
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Genet. , 23 May 2023
Sec. Livestock Genomics
Volume 14 - 2023 | https://doi.org/10.3389/fgene.2023.1107462
This article is part of the Research Topic Insights in Livestock Genomics: 2022 View all 9 articles
 Juber Herrera-Uribe1†
Juber Herrera-Uribe1† Kyu-Sang Lim1,2†
Kyu-Sang Lim1,2† Kristen A. Byrne3
Kristen A. Byrne3 Lance Daharsh1
Lance Daharsh1 Haibo Liu1
Haibo Liu1 Ryan J. Corbett1
Ryan J. Corbett1 Gianna Marco1
Gianna Marco1 Martine Schroyen1
Martine Schroyen1 James E. Koltes1
James E. Koltes1 Crystal L. Loving3
Crystal L. Loving3 Christopher K. Tuggle1*
Christopher K. Tuggle1*Neutrophils are vital components of the immune system for limiting the invasion and proliferation of pathogens in the body. Surprisingly, the functional annotation of porcine neutrophils is still limited. The transcriptomic and epigenetic assessment of porcine neutrophils from healthy pigs was performed by bulk RNA sequencing and transposase accessible chromatin sequencing (ATAC-seq). First, we sequenced and compared the transcriptome of porcine neutrophils with eight other immune cell transcriptomes to identify a neutrophil-enriched gene list within a detected neutrophil co-expression module. Second, we used ATAC-seq analysis to report for the first time the genome-wide chromatin accessible regions of porcine neutrophils. A combined analysis using both transcriptomic and chromatin accessibility data further defined the neutrophil co-expression network controlled by transcription factors likely important for neutrophil lineage commitment and function. We identified chromatin accessible regions around promoters of neutrophil-specific genes that were predicted to be bound by neutrophil-specific transcription factors. Additionally, published DNA methylation data from porcine immune cells including neutrophils were used to link low DNA methylation patterns to accessible chromatin regions and genes with highly enriched expression in porcine neutrophils. In summary, our data provides the first integrative analysis of the accessible chromatin regions and transcriptional status of porcine neutrophils, contributing to the Functional Annotation of Animal Genomes (FAANG) project, and demonstrates the utility of chromatin accessible regions to identify and enrich our understanding of transcriptional networks in a cell type such as neutrophils.
Neutrophils originate from myeloid progenitors in the bone marrow and go through numerous phases of development, including myeloblast, promyelocyte, myelocyte, metamyelocyte, band cell, and eventually polymorphonuclear neutrophil (Hong, 2017). Neutrophils are one of the most common and important immune cell types in the vertebrate bloodstream (Fingerhut et al., 2020); in human they represent 50%–70% of all leukocytes (Mortaz et al., 2018). Neutrophils are recruited to areas of inflammation from the bloodstream, migrating through chemical signal gradients in a process known as chemotaxis (de Oliveira et al., 2016). Neutrophils are among the most specific immune cell responding to chemotaxic signals, and serve as an overarching model for eukaryotic chemotaxis (Metzemaekers et al., 2020a). Neutrophils respond to chemoattractant signals by secreting granules (degranulation), ingesting microorganisms and other particles (phagocytosis), and forming neutrophil extracellular traps that catch and kill extracellular bacteria once they arrive at the inflammation site (Gierlikowska et al., 2021). Importantly, neutrophils are among the most fully differentiated immune cells (Hong, 2017) and the resting state is highly informative for neutrophil function. Thus, multiple pathways, many unique to the neutrophils, are fully deployed in non-activated neutrophils and the transcription regulation to effect neutrophil activity is poorly understood in the pig.
Genome-wide transcriptomic approaches can identify transcriptional signatures in whole blood (Liu et al., 2017) and specific populations of porcine immune cells (Herrera-Uribe et al., 2021), including neutrophils (Huang et al., 2020). Transcriptomic analyses have also been employed to explore different immune populations in pigs under different conditions such as infections, differentiation, tissue niche, and health status (do Nascimento et al., 2018; Dong et al., 2021; Herrera-Uribe et al., 2020; Summers et al., 2020). Recently, bulk RNA sequencing (bulk RNA-seq) and single-cell RNA sequencing (scRNA-seq) was used to identify genes expressed in different subsets and co-expressed clusters of porcine immune cells from peripheral mononuclear cells (PBMC) (Herrera-Uribe et al., 2021). However, as PBMC preparations exclude neutrophils, there is still limited information cataloging the porcine neutrophil transcriptome. So far, only one RNA-seq-based transcriptomic study (Huang et al., 2020) and two array-based studies (Sanz-Santos et al., 2011; Wang et al., 2017) have been published, but no study exploring the regulatory elements involved in the transcriptional network controlling such RNA expression patterns has been reported.
Genome-wide chromatin accessibility assays can identify genomic regions physically accessible to transcriptional machinery and provide clues to cell-specific gene expression mechanisms that determine the cell identity and function (Yan et al., 2020). For example, recruitment of regulatory proteins to specific transcription factor binding motifs (TFBM) found in such accessible regions provides the opportunity to activate or maintain specific cellular functions (Stein et al., 2010). Therefore, identifying open chromatin regions helps to identify possible regulatory elements and predict cell functions and regulatory pathways in different cell types (Natarajan et al., 2012). Several epigenomic assays can identify regulatory regions genome-wide, such as the DNase I hypersensitivity assay (Pipkin and Lichtenheld, 2006), and more recently the Assay for Transposase Accessible Chromatin (ATAC-seq) (Buenrostro et al., 2013). Combining epigenetic assays, such as ATAC-seq with gene expression assays such as RNA-seq, provides a powerful approach to identify accessible chromatin regions genome-wide, associates the pattern of such regions with RNA expression, and establishes transcriptional regulatory networks to identify specific regulatory functions (Lowe et al., 2019).
A combined genome-wide annotation of chromatin accessibility and transcriptome of porcine neutrophils will contribute to the functional annotation of the porcine genome (part of the FAANG project), enriching our understanding of the molecular regulation of neutrophils, and provide tools for increased utilization of the pig as a model to explore human immunity. In this study, we used ATAC-seq to identify open chromatin regions, and RNA-seq to describe the specific gene expression pattern in porcine neutrophils through comparison to published transcriptomic data of eight other immune cell types that were magnetically and flow cytometrically sorted from peripheral blood mononuclear cells (Myeloid, NK, and several populations of T and B cells) of the same animals. In summary, our analysis revealed co-expressed and specific transcriptional patterns in porcine neutrophils, including transcription factors that were predicted to bind to accessible promoter regions of neutrophil-specific genes (NSGs).
Two crossbred (predominantly Large White and Landrace heritage) male pigs of 6 months of age were used for RNA-seq and ATAC-seq, and four crossbred adults (>6 months of age, unknown sex) were used for Fluidigm Reverse Transcriptase Polymerase Chain Reaction (RT-qPCR) validation. Pigs were housed in BLS2 rooms at the National Animal Disease Center (Ames, IA) and all animal procedures were performed in compliance with and approval by Institutional Animal Care and Use Committee. Pigs were observed daily by animal care staff and were bright, active, and alert during their life. No veterinary treatment was warranted the month prior to sample collection. Upon necropsy, no gross pulmonary lesions suggestive of pneumonia were noted. Up to 120 mL of blood was drawn into BD Vacutainer mononuclear Cell Preparation Tubes—Sodium Citrate (CPTTM, Becton Dickson) or 60-mL syringes containing 6 mL of acid citrate dextrose (ACD). PBMCs were isolated, enumerated, and viability assessed as previously described (Byrne et al., 2021).
Neutrophils were isolated from whole blood in CPTTM tubes as previously described (Heit et al., 2006; Lindblom et al., 2018). Briefly, whole blood was subjected first to dextran sedimentation using 6% Dextran/0.9% NaCl solution at room temperature for 45-60 min. Supernatant containing neutrophils and mononuclear cells was transferred to a conical tube and centrifuged for 12 min at 300 RCF, 4°C with low brake. Neutrophils and mononuclear cells were separated from the erythrocyte-rich pellet by lysing erythrocytes with Ammonium-Chloride-Potassium (ACK) Lysing buffer (ThermoFisher). The pellet was resuspended in phosphate buffered saline (PBS) and the cell suspension was layered over Ficoll-Histopaque-1077 (Catalog No.1077, Sigma) and centrifuged for 30 min at 450 RCF, room temperature, low brake. After centrifugation, PBMCs were removed, and the pellet that contained neutrophils was further processed. Neutrophils were rinsed with 4 mL of Hanks’ Balance Salt Solution (HBSS), centrifuged at 450 RCF for 5 min and resuspended in 2 mL HBSS. Cell count and viability data were obtained using the MUSE cell analyzer system (Millipore). Cells were cryopreserved using 30% dimethyl sulfoxide (DMSO) in RPMI medium and thawed for later use following a previously reported protocol (Milani et al., 2016). A flowchart of the methods is shown in the Supplementary Figure S1.
Isolated neutrophils were enumerated and immediately lysed in RLT-plus buffer for bulk RNA sequencing (bulkRNA-seq). RNA extraction for RLT-plus samples was performed using the AllPrep DNA/RNA MiniKit (QIAGEN) following manufacturer’s instructions. Eluted RNA was treated with RNase-Free DNase (QIAGEN). RNA quantity and integrity were assessed with the Agilent 2200 TapeStation system (Agilent Technologies). RNA samples with RNA integrity numbers (RINs) ≥ 8.2 were used for bulk RNA-seq. Neutrophil RNA extraction for Fluidigm RT-PCR validation were performed using PowerLyzer UltraClean Tissue and Cells RNA Isolation Kit with On-Spin Column DNase I Kit according to manufacturer’s instructions (MoBio, Carlsbad, CA) according to kit instructions.
RNA was fragmented and prepared into libraries using the TruSeq Stranded Total RNA Sample Preparation Kit (Illumina). The two neutrophil libraries were diluted and pooled in approximately equimolar amounts with other eight sorted immune cells previously reported (Herrera-Uribe et al., 2021), Pooled libraries were sequenced in paired-end mode (2 × 150-bp reads) using an Illumina NextSeq 500 sequencer (300 cycle kit).
RNA-seq data processing was performed as reported (Herrera-Uribe et al., 2020). The GTF file for the pig reference genome Sscrofa11.1 (Ensembl, version 97) was used as the genome annotation file to be consistent with the previous report, and because version 97 may be more complete for some important immune genes than later versions (Herrera-Uribe et al., 2021). Mapped read counts per gene from pair-end reads and single-end reads generated from initial trimming of each sample were added together to get the final count table. Given that different types of immune cells have quite different transcriptome profiles (Hicks and Irizarry, 2015), YARN (Paulson et al., 2017), a tissue type-aware RNA-seq data normalization tool, was used to filter and normalize the count table. Genes with extremely low expression levels were filtered out using the filterLowGenes function such that only genes with more than 4 read counts in at least one replicate were kept. The final count table contained 10,974 genes, which was then normalized using the normalizeTissueAware function, which leverages the smooth quantile normalization method (Hicks et al., 2018).
Exploratory analysis of RNA-seq data was performed using the DESeq2 package (version 1.24.0) (Love et al., 2014) within the RStudio software (v1.2.1335). First, regularized log-transformation was applied to the normalized count table with the rld function. Then principal component analysis (PCA) and sample similarity analysis was carried out and results were visualized using the plotPCA and distancePlot functions. Heatmaps to display enriched genes were created using the pheatmap package (v1.0.12).
We identified genes with cell type-enriched expression using the method previously reported (Herrera-Uribe et al., 2021). Briefly, a gene was labeled as cell-type enriched if the expression level (averaged across cell types) of a given gene in one cell type was at least two-times higher than the averaged expression level of the given gene across all remaining cell types and the adjusted p-value of this contrast was less than 0.05 (Benjamini and Hochberg, 1995). Heatmaps to display enriched genes were created as mentioned above.
For cross species comparison, human and mouse neutrophil (Haemopedia) RNA-seq expression data (Hilton Laboratory at the Walter and Eliza Hall Institute) was used (Choi et al., 2019). Neutrophils for these data sets were isolated using a similar method as described above: depleting red blood cells from the erythrocyte-rich pellet by lysing erythrocytes, plus negative selection of eosinophils and further cell purity confirmation by flow cytometry. Only orthologous genes with one-to-one matches between human and pig (orthologs gene list obtained from Ensembl BioMart (Durinck et al., 2009) were compared. Spearman’s rank correlation analysis was performed to identify correlation between expression levels (transcript per million, TPM) of orthologs gene in pig and human neutrophils. The significance level was set at p < 0.05 and the level of Spearman’s rank correlation coefficient (rho, ρ) was defined as low (ρ < 0.3), moderate (0.3 ≤ ρ < 0.5), and strong (ρ ≥ 0.5) correlation.
Gene Ontology (GO) analysis of the top 25% enriched genes was performed using Metascape (Zhou et al., 2019). The threshold p-value was set to 0.01. Several terms were clustered into the most enriched GO term. Term pairs with Kappa similarity score above 0.3 were displayed as a network to show relationship among enriched terms. Protein-protein interaction enrichment analysis of the same gene list was performed using STRING (Szklarczyk et al., 2019), BioGrid (Stark et al., 2006), OmniPath (Türei et al., 2016) and InWeb_IM (Li et al., 2017) databases.
The Molecular Complex Detection (MCODE) algorithm was applied to identify densely connected protein network components (Bader and Hogue, 2003). The three best scoring terms by p-value were retained as the functional description of the corresponding components. Only orthologous genes were compared. All Ensembl gene IDs with detectable expression level in neutrophils as defined above were used as the background reference. All networks displayed were visualized using Cytoscape.
Briefly, RT-qPCR analysis was run on the Fluidigm BioMark HD System, using 48 × 48 Fluidigm Dynamic Arrays (Fluidigm, South San Francisco, CA). Primer efficiency was tested simultaneously. Four porcine neutrophils and PBMC RNA samples were included in the assay. Also, serial dilutions of a neutrophil-PBMC pooled sample were included in the Fluidigm plate for primer efficiency calculation. The set of genes assayed were part of a previously developed assay dataset (Schroyen, Marco and Tuggle, unpublished results). Within this independent dataset, we selected all neutrophil significantly enriched genes (SEG, defined below) present (a total of 14) and also selected five genes that were not present in the SEG list for further analysis. The cDNA was pre-amplified for 14 cycles (10 min at 95°C followed by 14 cycles of 95°C for 15 s and 4 min at 60°C) in reactions primed by a master mix of 48 TaqMan Gene Expression Assays (Applied Biosystem) using PreAmp Master Mix (Applied Biosystem). Following a 5-fold dilution of pre-amplified product in water, samples and Assays were loaded onto the plates using the 48.48 MX IFC Controller (Fluidigm). Real-time PCR was performed using the BioMark System for Genetic Analysis (Fluidigm). Cycle threshold (Ct) values were determined using real-time PCR analysis v3.1.3 software (Fluidigm). Ct values corresponding to transcripts encoding ACTB and RPL32 were used as endogenous controls. Changes in transcript expression were calculated using the ΔΔCt method (Livak and Schmittgen, 2001) and converted to log2 scale. Graphs and Student’s t-test statistical analyses were generated using Prism 6 (GraphPad). p-values <0.05 were considered statistically significant. Additionally, a Pearson correlation analysis was performed to determine concordance between RNA-seq data and Fluidigm qPCR gene expression values. The significance level for Pearson correlation analysis was set at p < 0.05.
The normalized gene expression levels (transcripts per million; TPM) (Patro et al., 2017) of Significant Enriched Genes (SEGs) (n = 832) across 9 cell types were used in the co-expression network analysis using the partial correlation coefficient with information theory (PCIT) algorithm (Reverter and Chan, 2008; Koesterke et al., 2013). The gene co-expression networks of 681 genes (nodes) with the absolute value of the correlation coefficient greater than 0.95 were visualized using Cytoscape v3.8.2 software (Shannon et al., 2003) (Figure 2A). The log2 transformed (TPM+1) values were used to create heatmaps using the pheatmap package in R to show expression patterns across cell types (Figure 2B). To identify candidate neutrophils specific regulators, transcription factors (n = 30) in cluster 1 which showed myeloid specific expression were used in another heatmap (Figure 3). The cell expression data from human and mouse (Choi et al., 2019) were plotted into Figure 3 to compare cell type specific expression patterns across species. The gene expression values (TPM) were normalized relative to the gene expression level in neutrophils for comparison purposes across cell types.
After thawing, neutrophils were immediately lysed, nuclei were counted and transposed with Tn5 transposase following the standard protocol (Buenrostro et al., 2015). In brief, 50,000 nuclei were incubated for 30 min at 37°C in transposase reaction buffer (25 µL TD-2x reaction buffer, 2.5 µL TDR1-Nextera Tn5 Transposase and 22.5 µL Nuclease Free H2O). Transposed DNA was isolated using MiniElute PCR Purification Kit (Qiagen) and eluted with 10 µL of Elution buffer (Qiagen). DNA size selection for 100 to 800-bp fragments from the purified libraries was performed using BluePippin cassettes (Sage Science). Libraries were pooled and sequenced on an Illumina HiSeq 3000 sequencer to generate 50-bp paired-end reads at the Iowa State University DNA facility.
ATAC-seq data processing was performed as previously reported using the nf-core pipeline nf-core/atacseq (Ewels et al., 2020). Read quality was checked using MultiQC version 0.11.7 (Ewels et al., 2016). Illumina sequencing adapters and low-quality bases were trimmed using Trim Galore (version 0.6.5). Paired-end, trimmed reads were separately mapped to the Sus scrofa 11.1 pig reference genome using the BWA mem aligner (Li and Durbin, 2010). Sequence duplicates from the paired-end and singleton reads were marked in each individual BAM file, and then BAM files were merged for each sample. Library insert size and duplication rate were checked using picard tools1. Duplicate and mitochondrial reads were removed from the aligned reads. The BAM files were used for peak calling with the MACS2 tool, and either broad or narrow peaks were detected with a q-value cutoff of 0.052. HOMER was used to annotate peaks relative to known genomic features3. The fraction of reads in peaks (FRiP) was calculated using the combined alignments from both replicates. The Integrative Genomics Viewer (IGV) was used to performed visual inspection of the data and visualize the open chromatin regions using bigWig files. Visualization of genome-wide chromatin open regions enrichment at the TSS and across the gene body were visualized using deepTools (Ramírez et al., 2014). Overlapping peaks from all samples were merged to form a consensus peak list.
The read counts of narrow peaks from the ATAC-seq data were standardized as reads per kilobase. The read counts per kilobase were normalized by the trimmed mean of M values (TMM) and then by count per million values, computed for each peak using the EdgeR package in R. An intensity of cut site at open chromatin region (OCR) in the promoter of each gene was assessed by the sum of the normalized count values of the peaks overlapping with a given gene’s promoter region which were defined as the transcription start site (TSS) ± 3 kb. The length of OCRs in the promoter region (TSS-OCRs) for each gene was also calculated. To evaluate the effects of the cut site intensity and the length of accessible regions within promoters on gene expression in neutrophils, Pearson correlations were computed between ATAC-seq peaks either for all expressed genes or for neutrophils enriched genes. The log2 transformed values of gene expression and TSS-OCRs cut intensity and length were used in the correlation analysis.
To find transcription factors exhibiting neutrophil specific expression, transcription factor binding motifs (TFBM) were identified by enrichment analysis of the neutrophil enriched genes TSS-OCR using the HOMER command findMotifsGenome.pl4. The binding motifs for three (KLF5, GFI1B, and GATA1) of the four neutrophil-specific transcription factors identified were annotated in the HOMER database, and these were further investigated to find neutrophil specific genes that had the corresponding motifs in their promoter regions. In addition, protein-protein interactions among neutrophil specific genes were investigated in the STRING database (Szklarczyk et al., 2019). We constructed the network focusing on the four transcription factors identified above with their target genes based on binding motifs and protein interactions.
We compared neutrophil gene expression and chromatin accessibility data with previously published DNA methylation data from the same cell populations (Corbett et al., 2022). Average DNA methylation profiles for neutrophil-enriched genes and neutrophil ATAC peaks were generated using deepTools ComputeMatrix command (Ramírez et al., 2016) with default parameters and including 2-kb regions flanking these genes and peaks. Enrichment of neutrophil SEGs and HEGs within neutrophil lowly methylated genes in different genomic feature contexts was calculated using hypergeometric tests. Regions of overlap between neutrophil lowly methylated regions (LMRs) and ATAC peaks were identified using bedtools intersect (Quinlan and Hall, 2010), and overlap enrichment was calculated using hypergeometric tests.
The genome-wide transcriptome profile of porcine neutrophils was determined using RNA-seq, and the data was compared with eight different immune cell transcriptomic profiles to identify genes enriched for expression within the neutrophil cell type. RNA-seq libraries contained an average of 40 M clean reads, of which 94.4% aligned to the Sus scrofa reference genome after filtering (Supplementary File S1). Relationships among the nine porcine immune cell transcriptomes were assessed by principal component analysis (PCA) and hierarchical clustering (Supplementary Figure S2). While erythrocyte contamination of neutrophils is highly likely in the dataset given the neutrophil isolation method used, we only found low expression of two red blood gene markers (HBB: 37 TPM and HEMGN: 1 TPM) identified previously by single cell analysis of human red cells (Jain et al., 2022). Read counts for other red blood cell markers such as HBA, HBD, HBG, NIX, ACVR2B, AHSP and HEMGN were zero in the porcine neutrophil transcriptome for these samples (Supplementary File S1). These results indicated a low level of erythrocyte contamination in the neutrophil samples, and neither HBB nor HEMGN were found in further differential gene expression analysis. In the PCA analysis, neutrophils appeared closest to monocytes, and both were clearly distinguished from other cell types.
The total number of expressed genes detected in neutrophils was 10,974, which were used for differential gene expression (DGE) analysis across cell types. Significantly enriched genes (SEGs) were identified in neutrophils as genes whose expression was both significantly different (adjusted p-value<0.05) and a minimum of two-fold higher in expression than the average of all other cell populations (see Methods). In total, we identified 832 SEGs (Supplementary File S1). A subset of the SEGs were selected as highly enriched genes (HEG, 25% highest in log2 FC value) and neutrophil HEG expression across all cell types was visualized (Figure 1A). Although the high expression of these 208 HEG in neutrophils is evident in comparison with the other previously reported cell types, we also observed gene expression similarities with the myeloid (monocytes and dendritic cells) population for many HEG genes (Figure 1A; Supplementary Figure S2). GO analyses using neutrophil HEG indicated enrichment for biological processes such as myeloid leukocyte activation, cytokine-cytokine receptor interaction, response to bacterium and phagocytosis among others, which is characteristic of the neutrophils, and depicted as networks of similar terms (Figure 1B and Supplementary File S2). Protein-protein interaction (PPI) analysis shows proteins in networks that are believed to interact with each other to promote specific biological processes on the basis of experimental and computational data (see Methods). The PPI analysis of HEGs detected 16 subgroups of protein interactions (Figure 1C and Supplementary File S2) such as regulating exocytosis, neutrophil degranulation, type I interferon and NF-kappa B signaling pathways, among others. Overall, evaluation of predicted functional annotations within the neutrophil HEGs provided evidence of genes enriched in functions very consistent with neutrophils, indicating that our isolated cell population and the RNA-seq data obtained represent the expected immune cell type.
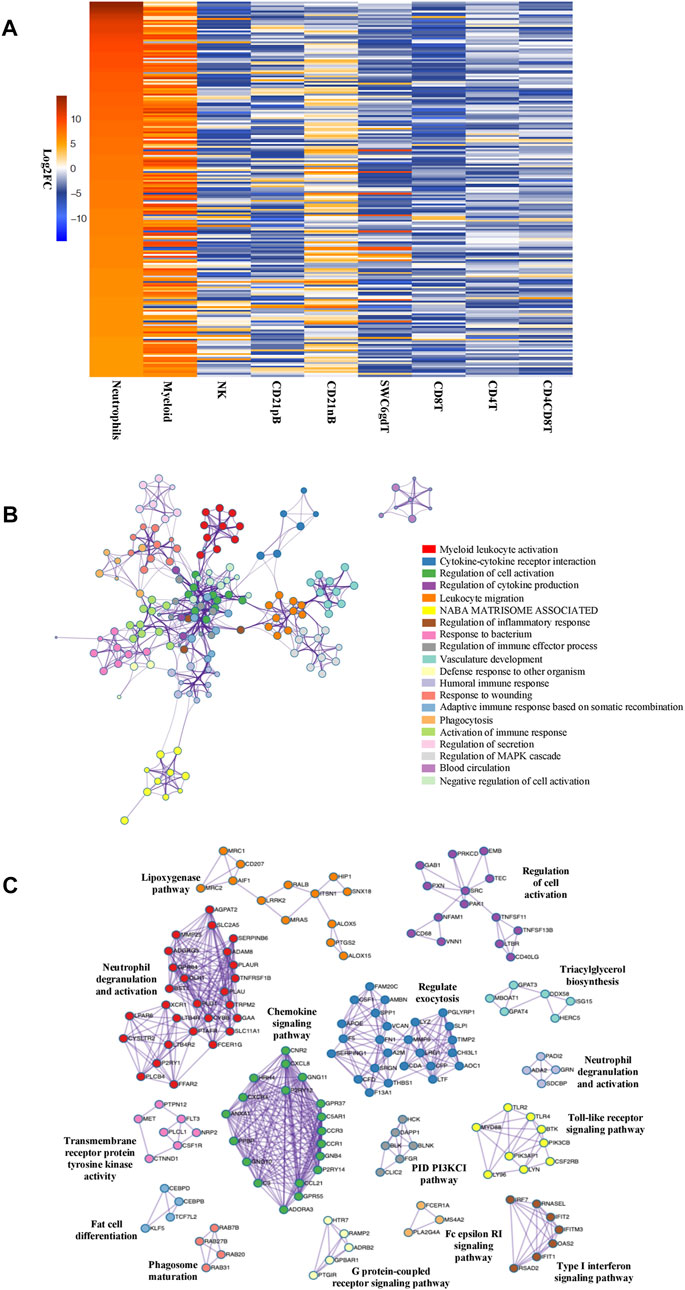
FIGURE 1. Genes enriched in porcine neutrophil transcriptome are linked to specific GO pathways and PPI networks. (A) Heatmap showing the top 25% of HEGs in Neutrophils and eight sorted cells, in decreasing order of fold change difference in expression. (B) Ontology enrichment clusters of SEGs. The most statistically significant term within cluster was chosen to represent the cluster. Term color is given by cluster ID and the size of the node is proportional to–log10 p-value of enrichment. The stronger the similarity among terms, the thicker the edges between them. (C) PPI networks of SEGs. A unique color was assigned to each MCODE network.
Four independent porcine neutrophil and PBMC samples were obtained to validate the RNA-seq expression results. RNA isolation and qPCR analysis of 14 genes was performed using available data from a previously validated assay (Schroyen, Marco and Tuggle, unpublished results). The estimate of fold change ratio calculated by analyzing the genes expression of 19 genes between neutrophil and PBMC samples using qPCR was compared to the fold change ratio of SEGs in neutrophil (see methods) using RNA-seq. In total, fourteen genes within the SEG list (ALOX5AP, BCL6, CHI3L1, CDF2RB, GRN, IL13RA1, ILR2, IL18RAP, PLXNC1, TLR8, TSPAN2, SERPING1, SOD2 and VNN2) and five genes that were not SEGs in neutrophils (CTSS, GMPR, PIM1, NCF2 and UBN1) had gene expression levels that were very consistent with the DGE analysis and confirm the higher expression of SEGs in neutrophils compared to PBMC (Supplementary Figure S3; Supplementary Figure S4). These 19 genes exhibited strong and highly significant positive correlation (rho = 0.71, p-value 0.00074) between the RNA-seq and Fluidigm analyses (Supplementary Figure S5). In conclusion, gene expression enrichment found using RNA-seq was confirmed using the Fluidigm qPCR assay in an independent neutrophil dataset.
RNA expression patterns reflect a significant correlation structure in the transcriptome, and co-expressed genes are frequently similar in biological function (van Dam et al., 2018). Hence, we hypothesized that defining co-expressed genes, and identifying those in the co-expressed network that are enriched in neutrophils, would help us to identify specific genes and transcription factors that would be driving the gene expression important for specific porcine neutrophil functions. To address this hypothesis, we defined co-expressed genes across the nine major immune cell type transcriptomes available. Co-expression networks across these cell types contained 681 genes (r ≥ 0.95) and were constructed based on the PCIT algorithm (Reverter and Chan, 2008; Koesterke et al., 2013). A co-expression network for all SEGs was constructed from this analysis (Figure 2A). Three easily distinguishable co-expression subnetwork clusters were detected, of which cluster 1 (n = 598) showed uniquely higher expressions in myeloid cell types such as neutrophils and monocytes compared to very low expression in lymphoid cell types (Figure 2B and Supplementary File S3). A subset of neutrophil co-expressed genes was also present in the other two clusters that also show expression in other cell types. Genes from cluster 2 genes are expressed in SWC6 (Swine workshop cluster 6) γδ T and CD21 nB B cells, while cluster 3 genes were also expressed in NK cells and B cells (CD21 pB and CD21 nB) (Figures 2A,B, Supplementary File S3).
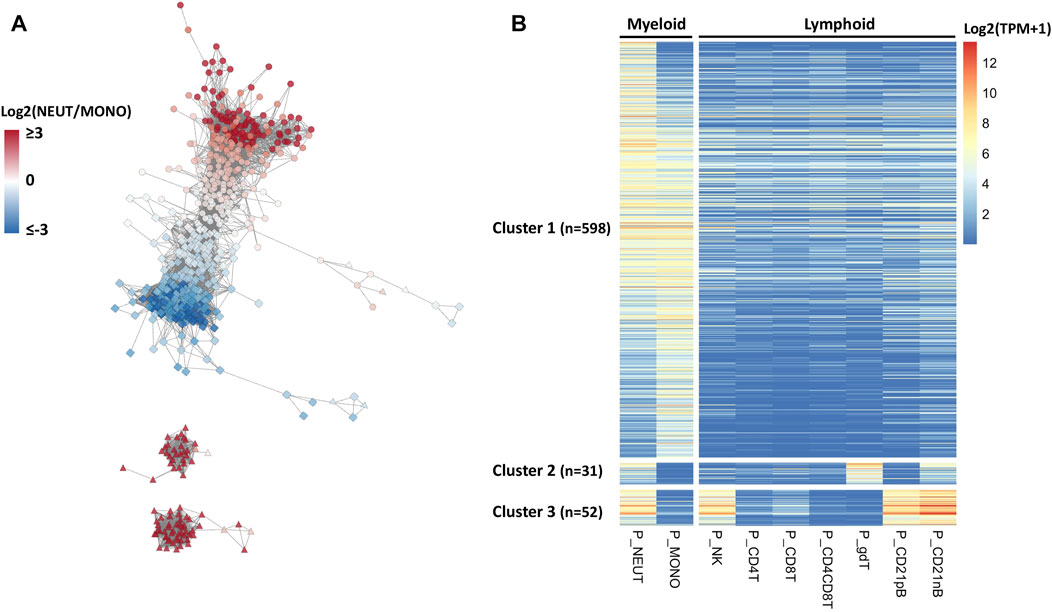
FIGURE 2. Co-expression network analysis identifying myeloid gene clusters across porcine immune cells. (A) Co-expression network analysis (edges between gene nodes shown for correlation r > 0.95) among genes whose expressions were enriched for neutrophils across nine porcine immune cell types and their expression patterns. In the network, the gene nodes (n = 681), and node colors indicate log2 fold change (log2FC) between myeloid cells, neutrophils and monotypes (e.g., positive value indicates higher expression in neutrophils). The node shapes represent the cell type with the highest expression for that gene; circle, square, and triangle indicate neutrophils, monotypes, and other cell types, respectively. (B) In the heatmap, gene rows were clustered based on the correlation levels and sorted by log2FC in neutrophils.
To achieve a more refined understanding of the neutrophil biology underlying the co-expression network, we used a more stringent criterion to determine neutrophil-specific genes (NSGs) for further analysis. NSGs were defined as SEGs that were a) co-expressed (r ≥ 0.95), b) had highest expression in neutrophils, and c) were statistically significantly (q < 0.05) two-fold higher in expression than in monocytes/dendritic cells. A total of 80 NSGs were identified based on these criteria (Supplementary File S3), of which 30 have been annotated as a transcription factor (Figure 3). Four of these NSGs were designated as neutrophil specific transcription factors (NSTF) in pig, and include GATA1, MXD1, KLF5, and GFI1B (Figure 3). Additionally, the pig neutrophil data allowed us to also identify porcine myeloid specific transcription factors (MSTF) (TF with highest expression in neutrophils or monocytes/dendritic cells and were statistically significantly (q < 0.05) two-fold higher in expression than the other seven porcine cell types, Figure 3 and Supplementary File S3). Thus, porcine MSTF were also identified (DACH1, ZNF648, CEBPB, CEBPA, CEBPD, LTF, LITAF, VDR, NFE2, BATF2, IRF7, TCF7L2, SPI1 and SMAD9) (Figure 3). Interestingly, HHEX was identified as part of the myeloid co-expression network in Figure 2A but was also highly expressed in B cells; similar TF expression was observed for FOSL2 and BCL6 that were part of the myeloid co-expression network and were highly expressed in NK cells.
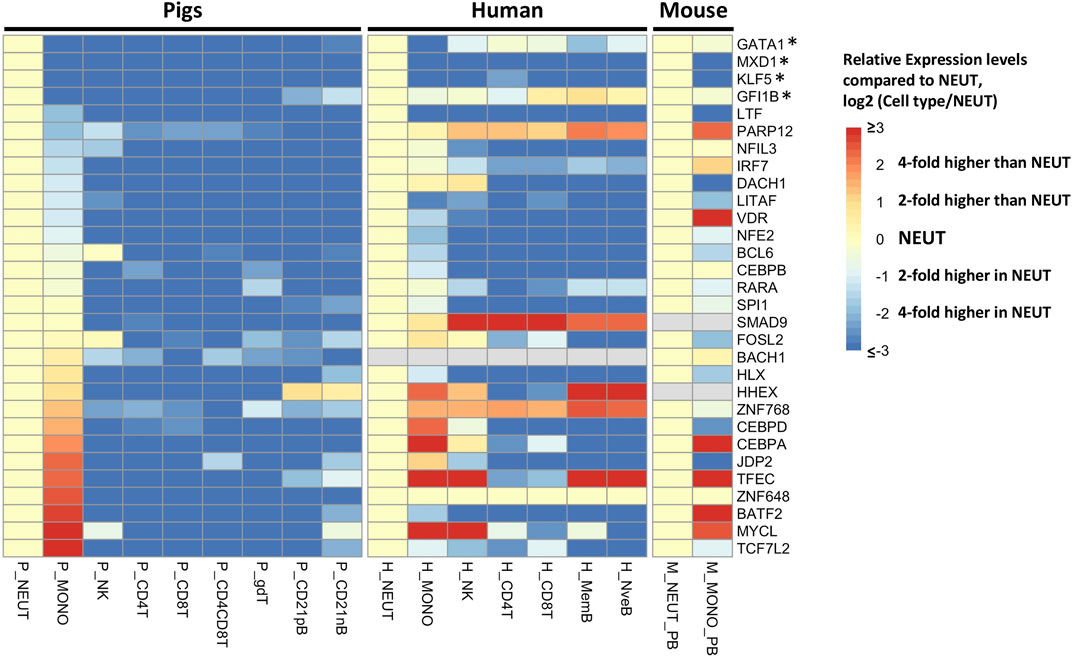
FIGURE 3. Gene expression pattern of selected transcription factors (n = 30) in cluster 1 of the neutrophil network (r > 0.95). The human and mouse datasets (Choi et al., 2019) were compared to the pig data, and expression levels across cell types were standardized by the expression levels in neutrophils of each gene to visualize the relative gene expression level compared to that in neutrophils. The asterisk indicates the transcription factors with neutrophil-specific expression in pigs.
Finally, we compared the expression of all TFs on the NSGs list with orthologous human and mouse genes from Haemopedia (Choi et al., 2019), finding two NSTF that have higher expression in neutrophil versus monocytes across species, MXD1 and KLF5 (Figure 3). Also, LTF, LITAF and NFE2 showed higher expression in neutrophils for all three species, but not as high as MXD1 and KLF5 in porcine cells. In addition, two MSTF have higher expression in pig and human (CEBPD) and in all three species (CEBPA).
To define the accessible chromatin in neutrophils from healthy, non-stimulated pigs, we performed ATAC-seq from duplicate samples to those used for neutrophil RNA-seq analysis. We generated approximately 227 million ATAC-seq reads in total, of which 99% were aligned to the Sus scrofa reference genome (Supplementary File S4). ATAC-seq libraries contain short fragments (<100 bp), which represent nucleosome-free regions, and larger fragments that span one or more nucleosomes. Clear nucleosome distribution was observed (Figure 4A). Such fragments are mapped to the genome and merged into peaks when sufficient signal is present (Methods). On average, we identified a total of ∼134,000 accessible chromatin elements, of which 57,567 were designed as consensus broad peaks in both library samples meeting the ENCODE standards for ATAC-seq5. The ENCODE quality control measure, FRiP score, was over 0.4 in our dataset, which is higher than the minimum of 0.2 used for ENCODE ATAC-seq data (Ou et al., 2018). Consensus narrow peaks averaged around 1 Kb in width, and ATAC-seq signal from both replicates were highly significant and strongly correlated (r = 0.93, p < 2.2e-16) (Supplementary Figure S6). To confirm and annotate the distribution pattern of the ATAC-seq peaks, we analyzed the ATAC-seq reads distribution across transcriptional start and end sites (TSS, TES) (±3.0 Kb) in Sus scrofa reference genome and assigned peaks to genomic features (Figures 4B,C). Although most peaks were located at the intronic or intergenic space (Figure 4C), a clear enrichment of peaks was seen very close to the TSS in the gene-proximal space (Figure 4B), indicating we were able to detect accessible chromatin at active genes. Furthermore, we observed accessible chromatin around TSS regions of enriched genes that are typically involved in pathogen recognition (TLR4), DNA-binding transcription activator activity (CEBPB, KLF5), and interferon signaling pathway (ISG15) (Figure 4D), as well as a lack of accessible chromatin at genes with low or no expression in neutrophils (such as ZP3 and FABP6) (Supplementary Figure S7). These results demonstrate the high sensitivity of ATAC-seq to detect regions associated with active gene promoters.
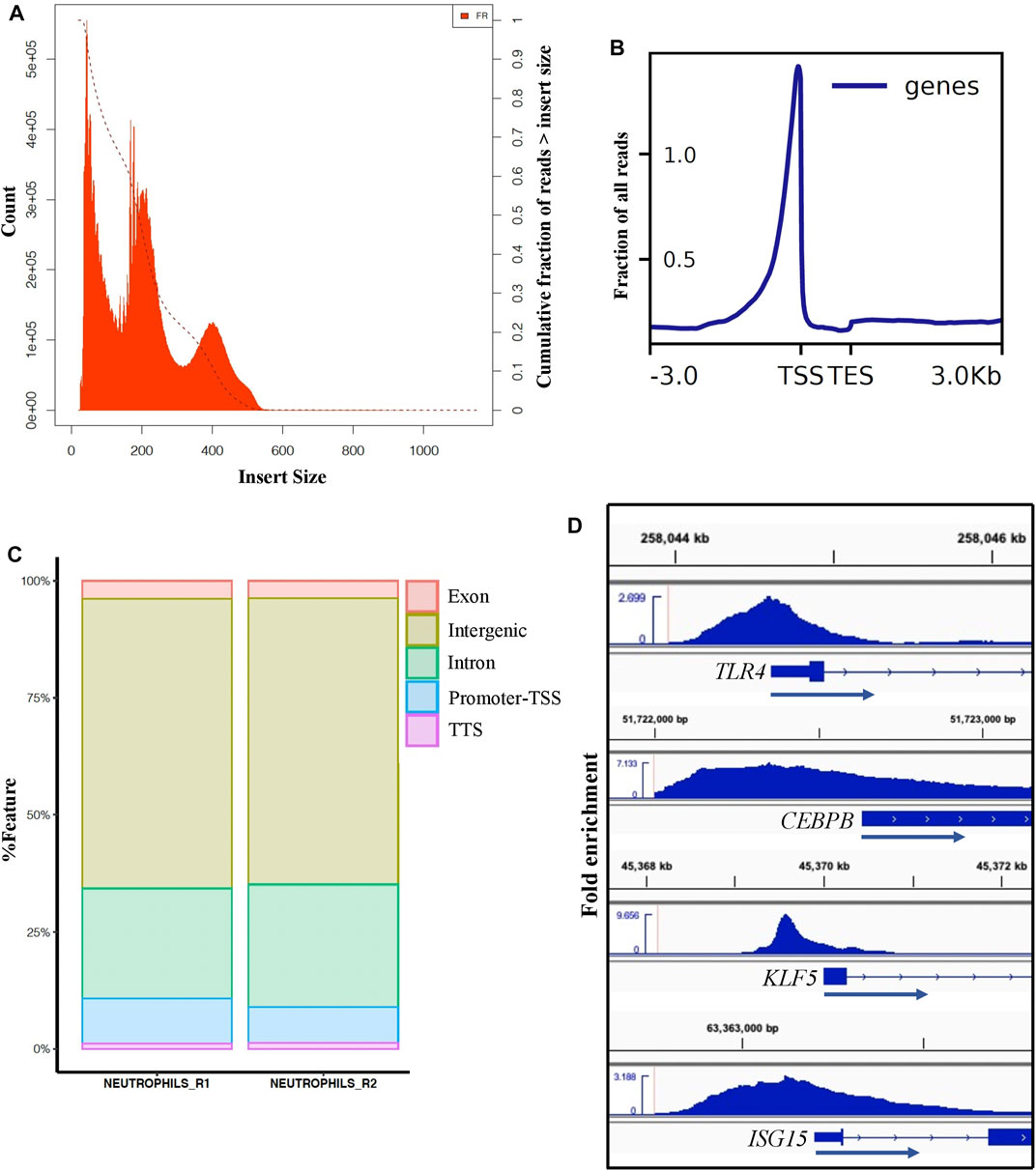
FIGURE 4. ATAC-seq analysis of porcine neutrophils. (A) Distribution of ATAC-seq data fragment length from one neutrophil replicate. Fragment size distribution plot shows enrichment less than 100 bp and around 100-200 bp, indicating nucleosome-free region and mono-nucleosome-bound fragments, respectively. Similar distribution was observed for the other replicate (data not shown). (B) TSS enrichment plot from one neutrophil replicate shows that nucleosome-free fragments are enriched at TSS. (C) Peak annotation plot shows ATAC-seq peaks distribution genome-wide. (D) Visualization of ATAC-seq peaks using the Integrative Genomics Viewer (IGV) near example SEGs in porcine neutrophils (TLR4 FC: 7.2, CEBPB FC: 3, KLF5 FC: 5.9 and ISGS15 FC: 4.4).
To expand comparison of ATAC-seq signals and gene expression genome-wide, Pearson correlation analysis was performed to test the relationship between gene expression and ATAC-seq signal intensity and length around TSS (±3 kb). Highly significant but relatively low correlation (r = 0.23, p-value <2.2e-16) was found between the ATAC-signal cut density and all genes used in comparative analysis in this study across cell types (n = 11,151) (Figure 5A). Interestingly, similarly significant and moderate correlation (r = 0.30, p-value <2.2e-16) was observed for enriched genes in neutrophils, (Figure 5B). For both analyses, some expressed genes did not show accessibility within promoter regions, over a range of expression levels. Therefore, we calculated the correlation between enriched genes and accessible regions that had at least one TSS-OCR. As a result, we found the highest correlation with similar significance (r = 0.41, p-value <2.2e-16) (Figure 5C). Finally, we also identified a weak correlation between gene expression and the length of TSS-OCR in the same comparisons that were performed for ATAC-seq signal intensity (Supplementary Figure S8). Overall, ATAC-seq analysis demonstrated high quality and enrichment for ATAC-seq regions around regulatory regions such as TSS genome-wide.
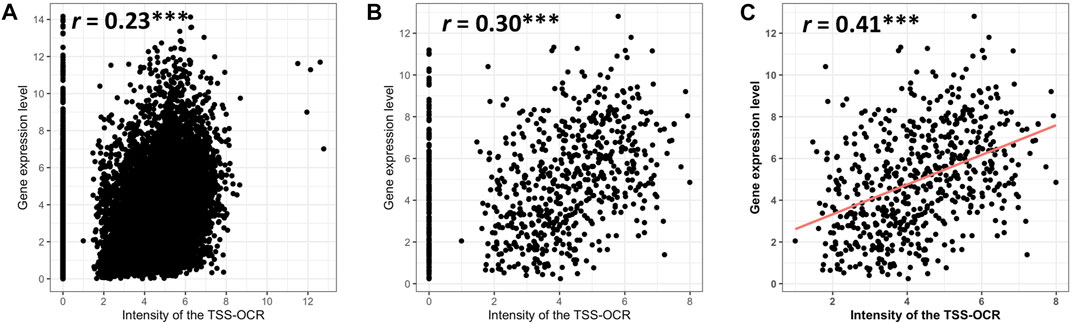
FIGURE 5. Relationship of gene expression with the intensity of ATAC-seq peak overlapping the gene’s promoter region (TSS-OCR). (A) All genes. (B) SEGs. (C) NSGs that have at least one TSS-OCR (open chromatin regions around ±3 kb from the TSS).
We used published DNA methylation data generated from samples collected from the same pigs in parallel (Corbett et al., 2022) to determine whether neutrophil DNA methylation was related to gene expression and accessible chromatin regions. Also, we tested the significant enrichment between differentially methylated regions (DMR) (calculated across the same immune cell populations used in this study) and SEGs and HEGs. As is shown in Figure 6A, neutrophils SEGs exhibit low DNA methylation patterns across TSS regions, which indicates activation of gene expression in both neutrophil replicates. Lowly methylated regions (LMRs) were highly enriched among neutrophil SEGs (p = 2.4 × 10-3). In total, 119 neutrophil SEGs showed LMRs in comparison with other porcine immune cell methylomes reported previously (Supplementary File S6) (Corbett et al., 2022). LMRs across SEGs gene features showed that promoters and transcription termination sires (TTS) have higher enrichment with the expression of SEGs (Figure 6B). However, significant high enrichment of LMRs in intragenic regions of SEGs was also evident, indicating that low methylation was associated with neutrophils SEGs outside promoter regions Figure 6B. The strongest LMR enrichment was observed around promoter of HEGs (p = 7.19E-4), which are the top 25% of SEGs, demonstrating a stronger association between DNA methylation and gene expression. Additionally, we tested the relationship of genome-wide neutrophil DNA methylation distribution and chromatin accessibility (ATAC-seq signal) across expressed genes in neutrophils. In general, we observed a depletion of DNA methylation in all sequence contexts near open chromatin peaks of expressed genes in neutrophils (Figure 6C). Indeed, we detected 7.4-fold enrichment of LMRs in ATAC-seq peaks (p < 1 × 10-16), which demonstrate a high association between DNA methylation and chromatin accessibility in porcine neutrophils. There was a trend towards higher ATAC-seq signal among peaks overlapping neutrophil LMRs compared to those not overlapping LMRs (Figure 6D), indicating that, on average, chromatin accessibility increases when coupled with DNA hypomethylation. Collectively, these results link DNA methylation with control of chromatin accessibility and gene expression patterns in porcine neutrophils.
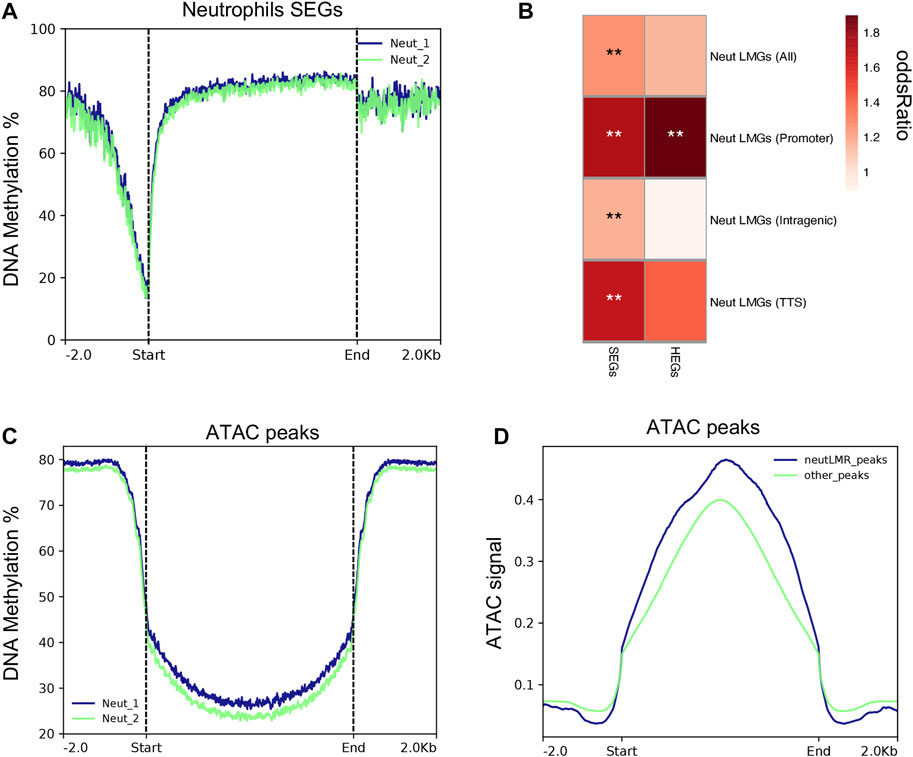
FIGURE 6. Porcine neutrophil methylation data is associated with neutrophil gene expression and ATAC-seq peaks. (A) Methylation rates genome wide across the transcription start site of neutrophils SEGs for each replicate sample. (B) Heatmap of normalized enrichment p-values of neutrophils lowly methylated regions (LMRs)m SEGs and HEGs overlapping different genomic features. (C) Methylation rates genome wide gene across ATAC-seq peaks that were detected in porcine neutrophils. (D) Fold enrichment of LMRs genome wide across ATAC-seq peaks.
Accessible chromatin is expected to contain regulatory elements bound by transcription factors controlling expression of nearby and remote genes. To identify the potential regulatory role of NSTFs in NSG promoter regions, we determined the enrichment of transcription factor binding motifs (TFBMs) within narrow ATAC-seq peaks around promoter regions (±3000 bp from TSSs) of NSGs. Within 8,415 peaks around the promoter regions of 7,868 genes expressed in neutrophils, 65 peaks overlapped promoter regions of 58 of the total 80 NSGs (Supplementary Figure S9). TFBM analysis focused on known binding motif on those 65 TSS-OCR revealed that NSTFs such as GATA1, GFI1B, and KLF5 were predicted to bind motifs enriched in the TSS-OCR. A total of 52 NSGs were predicted to have motifs within their open chromatin regions corresponding to these NSTF (Figure 7). Most NSGs had predicted enrichment for KLF5 motifs (46), followed by GATA1 (13) and GFI1B (10), with a limited number of genes with more than one TF motif predicted (Figure 7A). A network of the NSG predicted to be regulated by these NSTF was then constructed (Figure 7B). Interesting, 17 NSGs were predicted to be regulated by more than one NSTF, including CD101, CERS4 and SPHK1, CAMK1, IFIT1, MARCKS, TMEM92 and several unannotated genes. The NSTF gene MXD1 was predicted to be regulated by both GATA1 and GFI1B. Interestingly, several genes in this doubly-regulated group have been annotated to interact with other members of this group, including ACSL1 (interaction partner: ACSL4), CERS4 (interaction partner SPHK1), and BST1 (two interaction partners: ADAM18 and C4BPA) (Figure 7B). The latter interaction network is interesting as three other predicted NSTF target genes interact with BST1, indicating that BST1 may be an important protein in neutrophil function. An additional interaction network was seen for IFIT1 which interacts with predicted targets ISG15 and HERC5, as well as non-enriched gene IFIT2. Finally, several other genes are in the diagram that were not shown to have enrichment of nearby NSTF motifs (such as MMP9, PDLIM1, TIAM2 and ARHGAP15), but nevertheless are predicted to interact with other genes predicted to be transcriptionally regulated by NSTF (Figure 7B).
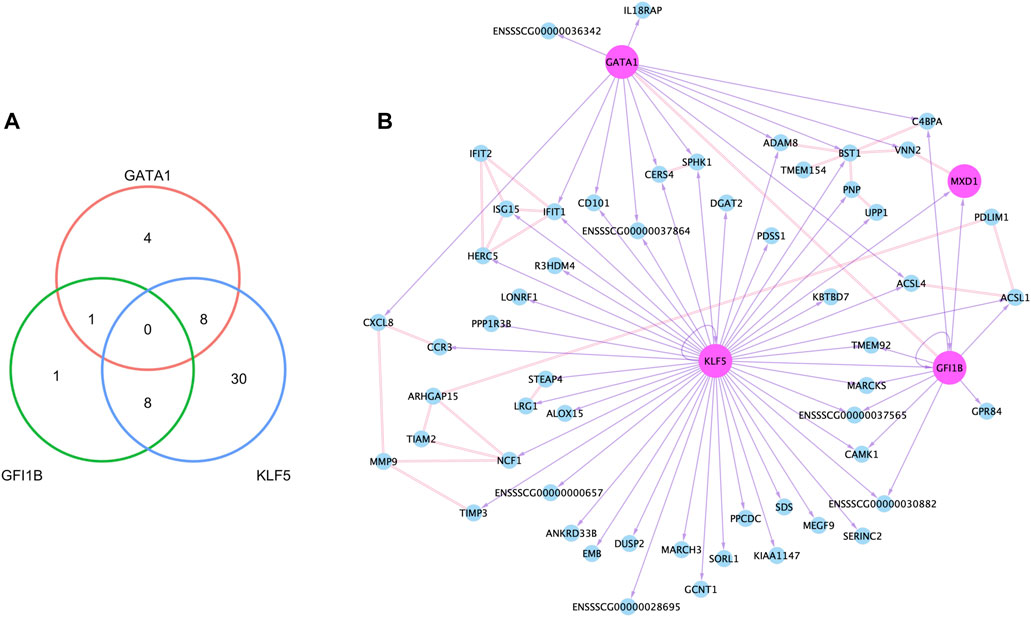
FIGURE 7. Neutrophil co-expression network created with epigenetic and transcriptomic data. (A) The number of genes that had binding motifs for the three neutrophil-specific transcription factors (TFs) within TSS-OCRs, and the network based on binding motifs and protein-protein interactions. In the network (B), the node (circle) indicates each gene, and the pink color indicates TFs. The double line between nodes represents the interaction in the protein level, and the purple arrow with a solid line point to a gene that had a binding motif of the source node.
In this study, we report the first comprehensive integrative analysis of chromatin accessibility and transcriptome of neutrophils isolated from non-stimulated pigs to expand the epigenetic and transcriptomic annotations of this important porcine innate immune cell. We then combined RNA-seq and ATAC-seq data to link neutrophil transcriptomic with genome-wide accessible chromatin data to identify potential regulatory relationships controlling neutrophil-specific gene expression. In consequence, we identified porcine neutrophil networks based on chromatin accessibility combined with TF and potential target genes enriched in neutrophil expression that may regulate this specific porcine neutrophil transcriptional program.
The RNA analysis of porcine neutrophils demonstrated these cells have a very different transcriptional pattern compared to previously reported porcine mononuclear cell transcriptomes (Herrera-Uribe et al., 2021) including three types of T cells (CD4−CD8+, CD4+CD8a−, CD8+CD4+, γδT-cells), NK and two types of B cells. However, the RNA expression patterns were more similar to the monocytes/DC population reported in that study. Further, the GO terms associated with genes whose expression was highly enriched in neutrophils were characteristic of known neutrophil function and pathways.
Based on the hypothesis that correlated expressed genes are more likely to have a similar biological role than genes with diverse expression pattern (Mola et al., 2020), we identified co-expressed gene clusters across the nine types of porcine immune cells mentioned above. Initially, we identified a myeloid cluster (for neutrophils and monocytes/DC), and by increasing the stringency, we identified a list of NSGs co-expressed specifically in neutrophils (SEG and co-expressed genes). The NSGs and myeloid dataset lists published here might be utilized in comparative analyses of neutrophils across species, to predict neutrophil content in whole blood via machine learning methods (Maslova et al., 2020).
Within the co-expressed neutrophil specific genes, it was interesting to identify a number of genes encoding TF. In fact, multiple TF with known roles within the innate immune system were found, especially TFs with myeloid and neutrophil functions given that both cell types are differentiated from the same bone marrow-residing precursor cell (Mehta et al., 2014). The well-known examples are essential TFs for myeloid and neutrophil differentiation such as SPI1 (which encodes PU.1), CEPA/B, GATA1, KLF5 and GFI1B (Bjerregaard et al., 2003; Duan and Horwitz, 2003; Shahrin et al., 2016; Denholtz et al., 2020; Khoyratty et al., 2021). Red and white blood cell differentiation is driven by GATA1 and PU.1, which act by mutual repression in the myeloid lineage (Nerlov et al., 2000). Recently, Fischer and others demonstrated that PU.1 drives an inhibitor epigenetic program in neutrophils which prevent the induction of an excessive innate immune response (Fischer et al., 2019).
GFI1B, a porcine NSTF found in our study has been shown to be an essential TF for neutrophil differentiation in mice; it is also necessary for the formation of both erythroid and megakaryocytic lineages in knockout mice (Saleque et al., 2002; Vassen et al., 2007). In human and mice, GFI1B mutations are linked to severe neutropenia and platelet shape, number, and function (Stevenson et al., 2013; Anguita et al., 2017; Geissler et al., 2018). The molecular function of GFI1B is to repress the expression of target genes (Vassen et al., 2005). GFI1B binds to the DNA, recruiting histone deacetylases and demethylases, which decrease gene expression (van der Meer et al., 2010). Another fundamental TF for neutrophil differentiation and commitment is KLF5, which was also found as a NSTF in our study. In vivo studies have revealed that loss of KLF5 expression was associated with attenuated neutrophil differentiation (Shahrin et al., 2016). Additionally, reduction of KLF5 expression was also reported in acute myeloid leukemia cancer (Humbert et al., 2011), where the neutrophils counts are also reduced (Zhang et al., 2021). Our approach further identified TFs highly expressed in porcine neutrophils and in other immune cell types. For example, HHEX and FOSL2, which in concordance with previous studies are highly expressed in neutrophils, B cells (Laidlaw et al., 2020) and NK cells (Grassi et al., 2018; Li et al., 2020), respectively. Additionally, we were able to find MSTFs such as SPI1, CEBPB and CEBPA, which also have been reported as highly expressed in human and mice myeloid lineages (Rosenbauer and Tenen, 2007; Mildner et al., 2017; Braun et al., 2019). Overall, these study results suggest a group of NSTFs in pig that have functional roles in the granulocyte lineage in other species such as mouse and human and therefore confirm the value of the porcine NSTFs list identified in this study.
We observed global chromatin accessibility regions that are comparable to those reported in mice (Fischer et al., 2019). Although ATAC-seq studies have been performed on human neutrophils, the depth of this analysis compared to our study was unclear as the number of total peaks in non-stimulated human neutrophils were not reported (Mistry et al., 2019; Ram-Mohan et al., 2021). In this study, we have detected slightly more accessible regions compared to those detected in mouse neutrophils. Khoyratty, et al., 2021 reported 47,164 ATAC-seq peaks across different murine neutrophil developmental stages (Khoyratty et al., 2021; Fischer et al., 2019) found around 20,000 ATAC-seq peaks (Fischer et al., 2019), in non-stimulated mouse neutrophils. Chromatin spatial and peak genomic distribution was similar to those reported in human neutrophils under non-stimulated conditions (Chen et al., 2016; Ram-Mohan et al., 2021). Interestingly, neutrophils plasticity has been attributed to changes in the chromatin structure, and the openness and closeness of the TFBM are responsible for the specific neutrophil response against a specific stimulus (Chen et al., 2016; Ram-Mohan et al., 2021). Ram-Mohan et al., 2021 identified specific chromatin changes in human neutrophils against different toll-like receptor (TLR) activators and E. coli, suggesting that highly differentiated neutrophils could modify their chromatin to affect transcriptional changes (reviewed in Rosales, 2018). Taken together, we provided for the first time the chromatin accessibility patterns in porcine neutrophils under non-stimulated conditions. Identifying the accessible chromatin regions and chromatin states with different pathogens or different TLR mimics in these cells would further evaluate neutrophil plasticity at the epigenomic and transcriptomic level in the pig.
By the integration of transcriptomic and chromatin accessibility data from biological replicate samples, we developed more detailed information on the transcriptional mechanics controlling gene expression in porcine neutrophils. First, we investigated the correlation between the promoter accessibility of a gene and its respective expression. We identified a significant positive correlation, which showed consistent correlation for all expressed genes and SEG. However, there were genes that showed an opposite pattern of promoter accessibility (no peak detected around promoter region) and expression (expressed in porcine neutrophils). Different studies have shown that low (or high) gene expression is not always associated with a lack of chromatin accessibility (Hughes et al., 2017; Su et al., 2017; Rajbhandari et al., 2018). As well, accessible chromatin regions around gene promoters are not always associated with expressed genes or regions marked with active histone modifications (Ackermann et al., 2016; Daugherty et al., 2017). Additionally, incomplete promoter annotation, especially in farm animals (Kern et al., 2021), could affect the correlation between accessible chromatin regions and gene expression.
We used recently published DNA methylation data from the same porcine neutrophil sample (and other eight porcine immune cells, see methods) (Corbett et al., 2022) to interrogate the relationship between DNA methylation levels, gene expression and chromatin accessibility. The integration of these data revealed that promoter regions with accessible chromatin exhibited low methylation, and highly neutrophil-enriched genes had a strong correlation with low methylated DNA in different genomic features, particularly with promoter regions. This negative association between DNA methylation, gene expression and chromatin accessibility have been observed in other immune cells and tissues (Pan et al., 2021; Roy et al., 2021; Corbett et al., 2022; Liu et al., 2022). Interestingly, only 119 of 832 SEG in neutrophils had LMRs around promoters in comparison with the eight porcine immune cells mentioned above. This suggest that highly expressed genes in neutrophils are likely controlled by a mechanism at least partially independent of DNA methylation, as it has been shown in other studies that DNA methylation and chromatin accessibility do not always follow an inverse correlation with gene expression (Lim et al., 2017; Spainhour et al., 2019). This result suggests that methylation and chromatin accessibility are two of multiple levels of transcriptional regulation (Thurman et al., 2012) for the porcine neutrophil transcriptome. Overall, while these data suggest that gene expression can be explained by chromatin accessibility around promoter regions, a further integrated analysis including more epigenetic data such as histone modifications and chromatin interaction data could complement current understanding regarding gene regulation in the pig. Such future work could also pay special attention to non-coding regions, which also shape gene expression patterns through enhancers and non-coding RNA activity (Chen et al., 2017; Panigrahi and O'Malley, 2021) as demonstrated in human neutrophils (Ecker et al., 2017).
As a main objective of this study was to identify the specific transcriptional network in porcine neutrophils according to gene expression and chromatin accessibility patterns, we also performed a TFBM enrichment analysis (using NSTF) on chromatin accessible regions around promoters of NSGs to predict networks of TF and their putative neutrophil specific target genes. Indeed, this approach identified several known lineage-specific TFs such as GATA1, KLF5, MXD1 and GFI1B that are likely involved in maintaining neutrophils commitment and essential functions associated with those commitments. All these NSTF have been found expressed in other human mature neutrophil datasets (Monaco et al., 2019) and in early committed mice neutrophil progenitors (Kwok et al., 2020).
The combined analysis revealed that GATA1 was predicted to bind the promoter region of the principal neutrophil-attracting chemokine and major mediator of the inflammation, CXCL8 (Metzemaekers et al., 2020b). As well as KLF5 was predicted to interact with promoters of ISG15 (IFN-stimulated gene 15) and IFIT1 (IFN-induce protein with tetratricopeptide repeats 1) (also predicted target of GATA1) and CCR3 that are involved in the antiviral responses of neutrophils as a frontline of the innate immune system (Tamassia et al., 2008; Rudd et al., 2019). The putative target genes IFIT1 and ISG15 are also induced in response to bacteria in neutrophils (Xie et al., 2020). In general, our integrative approach identified TFs and promoter accessible regions of target genes within NSTF. However, the predictions that these TFs regulate the genes with motif-enriched promoters enriched would need to be validated with genetic/biochemical experiments to provide more insight into the regulatory mechanisms of gene regulation in porcine neutrophils.
Interestingly, we identified a TF that has not been widely studied in neutrophils, MAX dimerization protein 1 gene (MXD1), as a NSTF potentially involved in neutrophil regulatory networks. Interestingly, MXD1 was predicted to be controlled by other two NSTFs, KLF5 and GFI1B. Lummertz da Rocha, E et al., 2018 identified increase of the MXD1 gene expression in the later stages of mature neutrophil differentiation from mouse bone marrow scRNA-seq data (Lummertz da Rocha et al., 2018). Further, MXD1 expression was significantly associated with the low proportion of neutrophils in tumor immune microenvironment data which included information from 22 cell types (Du et al., 2021). In the Lummertz da Rocha et al., 2018 study, a gene regulatory network analysis linked MMP8, MMP9, RETNLG and CD52 genes as predicted targets of MXD1. In our experimental approach using ATAC-seq data, we were able to identify MMP9 (which mediates neutrophil migration in infection (Bradley et al., 2012)) as a SEG that interact with other NSGs (CXCL8, NCF1 and TIMP3) in porcine neutrophils, but we did not detect a predicted binding motif for NSTFs in the MMP9 TSS-OCR.
Some of the genes displayed in the specific transcriptional network in porcine neutrophils interacted with other NSGs and interact with other predicted NSTF target genes. All these multiple interactions in the network suggest a collaborative and important function of these genes in neutrophils. For example, BST1 (CD157) protein is highly expressed on the surface of most human circulating neutrophils and plays a fundamental role in neutrophil adhesion and migration (Funaro et al., 2004; Ortolan et al., 2006). Additionally, we identified a group of genes that interact with other predicted transcriptional regulated genes, although they were not predicted to be transcriptionally regulated by any of NSTF. Two of these genes could illustrate the non-activated state of the neutrophils we used in our study, for example, PDLM1 and ARHGAP15. The PDLIM1 protein, a member of the LIM domain-binding protein family, negatively regulates NF-κB-activation in the cytoplasm by sequestration of p65 (subunit of NF-κB), suppressing NF-κB translocation to the nuclei (Ono et al., 2015). ARHGAP15, a GTPase of the Rac family is also a negative regulator of neutrophil function (Costa et al., 2011). Costa et al. (2011) observed in mice lacking ARHGAP15 an increase of neutrophil recruitment to the site of infection, reducing systemic inflammation and improving mice survival. They also found enhancement of chemotactic activity, straighter directional migration, amplified reactive oxygen species production, increase of phagocytosis and bacterial killing in vitro in cells lacking ARHGAP15. Together, our study identified a gene network in porcine neutrophils that contains several well-known essential genes for neutrophil functions as well as potential novel gene candidates that could expand the understanding of the neutrophil, especially in pig.
The epigenetic (histone ChIP, ATAC-seq and DNA methylation analyses) and transcriptional data (RNA-seq) provided by the FAANG project is contributing to the identification of regulatory regions in the porcine genome (Giuffra et al., 2019; Herrera-Uribe et al., 2021; Kern et al., 2021; Pan et al., 2021; Corbett et al., 2022). Such data is very powerful to detect regulatory regions, but it is time-intensive and comes with significant cost. Our strategy using only RNA-seq and ATAC-seq may be more generally applied to identify many of these elements and so improve the pig genome annotation of other porcine circulating immune cells requiring many biological states. These states would include resting and different immune state conditions such as response to bacterial and viral infections or immune-related diseases. We also demonstrated how these data can be utilized to inform researchers to link TFs, open chromatin, and gene expression to predict the interactions of these regulatory elements to form transcriptional networks. Thus, the integrative analysis presented in this manuscript will be a useful tool to extend the epigenetic and transcriptomic annotations to the other porcine immune cell types.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://www.ebi.ac.uk/ena, PRJEB57595; https://www.ebi.ac.uk/ena, PRJEB51699.
The animal study was reviewed and approved by USDA—National Animal Disease Center (Ames, IA).
CL, CT, and JK conceptualized and supervised research. JH-U, KB, and CL collected samples and isolated cells. JH-U performed bulk RNA isolations and performed ATAC-seq library preparation. MS and GV performed Fluidigm analyses. JH-U, LD, and HL performed bulk RNA-seq analyses. K-SL performed co-expression, cross-species gene expression, and transcription factor analyses. LD performed ATAC-seq analyses. RC performed methylation analyses. JH-U, K-SL, JK, CL, and CT interpreted the data and drafted the manuscript. All authors contributed to the writing of the materials and methods, edited the manuscript, and approved the final version. All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
This work was supported by the National Institute of Food and Agriculture (NIFA) Project 2018-67015-270, and by USDA-ARS CRIS project #5030-3200-225-00D.
We acknowledge the DNA facility of the Iowa State University for provision of technical support and sequencing platforms utilized in this study. We are grateful to the NADC animal care staff for their efforts. We thank Catherine Ernst at Michigan State University for providing the Yorkshire pigs used in the bulk RNA-seq and ATAC-seq study. We also thank Zahra Bond for technical assistance with neutrophil isolation.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgene.2023.1107462/full#supplementary-material
ACD, Acid Citrate Dextrose; ACK, Ammonium-Chloride-Potassium; ATAC-seq, Assay for Transposase Accessible Chromatin; DC, Dendritic Cells; DGE, Differential Gene Expression; DMSO, Dimethyl sulfoxide; FAANG, Functional Annotation of Animal Genomes; FRiP, Fraction of reads in called peak regions; GO, Gene oOntology; HBSS, Hanks’ Balance Salt Solution; HEGs, Highly Enriched Genes; IGV, Integrative Genomics Viewer; LMR, Low Methylated Region; MCODE, Molecular Complex Detection; MSTF, Myeloid Specific Transcription Factor; NSGs, Neutrophil Specific Genes; NSTF, Neutrophil Specific Transcription Factor; PBMCs, Peripheral Blood Mononuclear Cells; PCA, Principal Component Analysis; PPI, Protein-Protein Interaction; OCRs, Open Chromatin Regions; RT-qPCR, Reverse Transcriptase Polymerase Chain Reaction; TLR, Toll-Like Receptor; TF, Transcription Factor; TFBM, Transcription Factor Binding Motifs; TMM, Trimmed Mean of M Values; TPM, Transcript Per Million; TSS, Transcription Start Site; SEGs, Significantly Enriched Genes.
1https://broadinstitute.github.io/picard/
2https://hbctraining.github.io/Intro-to-ChIPseq/lessons/05_peak_calling_macs.html
3http://homer.ucsd.edu/homer/ngs/annotation.html
5https://www.encodeproject.org/atac-seq/
Ackermann, A. M., Wang, Z., Schug, J., Naji, A., and Kaestner, K. H. (2016). Integration of ATAC-seq and RNA-seq identifies human alpha cell and beta cell signature genes. Mol. Metab. 5, 233–244. doi:10.1016/j.molmet.2016.01.002
Anguita, E., Candel, F. J., Chaparro, A., and Roldán-Etcheverry, J. J. (2017). Transcription factor GFI1B in health and disease. Front. Oncol. 7, 54. doi:10.3389/fonc.2017.00054
Bader, G. D., and Hogue, C. W. (2003). An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinforma. 4, 2. doi:10.1186/1471-2105-4-2
Benjamini, Y., and Hochberg, Y. (1995). Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B Stat. Methodol. 57, 289–300. doi:10.1111/j.2517-6161.1995.tb02031.x
Bjerregaard, M. D., Jurlander, J., Klausen, P., Borregaard, N., and Cowland, J. B. (2003). The in vivo profile of transcription factors during neutrophil differentiation in human bone marrow. Blood 101, 4322–4332. doi:10.1182/blood-2002-03-0835
Bradley, L. M., Douglass, M. F., Chatterjee, D., Akira, S., and Baaten, B. J. (2012). Matrix metalloprotease 9 mediates neutrophil migration into the airways in response to influenza virus-induced toll-like receptor signaling. PLoS Pathog. 8, e1002641. doi:10.1371/journal.ppat.1002641
Braun, T. P., Okhovat, M., Coblentz, C., Carratt, S. A., Foley, A., Schonrock, Z., et al. (2019). Myeloid lineage enhancers drive oncogene synergy in CEBPA/CSF3R mutant acute myeloid leukemia. Nat. Commun. 10, 5455. doi:10.1038/s41467-019-13364-2
Buenrostro, J. D., Giresi, P. G., Zaba, L. C., Chang, H. Y., and Greenleaf, W. J. (2013). Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods. 10, 1213–1218. doi:10.1038/nmeth.2688
Buenrostro, J. D., Wu, B., Chang, H. Y., and Greenleaf, W. J. (2015). ATAC-seq: A method for assaying chromatin accessibility genome-wide. Curr. Protoc. Mol. Biol. 109, 21. doi:10.1002/0471142727.mb2129s109
Byrne, K. A., Tuggle, C. K., and Loving, C. L. (2021). Differential induction of innate memory in porcine monocytes by β-glucan or bacillus Calmette-Guerin. Innate Immun. 08 (27), 448–460. doi:10.1177/1753425920951607
Chen, X., Shen, Y., Draper, W., Buenrostro, J. D., Litzenburger, U., Cho, S. W., et al. (2016). ATAC-see reveals the accessible genome by transposase-mediated imaging and sequencing. Nat. Methods 13, 1013–1020. doi:10.1038/nmeth.4031
Chen, H., Du, G., Song, X., and Li, L. (2017). Non-coding transcripts from enhancers: New insights into enhancer activity and gene expression regulation. Genomics Proteomics Bioinforma. 15, 201–207. doi:10.1016/j.gpb.2017.02.003
Choi, J., Baldwin, T., Wong, M., Bolden, J., Fairfax, K., Lucas, E., et al. (2019). Haemopedia RNA-seq: A database of gene expression during haematopoiesis in mice and humans. Nucleic Acids Res. 47, D780–D785. doi:10.1093/nar/gky1020
Corbett, R. J., Luttman, A. M., Herrera-Uribe, J., Liu, H., Raney, N. E., Grabowski, J. M., et al. (2022). Assessment of DNA methylation in porcine immune cells reveals novel regulatory elements associated with cell-specific gene expression and immune capacity traits. BMC Genomics 23, 575. doi:10.1186/s12864-022-08773-5
Costa, C., Germena, G., Martin-Conte, E. L., Molineris, I., Bosco, E., Marengo, S., et al. (2011). The RacGAP ArhGAP15 is a master negative regulator of neutrophil functions. Blood 118, 1099–1108. doi:10.1182/blood-2010-12-324756
Daugherty, A. C., Yeo, R. W., Buenrostro, J. D., Greenleaf, W. J., Kundaje, A., and Brunet, A. (2017). Chromatin accessibility dynamics reveal novel functional enhancers in C. elegans. Genome Res. 12 (27), 2096–2107. doi:10.1101/gr.226233.117
de Oliveira, S., Rosowski, E. E., and Huttenlocher, A. (2016). Neutrophil migration in infection and wound repair: Going forward in reverse. Nat. Rev. Immunol. 27 (16), 378–391. doi:10.1038/nri.2016.49
Denholtz, M., Zhu, Y., He, Z., Lu, H., Isoda, T., Döhrmann, S., et al. (2020). Upon microbial challenge, human neutrophils undergo rapid changes in nuclear architecture and chromatin folding to orchestrate an immediate inflammatory gene program. Genes Dev. 34, 149–165. doi:10.1101/gad.333708.119
do Nascimento, N. C., Guimaraes, A. M. S., Dos Santos, A. P., Chu, Y., Marques, L. M., and Messick, J. B. (2018). RNA-Seq based transcriptome of whole blood from immunocompetent pigs (Sus scrofa) experimentally infected with Mycoplasma suis strain Illinois. Vet. Res. 49:49. doi:10.1186/s13567-018-0546-6
Dong, Q., Lunney, J. K., Lim, K. S., Nguyen, Y., Hess, A. S., Beiki, H., et al. (2021). Gene expression in tonsils in swine following infection with porcine reproductive and respiratory syndrome virus. BMC Vet. Res. 17, 88. doi:10.1186/s12917-021-02785-1
Du, F., Dong, D., Zhang, X., and Jia, J. (2021). MXD1 is a potential prognostic biomarker and correlated with specific molecular change and tumor microenvironment feature in esophageal squamous cell carcinoma. Technol. Cancer Res. Treat. 20, 15330338211052142, doi:10.1177/15330338211052142
Duan, Z., and Horwitz, M. (2003). Gfi-1 oncoproteins in hematopoiesis. Hematology. 8, 339–344. doi:10.1080/10245330310001612116
Durinck, S., Spellman, P. T., Birney, E., and Huber, W. (2009). Mapping identifiers for the integration of genomic datasets with the R/Bioconductor package biomaRt. Nat. Protoc. 4, 1184–1191. doi:10.1038/nprot.2009.97
Ecker, S., Chen, L., Pancaldi, V., Bagger, F. O., Fernández, J. M., Carrillo de Santa Pau, E., et al. (2017). Genome-wide analysis of differential transcriptional and epigenetic variability across human immune cell types. Genome Biol. 18, 18. doi:10.1186/s13059-017-1156-8
Ewels, P., Magnusson, M., Lundin, S., and Käller, M. (2016). MultiQC: Summarize analysis results for multiple tools and samples in a single report. Bioinformatics 32, 3047–3048. doi:10.1093/bioinformatics/btw354
Ewels, P. A., Peltzer, A., Fillinger, S., Patel, H., Alneberg, J., Wilm, A., et al. (2020). The nf-core framework for community-curated bioinformatics pipelines. Nat. Biotechnol. 38, 276–278. doi:10.1038/s41587-020-0439-x
Fingerhut, L., Dolz, G., and de Buhr, N. (2020). What is the evolutionary fingerprint in neutrophil granulocytes? Int. J. Mol. Sci. 25, 4523. doi:10.3390/ijms21124523
Fischer, J., Walter, C., Tönges, A., Aleth, H., Jordão, M. J. C., Leddin, M., et al. (2019). Safeguard function of PU.1 shapes the inflammatory epigenome of neutrophils. Nat. Immunol. 20, 546–558. doi:10.1038/s41590-019-0343-z
Funaro, A., Ortolan, E., Ferranti, B., Gargiulo, L., Notaro, R., Luzzatto, L., et al. (2004). CD157 is an important mediator of neutrophil adhesion and migration. Blood 104, 4269–4278. doi:10.1182/blood-2004-06-2129
Geissler, S., Textor, M., Stumpp, S., Seitz, S., Lekaj, A., Brunk, S., et al. (2018). Loss of murine Gfi1 causes neutropenia and induces osteoporosis depending on the pathogen load and systemic inflammation. PLoS One 13, e0198510. doi:10.1371/journal.pone.0198510
Gierlikowska, B., Stachura, A., Gierlikowski, W., and Demkow, U. (2021). Phagocytosis, degranulation and extracellular traps release by neutrophils-the current knowledge, pharmacological modulation and future prospects. Front. Pharmacol. 12, 666732. doi:10.3389/fphar.2021.666732
Giuffra, E., Tuggle, C. K., and Consortium, F. (2019). Functional annotation of animal genomes (FAANG): Current achievements and roadmap. Annu. Rev. Anim. Biosci. 7, 65–88. doi:10.1146/annurev-animal-020518-114913
Grassi, L., Pourfarzad, F., Ullrich, S., Merkel, A., Were, F., Carrillo-de-Santa-Pau, E., et al. (2018). Dynamics of transcription regulation in human bone marrow myeloid differentiation to mature blood neutrophils. Cell Rep. 24, 2784–2794. doi:10.1016/j.celrep.2018.08.018
Heit, B., Jones, G., Knight, D., Antony, J. M., Gill, M. J., Brown, C., et al. (2006). HIV and other lentiviral infections cause defects in neutrophil chemotaxis, recruitment, and cell structure: Immunorestorative effects of granulocyte-macrophage colony-stimulating factor. J. Immunol. 177, 6405–6414. doi:10.4049/jimmunol.177.9.6405
Herrera-Uribe, J., Liu, H., Byrne, K., Bond, Z., Loving, C., and Tuggle, C. (2020). Changes in H3K27ac at gene regulatory regions in porcine alveolar macrophages following LPS or PolyIC exposure. Front. Genet. 11, 817. doi:10.3389/fgene.2020.00817
Herrera-Uribe, J., Wiarda, J., Sivasankaran, S., Daharsh, L., Liu, H., Byrne, K., et al. (2021). Reference transcriptomes of porcine peripheral immune cells created through bulk and single-cell RNA sequencing. Front. Genet. 12, 689406. doi:10.3389/fgene.2021.689406
Hicks, S. C., and Irizarry, R. A. (2015). quantro: a data-driven approach to guide the choice of an appropriate normalization method. Genome Biol. 16, 117. doi:10.1186/s13059-015-0679-0
Hicks, S. C., Okrah, K., Paulson, J. N., Quackenbush, J., Irizarry, R. A., and Bravo, H. C. (2018). Smooth quantile normalization. Biostat. Oxf. Engl. 19, 185–198. doi:10.1093/biostatistics/kxx028
Hong, C. W. (2017). Current understanding in neutrophil differentiation and heterogeneity. Immune Netw. 17, 298–306. doi:10.4110/in.2017.17.5.298
Huang, T., Jiang, C., Yang, M., Xiao, H., Huang, X., Wu, L., et al. (2020). Salmonella enterica serovar Typhimurium inhibits the innate immune response and promotes apoptosis in a ribosomal/TRP53-dependent manner in swine neutrophils. Vet. Res. 27, 105. doi:10.1186/s13567-020-00828-3
Hughes, A. E., Enright, J. M., Myers, C. A., Shen, S. Q., and Corbo, J. C. (2017). Cell type-specific epigenomic analysis reveals a uniquely closed chromatin architecture in mouse rod photoreceptors. Sci. Rep. 7, 43184. doi:10.1038/srep43184
Humbert, M., Halter, V., Shan, D., Laedrach, J., Leibundgut, E. O., Baerlocher, G. M., et al. (2011). Deregulated expression of Kruppel-like factors in acute myeloid leukemia. Leuk. Res. 35, 909–913. doi:10.1016/j.leukres.2011.03.010
Jain, V., Yang, W. H., Wu, J., Roback, J. D., Gregory, S. G., and Chi, J. T. (2022). Single cell RNA-seq analysis of human red cells. Front. Physiol. 13, 828700. doi:10.3389/fphys.2022.828700
Kern, C., Wang, Y., Xu, X., Pan, Z., Halstead, M., Chanthavixay, G., et al. (2021). Functional annotations of three domestic animal genomes provide vital resources for comparative and agricultural research. Nat. Commun. 23 (12), 1821. doi:10.1038/s41467-021-22100-8
Khoyratty, T. E., Ai, Z., Ballesteros, I., Eames, H. L., Mathie, S., Martín-Salamanca, S., et al. (2021). Distinct transcription factor networks control neutrophil-driven inflammation. Nat. Immunol. 22, 1093–1106. doi:10.1038/s41590-021-00968-4
Koesterke, L., Koltes, J. E., Weeks, N. T., Milfeld, K., Vaughn, M., Reecy, J. M., et al. (2013). “Optimizing the PCIT algorithm on Stampede’s Xeon and Xeon Phi processors for faster discovery of biological networks,” in Proceedings of the XSEDE2013 Conference (ACM).
Kwok, I., Becht, E., Xia, Y., Ng, M., Teh, Y. C., Tan, L., et al. (2020). Combinatorial single-cell analyses of granulocyte-monocyte progenitor heterogeneity reveals an early uni-potent neutrophil progenitor. Immunity 53, 303–318. doi:10.1016/j.immuni.2020.06.005
Laidlaw, B. J., Duan, L., Xu, Y., Vazquez, S. E., and Cyster, J. G. (2020). The transcription factor Hhex cooperates with the corepressor Tle3 to promote memory B cell development. Nat. Immunol. 21, 1082–1093. doi:10.1038/s41590-020-0713-6
Li, H., and Durbin, R. (2010). Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595. doi:10.1093/bioinformatics/btp698
Li, T., Wernersson, R., Hansen, R. B., Horn, H., Mercer, J., Slodkowicz, G., et al. (2017). A scored human protein-protein interaction network to catalyze genomic interpretation. Nat. Methods 14, 61–64. doi:10.1038/nmeth.4083
Li, K., Wu, Y., Li, Y., Yu, Q., Tian, Z., Wei, H., et al. (2020). Landscape and dynamics of the transcriptional regulatory network during natural killer cell differentiation. Genomics Proteomics Bioinforma. 18, 501–515. doi:10.1016/j.gpb.2020.12.003
Lim, Y. C., Li, J., Ni, Y., Liang, Q., Zhang, J., Yeo, G. S. H., et al. (2017). A complex association between DNA methylation and gene expression in human placenta at first and third trimesters. PLoS One 12, e0181155. doi:10.1371/journal.pone.0181155
Lindblom, S. C., Gabler, N. K., Dilger, R. N., Olson, Z. F., Loving, C. L., and Kerr, B. J. (2018). Influence of feeding thermally peroxidized soybean oil on oxidative status in growing pigs. J. Anim. Sci. 96, 545–557. doi:10.1093/jas/sky005
Liu, H., Smith, T. P. L., Nonneman, D. J., Dekkers, J. C. M., and Tuggle, C. K. (2017). A high-quality annotated transcriptome of swine peripheral blood. BMC Genomics 18, 479. doi:10.1186/s12864-017-3863-7
Liu, S., Gao, Y., Canela-Xandri, O., Wang, S., Yu, Y., Cai, W., et al. (2022). A multi-tissue atlas of regulatory variants in cattle. Nat. Genet. 54, 1438–1447. doi:10.1038/s41588-022-01153-5
Livak, K. J., and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408. doi:10.1006/meth.2001.1262
Love, M. I., Huber, W., and Anders, S. (2014). Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550. doi:10.1186/s13059-014-0550-8
Lowe, E. K., Cuomo, C., Voronov, D., and Arnone, M. I. (2019). Using ATAC-seq and RNA-seq to increase resolution in GRN connectivity. Methods Cell Biol. 151, 115–126. doi:10.1016/bs.mcb.2018.11.001
Lummertz da Rocha, E., Rowe, R. G., Lundin, V., Malleshaiah, M., Jha, D. K., Rambo, C. R., et al. (2018). Reconstruction of complex single-cell trajectories using CellRouter. Nat. Commun. 9, 892. doi:10.1038/s41467-018-03214-y
Maslova, A., Ramirez, R. N., Ma, K., Schmutz, H., Wang, C., Fox, C., et al. (2020). Deep learning of immune cell differentiation. Proc. Natl. Acad. Sci. U. S. A. 117, 25655–25666. doi:10.1073/pnas.2011795117
Mehta, H. M., Glaubach, T., and Corey, S. J. (2014). Systems approach to phagocyte production and activation: Neutrophils and monocytes. Adv. Exp. Med. Biol. 844, 99–113. doi:10.1007/978-1-4939-2095-2_6
Metzemaekers, M., Gouwy, M., and Proost, P. (2020a). Neutrophil chemoattractant receptors in health and disease: Double-edged swords. Cell Mol. Immunol. 17, 433–450. doi:10.1038/s41423-020-0412-0
Metzemaekers, M., Vandendriessche, S., Berghmans, N., Gouwy, M., and Proost, P. (2020b). Truncation of CXCL8 to CXCL8(9-77) enhances actin polymerization and in vivo migration of neutrophils. J. Leukoc. Biol. 107, 1167–1173. doi:10.1002/JLB.3AB0220-470R
Milani, P., Escalante-Chong, R., Shelley, B. C., Patel-Murray, N. L., Xin, X., Adam, M., et al. (2016). Cell freezing protocol suitable for ATAC-Seq on motor neurons derived from human induced pluripotent stem cells. Sci. Rep. 6, 25474. doi:10.1038/srep25474
Mildner, A., Schönheit, J., Giladi, A., David, E., Lara-Astiaso, D., Lorenzo-Vivas, E., et al. (2017). Genomic characterization of murine monocytes reveals C/EBPβ transcription factor dependence of Ly6C. Immunity 46, 849–862.e847. doi:10.1016/j.immuni.2017.04.018
Mistry, P., Nakabo, S., O'Neil, L., Goel, R. R., Jiang, K., Carmona-Rivera, C., et al. (2019). Transcriptomic, epigenetic, and functional analyses implicate neutrophil diversity in the pathogenesis of systemic lupus erythematosus. Proc. Natl. Acad. Sci. U. S. A. 116, 25222–25228. doi:10.1073/pnas.1908576116
Mola, S., Foisy, S., Boucher, G., Major, F., Beauchamp, C., Karaky, M., et al. (2020). A transcriptome-based approach to identify functional modules within and across primary human immune cells. PLoS One 15, e0233543. doi:10.1371/journal.pone.0233543
Monaco, G., Lee, B., Xu, W., Mustafah, S., Hwang, Y. Y., Carré, C., et al. (2019). RNA-seq signatures normalized by mRNA abundance allow absolute deconvolution of human immune cell types. Cell Rep. 26, 1627–1640. doi:10.1016/j.celrep.2019.01.041
Mortaz, E., Alipoor, S. D., Adcock, I. M., Mumby, S., and Koenderman, L. (2018). Update on neutrophil function in severe inflammation. Front. Immunol. 9, 2171. doi:10.3389/fimmu.2018.02171
Natarajan, A., Yardimci, G. G., Sheffield, N. C., Crawford, G. E., and Ohler, U. (2012). Predicting cell-type-specific gene expression from regions of open chromatin. Genome Res. Sep. 22, 1711–1722. doi:10.1101/gr.135129.111
Nerlov, C., Querfurth, E., Kulessa, H., and Graf, T. (2000). GATA-1 interacts with the myeloid PU.1 transcription factor and represses PU.1-dependent transcription. Blood 95, 2543–2551. doi:10.1182/blood.v95.8.2543.008k19_2543_2551
Ono, R., Kaisho, T., and Tanaka, T. (2015). PDLIM1 inhibits NF-κB-mediated inflammatory signaling by sequestering the p65 subunit of NF-κB in the cytoplasm. Sci. Rep. 5, 18327. doi:10.1038/srep18327
Ortolan, E., Tibaldi, E. V., Ferranti, B., Lavagno, L., Garbarino, G., Notaro, R., et al. (2006). CD157 plays a pivotal role in neutrophil transendothelial migration. Blood 108, 4214–4222. doi:10.1182/blood-2006-04-017160
Ou, J., Liu, H., Yu, J., Kelliher, M. A., Castilla, L. H., Lawson, N. D., et al. (2018). ATACseqQC: A bioconductor package for post-alignment quality assessment of ATAC-seq data. BMC Genomics 19, 169. doi:10.1186/s12864-018-4559-3
Pan, Z., Yao, Y., Yin, H., Cai, Z., Wang, Y., Bai, L., et al. (2021). Pig genome functional annotation enhances the biological interpretation of complex traits and human disease. Nat. Commun. 12, 5848. doi:10.1038/s41467-021-26153-7
Panigrahi, A., and O'Malley, B. W. (2021). Mechanisms of enhancer action: The known and the unknown. Genome Biol. 22, 108. doi:10.1186/s13059-021-02322-1
Patro, R., Duggal, G., Love, M. I., Irizarry, R. A., and Kingsford, C. (2017). Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods. 14, 417–419. doi:10.1038/nmeth.4197
Paulson, J. N., Chen, C. Y., Lopes-Ramos, C. M., Kuijjer, M. L., Platig, J., Sonawane, A. R., et al. (2017). Tissue-aware RNA-Seq processing and normalization for heterogeneous and sparse data. BMC Bioinforma. 18, 437. doi:10.1186/s12859-017-1847-x
Pipkin, M. E., and Lichtenheld, M. G. (2006). A reliable method to display authentic DNase I hypersensitive sites at long-ranges in single-copy genes from large genomes. Nucleic Acids Res. 34, e34. doi:10.1093/nar/gkl006
Quinlan, A. R., and Hall, I. M. (2010). BEDTools: A flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842. doi:10.1093/bioinformatics/btq033
Rajbhandari, P., Thomas, B. J., Feng, A. C., Hong, C., Wang, J., Vergnes, L., et al. (2018). IL-10 signaling remodels adipose chromatin architecture to limit thermogenesis and energy expenditure. Cell 172, 218–233. doi:10.1016/j.cell.2017.11.019
Ramírez, F., Dündar, F., Diehl, S., Grüning, B. A., and Manke, T. (2014). deepTools: a flexible platform for exploring deep-sequencing data. Nucleic Acids Res. 42, W187–W191. doi:10.1093/nar/gku365
Ramírez, F., Ryan, D. P., Grüning, B., Bhardwaj, V., Kilpert, F., Richter, A. S., et al. (2016). deepTools2: a next generation web server for deep-sequencing data analysis. Nucleic Acids Res. 44, W160–165.
Ram-Mohan, N., Thair, S. A., Litzenburger, U. M., Cogill, S., Andini, N., Yang, X., et al. (2021). Profiling chromatin accessibility responses in human neutrophils with sensitive pathogen detection. Life Sci. Alliance 08, e202000976. doi:10.26508/lsa.202000976
Reverter, A., and Chan, E. K. (2008). Combining partial correlation and an information theory approach to the reversed engineering of gene co-expression networks. Bioinformatics 24, 2491–2497. doi:10.1093/bioinformatics/btn482
Rosales, C. (2018). Neutrophil: A cell with many roles in inflammation or several cell types? Front. Physiol. 9, 113. doi:10.3389/fphys.2018.00113
Rosenbauer, F., and Tenen, D. G. (2007). Transcription factors in myeloid development: Balancing differentiation with transformation. Nat. Rev. Immunol. 7, 105–117. doi:10.1038/nri2024
Roy, R., Ramamoorthy, S., Shapiro, B. D., Kaileh, M., Hernandez, D., Sarantopoulou, D., et al. (2021). DNA methylation signatures reveal that distinct combinations of transcription factors specify human immune cell epigenetic identity. Immunity 54, 2465–2480.e5. doi:10.1016/j.immuni.2021.10.001
Rudd, J. M., Pulavendran, S., Ashar, H. K., Ritchey, J. W., Snider, T. A., Malayer, J. R., et al. (2019). Neutrophils induce a novel chemokine receptors repertoire during influenza pneumonia. Front. Cell Infect. Microbiol. 9, 108. doi:10.3389/fcimb.2019.00108
Saleque, S., Cameron, S., and Orkin, S. H. (2002). The zinc-finger proto-oncogene Gfi-1b is essential for development of the erythroid and megakaryocytic lineages. Genes Dev. 16, 301–306. doi:10.1101/gad.959102
Sanz-Santos, G., Jiménez-Marín, A., Bautista, R., Fernández, N., Claros, G. M., and Garrido, J. J. (2011). Gene expression pattern in swine neutrophils after lipopolysaccharide exposure: A time course comparison. BMC Proc. 5 Suppl 4, S11. doi:10.1186/1753-6561-5-S4-S11
Shahrin, N. H., Diakiw, S., Dent, L. A., Brown, A. L., and D'Andrea, R. J. (2016). Conditional knockout mice demonstrate function of Klf5 as a myeloid transcription factor. Blood 128, 55–59. doi:10.1182/blood-2015-12-684514
Shannon, P., Markiel, A., Ozier, O., Baliga, N. S., Wang, J. T., Ramage, D., et al. (2003). Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504. doi:10.1101/gr.1239303
Spainhour, J. C., Lim, H. S., Yi, S. V., and Qiu, P. (2019). Correlation patterns between DNA methylation and gene expression in the cancer genome atlas. Cancer Inf. 18, 1176935119828776. doi:10.1177/1176935119828776
Stark, C., Breitkreutz, B. J., Reguly, T., Boucher, L., Breitkreutz, A., and Tyers, M. (2006). BioGRID: A general repository for interaction datasets. Nucleic Acids Res. 34, D535–D539. doi:10.1093/nar/gkj109
Stein, G. S., Stein, J. L., Van Wijnen, A. J., Lian, J. B., Montecino, M., Croce, C. M., et al. (2010). Transcription factor-mediated epigenetic regulation of cell growth and phenotype for biological control and cancer. Adv. Enzyme Regul. 50, 160–167. doi:10.1016/j.advenzreg.2009.10.026
Stevenson, W. S., Morel-Kopp, M. C., Chen, Q., Liang, H. P., Bromhead, C. J., Wright, S., et al. (2013). GFI1B mutation causes a bleeding disorder with abnormal platelet function. J. Thromb. Haemost. 11, 2039–2047. doi:10.1111/jth.12368
Su, Y., Shin, J., Zhong, C., Wang, S., Roychowdhury, P., Lim, J., et al. (2017). Neuronal activity modifies the chromatin accessibility landscape in the adult brain. Nat. Neurosci. 20, 476–483. doi:10.1038/nn.4494
Summers, K. M., Bush, S. J., and Hume, D. A. (2020). Network analysis of transcriptomic diversity amongst resident tissue macrophages and dendritic cells in the mouse mononuclear phagocyte system. PLoS Biol. 10, e3000859. doi:10.1371/journal.pbio.3000859
Szklarczyk, D., Gable, A. L., Lyon, D., Junge, A., Wyder, S., Huerta-Cepas, J., et al. (2019). STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 47, D607–D613. doi:10.1093/nar/gky1131
Tamassia, N., Le Moigne, V., Rossato, M., Donini, M., McCartney, S., Calzetti, F., et al. (2008). Activation of an immunoregulatory and antiviral gene expression program in poly(I:C)-transfected human neutrophils. J. Immunol. 181, 6563–6573. doi:10.4049/jimmunol.181.9.6563
Thurman, R. E., Rynes, E., Humbert, R., Vierstra, J., Maurano, M. T., Haugen, E., et al. (2012). The accessible chromatin landscape of the human genome. Nature 489, 75–82. doi:10.1038/nature11232
Türei, D., Korcsmáros, T., and Saez-Rodriguez, J. (2016). OmniPath: Guidelines and gateway for literature-curated signaling pathway resources. Nat. Methods 13, 966–967. doi:10.1038/nmeth.4077
van Dam, S., Võsa, U., van der Graaf, A., Franke, L., and de Magalhães, J. P. (2018). Gene co-expression analysis for functional classification and gene-disease predictions. Brief. Bioinform. 19, 575–592. doi:10.1093/bib/bbw139
van der Meer, L. T., Jansen, J. H., and van der Reijden, B. A. (2010). Gfi1 and Gfi1b: Key regulators of hematopoiesis. Leukemia 24, 1834–1843. doi:10.1038/leu.2010.195
Vassen, L., Fiolka, K., Mahlmann, S., and Möröy, T. (2005). Direct transcriptional repression of the genes encoding the zinc-finger proteins Gfi1b and Gfi1 by Gfi1b. Nucleic Acids Res. 33, 987–998. doi:10.1093/nar/gki243
Vassen, L., Okayama, T., and Möröy, T. (2007). Gfi1b:green fluorescent protein knock-in mice reveal a dynamic expression pattern of Gfi1b during hematopoiesis that is largely complementary to Gfi1. Blood 109, 2356–2364. doi:10.1182/blood-2006-06-030031
Wang, H., Zhang, Q., Yin, L., Liu, X., Zhao, S., Zhu, M., et al. (2017). Transcriptomic basis of neutrophil ratio variation induced by poly I:C stimulation in porcine peripheral blood. Front. Agric. Sci. Eng. 4, 342–352. doi:10.15302/j-fase-2017162
Xie, X., Shi, Q., Wu, P., Zhang, X., Kambara, H., Su, J., et al. (2020). Single-cell transcriptome profiling reveals neutrophil heterogeneity in homeostasis and infection. Nat. Immunol. 21, 1119–1133. doi:10.1038/s41590-020-0736-z
Yan, F., Powell, D. R., Curtis, D. J., and Wong, N. C. (2020). From reads to insight: A hitchhiker's guide to ATAC-seq data analysis. Genome Biol. 21, 22. doi:10.1186/s13059-020-1929-3
Zhang, Q., Yang, Q., Weng, Y., Huang, Z., Chen, R., Zhu, Y., et al. (2021). Neutrophil-to-lymphocyte ratio correlates with prognosis and response to chemotherapy in patients with non-M3 de novo acute myeloid leukemia. Transl. Cancer Res. 10, 1013–1024. doi:10.21037/tcr-20-2179
Keywords: pig, transcriptome, bulk-RNA-seq, co-expressed genes, transcription factor (TF), gene regulation, immune cells, ATAC-seq
Citation: Herrera-Uribe J, Lim K-S, Byrne KA, Daharsh L, Liu H, Corbett RJ, Marco G, Schroyen M, Koltes JE, Loving CL and Tuggle CK (2023) Integrative profiling of gene expression and chromatin accessibility elucidates specific transcriptional networks in porcine neutrophils. Front. Genet. 14:1107462. doi: 10.3389/fgene.2023.1107462
Received: 24 November 2022; Accepted: 27 April 2023;
Published: 23 May 2023.
Edited by:
Martino Cassandro, University of Padua, ItalyReviewed by:
Brittney Keel, Agricultural Research Service (USDA), United StatesCopyright © 2023 Herrera-Uribe, Lim, Byrne, Daharsh, Liu, Corbett, Marco, Schroyen, Koltes, Loving and Tuggle. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Christopher K. Tuggle, Y2t0dWdnbGVAaWFzdGF0ZS5lZHU=
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.