- 1Laboratory of Animal Physiology and Biochemistry, Animal Embryo Center, College of Animal Science, Qingdao Agricultural University, Qingdao, China
- 2Laboratory of Animal Molecular Shandong Black Cattle Breeding Engineering Technology Center, College of Animal Science, Qingdao Agricultural University, Qingdao, China
Shandong black cattle is a new breed of cattle that is developed by applying modern biotechnology, such as somatic cloning, and conventional breeding methods to Luxi cattle. It is very important to study the function and regulatory mechanism of circRNAs in muscle differentiation among different breeds to improve meat quality and meat production performance and to provide new ideas for beef cattle meat quality improvements and new breed development. Therefore, the goal of this study was to sequence and identify circRNAs in muscle tissues of different breeds of cattle. We used RNA-seq to identify circRNAs in the muscles of two breeds of cattle (Shandong black and Luxi). We identified 14,640 circRNAs and found 655 differentially expressed circRNAs. We also analyzed the classification and characteristics of circRNAs in muscle tissue. Gene Ontology and Kyoto Encyclopedia of Genes and Genomes analyses were used on the parental genes of circRNAs. They were mainly involved in a variety of biological processes, such as muscle fiber development, smooth muscle cell proliferation, bone system morphogenesis, tight junctions and the MAPK, AMPK, and mTOR signaling pathways. In addition, we used miRanda to predict the interactions between 14 circRNAs and 11 miRNAs. Based on the above assays, we identified circRNAs (circ0001048, circ0001103, circ0001159, circ0003719, circ0003424, circ0003721, circ0003720, circ0001519, circ0001530, circ0005011, circ0014518, circ0000181, circ0000190, circ0010558) that may play important roles in the regulation of muscle growth and development. Using real-time quantitative PCR, 14 circRNAs were randomly selected to verify the real circRNAs. Luciferase reporter gene system was used to verify the binding site of miR-1 in circ0014518. Our results provide more information about circRNAs regulating muscle development in different breeds of cattle and lay a solid foundation for future experiments.
Introduction
CircRNA is a unique kind of non-coding RNA that has no 5′ terminal cap or 3′ terminal poly (a) tail and presents a closed ring structure (Rong et al., 2017). Kolakofsky (1976) and Sanger et al. (1976) first discovered circular RNAs (showed closed loop) in plant viroid and parainfluenza virus particles by electron microscopy. It was found that most of the circRNAs originate from exons and a few from introns. In terms of function, circRNAs mainly adsorb miRNAs through the “molecular sponge” mechanism, thus inhibiting the regulatory role of miRNAs and enhancing the expression levels of target genes (Memczak et al., 2013; Traurig et al., 2013; Zhang et al., 2016). In recent years, circRNA has become a new research hotspot in the field of scientific research. It has been found that circRNAs are involved in many biological processes, including growth and development, and diseases, among others. Ivano sequenced human and mouse myoblasts and compared the expression of circRNAs in myoblasts and myoblasts of Duchenne muscular dystrophy. It was the first time that circRNA was expressed during myogenic differentiation. Thirty one significantly different circRNAs were screened out, which laid a foundation for the study of circRNA in skeletal muscle development (Legnini et al., 2017). Subsequent studies found that circRNA was expressed in the skeletal muscle of pigs, cattle and other animals, and its expression was spatiotemporal and interspecific. Li found that the circRNAs circFUT1O and circFGFR4 could regulate the proliferation and differentiation of bovine skeletal muscle cells by absorbing miR-133a and miR-107, respectively (Li et al., 2017, 2018). Wei found that circLMO7 can regulate the differentiation and apoptosis of bovine skeletal muscle cells by absorbing miR-378a-3p (Wei et al., 2017). In conclusion, circRNAs play an important role in the development of bovine muscle. There are almost no reports about the regulation by circRNAs of the development of bovine skeletal muscle. In addition, we found that the current research on circRNAs is mainly focused on cancer, while the research on livestock and poultry mainly focuses on different periods (fetal vs adult), and there is little research on different varieties. This study is mainly about identification and characterization of circRNA in different breeds of cattle, which has certain theoretical significance for breeding.
Shandong black cattle are the first embryo transfer calves in China and are obtained by vitrified frozen somatic cell cloning embryos. According to the breeding goal, the new generation core breeding group was established by filial generation of Shandong black cattle. Then the new generation core breeding group hybridized with Shandong black cattle. Then the ideal black cattle and Shandong black cattle were selected from the second generation group and entered the cross-cross fixation stage. After four generations, it was finally bred into a new Shandong black cattle variety matching line and used as a bull. After careful cultivation by researchers, Shandong black cattle bulls were used in breeding. At present, there are 351 breeding bulls and 694 basic cows that meet the requirements of breeding objectives. Their average weight is 920 kg bulls and 510 kg cows. In 2015, Shandong black cattle was approved by the National Animal and Poultry Genetic Resources Committee as a new population and successfully established a new variety cultivation Chinese base. With the approval of the local government, we have established the local standard of Shandong black cattle. The major genes CAST, MSTN, and CAPN1 related to fat deposition, muscle tenderness and marbling have been preliminarily screened, As a molecular marker for early screening of cattle, ultrasonic was used to determine marbling, eye muscle area, back fat thickness, and intramuscular fat content. The purpose of this study was to investigate the identification and characterization of cirRNA in Hybrid Progenies (Shandong black cattle) and primordial maternal generation (Luxi cattle), and further explore its regulation related to production performance. Finally, high-quality beef cattle were selected to realize the leap from new varieties to new varieties.
We selected Shandong black cattle and Luxi cattle for this research study, identified circRNAs in muscle tissue, analyzed their genomic characteristics, expression differences, etc. Through high-throughput sequencing technology combined with functional verification tests, we revealed the function and regulatory mechanism of circRNA in muscle development and combined these findings with the characteristics of different breeds to cultivate fast-growing and high meat production rate beef cattle varieties to increase the speed of beef cattle breeding and provide a theoretical basis for the development of China’s beef industry.
Materials and Methods
Sample Preparation
The animals used in this research institute, Shandong black cattle and Luxi cattle, were selected from Shandong black Cattle Technology Co., Ltd., and Dadi yellow cattle. Three healthy Shandong black cattle and three Luxi cattle animals at 18 months old were selected. All experimental cattle were raised in the same farm environment, and the samples were stored in liquid nitrogen to extract total RNA. Muscle tissue samples were fixed in 4% formaldehyde and stained with hematoxylin and eosin (HE) for histological observation. All experimental and surgical procedures involved in this study followed the “guidelines for experimental animals” of the Ministry of Science and Technology (Beijing, China). All procedures and animal care were in line with the recommendations of the European Commission (1997) and were approved by the Experimental Animal Ethics Committee of Qingdao Agricultural University.
HE Staining of Muscle Tissue
Paraffin sections were made from muscle tissue fixed with 4% paraformaldehyde. The HE staining protocol included dewaxing, covering with water, hematoxylin staining, washing with water, 5% acetic acid differentiation, back blue, eosin staining, dehydration, natural drying, sealing, and image acquisition. The specific steps have been described previously (Cao et al., 2014). HE staining images were used for counting, and the surface area was measured by Image-Pro Plus software. SPASS software was used for statistical analysis to calculate significant differences.
circRNA Sequencing
High-throughput full transcriptome sequencing and subsequent bioinformatics analysis were carried out by Annoload Technologies (Beijing, China) as follows. Total RNA was extracted from the longissimus dorsi muscle of Shandong black cattle (B) and Luxi cattle (L) using TRIzol reagent (TIANGEN) according to the manufacturer’s instructions, and genomic DNA was removed with DNase I (Takara). RNA quality (RNA integrity number, RIN) was determined by an Agilent 2100 Bioanalyzer, and ND-2000 (NanoDrop) was quantified. Using a Ribo-Zero Gold Kit to remove rRNA from samples, and according to the specifications for the NEBNext Ultra Directional RNA Library Preparation Kit for Illumina (NEB, Ipswich, MA, USA), different index tags were selected to construct the library, and Illumina was used to sequence the constructed library.
Read Quality Control and Mapping
The original paired-end reads were trimmed and filtered for quality Trimmomatic using the default parameters.1 Then, TopHat software2 was used to align the clean reads with the reference bovine genome3 and obtain the orientation pattern. The software was used to align the RNA sequence reads with the genome to detect gene expression and exon splicing. The genome was constructed on the superfast short read mapper Bowtie 2 for mapping with default parameters.
Identification of Differentially Expressed circRNAs
We used SRPBM [spliced reads per Billion Mapping, defined as number of circular reads/(number of mapped reads × read length)] as a standardized method to quantify the expression of circRNA, and DEseq2 was used to analyze the differential expression of circRNA (Love et al., 2014). In pairwise comparisons, circRNAs with P < 0.05 and absolute fold change value greater than 1.5 were considered to be significantly differentially expressed, and finally, the number of upregulated and downregulated circRNAs was obtained.
Enrichment Analysis of GO, KEGG, and PPI Pathways
Gene Ontology (GO) analysis and KEGG pathway analysis of the parental genes of differentially expressed circRNAs were used for annotation. The Blast2GO method was used for GO function analysis. KOBAS software was used to test the statistical enrichment of differential gene expression in the KEGG pathway. When P < 0.05, GO terms and KEGG pathways were considered to be significantly enriched.
Prediction of miRNA Targets of circRNAs
To explore the function of circRNAs and predict which circRNA acts as a miRNA sponge, we used miRanda (3.3a)4 to predict the target relationship (John et al., 2004). In view of the published reports and the extractability of the sequences, we only selected the type of classical (when the formation site of the circRNA was exactly on the boundaries of exons) and antisense (when the circRNA was formed by the antisense strand of the gene) circRNAs to predict the miRNA targeting relationship.
Experimental Verification of circRNA
Real-time quantitative PCR (qRT-PCR) was used to verify the expression of circRNA. The expression levels of the selected circRNAs were standardized with the levels of the housekeeping gene GAPDH. Primer 3.0 software was used to design primers (Supplementary Table 7), which were synthesized by SANGON Biotechnology Co., Ltd. (Shanghai, China). Total RNA was converted into cDNA using random hexamers with Transcriptor First Strand cDNA Synthesis Kit (Roche, Australia). The qRT-PCR reaction (20 μL) consisted of 1 μL template cDNA, 5 ml (5 × 1 ml vials) 10 μL upstream and downstream primers, respectively, RNAse-free water. The procedure was as follows: 94°C for 10 min and 40 cycles of 94°C for 30 s, 60°C for 30 s, and 72°C for 40 s. The expression of circRNAs were calculated by the 2–ΔΔ CT value method.
Luciferase Reporter Assay
Cells were seeded in 96-well plates at a density of 5 × 103 cells (HEK-293T) per well 24 h before transfection. The cells were co-transfected with a mixture of 50 ng FL reporter vectors, 5 ng Renilla luciferase (RL) reporter vectors (pRL-TK), and miRNA mimics at the indicated concentration. The miRNA mimic were obtained from Life Technologies. After 48 h, the luciferase activity was measured with a dual luciferase reporter assay system (Promega, Madison, WI, United States). In this study, the double luciferase vector psi check-2 was selected, and the bta-circ0014629 sequence was cloned into the reporter gene vector psicheck to synthesize the predicted miRNA mimics and control. MiRNA mimics and psicheck-bta-circ0014629 were transfected into 293T cells at the same time. The expression level of reporter genes was detected by multi-functional enzyme labeling instrument, and the miRNAs with down-regulation of reporter gene expression were screened. It is necessary to clone the potential binding target of miRNA into the 3′UTR region of r-luciferase (hrluc), and then co transfect with the miRNA to determine the activity of R-Luciferase. F-Luciferase (hluc+) is used as the internal reference to correct the transfection efficiency between different samples.
Results
Apparent Differences in Muscle Fibers in Different Breeds of Beef Cattle
The muscle fibers of the longissimus dorsi in the back of Shandong black cattle and Luxi cattle were significantly different in the apparent observation of paraffin sections stained by HE (Figure 1). The length of single muscle fibers of Shandong black cattle was significantly longer than that of Luxi cattle, and the number of nucleus in each muscle fiber was also greater. The boundary between the muscle fibers of Shandong black cattle was clearer and rounder than that of Luxi cattle. IPP software analysis showed that there were significant differences in the muscle fiber diameter, length and weight (P < 0.05) but no significant differences in other muscle fiber properties (P > 0.05) (Table 1).
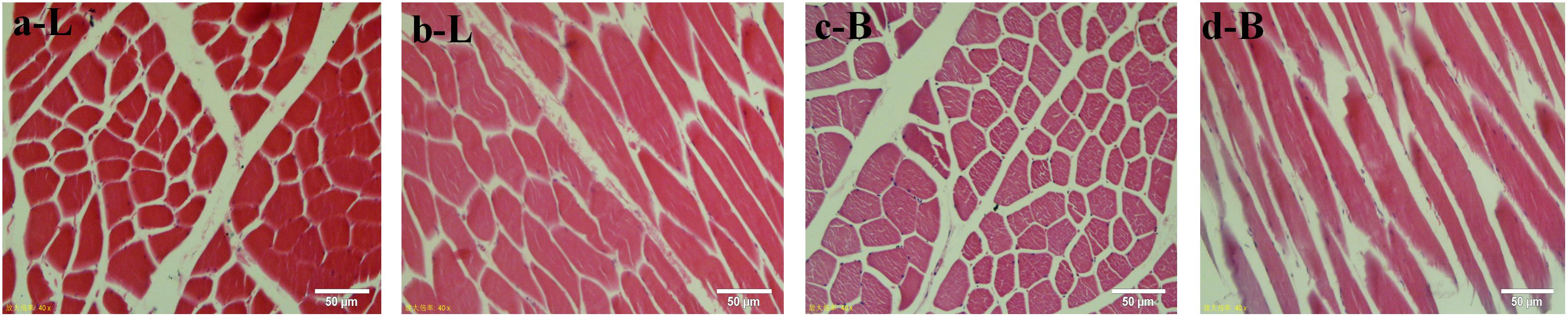
Figure 1. HE staining of muscle tissue paraffin sections. Red represents a single muscle fiber, blue represents a nucleus (marked by arrows); a and c stand for tissue crosscutting, b and d stand for tissue longitudinal cutting; L stands for Luxi cattle, B stands for Shandong black cattle.
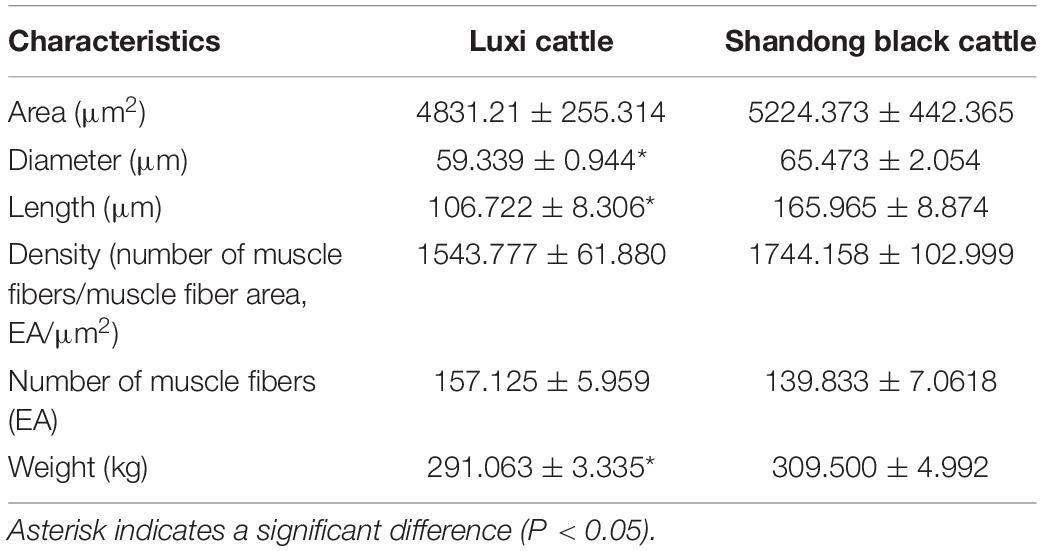
Table 1. Comparison of muscle fiber characteristics and growth characteristics of different breeds of cattle.
Transcriptome Quantification
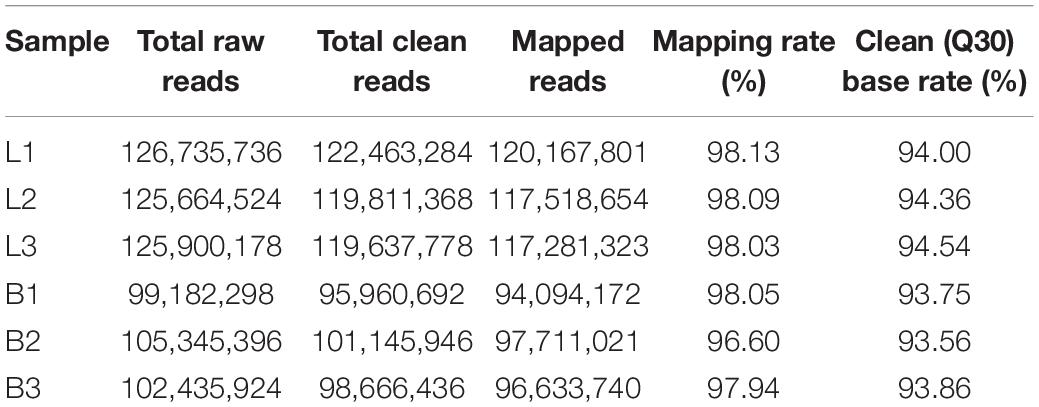
To examine the expression profile of circRNAs in the longissimus dorsi muscle of different breeds of beef cattle, we established a cDNA library of six longissimus dorsi muscle samples from Luxi cattle (L) and Shandong black cattle (B) and obtained the raw data after Illumina sequencing. After removing the low-quality reads and the reads with adapter sequences from the RNA-seq raw reads, 223,609,230 clean reads (B: 122,463,284, L: 101,145,946; Table 2) were obtained. Then, we queried the clean reads of the latest reference genome and used TopHat (see text footnote 2) for mapping. In the B and L samples, 98.08 and 97.53% of the reads were located in the reference genome, and the clean (Q30) base rates were 94.30 and 93.72%, respectively.
DEG Analysis and Functional Annotation
In this study, 14640 circRNAs and 4201 parental genes (Supplementary Table 1) were detected, of which 1591 (10.86%) were expressed in all samples (Figure 2A). The SRPBM values (one billionth of the splicing reading) were used to calculate the expression levels of circRNAs. We list the 30 highest expression circRNAs in each group (Table 3). According to their position in the genome, 14856 circRNAs were divided into six types: classical: when the formation site of the circRNA was exactly on the boundaries of exons (82.9%), alter exon: when one end of the circRNA formation site was on the exon boundary, and the other end was inside the exon (4.5%), intron: when the formation site of the circRNA was completely in the intron region (2.1%), overlap exon: when the formation site of the circRNA spanned the exon region (7.6%), antisense: when the circRNA was formed by the antisense strand of the gene (0.5%), and intergenic: when the formation site of circRNA was completely inside the intergenic region (2.3%) (Figure 2B). In a single exon, the length of the exon was significantly longer than that of the circular RNA composed of multiple exons (Figure 2C). circRNAs were mainly encoded by two to four exons (Figure 2D).
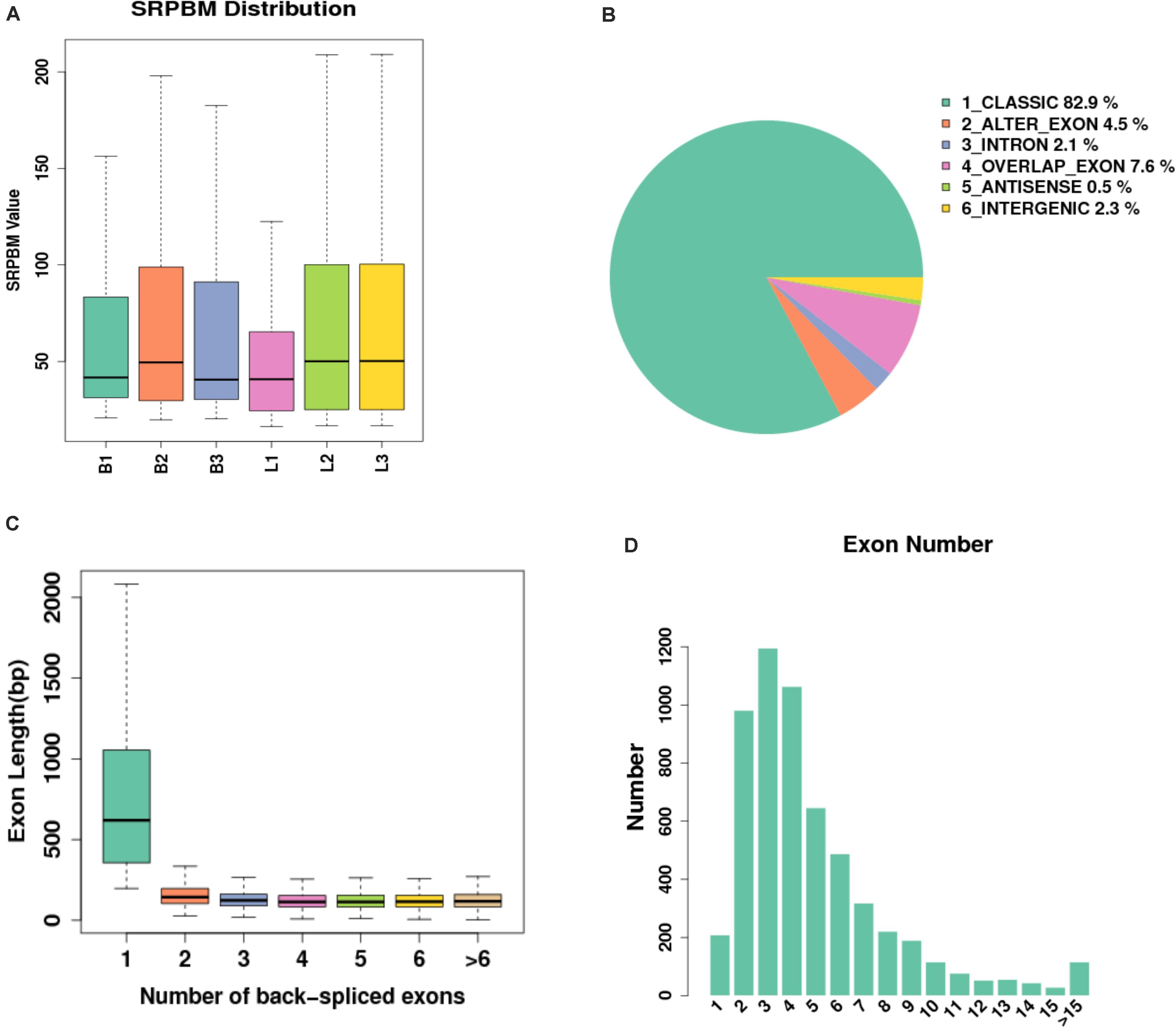
Figure 2. Expression level of sample circRNAs. (A) SRPBM density: distribution density of circRNA expression in the sample; SRPBM value: the normalized count value of circRNAs, calculated by a formula [SRPBM = (SR×109)/N] in which SR is the number of spliced reads, and N is the total number of mapped reads in samples. (B) Distribution pie chart of the six types of circRNAs; (C) Box plot of the single exon length distribution of circular RNAs. The abscissa represents the number of exons contained in the circRNA, and the ordinate represents the length of a single exon; (D) The distribution of the number of exons contained in circular RNAs in the sample, the abscissa represents the number of exons, and the ordinate represents the number of circRNAs corresponding to the number of exons.
Differential Expression of circRNAs
In this study, 655 differential circRNAs and 467 parental genes (Supplementary Table 2) were detected, of which 267 were upregulated and 388 were downregulated (Figure 3A). circRNAs with significant differential expression are visualized by a red volcano map (Figure 3B) and cluster heat map (Figure 3C) to illustrate the distribution of differential circRNAs.
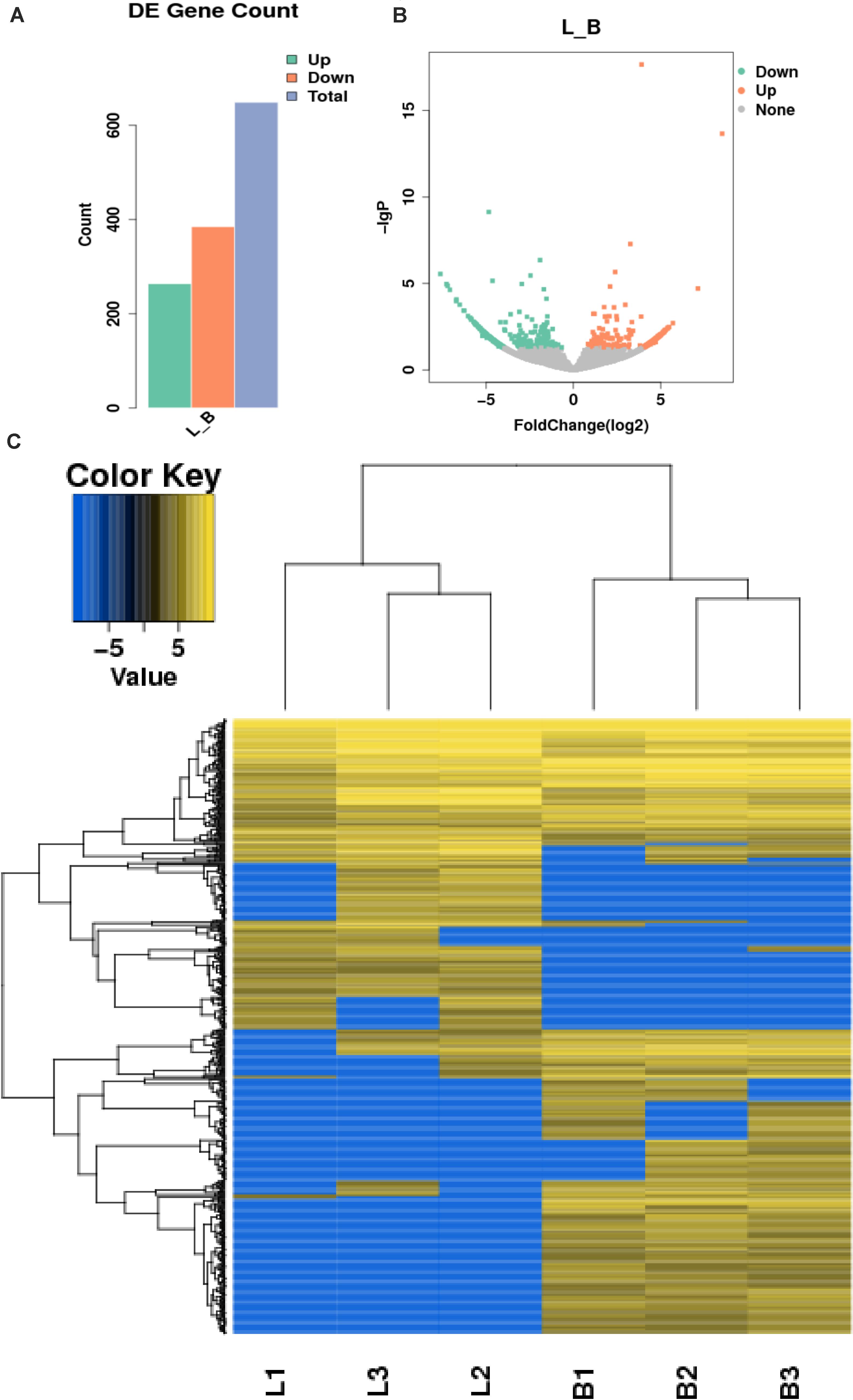
Figure 3. Differential expression of circRNAs in the samples. (A) Statistical chart of the differential expression of 655 DE circRNAs, of which 267 were upregulated and 388 were downregulated; (B) Volcanic chart of differential circRNAs, the abscissa is the fold change of expression in L and B samples, the ordinate is the statistical significance of the change of expression. Different colors indicate different classifications: green (downregulated), orange (upregulated), and gray (the difference is not obvious); (C) Cluster heat map of differentially expressed genes. The change in expression quantity is represented by the change in color, blue represents lower expression levels, and yellow represents higher expression levels.
Enrichment Analysis by GO, KEGG, and SPONG
The function of a circRNA is reflected by its parental genes. To determine the function of these genes, GO analysis was carried out. According to the statistical data of differentially expressed circRNAs and their parental genes, we annotated the parental genes of circRNAs instead of the circRNAs because there is currently no information on the annotation of circRNAs. The parental genes of differentially expressed circRNAs were annotated by 65 different GO terms (Figure 4A). The most annotated GO terms were cellular process (BP), single organization process (BP), cell part (CC), organelle (CC), binding (MF), and catalytic (MF). To understand the functions of DEGs, goatools5 were used to conduct functional enrichment GO analyses. The results showed that 142 differentially expressed parental genes of circRNAs were significantly enriched. The highest enrichment was cell process, regulation of major metabolic processes, intracellular part, intracellular organelle, membrane-bound organelle and egg white matter binding (Figures 4B–D). The significant enrichment results of the three GO categories are listed in Supplementary Table 3. According to the statistical results of the GO functional enrichment analysis (Supplementary Table 3), we identified 29 terms of 60 different parental genes related to muscle growth and development. See Supplementary Table 4 for details. According to statistics of the enrichment degrees of the 60 DE parental genes in each term (Figure 5A), it is found that the genes with higher enrichment degrees were gene 1570 (TTN), gene 23041 (MYBPC2), gene 6914 (MYBPC1), gene 1832 (NEB), and gene e22584 (MYH15), while the genes with lower enrichment degrees were gene 7152 (ZBCC9), gene 5917 (ACTR3b), and gene 5819 (EZH2), all of which are involved in the process of muscle growth and development, but the enrichment degree was highly different.
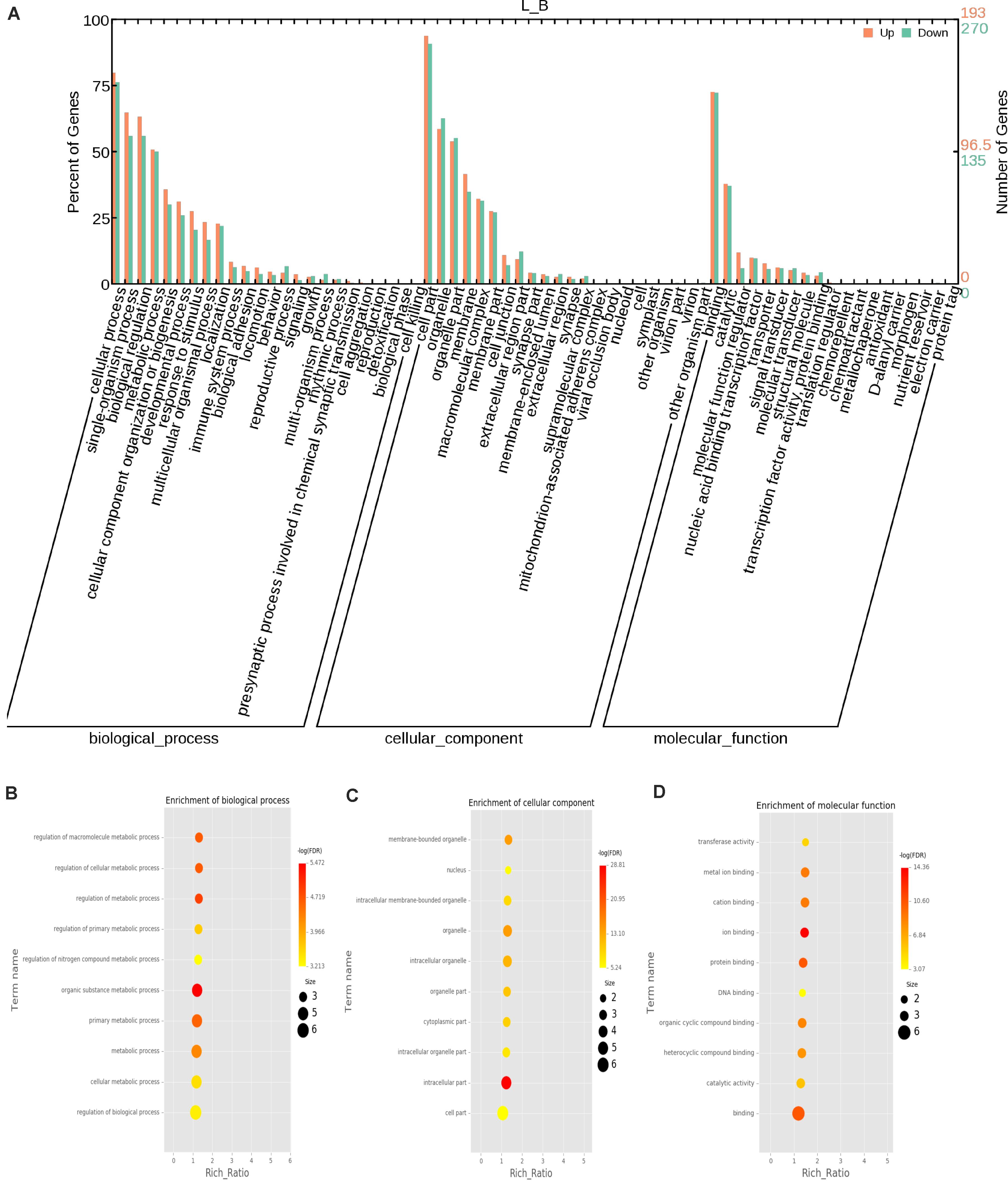
Figure 4. GO annotation of differential circRNAs and enrichment analysis. (A) GO annotation of the parental genes of DE circRNAs. The abscissa is the ontology classification, and the ordinate is the proportion of genes annotated to this term among all annotated genes; (B–D) Q-value enrichment map of single group GO entries. Each point represents the estimated enrichment degree of the corresponding GO term, and the closer the color is to red, the higher the enrichment degree. The size of each point indicates the number of genes enriched in the GO term, and the larger the point is, the more genes enriched in the GO term, and vice versa.

Figure 5. Enrichment degree of the identified parental genes in GO terms (A) and KEGG pathways (B). Different colors indicate the different enrichment degrees of parental genes in terms related to muscle growth and development. The darker the color, the higher the enrichment degree and the lighter the color, the lower the enrichment degree; the enrichment degree of source genes is arranged from high at the top to low at the bottom.
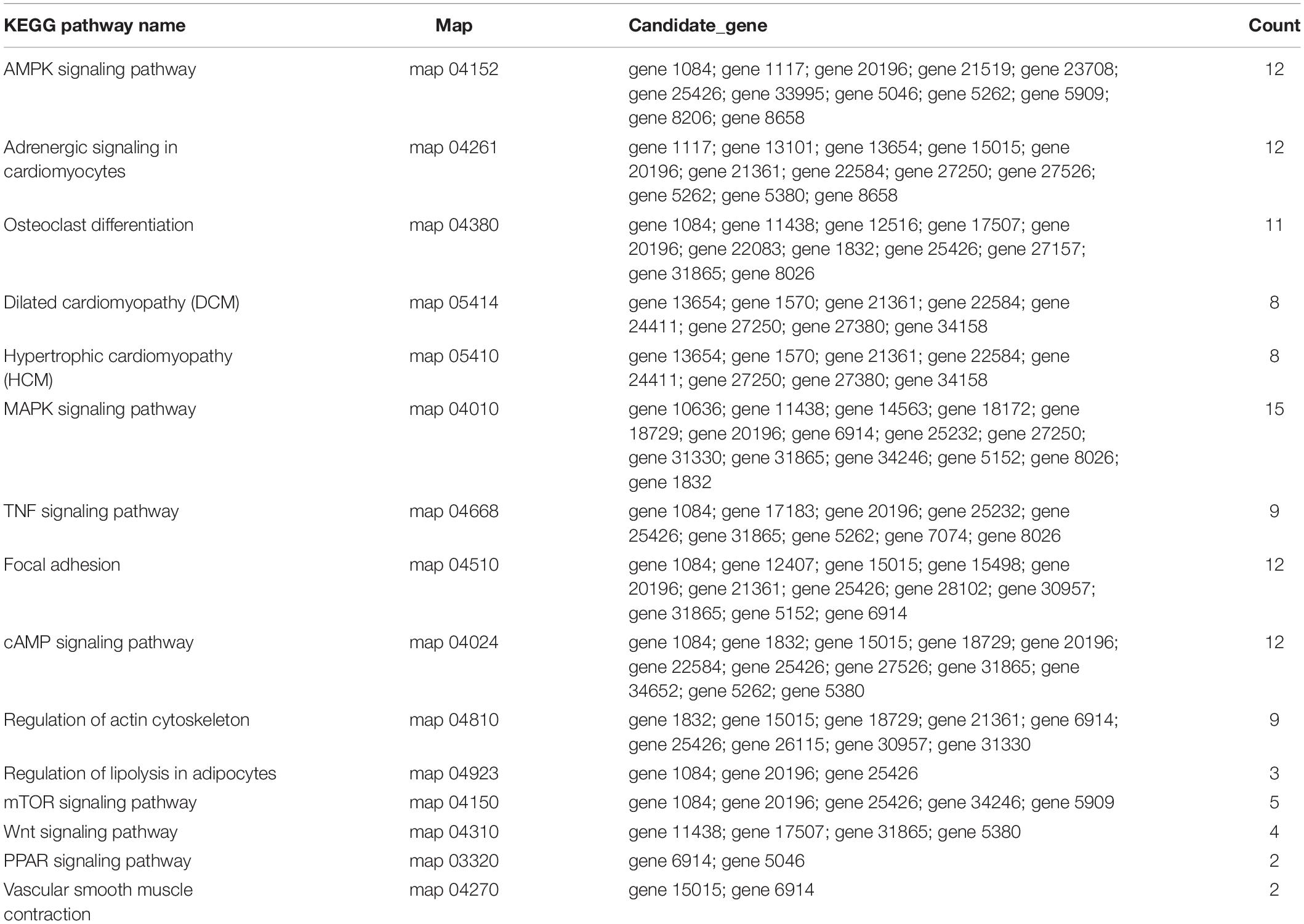
To predict the functions of significantly enriched parental genes, pathway analysis was conducted based on the KEGG pathway database. Among the 36 pathways with significant enrichment, the AMPK signaling pathway, cellular pathway, and cellular pathway and adaptive signaling in cardiomyocytes were the most significantly enriched (Figure 6A and Supplementary Table 6). All the samples were enriched by KOBAS, and the distribution map was made according to the significance of the Q-values of the KOBAS enrichment analysis (Figure 6B). Based on the above enrichment results, we identified 15 pathways (AMPK signaling pathway, adaptive signaling in cardiometrics, osteoblast differentiation, differentiated cardiopathy (DCM), hypertonic cardiopathy (HCM), MAPK signaling pathway, TNF signaling pathway, focal adhesion, and cAMP signaling pathway. These pathways were enriched for 49 parental genes (Table 4). One parental gene was involved in the regulation of multiple pathways. According to the statistics of the pathway enrichment analysis of the 49 parental genes (Figure 5B), the genes with higher enrichment were gene 20196 (AKT3), gene 1084 (PIK3CB), gene 25426 (PIK3R1), gene 31865 (MAPK8), and gene 21361 (MYL2), and the genes with lower enrichment were gene 6914 (MYBPC1), gene 7074 (BNM1L), gene 8206 (TBC1D1), and gene 822 (MECOM), all of which are involved in muscle processes. The pathways related to meat growth and development were also regulated, but the degree of enrichment was different.
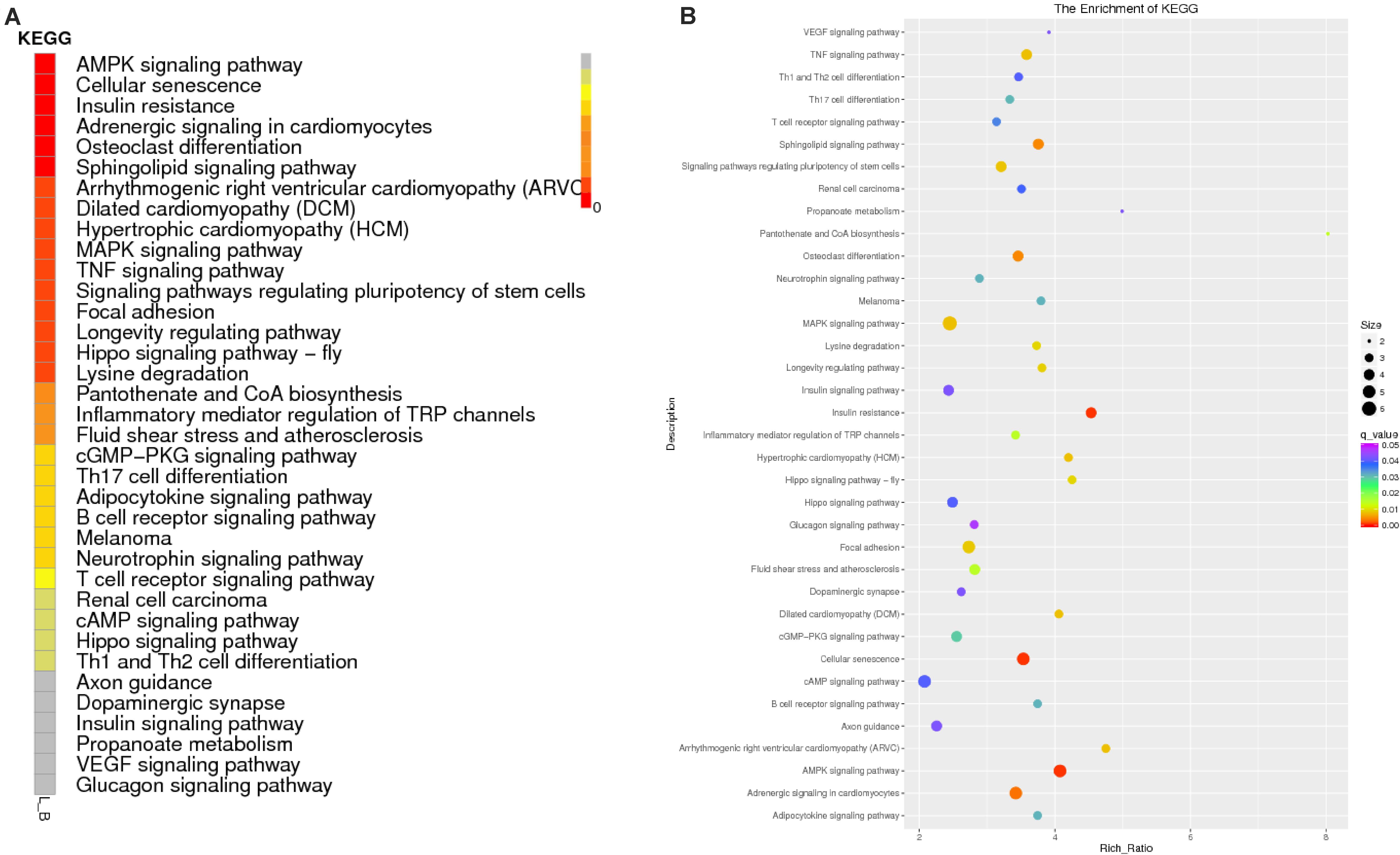
Figure 6. Enrichment results of the KEGG analysis. (A) Enrichment path Q-value distribution map, in which the vertical coordinate is the KEGG pathway entry, horizontal coordinate is the name of the sample, and different colors represent different enrichment degrees; (B) KEGG enrichment Q-value results for single groups, whereby each point represents the enrichment degree of the KEGG entry, and the closer the color is to red, the higher the enrichment degree. The size of each point indicates the number of genes enriched in the KEGG entry, and the larger the point is, the more genes enriched in the KEGG entry, and vice versa.
Target miRNAs of Differentially Expressed circRNAs in Different Breeds of Beef Cattle
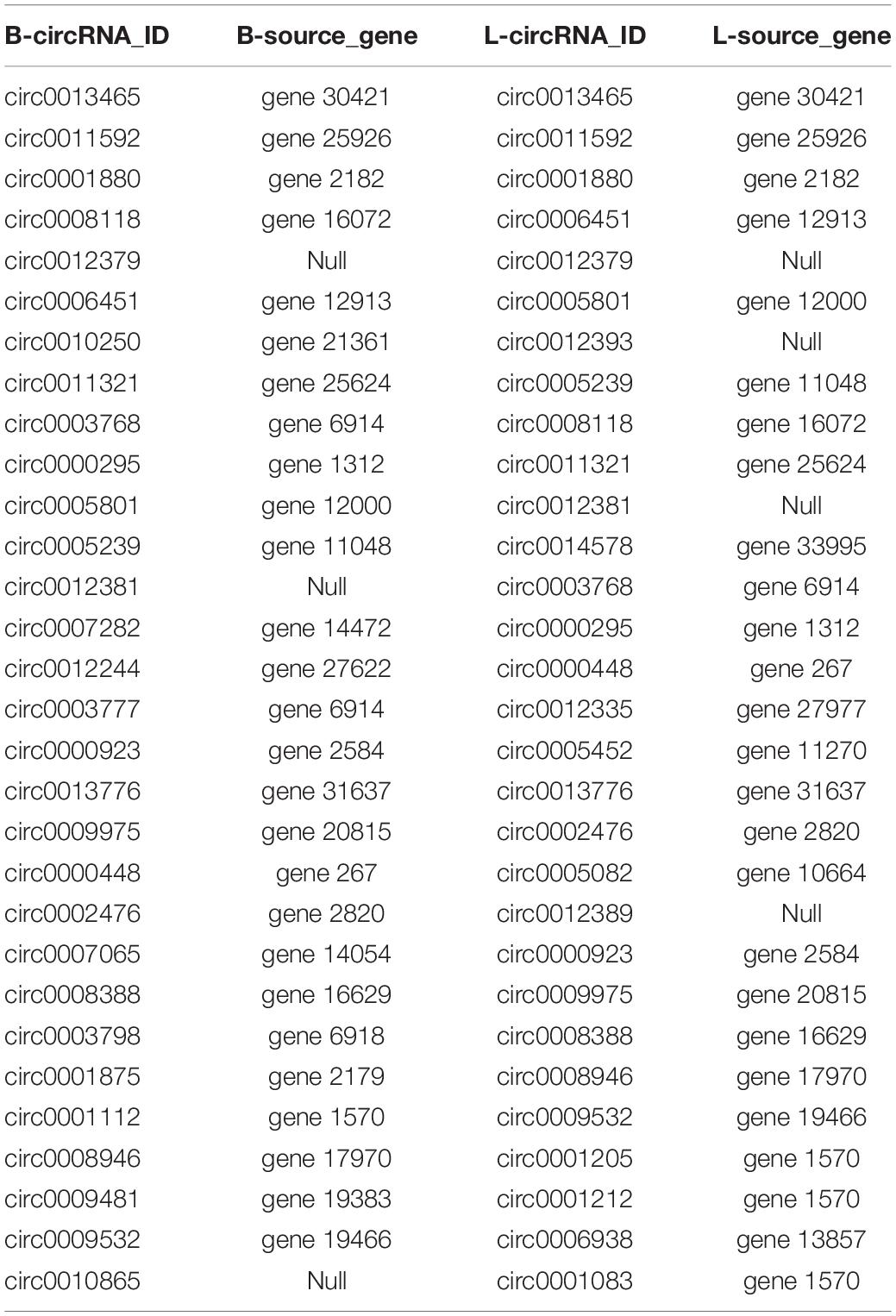
To further understand the function of circRNAs, we used miRanda (see text footnote 4) to predict the interactions between circRNAs and miRNAs. According to the interaction data and differential expression of miRNAs and circRNAs, the interaction network data files were generated and imported into Cytoscape software. The attributes of the target circRNAs were visualized in the network, and the topological attributes of some networks are marked (Figure 7A). A total of 1799 circRNAs and 652 miRNAs were obtained to generate 5037 pairs of interactions between miRNAs and circRNAs (Supplementary Table 6), in which circ0013807 interacts with 37 miRNAs, circ0006152 interacts with 27 miRNAs, miR-11988 interacts with 449 circRNAs, and miR-11986b interacts with 376 circRNAs. According to the above differential expression and GO/KEGG/PPI enrichment analyses, we identified seven genes of different sources related to muscle growth, corresponding to 14 circRNAs and 11 target miRNAs (Figure 7B). It was found that there are multiple binding sites of miRNAs in some circRNAs (such as MYBPC1 and miR-11986b, RYR1 and miR-1) sequences. After a miRNA is adsorbed, it cannot regulate its corresponding target gene, thus a circRNA acts as a miRNA molecular sponge.
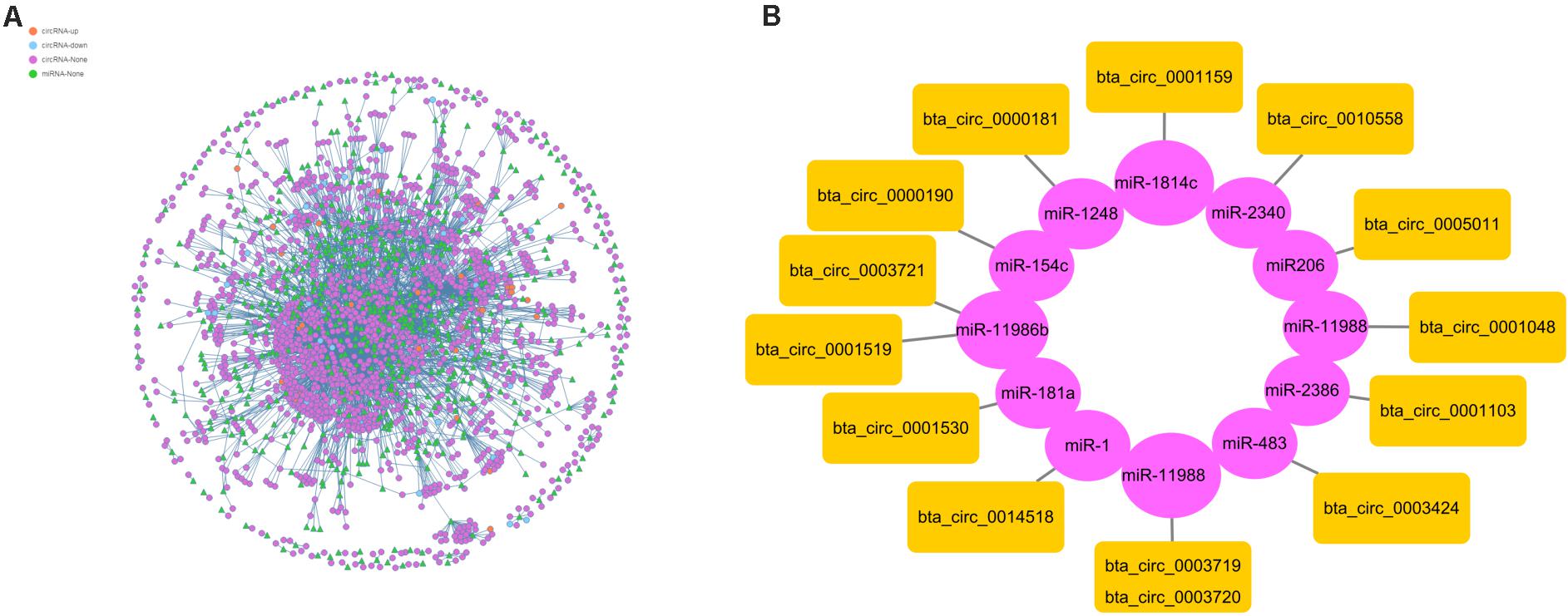
Figure 7. Interaction between circRNAs and miRNAs. (A) The circle nodes represent circRNAs, and the triangle nodes represent miRNAs. The lines represent the relationship between circRNAs and miRNAs. (B) Prediction of the target relationships between miRNAs and circRNAs.
Confirmation of circRNA Expression by qRT-PCR
To verify the expression level of differentially expressed circRNAs, we randomly selected six highly expressed circRNAs and detected their expression level by qRT-PCR (Supplementary Table 7). These results were consistent with the trends observed in RNA-seq data (Figure 8A), with a correlation coefficient R2 = 0.9982 indicating that the RNA-seq results were reliable (Figure 8B). In this study, the cDNA generated by RNA reverse transcription was collected again after RNase R digestion, and the presence of circRNA was still detected in subsequent experiments. qRT-PCR quantitative detection results (Figure 8C) showed that compared with the control group (GAPDH), the expression of circRNA in RNase R digestion group was increased, and the expression of GAPDH was almost undetectable after digestion by exonuclease.
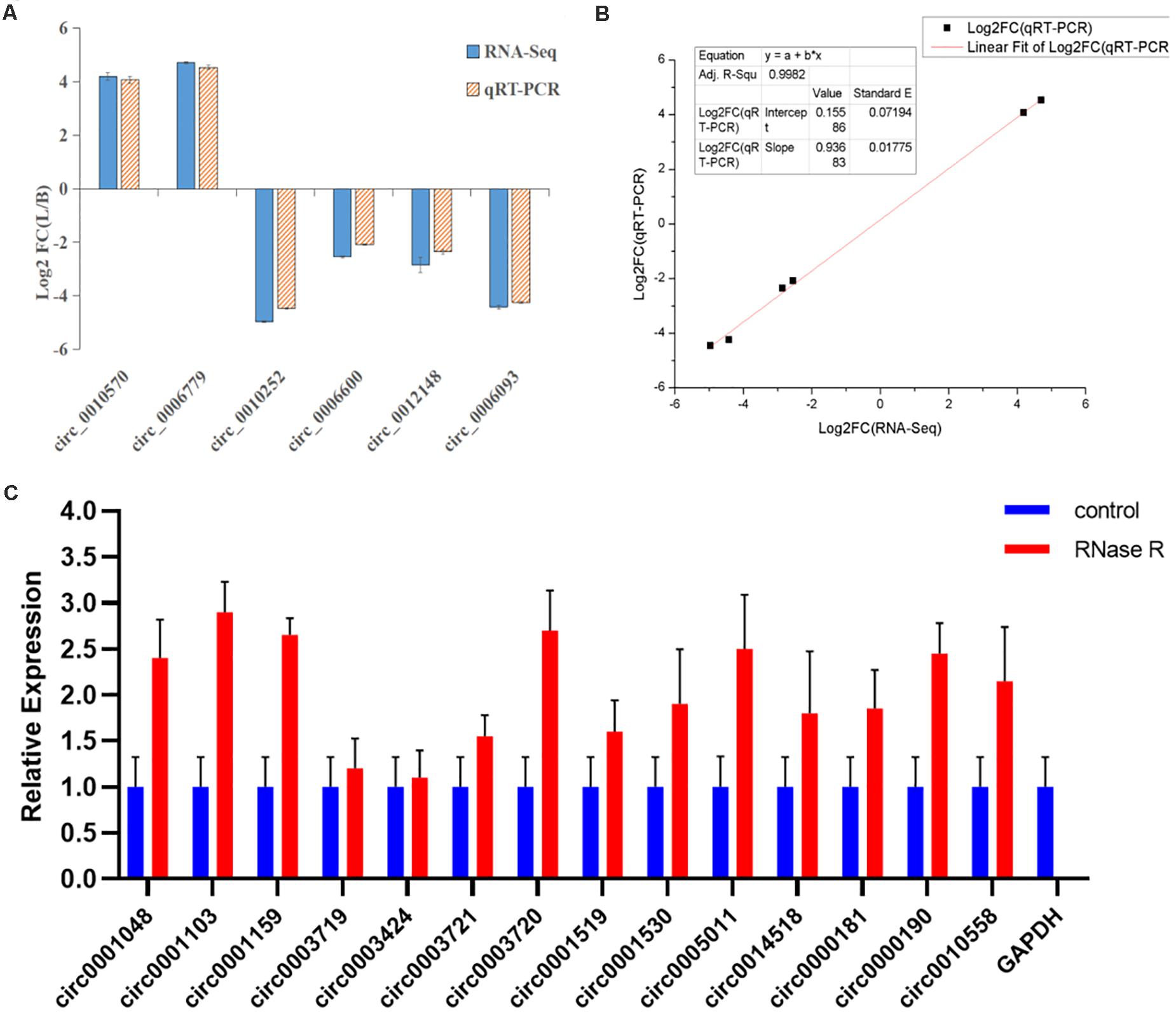
Figure 8. Linear fitting of RNA-seq and qRT-PCR circRNA expression data. (A) The circRNAs related to muscle growth and development were selected for qRT-PCR analysis. (B) The six selected circRNAs showed consistent results between the RNA-seq and qRT-PCR analyses, with a correlation coefficient (R2) = 0.9982. (C) After RNase R treatment, the expression level of circRNAs and GAPDH were measured. The X-axis indicates circRNAs, and the Y-axis indicates the relative expression level of circRNAs and GAPDH. Error bars indicates ±SD.
Detection of Luciferase Activity of circ0014518 and bta-miR-1
The miRNA binding site of circ0014518 was screened according to the sequencing data and bioinformatics analysis results. It was found that circ0014518 contained bta-miR-1 binding site. Both RNA22 and RNAhybird predicted that circ0014518 contained bta-miR-1 binding sites (Figure 9A). In order to further verify the binding site of circ0014518 and miR-1, the psi-circ0014518 vector containing bta-miR-1 binding site was constructed, and the binding site was mutated from acauuc to ucuugc by site directed mutagenesis kit. Double luciferase reporter gene system was used to verify the binding site of circ0014518 and bta-miR-1. The results (Figure 9B) showed that the luciferase activity of bta-miR-1 and psi-circ0014518 was significantly lower than that of the control group (P < 0.01), but the luciferase activity of the mutant vector and bta-miR-1 co-transfected group was not significantly different from that of the control group, indicating that circ0014518 has a bta-miR-1 binding site.
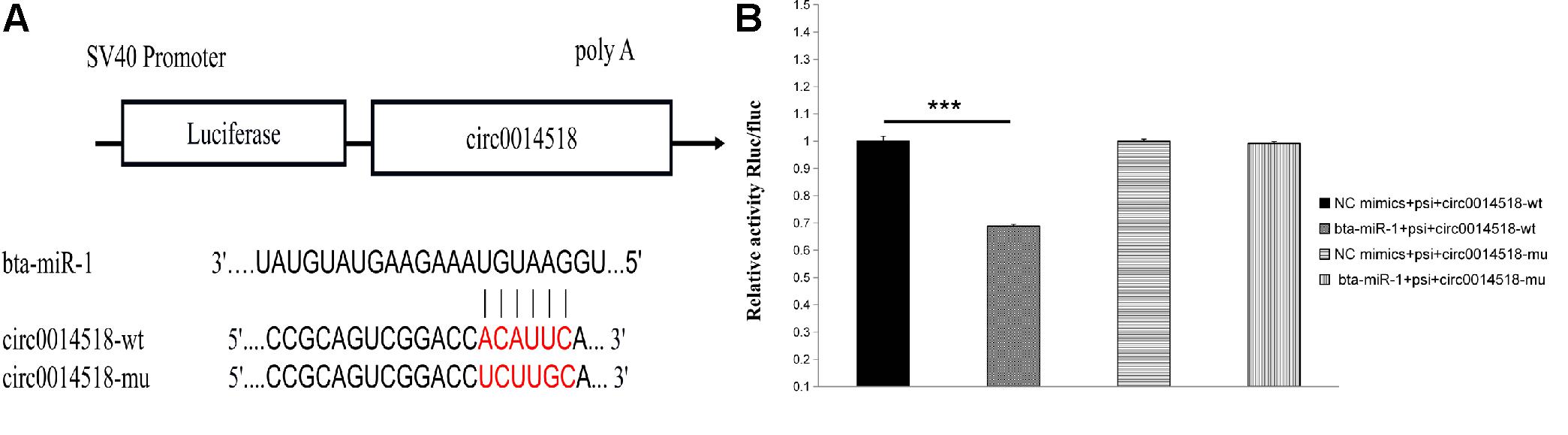
Figure 9. The prediction and verification binding site of circ0014518 and bta-miR-1. (A) The predicted binding site of between circ0014518 and bta-miR-1. (B) Verification binding site of circ0014518 and bta-miR-1 the luciferase reporter assay. ***P < 0.01.
Discussion
In this study, we first examined the apparent differences in muscle fibers of different breeds of beef cattle. The results showed that there were obvious differences in the apparent observation of the muscle fibers of HE stained paraffin sections of the longissimus dorsi of Shandong black cattle and Luxi cattle. The length of a single muscle fiber of Shandong black cattle was significantly longer than that of Luxi cattle, and the number of nuclei in each muscle fiber was also greater. The border between the muscle fibers of Shandong black cattle was clearer and rounder than that of Luxi cattle. IPP software analysis showed that there were significant differences in the diameter and length of muscle fibers (P < 0.05) but no significant differences in the number, density or area of other muscle fibers (P > 0.05). The occurrence of these differences may be the key factors leading to the differences in meat production performance and meat quality of the two breeds of cattle after birth, which was also the research basis of this study to explore the underlying molecular regulatory mechanism.
We used RNA-seq technology to study the expression of circRNA in the longissimus dorsi muscle of different breeds of beef cattle. A total of 14,640 circRNAs and 4201 parental genes were detected. circ0013465 (UBE2D1), circ0011592 (UBE3A), and circ0001880 (MYL1) were the most highly expressed in the two libraries. PROSITE-ProRule annotation of these proteins (UBE2D1 and UBE3A) showed that they are involved in protein ubiquitination, which is part of the protein modifications that regulate cell metabolism within eukaryotes (Polge et al., 2016). MYL1 is a crucial protein for adequate skeletal muscle function and belongs to the myosin family (Ravenscroft et al., 2018). The ubiquitin proteasome system (UPS) is mainly responsible for the increased protein breakdown observed in muscle wasting. The Ube family of E3 ligases is a class of enzymes (i.e., troponin I, myosin heavy chains, and actin) that can guide the degradation of major contractile proteins. Their catalytic activity depends on the covalent binding of polyubiquitin chains catalyzed by a specific E2 on the substrate (Uhler et al., 2014). Studies have shown that UPS can control almost any muscle mass and recovery process in catabolism. The muscle-specific E3 ligase UBE family participates in the targeting of actin, myosin, troponin, and other major contractile proteins (Ravenscroft et al., 2018), indicating that the high expression of circRNAs plays a certain role in muscle development and redifferentiation.
According to the fold change >1.5 and P < 0.05 criteria, 655 differentially expressed circRNAs were identified, corresponding to 467 parental genes, 267 of which were upregulated and 388 downregulated in Luxi cattle. The function of a circRNA is reflected in its parental gene. Because there is no information about the annotation of circRNAs at present, we annotated the parental genes of differentially expressed circRNAs. As a result, is the genes were annotated in 65 different GO terms, which mainly play a role in biological processes such as regulation of cell process, regulation of metabolic process and part in the cell. We identified 29 related terms. There were 60 different genes associated with muscle growth and development. The genes with higher enrichment were gene 1570 (TTN), gene 23041 (MYBPC2), gene 6914(MYBPC1), gene 1832 (NEB), and gene 22584 (MYH15). All of the above genes participate in muscle growth and development, and their corresponding circRNAs also play a role in this process. The number of circRNA parental genes in the different samples was significantly different. The difference reflects the cumulative effect of circRNAs on expression characteristics. Based on the KEGG pathway database, we further analyzed the circRNAs and found that the AMPK signaling pathway, cellular signaling pathway, and cellular signaling and alternative signaling in cardiomyocytes were the most significantly enriched pathways. In our study, based on the above enrichment results, we identified 15 distinct enriched pathways related to muscle growth and development, including the AMPK signaling pathway, MAPK signaling pathway and adaptive signaling in cardiometrics, which influence muscle fiber processes (O’Connell et al., 2013; Zeng, 2016); the MTOR signaling pathway and Wnt signaling pathway, which are involved in the regulation of skeletal muscle development and regeneration (Fernández-Martos et al., 2011; Hong-Bo et al., 2017; Cong, 2018); the PPAR signaling pathway, which is involved in the regulation of intramuscular fat deposition (Li, 2018) and the cytoskeletal signaling pathway. According to the statistical data, the genes with higher enrichment levels were gene 20196 (AKT3), gene 1084 (PIK3CB), gene 25426 (PIK3R1), gene 31865 (MAPK8), gene 21361 (MYL2), and gene 6914 (MYBPC1), which could indicate that the circRNAs produced by these genes may play a role in the growth and development of muscle through these pathways. Combining these results with the above results, we identified seven parental genes (TNN, MYBPC1, NEB, MEF2C, MYH7, PPP2R3A, and RYR1) and 38 corresponding circRNAs. Comparing these results with previous research results, significant differences were observed in the expression of circRNAs related to the muscle development of different breeds of cattle, suggesting that circRNAs may play an important role in muscle development. Whether these circRNAs have specific functions and what the functional mechanism is need to be studied further.
circRNAs can play important roles by regulating the transcription and expression of their parental genes (Zheng et al., 2017). At present, there is a relatively limited understanding of the details of the formation of circRNA and its functional mechanism. circRNA can be obtained by transcription of protein-coding genes or intergenic regions (Qu et al., 2015). The formation of circRNA from a protein-coding gene is caused by the variable splicing of the parental gene (Ashwal-Fluss et al., 2014). Therefore, there should be a certain correlation between a circRNA and its parental gene expression. We found that one source gene may produce multiple circRNA subtypes. For example, the MYBPC1 gene can produce nine different circRNA subtypes. We obtained the FPKM value of the two varieties and found that both were differentially expressed. Although one source gene may produce multiple circRNA subtypes at the same time, only 3 or 4 of them have high expression levels, and the rest have low expression levels, which indicates that the cyclization of RNA in muscle is strictly regulated. To further understand the biological function and molecular function of the parental genes of significantly differentially expressed circRNAs, we predicted the interaction between circRNAs and miRNAs and constructed a network from the interaction data. The interaction network showed that a single miRNA may be correlated with multiple differentially expressed circRNAs, and there have been reports that circRNAs can competitively adsorb miRNAs (Zheng et al., 2016). Based on the high-throughput sequencing results, we selected 14 circRNAs related to muscle development as candidate circRNAs (circ0001048, circ0001103, circ0001159, circ0003719, circ0003424, circ0003721, circ0003720, circ0001519, circ0001530, circ0005011, circ0014518, circ0000181, circ0000190, circ0010558). In addition, the target miRNAs were predicted, the corresponding mRNA targets of the miRNAs were predicted, and the circRNA-miRNA-mRNA network was constructed to further study the regulation of muscle development. We will further verify this network in future experiments, which provides a new basis for the study of muscle development in cattle.
In the study, the 14 circRNAs quantitatively detected were required to identify whether they were real circRNAs. The results showed that circRNA could resist RNase R digestion, but linear RNA could not. Therefore, the candidate circRNAs (circ0001048, circ0001103, circ0001159, circ0003719, circ0003424, circ0003721, circ0003720, circ0001519, circ0001530, circ0005011, circ0014518, circ0000181, circ0000190, circ0010558) are circular RNAs. At the same time, a circRNA was screened for functional verification. Luciferase reporter gene system was used to verify the binding site of miR-1 in circ0014518, indicating that circ0014518 can be used as a molecular sponge of miR-1. It has been reported that miR-1 participates in the regulation of skeletal muscle development through target gene Pax7. We intend to further verify this in future experiments. In addition to the above findings, there were pathways that were not found in our study that are known to be important and enriched for many parental genes and some that have been reported in previous studies to regulate muscle growth and development. The miRNAs that have been shown to be expressed specifically or preferentially in muscles are called muscle-specific miRNAs (muscle-specific microRNAs, myomiRs) (Horak et al., 2016) and include miR-1, miR-206, miR-128, miR-483, miR-2425-5p, miR-181a, miR-208a, miR-208b, and miR-486 (Sempere et al., 2004; Van Rooij et al., 2009; Small et al., 2010). MiR-206 is specifically expressed only in skeletal muscle, and its targeted circRNAs (circ0001651, circ0010874, circ0010882, circ0010890, circ0010896, circ0012793) were not significantly differentially expressed. Whether these circRNAs have specific functions and what their functional mechanisms are need further study.
Conclusion
In this study, we investigated the expression of circRNAs in muscle tissues of different breeds of beef cattle, obtained 655 differentially expressed circRNAs and 467 parental genes, selected 15 circRNAs related to muscle development as candidate circRNAs, and predicted the target miRNAs. These findings may provide clues for further research on muscle development in different breeds of beef cattle.
Data Availability Statement
We have stored the RNA sequence data in the public domain GEO NCBI, GEO accession numbers: SRR11819095, SRR11819094, SRR11819093, SRR11819092, SRR11819091, and SRR11819090.
Ethics Statement
The animal study was reviewed and approved by the Animal Experiments of Qingdao Agricultural University IACUC (Institutional Animal Care and Use Committee).
Author Contributions
YD, XB, CX, and RL designed the study. RL and XL conducted the experiment. RL, XL, and XB performed and collected the data. RL analyzed the data and wrote the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by the Shandong Modern Agricultural Industry Technology System Cattle Industry Innovation Team (668-2216030).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This manuscript has been released as a pre-print at Epigenetics & Genomics, Liu et al. (2020).
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgene.2020.565085/full#supplementary-material
Abbreviations
“HE staining”, hematoxylin and eosin staining; GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes; PPI, Protein-protein interaction networks; SRPBM, spliced reads per billion mapping; RNA-seq, RNA sequencing technology; qRT-PCR, real-time quantitative PCR; SPONG, the interaction network between miRNA and circRNA.
Footnotes
- ^ http://www.usadellab.org/cms/index.php?page=Trimmomatic
- ^ http://tophat.cbcb.umd.edu/
- ^ https://www.ncbi.nlm.nih.gov/genome/82?genomeassemblyID=371813
- ^ http://www.microrna.org/microrna/home.do
- ^ https://pypi.org/project/goatools/
References
Ashwal-Fluss, R., Meyer, M., Pamudurti, N. R., Ivanov, A., Bartok, O., Hanan, M., et al. (2014). circRNA Biogenesis Competes with Pre-mRNA Splicing. Mol. Cell 56, 55–66. doi: 10.1016/j.molcel.2014.08.019
Cao, T., Shi, L. G., and Zhang, L. L. (2014). Comparative Study on Fetal Muscle Fiber of Wuzhishan Pig and Changbai Pig during 65d Gestation. J. Anim. Ecol. 35, 37–40.
Cong, X. X. (2018). The Role and Regulation Mechanism of mTOR Signaling Pathway in Muscle Regeneration and Tendon Differentiation. Zhejiang: Zhejiang University.
Fernández-Martos, C. M., González-Fernández, C., González, P., Maqueda, A., Arenas, E., Rodríguez, F. J., et al. (2011). Differential Expression of Wnts after Spinal Cord Contusion Injury in Adult Rats. PLoS One 6:e27000. doi: 10.1371/journal.pone.0027000
Hong-Bo, F., Zhen-Ya, Z., Xiang-Guang, L., Chun-Qi, G., Hui-Chao, Y., Zhe-Sheng, C., et al. (2017). CDX2 Stimulates the Proliferation of Porcine Intestinal Epithelial Cells by Activating the mTORC1 and Wnt/β-Catenin Signaling Pathways. Int. J. Mol. Sci. 18:2447. doi: 10.3390/ijms18112447
Horak, M., Novak, J., and Bienertova-Vasku, J. (2016). Muscle-specific microRNAs in skeletal muscle development. Dev. Biol. 410, 1–13. doi: 10.1016/j.ydbio.2015.12.013
John, B., Enright, A.J., Aravin, A., Tuschl, T., Sander, C., Marks, D., et al. (2004). Human MicroRNA Targets. PLoS Biol. 2:e363. doi: 10.1371/journal.pbio.0020363
Kolakofsky, D. (1976). Isolation and characterization of Sendai virus DI-RNAs. Cell 8, 547–555. doi: 10.1016/0092-8674(76)90223-3
Legnini, I., Di, G., Rossi, F., Morlando, M. A., Briganti, F., Sthandier, O., et al. (2017). Circ-ZNF609 Is a Circular RNA that Can Be Translated and Functions in Myogenesis. Mol. Cell 66, 22.e–37.e.
Li, H., Wei, X., Yang, J., Dong, D., Hao, D., Huang, Y., et al. (2018). CircFGFR4 Promotes Differentiation of Myoblasts via Binding miR-107 to Relieve Its Inhibition of Wnt3a. Mol. Ther. Nucl. Acids 11, 272–283. doi: 10.1016/j.omtn.2018.02.012
Li, H., Yang, J., Wei, X., Song, C., Dong, D., Huang, Y., et al. (2017). CircFUT10 reduces proliferation and facilitates differentiation of myoblasts by sponging miR-133a. J. Cell. Physiol. 233, 4643–4651. doi: 10.1002/jcp.26230
Li, Y. L. (2018). The role and mechanism of PPAR signaling pathway in regulating the differential deposition of subcutaneous fat and intramuscular fat in pigs. Xianyang: Northwest A & F University.
Liu, R. L., Liu, X. X., Bai, X. J., Xiao, C. Z., and Dong, Y. J. (2020). Identification and characterization of circRNA during muscle development in different breeds of cattle. Preprint.
Love, M. I., Huber, W., and Anders, S. (2014). Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15:550.
Memczak, S., Jens, M., Elefsinioti, A., Torti, F., Krueger, J., Rybak, A., et al. (2013). Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 495, 333–338. doi: 10.1038/nature11928
O’Connell, T. D., Jensen, B. C., Baker, A. J., and Simpson, P. C. (2013). Cardiac Alpha1-Adrenergic Receptors: Novel Aspects of Expression, Signaling Mechanisms, Physiologic Function, and Clinical Importance. Pharmacol. Rev. 66, 308–333. doi: 10.1124/pr.112.007203
Polge, C., Koulmann, N., Claustre, A., Jarzaguet, M., Serrurier, B., Combaret, L., et al. (2016). UBE2D2 is noninvolved in MuRF1-dependent muscle wasting during hindlimb suspension. Int. J. Biochem. Cell Biol. 79, 488–493. doi: 10.1016/j.biocel.2016.06.019
Qu, S., Yang, X., Li, X., Wang, J., Gao, Y., Shang, R., et al. (2015). Circular RNA: a new star of noncoding RNAs. Cancer Lett. 365, 141–148. doi: 10.1016/j.canlet.2015.06.003
Ravenscroft, G., Zaharieva, I., Bortolotti, C. A., Lambrughi, M., Pignataro, M., Borsari, M., et al. (2018). Bi-allelic mutations in MYL1 cause a severe congenital myopathy. Hum. Mol. Genet. 27, 4263–4272.
Rong, D., Sun, H., Li, Z., Liu, S., Dong, C., Fu, K., et al. (2017). An emerging function of circRNA-miRNAs-mRNA axis in human diseases. Oncotarget 8, 73271–73281. doi: 10.18632/oncotarget.19154
Sanger, H. L., Klotz, G., Riesner, D., Hans, J., and Kleinschmidt, G. A. K. (1976). Viroids are single-stranded covalently closed circular RNA molecules existing as highly base-paired rod-like structures. Proc. Natl. Acad. Sci. 73, 3852–3856. doi: 10.1073/pnas.73.11.3852
Sempere, L. F., Freemantle, S., Pitha-Rowe, I., Moss, E., Ambros, V., et al. (2004). Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 5:R13.
Small, E. M., O”Rourke, J. R., Moresi, V., Sutherland, L. B., McAnally, J., Gerard, R. D., et al. (2010). Regulation of PI3-Kinase/Akt Signaling by Muscle-enriched microRNA-486. Proc. Natl. Acad. Sci. U S A. 107, 4218–4223. doi: 10.1073/pnas.1000300107
Traurig, M., Ossowski, V., Fleming, J. M., Mullins, M., and Baier, L. J. (2013). Human adipose microRNA-221 is upregulated in obesity and affects fat metabolism downstream of leptin and TNF-α. Diabetologia 56, 1971–1979. doi: 10.1007/s00125-013-2950-9
Uhler, J. P., Spåhr, H., Farge, G., Clavel, S., Larsson, N. G., Falkenberg, M., et al. (2014). The UbL protein UBTD1 stably interacts with the UBE2D family of E2 ubiquitin conjugating enzymes. Biochem. Biophys. Res. Commun. 443, 7–12. doi: 10.1016/j.bbrc.2013.10.137
Van Rooij, E., Quiat, D., Johnson, B. A., Sutherland, L. B., Qi, X., Richardson, J. A., et al. (2009). A Family of microRNAs Encoded by Myosin Genes Governs Myosin Expression and Muscle Performance. Dev. Cell 17, 0–673.
Wei, X., Li, H., Yang, J., Hao, D., Dong, D., Huang, Y., et al. (2017). Circular RNA profiling reveals an abundant circLMO7 that regulates myoblasts differentiation and survival by sponging miR-378a-3p. Cell Death Dis. 8:3153.
Zeng, Y. Y. (2016). Effect of Low MEF2C Expression on C2C12 Myofibroblast Fiber Composition and Molecular Mechanism. Jinzhou: Jinzhou Medical University.
Zhang, B. W., Cai, H. F., Wei, X. F., Sun, J. J., Lan, X. Y., Lei, C. Z., et al. (2016). miR-30-5p Regulates Muscle Differentiation and Alternative Splicing of Muscle-Related Genes by Targeting MBNL. Int. J. Mol. Sci. 17:182. doi: 10.3390/ijms17020182
Zheng, J., Liu, X., Xue, Y., Gong, W., Ma, J., Xi, Z., et al. (2017). TTBK2 circular RNA promotes glioma malignancy by regulating miR-217/HNF1β/Derlin-1 pathway. J. Hematol. Oncol. 10:52.
Keywords: Luxi cattle, circRNA, skeletal muscle, Shandong black cattle, identification
Citation: Liu R, Liu X, Bai X, Xiao C and Dong Y (2020) Identification and Characterization of circRNA in Longissimus Dorsi of Different Breeds of Cattle. Front. Genet. 11:565085. doi: 10.3389/fgene.2020.565085
Received: 23 June 2020; Accepted: 27 October 2020;
Published: 26 November 2020.
Edited by:
Xianyong Lan, Northwest A&F University, ChinaReviewed by:
XiuKai Cao, Yangzhou University, ChinaBinglin Yue, Northwest A&F University, China
Chengchuang Song, Northwest A&F University, China
Copyright © 2020 Liu, Liu, Bai, Xiao and Dong. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yajuan Dong, etcenter@126.com
 Ruili Liu1
Ruili Liu1 Xianxun Liu
Xianxun Liu Yajuan Dong
Yajuan Dong