
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Cardiovasc. Med. , 21 October 2021
Sec. General Cardiovascular Medicine
Volume 8 - 2021 | https://doi.org/10.3389/fcvm.2021.747620
This article is part of the Research Topic CardioNeurology: Basic, translational and clinical research View all 20 articles
 Jonathan V. Mui1†
Jonathan V. Mui1† Jiandong Zhou2†
Jiandong Zhou2† Sharen Lee1
Sharen Lee1 Keith Sai Kit Leung3
Keith Sai Kit Leung3 Teddy Tai Loy Lee3
Teddy Tai Loy Lee3 Oscar Hou In Chou3
Oscar Hou In Chou3 Shek Long Tsang1
Shek Long Tsang1 Abraham Ka Chung Wai3
Abraham Ka Chung Wai3 Tong Liu4
Tong Liu4 Wing Tak Wong5
Wing Tak Wong5 Carlin Chang6
Carlin Chang6 Gary Tse4,7*
Gary Tse4,7* Qingpeng Zhang1*
Qingpeng Zhang1*Introduction: The effects of sodium-glucose cotransporter 2 inhibitors (SGLT2I) and dipeptidyl peptidase-4 inhibitors (DPP4I) on new-onset cognitive dysfunction in type 2 diabetes mellitus remain unknown. This study aimed to evaluate the effects of the two novel antidiabetic agents on cognitive dysfunction by comparing the rates of dementia between SGLT2I and DPP4I users.
Methods: This was a population-based cohort study of type 2 diabetes mellitus patients treated with SGLT2I and DPP4I between January 1, 2015 and December 31, 2019 in Hong Kong. Exclusion criteria were <1-month exposure or exposure to both medication classes, or prior diagnosis of dementia or major neurological/psychiatric diseases. Primary outcomes were new-onset dementia, Alzheimer's, and Parkinson's. Secondary outcomes were all-cause, cardiovascular, and cerebrovascular mortality.
Results: A total of 13,276 SGLT2I and 36,544 DPP4I users (total n = 51,460; median age: 66.3 years old [interquartile range (IQR): 58–76], 55.65% men) were studied (follow-up: 472 [120–792] days). After 1:2 matching (SGLT2I: n = 13,283; DPP4I: n = 26,545), SGLT2I users had lower incidences of dementia (0.19 vs. 0.78%, p < 0.0001), Alzheimer's (0.01 vs. 0.1%, p = 0.0047), Parkinson's disease (0.02 vs. 0.14%, p = 0.0006), all-cause (5.48 vs. 12.69%, p < 0.0001), cerebrovascular (0.88 vs. 3.88%, p < 0.0001), and cardiovascular mortality (0.49 vs. 3.75%, p < 0.0001). Cox regression showed that SGLT2I use was associated with lower risks of dementia (hazard ratio [HR]: 0.41, 95% confidence interval [CI]: [0.27–0.61], P < 0.0001), Parkinson's (HR:0.28, 95% CI: [0.09–0.91], P = 0.0349), all-cause (HR:0.84, 95% CI: [0.77–0.91], P < 0.0001), cardiovascular (HR:0.64, 95% CI: [0.49–0.85], P = 0.0017), and cerebrovascular (HR:0.36, 95% CI: [0.3–0.43], P < 0.0001) mortality.
Conclusions: The use of SGLT2I is associated with lower risks of dementia, Parkinson's disease, and cerebrovascular mortality compared with DPP4I use after 1:2 ratio propensity score matching.
Type-2 diabetes mellitus is a complex multi-systemic disorder with wide-ranging complications affecting the retinal, cardiovascular, renal, and peripheral nervous systems (1–5). Increasingly, cognitive dysfunction is being recognized as a clinically important complication of type-2 diabetes (6). Diabetic patients are associated with a 1.5-fold increased risk of cognitive dysfunction, 1.9-fold increased risk of dementia, and 2.2-fold increased risk of stroke (7–9). While the underlying pathophysiology is still unclear, several mechanisms have been proposed including insulin resistance, hypoglycemia, hyperglycemia-induced cerebral microvascular and macrovascular dysfunction, as well as amyloid deposition (10, 11). It is highly likely that the cognitive dysfunction is multifactorial and caused by a combination of these mechanisms specific to the demographic and comorbidities of the patient.
Several studies have suggested that improved glycemic control, reduced HbA1c levels, and use of anti-diabetic medication are associated with a reduced risk of cognitive dysfunction (12–15). This has consequently raised the prospect of anti-diabetic agents reducing cognitive dysfunction in type 2 diabetes patients. Of interest are novel second-line anti-diabetic agents including sodium-glucose cotransporter 2 inhibitors (SGLT2I) and dipeptidyl peptidase-4 inhibitors. Multiple preclinical studies have suggested that DPP4I and SGLT2I improve cognition in animal models via a variety of mechanisms (16–20). However, few clinical studies have explored SGLT2I and DPP4I in their effects on cognitive dysfunction in diabetic patients. A randomized controlled trial in 2018 found no cognitive decline in SGLT2I and DPP4I users within 12 months while a case-control study in 2019 found that DPP4I and SGLT2I use are associated with a lower risk of dementia compared with other anti-diabetic agents (21, 22). Until recent times, no study has directly compared the risk of cognitive dysfunction and major neurocognitive disorders among SGLT2I and DPP4I users.
Therefore, the present study aimed to compare the incidence of dementia in SGLT2 users against DPP4I users in a Chinese population to evaluate the effects of the two novel antidiabetic agents on cognitive dysfunction.
This was a retrospective, territory-wide cohort study of type-2 diabetes mellitus patients with SGLT2I/DPP4I use between January 1, 2015, and December 31, 2019 in Hong Kong (Figure 1). Patients during the aforementioned period were enrolled and followed up until December 31, 2019, or until death. Patients with <1 month SGLT2I/DPP4I exposure (N = 3,225), with both SGLT2I and DPP4I therapy (N = 15,276), or with a prior diagnosis of all-cause dementia, Alzheimer's disease, dementia with Lewy bodies, vascular dementia, frontotemporal dementia, or other major neurological/psychiatric diseases (N = 2,785) were excluded.
The patients were identified from the Clinical Data Analysis and Reporting System (CDARS), a city-wide database that centralizes patient information from individual local hospitals to establish comprehensive medical data, including clinical characteristics, disease diagnosis, laboratory results, and drug treatment details. The system has been previously used by both our team and other teams in Hong Kong (23–25). Clinical and biochemical data were extracted for the present study. The demographics of the patients include gender and age of initial drug use (baseline). Prior comorbidities were extracted based on standard International Classification of Diseases Ninth Edition (ICD-9) codes (Supplementary Table 1). The Charlson comorbidity index and neutrophil-to-lymphocyte ratio (NLR) were calculated. Mortality was recorded using the International Classification of Diseases Tenth Edition (ICD-10) coding. ICD-10 codes I00-I09, I11, I13, I20-I51 were used to identify cardiovascular mortality outcomes. ICD-10 codes I60-I69 identified cerebrovascular mortality. Medication histories and baseline laboratory examinations were extracted. Mortality data were obtained from the Hong Kong Death Registry, a population-based official government registry with the registered death records of all Hong Kong citizens linked to CDARS.
The primary outcomes were new-onset dementia, new-onset Alzheimer's disease, and new-onset Parkinson's disease. The secondary outcomes were all-cause mortality, cardiovascular mortality, and cerebrovascular mortality. Descriptive statistics were used to summarize baseline clinical and biochemical characteristics of patients with SGLT2I and DPP4I use. For baseline clinical characteristics, the continuous variables were presented as median (95% confidence interval [CI]/interquartile range [IQR]) and the categorical variables were presented as total number (percentage). Continuous variables were compared using the two-tailed Mann-Whitney U test, while the two-tailed Chi-square test with Yates' correction was used to test 2 × 2 contingency data. Propensity score matching with 1:2 ratio between SGLT2I and DPP4I users based on demographics, Charlson comorbidity index, prior comorbidities, non-SGLT2I/DPP4I medications, baseline fasting glucose, and HbA1c tests were performed using the nearest neighbor search strategy. Propensity score matching results between treatment-group (SGLT2I) vs. control-group (DPP4I) before and after matching are shown in Supplementary Figure 1. Propensity score matching adjustment approaches including propensity score stratification (26), propensity score matching with inverse probability weighting (27) and high-dimensional propensity score (28) were also performed.
Cox regression models were used to identify significant risk predictors for the study outcomes. Competing risk analysis models (cause-specific and sub-distribution) were considered. The hazard ratio (HR), 95% CI, and P-value were reported. Statistical significance is defined as P < 0.05. All statistical analyses were performed with R studio (Boston, MA, Version 1.1.456) and Python (Scotts Valley, CA, Version 3.6).
The study cohort included 13,276 SGLT2I users and 36,544 DPP4I users (total n = 51,460; median age: 66.3 years old [IQR: 58–76], 55.65% men). After a mean follow-up of 472 days (IQR: 120–792), 724 (1.45%) developed new-onset dementia, 107 (0.21%) developed new-onset Alzheimer's disease, 77 (0.15%) developed with new onset Parkinson's disease, and in total, 5,687 (11.41%) died from all-causes in which 833 (1.67%) died with cardiovascular causes and 217 (0.43%) died with cerebrovascular causes.
The baseline and clinical characteristics of DPP4I and SGLT2I users before and after 1:2 propensity score matching are shown in Table 1. Both before and after 1:2 propensity score matching, SGLT2I users had lower incidences of new-onset dementia (0.19 vs. 0.78%, p < 0.0001), new onset Alzheimer's disease (0.01 vs. 0.1%, p = 0.0047), new onset Parkinson's disease (0.02 vs. 0.14%, p = 0.0006), all-cause mortality (5.48 vs. 12.69%, p < 0.0001), cardiovascular mortality (0.49 vs. 3.75%, p < 0.0001), and cerebrovascular mortality (0.88 vs. 3.88%, p < 0.0001) compared with DPP4I users. The balancing comparisons of treated (SGLT2I) and controls (DPP4I) after 1:2 propensity matching with nearest neighbor search strategy are shown in Supplementary Table 2. None of the confounding characteristics remained significant after propensity matching.
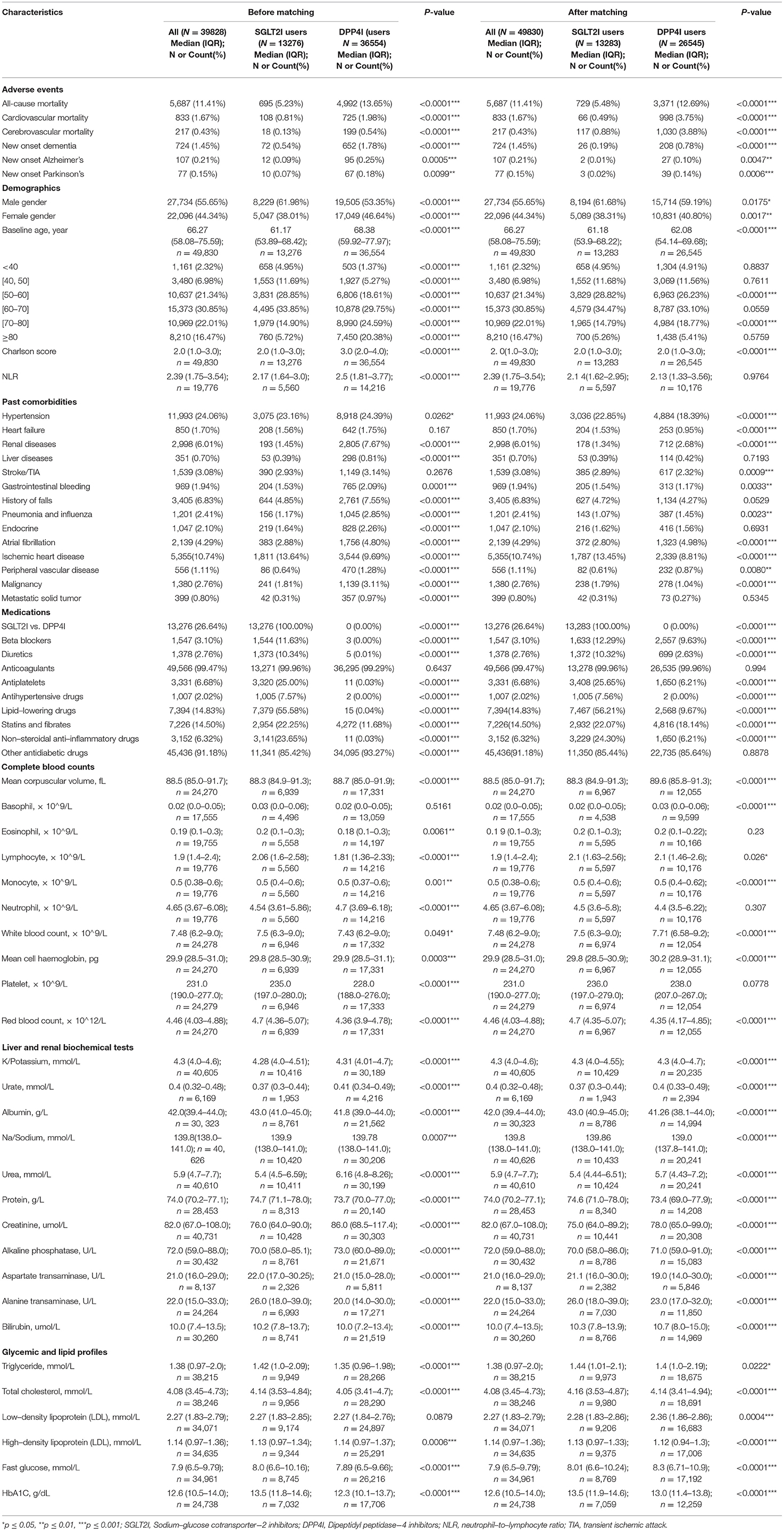
Table 1. Baseline and clinical characteristics of patients with DPP4I vs. SGLT2I uses before and after propensity score matching (1:2).
The baseline and clinical characteristics of patients with new-onset dementia, new-onset Alzheimer's, and new-onset Parkinson's before and after 1:2 propensity score matching are shown in Table 2. The cumulative incidence curves for new-onset cognitive dysfunction and mortality outcomes stratified by the drug use of SGLT2I and DPP4I after 1:2 propensity score matching are shown in Figures 2, 3, respectively.
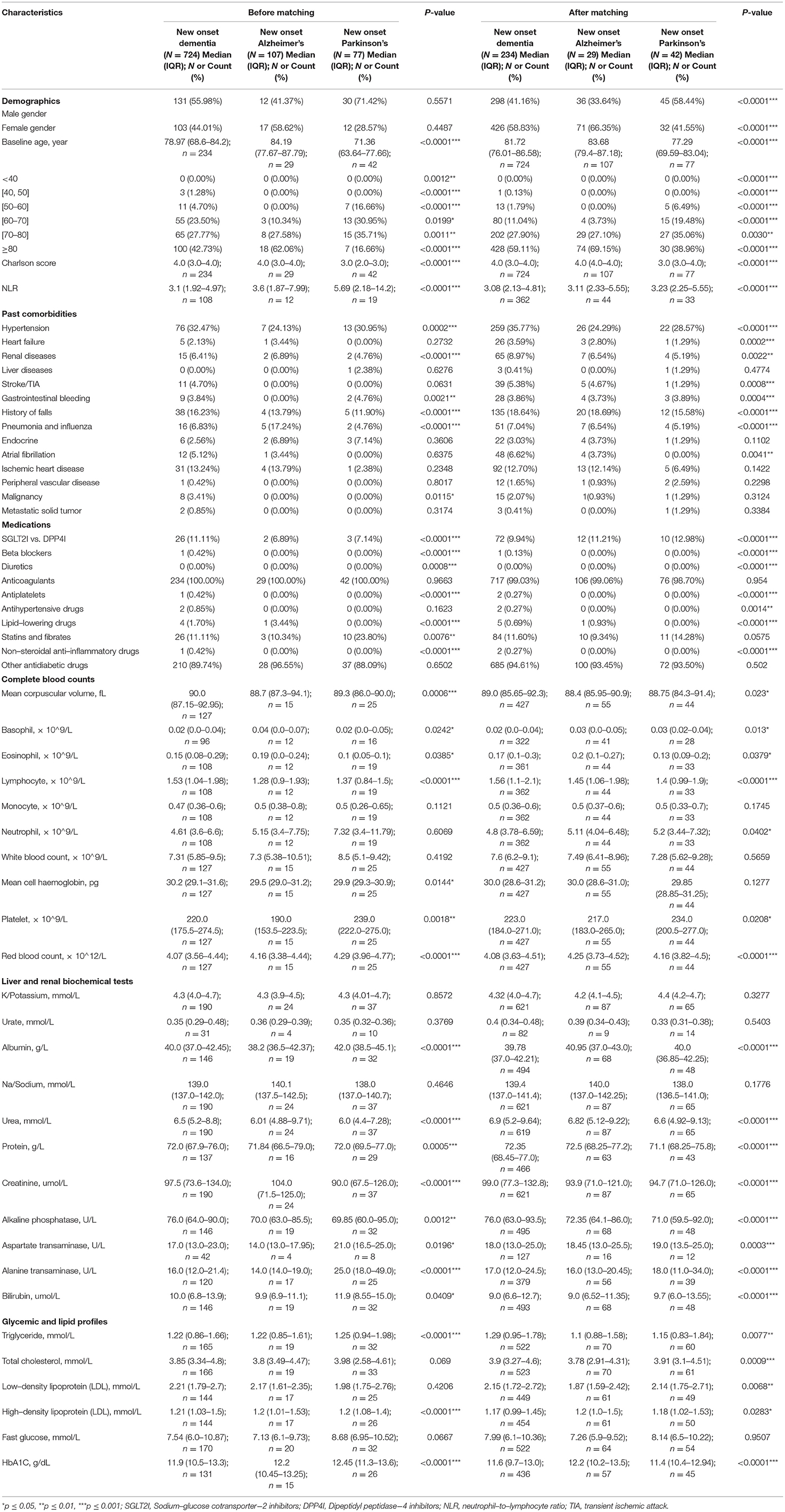
Table 2. Baseline and clinical characteristics of patients with new–onset dementia, Alzheimer's, and Parkinson's before and after propensity score matching (1:2).
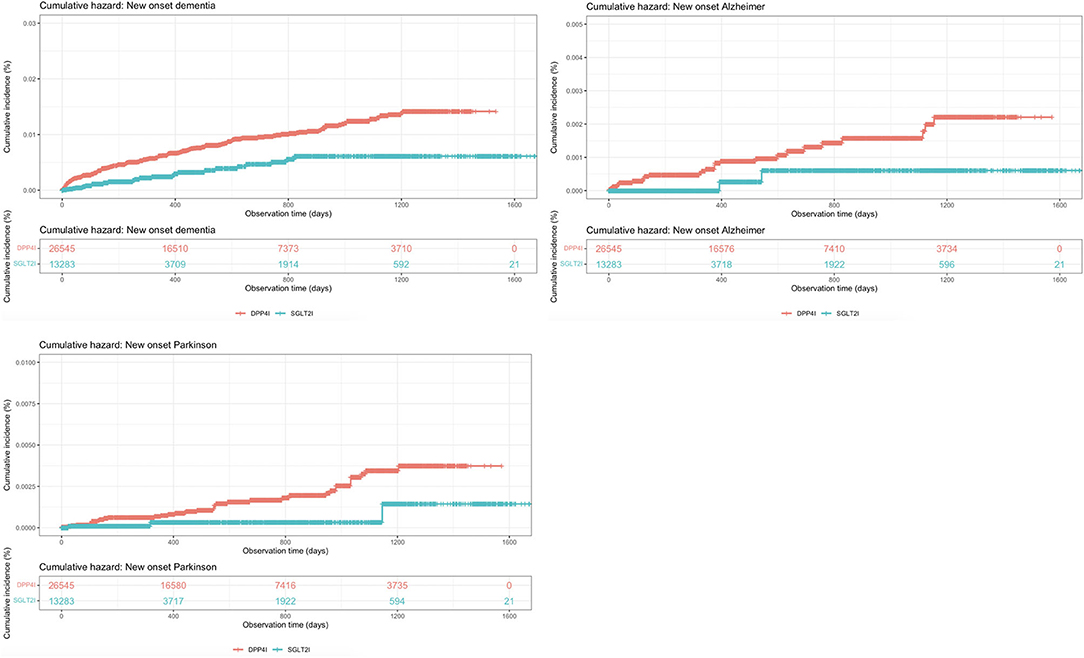
Figure 2. Cumulative incidence curves for new-onset cognitive dysfunctions stratified by the drug use of SGLT2I and DPP4I after propensity score matching (1:2).

Figure 3. Cumulative incidence curves for mortality outcomes stratified by the drug use of SGLT2I and DPP4I after propensity score matching (1:2).
The univariate Cox analyses of significant risk factors for new-onset dementia, Alzheimer's, and Parkinson's disease are shown in Supplementary Table 3 while the univariate Cox analyses of significant risk factors for all-cause, cardiovascular, and cerebrovascular mortality are shown in Supplementary Table 4. Compared with DPP4I, SGLT2I use demonstrated significant protective effects against new onset dementia (HR:0.41, 95% CI: [0.27, 0.61], P <0.0001) and new onset Parkinson's disease (HR:0.28, 95% CI: [0.09, 0.91], P = 0.0349), but not new onset Alzheimer's disease (HR:0.25, 95% CI: [0.06, 1.04], P = 0.0569). SGLT2 use was also associated with significantly lower incidence of all-cause mortality (HR:0.84, 95% CI: [0.77, 0.91], P < 0.0001), cardiovascular mortality (HR:0.64, 95% CI: [0.49, 0.85], P = 0.0017), and cerebrovascular mortality (HR:0.36, 95% CI: [0.30, 0.43], P < 0.0001).
Competing for risk analyses using cause-specific and subdistribution hazard models were conducted on the matched cohorts as presented in Table 3. Both models confirmed the findings from the univariate Cox analyses that SGLT2I use is associated with lower incidence of new-onset dementia, new-onset Parkinson's, all-cause mortality, cardiovascular mortality, and cerebrovascular mortality, but not new-onset Alzheimer's disease compared with DPP4I use. In addition, sensitivity analyses were further conducted using Cox proportional hazard model on the matched cohorts with 1-year lag time, as presented in Supplementary Table 5.
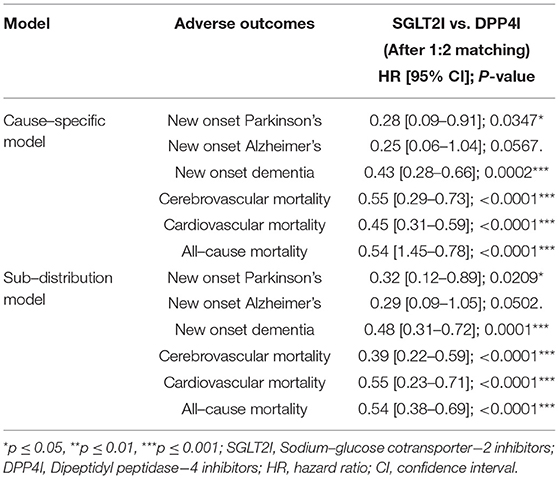
Table 3. HRs (and 95% CIs) of SGLT2I vs. DPP4I from cause–specific and subdistribution hazard models for cognitive dysfunction and mortality risks after 1:2 propensity score matching.
Finally, different propensity score matching adjustment approaches were performed as presented in Table 4. Again, the three approaches confirmed the findings from the univariate Cox analyses that SGLT2I users have a lower risk of new-onset dementia, new-onset Parkinson's, all-cause mortality, cardiovascular mortality, and cerebrovascular mortality, but not new-onset Alzheimer's disease compared with DPP4I users.
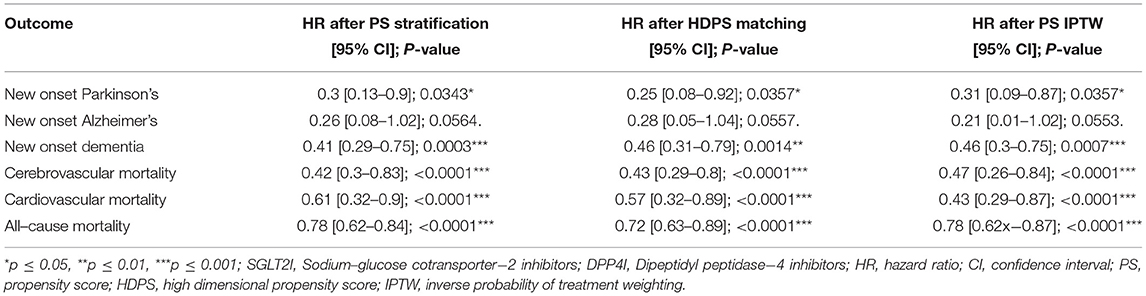
Table 4. Risk of incident adverse cognitive dysfunction events, and mortality outcomes in matched cohorts associated with the treatment of SGLT2I vs. DPP4I using different matching approaches.
A subgroup analysis was performed on SGLT2I and DPP4I users with concurrent type-2 diabetes and cardiovascular disease (defined as heart failure, myocardial infarction, ischemic heart disease, peripheral vascular disease, atrial fibrillation, or cardiovascular medication use) (Table 5). Patients with new-onset cardiovascular disease after SGLT2I/DPP4I use were excluded.
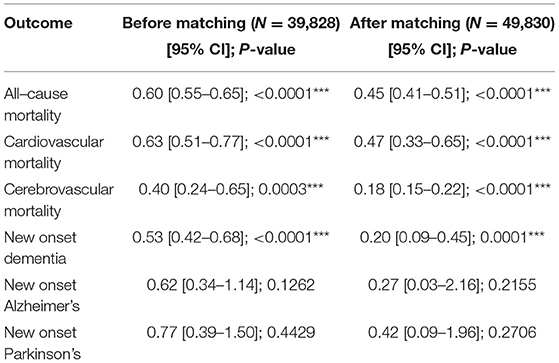
Table 5. Subgroup analysis: Treatment effects of SGLT2I vs. DPP4I for incident adverse cognitive dysfunction events, and mortality outcomes in patients with both type−2 diabetes mellitus and cardiovascular diseases before and after propensity score matching (1:2).
After propensity-score matching, SGLT2I users had a median follow-up time of 459 days (IQR: 42–849) while DPP4I users had a median follow-up time of 522 days (IUQ: 74–1,004). SGLT2I users had a significantly lower risk of new-onset dementia (HR:0.2, 95% CI: [0.09, 0.45], P < 0.0001) but not new-onset Alzheimer's disease (HR:0.27, 95% CI: [0.03, 2.16], P = 0.2155) and Parkinson's disease (HR:0.42, 95% CI: [0.09, 1.96], P = 0.2706) compared with DPP4I users.
This study demonstrated several major findings. Firstly, SGLT2I users had a lower risk of new-onset dementia, Alzheimer's disease, and Parkinson's disease compared with DPP4I users. Secondly, SGLT2I users had a lower risk of all-cause mortality, as well as cerebrovascular and cardiovascular mortality. All of these were confirmed by univariate Cox regression analysis and competing risk analysis models apart from the association with Alzheimer's disease, which was not significantly reduced in SGLT2I users compared with DPP4I users.
The superior protective effect of DPP4I on dementia compared with other second-line anti-diabetic medication has been demonstrated by multiple studies (29–32). To our knowledge, no study so far has attempted a direct head-to-head between DPP4I and SGLT2I users for new-onset dementia, although a recent case-control study indirectly compared them when considering the risk of dementia associated with different antidiabetic medications (22). They found that while both DPP4I and SGLT2I were associated with lower odds of dementia, the odds ratio for dementia were 0.8 and 0.58 for DPP4I and SGLT2I, respectively. This is consistent with our findings that SGLT2I is superior to DPP4I in lowering dementia risk in diabetic patients. There are several possible explanations for the superior dementia-protective effects of SGLT2I. Firstly, both obesity and diabetes are independent risk factors for dementia due to shared pathophysiological mechanisms such as oxidative stress, inflammation, and insulin resistance (33, 34). Therefore, the increased reduction in weight and HbA1c observed in SGLT2I compared with DPP4I may account for the greater reduction in dementia risk (35, 36). Secondly, animal studies have proposed different neuroprotective mechanisms of SGLT2I and DPP4I which may account for their different efficacy in reducing dementia risk. DPP4I predominantly reduced amyloid deposition, tau phosphorylation, while increased GLP-1 and stromal-derived factor-1 which promoted neurogenesis (16, 37). In contrast, SGLT2I improved brain mitochondrial function, hippocampal synaptic plasticity and inhibited acetylcholinesterase (18, 20, 38).
Alzheimer's disease and diabetes are closely linked by mechanisms such as oxidative stress, amyloid deposition, and tau hyperphosphorylation, so much so that some have termed Alzheimer's as “Type-3 diabetes” (39, 40). There has been growing interest in DPP4I as a potential new therapy against Alzheimer's, with animal studies showing that it reduces amyloid β protein, tau phosphorylation, inflammatory cytokines, and neuronal cell apoptosis in the brain (37, 41–43). This is consistent with clinical studies which found that DPP4I use is associated with the reduced rate of memory decline and increased mini-mental state examination (MMSE) score in Alzheimer's patients compared with metformin use (44, 45). Research on the role of SGLT2I in Alzheimer's disease so far has been based predominantly on animal models, with promising studies suggesting that SGLT2 reduces the amyloid burden, tau pathology, and brain atrophy volume (46). Our finding that SGLT2I use was associated with lower or similar risks of Alzheimer's compared with DPP4I suggested that both may have potential roles as novel therapeutic approaches for Alzheimer's patients and the role of SGLT2I in Alzheimer's should be further explored. The subgroup analysis on patients with both type 2 diabetes and cardiovascular disease showed SGLT2I did not significantly reduce the risk of Alzheimer's disease and Parkinson's disease. This could be a reflection of the equally strong association between cardiovascular disease and such cognitive pathologies (47, 48), as well as their link with type-2 diabetes.
Parkinson's disease is another neurodegenerative disorder closely associated with diabetes, sharing pathophysiological mechanisms such as insulin dysregulation, amyloid deposition, microglial activation, and mitochondrial dysfunction (49). This has been confirmed clinically by several cohort studies which demonstrate type 2 diabetes is associated with an increased risk of Parkinson's (50, 51). Whilst the interest in this is much lower than that of Alzheimer's, several recent studies have suggested beneficial effects of DPP4I in diabetic patients with Parkinson's. A retrospective longitudinal cohort study found a strong protective association between DPP4I and GLP-1 agonist use and Parkinson's disease while another retrospective study found that DPP4I use was associated with increased dopamine transporter availability, slower increase in levodopa dose, and lower risk of levodopa-induced dyskinesia in diabetic patients with Parkinson's disease (52, 53). Our study is the first to compare DPP4I and SGLT2I in their associated Parkinson's risk and demonstrated that SGLT2I has superior protective effects against Parkinson's. Due to the close association and overlapping pathophysiology between Parkinson's disease and dementia with Lewy bodies, it could be inferred that SGLT2I also has superior protective effects against dementia with Lewy Bodies and Parkinson's disease dementia compared with DPP4I (54–56). To date, no study has examined the role of SGLT2I in Parkinson's disease or dementia with Lewy Bodies and our finding suggests that this is an exciting area of research that warrants further investigation.
Several limitations should be noted for the present study. First, given its observational nature, there was inherent information bias due to under-coding, coding errors, and missing data. Additionally, the drug compliance of the patient can only be assessed indirectly through prescription refills, which were ultimately not a direct measurement of drug exposure. Second, residual and post-baseline confounding might be present despite robust propensity-matching, particularly with the unavailability of information on lifestyle cardiovascular risk factors, e.g., smoking. The drug exposure duration among the patients has not been controlled, which might affect their risk against the study outcomes. Finally, the occurrence of cognitive dysfunction outcomes out of the hospital was not accounted for.
The use of SGLT2I is associated with a significantly lower risk of dementia, Parkinson's disease, all-cause mortality, cardiovascular mortality, and cerebrovascular mortality compared with DPP4 use.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
The studies involving human participants were reviewed and approved by the Joint Chinese University of Hong Kong–New Territories East Cluster Clinical Research Ethics Committee. Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
JM and JZ: conception of study and literature search, preparation of figures, study design, data collection, data contribution, statistical analysis, data interpretation, manuscript drafting, and critical revision of manuscript. SL, KL, TLe, OC, ST, AW, TLi, WW, CC, GT, and QZ: conception of study and literature search, study design, data collection, data analysis, data contribution, manuscript drafting, critical revision of manuscript, and study supervision. All authors contributed to the article and approved the submitted version.
This study was supported by the National Natural Science Foundation of China (NSFC) Grant Nos. 72042018, 71972164, and 71672163, in part by the Health and Medical Research Fund Grant (HMRF), the Food and Health Bureau, the Government of the Hong Kong Special Administrative Region No. 16171991, and in part by the Theme-Based Research Scheme of the Research Grants Council of Hong Kong Grant No. T32-102/14N.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcvm.2021.747620/full#supplementary-material
1. Fung ACH, Tse G, Cheng HL, Lau ESH, Luk A, Ozaki R, et al. Depressive symptoms, co-morbidities, and glycemic control in Hong Kong Chinese elderly patients with Type 2 diabetes mellitus. Front Endocrinol. (2018) 9:261. doi: 10.3389/fendo.2018.00261
2. Lee S, Zhou J, Wong WT, Liu T, Wu WKK, Wong ICK, et al. Glycemic and lipid variability for predicting complications and mortality in diabetes mellitus using machine learning. BMC Endocr Disord. (2021) 21:94. doi: 10.1186/s12902-021-00751-4
3. Kovesdy CP, Isaman D, Petruski-Ivleva N, Fried L, Blankenburg M, Gay A, et al. Chronic kidney disease progression among patients with type 2 diabetes identified in US administrative claims: a population cohort study. Clin Kidney J. (2021) 14:1657–64. doi: 10.1093/ckj/sfaa200
4. Folkerts K, Kelly AMB, Petruski-Ivleva N, Fried L, Blankenburg M, Gay A, et al. Cardiovascular and renal outcomes in patients with type-2 diabetes and chronic kidney disease identified in a United States administrative claims database: a population cohort study. Nephron. (2021) 145:342–52. doi: 10.1159/000513782
5. Shin JY, Roh SG, Lee NH, Yang KM. Influence of epidemiologic and patient behavior-related predictors on amputation rates in diabetic patients: systematic review and meta-analysis. Int J Low Extrem Wounds. (2017) 16:14–22. doi: 10.1177/1534734617699318
6. van Sloten TT, Sedaghat S, Carnethon MR, Launer LJ, Stehouwer CDA. Cerebral microvascular complications of type 2 diabetes: stroke, cognitive dysfunction, and depression. Lancet Diabetes Endocrinol. (2020) 8:325–36. doi: 10.1016/S2213-8587(19)30405-X
7. Cukierman T, Gerstein HC, Williamson JD. Cognitive decline and dementia in diabetes–systematic overview of prospective observational studies. Diabetologia. (2005) 48:2460–9. doi: 10.1007/s00125-005-0023-4
8. Ott A, Stolk RP, van Harskamp F, Pols HA, Hofman A, Breteler MM. Diabetes mellitus and the risk of dementia: the Rotterdam Study. Neurology. (1999) 53:1937–42. doi: 10.1212/WNL.53.9.1937
9. Emerging Risk Factors C, Sarwar N, Gao P, Seshasai SR, Gobin R, Kaptoge S, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet. (2010) 375:2215–22. doi: 10.1016/S0140-6736(10)60484-9
10. Kodl CT, Seaquist ER. Cognitive dysfunction and diabetes mellitus. Endocr Rev. (2008) 29:494–511. doi: 10.1210/er.2007-0034
11. Munshi MN. Cognitive dysfunction in older adults with diabetes: what a clinician needs to know. Diabetes Care. (2017) 40:461–7. doi: 10.2337/dc16-1229
12. Crane PK, Walker R, Larson EB. Glucose levels and risk of dementia. N Engl J Med. (2013) 369:1863–4. doi: 10.1056/NEJMc1311765
13. Liu S, Lu Y, Cai X, Cong R, Li J, Jiang H, et al. Glycemic control is related to cognitive dysfunction in elderly people with Type 2 diabetes mellitus in a rural chinese population. Curr Alzheimer Res. (2019) 16:950–62. doi: 10.2174/1567205016666191023110712
14. Kim JY, Ku YS, Kim HJ, Trinh NT, Kim W, Jeong B, et al. Oral diabetes medication and risk of dementia in elderly patients with type 2 diabetes. Diabetes Res Clin Pract. (2019) 154:116–23. doi: 10.1016/j.diabres.2019.07.004
15. Zheng F, Yan L, Yang Z, Zhong B, Xie W. HbA(1c), diabetes and cognitive decline: the english longitudinal study of ageing. Diabetologia. (2018) 61:839–48. doi: 10.1007/s00125-017-4541-7
16. Chen S, Zhou M, Sun J, Guo A, Fernando RL, Chen Y, et al. DPP-4 inhibitor improves learning and memory deficits and AD-like neurodegeneration by modulating the GLP-1 signaling. Neuropharmacology. (2019) 157:107668. doi: 10.1016/j.neuropharm.2019.107668
17. Ide M, Sonoda N, Inoue T, Kimura S, Minami Y, Makimura H, et al. The dipeptidyl peptidase-4 inhibitor, linagliptin, improves cognitive impairment in streptozotocin-induced diabetic mice by inhibiting oxidative stress and microglial activation. PLoS One. (2020) 15:e0228750. doi: 10.1371/journal.pone.0228750
18. Lin B, Koibuchi N, Hasegawa Y, Sueta D, Toyama K, Uekawa K, et al. Glycemic control with empagliflozin, a novel selective SGLT2 inhibitor, ameliorates cardiovascular injury and cognitive dysfunction in obese and type 2 diabetic mice. Cardiovasc Diabetol. (2014) 13:148. doi: 10.1186/s12933-014-0148-1
19. Wang S, Liu Y, Zhang H, Guo Y, Li M, Gao W, et al. Effects of an SGLT2 inhibitor on cognition in diabetes involving amelioration of deep cortical cerebral blood flow autoregulation and pericyte function. Alzheimer's Dementia. (2020) 16:e037056. doi: 10.1002/alz.037056
20. Sa-Nguanmoo P, Tanajak P, Kerdphoo S, Jaiwongkam T, Pratchayasakul W, Chattipakorn N, et al. SGLT2-inhibitor and DPP-4 inhibitor improve brain function via attenuating mitochondrial dysfunction, insulin resistance, inflammation, and apoptosis in HFD-induced obese rats. Toxicol Appl Pharmacol. (2017) 333:43–50. doi: 10.1016/j.taap.2017.08.005
21. Perna S, Mainardi M, Astrone P, Gozzer C, Biava A, Bacchio R, et al. 12-month effects of incretins versus SGLT2-Inhibitors on cognitive performance and metabolic profile. a randomized clinical trial in the elderly with Type-2 diabetes mellitus. Clin Pharmacol. (2018) 10:141–51. doi: 10.2147/CPAA.S164785
22. Wium-Andersen IK, Osler M, Jørgensen MB, Rungby J, Wium-Andersen MK. Antidiabetic medication and risk of dementia in patients with type 2 diabetes: a nested case-control study. Eur J Endocrinol. (2019) 181:499–507. doi: 10.1530/EJE-19-0259
23. Lee S, Liu T, Zhou J, Zhang Q, Wong WT, Tse G. Predictions of diabetes complications and mortality using hba1c variability: a 10-year observational cohort study. Acta Diabetol. (2021) 58:171–80. doi: 10.1007/s00592-020-01605-6
24. Zhou J, Lee S, Guo CL, Chang C, Liu T, Leung KSK, et al. Anticoagulant or antiplatelet use and severe COVID-19 disease: a propensity score-matched territory-wide study. Pharmacol Res. (2021) 165:105473. doi: 10.1016/j.phrs.2021.105473
25. Zhou J, Wang X, Lee S, Wu WKK, Cheung BMY, Zhang Q, et al. Proton pump inhibitor or famotidine use and severe COVID-19 disease: a propensity score-matched territory-wide study. Gut. (2020) 70:gutjnl-2020-323668. doi: 10.1136/gutjnl-2020-323668
26. Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. (2011) 46:399–424. doi: 10.1080/00273171.2011.568786
27. Austin PC, Stuart EA. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat Med. (2015) 34:3661–79. doi: 10.1002/sim.6607
28. Schneeweiss S, Rassen JA, Glynn RJ, Avorn J, Mogun H, Brookhart MA. High-dimensional propensity score adjustment in studies of treatment effects using health care claims data. Epidemiology. (2009) 20:512–22. doi: 10.1097/EDE.0b013e3181a663cc
29. Kim YG, Jeon JY, Kim HJ, Kim DJ, Lee KW, Moon SY, et al. Risk of dementia in older patients with type 2 diabetes on dipeptidyl-peptidase IV inhibitors versus sulfonylureas: a real-world population-based cohort study. J Clin Med. (2018) 8:28. doi: 10.3390/jcm8010028
30. Kim WJ, Noh JH, Han K, Park CY. The association between second-line oral antihyperglycemic medication on types of dementia in Type 2 diabetes: a nationwide real-world longitudinal study. J Alzheimers Dis. (2021) 81:1263–72. doi: 10.3233/JAD-201535
31. Rizzo MR, Barbieri M, Boccardi V, Angellotti E, Marfella R, Paolisso G. Dipeptidyl peptidase-4 inhibitors have protective effect on cognitive impairment in aged diabetic patients with mild cognitive impairment. J Gerontol A Biol Sci Med Sci. (2014) 69:1122–31. doi: 10.1093/gerona/glu032
32. Zhou JB, Tang X, Han M, Yang J, Simó R. Impact of antidiabetic agents on dementia risk: a Bayesian network meta-analysis. Metabolism. (2020) 109:154265. doi: 10.1016/j.metabol.2020.154265
33. Baglietto-Vargas D, Shi J, Yaeger DM, Ager R, LaFerla FM. Diabetes and Alzheimer's disease crosstalk. Neurosci Biobehav Rev. (2016) 64:272–87. doi: 10.1016/j.neubiorev.2016.03.005
34. Kivipelto M, Ngandu T, Fratiglioni L, Viitanen M, Kåreholt I, Winblad B, et al. Obesity and vascular risk factors at midlife and the risk of dementia and Alzheimer disease. Arch Neurol. (2005) 62:1556–60. doi: 10.1001/archneur.62.10.1556
35. Bailey CJ, Del Prato S, Wei C, Reyner D, Saraiva G. Durability of glycaemic control with dapagliflozin, an SGLT2 inhibitor, compared with saxagliptin, a DPP4 inhibitor, in patients with inadequately controlled type 2 diabetes. Diabetes Obes Metab. (2019) 21:2564–9. doi: 10.1111/dom.13841
36. Pinto LC, Rados DV, Remonti LR, Kramer CK, Leitao CB, Gross JL. Efficacy of SGLT2 inhibitors in glycemic control, weight loss and blood pressure reduction: a systematic review and meta-analysis. Diabetol Metab Syndr. (2015) 7(Suppl. 1):A58. doi: 10.1186/1758-5996-7-S1-A58
37. Kosaraju J, Gali CC, Khatwal RB, Dubala A, Chinni S, Holsinger RM, et al. Saxagliptin: a dipeptidyl peptidase-4 inhibitor ameliorates streptozotocin induced Alzheimer's disease. Neuropharmacology. (2013) 72:291–300. doi: 10.1016/j.neuropharm.2013.04.008
38. Shaikh S, Rizvi SM, Shakil S, Riyaz S, Biswas D, Jahan R. Forxiga (dapagliflozin): plausible role in the treatment of diabetes-associated neurological disorders. Biotechnol Appl Biochem. (2016) 63:145–50. doi: 10.1002/bab.1319
39. Nguyen TT, Ta QTH, Nguyen TKO, Nguyen TTD, Giau VV. Type 3 diabetes and its role implications in Alzheimer's disease. Int J Mol Sci. (2020) 21:3165. doi: 10.3390/ijms21093165
40. Kandimalla R, Thirumala V, Reddy PH. Is Alzheimer's disease a Type 3 diabetes? A critical appraisal. Biochim Biophys Acta Mol Basis Dis. (2017) 1863:1078–89. doi: 10.1016/j.bbadis.2016.08.018
41. Kosaraju J, Murthy V, Khatwal RB, Dubala A, Chinni S, Muthureddy Nataraj SK, et al. Vildagliptin: an anti-diabetes agent ameliorates cognitive deficits and pathology observed in streptozotocin-induced Alzheimer's disease. J Pharm Pharmacol. (2013) 65:1773–84. doi: 10.1111/jphp.12148
42. D'Amico M, Di Filippo C, Marfella R, Abbatecola AM, Ferraraccio F, Rossi F, et al. Long-term inhibition of dipeptidyl peptidase-4 in Alzheimer's prone mice. Exp Gerontol. (2010) 45:202–7. doi: 10.1016/j.exger.2009.12.004
43. Kosaraju J, Holsinger RMD, Guo L, Tam KY. Linagliptin, a dipeptidyl Peptidase-4 inhibitor, mitigates cognitive deficits and pathology in the 3xTg-AD mouse model of Alzheimer's disease. Mol Neurobiol. (2017) 54:6074–84. doi: 10.1007/s12035-016-0125-7
44. Isik AT, Soysal P, Yay A, Usarel C. The effects of sitagliptin, a DPP-4 inhibitor, on cognitive functions in elderly diabetic patients with or without Alzheimer's disease. Diabetes Res Clin Pract. (2017) 123:192–8. doi: 10.1016/j.diabres.2016.12.010
45. Wu CY, Ouk M, Wong YY, Anita NZ, Edwards JD, Yang P, et al. Relationships between memory decline and the use of metformin or DPP4 inhibitors in people with type 2 diabetes with normal cognition or Alzheimer's disease, and the role APOE carrier status. Alzheimers Dement. (2020) 16:1663–73. doi: 10.1002/alz.12161
46. Wiciński M, Wódkiewicz E, Górski K, Walczak M, Malinowski B. Perspective of SGLT2 inhibition in treatment of conditions connected to neuronal loss: focus on Alzheimer's disease and ischemia-related brain injury. Pharmaceuticals. (2020) 13:379. doi: 10.3390/ph13110379
47. Potashkin J, Huang X, Becker C, Chen H, Foltynie T, Marras C. Understanding the links between cardiovascular disease and Parkinson's disease. Mov Disord. (2020) 35:55–74. doi: 10.1002/mds.27836
48. Stampfer MJ. Cardiovascular disease and Alzheimer's disease: common links. J Intern Med. (2006) 260:211–23. doi: 10.1111/j.1365-2796.2006.01687.x
49. Cheong JLY, de Pablo-Fernandez E, Foltynie T, Noyce AJ. The Association between Type 2 diabetes mellitus and Parkinson's disease. J Parkinsons Dis. (2020) 10:775–89. doi: 10.3233/JPD-191900
50. Hu G, Jousilahti P, Bidel S, Antikainen R, Tuomilehto J. Type 2 diabetes and the risk of Parkinson's disease. Diabetes Care. (2007) 30:842–7. doi: 10.2337/dc06-2011
51. Xu Q, Park Y, Huang X, Hollenbeck A, Blair A, Schatzkin A, et al. Diabetes and risk of Parkinson's disease. Diabetes Care. (2011) 34:910–5. doi: 10.2337/dc10-1922
52. Brauer R, Wei L, Ma T, Athauda D, Girges C, Vijiaratnam N, et al. Diabetes medications and risk of Parkinson's disease: a cohort study of patients with diabetes. Brain. (2020) 143:3067–76. doi: 10.1093/brain/awaa262
53. Jeong SH, Chung SJ, Yoo HS, Hong N, Jung JH, Baik K, et al. Beneficial effects of dipeptidyl peptidase-4 inhibitors in diabetic Parkinson's disease. Brain. (2021) 144:1127–37. doi: 10.1093/brain/awab015
54. Aarsland D. Cognitive impairment in Parkinson's disease and dementia with Lewy bodies. Parkinsonism Relat Disord. (2016) 22(Suppl. 1):S144–8. doi: 10.1016/j.parkreldis.2015.09.034
55. McKeith IG, Mosimann UP. Dementia with Lewy bodies and Parkinson's disease. Parkinsonism Relat Disord. (2004) 10(Suppl. 1):S15–8. doi: 10.1016/j.parkreldis.2003.12.005
Keywords: SGLT2, SGLT2 (sodium-glucose cotransporter 2) inhibitor, DPP4, DPP4 inhibitor, dementia, cognitive dysfunction, Alzheimer's disease, Parkinson's disease
Citation: Mui JV, Zhou J, Lee S, Leung KSK, Lee TTL, Chou OHI, Tsang SL, Wai AKC, Liu T, Wong WT, Chang C, Tse G and Zhang Q (2021) Sodium-Glucose Cotransporter 2 (SGLT2) Inhibitors vs. Dipeptidyl Peptidase-4 (DPP4) Inhibitors for New-Onset Dementia: A Propensity Score-Matched Population-Based Study With Competing Risk Analysis. Front. Cardiovasc. Med. 8:747620. doi: 10.3389/fcvm.2021.747620
Received: 26 July 2021; Accepted: 17 September 2021;
Published: 21 October 2021.
Edited by:
Leonardo Roever, Federal University of Uberlandia, BrazilReviewed by:
Jingyi Ren, China-Japan Friendship Hospital, ChinaCopyright © 2021 Mui, Zhou, Lee, Leung, Lee, Chou, Tsang, Wai, Liu, Wong, Chang, Tse and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gary Tse, Z2FyeXRzZTg2QGdtYWlsLmNvbQ==; Qingpeng Zhang, cWluZ3BlbmcuemhhbmdAY2l0eXUuZWR1Lmhr
†These authors share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.