
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Endocrinol. , 20 October 2022
Sec. Reproduction
Volume 13 - 2022 | https://doi.org/10.3389/fendo.2022.942368
This article is part of the Research Topic Endometriosis: Pathogenesis, Diagnosis and Treatment, volume II View all 10 articles
 Sung-Jin Bae1†
Sung-Jin Bae1† Yunju Jo2†
Yunju Jo2† Min Kyoung Cho3
Min Kyoung Cho3 Jung-Sook Jin3
Jung-Sook Jin3 Jin-Young Kim4
Jin-Young Kim4 Jaewon Shim5
Jaewon Shim5 Yun Hak Kim6,7
Yun Hak Kim6,7 Jang-Kyung Park8
Jang-Kyung Park8 Dongryeol Ryu2
Dongryeol Ryu2 Hyun Joo Lee9
Hyun Joo Lee9 Jongkil Joo9*
Jongkil Joo9* Ki-Tae Ha3,4*
Ki-Tae Ha3,4*Endometriosis is a gynecological disease prevalent in women of reproductive age, and it is characterized by the ectopic presence and growth of the eutopic endometrium. The pathophysiology and diagnostic biomarkers of endometriosis have not yet been comprehensively determined. To discover molecular markers and pathways underlying the pathogenesis of endometriosis, we identified differentially expressed genes (DEGs) in three Gene Expression Omnibus microarray datasets (GSE11691, GSE23339, and GSE7305) and performed gene set enrichment analysis (GSEA) and protein–protein interaction (PPI) network analyses. We also validated the identified genes via immunohistochemical analysis of tissues obtained from patients with endometriosis or healthy volunteers. A total of 118 DEGs (79 upregulated and 39 downregulated) were detected in each dataset with a lower (fold change) FC cutoff (log2|FC| > 1), and 17 DEGs (11 upregulated and six downregulated) with a higher FC cutoff (log2|FC| > 2). KEGG and GO functional analyses revealed enrichment of signaling pathways associated with inflammation, complement activation, cell adhesion, and extracellular matrix in endometriotic tissues. Upregulation of seven genes (C7, CFH, FZD7, LY96, PDLIM3, PTGIS, and WISP2) out of 17 was validated via comparison with external gene sets, and protein expression of four genes (LY96, PDLIM3, PTGIS, and WISP2) was further analyzed by immunohistochemistry and western blot analysis. Based on these results, we suggest that TLR4/NF-κB and Wnt/frizzled signaling pathways, as well as estrogen receptors, regulate the progression of endometriosis. These pathways may be therapeutic and diagnostic targets for endometriosis.
Endometriosis is a common gynecological disorder in which endometrial tissue grows outside the uterus (1). Endometriosis affects around 10% of women of reproductive age and often causes dysmenorrhea, chronic pelvic pain, and infertility (1, 2). Despite the widespread acceptance of the retrograde menstruation theory proposed by Sampson in 1927, the pathogenesis of endometriosis remains poorly understood (3). Thus, various other factors, including genetic, epigenetic, stem cell, inflammatory, angiogenic, and immunological factors, should be considered to better understand the complex pathophysiology of endometriosis (3, 4). Gynecological surgery is the major therapeutic option for endometriosis treatment. Oral contraceptives, progestins, nonsteroidal anti-inflammatory drugs, and gonadotropin-releasing hormone agonists are alternative treatment options (2, 5). However, the efficacy of these treatment strategies, whether surgical or non-surgical, is still limited due to the high recurrence rate of the disease.
Despite its high prevalence, the diagnosis of endometriosis is often delayed as it has no symptoms distinct from those of ordinary menstrual cramps (6, 7). Histopathological examination by invasive laparoscopy or surgery is the gold standard for diagnosis of endometriosis (2). The identification of diagnostic biomarkers is thus urgently required to improve the diagnosis and treatment of patients with endometriosis. Previous reviews on this topic have focused on identification of potential biomarker candidates from specimens such as peritoneal fluid, blood, urine, and endometrial biopsies (6–9), highlighting several factors as noninvasive diagnostic biomarkers, including growth factors, hormones, cytokines, complements, glycoproteins, and antibodies. However, these biomarkers are merely used to supplement diagnosis of endometriosis, as none of them has demonstrated sufficient sensitivity and specificity (10).
Integration of different types of omics data is routinely utilized to discover and validate novel disease biomarkers (11, 12). Potential diagnostic biomarkers and therapeutic targets of endometriosis have been proposed in such integrative bioinformatics studies based on the identification of differentially expressed genes (DEGs) (13–16). However, identification of common biomarkers that are consistently detected in all datasets is difficult due to the heterogeneity among independent datasets. Here, three microarray datasets (GSE11691, GSE23339, and GSE7305), which include gene expression data from normal endometrial and endometriosis tissues, were obtained from the Gene Expression Omnibus (GEO) database. Non-biased bioinformatics analyses, including identification of DEGs, gene set enrichment analysis (GSEA), and protein–protein interaction (PPI) network analysis, were conducted, and the findings were further validated by analyzing immunohistochemistry (IHC) and western blot analysis of tissue specimens obtained from patients with endometriosis or healthy volunteers. Based on the obtained results, we propose six biomarkers as potential targets for the diagnosis and treatment of endometriosis.
The datasets for analysis in this study were chosen according to the inclusion and exclusion criteria summarized in Supplementary Figure 1. Briefly, the gene sets were obtained by searching the Gene Expression Omnibus (GEO; https://www.ncbi.nlm.nih.gov/geo) database and filtered by three different criteria, including characteristics of data, experiment, and sample. Among six datasets filtered by three criteria, GSE11691, GSE23339, and GSE7305 were applied for DEG and GSEA analysis. Whereas GSE135485 and GSE25628 were used as validation datasets due to their imbalanced and small sample sizes, GSE6364 was filtered out as it contained the data from normal endometrium but not endometriosis. The gene expression data used in this study (GSE11691, GSE23339, GSE7305, GSE135485, and GSE25628) were downloaded from the GEO database, and a total of 128 samples were collected. GSE11691 using the GPL96 platform includes data obtained from nine endometriosis and normal uterine endometrium samples, respectively (17). GSE23339 includes data derived from 10 endometriosis and nine normal uterine endometrium samples using the GPL6102 platform I (18). GSE7305 using the GPL570 platform includes data collected from 10 endometriosis and normal uterine endometrium samples, respectively (19). GSE135485 includes data collected from 54 endometriosis and four normal uterine endometrium samples using the GPL21290 platform. GSE25628 includes data from seven endometriosis and six normal uterine endometrium samples using the GPL571 platform (20). Detailed information on the datasets is summarized in Table 1.
Transcriptome analysis was conducted using R (version 4.1.1) via RStudio (Desktop version, 1.4.1717). The three datasets selected for DEG identification (GSE11691, GSE23339, and GSE7305) were downloaded from the GEO database of the National Center for Biotechnology Information using the GEOquery R package (17–19, 21). Multiple probes related to the same gene were reduced to one, and summarized as median values for further analysis. Since gene expression profiles differed between samples included in GSE11691, quantile normalization was applied using the preprocessCore R package (https://github.com/bmbolstad/preprocessCore). DEGs were defined as genes with adjusted p-values and Log2|FoldChange| (Log|FC|) less and greater than 0.05 and 1, respectively. We adjusted p-value to correct the false positive error caused by the multiple tests and calculated it by the Benjamini & Hochberg method (22), which is one of the popular tools to minimize the false discovery rate. The cutoff criteria that we applied was 0.1. For analysis of the pathway and PPI of DEGs, we adopted Log|FC| > 1 and used Log|FC| > 2 for heatmap and network analysis.
Analysis of Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) biological processes of DEGs were performed using the JEPETTO plugin (version 1.3.1) of Cytoscape (version 3.8.2). Visualization was performed by constructing a scatter plot with XD-score and q-value as axes. q-value < 0.25 was used as the cutoff criterion, according to GSEA guidance (https://software.broadinstitute.org/cancer/software/gsea/wiki/index.php/FAQ).
The interactions of proteins encoded by the DEGs were identified using the STRING plugin (version 1.7.0) of Cytoscape by using “Homo sapiens” as search keyword, and a confidence score cutoff higher than 0.4. GeneMANIA plugin (version 3.5.2) was used to identify physical interactions. Visualization of the network was performed using Cytoscape.
A heatmap of the top 17 DEGs identified in each dataset was generated using Morpheus, a versatile matrix visualization and analysis software from the Broad Institute at the Massachusetts Institute of Technology (https://software.broadinstitute.org/morpheus/).
Network analysis was performed as previously described (23). Briefly, the pattern of co-expression between the normal and endometriosis groups was visualized based on Spearman’s correlation. For this purpose, dplyr, stringr, ggpubr, ggplot2, igraph, ggraph, corrr, corrplot, tidyverse, and reshape2 R packages were applied.
GSEA of each gene expression dataset was performed using the GSEA software (version 4.1.0) from the Broad Institute at the Massachusetts Institute of Technology (https://www.gsea-msigdb.org/gsea/index.jsp). The absolute value of the normalized enrichment score (NES), the enrichment score for the gene set after it had been normalized across analyzed gene sets, was set to > 1.5 as the cutoff criteria. The false discovery rate (FDR) q-value, which represents the estimated probability that the normalized enrichment score constitutes a false-positive finding, was set to < 0.02. GO network analyses were performed on each GSEA by utilizing Cytoscape to visualize enrichment maps.
Laparoscopic surgeries were performed at the Department of Obstetrics and Gynecology of Pusan National University Hospital (Busan, Korea) between June 2019 and December 2020. The surgical process was performed during the early follicular phase to rule out the potential early pregnancy and the possibility of the ovarian cyst being functional (24, 25). To obtain normal uterine endometrial tissue as the control group, we gathered the patients with male-factor infertility with normal gynecologic anatomy who underwent endometrial scratch prior to proceeding to programmed or natural embryo-transfer cycle, and the patients with hormone-independent ovarian cysts such as mature teratoma or cystadenoma who underwent laparoscopy and presented no endometriotic lesion in the pelvic cavity. The exclusion criteria were irregular menstrual periods, the presence of endocrine disorders such as hyperprolactinemia or thyroid dysfunction, and medication history with dysmenorrhea management such as GnRH analogs, oral contraceptives, or progestins during the past three months to the recruitment. Eligible patients were further examined and proven morphologically free from possible asymptomatic endometriosis and other hormone-dependent gynecological pathologies, such as uterine adenomyosis and leiomyoma, by undergoing imaging studies including gynecological ultrasonography, abdominal computed tomography scans and/or pelvic magnetic resonance imaging. All specimens from the control group were further histologically assessed and confirmed as normal endometrial tissue by pathologists. Endometriosis was pathologically diagnosed in tissue specimens derived from 32 patients. Endometriosis was classified into stages I-IV according to the revised American Society for Reproductive Medicine (r-ASRM) classification system. The average stage detected in the samples was approximately III. The average age of the patients was 33.33 ± 7.53 years, whereas the average BMI was 21.51 ± 3.52. Samples of normal endometrial tissue were obtained from ten healthy volunteers who have not been diagnosed with endometriosis. The average age of the volunteers was 30.3 ± 4.9 years, whereas the average BMI was 22.06 ± 2.06. There was no statistically significant difference between the average age and BMI values of the two groups (with p-values of 0.16 and 0.56). Tissue specimens were deposited in the Biobank of the Pusan National University Hospital. Detailed information on the patient and normal volunteer is summarized in Supplementary Table 1.
This study was approved by the Institutional Review Board (IRB) of Pusan National University Hospital (2104-009-101). All patients signed informed consents for the study protocol. All procedures were conducted in accordance with IRB guidelines.
Fresh tissues collected by the laparoscopic surgeries were rinsed with normal saline to remove blood and impurities, and rapidly frozen and stored in the Biobank of Pusan National University Hospital. The donated specimens were fixed with 4% formaldehyde solution, and processed to obtain paraffin embedded tissue blocks. Five–μm thick sections of tissue blocks were deparaffinized by soaking in xylene and gradient ethanol solution. The sections were then incubated with primary antibodies, including anti-lymphocyte antigen 96 (LY96; 1:200; ab22048, Abcam), anti-PDZ And LIM Domain 3 (PDLIM3; 1:200; HPA004749, Atlas Antibodies, Bromma, Sweden), anti-prostaglandin I2 synthase (PTGIS; 1:200; ab23668, Abcam), and anti-WNT1-inducible-signaling pathway protein 2 (WISP2; 1:200; ab28317, Abcam) at 4°C overnight. The sections were then incubated with Dako REAL EnVision Detection System (K5007; Dako, Jena, Germany) for 1 h. After rinsing with phosphate-buffered saline (PBS), immunostaining was visualized using DAB+ chromogen buffer (K5007, Dako). The slides were counterstained with hematoxylin solution, and representative images were taken using an optical microscope (Axio Scope A1; Carl Zeiss, Oberkochen, Germany). Histopathological scoring was performed through examination under a light microscope by a pathologist. Staining intensity was classified as follows: 0, negative immunostaining; 1, weak expression level; 2, moderate expression level; 3, strong expression level; and 4, very strong expression level.
Total proteins were extracted from frozen tissue specimens using protein lysis buffer containing 10 mM HEPES pH 7.45, 150 mM sodium chloride, 1% (w/v) NP−40, 5 mM sodium pyrophosphate, 5 mM sodium fluoride, and 2 mM sodium vanadate with a protease inhibitor cocktail (Roche Applied Science, Penzburg, Germany). 30 μg of protein lysates was electrophoresed by sodium dodecyl sulfate−polyacrylamide gel and transferred onto nitrocellulose membranes (0.45 μm; ThermoFisher Scientific, Waltham, MA). The membranes were blocked with 5% (w/v) non-fat dry milk and incubated with primary antibodies against target proteins, including LY96, PDLIM3, PTGIS, and WISP2 at 4°C overnight. The membranes were washed and incubated with proper secondary antibodies conjugated with horseradish peroxidase. The bands of interesting proteins were detected with the enhanced chemiluminescence Plus kit (ThermoFisher Scientific) using ImageQuant LAS 4000 imaging system (GE healthcare, Chicago, IL).
The Drug-Gene Interaction Database (DGIdb, http://www.dgidb.org) is an online database that facilitates interpretation of the results of genome-wide studies, and generation of hypotheses in the context of druggable genome (26). DGIbd was used to identify potentially druggable genes from an input list of genes including the statistically significant DEGs.
To evaluate the statistical difference between the two groups, Student’s t-test was performed using GraphPad Prism (version 5.01; GraphPad Software, San Diego, CA, USA). Statistical significance was set at p < 0.05.
We first determined the distributions of gene expression levels in three human endometrial transcriptome datasets, GSE11691, GSE23339, and GSE7305 (17–19). The transcriptome profiles of GSE11691 showed the highest level of variation among the three transcriptomes. Thus, we conducted quantile normalization to avoid artifacts in subsequent analyses. The gene expression data included in GSE23339 and GSE7305 datasets were converted to Log2 scale (Figure 1A), and DEGs were then identified using these three datasets. As highlighted in the colored volcano plots (Figure 1B), a total of 536 (361 upregulated and 175 downregulated), 1,042 (562 upregulated and 480 downregulated), and 1,515 (882 upregulated and 633 downregulated) DEGs were identified in GSE11691, GSE23339, and GSE7305, respectively (Figure 1C). Moreover, integration of DEGs shared across the datasets revealed 118 (79 upregulated and 39 downregulated; Figure 1C) and 17 (11 upregulated and six downregulated; Figure 1D) common DEGs with a lower (fold change) FC cutoff (log2|FC| > 1) and a higher FC cutoff (log2|FC| > 2), respectively (Table 2).
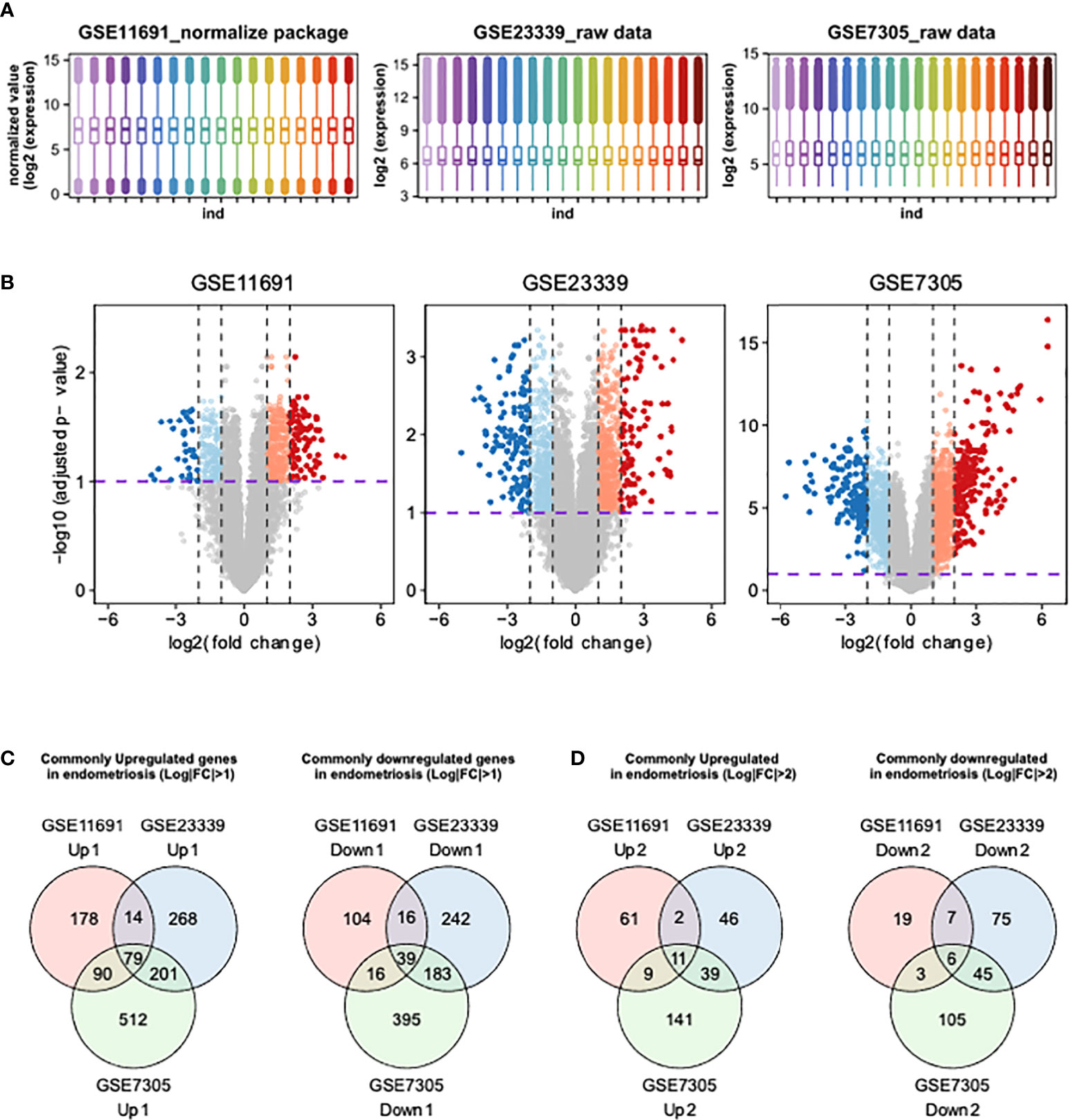
Figures 1 Identification of DEGs using integrative bioinformatics analysis. (A) The distribution of transcriptome obtained from GSE11691, GSE23339, and GSE7305 were shown. (B) The volcano plots show the DEGs from GSE11691, GSE23339, and GSE7305. Red indicates relative upregulated genes with p-value < 0.05 and Log2|FC| > 2, orange indicates relative upregulated genes with p-value < 0.05 and 1 < Log2|FC| ≤ 2, blue indicates relative downregulated genes with p < 0.05 and Log2|FC| > 2, and light cyan indicates relative downregulated genes with p < 0.05 and 1 < Log2|FC| ≤ 2. (C, D) Venn diagrams of upregulated and downregulated genes DEGs from three indicated datasets. The cutoff values of Log2|FC| were set to 1 (C) and 2 (D).
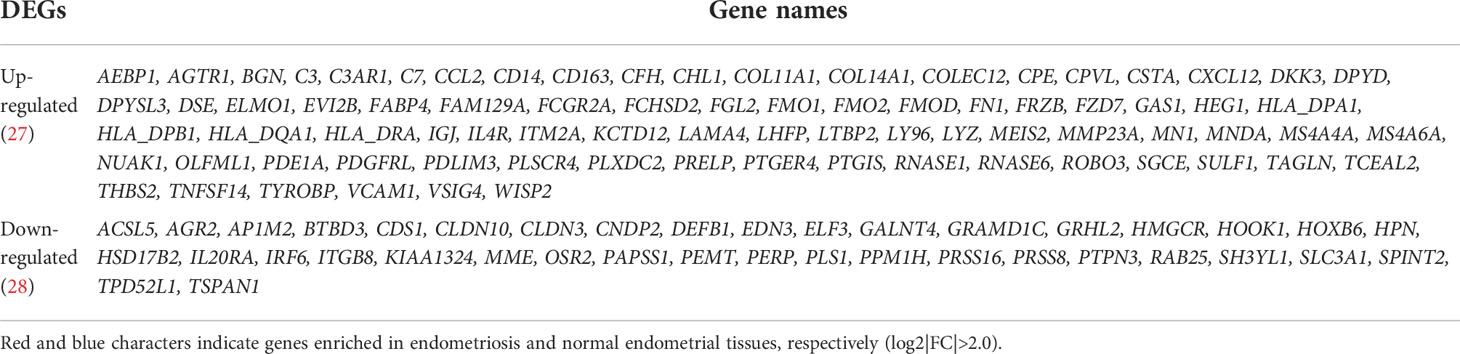
Table 2 The list of identified DEGs in three endometriosis datasets, GSE11691, GSE23339, and GSE7305.
To elucidate the pathways and molecular functions related to the 118 common DEGs identified above, KEGG and GO enrichment analyses were conducted. The analysis results revealed the involvement of the identified DEGs in the extracellular matrix, cell adhesion, complement activation, immune response, and inflammation processes (Figure 2A).
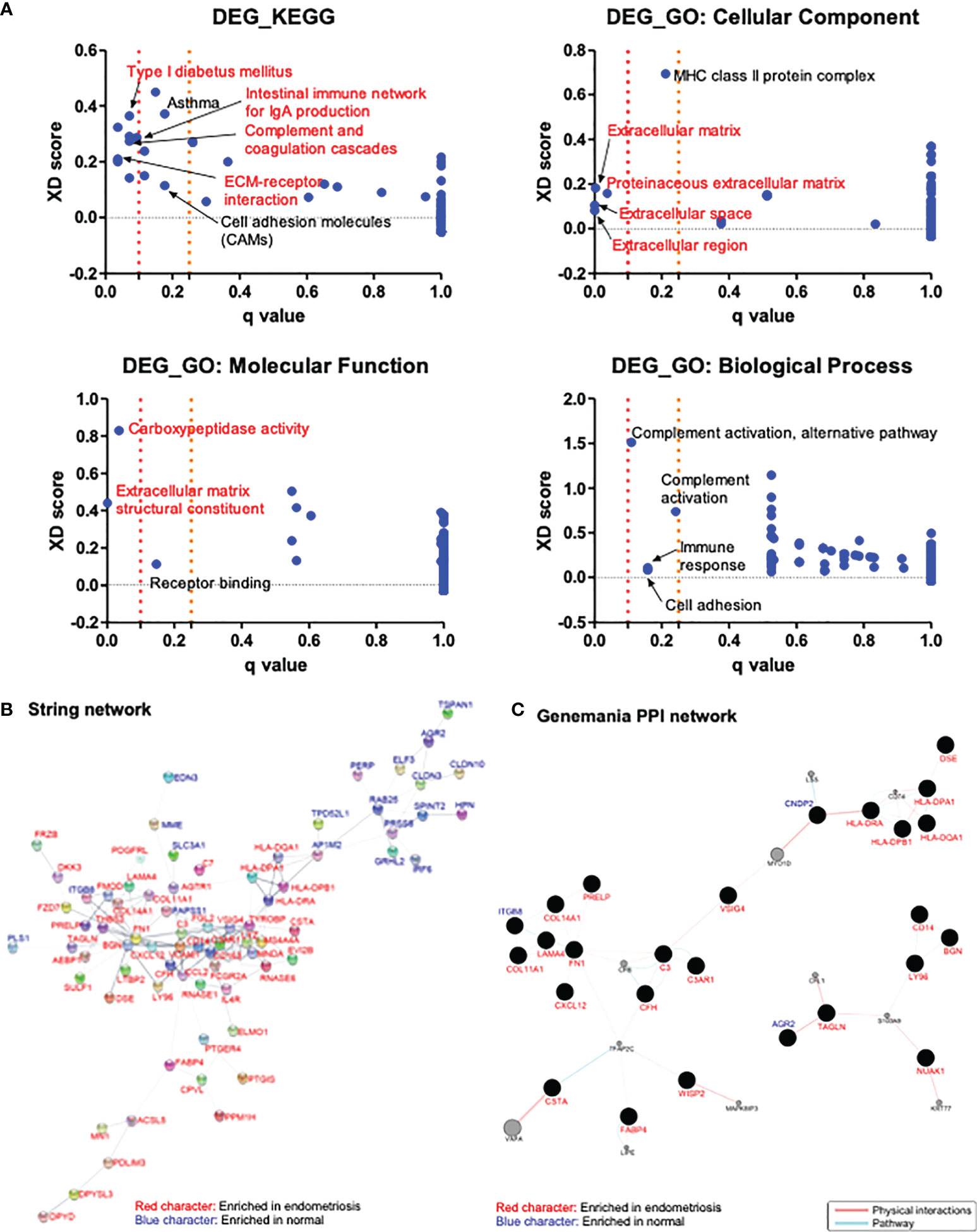
Figure 2 Analysis of the enriched pathways and interaction networks of DEGs in endometriosis. (A) KEGG and GO pathway analysis of DEGs from GSE11691, GSE23339, and GSE7305 datasets with Log2|FC| > 1 were performed with JEPETTO plugin of Cytoscape, and scatter plot were constructed with q-value and XD-score as x- and y-axis, respectively. Red character indicates the pathways with q-value < 0.1 and black character indicates the pathways with 0.1 < q-value < 0.25. (B, C) Protein–protein interaction network of DEGs from three datasets with Log2|FC| > 1 were analyzed in Cytoscape with plugins of STRING (B) and GeneMANIA (C). Red character indicates genes enriched in endometriosis and blue character indicates genes enriched in normal.
Interactions between proteins encoded by 118 DEGs identified above were analyzed using the STRING and GeneMANIA plugins of Cytoscape. STRING analysis resulted in a network comprising 118 nodes (genes) and 197 edges (interactions). A total of 37 nodes did not have any edges, and four nodes had only few edges. The rest of the network, including 77 genes and 195 interactions is shown in Figure 2B. The genes AP1M2, BGN, C3, C3AR1, CCL2, CD14, CD163, COL14A1, FCGR2A, FGL2, FN1, LYZ, MS4A4A, RNASE6, TYROBP, and VCAM1 had relatively higher number of interactions (over four edges). GeneMANIA analysis resulted in a network consisting of 138 nodes and 72 edges. Among these nodes, 77 genes did not have any physical or pathway interactions, and 24 genes had merely simple interactions. The main interaction network harboring 37 nodes and 57 edges is shown in Figure 2C. The genes showing relatively higher number of interactions (over three edges) in the network were identified to be C3, CFH, CNDP2, COL11A1, COL14A1, FABP4, FN1, HLA-DPA1, HLA-DPB1, HLA-DQA1, HLA-DRA, ITGB8, LAMA4, LY96, and TAGLN. STRING analysis revealed CCL2 as significant (with over three edges), based on 15 common DEGs with a cutoff value of Log2|FC| > 2. On the other hand, GeneMANIA revealed that three proteins encoded by AGR2, FZD7, and LY96 were in physical or pathway contact with each other (Supplementary Figure 2).
The heatmaps (Figure 3A) depicted the expression levels of the 17 frequent DEGs with p-values less than 0.05 and Log2|FC| more than 2, clearly illustrating the differential expression patterns of those selected genes in endometriosis compared to normal control across three independent datasets. In addition, gene networks showing the co-expression patterns of the selected genes were constructed based on Spearman’s correlation. The 17 DEGs were highly associated with one another in three independent datasets from normal tissues, and thereby generated a massive co-expression gene network (Figure 3B). On the other hand, the number of correlations, shown as edges, was drastically reduced in ectopic lesions of endometriosis patients compared to normal sites. Interestingly, these patterns of gene networks with decreased correlations under endometriosis were comparable across the three independent datasets. Hence, the decrease in correlations between these 17 DEGs may contribute to the pathophysiology of endometriosis.
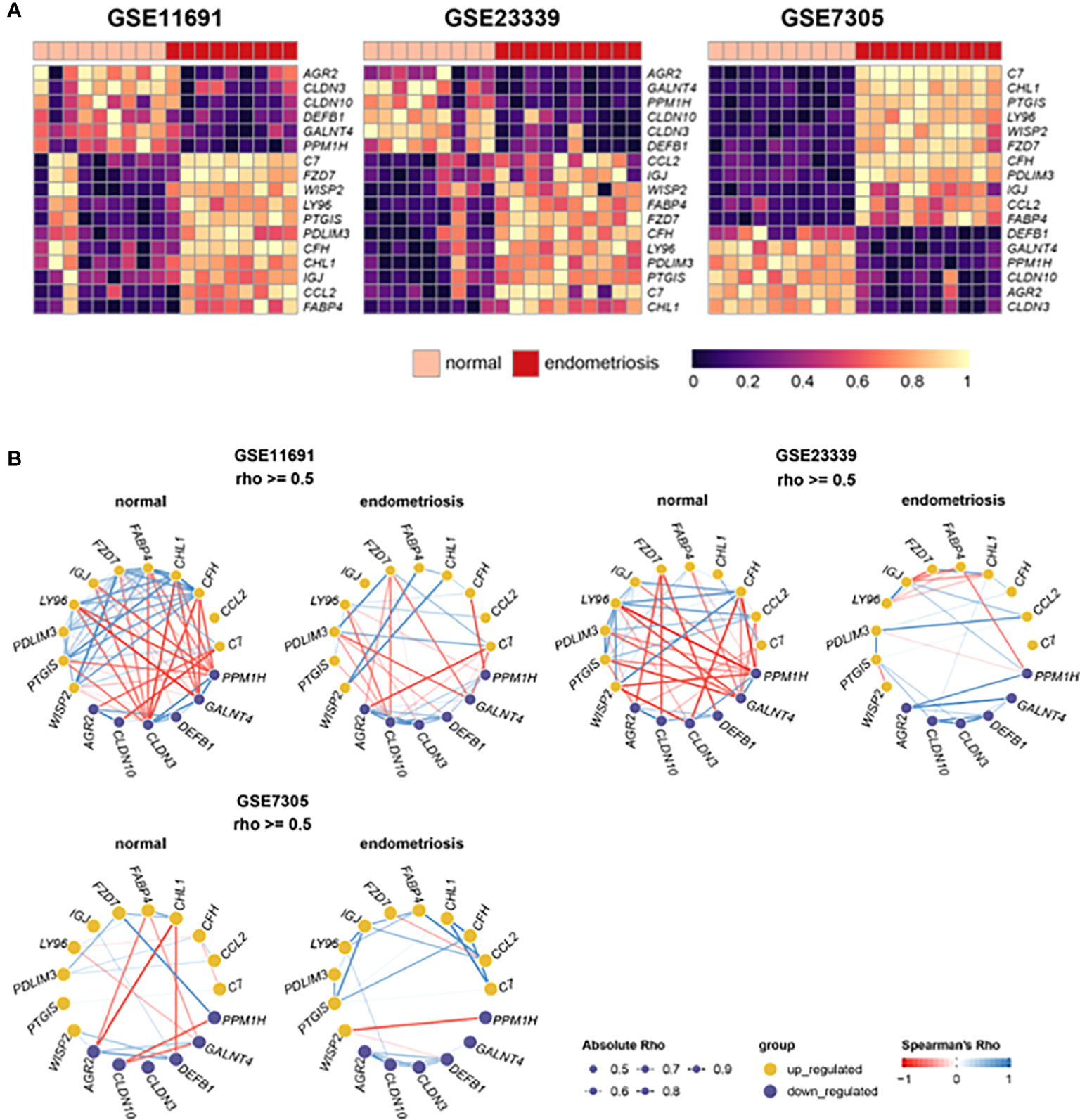
Figure 3 Expression and correlation of 22 DEGs with Log2|FC| > 2. (A) Heat maps show the expression levels of 22 DEGs in datasets, GSE11691, GSE23339, and GSE7305. (B) Gene networks display correlations of 22 DEGs in the normal and endometrial tissues of each dataset. The depth of edges indicates absolute Rho ranging from 0.5 to 1. The color of edges indicates co-expression ranging from -1 (red) to 1 (blue) by Spearman’s Rho.
GSEA was carried out to identify functional pathways in endometriosis that are shared across the three datasets analyzed (GSE11691, GSE23339, and GSE7305). A total of 11 common pathways were identified in the hallmark analysis, including 9 upregulated and 2 downregulated pathways (Figure 4A). Inflammation-related pathways such as IL6-JAK/STAT3 signaling, inflammatory response, interferon α response, interferon γ response, and TNFα signaling via NF-κB, were found to be significantly upregulated in patients with endometriosis. In addition, nine common pathways were found to be upregulated in KEGG analysis (Figure 4B). Further, inflammation-related pathways including chemokine signaling pathway and NOD-like receptor signaling pathway, cytokine-cytokine receptor interaction, viral myocarditis, Leishmania infection, and asthma were found to be upregulated in patients with endometriosis. In GO network analysis, inflammation-related pathways, such as immune response, chemotaxis, and immune cell migration, and cell-cell interaction-related pathways, such as extracellular matrix, proteoglycan, and endocytosis, were commonly enriched for genes of three datasets with endometriosis (Figure 4C). Phagocytosis-related pathways were upregulated in endometriosis in two datasets, GSE23339 and GSE7305. The small portions of pathways related to platelets and coagulation were upregulated in endometriosis of the two gene sets, GSE11691 and GSE7305. GO pathways, including mitochondria, ribosomes, and RNA splicing and/or processing, were commonly enriched in three normal datasets. Finally, the GO pathways involved in the cell cycle and microtubules were downregulated in two datasets, GSE23339 and GSE7305.
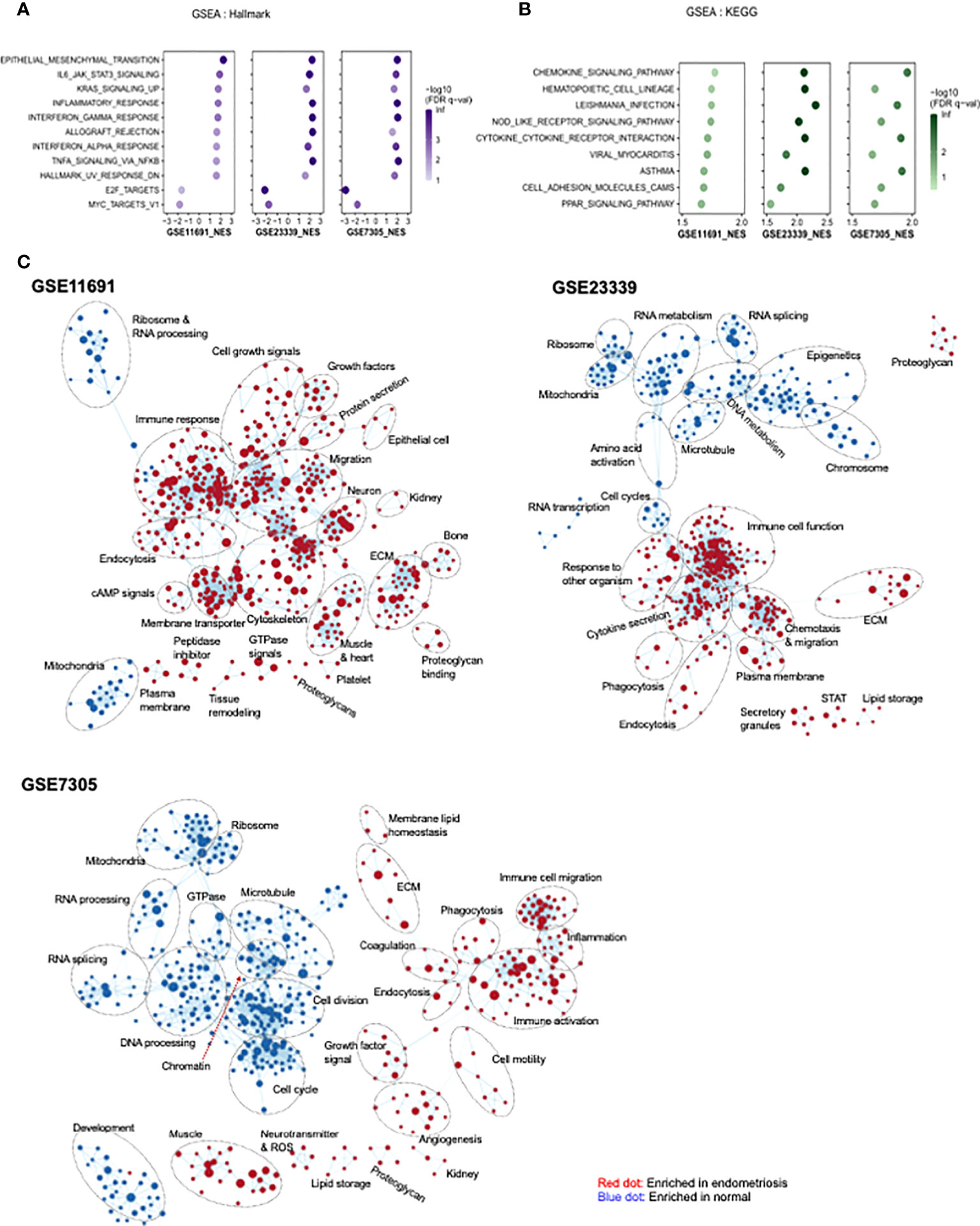
Figure 4 Identification of pathway networks by GSEA. (A, B) Common pathway categories identified by GSEA with gene set database of Hallmark (A) and KEGG (B) are presented. (C) GSEA was performed to obtain enriched GO-terms and visualized using Enrichment Map plugin of Cytoscape. The size of each node indicates the size of gene set. Red and blue indicate the node enriched in endometriosis and normal tissue, respectively.
To further evaluate the reliability of the results from integrative analysis of GSE11691, GSE23339, and GSE7305, the expression levels of 17 common DEGs in two external datasets, GSE135485 and GSE25628, were determined. The expression levels of 10 genes in GSE135485, including C7, CFH, CHL1, CLDN3, FZD7, IGJ, LY96, PDLIM3, PTGIS, and WISP2 were correlated with those in the validation sets (Supplementary Table 2). However, the expression levels of other genes did not correlate with those in the integrated data. In the GSE25628 dataset, expression levels of 11 genes, including AGR2, C7, CFH, FABP4, FZD7, GALNT4, LY96, PDLIM3, PPM1H, PTGIS, and WISP2 were in line with integrative analysis results (Supplementary Table 3). According to results from two external datasets, GSE135485 and GSE25628, the expression levels of seven genes, including C7, CFH, FZD7, LY96, PDLIM3, PTGIS, and WISP2 were correlated with training sets listed in Table 2.
The roles of C7, CFH, and FZD7 in endometriosis have been reported previously (8, 29). These studies revealed higher expression of C7, CFH, and FZD7 proteins in tissues with endometriosis compared to normal endometrium by IHC analysis. High expression of PDLIM3 in endometriosis has also been reported in several previous bioinformatics studies with little supportive experimental evidence (15, 30, 31). Thus, we conducted IHC and western blot analysis on tissues from normal endometrium and endometriosis foci to further evaluate the expression of proteins encoded by the identified genes, including LY96, PDLIM3, PTGIS, and WISP2. None of these genes have yet been evaluated in terms of their expression profiles in endometriosis tissue. Quantitative assessments of histological images clearly demonstrated higher expression levels of LY96, PDLIM3, PTGIS, and WISP2, proteins in tissues from patients with endometriosis, in line with integrative analysis results (Figure 5). According to western blot analysis, the protein levels of LY96, PDLIM3, and PTGIS were higher in the tissues of endometriosis patients. However, the expression of WISP2 was not correlated with the IHC analysis (Supplementary Figure 3).
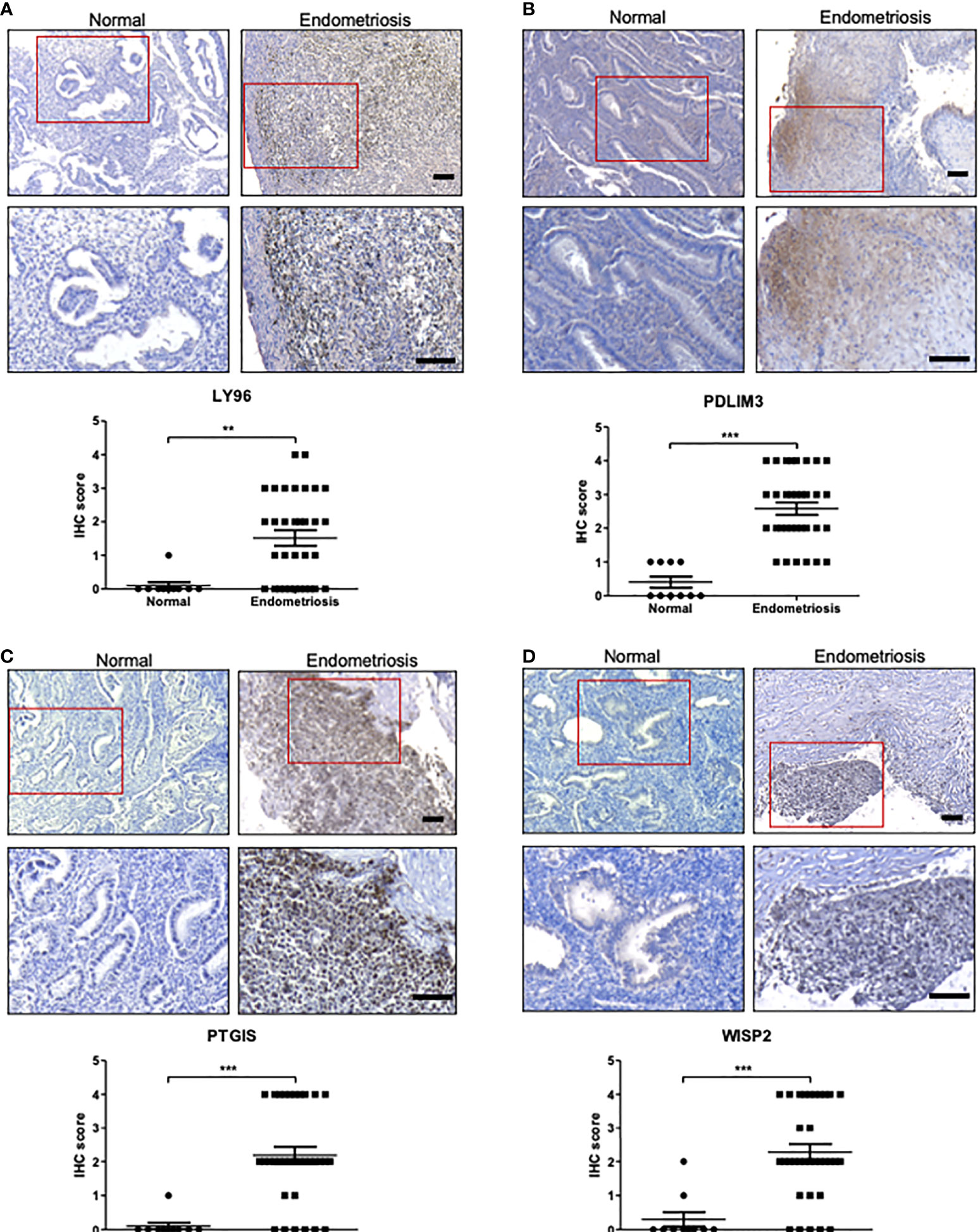
Figure 5 The protein expression of LY96, PDLIM3, PTGIS, and WISP2 in endometriosis tissue. (A–D) The protein expression of (A) LY96, (B) PDLIM3, (C) PTGIS, and (D) WISP2 was measured by IHC analysis of endometrial tissues from healthy volunteers or endometriosis patients. The representative IHC images are shown in upper panel (bar = 100 μm). The graphs in lower panel represent IHC score. **p < 0.01 and ***p < 0.001.
To further identify relevant drug-gene interactions and potential druggable target genes, we utilized the DGIdb by applying C7, CFH, FZD7, LY96, PDLIM3, and PTGIS as queries. Three genes, FZD7, LY96, and PTGIS, were found to be associated with drugs vantictumab, eritoran tetrasodium, and phenylbutazone, respectively (Table 3). CFH was predicted to be a target gene for three antibody drugs: bevacizumab, eculizumab, and ranibizumab. No potential gene-drug interactions were identified for C7 and PDLIM3.
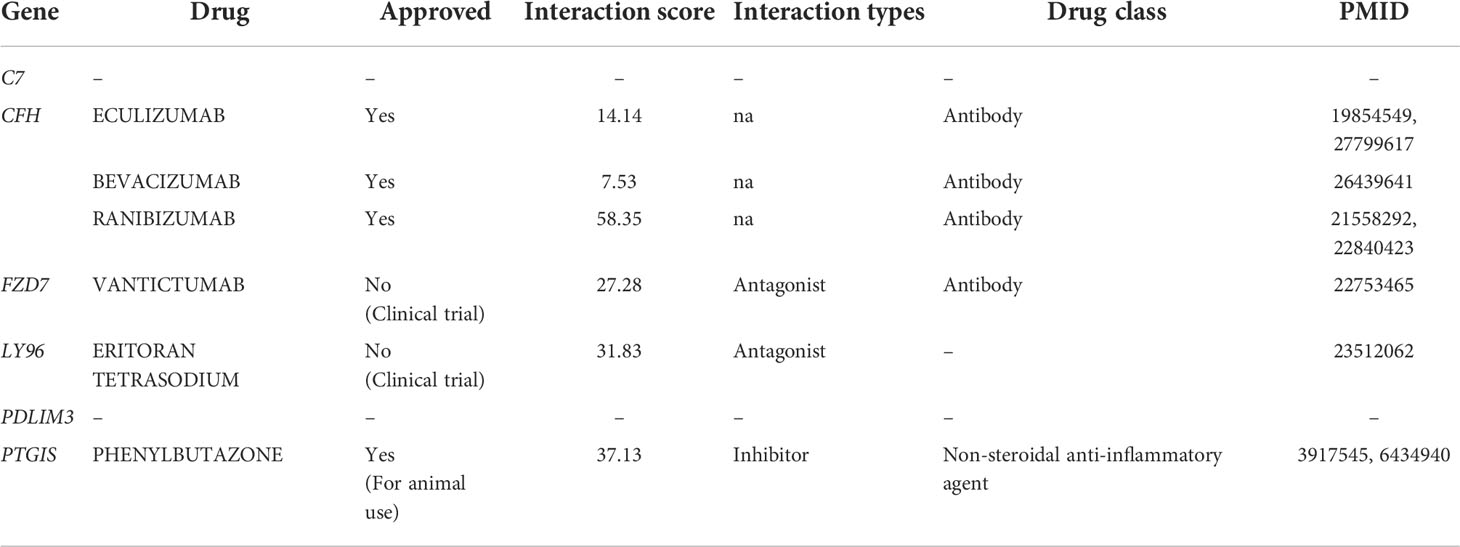
Table 3 Potent drugs identified in DGIdb corresponding to consistently upregulated six DEGs in endometriosis.
Endometriosis is a chronic inflammatory disease. The infiltration of immune cells and secretion of inflammatory mediators in the peritoneal microenvironment cause symptoms and signs observed in endometriosis patients (32). Endometriosis is characterized by inflammation and subsequent fibrosis, which eventually lead to pelvic discomfort, bowel and urinary problems, and infertility (32, 33). In contrast to normal endometrium, accumulated hemorrhage and tissue injury are key steps for initiating inflammation in the endometrial lesion and peritoneal cavity (33, 34). Hormonal alterations, particularly cyclic estrogen fluctuations, also contribute to an inflammatory imbalance in endometriosis (35). In contrast to other inflammatory diseases, increased estradiol production and estrogen receptor β (ERβ) expression in ectopic endometriosis lesions jointly activate nuclear factor-κB (NF-κB), a key inflammatory regulator (36, 37). Thus, the combination of oral contraceptives and non-steroidal anti-inflammatory drugs (NSAIDs) has been advised as the initial medical treatment option (10, 38). However, the clinical efficacy of this combination is suboptimal due to its low potency and side effects.
In this study, we identified six genes that may be utilized as diagnostic and/or therapeutic targets in endometriosis. STRING and GeneMANIA analyses revealed no evidence of any potent protein–protein interaction between these genes. Complement 7 (C7) and complement factor H (CFH), both of which are components of the complement system, have previously been identified as potent indicators of complement activation in endometriosis and endometriosis-associated ovarian cancer (29). C3, another member of the complement system, has been reported to be upregulated in endometriosis (39) and was also identified in our study as a common upregulated gene. Other members of the complement activation pathway, such as complement C3a receptor 1 (C3AR1) and V-set and immunoglobulin domain containing 4 (VSIG4), are also known in endometriosis, and upregulation of complement and coagulation pathways in endometriosis has been well-reported in several studies (15, 29, 39, 40). These studies suggested that autoimmune response in endometriosis is associated with the complement system (29, 39). In addition, the membrane attack complex in the complement system, which is composed of several complement proteins including C5b, C6, C7, C8, and C9, may also cause tissue damage and thereby induce inflammation in endometrial lesions.
Lymphocyte antigen 96 (LY96), also known as myeloid differentiation protein 2 (MD-2), has been reported its expression in the uterine endometrium (28, 41) and is a coreceptor of Toll-like receptor 4 (TLR4) (42). The TLR4 system is crucial for pathogen recognition and the activation of innate immunity. The complex of bacterial lipopolysaccharide (LPS) and LPS-binding protein interacts with cluster of differentiation 14 (CD14), a glycosylphosphatidylinositol-anchored membrane protein, and transfers LPS to LY96, consequently facilitating the dimerization of TLR4 (43). Damage-associated molecular patterns (DAMPs) secreted by most types of damaged tissues also bind to the TLR4 system and activate downstream pro-inflammatory signals similar to pathogen-associated molecular patterns, including bacterial LPS (44). Prostaglandin I2 synthase (PTGIS), also known as prostacyclin synthase (PGIS) or cytochrome P450 isomerase 8A1 (CYP8A1), is an enzyme that converts prostaglandin H2 to prostaglandin I2, and thereby modulates the inflammatory response (45). The role of PTGIS in inflammatory diseases is controversial, as it promotes progression of rheumatoid arthritis yet suppresses progression of pulmonary vascular disease and atherosclerosis (46). Although PTGIS was identified as a transcriptional target gene of NF-κB (47), the regulation of PTGIS mRNA expression does not exactly correlate to those of typical NF-κB targets (48, 49). In the vascular system and uterine endometrium, estrogen receptors work in concert with NF-κB to regulate the activity of prostaglandin-synthesizing enzymes, including COX-2 and PTGIS (50, 51). Furthermore, high levels of prostacyclin and its derivative, 6-keto-prostaglandin F1α, have been detected in the peritoneal fluid of endometriosis patients (52, 53). PTGIS expression was shown to be downregulated in a murine model of implanted endometrium (54); however, proteomics analyses demonstrated upregulated PTGIS expression in human ovarian endometrioma samples (55). In this study, we demonstrated the increased expression of LY96 and PTGIS via integrative transcriptome analysis, and further validated by IHC analysis of human-derived tissues.
Frizzled 7 (FZD7) is a member of the frizzled family and an atypical G protein-coupled receptor for Wnt proteins. FZD7 interacts with Dishevelled (Dvl) and lipoprotein receptor-related proteins (LRPs) in the presence of canonical Wnt signaling, and thereby promotes β-catenin signaling (56). This signaling pathway is closely related to embryonic development, cell proliferation, epithelial-to-mesenchymal transition, and carcinogenesis (57, 58). Wnt signaling is also involved in the production of enzymes related to prostaglandin metabolism in bone and skin, including COX-2 and PTGIS (59, 60). The crosstalk between the Wnt/frizzled and TLR4/NF-κB signaling pathways is well-established in chronic inflammation, development, and tumorigenesis (61, 62). PDZ and LIM domain 3 (PDLIM3) are involved in cytoskeleton assembly, in particular the formation of Z-disks in skeletal muscles (63). Although several microarray and proteome analyses have already revealed high PDLIM3 expression in this regard (15, 30, 31, 55), the exact role of PDLIM3 in endometriosis remains to be elucidated. The expression of Wnt1-inducible signaling pathway protein 2 (WISP2) might be another evidence of the connection between Wnt/frizzled and TLR4/NF-κB signaling pathway. The protein, also known as cellular communication network factor 5 (CCN5), is a secretory protein and a member of the connective tissue growth factor family (64). The expression of WISP2 is induced by Wnt signaling, and restricts cell growth, migration, adhesion, and differentiation, particularly in the vascular system and cancer cells (65–67). Secreted WISP2 is also involved in the activation of the canonical Wnt signaling pathway (68). However, our examination of WISP2 expression was not consistent between IHC and Western blot analysis. the delicate role of the WISP2 in the endometriosis still remains ambiguous and further extensive studies are required.
In endometriosis, chronic inflammation is co-regulated by ERβ-related signaling and the classical NF-κB signaling pathway (36, 37). Estrogen stimulation increases the expression of PDLIM3 in human prostate cancer (69). However, even if the expression level of PTGIS does not perfectly match that of the ERβ, it may be negatively influenced by ERβ (70, 71). We thus propose here the existence of signaling interactions between the validated DEGs including C7, CFH, FZD7, LY96, PDLIM3, and PTGIS, and pathways including TLR4/NF-κB, Wnt/frizzled, and estrogen receptors (Figure 6). Further research is needed to elucidate the precise roles of the identified genes in endometriosis.
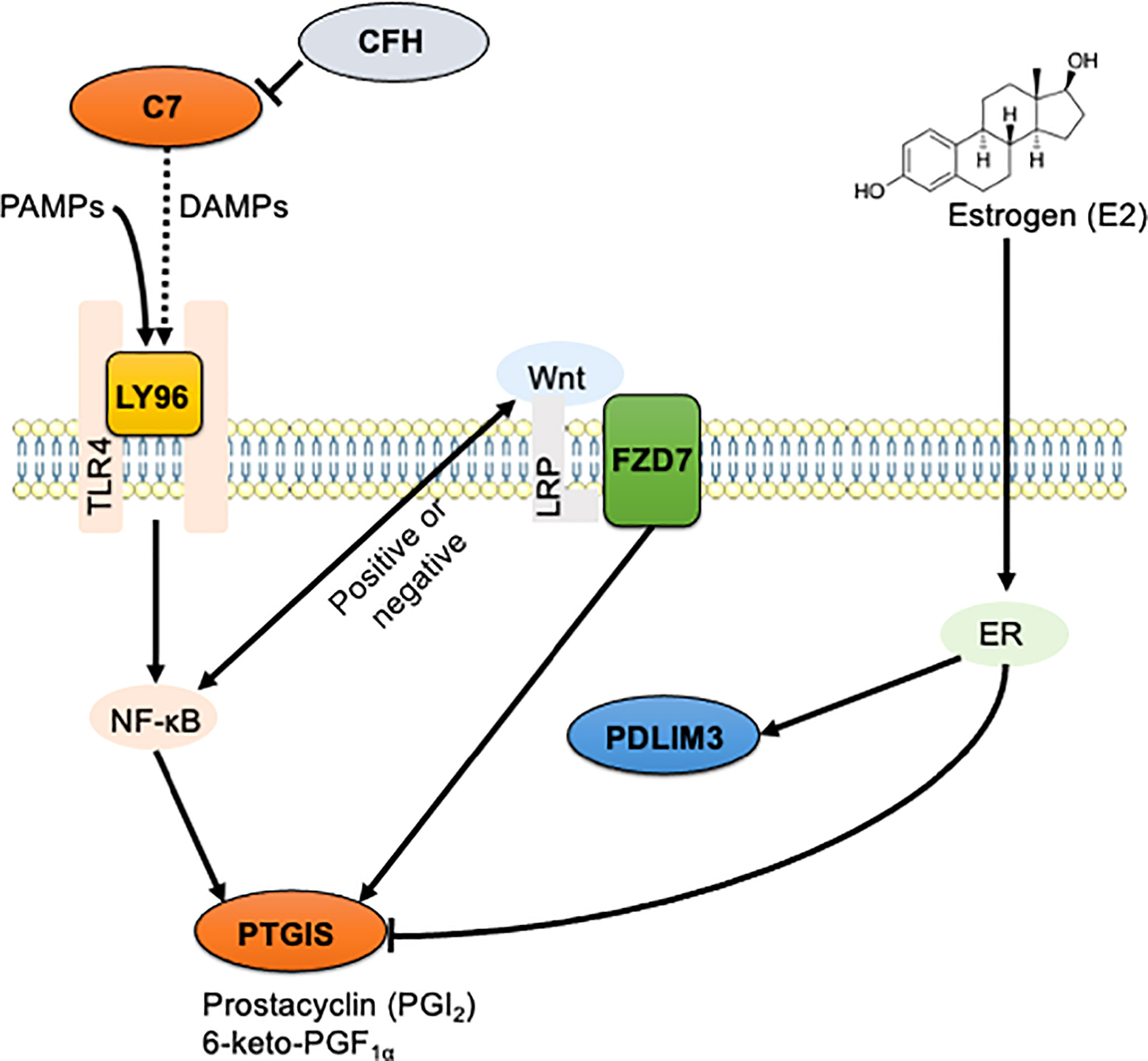
Figure 6 The schematic illustration of the hypothesized signaling network in endometriosis. Signaling interaction between six identified DEGs including C7, CFH, FZD7, LY96, PDLIM3, and PTGIS, and three enriched pathways including TLR4/NF-κB, Wnt/frizzled, and estrogen receptors were hypothesized and schematically illustrated. Solid line indicates direct interaction, and dashed line indicates indirect and/or proposed interaction.
We also found four druggable genes: FZD7, LY96, PTGIS, and CFH. FZD7 is a direct target of vantictumab, a neutralizing antibody currently being developed as an anticancer agent, particularly for triple-negative breast cancer (72, 73). In addition to vantictumab, an oligopeptide Fz7-21 and a small molecule SRI37892 have been evaluated as a new Fzd7-targeting agent to disrupt the Wnt signaling pathway for inhibiting intestinal stem cell function and cancer progression (74, 75). We suspect that these agents may have been identified for candidate drugs if registration in DGIdb. LY96 has been identified as a target of eritoran tetrasodium, a TLR4 antagonist drug that has been indicated for the treatment of sepsis in several clinical studies (76). However, eritoran tetrasodium was not successful due to its similar mortality compared to that of placebo (42). Instead of eritoran tetrasodium, we suggest that MD2-IN-1, isofraxidin, and L48H37, studied in the inflammation and cancer research field at the preclinical level, might be applied to treat endometriosis as LY96-targeting agents (77–79). Phenylbutazone is a non-steroidal anti-inflammatory drug (NSAID) that inhibits prostaglandin H synthases (PTGS1 and PTGS2) and PTGIS through peroxide-mediated deactivation (27). Although it has been approved for the treatment of backache and ankylosing spondylitis, phenylbutazone is currently withdrawn from human medicine, as it can cause severe adverse effects such as suppression of white blood cell production and aplastic anemia (80). For CFH-targeting drugs, DGIdb has suggested two VEGF-neutralizing antibodies, including ranibizumab and bevacizumab, and one C5-antagonizing antibody, eculizumab. As CFH is a non-specific off-target for these antibodies, its clinical application may result in significant adverse effects, particularly in patients with a mutated CFH gene (81–85). None of these drugs for targeting identified genes have yet been used to target the identified genes for endometriosis treatment. Additional in vivo and clinical studies are needed to determine the efficacy of these drugs in endometriosis treatment.
The datasets should ideally be comparable in terms of sample collection, underlying disease, menstrual cycle, and experiment types for the analysis of the collection of public gene sets. These elements of the datasets used for DEG analysis varied, particularly with regard to the menstrual cycle and underlying diseases such leiomyoma. We tried to select datasets using a filter of three objective criteria despite the restriction of only being able to access the datasets from public databases. In addition, we meticulously gathered normal control samples during the early follicular phase from healthy volunteers who were free of any gynecological diseases. However, there is almost no significant chance of finding genes that follow the menstrual cycle or underlying disease. The results clearly indicate that the genes discovered by DEG analysis may be important for conditions beyond than underlying disease and/or the menstrual cycle.
In conclusion, we identified 118 DEGs (79 upregulated and 39 downregulated) that may be involved in endometriosis pathogenesis. KEGG and GO functional analyses revealed enrichment of inflammation, complement activation, cell adhesion, and extracellular matrix pathways in endometriosis. Six genes, C7, CFH, FZD7, LY96, PDLIM3, and PTGIS were verified as upregulated DEGs by comparison to external gene sets, IHC and western blot analyses further confirmed the elevated protein expression levels of LY96, PDLIM3, and PTGIS in human endometrial lesions. We further analyzed the involvement of these genes in signaling pathways including TLR4/NF-κB, Wnt/frizzled, and estrogen receptors. We expect that subsequent studies will confirm the genes identified here as essential biomarkers for endometriosis diagnosis and treatment.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
The studies involving human participants were reviewed and approved by Institutional Review Board (IRB) of Pusan National University Hospital (2104-009-101). The patients/participants provided their written informed consent to participate in this study.
YJ analyzed informatics data. MC and J-SJ conducted IHC experiments. S-JB, J-YK, and JS analyzed the data. YK and DR validated and visualized the results from the informatics analysis. J-KP, HL, and JJ collected human samples and analyzed the clinical signatures and statistics. S-JB and YJ wrote the draft of this manuscript. S-JB, J-YK, and JS revised the manuscript. JJ and K-TH conceptualized this study and reviewed the manuscript. All authors read and approved the final manuscript.
This research was supported by a grant from Kosin University College of Medicine (2021) and by the National Research Foundation of Korea (NRF) grants funded by the Korean government (MIST: NRF-2020R1C1C1003703 to S-JB, NRF-2022R1I1A1A01063460 and NRF-2021R1A5A8029876 to YJ, NRF-2020R1F1A1067185 to JJ, and NRF-2021R1A4A1025662 to S-JB and K-TH).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2022.942368/full#supplementary-material
1. Taylor HS, Kotlyar AM, Flores VA. Endometriosis is a chronic systemic disease: Clinical challenges and novel innovations. Lancet (2021) 397(10276):839–52. doi: 10.1016/S0140-6736(21)00389-5
2. Parasar P, Ozcan P, Terry KL. Endometriosis: Epidemiology, diagnosis and clinical management. Curr Obstet Gynecol Rep (2017) 6(1):34–41. doi: 10.1007/s13669-017-0187-1
3. Kapoor R, Stratopoulou CA, Dolmans MM. Pathogenesis of endometriosis: New insights into prospective therapies. Int J Mol Sci (2021) 22(21):11700. doi: 10.3390/ijms222111700
4. Augoulea A, Alexandrou A, Creatsa M, Vrachnis N, Lambrinoudaki I. Pathogenesis of endometriosis: The role of genetics, inflammation and oxidative stress. Arch Gynecol Obstet (2012) 286(1):99–103. doi: 10.1007/s00404-012-2357-8
5. Taylor HS, Kotlyar AM, Flores VA. Endometriosis is a chronic systemic disease: Clinical challenge and novel innovations. Lancet (2021) 397(10276):839–52. doi: 10.1016/S0140-6736(21)00389-5
6. Ahn SH, Singh V, Tayade C. Biomarkers in endometriosis: Challenges and opportunities. Fertil Steril (2017) 107(3):523–32. doi: 10.1016/j.fertnstert.2017.01.009
7. Anastasiu CV, Moga MA, Elena Neculau A, Balan A, Scarneciu I, Dragomir RM, et al. Biomarkers for the noninvasive diagnosis of endometriosis: State of the art and future perspectives. Int J Mol Sci (2020) 21(5):1750. doi: 10.3390/ijms21051750
8. MDH T, Fassbender A, F O D, Vanhie A. Endometriosis biomarkers: Will codevelopment in academia-industry partnerships result in new and robust noninvasive diagnostic tests? Biol Reprod (2019) 101(6):1140–5. doi: 10.1093/biolre/ioz016
9. Tian Z, Chang XH, Zhao Y, Zhu HL. Current biomarkers for the detection of endometriosis. Chin Med J (Engl) (2020) 133(19):2346–52. doi: 10.1097/CM9.0000000000001063
10. Burghaus S, Schafer SD, Beckmann MW, Brandes I, Brunahl C, Chvatal R, et al. Diagnosis and treatment of endometriosis. guideline of the dggg, sggg and oeggg (S2k level, awmf registry number 015/045, august 2020). Geburtshilfe Frauenheilkd (2021) 81(4):422–46. doi: 10.1055/a-1380-3693
11. Goulielmos GN, Matalliotakis M, Matalliotaki C, Eliopoulos E, Matalliotakis I, Zervou MI. Endometriosis research in the -omics era. Gene (2020) 741:144545. doi: 10.1016/j.gene.2020.144545
12. Sun YV, Hu YJ. Integrative analysis of multi-omics data for discovery and functional studies of complex human diseases. Adv Genet (2016) 93:147–90. doi: 10.1016/bs.adgen.2015.11.004
13. Cui D, Liu Y, Ma J, Lin K, Xu K, Lin J. Identification of key genes and pathways in endometriosis by integrated expression profiles analysis. PeerJ (2020) 8:e10171. doi: 10.7717/peerj.10171
14. Lu ZZ, Gao Y. Screening differentially expressed genes between endometriosis and ovarian cancer to find new biomarkers for endometriosis. Ann Med (2021) 53(1):1377–89. doi: 10.1080/07853890.2021.1966087
15. Chen M, Zhou Y, Xu H, Hill C, Ewing RM, He D, et al. Bioinformatic analysis reveals the importance of epithelial-mesenchymal transition in the development of endometriosis. Sci Rep (2020) 10(1):8442. doi: 10.1038/s41598-020-65606-9
16. Dai FF, Bao AY, Luo B, Zeng ZH, Pu XL, Wang YQ, et al. Identification of differentially expressed genes and signaling pathways involved in endometriosis by integrated bioinformatics analysis. Exp Ther Med (2020) 19(1):264–72. doi: 10.3892/etm.2019.8214
17. Hull ML, Escareno CR, Godsland JM, Doig JR, Johnson CM, Phillips SC, et al. Endometrial-peritoneal interactions during endometriotic lesion establishment. Am J Pathol (2008) 173(3):700–15. doi: 10.2353/ajpath.2008.071128
18. Hawkins SM, Creighton CJ, Han DY, Zariff A, Anderson ML, Gunaratne PH, et al. Functional microrna involved in endometriosis. Mol Endocrinol (2011) 25(5):821–32. doi: 10.1210/me.2010-0371
19. Hever A, Roth RB, Hevezi P, Marin ME, Acosta JA, Acosta H, et al. Human endometriosis is associated with plasma cells and overexpression of b lymphocyte stimulator. Proc Natl Acad Sci U.S.A. (2007) 104(30):12451–6. doi: 10.1073/pnas.0703451104
20. Crispi S, Piccolo MT, D'Avino A, Donizetti A, Viceconte R, Spyrou M, et al. Transcriptional profiling of endometriosis tissues identifies genes related to organogenesis defects. J Cell Physiol (2013) 228(9):1927–34. doi: 10.1002/jcp.24358
21. Davis S, Meltzer PS. Geoquery: A bridge between the gene expression omnibus (Geo) and bioconductor. Bioinformatics (2007) 23(14):1846–7. doi: 10.1093/bioinformatics/btm254
22. Benjamini Y, Hochberg Y. Controlling the false discovery rate - a practical and powerful approach to multiple testing. J R Stat Soc B (1995) 57(1):289–300. doi: 10.1111/j.2517-6161.1995.tb02031.x
23. Kim J, Lee H, Jin EJ, Jo Y, Kang BE, Ryu D, et al. A microfluidic device to fabricate one-step cell bead-laden hydrogel struts for tissue engineering. Small (2022) 18(1):e2106487. doi: 10.1002/smll.202106487
24. Hwu YM, Wu FS, Li SH, Sun FJ, Lin MH, Lee RK. The impact of endometrioma and laparoscopic cystectomy on serum anti-mullerian hormone levels. Reprod Biol Endocrinol (2011) 9:80. doi: 10.1186/1477-7827-9-80
25. Schweppe KW, Ring D. Peritoneal defects and the development of endometriosis in relation to the timing of endoscopic surgery during the menstrual cycle. Fertil Steril (2002) 78(4):763–6. doi: 10.1016/s0015-0282(02)03431-3
26. Griffith M, Griffith OL, Coffman AC, Weible JV, McMichael JF, Spies NC, et al. Dgidb: Mining the druggable genome. Nat Methods (2013) 10(12):1209–10. doi: 10.1038/nmeth.2689
27. Reed GA, Griffin IO, Eling TE. Inactivation of prostaglandin h synthase and prostacyclin synthase by phenylbutazone. requirement for peroxidative metabolism. Mol Pharmacol (1985) 27(1):109–14.
28. Sheldon IM, Cronin J, Goetze L, Donofrio G, Schuberth HJ. Defining postpartum uterine disease and the mechanisms of infection and immunity in the female reproductive tract in cattle. Biol Reprod (2009) 81(6):1025–32. doi: 10.1095/biolreprod.109.077370
29. Suryawanshi S, Huang X, Elishaev E, Budiu RA, Zhang L, Kim S, et al. Complement pathway is frequently altered in endometriosis and endometriosis-associated ovarian cancer. Clin Cancer Res (2014) 20(23):6163–74. doi: 10.1158/1078-0432.Ccr-14-1338
30. Kobayashi H, Yamashita Y, Iwase A, Yoshikawa Y, Yasui H, Kawai Y, et al. The ferroimmunomodulatory role of ectopic endometriotic stromal cells in ovarian endometriosis. Fertil Steril (2012) 98(2):415–22.e1-12. doi: 10.1016/j.fertnstert.2012.04.047
31. Logan PC, Yango P, Tran ND. Endometrial stromal and epithelial cells exhibit unique aberrant molecular defects in patients with endometriosis. Reprod Sci (2018) 25(1):140–59. doi: 10.1177/1933719117704905
32. Lin YH, Chen YH, Chang HY, Au HK, Tzeng CR, Huang YH. Chronic niche inflammation in endometriosis-associated infertility: Current understanding and future therapeutic strategies. Int J Mol Sci (2018) 19(8):2385. doi: 10.3390/ijms19082385
33. Wang Y, Nicholes K, Shih IM. The origin and pathogenesis of endometriosis. Annu Rev Pathol (2020) 15:71–95. doi: 10.1146/annurev-pathmechdis-012419-032654
34. Lu J, Wang Z, Cao J, Chen Y, Dong Y. A novel and compact review on the role of oxidative stress in female reproduction. Reprod Biol Endocrinol (2018) 16(1):80. doi: 10.1186/s12958-018-0391-5
35. Garcia-Gomez E, Vazquez-Martinez ER, Reyes-Mayoral C, Cruz-Orozco OP, Camacho-Arroyo I, Cerbon M. Regulation of inflammation pathways and inflammasome by sex steroid hormones in endometriosis. Front Endocrinol (Lausanne) (2019) 10:935. doi: 10.3389/fendo.2019.00935
36. Gou Y, Li X, Li P, Zhang H, Xu T, Wang H, et al. Estrogen receptor beta upregulates Ccl2 Via nf-kappab signaling in endometriotic stromal cells and recruits macrophages to promote the pathogenesis of endometriosis. Hum Reprod (2019) 34(4):646–58. doi: 10.1093/humrep/dez019
37. Han SJ, Lee JE, Cho YJ, Park MJ, O'Malley BW. Genomic function of estrogen receptor beta in endometriosis. Endocrinology (2019) 160(11):2495–516. doi: 10.1210/en.2019-00442
38. Koninckx PR, Fernandes R, Ussia A, Schindler L, Wattiez A, Al-Suwaidi S, et al. Pathogenesis based diagnosis and treatment of endometriosis. Front Endocrinol (Lausanne) (2021) 12:745548. doi: 10.3389/fendo.2021.745548
39. Rekker K, Saare M, Eriste E, Tasa T, Kukuskina V, Roost AM, et al. High-throughput mrna sequencing of stromal cells from endometriomas and endometrium. Reproduction (2017) 154(1):93–100. doi: 10.1530/REP-17-0092
40. Jiang L, Zhang MM, Wang SX, Han YY, Fang XL. Common and specific gene signatures among three different endometriosis subtypes. Peerj (2020) 8:e8730. doi: 10.7717/peerj.8730
41. Hirata T, Osuga Y, Hirota Y, Koga K, Yoshino O, Harada M, et al. Evidence for the presence of toll-like receptor 4 system in the human endometrium. J Clin Endocrinol Metab (2005) 90(1):548–56. doi: 10.1210/jc.2004-0241
42. Opal SM, Laterre PF, Francois B, LaRosa SP, Angus DC, Mira JP, et al. Effect of eritoran, an antagonist of Md2-Tlr4, on mortality in patients with severe sepsis: The access randomized trial. JAMA (2013) 309(11):1154–62. doi: 10.1001/jama.2013.2194
43. Lu YC, Yeh WC, Ohashi PS. Lps/Tlr4 signal transduction pathway. Cytokine (2008) 42(2):145–51. doi: 10.1016/j.cyto.2008.01.006
44. Lee KM. Seong SY. partial role of Tlr4 as a receptor responding to damage-associated molecular pattern. Immunol Lett (2009) 125(1):31–9. doi: 10.1016/j.imlet.2009.05.006
45. Wang B, Wu L, Chen J, Dong L, Chen C, Wen Z, et al. Metabolism pathways of arachidonic acids: Mechanisms and potential therapeutic targets. Signal Transduct Target Ther (2021) 6(1):94. doi: 10.1038/s41392-020-00443-w
46. Stitham J, Midgett C, Martin KA, Hwa J. Prostacyclin: An inflammatory paradox. Front Pharmacol (2011) 2:24. doi: 10.3389/fphar.2011.00024
47. Pahl HL. Activators and target genes of Rel/Nf-kappab transcription factors. Oncogene (1999) 18(49):6853–66. doi: 10.1038/sj.onc.1203239
48. Zhao H, Hiroi T, Hansen BS, Rade JJ. Cyclic stretch induces cyclooxygenase-2 gene expression in vascular endothelial cells Via activation of nuclear factor kappa-beta. Biochem Biophys Res Commun (2009) 389(4):599–601. doi: 10.1016/j.bbrc.2009.09.028
49. Gurgul-Convey E, Lenzen S. Protection against cytokine toxicity through endoplasmic reticulum and mitochondrial stress prevention by prostacyclin synthase overexpression in insulin-producing cells. J Biol Chem (2010) 285(15):11121–8. doi: 10.1074/jbc.M109.054775
50. Shah BH. Estrogen stimulation of cox-2-Derived Pgi2 confers atheroprotection. Trends Endocrinol Metab (2005) 16(5):199–201. doi: 10.1016/j.tem.2005.05.008
51. King AE, Collins F, Klonisch T, Sallenave JM, Critchley HO, Saunders PT. An additive interaction between the nfkappab and estrogen receptor signalling pathways in human endometrial epithelial cells. Hum Reprod (2010) 25(2):510–8. doi: 10.1093/humrep/dep421
52. Drake TS, O'Brien WF, Ramwell PW, Metz SA. Peritoneal fluid thromboxane B2 and 6-Keto-Prostaglandin F1 alpha in endometriosis. Am J Obstet Gynecol (1981) 140(4):401–4. doi: 10.1016/0002-9378(81)90033-8
53. Ylikorkala O, Makila UM. Prostacyclin and thromboxane in gynecology and obstetrics. Am J Obstet Gynecol (1985) 152(3):318–29. doi: 10.1016/s0002-9378(85)80221-0
54. Kusakabe KT, Abe H, Kondo T, Kato K, Okada T, Otsuki Y. DNA Microarray analysis in a mouse model for endometriosis and validation of candidate factors with human adenomyosis. J Reprod Immunol (2010) 85(2):149–60. doi: 10.1016/j.jri.2010.02.008
55. Vehmas AP, Muth-Pawlak D, Huhtinen K, Saloniemi-Heinonen T, Jaakkola K, Laajala TD, et al. Ovarian endometriosis signatures established through discovery and directed mass spectrometry analysis. J Proteome Res (2014) 13(11):4983–94. doi: 10.1021/pr500384n
56. Yang L, Wu X, Wang Y, Zhang K, Wu J, Yuan YC, et al. Fzd7 has a critical role in cell proliferation in triple negative breast cancer. Oncogene (2011) 30(43):4437–46. doi: 10.1038/onc.2011.145
57. Yang Y. Wnt signaling in development and disease. Cell Biosci (2012) 2(1):14. doi: 10.1186/2045-3701-2-14
58. Sompel K, Elango A, Smith AJ, Tennis MA. Cancer chemoprevention through frizzled receptors and emt. Discovery Oncol (2021) 12(1):32. doi: 10.1007/s12672-021-00429-2
59. Robinson JA, Chatterjee-Kishore M, Yaworsky PJ, Cullen DM, Zhao W, Li C, et al. Wnt/Beta-catenin signaling is a normal physiological response to mechanical loading in bone. J Biol Chem (2006) 281(42):31720–8. doi: 10.1074/jbc.M602308200
60. Lombardi A, Arseni L, Carriero R, Compe E, Botta E, Ferri D, et al. Reduced levels of prostaglandin I2 synthase: A distinctive feature of the cancer-free trichothiodystrophy. Proc Natl Acad Sci USA (2021) 118(26):e2024502118. doi: 10.1073/pnas.2024502118
61. Du Q, Geller DA. Cross-regulation between wnt and nf-kappab signaling pathways. For Immunopathol Dis Therap (2010) 1(3):155–81. doi: 10.1615/ForumImmunDisTher.v1.i3
62. Ma B, Hottiger MO. Crosstalk between Wnt/Beta-catenin and nf-kappab signaling pathway during inflammation. Front Immunol (2016) 7:378. doi: 10.3389/fimmu.2016.00378
63. Pashmforoush M, Pomies P, Peterson KL, Kubalak S, Ross J Jr., Hefti A, et al. Adult mice deficient in actinin-associated lim-domain protein reveal a developmental pathway for right ventricular cardiomyopathy. Nat Med (2001) 7(5):591–7. doi: 10.1038/87920
64. Sabbah M, Prunier C, Ferrand N, Megalophonos V, Lambein K, De Wever O, et al. Ccn5, a novel transcriptional repressor of the transforming growth factor beta signaling pathway. Mol Cell Biol (2011) 31(7):1459–69. doi: 10.1128/MCB.01316-10
65. Russo JW, Castellot JJ. Ccn5: Biology and pathophysiology. J Cell Commun Signal (2010) 4(3):119–30. doi: 10.1007/s12079-010-0098-7
66. Yoon PO, Lee MA, Cha H, Jeong MH, Kim J, Jang SP, et al. The opposing effects of Ccn2 and Ccn5 on the development of cardiac hypertrophy and fibrosis. J Mol Cell Cardiol (2010) 49(2):294–303. doi: 10.1016/j.yjmcc.2010.04.010
67. Fritah A, Saucier C, De Wever O, Bracke M, Bieche I, Lidereau R, et al. Role of wisp-2/Ccn5 in the maintenance of a differentiated and noninvasive phenotype in human breast cancer cells. Mol Cell Biol (2008) 28(3):1114–23. doi: 10.1128/MCB.01335-07
68. Grunberg JR, Hammarstedt A, Hedjazifar S, Smith U. The novel secreted adipokine Wnt1-inducible signaling pathway protein 2 (Wisp2) is a mesenchymal cell activator of canonical wnt. J Biol Chem (2014) 289(10):6899–907. doi: 10.1074/jbc.M113.511964
69. Chakravarty D, Sboner A, Nair SS, Giannopoulou E, Li R, Hennig S, et al. The oestrogen receptor alpha-regulated lncrna Neat1 is a critical modulator of prostate cancer. Nat Commun (2014) 5:5383. doi: 10.1038/ncomms6383
70. Rebordao MR, Galvao A, Pinto-Bravo P, Pinheiro J, Gamboa S, Silva E, et al. Endometrial prostaglandin synthases, ovarian steroids, and oxytocin receptors in mares with oxytocin-induced luteal maintenance. Theriogenology (2017) 87:193–204. doi: 10.1016/j.theriogenology.2016.08.028
71. Ospina JA, Duckles SP, Krause DN. 17beta-estradiol decreases vascular tone in cerebral arteries by shifting cox-dependent vasoconstriction to vasodilation. Am J Physiol Heart Circ Physiol (2003) 285(1):H241–50. doi: 10.1152/ajpheart.00018.2003
72. Diamond JR, Becerra C, Richards D, Mita A, Osborne C, O'Shaughnessy J, et al. Phase ib clinical trial of the anti-frizzled antibody vantictumab (Omp-18r5) plus paclitaxel in patients with locally advanced or metastatic Her2-negative breast cancer. Breast Cancer Res Treat (2020) 184(1):53–62. doi: 10.1007/s10549-020-05817-w
73. Gurney A, Axelrod F, Bond CJ, Cain J, Chartier C, Donigan L, et al. Wnt pathway inhibition Via the targeting of frizzled receptors results in decreased growth and tumorigenicity of human tumors. Proc Natl Acad Sci U.S.A. (2012) 109(29):11717–22. doi: 10.1073/pnas.1120068109
74. Nile AH, de Sousa EMF, Mukund S, Piskol R, Hansen S, Zhou L, et al. A selective peptide inhibitor of frizzled 7 receptors disrupts intestinal stem cells. Nat Chem Biol (2018) 14(6):582–90. doi: 10.1038/s41589-018-0035-2
75. Zhang W, Lu W, Ananthan S, Suto MJ, Li Y. Discovery of novel frizzled-7 inhibitors by targeting the receptor's transmembrane domain. Oncotarget (2017) 8(53):91459–70. doi: 10.18632/oncotarget.20665
76. Barochia A, Solomon S, Cui X, Natanson C, Eichacker PQ. Eritoran tetrasodium (E5564) treatment for sepsis: Review of preclinical and clinical studies. Expert Opin Drug Metab Toxicol (2011) 7(4):479–94. doi: 10.1517/17425255.2011.558190
77. Jin J, Yu X, Hu Z, Tang S, Zhong X, Xu J, et al. Isofraxidin targets the Tlr4/Md-2 axis to prevent osteoarthritis development. Food Funct (2018) 9(11):5641–52. doi: 10.1039/c8fo01445k
78. Wang Y, Shan X, Dai Y, Jiang L, Chen G, Zhang Y, et al. Curcumin analog L48h37 prevents lipopolysaccharide-induced Tlr4 signaling pathway activation and sepsis Via targeting Md2. J Pharmacol Exp Ther (2015) 353(3):539–50. doi: 10.1124/jpet.115.222570
79. Zhang Y, Wu J, Ying S, Chen G, Wu B, Xu T, et al. Discovery of new Md2 inhibitor from chalcone derivatives with anti-inflammatory effects in lps-induced acute lung injury. Sci Rep (2016) 6:25130. doi: 10.1038/srep25130
80. Worboys M, Toon E. Phenylbutazone (Bute, pbz, epz): One drug across two species. Hist Philos Life Sci (2018) 40(2):27. doi: 10.1007/s40656-018-0191-4
81. Medina FM, Alves Lopes da Motta A, Takahashi WY, Carricondo PC, Dos Santos Motta MM, Melo MB, et al. Pharmacogenetic effect of complement factor h gene polymorphism in response to the initial intravitreal injection of bevacizumab for wet age-related macular degeneration. Ophthalmic Res (2015) 54(4):169–74. doi: 10.1159/000439172
82. Smailhodzic D, Muether PS, Chen J, Kwestro A, Zhang AY, Omar A, et al. Cumulative effect of risk alleles in cfh, Arms2, and vegfa on the response to ranibizumab treatment in age-related macular degeneration. Ophthalmology (2012) 119(11):2304–11. doi: 10.1016/j.ophtha.2012.05.040
83. McKibbin M, Ali M, Bansal S, Baxter PD, West K, Williams G, et al. Cfh, vegf and Htra1 promoter genotype may influence the response to intravitreal ranibizumab therapy for neovascular age-related macular degeneration. Br J Ophthalmol (2012) 96(2):208–12. doi: 10.1136/bjo.2010.193680
84. Fakhouri F, Fila M, Provot F, Delmas Y, Barbet C, Chatelet V, et al. Pathogenic variants in complement genes and risk of atypical hemolytic uremic syndrome relapse after eculizumab discontinuation. Clin J Am Soc Nephrol (2017) 12(1):50–9. doi: 10.2215/CJN.06440616
85. Davin JC, Gracchi V, Bouts A, Groothoff J, Strain L, Goodship T. Maintenance of kidney function following treatment with eculizumab and discontinuation of plasma exchange after a third kidney transplant for atypical hemolytic uremic syndrome associated with a cfh mutation. Am J Kidney Dis (2010) 55(4):708–11. doi: 10.1053/j.ajkd.2009.08.011
Keywords: endometriosis, LY96, PDLIM3, PTGIS, TLR4/NF-κB, Wnt/frizzled, estrogen receptor
Citation: Bae S-J, Jo Y, Cho MK, Jin J-S, Kim J-Y, Shim J, Kim YH, Park J-K, Ryu D, Lee HJ, Joo J and Ha K-T (2022) Identification and analysis of novel endometriosis biomarkers via integrative bioinformatics. Front. Endocrinol. 13:942368. doi: 10.3389/fendo.2022.942368
Received: 12 May 2022; Accepted: 07 October 2022;
Published: 20 October 2022.
Edited by:
Rajakumar Anbazhagan, Eunice Kennedy Shriver National Institute of Child Health and Human Development (NIH), United StatesReviewed by:
Asamanja Chattoraj, Kazi Nazrul University, IndiaCopyright © 2022 Bae, Jo, Cho, Jin, Kim, Shim, Kim, Park, Ryu, Lee, Joo and Ha. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jongkil Joo, amtqb29AcHVzYW4uYWMua3I=; Ki-Tae Ha, aGFnaXNAcHVzYW4uYWMua3I=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.