- 1Beijing University of Chinese Medicine, Beijing, China
- 2Department of Oncology, China-Japan Friendship Hospital, Beijing, China
- 3Department of Oncology, Fangshan Hospital, Beijing, China
Background: Lung squamous cell carcinoma (LUSC) shares less typical onco-drivers and target resistance, but a high overall mutation rate and marked genomic complexity. Mismatch repair (MMR) deficiency leads to microsatellite instability (MSI) and genomic instability. MSI is not an ideal option for prognosis of LUSC, whereas its function deserves exploration.
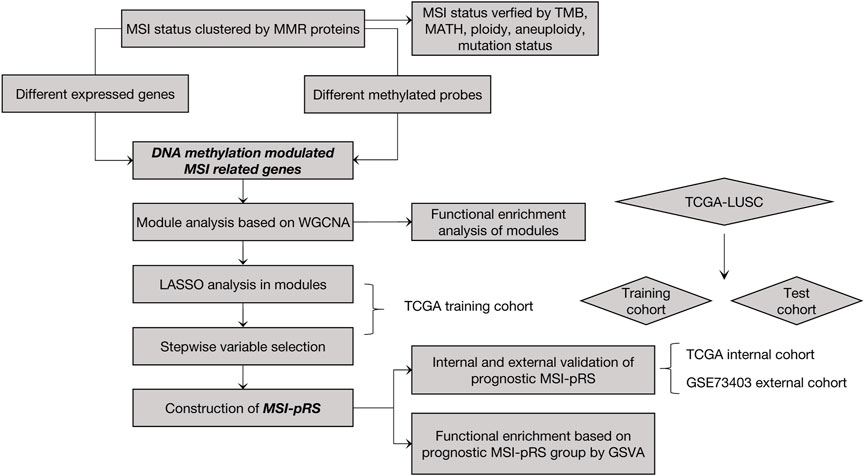
Method: MSI status was classified by MMR proteins using unsupervised clustering in the TCGA–LUSC dataset. The MSI score of each sample was determined by gene set variation analysis. Intersections of the differential expression genes and differential methylation probes were classified into functional modules by weighted gene co-expression network analysis. Least absolute shrinkage and selection operator regression and stepwise gene selection were performed for model downscaling.
Results: Compared with the MSI-low (MSI-L) phenotype, MSI-high (MSI-H) displayed higher genomic instability. The MSI score was decreased from MSI-H to normal samples (MSI-H > MSI-L > normal). A total of 843 genes activated by hypomethylation and 430 genes silenced by hypermethylation in MSI-H tumors were classified into six functional modules. CCDC68, LYSMD1, RPS7, and CDK20 were used to construct MSI-related prognostic risk score (MSI-pRS). Low MSI-pRS was a protective prognostic factor in all cohorts (HR = 0.46, 0.47, 0.37; p-value = 7.57e-06, 0.009, 0.021). The model contains tumor stage, age, and MSI-pRS that showed good discrimination and calibration. Decision curve analyses indicated that microsatellite instability-related prognostic risk score added extra value to the prognosis. A low MSI-pRS was negatively correlated with genomic instability. LUSC with low MSI-pRS was associated with increased genomic instability and cold immunophenotype.
Conclusion: MSI-pRS is a promising prognostic biomarker in LUSC as the substitute of MSI. Moreover, we first declared that LYSMD1 contributed to genomic instability of LUSC. Our findings provided new insights in the biomarker finder of LUSC.
1 Introduction
Lung squamous cell carcinoma (LUSC) comprises 20% of non-small cell lung cancer (NSCLC) cases (Santos and Rodriguez, 2022). Compared with lung adenocarcinoma (LUAD), targetable genetic aberrations are not typical and target therapy is not ideal in LUSC. Immune checkpoint inhibitor (ICI)-based combination regimens have been moved into the first-line option, which led to a landmark change in the treatment of LUSC (Santos and Rodriguez, 2022). In contrast to LUAD, for smoking or other chemical exposures, the molecular profile of all LUSC stages is characterized by highly heterogeneous malignancy, with high genomic instability contributing to the high tumor mutational burden (Heist et al., 2012; Chen et al., 2022).
Genomic instability is an enabling hallmark of tumorigenesis and is the consequence of the DNA damage repair (DDR) system deficiency. DDR deficiency results from mutations of large-scale upstream cancer suppressor genes, such as TP53, which is commonly mutated across pan-cancer and in at least 80% of LUSC cases, and is enhanced by the following clonal evolution of cells (Mandal et al., 2019). DDR defects conventionally lead to either chromosomal instability (CIN) or microsatellite instability (MSI). CIN describes a wide variety of chromosomal abnormalities, including chromosomal rearrangements, deletions, insertions, and amplifications. MSI deriving from deficiency of DNA mismatch repair (MMR) manifests as the insertion of a few base pairs or deletion mutations, specifically at a repetitive sequence during DNA replication and genetic recombination (Zhao et al., 2019).
MMR involves a series of proteins which act in the manner of homodimers. MutS homologs (MSH2, MSH3, and MSH6) are responsible for detecting, recognizing, and binding mismatch errors. MutL homologs (MLH1 and PMS2) participate in the excision and synthesis of corrected DNA bases. Repression of transcription or functional defects in one or more MMR enzymes results in a systemic MMR deficiency (MMR-d). Hypermethylation and deletion mutations of MMR genes, especially those of MLH1 and MSH2, lead to transcriptional silence accounting for the majority of MMR-d. Alternations of MSH6 and PMS2 contribute a part of the remainder (Xiao et al., 2014; Yanagawa et al., 2021).
In the absence of an efficient correction system, tumors with MMR-d backgrounds are particularly sensitive to DNA mismatch errors and manifest as the accumulation of mutations in brief repetitive DNA sequences (microsatellite sites), which is acknowledged as microsatellite instability-high (MSI-H). MSI-H occurs in about 10%–25% of colorectal cancers, in about 5%–20% gastric cancers, and in about 13%–30% of endometrial cancers. MSI-H is associated with TP53 mutations and high tumor mutation burden (TMB), which leads to tumor immunogenicity and stimulates the host anti-tumor immune response, thereby being sensitive to immune checkpoint inhibitors (ICIs).
The prevalence of MSI-H in NSCLC is not as prevalent as in the previously described cancers with frequencies of 0.17%–0.8% (Warth et al., 2016; De Marchi et al., 2022). MSI-H NSCLC samples were frequently associated with heavy smoking history and tended to be LUSC or sharing squamous components (Woenckhaus et al., 2003). MSI may not act as the driver factor as that in the inherited cancer and tend to represent a type of genomic instability in lung cancer (Pastuszak-Lewandoska et al., 2016). The correlation of genome instability and the response to ICIs has been attached with great importance in NSCLC. A past pan-cancer study declared a positive association between the DDR-associated gene defect and the prevalence of programmed cell death-ligand 1 (PD-L1) in NSCLC. In addition, patients with a DDR defect acquired clinical benefit from ICIs with improved median progression-free survival (mPFS) and median overall survival (mOS) (Chae et al., 2019). Compared with LUAD, LUSC shares more complex genomic instability. It was reported that a direct relationship between DDR gene variants and T cell activation was observed in LUSC rather than in LUAD (Kim et al., 2020). Therefore, the MSI-H phenotype is presumably meaningful in LUSC. Distribution of MMR expression potentially reflects the de novo mechanism of genome instability formation; thus, it remains to be a potential indicator in NSCLC.
So far, the clinical implication of MSI in LUSC remains unclear. In this study, MSI-related prognostic risk score (MSI-pRS) was established by machine learning and bioinformatics methods. The MSI status of LUSC samples was distinguished based on the expression of MMR systems. According to the MSI status, differentially expressed genes (DEGs) were identified and then classified into the functional gene modules by weighted correlation network analysis (WGCNA). Ultimately, four key MSI-related prognostic genes, CCDC68, LYSMD1, RPS7, and CDK20, were screened and used to construct a new risk score model named MSI-pRS to predict LUSC. We further analyzed the genomic features, immune infiltration, and the association with driver genes. Internal and external dataset validations were used to further verify the MSI-pRS model.
2 Methods
2.1 Data collection and processing
TCGA–LUSC level 3 RNA-seq data (HTSeq-Counts) were directly downloaded using the GDC data transfer tool (https://portal.gdc.cancer.gov/). The TCGA cohort was randomly assigned into a training cohort and a validation cohort at a ratio of 3:2 using the caTools package. GSE73403 (Agilent-014850, whole human genome microarray 4x44K G4112F) was downloaded from Gene Expression Omnibus (GEO) datasets (https://www.ncbi.nlm.nih.gov/). GSE135222 was used for exploring the association between ICI response and gene expression (Ritchie et al., 2015). HTSeq-counts were transformed into log-2-transformed transcripts per kilo-base per million mapped reads (TPM). Gene length was calculated as the sum of lengths of the non-redundant exon. Agilent one-color microarray intensity data were read by the “read.maimages” function, background-correlated by “backgroundCorrect” function, and normalized by the “normalizeBetweenArrays” function. The processes described previously were all implemented in the limma package (Aryee et al., 2014).
TCGA–LUSC DNA methylation data (IDATs) were also downloaded and read by the “read.metharray.exp” function applied in minfi and filtered by the “champ.filter” function in the ChAMP R package (Wilkerson and Hayes, 2010; Tian et al., 2017). Particularly, poor performing probes with p-value above 0.05, belonging to a sex chromosome, known to have common SNPs at the CpG sites, or having been demonstrated to be mapped to multiple places in the genome were removed before differential methylation analysis. Normalization was performed by the BMIQ method with the “champ.norm” function (Chae et al., 2019). TCGA–LUSC minus germline somatic copy number alternations (CNAs) and merged somatic simple-nucleotide variation (sSNV) segmented data of the cohort were downloaded from GDAC Firehose (Broad Institute TCGA Genome Data Analysis Center, https://gdac.broadinstitute.org/) for analysis of mutation status.
2.2 Unsupervised classification of TCGA–LUSC samples for MSI status
An unsupervised clustering algorithm was applied to classify the MSI status of TCGA–LUSC samples based on the expression of seven genes encoding MMR proteins (MSH2, MSH3, MSH6, MLH1, MLH3, PMS2, and PMS1). The median absolute deviation (MAD) of the data matrix was used for further cluster analysis. 1,000 time repetitions were applied for guaranteeing the stability of classification. The agglomerative hierarchical clustering algorithm was based upon Pearson’s correlation distance. The highest cluster group was set as 6 (k = 6). The heatmap of consensus matrices, cluster-consensus plot, and item-consensus plot were used for defining the ultimate MSI clusters by taking the stability and purity of clusters into consideration (Hänzelmann et al., 2013). The aforementioned steps were carried out using the ConsensusClusterPlus package.
Gene set variation analysis (GSVA) was performed to derive the MSI score based on the MMR system gene set that contained the seven genes to identify the MSI status of each sample (Chalmers et al., 2017). Genomic instability of different groups based on MSI status was characterized and compared by measuring TMB, mutant-allele tumor heterogeneity (MATH), DNA ploidy status, and aneuploidy score. TMB was calculated as the rate of somatic non-synonymous mutations per megabase of sequenced DNA. The exome size was estimated as 38 Mb (Mayakonda et al., 2018). To evaluate tumor genomic heterogeneity, MATH was calculated as the MAD and the median of variant allele frequencies of non-synonymous variants using the “inferHeterogeneity” implemented in maftools (Carter et al., 2012). DNA content is the main biologic index of tumor multiplication potentiality. Ploidy reflects the actual DNA content of cancer cells (Taylor et al., 2018). Aneuploidy reflects the imbalance and complication of DNA replication. DNA ploidy calculated using the Absolute algorithm and the aneuploidy score of TCGA–LUSC samples was directly downloaded from https://gdc.cancer.gov/about-data/publications/panimmune (Langfelder and Horvath, 2008).
2.3 Identification of MSI-related genes regulated by DNA methylation
DNA methylation is the critical epigenetical mechanism of regulating MSI through transcriptionally silenced or activated hub gene expression in the MSI-related signaling by hyper- or hypo-DNA methylation of gene promoters, including TSS200, TSS1500, 1stexon, and 5′UTR. We further obtained the MSI-related genes regulated by DNA methylation in the following three steps. First, differentially expressed genes (DEGs) were identified from the intersection of results calculated by two methods based on limma and DESeq2. Second, similar ways were used to obtain different methylation probes (DMPs) using ChAMP and minfi packages (Pastuszak-Lewandoska et al., 2016; Chae et al., 2019). Third, the overlapping of genes targeted by DMPs in promoters and DEGs was obtained. Genes with the reverse methylation and expression status were identified as MSI-related, which were regulated by DNA methylation and used for further prognostic analysis.
Gene annotation was based on Homo. sapiens GRCh38.p13 GFF3 (v35) file (GENCODE website, https://www.gencodegenes.org/). The ensemble ID was converted into a gene symbol. Genes with duplicate annotation were represented by genes on the minor chromosomes. The threshold of DEGs and DMPs was defined as |logFC| > 0 and p-value <0.05.
2.4 Functional classification of epigenetically regulated MSI-related genes
MSI-related genes regulated by DNA methylation were classified into functional modules by gene co-expression networks using the WGCNA R package (Kuleshov et al., 2016). In this way, genes with similar patterns were grouped into the same module to realize feature dimension reduction. The soft thresholding power was set to 5 on the criterion of approximate scale-free topology by the “pickSoftThreshold” function. The weighted adjacency matrix was transformed into a topological overlap degree matrix (TOM). Hierarchical clustering was used to produce a hierarchical clustering tree of genes whose densely interconnected branches were highly co-expressed genes. Modules having shared similar expression profiles were simplified by a dynamic tree cut. Different colors represent different modules. We then quantified the associations of modules with an MSI phenotype to identify the MSI-related gene modules. Gene significance (GS) was defined as the absolute value of the correlation between the gene and the clinical phenotype. Module membership (MM) was defined as the correlation between the summary profile of the module and gene expression. The biological functions of gene modules were characterized by gene ontology (GO) using EnrichR (Gu et al., 2022).
2.5 Development of the MSI-related prognostic risk score (MSI-pRS)
The least absolute shrinkage and selection operator (LASSO) regression was performed using the glmnet R package for downscaling prognostic genes (Chen et al., 2021). Particularly, LASSO regression analyses were applied to the genes included in the functional modules classified in the 2.4 of the training cohort. The fitted lambda value for the model was screened by cross-validation. Prognostic genes derived from the modules were integrated for further stepwise variable selection procedure based on the multivariate Cox model to construct the ultimate MSI-pRS using the My.stepwise R package.
The expression matrix of the selected genes for the model was extracted, and the MSI-PRS of each sample was calculated using the following formula:
The MSI-pRS of sample i was calculated as the expression of candidate gene j in sample i, weighted by the coefficient in the multivariate Cox regression model. All the samples were stratified into high- and low- MSI-pRS groups. The cutting points were selected by the “surv-cutpoint” function implemented in the survminer R package, according to Chen et al. (Blanche et al., 2013). All potential cutting points were repeatedly tested to find the maximum rank statistic to reduce the calculated batch effect. We divided the whole cohorts based on the MSI-pRS to further explore the prognostic value of the MSI-pRS for convenient routine use. The “surv-cutpoint” function was used to dichotomize the MSI-pRS, and the Kaplan–Meier method was used and log-rank tests between groups were performed. Hazard ratios (HRs) of the MSI-pRS were derived from univariate Cox regression. Subgroup analysis of MSI-pRS groups was performed to eliminate the interference of interactive variables. The multivariate Cox model fitting into age, stage, and MSI-pRS was applied in training, internal validation, and external validation cohorts. Receiver operating characteristic (ROC) curves at years 1, 3, and 5 of the model were drawn to evaluate the discriminative ability of the MSI-pRS using the timeROC R package (Vickers and Elkin, 2006). The efficiency of the MSI-pRS was assessed by comparing the decision curve analyses (DCA) and curves of models with or without the MSI-pRS group (Charoentong et al., 2017). Statistical analysis was conducted using R software (version 4, 4.1.2). Survival analyses were carried out using the survival R package, and the forest plots were pictured using the forestplot R package.
2.6 Functional enrichment analysis
Tumor-infiltrated cells were estimated by single-sample gene set enrichment analysis (ssGSEA) using the GSVA package (Hänzelmann et al., 2013). Transcriptional data of tumor-infiltrating cells used for functional analysis were derived from Charoentong et al. (Rooney et al., 2015). The positive immune regulators were defined as the collection of “effector” cells, active dendritic cells (aDCs), natural killer cells (NKs), and natural killer T cells (NKTs). Negative immune regulators were defined as the collection of regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs). The “effector” cells were defined as active T cells (aCD4+T and aCD8+T) and effector memory T cells (CD4+Tem and CD8+Tem). Cytolytic activity (CYT) was used for evaluating immune activity and calculated as the geometric mean of granzyme A (GZMA) and perforin (PRF1) expression levels as previously defined (Cancer Genome Atlas Research Network, 2012). Functional enrichment analysis between groups was realized by GSVA based on gene expression data matrices.
3 Results
3.1 MSI status and the genomic instability features of LUSC
The construction process of the MSI-pRS is shown in Figure 1. A total of 551 samples consisting of 502 tumor samples and 49 normal samples were contained in the TCGA–LUSC RNA-seq dataset. A total of 472 LUSC patients with clinical outcomes, transcriptomics, and mutation data were included for the analysis. The expression data of 49 normal samples were used for the control.
The MSI status was determined by the expression data of the MMR proteins in the following process: (Santos and Rodriguez, 2022) the relative MSI status in the TCGA–LUSC cohort was identified by consensus clustering using k-means as the base method (Chen et al., 2022). The MSI score of each sample was calculated by GSVA, according to MMR proteins. We tested clustering for K = 2–6 and chose the optimal number of subgroups using consensus matrices. According to the item-consensus plot and cluster-consensus plot, K = 2 showed crisp clusters with acceptable stability and purity in both groups (Figures 2A–C). The patients were divided into two robust groups with low MSI (MSI-L) containing 196 samples and high MSI (MSI-H) containing 276 samples.
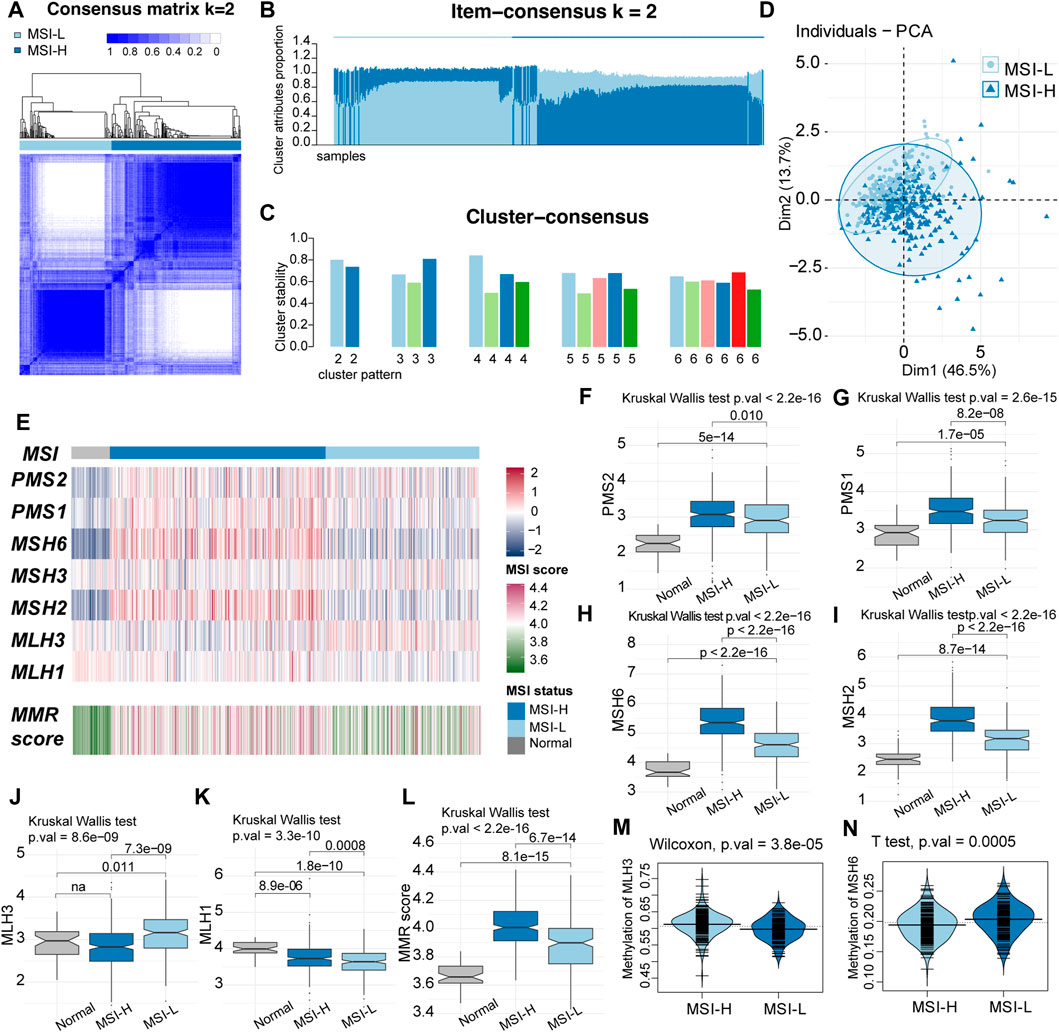
FIGURE 2. Two-group pattern of microsatellite instability (MSI) status by unsupervised classification in TCGA LUSC cohort. (A) Heatmap of consensus matrix when the classification pattern was two-group (k = 2). (B) Item consensus plot when k = 2 showed that the cluster pattern shared the acceptable purity in both groups. (C) Cluster consensus plot when k = 2 indicated the stability of the cluster patterns. (D) Principal component analysis (PCA) plot when k = 2 with 196 samples in MSI-L groups and 276 samples in the MSI-H group. (E) Comparation of the MMR proteins expression and the MSI score between MSI-H and MSI-L groups. MSI-H tended to had higher expression of MMR proteins and MSI score. (F–K) Comparation of MMR proteins among MSI-H, MSI-L and normal samples. (L) Boxplot of MMR score. MSI score was decreased from MSI-H samples to normal samples (MSI-H > MSI-L > normal). (M–N) Beanplot of methylation level of MLH3 and MSH6.
The principal component analysis (PCA) plot preliminarily showed that the two groups had some difference in the expression of MMR proteins that somehow overlapped (Figure 2D). Particularly, the expression of MMR proteins and MSI scores were compared among normal samples, MSI-H, and MSI-L LUSC samples. Among the seven MMR proteins, PMS2, PMS1, MSH6, and MSH2 were significantly highest in the MSI-H group, while the expression level of the normal group was lowest (MSI-H > MSI-L > normal, Figures 2E–I). Expression of MLH3 was highest in MSI-L samples (Figures 2E, J), and that of MLH1 was highest in the normal samples and lowest in the MSI-H samples (Figures 2E, K). MSH3 was not significantly different among the groups. The MSI score was highest in the MSI-H group and lowest in the normal samples (MSI-H > MSI-L > normal, Figures 2E, L). The difference in the promoter methylation level of MMR genes was observed in MLH3, and MSH6 was consistent with the expression of the two proteins (Figures 2M, N). No significant difference in clinical characteristics was discovered between these two clusters of patients (Supplementary Table S1).
Genomic features between the two clusters were evaluated by TMB, tumor heterogeneity, DNA content, and aneuploidy status. The MSI-H group tended to obtain a subpopulation of heterogenous tumors with higher median MATH (Mann–Whitney U-test, 34.89 vs. 38.38, p-value = 0.002, Figure 3A). Higher TMB, DNA content (ploidy), and aneuploidy score were observed in the MSI-H group (Mann–Whitney U-test, 7.63/mb vs. 6.09/mb, p-value = 1.745e-05, Figure 3B; 2.88 vs. 2.05, p-value = 0.013; Figure 3C; 17 vs. 14, p-value = 0.001; Figure 3D). TP53, the alternation of which was the most universal mutation in LUSC patients, was more frequently mutated in the MSI-H group (chi-square test, p-value = 0.0009 Figure 3E). Moreover, the MSI score was higher in the patients of the TP53 mut group (Student’s t-test, p-value = 4.4e-07, Figure 3F). To sum up, MSI-H in LUSC indicated a subgroup with higher genetic instability.
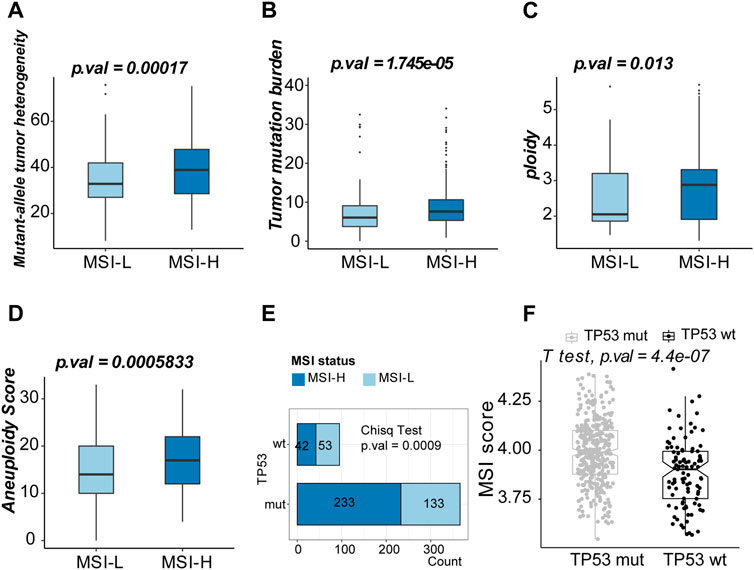
FIGURE 3. Exploration of the genomic features of MSI-H and MSI-L groups. (A) Boxplot of mutant–allele heterogeneity (MATH) (indicating intratumor heterogeneity). (B) Boxplot of tumor mutation burden (TMB). (C) Boxplot of ploidy (DNA content). (D) Boxplot of the aneuploidy score. (E) Comparison of the MSI scores between TP53 mut and TP53 wt groups. (F) Top 10 mutated genes in the MSI-L group. (G) Top ten mutated genes in the MSI-H group.
3.2 Identification of epigenetically regulated functional MSI-related genes
There were 7,299 and 4,013 upregulated genes recognized by limma and DESeq2 methods, respectively. The number of downregulated genes was 6,663 and 5,607. Ultimately, 3,811 DEGs were upregulated and 4,446 DEGs were downregulated in MSI-H samples. A total of 63,531 and 16,197 hypermethylated probes were calculated using minfi and ChAMP, while 63,547 and 45,387 probes were hypomethylated. The final DMPs were 14,090 hypermethylated probes and 33,501 hypomethylated probes in MSI-H samples. We eventually identified 843 genes activated by hypomethylation and 430 genes silenced by hypermethylation in MSI-H tumors (Supplemenatry Tables S2, S3; Figures 4A–C).
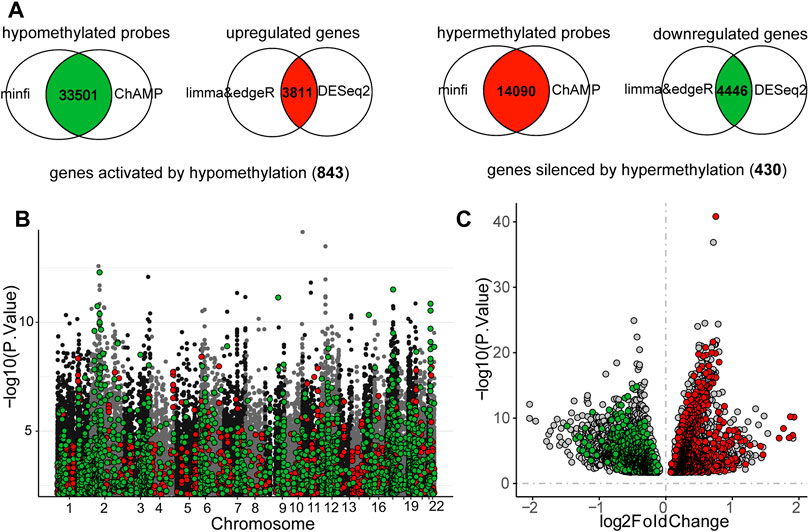
FIGURE 4. Intersection of the differentially expressed genes (DEGs) and different methylation probes (DMPs) to identify the DNA methylation regulated MSI-related genes. (A) Process of recognizing DEGs and DMPs. (B) Distribution of DNA methylation on chromosomes. MSI-related genes which were regulated by DNA methylation were highlighted. Red points represented probes that were hypermethylated in MSI-H, whereas green points indicated the probes were hypomethylated in MSI-H. (C) Volcano plot of DEGs between MSI-H and MSI-L groups. MSI-related genes which were regulated by DNA methylation were highlighted. Red points represented the upregulated MSI genes, and the green points represented the downregulated genes. Threshold of DEGs and DMPs was defined as: |logFC| > 0 and p-value <0.05.
We focused on the MSI-related genes which are epigenomically regulated by DNA methylation in the gene promoters and classified them into functional modules by gene co-expression networks. Six functional gene modules correlated with the MSI phenotype were identified. The gray module was the cluster of genes not related to any of the modules. Genes in the brown, turquoise, and yellow modules were more likely to be upregulated through DNA hypomethylation in the MSI-H group, whereas genes in blue and green modules tended to be downregulated by DNA hypermethylation in the MSI-H group (Figures 5A, B; Supplementary Figures S1A–D).
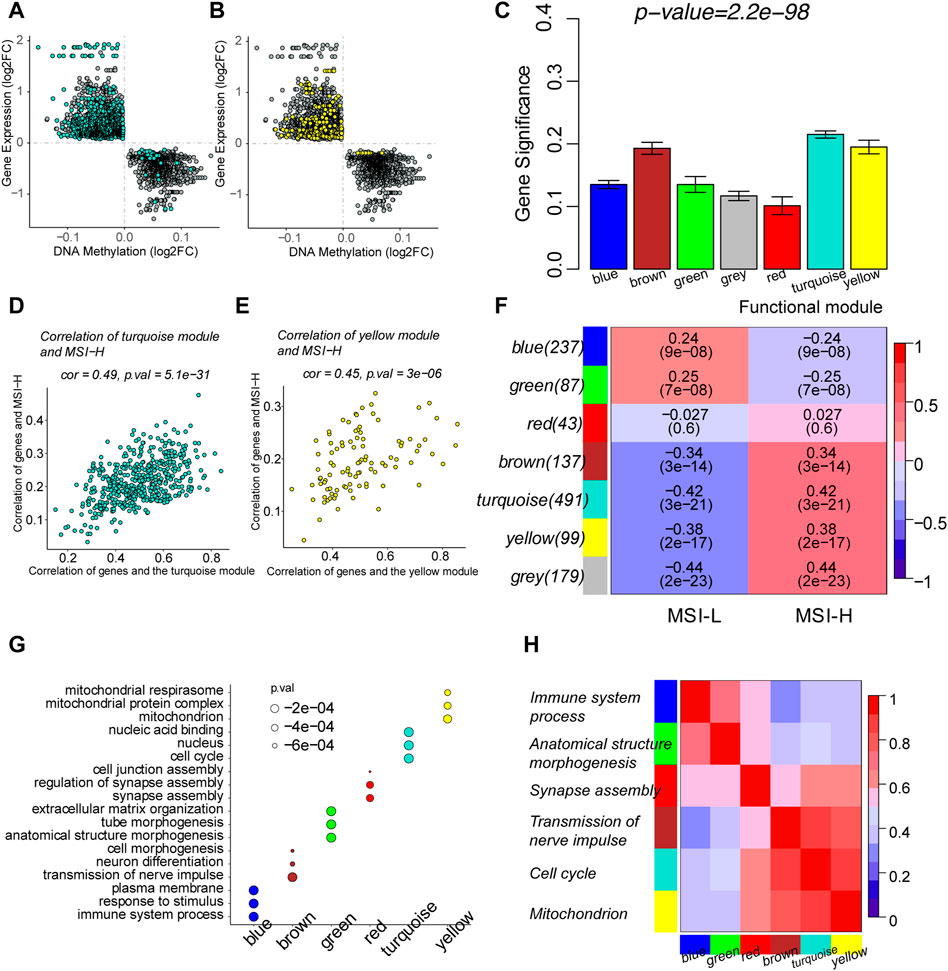
FIGURE 5. Identification of MSI-related functional gene modules by weighted correlation network analysis (WGCNA). (A) Scatterplots of the log2FC of DMPs and DEGs in the turquoise module. (B) Scatterplots of the log2FC of DMPs and DEGs in the yellow module. (C) Barplot of mean gene significance (GS) of each module. GS was defined as the absolute value of the correlation between the gene and the clinical phenotype. Since modules were clusters of functional genes, the mean GS of each module was correlated with GS of each gene in the module. (D) Scatterplots showed the correlation between GS and module membership (MM) in the turquoise module. MM was defined as the correlation between the summary profile of the module and the gene expression. (E) Scatterplots showed the correlation between GS and MM in the yellow module. (F) Relationship between functional modules and MSI phenotypes. Each row corresponded to a module containing functional genes, column to the MSI status. Each cell contains the corresponding correlation and p-value. The table is color-coded by correlation, according to the color legend. (G) Top three enriched biological processes of every module by functional enrichment of genes in each module. (H) Correlation among the functional modules. The top enriched biological process was used to represent the module in the y-axis.
The correlation between modules and MSI was measured by MM and GS. MM indicated the correlation between the defined module and gene expression (Supplementary Table S4). GS represented the correlation between the gene and the clinical phenotype (Supplementary Table S4). The mean GS of each module was determined by the GS of each gene in the module (Figure 5C). The functional modules correlated with MSI were characterized by the high correlation of GS and MM of the genes in the module. Scatterplots of modules depicted the correlation between GS and MM (Figures 5D, E; Supplementary Figures S1E–H). GS and MM of genes in the yellow and turquoise modules are highly correlated with each other, illustrating that the genes in the modules are the central elements associated with the MSI. The association of the module and MSI is shown in the heatmap. The blue and green modules are negatively correlated with the MSI-H phenotype, while the brown, turquoise, and yellow modules were positively correlated with the MSI-H phenotype. A total of 179 genes were classified into the gray module and were thereby removed. Since there was no apparent correlation between the red module and MSI status, genes in the red module were waived for further prognostic analysis (Figure 5F). Functional enrichment was performed to illustrate the biological function of each module (Supplementary Table S5). The turquoise module represented a combination of genes that participated in cell cycle and DNA replication. The yellow module was crucially related to mitochondrion metabolism to provide energy for the biological process. The blue module gathered genes participating in the immune processes (Figures 4G, H).
3.3 Stepwise prognostic analysis of MSI-related genes and the MSI-pRS construction
The TCGA–LUSC cohort was randomly divided into training (n = 314) and validation (n = 158) cohorts for survival analysis. GSE73403 was used as the external validation cohort. The baseline clinicopathological features of the training cohort and validation cohorts are shown in Supplementary Table S6. MSI-related genes in the functional modules were selected to construct the MSI-pRS under the following two steps.
First, LASSO analyses based on the multivariate Cox model were carried out in the functional modules, except for the red module which is defined in the previously described process in the TCGA training cohort (Supplementary Figure S2). As a result, three genes,namely, ribosomal protein S7 (RPS7), cyclin-dependent kinase 20 (CDK20), and LysM domain containing 1 (LYSMD1) in the yellow module and three genes, namely, coiled-coil domain containing 68 (CCDC68), aldehyde dehydrogenase three family member B1 (ALDH3B1), and phosphodiesterase 1B (PDE1B) in the blue module were identified using a LASSO filtering (Supplementary Figures S2A–D). Genes in the turquoise, brown, and green modules failed to construct the model (Supplementary Figures S2E–J).
Second, genes selected in the previous step were brought into two-way stepwise regression to further simplify the model. Ultimately, four MSI-related genes, namely, CCDC68, LYSMD1, RPS7, and CDK20, were used to develop the MSI-pRS model (p-value = 1e-04, Table 1). Univariate Cox analysis indicated that the four genes were prognostic elements. LYSMD1 and RPS7 were protective factors, while CCDC68 and CDK20 were risk factors (Figure 6A). The coefficients obtained from the multivariate Cox regression were utilized as multiplicators, and the MSI-pRS was calculated as follows: MSI-PRS = CCDC68 expression level * 0.29 + LYSMD1 expression level * (−0.57) + RPS7 expression level * (−0.24) + CDK20 expression level * (0.43).
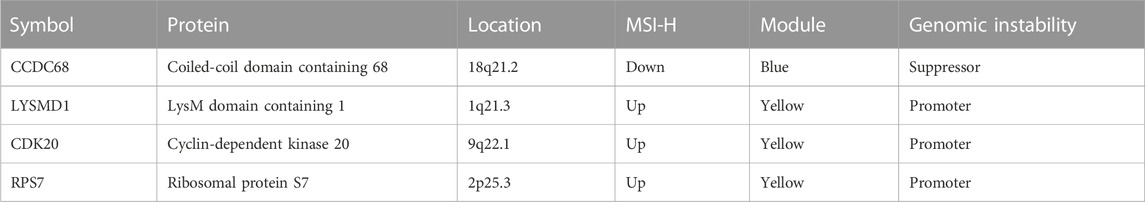
TABLE 1. Details of genes used for the construction of the MSI-related prognostic risk score (MSI-PRS).
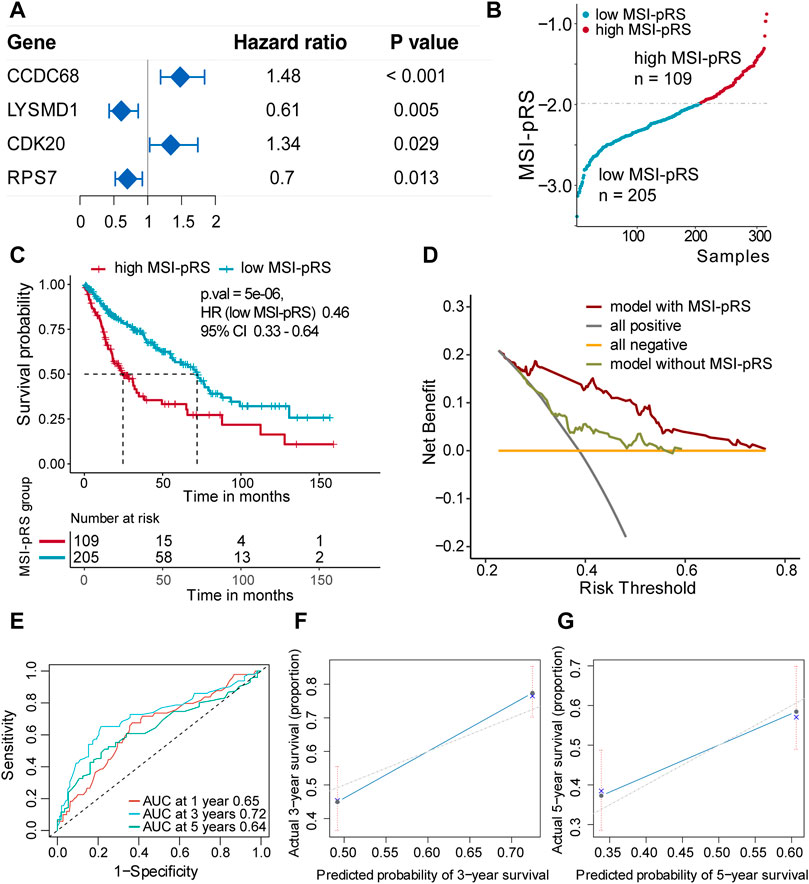
FIGURE 6. Construction of MSI-related prognostic risk score (MSI-pRS) in the TCGA training cohort. (A) Forest plot of prognostic genes used to construct MSI-PRS. Hazard ratio (HR) of each gene was derived from the univariate Cox regression of the four genes. (B) TCGA LUSC training cohort was divided into two groups according to the MSI-pRS. (C) Kaplan-Meier curves of the high MSI-pRS and low MSI-pRS groups in TCGA LUSC training cohort. The overall survival (OS) was used as the primary point. (D) Decision curves of the multivariate Cox model with or without MSI-pRS. The model with MSI-pRS declared a better performance than the model without the MSI-pRS group. (E) Receiver operating characteristic (ROC) curves at years 1, 3, and 5 of the model including age, tumor stage, and MSI-pRS group. (F and G) Calibration curves of the model, including age, tumor stage, and MSI-pRS group.
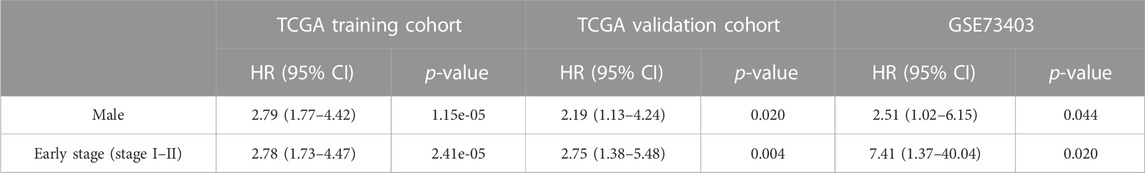
The MSI-pRS was a negative prognostic indicator according to univariate Cox regression in TCGA training, internal validation, and external validation cohorts (HR = 2.72, 95% CI 1.82–4.07, p-value = 1.22e-06; HR = 1.86 95% CI 1.01–3.43, p-value = 0.046; HR = 2.60, 95% CI 1.06–6.40, p-value = 0.037). The exploratory subgroup analyses in the three cohorts were performed (Supplementary Table S7). The low MSI-pRS was a protective factor in the male subgroup and the early stage (Stages I–II), which was consistent in all cohorts (Table 2).
Patients were assigned into two groups according to the MSI-pRS (Figures 6B, 7A, 8A). Low MSI-pRS was a protective prognostic factor in all cohorts (HR = 0.46, p-value = 7.57e-06, Figure 6C; HR = 0.47, p-value = 0.009; Figure 7B; HR = 0.37, p-value = 0.021; Figure 8B). The C-index of the univariate model of the MSI-pRS group was 0.72 (95% CI 0.64–0.80), 0.65 (95% CI 0.51–0.78), and 0.70 (95% CI 0.50–0.90) in the three cohorts. We then brought age, tumor stage, and MSI-pRS group into the multivariate Cox model to explore whether MSI-pRS added an incremental discriminative value to the clinical use. The model with the MSI-pRS was superior to the one without the MSI-pRS in all cohorts (p-value = 4.64−06, 0.008, 0.040). DCA curves of the two models demonstrated that the model with the MSI-pRS achieved better performance in all cohorts (Figures 6D, 7C, 8C).
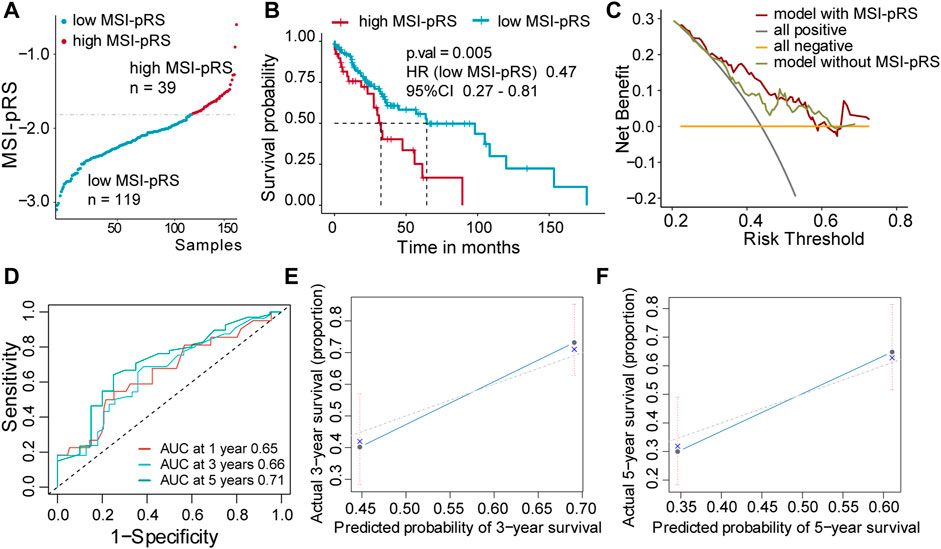
FIGURE 7. Validation of MSI-related prognostic risk score (MSI-pRS) in the internal test cohort (TCGA test cohort). (A) TCGA LUSC test cohort was divided into two groups according to MSI-pRS. (B) Kaplan-Meier curves of the high MSI-pRS and low MSI-pRS groups in TCGA LUSC test cohort. (C) Decision curves of the multivariate Cox model with or without MSI-PRS. The model with MSI-pRS declared a better performance than the model without the MSI-pRS group. (D) Receiver operating characteristic (ROC) curves at years 1, 3, and 5 of the model including age, tumor stage, and MSI-pRS group. (E and F) Calibration curves of the model, including age, tumor stage, and MSI-pRS group.
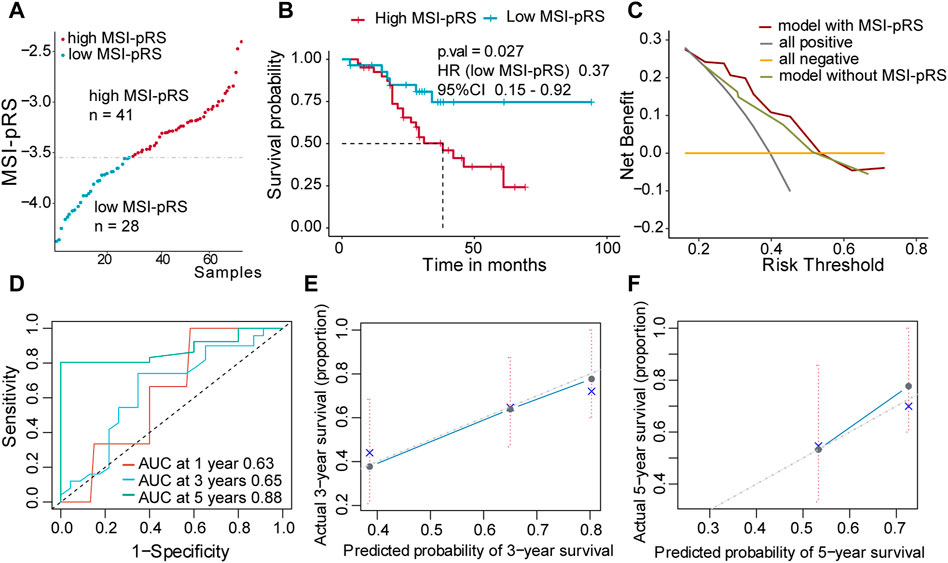
FIGURE 8. Validation of the MSI-related prognostic risk score (MSI-pRS) in the external test cohort (GSE73403 cohort). (A) All patients were divided into MSI-pRS high and MSI-pRS low groups. (B) Kaplan-Meier curves of the MSI-pRS high and MSI-pRS low groups in the external test cohort. (C) Decision curves of the multivariate Cox model with or without MSI-pRS. The model with the MSI-pRS declared a better performance than the model without the MSI-pRS group. (D) Receiver operating characteristic (ROC) curves at years 1, 3, and 5 of the model including age, tumor stage, and MSI-pRS group. (E and F) Calibration curves of the model including age, tumor stage, and MSI-pRS group.
The predictive accuracy of the MSI-pRS in the multivariate Cox model was evaluated by the time-dependent ROC and C-index. The C-index of the multivariate model was 0.64 (95% CI 0.59–0.69), 0.62 (95% CI 0.54–0.69), and 0.65 (95% CI 0.55–0.75) in the three cohorts. The area under the ROC curve (AUC) at 1 year, 3 year, and 5 year of the models with the MSI-pRS was 0.65, 0.72, and 0.64, respectively, in the TCGA training cohort; 0.65, 0.66, and 0.71 in TCGA validation cohort; 0.63, 0.65, and 0.88 in the external validation cohort (Figures 6E, 7D, 8D). The calibration curves showed that the model presents satisfied coherence between the actual survival and predicted survival rates (Figures 6F, G, 7E, F, 8E, F).
3.4 Correlation between the MSI-PRS and MSI status in LUSC
Patients with MSI-H tended to have lower MSI-pRS (Student’s t-test, mean MSI-PRS: −2.18 vs. −2.019, p-value = 2.99e-05, Figure 9A; Supplementary Figure S3A). The MSI-pRS of normal samples was calculated for control. Compared with normal patients, LUSC samples had lower MSI-pRS (ANOVA test, p-value <2.2e-06, Figure 9B). The MSI-pRS was negatively correlated with MSI score. The correlation of the MSI-pRS and MSI score in the MSI-L group was higher than that of the MSI-H group (MSI-L: r = −0.60, 95% CI −0.68∼−0.50, p-value <2.2e-16; MSI-H: r = −0.38, 95% CI −0.48∼−0.27, p-value = 7.266e-11; Figure 9C).
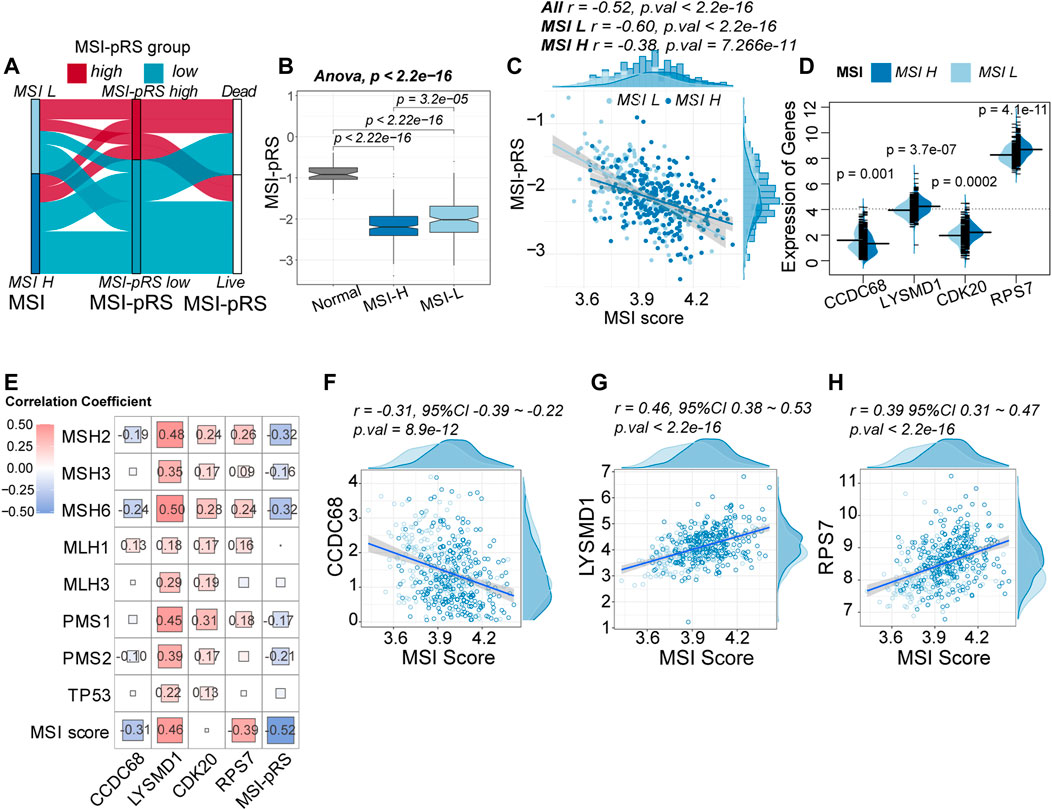
FIGURE 9. Relationship between the MSI-related prognostic risk score (MSI-pRS) and MSI status. (A) Flow diagram depicting the correlation among MSI status, MSI-pRS group, and clinical outcome in the TCGA LUSC training cohort. (B) Comparison of the MSI-pRS among normal samples, MSI-L samples, and MSI-H samples. MSI-pRS was decreased from the normal samples to the MSI-H samples (normal > MSI-L > MSI-H). (C) Correlation between MSI score and MSI-pRS. MSI-pRS was negatively correlated with MSI score. (D) Comparison of the four elements used to construct the MSI-pRS model between MSI-H and MSI-L groups. LYSMD1, CDK20, and RPS7 were higher in the MSI-H group, while CCDC68 was higher in the MSI-L group. (E) Relationship between MMR systems and MSI-pRS. (F) Correlation of CCDC68 and MSI scores. (G) Correlation of LYSMD1 and MSI scores. (H) Correlation of RPS7 and MSI scores.
The expression of the four genes used for constructing the MSI-PRS model was compared between the MSI-H and MSI-L group. Except for CCDC68, the other three genes were upregulated in the MSI-H group (Figure 9D). The correlation between MMR proteins and four genes used to construct the MSI-pRS was analyzed (Figure 9E; Supplementary Figure S3C, D). LYSMD1 was significantly positively related with MMR proteins, especially in the TP53 wt cohort (Supplementary Figure S3C, D). The same trend was also observed in CDK20. Despite the positive correlation between CDK20 and MMR proteins, the correlation between CDK20 and MSI score was weak. LYSMD1 and RPS7 were positively correlated with MSI score, whereas CCDC68 was negatively correlated with MSI score (Figures 9F–H).
3.5 Low MSI-pRS related to TP53 mutation and DNA hypomethylation of TP53
TP53 mut tended to have lower MSI-pRS (Student’s t-test, p-value = 4.4e-07, Figure 10A). TP53 had a higher mutation rate in the MSI-pRS low group in the training and internal validation cohorts (chi-square test; p-value = 0.0004, 0.008; Figure 10B; Supplementary Figure S4A). The comparison of four genes between the TP53 mut and TP53 wt groups was performed. The expression of LYSMD1 was significantly higher in the TP53 mut group, whereas those of the other three genes showed no difference (Mann–Whitney’s U-test, p-value = 3.313e-06; Figure 10C).
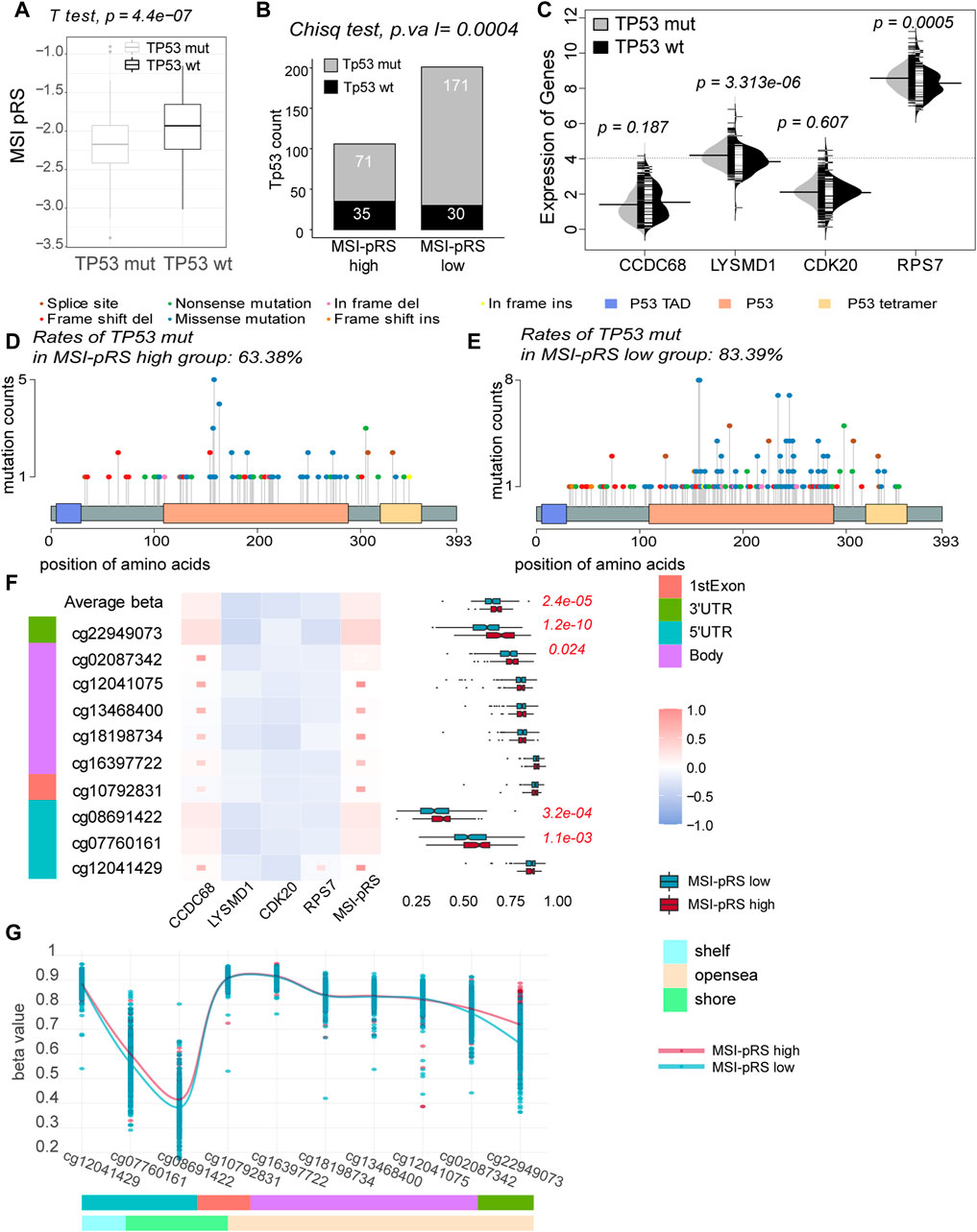
FIGURE 10. Relationship between the MSI-related prognostic risk score (MSI-pRS) and TP53 status. (A) TP53 mut had lower MSI-pRS than the TP53 wt. (B) Incidence of TP53 mutations was more frequent in the low MSI-pRS group in the TCGA training cohort. (C) Comparison of the four elements used to construct the MSI-pRS model between TP53 mut and TP53 wt groups. LYSMD1 and RPS7 were higher in the TP53 mut group. (D) Landscape of TP53 mutations in the MSI-pRS high group. (E) Landscape of TP53 mutations in the MSI-pRS low group. (F) Correlation of the MSI-PRS and its elements with DNA methylation probes on TP53 genes. MSI-pRS and CCDC68 were positively correlated with TP53 DNA methylation, whereas LYSMD1, CDK20, and RPS7 were negatively correlated with TP53 DNA methylation. (G) Comparison of DNA methylation on TP53 genes between MSI-pRS high and low groups in TCGA LUSC cohort.
Transactivation domains (TADs) mediate the transcriptional activity. A total of three and nine mutations on TADs were, respectively, observed in the MSI-pRS high and MSI-pRS low groups. There was no difference in the mutations of TADs in the two groups. The DNA-binding domain (DBD) enables p53 protein sequence-specific binding to DNA with a highly conserved structure. Mutations of TP53 concentrated on the DBD. Missense mutation was the most common mutation type in both groups (chi-square test; MSI-pRS high: 54.1%, MSI-pRS low: 64.5%; p-value = 0.083; Figures 10D, E). The frequent mutations at six hotspots, i.e., codons R175, G245, R248, R249, R273, and R282, were 9.1% in the MSI-pRS high group while 16.4% in MSI-pRS low group (Chi-square test; p-value = 0.064; Figures 10D, E).
DNA hypomethylation commonly removes suppression of genes. The average beta value of DNA methylation on TP53 genes was lower in the MSI-pRS low group (Mann–Whitney’s U-test, p-value = 2.4e-05), especially that on the promoters of TP53 (cg12041429, cg07760161, cg08691422, and cg10792831, Mann–Whitney’s U-test, p-value = 2.0e-04, Figures 10F, G). The expression of TP53 was not significantly different in the MSI-pRS high and -low groups (Mann–Whitney’s U-test; p-value = 0.151). MSI-PRS and CCDC68 were positively correlated with DNA methylation of TP53, while LYSMD1, CDK20, and RPS7 were negatively correlated with DNA methylation of TP53.
3.6 Low MSI-PRS associated with genomic instability
Chromosome 3p alternations are an acknowledged feature of LUSC, with chromosome 3p loss and 3q amplification involved in the tumorigenesis of LUSC. The MSI-pRS was lower in the 3p deletion and 3q amplification group (Student’s t-test; 3p loss vs. 3p normal: −2.15 vs. −1.85, p-value = 0.0003; 3q amp vs. 3q normal: −2.11 vs. −1.89, p-value = 0.002; Figure 11A, B). The MSI-pRS low group had higher TMB, MATH, and aneuploidy scores (Mann–Whitney’s U-test; p-value = 0.0004, 0.019, 0.015; Supplementary Figures S4B–D). The MSI-pRS high group had higher subclonal genome fraction (Mann–Whitney’s U-test; p-value = 0.006; Supplementary Figures S4E). DNA content had no significant difference between MSI-pRS groups (Supplementary Figures S4F). LYSMD1 and RPS7 were higher in the 3p loss and 3q amplification groups than in the chromosome 3 normal samples, whereas CCDC68 was higher in the chromosome normal samples (Mann–Whitney’s U-test; Figures 11C, D).
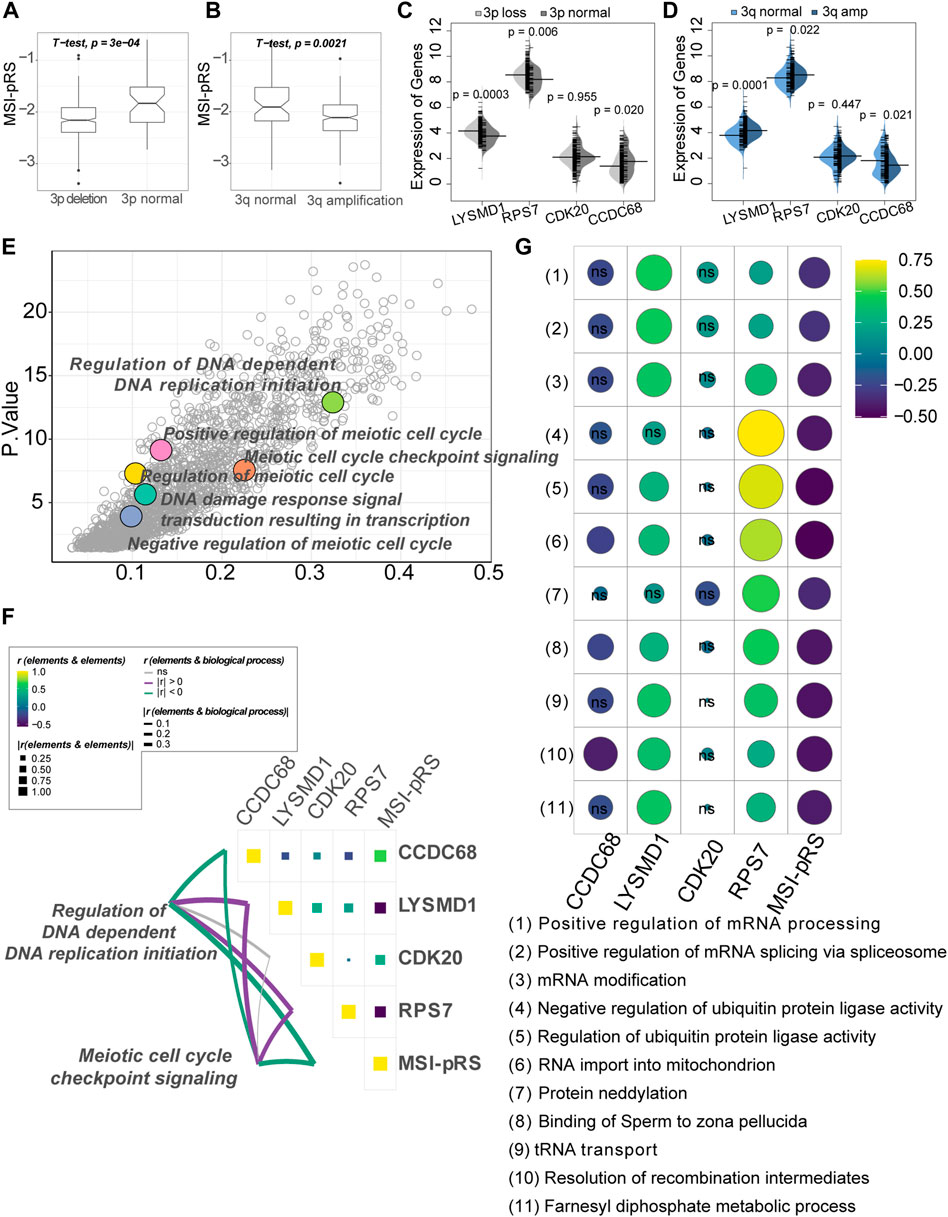
FIGURE 11. Genomic feature exploration of the MSI-related prognostic risk score (MSI-PRS). (A) MSI-pRS was lower in the chromosome 3p deletion LUSC. (B) MSI-pRS was lower in the chromosome 3q amplification LUSC. (C) Comparison of the four elements used to construct the MSI-pRS model between 3p loss and 3p normal groups. LYSMD1 and RPS7 were higher in the 3p loss group, while CCDC68 was lower in the 3p loss group. (D) Comparison of the four elements used to construct the MSI-pRS model between 3q amplification and 3q normal groups. LYSMD1 and RPS7 were higher in the 3q amplification group, while CCDC68 was lower in the 3q amplification group. (E) DNA replication, DNA damage response, and cell cycle checkpoint were upregulated in the MSI-pRS low group. Low MSI-pRS was associated with high genomic instability with active DNA damage repair response, whereas high MSI-pRS was vice versa. (G) Correlation of MSI-pRS and the transcriptional/ translational control associated biological processes. High MSI-pRS was negatively associated with transcriptional/translational control. LYSMD1 and RPS7 were positively correlated with the previously described processes. (F) Correlation of the MSI-pRS and its four elements. LYSMD1 and RPS7 were negatively correlated with the MSI-pRS, while CCDC68 and CDK20 were positively correlated with the MSI-pRS. Correlation of the MSI-pRS and four elements and DNA replication and cell cycle checkpoint processes. MSI-pRS and CCDC68 negatively related to the processes, whereas LYSMD1 and RPS7 were positively correlated.
Functional exploration was performed in the TCGA–LUSC cohort and GSE73403 validation cohort using the GO BP dataset by GSVA, according to the MSI-pRS group. Results obtained from the three cohorts (the training cohort, internal test cohort, and external test cohort) are intersected to obtain the final functional enrichment results. DNA-dependent DNA replication initiation and DNA damage response signal transduction resulting in transcription were upregulated in the MSI-pRS low group (Figure 11E). In particular, a number of meiotic cell cycle-related processes were upregulated in the MSI-pRS low group in all cohorts.
Biological processes that highly correlated with MSI-pRS and the expression of genes used to construct the MSI-pRS were explored. MSI-pRS was negatively correlated with meiotic cell cycle checkpoint signaling and DNA-dependent DNA replication initiation (Figure 11F; Supplementary Figure S5A). Moreover, MSI-pRS was negatively correlated with transcriptional and translational control, including mRNA and protein modification (Figure 11F; Supplementary Figures S5B).
Among the four genes comprising MSI-PRS, LYSMD1 and RPS7 were negatively related to DNA replication initiation and meiotic cell cycle checkpoint signaling and positively correlated with the transcriptional control (Figures 11F, G; Supplementary Figures S5B, C). LYSMD1 was highly correlated with the mRNA modification in the TCGA–LUSC cohort and the GSE73403 cohort, while RPS7 was tightly correlated with ubiquitin protein ligase activity in all cohorts (Figures 11F, G; Supplementary Figures S5B). CCDC68 was negatively correlated with the DNA replication initiation process and meiotic cell cycle checkpoint signaling but had a weak relationship with transcriptional and translational control processes. Although the expression of CDK20 was weakly correlated with all explored functional processes, it was the critical elements that composed the MSI-pRS (TCGA-LUSC: r = 0.38, p-value < 2.2e-16; GSE73403: r = 0.52, p-value = 4.2e-06; Figures 11F, G; Supplementary Figures S5A, B).
3.7 High MSI-pRS was characterized with an inflamed TME
We then explored the immune features of the MSI-pRS group. CYT represented the ultimate anti-tumoral cytotoxicity and was higher in the MSI-pRS high group in all cohorts (Mann–Whitney’s U-test; p-value = 0.021, 0.023, 0.033; Figure 12A; Supplementary Figures S6A, B), indicating a stronger immune response in the MSI-pRS high group. Tumors with high MSI-pRS were infiltrated by a higher abundance of immune cells, including both positive immune executors and negative immune regulators in all cohorts (Figure 12B; Supplementary Figure S6C). Functional enrichment by GSVA showed that T cell activation, especially through the major histocompatibility complex (MHC) II class process, was upregulated in the MSI-pRS high group, while processes involved in protein translation were downregulated in all cohorts (Figure 12C). Moreover, blood vessel remodeling and maturation were upregulated in the MSI-pRS high group (Figure 12C). CCDC68 was positively correlated with T cell activation, while LYSMD1 and RPS7 were negatively correlated (Figure 12D, Supplementary Figure S6D).
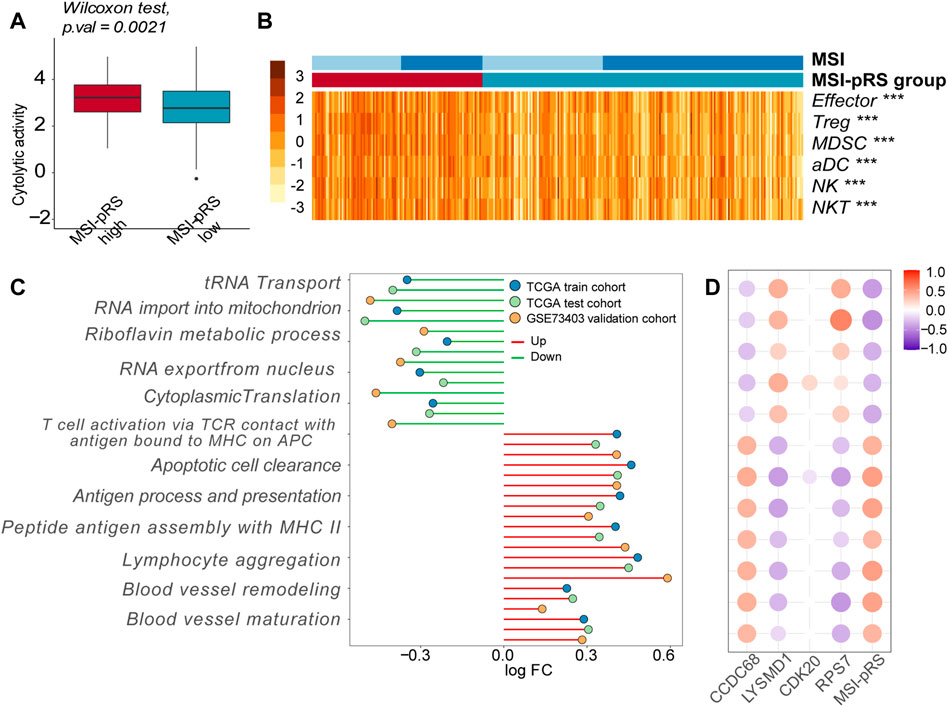
FIGURE 12. Immune features exploration of the MSI-related prognostic risk score (MSI-pRS). (A) Cytolytic activity (CYT) was higher in the MSI-pRS high group in the training cohort. (B) Heatmap of immune cells in tumor microenvironment in the training cohort. (C) Lollipop plot of the altered biological processes concentrating on the immune process and cell cycle, according to the MSI-pRS group in the three cohorts. (D) Correlation plot of the four genes in the MSI-pRS model and the altered biological processes in the TCGA training cohort. Abbreviation: LUSC, lung squamous cell carcinoma; MSI, microsatellite instability; MSI-pRS, MSI-related prognostic risk score; ICIs, immune checkpoint inhibitors; CYT, cytolytic activity; TME, tumor microenvironment; TP53, tumor protein p53; PD-L1, programmed death-ligand 1; MLH, MutL homologs; MSH, MutS homologs; TCGA, The Cancer Genome Atlas; GEO, Gene Expression Omnibus; GSEA, gene set enrichment analysis; WGCNA, weighted correlation network analysis; GO, Gene Ontology; OS, overall survival time; PFS, progression-free survival; DCA, decision curve analyses; HR, hazard ratios; ROC, receiver operating characteristic; AUC, area under the ROC curve; DEGs, differentially expressed genes; DMPs, different methylation probes; MHC, major histocompatibility complex; CCDC68, coiled-coil domain containing 68; RPS7, ribosomal protein S7; CDK20, cyclin-dependent kinase 20; and LYSMD1, LysM domain containing 1.
4 Discussion
Lung squamous cell carcinoma comprises approximately 30% of NSCLC with a high rate of protein-altering mutations (Gerber et al., 2015). In contrast to better-known driver alterations in adenocarcinoma, such as EGFR, KRAS, and ALK, passenger mutations seem to contribute to the high somatic mutation rate in LUSC (Perez-Moreno et al., 2012). Passenger mutations dynamically accumulate under the background alternations of oncological gene drivers on the upstream of the cell cycle, such as TP53, for which target therapy is always invalid in LUSC. Based on the intricate genomic features of LUSC, the study explored the genome instability of LUSC and established a prognostic signature associated with it.
MSI is the molecular feature of the cancers with MMR deficiency. Ensuring high-fidelity DNA replication is essential for maintaining genome stability. The newly synthesized strand containing mismatches that have escaped proofreading by excision followed by resynthesis and ligation during DNA replication are corrected by MMR with the help of DNA replicative polymerase and DNA ligase (Hsieh and Zhang, 2017). In addition to its roles in editing replication errors, the MMR system also triggers cell cycle arrest and apoptosis in DDR. Loss of MMR results in inherited cancer susceptibility, such as Lynch syndrome, as well as an increased incidence of sporadic cancers (Knijnenburg et al., 2018; Xiao et al., 2021).
The major alternation leading to MMR-d is DNA hypermethylation in the MLH1. DNA hypermethylation or mutations (single-nucleotide polymorphism, deletion, etc) of MSH2 and other MMR genes may explain a portion of the silencing of MMR proteins and the fellow molecules. It was reported that the methylation rate of the MLH1 promoter CpG islands was 72.9% in gastric cancer and 89% in endometrial cancer. When it comes to NSCLC, the methylation rate of MLH1 was reported as 27%–35.7%, whereas the incidence of MSI-H was rare (Seng et al., 2008; Pastuszak-Lewandoska et al., 2016). The previously described phenomena indicated that epigenetical markers of MMR proteins were not consistent with MSI status and genomic instability. Generally, despite the complexity of the LUSC genome with high mutation rates, MSI is not commonplace with the incidence of less than 1% (Xiao et al., 2014; Yanagawa et al., 2021). We proposed that MSI status potentially performs its roles in genomic instability with other mechanisms.
The MSI phenotypes were defined in the LUSC by the unsupervised classification. TCGA–LUSC data were divided into MSI-H and MSI-L groups, according to MMR protein expression. The MSI status of each sample was identified by MSI score based on the expression of MMR proteins. Compared with normal samples, LUSC samples had higher MSI score, and patients with an MSI-H phenotype had higher MSI score. The MSI-H phenotype was characterized by high TMB, DNA content, and rate of aneuploidy. In addition, TP53 was at a high rate of aberrations in the MSI-H cluster. MSI-H tumors displayed high genomic instability, which was tightly associated with the clonally expanded mutations in cancer driver genes and led to tumor heterogeneity. The MSI status defined by MMR proteins in this study revealed the severity of genomic instability in LUSC.
The MSI status was relatively clustered according to MMR proteins in the dataset and discriminated the genomic features of LUSC in some respects. The MSI-pRS defined the clinical outcome of LUSC patients based on the MSI status. The MSI-pRS was composed of CCDC68, LYSMD1, RPS7, and CDK20 and was negatively correlated with the MSI score. Compared with tumor tissues, the MSI-pRS was higher in normal tissues. The MSI-pRS of the MSI-H group was lower than that of the MSI-L group. The MSI-pRS was a negative prognostic indicator with good discrimination and calibration. Patients with low MSI-pRS tended to have an improved OS. DCA curves suggested that the model added extra value to the prognosis not only in the training cohort but also in the internal and external test cohorts.
The association of MSI-pRS and MSI status was explored. Low MSI-pRS was associated with high genomic instability with high tumor heterogeneity and TMB. The TP53 gene encodes the p53 protein which acts as the guardian of the genome to preserve genomic integrity (Marei et al., 2021). The MSI-pRS low group had a higher incidence of TP53 mutation and DNA hypomethylation of TP53. Aneuploidy is defined as the unbalanced number of chromosomes and is a salient feature of cancer genomes. Chromosome 3 alterations including 3p loss and 3q amplification participated in the tumorigenesis of LUSC. The incidence of aneuploidy was detected more frequently in the MSI-pRS low group. Moreover, LUSC with 3p loss or 3q amplification had a higher MSI-pRS. Functional enrichment declared that low MSI-pRS was associated with the upregulation of a DNA damage response signal, DNA replication initiation, cell cycle checkpoint signaling, and transcriptional and translational control. Past studies confirmed that MSI-H is associated with better survival in colorectal cancers but is inconclusive in other cancers. Low MSI-pRS was characterized by genomic instability and was associated with better clinical outcomes (Popat et al., 2005). The association of genomic stability and clinical outcomes found in the study was consistent with that of the previous study. The MSI-pRS shows promise to be an optional prognostic genomic biomarker in LUSC as the substitute of MSI.
Tumors with high MSI-pRS are displayed as an inflamed immune phenotype with acquired immune escape. Both immune effectors (active T cells and effect memorial T cells) and immune regulators (Tregs and MDSCs) were also highly infiltrated in the MSI-pRS high group. Functional enrichment indicated that processes associated with antigen presentation by MHC II were upregulated. Moreover, blood vessel remodeling and maturation might contribute to high-grade malignancy. The MSI-pRS high group was characterized by an inflamed immunophenotype, while the MSI-pRS low group was characterized by a cold one. Patients with high MSI-pRS might be the potential subpopulation that derives profit from ICIs.
The elements of MSI-pRS were explored. CCDC68 was positively correlated with MSI-pRS and is a tumor-suppressive gene by reducing cell proliferation and enhancing apoptosis, which is strongly expressed in the lung cancer tissues (Hua et al., 2020). The gene predicted short OS in LUSC patients. MSI-H, 3p loss, and 3q amp samples tended to have lower CCDC68. The gene was negatively correlated with DNA replication initiation and cell cycle checkpoint, but positively correlated with tumorigenesis-associated immune response and tumorous vessel modification. The findings declared that CCDC68 had a suppressive role in the genomic instability and a promoting role in the inflamed TME.
LYSMD1 encodes a highly conservative receptor containing a lysin motif domain, the role of which in cancerogenesis has not been elucidated so far. In the study, we first declared that LYSMD1 was a promoter of genomic instability. LYSMD1 was a positive prognostic biomarker and was negatively correlated with MSI-pRS. LUSC samples with MSI-H, TP53 mut, 3p loss, and 3q amplification were more likely to have a higher expression of LYSMD1. The gene was positively correlated with MMR proteins and participated in active DNA replication and transcription. Nevertheless, LYSMD1 was negatively correlated with anti-cancer immunity, which might contribute to cold TME and immunosuppression.
RPS7 encodes a component of the small 40 S subunit of the ribosome, which is a critical performer in protein translation (Wu et al., 2021). The gene was a positive prognostic biomarker and negatively correlated with MSI-pRS. RPS7 is upregulated in the MSI-H, TP53 mut, 3p loss, and 3q amplification LUSC samples. The gene also contributed to active DNA replication, transcription, and translation, especially in the activity of ubiquitin ligase in the post-translational modification. RPS7 might contribute to the cold TME with the negative correlation with immune cells in the TME.
CDK20 was once reported as a promoter of the G1/S transition and a regulator of G0/G1 checkpoint. Overexpression of CDK20 promotes proliferation and is regarded as a tumorigenesis-related factor in many cancers (Lai et al., 2020). CDK20 was a risk factor of clinical outcomes and positively correlated with MSI-pRS. The MSI-pRS was higher in the MSI-H and was positively correlated with the expression of MMR proteins, especially in the TP53 wt group. However, TP53 mut, 3p loss, and 3q amplification LUSC samples had no difference in CDK20 compared with the wild-type samples.
There were some limitations in the study. First, the MSI status defined in this study was a relative concept for it was classified based on the expression of MMR proteins in the TCGA–LUSC dataset. As it was declared before, MSI-H in LUSC represents a type of genetic instability. We observed the higher expression of MMR proteins in the MSI-H samples compared with the normal and MSI-L samples, which was different from that seen in colorectal cancer. The consistent trend of DNA methylation on the promoters and expression was observed in MSH3 and MSH6. MMR proteins might be activated by the genomic instability in the MSI-H group. So far, the most widely used MSI detection method was polymerase chain reaction (PCR) amplification of microsatellite markers using different panels available comprising a combination of mononucleotide and dinucleotide repeats. Tumors with instability at two or more of these markers were defined as being MSI-H, whereas those with instability at one repeat or showing no instability were defined as MSI-L tumors (Buhard et al., 2006). Evaluating the expression of the MMR proteins by immunohistochemistry (IHC) on histological tissue sections has been regarded as a valid surrogate to identify tumors with a higher probability of instability. The two methods also involved a discordance because MSI also resulted from other mechanisms (Cherri et al., 2022). The two methods are inappropriate for the cancers with low incidence of MSI and MMR dysfunction. With the widespread use of next-generation sequencing (NGS), it has been an alternative molecular test for assessing MSI and other genomic features, such as TMB and MATH, as well as revealing the molecular mechanisms leading to genomic instability. A panel of MSI-associated gene expression of tumors based on the transcriptomic data might identify the MSI status of cancer with low incidence of MSI (Li et al., 2020). Therefore, we suggested that MSI status in LUSC could be distinguished according to the expression of MMR.
Second, MSI-H tumors in the study were characterized by high DNA ploidy and aneuploidy scores, which were commonly derived from CIN. Generally, CIN represents abbreviations in DNA content and is mutually exclusive with MSI (Yoshioka et al., 2019). Both CIN and MSI result from genomic instability and derive from replication stress-associated DNA double-strand breaks (DSBs). CIN develops when DSBs are not effectively repaired by homologous recombination under the circumstance of an MMR proficient background (Matsuno et al., 2019). MSI and hypermutation generate by erroneously repaired DSBs when MMR systems are deficient concurrently. Based on the previously described processes, we assumed that MMR proteins acted as regulators of genomic instability in LUSC, which is reflected by MSI status clustered by expression of MMR proteins.
Furthermore, the MSI-pRS was constructed with an aim of excavating the meaning of MSI in LUSC profoundly. More prospective cohort studies are needed to estimate the clinical significance of the MSI-pRS signature, which might be accomplished by direct immunohistochemistry or gene sequencing of tissues. In the study, we proposed bioinformatics evidence for the correlation between the MSI-pRS and immune phenotype and inferred that LUSC patients with high MSI-pRS could derive benefits from ICIs and the combination of ICIs and angiogenetic therapy. However, the molecular mechanism needs more experimental verification.
5 Conclusion
In conclusion, we performed an integrative analysis to explore the MSI status in LUSC. Four hub MSI-related genes, namely, CCDC68, LYSMD1, RPS7, and CDK20, were identified and used to establish a prognostic score related to genomic instability named MSI-pRS in LUSC. Low MSI-pRS predicted better OS. LUSC with low MSI-pRS was associated with increased genomic instability and cold immunophenotype. MSI-pRS is a promising prognostic biomarker in LUSC as the substitute of MSI. In addition, we first declared the promotive role of LYSMD1 in genomic instability of LUSC. Our findings provided new insights in the biomarker finder of LUSC.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material; further inquiries can be directed to the corresponding author.
Ethics statement
The study was carried out with the ethical approval of the institutional Ethics Committee of the Faculty of Medicine at China–Japan Friendship Hospital (2021-122-K80). Publicly available data were used in the article, so the ethical approval was waived.
Author contributions
ZH and HC contributed to the idea and design. ZH, XL, and JL help in the research and download of the data. ZH and ZL contributed to data excavation. ZH and HC accomplished the writing and revision of the manuscript. JZ and YP provided writing guidance to the manuscript. All authors have read and approved the submitted version.
Funding
This work was supported by the Capital Health Development Scientific Research Project: Combination of Gegen Qinlian tablets with immune checkpoint inhibitors and chemotherapy in advanced lung squamous cell carcinoma: a clinical study (2022-2-4065).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors, and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgene.2023.1061002/full#supplementary-material
Supplementary Figure S1 | Genes in the blue, green, brown and red modules.
Supplementary Figure S2 | The process of Least shrinkage and selection operator.
Supplementary Figure S3 | Relationship between MSI-related prognostic risk score (MSI-pRS) and MSi status.
Supplementary Figure S4 | Genomic feature exploration of MSI-related prognostic risk score (MSI-pRS).
Supplementary Figure S5 | Functional enrichment in the GSE73403 external validation cohort.
Supplementary Figure S6 | Immune features of MSI-pRS high and MSI-pRS low group in the TCGA-LUSC internal validation cohort and the GSE73403 external validation cohort.
References
Aryee, M. J., Jaffe, A. E., Corrada-Bravo, H., Ladd-Acosta, C., Feinberg, A. P., Hansen, K. D., et al. (2014). Minfi: A flexible and comprehensive bioconductor package for the analysis of Infinium DNA methylation microarrays. Bioinformatics 30 (10), 1363–1369. doi:10.1093/bioinformatics/btu049
Blanche, P., Dartigues, J. F., and Jacqmin-Gadda, H. (2013). Estimating and comparing time-dependent areas under receiver operating characteristic curves for censored event times with competing risks. Stat. Med. 32 (30), 5381–5397. doi:10.1002/sim.5958
Buhard, O., Cattaneo, F., Wong, Y. F., Yim, S. F., Friedman, E., Flejou, J. F., et al. (2006). Multipopulation analysis of polymorphisms in five mononucleotide repeats used to determine the microsatellite instability status of human tumors. J. Clin. Oncol. 24 (2), 241–251. doi:10.1200/JCO.2005.02.7227
Cancer Genome Atlas Research Network (2012). Comprehensive genomic characterization of squamous cell lung cancersRogers, Kristen [corrected to Rodgers, Kristen. Nature 489 (7417), 491288519–491288525. doi:10.1038/nature11404
Carter, S. L., Cibulskis, K., Helman, E., McKenna, A., Shen, H., Zack, T., et al. (2012). Absolute quantification of somatic DNA alterations in human cancer. Nat. Biotechnol. 30 (5), 413–421. doi:10.1038/nbt.2203
Chae, Y. K., Anker, J. F., Oh, M. S., Bais, P., Namburi, S., Agte, S., et al. (2019). Mutations in DNA repair genes are associated with increased neoantigen burden and a distinct immunophenotype in lung squamous cell carcinoma. Sci. Rep. 9 (1), 3235. Published 2019 Mar 1. doi:10.1038/s41598-019-39594-4
Chalmers, Z. R., Connelly, C. F., Fabrizio, D., Gay, L., Ali, S. M., Ennis, R., et al. (2017). Analysis of 100,000 human cancer genomes reveals the landscape of tumor mutational burden. Genome Med. 9 (1), 34. doi:10.1186/s13073-017-0424-2
Charoentong, P., Finotello, F., Angelova, M., Mayer, C., Efremova, M., Rieder, D., et al. (2017). Pan-cancer immunogenomic analyses reveal genotype-immunophenotype relationships and predictors of response to checkpoint blockade. Cell Rep. 18 (1), 248–262. doi:10.1016/j.celrep.2016.12.019
Chen, B., Li, R., Zhang, J., Xu, L., and Jiang, F. (2022). Genomic landscape of metastatic lymph nodes and primary tumors in non-small-cell lung cancer. Pathol. Oncol. Res. 28, 1610020. Published 2022 Jun 16. doi:10.3389/pore.2022.1610020
Chen, H., Yao, J., Bao, R., Dong, Y., Zhang, T., Du, Y., et al. (2021). Cross-talk of four types of RNA modification writers defines tumor microenvironment and pharmacogenomic landscape in colorectal cancer. Mol. Cancer 20 (1), 29. Published 2021 Feb 8. doi:10.1186/s12943-021-01322-w
Cherri, S., Oneda, E., Noventa, S., Melocchi, L., and Zaniboni, A. (2022). Microsatellite instability and chemosensitivity in solid tumours. Ther. Adv. Med. Oncol. 14 (17588359221099347), 17588359221099347. Published 2022 May 21. doi:10.1177/17588359221099347
De Marchi, P., Berardinelli, G. N., Cavagna, R. O., Pinto, I. A., da Silva, F. A. F., Duval da Silva, V., et al. (2022). Microsatellite instability is rare in the admixed Brazilian population of non-small cell lung cancer: A cohort of 526 cases. Pathobiology 89 (2), 101–106. doi:10.1159/000520023
Gerber, D. E., Paik, P. K., and Dowlati, A. (2015). Beyond adenocarcinoma: Current treatments and future directions for squamous, small cell, and rare lung cancer histologies. Am. Soc. Clin. Oncol. Educ. Book, 147–162. doi:10.14694/EdBook_AM.2015.35.147
Gu, X., Chu, L., and Kang, Y. (2022). Angiogenic factor-based signature predicts prognosis and immunotherapy response in non-small-cell lung cancer. Front. Genet. 13, 894024. Published 2022 May 18. doi:10.3389/fgene.2022.894024
Hänzelmann, S., Castelo, R., and Guinney, J. (2013). Gsva: Gene set variation analysis for microarray and RNA-seq data. BMC Bioinforma. 14, 7. doi:10.1186/1471-2105-14-7
Heist, R. S., Sequist, L. V., and Engelman, J. A. (2012). Genetic changes in squamous cell lung cancer: A review. J. Thorac. Oncol. 7 (5), 924–933. doi:10.1097/JTO.0b013e31824cc334
Hsieh, P., and Zhang, Y. (2017). The Devil is in the details for DNA mismatch repair. Proc. Natl. Acad. Sci. U. S. A. 114 (14), 3552–3554. doi:10.1073/pnas.1702747114
Hua, T., Ding, J., Xu, J., Fan, Y., Liu, Z., and Lian, J. (2020). Coiled-coil domain-containing 68 promotes non-small cell lung cancer cell proliferation in vitro. Oncol. Lett. 20 (6), 356. doi:10.3892/ol.2020.12220
Kim, J. Y., Choi, J. K., and Jung, H. (2020). Genome-wide methylation patterns predict clinical benefit of immunotherapy in lung cancer. Clin. Epigenetics 12 (1), 119. Published 2020 Aug 6. doi:10.1186/s13148-020-00907-4
Knijnenburg, T. A., Wang, L., Zimmermann, M. T., Chambwe, N., Gao, G. F., Cherniack, A. D., et al. (2018). Genomic and molecular landscape of DNA damage repair deficiency across the cancer genome Atlas. Cell Rep. 23 (1), 239–254.e6. doi:10.1016/j.celrep.2018.03.076
Kuleshov, M. V., Jones, M. R., Rouillard, A. D., Fernandez, N. F., Duan, Q., Wang, Z., et al. (2016). Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44 (W1), W90–W97. doi:10.1093/nar/gkw377
Lai, L., Shin, G. Y., and Qiu, H. (2020). The role of cell cycle regulators in cell survival-dual functions of cyclin-dependent kinase 20 and p21Cip1/waf1. Int. J. Mol. Sci. 21 (22), 8504. Published 2020 Nov 12. doi:10.3390/ijms21228504
Langfelder, P., and Horvath, S. (2008). Wgcna: an R package for weighted correlation network analysis. BMC Bioinforma. 9, 559. Published 2008 Dec 29. doi:10.1186/1471-2105-9-559
Li, L., Feng, Q., and Wang, X. (2020). PreMSIm: An R package for predicting microsatellite instability from the expression profiling of a gene panel in cancer. Comput. Struct. Biotechnol. J. 18, 668–675. Published 2020 Mar 19. doi:10.1016/j.csbj.2020.03.007
Mandal, R., Samstein, R. M., Lee, K. W., Havel, J. J., Wang, H., Krishna, C., et al. (2019). Genetic diversity of tumors with mismatch repair deficiency influences anti-PD-1 immunotherapy response. Science 364 (6439), 485–491. doi:10.1126/science.aau0447
Marei, H. E., Althani, A., Afifi, N., Hasan, A., Caceci, T., Pozzoli, G., et al. (2021). p53 signaling in cancer progression and therapy. Cancer Cell Int. 21 (1), 703. Published 2021 Dec 24. doi:10.1186/s12935-021-02396-8
Matsuno, Y., Atsumi, Y., Shimizu, A., Katayama, K., Fujimori, H., Hyodo, M., et al. (2019). Replication stress triggers microsatellite destabilization and hypermutation leading to clonal expansion in vitro. Nat. Commun. 10 (1), 3925. Published 2019 Sep 2. doi:10.1038/s41467-019-11760-2
Mayakonda, A., Lin, D. C., Assenov, Y., Plass, C., and Koeffler, H. P. (2018). Maftools: Efficient and comprehensive analysis of somatic variants in cancer. Genome Res. 28 (11), 1747–1756. doi:10.1101/gr.239244.118
Pastuszak-Lewandoska, D., Kordiak, J., Antczak, A., Migdalska-Sek, M., Czarnecka, K. H., Gorski, P., et al. (2016). Expression level and methylation status of three tumor suppressor genes, DLEC1, ITGA9 and MLH1, in non-small cell lung cancer. Med. Oncol. 33 (7), 75. doi:10.1007/s12032-016-0791-3
Perez-Moreno, P., Brambilla, E., Thomas, R., and Soria, J. C. (2012). Squamous cell carcinoma of the lung: Molecular subtypes and therapeutic opportunities. Clin. Cancer Res. 18 (9), 2443–2451. doi:10.1158/1078-0432.CCR-11-2370
Popat, S., Hubner, R., and Houlston, R. S. (2005). Systematic review of microsatellite instability and colorectal cancer prognosis. J. Clin. Oncol. 23 (3), 609–618. doi:10.1200/JCO.2005.01.086
Ritchie, M. E., Phipson, B., Wu, D., Hu, Y., Law, C. W., Shi, W., et al. (2015). Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43 (7), e47. doi:10.1093/nar/gkv007
Rooney, M. S., Shukla, S. A., Wu, C. J., Getz, G., and Hacohen, N. (2015). Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell 160 (1-2), 48–61. doi:10.1016/j.cell.2014.12.033
Santos, E. S., and Rodriguez, E. (2022). Treatment considerations for patients with advanced squamous cell carcinoma of the lung. Clin. Lung Cancer S1525-7304 (22), 457–466. [published online ahead of print, 2022 Jun 21]. doi:10.1016/j.cllc.2022.06.002
Seng, T. J., Currey, N., Cooper, W. A., Lee, C. S., Chan, C., Horvath, L., et al. (2008). DLEC1 and MLH1 promoter methylation are associated with poor prognosis in non-small cell lung carcinoma. Br. J. Cancer 99 (2), 375–382. doi:10.1038/sj.bjc.6604452
Taylor, A. M., Shih, J., Ha, G., Gao, G. F., Zhang, X., Berger, A. C., et al. (2018). Genomic and functional Approaches to understanding cancer aneuploidy. Cancer Cell 33 (4), 676–689.e3. doi:10.1016/j.ccell.2018.03.007
Tian, Y., Morris, T. J., Webster, A. P., Yang, Z., Beck, S., Feber, A., et al. (2017). ChAMP: Updated methylation analysis pipeline for Illumina BeadChips. Bioinformatics 33 (24), 3982–3984. doi:10.1093/bioinformatics/btx513
Vickers, A. J., and Elkin, E. B. (2006). Decision curve analysis: A novel method for evaluating prediction models. Med. Decis. Mak. 26 (6), 565–574. doi:10.1177/0272989X06295361
Warth, A., Körner, S., Penzel, R., Muley, T., Dienemann, H., Schirmacher, P., et al. (2016). Microsatellite instability in pulmonary adenocarcinomas: A comprehensive study of 480 cases. Virchows Arch. 468 (3), 313–319. doi:10.1007/s00428-015-1892-7
Wilkerson, M. D., and Hayes, D. N. (2010). ConsensusClusterPlus: A class discovery tool with confidence assessments and item tracking. Bioinformatics 26 (12), 1572–1573. doi:10.1093/bioinformatics/btq170
Woenckhaus, M., Stoehr, R., Dietmaier, W., Wild, P. J., Zieglmeier, U., Foerster, J., et al. (2003). Microsatellite instability at chromosome 8p in non-small cell lung cancer is associated with lymph node metastasis and squamous differentiation. Int. J. Oncol. 23 (5), 1357–1363. doi:10.3892/ijo.23.5.1357
Wu, L., Kou, F., Ji, Z., Zhang, B., Guo, Y., et al. (2021). SMYD2 promotes tumorigenesis and metastasis of lung adenocarcinoma through RPS7. Cell Death Dis. 12 (5), 439. Published 2021 May 2. doi:10.1038/s41419-021-03720-w
Xiao, X., Melton, D. W., and Gourley, C. (2014). Mismatch repair deficiency in ovarian cancer - molecular characteristics and clinical implications. Gynecol. Oncol. 132 (2), 506–512. doi:10.1016/j.ygyno.2013.12.003
Xiao, Y., Lu, D., Lei, M., Xie, W., Chen, Y., Zheng, Y., et al. (2021). Comprehensive analysis of DNA damage repair deficiency in 10,284 pan-cancer study. Ann. Transl. Med. 9 (22), 1661. doi:10.21037/atm-21-5449
Yanagawa, N., Yamada, N., Sugimoto, R., Osakabe, M., Uesugi, N., Shiono, S., et al. (2021). The frequency of DNA mismatch repair deficiency is Very low in surgically resected lung carcinoma. Front. Oncol. 11, 752005. Published 2021 Oct. doi:10.3389/fonc.2021.752005
Yoshioka, K. I., Matsuno, Y., Hyodo, M., and Fujimori, H. (2019). Genomic-destabilization-associated Mutagenesis and clonal evolution of cells with mutations in tumor-suppressor genes. Cancers (Basel) 11 (11), 1643. Published 2019 Oct 24. doi:10.3390/cancers11111643
Keywords: lung squamous cell carcinoma, genomic instability, mismatch repair system, microsatellite instability, TP53, prognostic biomarkers
Citation: Hu Z, Liu Z, Zheng J, Peng Y, Lu X, Li J, Tan K and Cui H (2023) Microsatellite instability-related prognostic risk score (MSI-pRS) defines a subset of lung squamous cell carcinoma (LUSC) patients with genomic instability and poor clinical outcome. Front. Genet. 14:1061002. doi: 10.3389/fgene.2023.1061002
Received: 04 October 2022; Accepted: 30 January 2023;
Published: 17 February 2023.
Edited by:
Rajiv Kumar, German Cancer Research Center (DKFZ), GermanyReviewed by:
Hanchu Xiong, Zhejiang Provincial People’s Hospital, ChinaBing Song, University of Texas Southwestern Medical Center, United States
Copyright © 2023 Hu, Liu, Zheng, Peng, Lu, Li, Tan and Cui. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Huijuan Cui, Y3VpaGoxOTYzQHNpbmEuY29t
 Zixin Hu
Zixin Hu Zhening Liu1,2
Zhening Liu1,2 Yanmei Peng
Yanmei Peng Xingyu Lu
Xingyu Lu Jia Li
Jia Li Huijuan Cui
Huijuan Cui