- 1Shenzhen Institute of Dermatology, Shenzhen Center for Chronic Disease Control, Shenzhen, China
- 2NHC Key Laboratory of Systems Biology of Pathogens, National Institute of Pathogen Biology, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China
- 3National Institute of Pathogen Biology and Key Laboratory of Respiratory Disease Pathogenomics, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China
- 4Key Laboratory of Pathogen Infection Prevention and Control (Ministry of Education), State Key Laboratory of Respiratory Health and Multimorbidity, National Institute of Pathogen Biology, Chinese Academy of Medical Sciences& Peking Union Medical College, Beijing, China
Introduction: This study aims to determine the etiological, sociodemographic, and clinical characteristics of STIs, and the level of resistance in M. genitalium in Shenzhen, a representative first-tier city of southern China.
Methods: A multicenter cross-sectional study was conducted and 7886 sexually active participants attending STI-related departments were involved from 22 hospitals. Nine STI-related organisms including N. gonorrhoeae, C. trachomatis, T. vaginalis, M. genitalium, HSV-1, HSV-2, M. hominis, U. parvum, and U. urealyticum were screened.
Results: Being single or divorced was associated with increased detection of N. gonorrhoeae, C. trachomatis, M. genitalium, HSV-1, HSV-2 and M. hominis. Lower education level was associated with increased detection of C. trachomatis, HSV-2 and M. hominis. No insurance coverage was an independent risk factor for T. vaginalis, M. hominis and U. parvum positivity. Three resistance-determining regions related to macrolide and fluoroquinolone were sequenced in 154 M. genitalium positive samples, among which 90.3% harbored mutations related to macrolide or fluroquinolone resistance and 67.5% were multidrug-resistant M. genitalium. A2072G in 23S rRNA and Ser83Ile in parC were the most common mutations. M. hominis was associated with manifestations of bacterial vaginosis in female and epididymitis in male.
Conclusions: Single or divorced individuals, those with lower education level and individuals without insurance are higher-risk key populations for STIs. The prevalence of antimicrobial-resistant M. genitalium in Shenzhen is high. Detection of M. hominis increased significantly with lower education level and no health insurance coverage, and it is associated with bacterial vaginosis or epididymitis, indicating that M. hominis deserves further attention.
1 Introduction
The high burden of sexually transmitted infections (STIs) remains a current public health issue worldwide. In 2019, there were 769.85 million incident cases of STIs globally (Zheng et al., 2022), although less likely to be fatal, they seriously affect the quality of life and lead to considerable neonatal deaths and infertility. Besides gonorrhea and chlamydia, infections of antimicrobial-resistant Mycoplasma genitalium and herpes simplex virus (HSV) are also emerging global health problem. Mycoplasma hominis, Ureaplasma parvum and Ureaplasma urealyticum are microorganisms frequently found in human urogenital tract and their roles in causing specific symptoms of STIs remains controversial (Horner et al., 2018).
The prevalence of STI-related organisms varies globally, demonstrating the regional differences in socioeconomics, demographics, sexual orientation and cultural factors (World Health Organization, 2016; Van Gerwen et al., 2022). To date, studies in China mainly described the prevalence of common STI pathogens with limited emphasis on their correlation with sociodemographic characteristics and clinical presentations (Yin et al., 2018; Li et al., 2020; Han et al., 2021). The absence of these data undermines the formulation of region-specific, evidence-based interventions and policies.
In this study, a large-scale screening of STI-related organisms was performed in Shenzhen, a representative first-tier city of southern China. The participants included healthy and symptomatic individuals of both genders with varied symptoms of STI. Nine STI-related organisms (N. gonorrhoeae, C. trachomatis, Trichomonas vaginalis, M. genitalium, HSV-1, HSV-2, M. hominis, U. parvum, U. urealyticum) were detected using a multiplex PCR coupled with mass spectrum method, STI-MS (sexually transmitted infection-mass spectrometry) (Xiu et al., 2019). M. genitalium-positive samples underwent screening to determine the presence of mutations associated with macrolide and fluoroquinolone resistance. The aim of the study was to determine the sociodemographic and clinical features of higher-risk key populations with different STIs, and to describe the level of resistance in M. genitalium. The findings can contribute to the formulation of screening strategies based on key populations and specific symptoms.
2 Materials and methods
2.1 Study design and population
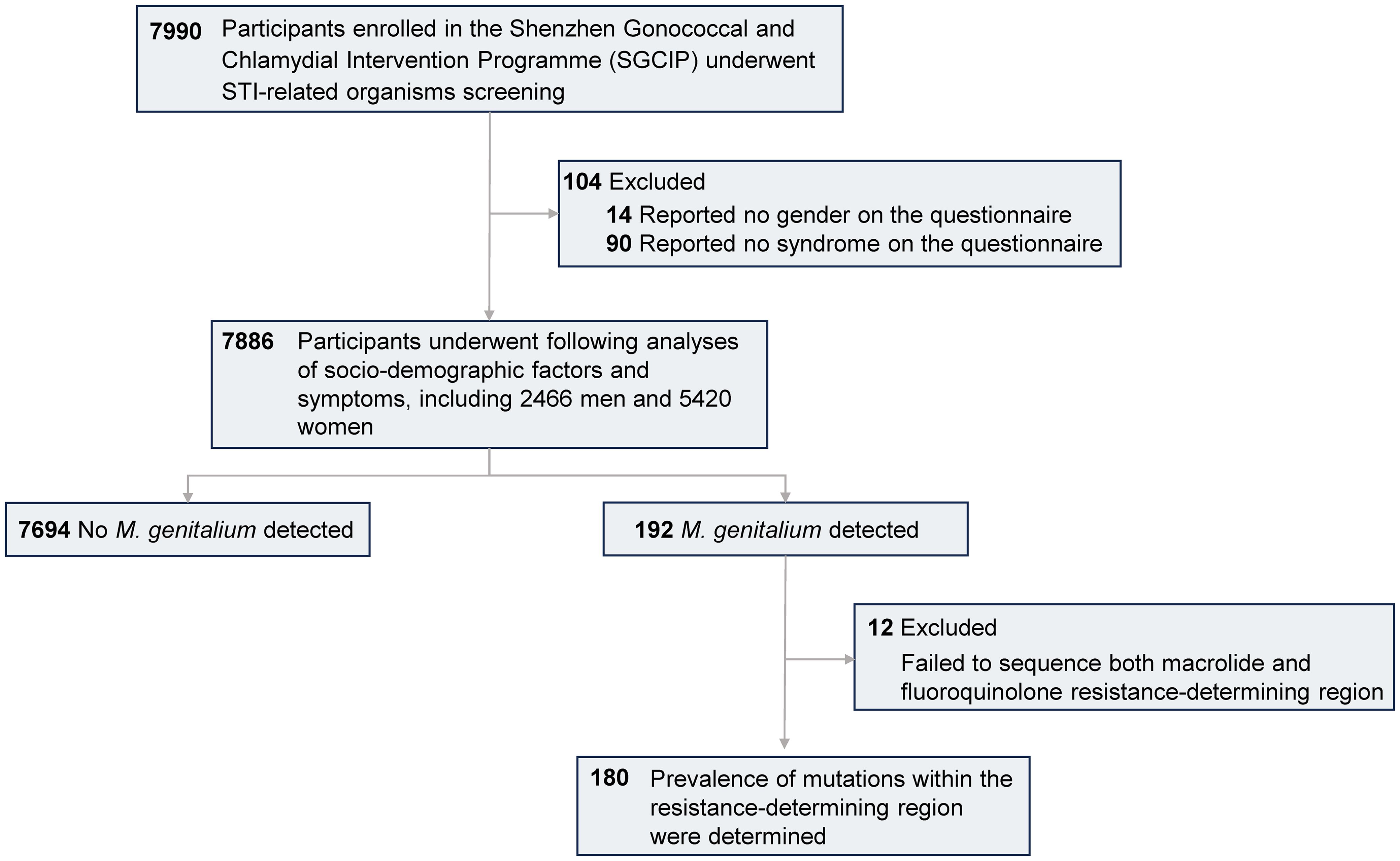
This cross-sectional study was conducted among subjects enrolled in the Shenzhen Gonococcal and Chlamydial Intervention Programme (SGCIP). Twenty-two hospitals situated across six districts in Shenzhen were included in the programme (Supplementary Table 1). Individuals attending STI-related departments including dermatology, urology, gynecology and obstetrics in 2018 were evaluated. The inclusion criteria were as follows: aged ≥ 16 years, sexually active, and had not used antibiotics within the past two weeks. All participants completed a questionnaire regarding their sociodemographic characteristics. Urethral swabs were collected from males and cervical swabs were collected from females. Clinicians conducted physical examinations and documented the symptoms. A total of 7990 participants enrolled in the SGCIP underwent STI-related organisms screening and 104 participants who failed to correctly fill the gender and symptoms on the questionnaire were excluded. The remaining 2466 men and 5420 women were eligible (Figure 1). Participants without any of the above symptoms were defined as controls.
This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.
2.2 Sample preparation and mass spectrum test
Swabs were rotated in 500μL phosphate-buffered saline and 200μL were used for DNA extraction with MagNA Pure LC 2.0 instrument and MagNA Pure LC Total Nucleic Acid Isolation Kit (Roche Diagnostics, Mannheim, Germany). N. gonorrhoeae, C. trachomatis, T. vaginalis, M. genitalium, HSV-1, HSV-2, M. hominis, U. parvum, and U. urealyticum were simultaneously screened using the STI-MS method (Xiu et al., 2019).
2.3 Sequencing of the macrolide and fluoroquinolone resistance-determining region
Sequences of the macrolide RDR (23S rRNA) and fluoroquinolone RDR (parC and gyrA gene) were amplified using nested PCR from the M. genitalium-positive samples. Sanger sequencing was performed on amplicons (Tsingke Biotechnology, Beijing, China). PCR reactions were conducted with the Roche FastStart High Fidelity PCR System (Roche Diagnostics). Primers were listed in Supplementary Table 2.
2.4 Statistical analysis
The 95% confidence intervals for the detection rate of each STI-related organism were calculated using the VassarStats website (http://vassarstats.net/index.html). The chi-square tests were used to compare the difference in the detection rate of each STI-related organism in the male and female populations. Sociodemographic factors associated with the detection of STI-related organism and antimicrobial-resistant M. genitalium were first investigated using the univariable logistic regression analyses, and factors with a significance level of P < 0.1 were included in the multivariable model. All the nine STI-related organisms detected in the study were included in the multivariable logistic regression model to investigate their associations with the urogenital symptoms of male and female participants respectively. The chi-squared tests and logistic regression analyses were performed using SPSS 26.0
3 Results
3.1 Sociodemographic and clinical characteristics
Of the 7886 eligible participants, 5420 (68.7%) were women and the median age was 31 years (interquartile range, 27-36). The majority of participants were married (74.5%), with the most common educational attainment being college or graduate level (38.2%). Most participants had health insurance (61.4%), with 16.6% reporting a monthly income of less than US$500. 96.7% of the participants were heterosexual and 36% reported having one or more casual sexual partners in the past three months. Other sociodemographic characteristics, including residency, living time in Shenzhen, and occupation, are listed in Supplementary Table 3.
Among eligible participants, 1290 males and 3454 females had at least one clinical sign or symptom. For male, urethral burning or irritation was the most common symptom (25.1%), while for female, the most common symptom was abnormal vaginal discharge (53.8%). The statistics of clinical signs and symptoms were listed in Supplementary Table 4.
3.2 Detection of STI-related organisms
According to the results of STI-MS, among the 7886 eligible participants, 1095 (44.4%) males and 3572 (65.9%) females were positive for at least one microorganism. C. trachomatis was the most common detected STI pathogen, with the detection rate of 11.6% in male and 10.0% in female; U. parvum was the most frequently detected STI-related organism, with the detection rate of 17.3% in male and 51.2% in female. Among STI pathogens, the prevalence of N. gonorrhoeae and C. trachomatis of men was significantly higher than women, but for T. vaginalis, the prevalence of women was higher. The detection rates of M. hominis, U. parvum and U. urealyticum in women were significantly higher than men (Supplementary Table 5). The coinfection rate of six STI pathogens was 4.1% (100/2466) for male and 2.6% (139/5420) for female. Taking M. hominis, U. parvum and U. urealyticum into account, the coinfection rate was 14.4% (355/2466) and 23.2% (1255/5420) for male and female respectively.
3.3 Sociodemographic characteristics associated with detection of STI-related organisms
Univariable analyses showed that N. gonorrhoeae and C. trachomatis infections were found more frequently in males than females, while for T. vaginalis, H. hominis, U. parvum, and U. urealyticum, the situation was reversed. No health insurance coverage and marital status as single or divorced emerged as risk factors for a broader spectrum of pathogen infections. Higher level of education, income and older age were primary protective factors. Casual sex partners, mobility of population (those without residency or with short living time) were associated with increased organism positivity. Compared to unemployment, government staff and housewife/househusband were linked to decreased organism positivity (Supplementary Tables 6-8).
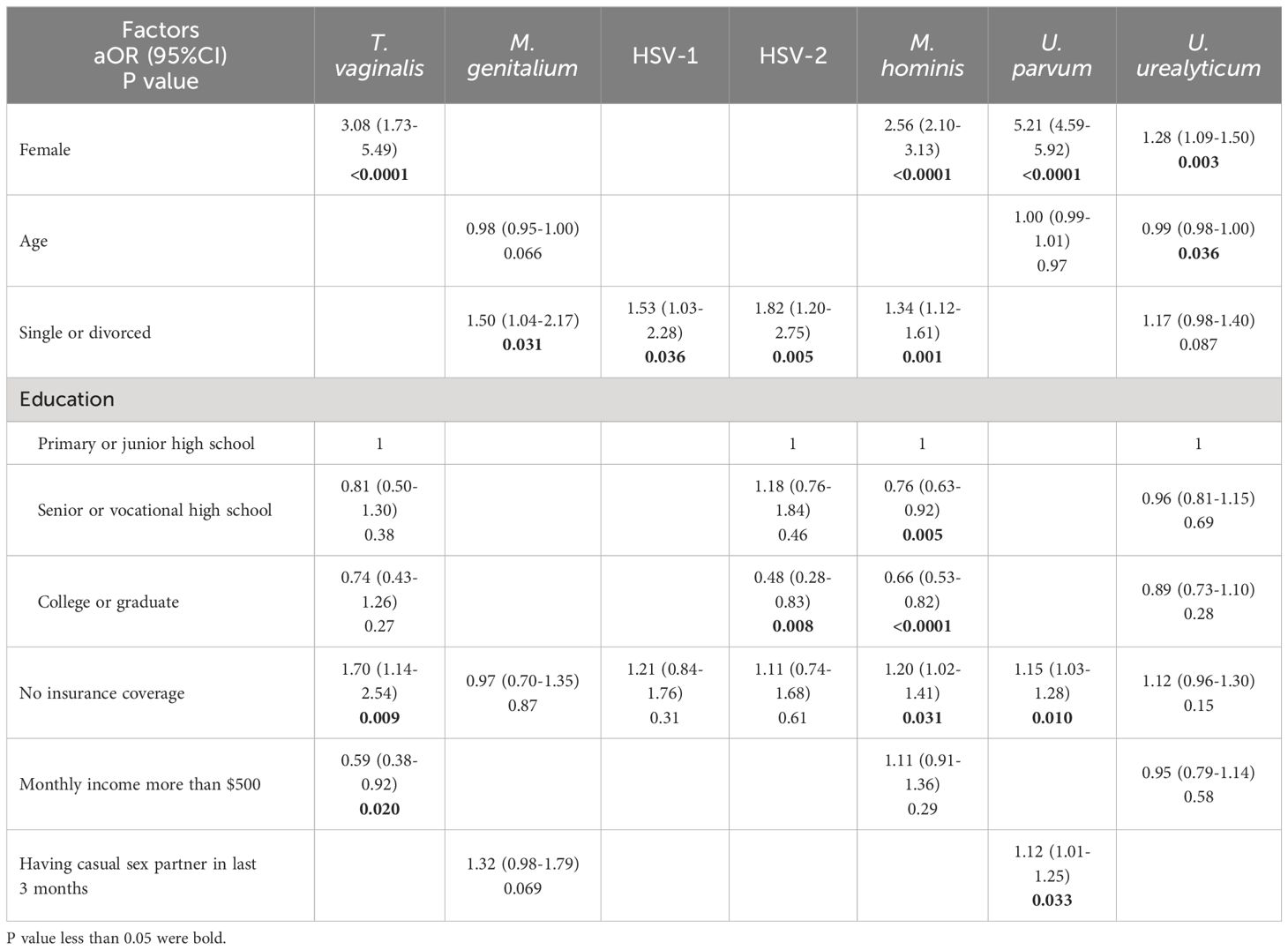
Results of multivariate analyses of sociodemographic factors associated with N. gonorrhoeae and C. trachomatis infections were shown in Supplementary Table 9, and the findings aligned with previous studies conducted on the same population (Ning et al., 2022; Wang et al., 2022). For the other seven STI-related organisms, T. vaginalis, M. hominis, U. parvum and U. urealyticum were found more frequently among females. Being single or divorced was an important factor related to infection, which was associated with increased detection of M. genitalium (aOR=1.50, 95%CI: 1.04-2.17), HSV-1 (aOR=1.53, 95%CI: 1.03-2.28), HSV-2 (aOR=1.82, 95%CI: 1.20-2.75) and M. hominis (aOR=1.34, 95%CI: 1.12-1.61). Elevated educational level was significantly correlated with reduced odds of HSV-2 and M. hominis infection. No insurance coverage was an independent risk factor for T. vaginalis, M. hominis and U. parvum positivity. Moreover, monthly income more than $500 was associated with decreased detection of T. vaginalis (aOR=0.59, 95%CI: 0.38-0.92). The prevalence of U. parvum was higher in participants with casual sex partner in last 3 months (Table 1). When other sociodemographic factors were included, the association of occupation, mobility of population and sexual orientation with infections weakened (Supplementary Table 10).
3.4 Detection of macrolide and fluoroquinolone resistance-associated mutations in M. genitalium-positive samples
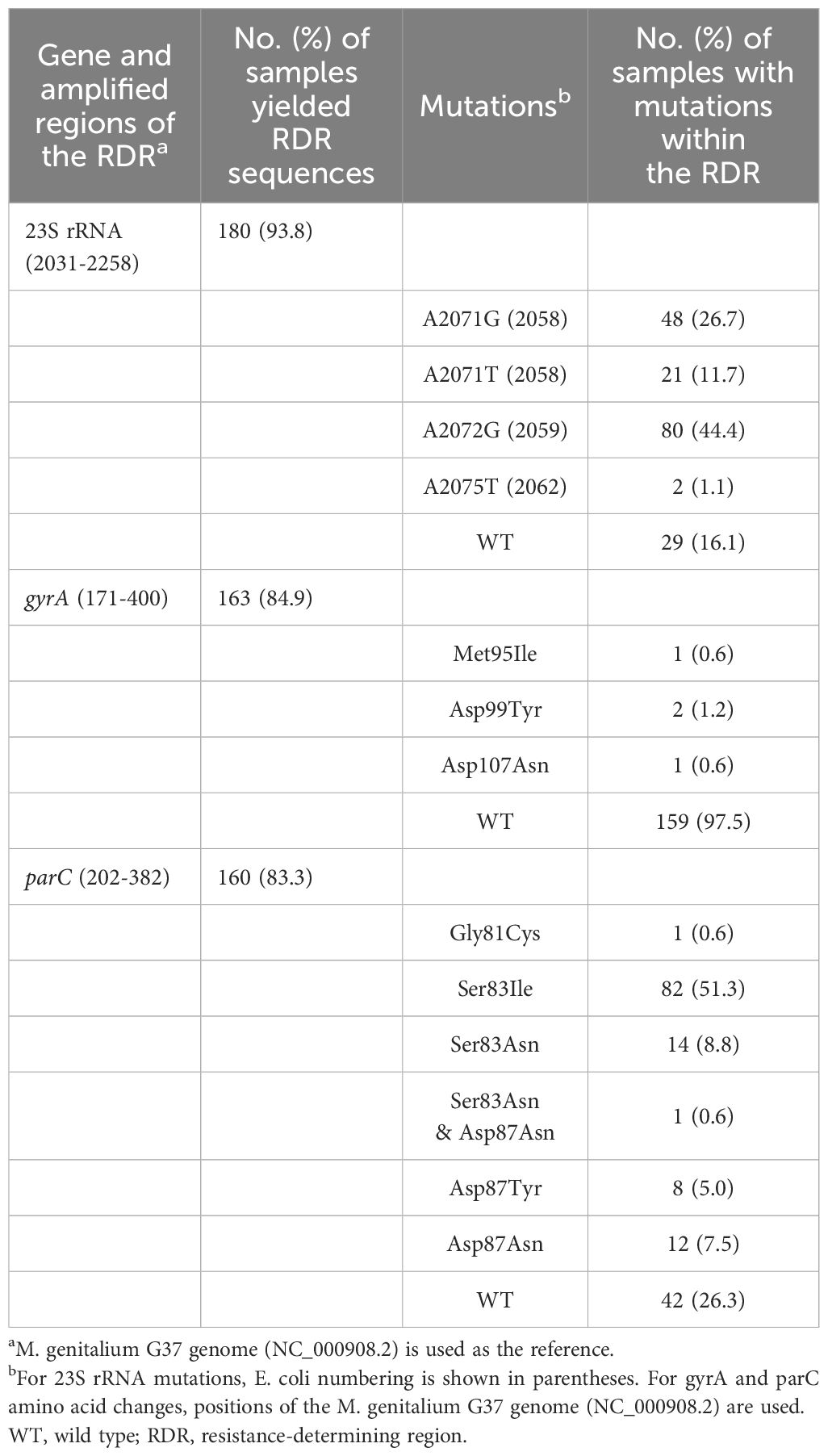
Among the 192 M. genitalium-positive samples, successful amplification and sequencing of the macrolide RDR were achieved in 180 (93.8%) samples (Figure 1), with mutations in 83.9% (151/180) of these samples. Four mutations were identified and A2072G was the most common (44.4%, 80/180). Other mutations included A2071G (n=48), A2071T (n=21) and A2075T (n=2) (Table 2).
Successful amplification and sequencing of the parC gene were achieved in 160 (83.3%) M. genitalium-positive samples, with missense mutations in 73.8% (118/160) of these samples. Five missense mutations were detected and Ser83Ile was the most common, with a detection rate of 51.3% (82/160). Other missense mutations included Ser83Asn (n=14), Asp87Asn (n=12), Asp87Tyr (n=8) and Gly81Cys (n=1). One sample harbored a double parC gene missense mutation (Ser83Asn and Asp87Asn) (Table 2).
For the gyrA gene, amplification and sequencing were successful in 163 (84.9%) M. genitalium-positive samples, with missense mutations in 2.5% (4/163) of these samples, including Asp99Tyr (n=2), Met95Ile (n=1) and Asp107Asn (n=1). All the samples harboring missense mutations in the gyrA gene exhibited missense mutations in the parC gene (Table 2).
Successful amplification and sequencing of the three RDRs were achieved in 154 samples. Among those samples, 16.2% (25/154) were macrolide-resistant M. genitalium infection (with mutations solely in the macrolide RDR); 6.5% (10/154) were fluoroquinolone-resistant M. genitalium infection (with mutations solely in the fluoroquinolone RDR); 67.5% (104/154) were multidrug-resistant M. genitalium infection; 9.7% (15/154) exhibited wild-type sequences of the three RDRs.
The prevalence of macrolide or fluoroquinolone-resistant M. genitalium infection and associations with sociodemographic factors, co-infecting organisms, and urogenital symptoms were determined with univariate and multivariate analysis. Univariate regression analysis showed participants lacking Shenzhen residency [OR=2.75 (1.08-6.99), p=0.034] and without insurance coverage [OR=2.41 (1.14-5.10), p=0.021] exhibited a higher rate of fluoroquinolone-resistant M. genitalium infection, while the multivariate analysis included Shenzhen residency and insurance showed the two factors were not independent for increased fluoroquinolone-resistant M. genitalium prevalence (p=0.138 and 0.087 respectively). No specific co-infecting organisms and urogenital symptoms were significantly associated with macrolide or fluoroquinolone-resistant M. genitalium infection.
3.5 Urogenital symptoms associated with STI-related organisms
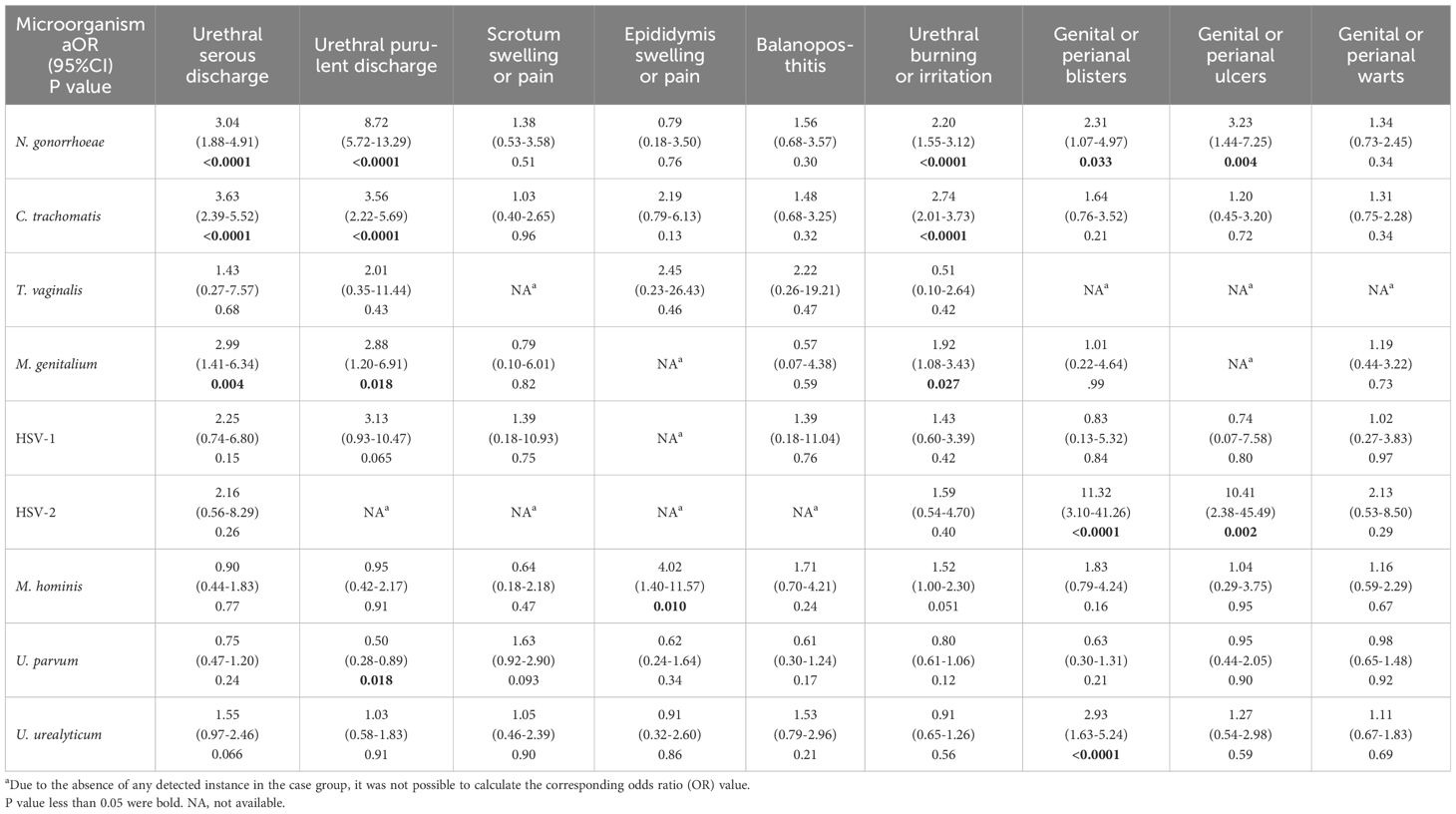
As accepted causes of male urethritis, N. gonorrhoeae, C. trachomatis, and M. genitalium were associated with urethral serous discharge, urethral purulent discharge, and urethral burning or irritation. HSV-2 infection in male was strongly associated with genital or perianal blisters (aOR=11.32, 95%CI: 3.10–41.26) and ulcers (aOR=10.41, 95%CI: 2.38–45.49), indicating in males with genital lesions, if tests for pathogens that cause such lesions, including Treponema pallidum, are negative, HSV should be considered the primary causative organism, necessitating antiviral treatment. Furthermore, the detection of M. hominis, U. parvum, and U. urealyticum was found to be associated with certain urogenital symptoms. In this study, epididymis swelling or pain was solely associated with M. hominis (aOR=4.02, 95%CI: 1.40–11.57). U. urealyticum was observed to be associated with genital or perianal blisters (aOR=2.93, 95%CI: 1.63–5.24), whereas U. parvum was associated with a decreased risk of urethral purulent discharge (aOR=0.5, 95%CI: 0.28–0.89) (Table 3).
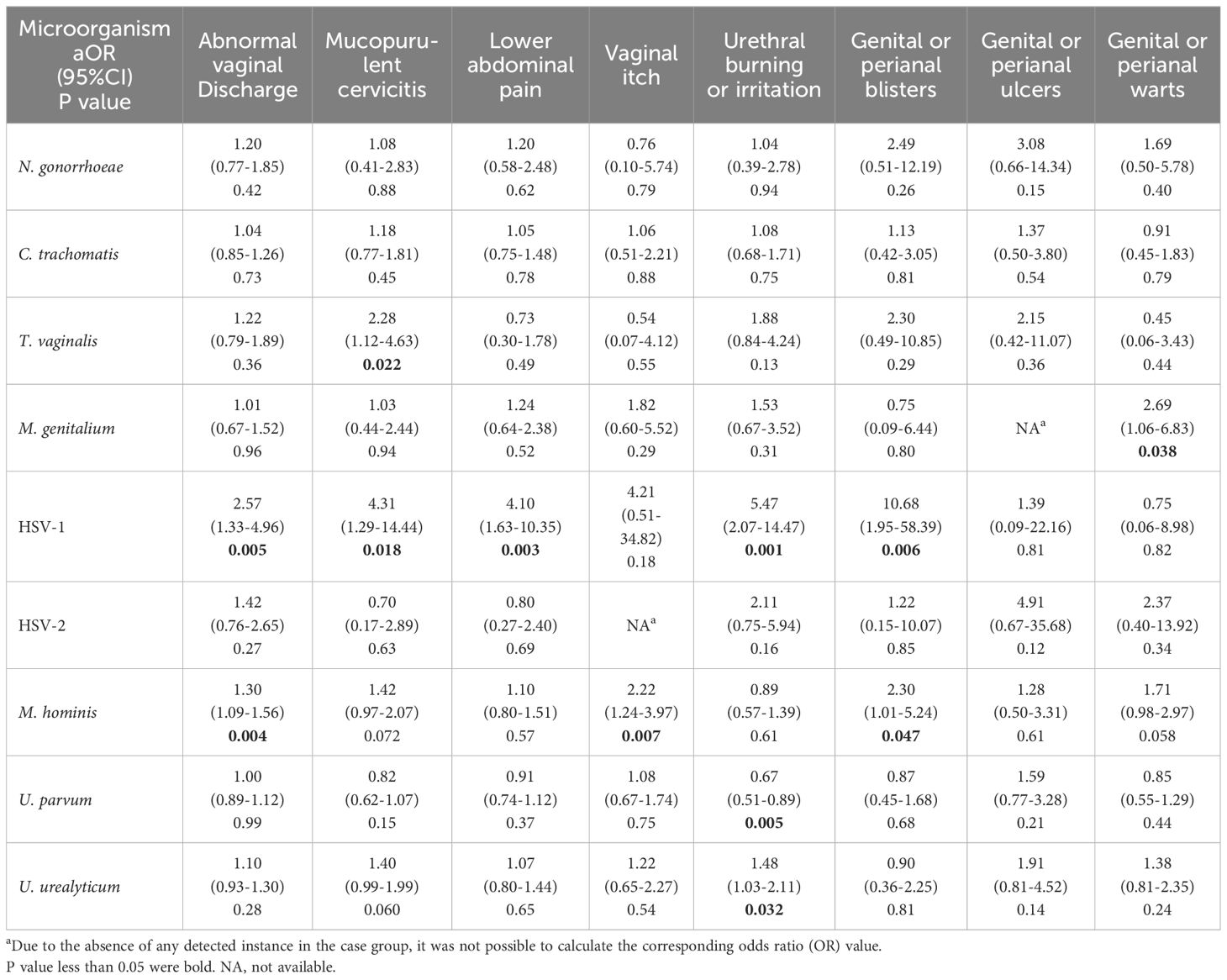
Females demonstrated a different pattern in symptom-organism associations compared to males. Infections of causative agents of male urethritis (N. gonorrhoeae, C. trachomatis, and M. genitalium) were not linked to urethritis symptoms in female, indicating most infections of theses pathogens were asymptomatic. We found that HSV-1 and M. hominis were more frequently detected in female participants with various symptoms. Specifically, HSV-1 was associated with abnormal vaginal discharge (aOR=2.57, 95%CI: 1.33-4.96), mucopurulent cervicitis (aOR=4.31, 95%CI: 1.29-14.44), lower abdominal pain (aOR=4.10, 95%CI: 1.63-10.35), urethral burning or irritation (aOR=5.47, 95%CI: 2.07-14.47), and genital or perianal blisters (aOR=10.68, 95%CI: 1.95-58.39); M. hominis was correlated with abnormal vaginal discharge (aOR=1.30, 95%CI: 1.09-1.56), vaginal itch (aOR=2.22, 95%CI: 1.24-3.97), and genital or perianal blisters (aOR=2.30, 95%CI: 1.01-5.24). U. parvum was considered to be a protective factor of urethral burning or irritation with 0.67-fold decreased risk (95%CI: 0.5-0.89). Detailed results of symptom-organism associations of female were shown in Table 4.
Detailed results of symptom-organism associations were shown in Tables 3 and 4. Proportions of STI-related organisms in cases and controls were shown in Supplementary Tables 11 and 12.
4 Discussion
Given that different sexually transmitted organisms share the same route of transmission and their infections have similar symptoms. Accurate identification of the causative agents, rather than relying on empiric treatment, can promote responsible antimicrobial use and stewardship. Therefore, targeted screening within higher-risk key population groups can mitigate the socioeconomic burden, and early diagnosis can alleviate patients’ anxiety with multiple clinic visits. Currently, there is a lack of large-scale research in China investigating high-risk populations for diverse STI-related organism infections and their sociodemographic features. Here, we revealed that unmarried status, lower education level, and absence of health insurance coverage were associated with an elevated positivity rate for multiple STI-related organisms. These organisms include not only well-established pathogens like N. gonorrhoeae, C. trachomatis, and M. genitalium, but also HSVs, which are rarely selected as routine screening targets (Karellis et al., 2022; Pereyre et al., 2022) and organisms with controversial pathogenicity, such as M. hominis (Taylor-Robinson, 2017; Hughes and Saunders, 2018). Previous studies on the prevalence of sexually transmitted pathogens have primarily been country-based and have found that low- and middle-income countries bear a higher disease burden (Rowley et al., 2019). Shenzhen, where this study was conducted, is located in one of China’s most economically developed regions. Despite this, significant differences were observed in the detection rates of various STI-related microorganisms. These findings indicated that individuals with lower incomes and education levels in large cities also required significant attention and medical resources. The higher prevalence of STIs among single or divorced individuals and those with lower education levels can be attributed to a combination of behavioral and social factors (Nahmias and Nahmias, 2011). The behavioral factors include sexual practices, contraceptive use and health seeking behavior (Pouget, 2017), while the social factors include socioeconomic status, social network and the availability to knowledge about STIs (Wayal et al., 2019).
The prevalence of bacterial sexually transmitted pathogens among women of reproductive age exhibits geographical variability across different regions of the world. Several studies with similar populations to ours (sexually active adult women attending STI clinics) employed molecular tests to obtain and the detection rates of STI pathogens. In an Australian study, N. gonorrhoeae and C. trachomatis were detected in 1% and 8% of the enrolled women, and the prevalence of M. genitalium and T. vaginalis were 6% and 0.87% respectively (Latimer et al., 2022). In another epidemiological study of STI pathogens conducted in two cities in southern China, the prevalence of N. gonorrhoeae, C. trachomatis, M. genitalium and T. vaginalis were 1.8%, 15.4%, 1.9% and 1.8% among female participants respectively (Kang et al., 2022). In this study, the infection rates of N. gonorrhoeae, C. trachomatis, M. genitalium and T. vaginalis among female participants were 1.8%, 10.0%, 2.3% and 2.1%, respectively. Based on the above studies, the prevalence of C. trachomatis is the highest, reaching or surpassing 10%, whereas the prevalence of M. genitalium exhibits significant variability.
There were 831 male participants with symptoms of urethritis (urethral discharge or irritation), of which 126 (15.2%) were detected with N. gonorrhoeae and the remaining 705 N. gonorrhoeae-negative males were defined as nongonococcal urethritis (NGU). In this study, the prevalence of C. trachomatis, M. genitalium and T. vaginalis among NGU was 17.2% (N=121), 5.1% (N=36) and 0.6% (N=4) respectively. N. gonorrhoeae and C. trachomatis are well-accepted etiologic agents of urethritis and in United States, about 5%-20% of urethritis is caused by N. gonorrhoeae infection (Bachmann et al., 2015), while C. trachomatis infection accounts for the highest proportion of NGU ranging from 15% to 40% (Wetmore et al., 2011). The prevalence of the two agents in our study were similar to those in United States. M. genitalium is now recognized as an established etiology of NGU and it is estimated to cause 13% to 31% of NGU cases in the US (Schwebke et al., 2011; Manhart et al., 2013). Furthermore, we also reported a lower detection rate of T. vaginalis compared to several published studies (Schwebke et al., 2011; Gaydos et al., 2013; Manhart et al., 2013). The comparatively lower detection rates of T. vaginalis and M. genitalium among urethritis cases suggest variations in their prevalence among different countries.
We believe that females infected with M. hominis deserved further attention. While its involvement in urogenital infections and its impact on reproductive health remain controversial, previous studies have reported associations between bacterial vaginosis (BV) and suggested M. hominis might promote the proliferation of other BV-associated bacteria (Taylor-Robinson, 2017; Rumyantseva et al., 2019; Plummer et al., 2021). In our study, detection of M. hominis was linked to abnormal vaginal discharge and vaginal itch, which are manifestations of BV, thereby supporting the association between M. hominis and BV. BV is associated with an overgrowth of Gardnerella vaginalis. Testing G. vaginalis and exploring the relationship between its level and M. hominis will help further clarify the role of M. hominis in BV.
Furthermore, the increased detection of M. hominis exhibited a significant correlation with lower education level and the absence of health insurance coverage. This underlines the potential substantial prevalence of M. hominis in China, or other populous developing countries. Additionally, we demonstrated a significant association between M. hominis and epididymis swelling or pain, which has provided insights into the etiology of epididymitis (Pilatz et al., 2015).
M. genitalium has rapidly acquired resistance against recommended treatment, US CDC has escalated it to the antimicrobial resistance threats watch list in 2019 (Centers for Disease Control and Prevention, 2019), but the surveillance data in China were limited. In our study, macrolide-resistant M. genitalium were found in 83.9% M. genitalium-positive samples, close to the prevalence reported in Nanjing (Li et al., 2020), higher than those reported in the United States (59.1% (Manhart et al., 2023) and 52% (Getman et al., 2023)), Australia (62%) (Sweeney et al., 2019), and Uganda (10.7%) (Melendez et al., 2022). Fluoroquinolone resistance is conferred by mutations in ParC and GyrA. Our study and others in Australia and China reported that less than 10% of M. genitalium had GyrA mutations, with all of these cases cooccurring with ParC mutations (Murray et al., 2017; Ke et al., 2020; Murray et al., 2023). Therefore, ParC, especially the Ser83Ile mutation, serves as a valuable marker to guide the clinical use of fluoroquinolones (Sweeney et al., 2022). Based on our study, the prevalence of wild-type ParC and ParC Ser83 were 26.7% and 39.8%, close to those reported in Guangzhou. These data indicate large cities in southern China have a low prevalence of wild-type of ParC, which will result in a higher risk of fluoroquinolone treatment failure. We also found that in participants lack of health insurance, there was a significant rise in the proportion of quinolone-resistant M. genitalium. This is likely attributed to patients seeking care from non-formal medical facilities or self-medicating.
HSV is the common cause of viral STI. A study in the United States found no significant differences in the manifestations of genital tract infection caused by HSV-1 and HSV-2 (Bernstein et al., 2013). However, in our study, HSV-1 exhibited a greater correlation with female urogenital symptoms compared to HSV-2. While the association between genital lesions and HSV-2 was only observed in male participants. The study in the United States has demonstrated variations in the epidemiological features of HSV across different age groups and racial populations. Consequently, the prevalence of HSV in China may exhibit distinct characteristics compared to United States. The data presented here will contribute novel evidence regarding the pathogenicity of HSV.
A key strength of this study is the large sample size, comprising 7886 participants, with both men and women. The participants encompassed both asymptomatic and symptomatic individuals presenting with a range of urogenital symptoms. Another strength is the broad screening of STI-related organisms using a highly sensitive mass spectrometry-based method. Finally, we systematically gathered sociodemographic information including marital status, educational attainment, health insurance coverage, income, etc. and these characteristics were found to be correlated with specific STI-related organisms.
One limitation of the study was the incapacity of mass spectrometry-based screening methods to quantify bacterial loads. Previous studies have reported associations between high bacterial loads of U. urealyticum and U. parvum with nongonococcal urethritis in men (Deguchi et al., 2015; Frolund et al., 2016). Additionally, high bacterial loads of M. hominis have been linked to dysbiosis in patients with BV (Cox et al., 2016; Taylor-Robinson, 2017). Therefore, by understanding bacterial loads, we can uncover a more detailed link between infectious organisms and specific symptoms. Through identifying the appropriate cut-off level of bacterial load, the role of infected organisms in patients with urogenital symptoms can be determined. Furthermore, investigating the correlation between sociodemographic characteristics and the bacterial load is a topic meriting exploration. Another limitation of the study was that urethral or cervical swabs were used as the only sample type for STI-related organism screening. Although there have been currently limited researches on the distribution of STI-related organisms across different sample types, Getman et al. found that the distribution of M. genitalium from female urogenital tract was highly complex, with diverse phenotypic combinations at different anatomic sites (Getman et al., 2023). Therefore, other STI-related organisms may exhibit distinctive distribution patterns across different sample types.
5 Conclusion
This study investigated the prevalence of STI-related organisms in Shenzhen, a representative large city in southern China and revealed a high prevalence of antimicrobial-resistant M. genitalium. Unmarried status, lower education level, and absence of health insurance coverage were important sociodemographic factors associated with an elevated positivity rate for multiple STI-related organisms. We found evidence supporting the association between M. hominis and BV or epididymitis. M. hominis exhibited a significant correlation with lower education level and the absence of health insurance coverage, which underlines the potential substantial prevalence of M. hominis in China. For HSV infection, we found HSV-1 exhibited a greater correlation with female urogenital symptoms. Additionally, we found a possible association between quinolone-resistant M. genitalium and lack of health insurance. These findings offer valuable insights into higher-risk key population groups, aiding the development of screening strategies and personalized guidelines for M. genitalium treatment.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
This study was performed in accordance with the recommendations of the national ethics regulations and approved by the Medical Ethics Committee of Shenzhen Center for Chronic Disease Control (20180301). All the participants provided written informed consent.
Author contributions
FFW: Conceptualization, Data curation, Formal Analysis, Project administration, Resources, Supervision, Writing – original draft, Writing – review & editing. CZ: Data curation, Formal Analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing. LX: Methodology, Validation, Writing – review & editing. YML: Methodology, Validation, Writing – review & editing. YZ: Methodology, Resources, Writing – review & editing. YZL: Methodology, Resources, Writing – review & editing. YC: Conceptualization, Data curation, Project administration, Resources, Supervision, Validation, Writing – review & editing. JP: Conceptualization, Funding acquisition, Investigation, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported by the CAMS Innovation Fund for Medical Sciences (CIFMS) (2021-I2M-1-037 and 2021-I2M-1-038).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcimb.2024.1407124/full#supplementary-material
References
Bachmann, L. H., Manhart, L. E., Martin, D. H., Sena, A. C., Dimitrakoff, J., Jensen, J. S., et al. (2015). Advances in the understanding and treatment of male urethritis. Clin. Infect. Dis. 61 Suppl 8, S763–S769. doi: 10.1093/cid/civ755
Bernstein, D. I., Bellamy, A. R., Hook, E. W., 3rd, Levin, M. J., Wald, A., Ewell, M. G., et al. (2013). Epidemiology, clinical presentation, and antibody response to primary infection with herpes simplex virus type 1 and type 2 in young women. Clin. Infect. Dis. 56, 344–351. doi: 10.1093/cid/cis891
Centers for Disease Control and Prevention (2019). Antibiotic Resistance Threats in the United States 2019. Available at www.cdc.gov/DrugResistance/Biggest-Threats.html.
Cox, C., Watt, A. P., Mckenna, J. P., Coyle, P. V. (2016). Mycoplasma hominis and Gardnerella vaginalis display a significant synergistic relationship in bacterial vaginosis. Eur. J. Clin. Microbiol. Infect. Dis. 35, 481–487. doi: 10.1007/s10096-015-2564-x
Deguchi, T., Shimada, Y., Horie, K., Mizutani, K., Seike, K., Tsuchiya, T., et al. (2015). Bacterial loads of Ureaplasma parvum contribute to the development of inflammatory responses in the male urethra. Int. J. STD AIDS 26, 1035–1039, 26. doi: 10.1177/0956462414565796
Frolund, M., Lidbrink, P., Wikstrom, A., Cowan, S., Ahrens, P., Jensen, J. S. (2016). Urethritis-associated pathogens in urine from men with non-gonococcal urethritis: A case-control study. Acta Derm Venereol 96, 689–694. doi: 10.2340/00015555-2314
Gaydos, C. A., Barnes, M. R., Quinn, N., Jett-Goheen, M., Hsieh, Y. H. (2013). Trichomonas vaginalis infection in men who submit self-collected penile swabs after internet recruitment. Sex Transm Infect. 89, 504–508. doi: 10.1136/sextrans-2012-050946
Getman, D., Cohen, S., Jiang, A. (2023). Distribution of macrolide resistant mycoplasma genitalium in urogenital tract specimens from women enrolled in a US clinical study cohort. Clin. Infect. Dis. 76, e776–e782. doi: 10.1093/cid/ciac602
Han, Y., Yin, Y. P., Liu, J. W., Chen, K., Zhu, B. Y., Zhou, K., et al. (2021). Rectal mycoplasma genitalium in patients attending sexually transmitted disease clinics in China: an infection that cannot be ignored. Infect. Drug Resist. 14, 2509–2515. doi: 10.2147/IDR.S314775
Horner, P., Donders, G., Cusini, M., Gomberg, M., Jensen, J. S., Unemo, M. (2018). Should we be testing for urogenital Mycoplasma hominis, Ureaplasma parvum and Ureaplasma urealyticum in men and women? - a position statement from the European STI Guidelines Editorial Board. J. Eur. Acad. Dermatol. Venereol 32, 1845–1851. doi: 10.1111/jdv.15146
Hughes, G., Saunders, J. (2018). Mycoplasma genitalium: the next sexually transmitted superbug? BMJ 363, k4376. doi: 10.1136/bmj.k4376
Kang, W. T., Xu, H., Liao, Y., Guo, Q., Huang, Q., Xu, Y., et al. (2022). Qualitative and quantitative detection of multiple sexually transmitted infection pathogens reveals distinct associations with cervicitis and vaginitis. Microbiol. Spectr. 10, e0196622. doi: 10.1128/spectrum.01966-22
Karellis, A., Naeem, F., Nair, S., Mallya, S. D., Routy, J. P., Gahagan, J., et al. (2022). Multiplexed rapid technologies for sexually transmitted infections: a systematic review. Lancet Microbe 3, e303–e315. doi: 10.1016/S2666-5247(21)00191-9
Ke, W., Li, D., Tso, L. S., Wei, R., Lan, Y., Chen, Z., et al. (2020). Macrolide and fluoroquinolone associated mutations in Mycoplasma genitalium in a retrospective study of male and female patients seeking care at a STI Clinic in Guangzhou, China 2016-2018. BMC Infect. Dis. 20, 950. doi: 10.1186/s12879-020-05659-3
Latimer, R. L., Vodstrcil, L. A., Plummer, E. L., Doyle, M., Murray, G. L., Fairley, C. K., et al. (2022). The clinical indications for testing women for Mycoplasma genitalium. Sex Transm Infect. 98, 277–285. doi: 10.1136/sextrans-2020-054818
Li, Y., Su, X., Le, W., Li, S., Yang, Z., Chaisson, C., et al. (2020). Mycoplasma genitalium in symptomatic male urethritis: macrolide use is associated with increased resistance. Clin. Infect. Dis. 70, 805–810. doi: 10.1093/cid/ciz294
Manhart, L. E., Gillespie, C. W., Lowens, M. S., Khosropour, C. M., Colombara, D. V., Golden, M. R., et al. (2013). Standard treatment regimens for nongonococcal urethritis have similar but declining cure rates: a randomized controlled trial. Clin. Infect. Dis. 56, 934–942. doi: 10.1093/cid/cis1022
Manhart, L. E., Leipertz, G., Soge, O. O., Jordan, S. J., Mcneil, C., Pathela, P., et al. (2023). Mycoplasma genitalium in the US (MyGeniUS): surveillance data from sexual health clinics in four US regions. Clin. Infect. Dis 77, 1449–1459. doi: 10.1093/cid/ciad405
Melendez, J. H., Hardick, J., Onzia, A., Yu, T., Kyambadde, P., Parkes-Ratanshi, R., et al. (2022). Retrospective analysis of Ugandan men with urethritis reveals mycoplasma genitalium and associated macrolide resistance. Microbiol. Spectr. 10, e0230421. doi: 10.1128/spectrum.02304-21
Murray, G. L., Bradshaw, C. S., Bissessor, M., Danielewski, J., Garland, S. M., Jensen, J. S., et al. (2017). Increasing macrolide and fluoroquinolone resistance in mycoplasma genitalium. Emerg. Infect. Dis. 23, 809–812. doi: 10.3201/eid2305.161745
Murray, G. L., Plummer, E. L., Bodiyabadu, K., Vodstrcil, L. A., Huaman, J. L., Danielewski, J. A., et al. (2023). gyrA mutations in mycoplasma genitalium and their contribution to moxifloxacin failure: time for the next generation of resistance-guided therapy. Clin. Infect. Dis. 76, 2187–2195. doi: 10.1093/cid/ciad057
Nahmias, S. B., Nahmias, D. (2011). Society, sex, and STIs: human behavior and the evolution of sexually transmitted diseases and their agents. Ann. N Y Acad. Sci. 1230, 59–73. doi: 10.1111/j.1749-6632.2011.06079.x
Ning, N., Weng, R., Zhang, C., Wen, L., Wang, H., Ye, J., et al. (2022). Cluster analysis for symptomatic management of Neisseria gonorrhoea and Chlamydia trachomatis in sexually transmitted infections related clinics in China. Front. Public Health 10. doi: 10.3389/fpubh.2022.1005481
Pereyre, S., Camelena, F., Henin, N., Bercot, B., Bebear, C. (2022). Clinical performance of four multiplex real-time PCR kits detecting urogenital and sexually transmitted pathogens. Clin. Microbiol. Infect. 28, 733.e7–733.e13. doi: 10.1016/j.cmi.2021.09.028
Pilatz, A., Hossain, H., Kaiser, R., Mankertz, A., Schuttler, C. G., Domann, E., et al. (2015). Acute epididymitis revisited: impact of molecular diagnostics on etiology and contemporary guideline recommendations. Eur. Urol 68, 428–435. doi: 10.1016/j.eururo.2014.12.005
Plummer, E. L., Vodstrcil, L. A., Bodiyabadu, K., Murray, G. L., Doyle, M., Latimer, R. L., et al. (2021). Are Mycoplasma hominis, Ureaplasma urealyticum and Ureaplasma parvum Associated With Specific Genital Symptoms and Clinical Signs in Nonpregnant Women? Clin. Infect. Dis. 73, 659–668. doi: 10.1093/cid/ciab061
Pouget, E. R. (2017). Social determinants of adult sex ratios and racial/ethnic disparities in transmission of HIV and other sexually transmitted infections in the USA. Philos. Trans. R Soc. Lond B Biol. Sci. 372. doi: 10.1098/rstb.2016.0323
Rowley, J., Vander Hoorn, S., Korenromp, E., Low, N., Unemo, M., Abu-Raddad, L. J., et al. (2019). Chlamydia, gonorrhoea, trichomoniasis and syphilis: global prevalence and incidence estimates 2016. Bull. World Health Organ 97, 548–562P. doi: 10.2471/BLT.18.228486
Rumyantseva, T., Khayrullina, G., Guschin, A., Donders, G. (2019). Prevalence of Ureaplasma spp. and Mycoplasma hominis in healthy women and patients with flora alterations. Diagn. Microbiol. Infect. Dis. 93, 227–231. doi: 10.1016/j.diagmicrobio.2018.10.001
Schwebke, J. R., Rompalo, A., Taylor, S., Sena, A. C., Martin, D. H., Lopez, L. M., et al. (2011). Re-evaluating the treatment of nongonococcal urethritis: emphasizing emerging pathogens–a randomized clinical trial. Clin. Infect. Dis. 52, 163–170. doi: 10.1093/cid/ciq074
Sweeney, E. L., Bradshaw, C. S., Murray, G. L., Whiley, D. M. (2022). Individualised treatment of Mycoplasma genitalium infection-incorporation of fluoroquinolone resistance testing into clinical care. Lancet Infect. Dis. 22, e267–e270. doi: 10.1016/S1473-3099(21)00629-0
Sweeney, E. L., Trembizki, E., Bletchly, C., Bradshaw, C. S., Menon, A., Francis, F., et al. (2019). Levels of mycoplasma genitalium antimicrobial resistance differ by both region and gender in the state of queensland, Australia: implications for treatment guidelines. J. Clin. Microbiol. 57. doi: 10.1128/JCM.01555-18
Taylor-Robinson, D. (2017). Mollicutes in vaginal microbiology: Mycoplasma hominis, Ureaplasma urealyticum, Ureaplasma parvum and Mycoplasma genitalium. Res. Microbiol. 168, 875–881. doi: 10.1016/j.resmic.2017.02.009
Van Gerwen, O. T., Muzny, C. A., Marrazzo, J. M. (2022). Sexually transmitted infections and female reproductive health. Nat. Microbiol. 7, 1116–1126. doi: 10.1038/s41564-022-01177-x
Wang, H., Weng, R., Zhang, C., Ye, J., Wen, L., Li, J., et al. (2022). High chlamydia infection and its associated factors among patients seeking clinic-based STI services in Southern China: A preliminary cross-sectional study. Front. Public Health 10. doi: 10.3389/fpubh.2022.1005334
Wayal, S., Reid, D., Weatherburn, P., Blomquist, P., Fabiane, S., Hughes, G., et al. (2019). Association between knowledge, risk behaviours, and testing for sexually transmitted infections among men who have sex with men: findings from a large online survey in the United Kingdom. HIV Med. 20, 523–533. doi: 10.1111/hiv.12753
Wetmore, C. M., Manhart, L. E., Lowens, M. S., Golden, M. R., Whittington, W. L., Xet-Mull, A. M., et al. (2011). Demographic, behavioral, and clinical characteristics of men with nongonococcal urethritis differ by etiology: a case-comparison study. Sex Transm Dis. 38, 180–186. doi: 10.1097/OLQ.0b013e3182040de9
World Health Organization (2016). Global Health Sector Strategy On Sexually Transmitted Infections 2016–2021. Available at https://www.who.int/publications/i/item/WHO-RHR-16.09
Xiu, L., Zhang, C., Li, Y., Wang, F., Peng, J. (2019). Simultaneous detection of eleven sexually transmitted agents using multiplexed PCR coupled with MALDI-TOF analysis. Infect. Drug Resist. 12, 2671–2682. doi: 10.2147/IDR.S219580
Yin, Y. P., Han, Y., Dai, X. Q., Zheng, H. P., Chen, S. C., Zhu, B. Y., et al. (2018). Susceptibility of Neisseria gonorrhoeae to azithromycin and ceftriaxone in China: A retrospective study of national surveillance data from 2013 to 2016. PloS Med. 15, e1002499. doi: 10.1371/journal.pmed.1002499
Keywords: sexually transmitted infections, sociodemographic characteristics, urogenital symptoms, Mycoplasma genitalium, antimicrobial resistance
Citation: Wang F, Zhang C, Xiu L, Li Y, Zeng Y, Li Y, Cai Y and Peng J (2024) Etiological, sociodemographic and clinical characteristics of sexually transmitted infections and M. genitalium resistance in Shenzhen: a multicenter cross-sectional study in China. Front. Cell. Infect. Microbiol. 14:1407124. doi: 10.3389/fcimb.2024.1407124
Received: 26 March 2024; Accepted: 28 June 2024;
Published: 25 July 2024.
Edited by:
Sherry Dunbar, Luminex (United States), United StatesReviewed by:
Fulvia Pimpinelli, San Gallicano Dermatological Institute IRCCS, ItalyYajie Wang, Capital Medical University, China
Copyright © 2024 Wang, Zhang, Xiu, Li, Zeng, Li, Cai and Peng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Junping Peng, cGVuZ2pwQGhvdG1haWwuY29t; Yumao Cai, NjQxNjU0NjlAcXEuY29t
†These authors have contributed equally to this work
 Feng Wang
Feng Wang Chi Zhang
Chi Zhang Leshan Xiu
Leshan Xiu Yamei Li2
Yamei Li2 Yumao Cai
Yumao Cai Junping Peng
Junping Peng