- Department of Neurobiology, University of Alabama at Birmingham, Birmingham, AL, United States
The expression of genetic material governs brain development, differentiation, and function, and targeted manipulation of gene expression is required to understand contributions of gene function to health and disease states. Although recent improvements in CRISPR/dCas9 interference (CRISPRi) technology have enabled targeted transcriptional repression at selected genomic sites, integrating these techniques for use in non-dividing neuronal systems remains challenging. Previously, we optimized a dual lentivirus expression system to express CRISPR-based activation machinery in post-mitotic neurons. Here we used a similar strategy to adapt an improved dCas9-KRAB-MeCP2 repression system for robust transcriptional inhibition in neurons. We find that lentiviral delivery of a dCas9-KRAB-MeCP2 construct driven by the neuron-selective human synapsin promoter enabled transgene expression in primary rat neurons. Next, we demonstrate transcriptional repression using CRISPR sgRNAs targeting diverse gene promoters, and show superiority of this system in neurons compared to existing RNA interference methods for robust transcript specific manipulation at the complex Brain-derived neurotrophic factor (Bdnf) gene. Our findings advance this improved CRISPRi technology for use in neuronal systems for the first time, potentially enabling improved ability to manipulate gene expression states in the nervous system.
Introduction
Brain function and development relies on tightly coordinated transcriptional programs, and dysregulated gene expression patterns are linked to many neurological and psychological disorders (Xu et al., 2006; Liang et al., 2008; McClung and Nestler, 2008; De Jong et al., 2012; Glatt et al., 2012; Lewis and Cookson, 2012; Campbell et al., 2013; Winkler and Fox, 2013; Alberini and Kandel, 2014; Bettencourt et al., 2014; Pramparo et al., 2015; MacMullen et al., 2016; Woo et al., 2018; Yap and Greenberg, 2018). Understanding the functional relevance and contributions of divergent expression states requires the ability to manipulate gene expression in a targeted and specific manner. A classic approach is to delete an individual candidate gene implicated in a biological process or disease state and characterize the resulting phenotype. Although powerful, this approach often carries significant drawbacks (Eisener-Dorman et al., 2009), such as those arising from genetic background (Crusio, 2004), genetic compensation unrelated to the loss of the targeted gene's protein function (El-Brolosy and Stainier, 2017), an inability to assess genes for which a knockout is lethal, and technical challenges in achieving homogenous knockouts at all loci due to ploidy (Boettcher and McManus, 2015). It is also often difficult to distinguish the functional role of a gene of interest in behavior from its role in development (Eisener-Dorman et al., 2009) due to the irreversibility of genetic manipulations (Boettcher and McManus, 2015). Further, this approach suffers from an inherent lack of specificity in cases where the entire genomic locus is perturbed rather a specific transcript isoform. RNA interference (RNAi) based methods of gene suppression can overcome many of these challenges, but also possess extensive sequence-dependent and sequence-independent off-target effects (Bridge et al., 2003; Jackson et al., 2003; Sledz et al., 2003; Fish and Kruithof, 2004; Khan et al., 2009; Ui-Tei, 2013; Olejniczak et al., 2016). These limitations can make interpretations of knockout and RNAi studies challenging, and highlight the need for more robust strategies to manipulate gene expression (Bridge et al., 2003; Sledz et al., 2003; Fish and Kruithof, 2004; Judge et al., 2005; Read et al., 2009; Ui-Tei, 2013; Olejniczak et al., 2016).
Advances in CRISPR/Cas9 technology have revolutionized functional investigations of gene expression (Heidenreich and Zhang, 2016; Huang et al., 2018; Knott and Doudna, 2018; Zhang, 2019). Cas9, an RNA-directed endonuclease, can be localized to selected genomic sites with a single guide RNA (sgRNA) complementary to the DNA location of interest, provided a protospacer adjacent motif (PAM) sequence is nearby (5′-NGG-3' for the widely used Streptococcus pyogenes Cas9). Once trafficked, it will induce a double stranded break and can thereby be used to create targeted permanent genomic alterations (Jiang and Doudna, 2017). Mutation of the catalytic domain of Cas9 to generate nuclease-dead Cas9 (dCas9) preserves its homing function and also enables a diverse array of applications (Liu et al., 2016; Savell and Day, 2017; Pickar-Oliver and Gersbach, 2019; Zhang, 2019). For example, dCas9 targeted to the transcriptional start sites of genes physically impedes the transcription process without altering the DNA sequence itself, a process termed CRISPR interference (CRISPRi) (Qi et al., 2013). This strategy can be preferable to Cas9 approaches when assaying gene expression due to the avoidance of cellular toxicity arising from the induced double-stranded DNA breaks, the increased specificity of altering transcription while preserving the genetic structure, and the potential reversibility of the resultant transcriptional modifications (Gilbert et al., 2013, 2014; Qi et al., 2013; La Russa and Qi, 2015; Mandegar et al., 2016; Thakore et al., 2016; Martella et al., 2019). Subsequent efforts have improved the transcriptional silencing function of CRISPRi via fusion of potent transcriptional inhibitors to dCas9 (Gilbert et al., 2013; Pickar-Oliver and Gersbach, 2019), such as a Krüppel-associated box (KRAB) repressor domain. While this approach has been adopted for targeted as well as multiplexed gene silencing in the nervous system (Zheng et al., 2018), these approaches have not led to consistent gene silencing at other targets (Savell et al., 2019a), highlighting the need for continued optimization.
A recent unbiased screen comparing multiple dCas9 fusion systems demonstrated robust and selective gene repression via fusion of a dimer containing a KRAB repressor domain in addition to methyl-CpG binding protein 2 (MeCP2). CRISPRi with dCas9-KRAB-MeCP2 resulted in significant improvement over existing dCas9 interference approaches, as well as gene knockdown using classic RNAi technology (Yeo et al., 2018). However, the adoption of this technology for use in neuronal systems requires surpassing significant barriers due to difficulties in transgene delivery and nuclear localization in post-mitotic neurons (Suzuki et al., 2016; Savell and Day, 2017; Savell et al., 2019a). Recently, we established an approach for CRISPR activation (CRISPRa) to achieve specific, robust, and multiplexable gene induction in neurons both in vitro and in vivo by engineering an optimized dual-lentivirus technique (Savell et al., 2019a). Here, we apply these advances to translate this second-generation dCas9-KRAB-MeCP2 CRISPRi technology for functional use in neuronal systems.
Methods and Materials
Animals
All experiments were performed in accordance with the University of Alabama at Birmingham Institutional Animal Care and Use Committee. Sprague-Dawley timed pregnant rat dams were purchased from Charles River Laboratories. Dams were individually housed until embryonic day 18 for cell culture harvest in an AAALAC-approved animal care facility on a 12-h light/dark cycle with ad libitum food and water.
Primary Rat Brain Cultures
Primary rat cell cultures were generated from embryonic day 18 (E18) rat striatal and hippocampal tissue, as described previously (Day et al., 2013; Savell et al., 2016, 2019a). Briefly, cell culture plates (Denville Scientific Inc.) were coated overnight with poly-L-lysine (Sigma-Aldrich; 50 μg/ml) supplemented with laminin (Sigma-Aldrich; 7.5 μg/mL) and rinsed with diH2O. Dissected cortical or hippocampal tissue was incubated with papain (Worthington LK003178) for 25 min at 37°C. After rinsing in complete Neurobasal media (Neurobasal Medium (Gibco; #21103049), supplemented with B27 (Gibco; #17504044, 1X concentration) and L-glutamine (Gibco; # 25030149, 0.5 mM), a single cell suspension was prepared by sequential trituration through large to small fire-polished Pasteur pipettes and filtered through a 100 μm cell strainer (Fisher Scientific). Cells were pelleted, re-suspended in fresh media, counted, and seeded to a density of 125,000 cells per well on 24-well culture plates (65,000 cells/cm2). Cells were grown in complete Neurobasal media for 11 days in vitro (DIV 11) in a humidified CO2 (5%) incubator at 37°C with half media changes at DIV 1 and 10. Lentiviral transduction occurred on either DIV 4 or 5 when 330 μl of culture media was removed from each culture well and lentivirus was delivered for an incubation period of 8–12 h. Following this transduction period, 600 μl of fresh complete Neurobasal media wash occurred before replacement with a mixture of 300 μl of fresh complete Neurobasal and 300 μl of the culture media removed prior to transduction. On DIV 11, media was removed and RNA extraction either occurred immediately or the culture plate was stored at −80°C for RNA extraction at a later date. For the Reln targeting experiment, data is shown from neurons that received media supplementation (10 μL Neurobasal media) 1 h prior to RNA extraction.
HEK293T Cell Line
HEK293T cells were obtained from American Type Culture Collection (ATCC catalog #CRL-3216, RRID:CVCL_0063) and were maintained in standard HEK Media: DMEM (DMEM High glucose, pyruvate; Gibco 11995081) + 10% FBS (Qualified US Origin; BioFluid 200-500-Q) + 1U Penicillin-Streptomycin (Gibco 15140122). Cells were passaged in T75 or T225 tissue culture flasks at 70–80% confluence no more than 25 times and were checked for mycoplasma contamination periodically. HEK293T cells were utilized in luciferase assay experiments and for lentiviral production.
Plasmid Design and Construction
To generate a lentivirus-compatible dCas9-KRAB-MeCP2 construct capable of robust neuronal expression, a Gibson assembly cloning strategy was performed using the XhoI and EcoRI restriction sites in our previously published lenti SYN-FLAG-dCas9-VPR backbone (Addgene plasmid #114196 Savell et al., 2019a,b, 2020), substituting VPR for KRAB-MeCP2 via PCR amplification from the original dCas9-KRAB-MeCP2 vector [a gift from Alejandro Chavez & George Church (Addgene plasmid #110821 Yeo et al., 2018). The full sequence of the updated lentivirus compatible SYN-dCas9-KRAB-MeCP2 is provided in Supplementary Material. SYN-dCas9 (generated in this study) and SYN-KRAB-dCas9/EGFP (previously published Savell et al., 2019a) construct sequences used in luciferase experiments are also provided in Supplementary Material. The SYN-dCas9 construct was generated from the lenti SYN-FLAG-dCas9-VPR backbone construct described above.
A Fos-Firefly Luciferase reporter plasmid was generated via amplification of a portion of the Fos promoter (−722bp to +97bp of the Rn6 annotated Fos transcription start site) from rat genomic DNA. PCR amplification occurred using the forward primer tgctagtggatccTTGTAGGTAAAGCGGGTTATTGA and reverse primer tgctagtaagcttGGGTAGACACTGGTGGGA, followed by a BamHI or HindIII digestion and ligation reaction to insert this sequence upstream of a firefly luciferase construct (a gift from Michael Rehli). The full sequence of the Fos-Firefly Luciferase reporter construct is found in Supplementary Material.
All sgRNAs used here were cloned into our previously published lenti U6-sgRNA/EF1α-mCherry vector using BbsI restriction digest and sense/antisense oligos containing the target sequence, as previously described [Addgene plasmid #114199 (Savell et al., 2019a,b, 2020)]. New sgRNAs generated for this manuscript were designed using the online tool ChopChop v3 (https://chopchop.cgu.uib.no; Labun et al., 2019) using search options for CRISPRa or CRISPRi, screened against the Rn6 rat genome assembly. With few exceptions, promoter-targeted sgRNAs were restricted to within 500bp upstream of the target gene TSS for CRISPRa, and ± 500 bp of the TSS for CRISPRi. All sgRNAs were examined for genome-wide sequence specificity using Cas-OFFinder (Bae et al., 2014). Information related to CRISPR sgRNA sequences, genomic targets, targeting strand, distance from the gene TSS, original design strategy (CRISPRa or CRISPRi), and potential off-target sites identified from Cas-OFFinder (Bae et al., 2014) is presented in Supplementary Table 1. shRNA construction followed a similar approach, utilizing the Broad TRC shRNA design tool (http://portals.broadinstitute.org/gpp/public/) and inserted into a lenti U6-shRNA/EF1α-mCherry vector (Zipperly et al., 2020). shRNAs were assessed for specificity using BLAST. A list of all shRNA target sequences is provided in Supplementary Table 1.
Luciferase Assay
80,000 HEK293T cells were plated in 500 μl HEK Media and 24 h later 500 ng total plasmid DNA was transfected with 1.5 μl FuGene HD (Promega) as follows: 50 ng of luciferase plasmid, 450 ng in 1:2 molar ratio of total sgRNA:dCas9-KRAB-MeCP2. 24 h following transfection, a luciferase glow assay was performed according to manufacturer's instructions (Thermo Scientific Pierce Firefly Glow Assay; Thermo Scientific 16177). Briefly, cells were lysed in 200 μl 1x Cell Lysis Buffer while shaking at low speed and protected from light for 45 min. 20 μl of lysate was then added to an opaque 96-well microplate (Corning 353296) and combined with 50 μl 1x D-Luciferin Working Solution supplemented with 1x Firefly Signal Enhancer (Thermo Scientific Pierce Firefly Signal Enhancer; Thermo Scientific 16180). Following a 10 min dark incubation period to allow for signal stabilization, luminescence was recorded using a Synergy 2 Multi-Detection Microplate Reader (BioTek) with a read height of 1 mm, a 1 s integration time, and a 100 ms delay.
Lentivirus Production
Packaging and concentration of the dCas9-KRAB-MeCP2, sgRNA, and shRNA constructs into lentiviruses occurred as described previously (Savell et al., 2019a). Under sterile BSL-2 conditions, HEK293T cells were transfected with either the dCas9-KRAB-MeCP2, sgRNA, or shRNA constructs in combination with the psPAX2 packaging and pCMV-VSV-G envelope plasmids (Addgene plasmid #12260 and #8454) using FuGene HD (Promega) in fresh HEK media. 48 h following transfection, supernatant was removed, large debris were removed by a 10 min spin at 2,300 rcf, followed by filtration through a 0.45 μm filter, and centrifugation for 1 h 45 min at 106,883 rcf at 4°C. The viral pellet was allowed to resuspend in sterile PBS at 4°C overnight and stored at −80°C. Genomic titer was determined using the Lenti-Z qRT-PCR Titration Kit according to manufacturer's instructions (Takara #631235). Smaller scale virus preparation for sgRNAs and shRNAs was performed through a similar transfection in a 12-well culture plate. After 48 hrs lentiviruses were concentrated with Lenti-X concentrator (Takara), resuspended in sterile PBS over 24–48 h and used immediately. dCas9-KRAB-MeCP2 lentivirus was used at a genomic titer of >2,000 genomic copies/cell unless otherwise indicated in combination with sgRNAs either concentrated to >1,000 genomic copies/cell or produced in small scale preparation as indicated above and divided across 3 culture plate wells. shRNA-expressing lentiviruses were produced using the small-scale preparation described above and divided across 2 culture well per experiment. All viral titers reported here are physical titers (GC/ml), not functional titers (e.g., TU/ml, another commonly reported measure of viral titer). The multiplicity of infection used in our experiments (1,000–4,000 GC/cell) accounts for 10–100-fold differences in physical and functional titer.
Immunocytochemistry
After removal of neuronal culture media, cells were washed with PBS and incubated at room temperature for 20 min in freshly prepared 4% paraformaldehyde in PBS. After fixation, cells were washed twice with PBS and neuronal membranes were permeabilized with PBS containing 0.25% Triton X-100 for 15 min at room temperature. Cells were then washed three times in PBS, blocked for 1 h (10% Thermo Blocker bovine serum albumin (BSA) #37525, 0.05% Tween-20, and 300 mM glycine in PBS) and co-incubated with DYDDDDK Tag (FLAG epitope) Monoclonal Antibody (FG4R) (1:5,000 in PBS with 10% Thermo Blocker BSA; Thermo Fisher catalog #MA1-91878, RRID: AB_1957945) overnight at 4°C. Cells were then washed three times with PBS before a 45 min incubation in IRDye 680RD Goat anti-Mouse IgG Secondary Antibody (1:250 in PBS with 10% Thermo Blocker BSA; Li-Cor catalog #925-68070, RRID: AB_2651128). Cells were then washed a final three times with PBS for 5 min. Slide covers slips with Prolong Gold anti-fade medium (Invitrogen) containing 4,6-diamidino-2-phenylindole (DAPI) stain were placed atop the culture wells. A Nikon TiS inverted fluorescent microscope was used to capture 10X magnification (1,888 mm2 field of view) images from a 24-well culture plate.
RNA Extraction and RT-qPCR
Total RNA was extracted (RNAeasy kit, Qiagen) and reverse-transcribed (iScript cDNA Synthesis Kit, Bio-Rad) following the manufacturers' instructions. cDNA was subject to RT-qPCR for genes of interest in duplicate using a CFX96 real-time PCR system (Bio-Rad) at 95°C for 3 min, followed by 40 cycles of 95°C for 10 s and 58°C for 30 s, followed by real-time melt analysis to verify product specificity, as described previously (Savell et al., 2016, 2019a; Duke et al., 2020). Gapdh was used for normalization via the ΔΔCt method (Livak and Schmittgen, 2001). A list of PCR primer sequences is provided in Supplementary Table 1.
Statistical Analysis
Gene expression differences from RT-qPCR experiments were compared with either an unpaired t-test or One- or Two-way ANOVA with Tukey's or Sidak's post-hoc test where appropriate. Statistical significance was designated at α = 0.05 for all analyses. Statistical and graphical analyses were performed with Prism software (GraphPad). Statistical assumptions (e.g., normality and homogeneity for parametric tests) were formally tested and examined via boxplots except in the MOI experiment presented in Figure 2B where low sample sizes (n = 2 per group) prevented this analysis.
Results
A SYN-Driven dCas9-KRAB-MeCP2 Construct Suppresses Gene Expression at a Luciferase Reporter in HEK293T Cells
As highlighted in previous studies, catalytically inactivated Cas9 (dCas9) fusion systems recruiting KRAB and MeCP2 domains provide improved gene-specific transcriptional knockdown (CRISPRi) in mammalian cell lines (Yeo et al., 2018; Xiong et al., 2019; Figure 1A). To leverage this second-generation construct for efficient use in neuronal systems, we adopted a dual lentiviral approach segregating the dCas9-KRAB-MeCP2 and the sgRNA onto separate vectors (Figure 1B). This approach enables efficient packaging of the large dCas9-KRAB-MeCP2 fusion with the robust neuron-selective human synapsin 1 promoter (SYN) and provides the flexibility to readily exchange and combine sgRNA scaffold constructs (Savell et al., 2019a). To confirm functionality of the dCas9-KRAB-MeCP2 construct, we engineered a luciferase reporter system containing firefly luciferase positioned downstream of the truncated rat Fos promoter in HEK293T cells. Transfecting sgRNAs designed to target the adapted rat Fos promoter region produced robust suppression of luciferase signal relative to control sgRNAs targeted to the bacterial gene lacZ (a sequence that does not exist in the mammalian genome; Figure 1C), demonstrating that the adapted SYN-dCas9-KRAB-MeCP2 fusion system produces efficient targeted gene suppression that translates into a loss of functional protein. The SYN-dCas9-KRAB-MeCP2 fusion system achieved more robust luciferase knockdown (83.3%) relative to SYN-dCas9 (17.8%) and SYN-KRAB-dCas9/EGFP (68.2%) fusions when compared against individual lacZ sgRNA controls. This confirms and supports findings from Yeo et al. demonstrating that the updated dCas9-KRAB-MeCP2 construct is often more effective at targeted gene suppression than dCas9 or dCas9-KRAB only fusions (Yeo et al., 2018).
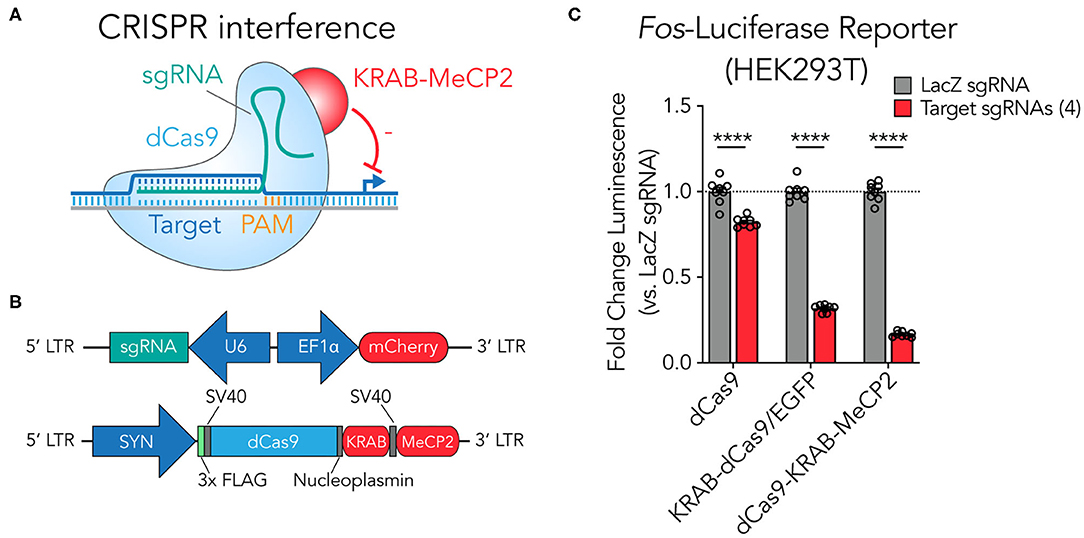
Figure 1. A SYN-driven dCas9-KRAB-MeCP2 construct suppresses gene expression at a luciferase reporter in HEK293T cells. (A) Illustration of the dCas9-KRAB-MeCP2 suppression strategy. An sgRNA with a spacer complementary to the targeted genomic site adjacent to a PAM motif directs the dCas9-KRAB-MeCP2 transcriptional suppresser to targeted genetic loci. (B) The dual vector lentiviral construct designs. The U6 polymerase 3 promoter drives expression of the sgRNA which can be adapted to target specific genetic loci. The EF1α promoter drives expression of mCherry, useful for rapid assessment of lentiviral expression. The second construct contains the dCas9-KRAB-MeCP2 fusion driven by the neuron-selective SYN promoter. This fusion contains a FLAG epitope so that construct expression can be visualized readily by immunocytochemistry. (C) Luciferase assay confirms luciferase reporter suppression (from 4 multiplexed targeting sgRNAs) by the dCas9-KRAB-MeCP2 system in HEK293T cells relative to non-targeted (lacZ) sgRNA controls, with more robust repression than first-generation CRISPRi tools [dCas9 alone or KRAB-dCas9/EGFP; n = 8, unpaired t-test; dCas9 t(14) = 6.602, p = 0.000012; KRAB-dCas9/EGFP t(14) = 32.89, p < 0.000001; dCas9-KRAB-MeCP2 t(14) = 42.14, p < 0.000001]. All data are expressed as mean ± s.e.m. Individual comparisons; ****p < 0.0001.
dCas9-KRAB-MeCP2 Is Capable of Strong Gene Suppression at Multiple Genes in Primary Neuronal Cultures
To examine functionality of the adapted dCas9-KRAB-MeCP2 system in neuronal systems, plasmid constructs were packaged into high-titer lentiviruses (5.47*1010 to 1.91*1011 genomic copies per mL) and transduced into primary rat neuronal cultures (Figure 2). The dCas9-KRAB-MeCP2 fusion was engineered to contain the FLAG epitope, which readily enables immunocytochemistry (ICC) visualization studies. ICC against the FLAG epitope indicated neuronal expression and nuclear localization of the dCas9-KRAB-MeCP2 fusion in rat primary striatal cultures (Figure 2A). To confirm sgRNA-targeted repression and better understand the lentiviral load required when utilizing this system in neurons, we varied the lentiviral genomic copies delivered per cell while targeting the immediate early gene Fosb in primary rat hippocampal cultures (Figure 2B). A Two-way ANOVA revealed a significant main effect of sgRNA for the Fosb groups relative to lacZ controls, suggesting effective gene silencing. In contrast, there was no main effect of lentiviral genome copy number, indicating that 1,000 genome copies per cell was sufficient for primary neuronal culture experiments. To examine the capability of this system to downregulate genes of diverse functional classes, we developed and employed sgRNAs to traffic the system to the lysine methyltransferase Kmt2b, the extracellular matrix protein Reln, and the signaling neuropeptide Npy (Figure 2C). Comparison of gene expression with gene-specific RT-qPCR primer sets revealed significant decreases in gene expression at each target compared to lacZ control sgRNAs, with no significant effect at non-targeted genes. These results confirm that robust targeted gene repression can be achieved using the dCas9-KRAB-MeCP2 system across diverse gene targets in neuronal systems.
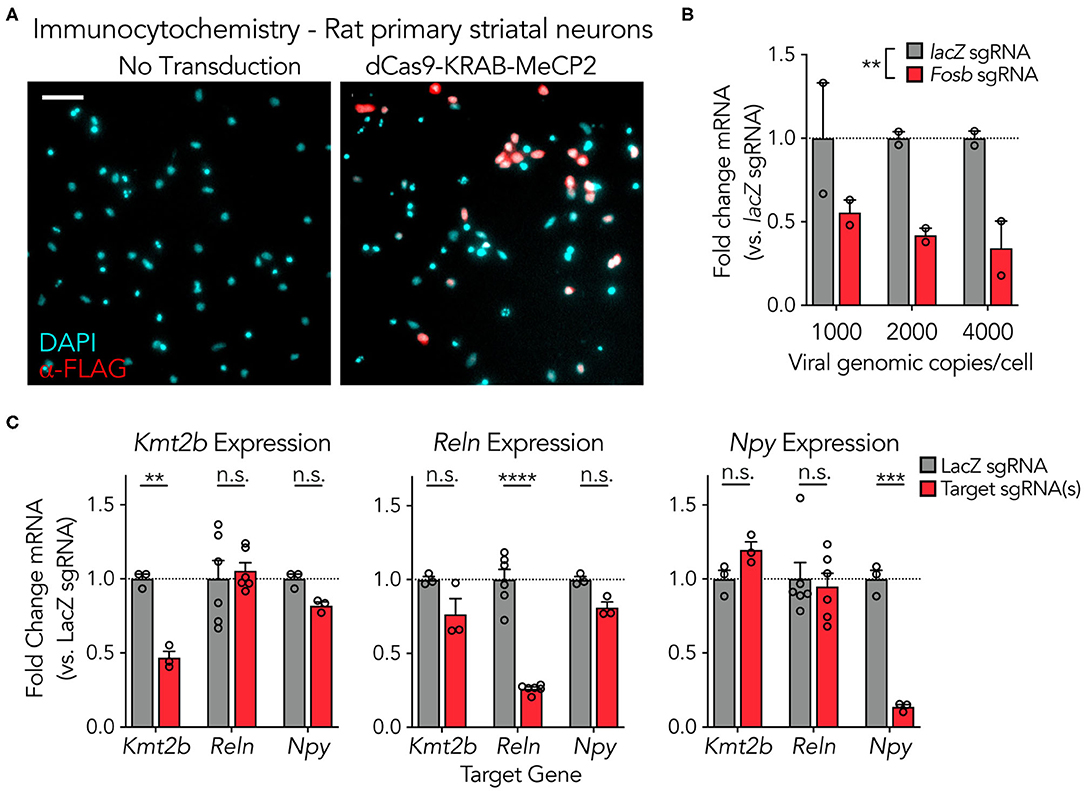
Figure 2. dCas9-KRAB-MeCP2 is capable of strong gene suppression at multiple genes in primary neuronal cultures. (A) Immunocytochemistry demonstrating expression of the lentiviral SYN-driven dCas9-KRAB-MeCP2 construct in primary rat striatal cultures. Scale bar = 200 μm. (B) dCas9-KRAB-MeCP2 induces targeted gene suppression at Fosb (recruited by an individual sgRNA) relative to lacZ sgRNA controls in primary rat hippocampal cultures as revealed by RT-qPCR. There was no main effect of viral genome copies per cell [n = 2, Two-Way ANOVA; sgRNA F(1,6) = 19.17, p = 0.0047; viral genomic copies per cell F(2,6) = 0.2380, p = 0.7953, interaction F(2,6) = 0.2380, p = 0.7953]. (C) RT-qPCR demonstrates targeted gene suppression in striatal cultures relative to non-targeted controls across multiple genes, with either 6 sgRNAs targeting Kmt2b, 1 sgRNA targeting Reln, or 3 sgRNAs targeting Npy [n = 3–6, Two-Way ANOVA; left (Kmt2b expression): lacZ vs targeted sgRNA(s) F(1,18) = 8.088, p = 0.0108, and gene targeted F(2,18) = 5.400, p = 0.0146, Sidak's multiple comparisons test: Kmt2b t(18) = 3.642, p = 0.0056; Reln t(18) = 0.5278, p = 0.9380; Npy t(18) = 1.228, p = 0.5529; middle (Reln expression); lacZ vs. targeted sgRNA(s) F(1,18) = 61.82, p < 0.0001, and gene targeted F(2,18) = 15.92, p = 0.0001, Sidak's multiple comparisons test: Kmt2b t(18) = 2.511, p = 0.0640; Reln t(18) = 11.17, p < 0.0001; Npy t(18) = 2.022, p = 0.1648; right (Npy expression); lacZ vs targeted sgRNA(s) F(1,18) = 8.160, p = 0.0105, and gene targeted F(2,18) = 12.67, p = 0.0004, Sidak's multiple comparisons test: Kmt2b t(18) = 1.237, p = 0.5470; Reln t(18) = 0.4606, p = 0.9573; Npy t(18) = 5.428, p = 0.0001]. All data are expressed as mean ± s.e.m. Individual comparisons, **p < 0.01, ***p < 0.001, ****p < 0.0001.
dCas9-KRAB-MeCP2 Induces Targeted Transcript-Specific Bdnf Gene Repression in Primary Rat Hippocampal Cultures
Gene expression in neuronal systems is complex, with many genes utilizing alternative splicing in critical ways to control development and synaptic plasticity (Raj and Blencowe, 2015; Vuong et al., 2016; Le François et al., 2018; Weyn-Vanhentenryck et al., 2018). One example of this complexity occurs at the brain-derived neurotrophic factor gene Bdnf, which uses divergent non-coding exons (1-Xa) combined with a single coding exon (IX) in multiple alternatively spliced forms produced from 9 distinct gene promoters (Figure 3A; Aid et al., 2007). The most highly expressed transcripts employ either the non-coding exon I or IV, and the specific functionality of this transcript heterogeneity remains an area of active investigation (Maynard et al., 2017, 2018; Hallock et al., 2019; Savell et al., 2019a). We designed short hairpin RNAs (shRNAs) and CRISPRi sgRNAs to compare these targeted transcript suppression methods at Bdnf I and Bdnf IV transcripts. shRNA targeting of the Bdnf I transcript was unsuccessful at decreasing its expression, but instead resulted in an unintended decrease in the expression of Bdnf IV (Figure 3B). Bdnf IV shRNA successfully repressed Bdnf IV expression and resulted in a surprising increase of Bdnf I transcript expression, perhaps as a result of genetic compensation for Bdnf IV knockdown. Both shRNAs failed to reduce total coding Bdnf levels as measured by RT-qPCR primers located in the Bdnf IX coding region. Taken together, these findings suggest that while capable of some transcript-specific knockdown, these shRNAs produced only modest effects at the targeted transcripts, a feature commonly observed in RNAi strategies (Boettcher and McManus, 2015; La Russa and Qi, 2015). To examine if a dCas9-KRAB-MeCP2 strategy could improve transcript-specific knockdown levels, we next employed individual sgRNAs designed to target regions upstream of the Bdnf I and Bdnf IV exons in hippocampal neurons (Figure 3C). Strikingly, dCas9-KRAB-MeCP2 targeting induced a 96% transcriptional knockdown at Bdnf I and a 92% knockdown at Bdnf IV relative to lacZ controls. Interestingly, targeting the Bdnf IV upstream region did not affect levels of Bdnf I, but targeting the Bdnf I upstream region resulted in a 21.9% reduction of Bdnf IV levels. Targeting either region resulted in a significant reduction in Bdnf IX levels (35.8% for the Bdnf I sgRNA and 42.7% for the Bdnf IV sgRNA). Taken together, these findings indicate that the dCas9-KRAB-MeCP2 system is capable of transcript-selective knockdown and can outperform widely utilized RNAi knockdown methods in neuronal systems.
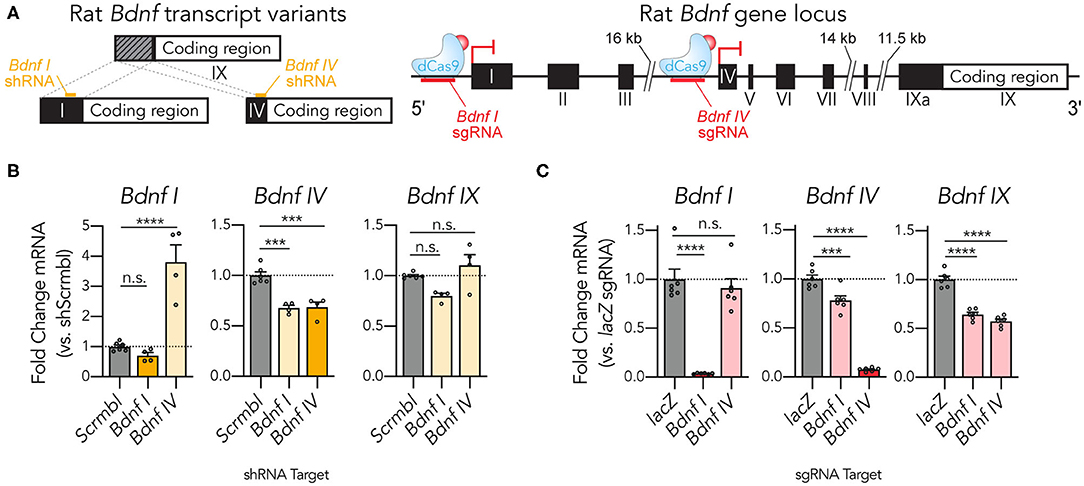
Figure 3. dCas9-KRAB-MeCP2 induces targeted transcript specific Bdnf gene repression in primary rat hippocampal cultures. (A) Complex structure of the rat Bdnf gene produces spliced transcripts from multiple promoter regions using non-coding exons (I-IXa) and a common coding exon (IX). shRNAs (left) or sgRNAs (right) can be designed for targeted transcript suppression. (B) shRNA targeting the Bdnf I variant did not significantly decrease its expression as assessed by RT-qPCR, while shRNA designed against the Bdnf IV variant increased expression of the Bdnf I variant [left, Bdnf I RT-qPCR: n = 4–6, One-Way ANOVA, F(2,11) = 32.55, p < 0.0001. Middle, Bdnf IV RT-qPCR: n = 4–6, One-Way ANOVA, F(2,11) = 25.19, p < 0.0001]. Targeting either variant did not alter the expression levels of total Bdnf as measured using qPCR primers designed for the Bdnf IX common coding region [right, Bdnf IX RT-qPCR: n = 4–6, One-Way ANOVA, F(2,11) = 7.266, p = 0.0097]. (C) Individual sgRNAs designed upstream of the Bdnf I and Bdnf IV exons recruit dCas9-KRAB-MeCP2 to induce transcript specific gene repression of the Bdnf I and Bdnf IV transcript variants as assessed by RT-qPCR. Both sgRNAs resulted in a significant decrease in total Bdnf IX expression [left, Bdnf I RT-qPCR: n = 6, One-Way ANOVA, F(2,15) = 41.87, p < 0.0001. Middle, Bdnf IV RT-qPCR: n = 6, One-Way ANOVA, F(2,15) = 181.9, p < 0.0001. Right, Bdnf IX RT-qPCR: n = 6, one-way ANOVA, F(2,15) = 74.56, p < 0.0001]. Tukey's multiple comparisons test was used for individual comparisons. All data are expressed as mean ± s.e.m. Individual comparisons; ***p < 0.001, ****p < 0.0001.
Discussion
Here, we describe an adapted CRISPRi system in which a recently optimized dCas9-KRAB-MeCP2 transcriptional repressor is driven by the human synapsin promoter. Lentiviral expression of sgRNAs targeting this system to unique gene promoters produced robust knockdown at multiple gene targets in two neuronal culture platforms. This optimized CRISPRi system also enabled transcript-specific manipulations at the Bdnf gene locus, revealing efficient silencing of a complex target relevant to learning, memory, and neuropsychiatric disease (Aid et al., 2007; Bekinschtein et al., 2008, 2014; Lubin et al., 2008; Roth et al., 2009; Zagrebelsky and Korte, 2014; Hing et al., 2018; Lughetti et al., 2018; Lima Giacobbo et al., 2019).
It is worth highlighting the extent of gene depletion achieved by a single sgRNA in these experiments. For instance, near knockout levels of transcript-selective suppression were achieved at Bdnf without disrupting the genetic locus itself (96% at Bdnf I and 92% at Bdnf IV). However, not all targeted gene transcripts investigated were suppressed to the same level. The majority of the sgRNAs utilized here were originally designed for transcriptional activation by placing them in the promoter region upstream of gene transcription start sites (TSS) (Savell et al., 2019a), not downstream of the TSS as is commonly suggested for CRISPRi (Dominguez et al., 2016). Enhanced silencing strength could likely be achieved by using sgRNAs designed specifically to interfere with transcription elongation and transcription factor binding (Dominguez et al., 2016), or by multiplexing multiple sgRNAs together to target genes of interest (Yeo et al., 2018). However, our observation that upstream TSS sgRNAs could be useful for CRISPRi may also reflect an advantage of this optimized system, permitting bidirectional gene modulation (i.e., CRISPRa and CRISPRi) using the same sgRNA in parallel experiments.
Recently, we harnessed a neuron-optimized dCas9 transcriptional activator system to target and induce 16 genes simultaneously to mimic the genetic signature of acute dopamine receptor activation (Savell et al., 2019b). Such strategies enable an unprecedented ability to investigate larger transcriptional networks rather than relying on studies of individual genes. Likewise, recent studies have devoted additional attention to modulation of non-coding RNA and cis-regulatory element function using dCas9 strategies (Carullo et al., 2020; Li et al., 2020). The adapted CRISPRi dCas9-KRAB-MeCP2 system described here is compatible with these approaches and can easily be harnessed for expanded gene silencing via the use of multiplexed sgRNA expression cassettes, targeting non-coding RNA loci, or trafficking the system to regulatory regions of interest.
The system described here uses a neuron-selective promoter, which will enable selective targeting of neurons when applied for in vivo uses (Savell et al., 2019a). Given the cellular diversity of the nervous system, future extensions of this technology to target individual cell populations through combination with cre-recombinase systems is of high interest. Recently, the lentiviral delivery of the first generation dCas9-KRAB construct was employed to selectively modify expression states in either glutamatergic or GABAergic neurons by using mouse CaMKIIα and VGAT selective promoters (Zheng et al., 2018). Our construct could be adapted similarly for targeted transcriptional manipulations in specific neuronal populations. This report also demonstrated that the first generation dCas9-KRAB system provided more robust silencing as compared to shRNAs at multiple genes in primary neurons (Zheng et al., 2018). Thus, our results add to accumulating evidence supporting increased efficiency of CRISPRi over RNAi in achieving robust gene repression (Boettcher and McManus, 2015; Yeo et al., 2018).
As this system does not permanently alter the genetic loci of interest when suppressing transcription, it is possible that transient silencing can be achieved. This has broad appeal for investigations of transient experience-dependent transcription, as well as for critical windows of development. Through combination with drug- (Gao et al., 2016) or light-inducible (Konermann et al., 2013; Polstein and Gersbach, 2015; Bubeck et al., 2018) techniques, these approaches may enable temporally specific manipulation that more thoroughly mimics endogenous gene expression patterns. Finally, given the increased transcriptional complexity and alternative splicing that occurs at genes highly expressed in brain tissue (Wang et al., 2008; Mazin et al., 2013; Raj and Blencowe, 2015; Vuong et al., 2016; Weyn-Vanhentenryck et al., 2018), this technology extends our ability to dissect the distinct function of unique transcripts. The need for such technology is ever more apparent as the association of alternative spliced transcripts with neuropsychiatric disease becomes increasingly appreciated (Mazin et al., 2013; Raj and Blencowe, 2015; Li et al., 2016; Lin et al., 2016; Jutzi et al., 2018; Le François et al., 2018; Raj et al., 2018; Latorre et al., 2019). It should be noted however, that CRISPRi technologies do not target the individual transcript itself, but rather the genomic locus from which different isoforms arise. Thus, this approach should be seen as a complement to RNAi approaches rather than a replacement when investigating specific RNA transcript isoforms. In summary, our results demonstrate robust gene silencing in neurons using a second-generation CRISPRi system, and suggest that continued development of this approach will enable novel experimental strategies in neuronal systems.
Data Availability Statement
The SYN-dCas9-KRAB-MeCP2 construct generated in this study will be made available on Addgene (#155365) for use by the broader research community. All relevant data that support the findings of this study are available by request from the corresponding author.
Ethics Statement
All experiments were performed in accordance with the University of Alabama at Birmingham Institutional Animal Care and Use Committee.
Author Contributions
CD conceived of the SYN-dCas9-KRAB-MeCP2 construct, which was created with a cloning strategy designed by JR, with assistance from NS and NC. FS conceived, designed, constructed the Fos-luciferase construct, and the general shRNA and sgRNA backbones. CD completed and analyzed the luciferase assay and ICC experiments. SB designed and constructed the Bdnf sgRNA and shRNA constructs and completed and analyzed all Bdnf and Fosb experiments. MD completed and analyzed the Reln targeting experiment with assistance from RP. CD completed and analyzed the Kmt2b and Npy targeting experiment. NC designed and constructed the Kmt2b sgRNA constructs. Npy sgRNAs were designed by JD and constructed by AB. All projects were supervised by JD. CD and JD wrote the main text of the manuscript. All authors have approved the final version of the manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was supported by NIH grants DP1-DA039650, R00-DA034681, and R01-MH114990 (JD), F32-MH112304 (SB), F32-DA041778 (FS), T32-GM008361, and T32-NS061788 (CD). Additional assistance to JD was provided by the UAB Pittman Scholars Program. We thank all current and former Day Lab members for assistance and support.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgeed.2020.00009/full#supplementary-material
References
Aid, T., Kazantseva, A., Piirsoo, M., Palm, K., and Timmusk, T. (2007). Mouse and rat BDNF gene structure and expression revisited. J. Neurosci. Res. 85, 525–535. doi: 10.1002/jnr.21139
Alberini, C. M., and Kandel, E. R. (2014). The regulation of transcription in memory consolidation. Cold Spring Harb. Perspect. Biol 7:a021741. doi: 10.1101/cshperspect.a021741
Bae, S., Park, J., and Kim, J.-S. (2014). Cas-OFFinder: a fast and versatile algorithm that searches for potential off-target sites of Cas9 RNA-guided endonucleases. Bioinformatics 30, 1473–1475. doi: 10.1093/bioinformatics/btu048
Bekinschtein, P., Cammarota, M., Katche, C., Slipczuk, L., Rossato, J. I., Goldin, A., et al. (2008). BDNF is essential to promote persistence of long-term memory storage. Proc. Natl. Acad. Sci. U.S.A. 105, 2711–2716. doi: 10.1073/pnas.0711863105
Bekinschtein, P., Cammarota, M., and Medina, J. H. (2014). BDNF and memory processing. Neuropharmacology 76 Pt C, 677–683. doi: 10.1016/j.neuropharm.2013.04.024
Bettencourt, C., Ryten, M., Forabosco, P., Schorge, S., Hersheson, J., Hardy, J., et al. (2014). Insights from cerebellar transcriptomic analysis into the pathogenesis of ataxia. JAMA Neurol. 71, 831–839. doi: 10.1001/jamaneurol.2014.756
Boettcher, M., and McManus, M. T. (2015). Choosing the right tool for the job: RNAi, TALEN, or CRISPR. Mol. Cell 58, 575–585. doi: 10.1016/j.molcel.2015.04.028
Bridge, A. J., Pebernard, S., Ducraux, A., Nicoulaz, A.-L., and Iggo, R. (2003). Induction of an interferon response by RNAi vectors in mammalian cells. Nat. Genet. 34, 263–264. doi: 10.1038/ng1173
Bubeck, F., Hoffmann, M. D., Harteveld, Z., Aschenbrenner, S., Bietz, A., Waldhauer, M. C., et al. (2018). Engineered anti-CRISPR proteins for optogenetic control of CRISPR-Cas9. Nat. Methods 15, 924–927. doi: 10.1038/s41592-018-0178-9
Campbell, M. G., Kohane, I. S., and Kong, S. W. (2013). Pathway-based outlier method reveals heterogeneous genomic structure of autism in blood transcriptome. BMC Med. Genomics 6:34. doi: 10.1186/1755-8794-6-34
Carullo, N. V. N., Phillips, R. A. III, Simon, R. C., Roman Soto, S. A., Hinds, J. E., Salisbury, A. J., et al. (2020). Enhancer RNAs predict enhancer-gene regulatory links and are critical for enhancer function in neuronal systems. bioRxiv 11, e0157086–e0157025. doi: 10.1101/270967
Crusio, W. E. (2004). Flanking gene and genetic background problems in genetically manipulated mice. BPS 56, 381–385. doi: 10.1016/j.biopsych.2003.12.026
Day, J. J., Childs, D., Guzman-Karlsson, M. C., Kibe, M., Moulden, J., Song, E., et al. (2013). DNA methylation regulates associative reward learning. Nat. Neurosci 16, 1445–1452. doi: 10.1038/nn.3504
De Jong, S., Boks, M. P. M., Fuller, T. F., Strengman, E., Janson, E., de Kovel, C. G. F., et al. (2012). A gene co-expression network in whole blood of schizophrenia patients is independent of antipsychotic-use and enriched for brain-expressed genes. PLoS ONE 7:e39498. doi: 10.1371/journal.pone.0039498
Dominguez, A. A., Lim, W. A., and Qi, L. S. (2016). Beyond editing: repurposing CRISPR-Cas9 for precision genome regulation and interrogation. Nat. Rev. Mol. Cell Biol. 17, 5–15. doi: 10.1038/nrm.2015.2
Duke, C. G., Savell, K. E., Tuscher, J. J., Phillips, R. A., and Day, J. J. (2020). Blue light-induced gene expression alterations in cultured neurons are the result of phototoxic interactions with neuronal culture media. eNeuro 7, ENEURO.0386–19.2019. doi: 10.1523/ENEURO.0386-19.2019
Eisener-Dorman, A. F., Lawrence, D. A., and Bolivar, V. J. (2009). Cautionary insights on knockout mouse studies: the gene or not the gene? Brain Behav. Immun. 23, 318–324. doi: 10.1016/j.bbi.2008.09.001
El-Brolosy, M. A., and Stainier, D. Y. R. (2017). Genetic compensation: a phenomenon in search of mechanisms. PLoS Genet. 13:e1006780. doi: 10.1371/journal.pgen.1006780
Fish, R. J., and Kruithof, E. K. O. (2004). Short-term cytotoxic effects and long-term instability of RNAi delivered using lentiviral vectors. BMC Mol. Biol. 5:9. doi: 10.1186/1471-2199-5-9
Gao, Y., Xiong, X., Wong, S., Charles, E. J., Lim, W. A., and Qi, L. S. (2016). Complex transcriptional modulation with orthogonal and inducible dCas9 regulators. Nat. Methods 13, 1043–1049. doi: 10.1038/nmeth.4042
Gilbert, L. A., Horlbeck, M. A., Adamson, B., Villalta, J. E., Chen, Y., Whitehead, E. H., et al. (2014). Genome-scale CRISPR-mediated control of gene repression and activation. Cell 159, 647–661. doi: 10.1016/j.cell.2014.09.029
Gilbert, L. A., Larson, M. H., Morsut, L., Liu, Z., Brar, G. A., Torres, S. E., et al. (2013). CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 154, 442–451. doi: 10.1016/j.cell.2013.06.044
Glatt, S. J., Tsuang, M. T., Winn, M., Chandler, S. D., Collins, M., Lopez, L., et al. (2012). Blood-based gene expression signatures of infants and toddlers with autism. J. Am. Acad. Child Adolesc. Psychiatry 51, 934–44.e2. doi: 10.1016/j.jaac.2012.07.007
Hallock, H. L., Quillian, H. M., Mai, Y., Maynard, K. R., Hill, J. L., and Martinowich, K. (2019). Manipulation of a genetically and spatially defined sub-population of BDNF-expressing neurons potentiates learned fear and decreases hippocampal-prefrontal synchrony in mice. Neuropsychopharmacology 44, 2239–2246. doi: 10.1038/s41386-019-0429-1
Heidenreich, M., and Zhang, F. (2016). Applications of CRISPR-Cas systems in neuroscience. Nat. Rev. Neurosci. 17, 36–44. doi: 10.1038/nrn.2015.2
Hing, B., Sathyaputri, L., and Potash, J. B. (2018). A comprehensive review of genetic and epigenetic mechanisms that regulate BDNF expression and function with relevance to major depressive disorder. Am. J. Med. Genet. B Neuropsychiatr. Genet. 177, 143–167. doi: 10.1002/ajmg.b.32616
Huang, C.-H., Lee, K.-C., and Doudna, J. A. (2018). Applications of CRISPR-cas enzymes in cancer therapeutics and detection. Trends Cancer 4, 499–512. doi: 10.1016/j.trecan.2018.05.006
Jackson, A. L., Bartz, S. R., Schelter, J., Kobayashi, S. V., Burchard, J., Mao, M., et al. (2003). Expression profiling reveals off-target gene regulation by RNAi. Nat. Biotechnol. 21, 635–637. doi: 10.1038/nbt831
Jiang, F., and Doudna, J. A. (2017). CRISPR–Cas9 structures and mechanisms. Annu. Rev. Biophys. 46, 505–529. doi: 10.1146/annurev-biophys-062215-010822
Judge, A. D., Sood, V., Shaw, J. R., Fang, D., McClintock, K., and MacLachlan, I. (2005). Sequence-dependent stimulation of the mammalian innate immune response by synthetic siRNA. Nat. Biotechnol. 23, 457–462. doi: 10.1038/nbt1081
Jutzi, D., Akinyi, M. V., Mechtersheimer, J., Frilander, M. J., and Ruepp, M.-D. (2018). The emerging role of minor intron splicing in neurological disorders. Cell Stress 2, 40–54. doi: 10.15698/cst2018.03.126
Khan, A. A., Betel, D., Miller, M. L., Sander, C., Leslie, C. S., and Marks, D. S. (2009). Transfection of small RNAs globally perturbs gene regulation by endogenous microRNAs. Nat. Biotechnol. 27, 549–555. doi: 10.1038/nbt.1543
Knott, G. J., and Doudna, J. A. (2018). CRISPR-Cas guides the future of genetic engineering. Science 361, 866–869. doi: 10.1126/science.aat5011
Konermann, S., Brigham, M. D., Trevino, A., Hsu, P. D., Heidenreich, M., Cong, L., et al. (2013). Optical control of mammalian endogenous transcription and epigenetic states. Nature 500, 472–476. doi: 10.1038/nature12466
La Russa, M. F., and Qi, L. S. (2015). The new state of the art: Cas9 for gene activation and repression. Mol. Cell. Biol. 35, 3800–3809. doi: 10.1128/MCB.00512-15
Labun, K., Montague, T. G., Krause, M., Torres Cleuren, Y. N., Tjeldnes, H., and Valen, E. (2019). CHOPCHOP v3: expanding the CRISPR web toolbox beyond genome editing. Nucleic Acids Res. 47, W171–W174. doi: 10.1093/nar/gkz365
Latorre, E., Mesonero, J. E., and Harries, L. W. (2019). Alternative splicing in serotonergic system: implications in neuropsychiatric disorders. J. Psychopharmacol. (Oxford) 33, 1352–1363. doi: 10.1177/0269881119856546
Le François, B., Zhang, L., Mahajan, G. J., Stockmeier, C. A., Friedman, E., and Albert, P. R. (2018). A novel alternative splicing mechanism that enhances human 5-HT1A receptor RNA stability is altered in major depression. J. Neurosci. 38, 8200–8210. doi: 10.1523/JNEUROSCI.0902-18.2018
Lewis, P. A., and Cookson, M. R. (2012). Gene expression in the Parkinson's disease brain. Brain Res. Bull. 88, 302–312. doi: 10.1016/j.brainresbull.2011.11.016
Li, K., Liu, Y., Cao, H., Zhang, Y., Gu, Z., Liu, X., et al. (2020). Interrogation of enhancer function by enhancer-targeting CRISPR epigenetic editing. Nat. Commun 11, 485. doi: 10.1038/s41467-020-14362-5
Li, Y. I., van de Geijn, B., Raj, A., Knowles, D. A., Petti, A. A., Golan, D., et al. (2016). RNA splicing is a primary link between genetic variation and disease. Science 352, 600–604. doi: 10.1126/science.aad9417
Liang, W. S., Reiman, E. M., Valla, J., Dunckley, T., Beach, T. G., Grover, A., et al. (2008). Alzheimer's disease is associated with reduced expression of energy metabolism genes in posterior cingulate neurons. Proc. Natl. Acad. Sci. U.S.A. 105, 4441–4446. doi: 10.1073/pnas.0709259105
Lima Giacobbo, B., Doorduin, J., Klein, H. C., Dierckx, R. A. J. O., Bromberg, E., and de Vries, E. F. J. (2019). Brain-derived neurotrophic factor in brain disorders: focus on neuroinflammation. Mol. Neurobiol. 56, 3295–3312. doi: 10.1007/s12035-018-1283-6
Lin, L., Park, J. W., Ramachandran, S., Zhang, Y., Tseng, Y.-T., Shen, S., et al. (2016). Transcriptome sequencing reveals aberrant alternative splicing in Huntington's disease. Hum. Mol. Genet. 25, 3454–3466. doi: 10.1093/hmg/ddw187
Liu, X. S., Wu, H., Ji, X., Stelzer, Y., Wu, X., Czauderna, S., et al. (2016). Editing DNA methylation in the mammalian genome. Cell 167, 233–247.e17. doi: 10.1016/j.cell.2016.08.056
Livak, K. J., and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25, 402–408. doi: 10.1006/meth.2001.1262
Lubin, F. D., Roth, T. L., and Sweatt, J. D. (2008). Epigenetic regulation of BDNF gene transcription in the consolidation of fear memory. J. Neurosci. 28, 10576–10586. doi: 10.1523/JNEUROSCI.1786-08.2008
Lughetti, L., Lucaccioni, L., Fugetto, F., Predieri, B., Berardi, A., and Ferrari, F. (2018). Brain-derived neurotrophic factor and epilepsy: a systematic review. Neuropeptides 72, 23–29. doi: 10.1016/j.npep.2018.09.005
MacMullen, C. M., Vick, K., Pacifico, R., Fallahi-Sichani, M., and Davis, R. L. (2016). Novel, primate-specific PDE10A isoform highlights gene expression complexity in human striatum with implications on the molecular pathology of bipolar disorder. Transl. Psychiatry 6, e742–e742. doi: 10.1038/tp.2016.3
Mandegar, M. A., Huebsch, N., Frolov, E. B., Shin, E., Truong, A., Olvera, M. P., et al. (2016). CRISPR interference efficiently induces specific and reversible gene silencing in human iPSCs. Cell Stem Cell 18, 541–553. doi: 10.1016/j.stem.2016.01.022
Martella, A., Firth, M., Taylor, B. J. M., Göppert, A., Cuomo, E. M., Roth, R. G., et al. (2019). Systematic evaluation of CRISPRa and CRISPRi modalities enables development of a multiplexed, orthogonal gene activation and repression system. ACS Synth. Biol. 8, 1998–2006. doi: 10.1021/acssynbio.8b00527
Maynard, K. R., Hobbs, J. W., Phan, B. N., Gupta, A., Rajpurohit, S., Williams, C., et al. (2018). BDNF-TrkB signaling in oxytocin neurons contributes to maternal behavior. Elife 7:525. doi: 10.7554/eLife.33676
Maynard, K. R., Hobbs, J. W., Sukumar, M., Kardian, A. S., Jimenez, D. V., Schloesser, R. J., et al. (2017). Bdnf mRNA splice variants differentially impact CA1 and CA3 dendrite complexity and spine morphology in the hippocampus. Brain Struct. Funct. 222, 3295–3307. doi: 10.1007/s00429-017-1405-3
Mazin, P., Xiong, J., Liu, X., Yan, Z., Zhang, X., Li, M., et al. (2013). Widespread splicing changes in human brain development and aging. Mol. Syst. Biol. 9:633. doi: 10.1038/msb.2012.67
McClung, C. A., and Nestler, E. J. (2008). Neuroplasticity mediated by altered gene expression. Neuropsychopharmacology 33, 3–17. doi: 10.1038/sj.npp.1301544
Olejniczak, M., Urbanek, M. O., Jaworska, E., Witucki, L., Szczesniak, M. W., Makalowska, I., et al. (2016). Sequence-non-specific effects generated by various types of RNA interference triggers. Biochim. Biophys. Acta 1859, 306–314. doi: 10.1016/j.bbagrm.2015.11.005
Pickar-Oliver, A., and Gersbach, C. A. (2019). The next generation of CRISPR-Cas technologies and applications. Nat. Rev. Mol. Cell Biol. 20, 490–507. doi: 10.1038/s41580-019-0131-5
Polstein, L. R., and Gersbach, C. A. (2015). A light-inducible CRISPR-Cas9 system for control of endogenous gene activation. Nat. Chem. Biol 11, 198–200. doi: 10.1038/nchembio.1753
Pramparo, T., Pierce, K., Lombardo, M. V., Carter Barnes, C., Marinero, S., Ahrens-Barbeau, C., et al. (2015). Prediction of autism by translation and immune/inflammation coexpressed genes in toddlers from pediatric community practices. JAMA Psychiatry 72, 386–394. doi: 10.1001/jamapsychiatry.2014.3008
Qi, L. S., Larson, M. H., Gilbert, L. A., Doudna, J. A., Weissman, J. S., Arkin, A. P., et al. (2013). Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 152, 1173–1183. doi: 10.1016/j.cell.2013.02.022
Raj, B., and Blencowe, B. J. (2015). Alternative splicing in the mammalian nervous system: recent insights into mechanisms and functional roles. Neuron 87, 14–27. doi: 10.1016/j.neuron.2015.05.004
Raj, T., Li, Y. I., Wong, G., Humphrey, J., Wang, M., Ramdhani, S., et al. (2018). Integrative transcriptome analyses of the aging brain implicate altered splicing in Alzheimer's disease susceptibility. Nat. Genet. 50, 1584–1592. doi: 10.1038/s41588-018-0238-1
Read, M. L., Mir, S., Spice, R., Seabright, R. J., Suggate, E. L., Ahmed, Z., et al. (2009). Profiling RNA interference (RNAi)-mediated toxicity in neural cultures for effective short interfering RNA design. J. Gene Med 11, 523–534. doi: 10.1002/jgm.1321
Roth, T. L., Lubin, F. D., Funk, A. J., and Sweatt, J. D. (2009). Lasting epigenetic influence of early-life adversity on the BDNF gene. BPS 65, 760–769. doi: 10.1016/j.biopsych.2008.11.028
Savell, K., Sultan, F., and Day, J. (2019b). A novel dual lentiviral crispr-based transcriptional activation system for gene expression regulation in neurons. Bio-Protocol. 9, 1–18. doi: 10.21769/BioProtoc.3348
Savell, K. E., Bach, S. V., Zipperly, M. E., Revanna, J. S., Goska, N. A., Tuscher, J. J., et al. (2019a). A neuron-optimized CRISPR/dCas9 activation system for robust and specific gene regulation. eNeuro 6, ENEURO.0495–18.2019. doi: 10.1523/ENEURO.0495-18.2019
Savell, K. E., and Day, J. J. (2017). Applications of CRISPR/Cas9 in the mammalian central nervous system. Yale J. Biol. Med. 90, 567–581. doi: 10.1016/j.neuron.2014.07.043
Savell, K. E., Gallus, N. V. N., Simon, R. C., Brown, J. A., Revanna, J. S., Osborn, M. K., et al. (2016). Extra-coding RNAs regulate neuronal DNA methylation dynamics. Nat. Commun. 7:12091. doi: 10.1038/ncomms12091
Savell, K. E., Zipperly, M. E., Tuscher, J. J., Duke, C. G., Phillips, R. A. III, Bauman, A. J., et al. (2020). A dopamine-induced gene expression signature regulates neuronal function and cocaine response. Sci. Adv. 6:eaba4221. doi: 10.1136/sciadv.aba4221
Sledz, C. A., Holko, M., de Veer, M. J., Silverman, R. H., and Williams, B. R. G. (2003). Activation of the interferon system by short-interfering RNAs. Nat. Cell Biol. 5, 834–839. doi: 10.1038/ncb1038
Suzuki, K., Tsunekawa, Y., Hernandez-Benitez, R., Wu, J., Zhu, J., Kim, E. J., et al. (2016). In vivo genome editing via CRISPR/Cas9 mediated homology-independent targeted integration. Nature 540, 144–149. doi: 10.1038/nature20565
Thakore, P. I., Black, J. B., Hilton, I. B., and Gersbach, C. A. (2016). Editing the epigenome: technologies for programmable transcription and epigenetic modulation. Nat. Methods 13, 127–137. doi: 10.1038/nmeth.3733
Ui-Tei, K. (2013). Optimal choice of functional and off-target effect-reduced siRNAs for RNAi therapeutics. Front. Genet 4:107. doi: 10.3389/fgene.2013.00107
Vuong, C. K., Black, D. L., and Zheng, S. (2016). The neurogenetics of alternative splicing. Nat. Rev. Neurosci. 17, 265–281. doi: 10.1038/nrn.2016.27
Wang, E. T., Sandberg, R., Luo, S., Khrebtukova, I., Zhang, L., Mayr, C., et al. (2008). Alternative isoform regulation in human tissue transcriptomes. Nature 456, 470–476. doi: 10.1038/nature07509
Weyn-Vanhentenryck, S. M., Feng, H., Ustianenko, D., Duffi,é, R., Yan, Q., Jacko, M., et al. (2018). Precise temporal regulation of alternative splicing during neural development. Nat. Commun. 9:2189. doi: 10.1038/s41467-018-04559-0
Winkler, J. M., and Fox, H. S. (2013). Transcriptome meta-analysis reveals a central role for sex steroids in the degeneration of hippocampal neurons in Alzheimer's disease. BMC Syst. Biol. 7:51. doi: 10.1186/1752-0509-7-51
Woo, H. I., Lim, S.-W., Myung, W., Kim, D. K., and Lee, S.-Y. (2018). Differentially expressed genes related to major depressive disorder and antidepressant response: genome-wide gene expression analysis. Exp. Mol. Med. 50, 92. doi: 10.1038/s12276-018-0123-0
Xiong, K., Marquart, K. F., la Cour Karottki, K. J., Li, S., Shamie, I., Lee, J. S., et al. (2019). Reduced apoptosis in Chinese hamster ovary cells via optimized CRISPR interference. Biotechnol. Bioeng. 116, 1813–1819. doi: 10.1002/bit.26969
Xu, P.-T., Li, Y.-J., Qin, X.-J., Scherzer, C. R., Xu, H., Schmechel, D. E., et al. (2006). Differences in apolipoprotein E3/3 and E4/4 allele-specific gene expression in hippocampus in Alzheimer disease. Neurobiol. Dis. 21, 256–275. doi: 10.1016/j.nbd.2005.07.004
Yap, E.-L., and Greenberg, M. E. (2018). Activity-regulated transcription: bridging the gap between neural activity and behavior. Neuron 100, 330–348. doi: 10.1016/j.neuron.2018.10.013
Yeo, N. C., Chavez, A., Lance-Byrne, A., Chan, Y., Menn, D., Milanova, D., et al. (2018). An enhanced CRISPR repressor for targeted mammalian gene regulation. Nat. Methods 15, 611–616. doi: 10.1038/s41592-018-0048-5
Zagrebelsky, M., and Korte, M. (2014). Form follows function: BDNF and its involvement in sculpting the function and structure of synapses. Neuropharmacology 76 Pt C, 628–638. doi: 10.1016/j.neuropharm.2013.05.029
Zhang, F. (2019). Development of CRISPR-Cas systems for genome editing and beyond. Q. Rev. Biophys 52, 1–31. doi: 10.1017/S0033583519000052
Zheng, Y., Shen, W., Zhang, J., Yang, B., Liu, Y.-N., Qi, H., et al. (2018). CRISPR interference-based specific and efficient gene inactivation in the brain. Nat. Neurosci 21, 447–454. doi: 10.1038/s41593-018-0077-5
Keywords: CRISPRi, dCas9, neurons, KRAB-MeCP2, gene regulation, transcription
Citation: Duke CG, Bach SV, Revanna JS, Sultan FA, Southern NT, Davis MN, Carullo NVN, Bauman AJ, Phillips RA III and Day JJ (2020) An Improved CRISPR/dCas9 Interference Tool for Neuronal Gene Suppression. Front. Genome Ed. 2:9. doi: 10.3389/fgeed.2020.00009
Received: 27 May 2020; Accepted: 03 August 2020;
Published: 15 September 2020.
Edited by:
Gabriele Lignani, University College London, United KingdomReviewed by:
Max A. Horlbeck, Boston Children's Hospital and Harvard Medical School, United StatesMarta Olejniczak, Institute of Bioorganic Chemistry (PAS), Poland
Copyright © 2020 Duke, Bach, Revanna, Sultan, Southern, Davis, Carullo, Bauman, Phillips and Day. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jeremy J. Day, ampkYXlAdWFiLmVkdQ==
 Corey G. Duke
Corey G. Duke Svitlana V. Bach
Svitlana V. Bach Jasmin S. Revanna
Jasmin S. Revanna Nicholas T. Southern
Nicholas T. Southern M. Natalie Davis
M. Natalie Davis Nancy V. N. Carullo
Nancy V. N. Carullo Allison J. Bauman
Allison J. Bauman Robert A. Phillips III
Robert A. Phillips III Jeremy J. Day
Jeremy J. Day