- Shanghai Clinical Research Center of Bone Diseases, Department of Osteoporosis and Bone Diseases, Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China
Objective: To summarize the clinical features and bone complications in a patient from a large family with X-linked congenital adrenocortical hypoplasia (AHC) and evaluate the efficacy of different treatment regimens on the prognosis of secondary osteoporosis caused by AHC at a 5-year follow-up.
Methods: A large family with AHC was recruited, and the causative gene mutation was identified by Sanger sequencing in the proband. Clinical features as well as radiological examinations and laboratory indices of osteoporosis secondary to AHC were analyzed in this study. Meanwhile, the proband was treated with classical antiresorptive drugs (bisphosphonates) for 2 years and switched to a vitamin K2 analogue for another 3 years, during which the efficacy of the drugs was evaluated.
Results: The proband was identified as carrying a homozygous insertion mutation (p. Thr193GlyfsX13) in the NR0B1 (nuclear receptor subfamily 0, group B, member 1) gene, resulting in a premature stop codon due to a frameshift mutation. During treatment and follow-up, the proband did not respond well to bisphosphonate and developed atypical femoral fractures. Vitamin K2 improved clinical symptoms. In terms of bone mineral density (BMD), there is no evidence of any effect of vitamin K2 on the neck of femur, though some minor effects on spinal BMD cannot be excluded.
Conclusions: Secondary osteoporosis induced by AHC deserves clinical attention. Unlike in primary osteoporosis, the curative effect of bisphosphonates was unsatisfactory and was more likely to cause atypical femoral fractures in long-term treatment. It is suggested that bone anabolic drugs may be better alternatives.
Introduction
Osteoporosis is characterized by low-energy fractures resulting from low bone mass and damaged bone microarchitecture. It is especially prevalent in postmenopausal women and older men, and can be evaluated by dual-energy x-ray absorptiometry (DXA). It is crucial to distinguish primary from secondary osteoporosis, which directly affects the choice of treatment options, appraisal of therapeutic efficacy, the incidence of side effects, and prediction of prognosis. Secondary osteoporosis occurs in almost 2/3 of men, more than half of premenopausal and perimenopausal women, and approximately 1/5 of postmenopausal women (1). Multiple diseases or drugs affecting bone metabolism can lead to secondary osteoporosis, of which hypogonadism is the most common cause in men (2, 3).
In this study, we report an adult patient with osteoporosis secondary to congenital adrenal dysplasia complaining of low BMD. Congenital adrenal dysplasia is a rare disorder inherited in X-linked (OMIM 300200) and autosomal recessive (OMIM 240200) forms that also occurs as part of the Xp21 deletion syndrome. Mutations in the NR0B1 gene mapped on chromosome Xp21 are responsible for AHC (4). NR0B1 encodes the DAX-1 (dosage sensitive sex reversal, adrenal hypoplasia, critical region on the X chromosome, gene 1) protein, which is mainly expressed in steroidogenic tissue (gonads and adrenal cortex), as well as the hypothalamus and pituitary. It plays an essential role in the development and regulation of the adrenal gland and reproductive axis (5).
AHC has an approximate incidence of 1:12,500 live births in Australia (6). A study from the UK demonstrated that the incidence rate in children is 1:140,000–1:1,200,000, of which the incidence rate in male patients was 1:70,000–1:600,000 (7). Furthermore, AHC displayed a two-peak profile divided by the age of onset (4). The majority of patients with AHC have an early onset, usually within 2 months of life, accompanied by severe salt wasting and high mortality. Late onset occurs in the age range of 2 to 9 years, and clinical symptoms are more atypical and insidious (4, 8, 9). In general, male patients with X-linked AHC usually present primary adrenal insufficiency (PAI) or isolated mineralocorticoid deficiency at an early age, with or without hypogonadotropic hypogonadism (HHG) (10), while female patients are almost always asymptomatic carriers.
Apart from typical clinical symptoms, such as PAI and HHG, attention should be given to skeletal complications such as osteoporosis, which is easily overlooked. The mechanism of osteoporosis secondary to AHC is still not fully elucidated, and hypogonadism is one of the contributors (11). In the present study, the AHC patient with severe osteoporosis had been followed up for almost 5 years. A poor response to bisphosphonates was observed, and atypical femoral fractures (AFFs) occurred during the first 2 years of treatment. Given the primary disease and concomitant therapy of steroids in this patient, it was important to choose an optimal therapeutic regimen.
Here, the clinical characteristics of osteoporosis secondary to AHC are analyzed, and the choice of drugs for such diseases is discussed. The purpose of this case report is to attract the attention of clinicians to select appropriate drugs, avoid serious complications, and obtain the best curative effect.
Materials and methods
Subject
The present study was approved by the Ethics Committee of Shanghai Sixth People’s Hospital Affiliated with Shanghai Jiao Tong University School of Medicine. We investigated a middle-aged male patient from an AHC family who complained of low BMD (Figure 1). Written informed consent was obtained from the participant.
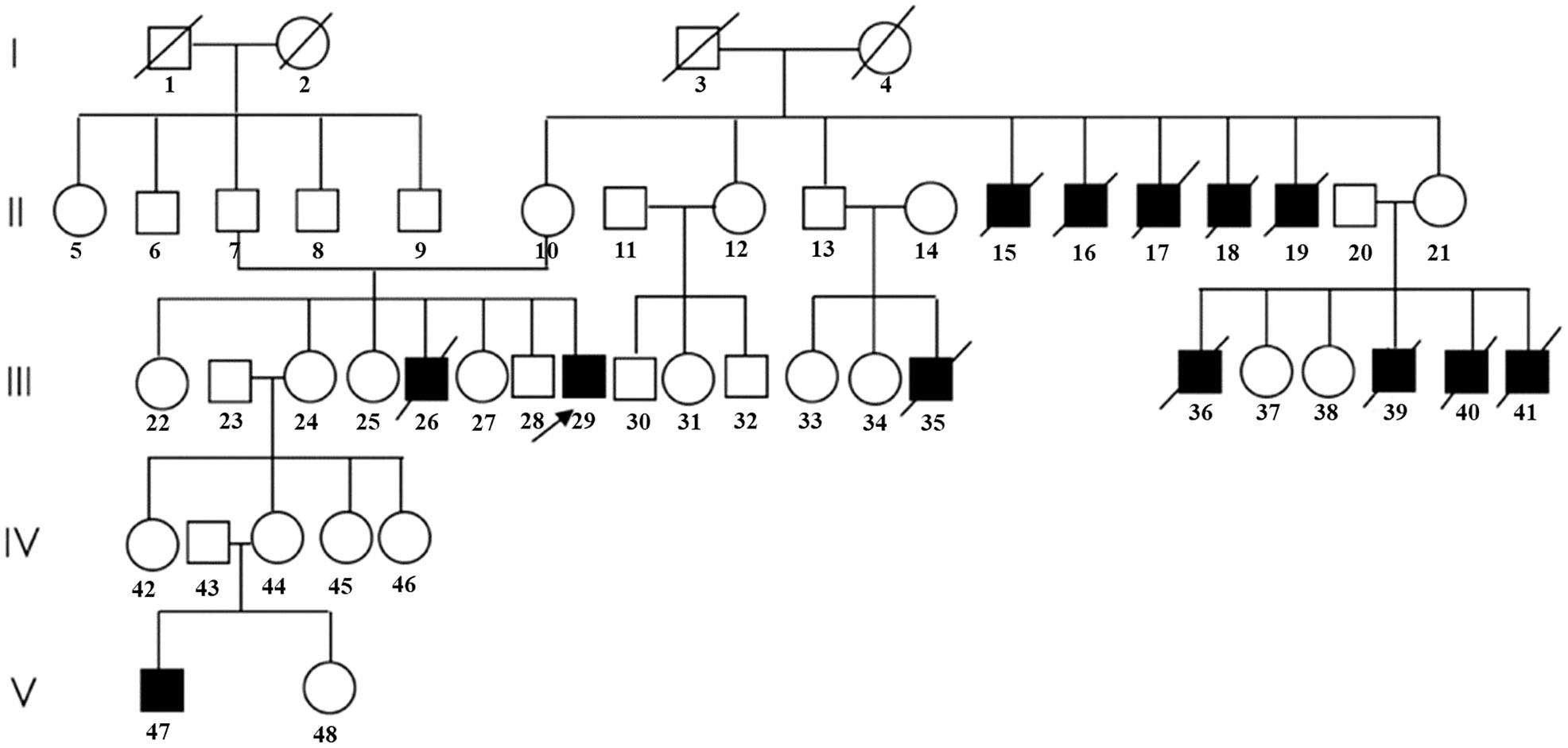
Figure 1 Pedigree of the family in the present study. (Patients with typical clinical symptoms of AHC are shown by darkened symbols. The black arrow indicates the proband.).
Clinical, laboratory, and imaging evaluation
Anthropology measurements (including height, weight, and age) and detailed medical history were recorded. Complete medical check-ups were performed, including skin pigmentation and secondary sexual signs. Blood samples were extracted for routine biochemical indicators, sex hormones, and bone turnover markers (BTMs). Radiographs of the thoracolumbar spine, abdominal computed tomography (CT), cerebral magnetic resonance imaging (MRI), and bone scintigraphy were performed. BMD (g/cm2) of the lumbar spine 1–4 (L1–4), left femoral neck, and total hip was measured by Lunar Prodigy DXA (GE Healthcare, Madison, USA). The LSC (least significant change) was calculated according to International Society for Clinical Densitometry (ISCD) recommendations (12). To be considered significant, the BMD difference before and after medication should be greater than the LSC. The LSC (lumber spine, femur neck, and total hip) at our center was 0.028 g/cm2.
Genetic analysis
Genomic DNA was extracted from peripheral blood using a conventional method. The primers were designed by Primer3 software (http://bioinfo.ut.ee/primer3-0.4.0/, Supplementary Table 1). After predenaturation at 95°C for 2 min, PCR amplifications were performed for 35 cycles (96°C for 10 s, 68°C for 1 min, and 72°C for 1 min). PCR products were purified and sequenced on an ABI3730XL platform with the BigDye3.1 Kit (ABI company, USA).
Case report
Proband III-29 (Figure 1), a 46-year-old man, was born at term from an uneventful pregnancy with a birth weight of 3.6 kg, and the parents were nonconsanguineous, healthy, and of average height. By age 8, he developed recurrent episodes of fever, chills, and fatigue with unknown causes. Growth retardation was observed, and hyperpigmented macules grew, especially on the lips, ears, and joints of the fingers. Immediately, he was diagnosed with PAI (specific data were not available) at the local hospital and treated with prednisone for nearly 30 years, with a maximum dose of up to 30 mg per day maintained until the present. During this time, acute adrenal crises occurred twice due to improper drug withdrawal. Due to the lack of secondary sexual characteristics, HHG was diagnosed, and testosterone undecanoate was administered 10 years ago, during which he recovered his sexual function slightly. Backache, fatigue, and muscle cramps gradually developed at the age of 35. Alendronate was administered for 4 years and 10 months, withdrawn for 3 years, and then restarted for 2 years. However, the treatment effect was not satisfactory, and neither the bone pain nor the BMD improved, so he came to our clinic for help (specific data were not available).
At his first visit to our clinic, he mentioned that his family members (III-26, 35, 36, 39, 40, and 41) also presented the same symptoms as him, including skin pigmentation and salt wasting. Unfortunately, all of them passed away at ages ranging from 7 to 11. Another patient (V-47) was 14 years old and presented with weight loss and progressive skin pigmentation. The height, fingertip distance, and weight of the proband were 156 cm (−4.5 SD), 168 cm, and 48 kg, respectively. Physical examination revealed moderate hyperpigmentation on the buccal mucosa, lips, and gums; normal olfaction; sparse pubic hair; small penis; and low testicular volumes of 4 ml on the left and 5 ml on the right (Tanner stage 2). Imaging examination indicated multiple compression fractures of the thoracolumbar spine and crumpled adrenal glands without calcification (Figure 2), which were detected by x-ray, bone scintigraphy, and abdominal CT scans. To evaluate the changes in the hypothalamus and pituitary, the patient was examined by brain MRI without any abnormalities. In addition, DXA revealed a low BMD (L1–L4, Z score −2.3 SD; femoral neck, Z score −2.9 SD; total hip, Z score −2.6 SD).
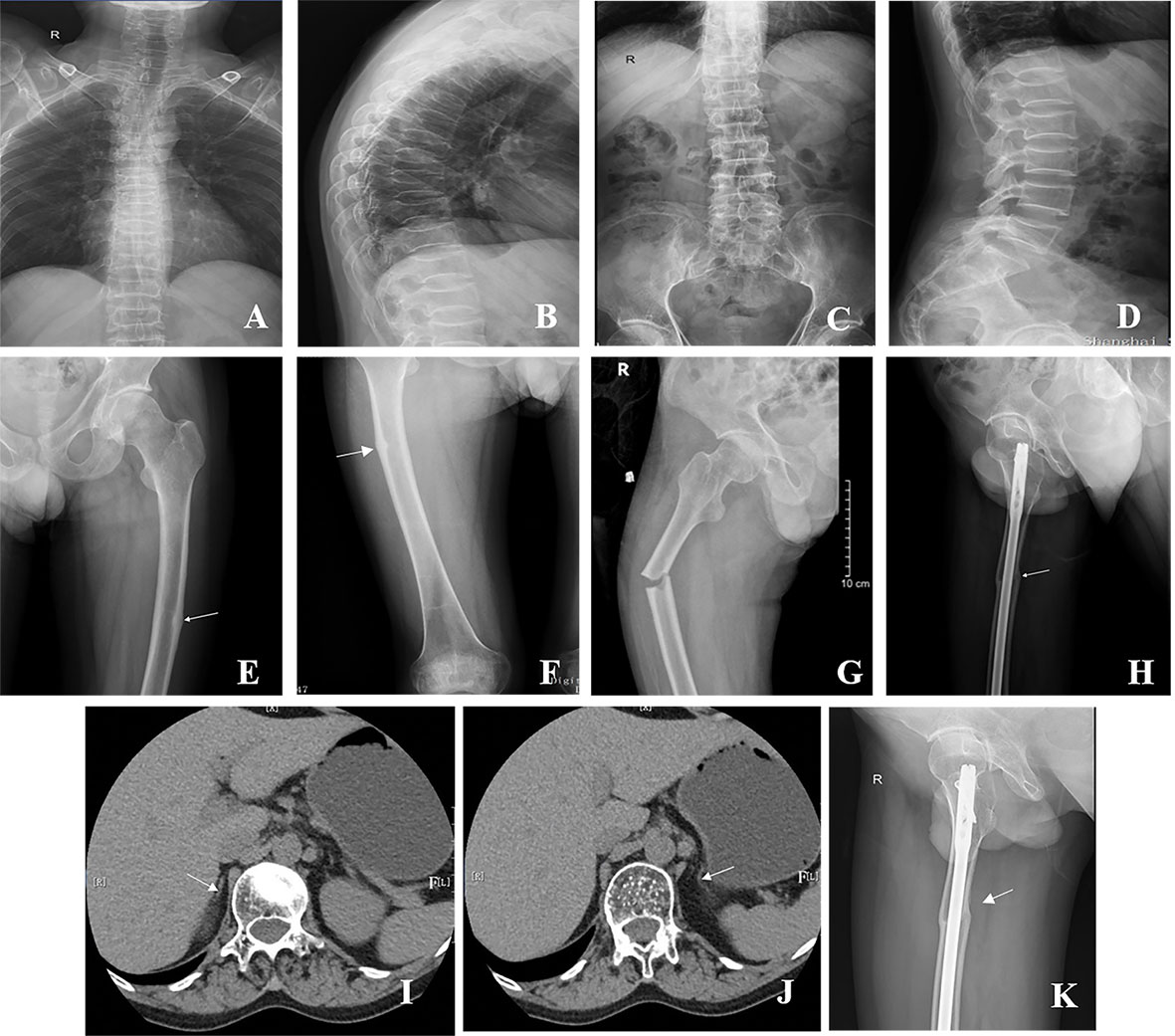
Figure 2 Radiographs of the patient with AHC. (A–D) Multiple vertebral compression fractures. (E, F) The white arrow shows the pseudofracture line in the left and right femurs. (G) Right femoral shaft fracture at the site of the pseudofracture line. (H) Right femoral shaft fracture after internal fixation. (I, J) The white arrow shows that the adrenal glands on both sides were small, and the structure was unclear. (K) Right femoral shaft fracture healing in 2022.
Laboratory parameters are summarized in Supplementary Table 2. As a clinical sign of PAI, the patient showed low levels of serum cortisol at 8 o’clock in the morning and elevated serum levels of ACTH. The plasma concentrations of renin supine and angiotensin II exceeded the upper limit of the normal reference value, while aldosterone was below the lower limit of the normal reference value. The patient exhibited low serum levels of T and LH. Sanger sequencing of NR0B1 revealed an inserted mutation (c.572_575dup) in exon 1, resulting in a frameshift with premature termination (p.Thr193GlyfsX13, Supplementary Figure 1). Combined with a molecular diagnosis, clinical characteristics, and laboratory indices, the diagnosis of AHC was established.
Given the high levels of BTMs and poor efficacy probably resulting from the intermittent use of bisphosphonates, a regular oral alendronate treatment (70 mg, alendronate sodium, and vitamin D3 tablets, Merck Sharp & Dohme Ltd, USA) was advised. The BTMs and BMD (Table 1) were rechecked 1 year later, and the results indicated that the effect of oral drugs was not ideal even when taking them regularly. Therefore, intravenous zoledronate (5 mg, Aclasta, Novartis Pharma Stein AG, Switzerland) yearly was recommended in the next year. After treatment with zoledronic acid for nearly 1 year, the patient complained of pain in the right proximal femur, and a pseudofracture line was detected by x-ray (Figure 2E). One month later, an AFF occurred in the right femur at the site of the pseudofracture line due to an accidental fall, and internal fixation was performed (Figure 2H). A pseudofracture line in the middle of the left femur and a significant decrease in BMD of the femur neck were also found in the same year. Based on the conditions described above, bisphosphonates were not appropriate for the patient, and treatment was changed to vitamin K2 analogues (Menatetrenone, Eisai, Japan, 15 mg tid p.o.). During the subsequent follow-up, the level of serum osteocalcin was significantly elevated, bone pain was relieved, and no pathological fractures appeared (Figure 3). Subsequently, BMD in the lumbar spine 1–4 and total hip increased from 2019 to 2022 (Table 1). The difference in BMD in the lumbar spine and total hip from 2019 to 2022 exceeded the LSC (0.077 and 0.082 g/cm2 compared with 0.028 g/cm2). However, the difference in BMD in the neck of the femur from 2019 to 2022 was below the LSC (0.007 g/cm2 compared with 0.028 g/cm2).
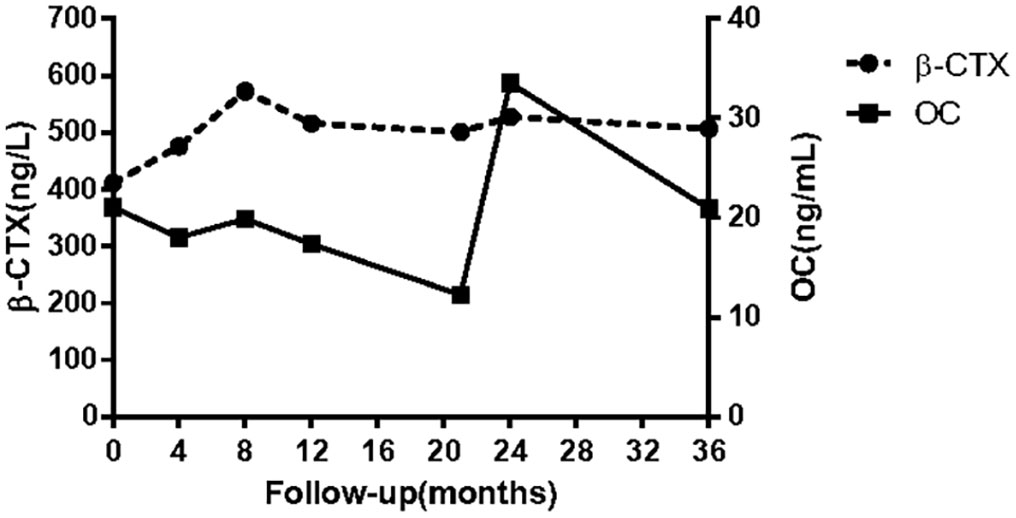
Figure 3 Bone turnover markers during treatment (alendronate orally at the beginning of treatment; a single infusion of intravenous zoledronic acid by the 12th month; right femur fracture by the 14th month; atypical fracture of left femur by the 20th month; menatetrenone orally by the 21st month). β-CTX: β-isomerized C-terminal telopeptide of type I collagen; OC: serum osteocalcin in the form of an N-terminal mid-molecule fragment.
Discussion
AHC, a special type of PAI, was first documented by the pathologist Sikl in 1948 (8). In 1994, Muscatelli et al. found that the candidate gene was localized at the Xp21.3 region (13). The NR0B1 gene consists of two exons separated by a single intron and encodes the DAX-1 protein with a length of 470 amino acids, an orphan member of the nuclear receptor superfamily (14). Instead of the highly conserved zinc finger DNA binding domain (DBD) of other classical nuclear receptors, the N-terminal of the DAX-1 protein contains 3.5 alanine/glycine-rich repeats of a 65–70 amino acid motif, which plays a vital role in the transcriptional regulation of NR0B1 (15–17). In contrast, the highly conserved C-terminal domain is homologous to the ligand-binding domain (LBD) of the nuclear receptor superfamily and mediates ligand binding, dimerization, and nuclear localization (10).
To date, over 300 individuals and families with AHC have been reported, most of which harbor frameshift or nonsense mutations (18–22). It was speculated that the hot spot mutation of NR0B1 was located in the putative LBD, causing premature truncation of the DAX-1 protein (7, 22). Previous studies indicated that clinical phenotypic heterogeneity was detected in patients with AHC, even when caused by the same mutation of the NR0B1 gene (23, 24). Affected individuals in this pedigree manifested various clinical phenotypes, which supported the above conclusion and hinted at the important role of genetic heterogeneity and epigenetic factors in the phenotype and prognosis of AHC. Further studies are needed to reveal the complex genotype–phenotype correlation.
Bertalan reported two brothers with serious osteoporosis secondary to AHC at the ages of 51 and 43 and revealed two novel NR0B1 mutations (c.568-571del and c.572-575del). However, the treatment method was not mentioned (25). Previous studies did not focus on osteoporosis secondary to AHC. In fact, it is an inevitable complication of AHC with serious consequences. Our study focused on osteoporosis secondary to AHC for the first time and discussed the choice of effective treatment.
This case is not simple secondary osteoporosis and has many clinical and biochemical characteristics different from classical GIOP. The long-term effect of GC on bone metabolism is the inhibition of osteogenesis. Theoretically, the BTMs of the patient should decrease. However, the bone metabolism of this patient was not inhibited, and treatment with bisphosphonates failed to effectively reduce the level of BTMs. At the same time, we did not expect that AFFs would occur when the BTMs were not suppressed. The mechanism of osteoporosis secondary to AHC has not been fully elucidated. One explanation was that severe osteoporosis was induced by the lack of a multitude of hormones, especially androgen (26). However, some studies reported patients with HH accompanied by osteoporosis or osteopenia, where bone loss was irreversible even with sufficient androgen substitution (27–29). On the other hand, long-term corticosteroid replacement therapy of this patient also had an adverse impact on BMD (30). Therefore, choosing an appropriate drug is the key point for the prevention and treatment of osteoporosis secondary to AHC.
Antiresorptive drugs, such as alendronate or zoledronate, are classical anti-osteoporosis medications that show satisfactory therapeutic effects in most patients with osteoporosis (31). To evaluate the efficacy of drugs for osteoporosis, BTMs are often measured in clinical practice (32). In this patient, long-term high levels of BTMs (OC and β-CTX) indicated that the efficacy of bisphosphonates, either oral or intravenous, was somewhat disappointing. More seriously, AFFs occurred during the treatment. Generally, bisphosphonates are effective in reducing the risk of osteoporotic fractures, and the risk of AFF increases gradually with the length of treatment. However, it usually occurs after continuous use for more than 5 years (33). Moreover, a cohort study indicated that women with osteoporosis who took bisphosphonate holidays were less likely to sustain AFFs (0.03%) than women in the persistent and nonpersistent groups (0.15%). The average drug holiday was 3.1 ± 1.6 years, which occurred after 5.2 ± 1.8 years of bisphosphonate use, and medication was restarted for an additional mean time of 2.5 ± 2.0 years (34). The patient in our study received 4 years of continuous treatment after a 3-year bisphosphonate holiday; not only was the drug effect unsatisfactory, but serious adverse events also occurred. There might be several possibilities to explain the case. First, despite a 3-year drug holiday, the cumulative duration of medication was too long to attenuate its efficacy. Second, the NR0B1 gene with the mutation site (c.572_575dup) encodes a truncated protein, which is related to bone metabolism and the function of osteoclasts and osteoblasts through an unknown mechanism. Hence, further pharmacogenomic analysis and functional research both in vitro and in vivo need to be performed.
Long-term hormone replacement and symptomatic treatment were often administered in AHC patients. Before puberty, male patients with HHG were treated with androgen to develop secondary sexual characteristics and improve their quality of life. Previous studies indicated that bone loss was irreversible even after sufficient testosterone replacement after puberty (28, 35). Therefore, we hypothesized that the bone health of AHC patients should be considered before skeletal maturation. Furthermore, with a diagnosis of osteopenia or osteoporosis, it is necessary to closely monitor the patient and be cautious about side effects, such as AFF and osteonecrosis of the jaw (ONJ), when using anti-osteoporotic drugs, especially bisphosphonates (33).
Anabolic drugs are another type of medication that act through changes in bone remodeling, bone modeling, or both and an increase in bone formation (36). Teriparatide, as a classical anabolic drug, is not ideal in patients at high risk of femoral neck or other cortical fractures. In addition, teriparatide is not approved for male patients or secondary osteoporosis in China. Given the above, the patient was advised to take menatetrenone instead. Menatetrenone has been proven to reduce osteoporotic fractures and improve hip bone strength despite its modest effect on BMD (37, 38). The mechanism of menatetrenone aims to promote osteocalcin γ-carboxylation, and only carboxylated osteocalcin has a weak osteogenic effect, which explains its mild effect on bone formation and increases in the osteocalcin level (37). After 3 months of treatment, the patient presented with greatly relieved bone pain, and the level of osteocalcin (bone formation marker) had increased substantially.
There is an explanation for the inconsistent changes in BMD in different bone sites after treatment. When this patient was initially treated with alendronate, the BMD of the femoral neck increased significantly (Z score from −2.9 to −2.2), which indicated that bisphosphonate treatment was effective. Because zoledronic acid is more potent than alendronate, we administered zoledronic acid to increase BMD and reduce BTMs. However, unexpectedly, atypical fractures occurred. Improvement in the BMD of the femoral neck requires weight-bearing and mechanical stimulation in addition to drug effects. Due to the AFF, lower extremity pain led to limited movement of the patient, which was also the reason for the decrease in BMD in the femoral neck (Z score from −2.2 to −2.6). After surgical treatment, his mobility was significantly improved and combined with a bone formation agent, he had an increased BMD in his femoral neck (Z score from −2.6 to −2.2). However, Z score dropped to −2.5 by 2022, and therefore there is no evidence of any effect of vitamin K on the neck of the femur.
In conclusion, the present study described the clinical features and bone complications in a male patient with secondary osteoporosis induced by AHC who was followed up for 5 years and adjusted anti-osteoporosis treatments three times. It is suggested that for adult patients with AHC, close attention should be paid to skeletal complications, especially when combined with glucocorticoid therapy. It is of great importance to evaluate bone mass and provide treatment as early as possible, accompanied by regular follow-up. Furthermore, anabolic agents may be a better choice for the treatment of osteoporosis secondary to AHC.
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
Ethics statement
The studies involving human participants were reviewed and approved by the Ethics Committee of Shanghai Jiao Tong University school of medicine Affiliated Sixth People’s Hospital. The patients/participants provided their written informed consent to participate in this study.
Author contributions
XT and TX drafted the manuscript. LL, XL, XT, and TX collected follow-up data. ZZ, HY, XT, and TX revised the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by the National Key Research and Development Program of China (No. 2018YFA0800801); the National Natural Science Foundation of China (NSFC) (Nos.81974126; 81770874); the Clinical Science and Technology Innovation Project of Shanghai Shenkang Hospital Development Center (No. SHDC12018120); and the Shanghai Key Clinical Center for Metabolic Disease, Shanghai Health Commission Grant (No. 2017ZZ01013).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2022.961322/full#supplementary-material
References
1. Painter SE, Kleerekoper M, Camacho PM. Secondary osteoporosis: A review of the recent evidence. Endocrine Pract (2006) 12:436–45. doi: 10.4158/EP.12.4.436
2. Lewiecki EM. Evaluating patients for secondary causes of osteoporosis. Curr Osteoporosis Rep (2022) 20:1–12. doi: 10.1007/s11914-022-00717-y
3. Shevroja E, Cafarelli FP, Guglielmi G, Hans D. DXA parameters, trabecular bone score (TBS) and bone mineral density (BMD), in fracture risk prediction in endocrine-mediated secondary osteoporosis. Endocrine (2021) 74:20–8. doi: 10.1007/s12020-021-02806-x
4. Reutens AT, Achermann JC, Ito M, Ito M, Gu WX, Habiby RL, et al. Clinical and functional effects of mutations in the DAX-1 gene in patients with adrenal hypoplasia congenita. J Clin Endocrinol Metab (1999) 84:504–11. doi: 10.1210/jc.84.2.504
5. Guo WW, Burris TP, Mccabe ERB. Expression of DAX-1, the gene responsible for X-linked adrenal hypoplasia congenita and hypogonadotropic hypogonadism, in the hypothalamic-Pituitary-Adrenal/Gonadal axis. Biochem Mol Med (1995) 56:8–13. doi: 10.1006/bmme.1995.1049
6. Laverty CRA, Fortune DW, Beischer NA. Congenital idiopathic adrenal hypoplasia. Obstet Gynecol (1973) 41:655–64. doi: 10.1016/S0031-3025(16)39226-1
7. Lin L, Gu WX, Ozisik G, To WS, Owen CJ, Jameson JL, et al. Analysis of DAX1 (NR0B1) and steroidogenic factor-1 (NR5A1) in children and adults with primary adrenal failure: Ten years' experience. J Clin Endocrinol Metab (2006) 91:3048–54. doi: 10.1210/jc.2006-0603
8. Sikl H. Addison's disease due to congenital hypoplasia of the adrenals in an infant aged 33 days. J Pathol Bacteriol (1948) 60:323. doi: 10.1002/path.1700600220
9. Kyriakakis N, Shonibare T, Kyaw-Tun J, Lynch J, Lagos CF, Achermann JC, et al. Late-onset X-linked adrenal hypoplasia (DAX-1, NR0B1): two new adult-onset cases from a single center. Pituitary (2017) 20:585–93. doi: 10.1007/s11102-017-0822-x
10. Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schütz G, Umesono K, et al. The nuclear receptor superfamily: The second decade. Cell (1995) 83:835–9. doi: 10.1016/0092-8674(95)90199-X
11. Vilaca T, Eastell R, Schini M. Osteoporosis in men. Lancet Diabetes Endocrinol (2022) 10:273–83. doi: 10.1016/S2213-8587(22)00012-2
12. Baim S, Wilson CR, Lewiecki EM, Luckey MM, Downs RW, Lentle BC. Precision assessment and radiation safety for dual-energy X-ray absorptiometry: position paper of the international society for clinical densitometry. J Clin Densitometry (2005) 8:371–8. doi: 10.1385/JCD:8:4:371
13. Muscatelli F, Strom TM, Walker AP, Zanaria E, Récan D, Meindl A, et al. Mutations in the DAX-1 gene give rise to both X-linked adrenal hypoplasia congenita and hypogonadotropic hypogonadism. Nature (1994) 372:672–6. doi: 10.1038/372672a0
14. Guo W, Burris TP, Zhang YH, Huang BL, Mason J, Copeland KC, et al. Genomic sequence of the DAX1 gene: An orphan nuclear receptor responsible for X-linked adrenal hypoplasia congenita and hypogonadotropic hypogonadism. J Clin Endocrinol Metab (1996) 81:2481–6. doi: 10.1210/jc.81.7.2481
15. Burris TP, Guo W, Mccabe ER. The gene responsible for adrenal hypoplasia congenita, DAX-1, encodes a nuclear hormone receptor that defines a new class within the superfamily. Recent Prog Horm Res (1996) 51:241–59; discussion 259-60.
16. García-Acero M, Molina M, Moreno O, Ramirez A, Forero C, Céspedes C, et al. Gene dosage of DAX-1, determining in sexual differentiation: duplication of DAX-1 in two sisters with gonadal dysgenesis. Mol Biol Rep (2019) 46:2971–8. doi: 10.1007/s11033-019-04758-y
17. Lalli E. Role of orphan nuclear receptor DAX-1/NR0B1 in development, physiology, and disease. Adv Biol (2014) 2014:582749. doi: 10.1155/2014/582749
18. Habiby RL, Boepple P, Nachtigall L, Sluss PM, Crowley ,WF Jr., Jameson JL. Adrenal hypoplasia congenita with hypogonadotropic hypogonadism: evidence that DAX-1 mutations lead to combined hypothalmic and pituitary defects in gonadotropin production. J Clin Invest (1996) 98:1055–62. doi: 10.1172/JCI118866
19. Li N, Liu R, Zhang H, Yang J, Sun S, Zhang M, et al. Seven novel DAX1 mutations with loss of function identified in Chinese patients with congenital adrenal hypoplasia. J Clin Endocrinol Metab (2010) 95:E104–11. doi: 10.1210/jc.2009-2408
20. Krone N, Riepe FG, Dörr HG, Morlot M, Rudorff KH, Drop SL, et al. Thirteen novel mutations in the NR0B1 (DAX1) gene as cause of adrenal hypoplasia congenita. Hum Mutat (2005) 25:502–3. doi: 10.1002/humu.9331
21. Buonocore F, Achermann JC. Primary adrenal insufficiency: New genetic causes and their long-term consequences. Clin Endocrinol (Oxf) (2020) 92:11–20. doi: 10.1111/cen.14109
22. Goodfellow PN, Camerino G. DAX-1, an 'antitestis' gene. Cell Mol Life Sci (1999) 55:857–63. doi: 10.1210/jc.81.7.2481
23. Wu SM, Gao JZ, He B, Long WJ, Luo XP, Chen L. A novel NR0B1 gene mutation causes different phenotypes in two Male patients with congenital adrenal hypoplasia. Curr Med Sci (2020) 40:172–7. doi: 10.1007/s11596-020-2161-9
24. Laissue P, Copelli S, Bergada I, Bergada C, Barrio G, Karaboga S, et al. Partial defects in transcriptional activity of two novel DAX-1 mutations in childhood-onset adrenal hypoplasia congenita. Clin Endocrinol (Oxf) (2006) 65:681–6. doi: 10.1111/j.1365-2265.2006.02649.x
25. Bertalan R, Bencsik Z, Mezei P, Vajda Z, Butz H, Patócs A. Novel frameshift mutation of the NR0B1(DAX1) in two tall adult brothers. Mol Biol Rep (2019) 46:4599–604. doi: 10.1007/s11033-019-04688-9
26. Golds G, Houdek D, Arnason T. Male Hypogonadism and osteoporosis: The effects, clinical consequences, and treatment of testosterone deficiency in bone health. Int J Endocrinol (2017) 2017:4602129. doi: 10.1155/2017/4602129
27. Simsek-Kiper PO, Dikoglu E, Campos-Xavier B, Utine GE, Bonafe L, Unger S, et al. Positive effects of an angiotensin II type 1 receptor antagonist in camurati-engelmann disease: A single case observation. Am J Med Genet A (2014) 164a:2667–71. doi: 10.1002/ajmg.a.36692
28. De Rosa ,M, Paesano ,L, Nuzzo ,V, Zarrilli ,S, Del Puente ,A, Oriente ,P, et al. Bone mineral density and bone markers in hypogonadotropic and hypergonadotropic hypogonadal men after prolonged testosterone treatment. J Endocrinol Invest (2001) 24:246–52. doi: 10.1007/BF03343854
29. Francke U, Harper JF, Darras BT, Cowan JM, Mccabe ER, Kohlschütter A, et al. Congenital adrenal hypoplasia, myopathy, and glycerol kinase deficiency: molecular genetic evidence for deletions. Am J Hum Genet (1987) 40:212–27.
30. Chotiyarnwong P, Mccloskey EV. Pathogenesis of glucocorticoid-induced osteoporosis and options for treatment. Nat Rev Endocrinol (2020) 16:437–47. doi: 10.1038/s41574-020-0341-0
31. Eastell R, Szulc P. Use of bone turnover markers in postmenopausal osteoporosis. Lancet Diabetes Endocrinol (2017) 5:908–23. doi: 10.1016/S2213-8587(17)30184-5
32. Vlot MC, Den Heijer ,M, De Jongh ,RT, Vervloet MG, Lems WF, De Jonge ,R, et al. Clinical utility of bone markers in various diseases. Bone (2018) 114:215–25. doi: 10.1016/j.bone.2018.06.011
33. Black DM, Geiger EJ, Eastell R, Vittinghoff E, Li BH, Ryan DS, et al. Atypical femur fracture risk versus fragility fracture prevention with bisphosphonates. New Engl J Med (2020) 383:743–53. doi: 10.1056/NEJMoa1916525
34. Adams AL, Adams JL, Raebel MA, Tang BT, Kuntz JL, Vijayadeva V, et al. Bisphosphonate drug holiday and fracture risk: A population-based cohort study. J Bone Mineral Res (2018) 33:1252–9. doi: 10.1002/jbmr.3420
35. Wong FH, Pun KK, Wang C. Loss of bone mass in patients with klinefelter's syndrome despite sufficient testosterone replacement. Osteoporos Int (1993) 3:3–7. doi: 10.1007/BF01623169
36. Reid IR, Billington EO. Drug therapy for osteoporosis in older adults. Lancet (London England) (2022) 399:1080–92. doi: 10.1016/S0140-6736(21)02646-5
37. Iwamoto J, Takeda T, Sato Y. Menatetrenone (vitamin K2) and bone quality in the treatment of postmenopausal osteoporosis. Nutr Rev (2006) 64:509–17. doi: 10.1111/j.1753-4887.2006.tb00184.x
Keywords: secondary osteoporosis, X-linked congenital adrenocortical hypoplasia, gene mutation, NR0B1 gene, efficacy of treatment
Citation: Tao X, Xu T, Liu L, Lin X, Zhang Z and Yue H (2022) Case report: Clinical characteristics and treatment of secondary osteoporosis induced by X-linked congenital adrenal dysplasia. Front. Endocrinol. 13:961322. doi: 10.3389/fendo.2022.961322
Received: 04 June 2022; Accepted: 21 November 2022;
Published: 08 December 2022.
Edited by:
Elżbieta Skowrońska-Jóźwiak, Medical University of Łódź, PolandReviewed by:
Voraluck Phatarakijnirund, Phramongkutklao Hospital, ThailandVolha Zhukouskaya, Universitéde Paris, France
Copyright © 2022 Tao, Xu, Liu, Lin, Zhang and Yue. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hua Yue, eXVleWluZ2xvbmdodWFAMTYzLmNvbQ==; Zhenlin Zhang, emhhbmd6bEBzanR1LmVkdS5jbg==
†These authors have contributed equally to this work and share first authorship
 Xiaohui Tao
Xiaohui Tao Tian Xu
Tian Xu Li Liu
Li Liu Xiaoyun Lin
Xiaoyun Lin Zhenlin Zhang
Zhenlin Zhang Hua Yue
Hua Yue