticosteroids in coronary artery aneurysm (64, 95). In pneumonia-associated ARDS, early treatment with corticosteroids and/or IVIG may reduce the aberrant immune responses that have been described (30). This may be also applicable to the treatment of COVID-19-related ARDS. For instance, a combination of pulse corticosteroids and IVIG therapy has been shown to rescue patients with tocilizumab-resistant severe COVID-19 (96). High dose IVIG regimen (2 gm/Kg) is largely demonstrated as more effective (42, 94), however, early IVIG therapy for KD within 4 days did not provide better protection from the development of CAL (97, 98). Whether the earlier and higher doses of immunotherapy for MIS-C and KD responsible for better outcomes deserves further studies.
Intravenous immunoglobulin resistance in the acute stage is frequently associated with the development of CAL in patients with KD. A few different scoring systems have been developed to predict IVIG resistance (99–101), and to provide a precise anti-inflammatory regimen, such as to infliximab (or anakinra) in addition to aspirin, IVIG, and corticosteroids therapy (101). Unfortunately, a scoring system (Kobayashi Score) which is successfully used to predict and prevent CAL in Japan (99) performs poorly in sensitivity and specificity in Western countries (100). MIS-C is IVIG resistant in most patients, therefore IVIG plus corticosteroids is used to treat the life-threatening condition (4–9, 96).
Certain unique complications of KD, such as shock, macrophage activation syndrome (MAS), or coronary aneurysm are usually associated with IVIG resistance and require additional anti-inflammatory regimens, such as cyclosporin, anti-IL1, or anti-IL6 treatment (101, 102). It is reasonable to add anti-IL6 for KD or MIS-C for IVIG resistance because serum IL6 levels correlate with IVIG resistance (45, 103). However, in a study of four patients with IVIG-resistant KD who are responsive to anti-IL6 treatment but affected by coronary artery aneurysms in 2 of them (104). The patients with KD with IVIG-resistance usually respond to anti-TNF, anti-IL-1, or steroid pulse therapy (101–104). Furthermore, anti-cytokines, such as tocilizumab and anakinra or anti-coagulopathy regimens have been used for COVID-19 hyperinflammatory syndrome in adults and resulted in favorable outcomes (105, 106), and are suggested to be used in MIS-C with IVIG resistance (9, 19, 28). Apparently, a scoring system based on symptoms and biochemistry to predict IVIG resistance may not be enough. Some studies had identified IVIG resistance associated with elevated IL-6, IL-10, and/or TNF-α levels in KD with and without shock syndrome (27, 45, 48), and we showed that lower IL-5 levels associated with lower eosinophil number was correlated to IVIG resistance (46), and allele rs2243250T of the IL-4 gene conferred protection against coronary artery lesions in KD (85). Patients with KD or MIS-C in different countries or races may require varied criteria for the prediction of the resistance to IVIG or anti-cytokine treatment. A new scoring system should include conventional symptoms and individuals' immunological parameters to provide a better guide to decrease IVIG resistance and increase effectiveness of the additional anti-inflammatory therapy. In addition to hyperinflammation and shock syndrome, patients with KDSS (20–22) or MIS-C (9, 19) usually manifest with coagulopathy, embolism, and thrombosis. It remains to be determined whether the embolism, thrombosis, and/or coagulopathy in MIS-C require certain anti-thrombotic therapy, and whether patients with MIS-C with coronary involvement require a long term aspirin treatment, which is a regimen for treating patients with KD who develop coronary abnormalities (42, 93).
Until July 2020, more than 1,000 cases of MIS-C had been reported (107). However, the definition and treatment regimens are not standardized yet! While COVID-19 remains pandemic, MIS-C cases will increase further. It is also a concern that this augmentation of Th17/Treg imbalance in MIS-C after COVID-19 may be extended by a COVID-19 mass vaccination in which the vaccine antigen with adjuvant may increase the risk of MIS-C. Both KD and MIS-C are non-contagious but potentially virus or antigen- (PAMP-) induced autoimmunity in genetically susceptible individuals. Patients from different genetic backgrounds and environments including pre-existing subneutralized antibodies or abnormal autoantibodies directed against different compartments, such as endoglin, EDIL3, and or casein kinases of endothelial cells, and so on, may influence the development of MIS-C or KD (108). Both patients with MIS-C and KD have augmented IL-6, IL-17A, and IP-10 production (27, 28, 45, 108), but the levels of SCF, TWEAK, and ADA significantly decreased in patients with KD but not in patients with MIS-C (108), suggesting they have similar immune activation pathways but different regulatory (suppressive) pathways. Taken together, we postulate immunopathogenesis of the COVID-19 associated MIS-C begins with innate immunity of SARS-CoV-2 infection to cells via ACE2 (Figure 1A), followed by adaptive immunity of COVID-19 with antigen presentation through HLA for T-cell differentiation toward an effective immunity or altered Th17 response (Figure 1B), and leading to individual autoimmunity with MIS-C in the convalescent stage (Figure 1C).
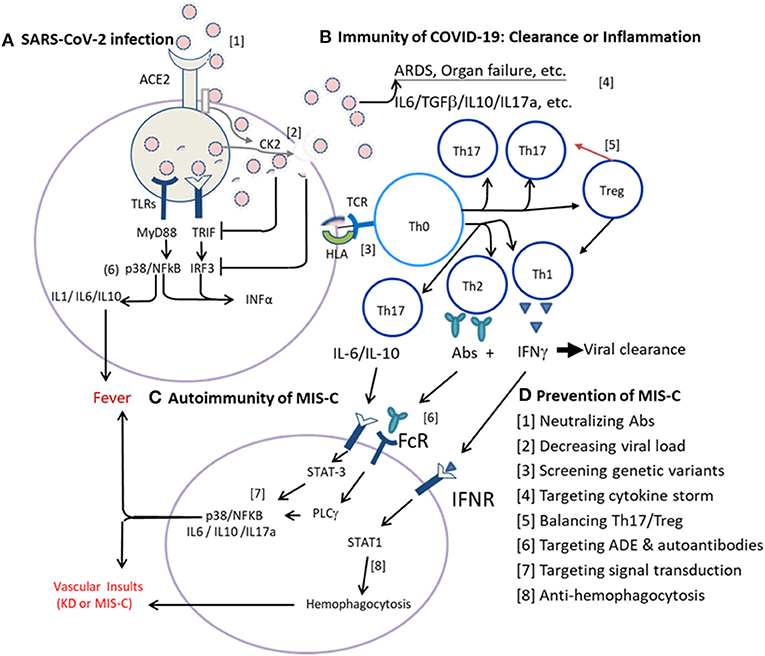
Figure 1. Immunotherapies of multisystem inflammatory syndrome in children (MIS-C) based on infection immunity and autoimmunity. (A) Innate immunity of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection to cells via ACE2. The SARS-CoV-2 virus enters host cells via ACE2 where a serine protease cleaves the viral spike (S) protein and allows the virus to fuse with the plasma membrane for internalization. Host RNA sensing receptors, such as RIG-1 (DDX58), Toll-like receptor-3 (TLR3), TLR7, and/or TLR8, detect the internalized virus and induce the production of interferons (IFNs) via MyD88, TRIF (TICAM1), IRF3, and/or IRF7 pathways, promote production of proinflammatory cytokines via MAPK (e.g., p38) and NFkB pathways. Normally, the host RNA sensing receptor(s) of RIG-1 and TLR7 signaling pathways will mediate an effective induction of IFNs for virus clearance. While the virus hijacks RNA sensing receptors and pathways or activates casein kinase 2 (CK2) for filopodial protrusion of budding viral particles, the virus multiplies rapidly and the infection spreads systemically. (B) Adaptive immunity of coronavirus disease-19 (COVID-19). Upon antigen presentation for T-cell adaptive immunity via human leukocyte antigen (HLA), an optimal adaptive immunity with T-cell immunity and B-cell production of neutralizing antibodies (Abs) for virus clearance is normally elicited. While the initial virus load is high, or the TLR is either of congenital deficit or of acquired deficit as in the elderly, or the viral glycoproteins (antigens) could suppress the MyD88, TRIF, IRF3, and/or IRF7 signaling pathways, the antigen-presenting cells (APCs) are hijacked and present the viral antigen with an altered signal for the polarization of naïve T helper cells (Tho) toward Th17 response with inflammatory cytokines production, but not Treg regulation for a proper Th1 cell immunity and/or Th2 humoral (B cell) response for neutralizing antibody production. (C) Individual autoimmunity with MIS-C. In convalescence, most patients recovered due to efficient adaptive immunity of Th1 cell immunity and Th2 neutralizing Abs unless individual subjects with genetic susceptibility or altered endogenous milieu. For instance, the viral antigens (PAMPs) interact with individual HLA subtype(s) of APCs and induce altered autoimmunity with Th17/Treg imbalance and augmented cytokine storm of IL-6, IL-17A, and/or IL-10 expression or abnormal autoantibodies resulting in MIS-C with systemic vasculitis, thrombosis, and shock, as seen in MIS-C. In addition, certain host milieu, e.g., abnormal homeostasis of vitamins and microbiota which could compromise Treg responses and enhance Th17/Treg imbalance. Alternatively, altered FcγR or subneutralized IgG antibodies might induce antibody-dependent enhancement (ADE) of immune reaction, and autoantibodies to form immune complex or to bind endothelial cells and induce abnormal hyperinflammation. In summary, the autoimmune vasculitis of KD or MIS-C can be mediated by a genetic variant of HLA, FcγR, and/or ADE resulting in hyperinflammation with Th17/Treg imbalance. (D) Prevention of MIS-C can be made based on infection immunity and autoimmunity described above. A series of sequential steps ([1]–[8]) as indicated can be utilized to prevent the life-threatening MIS-C.
As shown in Figure 1A, the SARS-CoV-2 virus enters the host cells via ACE2 where a serine protease cleaves the viral spike (S) protein and allows the virus to fuse with the plasma membrane for internalization (109). Host RNA sensing receptors, such as RIG-1 (DDX58), TLR3, TLR7, and/or TLR8, detect the internalized virus, induce IFN production via MyD88, TRIF (TICAM1), IRF3, and/or IRF7 pathways, and promote the production of proinflammatory cytokines via MAPK (e.g., p38) and NFkB pathways (110, 111). A virulent virus can hijack RNA sensing receptors and pathways or activate casein kinase 2 (CK2) for the enhancement of budding viral particles (112). While the initial virus load is low or the virus belongs to a less virulent strain, the host RNA sensing receptor(s) of RIG-1 and TLR7 signaling pathways will mediate an effective induction of IFNs and antigen presentation for an optimal adaptive immunity with T-cell immunity and B-cell production of neutralizing antibodies (Abs) for virus clearance. Alternatively, while the virus load is high, or the TLR7 is either of congenital deficit (88) or of acquired deficit as in the elderly (113), or the viral glycoproteins (antigens) could suppress the MyD88, TRIF, IRF3, and/or IRF7 signaling pathways (111, 114), the antigen-presenting cells (APCs) are hijacked and present the viral antigen with an altered signal for the polarization of naïve Th cells (Tho) toward Th17 response with inflammatory cytokines production, but not Treg regulation for a proper Th1 cell immunity and/or Th2 humoral (B cell) response for neutralizing antibody production. The virus will multiply effectively, and a large number of antigens (PAMPs) will be released to augment Th17/Treg imbalance and promote cytokine storm, leading to ARDS with neutrophilia and lymphopenia, epithelial cell damage, vascular leakage, and/or coagulopathy in COVID-19 (Figure 1B).
In convalescence (Figure 1C), most patients recovered due to efficient adaptive immunity of Th1 cell immunity and Th2 neutralizing antibodies. While the viral antigens (PAMPs) interact with certain HLA subtype(s) of APCs and induce altered autoimmunity with Th17/Treg imbalance, in which an augmented cytokine storm of IL-6, IL-17A, and/or IL-10 expression and abnormal autoantibodies could result in MIS-C with systemic vasculitis, thrombosis, and shock as seen in MIS-C. The abnormal virus-host response may not only depend on genetic variants, e.g., HLA subtypes (87) and TLR7 variants (88), but also host milieu, e.g., homeostasis of vitamins and microbiota which could maintain better Treg responses for anti-inflammatory reactions (115–117). Alternatively, altered FcγR or subneutralized IgG antibodies might induce ADE of immune reaction. ADE resulting from the interaction of the FcγRIIA with a variant polymorphism had been found in SARS-CoV-1 infections (118), and a similar mechanism has been demonstrated in vitro in COVID-19 (119). It is possible that the autoimmune vasculitis of KD or MIS-C is mediated by a genetic variant of HLA, FcγR, and/or ADE resulting in hyperinflammation with Th17/Treg imbalance. However, the Th17/Treg imbalance may be different between MIS-C and KD because the Th17 mediators were elevated in both the diseases but the immunosuppressive mediators: SCF, TWEAK, and ADA were lower in KD than in MIS-C (108). In patients with KD or MIS-C with failure of IVIG and corticosteroids treatment, additional immunotherapies might be applicable by targeting different Th17/Treg imbalances.
Based on the postulated immunopathogenesis of COVID-19 associated MIS-C described above, we could make a series of sequential steps ([1]–[8]) to prevent the life-threatening MIS-C as indicated in Figure 1D and as described below:
[1] Blocking virus entry by neutralizing Abs. In a meta-analysis of 12 controlled trials with more than 4,000 participants, transfusions of convalescent plasma with neutralizing Abs interrupted the virus-ACE2 interaction. The treatment in hospitalized COVID-19 patients reduced the mortality rate by 57% (10 vs. 22%; OR: 0.43, p < 0.001) (120). Similarly, convalescent plasma or neutralizing monoclonal antibodies (MoAbs) have also rescued patients with Ebola (121), SARS (122), and Middle East respiratory syndrome (MERS) (123). Thus, early administration of hyperimmune or recombinant MoAbs with neutralizing Abs directed against SARS-CoV-2 should be able to decrease virus load and raise better immune response toward balanced Th17/Treg reaction resulting in less severity and less autoimmunity.
[2] Decreasing viral load. There are many in vitro studies showing that several potential anti-COVID-19 agents could block the entry, replication, and/or shedding of SARS-CoV-2 (112, 124). The decrease of viral replication and shedding could be made by the inhibition of virus-cell fusion, virus and host proteases, lysosome acidification, RNA synthetase, and virus budding (124, 125). A proper regimen (e.g., remdesivir, avigan, or silmitasertib) to decrease the virus transmission between the infected and non-infected cells may enhance immune response and mitigate the possible autoimmunity. A combination of neutralizing MoAbs and anti-virus agent may induce a synergistic effect.
[3] Screening genetic variants. Similar to KD which has been linked to certain alleles of HLA subtypes in regard to disease susceptibility and severity, the severity of COVID-19 has been proposed to be associated with HLA-B*46:01 in a computational simulation by using SARS-CoV-2 whole-genome peptides for simulating their binding to 145 MHC class I HLA-A, -B, and -C genotypes (87). Moreover, a recent report showed that a mutant (D839Y/N/E) from a European strain of SARS-CoV-2 could serve as a superantigen to induce T-cell receptor activation, resulting in hyperinflammatory response, which may be implicated in the development of MIS-C as well as cytokine storm in adult patients with COVID-19 (126). Deletion or mutation of TLR7 has also been attributed to more severity of COVID-19 in young adults (88). Further studies to identify the risk genetic variants for severity and/or autoimmunity of COVID-19 would help develop a screening genetic test for protecting susceptible children from contacts of COVID-19 and incubate better Th17/Treg balance by nurturing internal milieu with proper homeostasis of vitamin D, vitamin A, and microbiota (104–106).
[4] Targeting cytokine storm. In an early trial with anti-IL6R for patients with COVID-19 hospitalized with cardiopulmonary exacerbation showed potential benefits in decreasing CRP levels, fever, and severity (127). However, later randomized trials demonstrated no significant effects on the severity or fatality of COVID-19 (128). Taken together, aiming at a single target of one cytokine action may be ineffective but a combined regimen or sequential targeting may be required for eliminating the cytokine storm mediated by a couple of hyperinflammatory cytokines in COVID-19 or MIS-C. Th17 mediators, Il-6 and IL-17A and Th1 down-stream mediators, TNF-α and IP-10, more prominently increased in KD than in MIS-C (27, 28, 45, 108), suggesting that targeting IL-17A by Secukinumab or anti-TNFα could be considered in patients with KD with IVIG resistance or with KDSS. Children with MIS-C, who did not have IL-17A or TNFα overexpression (108), may be treated with a combination of IVIG, corticosteroids, and recombinant IL-1-receptor antagonist, Anakinra.
[5] Balancing Th17/Treg immune response. Abnormal immune regulation has been shown in patients with KD or MIS-C (46–50). Both genetic and epigenetic alterations in Treg pathways have been demonstrated in patients with KD (53– 56). The induction and/or stabilization of Treg cell development is affected by endogenous milieu, such as vitamins and metabolites from microbiota (115– 117, 129–131). Vitamin D levels have been shown lower in many patients with COVID-19 and associated with increased inflammatory cytokines and an increased risk of pneumonia (129). The lower vitamin D concentration is not only linked to higher severity of COVID-19 (130, 131), but also associated with an increase in thrombotic episodes (132, 133), which are frequently observed in COVID-19 associated MIS-C (4–9). Vitamin D deficiency has been also shown to be associated with KD with IVIG resistance (134). Moreover, microbiota have recently been shown to coordinate adipocyte-derived mesenchymal stem cells to combat autoimmunity of Type 1 diabetes in mice (135), and mesenchymal stem cells (MSC) or their exosomes have been proposed to eliminate hyperinflammation of COVID-19 (136, 137). We have also recently shown that exosomes derived from MSCs (MSC-EVs) could rescue inflammatory neuropathic pain (138). Evidence accumulated has demonstrated that the effects of MSCs and exosomes derived from MSCs are useful in treating inflammatory diseases and fibrosis (139, 140). This regimen may be suitable for not only cytokine storm but also post-infectious pulmonary fibrosis. In addition, epigenetic modulations of FOXP3 expression by DNA methylation and/or miRNA expression (54–60), may also be applicable to correct the Th17/Treg imbalance.
[6] Targeting ADE and autoantibodies. ADE of immunopathology has been concerned to potentially happen in dengue virus, Zika virus, Ebola virus, respiratory syncytial virus (RSV), and coronaviruses (119). The potential ADE in MIS-C could be treated by using IVIG with and without corticosteroids as shown in patients with MIS-C (4–9), or might be rescued by the elimination of the glycosylation site at N297 of the IgG Fc portion or by a mutation in the Fc region resulting in an effective antibody neutralization but not ADE (119). Several autoantibodies, such as autoantibodies to MAP2K2, CSNK1A1, CSNK2A1, and CSNK1E1 were notably found in patients with MIS-C, and autoantibodies directed against EDIL3 were exclusively found in patients with KD (108), suggesting these autoantibodies might be used as biomarkers for differential diagnosis and their anti-idiotypic antibodies might be used for prevention of autoimmune vasculitis.
[7] Targeting signal transduction pathways. COVID-19 has been shown to induce hyperactivation of TLR-mediated MAPK pathway and CK2-mediated filopodial protrusion of viral shedding (112). Inhibition of p38 activation has been shown to decrease viral replication and cytokine induction in in vitro cell model (112). This is further supported by a recent report showing autoantibodies to MAP2K2, and three members of the casein kinase family (CSNK1A1, CSNK2A1, and CSNK1E1) are notable in children with MIS-C (108). Inhibitors of the phosphokinases which are activated in an in vitro model of SARS-CoV-2 infection, including CK2, CDK, AXL, and PIKFYVE kinases, may possess antiviral efficacy. A combination of different inhibitors of the kinases may have a synergistic effect on anti-viral and anti-inflammatory responses. A recent study showed a combination of a viral protease inhibitor, GC376, and the RNA-dependent RNA synthetase inhibitor, remdesivir, offered sterilizing additive effects (125). In addition, a proteasome inhibitor MG132 which could inhibit IL-6/TGF-β-mediated downregulation of FOXP3 protein may potentially raise the Treg activity (60).
[8] Anti-hemophagocytosis. Hemophagocytosis syndrome also called MAS usually occurs in patients with autoimmune disorders (141, 142). Interestingly, patients with KDSS (20–22) or MIS-C (9, 19) can have IVIG resistance associated with the hemophagocytosis with anemia, elevated Th1 mediator, such as IFNγ, associated with hyperferritinemia and hypertriglyceridemia. In this situation, a combination of IVIG with cyclosporin-A, anti-TNF-α, and/or MSC administration may be required (9, 30, 98–101).
In summary, the autoimmune vasculitis of KD, KDSS, or MIS-C is mediated by a genetic variant of HLA, FcγR, and/or ADE resulting in hyperinflammation with Th17/Treg imbalance with augmented Th17/Th1 mediators: IL-6, IL-10, IP-10, IFNγ, and IL-17A, and a lower expression of Treg-signaling molecules, FoxP3 and TGF-β, and other suppressive immune mediators. Th17/Treg imbalances among them share similar activation pathways but different regulatory (suppressive) pathway. Based on the similar and different immunopathogenesis, we can make early protection, prevention, and precision treatment of the diseases beyond IVIG and corticosteroids therapies. Evolution of immunotherapies for the diseases has shown that IVIG alone or combined with corticosteroids is the standard treatment for KD, KDSS, and MIS-C. However, some patients are resistant to these therapies, and these susceptible individuals must be detected and given the treatment which can render an early block of viral entry, viral replication and/or shedding, and combat Th17/Treg imbalance by anti-cytokine or pro-Treg for reversing the hyperinflammation and IVIG resistance. Clarifying phenotypes, genetic susceptibility, and hyperinflammatory mechanisms of KD, KDSS, and MIS-C with and without IVIG resistance may help develop a so-called “Know thyself, enemy (pathogen) and ever-victorious” strategy for prevention and immunotherapy for KD and/or MISC.
The original contributions presented in the study are cited properly in the references, further inquiries can be directed to the corresponding author/s.
M-RC collected the references and summarized the KD studies in Mackay Children's Hospital for drafting the manuscript. H-CK collected the references and summarized the KD studies in Kaohsiung Chang Gung Memorial Hospital for drafting the manuscript. Y-JL drafted the scheme and revised the manuscript. HC provided the information and references regarding KDSS and compared the phenotypes among KD, KDSS, and MIS-C. SL provided the references and suggestions for the section regarding epigenetic controls of Th17/Treg balance on the autoimmunity of KD and MIS-C. H-CL drafted the section of Treg balance influenced by milieu conditions, such as homeostasis of vitamin D and microbiota. KY designed the article scheme and organized the information for the completion of the article approved by all authors before submission. All authors contributed to the article and approved the submitted version.
This work was supported by a grant from MOST 108-2314-B-195-021-MY3 (for KY).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
1. Dong Y, Mo X, Hu Y, Qi X, Jiang F, Jiang Z, et al. Epidemiology of Covid-19 among children in China. Pediatrics. (2020) 145:e20200702. doi: 10.1542/peds.2020-0702
2. Ludvigsson JF. Systematic review of Covid-19 in children shows milder cases and a better prognosis than adults. Acta Paediatr. (2020) 109:1088–95. doi: 10.1111/apa.15270
3. Tung Ho CL, Oligbu P, Ojubolamo O, Pervaiz M, Oligbu G. Clinical characteristics of children with Covid-19. AIMS Public Health. (2020) 7:258–273. doi: 10.3934/publichealth.2020022
4. Riphagen S, Gomez X, Gonzales-Martinez C, Wilkinson N, Theocharis P. Hyperinflammatory shock in children during Covid-19 pandemic. Lancet. (2020) 395:1607–8. doi: 10.1016/S0140-6736(20)31094-1
5. Verdoni L, Mazza A, Gervasoni A, Martelli L, Ruggeri M, Ciuffreda M, et al. An outbreak of severe Kawasaki-like disease at the Italian epicentre of the SARS-CoV-2 epidemic: an observational cohort study. Lancet. (2020) 395:1771–8. doi: 10.1016/S0140-6736(20)31103-X
6. Cheung EW, Zachariah P, Gorelik M, Boneparth A, Kernie SG, Orange JS, et al. Multisystem inflammatory syndrome related to Covid-19 in previously healthy children and adolescents in New York City. JAMA. (2020) 8:e2010374. doi: 10.1001/jama.2020.10374
7. Pouletty M, Borocco C, Ouldali N, Caseris M, Basmaci R, Lachaume N, et al. Paediatric multisystem inflammatory syndrome temporally associated with SARS-CoV-2 mimicking Kawasaki disease (Kawa-Covid-19): a multicentre cohort. Ann Rheum Dis. (2020) 79:999–1006. doi: 10.1136/annrheumdis-2020-217960
8. Belhadjer Z, Méot M, Bajolle F, Khraiche D, Legendre A, Abakka S, et al. Acute heart failure in multisystem inflammatory syndrome in children (MIS-C) in the context of global SARS-CoV-2 pandemic. Circulation. (2020) 142:429–36. doi: 10.1161/CIRCULATIONAHA.120.048360
9. Feldstein LR, Rose EB, Horwitz SM, Collins JP, Newhams MM, Son MBF, et al. Multisystem inflammatory syndrome in U.S. children and adolescents. N Engl J Med. (2020) 383:334–46. doi: 10.1056/NEJMoa2021680
10. Kawasaki T, Kosaki F, Okawa S, Shigematsu I, Yanagawa H. A new infantile acute febrile mucocutaneous lymph node syndrome (MLNS) prevailing in Japan. Pediatrics. (1974) 54:271–6.
11. Wang CL, Wu YT, Liu CA, Kuo HC, Yang KD. Kawasaki disease: infection, immunity and genetics. Pediatr Infect Dis J. (2005) 24:998–1004. doi: 10.1097/01.inf.0000183786.70519.fa
12. Lin MT, Wu MH. The global epidemiology of Kawasaki disease: Review and future perspectives. Glob Cardiol Sci Pract. (2017) 3:e201720. doi: 10.21542/gcsp.2017.20
13. Witt MT, Minich LL, Bohnsack JF, Young PC. Kawasaki disease: more patients are being diagnosed who do not meet American Heart Association criteria. Pediatrics. (1999) 104:e10. doi: 10.1542/peds.104.1.e10
14. Singh S, Agarwal S, Bhattad S, Gupta A, Suri D, Rawat A, et al. Kawasaki disease in infants below 6 months: a clinical conundrum? Int J Rheum Dis. (2016) 19:924–8. doi: 10.1111/1756-185X.12854
15. Sudo D, Monobe Y, Yashiro M, Mieno MN, Uehara R, Tsuchiya K, et al. Coronary artery lesions of incomplete Kawasaki disease: a nationwide survey in Japan. Eur J Pediatr. (2012) 171:651–6. doi: 10.1007/s00431-011-1630-3
16. Burns JC. History of the worldwide emergence of Kawasaki disease. Int J Rheum Dis. (2018) 21:13–15. doi: 10.1111/1756-185X.13214
17. Kuo HC, Yang KD, Chang WC, Ger LP, Hsieh KH. Kawasaki Disease: An update on diagnosis and treatment. Pediatr Neonatol. (2012) 53:4–11. doi: 10.1016/j.pedneo.2011.11.003
18. Nakamura Y. Kawasaki disease: epidemiology and the lessons from it. Int J Rheum Dis. (2018) 21:16–9. doi: 10.1111/1756-185X.13211
19. Whittaker E, Bamford A, Kenny J, Kaforou M, Jones CE, Shah P, et al. Clinical characteristics of 58 children with a pediatric inflammatory multisystem syndrome temporally associated With SARS-CoV-2. JAMA. (2020) 8:e2010369. doi: 10.1001/jama.2020.10369
20. Kanegaye JT, Wilder MS, Molkara D, Frazer JR, Pancheri J, Tremoulet AH, et al. Recognition of a Kawasaki disease shock syndrome. Pediatrics. (2009) 123:e783–9. doi: 10.1542/peds.2008-1871
21. Chen PS, Chi H, Huang FY, Peng CC, Chen MR, Chiu NC. Clinical manifestations of Kawasaki disease shock syndrome: a case-control study. J Microbiol Immunol Infect. (2015) 48:43–50. doi: 10.1016/j.jmii.2013.06.005
22. Gamez-Gonzalez LB, Moribe-Quintero I, Cisneros-Castolo M, Varela-Ortiz J, Muñoz-Ramírez M, Garrido-García M, et al. Kawasaki disease shock syndrome: Unique and severe subtype of Kawasaki disease. Pediatr Int. (2018) 60:781–90. doi: 10.1111/ped.13614
23. Weisberg SP, Connors T, Zhu Y, Baldwin M, Lin WH, Wontakal S, et al. Antibody responses to SARS-CoV2 are distinct in children with MIS-C compared to adults with Covid-19. MedRxiv. (2020) 2020.07.12.20151068.
24. Low DE. Toxic shock syndrome: major advances in pathogenesis, but not treatment. Crit Care Clin. (2013) 29:651–75. doi: 10.1016/j.ccc.2013.03.012
25. Esper F, Shapiro ED, Weibel C, Ferguson D, Landry ML, Kahn JS. Association between a novel human coronavirus and Kawasaki disease. J Infect Dis. (2005) 191:499–502. doi: 10.1086/428291
26. Chang LY, Lu CY, Shao PL, Lee PI, Lin MT, Fan TY, et al. Viral infections associated with Kawasaki disease. J Formos Med Assoc. (2014) 113:148–54. doi: 10.1016/j.jfma.2013.12.008
27. Li Y, Zheng Q, Zou L, Wu J, Guo L, Teng L, et al. Kawasaki disease shock syndrome: clinical characteristics and possible use of IL-6, IL-10 and IFN-γ as biomarkers for early recognition. Pediatr Rheumatol Online J. (2019) 17:1. doi: 10.1186/s12969-018-0303-4
28. Diorio C, Henrickson SE, Vella LA, McNerney KO, Weirick ME, Gouma S, et al. Multisystem inflammatory syndrome in children and Covid-19 are distinct presentations of SARS-CoV-2. J Clin Invest. (2020) 130:5967–75. doi: 10.1172/JCI140970
29. Lee KY, Rhim JW, Kang JH. Immunopathogenesis of COVID-19 and early immunomodulators. Clin Exp Pediatr. (2020) 63:239–50. doi: 10.3345/cep.2020.00759
30. Lee KY. Pneumonia, acute respiratory distress syndrome, and early immune-modulator therapy. Int J Mol Sci. (2017) 18:388. doi: 10.3390/ijms18020388
31. Rhim JW, Kang HM, Han JW, Lee KY. A presumed etiology of Kawasaki disease based on epidemiological comparison with infectious or immune-mediated diseases. Front Pediatr. (2019) 7:202. doi: 10.3389/fped.2019.00202
32. Meckiff BJ, Ramírez-Suástegui C, Fajardo V, Chee SJ, Kusnadi A, Simon H, et al. Imbalance of regulatory and cytotoxic SARS-CoV-2-reactive CD4(+) T Cells in COVID-19. Cell. (2020) 183:1340–53.e16. doi: 10.1016/j.cell.2020.10.001
33. Marrani E, Burns JC, Cimaz R. How should we classify Kawasaki disease? Front Immunol. (2018) 9:2974. doi: 10.3389/fimmu.2018.02974
34. Sakurai Y. Autoimmune aspects of Kawasaki disease. J Investig Allergol Clin Immunol. (2019) 29:251–61. doi: 10.18176/jiaci.0300
35. Liu L, Wei Q, Lin Q, Fang J, Wang H, Kwok H, et al. Anti-spike IgG causes severe acute lung injury by skewing macrophage responses during acute SARS-CoV infection. JCI Insight. (2019) 4:e123158. doi: 10.1172/jci.insight.123158
36. Jaume M, Yip MS, Cheung CY, Leung HL, Li PH, Kien F, et al. Anti-severe acute respiratory syndrome coronavirus spike antibodies trigger infection of human immune cells via a pH- and cysteine protease-independent FcγR pathway. J Virol. (2011) 85:10582–97. doi: 10.1128/JVI.00671-11
37. Yuan FF, Tanner J, Chan PKS, Biffin S, Dyer WB, Geczy AF, et al. Influence of FcγRIIA and MBL polymorphisms on severe acute respiratory syndrome. Tissue Antigens. (2005) 66:291–6. doi: 10.1111/j.1399-0039.2005.00476.x
38. Lee CH, Chen RF, Liu JW, Yeh WT, Chang JC, Liu PM, et al. Altered p38 mitogen-activated protein kinase expression in different leukocytes with increment of immunosuppressive mediators in patients with severe acute respiratory syndrome. J Immunol. (2004) 172:7841–7. doi: 10.4049/jimmunol.172.12.7841
39. Lee YS, Chen CH, Chao A, Chen ES, Wei ML, Chen LK, et al. Molecular signature of clinical severity in recovering patients with severe acute respiratory syndrome coronavirus (SARS-CoV). BMC Genomics. (2005) 6:132. doi: 10.1186/1471-2164-6-132
40. Wang CL, Wu YT, Lee CJ, Liu HC, Huang LT, Yang KD. Decreased nitric oxide production after intravenous immunoglobulin treatment in patients with Kawasaki disease. J Pediatr. (2002) 141:560–5. doi: 10.1067/mpd.2002.127505
41. Wang CL, Wu YT, Liu CA, Lin MW, Lee CJ, Huang LT, et al. Expression of CD40 ligand on CD4+ T-cells and platelets correlated to the coronary artery lesion and disease progress in Kawasaki disease. Pediatrics. (2003) 111:E140–7. doi: 10.1542/peds.111.2.e140
42. McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation. (2017) 135:e927–99. doi: 10.1161/CIR.0000000000000484
43. Eleftheriou D, Levin M, Shingadia D, Tulloh R, Klein NJ, Brogan PA. Management of Kawasaki disease. Arch Dis Child. (2014) 99:74–83. doi: 10.1136/archdischild-2012-302841
44. Kato H, Koike S, Yokoyama T. Kawasaki disease: effect of treatment on coronary artery involvement. Pediatrics. (1979) 63:175–9.
45. Hiromichi Hamada, Hiroyuki Suzuki, Jun Abe, et al. Inflammatory cytokine profiles during Cyclosporin treatment for immunoglobulin-resistant Kawasaki disease. Cytokine. (2012) 60:681–5. doi: 10.1016/j.cyto.2012.08.006
46. Kuo HC, Wang CL, Liang CD, Yu HR, Huang CF, Wang L, et al. Association of lower eosinophil-related T helper 2 (Th2) cytokines with coronary artery lesions in Kawasaki disease. Pediatr Allergy Immunol. (2009) 20:266–72. doi: 10.1111/j.1399-3038.2008.00779.x
47. Guo MM, Tseng WN, Ko CH, Pan HM, Hsieh KS, Kuo HC. Th17- and Treg-related cytokine and mRNA expression are associated with acute and resolving Kawasaki disease. Allergy. (2015) 70:310–8. doi: 10.1111/all.12558
48. Cho HJ, Bak SY, Kim SY, Yoo R, Baek HS, Yang S, et al. High neutrophil: lymphocyte ratio is associated with refractory Kawasaki disease. Pediatr Int. (2017) 59:669–74. doi: 10.1111/ped.13240
49. Li S, Jiang L, Li X, Lin F, Wang Y, Li B, et al. Clinical and pathological investigation of patients with severe Covid-19. JCI Insight. (2020) 5:e138070. doi: 10.1172/jci.insight.138070
50. Lin L, Lu L, Cao W, Li T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection-a review of immune changes in patients with viral pneumonia. Emerg Microbes Infect. (2020) 9:727–32. doi: 10.1080/22221751.2020.1746199
51. Gao Y, Tang J, Chen W, Li Q, Nie J, Lin F, et al. Inflammation negatively regulates FOXP3 and regulatory T-cell function via DBC1. Proc Natl Acad Sci USA. (2015) 112:E3246–54. doi: 10.1073/pnas.1421463112
52. Gao Z, Gao Y, Li Z, Chen Z, Lu D, Tsun A, et al. Synergy between IL-6 and TGF-β signaling promotes FOXP3 degradation. Int J Clin Exp Pathol. (2012) 5:626–33.
53. Kuo HC, Onouchi Y, Hsu YW, Chen WC, Huang JD, Huang YH, et al. Polymorphisms of transforming growth factor-β signaling pathway and Kawasaki disease in the Taiwanese population. J Hum Genet. (2011) 56:840–5. doi: 10.1038/jhg.2011.113
54. Kuo HC, Chang JC, Kuo HC, Yu HR, Wang CL, Lee CP, et al. Identification of an association between genomic hypomethylation of FCGR2A and susceptibility to Kawasaki disease and intravenous immunoglobulin resistance by DNA methylation array. Arthritis Rheumatol. (2015) 67:828–36. doi: 10.1002/art.38976
55. Huang YH, Li SC, Huang LH, Chen PC, Lin YY, Lin CC, et al. Identifying genetic hypomethylation and upregulation of toll-like receptors in Kawasaki disease. Oncotarget. (2017) 8:11249–58. doi: 10.18632/oncotarget.14497
56. Chen KD, Huang YH, Ming-Huey Guo M, Lin TY, Weng WT, Yang HJ, et al. The human blood DNA methylome identifies crucial role of beta-catenin in the pathogenesis of Kawasaki disease. Oncotarget. (2018) 9:28337–50. doi: 10.18632/oncotarget.25305
57. Huang LH, Kuo HC, Pan CT, Lin YS, Huang YH, Li SC. Multiomics analyses identified epigenetic modulation of the S100A gene family in Kawasaki disease and their significant involvement in neutrophil transendothelial migration. Clin Epigenet. (2018) 10:135. doi: 10.1186/s13148-018-0557-1
58. Kuo HC, Hsieh KS, Ming-Huey Guo M, Weng KP, Ger LP, Chan WC, et al. Next-generation sequencing identifies micro-RNA-based biomarker panel for Kawasaki disease. J Allergy Clin Immunol. (2016) 138:1227–30. doi: 10.1016/j.jaci.2016.04.050
59. Ni FF, Li CR, Li Q, Xia Y, Wang GB, Yang J. Regulatory T cell microRNA expression changes in children with acute Kawasaki disease. Clin Exp Immunol. (2014) 178:384–93. doi: 10.1111/cei.12418
60. Freudenberg K, Lindner N, Dohnke S, Garbe AI, Schallenberg S, Kretschmer K. Critical role of TGF-β and IL-2 receptor signaling in Foxp3 induction by an inhibitor of DNA methylation. Front Immunol. (2018) 9:125. doi: 10.3389/fimmu.2018.00125
61. Shevach EM, Thornton AM. tTregs, pTregs, and iTregs: similarities and differences. Immunol Rev. (2014) 259:88–102. doi: 10.1111/imr.12160
62. Zhao C, Li X, Yang Y, Li Z, Li M, Tan Q, et al. An analysis of Treg/Th17 cells imbalance associated microRNA networks regulated by moxibustion therapy on Zusanli (ST36) and Shenshu (BL23) in mice with collagen induced arthritis. Am J Transl Res. (2019) 11:4029–45. PMCID: PMC6684903
63. Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. (2006) 441:235–8. doi: 10.1038/nature04753
64. Chen S, Dong Y, Yin Y, Krucoff MW. Intravenous immunoglobulin plus corticosteroid to prevent coronary artery abnormalities in Kawasaki disease: a meta-analysis. Heart. (2013) 99:76–82. doi: 10.1136/heartjnl-2012-302126
65. Kim L, Whitaker M, O'Halloran A, Kambhampati A, Chai SJ, Reingold A, et al. Hospitalization rates and characteristics of children aged <18 years hospitalized with laboratory-confirmed Covid-19–Covid-NET, 14 States, March 1–July 25, 2020. MMWR Morb Mortal Wkly Rep. (2020) 69:1081–8. doi: 10.15585/mmwr.mm6932e3
66. Jones I, Bell LCK, Manson JJ, Last A. An adult presentation consistent with PIMS-TS. Lancet Rheumatol. (2020) 2:e520–1. doi: 10.1016/S2665-9913(20)30234-4
67. Shaigany S, Gnirke M, Guttmann A, Chong H, Meehan S, Raabe V, et al. An adult with Kawasaki-like multisystem inflammatory syndrome associated with Covid-19. Lancet. (2020) 396:e8–10. doi: 10.1016/S0140-6736(20)31526-9
68. Galeotti C, Bayry J. Autoimmune and inflammatory diseases following Covid-19. Nat Rev Rheumatol. (2020) 16:413–4. doi: 10.1038/s41584-020-0448-7
69. Gomard-Mennesson E, Landron C, Dauphin C, Epaulard O, Petit C, Green L, et al. Kawasaki disease in adults: report of 10 cases. Medicine (Baltimore). (2010) 89:149–58. doi: 10.1097/MD.0b013e3181df193c
70. Mitani Y, Tsuda E, Kato H, Higaki T, Fujiwara M, Ogawa S, et al. Emergence and characterization of acute coronary syndrome in adults after confirmed or missed history of Kawasaki disease in Japan: a Japanese nationwide survey. Front Pediatr. (2019) 7:275. doi: 10.3389/fped.2019.00275
71. Shimizu C, Sood A, Lau HD, Oharaseki T, Takahashi K, Krous HF, et al. Cardiovascular pathology in 2 young adults with sudden, unexpected death due to coronary aneurysms from Kawasaki disease in childhood. Cardiovasc Pathol. (2015) 24:310–6. doi: 10.1016/j.carpath.2015.02.006
72. Burns JC, Shike H, Gordon JB, Malhotra A, Schoenwetter M, Kawasaki T. Sequelae of Kawasaki disease in adolescents and young adults. J Am Coll Cardiol. (1996) 28:253–7. doi: 10.1016/0735-1097(96)00099-x
73. Wichmann D, Sperhake JP, Lütgehetmann M, Steurer S, Edler C, Heinemann A, et al. Autopsy findings and venous thromboembolism in patients with Covid-19. Ann Intern Med. (2020) 173:268–77. doi: 10.7326/M20-2003
74. Adachi T, Chong JM, Nakajima N, Sano M, Yamazaki J, Miyamoto I, et al. Clinicopathologic and immunohistochemical findings from autopsy of patients with Covid-19, Japan. Emerg Infect Dis. (2020) 26:2157–61. doi: 10.3201/eid2609.201353
75. Nicholls JM, Poon LL, Lee KC, Ng WF, Lai ST, Leung CY, et al. Lung pathology of fatal severe acute respiratory syndrome. Lancet. (2003) 361:1773–8. doi: 10.1016/s0140-6736(03)13413-7
76. Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. (2020) 383:120–8. doi: 10.1056/NEJMoa2015432
77. Takahashi K, Oharaseki T, Naoe S, Wakayama M, Yokouchi Y. Neutrophilic involvement in the damage to coronary arteries in acute stage of Kawasaki disease. Pediatr Int. (2005) 47:305–10. doi: 10.1111/j.1442-200x.2005.02049.x
78. Kobayashi M, Matsumoto Y, Ohya M, Harada K, Kanno H. Histologic and immunohistochemical evaluation of infiltrating inflammatory cells in Kawasaki disease arteritis lesions. Appl Immunohistochem Mol Morphol. (2020) 29:62–7. doi: 10.1097/PAI.0000000000000860
79. Onouchi Y. The genetics of Kawasaki disease. Int J Rheum Dis. (2018) 21:26–30. doi: 10.1111/1756-185X.13218
80. Kato S, Kimura M, Tsuji K, Kusakawa S, Asai T, Juji T, et al. HLA Antigens in Kawasaki Disease. Pediatrics. (1978) 61:252–5.
81. Oh J, Han JW, Lee SJ, Lee KY, Suh BK, Koh DK, et al. Polymorphisms of HLA genes in Korean children with Kawasaki disease. Pediatr Cardiol. (2008) 29:402–8. doi: 10.1007/s00246-007-9146-3
82. Onouchi Y. Molecular genetics of Kawasaki disease. Pediatr Res. (2009) 65:46R−54R. doi: 10.1203/PDR.0b013e31819dba60
83. Huang FY, Chang TY, Chen MR, Hsu CH, Lee HC, Lin SP, et al. Genetic variations of HLA-DRB1 and susceptibility to Kawasaki disease in Taiwanese children. Hum Immunol. (2007) 68:69–74. doi: 10.1016/j.humimm.2006.10.018
84. Huang FY, Lee YJ, Chen MR, Hsu CH, Lin SP, Sung TC, et al. Polymorphism of transmembrane region of MICA gene and Kawasaki disease. Exp Clin Immunogenet. (2000) 17:130–7. doi: 10.1159/000019132
85. Chen MR, Chang ZY, Chiu NC, Chi H, Yang KD, Chang L, et al. Validation of genome-wide associated variants for Kawasaki disease in a Taiwanese case-control sample. Sci Rep. (2020) 10:11756. doi: 10.1038/s41598-020-68673-0
86. Kuo HC, Chang JC, Guo MM, Hsieh KS, Yeter D, Li SC, et al. Gene-gene associations with the susceptibility of Kawasaki disease and coronary artery lesions. PLoS ONE. (2015) 10:e0143056. doi: 10.1371/journal.pone.0143056
87. Nguyen A, David JK, Maden SK, Wood MA, Weeder BR, Nellore A, et al. Human leukocyte antigen susceptibility map for severe acute respiratory syndrome coronavirus 2. J Virol. (2020) 94:e00510-20. doi: 10.1128/JVI.00510-20
88. van der Made CI, Simons A, Schuurs-Hoeijmakers J, van den Heuvel G, Mantere T, Kersten S, et al. Presence of genetic variants among young men with severe Covid-19. JAMA. (2020) 324:1–11. doi: 10.1001/jama.2020.13719
89. Poulas K, Farsalinos K, Zanidis C. Activation of TLR7 and innate immunity as an efficient method against Covid-19 pandemic: imiquimod as a potential therapy. Front Immunol. (2020) 11:1373. doi: 10.3389/fimmu.2020.01373
90. Zhao J, Yang Y, Huang H, Li D, Gu D, Lu X, et al. Relationship between the ABO blood group and the Covid-19 susceptibility. Clin Infect Dis. (2020) 509:220–223. doi: 10.1093/cid/ciaa1150
91. Fan Q, Zhang W, Li B, Li DJ, Zhang J, Zhao F. Association between ABO blood group system and Covid-19 susceptibility in Wuhan. Front Cell Infect Microbiol. (2020) 10:404. doi: 10.3389/fcimb.2020.00404
92. Latz CA, DeCarlo C, Boitano L, Png CYM, Patell R, Conrad MF, et al. Blood type and outcomes in patients with Covid-19. Ann Hematol. (2020) 99:2113–8. doi: 10.1007/s00277-020-04169-1
93. Suda K. Thromboprophylaxis in patients with coronary aneurysms caused by Kawasaki disease. Nihon Rinsho. (2014) 72:1659–63.
94. Terai M, Shulman ST. Prevalence of coronary artery abnormalities in Kawasaki disease is highly dependent on gamma globulin dose but independent of salicylate dose. J Pediatr. (1997) 131:888–93. doi: 10.1016/s0022-3476(97)70038-6
95. Newburger JW, Sleeper LA, McCrindle BW, Maximilian Png CY, Patell R, Conrad MF, et al. Randomized trial of pulsed corticosteroid therapy for primary treatment of Kawasaki disease. N Engl J Med. (2007) 356:663–75. doi: 10.1056/NEJMoa061235
96. Sheianov MV, Udalov YU, Ochkin SS, Bashkov AN, Samoilov AS. Pulse therapy with corticosteroids and intravenous immunoglobulin in the management of severe tocilizumab-resistant Covid-19: A report of three clinical cases. Cureus. (2020) 12:e9038. doi: 10.7759/cureus.9038
97. Kuwabara M, Yashiro M, Ae R, Yanagawa H, Nakamura Y. The effects of early intravenous immunoglobulin therapy for Kawasaki disease: the 22nd nationwide survey in Japan. Int J Cardiol. (2018) 269:334–8. doi: 10.1016/j.ijcard.2018.07.092
98. Yan F, Zhang H, Xiong R, Cheng X, Chen Y, Zhang F. Effect of early intravenous immunoglobulin therapy in Kawasaki disease: a systematic review and meta-analysis. Front Pediatr. (2020) 8:593435. doi: 10.3389/fped.2020.593435
99. Kobayashi T, Saji T, Otani T, Takeuchi K, Nakamura T, Arakawa H, et al. Efficacy of immunoglobulin plus prednisolone for prevention of coronary artery abnormalities in severe Kawasaki disease (RAISE study): a randomised, open-label, blinded-endpoints trial. Lancet. (2012) 379:1613–20. doi: 10.1111/jpc.12048
100. Arane K, Mendelsohn K, Mimouni M, Mimouni F, Koren Y, Brik Simon D, et al. Japanese scoring systems to predict resistance to intravenous immunoglobulin in Kawasaki disease were unreliable for Caucasian Israeli children. Acta Paediatr. (2018) 107:2179–84. doi: 10.1111/apa.14418
101. Tremoulet AH. Adjunctive therapies in Kawasaki disease. Int J Rheum Dis. (2018) 21:76–9. doi: 10.1111/1756-185X.13208
102. García-Pavón S, Yamazaki-Nakashimada MA, Báez M, Borjas-Aguilar KL, Murata C. Kawasaki disease complicated with macrophage activation syndrome: a systematic review. J Pediatr Hematol Oncol. (2017) 39:445–51. doi: 10.1097/MPH.0000000000000872
103. Xie T, Wang Y, Fu S, Wang W, Xie C, Zhang Y, et al. Predictors for intravenous immunoglobulin resistance and coronary artery lesions in Kawasaki disease. Pediatr Rheumatol Online J. (2017) 15:17. doi: 10.1186/s12969-017-0149-1
104. Nozawa T, Imagawa T, Ito S. Coronary-artery aneurysm in Tocilizumab-treated children with Kawasaki's disease. N Engl J Med. (2017) 377:1894–96. doi: 10.1056/NEJMc1709609
105. Magro G. Covid-19: Review on latest available drugs and therapies against SARS-CoV-2. Coagulation and inflammation cross-talking. Virus Res. (2020) 286:198070. doi: 10.1016/j.virusres.2020.198070
106. Giudice V, Pagliano P, Vatrella A, Masullo A, Poto S, Polverino BM, et al. Combination of ruxolitinib and eculizumab for treatment of severe SARS-CoV-2-related acute respiratory distress syndrome: a controlled study. Front Pharmacol. (2020) 11:857. doi: 10.3389/fphar.2020.00857
107. Levin M. Childhood multisystem inflammatory syndrome–a new challenge in the pandemic. N Engl J Med. (2020) 383:393–5. doi: 10.1056/NEJMe2023158
108. Consiglio CR, Cotugno N, Sardh F, Pou C, Amodio D, Rodriguez L, et al. The immunology of multisystem inflammatory syndrome in children with COVID-19. Cell. (2020) 183:968–81.e7. doi: 10.1016/j.cell.2020.09.016
109. Hussain M, Jabeen N, Raza F, Shabbir S, Baig AA, Amanullah A, et al. Structural variations in human ACE2 may influence its binding with SARS-CoV-2 spike protein. J Med Virol. (2020) 24:221–5. doi: 10.1002/jmv.25832
110. Jensen S, Thomsen AR. Sensing of RNA viruses: a review of innate immune receptors involved in recognizing RNA virus invasion. J Virol. (2012) 86:2900–10. doi: 10.1128/JVI.05738-11
111. Schulz KS, Mossman KL. Viral evasion strategies in type I IFN signaling–a summary of recent developments. Front Immunol. (2016) 7:498. doi: 10.3389/fimmu.2016.00498
112. Bouhaddou M, Memon D, Meyer B, White KM, Rezelj VV, Correa Marrero M, et al. The global phosphorylation landscape of SARS-CoV-2 infection. Cell. (2020) 182:685–712.e19. doi: 10.1016/j.cell.2020.06.034
113. Dunston CR, Griffiths HR. The effect of ageing on macrophage Toll-like receptor-mediated responses in the fight against pathogens. Clin Exp Immunol. (2010) 161:407–16. doi: 10.1111/j.1365-2249.2010.04213.x
114. Shi CS, Qi HY, Boularan C, Huang NN, Abu-Asab M, Shelhamer JH, et al. SARS-coronavirus open reading frame-9b suppresses innate immunity by targeting mitochondria and the MAVS/TRAF3/TRAF6 signalosome. J Immunol. (2014) 193:3080–9. doi: 10.4049/jimmunol.1303196
115. Castellani ML, Shaik-Dasthagirisaheb YB, Tripodi D, Anogeianaki A, Felaco P, Toniato E, et al. Interrelationship between vitamins and cytokines in immunity. J Biol Regul Homeost Agents. (2010) 24:385–90.
116. Mucida D, Park Y, Kim G, Turovskaya O, Scott I, Kronenberg M, et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science. (2007) 317:256–60. doi: 10.1126/science.1145697
117. Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature. (2013) 504:451–5. doi: 10.1038/nature12726
118. Wang SF, Tseng SP, Yen CH, Yang JY, Tsao CH, Shen CW, et al. Antibody-dependent SARS coronavirus infection is mediated by antibodies against spike proteins. Biochem Biophys Res Commun. (2014) 451:208–14. doi: 10.1016/j.bbrc.2014.07.090
119. Eroshenko N, Gill T, Keaveney MK, Church GM, Trevejo JM, Rajaniemi H. Implications of antibody-dependent enhancement of infection for SARS-CoV-2 countermeasures. Nat Biotechnol. (2020) 38:789–91. doi: 10.1038/s41587-020-0577-1
120. Joyner MJ, Klassen SA, Senefeld J, Johnson PW, Carter RE, Wiggins CC, et al. Evidence favouring the efficacy of convalescent plasma for Covid-19 therapy. medRxiv. (2020). doi: 10.1101/2020.07.29.20162917
121. Mulangu S, Dodd LE, Davey RT Jr, Mbaya OT, Proschan M, Mukadi D, et al. A randomized, controlled trial of Ebola virus disease therapeutics. N Engl J Med. (2019) 381:2293–303. doi: 10.1056/NEJMoa1910993
122. Cheng Y, Wong R, Soo YOY, Wong WS, Lee CK, M Ng MHL, et al. Use of convalescent plasma therapy in SARS patients in Hong Kong. Eur J Clin Microbiol Infect Dis. (2005) 24:44–6. doi: 10.1007/s10096-004-1271-9
123. Arabi YM, Hajeer AH, Luke T, Raviprakash K, Balkhy H, Johani S, et al. Feasibility of using convalescent plasma immunotherapy for MERS-CoV infection, Saudi Arabia. Emerging Infect Dis. (2016) 22:1554–61. doi: 10.3201/eid2209.151164
124. Vabret N, Britton GJ, Gruber C, Hegde S, Kim J, Kuksin M, et al. Immunology of Covid-19: current state of the science. Immunity. (2020) 52:910–41. doi: 10.1016/j.immuni.2020.05.002
125. Fu L, Ye F, Feng Y, Yu F, Wang Q, Wu Y, et al. Both Boceprevir and GC376 efficaciously inhibit SARS-CoV-2 by targeting its main protease. Nat Commun. (2020) 11:4417. doi: 10.1038/s41467-020-18233-x
126. Cheng MH, Zhang S, Porritt RA, Noval Rivas M, Paschold L, Willscher E, et al. Superantigenic character of an insert unique to SARS-CoV-2 spike supported by skewed TCR repertoire in patients with hyperinflammation. Proc Natl Acad Sci USA. (2020) 117:25254–62. doi: 10.1073/pnas.2010722117
127. Xu X, Han M, Li T, Wei Sun W, Wang D, Fu B, et al. Effective treatment of severe Covid-19 patients with tocilizumab. Proc Natl Acad Sci USA. (2020) 117:10970–5. doi: 10.1073/pnas.2005615117
128. NIH. Covid-19 Treatment Guidelines. Available online at: https://www.covid19treatmentguidelines.nih.gov/immune-based-therapy/immunomodulators/interleukin-6-inhibitors/ (accessed February 09, 2021).
129. Jun JS, Jung YK, Lee DW. Relationship between vitamin D levels and intravenous immunoglobulin resistance in Kawasaki disease. Korean J Pediatr. (2017) 60:216–20. doi: 10.3345/kjp.2017.60.7.216
130. Ilie PC, Stefanescu S, Smith L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin Exp Res. (2020) 32:1195–8. doi: 10.1007/s40520-020-01570-8
131. Biesalski HK. Vitamin D deficiency and co-morbidities in Covid-19 patients – a fatal relationship? Nfs J. (2020) 20:10–21. doi: 10.1016/j.nfs.2020.06.001
132. Rhodes JM, Subramanian S, Laird E, Griffin G, Kenny RA. Perspective: vitamin D deficiency and Covid-19 severity–plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2, and thrombosis. J Intern Med. (2021) 289:97–115. doi: 10.1111/joim.13149
133. Mohammad S, Mishra A, Ashraf MZ. Emerging Role of Vitamin D and its associated molecules in pathways related to pathogenesis of thrombosis. Biomolecules. (2019) 9:649. doi: 10.3390/biom9110649
134. Stagi S, Rigante D, Lepri G, Matucci Cerinic M, Falcini F. Severe vitamin D deficiency in patients with Kawasaki disease: a potential role in the risk to develop heart vascular abnormalities? Clin Rheumatol. (2016) 35:1865–72. doi: 10.1007/s10067-015-2970-6
135. Bassi ÊJ, Moraes-Vieira PM, Moreira-Sá CS, Almeida DC, Vieira LM, Cunha CS, et al. Immune regulatory properties of allogeneic adipose-derived mesenchymal stem cells in the treatment of experimental autoimmune diabetes. Diabetes. (2012) 61:2534–45. doi: 10.2337/db11-0844
136. Tsuchiya A, Takeuchi S, Iwasawa T, Kumagai M, Sato T, Motegi S, et al. Therapeutic potential of mesenchymal stem cells and their exosomes in severe novel coronavirus disease 2019 (Covid-19) cases. Inflamm Regen. (2020) 40:14. doi: 10.1186/s41232-020-00121-y
137. Sengupta V, Sengupta S, Lazo A, Woods P, Nolan A, Bremer N. Exosomes derived from bone marrow mesenchymal stem cells as treatment for severe Covid-19. Stem Cells Dev. (2020) 29:747–54. doi: 10.1089/scd.2020.0080
138. Shiue SJ, Rau RH, Shiue HS, Hung YW, Li ZX, Yang KD, et al. Mesenchymal stem cell exosomes as a cell-free therapy for nerve injury-induced pain in rats. Pain. (2019) 160:210–23. doi: 10.1097/j.pain.0000000000001395
139. Li Y, Li H, Cao Y, Wu F, Ma W, Wang Y, et al. Placenta-derived mesenchymal stem cells improve airway hyperresponsiveness and inflammation in asthmatic rats by modulating the Th17/Treg balance. Mol Med Rep. (2017) 16:8137–45. doi: 10.3892/mmr.2017.7605
140. Hough KP, Chanda D, Duncan SR, Thannickal VJ, Deshane JS. Exosomes in immunoregulation of chronic lung diseases. Allergy. (2017) 72:534–44. doi: 10.1111/all.13086
141. Crayne CB, Albeituni S, Nichols KE, Cron RQ. The immunology of macrophage activation syndrome. Front Immunol. (2019) 10:119. doi: 10.3389/fimmu.2019.00119
Keywords: Kawasaki disease, multisystem inflammatory syndrome in children, susceptibility, autoimmunity, immunotherapy, coronavirus disease-19
Citation: Chen M-R, Kuo H-C, Lee Y-J, Chi H, Li SC, Lee H-C and Yang KD (2021) Phenotype, Susceptibility, Autoimmunity, and Immunotherapy Between Kawasaki Disease and Coronavirus Disease-19 Associated Multisystem Inflammatory Syndrome in Children. Front. Immunol. 12:632890. doi: 10.3389/fimmu.2021.632890
Received: 24 November 2020; Accepted: 22 January 2021;
Published: 26 February 2021.
Edited by:
Joshua Daniel Ooi, Monash University, AustraliaReviewed by:
Kyung Lim Yoon, Kyung Hee University, South KoreaCopyright © 2021 Chen, Kuo, Lee, Chi, Li, Lee and Yang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kuender D. Yang, eWFuZ2tkLnllaEBob3RtYWlsLmNvbQ==
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.