
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
MINI REVIEW article
Front. Cell. Infect. Microbiol. , 01 December 2023
Sec. Extra-intestinal Microbiome
Volume 13 - 2023 | https://doi.org/10.3389/fcimb.2023.1295111
This article is part of the Research Topic Reviews in Microbiome in Health & Disease View all 13 articles
 Guangyu Ma1
Guangyu Ma1 Yuguo Shi1
Yuguo Shi1 Lulu Meng1
Lulu Meng1 Haolong Fan2
Haolong Fan2 Xiaomei Tang1
Xiaomei Tang1 Huijuan Luo1
Huijuan Luo1 Dongju Wang1
Dongju Wang1 Juan Zhou1
Juan Zhou1 Xiaomin Xiao1*
Xiaomin Xiao1*In recent years, it has become evident that early-life intestinal flora plays a pivotal role in determining human health. Consequently, it is imperative to explore the establishment of neonatal intestinal flora and its influencing factors. Early neonatal intestinal flora is influenced by a multitude of factors, including maternal and infant-related factors, as well as external environment. This review summarizes the colonization mechanism of intestinal flora in the early life of newborns and discussed their influence on the establishment of neonatal intestinal flora, taking into account factors such as delivery mode, gestational age and feeding mode. Additionally, this review delves into the natural or artificial reconstruction of intestinal flora colonization defects in infants born via cesarean section and premature infants, with the goal of establishing a theoretical foundation for preventing and treating issues related to neonatal intestinal flora colonization and associated diseases.
The establishment of the human intestinal microbiota during early life plays a critical role in determining human, exerting profound effects on metabolic processes, growth, immune function, and even behavioral outcomes (Clemente et al., 2012). In recent years, there has been a substantial body of research focused on exploring the composition and functionality of the infant gut microbiota, revealing a distinct correlation between reduced microbial diversity and aberrant structural composition of the gut microbiota and the development of various diseases that may manifest in infancy or later in life, including conditions such as asthma, inflammatory bowel diseases, and metabolic disorders (Milani et al., 2017). Consequently, it is imperative to comprehensively investigate the governing principles and contributing factors that shape the establishment of the neonatal gut microbiota during early life. With the rapid advancements in metagenomic sequencing technology, it is possible to explore the intricate internal patterns and interrelationships within the highly complex gut microbiota. In this review, we have undertaken a comprehensive review of the research progress pertaining to the colonization mechanisms of the early-life intestinal microbiota in newborns. We have delved into the extensive discussion of its profound influence on the establishment of the neonatal intestinal microbiota, considering critical factors such as delivery mode, gestational age, and feeding practices. Furthermore, we have conducted a rigorous analysis of both natural and artificial strategies for rectifying colonization deficiencies within the intestinal microbiota, specifically in the context of infants born via cesarean section and preterm birth. Our primary objective is to furnish a well-founded theoretical framework that can inform and guide proactive measures for the prevention and treatment of intestinal microbiota colonization anomalies and the resultant diseases affecting clinical infants.
The traditional view suggests that the fetal environment in utero is sterile, and the colonization of the intestinal flora commences at birth. During childbirth, the neonate first encounters maternal intestinal, vaginal, and skin flora, as well as the surrounding environment. This initial contact facilitates the colonization and proliferation of the intestinal flora, which gradually evolves and establishes a mature microflora structure postnatally (Adlerberth, 2008). It is evident that maternal symbiotic bacteria play a crucial role in the early-life establishment of the neonatal intestinal flora. However, in line with pertinent research (Jiménez et al., 2005; DiGiulio et al., 2008; Aagaard et al., 2014; Collado et al., 2016; Chu et al., 2017b), bacteria were found in the placenta, umbilical cord and amniotic fluid in full-term pregnancy, which suggests that the fetus may not be entirely sterile in utero and that the initial colonization of the fetal intestinal flora may indeed commence within the maternal uterus. Jimenez E (Jiménez et al., 2005) conducted tests on umbilical cord blood flora from neonates delivered by cesarean section, revealing the presence of bacteria belonging to Enterococcus, Streptococcus, Staphylococcus, and Propionibacterium. Additionally, in animal experiments, the intestines of pregnant mice were inoculated with genetically labeled enterococcus faecium, and the labeled enterococcus faecium was detected in the feces of young mice delivered by cesarean section, suggesting the potential transmission of maternal microorganisms to the fetus in utero (Jiménez et al., 2008). The first meconium of a newborn can reflect the fetal situation in utero, and bacteria can be detected in the first meconium of a newborn (Kennedy et al., 2021; Turunen et al., 2021). These investigations provide compelling evidence supporting the concept of fetal intestinal flora colonization initiating in utero.
However, several studies have raised questions regarding whether fetal gut flora colonization truly initiates in utero. For instance, Kennedy KM (Kennedy et al., 2021) reported that they did not detect bacteria in the meconium collected from healthy newborns born via cesarean section. This suggests that, although microbial presence was observed in the fetal meconium for the first time, the majority of fetuses were delivered vaginally. The meconium collection typically occurs several hours to a few days after birth, and the microflora found in meconium may colonize during delivery or after birth. Therefore, it is plausible that the microflora found in meconium might colonize during delivery or in the postnatal period. Some studies have suggested an absence of flora in the amniotic fluid and placenta. RCR technology was conducted on the amniotic fluid of 344 women from 15 to 22 weeks of pregnancy, and the results showed that mycoplasma, bacteria and fungi in the amniotic fluid samples were negative (Rowlands et al., 2017). The amniotic fluid samples of 10 women who had cesarean section were tested, and the test results showed that the bacterial load in the amniotic fluid was no different from that in the blank control group (sterile Nacl), but the bacterial load in the amniotic fluid samples of 14 women with rupture of fetal membrane was 10 times higher than that of those who had cesarean section (Rehbinder et al., 2018). Amniotic fluid remains sterile due to the protective membrane. For infants with premature rupture of the membrane, intestinal flora colonization may initiate in utero, and fetal intestinal flora colonization may begin after the rupture of the membrane. Additionally, 16S rRNA sequencing technology was used to examine placenta-like flora in 29 women who did not give birth to full-term infants via cesarean section, and the results showed that there was no significant difference in the composition and structure of the bacterial spectrum between the placenta samples and the control group (Theis et al., 2019). Some scholars have raised concerns that bacterial flora determination technology primarily relies on bacterial 16S rRNA gene amplification, and the reagents and DNA extraction kits used may be susceptible to contamination from environmental bacteria. Such contamination could potentially affect the accuracy of bacterial flora determination and may not fully reflect the actual bacterial flora in the sample (Mühl et al., 2010; Salter et al., 2014). There is still a lot of controversy about whether fetal intestinal flora colonizes in utero or after delivery.
To investigate the origin of intestinal microflora in infants and the contribution of various maternal bacterial flora from different body sites to the establishment of infant intestinal microflora, Ferretti P and colleagues (Ferretti et al., 2018) collected the bacterial samples from the mother’s gut, vagina, skin and oral cavity. The results revealed that bacterial flora from these maternal sites contributed to the establishment of infant’s intestinal microflora. Maternal origins of neonatal gut microbiota are depicted in Figure 1. Notably, the maternal fecal microbiome accounted for the largest proportion of the contribution at 22.1%, followed by vagina (16.3%), mouth (7.2%) and skin (5%). Over time, the contribution of the maternal fecal microbiome increased, while the contributions from the vagina, mouth, and skin decreased. The colonization of the infant’s intestinal tract by the mother’s vaginal, oral, and skin flora was relatively transient. Interestingly, species from the maternal vaginal flora constituted approximately 3.5% of the total fecal microbial population in infants at 3 days after birth, but they were no longer detectable in the infant’s feces by the first week. This phenomenon may be attributed to the differences between the vaginal and intestinal environments, as the maternal vaginal strains faced challenges in establishing a lasting presence in the infant’s gut. The infant’s intestinal environment. Given the transient nature of maternal vaginal colonization in the infant’s gut, the feasibility and potential benefits of “vaginal seeding” warrant further discussion.
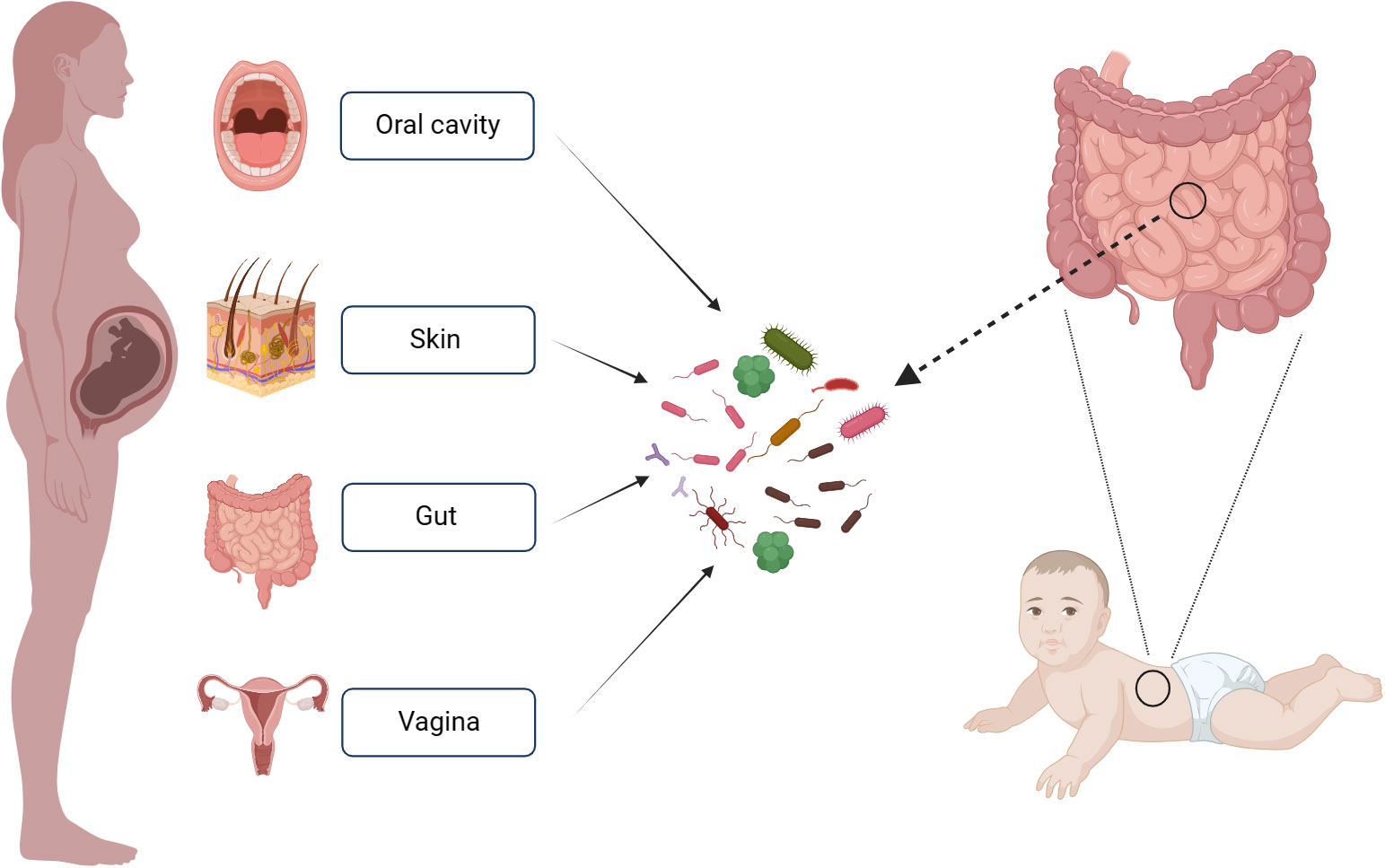
Figure 1 Maternal Sources of Neonatal Gut Microbiota. This figure was created with BioRender.com.
The microbial environment characterized by aerobic or facultative anaerobic bacteria, such as Enterobacter, Enterococcus and Staphylococcus, is established in the early stages of a newborn’s intestine. These bacteria consume oxygen during their growth, thereby altering the intestinal microenvironment to favor the proliferation of anaerobic bacteria, including Bifidobacterium, Clostridium, and Bacteroides (Adlerberth, 2008). Within the first week of birth, the infant’s intestinal flora undergoes changes influenced by the mother’s vaginal and skin flora. The maternal intestinal flora becomes increasingly dominant in colonizing the newborn’s intestinal tract. The alpha diversity of the initial flora decreases initially but gradually increases from the first to the third month of life. During this period, the infant’s microflora is primarily composed of Bifidobacterium and Bacteroides. It’s noteworthy that a stable gut microbiota resembling that of adults is typically established between the ages of 2 to 4 years (Stewart et al., 2018; O’Neill et al., 2020). Thus, neonates undergo a developmental process to establish their intestinal flora. In the initial stage, the intestinal flora is relatively simple and fragile, characterized by low species diversity, rendering it susceptible to external factors. Over time, however, the intestinal flora becomes relatively stable. This underscores the complexity of gut microbiota changes, with our current understanding representing only a fraction of the larger picture.
The early neonatal intestinal flora is influenced by various factors, including maternal and infant-related factors and external environment (O’Neill et al., 2020). This article delineates the influence of factors such as delivery mode and gestational age on the establishment of the infant’s intestinal microbiota (Figure 2). A schematic diagram illustrates how bacterial colonization in the infant gut microbiota changes under various influencing factors (Figure 3).
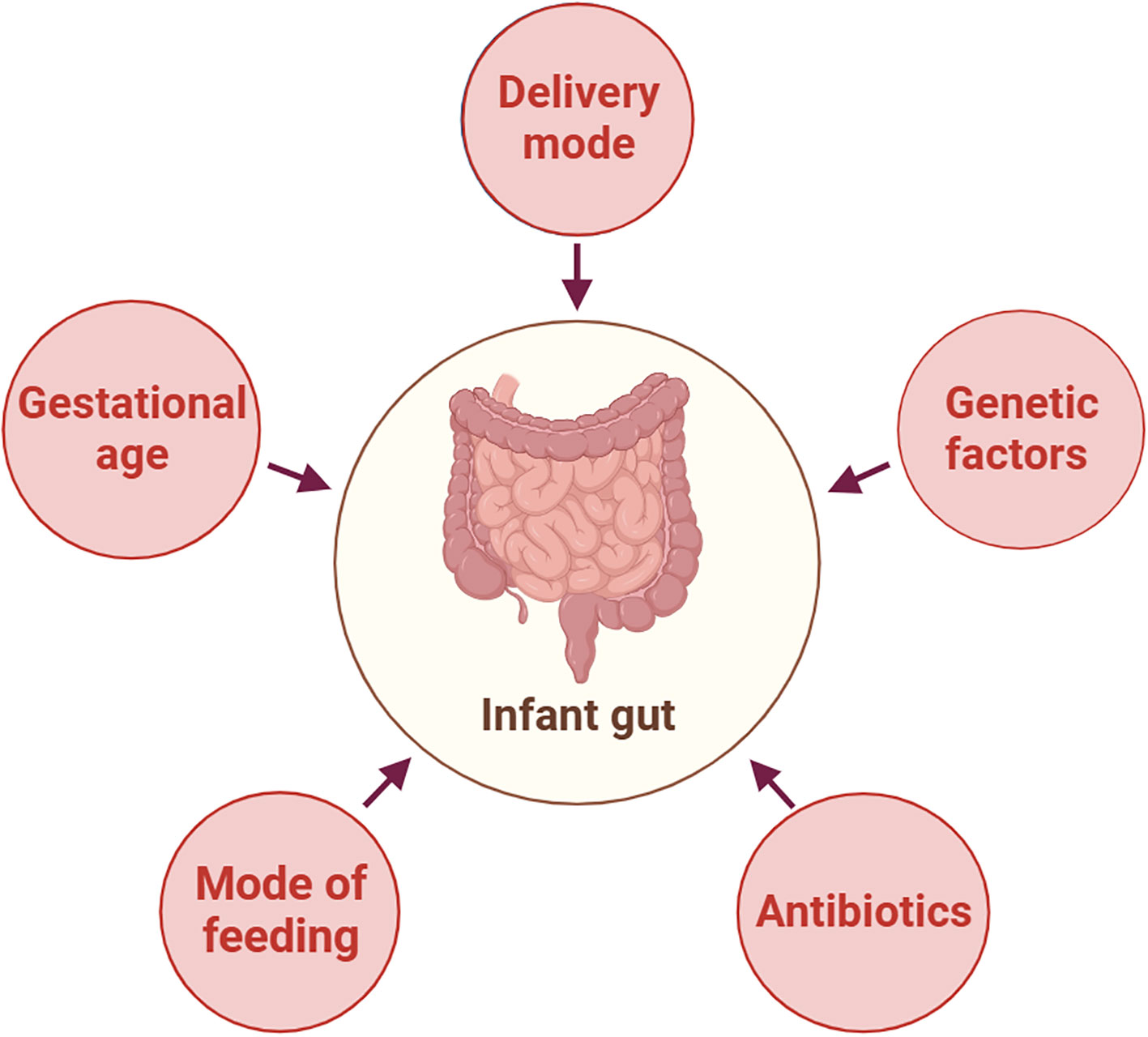
Figure 2 Factors Affecting the Composition of Neonatal Gut Microbiota. This figure was created with BioRender.com.
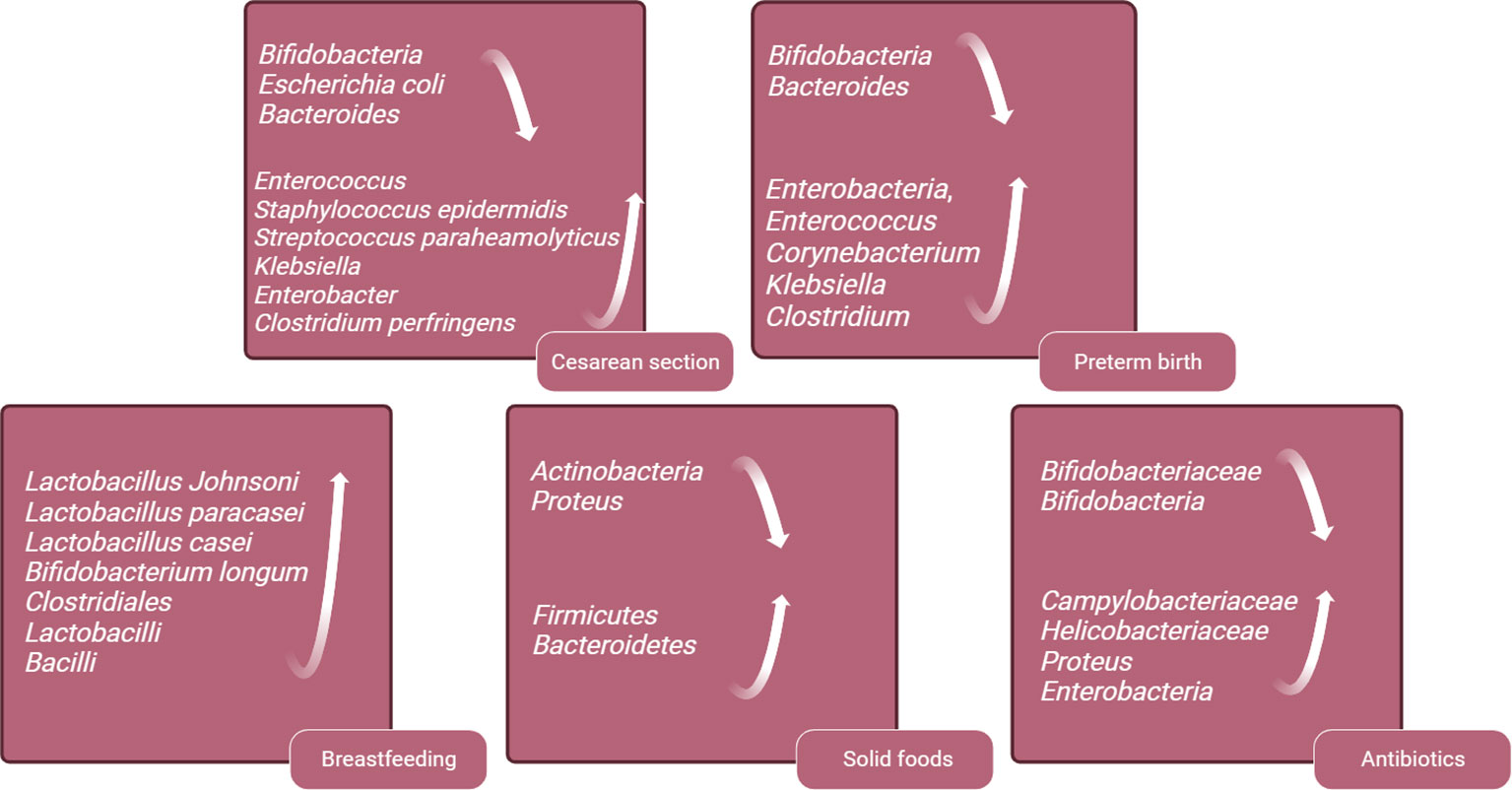
Figure 3 Schematic Diagram of Changes in Infant Gut Microbiota Bacteria Under Various Influencing Factors. This figure was created with BioRender.com.
Intestinal flora plays a pivotal role in the early development of the neonatal immune system and metabolism. The initial colonization of the gut microbiota during the perinatal period represents a critical juncture in the human gut microbiome’s ecological transformation. Fetuses born via cesarean section may experience colonization defects in their intestinal flora, which can have enduring impacts on host’s metabolism and immune development (Olszak et al., 2012). A comprehensive meta-analysis of 23 studies conducted in Northern Ireland has revealed that infants delivered via cesarean section face a 20% higher risk of developing asthma in both childhood and adulthood, in compared to those born through vaginal delivery (Thavagnanam et al., 2008). These findings have been meticulously adjusted to account for potential confounding factors such as maternal smoking, low birth weight, and the duration of breastfeeding. Furthermore, additional studies have noted a heightened susceptibility to allergic rhinitis and atopic dermatitis in children born through cesarean section (Pistiner et al., 2008; Thavagnanam et al., 2008).
In addition, there is a growing focus on exploring alterations in the colonization patterns of gut microbiota and their potential links to long-term risks of obesity and metabolic syndrome. A prospective cohort study found that, even after accounting for influencing factors such as maternal weight and birth weight, children born via cesarean section exhibit a significantly higher incidence of obesity by the age of 3, compared to those delivered vaginally (Huh et al., 2012). In Brazil, another cohort study has reported an increased occurrence of central and peripheral obesity in adults associated with cesarean section (Mesquita et al., 2013). Moreover, infants born via cesarean section have a 23% higher risk of developing type 1 diabetes during childhood (Cho and Norman, 2013). Fetuses born via cesarean section are not exposed to the mother’s vaginal flora but are initially exposed to flora from the mother’s skin or the birthing environment. Early fecal samples from vaginally delivered infants are dominated by Bifidobacteria, Escherichia coli and Bacteroides, accounting for approximately 68.3% of the neonatal gut microbiome. In contrast, infants delivered by cesarean section exhibit depleted populations of these gut microbiome genera, which are replaced by Enterococcus, Staphylococcus epidermidis, Streptococcus paraheamolyticus, Klebsiella, Enterobacter and Clostridium perfringens. These microbiotas are typically found on the skin and in the hospital environment, with 83.7% of infants born via cesarean section carrying opportunistic pathogens during the neonatal period, compared to 49.4% of vaginally born infants. This difference may increase the risk of neonatal infection (Lax et al., 2017; Wampach et al., 2018; Shao et al., 2019).
Cesarean section interrupts the normal transmission route of maternal symbiotic bacteria to infants, potentially increasing the chance of opportunistic pathogen colonization in the intestines. The increasing prevalence of cesarean section in recent years not only increases the risk of pregnancy in the scar area of cesarean section in the second pregnancy, but may also lead to defects in the colonization of fetal intestinal flora. Chu et al. (Chu et al., 2017a) demonstrated that the effect of delivery mode on fetal intestinal flora only lasts for the first 6 months, with no significant difference in flora after this period. Shao et al.(Shao et al., 2019) suggest that although the impact of cesarean section on infants’ intestinal flora diminishes over time, microbiome differences can still be detected up to the age of 1 year. Some studies have even found that differences in gut microbiota between cesarean section and vaginal delivery persist until the age of 7 (Penders et al., 2006; Neu and Rushing, 2011). The short-term or permanent effects of delivery mode on fetal microbiota still require long-term follow-up. Additionally, the routine use of antibiotics during cesarean section is a standard diagnosis and treatment process, so it remains uncertain whether antibiotics, as an interfering factor in comparing intestinal flora between vaginal and cesarean-born infants, impact the results.
The study conducted by Santos et al. presents a noteworthy finding that challenges the conventional understanding of the relationship between the maternal vaginal microbiome and the early development of the infant gut microbiome(Dos Santos et al., 2023). Traditionally, it has been believed that infants acquire their initial microbial communities during birth, particularly through exposure to maternal vaginal flora. However, the study’s results suggest otherwise, indicating that the composition of the maternal vaginal microbiome may not have a significant impact on the early years of the infant gut microbiome. This finding sparks further inquiry into the multifaceted factors that shape early gut microbiota and their implications for child health and development.
Gestational age at delivery is another crucial factor that impacts the establishment of intestinal flora in infants. Gestational age at birth significantly influences the formation and development of the intestinal microbiota, particularly in preterm infants. Research indicates that gestational age at birth plays a crucial role in shaping the structure and diversity of the intestinal microbiota. Gregory et al. (Gregory et al., 2016) proposed that gestational age at birth determines the trajectory of microbiota development within the first three weeks after birth. Chernikova et al.(Chernikova et al., 2018) demonstrated that gestational age at birth has a notable impact on the composition of the intestinal microbiota, with extremely preterm infants having a Simpson index of 0.35, compared to 0.65 in preterm infants. Furthermore, Drell et al. (Drell et al., 2014) emphasized a positive correlation between corrected gestational age and microbiota diversity, highlighting the role of gestational age in the evolution of the microbiota during early childhood.
Many diseases in premature infants are closely associated with their intestinal flora, such as necrotizing enterocolitis (NEC) (Aguilar-Lopez et al., 2021). Impaired composition and functionality of the intestinal flora have a direct impact on the health and potential complications of preterm infants. A better understanding of the differences in intestinal flora between preterm and full-term infants can help address the intestinal flora deficiencies in preterm infants and reduce related diseases.
In comparison with full-term infants, preterm infants have lower intestinal flora diversity, delayed colonization of Bifidobacteria and Bacteroides, and an increase in opportunistic pathogens like Enterobacteria, Enterococcus, Corynebacterium, Klebsiella and Clostridium (Mai et al., 2013; Arboleya et al., 2015). In addition, studies have found that the levels of short-chain fatty acids, which are metabolites products of the flora, are lower in the gut of preterm infants compared to full-term infants (Bergström et al., 2014). Premature infants exhibit significant differences in their intestinal microbiota compared to full-term infants. These distinctions may render premature infants more susceptible to microbial imbalances, leading to alterations in metabolic byproducts and potential ramifications for immune system development, as well as the risk of related diseases. These disparities are often attributed to the influence of various medical factors that premature infants are frequently exposed to, including cesarean section delivery, lack of breastfeeding, mechanical ventilation, and the frequent use of antibiotics. These factors can contribute to variations in the composition of the intestinal microbiota(Cuna et al., 2021). To improve the deficiency of intestinal flora in premature infants, this review will provide the following information.
In recent years, the research and findings have primarily focused on the influence of maternal symbiotic flora on neonatal intestinal flora colonization but have often overlooked the impact of acquired breastfeeding. Breast milk is the best source of nutrition for infants, and the World Health Organization advocates exclusive breastfeeding until six months of age, with complementary foods gradually added, and breastfeeding continuing for at least two years (Binns et al., 2016).
When compared to formula-fed infants delivered vaginally, exclusively breastfed infants have a higher presence of probiotics in their gut, including Lactobacillus Johnsoni, Lactobacillus paracasei, Lactobacillus casei and Bifidobacterium longum (Bäckhed et al., 2015). The imbalance of intestinal flora in infants resulting from cesarean delivery can be corrected through subsequent breastfeeding. At six months postpartum, the composition and structure of the intestinal microbiome in infants delivered via cesarean section differ from those delivered vaginally, with fewer Bacteroides and Shigella genera, and more Klebsiella, Veillonella and Enterococcus faecalis. The differences in intestinal microbiome caused by delivery the mode of delivery can be mitigated by acquired exclusive breastfeeding (Liu et al., 2023). Breastfeeding can reduce the incidence of NEC and sepsis in preterm infants (Quigley et al., 2018). Breastfeeding is vital for the establishment of intestinal flora in preterm infants. Studies have shown that preterm infants who are breastfed have increased gut microbiome alpha diversity compared to formula feeding (Cong et al., 2016). There are more Clostridiales, Lactobacilli and Bacilli in the intestines of premature infants who are breastfed compared to formula feeding and donor milk feeding (Cong et al., 2017). Breast milk is beneficial to the intestinal microbiome development of premature infants and increases the microbial diversity in early life, and it is recommended to support breast-feeding for premature infants after birth.
The weaning process represents the final trajectory of infant gut microbiota formation. In this process, diet plays a crucial role in regulating the microbial community (Kumbhare et al., 2019). Before weaning, the gut microbiota’s functional repertoire primarily involves gene expression related to the utilization of lactose (Tanaka and Nakayama, 2017). However, with the introduction of solid foods, the gene expression of the gut microbiota begins to increase in functions such as carbohydrate utilization, vitamin synthesis, and xenobiotic degradation. This transitional process is consistent with the rapid development of the immune system and changes in dietary intake.
The introduction of solid foods is another key factor in regulating the composition of the gut microbiota and marks the beginning of its maturation phase. This maturation of the microbial community is a gradually evolving process that ultimately leads to a more diverse and stable microbial profile (Davis et al., 2020). The introduction of solid foods triggers changes in the infant gut microbiota, and the supplementation of new nutrients increases the alpha -diversity of the gut microbiota, with Firmicutes and Bacteroidetes replacing Actinobacteria and Proteus as dominant phyla (Fallani et al., 2011; Koenig et al., 2011; Martin et al., 2016). Additionally, the total amount of short-chain fatty acids increases, with a significant rise in butyrate levels. The introduction of solid foods also increases the intake of plant-derived polysaccharides. During this stage, the composition of Bifidobacterium gradually shifts towards strains capable of metabolizing plant-derived polysaccharides, such as Bifidobacterium longum and Bifidobacterium adolescentis (O’Callaghan and van Sinderen, 2016). Simultaneously, Bifidobacterium bifidum redirects its metabolic capacity from human milk oligosaccharides (HMOs) towards mucin degradation.
Increased protein intake leads to an increase in the abundance of Lachnospira and a decrease in Bifidobacterium (Laursen et al., 2016). On the other hand, high fiber intake is associated with changes in the genus Prevotella. The addition of solid foods also marks the beginning of the transition of the infant gut microbiota structure towards an adult-like structure, with a microbial community becoming more complex to adapt to the plant-derived polysaccharides present in the adult diet, promoting mutualistic symbiosis between the host and microbiota (Koenig et al., 2011). This process continues until the age of two when the gut microbiota structure of infants reaches a stable state similar to that of adults.
Perinatal exposure to antibiotics has a significant impact on the early establishment of gut microbiota in offspring and increases the risk of childhood asthma, allergies, and obesity (Cox et al., 2014; Leclercq et al., 2017). Antibiotics affect an infant's gut microbiota in two ways: (1) Antibiotics can reach the fetal bloodstream through the umbilical cord. (2) Antibiotics alter the maternal vaginal and intestinal microbiome, leading to vertical transmission of infant intestinal flora abnormalities. A study on the effects of intrauterine antibiotic use on neonatal intestinal microbiota found that infants exposed to intrauterine antibiotic had decreased levels of Bifidobacteriaceae and an increased the proportion of potentially pathogenic microorganisms, including Campylobacteriaceae or Helicobacteriaceae (Nogacka et al., 2017). Another similar study found that infants treated with antibiotics during delivery had lower gut bacterial diversity, a decreased relative abundance of actinomycetes (especially Bifidobacteriaceae), and a higher relative abundance of Proteus in the gut microbiota (Zimmermann and Curtis, 2020). In full-term infants, the administration of antibiotics within a few hours after birth can reduce the level of Bifidobacteria in the gut and increase the level of Enterobacteria (Arboleya et al., 2015). The use of antibiotics during and after delivery can significantly affect the intestinal flora of infants, and it is recommended to avoid antibiotics unless medically necessary to reduce infants' exposure to antibiotics.
Given that premature infants tend to have relatively fragile immune systems and are more susceptible to infections, antibiotics are commonly administered for therapeutic purposes (Groer et al., 2014; Yasmin et al., 2017). In fact, antibiotics rank among the most frequently prescribed medications in the Neonatal Intensive Care Unit (NICU). However, the use of antibiotics presents a double-edged sword. While antibiotics can reduce the mortality rate associated with infectious diseases in premature infants, they often disrupts and disturbs the composition of the intestinal microbiota, consequently increasing the risk of various diseases (Morowitz et al., 2022). Additionally, research indicates that premature infants, particularly those with extremely low birth weights, face an increased risk of developing conditions like sepsis and NEC as a result of prolonged exposure to antibiotics (Cantey et al., 2018; Vatne et al., 2023).
As the prophylactic application of antibiotics to pregnant women during cesarean section is a routine obstetrical diagnosis and treatment process, antibiotics that can cross the placenta may reach the fetus through the placental barrier. In order to prevent intrauterine exposure of newborns to antibiotics, antibiotics are administered after umbilical cord cutting in some countries to minimize the exposure of newborns to antibiotics (Stinson et al., 2018). It is worth to study whether the timing of antibiotics for cesarean section prophylaxis can affect the gut microbes of infants or not, thus revealing the influence of antibiotic exposure before umbilical rupture to fetal gut flora. Relevant studies have compared the use of antibiotics before skin incision and after the umbilical cord is clamped in the gut microbiome composition of full-term infants born via elective cesarean section. Surprisingly, there were no significant difference in the gut microbiome composition observed at 9 months of age, and the potential long-term effects of antibiotics cannot be ruled out based on this study (Kamal et al., 2019). Similar results were found in another study, which reported no difference in intestinal microbiota composition between 10 and 9 months after birth (Dierikx et al., 2022). Prenatal antibiotic exposure in infants born via cesarean section does not appear to affect early-life microbiome development, but long-term follow-up is needed.
There is growing evidence that genetic factors influence the gut microbiota of infants. The similarity of intestinal flora between identical twins is higher than that between fraternal twins, indicating that genetic factors have an impact on fecal microbial community (Stewart et al., 2005). A study has proved that there are differences in the gut microbiome in males and females, with male babies having a lower gut microbiome diversity index at birth, while female babies have a higher gut microbiome diversity index, and female babies have a higher abundance of Clostridium difficile and lower abundance of Enterobacterium than male babies (Cong et al., 2016). Another similar study demonstrated that the relative abundance of Bacteroides in the intestinal flora of 3-month-old male infants was lower than that of female infants (Kozyrskyj et al., 2016). Genetic factors may play a potential role in the establishment of infant intestinal microbiota, and the mechanism of sex difference on intestinal microbiota community is still unclear.
In recent years, there has been growing interest in a research topic called ‘vaginal seeding’ as a way to help newborns born by cesarean section establish a healthy gut microbiome. This involves swabbing newborns with gauze containing maternal vaginal fluid, starting from the lips and proceeding to different parts of the body to mimic the natural birth process. The idea is to introduce beneficial vaginal microbes to cesarean-born babies, with the aim of making their gut flora more similar to that of babies born through vaginal delivery. Studies have shown that this ‘vaginal seeding’ can indeed help cesarean-born babies establish a gut microbiome that resembles that of babies born vaginally.
However, it’s important to note that current research has primarily focused on short-term effects, and we lack a clear understanding of the long-term consequences. There is also a concern about the potential risk of neonatal infection due to the introduction of vaginal bacteria during the ‘vaginal seeding’ process. Therefore, caution is needed, and more clinical studies are necessary to ensure the safety of this practice. If this practice is proven to be beneficial and safe, it’s important to assess its acceptance among expectant mothers.
Furthermore, researchers have explored different methods of introducing maternal vaginal microbes to cesarean-born babies. For example, a study by Wilson et al. (Wilson et al., 2021) attempted to give cesarean section newborns an oral solution containing maternal vaginal microbial. Surprisingly, the results showed no significant difference in the composition and development of the babies’ intestinal flora compared to those who received a standard oral saline solution. This suggests that the method of introducing mother’s vaginal microbes may not be as effective in reconstructing the gut flora of cesarean-born babies as previously thought. More experiments are needed to verify the impact of various seeding methods on the reconstruction of the newborns’ gut microbiome.
It has been reported that a newborn’s intestinal flora is primarily derived from the mother’s intestinal flora, with seeding of the maternal vaginal flora playing a secondary role in the colonization of the infant’s fecal flora (Sakwinska et al., 2017; Turunen et al., 2021). Ferretti P et al. (Jiménez et al., 2005) demonstrated that maternal intestinal, vaginal, skin and oral flora all contributed to the establishment of infant’s intestinal flora. Maternal fecal flora makes the most significant contribution to the infant’s intestinal flora, accounting for 22.1% of the infant’s intestinal microflora, followed by vaginal flora (16.3%). However, the contribution of vaginal flora species to infant feces diminishes over time. If vaginal microbiota isn’t the primary source of intestinal microbiota in infants, then the effectiveness of “vaginal seeding” in restoring intestinal microbiota colonization defects in newborns born by cesarean section is questionable. It is suspected that fecal microbiota transplantation (FMT) may correct the imbalance of intestinal microbiota in infants born by cesarean section. In an experimental setting, within 10 hours of birth, the mother’s fecal bacterial solution was mixed with breast milk and fed the cesarean-born infants, who did not experience adverse reactions. FMT treatment not only restored the intestinal microbiome of the cesarean-born infants but also led to the development of their gut microbiota being more similar to that of vaginally delivered infants. The microbiota of infants delivered by cesarean section with FMT in the early postpartum period was different from that of those delivered vaginally. However, from day 7 onwards, the gut microbiota of infants treated with FMT resembled that of infants delivered vaginally. Furthermore, FMT reduced the presence of intestinal opportunistic pathogens such as Enterococcus, Enterobacter, and Klebsiella in cesarean newborns (Korpela et al., 2020). Maternal intestinal flora transplantation has shown significant advantages in rectifying intestinal flora deficiencies in neonates born by cesarean section. Therefore, it may be feasible to consider the transplantation of maternal intestinal flora. However, the feasibility, safety, and clinical applicability of methods like ‘vaginal seeding’ and fecal bacteria transplantation still require further evaluation.
Further evaluation is needed to determine the feasibility and clinical applicability of ‘vaginal seeding’ and fecal bacteria transplantation as therapeutic methods to compensate for congenital defects in intestinal flora colonization.
Probiotics are live microorganisms that provide benefit to the host by colonizing the human body and changing the composition of the flora in a certain part of the host. The deficiency of newborn intestinal flora can be restored by probiotics. For premature infants, probiotics can enhance intestinal barrier function by regulating flora structure, reduce the colonization and migration of pathogenic bacteria, and promote the development and functional maturation of intestinal immune cells in newborns, thus reducing the incidence and mortality of NEC (Dermyshi et al., 2017; Gopalakrishna et al., 2019; Morgan et al., 2020). After oral administration of probiotics, preterm infants have a higher relative abundance of Bifidobacterium and Enterobacterium, and a lower relative abundance of Escherichia coli, Enterococcus and Klebsiella in the gut, and a lower incidence of NEC (Patole et al., 2014; van Best et al., 2020). Some studies have shown that early probiotic supplementation with Lacticaseibacillus rhamnosus GG (LGG) and Bifidobacterium animalis can affect the colonization of potential pathogens in infant intestines (Hui et al., 2021). Probiotics showed great benefit in restoring early intestinal flora colonization defects in preterm infants. It is recommended to apply probiotics in the early life of premature infants to prevent and treat intestinal flora defects and related diseases in premature infants.
Antibiotics can cause defects in infants’ gut microbiota and are associated with health problems in later life (Hviid et al., 2011; Metsälä et al., 2013; Azad et al., 2014; Chu et al., 2015). Probiotic supplementation can reshape antibiotic-treated intestinal microbiota disturbances in mice (Grazul et al., 2016). In the study (Zhong et al., 2021), it was found that exposure to piperacillin-tazobactam reduced the richness of the gut microbiota in full-term infants and may interfere with Bifidobacterium and Lactobacillus reproduction. It was also found that simultaneous treatment with probiotics and antibiotics was beneficial to the gut microbiota and could lead to an increase in Bifidobacterium. When supplementation with probiotics was delayed, there appeared to be no effect and no benefit from intervening in the gut microbiota disrupted by antibiotics. This study emphasizes that probiotics should be taken at the same time as antibiotics in the treatment of infants with intestinal flora disorders caused by antibiotics, and its therapeutic effect will be greater.
The alpha diversity of intestinal flora was significantly lower in newborns delivered by cesarean section compared to those born vaginally. However, after probiotic supplementation, the alpha diversity and beta diversity of intestinal flora in infants delivered by cesarean section become similar to those delivered vaginally. Additionally, there was a significant increase in the abundance of Lactobacillus and Bifidobacterium in the gut (Yang et al., 2021). The effect of probiotics on intestinal microbiota of newborns delivered by cesarean section also depends on the feeding mode of infants. In comparison to formula milk powder, breastfeeding led to an increase in intestinal Bifidobacterium and a decrease in Proteus and clostridium in cesarean-born infants (Korpela et al., 2018). Therefore, it is recommended to supplement probiotics to newborns delivered by cesarean section, and the promotion of infant breastfeeding is encouraged.
Overall, when compared to non-supplemented formulas, these prebiotic-supplemented ones increase the softness of stools (Skórka et al., 2018). They might also be able to decrease the incidence of enteric infections and diarrhea, reduce eczema, and increase Bifidobacteria counts. In premature infants, researchers have extensively studied various combinations of non-human milk galacto-, fructo-, and acidic oligosaccharides. These prebiotic mixtures have been found to modify the fecal microbiome, lower fecal pH, enhance gastric motility, reduce feeding intolerance, and increase fecal sIgA (Westerbeek et al., 2013). However, despite these positive effects, there is currently no related research on whether prebiotics can restore the colonization defects in the gut microbiota of newborns delivered by cesarean section, and this requires further investigation.
The question of whether fetal intestinal microbiota colonization occurs in utero or postnatally remains a subject of ongoing research with no definitive consensus. Traditional beliefs held that the fetal environment was sterile, with the infant’s gut primarily being colonized following birth through exposure to maternal and environmental microorganisms. However, recent investigations have introduced the possibility of some degree of microbial exposure or colonization in utero. Despite this, the extent and clinical significance of such prenatal microbial colonization remain subjects of investigation. Several studies have reported the presence of microbial DNA in amniotic fluid, placenta, and meconium (the initial fecal material passed by a newborn), sparking inquiries into the potential for prenatal microbial colonization. Yet, questions persist about the origins and roles of these microorganisms in the developing fetus. To gain a more comprehensive understanding of the timing and mechanisms underlying the establishment of the infant gut microbiota and the potential occurrence of prenatal colonization, further clinical studies are essential.
In shaping the infant’s gut microbiota, the review underscores the crucial role of the maternal-infant symbiotic relationship. Various microorganisms from different maternal body sites contribute to the development of the infant’s intestinal microflora, with the maternal gut microbiota making the most significant contribution. The infant’s gut microbiota undergoes multiple stages of change during the early period. It initially consists of aerobic or facultative anaerobic bacteria, gradually transitioning to anaerobic bacteria, such as Bifidobacterium, Clostridium, and Bacteroides. Understanding these transitions provides valuable insights into the formation of the infant’s gut microbiota.
While the colonization of the infant’s gut by maternal vaginal, oral, and skin flora is relatively short-lived, it has prompted discussions about the feasibility and potential benefits of the ‘vaginal seeding’ strategy. To determine when the infant’s gut microbiota tends to stabilize, resembling that of adults, further in-depth research is needed. This understanding contributes to a better comprehension of the evolution of the intestinal microbiota.
Future studies should delve deeper into the origins and dynamic changes of the infant’s gut microbiota to elucidate the potential impact of early microbial colonization on the health of children and adults. Additionally, there is a need for further research to explore the mechanisms and potential significance of prenatal microbial colonization.
Intestinal flora colonization is a complex process influenced by various factors, including delivery mode, gestational age, feeding mode, and antibiotic usage. The impact of delivery mode on newborn intestinal flora colonization is significant. Non-cesarean section indications should be advocated, and vaginal delivery should be selected as far as possible to avoid congenital colonization defects of infant intestinal flora. Understanding the impact of cesarean section on the neonatal gut microbiome and subsequent health outcomes is crucial. These findings shed light on the potential risks associated with cesarean section. Moreover, the need for long-term follow-up studies cannot be overstated. Such studies would allow us to gain a more comprehensive understanding of whether the effects observed persist over time or gradually diminish. This knowledge is crucial for healthcare professionals and parents alike. Exploring potential intervention measures to bridge the gap in gut microbiota between cesarean-born and vaginally born infants is another avenue for future research. This could include methods to promote the colonization of cesarean-born infants’ gut with flora closer to that of vaginally born infants or strategies to maintain a healthy balance of gut microbiota after cesarean section.
Preterm infants are typically delivered by cesarean section, and most receive antibiotic treatment while lacking the breastfeeding. Prolonged exposure to the hospital environment can exacerbate colonization defects in the intestinal flora of preterm infants. Future research needs to delve deeper into the diversity of the gut microbiota in preterm infants, including differences compared to full-term infants, in order to comprehensively understand its formation and evolution. Additionally, there is a need to enhance investigations into the connections between gut microbiota and diseases relevant to preterm infants, such as necrotizing enterocolitis, to determine the specific impact of the microbiota on these conditions, thus providing a basis for future prevention and treatment. It’s crucial not only to comprehend the composition of the gut microbiota but also to conduct in-depth research into their functions in metabolism, the immune system, and other physiological aspects to reveal more profound health associations. Long-term follow-up studies are indispensable to understand the extended development of the gut microbiota in preterm infants and their potential long-term health consequences, especially in adulthood, regarding potential risks related to the gut microbiota. Future research should focus on gaining a more comprehensive understanding of the formation and influence of the gut microbiota in preterm infants, with the goal of improving their health, reducing disease risks, offering more effective intervention measures, and enhancing their quality of life.
The use of antibiotics during the perinatal and postnatal periods can impact a baby’s gut microbiota. It is standard practice to administer prophylactic antibiotics to pregnant women during cesarean section. In order to minimize early antibiotic exposure for newborns, some guidelines recommend using antibiotics to prevent infection after umbilical cord cutting. Current studies suggest that the timing of antibiotic administration does not appear to affect the establishment of the baby’s gut microbiota. Breastfeeding is highly beneficial for establishing the intestinal flora in infants. It is recommended for newborns, and even in situations where breastfeeding is not feasible, it remains valuable. Breastfeeding contributes to the development of the intestinal microbiome of the intestinal microbiome in premature infants and enhances the microbial diversity in early life.
There are various perspectives on the source of neonatal intestinal flora colonization, including discussions about which components of the mother’s flora play a more beneficial role in compensating for congenital neonatal intestinal flora colonization defects. Debates persist regarding the feasibility of practices such as ‘vaginal seeding’, maternal fecal flora transplantation, and the choice between single-flora or multi-flora transplantation methods. More clinical studies are needed to validate these approaches. Breastfeeding, along with the use of probiotics and prebiotics, may have a beneficial effect in addressing colonization defects in the gut microbiota of preterm infants, newborns delivered by cesarean section, or infants affected by disorders related to gut microbiota establishment. Future research could consider gut microbes as a potential intervention to promote infant health.
GM: Writing – original draft. YS: Writing – original draft. LM: Writing – original draft. HF: Writing – original draft. XT: Writing – original draft. HL: Writing – original draft. DW: Writing – original draft. JZ: Writing – original draft. XX: Writing – review & editing.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study was supported by grants from the National Natural Science Foundation of China (No. 81771664). The content is solely the responsibility of the listed authors and does not necessarily represent the official views of the funding agency listed.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Aagaard, K., Ma, J., Antony, K. M., Ganu, R., Petrosino, J., Versalovic, J. (2014). The placenta harbors a unique microbiome. Sci. Transl. Med. 6, 237ra65. doi: 10.1126/scitranslmed.3008599
Adlerberth, I. (2008). Factors influencing the establishment of the intestinal microbiota in infancy. Nestle Nutr. Workshop Series Paediatric Programme 62, 13–29; discussion 29-33. doi: 10.1159/000146245
Aguilar-Lopez, M., Dinsmoor, A. M., Ho, T. T. B., Donovan, S. M. (2021). A systematic review of the factors influencing microbial colonization of the preterm infant gut. Gut Microbes 13, 1–33. doi: 10.1080/19490976.2021.1884514
Arboleya, S., Sánchez, B., Milani, C., Duranti, S., Solís, G., Fernández, N., et al. (2015). Intestinal microbiota development in preterm neonates and effect of perinatal antibiotics. J. Pediatr. 166, 538–544. doi: 10.1016/j.jpeds.2014.09.041
Azad, M. B., Bridgman, S. L., Becker, A. B., Kozyrskyj, A. L. (2014). Infant antibiotic exposure and the development of childhood overweight and central adiposity. Int. J. Obes. 38, 1290–1298. doi: 10.1038/ijo.2014.119
Bäckhed, F., Roswall, J., Peng, Y., Feng, Q., Jia, H., Kovatcheva-Datchary, P., et al. (2015). Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe 17, 690–703. doi: 10.1016/j.chom.2015.04.004
Bergström, A., Skov, T. H., Bahl, M. I., Roager, H. M., Christensen, L. B., Ejlerskov, K. T., et al. (2014). Establishment of intestinal microbiota during early life: a longitudinal, explorative study of a large cohort of Danish infants. Appl. Environ. Microbiol. 80, 2889–3000. doi: 10.1128/AEM.00342-14
Binns, C., Lee, M., Low, W. Y. (2016). The long-term public health benefits of breastfeeding. Asia Pac J. Public Health 28, 7–14. doi: 10.1177/1010539515624964
Cantey, J. B., Pyle, A. K., Wozniak, P. S., Hynan, L. S., Sánchez, P. J. (2018). Early antibiotic exposure and adverse outcomes in preterm, very low birth weight infants. J. Pediatr. 203, 62–67. doi: 10.1016/j.jpeds.2018.07.036
Chernikova, D. A., Madan, J. C., Housman, M. L., Zain-Ul-Abideen, M., Lundgren, S. N., Morrison, H. G., et al. (2018). The premature infant gut microbiome during the first 6 weeks of life differs based on gestational maturity at birth. Pediatr. Res. 84, 71–79. doi: 10.1038/s41390-018-0022-z
Cho, C. E., Norman, M. (2013). Cesarean section and development of the immune system in the offspring. Am. J. Obstet. Gynecol. 208, 249–254. doi: 10.1016/j.ajog.2012.08.009
Chu, D. M., Ma, J., Prince, A. L., Antony, K. M., Seferovic, M. D., Aagaard, K. M. (2017a). Maturation of the infant microbiome community structure and function across multiple body sites and in relation to mode of delivery. Nat. Med. 23, 314–326. doi: 10.1038/nm.4272
Chu, D., Stewart, C., Seferovic, M., Suter, M., Cox, J., Vidaeff, A., et al. (2017b). 26: Profiling of microbiota in second trimester amniotic fluid reveals a distinctive community present in the mid trimester and predictive of the placental microbiome at parturition. Am. J. Obstet. Gynecol. 216, S18–S19. doi: 10.1016/j.ajog.2016.11.917
Chu, S., Yu, H., Chen, Y., Chen, Q., Wang, B., Zhang, J. (2015). Periconceptional and gestational exposure to antibiotics and childhood asthma. PloS One 10, e0140443. doi: 10.1371/journal.pone.0140443
Clemente, J. C., Ursell, L. K., Parfrey, L. W., Knight, R. (2012). The impact of the gut microbiota on human health: an integrative view. Cell 148, 1258–1270. doi: 10.1016/j.cell.2012.01.035
Collado, M. C., Rautava, S., Aakko, J., Isolauri, E., Salminen, S. (2016). Human gut colonisation may be initiated in utero by distinct microbial communities in the placenta and amniotic fluid. Sci. Rep. 6, 23129. doi: 10.1038/srep23129
Cong, X., Judge, M., Xu, W., Diallo, A., Janton, S., Brownell, E. A., et al. (2017). Influence of feeding type on gut microbiome development in hospitalized preterm infants. Nurs. Res. 66, 123–133. doi: 10.1097/NNR.0000000000000208
Cong, X., Xu, W., Janton, S., Henderson, W. A., Matson, A., McGrath, J. M., et al. (2016). Gut microbiome developmental patterns in early life of preterm infants: impacts of feeding and gender. PloS One 11, e0152751. doi: 10.1371/journal.pone.0152751
Cox, L. M., Yamanishi, S., Sohn, J., Alekseyenko, A. V., Leung, J. M., Cho, I., et al. (2014). Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell 158, 705–721. doi: 10.1016/j.cell.2014.05.052
Cuna, A., Morowitz, M. J., Ahmed, I., Umar, S., Sampath, V. (2021). Dynamics of the preterm gut microbiome in health and disease. Am. J. Physiol. Gastrointest. Liver Physiol. 320, G411–G419. doi: 10.1152/ajpgi.00399.2020
Davis, E. C., Dinsmoor, A. M., Wang, M., Donovan, S. M. (2020). Microbiome composition in pediatric populations from birth to adolescence: impact of diet and prebiotic and probiotic interventions. Dig. Dis. Sci. 65, 706–722. doi: 10.1007/s10620-020-06092-x
Dermyshi, E., Wang, Y., Yan, C., Hong, W., Qiu, G., Gong, X., et al. (2017). The “Golden age” of probiotics: A systematic review and meta-analysis of randomized and observational studies in preterm infants. Neonatology 112, 9–23. doi: 10.1159/000454668
Dierikx, T., Berkhout, D., Eck, A., Tims, S., van Limbergen, J., Visser, D., et al. (2022). Influence of timing of maternal antibiotic administration during caesarean section on infant microbial colonisation: a randomised controlled trial. Gut 71, 1803–1811. doi: 10.1136/gutjnl-2021-324767
DiGiulio, D. B., Romero, R., Amogan, H. P., Kusanovic, J. P., Bik, E. M., Gotsch, F., et al. (2008). Microbial prevalence, diversity and abundance in amniotic fluid during preterm labor: a molecular and culture-based investigation. PloS One 3, e3056. doi: 10.1371/journal.pone.0003056
Dos Santos, S. J., Pakzad, Z., Albert, A. Y. K., Elwood, C. N., Grabowska, K., Links, M. G., et al. (2023). Maternal vaginal microbiome composition does not affect development of the infant gut microbiome in early life. Front. Cell. Infect. Microbiol. 13. doi: 10.3389/fcimb.2023.1144254
Drell, T., Lutsar, I., Stšepetova, J., Parm, U., Metsvaht, T., Ilmoja, M.-L., et al. (2014). The development of gut microbiota in critically ill extremely low birth weight infants assessed with 16S rRNA gene based sequencing. Gut Microbes 5, 304–312. doi: 10.4161/gmic.28849
Fallani, M., Amarri, S., Uusijarvi, A., Adam, R., Khanna, S., Aguilera, M., et al. (2011). Determinants of the human infant intestinal microbiota after the introduction of first complementary foods in infant samples from five European centres. Microbiol. (Reading) 157, 1385–1392. doi: 10.1099/mic.0.042143-0
Ferretti, P., Pasolli, E., Tett, A., Asnicar, F., Gorfer, V., Fedi, S., et al. (2018). Mother-to-infant microbial transmission from different body sites shapes the developing infant gut microbiome. Cell Host Microbe 24, 133–145.e5. doi: 10.1016/j.chom.2018.06.005
Gopalakrishna, K. P., Macadangdang, B. R., Rogers, M. B., Tometich, J. T., Firek, B. A., Baker, R., et al. (2019). Maternal IgA protects against the development of necrotizing enterocolitis in preterm infants. Nat. Med. 25, 1110–1115. doi: 10.1038/s41591-019-0480-9
Grazul, H., Kanda, L. L., Gondek, D. (2016). Impact of probiotic supplements on microbiome diversity following antibiotic treatment of mice. Gut Microbes 7, 101–114. doi: 10.1080/19490976.2016.1138197
Gregory, K. E., Samuel, B. S., Houghteling, P., Shan, G., Ausubel, F. M., Sadreyev, R. I., et al. (2016). Influence of maternal breast milk ingestion on acquisition of the intestinal microbiome in preterm infants. Microbiome 4, 68. doi: 10.1186/s40168-016-0214-x
Groer, M. W., Luciano, A. A., Dishaw, L. J., Ashmeade, T. L., Miller, E., Gilbert, J. A. (2014). Development of the preterm infant gut microbiome: a research priority. Microbiome 2, 38. doi: 10.1186/2049-2618-2-38
Huh, S. Y., Rifas-Shiman, S. L., Zera, C. A., Edwards, J. W. R., Oken, E., Weiss, S. T., et al. (2012). Delivery by caesarean section and risk of obesity in preschool age children: a prospective cohort study. Arch. Dis. Child 97, 610–616. doi: 10.1136/archdischild-2011-301141
Hui, Y., Smith, B., Mortensen, M. S., Krych, L., Sørensen, S. J., Greisen, G., et al. (2021). The effect of early probiotic exposure on the preterm infant gut microbiome development. Gut Microbes 13, 1951113. doi: 10.1080/19490976.2021.1951113
Hviid, A., Svanström, H., Frisch, M. (2011). Antibiotic use and inflammatory bowel diseases in childhood. Gut 60, 49–54. doi: 10.1136/gut.2010.219683
Jiménez, E., Fernández, L., Marín, M. L., Martín, R., Odriozola, J. M., Nueno-Palop, C., et al. (2005). Isolation of commensal bacteria from umbilical cord blood of healthy neonates born by cesarean section. Curr. Microbiol. 51, 270–274. doi: 10.1007/s00284-005-0020-3
Jiménez, E., Marín, M. L., Martín, R., Odriozola, J. M., Olivares, M., Xaus, J., et al. (2008). Is meconium from healthy newborns actually sterile? Res. Microbiol. 159, 187–193. doi: 10.1016/j.resmic.2007.12.007
Kamal, S. S., Hyldig, N., Krych, Ł., Greisen, G., Krogfelt, K. A., Zachariassen, G., et al. (2019). Impact of early exposure to cefuroxime on the composition of the gut microbiota in infants following cesarean delivery. J. Pediatr. 210, 99–105.e2. doi: 10.1016/j.jpeds.2019.03.001
Kennedy, K. M., Gerlach, M. J., Adam, T., Heimesaat, M. M., Rossi, L., Surette, M. G., et al. (2021). Fetal meconium does not have a detectable microbiota before birth. Nat. Microbiol. 6, 865–873. doi: 10.1038/s41564-021-00904-0
Koenig, J. E., Spor, A., Scalfone, N., Fricker, A. D., Stombaugh, J., Knight, R., et al. (2011). Succession of microbial consortia in the developing infant gut microbiome. Proc. Natl. Acad. Sci. U.S.A. 108 (Suppl 1), 4578–4585. doi: 10.1073/pnas.1000081107
Korpela, K., Helve, O., Kolho, K.-L., Saisto, T., Skogberg, K., Dikareva, E., et al. (2020). Maternal fecal microbiota transplantation in cesarean-born infants rapidly restores normal gut microbial development: A proof-of-concept study. Cell 183, 324–334.e5. doi: 10.1016/j.cell.2020.08.047
Korpela, K., Salonen, A., Vepsäläinen, O., Suomalainen, M., Kolmeder, C., Varjosalo, M., et al. (2018). Probiotic supplementation restores normal microbiota composition and function in antibiotic-treated and in caesarean-born infants. Microbiome 6, 182. doi: 10.1186/s40168-018-0567-4
Kozyrskyj, A. L., Kalu, R., Koleva, P. T., Bridgman, S. L. (2016). Fetal programming of overweight through the microbiome: boys are disproportionately affected. J. Dev. Origins Health Dis. 7, 25–34. doi: 10.1017/S2040174415001269
Kumbhare, S. V., Patangia, D. V. V., Patil, R. H., Shouche, Y. S., Patil, N. P. (2019). Factors influencing the gut microbiome in children: from infancy to childhood. J. Biosci. 44, 49. doi: 10.1007/s12038-019-9860-z
Laursen, M. F., Andersen, L. B. B., Michaelsen, K. F., Mølgaard, C., Trolle, E., Bahl, M. I., et al. (2016). Infant gut microbiota development is driven by transition to family foods independent of maternal obesity. mSphere 1, e00069-15. doi: 10.1128/mSphere.00069-15
Lax, S., Sangwan, N., Smith, D., Larsen, P., Handley, K. M., Richardson, M., et al. (2017). Bacterial colonization and succession in a newly opened hospital. Sci. Transl. Med. 9, eaah6500. doi: 10.1126/scitranslmed.aah6500
Leclercq, S., Mian, F. M., Stanisz, A. M., Bindels, L. B., Cambier, E., Ben-Amram, H., et al. (2017). Low-dose penicillin in early life induces long-term changes in murine gut microbiota, brain cytokines and behavior. Nat. Commun. 8, 15062. doi: 10.1038/ncomms15062
Liu, Y., Ma, J., Zhu, B., Liu, F., Qin, S., Lv, N., et al. (2023). A health-promoting role of exclusive breastfeeding on infants through restoring delivery mode-induced gut microbiota perturbations. Front. Microbiol. 14. doi: 10.3389/fmicb.2023.1163269
Mai, V., Torrazza, R. M., Ukhanova, M., Wang, X., Sun, Y., Li, N., et al. (2013). Distortions in development of intestinal microbiota associated with late onset sepsis in preterm infants. PloS One 8, e52876. doi: 10.1371/journal.pone.0052876
Martin, R., Makino, H., Cetinyurek Yavuz, A., Ben-Amor, K., Roelofs, M., Ishikawa, E., et al. (2016). Early-life events, including mode of delivery and type of feeding, siblings and gender, shape the developing gut microbiota. PloS One 11, e0158498. doi: 10.1371/journal.pone.0158498
Mesquita, D. N., Barbieri, M. A., Goldani, H. A. S., Cardoso, V. C., Goldani, M. Z., Kac, G., et al. (2013). Cesarean section is associated with increased peripheral and central adiposity in young adulthood: cohort study. PloS One 8, e66827. doi: 10.1371/journal.pone.0066827
Metsälä, J., Lundqvist, A., Virta, L. J., Kaila, M., Gissler, M., Virtanen, S. M. (2013). Mother’s and offspring’s use of antibiotics and infant allergy to cow’s milk. Epidemiol. (Cambridge Mass) 24, 303–309. doi: 10.1097/EDE.0b013e31827f520f
Milani, C., Duranti, S., Bottacini, F., Casey, E., Turroni, F., Mahony, J., et al. (2017). The first microbial colonizers of the human gut: composition, activities, and health implications of the infant gut microbiota. Microbiol. Mol. Biol. Reviews: MMBR 81, e00036-17. doi: 10.1128/MMBR.00036-17
Morgan, R. L., Preidis, G. A., Kashyap, P. C., Weizman, A. V., Sadeghirad, B., McMaster Probiotic, Prebiotic, and Synbiotic Work Group (2020). Probiotics reduce mortality and morbidity in preterm, low-birth-weight infants: A systematic review and network meta-analysis of randomized trials. Gastroenterology 159, 467–480. doi: 10.1053/j.gastro.2020.05.096
Morowitz, M. J., Katheria, A. C., Polin, R. A., Pace, E., Huang, D. T., Chang, C.-C. H., et al. (2022). The NICU antibiotics and outcomes (NANO) trial: a randomized multicenter clinical trial assessing empiric antibiotics and clinical outcomes in newborn preterm infants. Trials 23, 428. doi: 10.1186/s13063-022-06352-3
Mühl, H., Kochem, A.-J., Disqué, C., Sakka, S. G. (2010). Activity and DNA contamination of commercial polymerase chain reaction reagents for the universal 16S rDNA real-time polymerase chain reaction detection of bacterial pathogens in blood. Diagn. Microbiol. Infect. Dis. 66, 41–49. doi: 10.1016/j.diagmicrobio.2008.07.011
Neu, J., Rushing, J. (2011). Cesarean versus vaginal delivery: long-term infant outcomes and the hygiene hypothesis. Clin. Perinatol 38, 321–331. doi: 10.1016/j.clp.2011.03.008
Nogacka, A., Salazar, N., Suárez, M., Milani, C., Arboleya, S., Solís, G., et al. (2017). Impact of intrapartum antimicrobial prophylaxis upon the intestinal microbiota and the prevalence of antibiotic resistance genes in vaginally delivered full-term neonates. Microbiome 5, 93. doi: 10.1186/s40168-017-0313-3
O’Callaghan, A., van Sinderen, D. (2016). Bifidobacteria and their role as members of the human gut microbiota. Front. Microbiol. 7. doi: 10.3389/fmicb.2016.00925
Olszak, T., An, D., Zeissig, S., Vera, M. P., Richter, J., Franke, A., et al. (2012). Microbial exposure during early life has persistent effects on natural killer T cell function. Sci. (New York N.Y.) 336, 489–493. doi: 10.1126/science.1219328
O’Neill, I. J., Sanchez Gallardo, R., Saldova, R., Murphy, E. F., Cotter, P. D., McAuliffe, F. M., et al. (2020). Maternal and infant factors that shape neonatal gut colonization by bacteria. Expert Rev. Gastroenterol. Hepatol. 14, 651–664. doi: 10.1080/17474124.2020.1784725
Patole, S., Keil, A. D., Chang, A., Nathan, E., Doherty, D., Simmer, K., et al. (2014). Effect of Bifidobacterium breve M-16V supplementation on fecal bifidobacteria in preterm neonates–a randomised double blind placebo controlled trial. PloS One 9, e89511. doi: 10.1371/journal.pone.0089511
Penders, J., Thijs, C., Vink, C., Stelma, F. F., Sinjders, B., Kummeling, I., et al. (2006). Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics 118, 511–521. doi: 10.1542/peds.2005-2824
Pistiner, M., Gold, D. R., Abdulkerim, H., Hoffman, E., Celedón, J. C. (2008). Birth by cesarean section, allergic rhinitis, and allergic sensitization among children with a parental history of atopy. J. Allergy Clin. Immunol. 122, 274–279. doi: 10.1016/j.jaci.2008.05.007
Quigley, M., Embleton, N. D., McGuire, W. (2018). Formula versus donor breast milk for feeding preterm or low birth weight infants. Cochrane Database Sys. Rev. 6, 1–73. doi: 10.1002/14651858.CD002971.pub4
Rehbinder, E. M., Lødrup Carlsen, K. C., Staff, A. C., Angell, I. L., Landrø, L., Hilde, K., et al. (2018). Is amniotic fluid of women with uncomplicated term pregnancies free of bacteria? Am. J. Obstet. Gynecol. 219, 289.e1–289.e12. doi: 10.1016/j.ajog.2018.05.028
Rowlands, S., Danielewski, J. A., Tabrizi, S. N., Walker, S. P., Garland, S. M. (2017). Microbial invasion of the amniotic cavity in midtrimester pregnancies using molecular microbiology. Am. J. Obstet. Gynecol. 217, 71.e1–71.e5. doi: 10.1016/j.ajog.2017.02.051
Sakwinska, O., Foata, F., Berger, B., Brüssow, H., Combremont, S., Mercenier, A., et al. (2017). Does the maternal vaginal microbiota play a role in seeding the microbiota of neonatal gut and nose? Beneficial Microbes 8, 763–778. doi: 10.3920/BM2017.0064
Salter, S. J., Cox, M. J., Turek, E. M., Calus, S. T., Cookson, W. O., Moffatt, M. F., et al. (2014). Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 12, 87. doi: 10.1186/s12915-014-0087-z
Shao, Y., Forster, S. C., Tsaliki, E., Vervier, K., Strang, A., Simpson, N., et al. (2019). Stunted microbiota and opportunistic pathogen colonization in caesarean-section birth. Nature 574, 117–121. doi: 10.1038/s41586-019-1560-1
Skórka, A., Pieścik-Lech, M., Kołodziej, M., Szajewska, H. (2018). Infant formulae supplemented with prebiotics: Are they better than unsupplemented formulae? An updated systematic review. Br. J. Nutr. 119, 810–825. doi: 10.1017/S0007114518000120
Stewart, C. J., Ajami, N. J., O’Brien, J. L., Hutchinson, D. S., Smith, D. P., Wong, M. C., et al. (2018). Temporal development of the gut microbiome in early childhood from the TEDDY study. Nature 562, 583–588. doi: 10.1038/s41586-018-0617-x
Stewart, J. A., Chadwick, V. S., Murray, A. (2005). Investigations into the influence of host genetics on the predominant eubacteria in the faecal microflora of children. J. Med. Microbiol. 54, 1239–1242. doi: 10.1099/jmm.0.46189-0
Stinson, L. F., Payne, M. S., Keelan, J. A. (2018). A critical review of the bacterial baptism hypothesis and the impact of cesarean delivery on the infant microbiome. Front. Med. 5. doi: 10.3389/fmed.2018.00135
Tanaka, M., Nakayama, J. (2017). Development of the gut microbiota in infancy and its impact on health in later life. Allergol Int. 66, 515–522. doi: 10.1016/j.alit.2017.07.010
Thavagnanam, S., Fleming, J., Bromley, A., Shields, M. D., Cardwell, C. R. (2008). A meta-analysis of the association between Caesarean section and childhood asthma. Clin. Exp. Allergy 38, 629–633. doi: 10.1111/j.1365-2222.2007.02780.x
Theis, K. R., Romero, R., Winters, A. D., Greenberg, J. M., Gomez-Lopez, N., Alhousseini, A., et al. (2019). Does the human placenta delivered at term have a microbiota? Results of cultivation, quantitative real-time PCR, 16S rRNA gene sequencing, and metagenomics. Am. J. Obstet. Gynecol. 220, 267.e1–267.e39. doi: 10.1016/j.ajog.2018.10.018
Turunen, J., Tejesvi, M. V., Paalanne, N., Hekkala, J., Lindgren, O., Kaakinen, M., et al. (2021). Presence of distinctive microbiome in the first-pass meconium of newborn infants. Sci. Rep. 11, 19449. doi: 10.1038/s41598-021-98951-4
van Best, N., Trepels-Kottek, S., Savelkoul, P., Orlikowsky, T., Hornef, M. W., Penders, J. (2020). Influence of probiotic supplementation on the developing microbiota in human preterm neonates. Gut Microbes 12, 1–16. doi: 10.1080/19490976.2020.1826747
Vatne, A., Hapnes, N., Stensvold, H. J., Dalen, I., Guthe, H. J., Støen, R., et al. (2023). Early empirical antibiotics and adverse clinical outcomes in infants born very preterm: A population-based cohort. J. Pediatr. 253, 107–114.e5. doi: 10.1016/j.jpeds.2022.09.029
Wampach, L., Heintz-Buschart, A., Fritz, J. V., Ramiro-Garcia, J., Habier, J., Herold, M., et al. (2018). Birth mode is associated with earliest strain-conferred gut microbiome functions and immunostimulatory potential. Nat. Commun. 9, 5091. doi: 10.1038/s41467-018-07631-x
Westerbeek, E., Slump, R. A., Lafeber, H. N., Knol, J., Georgi, G., Fetter, W. P. F., et al. (2013). The effect of enteral supplementation of specific neutral and acidic oligosaccharides on the faecal microbiota and intestinal microenvironment in preterm infants. Eur. J. Clin. Microbiol. Infect. Dis. 32, 269–276. doi: 10.1007/s10096-012-1739-y
Wilson, B. C., Butler, É. M., Grigg, C. P., Derraik, J. G. B., Chiavaroli, V., Walker, N., et al. (2021). Oral administration of maternal vaginal microbes at birth to restore gut microbiome development in infants born by caesarean section: A pilot randomised placebo-controlled trial. EBioMedicine 69, 103443. doi: 10.1016/j.ebiom.2021.103443
Yang, W., Tian, L., Luo, J., Yu, J. (2021). Ongoing Supplementation of Probiotics to Cesarean-Born Neonates during the First Month of Life may Impact the Gut Microbial. Am. J. Perinatol. 38, 1181–1191. doi: 10.1055/s-0040-1710559
Yasmin, F., Tun, H. M., Konya, T. B., Guttman, D. S., Chari, R. S., Field, C. J., et al. (2017). Cesarean section, formula feeding, and infant antibiotic exposure: Separate and combined impacts on gut microbial changes in later infancy. Front. Pediatr. 5. doi: 10.3389/fped.2017.00200
Zhong, H., Wang, X.-G., Wang, J., Chen, Y.-J., Qin, H.-L., Yang, R. (2021). Impact of probiotics supplement on the gut microbiota in neonates with antibiotic exposure: an open-label single-center randomized parallel controlled study. World J. Pediatrics: WJP 17, 385–393. doi: 10.1007/s12519-021-00443-y
Keywords: newborn, intestinal flora, caesarean section, premature delivery, breast milk
Citation: Ma G, Shi Y, Meng L, Fan H, Tang X, Luo H, Wang D, Zhou J and Xiao X (2023) Factors affecting the early establishment of neonatal intestinal flora and its intervention measures. Front. Cell. Infect. Microbiol. 13:1295111. doi: 10.3389/fcimb.2023.1295111
Received: 15 September 2023; Accepted: 17 November 2023;
Published: 01 December 2023.
Edited by:
Suhana Chattopadhyay, University of Maryland, College Park, United StatesReviewed by:
Kathyayini Gopalakrishna, California Institute of Technology, United StatesCopyright © 2023 Ma, Shi, Meng, Fan, Tang, Luo, Wang, Zhou and Xiao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiaomin Xiao, am51eGlhb3hpYW9taW5AMTYzLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.