
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Immunol. , 25 November 2020
Sec. Vaccines and Molecular Therapeutics
Volume 11 - 2020 | https://doi.org/10.3389/fimmu.2020.586572
 Xia Xiao1,2,3†
Xia Xiao1,2,3† Conghui Wang1,2,3†
Conghui Wang1,2,3† De Chang4†
De Chang4† Ying Wang1,2,3
Ying Wang1,2,3 Xiaojing Dong1,2,3
Xiaojing Dong1,2,3 Tao Jiao1,2,3
Tao Jiao1,2,3 Zhendong Zhao1
Zhendong Zhao1 Lili Ren1,2,3
Lili Ren1,2,3 Charles S. Dela Cruz5
Charles S. Dela Cruz5 Lokesh Sharma5*
Lokesh Sharma5* Xiaobo Lei1,2,3*
Xiaobo Lei1,2,3* Jianwei Wang1,2,3*
Jianwei Wang1,2,3*COVID-19 pandemic has infected millions of people with mortality exceeding >1 million. There is an urgent need to find therapeutic agents that can help clear the virus to prevent severe disease and death. Identifying effective and safer drugs can provide more options to treat COVID-19 infections either alone or in combination. Here, we performed a high throughput screening of approximately 1,700 US FDA-approved compounds to identify novel therapeutic agents that can effectively inhibit replication of coronaviruses including SARS-CoV-2. Our two-step screen first used a human coronavirus strain OC43 to identify compounds with anti-coronaviral activities. The effective compounds were then screened for their effectiveness in inhibiting SARS-CoV-2. These screens have identified 20 anti-SARS-CoV-2 drugs including previously reported compounds such as hydroxychloroquine, amlodipine besylate, arbidol hydrochloride, tilorone 2HCl, dronedarone hydrochloride, mefloquine, and thioridazine hydrochloride. Five of the newly identified drugs had a safety index (cytotoxic/effective concentration) of >600, indicating a wide therapeutic window compared to hydroxychloroquine which had a safety index of 22 in similar experiments. Mechanistically, five of the effective compounds (fendiline HCl, monensin sodium salt, vortioxetine, sertraline HCl, and salifungin) were found to block SARS-CoV-2 S protein-mediated cell fusion. These FDA-approved compounds can provide much needed therapeutic options that we urgently need during the midst of the pandemic.
Novel coronavirus (CoV)–mediated disease (COVID-19) emerged as a major pandemic and has spread across the world in such a short period since December 2019. As of October 5, 2020, more than 35.1 million confirmed infections have been reported with approximately 1 million deaths (WHO, https://covid19.who.int/). These numbers may be a vast underestimation as many of the infected patients may remain asymptomatic and can only be detected by antibody testing (1). Similarly, many of the deaths may not be accounted for due to a lack of testing. The disease is caused by a novel CoV termed SARS-CoV-2 which belongs to the Coronaviridae family and is the third major CoV pandemic in the last 20 years after Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS) (2–7). The lack of available therapeutic options is a major limiting factor in treating these infections, leading to excessive mortality.
Currently, there is an urgent and unmet need for effective antiviral therapy that can not only decrease the disease burden in the patient but can also decrease the ability of the person to infect others. It is not practical to develop a novel drug for urgent needs such as during the current pandemic, which may take years to confirm safety and efficacy. Alternatively, it sounds like a lucrative option to repurpose the US Food and Drug Administration (FDA) approved drugs for their efficacy against SARS-CoV-2. Earlier screens have found antiviral efficacy of approved therapies such as hydroxychloroquine (8–14); however, these therapies failed to provide any beneficial effects in COVID-19 due to their toxic side effects (13–20). Finding efficacy of an approved drug against SARS-CoV-2 with minimal toxicity can provide much needed therapeutic option to treat COVID-19.
Here we screened approximately 1,700 US FDA-approved compounds to test their ability to inhibit SARS-CoV-2 replication. Here, we report 20 compounds that are highly effective in inhibiting SARS-CoV-2 replication at concentrations that were significantly lower than those having cytotoxic effects. We also investigated the possible mechanism of these compounds.
LLC-MK2 cells (Rhesus monkey kidney cells), which was provided by the Laboratoire des Pathogènes Emergents (LPE), Fondation Mérieux, Lyon, France, were cultured in 64% Hank’s MEM and 32% Earle’s MEM (Gibco, New York, USA) supplemented with 3% fetal bovine serum (FBS) (Hyclone, Utah, USA) and 1% glutamine (Thermo, Massachusetts, USA). Vero cells (African green monkey kidney cell), purchased from ATCC, were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco) supplemented with 10% FBS.
Human coronavirus (HCoV) strain OC43, which is a gift from Peking Union Medical College Hospital, was propagated in LLC-MK2 cells in 0.5% FBS MEM and virus titers were determined via TCID50 with LLC-MK2 cells. SARS-CoV-2 virus was isolated from the respiratory samples of patients in Wuhan of Hubei Province (3). SARS-CoV-2 virus was propagated in Vero cells and used in this study. All experiments with SARS-CoV-2 virus were conducted in a BSL-3 laboratory.
HEK293T cells stably expressing recombinant human ACE2 (293T/hACE2) were maintained in Dulbecco’s MEM containing 10% FBS and 100 units penicillin, and 100 μg of streptomycin per milliliter.
Mouse polyclonal against the OC43 N antibody was prepared in the laboratory. Rabbit polyclonal against SARS-CoV-2 N protein antibody was purchased from Sino Biological (Beijing, China). Alexa Fluor 488–conjugated goat anti-mouse IgG, Alexa Fluor 488–conjugated goat anti-rabbit IgG were purchased from Thermo.
US FDA-approved drug library which contains 1,700 compounds were purchased from TargetMol (Massachusetts, USA). LLC-MK2 cells were seeded at 2 × 104 cells per well in 96-well plates and incubated at 37°C and 5% CO2. The next day, LLC-MK2 cells were treated with the compounds at a concentration of 10 μM. After 1 h of treatment, cells were infected with OC43 at MOI of 1. At 48 h post-infection (hpi), cells were fixed with 4% paraformaldehyde for 20 min at room temperature. Immunofluorescence staining was performed using mouse anti-OC43 NP antibody, followed by anti-mouse Alexa Fluor 488 and DAPI (Sigma, St. Louis, MO). Images were captured by Operetta (PerkinElmer, Massachusetts, USA) at the magnification of 20× objective. The infection ratios were calculated using automated image analysis software (Harmony 3.5.2, PerkinElmer). Remdesivir and DMSO were used as positive and negative controls, respectively.
The positively identified drugs from this screen were used to perform dose-response curves against OC43 on LLC-MK2 and against SARS-CoV-2 using Vero cells as described below.
LLC-MK2 cells (for OC43 infection) or Vero cells (for SARS-CoV-2 infection) were seeded in 96 wells plate one day before infection at the concentration of 2 × 104 cells/well or 1.4 × 104 cells/well, respectively. For IC50, cells were pre-treated for 1 h with each drug at concentrations 0.013, 0.041, 0.123, 0.370, 1.111, 3.333, 10, and 30 μM and then infected with the virus at MOI of 1. Cell culture media was then replaced containing same concentration of the drug to ensure presence of drug at the time of infection. At 48 hpi (OC43) or 24 hpi (SARS-CoV-2), cells were fixed with 4% paraformaldehyde for 20 min at room temperature. Immunofluorescence was conducted with mouse anti-OC43 N protein antibody or rabbit anti-SARS-CoV-2-NP antibody and followed by anti-mouse, or anti-rabbit Alexa Flour 488 and DAPI. Images were performed by Operetta with 20× objective. The IC50 was calculated using automated image analysis software (Harmony 3.5.2, PerkinElmer).
For CC50, cells were pre-treated with each drug at concentrations 0.013, 0.041, 0.123, 0.370, 1.111, 3.333, 10, and 30 μM, respectively. After 48 h (LLC-MK2 cells) or 24 h (Vero cells) post-treatment, cell viability was evaluated by using a CCK8 kit (Yeasen, Beijing, China) according to the manufacturer’s instructions. The selectivity index was calculated using the following formula: SI = CC50/IC50. Graphpad Prism 7.0 was used for analyzing IC50 and CC50.
Cells were fixed with 4% paraformaldehyde for 20 min at room temperature, and permeabilized with 0.5% Triton X-100 for 10 min. Cells were then blocked with 5% BSA and stained with primary antibodies, followed by staining with an Alexa Fluor 488 secondary antibodies. Nuclei were counterstained with DAPI.
Vero cells were pre-treated with indicated concentrations of drugs for 1 h and incubated with SARS-CoV-2 at 0.1 MOI for 1 h. Then, cells were washed with opti-MEM for one time and incubated with indicated concentrations of drugs. At 24 hpi, supernatants were collected and viral RNA in the cell supernatants were extracted by using Direct-zol RNA MiniPrep kit (Zymo Research, CA, USA) according to the manufacturer’s instructions. Viral copy numbers were measured by RT-PCR using primers and probe targeting the SARS-CoV-2 N gene. The reference standard was tenfold diluted from 1 × 109 copies to 1 × 104 copies. PCR amplification cycle was 50°C, 15 min, 95°C, 3 min; 95°C, 15 s, 60°C, 45 s+ Plate Read, 50 cycles. The amplification process, fluorescence signal detection, data storage, and analysis were all completed by quantitative fluorescence PCR and its own software (Bio-Rad CFX Manager). The copies of the virus were calculated according to the standard curve. The inhibition ratio was obtained by dividing the number of copies of the virus in the vehicle control group. The data were nonlinearly fitted by GraphPad 7.0 software to calculate the IC50 of each drug.
Cell-cell fusion assays were performed as described previously (20). Briefly, HEK-293T cells were co-transfected with SARS-CoV-2-S glycoprotein and eGFP. At 24 h post-transfection, cells were digested with trypsin (0.25%) and overlaid on a 50% confluent monolayer of 293T-ACE2 cells at a ratio of 1:1 which were treated with candidate drugs for 1 h. After 24 h incubation, syncytial images were captured with Operetta (PerkinElmer, Massachusetts, USA).
Initial screening was performed using HCoV-OC43 due to its low biosafety concerns. OC43 is a hCoV that usually causes mild disease in humans and cattle (21). The experimental protocol is demonstrated in Figure 1A using LLC-MK2 cells infected with OC43 at an MOI of 1 for 48 h in presence of the US FDA-approved compounds. The inhibitory potentials of these compounds were measured with the treatment of compounds at 10 μM for 48 h. The viral presence was detected by immunostaining for the virus and DAPI staining for the cell nuclei. The inhibitory capacity was measured using the ratio of viral fluorescence to the DAPI and is depicted in Figure 1B. The initial screen obtained 231 compounds that had the ability to inhibit OC43 replication >95%. The remdesivir was used as a positive control (Figures 1C, D).
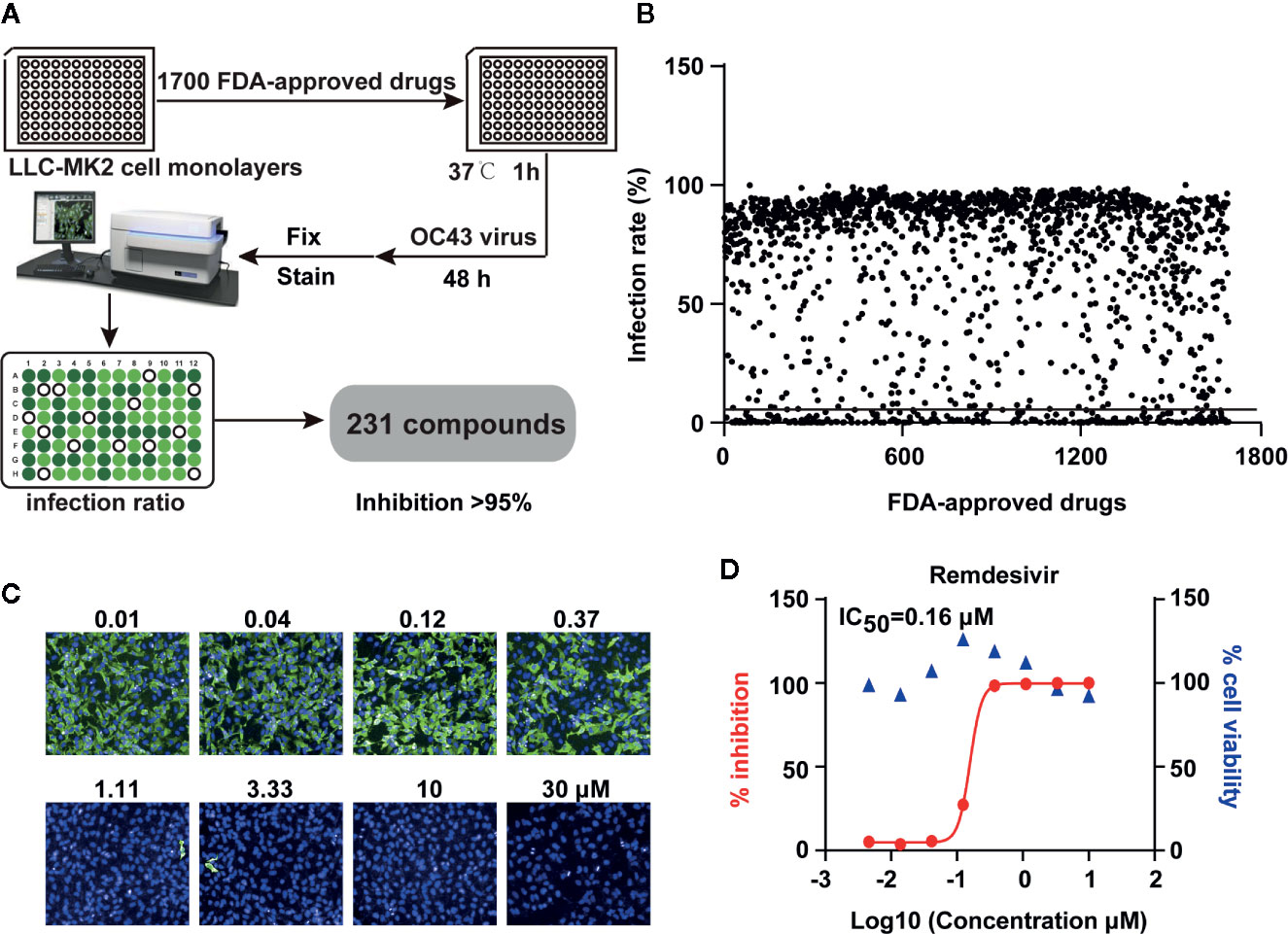
Figure 1 High-throughput screening of US FDA-approved drug library to inhibit human coronavirus OC43 replication in vitro. (A) The strategy of high throughput screening to identify antiviral drugs that effectively inhibit OC43 replication. LLC-MK2 cells were pretreated with FDA-approved drugs at 10 μM for 1 h and infected with 1 MOI of OC43 for 48 h. Cells were then fixed and stained to calculate the infection ratio with Operetta software. (B) Primary screening results of 1,700 FDA-approved drugs against OC43, each dot represents one compound along with the rate of OC43 inhibition. (C) Image samples show signals of OC43 infection in cell cultures. LLC-MK2 cells were treated with indicated doses of remdesivir for 1 h, and then cells were infected with OC43 for 48 h. (D) Cell viability of remdesivir to LLC-MK2 cells were measured by CCK-8 assays. The % inhibitions were calculated according to the data in (C).
Next, we sought to determine the effective concentrations of positively screened drugs in our initial approach and test the toxicity profile of these 231 drugs in relation to their viral inhibitory concentrations. Our data show that 56 of the positively screened drugs were effectively inhibited the viral replication at sub-micromolar concentrations including many of them can almost completely inhibit the viral replication at micromolar range (Figure 2). Surprisingly, the effective drugs against the CoV belonged to a wide range of therapeutic groups including those used for neurological diseases, hormones, enzymes, and antimicrobial agents among others (Table 1). The cytotoxic concentration to kill 50% of cells (CC50) was noted for these drugs by measuring cell viability over similar concentrations. The selective index in our study was found to be >600 for 5 of the screened compounds. The SI for the hydroxychloroquine was 22 in our study, indicating increased safety of newly identified drugs compared to the hydroxychloroquine.
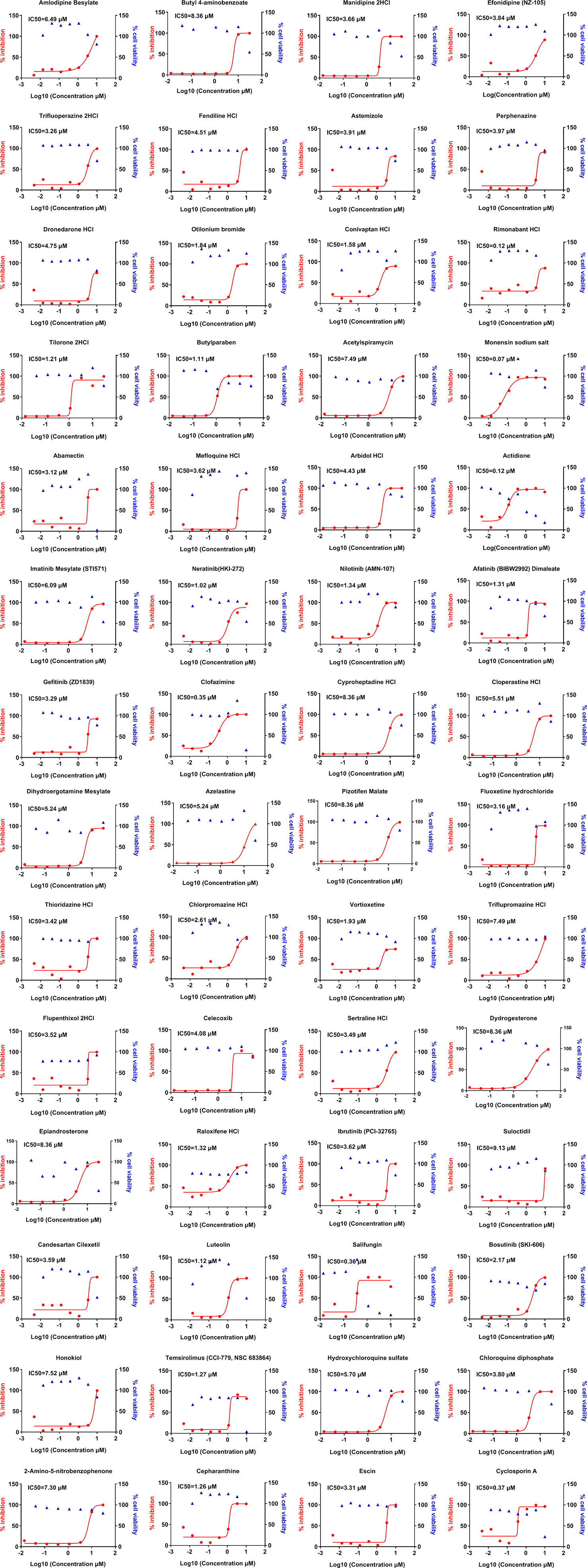
Figure 2 Dose-response curves of selected compounds from the hits against OC43 infection in vitro. LLC-MK2 cells were pretreated with indicated drugs at 37°C for 1 h with eight doses (0.014, 0.041, 0.123, 0.370, 1.111, 3.333, 10, 30 μM) with three-fold dilution followed by infection with OC43 at MOI of 1 for 48 h. In parallel, these compounds’ effects on the cell viability in LLC-MK2 cells were measured by CCK-8 assays. The left Y-axis of the graphs represents % inhibition of the infection (red dots) and the right Y-axis of the graphs presents % cell viability (blue triangles) in the presence of the drugs.
Finally, we sought to determine whether these 56 compounds can show efficacy against SARS-CoV-2, the causative agent of COVID-19. These 56 compounds from the initial screen were tested for their antiviral efficacy against SARS-CoV-2 in Vero cells. SARS-CoV-2 replicates within the Vero cells and causes cytopathic effects in these cells in the absence of any antiviral treatment. We generated the dose-response inhibition curves along with the cytotoxicity curves for these compounds in the presence of SARS-CoV-2 (Figure 3). Remdesivir was used as a positive control. Our data show that 20 of these compounds show significant efficacy in inhibiting SARS-CoV-2 replication with sub micromolar IC50 for many of these drugs such as nilotinib, clofazimine, and raloxifene. The effects also confirmed by immunofluorescence assay (data not shown). These compounds also belong to a wide variety of classes including cardiac glycosides, anti-malarial drug hydroxychloroquine, cyclooxygenase-2 inhibitors, and ion channel blockers. The IC50, CC50, and SI of these compounds are shown in Table 2.
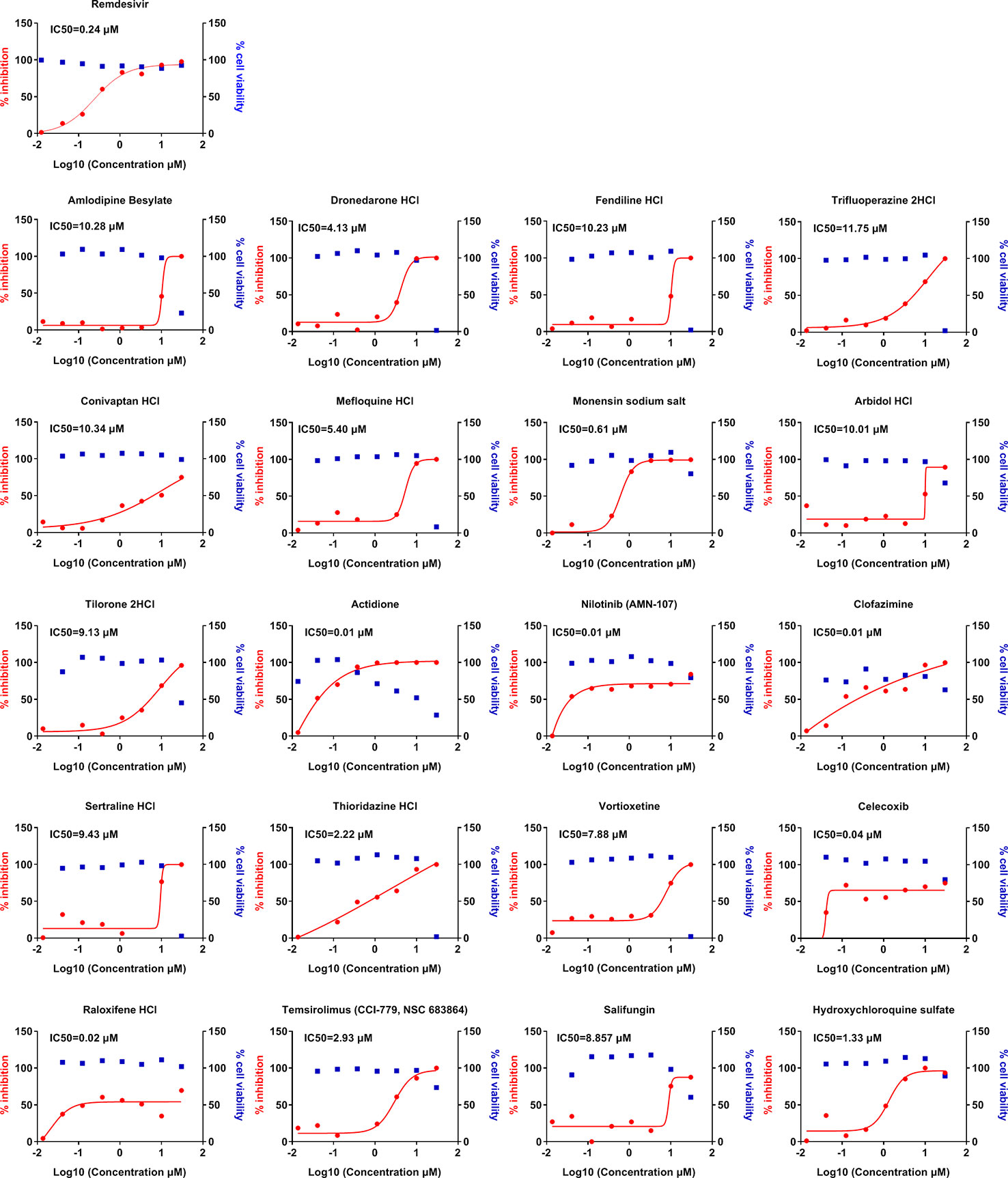
Figure 3 Dose-response curves of selected compounds against SARS-CoV-2 infection. Vero cells were pretreated with indicated drugs at 37°C for 1 h with eight doses (0.014, 0.041, 0.123, 0.370, 1.111, 3.333, 10, and 30 μM) with three-fold dilutions, followed by infection with SARS-CoV-2 at an MOI of 0.1 for 24 h. The viral load in the cell supernatant was quantified by qRT-PCR. Meanwhile, cell viability in the presence of these drugs was measured in Vero cells by CCK-8 assays. The left Y-axis of the graphs represents % inhibition of viral replication (red dots) and the right Y-axis of the graphs indicates % cell viability (blue squares) in the presence of the drugs.
Finally, to determine the mechanism by which the compounds inhibit SARS-CoV-2, we investigated these candidates’ effects on virus entry. First, we constructed the cell-cell fusion assays. As indicated in Figure 4, SARS-CoV-2 S protein expression can initiate cell fusion with ACE2-overexpressed cells, but the control vector did not. Then, we detected the effects of these indicated drugs on S protein-mediated cell fusion. Indicated drugs were added to cells at 10 μM before the co-incubation of the cells. We found that fendiline hydrochloride, monensin sodium salt, vortioxetine, sertraline hydrochloride, and salifungin inhibited the SARS-CoV-2 S protein-mediated cell fusion (Figure 4).
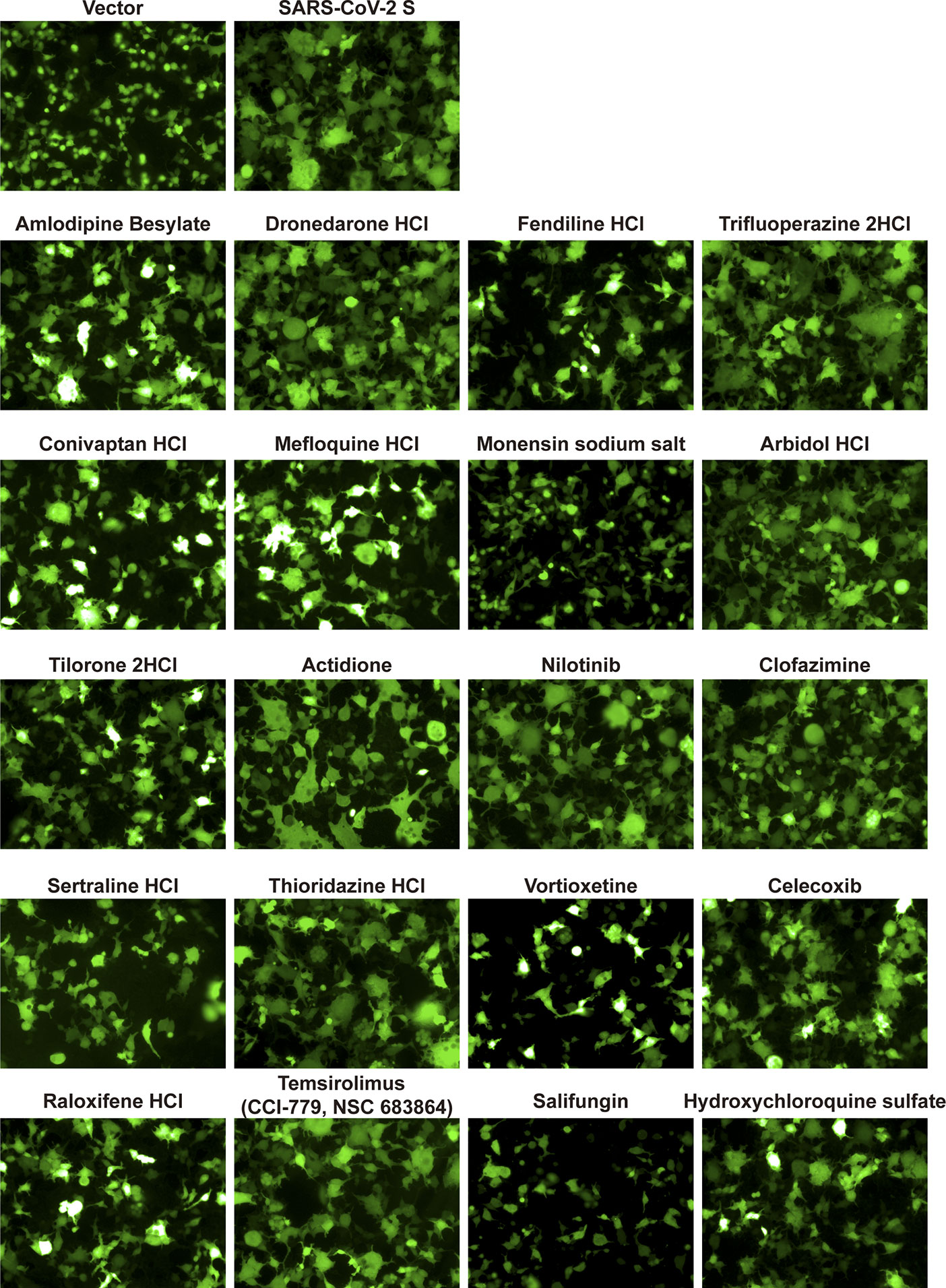
Figure 4 The effect of indicated drugs on cell-cell fusion mediated by SARS-CoV-2 S protein. HEK-293T cells were co-transfected with SARS-CoV-2-S glycoprotein and eGFP. 24 h post-transfection, cells were digested with trypsin (0.25%) and overlaid on a 50% confluent monolayer of 293T-ACE2 cells at a ratio of 1:1 which were treated with candidate drugs at 10 μM for 1 h. After 24-h incubation, images of syncytia were captured with Operetta and analyzed by Harmony software.
The current pandemic of COVID-19 is the third major outbreak in this century and the largest outbreak of the CoV in known human history. The three novel CoV outbreaks in such a short time are strong indicators of the potential threat posed by CoVs. While most respiratory viral infection research has been focused on influenza viruses that cause a huge burden of seasonal flu and occasionally pandemic outbreaks, CoV is likely to emerge as a similar or more severe pathogen than flu in long term.
Given the scale and devastation of the current COVID-19 outbreak and the persistent threat of CoVs in causing human disease, there is an urgent need to find effective and safe therapies to treat these patients. Currently, there are no approved therapies for CoVs including SARS-CoV-2. The experimental therapies being used with known antiviral agents either show limited efficacy (remdesivir) or have high systemic toxicity (hydroxychloroquine), limiting their usefulness (22–24). Finding new therapies that are effective and safe are urgently needed. In this study, we have identified many FDA-approved therapies that are highly effective against CoVs, including 20 of the effective agents against SARS-CoV-2. This screening confirms previous reports demonstrating anti-SARS-CoV-2 activity of hydroxychloroquine, amlodipine besylate, arbidol hydrochloride, tilorone 2HCl, dronedarone hydrochloride, mefloquine, celecoxib, and thioridazine hydrochloride (8–10, 25–27), while identifying additional 12 drugs. However, there are seven drugs have been reported when our manuscript was underreview (28–37). The underlying mechanisms of viral replication inhibition by these drugs are not clear. It is highly unlikely that these compounds will have similar antiviral mechanisms given the vast structural and pharmacological diversities of the effective antiviral compounds in our study. However, it is clear from other viral studies such as influenza or HIV, where antiviral drugs can affect various viral life cycle steps including attachment, entry, replication, assembly, and budding of viral progeny. Five drugs may inhibit S-mediated cell fusion as indicated by our data (Figure 4). Further studies are required to understand the precise mechanisms of each of the effective compounds found in this study.
Toxicity is one of the limiting factors in the therapeutic application of many drugs despite their known antiviral activities. Many of these drugs had SI of >600, showing promise of their usefulness at safe doses. For comparison, the SI of hydroxychloroquine was found to be 22 in our study while SI of amlodipine besylate was found to be ~3, demonstrating a much lower safety profile of this drug. Similarly, other drugs known to have low selective index such as digoxin for their approved use, also show lower SI in our screen. Five of the drugs with SI of >600 include tyrosine kinase inhibitor nilotinib, antibiotics such as clofazimine and actidione, and selective estrogen receptor modulators raloxifene, and non-steroidal anti-inflammatory drug celecoxib.
Betacoronaviruses have raised great public health threats to human beings, as most known HCoVs including all the three virulent HCoVs (SARS-CoV, MERS-CoV, and SARS-CoV-2) and two seasonal HCoVs (OC43 and HKU1) belong to this species (3–7, 21, 38). It is of great value to identify antivirals against a broad spectrum of HCoVs, particularly the Betacoronaviruses, to tackle such threats by pharmaceutical interventions (39, 40). To this end, we first screened the compounds which showed apparent activity of anti-OC43, the most prevalent HCoV circulates worldwide (41). We then narrowed down the candidates by the screening on SARS-CoV-2, resulting in the identification of 20 compounds which can inhibit both OC43 and SARS-CoV-2. Our study provides a foundation for subsequent anti-HCoVs drug screening of a broad spectrum. However, further tests are warranted to verify their efficacies.
In summary, our screen identified 14 previously unknown FDA-approved compounds that are effective in inhibiting SARS-CoV-2 besides confirming the antiviral properties of 7 previously reported compounds, validating our approach. This screen identified five new compounds highly efficacious in inhibiting the viral replication of SARS-CoV-2 with SI >600. Further studies are needed to confirm these drugs’ in vivo efficacy in COVID-19 relevant mouse models such as those with human ACE2 transgene (42) and human clinical studies.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Project conception: JW, XL, and LS. Experimental design: JW, XL, DC, LS, ZZ, and LR. Experimental work: XX, CW, YW, XD, and TJ. Data analysis: JW, XL, LR, CDC, XX, and ZZ. Writing original draft: JW, XL, LS, DC, and XX. Writing review and editing: JW, XL, DC, LS, and CC. All authors contributed to the article and approved the submitted version.
This work was supported by grants from the National Major Sciences & Technology Project for Control and Prevention of Major Infectious Diseases in China (2018ZX10301401 to XL), the National Natural Science Foundation of China (81930063 and 81971948 to JW and XL), Chinese Academy of Medical Sciences (CAMS) Innovation Fund for Medical Sciences (2016-I2M-1-014 and 2016-I2M-1-005 to JW and XL), National Key R&D Program of China (2020YFA0707600 to XL), and Beijing Municipal Science and Technology Project (Z2011000010200005). LS is supported by Parker B Francis Fellowship and American Lung Association Catalyst Award.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
This manuscript has been released as a pre-print at bioRxiv (43).
1. Guo L, Ren L, Yang S, Xiao M, Chang D, Yang F, et al. Profiling Early Humoral Response to Diagnose Novel Coronavirus Disease (COVID-19). Clin Infect Dis (2020) 71(15):778–85. doi: 10.1093/cid/ciaa310
2. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (2020) 395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5
3. Ren L, Wang Y, Wu Z, Xiang Z, Guo L, Xu T, et al. Identification of a novel coronavirus causing severe pneumonia in human: a descriptive study. Chin Med J (Engl) (2020) 133:1015–24. doi: 10.1097/CM9.0000000000000722
4. Drosten C, Gunther S, Preiser W, Van Der Werf S, Brodt HR, Becker S, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med (2003) 348(20):1967–76. doi: 10.1056/NEJMoa030747
5. Zaki A, Van Boheemen S, Bestebroer T, Osterhaus A, Fouchier R. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med (2012) 367(19):1814–20. doi: 10.1056/NEJMoa1211721
6. Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med (2020) 382(8):727–33. doi: 10.1056/NEJMoa2001017
7. Ksiazek T, Erdman D, Goldsmith C, Zaki S, Peret T, Emery S, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med (2003) 348(20):1953–66. doi: 10.1056/NEJMoa030781
8. Caly L, Druce J, Catton M, Jans D, Wagstaff K. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antiviral Res (2020) 178:104787. doi: 10.1016/j.antiviral.2020.104787
9. Lobo-Galo N, Terrazas-Lopez M, Martinez-Martinez A, Diaz-Sanchez AG. FDA-approved thiol-reacting drugs that potentially bind into the SARS-CoV-2 main protease, essential for viral replication. J Biomol Struct Dyn (2020) 1–9. doi: 10.1080/07391102.2020.1764393
10. Jeon S, Ko M, Lee J, Choi I, Byun S, Park S, et al. Identification of antiviral drug candidates against SARS-CoV-2 from FDA-approved drugs. Antimicrob Agents Chemother (2020) 64(7):e00819–20. doi: 10.1128/AAC.00819-20
11. Kandeel M, Al-Nazawi M. Virtual screening and repurposing of FDA approved drugs against COVID-19 main protease. Life Sci (2020) 251:117627. doi: 10.1016/j.lfs.2020.117627
12. Ferner RE, Aronson JK. Chloroquine and hydroxychloroquine in covid-19. BMJ (2020) 369:m1432. doi: 10.1136/bmj.m1432
13. Andreani J, Le Bideau M, Duflot I, Jardot P, Rolland C, Boxberger M, et al. In vitro testing of combined hydroxychloroquine and azithromycin on SARS-CoV-2 shows synergistic effect. Microb Pathog (2020) 145:104228. doi: 10.1016/j.micpath.2020.104228
14. Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, et al. In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis (2020) 71(15):732–9. doi: 10.1093/cid/ciaa237
15. Singh A, Singh A, Shaikh A, Singh R, Misra A. Chloroquine and hydroxychloroquine in the treatment of COVID-19 with or without diabetes: A systematic search and a narrative review with a special reference to India and other developing countries. Diabetes Metab Syndr (2020) 14(3):241–6. doi: 10.1016/j.dsx.2020.03.011
16. Badyal DK, Mahajan R. Chloroquine: Can it be a Novel Drug for COVID-19. Int J Appl Basic Med Res (2020) 10(2):128–30. doi: 10.4103/ijabmr.IJABMR_141_20
17. Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, et al. Compassionate Use of Remdesivir for Patients with Severe Covid-19. N Engl J Med (2020) 382(24):2327–36. doi: 10.1056/NEJMoa2007016
18. Guastalegname M, Vallone A. Could chloroquine /hydroxychloroquine be harmful in Coronavirus Disease 2019 (COVID-19) treatment? Clin Infect Dis (2020) 71(15):888–9. doi: 10.1093/cid/ciaa321
19. Gautret P, Lagier J, Parola P, Hoang V, Meddeb L, Mailhe M, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents (2020) 56(1):105949. doi: 10.1016/j.ijantimicag.2020.105949
20. Ou X, Liu Y, Lei X, Li P, Mi D, Ren L, et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat Commun (2020) 11(1):1620. doi: 10.1038/s41467-020-15562-9
21. Mcintosh K, Dees JH, Becker WB, Kapikian AZ, Chanock RM. Recovery in tracheal organ cultures of novel viruses from patients with respiratory disease. Proc Natl Acad Sci U S A (1967) 57(4):933–40. doi: 10.1073/pnas.57.4.933
22. Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet (2020b) 395(10236):1569–78. doi: 10.1016/S0140-6736(20)31022-9
23. Vinetz JM. Lack of efficacy of hydroxychloroquine in covid-19. BMJ (2020) 369:m2018. doi: 10.1136/bmj.m2018
24. Tang W, Cao Z, Han M, Wang Z, Chen J, Sun W, et al. Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: open label, randomised controlled trial. BMJ (2020) 369:m1849. doi: 10.1136/bmj.m1849
25. Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res (2020a) 30(3):269–71. doi: 10.1038/s41422-020-0282-0
26. Ke Y, Peng T, Yeh T, Huang W, Chang S, Wu S, et al. Artificial intelligence approach fighting COVID-19 with repurposing drugs. Biomed J (2020) 43(4):355–62. doi: 10.1016/j.bj.2020.05.001
27. Gimeno A, Mestres-Truyol J, Ojeda-Montes M, Macip G, Saldivar-Espinoza B, Cereto-Massague A, et al. Prediction of Novel Inhibitors of the Main Protease (M-pro) of SARS-CoV-2 through Consensus Docking and Drug Reposition. Int J Mol Sci (2020) 21(11):3793. doi: 10.3390/ijms21113793
28. Virdi R, Bavisotto R, Hopper N, Vuksanovic N, Melkonian T, Silvaggi N, et al. Discovery of Drug-like Ligands for the Mac1 Domain of SARS-CoV-2 Nsp3. SLAS Discov (2020) 2472555220960428. doi: 10.1177/2472555220960428
29. Gul S, Ozcan O, Asar S, Okyar A, Baris I, Kavakli I. In silico identification of widely used and well-tolerated drugs as potential SARS-CoV-2 3C-like protease and viral RNA-dependent RNA polymerase inhibitors for direct use in clinical trials. J Biomol Struct Dyn (2020) 1–20. doi: 10.1080/07391102.2020.1802346
30. Chandel V, Sharma P, Raj S, Choudhari R, Rathi B, Kumar D. Structure-based drug repurposing for targeting Nsp9 replicase and spike proteins of severe acute respiratory syndrome coronavirus 2. J Biomol Struct Dyn (2020) 1–14. doi: 10.1080/07391102.2020.1811773
31. Ruan Z, Liu C, Guo Y, He Z, Huang X, Jia X, et al. SARS-CoV-2 and SARS-CoV: Virtual screening of potential inhibitors targeting RNA-dependent RNA polymerase activity (NSP12). J Med Virol (2020). doi: 10.1002/jmv.26222. 10.1002/jmv.26222.
32. Jack A, Ferro L, Trnka M, Wehri E, Nadgir A, Costa K, et al. SARS CoV-2 nucleocapsid protein forms condensates with viral genomic RNA. bioRxiv (2020). doi: 10.1101/2020.09.14.295824. 2020.09.14.295824.
33. Chtita S, Belhassan A, Aouidate A, Belaidi S, Bouachrine M, Lakhlifi T. Discovery of Potent SARS-CoV-2 Inhibitors from Approved Antiviral Drugs via Docking Screening. Comb Chem High Throughput Screen (2020). doi: 10.2174/1386207323999200730205447
34. Galimberti S, Petrini M, Barate C, Ricci F, Balducci S, Grassi S, et al. Tyrosine Kinase Inhibitors Play an Antiviral Action in Patients Affected by Chronic Myeloid Leukemia: A Possible Model Supporting Their Use in the Fight Against SARS-CoV-2. Front Oncol (2020) 10:1428. doi: 10.3389/fonc.2020.01428
35. Smetana K Jr., Rosel D, Brabek J. Raloxifene and Bazedoxifene Could Be Promising Candidates for Preventing the COVID-19 Related Cytokine Storm, ARDS and Mortality. In Vivo (2020) 34:3027–8. doi: 10.21873/invivo.12135
36. Rasaeifar B, Gomez-Gutierrez P, Perez J. Molecular Features of Non-Selective Small Molecule Antagonists of the Bradykinin Receptors. Pharmaceut (Basel) (2020) 13(9):E259. doi: 10.3390/ph13090259
37. Elzupir AO. Inhibition of SARS-CoV-2 main protease 3CL(pro) by means of alpha-ketoamide and pyridone-containing pharmaceuticals using in silico molecular docking. J Mol Struct (2020) 1222:128878. doi: 10.1016/j.molstruc.2020.128878
38. Woo P, Lau S, Chu C, Chan K, Tsoi H, Huang Y, et al. Characterization and complete genome sequence of a novel coronavirus, coronavirus HKU1, from patients with pneumonia. J Virol (2005) 79(2):884–95. doi: 10.1128/JVI.79.2.884-895.2005
39. Bonam S, Kaveri S, Sakuntabhai A, Gilardin L, Bayry J. Adjunct Immunotherapies for the Management of Severely Ill COVID-19 Patients. Cell Rep Med (2020) 1(2):100016. doi: 10.1016/j.xcrm.2020.100016
40. Nichols C, Mcclenaghan C, Hanson A, Lee S. Coronavirus proteins as ion channels: Current and potential research. Front Immunol (2020) 11:573339. doi: 10.3389/fimmu.2020.573339
41. Zhang Y, Li J, Xiao Y, Zhang J, Wang Y, Chen L, et al. Genotype shift in human coronavirus OC43 and emergence of a novel genotype by natural recombination. J Infect (2015) 70(6):641–50. doi: 10.1016/j.jinf.2014.12.005
42. Bao L, Deng W, Huang B, Gao H, Liu J, Ren L, et al. The pathogenicity of SARS-CoV-2 in hACE2 transgenic mice. Nature (2020) 583(7818):830–3. doi: 10.1038/s41586-020-2312-y
Keywords: SARS-CoV-2, COVID-19, antiviral, therapy, US Food and Drug Administration-approved compounds
Citation: Xiao X, Wang C, Chang D, Wang Y, Dong X, Jiao T, Zhao Z, Ren L, Dela Cruz CS, Sharma L, Lei X and Wang J (2020) Identification of Potent and Safe Antiviral Therapeutic Candidates Against SARS-CoV-2. Front. Immunol. 11:586572. doi: 10.3389/fimmu.2020.586572
Received: 11 September 2020; Accepted: 13 October 2020;
Published: 25 November 2020.
Edited by:
Pedro A. Reche, Complutense University of Madrid, SpainReviewed by:
Fei Ye, National Institute for Viral Disease Control and Prevention (China CDC), ChinaCopyright © 2020 Xiao, Wang, Chang, Wang, Dong, Jiao, Zhao, Ren, Dela Cruz, Sharma, Lei and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jianwei Wang, d2FuZ2p3MjhAMTYzLmNvbQ==; Xiaobo Lei, ZnlsZWl4YkAxMjYuY29t; Lokesh Sharma, bG9rZXNoa3VtYXIuc2hhcm1hQHlhbGUuZWR1
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.