
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
OPINION article
Front. Endocrinol. , 22 July 2020
Sec. Thyroid Endocrinology
Volume 11 - 2020 | https://doi.org/10.3389/fendo.2020.00470
Protsiv et al. used three sets of data to demonstrate that human core body temperature had decreased by 0.03°C per decade since the industrial revolution in the US (1). They proposed that a 1.6% temperature drop over a period of almost 200 birth years had occurred. Anthropometrics, gender, or race were excluded as potential factors. The authors postulated that the principle contributor to this reduction was reduced inflammation, reflecting better, healthier environments and improved hygiene measures (1). Although hygiene has increased and hence reduced death from infectious disease, other factors in our environment have also changed significantly. Here we propose another plausible and potentially testable mechanism, that of the contribution of factors interfering with thyroid hormone (TH) metabolism.
TH is an essential physiological cue that acts at central and peripheral levels to affect internal temperature in endotherms (2). Humans strive to live at thermoneutral conditions, in which peripheral muscle metabolism generates sufficient heat as a by-product to maintain temperature without the need for additional heat-generating mechanisms (3). For us, the resting metabolic rate (RMR) is thus a crude proxy for core body temperature. TH directly affects the RMR by altering mitochondrial biogenesis and oxidative phosphorylation via TRα1, the principle TH receptor isoform in muscle (Figure 1) (4). TH fluctuations within the normal range alter the RMR in humans (5, 6), suggesting that subtle changes in TH homeostasis could have consequences for body temperature. Recent data indicate that TH also safeguards core body temperature at the central level. Peripheral sensors relay temperature information to hypothalamic nuclei where temperature-sensitive neurons further calibrate body temperature to a pre-fixed set point (7, 8). Mice in a thermoneutral environment that were given high doses of TH either systemically, or via direct hypothalamic injection, had an acutely elevated central temperature set point (9–11). These central TH effects are most-likely an example of pyrexia, or a controlled set point change, as opposed to uncontrolled hyperthermia (9). In mice lacking TRα1, the central thermostat was downregulated (12, 13) in addition to reduced metabolism and impaired heat dissipation (14). Of note, in small mammals like mice, TH also activates brown adipose tissue to generate additional heat through adaptive thermogenesis (15), a process that is possibly less relevant in healthy adult humans, though this point is under continual debate (16).
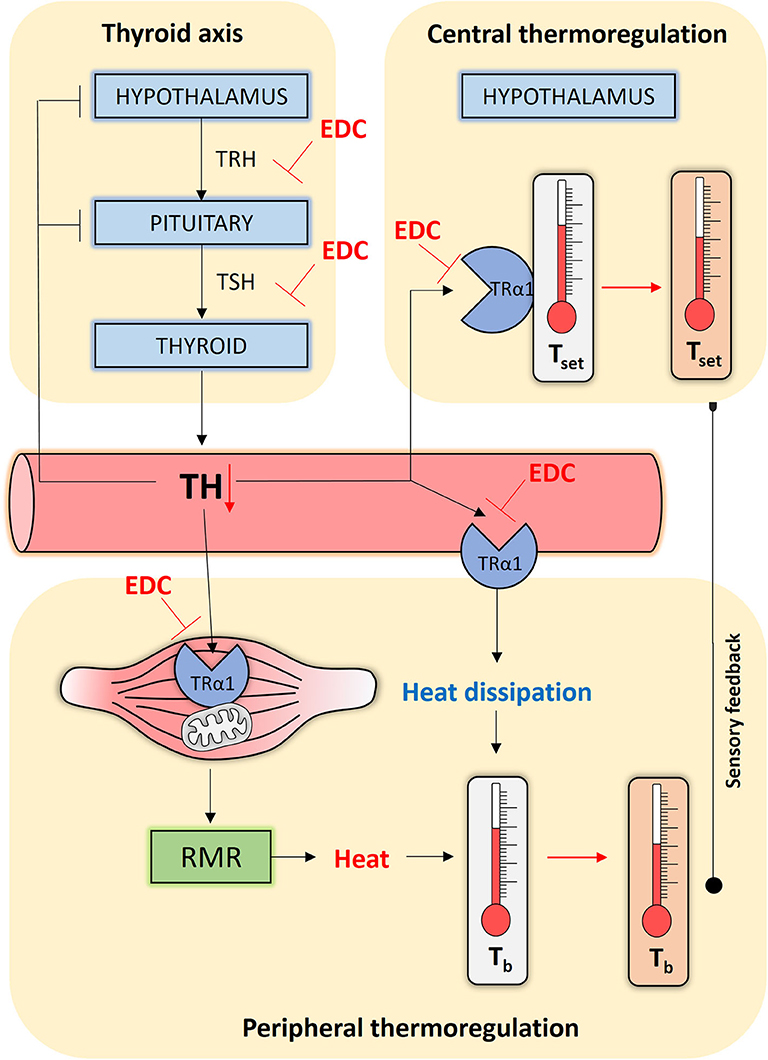
Figure 1. Simplified scheme of how endocrine disrupting chemicals (EDCs) might interfere with human thermoregulation. The Hypothalamic-Pituitary-Thyroid (HPT) axis, or thyroid axis, consists of neurons synthesizing thyrotropin-releasing hormone (TRH) that stimulates thyrotropes in the pituitary to release thyrotropin (TSH). TSH stimulates the thyroid gland to produce and secrete the thyroid hormones (THs), which negatively feedback at the level of the HPT axis. THs reach central and peripheral organs via the circulation. At the level of the hypothalamus, TH programs and safeguards the temperature set point (Tset). At the level of the muscle tissue, TH alters the resting metabolic rate (RMR) by acting on oxidative phosphorylation and mitochondrial biogenesis, generating heat in the process that maintains core body temperature (Tb). TH may additionally affect heat dissipation by regulating vasoconstriction. All the actions listed above, other than the thyroid axis set point, occur primarily through TRα1. EDCs can interfere at every point in this interconnected network and disrupt TH homeostasis or interfere with the activation of TRα1, causing impaired thermoregulation control through persistent temperature set point changes, a reduced RMR and altered heat dissipation.
But how do these actions affect core body temperature in humans? Back in 1894, a medical doctor T.C. Railton observed the significant impact TH can have on body temperature (17). His patient was a 14-year-old hypothyroid boy with an abnormally low body temperature of 35°C. After giving 36 grains of sheep's thyroid gland (thyroxin or T4 was not isolated until 1914 by Edward Kendall), his temperature rose to a staggering 39.1°C. Adjusting the dose to 5 grains twice a day normalized his body temperature to approximately 37°C (17). This one-person trial proved that the amount of thyroid tissue, and thus THs, dose-dependently alters body temperature. Similarly, the doctor treating the first-ever myxedema patient by hypodermic injection of thyroid extract, noted that her body temperature increased by several degrees (18). More recently, intravenous injection of high doses of levothyroxine reversed the low body temperatures (34–35.8°C) of seven comatose myxedema patients to more than 36°C (19).
Another intriguing case that demonstrates these temperature effects of TH is Wilson's Temperature Syndrome, first described in 1990. Patients display a form of chronic fatigue syndrome with symptoms that are hallmarks of hypothyroidism, as well as a lower-than-normal body temperature. Giving cyclic oral supra-physiological doses of T3 (up to 30 μg/day) elevated the average body temperature from 36.34 to 37°C in 11 tested patients (20). While the temperature fluctuations in Railton's case were in the range of several degrees, Friedman's data show that smaller doses of THs can induce body temperature changes around 0.5°C. It would be very interesting to know whether subclinical hypothyroidism, a condition marked by lower but within the normal range levels of TH that affects around 10% of the population (21), also alters average body temperature in an even narrower regimen. So far, only one cross-sectional study has been reported on the subject (22). This study included 306 subclinically hypothyroid subjects, however, the authors observed no body temperature differences with euthyroid individuals. Three potential caveats need to be mentioned here, first that temperature measurements only included one digit after the decimal point, whereas in the study carried out by Protsiv et al. (1) the authors extended their findings to two digits. The second is the small number of subclinical hypothyroid patients in the cohort tested and third, to be classed as subclinically hypothyroid, T4 levels must lie within the normal range, whereas TSH levels are elevated. As the cut-off range for elevated TSH varies according to region, but can easily be a 10 fold span, this emphasizes the difficulty of classing someone as subclinically hypothyroid.
If a similarly deregulated TH homeostasis lies at the base of the gradual temperature decrease during industrialization, what could have disrupted TH metabolism in the first place? A number of factors have changed in our environment since the industrial revolution. Chemical production has increased 300-fold since 1970, both in terms of diversity and quantity (UNEP, 2013—Global Chemicals Outlook - Toward Sound Management of Chemicals). These so-called endocrine disrupting chemicals (EDCs) affect each and every one of us in a gender-, race-, and anthropometric-independent manner (23). Apart from the range of effects on our daily body physiology, especially that of future generations, we hypothesize that the long-term use of chemical compounds might even have changed our own body's homeostatic mechanisms, including the RMR.
A myriad of studies have now unequivocally proven that this constant exposure to low doses of chemical mixtures can deregulate the thyroid axis, and alter human body homeostasis (24). The most evident adverse effect comprises impaired neurodevelopment, and human epidemiological data provide two lines of worrying evidence in that regard (25). First, deviating maternal TH levels, but within the normal range during fetal development lead to irreversible, structural changes in the central nervous system of infants and unfavorable outcomes such as lowered intelligence quotient (25, 26). Second, some of the investigated widely-used chemicals induce changes in TH levels (27) of the magnitude seen in the study of Korevaar et al. (26).
We speculate that chemically disrupted TH signaling interferes with the development and functioning of central thermoregulation (Figure 1). A population of parvalbuminergic neurons in the anterior hypothalamic area integrate temperature information to control heart rate and blood pressure (28). As abnormal maternal TH levels disrupt neuronal migration and differentiation, resulting in permanent hypertension and temperature-dependent tachycardia (29) it is possible that developmental disruption of maternal/fetal TH signaling by EDCs could adversely affect the fine-tuning of the temperature set point in a similar way. A clear-cut example is the persistent organochlorine pesticide dichlorodiphenyltrichloroethane (DDT) that is still found in significant levels in breast milk and amniotic fluid. Average concentrations of 3.49 ng DDT per g lipid, and 198.34 ng/g of its main metabolite DDE, were detected in pregnant women of a 2003–2004 cohort in the NANHES study (30). DDT is a very potent thyroid disruptor in animals (31, 32) and humans (33). Female mice exposed to 1.7 mg DDT per kg body weight during a two-week perinatal period, had a permanently lower core body temperature up until the age of 5 months (34). When combined with a high-fat diet, average body temperature in the adult mice dropped even more, causing many other complications such as insulin resistance and altered glucose/lipid metabolism (34). In obese people too, body temperature is lower than average (35). These comorbidities are associated with metabolic syndrome, a global health hazard that is mainly considered a non-communicable disease primarily caused by a sedentary lifestyle and calorie-rich diet, but also endocrine disruption. Such data suggest developmental misprogramming due to chemical exposure could be another trigger predisposing to metabolic syndrome (36).
Another possibility is that EDCs affect adult thermoregulation by interfering with TH-dependent central and peripheral thermoregulation in the adult (Figure 1) (37). Key to our reasoning is the fact that we are constantly exposed to low doses of complex EDC mixtures (38). On the one hand, EDCs can indirectly affect thermoregulation by disrupting thyroid axis-regulated TH homeostasis. Polychlorinated biphenyls, polybrominated diphenyl ethers, perchlorate, bisphenols, phthalates, pesticides, and perfluoroalkyls all cause persistent changes in circulating TH levels in animals and humans (39, 40). For example, the individuals of a cohort of 679 male pesticide applicators that were continuously exposed to the highest doses of the insecticide aldrin or the herbicide pendimethalin had higher TSH and lower T4 levels (41). These factors could thus alter the RMR or alter hypothalamic TH signaling (37). On the other hand, EDCs can interfere with the availability of THs to activate TRs (Figure 1). Adult mice shortly exposed to 100 μg/kg bisphenol A, an antagonist of TH action (42), displayed impaired muscle metabolism and lower body temperatures (43). Chemicals can also deregulate central control of metabolism and the energy balance (37), but whether they do so by disrupting local TH signaling remains to be investigated. In addition, EDCs also affect other hormone axes, as for instance glucocorticoids that are intimately implicated in metabolism and heat generation (44).
Data linking (developmental) chemical exposure, disrupted TH signaling and permanently reduced core body temperature are scarce, and need additional testing. A well-planned experiment should aim to causally link this chain of events to provide better proof for this paradigm. We recommend exposing mice to a well-known TH disrupting chemical (or mixture of chemicals) during perinatal development and follow core body temperature during postnatal life up until the adult stage. Simultaneously exposing mice to T3 during specific intervals could reveal critical time windows for establishment of the hypothalamic temperature set-point. Data should be coupled to mapping the neuroanatomical and (epi)genetic landscape in the (developing) hypothalamic nuclei. In addition, adult-onset exposure of adult wild-type and TRα1-deficient mice might reveal at which levels a particular EDC is most liable to induce disruption of thermoregulation. Such actions could be at the peripheral level by interfering with the RMR in muscle tissue, through impairing heat dissipation, or at the central level by altering the temperature set point, or possibly a combination of the above. To mimic effects on human thermoregulation best, experiments should be performed at thermoneutral conditions (45). Testing for dose-dependent effects on temperature will be subtle and requires a high “n” number per group. In the meantime, collecting data from large cohorts of patients with thyroid conditions, for instance of children born to (sub)clinically hypothyroxinemic mothers, could unravel similar correlations with offspring body temperature. To strengthen our hypothesis further, we should ideally have data from pre-industrialization or from emerging countries that have not yet fully industrialized to uncover possible trends. However, to the authors' knowledge no such data is available.
Our arguments remain merely speculative, but suggest yet another plausible mode of action of how EDCs can interfere with whole body homeostasis. While we are beginning to understand the complex mechanisms by which industrial chemicals endanger human health and wildlife preservation, we can only surmise the consequences of long-term exposure. With the advent of global crises such as climate change or the recent COVID-19 pandemic, it is however imperative to fully grasp the dynamic interaction between these factors so as to protect future generations from these threats that include chemical pollution.
BD worked out the concept of the paper. PV and BD both wrote the paper. All authors contributed to the article and approved the submitted version.
PV was funded by the Fondation pour la Recherche Médicale (FRM) (SPF201909009111) and has a basic science grant from the European thyroid association. This work also received support from the Centre National de la Recherche Scientifique (CNRS), the EU H2020 contract Thyrage (Grant n°666869), and the EU Horizon 2020 programme ATHENA (Grant n°825161).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
1. Protsiv M, Ley C, Lankester J, Hastie T, Parsonnet J. Decreasing human body temperature in the United States since the industrial revolution. Elife. (2020) 9:e49555. doi: 10.7554/eLife.49555
2. Mittag J. More than fever - novel concepts in the regulation of body temperature by thyroid hormones. Exp Clin Endocrinol Diabetes. (2019) 128:428–31. doi: 10.1055/a-1014-2510
3. Salvatore D, Simonides WS, Dentice M, Zavacki AM, Larsen PR. Thyroid hormones and skeletal muscle—new insights and potential implications. Nat Rev Endocrinol. (2014) 10:206–214. doi: 10.1038/nrendo.2013.238
4. Silva JE. Thermogenic mechanisms and their hormonal regulation. Physiol Rev. (2006) 86:435–64. doi: 10.1152/physrev.00009.2005
5. Al-Adsani H, Hoffer LJ, Silva JE. Resting energy expenditure is sensitive to small dose changes in patients on chronic thyroid hormone replacement 1. J Clin Endocrinol Metab. (1997) 82:1118–25. doi: 10.1210/jc.82.4.1118
6. Samuels MH, Kolobova I, Antosik M, Niederhausen M, Purnell JQ, Schuff KG. Thyroid function variation in the normal range, energy expenditure, and body composition in L-T4–Treated Subjects. J Clin Endocrinol Metab. (2017) 102:2533–42. doi: 10.1210/jc.2017-00224
7. Reitman ML. Of mice and men - environmental temperature, body temperature, and treatment of obesity. FEBS Lett. (2018) 592:2098–107. doi: 10.1002/1873-3468.13070
8. Zhao Z-DD, Yang WZ, Gao C, Fu X, Zhang W, Zhou Q, et al. A hypothalamic circuit that controls body temperature. Proc Natl Acad Sci USA. (2017) 114:2042–7. doi: 10.1073/pnas.1616255114
9. Dittner C, Lindsund E, Cannon B, Nedergaard J. At thermoneutrality, acute thyroxine-induced thermogenesis and pyrexia are independent of UCP1. Mol Metab. (2019) 25:20–34. doi: 10.1016/j.molmet.2019.05.005
10. Johann K, Cremer AL, Fischer AW, Heine M, Pensado ER, Resch J, et al. Thyroid-Hormone-Induced Browning of white adipose tissue does not contribute to thermogenesis and glucose consumption. Cell Rep. (2019) 27:3385–400.e3. doi: 10.1016/j.celrep.2019.05.054
11. Alvarez-Crespo M, Csikasz RI, Martínez-Sánchez N, Diéguez C, Cannon B, Nedergaard J, et al. Essential role of UCP1 modulating the central effects of thyroid hormones on energy balance. Mol Metab. (2016) 5:271–82. doi: 10.1016/j.molmet.2016.01.008
12. Marrif H, Schifman A, Stepanyan Z, Gillis M-A, Calderone A, Weiss RE, et al. Temperature homeostasis in transgenic mice lacking thyroid hormone receptor-α gene products. Endocrinology. (2005) 146:2872–84. doi: 10.1210/en.2004-1544
13. Wikstrom L. Abnormal heart rate and body temperature in mice lacking thyroid hormone receptor alpha 1. EMBO J. (1998) 17:455–61. doi: 10.1093/emboj/17.2.455
14. Warner A, Rahman A, Solsjo P, Gottschling K, Davis B, Vennstrom B, et al. Inappropriate heat dissipation ignites brown fat thermogenesis in mice with a mutant thyroid hormone receptor 1. Proc Natl Acad Sci USA. (2013) 110:16241–6. doi: 10.1073/pnas.1310300110
15. Bianco AC, Maia AL, da Silva WS, Christoffolete MA. Adaptive activation of thyroid hormone and energy expenditure. Biosci Rep. (2005) 25:191–208. doi: 10.1007/s10540-005-2885-6
16. Koskensalo K, Raiko J, Saari T, Saunavaara V, Eskola O, Nuutila P, et al. Human brown adipose tissue temperature and fat fraction are related to its metabolic activity. J Clin Endocrinol Metab. (2017) 102:1200–7. doi: 10.1210/jc.2016-3086
17. Railton TC. Sporadic cretinism treated by administration of the thyroid gland. BMJ. (1894) 1:1180–1. doi: 10.1136/bmj.1.1744.1180
18. Murray GR. Note on the treatment of myx dema by hypodermic injections of an extract of the thyroid gland of a sheep. BMJ. (1891) 2:796–7. doi: 10.1136/bmj.2.1606.796
19. Arlot S, Debussche X, Lalau JD, Mesmacque A, Tolani M, Quichaud J, et al. Myxoedema coma: response of thyroid hormones with oral and intravenous high-dose L-thyroxine treatment. Intensive Care Med. (1991) 17:16–8. doi: 10.1007/BF01708403
20. Friedman M, Miranda-Massari JR, Gonzalez MJ. Supraphysiological cyclic dosing of sustained release T3 in order to reset low basal body temperature. Health Sci J. (2006) 25:23–9.
21. Biondi B, Cappola AR, Cooper DS. Subclinical hypothyroidism. JAMA. (2019) 322:153. doi: 10.1001/jama.2019.9052
22. Ashizawa K, Imaizumi M, Usa T, Tominaga T, Sera N, Hida A, et al. Metabolic cardiovascular disease risk factors and their clustering in subclinical hypothyroidism. Clin Endocrinol. (2010) 72:689–95. doi: 10.1111/j.1365-2265.2009.03697.x
23. Demeneix B, Slama R. Endocrine Disruptors: From Scientific Evidence to Human Health Protection. Policy Department for Citizens' Rights and Constitutional Affairs. Directorate General for Internal Policies of the Union. PE 608.866, European Union (2019). Available online at: https://www.europarl.europa.eu/RegData/etudes/STUD/2019/608866/IPOL_STU(2019)608866_EN.pdf (accessed July 8, 2020).
24. Boas M, Feldt-Rasmussen U, Main KM. Thyroid effects of endocrine disrupting chemicals. Mol Cell Endocrinol. (2012) 355:240–8. doi: 10.1016/j.mce.2011.09.005
25. Bennett D, Bellinger DC, Birnbaum LS, Bradman A, Chen A, Cory-Slechta DA, et al. Project TENDR: targeting environmental neuro-developmental risks the TENDR consensus statement. Environ Health Perspect. (2016) 124:A118–22. doi: 10.1289/EHP358
26. Korevaar TIMM, Muetzel R, Medici M, Chaker L, Jaddoe VWV V, de Rijke YB, et al. Association of maternal thyroid function during early pregnancy with offspring IQ and brain morphology in childhood: a population-based prospective cohort study. Lancet Diabetes Endocrinol. (2016) 4:35–43. doi: 10.1016/S2213-8587(15)00327-7
27. Derakhshan A, Shu H, Peeters RP, Kortenkamp A, Lindh CH, Demeneix B, et al. Association of urinary bisphenols and triclosan with thyroid function during early pregnancy. Environ Int. (2019) 133:105123. doi: 10.1016/j.envint.2019.105123
28. Mittag J, Lyons DJ, Sällström J, Vujovic M, Dudazy-Gralla S, Warner A, et al. Thyroid hormone is required for hypothalamic neurons regulating cardiovascular functions. J Clin Invest. (2013) 123:509–16. doi: 10.1172/JCI65252
29. Harder L, Dudazy-Gralla S, Müller-Fielitz H, Hjerling Leffler J, Vennström B, Heuer H, et al. Maternal thyroid hormone is required for parvalbumin neurone development in the anterior hypothalamic area. J Neuroendocrinol. (2018) 30:e12573. doi: 10.1111/jne.12573
30. Woodruff TJ, Zota AR, Schwartz JM. Environmental chemicals in pregnant women in the United States: NHANES 2003–2004. Environ Health Perspect. (2011) 119:878–85. doi: 10.1289/ehp.1002727
31. Fini J-B, Mughal BB, Le Mével S, Leemans M, Lettmann M, Spirhanzlova P, et al. Human amniotic fluid contaminants alter thyroid hormone signalling and early brain development in xenopus embryos. Sci Rep. (2017) 7:43786. doi: 10.1038/srep43786
32. Tebourbi O, Hallègue D, Yacoubi MT, Sakly M, Rhouma KB. Subacute toxicity of p,p′-DDT on rat thyroid: Hormonal and histopathological changes. Environ Toxicol Pharmacol. (2010) 29:271–9. doi: 10.1016/j.etap.2010.03.002
33. Álvarez-Pedrerol M, Ribas-Fitó N, Torrent M, Carrizo D, Garcia-Esteban R, Grimalt JO, et al. Thyroid disruption at birth due to prenatal exposure to β-hexachlorocyclohexane. Environ Int. (2008) 34:737–40. doi: 10.1016/j.envint.2007.12.001
34. La Merrill M, Karey E, Moshier E, Lindtner C, La Frano MR, Newman JW, et al. Perinatal exposure of mice to the pesticide ddt impairs energy expenditure and metabolism in adult female offspring. PLoS ONE. (2014) 9:e103337. doi: 10.1371/journal.pone.0103337
35. Grimaldi D, Provini F, Pierangeli G, Mazzella N, Zamboni G, Marchesini G, et al. Evidence of a diurnal thermogenic handicap in obesity. Chronobiol Int. (2015) 32:299–302. doi: 10.3109/07420528.2014.983603
36. De Long NE, Holloway AC. Early-life chemical exposures and risk of metabolic syndrome. Diabetes Metab Syndr Obes. (2017) 10:101–9. doi: 10.2147/DMSO.S95296
37. Nadal A, Quesada I, Tudurí E, Nogueiras R, Alonso-Magdalena P. Endocrine-disrupting chemicals and the regulation of energy balance. Nat Rev Endocrinol. (2017) 13:536–46. doi: 10.1038/nrendo.2017.51
38. Kortenkamp A, Faust M. Regulate to reduce chemical mixture risk. Science. (2018) 361:224–6. doi: 10.1126/science.aat9219
39. Calsolaro V, Pasqualetti G, Niccolai F, Caraccio N, Monzani F. Thyroid disrupting chemicals. Int J Mol Sci. (2017) 18:2583. doi: 10.3390/ijms18122583
40. Leemans M, Couderq S, Demeneix B, Fini J-B. Pesticides with potential thyroid hormone-disrupting effects: a review of recent data. Front Endocrinol. (2019) 10:743. doi: 10.3389/fendo.2019.00743
41. Lerro CC, Beane Freeman LE, DellaValle CT, Kibriya MG, Aschebrook-Kilfoy B, Jasmine F, et al. Occupational pesticide exposure and subclinical hypothyroidism among male pesticide applicators. Occup Environ Med. (2018) 75:79–89. doi: 10.1136/oemed-2017-104431
42. Moriyama K, Tagami T, Akamizu T, Usui T, Saijo M, Kanamoto N, et al. Thyroid hormone action is disrupted by bisphenol a as an antagonist. J Clin Endocrinol Metab. (2002) 87:5185–90. doi: 10.1210/jc.2002-020209
43. Batista TM, Alonso-Magdalena P, Vieira E, Amaral MEC, Cederroth CR, Nef S, et al. Short-Term treatment with bisphenol-a leads to metabolic abnormalities in adult male mice. PLoS ONE. (2012) 7:e33814. doi: 10.1371/journal.pone.0033814
44. Scotney H, Symonds ME, Law J, Budge H, Sharkey D, Manolopoulos KN. Glucocorticoids modulate human brown adipose tissue thermogenesis in vivo. Metabolism. (2017) 70:125–32. doi: 10.1016/j.metabol.2017.01.024
Keywords: body temperature, thyroid hormone, thyroid hormone metabolism, thermoregulation, endocrine disrupting chemicals, industrialization, homeostasis
Citation: Vancamp P and Demeneix BA (2020) Is the Observed Decrease in Body Temperature During Industrialization Due to Thyroid Hormone-Dependent Thermoregulation Disruption? Front. Endocrinol. 11:470. doi: 10.3389/fendo.2020.00470
Received: 07 May 2020; Accepted: 15 June 2020;
Published: 22 July 2020.
Edited by:
Frédéric Flamant, Université de Lyon, FranceReviewed by:
Jens Mittag, University of Lübeck, GermanyCopyright © 2020 Vancamp and Demeneix. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Pieter Vancamp, cGlldGVyLnZhbmNhbXBAbW5obi5mcg==; Barbara A. Demeneix, YmFyYmFyYS5kZW1lbmVpeEBtbmhuLmZy
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.