- 1Department of Nutrition, Gillings School of Global Public Health and School of Medicine, The University of North Carolina at Chapel Hill, Chapel Hill, NC, United States
- 2Division of Pulmonary, Critical Care and Sleep Medicine, The Ohio State University Wexner Medical Center, Davis Heart and Lung Research Institute, Columbus, OH, United States
- 3Department of Microbial Infection and Immunity, The Ohio State University College of Medicine and Wexner Medical Center, Columbus, OH, United States
Obesity is a major independent risk factor for increased morbidity and mortality upon infection with Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV-2), which is responsible for the current coronavirus disease pandemic (COVID-19). Therefore, there is a critical need to identify underlying metabolic factors associated with obesity that could be contributing toward increased susceptibility to SARS-CoV-2 in this vulnerable population. Here, we focus on the critical role of potent endogenous lipid metabolites known as specialized pro-resolving mediators (SPMs) that are synthesized from polyunsaturated fatty acids. SPMs are generated during the transition of inflammation to resolution and have a vital role in directing damaged tissues to homeostasis; furthermore, SPMs display anti-viral activity in the context of influenza infection without being immunosuppressive. We cover evidence from rodent and human studies to show that obesity, and its co-morbidities, induce a signature of SPM deficiency across immunometabolic tissues. We further discuss how the effects of obesity upon SARS-CoV-2 infection are likely exacerbated with environmental exposures that promote chronic pulmonary inflammation and augment SPM deficits. Finally, we highlight potential approaches to overcome the loss of SPMs using dietary and pharmacological interventions. Collectively, this mini-review underscores the need for mechanistic studies on how SPM deficiencies driven by obesity and environmental exposures may exacerbate the response to SARS-CoV-2.
Introduction
Obesity is an independent risk factor for increased morbidity and mortality upon infection with the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) responsible for the current COVID-19 pandemic. Several studies underscore the notion that obesity, in addition to a range of other co-morbidities and dietary factors, may increase the risk for SARS-CoV-2 (1–10). As an example, in a study from Mexico, the odds of having COVID-19 among obese patients with a BMI > 30 kg/m2 was 61% higher than that of control non-obese patients (1). Generally, amongst patients with symptoms, those with severe or critical conditions had much higher BMI and prevalence of obesity than the normal population or COVID-19 negative patients (2–10). One study used the UK Biobank data (n = 285,817) to show that obesity almost doubled the risk of infection, adjusted for age, sex, ethnicity and socioeconomic status (9). Thus, it is clear that obesity results in a higher risk of increased severity of infection with SARS-CoV-2. These findings mirror influenza infection, as obesity also independently increases risk for influenza severity and death (11).
The high rate of obesity worldwide (e.g., in the U.S. over 40% of the adult population is obese) combined with the enhanced morbidity and mortality in obese individuals from infection with SARS-CoV-2 represents a public health emergency. Therefore, there is a critical need to identify the underlying factors by which obese patients are at high risk of infection and complications with SARS-CoV-2. In this mini-review, we focus on a unique aspect of fatty acid metabolism that may provide a link between obesity and immune dysregulation to SARS-CoV-2 infection. These significant insights could evoke new areas of investigation at a mechanistic level and ultimately therapeutic strategies for this vulnerable population.
Metabolites of the Specialized Pro-Resolving Mediator Family Are Critical in the Resolution of Viral Infection Through Multiple Mechanisms
A wide range of metabolic factors contribute toward impaired innate and adaptive immunity in obesity. Here, we discuss the role of fatty acid-derived metabolites belonging to the specialized pro-resolving mediator (SPM) family. These potent lipid autacoids known as resolvins, protectins, maresins, and lipoxins are synthesized during the transition of inflammation to resolution and are critical for turning damaged tissue to homeostasis (12). SPMs are predominately synthesized from the n-3 polyunsaturated fatty acids (PUFA) known as eicosapentaenoic (EPA) and docosahexaenoic (DHA) acids (Figure 1A). Some SPMs are also synthesized from arachidonic acid, an n-6 PUFA (Figure 1B). For further details on these metabolites and their immunoresolvants properties, we refer the reader to elegant reviews from Serhan et al. (12, 13).
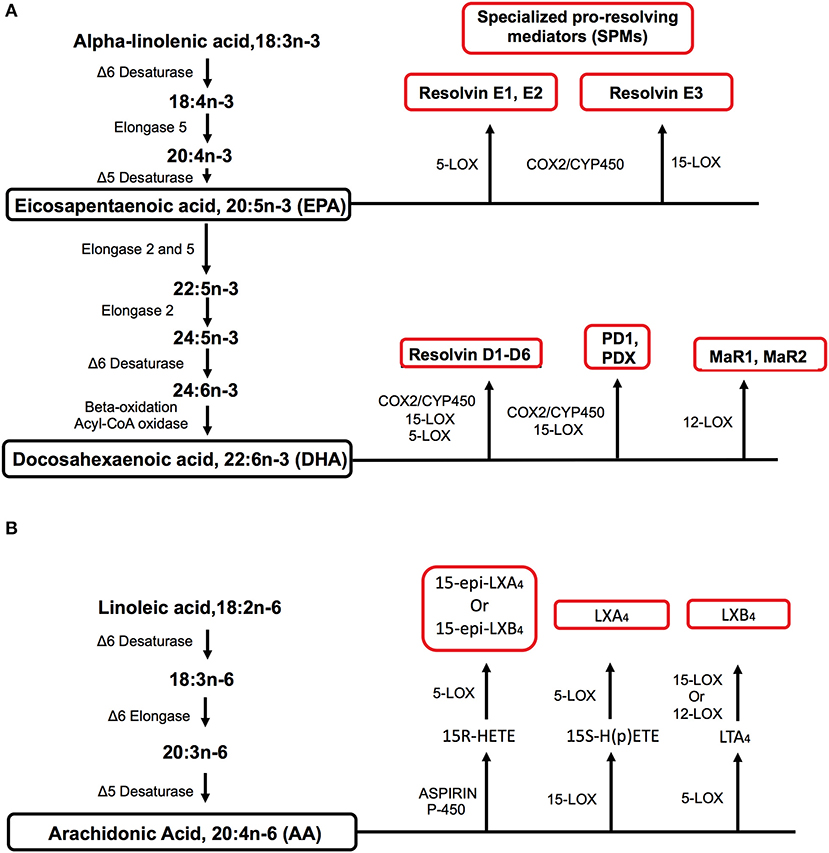
Figure 1. Metabolic pathways by which specialized pro-resolving mediators (SPMs) are synthesized from polyunsaturated fatty acids (PUFA). (A) EPA and DHA are long-chain n-3 PUFAs that serve as precursors for the biosynthesis of SPMs of the resolvin, protectin, and maresin families through the use of differing enzymes. EPA and DHA can be synthesized from the essential short-chain n-3 PUFA known as alpha-linolenic acid. (B) The biosynthesis of lipoxins from the n-6 PUFA arachidonic acid. Arachidonic acid can be synthesized from the essential n-6 PUFA linoleic acid. Key enzymes for fatty acid elongation and desaturation in addition to SPM biosynthesis are indicated for the n-3 and n-6 PUFA pathways. For simplicity, the biosynthesis of all SPM intermediates is not shown for the n-3 and n-6 pathways.
There is strong literature to support a role for SPMs in improving outcomes upon bacterial, parasitic, and viral infections (14, 15). To exemplify, the DHA-derived SPM known as protectin DX (PDX), an isomer of protectin D1 (PD1), enhanced mouse survival upon lethal H5N1 infection including under conditions where antiviral drugs failed to confer protection (16, 17). Mechanistically, PDX inhibited viral replication by targeting the nuclear export machinery for viral transcripts. PDX specifically blocked viral transcripts from being transported to NXF1, an mRNA transporter. Furthermore, pulmonary PDX levels were lowered upon influenza infection and were dependent on 12/15-lipoxygenase activity. These effects were unique to PDX as other PUFA-derived metabolites did not confer any improvement in survival.
Another study suggested that metabolites of the DHA-derived SPM family have utility as adjuvants for influenza vaccination. The SPM precursor 17-hydroxydocosahexaenoic acid (17-HDHA) increased antibody levels and improved survival upon pH1N1 influenza vaccination and infection in lean mice by promoting B cell differentiation toward the formation of CD138+ long-lived antibody secreting cells (18). At a molecular level, this was driven by 17-HDHA upregulating the expression of key transcription factors including Blimp-1, the master regulator of B cell differentiation toward antibody secreting plasma cells. Similarly, administration of dietary DHA ethyl esters, the parent compound of DHA-derived SPMs, also boost antibody levels of obese mice (19, 20). DHA improved antibody levels upon influenza infection by increasing the concentration of 14-hydroxydocosahexaenoic acid (14-HDHA), which in turn drove the formation of long-lived CD138+ antibody secreting cells (19). Therefore, these studies suggest that SPMs have a role in controlling influenza infection through differing mechanisms including improving aspects of humoral immunity. Furthermore, there is also in vitro evidence that the n-6 PUFA-derived SPM known as lipoxin B4 can stimulate antigen-specific IgG production from memory B cells in subjects that were vaccinated for influenza (21). In this case, lipoxin B4 upregulated the expression of Blimp-1 and XBP1 to increase the abundance of memory B cells.
The effects of SPMs are not just limited to influenza virus. For instance, aspirin-triggered resolvin D1 is reported to have anti-inflammatory effects on murine ocular inflammation driven by infection with herpes simplex virus (22). In addition, aspirin triggered resolvin D1 can clear mouse bacterial infections such as pulmonary pneumonia, which can lower the need for antibiotics (23, 24).
The cellular targets of SPMs in the context of viral infection and obesity are emerging. There is strong evidence for the role of SPMs in controlling chronic inflammation in obesity by targeting monocyte and macrophage polarization (25). This is particularly relevant for COVID-19 as adipose tissue presumably expresses high levels of the human angiotensin converting enzyme (ACE2), the receptor for SARS-CoV-2. ACE2 expression levels are likely higher in adipose tissue of the obese compared to the lungs, suggesting that adipose tissue may be a major target for SARS-CoV-2 (26). As described above, there is strong evidence on how SPMs drive B cell differentiation toward long-lived antibody secreting cells. However, it is unclear how SPMs influence other aspects of humoral immunity to promote antibody production. For instance, the abundance of T follicular helper cells, which are required to promote B cell activation and germinal center formation, is lowered in obesity (27). It remains unclear if SPMs could be targeting the abundance of these cells to improve germinal center formation and function. In addition, obesity impairs pulmonary outcomes upon influenza infection, including lung inflammation characterized by dysregulated memory CD8+ T cell metabolism (28). Given evidence to show that SPMs can control T cell differentiation and function, there is a need to understand the mechanisms by which SPMs may control the abundance and function of pulmonary T cell populations (29).
Obesity Promotes a Signature of SPM Deficiency
There is evidence that obesity generally drives a unique signature of SPM deficiency (19, 30–37). Table 1 summarizes the results of these studies. To exemplify, obese mice compared to lean controls display a rapid reduction in DHA-derived SPM precursors and SPMs in white adipose tissue within 4 days of consuming a high fat diet (37). Others have also reported a reduction of not only DHA-derived SPMs but also metabolites from the EPA pathway upon long term consumption of obesogenic diets in white adipose tissue and liver, which are central in driving complications of obesity (30, 32, 34, 42). As described below, these deficiencies can be overcome through dietary administration of EPA- or DHA-enriched marine oils. On the contrary, one study demonstrated that in a model of liver steatosis, select SPMs were elevated, which may be due to an attempt to lower chronic inflammation (38). However, in this study, the liver content of EPA and DHA, the parent fatty acids of SPMs, were lower in obese mice relative to lean controls.
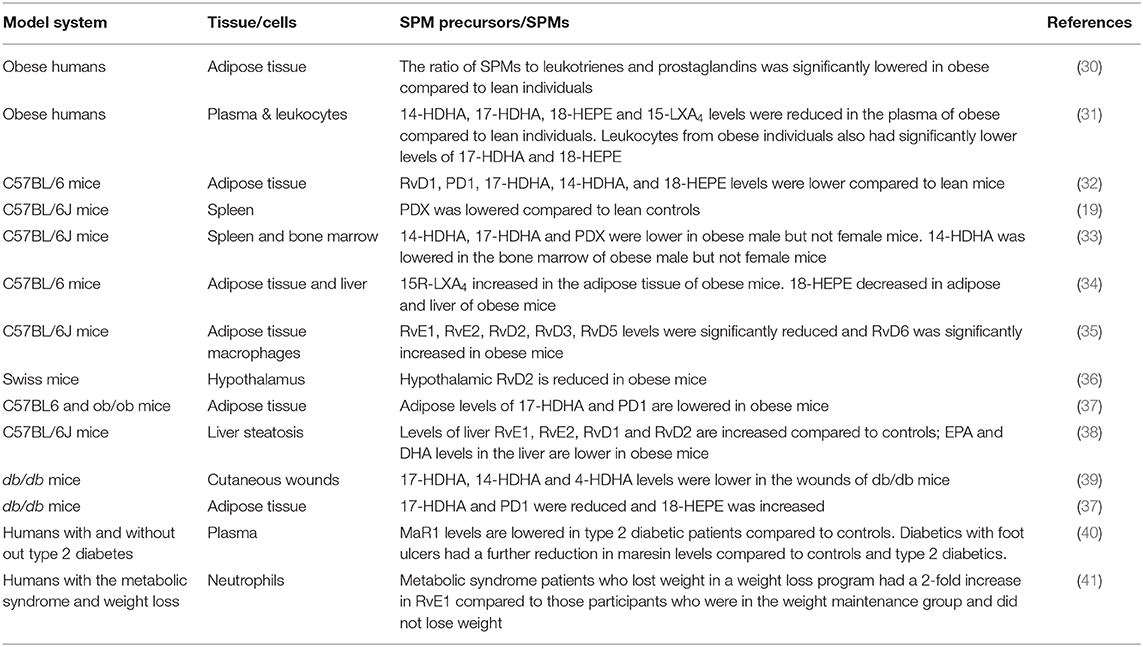
Table 1. Summary of the effects of obesity, diabetes, and weight loss on SPM levels across tissues of humans and mice.
SPM deficiencies are not just limited to adipose tissue and liver. When mice were fed a western diet, there was a significant loss of PDX in the spleen, which was reversed upon administration of DHA ethyl esters in the diet (19). A significant reduction of 14-HDHA, 17-HDHA, and PDX was also reported in mice consuming a high fat diet with a modest effect on 14-HDHA in the bone marrow (33). The effects were evident in male but not female obese C57BL/6J mice, suggesting sex differences in SPM deficiencies. In support of this notion, it is known that synthesis of DHA is higher in women than men (43). The notion of sex-differences in SPM metabolism is also consistent with a human study that showed females were protected from endothelial impairments driven by inflammation due to elevated levels of SPMs compared to males (44). The sex-differences are intriguing, as data on COVID-19 prevalence shows that males are disproportionally at higher risk for becoming infected than females across all ages (45).
Studies with human samples have validated murine studies by demonstrating that obese humans compared to lean controls display deficiencies of key SPM precursors in circulation. A major finding was that leukocytes isolated from obese patients had reduced levels of 17-HDHA and an unbalanced formation of DHA-derived resolvins along with an increased production of the potent chemokine leukotriene B4 (31). This study found impaired activity of 15-lipoxygenase, a key enzyme required for SPM biosynthesis to be the cause of the deficiency. Interestingly, the impairment was not due to reduced cellular uptake of DHA, consistent with rodent studies that show no impairment in DHA levels (33). Furthermore, when leukocytes were treated in vitro with 17-HDHA, the biosynthesis of downstream metabolites was rescued, demonstrating 15-lipoxygenase to be a potential therapeutic target for improving circulating levels of SPMs (31).
The observations on SPM deficiencies with obesity are generally consistent with models of type 2 diabetes, a major co-morbidity of obesity (Table 1). For instance, in wounds of db/db mice, select SPMs were lowered relative to littermate controls (39). In another study, 17-HDHA and PD1 were decreased in white adipose tissue of db/db mice, consistent with studies using diet-induced obese mice, although 18-HEPE levels were elevated compared to controls (37). In type 2 diabetic subjects, circulating maresin 1 (MaR1) levels were decreased compared to controls; furthermore, MaR1 was further decreased in those type 2 diabetics with foot ulcers (40). MaR1 is of significance given its role in regulating murine insulin sensitivity and adipose tissue inflammation in models of genetic and diet-induced obesity (46). Finally, a recent study showed weight loss elevated RvE1 levels in human subjects with metabolic syndrome (41), suggesting that the effects of obesity on SPMs could be potentially reversed through weight loss (Table 1).
Obese Individuals Have Increased Susceptibility to Environmental Exposures That Drive a State of SPM Deficiency
Recent studies have noted that individuals living in areas with higher levels of ambient air pollution are at a higher mortality risk from COVID-19 (47, 48). This was also noted with previous SARS pandemics (49). Obese individuals are uniquely susceptible to environmental exposures and it is currently unknown whether there is a higher rate of mortality from COVID-19 in obese patients that live in areas with increased air pollution. Epidemiological studies have indicated an association between obesity and air pollution (50, 51). Studies of obese humans and animal models have demonstrated a greater decrement in pulmonary function after exposure to the criteria air pollutant ozone (O3), enhanced production of proinflammatory cytokines, and markers of oxidative stress (52, 53). It is currently unclear why obese individuals are more susceptible to the health effects of environmental exposures. However, experimental data have noted that obese mice and humans exposed to air pollutants have increased pulmonary and systemic TNFα, IL-17, markers of lung injury, and airspace neutrophilia (54).
In addition to increased inflammation, acute exposure to O3 significantly reduces pulmonary and systemic DHA-derived SPM precursors and SPMs (55). Treatment of mice with 17-HDHA, 14-HDHA, and PDX significantly decreased O3-induced pulmonary inflammation (55). This suppression of SPM production was also noted in a murine model of nanotoxicity wherein obese mice exposed to nanoparticles had a significant suppression in pulmonary expression of 5-lipoxygenase and 12/15-lipoxygenase and the production of EPA- and DHA- derived SPMs (56). Taken together, these data suggest that the susceptibility of obese individuals to environmental lung diseases may drive an altered pulmonary immune response and a state of SPM deficiency that increases the morbidity and mortality to respiratory infections, including COVID-19.
Discussion
Given that SPM deficiencies in obesity are potentially contributing toward poor outcomes upon SARS-CoV-2 infection, administration of SPMs may be beneficial (57). This hypothesis assumes that SPMs would target key mechanisms by which SARS-CoV-2 drives an uncontrolled and dysregulated pulmonary response. SARS-CoV-2 can drive a cytokine storm, which may be a potential target for intervention as SPMs are known to have dual anti-inflammatory and pro-resolving properties including restricting excessive immune cell infiltration (12, 58). For instance, TNF-α, IL-6, IL-1β, IL-8, IL-12, monocyte chemoattractant protein 1 (MCP1), interferon-gamma inducible protein (IP10) and macrophage inflammatory protein 1A (MIP1A) have been implicated in driving complications associated with SARS-CoV-2 (59). Furthermore, uncontrolled infiltration of immune cells into the lungs, due to excessive reactive oxygen species and secretion of proteases promote pulmonary destruction and thereby lower blood oxygen upon SARS-CoV-2 infection (60). Thus, SPMs or their parent compounds may have utility in improving pulmonary cytokine production and recruitment of pulmonary immune cells upon infection. In support of this notion, in a mouse model of infection with non-typeable haemophilus influenzae, the aspirin triggered RvD1 decreased the concentration of pulmonary TNFα and IL-6 in addition to driving the clearance of macrophages (61).
There are several approaches that could increase levels of SPMs. One is through dietary intervention in which the parent compounds of SPMs, notably EPA and DHA, can be delivered as either over-the-counter supplements or as prescription supplements such as Lovaza, Vascepa, and Epanova. It is important to note that over-the-counter formulations of these fatty acids are not the same as prescriptions due to differences in dose, purity, and composition of the fatty acids. Nevertheless, a recent study showed that an SPM precursor containing marine oil strongly upregulated SPMs of the EPA and DHA series within hours of administration accompanied by enhanced neutrophil and monocyte phagocytosis of bacteria (62). However, a major limitation of this approach is that dietary EPA and DHA may not be as potent as direct intervention with SPMs (12). A more directed approach is to deliver SPMs rather than the parent compounds although the mode of delivery remains to be established. One recent study showed that SPMs were delivered using nanoparticles in a model of intestinal wound healing, which led to activation of pro-repair pathways in the colonic mucosa (63). Furthermore, changes in dietary patterns may be another viable option. The Western diet is associated with impaired pulmonary outcomes and a shift toward a Mediterranean diet may prevent a deficiency of SPMs (64).
An additional consideration is the potential role of n-6 PUFAs on outcomes related to SARS-CoV-2 infection. N-6 PUFAs are highly abundant in the western diet and there is some suggestion that select n-6 PUFAs such as linoleic acid could be driving SPM deficiencies due to competition between the n-6 and n-3 fatty acids for specific enzymes that control SPM biosynthesis (65, 66). This is particularly important to consider given that parenteral nutrition in a hospital setting is enriched in n-6 PUFA-enriched oils (67). Thus, increasing n-3 PUFA levels alone may not be enough to increase downstream SPMs in the obese but could require changes in the intake of n-6 PUFAs. Of course, n-6 PUFAs themselves are also critical for synthesis of SPMs such as lipoxins (12). Thus, additional studies on the complex relationship between dietary n-6 and n-3 PUFAs with downstream SPM biosynthesis, particularly in the context of viral infection are essential. Overall, there is no current evidence to support changes in dietary PUFA intake for improving outcomes upon SARS-CoV-2 infection, but is an important area of investigation at the pre-clinical and clinical level.
Finally, our understanding of the mechanisms by which SARS-CoV-2 exerts its effects are just emerging (60), although how the virus impairs outcomes in obese individuals currently remains unknown. There is no evidence for a role for SPMs in controlling the host's response upon SARS-CoV-2 infection. Therefore, there is a critical need to evaluate and understand the kinetics of SPM biosynthesis in human and animal models of obesity during SARS-CoV-2 infection using mass spectrometry-based lipidomics. Supporting experiments with gain and loss of function approaches in animal models are also required to establish that SPM deficiencies in obesity exacerbate the response to the infection. It is also important to consider the host genetic profile (34), which could be a major consideration in developing dietary or pharmacological approaches to overcoming SPM deficiencies and improving outcomes to SARS-CoV-2 for the obese.
Conclusion
In summary, SPMs are key players in inflammation resolution and the infectious response. Deficiencies in SPMs, driven by obesity, its co-morbidities, and chronic pulmonary environmental exposures, could exacerbate the SARS-CoV-2 induced morbidities and mortalities. Thus, there is an urgency for mechanistic studies on SPMs in the context of obesity and its co-morbidities upon SARS-CoV-2 infection. Ultimately, targeting SPM deficiencies through dietary and pharmacological interventions may be a therapeutic approach worth investigating in order to decrease the morbidity and mortality in response to SARS-CoV-2 infection in a highly vulnerable and metabolically impaired population.
Author Contributions
AP and KG wrote the manuscript. KO, MB, and SS wrote parts of the manuscript. SS assumes responsibility for the work. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by NIH R01AT008375 (SS), NIH R01ES031378 (KG and SS), and NIH R01AI134972 (KO).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Bello-Chavolla OY, Bahena-Lopez JP, Antonio-Villa NE, Vargas-Vázquez A, González-Díaz A, Márquez-Salinas A, et al. Predicting mortality due to SARS-CoV-2: A mechanistic score relating obesity and diabetes to COVID-19 outcomes in Mexico. J Clin Endocrinol Metab. (2020) 105:dgaa346. doi: 10.1210/clinem/dgaa346
2. Chen Q, Zheng Z, Zhang C, Zhang X, Wu H, Wang J, et al. Clinical characteristics of 145 patients with corona virus disease 2019 (COVID-19) in Taizhou, Zhejiang, China. Infection. (2020) 48:543–51. doi: 10.1007/s15010-020-01432-5
3. Bhatraju PK, Ghassemieh BJ, Nichols M, Kim R, Jerome KR, Nalla AK, et al. Covid-19 in critically ill patients in the seattle region - case series. N Engl J Med. (2020) 382:2012–22. doi: 10.1056/NEJMoa2004500
4. Li T, Zhang Y, Gong C, Wang J, Liu B, Shi L, et al. Prevalence of malnutrition and analysis of related factors in elderly patients with COVID-19 in Wuhan, China. Eur J Clin Nutri. (2020) 74:871–5. doi: 10.1038/s41430-020-0642-3
5. Liu M, He P, Liu H, Wang X, Li F, Chen S, et al. Clinical characteristics of 30 medical workers infected with new coronavirus pneumonia. Zhonghua Jie He He Hu Xi Za Zhi. (2020) 43:E016. doi: 10.3760/cma.j.issn.1001-0939.2020.0016
6. Peng YD, Meng K, Guan HQ, Leng L, Zhu RR, Wang BY, et al. [Clinical characteristics and outcomes of 112 cardiovascular disease patients infected by 2019-nCoV]. Zhonghua Xin Xue Guan Bing Za Zhi. (2020) 48:E004. doi: 10.3760/cma.j.cn112148-20200220-00105
7. Simonnet A, Chetboun M, Poissy J, Raverdy V, Noulette J, Duhamel A, et al. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity. (2020) 28:1195–9. doi: 10.1002/oby.22831
8. Wu J, Li W, Shi X, Chen Z, Jiang B, Liu J, et al. Early antiviral treatment contributes to alleviate the severity and improve the prognosis of patients with novel coronavirus disease (COVID-19). J Internal Med. (2020) 288:128–38. doi: 10.1111/joim.13063
9. Ho FK, Celis-Morales CA, Gray SR, Katikireddi SV, Niedzwiedz CL, Hastie C, et al. Modifiable and non-modifiable risk factors for COVID-19: results from UK Biobank. medRxiv [preprint]. (2020). doi: 10.1101/2020.04.28.20083295
10. Liao X, Chen H, Wang B, Li Z, Zhang Z, Li W, et al. Critical care for severe COVID-19: a population-based study from a province with low case-fatality rate in china. medRxiv [preprint]. (2020). doi: 10.1101/2020.03.22.20041277
11. Green WD, Beck MA. Obesity impairs the adaptive immune response to influenza virus. Ann Am Thorac Soc. (2017) 14:S406–9. doi: 10.1513/AnnalsATS.201706-447AW
12. Serhan CN, Levy BD. Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. J Clin Invest. (2018) 128:2657–69. doi: 10.1172/JCI97943
13. Serhan CN, Chiang N, Dalli J. New pro-resolving n-3 mediators bridge resolution of infectious inflammation to tissue regeneration. Mol Aspects Med. (2018) 64:1–17. doi: 10.1016/j.mam.2017.08.002
14. Basil MC, Levy BD. Specialized pro-resolving mediators: endogenous regulators of infection and inflammation. Nat Rev Immunol. (2016) 16:51–67. doi: 10.1038/nri.2015.4
15. Tam Vincent C, Quehenberger O, Oshansky Christine M, Suen R, Armando Aaron M, Treuting Piper M, et al. Lipidomic profiling of influenza infection identifies mediators that induce and resolve inflammation. Cell. (2013) 154:213–27. doi: 10.1016/j.cell.2013.05.052
16. Morita M, Kuba K, Ichikawa A, Nakayama M, Katahira J, Iwamoto R, et al. The lipid mediator protectin D1 inhibits influenza virus replication and improves severe influenza. Cell. (2013) 153:112–25. doi: 10.1016/j.cell.2013.02.027
17. Imai Y. Role of omega-3 PUFA-derived mediators, the protectins, in influenza virus infection. Biochim Biophys Acta. (2015) 1851:496–502. doi: 10.1016/j.bbalip.2015.01.006
18. Ramon S, Baker SF, Sahler JM, Kim N, Feldsott EA, Serhan CN, et al. The specialized proresolving mediator 17-HDHA enhances the antibody-mediated immune response against influenza virus: a new class of adjuvant? J Immunol. (2014) 193:6031–40. doi: 10.4049/jimmunol.1302795
19. Kosaraju R, Guesdon W, Crouch MJ, Teague HL, Sullivan EM, Karlsson EA, et al. B cell activity is impaired in human and mouse obesity and is responsive to an essential fatty acid upon murine influenza infection. J Immunol. (2017) 12:4738–52. doi: 10.4049/jimmunol.1601031
20. Teague H, Fhaner CJ, Harris M, Duriancik DM, Reid GE, Shaikh SR. n-3 PUFAs enhance the frequency of murine B-cell subsets and restore the impairment of antibody production to a T-independent antigen in obesity. J Lipid Res. (2013) 54:3130–8. doi: 10.1194/jlr.M042457
21. Kim N, Lannan KL, Thatcher TH, Pollock SJ, Woeller CF, Phipps RP. Lipoxin B4 enhances human memory b cell antibody production via upregulating cyclooxygenase-2 expression. J Immunol. (2018) 201:3343–51. doi: 10.4049/jimmunol.1700503
22. Rajasagi NK, Bhela S, Varanasi SK, Rouse BT. Frontline science: aspirin-triggered resolvin D1 controls herpes simplex virus-induced corneal immunopathology. J Leukoc Biol. (2017) 102:1159–71. doi: 10.1189/jlb.3HI1216-511RR
23. Chiang N, Fredman G, Backhed F, Oh SF, Vickery T, Schmidt BA, et al. Infection regulates pro-resolving mediators that lower antibiotic requirements. Nature. (2012) 484:524–8. doi: 10.1038/nature11042
24. Abdulnour RE, Sham HP, Douda DN, Colas RA, Dalli J, Bai Y, et al. Aspirin-triggered resolvin D1 is produced during self-resolving gram-negative bacterial pneumonia and regulates host immune responses for the resolution of lung inflammation. Mucosal Immunol. (2016) 9:1278–87. doi: 10.1038/mi.2015.129
25. Hellmann J, Tang Y, Kosuri M, Bhatnagar A, Spite M. Resolvin D1 decreases adipose tissue macrophage accumulation and improves insulin sensitivity in obese-diabetic mice. FASEB J. (2011) 25:2399–407. doi: 10.1096/fj.10-178657
26. Kassir R. Risk of COVID-19 for patients with obesity. Obes Rev. (2020) 21:e13034. doi: 10.1111/obr.13034
27. Farnsworth CW, Schott EM, Benvie A, Kates SL, Schwarz EM, Gill SR, et al. Exacerbated Staphylococcus aureus foot infections in obese/diabetic mice are associated with impaired germinal center reactions, Ig class switching, and humoral immunity. J Immunol. (2018) 201:560–72. doi: 10.4049/jimmunol.1800253
28. Rebeles J, Green WD, Alwarawrah Y, Nichols AG, Eisner W, Danzaki K, et al. Obesity-Induced changes in T-cell metabolism are associated with impaired memory T-cell response to influenza and are not reversed with weight loss. J Infect Dis. (2019) 219:1652–61. doi: 10.1093/infdis/jiy700
29. Chiurchiù V, Leuti A, Dalli J, Jacobsson A, Battistini L, Maccarrone M, et al. Proresolving lipid mediators resolvin D1, resolvin D2, and maresin 1 are critical in modulating T cell responses. Sci Transl Med. (2016) 8:353ra111. doi: 10.1126/scitranslmed.aaf7483
30. Titos E, Rius B, Lopez-Vicario C, Alcaraz-Quiles J, Garcia-Alonso V, Lopategi A, et al. Signaling and immunoresolving actions of resolvin D1 in inflamed human visceral adipose tissue. J Immunol. (2016) 197:3360–70. doi: 10.4049/jimmunol.1502522
31. Lopez-Vicario C, Titos E, Walker ME, Alcaraz-Quiles J, Casulleras M, Duran-Guell M, et al. Leukocytes from obese individuals exhibit an impaired SPM signature. FASEB J. (2019) 2019:fj201802587R. doi: 10.1096/fj.201802587R
32. Clària J, Dalli J, Yacoubian S, Gao F, Serhan CN. Resolvin D1 and resolvin D2 govern Local inflammatory tone in obese fat. J Immunol. (2012) 189:2597–605. doi: 10.4049/jimmunol.1201272
33. Crouch MJ, Kosaraju R, Guesdon W, Armstrong M, Reisdorph N, Jain R, et al. Frontline Science: A reduction in DHA-derived mediators in male obesity contributes toward defects in select B cell subsets and circulating antibody. J Leukoc Biol. (2019) 106:241–57. doi: 10.1002/JLB.3HI1017-405RR
34. Pal A, Al-Shaer AE, Guesdon W, Torres MJ, Armstrong M, Quinn K, et al. Resolvin E1 derived from eicosapentaenoic acid prevents hyperinsulinemia hyperglycemia in a host genetic manner. FASEB J. (in press). doi: 10.1101/848093
35. Bashir S, Sharma Y, Jairajpuri D, Rashid F, Nematullah M, Khan F. Alteration of adipose tissue immune cell milieu towards the suppression of inflammation in high fat diet fed mice by flaxseed oil supplementation. PLoS ONE. (2019) 14:e0223070. doi: 10.1371/journal.pone.0223070
36. Pascoal LB, Bombassaro B, Ramalho AF, Coope A, Moura RF, Correa-da-Silva F, et al. Resolvin RvD2 reduces hypothalamic inflammation and rescues mice from diet-induced obesity. J Neuroinflamm. (2017) 14:5. doi: 10.1186/s12974-016-0777-2
37. Neuhofer A, Zeyda M, Mascher D, Itariu BK, Murano I, Leitner L, et al. Impaired local production of proresolving lipid mediators in obesity and 17-HDHA as a potential treatment for obesity-associated inflammation. Diabetes. (2013) 62:1945–56. doi: 10.2337/db12-0828
38. Echeverria F, Valenzuela R, Espinosa A, Bustamante A, Alvarez D, Gonzalez-Manan D, et al. Reduction of high-fat diet-induced liver proinflammatory state by eicosapentaenoic acid plus hydroxytyrosol supplementation: involvement of resolvins RvE1/2 and RvD1/2. J Nutr Biochem. (2019) 63:35–43. doi: 10.1016/j.jnutbio.2018.09.012
39. Tang Y, Zhang MJ, Hellmann J, Kosuri M, Bhatnagar A, Spite M. Proresolution therapy for the treatment of delayed healing of diabetic wounds. Diabetes. (2013) 62:618–27. doi: 10.2337/db12-0684
40. Miao T, Huang B, He N, Sun L, Du G, Gong X, et al. Decreased plasma maresin 1 concentration is associated with diabetic foot ulcer. Mediators Inflamm. (2020) 2020:4539035. doi: 10.1155/2020/4539035
41. Barden A, Shinde S, Tsai IJ, Croft KD, Beilin LJ, Puddey IB, et al. Effect of weight loss on neutrophil resolvins in the metabolic syndrome. Prost Leukot Essent Fatty Acids. (2019) 148:25–9. doi: 10.1016/j.plefa.2019.07.001
42. González-Périz A, Horrillo R, Ferré N, Gronert K, Dong B, Morán-Salvador E, et al. Obesity-induced insulin resistance and hepatic steatosis are alleviated by ω-3 fatty acids: a role for resolvins and protectins. FASEB J. (2009) 23:1946–57. doi: 10.1096/fj.08-125674
43. Lohner S, Fekete K, Marosvolgyi T, Decsi T. Gender differences in the long-chain polyunsaturated fatty acid status: systematic review of 51 publications. Ann Nutr Metab. (2013) 62:98–112. doi: 10.1159/000345599
44. Rathod KS, Kapil V, Velmurugan S, Khambata RS, Siddique U, Khan S, et al. Accelerated resolution of inflammation underlies sex differences in inflammatory responses in humans. J Clin Investig. (2017) 127:169–82. doi: 10.1172/JCI89429
45. Jin JM, Bai P, He W, Wu F, Liu XF, Han DM, et al. Gender differences in patients with COVID-19: focus on severity and mortality. Front Public Health. (2020) 8:152. doi: 10.3389/fpubh.2020.00152
46. Martinez-Fernandez L, Gonzalez-Muniesa P, Laiglesia LM, Sainz N, Prieto-Hontoria PL, Escote X, et al. Maresin 1 improves insulin sensitivity and attenuates adipose tissue inflammation in ob/ob and diet-induced obese mice. FASEB J. (2017) 31:2135–45. doi: 10.1096/fj.201600859R
47. Conticini E, Frediani B, Caro D. Can atmospheric pollution be considered a co-factor in extremely high level of SARS-CoV-2 lethality in Northern Italy? Environ Pollut. (2020) 261:114465. doi: 10.1016/j.envpol.2020.114465
48. Wu X, Nethery RC, Sabath BM, Braun D, Dominici F. Exposure to air pollution and COVID-19 mortality in the United States: A nationwide cross-sectional study. medRxiv [preprint]. (2020). doi: 10.1101/2020.04.05.20054502
49. Cui Y, Zhang ZF, Froines J, Zhao J, Wang H, Yu SZ, et al. Air pollution and case fatality of SARS in the People's Republic of China: an ecologic study. Environ Health. (2003) 2:15. doi: 10.1186/1476-069X-2-15
50. Seo MY, Kim SH, Park MJ. Air pollution and childhood obesity. Clin Exp Pediatr. (2020). doi: 10.3345/cep.2020.00010. [Epub ahead of print].
51. Alemayehu YA, Asfaw SL, Terfie TA. Exposure to urban particulate matter and its association with human health risks. Environ Sci Pollut Res. (2020) 27:27491–506. doi: 10.1007/s11356-020-09132-1
52. Bennett WD, Hazucha MJ, Folinsbee LJ, Bromberg PA, Kissling GE, London SJ. Acute pulmonary function response to ozone in young adults as a function of body mass index. Inhal Toxicol. (2007) 19:1147–54. doi: 10.1080/08958370701665475
53. Williams AS, Mathews JA, Kasahara DI, Wurmbrand AP, Chen L, Shore SA. Innate and ozone-induced airway hyperresponsiveness in obese mice: role of TNF-alpha. Am J Physiol Lung Cell Mol Physiol. (2015) 308:L1168–77. doi: 10.1152/ajplung.00393.2014
54. Mancuso P. Obesity and lung inflammation. J Appl Physiol. (2010) 108:722–8. doi: 10.1152/japplphysiol.00781.2009
55. Kilburg-Basnyat B, Reece SW, Crouch MJ, Luo B, Boone AD, Yaeger M, et al. Specialized pro-resolving lipid mediators regulate ozone-induced pulmonary and systemic inflammation. Toxicol Sci. (2018) 163:466–77. doi: 10.1093/toxsci/kfy040
56. Alqahtani S, Kobos LM, Xia L, Ferreira C, Franco J, Du X, et al. Exacerbation of nanoparticle-induced acute pulmonary inflammation in a mouse model of metabolic syndrome. Front Immunol. (2020) 11:818. doi: 10.3389/fimmu.2020.00818
57. Panigrahy D, Gilligan MM, Huang S, Gartung A, Cortes-Puch I, Sime PJ, et al. Inflammation resolution: a dual-pronged approach to averting cytokine storms in COVID-19? Cancer Metastasis Rev. (2020) 39:337–40. doi: 10.1007/s10555-020-09889-4
58. Serhan CN. Pro-resolving lipid mediators are leads for resolution physiology. Nature. (2014) 510:92–101. doi: 10.1038/nature13479
59. Costela-Ruiz VJ, Illescas-Montes R, Puerta-Puerta JM, Ruiz C, Melguizo-Rodriguez L. SARS-CoV-2 infection: The role of cytokines in COVID-19 disease. Cytokine Growth Factor Rev. (2020). doi: 10.1016/j.cytogfr.2020.06.001. [Epub ahead of print].
60. Tay MZ, Poh CM, Renia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. (2020) 20:363–74. doi: 10.1038/s41577-020-0311-8
61. Croasdell A, Lacy SH, Thatcher TH, Sime PJ, Phipps RP. Resolvin D1 dampens pulmonary inflammation and promotes clearance of nontypeable haemophilus influenzae. J Immunol. (2016) 196:2742–52. doi: 10.4049/jimmunol.1502331
62. Souza PR, Marques RM, Gomez EA, Colas RA, De Matteis R, Zak A, et al. Enriched marine oil supplements increase peripheral blood specialized pro-resolving mediators concentrations and reprogram host immune responses: a randomized double-blind placebo-controlled study. Circulation Res. (2020) 126:75–90. doi: 10.1161/CIRCRESAHA.119.315506
63. Quiros M, Feier D, Birkl D, Agarwal R, Zhou DW, Garcia AJ, et al. Resolvin E1 is a pro-repair molecule that promotes intestinal epithelial wound healing. Proc Natl Acad Sci USA. (2020) 117:9477–82. doi: 10.1073/pnas.1921335117
64. Suratt BT, Ubags NDJ, Rastogi D, Tantisira KG, Marsland BJ, Petrache I, et al. An official american thoracic society workshop report: obesity and metabolism. An emerging frontier in lung health and disease. Ann Am Thorac Soc. (2017) 14:1050–9. doi: 10.1513/AnnalsATS.201703-263WS
65. Marchix J, Catheline D, Duby C, Monthean-Boulier N, Boissel F, Pedrono F, et al. Interactive effects of maternal and weaning high linoleic acid intake on hepatic lipid metabolism, oxylipins profile and hepatic steatosis in offspring. J Nutr Biochem. (2020) 75:108241. doi: 10.1016/j.jnutbio.2019.108241
66. Jandacek RJ. Linoleic acid: a nutritional quandary. Healthcare. (2017) 5:25. doi: 10.3390/healthcare5020025
Keywords: COVID-19, resolvins, protectins, maresins, lipoxins, antibodies
Citation: Pal A, Gowdy KM, Oestreich KJ, Beck M and Shaikh SR (2020) Obesity-Driven Deficiencies of Specialized Pro-resolving Mediators May Drive Adverse Outcomes During SARS-CoV-2 Infection. Front. Immunol. 11:1997. doi: 10.3389/fimmu.2020.01997
Received: 15 June 2020; Accepted: 23 July 2020;
Published: 11 August 2020.
Edited by:
Philip Calder, University of Southampton, United KingdomReviewed by:
Joan Clària, Hospital Clínic de Barcelona, SpainEsther Titos, Hospital Clínic of Barcelona, Spain
Copyright © 2020 Pal, Gowdy, Oestreich, Beck and Shaikh. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Saame Raza Shaikh, c2hhaWtoc2FAZW1haWwudW5jLmVkdQ==
†These authors have contributed equally to this work
 Anandita Pal
Anandita Pal Kymberly M. Gowdy
Kymberly M. Gowdy Kenneth J. Oestreich
Kenneth J. Oestreich Melinda Beck
Melinda Beck Saame Raza Shaikh
Saame Raza Shaikh