- 1Department of Psychiatry, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, China
- 2Department of Clinical Research Center, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, China
- 3Department of Cardiology, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, China
Objectives: Rapid eye movement (REM) sleep is closely related to all-cause mortality. The aim of this study is to explore the role of REM sleep on the incident heart failure (HF).
Methods: We selected 4490 participants (2480 women and 2010 men; mean age, 63.2 ± 11.0 years) from the Sleep Heart Health Study. HF was identified as the first occurrence during a mean follow-up period of 10.9 years. REM sleep including percentage of REM sleep and total REM sleep time were monitored using in-home polysomnography at baseline. Multivariable Cox regression analysis was utilized to explore the relationship between REM sleep and HF.
Results: In total, 436 (9.7%) cases of HF were observed during the entire follow-up period. After adjusting for potential covariates, an increased percentage of REM sleep (per 5%) was independently associated with a reduced incidence of HF [hazard ratio (HR) 0.88, 95% confidence interval (CI) 0.82–0.94, P < 0.001]. A similar result was also found between total REM sleep time (increased per 5 min) and incident HF (HR 0.97, 95% CI 0.95–0.99, P < 0.001). Moreover, the fourth quartile of both percentage of REM sleep (HR 0.65, 95% CI 0.48–0.88, P = 0.005) and total REM sleep time (HR 0.64, 95% CI 0.45–0.90, P = 0.010) had lower risk of incident HF when compared with the first quartile.
Conclusion: An increased percentage of REM sleep and total REM sleep time were associated with a reduced risk of HF. REM sleep may be a predictor of the incident HF.
Clinical Trial Registration: [ClinicalTrials.gov], identifier [NCT00005275].
Introduction
Sleep is an essential physiological phenomenon occurring in alternation with wakefulness, and good quality sleep is vital for maintaining health and homeostasis (1, 2). According to the World Health Organization, 27% of individuals worldwide suffer from sleep disorders. With the increasing pressure of work, study, and life, sleep issues have severely affected nearly 50–70 million people’s physical and mental health in the United States (3). There is growing evidence that poor sleep contributes to cardiovascular disease (CVD) and its risk factors, such as coronary artery disease, stroke, diabetes mellitus, hypertension, and obesity (4–8). However, the relationship between sleep and CVD deserves further investigation.
Rapid eye movement (REM) sleep and non-REM sleep are two critical stages of sleep. REM sleep, about 20–25% of total sleep time, is usually characterized by eye movement, increased heart rate and blood pressure, muscle relaxation, and low voltage and fast frequency in the electroencephalogram (9, 10). Recently, several studies have shown that individuals with a decreased percentage of REM sleep have high all-cause mortality (11, 12). Matthews et al. demonstrated that there was a close correlation between the percentage of REM sleep and sleep/wake ratios of blood pressure (13).
Despite the certainty of knowledge on the effects of REM sleep, the relationship between REM sleep and heart failure (HF) remains unclear. The purpose of this study was to explore whether there is an association between REM sleep (including percentage REM sleep and total REM sleep time) and incident HF based on the Sleep Heart Health Study (SHHS) datasets.
Materials and Methods
Study Population
Data were derived based on an existing dataset from the SHHS (ClinicalTrials.gov, identifier NCT00005275), which was a community-based multicenter prospective cohort study performed by the National Heart, Lung, and Blood Institute to investigate the cardiovascular and other consequences of sleep-disordered breathing between baseline (November 1, 1995 and January 31, 1998) and 2011. The detailed method and design of this study have been reported previously (14, 15). Briefly, a total of 6441 participants aged 40 years and older were recruited from several “parent” cohorts including the Atherosclerosis Risk in Communities study, the Cardiovascular Health Study, the Framingham Offspring and Omni study, the Strong Heart Study, the Health and Environment and Tucson Epidemiologic Study, and studies of hypertension in New York. All participants in the SHHS signed written consent, and the study protocol was approved by the institutional review board of each participating institution to collect sleep and questionnaire data. The data underlying this article are available in National Sleep Research Resource, at https://doi.org/10.25822/ghy8-ks59. The datasets were derived from sources in the public domain. Our access to the SHHS database was provided after acquiring a signed agreement with the Brigham and Women’s Hospital. The SHHS shared dataset from the National Sleep Research Resource does not include 637 individuals from the Strong Heart Study due to sovereignty issues. Exclusion criteria for our study were (1) previous CVD outcomes (n = 535); (2) participants who use CPAP or a mouthpiece (n = 7); and (3) no follow-up data (n = 772). Finally, 4490 participants were included in the present study (Supplementary Figure 1).
Sleep Parameters
Polysomnography (PSG) is essential for the diagnosis and management of many sleep parameters. All individuals in the current study underwent electroencephalography-based overnight unattended PSG (P-Series, Compumedics, Abbotsville, VIC, Australia) at home (15). Supplementary Material showed the details regarding the specific technical aspects of the PSG measurement. The time and percentage of REM sleep were captured using PSG monitoring. In addition, the percentage of REM sleep was categorized into quartiles: Q1 (<15.8%; n = 1113), Q2 (15.8–20.1%; n = 1134), Q3 (20.2–24.0%; n = 1145), and Q4 (>24.0%; n = 1098). Moreover, total REM sleep time was also divided into quartiles: Q1 (<54.0 min; n = 1113), Q2 (54.0–73.5 min; n = 1162), Q3 (73.6–91.5 min; n = 1110), and Q4 (>91.5 min; n = 1105). Other sleep structure parameters, including time in stage 1 (min), time in stage 2 (min), time in stage 3 (min), percentage stage 1, percentage stage 2, and percentage stage 3, were also included in the current analysis. Sleep duration was defined as the total time in bed (Time from lights off to lights on, rounded to nearest minute) based on the PSG record. The apnea–hypopnea index (AHI) obtained from the PSG was identified as the number of apneas or hypopneas recorded during the study per hour of sleep, accompanied by at least a 4% drop in oxygen saturation (15).
The Identification of Heart Failure
The criteria for HF was based on clinical signs and symptoms (such as rales, edema, dyspnea, and orthopnea), physiologic tests (decreased systolic function), and supportive findings (chest radiography or functional cardiac imaging), as previous study reported (16). HF was defined as the first occurrence during the average 10.9 years’ follow-up. The control group for HF patients was defined as participants who had no CVD outcomes at baseline and did not develop HF during the follow-up period when investigated the association between REM sleep and HF. Myocardial infarction (MI), stroke, and CVD death were also identified in the follow-up time. All the CVD events were evaluated in the parent cohorts using explicit protocols and identified for SHHS using follow-up interviews, written annual questionnaires, telephone contacts with study participants or next-of-kin, surveillance of local hospital records and community obituaries, and linkage with the Social Security Administration Death Master File (17).
Covariates
Covariates including age, sex, race, education, marital status, smoking status, body mass index (BMI), alcohol use, caffeine use, benzodiazepine use, hypertension, diabetes mellitus, triglyceride, cholesterol and high-density lipoprotein (HDL) cholesterol, percent time below oxygen desaturation 90% (T90), and AHI were obtained from the SHHS baseline examination.
Statistical Analyses
Continuous and categorical variables were summarized using Student’s t-test and Chi-squared tests, respectively. The results are presented as mean (±SD) and number (percentages). Moreover, unadjusted Kaplan–Meier survival curves were drawn to investigate the overall survival of individuals with different REM sleep quartiles. Cox regression analysis was used to calculate the hazard ratios (HRs) and 95% confidence intervals (CIs) in the relationship between REM sleep and HF. Multivariable Cox regression analysis adjusted for age, sex, race, education, marital status, smoking status, BMI, alcohol use, caffeine use, benzodiazepine use, hypertension, diabetes mellitus, triglyceride, cholesterol, HDL, sleep duration, T90, and AHI to examine the associations between REM sleep and HF. Subgroup analysis stratified by sex (men vs. women) and AHI (≥15 vs. <15 events/h) was performed when explore the role of REM sleep on the incidence of HF. All statistical analyses were conducted using SPSS (version 24.0; SPSS Inc.). A two-sided P < 0.05 was considered to be statistically significant.
Results
Study Population
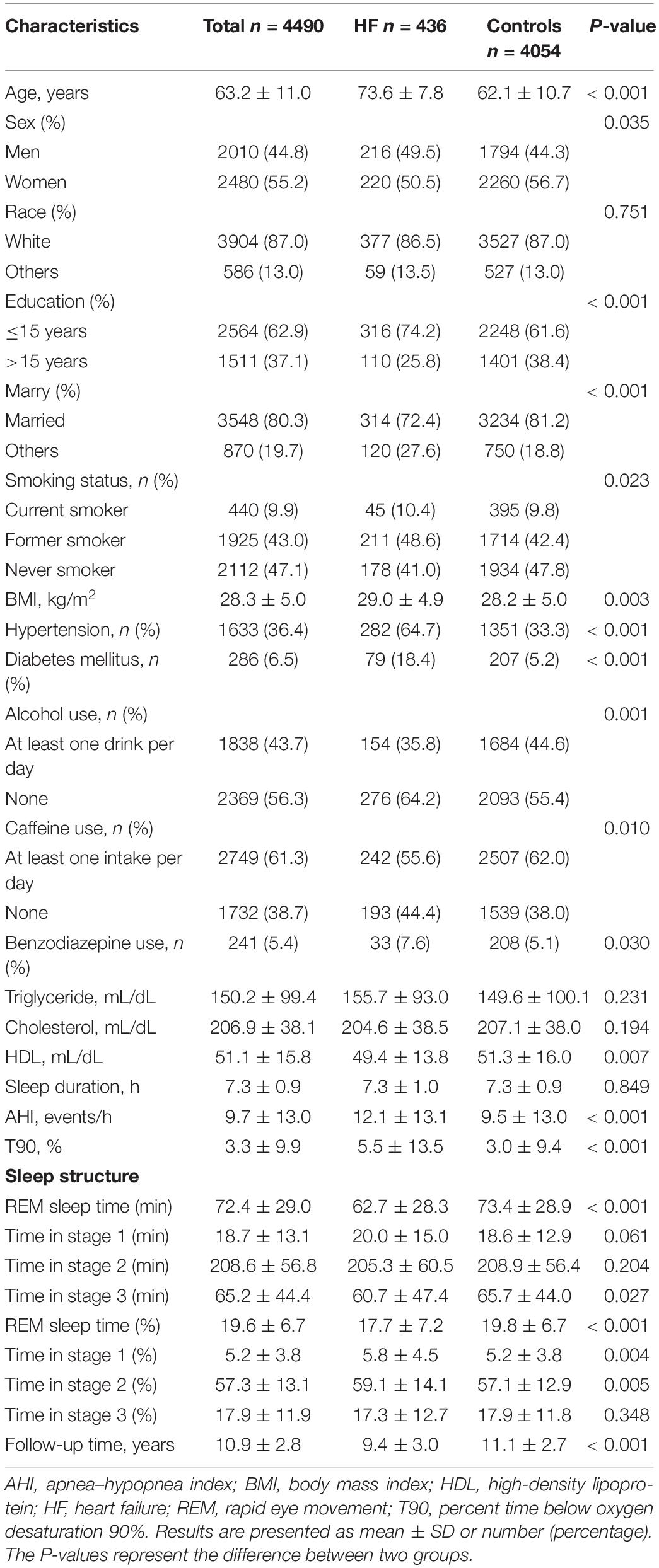
Table 1 shows the study characteristics in participants with and without HF. The present study included 4490 individuals [2480 women (55.2%)] with a mean age of 63.8 ± 11.0 years. A total of 436 (9.7%) cases of HF was observed during the follow-up period. Individuals with HF were older and had more smokers, hypertension, diabetes mellitus than controls. In addition, HF patients had low level of percentage of REM sleep, total REM sleep time, T90, and AHI compared with controls.
The Relationship Between Rapid Eye Movement Sleep and Incident Heart Failure
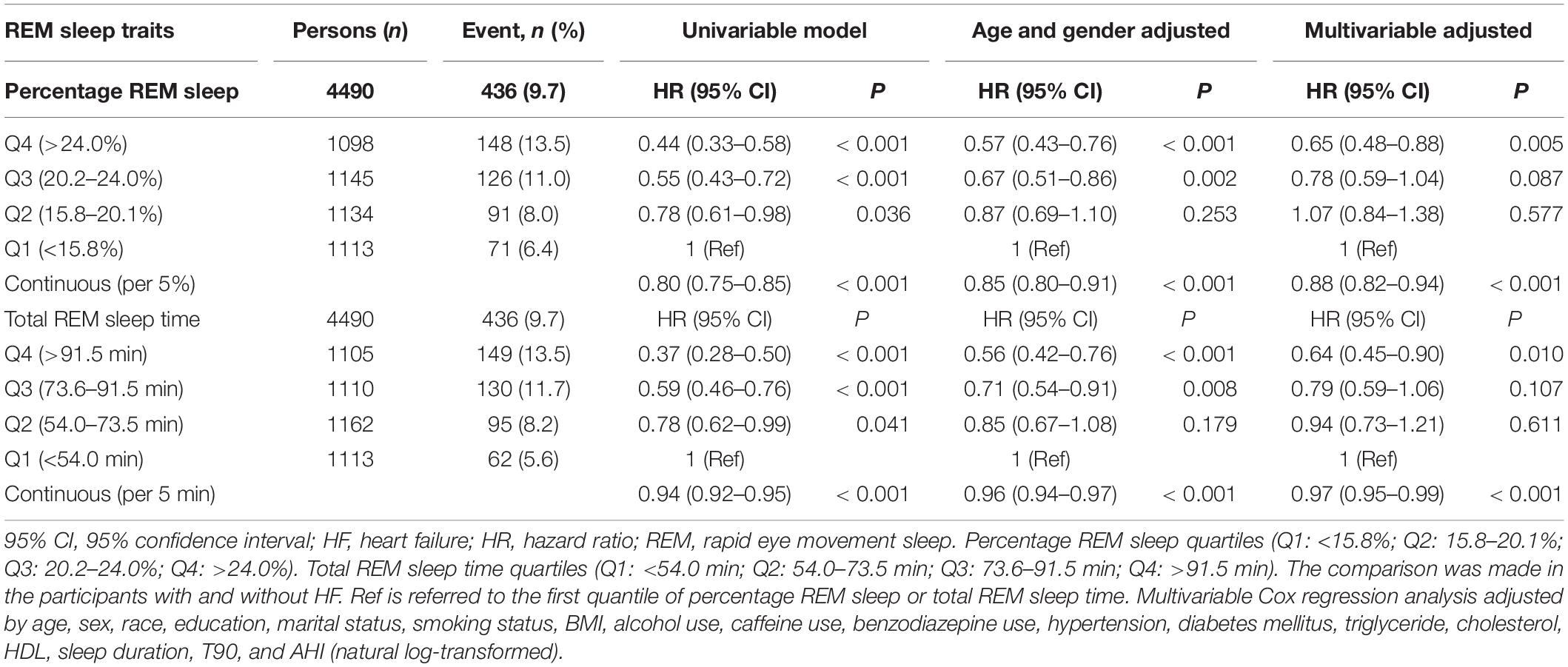
Unadjusted Kaplan–Meier survival curves showed that HF event rates increased with a decrease in REM sleep percentage (Figure 1A; Log-rank test: P < 0.001). After adjusting for age, sex, race, education, marital status, smoking status, BMI, alcohol use, caffeine use, benzodiazepine use, hypertension, diabetes mellitus, triglyceride, cholesterol, HDL, sleep duration, T90, and AHI (natural log-transformed), multivariable Cox regression analysis showed that an elevated percentage of REM sleep (per 5%) was significantly associated with a reduced risk of HF (HR 0.88, 95% CI 0.82–0.94, P < 0.001). Moreover, individuals in the fourth quartile of percentage REM sleep had a significantly lower risk of HF than those in the first quartile (HR 0.65, 95% CI 0.48–0.88, P = 0.005) (Table 2).
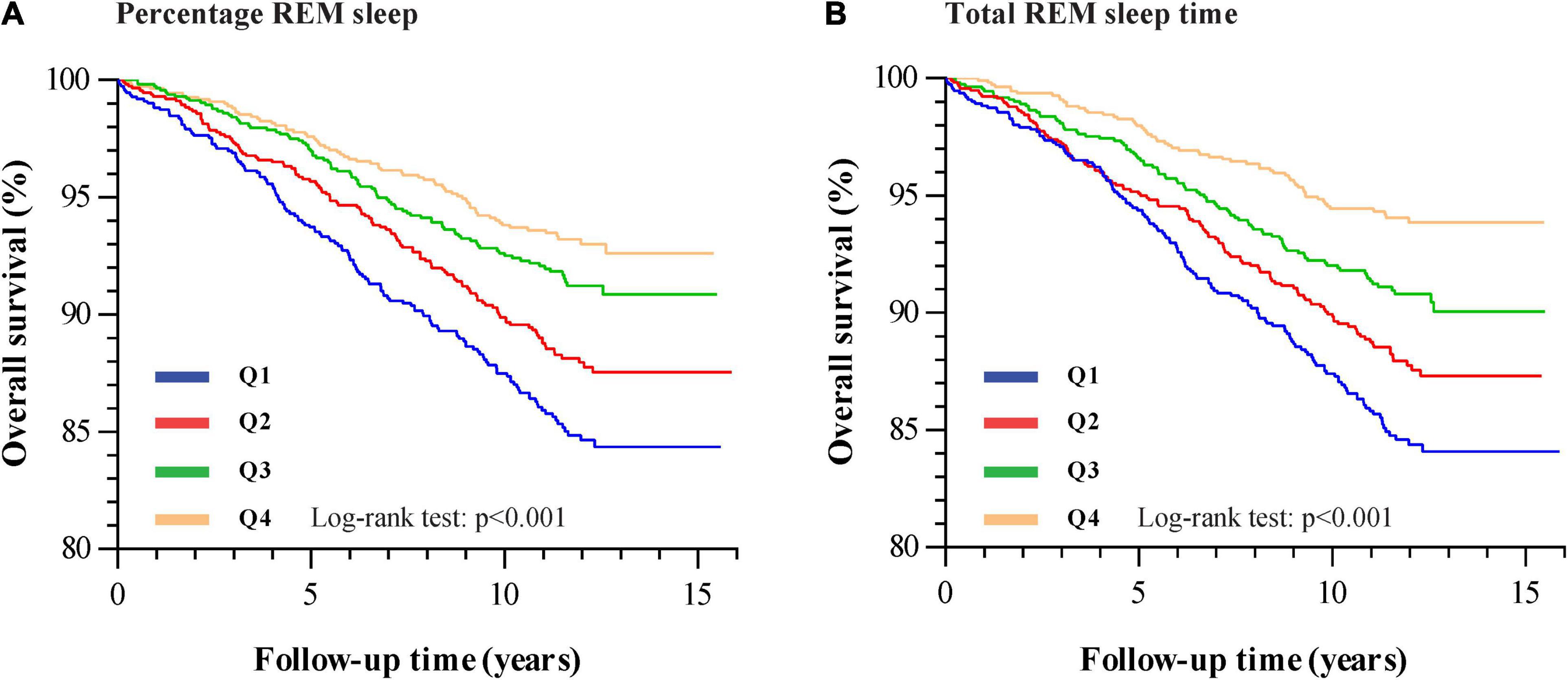
Figure 1. Kaplan–Meier plots of overall survival for HF stratified by percentage REM sleep quartiles [(A) Q1: <15.8%; Q2: 15.8–20.1%; Q3: 20.2–24.0%; Q4: >24.0%] and total REM sleep time quartiles [(B) Q1: <54.0 min; Q2: 54.0–73.5 min; Q3: 73.6–91.5 min; Q4: >91.5 min].
We also explored the association between total REM sleep time and HF. Similar to the results for REM sleep percentage, total REM sleep time (increased per 5 min) was found to be independently associated with HF (HR 0.97, 95% CI 0.95–0.99, P < 0.001). In addition, individuals in the fourth quartile of the total REM sleep time had the highest overall survival rate (Figure 1B; Log-rank test: P < 0.001). Total REM sleep time within the fourth quartile was associated with a lower risk of HF (HR 0.64, 95% CI 0.45–0.90, P = 0.010) than in the first quartile (Table 2). We also showed the results of final fully adjusted model in our Supplementary Tables 1, 2.
The Association of Rapid Eye Movement Sleep With Myocardial Infarction, Stroke, and Cardiovascular Disease Death
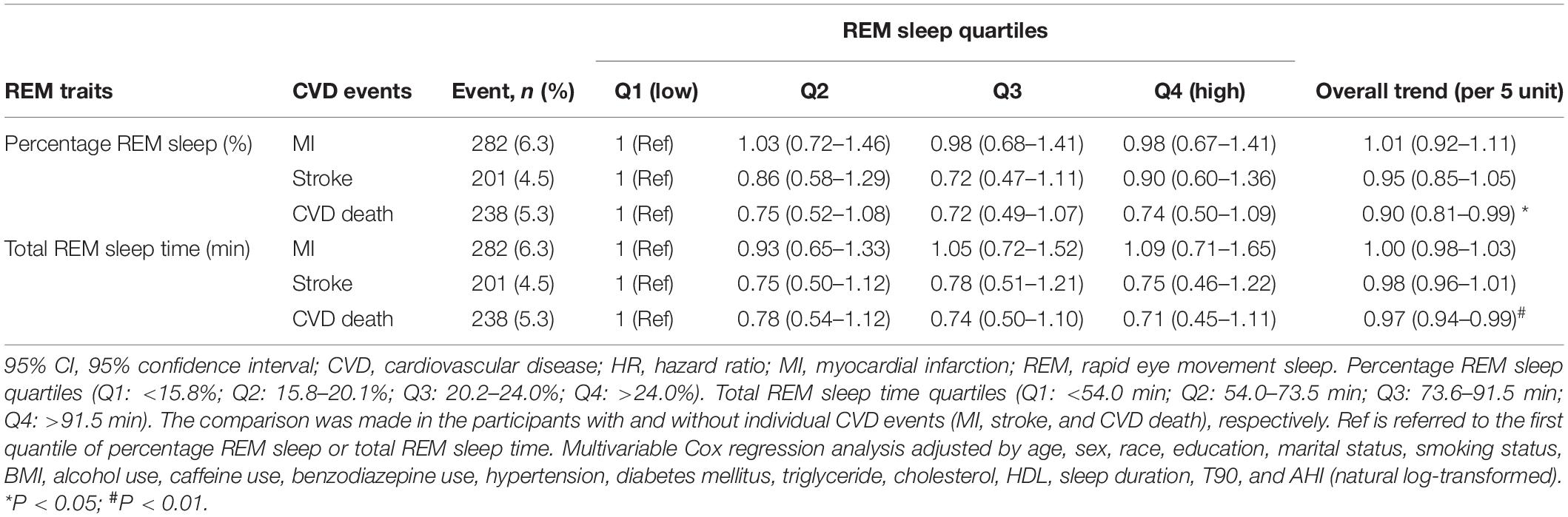
During the follow-up time, 282 cases of MI, 201 cases of stroke, and 238 cases of CVD death occurred. We also explored role of percentage REM sleep and total REM sleep time on the incidence of MI, stroke, and CVD death. Both percentage REM sleep (HR 0.90, 95% CI 0.81–0.99, P = 0.032) and total REM sleep time (HR 0.97, 95% CI 0.94–0.99, P = 0.010) were associated with decreased risk of CVD death, but not associated with MI and stroke (Table 3).
Other Sleep Structure Parameters and Cardiovascular Disease Events
We also investigated the role of sleep characteristics (including time in stage 1, time in stage 2, time in stage 3, percentage stage 1, percentage stage 2, and percentage stage 1) on the incidence of HF, MI, stroke, and CVD death. No significant association was found after adjusting for potential confounding variables (Supplementary Table 3).
Subgroup Analysis
We further conducted subgroup analysis stratified by AHI ≥ 15 and AHI < 15 events/h and sex (men vs. women) to further explore the role of REM sleep percentage and total REM sleep time on the incidence of HF. The results showed that both percentage and total time of REM sleep were still associated with incident HF in these subgroup analyses (Supplementary Tables 4, 5). Moreover, no significant interaction was found in these analyses (all the Pinteraction > 0.05).
Discussion
In the present study, we utilized a large-scale community-based population from SHHS to investigate the association between REM sleep and incidence of HF. Our study was a cohort-study design and the comparison of percentage REM sleep and total REM sleep time was made in the individuals with and without HF. REM sleep traits including percentage REM sleep and total REM sleep time were monitored by over-night PSG at home. Our multivariable Cox regression analysis demonstrated that middle-aged and older adults with an elevated percentage of REM sleep and total REM sleep time had a reduced risk of incident HF.
Poor lifestyles and behaviors are considered the main causes of negative cardiovascular outcomes (18). Sleep, a basic human behavior, is believed to be related to the risk of CVD and other health outcomes (19, 20). REM sleep is an important aspect of human sleep and often accompanied with vivid dreaming and high level of brain activity, which also has substantial effects on the physiological functions of the individual (21, 22). Previous studies showed that participants with a depressed mood spent less time in REM sleep (23). Besides, decreased REM sleep time in individuals is found to be closely related to worsening cognitive performance (24, 25). Matthews et al. also demonstrated a significant correlation between a lower proportion of REM sleep and a greater sleep/wake ratio of blood pressure (13). Increasing evidences have shown that REM sleep is vital to human health, but there was no evidence regarding the role of REM sleep on the incident HF. In this study, we provided evidences that both increased percentage REM sleep and total REM sleep time were significantly associated with low risk of incident HF. Sleep disordered breathing (SDB) is characterized by abnormal respiration during sleep and may influence the sleep continuity (26). SDB was also closely related to an increased risk of incident HF (27). Azarbarzin et al. found that sleep apnea-specific hypoxic burden was associated with the HF risk (28). We therefore adjusted AHI and T90 in our multivariable Cox regression analysis. Moreover, we performed subgroup analysis stratified by SDB severity (AHI ≥ 15 vs. AHI < 15 events/h) to examine whether SDB was potential confounders in the relationship between REM sleep and HF. The results revealed that percentage REM sleep and total REM sleep time were still associated with incidence of HF in subgroup analysis. Our findings indicated that REM sleep traits including percentage REM sleep and total REM sleep time might be marker to predict incident HF.
Previous studies showed that REM sleep is associated with all-cause mortality (11, 12). A decreased percentage of REM sleep was found to be associated with high CVD mortality in the Outcomes of Sleep Disorders in Older Men (MrOS) Sleep Study Cohort, but this relationship was not found in the Wisconsin Sleep Cohort (12). We also explored the role of REM sleep on the MI, stroke, and CVD death. Our results found a significant association of both percentage of REM sleep and total REM sleep time with CVD death based on SHHS, which could support the results of MrOS. However, no significant association was found between REM sleep and incidence of MI and stroke.
Previous studies revealed that the initiation and maintenance of REM sleep was related to the brainstem, forebrain, and hypothalamus (29). Gonnissen et al. showed that the changes of REM sleep time could be caused by circadian misalignment. Besides, reduced REM sleep may be associated with HPA-axis dysregulation and decreased insulin sensitivity (30). Additionally, decreased REM sleep was also found to have a high cortisol concentration (31). We speculate that the abnormal circadian rhythm and neuroendocrine function may contribute to the increased risk of CHF. The underlying biological mechanisms of REM sleep leading to an increased risk of HF still deserved further investigation.
The current study has some strengths. To our knowledge, it is the first to investigate the effect of REM sleep on the incidence of HF. REM sleep, including percentage and total time, was objectively monitored using PSG records, and our findings were based on a large community-based population. Nevertheless, this study also has several limitations. The objects of our analysis were mostly middle-aged and older Caucasian adults; therefore, the generalization of our conclusions to young people and other races merits careful consideration. Second, objective REM sleep was evaluated using a single-night PSG and may not fully reflect the significant value of REM sleep. Multiple long-term PSG monitoring may provide more accurate sleep parameters. Third, several parent cohorts oversampled snorers to increase the study-wide prevalence of SDB in SHHS. Therefore, our study population could not represent the general community population. Finally, we lack of data such as B-type natriuretic peptide (BNP), kidney function and left ventricular parameters such as ejection fraction, end-diastolic volume and end-systolic volume in the SHHS database that is closely related to the HF. We will investigate the effect of REM sleep on the changes of BNP, renal function and left ventricular function in our following study.
Conclusion
Our study provides evidence that increased percentage and total time of REM sleep were associated with a decreased risk of incident HF in middle-aged and older adults. Percentage REM sleep and total REM sleep time may be predictors for the incidence of HF. Monitoring REM sleep to fully understand the nocturnal autonomic nervous activity of people may contribute to prevent HF in the early stages.
Data Availability Statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://doi.org/10.25822/ghy8-ks59.
Ethics Statement
The studies involving human participants were reviewed and approved by the Boston University, Case Western Reserve University, Johns Hopkins University, Missouri Breaks Research, Inc., New York University Medical Center, University of Arizona, University of California at Davis, University of Minnesota – Clinical and Translational Science Institute, and University of Washington. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
BY and XM raised the idea for the study and handled the supervision in our study. BZ, XJ, JY, QM, LB, ZY, and WW contributed to the study design, writing, and review of the report. BY acquired the data in SHHS and participated in further data analysis. All authors approved the final version of the report.
Funding
This study was supported by the Natural Science Basic Research Program of Shaanxi (Nos. 2021JQ-395 and 2020ZDLSF02-10) and the Clinical Research Award of the First Affiliated Hospital of Xi’an Jiaotong University, China (No. XJTU1AF-CRF-2019-022). SHHS was supported by the National Heart, Lung, and Blood Institute cooperative agreements U01HL53916 (University of California, Davis), U01HL53931 (New York University), U01HL53934 (University of Minnesota), U01HL53937 and U01HL64360 (Johns Hopkins University), U01HL53938 (University of Arizona), U01HL53940 (University of Washington), U01HL53941 (Boston University), and U01HL63463 (Case Western Reserve University). The National Sleep Research Resource was supported by the National Heart, Lung, and Blood Institute (R24 HL114473 and 75N92019R002). SHHS was particularly grateful to the members of these cohorts who agreed to participate in SHHS as well.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We appreciate the Brigham and Women’s Hospital for sharing the Datasets of Sleep Heart Health Study (SHHS). SHHS recognizes all the investigators and staff who have contributed to its success. A list of SHHS investigators, staff and their participating institutions is available on the SHHS website, https://doi.org/10.25822/ghy8-ks59.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcvm.2022.771280/full#supplementary-material
Abbreviations
AHI, apnea–hypopnea index; BMI, body mass index; HF, heart failure; CI, confidence interval; CVD, cardiovascular disease; HDL, high-density lipoprotein; HR, hazard ratio; MI, myocardial infarction; MrOS, Outcomes of Sleep Disorders in Older Men; PSG, polysomnography; REM, rapid eye movement; SHHS, Sleep Heart Health Study; T90, percent time below oxygen desaturation 90%.
References
1. Kaufmann T, Elvsåshagen T, Alnæs D, Zak N, Pedersen PØ, Norbom LB, et al. The brain functional connectome is robustly altered by lack of sleep. NeuroImage. (2016) 127:324–32. doi: 10.1016/j.neuroimage.2015.12.028
2. Ibarra-Coronado EG, Pantaleon-Martinez AM, Velazquez-Moctezuma J, Prospéro-García O, Méndez-Díaz M, Pérez-Tapia M, et al. The bidirectional relationship between sleep and immunity against infections. J Immunol Res. (2015) 2015:678164. doi: 10.1155/2015/678164
3. Research IoMUCoSMa. Colten HR, Altevogt BM editors. Sleep Disorders and Sleep Deprivation: An Unmet Public Health Problem. Washington, DC: National Academies Press (US) (2006).
4. Wang C, Bangdiwala S, Rangarajan S, Lear SA, AlHabib KF, Mohan V, et al. Association of estimated sleep duration and naps with mortality and cardiovascular events: a study of 116 632 people from 21 countries. Eur Heart J. (2019) 40:1620–9. doi: 10.1093/eurheartj/ehy695
5. Wang Y, Wang J, Chen S, Li JQ, Lu QD, Vitiello MV, et al. Association of Longitudinal patterns of habitual sleep duration with risk of cardiovascular events and all-cause mortality. JAMA Netw Open. (2020) 3:e205246. doi: 10.1001/jamanetworkopen.2020.5246
6. Yan B, Zhao B, Fan Y, Yang J, Zhu F, Chen Y, et al. The association between sleep efficiency and diabetes mellitus in community-dwelling individuals with or without sleep-disordered breathing. J Diabetes. (2020) 12:215–23. doi: 10.1111/1753-0407.12987
7. Rosique-Esteban N, Papandreou C, Romaguera D, Warnberg J, Corella D, Martínez-González MÁ, et al. Cross-sectional associations of objectively-measured sleep characteristics with obesity and type 2 diabetes in the PREDIMED-Plus trial. Sleep. (2018) 41:zsy190. doi: 10.1093/sleep/zsy190
8. Li X, Sotres-Alvarez D, Gallo LC, Ramos AR, Aviles-Santa L, Perreira KM, et al. Associations of sleep-disordered breathing and insomnia with incident hypertension and diabetes. the hispanic community health study/study of latinos. Am J Respir Crit Care Med. (2021) 203:356–65. doi: 10.1164/rccm.201912-2330OC
9. Simor P, van der Wijk G, Nobili L, Peigneux P. The microstructure of REM sleep: why phasic and tonic? Sleep Med Rev. (2020) 52:101305. doi: 10.1016/j.smrv.2020.101305
10. Galbiati A, Sforza M, Fasiello E, Casoni F, Marrella N, Leitner C, et al. The association between emotional dysregulation and REM sleep features in insomnia disorder. Brain Cogn. (2020) 146:105642. doi: 10.1016/j.bandc.2020.105642
11. Zhang J, Jin X, Li R, Gao Y, Li J, Wang G. Influence of rapid eye movement sleep on all-cause mortality: a community-based cohort study. Aging (Albany NY). (2019) 11:1580–8. doi: 10.18632/aging.101858
12. Leary EB, Watson KT, Ancoli-Israel S, Redline S, Yaffe K, Ravelo LA, et al. Association of rapid eye movement sleep with mortality in middle-aged and older adults. JAMA Neurol. (2020) 77:1–12. doi: 10.1001/jamaneurol.2020.2108
13. Matthews KA, Kamarck TW, Hall MH, Strollo PJ, Owens JF, Buysse DJ, et al. Blood pressure dipping and sleep disturbance in African-American and caucasian men and women. Am J Hypertens. (2008) 21:826–31. doi: 10.1038/ajh.2008.183
14. Quan SF, Howard BV, Iber C, Kiley JP, Nieto FJ, O’Connor GT, et al. The sleep heart health study: design, rationale, and methods. Sleep. (1997) 20:1077–85.
15. Redline S, Sanders MH, Lind BK, Quan SF, Iber C, Gottlieb DJ, et al. Methods for obtaining and analyzing unattended polysomnography data for a multicenter study. sleep heart health research group. Sleep. (1998) 21:759–67. doi: 10.1093/sleep/21.7.759
16. Yan B, Wu Y, Fan X, Lu Q, Ma X, Bai L. Sleep fragmentation and incidence of congestive heart failure: the sleep heart health study. J Clin Sleep Med. (2021) 17:1619–25. doi: 10.5664/jcsm.9270
17. Punjabi NM, Caffo BS, Goodwin JL, Gottlieb DJ, Newman AB, O’Connor GT, et al. Sleep-disordered breathing and mortality: a prospective cohort study. PLoS Med. (2009) 6:e1000132. doi: 10.1371/journal.pmed.1000132
18. Writing Group M, Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, et al. Heart disease and stroke statistics-2016 update: a report from the american heart association. Circulation. (2016) 133:e38–360.
19. Lee SWH, Ng KY, Chin WK. The impact of sleep amount and sleep quality on glycemic control in type 2 diabetes: a systematic review and meta-analysis. Sleep Med Rev. (2017) 31:91–101. doi: 10.1016/j.smrv.2016.02.001
20. Covassin N, Singh P. Sleep duration and cardiovascular disease risk: epidemiologic and experimental evidence. Sleep Med Clin. (2016) 11:81–9. doi: 10.1016/j.jsmc.2015.10.007
21. Blumberg MS, Lesku JA, Libourel PA, Schmidt MH, Rattenborg NC. What is REM sleep? Curr Biol. (2020) 30:R38–49.
22. McCarley RW. Neurobiology of REM and NREM sleep. Sleep Med. (2007) 8:302–30. doi: 10.1016/j.sleep.2007.03.005
23. Smagula S, Reynolds C, Ancoli-Israel S, Barrett-Connor E, Dam TT, Hughes-Austin JM, et al. Sleep architecture and mental health among community-dwelling older men. Journals Gerontol Series B Psychol Sci Soc Sci. (2015) 70:673–81. doi: 10.1093/geronb/gbt125
24. Song Y, Blackwell T, Yaffe K, Ancoli-Israel S, Redline S, Stone K. Relationships between sleep stages and changes in cognitive function in older men: the MrOS sleep study. Sleep. (2015) 38:411–21. doi: 10.5665/sleep.4500
25. Scullin MK, Gao C, Fillmore P, Roberts RL, Pruett N, Bliwise DL. Rapid eye movement sleep mediates age-related decline in prospective memory consolidation. Sleep. (2019) 42:zsz055. doi: 10.1093/sleep/zsz055
26. Sanders MH, Newman AB, Haggerty CL, Redline S, Lebowitz M, Samet J, et al. Sleep and sleep-disordered breathing in adults with predominantly mild obstructive airway disease. Am J Respir Crit Care Med. (2003) 167:7–14. doi: 10.1164/rccm.2203046
27. Gottlieb DJ, Yenokyan G, Newman AB, O’Connor GT, Punjabi NM, Quan SF, et al. Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: the sleep heart health study. Circulation. (2010) 122:352–60. doi: 10.1161/circulationaha.109.901801
28. Azarbarzin A, Sands SA, Taranto-Montemurro L, Vena D, Sofer T, Kim SW, et al. The sleep Apnea-specific hypoxic burden predicts incident heart failure. Chest. (2020) 158:739–50. doi: 10.1016/j.chest.2020.03.053
29. Fraigne JJ, Grace KP, Horner RL, Peever J. Mechanisms of REM sleep in health and disease. Curr Opin Pulm Med. (2014) 20:527–32. doi: 10.1097/mcp.0000000000000103
30. Gonnissen HK, Mazuy C, Rutters F, Martens EA, Adam TC, Westerterp-Plantenga MS. Sleep architecture when sleeping at an unusual circadian time and associations with insulin sensitivity. PLoS One. (2013) 8:e72877. doi: 10.1371/journal.pone.0072877
Keywords: percentage of REM sleep, total REM sleep time, sleep heart health study, polysomnography, heart failue
Citation: Zhao B, Jin X, Yang J, Ma Q, Yang Z, Wang W, Bai L, Ma X and Yan B (2022) Increased Rapid Eye Movement Sleep Is Associated With a Reduced Risk of Heart Failure in Middle-Aged and Older Adults. Front. Cardiovasc. Med. 9:771280. doi: 10.3389/fcvm.2022.771280
Received: 06 September 2021; Accepted: 04 March 2022;
Published: 29 March 2022.
Edited by:
Tommaso Gori, Johannes Gutenberg University Mainz, GermanyReviewed by:
Eisuke Amiya, The University of Tokyo Hospital, JapanGian Domenico Pinna, Scientific Clinical Institute Maugeri (ICS Maugeri), Italy
Frederic Roche, Université Jean Monnet, France
Copyright © 2022 Zhao, Jin, Yang, Ma, Yang, Wang, Bai, Ma and Yan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bin Yan, eWFuYmluLjE5ODdAaG90bWFpbC5jb20=
†These authors have contributed equally to this work
 Binbin Zhao
Binbin Zhao Xiaoying Jin
Xiaoying Jin Jian Yang
Jian Yang Qingyan Ma
Qingyan Ma Zai Yang
Zai Yang Wei Wang
Wei Wang Ling Bai
Ling Bai Xiancang Ma
Xiancang Ma Bin Yan
Bin Yan