- 1Institute of Metabolic Diseases, Guang’anmen Hospital, China Academy of Chinese Medical Sciences, Beijing, China
- 2Graduate College, Beijing University of Chinese Medicine, Beijing, China
Traditional Chinese herbal medicine often exerts the therapeutic effect of “treating different diseases with the same method” in clinical practice; in other words, it is a kind of herbal medicine that can often treat two or even multiple diseases; however, the biological mechanism underlying its multi-path and multi-target pharmacological effects remains unclear. Growing evidence has demonstrated that gut microbiota dysbiosis plays a vital role in the occurrence and development of several diseases, and that the root cause of herbal medicine plays a therapeutic role in different diseases, a phenomenon potentially related to the improvement of the gut microbiota. We used local intestinal diseases, such as ulcerative colitis, and systemic diseases, such as type 2 diabetes, as examples; comprehensively searched databases, such as PubMed, Web of Science, and China National Knowledge Infrastructure; and summarized the related studies. The results indicate that multiple individual Chinese herbal medicines, such as Rhizoma coptidis (Huang Lian), Curcuma longa L (Jiang Huang), and Radix Scutellariae (Huang Qin), and Chinese medicinal compounds, such as Gegen Qinlian Decoction, Banxia Xiexin Decoction, and Shenling Baizhu Powder, potentially treat these two diseases by enriching the diversity of the gut microbiota, increasing beneficial bacteria and butyrate-producing bacteria, reducing pathogenic bacteria, improving the intestinal mucosal barrier, and inhibiting intestinal and systemic inflammation. In conclusion, this study found that a variety of traditional Chinese herbal medicines can simultaneously treat ulcerative colitis and type 2 diabetes, and the gut microbiota may be a significant target for herbal medicine as it exerts its therapeutic effect of “treating different diseases with the same method”.
1 Introduction
In the past decade or so, with the development of research projects such as the Human Microbiome Project and Human Intestinal Metagenome Project (Metagenomies of the Human Intestinal Tract, MetaHIT), human cognition in the field of gut microbiota has made significant progress, and the publication of relevant research results has increased exponentially (Illiano et al., 2020). Based on the evidence gathered so far, the impact of gut microbiota on health is profound and complex. Healthy gut microbiota not only prevent potentially harmful bacteria in the gut from entering the blood circulation, but also regulate metabolism and the immune system as well as provide nutrients and energy to the body. Furthermore, disorders associated with the gut microbiota are involved in the occurrence and development of various diseases, and altering this state by adjusting the diet, taking intestinal microecological agents, and even flora transplantation potentially reverse the development process of a variety of diseases to a certain extent, thus providing a novel idea for clinical treatment (Valdes et al., 2018).
Ulcerative colitis (UC) and type 2 diabetes mellitus (T2DM) are diseases closely linked to the state of the gut microbiota. As an important type of inflammatory bowel disease (IBD), UC is characterized by a continuous inflammatory response in the colorectal mucosa, which involves the rectum and damages the colon to varying degrees. Clinically, recurrent attacks and relief of alternating diarrhea, abdominal pain, mucus, pus, and blood stool are the main symptoms (Ananthakrishnan, 2015). T2DM is the main type of diabetes (accounting for > 90%), and its initial elevated blood sugar is often caused by insulin resistance. Over time, insulin production by islet beta cells gradually becomes insufficient, resulting in persistent hyperglycemia (Cho et al., 2018). Globally, the incidence of both diseases is increasing annually, and clinical evidence reveals that UC can significantly increase the risk of T2DM (Li et al., 2021c), which may be related to immune inflammation caused by intestinal flora disturbance (Jurjus et al., 2016).The combination of UC and T2DM is a difficult clinical problem. On the one hand, glucocorticoids, the first-choice treatment for UC, potentially aggravate blood-glucose fluctuation and render blood-glucose control difficult; on the other hand, hyperglycemia also makes it increasingly difficult to repair the damaged intestines of patients with UC, thus posing hidden threats to postoperative recovery (Maconi et al., 2014).
Traditional Chinese medicine (TCM) has a long history of application in China and East Asia and has significant advantages in the treatment of various complex diseases. “Treating different diseases using the same method” is an important constituent of the theoretical system of TCM; that is, when two or more diseases have the same core pathogenesis, TCM doctors may prescribe similar or exact treatments. In recent years, researchers have conducted several studies to explore the scientificity; for example, network pharmacology research has found that different diseases have related or the same pathogenesis, and TCM can play a therapeutic role in two or more diseases by regulating these core targets (Su et al., 2022). In addition, several omics studies have explored and confirmed the common characteristics of different diseases from the perspective of genes, proteins, and metabolites, and regulating these key targets is of great significance in disease treatment (Chang et al., 2018; Yang, 2019; Liu et al., 2020). Moreover, we found gut microbiota disorders to also be related to the emergence and development of a variety of diseases, and restoration of the intestinal microecology may also be a potentially important link to TCM‘s therapeutic role in “treating different diseases using the same method”.
Accumulating evidence suggests that many herbal medicines or their herbal compounds, such as Rhizoma Coptidis and Gegen Qinlian decoction, potentially play a therapeutic role in UC and T2DM (Zhang et al., 2013; Tian et al., 2019), and their pharmacological effects are related to the regulation of the gut microbiota. However, due to the lack of a comprehensive summary of relevant research, the utility of gut microbiota as a potential target to explain the effects of “treating different diseases with the same method” is yet to be fully elucidated. Therefore, the purpose of this review is to use UC and T2DM as examples to explain the role of the gut microbiota in the multi-pathway treatment mechanisms of TCM.
2 The Relationship Between UC and T2DM
2.1 Clinical Evidence of the Relationship Between the Two Diseases
Several studies have reported a correlation between UC onset and T2DM. A Danish, population-based cohort study found that patients with UC were significantly more likely to develop T2DM, and this risk was highest in the first year of IBD diagnosis and could persist for more than two decades (Jess et al., 2020); meanwhile, two cross-sectional studies also found the odds of suffering from T1D to increase significantly in patients with UC, a phenomenon possibly related to immune-system dysregulation (Kappelman et al., 2011; Halling et al., 2017). Moreover, a meta-analysis of five clinical studies confirmed that UC was associated with an increased risk of diabetes (odds ratio [OR]/relative risk [RR] = 1.33, 95% confidence interval [CI]: 1.03 to 1.71) (Jurjus et al., 2016). However, a retrospective cohort study from South Korea found that Crohn’s disease may increase the risk of developing diabetes; however, there was no statistical difference in patients with UC (Kang et al., 2019). In addition, UC may also promote the occurrence of other metabolic disorders besides diabetes, such as obesity, metabolic syndrome, and non-alcoholic fatty liver disease, among others (Hyun, 2021). What is the reason for this pathological phenomenon? To reveal the causes of this pathological phenomenon, researchers explored the topic from the perspective of genetics and found that there was some overlap in the susceptibility genes of patients with IBD and those of patients with metabolic diseases, such as diabetes (Lees et al., 2011).
2.2 Multiple Hypoglycemic Drugs Have Therapeutic Potential for UC
A retrospective study from Taiwan, China, found that patients with diabetes taking metformin had a significantly lower risk of developing UC, and a series of experimental studies found metformin to have a therapeutic effect on UC (Tseng, 2021), possibly by reducing intestinal tissue inflammation, maintaining normal mitochondrial structure, and protecting intestinal barrier integrity by regulating the TGF-β, NF-κB, LKB1/AMPK, and JNK pathways (Pandey et al., 2017; Deng et al., 2018; Samman et al., 2018; Wang et al., 2019; Liu et al., 2021). Another study found that the mucosal protective effect of metformin in UC mouse models was associated with an increased profusion of Akkermansia muciniphila (Ke et al., 2021). In addition, the simultaneous use of metformin and heat shock protein 90 potentially modulates HSP90/NLRP3 interaction and induces autophagy, thereby inhibiting colonic inflammation (Saber and El-Kader, 2021). Moreover, the co-administration of metformin and another hypoglycemic drug, empagliflozin, potentially exerts pharmacological effects on UC through the AMPKα/mTOR/NLRP3 signaling pathway and IL-23/IL-17 axis (Youssef et al., 2021).
In addition to metformin, other antidiabetic drugs may have potential therapeutic actions for UC. A randomized controlled clinical study confirmed that rosiglitazone, a thiazolidinedione hypoglycemic drug, was effective in the treatment of mild to moderately active UC (Lewis et al., 2008); however, a retrospective study did not support the conclusion that thiazolidinediones can treat UC (Lund et al., 2011). In addition, gliclazide, an insulin secretagogue, has also been shown to improve intestinal inflammation in UC rats via the PPARγ, NF-κB, and MAPK signaling pathways (Arafa et al., 2020). Several studies have also focused on the therapeutic potential of GLP-1 receptor agonists in UC, and Bang-Berthelsen et al. confirmed that GLP-1R mRNA tends to decrease by observing tissue samples from the colonic inflammation area of patients with IBD. GLP-1R-knockout IBD-model mice also showed the abnormal proliferation of microbial species in their feces and were more prone to intestinal damage, whereas the GLP-1 receptor agonist Exendin-4 and human GLP-1 analog liraglutide potentially reduced colonic inflammation through immunomodulation (Zatorski et al., 2019).
2.3 The Common Phenotypes Between UC and T2DM
2.3.1 Gut Microbiota
A substantial body of research has provided evidence for the role of gut microbiota in the progression of UC and T2DM. The proportion of certain anti-inflammatory butyrate-producing bacteria, such as Faecalibacterium prausnitzii and Roseburia spp., decreased in the gut of patients with UC and animal models. Although some proinflammatory bacteria, such as Fusobacterium and Escherichia coli, were enriched, the diversity of the gut microbiota also demonstrated a downward trend (Pavel et al., 2021). Butyrate is a type of short-chain fatty acid (SCFA) that plays a protective role in the intestinal barrier and maintains the barrier integrity of epithelial cells by regulating the proliferation and development of intestinal epithelial cells; due to the imbalance of the microbiota structure, the total content of butyrate in the gut of patients with UC also decreases (Xu et al., 2021b). In people with T2DM, there are certain similarities in gut-microbiota changes. The abundance of some beneficial bacteria, such as Bifidobacterium, Bacteroides, Roseburia, Faecalibacterium, and Akkermansia, showed a downward trend (Xu et al., 2021b). However, numerous other opportunistic pathogens showed an upward trend (Qin et al., 2012). Similarly, the abundance of butyrate-producing bacteria and content of butyrate in the intestinal tract also tended to decrease in some prediabetic and T2DM populations; when this trend was reversed, the disturbance of glycometabolism also improved (Arora and Tremaroli, 2021). In addition, other intestinal metabolites, such as tryptophan and bile acids, also cause disorders in patients with UC and T2DM (Liu et al., 2022).
2.3.2 Intestinal Barrier
The intestinal barrier comprises mechanical, chemical, immune, and biological barriers. Once the intestinal mucosa is damaged, bacteria and their derived toxins may infiltrate the blood circulation, enter the peripheral tissues, and induce metabolic inflammatory reactions. Intestinal-barrier damage in individuals with UC includes not only apoptosis, necrosis, and necroptosis in intestinal epithelial cells, but also disorders in tight junction protein expression, such as decreases in ZO-1 and claudin-4 and an increase in claudin-2 (Hyun, 2021). This pathological damage also occurs in individuals with T2DM. Through a retrospective analysis of capsule endoscopy data of patients with T2DM, Zhong et al. found that the incidence of intestinal villous edema and Lewis score (obtained based on intestinal villous edema, ulceration, and pathological strictures) were significantly higher than those in non-T2DM patients, and the Lewis score was positively correlated with the severity of insulin resistance (Zhong et al., 2016). Lipopolysaccharides (LPS) are important components of the cell wall of gram-negative bacteria and are released during the death and lysis of bacteria. Under physiological conditions, low concentrations of non-pathogenic LPS can be detected in circulation; however, in the state of gut-microbiota dysbiosis and intestinal mucosal damage, LPS molecules proliferate in the intestine and “leak” into the blood circulation (Cani et al., 2012). Through a prospective clinical analysis, Shen et al. found the serum LPS level in patients with T2DM to be significantly higher than that in non-T2DM patients, and LPS was positively correlated with blood-glucose fluctuation, indicating that poor blood sugar control in the short term potentially causes damage to the intestinal mucosa (Shen et al., 2019). Similarly, animal experiments further confirmed that the T2DM model also had abnormal tight junction protein expression and chronic immune inflammation in the intestinal tissue (Zhang et al., 2020); nonetheless, the degree of intestinal injury was far less than that in UC.
2.3.3 Chronic Inflammation
UC pathogenesis is the result of a combination of multiple pathogenic factors, and diffuse mucosal inflammation is the main symptom (Dai et al., 2021). Recent studies have found the abnormal production of inflammatory cytokines and activation of related signaling pathways in individuals with T2DM, and gut-microbiota disorders and intestinal-barrier injury both play important roles in the pathological process of inflammation, similar to that in UC (Hotamisligil, 2006). Normally, the inflammatory response in UC is limited to the colon and rectum, while chronic inflammation in T2DM is systemic, especially in tissues or organs closely related to the pathogenesis of T2DM, such as the liver, pancreas, and fatty tissue. There are numerous overlapping inflammatory signaling pathways involved in UC and T2DM, of which the nuclear factor kappa B (NF-κB)-related pathway is representative (Jurjus et al., 2016). NF-κB plays a central role in the development of various chronic inflammatory diseases. In the resting state, NF-κB bound to the kappa-B (I-κB) inhibitor is blocked in the cytoplasm in an inactive form; however, under the influence of bacterial components (e.g., LPS), proinflammatory cytokines (e.g., TNF-α, IL-1, etc.), or viruses, the inhibitor of NF-κB kinase is activated, inducing the phosphorylation of I-κB and promoting the nuclear translocation of NF-κB (Ben-Neriah and Karin, 2011). In individuals with UC, elevated NF-κB levels in the nucleus of colonic tissue promote the release of a series of proinflammatory factors encoded by the NF-κB signaling pathway, aggravate the local inflammatory response and tissue damage, and destroy the integrity of the intestinal barrier (Lu and Zhao, 2020). In the T2DM model, increased NF-κB levels widely exist in the intestine, pancreas, and insulin-target organs (Zhang et al., 2020), and are closely related to chronic inflammation, oxidative stress, insulin resistance, and several complications (Sahukari et al., 2020). In addition, multiple other inflammatory signaling pathways may be involved in these two diseases, such as transforming growth factor-β (TGF-β), tumor necrosis factor-α (TNF-α), and peroxisome proliferator-activated receptor (PPAR), among others (Jurjus et al., 2016; Hong et al., 2018).
2.4 Summary
Overall, clinical studies have found a significant correlation between the occurrence of UC and T2DM, and antidiabetic drugs such as metformin can also play a significant role in the treatment of UC. In addition to genetic factors, gut-microbiota disturbance may be the “common ground” that leads to the pathogenesis of both UC and T2DM, and it not only promotes the occurrence of local inflammation in the intestinal tract, but also damages the intestinal mucosal barrier, leading to the “leakage” of LPS and the occurrence of chronic systemic inflammatory response. Thus, targeting the gut microbiota potentially exerts therapeutic effects in both UC and T2DM.
3 TCM Research Progress in the Treatment of UC and T2DM
To comprehensively summarize the progress of TCM research regarding the treatment of UC and T2DM simultaneously, we searched databases such as PubMed, Web of Science, and China National Knowledge Infrastructure and identified individual herbs or Chinese herbal formulations (Tables 1, 2), and thoroughly excavated molecular mechanisms from the perspective of gut microbiota, intestinal barrier, and inflammatory response.
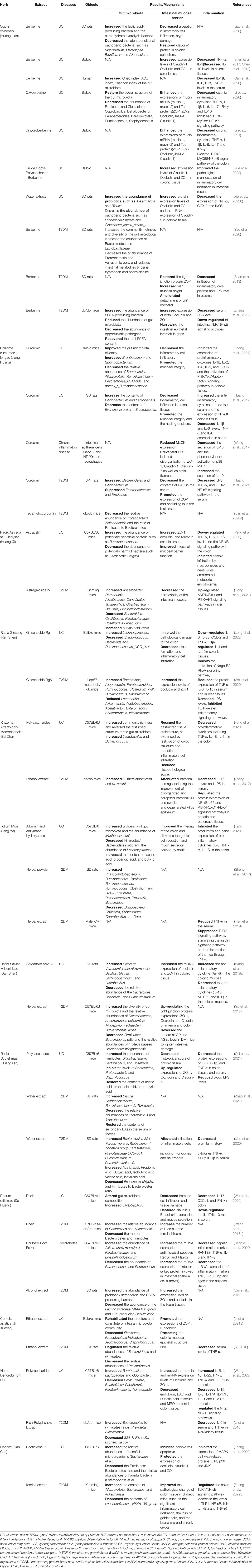
Table 1 Research progress of individual herb or herbal extracts for the simultaneous treatment of UC and T2DM.
3.1 Individual Herbs or Herbal Extracts
Berberine and curcumin are well-known herbal extracts whose hypoglycemic effects have been confirmed by multiple clinical studies (de Melo et al., 2018; Liang et al., 2019); moreover, these two extracts have also proven to improve intestinal inflammation (Zhang et al., 2016; Li et al., 2020). Berberine is an active alkaloid isolated from Rhizoma coptidis, and the regulation of the intestinal microbiota is a key target for berberine’s multiple pharmacological effects (Habtemariam, 2020; Cheng et al., 2021). In animal models of UC and T2DM, berberine increased the number of beneficial bacteria and reduced the potential pathogenic bacteria, thereby reducing inflammatory cell infiltration, repairing the intestinal mucosal barrier, and playing a role in the treatment of UC (Liao et al., 2020). In addition, it potentially reduces serum LPS levels, thus alleviating the chronic inflammatory state by downregulating the TLR/NF-κB signaling pathway (Zhang et al., 2019). Curcumin, a polyphenol extracted from Rhizoma curcumae longae, can repair dysregulated gut microbiota and exert various pharmacological effects. On the one hand, it can regulate the balance of Treg/Th17 cells in patients with UC and inhibit the expression of inflammatory factors, such as IL-6 and IL-17A (Zhong et al., 2021); on the other hand, it also prevents the LPS “leakage,” inhibits the occurrence of systemic inflammatory responses, and protects pancreatic islets and insulin target organs (Huang et al., 2021).
In the TCM theory system, Radix astragali seu Hedysari, Rhizoma atractylodis macrocephalae, and Radix ginseng are considered to have the effect of “invigorating qi” and potentially treat various debilitating diseases. Radix astragali seu Hedysari is a perennial Compositae plant, and its extract has been shown to increase butyrate-producing bacteria in the intestine, modulate the PI3K signaling pathway in T2DM mice (Gong et al., 2021), and repair the damaged intestinal mucosal barrier in mice with UC by reducing the pathogenic bacteria Escherichia spp. and Shigella spp. (Peng et al., 2020). Rhizoma atractylodis macrocephalae is often used as a beneficial spleen herb in China because of its unique advantages in improving gastrointestinal dysfunction (Yang et al., 2021a). Polysaccharides are important active ingredients of Atractylodes macrocephala, and Feng et al. found Atractylodes polysaccharides to increase beneficial bacteria, such as Lactobacillus and Butyricicoccus, affect the metabolic process of amino acids and bile acids, and reverse the damaged intestinal-tissue structure in UC model mice (Feng et al., 2020); in addition, its ethanol extract can also repair intestinal mucosal injury and reduce serum IL-1β and LPS levels in T2D mice (Zhang et al., 2017). Saponins are the main active components of ginseng and have anti-inflammatory and metabolic regulatory effects (Gao et al., 2020). In mouse models of UC and T2D, ginsenosides regulate gut microbiota, thereby improving intestinal mucosal damage and a series of inflammatory reactions (Wei et al., 2020; Long et al., 2022).
Furthermore, some herbs, such as Folium mori, Radix salviae miltiorrhizae, and Radix scutellariae, among others, also have dual therapeutic effects on UC and T2D, and more details are summarized in Table 1.
3.2 Chinese Herbal Formulae
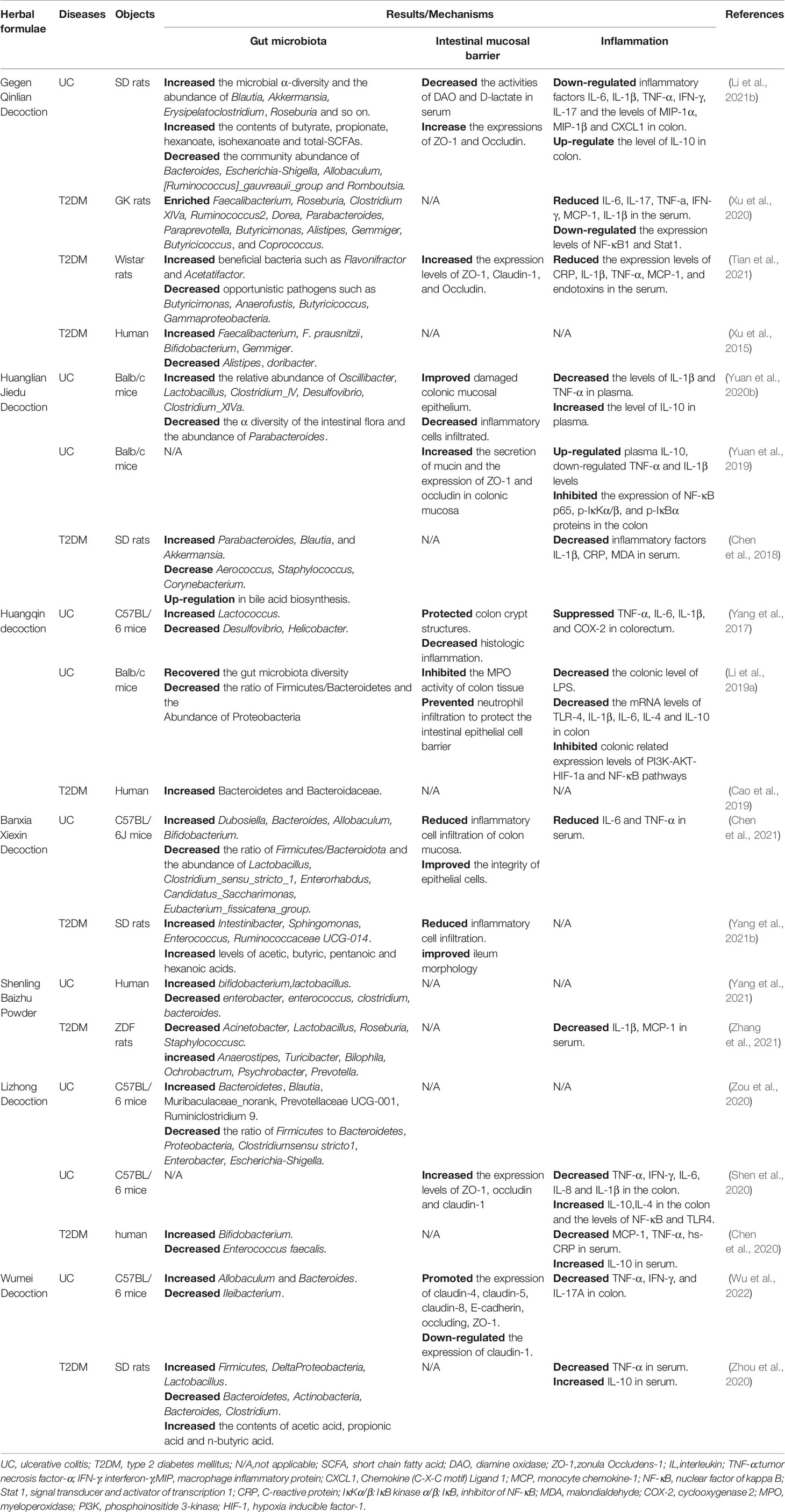
Chinese herbal formulae are an ingenious combination of several herbs under the guidance of TCM theory, which are characterized by multicomponent, multitarget, and synergistic effects (Qiu, 2007). Gegen Qinlian decoction (GGQLD), Huanglian Jiedu decoction (HLJDD), and Huangqin decoction (HQD) are considered to have the “clearing heat” effect, and modern research suggests that they have obvious anti-inflammatory effects and could be widely used in acute or chronic inflammatory diseases (Gao et al., 2017). GGQLD was first recorded in the book Treatise on Febrile and Miscellaneous Diseases written by Zhongjing Zhang, mainly comprising Gegen (Radix puerariae), Huangqin (Radix scutellariae), Huanglian (Rhizoma coptidis), and Gancao (Radix glycyrrhizae). The effects of GGQLD on the treatment of UC and T2DM are closely related to gut-microbiota regulation, and in the dextran sulfate sodium (DSS)-induced UC rat model, it potentially increased the relative abundance of the Lachnospiraceae_NK4A136_group and Roseburia (Li et al., 2021); in a T2DM rat model, it potentially increased butyric acid-producing bacteria, such as Faecalibacterium, Roseburia, Clostridium XIVa, and beneficial bacteria, such as Flavonifractor and Acetatifactor (Xu et al., 2020; Tian et al., 2021). In addition, GGQLD may also increase the expression of ZO-1 and occludin and downregulate inflammatory factors IL-6, IL-1β, TNF-α, IFN-γ, and IL-17 in both disease models, thereby repairing the intestinal mucosal barrier and reducing the level of inflammation in the intestine and circulation. Additionally, Shenling Baizhu Powder (SLBZP) and Lizhong Decoction (LZD) are considered to have the effect of “invigorating the spleen and regulating the stomach”, while Banxia Xiexin Decoction and Wumei Decoction are the representative formulae for treating diseases characterized by the cold-heat complex. In modern clinical practice, the above formulae have been widely used in various gastrointestinal and metabolic diseases, and the gut microbiota may be a potential key target. More details are listed in Table 2.
4 Discussion
4.1 Gut Microbiota: A Potential Key Target of Herbal Medicine in the Simultaneous Treatment of UC and T2DM
Over 1,000 species of gut microbiota constitute a complex ecosystem that interacts with the host, and some pathological changes, such as the imbalance of flora structure, proliferation of pathogenic bacteria, and disorder of flora metabolites, may disturb the host’s homeostasis, leading to the occurrence of a variety of diseases (Geng et al., 2022). By analyzing the above articles, it is evident that the regulatory effects of TCM on the gut microbiota are mainly manifested in the following aspects (Figure 1).
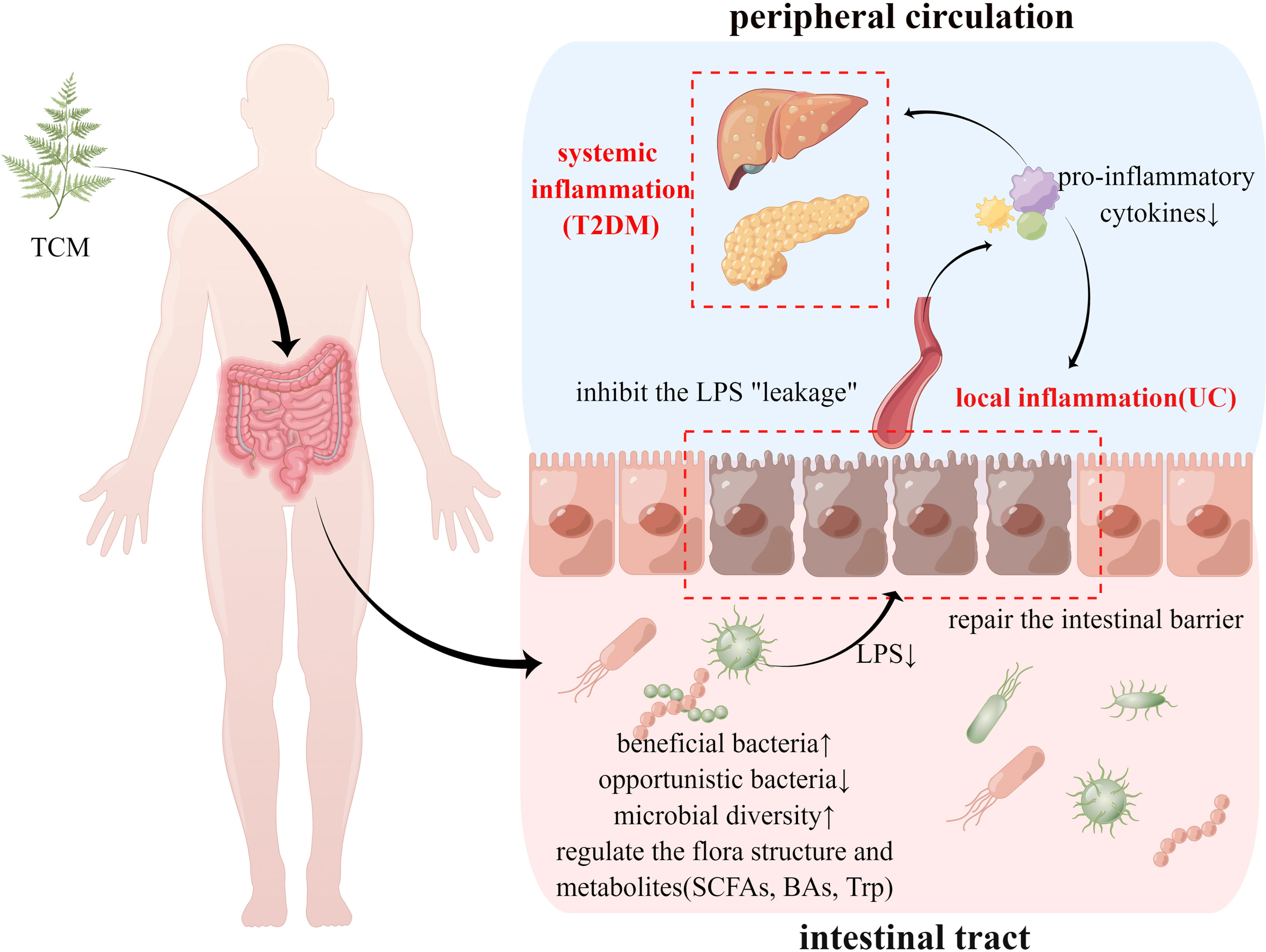
Figure 1 TCM’s therapeutic effect of treating UC and T2DM using the same method. TCM, traditional Chinese medicine; UC, ulcerative colitis; T2DM, type 2 diabetes mellitus; LPS, lipopolysaccharide; SCFAs, short chain fatty acids; BAs, bile acids; Trp, tryptophan.
4.1.1 Regulation of the Microbiota Structure
The diversity of the gut microbiota form the basis of its physiological functions, and a reduction in diversity has been confirmed to be closely related to the occurrence of various diseases. Both clinical and basic studies have confirmed that some patients with UC and UC mouse models induced by DSS decrease gut microbiota diversity (Xu et al., 2021a; Zhuang et al., 2021), and after exclusive enteral nutrition and chemical drug treatment, α-diversity indicators show an upward trend (Radhakrishnan et al., 2022). Similarly, decreased diversity has been observed in individuals with T2DM and in high-fat-fed mouse models (Nyavor et al., 2019; Balvers et al., 2021). As described in Tables 1, 2, most TCMs, such as curcumin and Rhizoma atractylodis macrocephalae, restore the diversity of flora, which may be one of the factors for treating different diseases using the same method. However, berberine (the main component of Rhizoma Coptidis) was confirmed to reduce bacterial diversity in both UC and T2DM models, possibly be due to its strong antibacterial efficacy (Li et al., 2021b), and its therapeutic effect on UC and T2DM may be similar to that of antibiotics. In addition, Huanglian Jiedu decoction, which uses Huanglian as the main drug, also reduced the bacterial diversity in UC mice.
Studies have demonstrated that the increased abundance of Firmicutes is associated with the development of obesity, and an upward trend in high-fat-fed animal models of T2DM has also been observed (Abenavoli et al., 2019). Some clinical studies have confirmed that the Bacteroidetes population is reduced in patients with UC, and this status could also be altered by microbiota transplantation; however, some studies have drawn contrary conclusions (Shi et al., 2016). As shown in the table, some TCMs have a clear regulatory effect on Firmicutes and Bacteroides, and most of them reduce the Firmicutes/Bacteroides ratio, which may be related to their therapeutic effect on the disease; however, considering the complexity of the gut microbiota, this conclusion requires further investigation.
4.1.2 Increasing Beneficial Bacteria
Bifidobacterium is an important probiotic that exists in human and animal intestines and is closely related to a variety of physiological and pathological phenomena in the body. In both UC and T2DM, the abundance of Bifidobacterium has exhibited a downward trend (Prosberg et al., 2016; Gurung et al., 2020), and probiotics containing Bifidobacterium potentially play an adjuvant role in the treatment of these two diseases (Tiderencel et al., 2020; Yao et al., 2021), which may be related to the regulation of intestinal immunity, inhibition of mucosal inflammation and oxidative stress, and protection of the intestinal barrier (Hampe and Roth, 2017). As indicated in the tables, the mechanism of action of curcumin and some prescriptions, such as SLBZP and LZD, on UC may be related to the regulation of Bifidobacterium spp. Akkermansia muciniphila, which has been a research topic of interest in recent years, can increase the integrity of the intestinal barrier by activating the Toll-like receptor 2 signaling pathway and promoting the production of IL-10, increasing the level of endocannabinoids in the intestine, among other substances. (Everard et al., 2013; Naito et al., 2018) Similar to that of Bifidobacterium, the Akkermansia muciniphila population was also significantly reduced in UC and T2DM individuals, and some Chinese herbal extracts, such as berberine and Rhein, could effectively increase the abundance of this bacterium in the gut. Lactobacillus is also a probiotic with various effects, such as mucosal protection and promotion of intestinal peristalsis. Compared to inactive UC, the abundance of Lactobacillus in the intestinal tract of active UC has also shown a downward trend (Prosberg et al., 2016), and several Chinese herbal extracts potentially promote the proliferation of Lactobacillus in the UC model, which may also be an important factor for the efficacy of Chinese herbs. Moreover, we noticed that although some Lactobacillus species are also beneficial for the treatment of T2DM (Miraghajani et al., 2017), the Chinese herbs and Chinese herbal formulations listed in Tables 1, 2 did not increase the abundance of Lactobacillus in the T2DM model.
4.1.3 Inhibition of Opportunistic Pathogens
Opportunistic pathogens in the gut microbiota are also important targets of TCM in the treatment of UC and T2DM. The impact of pathogenic Escherichia-Shigella on the host cannot be ignored; it not only affects the host through virulent factors, such as enterotoxins, adhesin fimbriae, and Shigella-like toxins, but also potentially damages the intestinal mucosa and affects the expression of intestinal tight junction proteins by destroying the host-cell actin cytoskeleton and stimulating the secretion of intestinal pro-inflammatory cytokines (Pawłowska and Sobieszczańska, 2017). In addition, the abundance of the opportunistic pathogens Enterobacter and Enterococcus was also negatively correlated with glucose and lipid metabolism and inflammatory indicators (Fei and Zhao, 2013; Patterson et al., 2016). TCM extracts, such as berberine and curcumin, and formula LZD potentially reduce the abundance of these bacteria, which may be the key factor for their therapeutic effect.
4.1.4 Regulation of Gut Microbiota Metabolites
In addition to the gut microbiota, the relationship between gut-related metabolites and diseases has attracted extensive interest from researchers (Liu et al., 2022). Butyric acid, an important component of SCFAs, plays an important role in inhibiting intestinal inflammation and maintaining intestinal mucosal barrier integrity. The protective effects of butyric acid on the intestinal barrier are multifaceted. First, it can activate G protein-coupled receptors, which in turn activate their downstream signaling pathways and affect the differentiation and migration of intestinal immune cells. Second, butyrate can also activate PPARs; promote the expression of tight junction proteins; ensure intestinal barrier function; inhibit the activation of the NF-κB signaling pathway, that is, the expression of inflammatory factors; and promote the secretion of intestinal antimicrobial peptides (Silva et al., 2018). In addition to its effects on the gut, butyrate can directly regulate the host’s glucose and lipid metabolism, body weight, and appetite, among others, and plays an important role in maintaining the host’s metabolic homeostasis (Zhang et al., 2021a). In the intestines of patients with UC and T2DM, the content of butyric acid and abundance of butyrate-producing bacteria tend to decrease, and the intake of dietary fiber, prebiotics/probiotics, or direct supplementation with butyric acid preparations all potentially play an adjuvant-treatment role (Coppola et al., 2021; Zhang et al., 2021a). The tables show that some Chinese herbs not only directly increase the content of butyric acid in the intestine but also promote the proliferation of the following butyric acid-producing bacteria: Prevotella, Faecalibacterium, Butyricicoccus, and Roseburia, thus exerting therapeutic effects in both diseases.
Tryptophan (Trp) is also a key gut metabolite that can affect various intestinal and metabolic diseases (Agus et al., 2018). The aromatic hydrocarbon receptor (AhR) ligand, the metabolite produced by intestinal Trp-decomposing microorganisms, showed a decreasing trend in the intestines of patients with IBD (Lamas et al., 2016), and recent studies have confirmed that, as a key immunomodulator, AhR ligands potentially induce a variety of cellular and epigenetic mechanisms to attenuate inflammation (Cannon et al., 2021). Trp is also important for glucose and lipid metabolism in the host. A clinical study found that the levels of hemoglobin A1c, total cholesterol, low-density lipoprotein cholesterol, and apolipoprotein B-100 were lower in the high tertile of Trp than in the low tertile (Wang et al., 2022). Indolepropionic acid and indoleacrylic acid are substances commonly formed after Trp decomposition, both of which are beneficial for restoring the function of the intestinal epithelial barrier and reducing the inflammatory response. Another study found that higher serum IPA levels were associated with a reduced risk of developing T2DM and improved insulin secretion (Ballan and Saad, 2021). In addition, abnormalities in synthesis, metabolism, and related signal-transduction pathways of bile acids (BAs) have been observed in patients with UC and T2DM. As the cometabolite of “host and gut microbiota,” BAs potentially promote the repair of intestinal mucosa and affect the secretion of intestinal hormones (e.g., GLP-1) through takeda G protein-coupled receptor 5 (TGR 5) and farnesoid X receptor (FXR) pathways (Bromke and Krzystek-Korpacka, 2021; Xie et al., 2021). LPS molecules are vital components of the outer membrane of Gram-negative bacteria. Excessive LPS levels in the intestinal tract and circulation can trigger an immune inflammatory response and lead to the progression of UC and T2D (Usuda et al., 2021). Overall, the imbalance of the above gut-related metabolites/derivatives caused by gut microbiota disorders is a common pathological link between UC and T2D and is also a potential target of TCM’s therapeutic effects.
4.2 Improvement of the Intestinal Barrier and Inflammatory Response: Important Links for Herbal Medicine in Treating UC and T2DM
The intestinal barrier effectively prevents harmful substances in the intestine, such as pathogenic microorganisms, various biological macromolecules, and antigens, from entering the blood circulation. In the above studies, intestinal tight junction proteins were the most commonly used indicators for assessing intestinal damage. When stimulated by factors such as inflammation and oxidative stress, the protein function is damaged and the density of the intestinal barrier is reduced, leading to the “leakage” of inflammatory substances and LPS molecules into the blood circulation, resulting in the occurrence of chronic systemic inflammation (Monaco et al., 2021). Occludin and claudin are transmembrane proteins and ZO is a peripheral membrane scaffold protein, all of which are indispensable components of intestinal tight junctions (Vancamelbeke and Vermeire, 2017). In the studies listed in the tables, various Chinese herbs appeared to modulate the expression of these proteins and play a role in enhancing intestinal permeability.
During UC onset, the production of inflammatory cytokines, such as TNF-α and INF-γ, by lymphocytes and lamina propria immune cells in the inflammatory intestinal epithelium potentially induces the expression of apoptosis-related proteins, such as caspase-1 in the epithelial cells, while inhibiting the expression of anti-apoptotic proteins, such as Bcl-2, thus inducing apoptosis of epithelial cells. At this time, the epithelial cells adjacent to the apoptotic cells cannot effectively seal the space left by the apoptotic cells; that is, the intestinal tight junctions are damaged and intestinal mucosal permeability is increased (Xie et al., 2022a). Presumably, intestinal mucosal damage is an inevitable result of an immune inflammatory response in patients with UC, thus eventually leading to the formation of a vicious circle between the two. However, in the T2DM model, increased permeability of the intestinal barrier is an important inducer of systemic inflammation, and LPS is an important mediator of the connection between the intestine and peripheral tissues. Studies have confirmed that CD14, an important component of the LPS-receptor complex, and its related molecules, TLR-2, TLR-4, and MD-2, are expressed in human and rat islets (Vives-Pi et al., 2003), and the activation of the receptor triggers the accumulation of numerous inflammatory factors through signaling pathways, such as NF-κB, which subsequently impair the normal secretion of insulin by downregulating the expression of PDX-1 and MafA (Amyot et al., 2012). In addition, LPS can also release inflammatory factors by combining with corresponding receptors in insulin target organs, such as the liver, and fat tissue, thereby disrupting the insulin signaling pathway and insulin receptor expression, ultimately reducing insulin efficacy and leading to the occurrence of T2DM (Singer-Englar et al., 2019).
5 Conclusion
TCM potentially plays a therapeutic role in different diseases; however, the underlying principle has not been explained from the perspective of modern science. In this study, we comprehensively searched Chinese medicines, their extracts, and Chinese medicine compounds that can treat both UC and T2DM and found that the gut microbiota and “gut-inflammation” axis may be the key targets and important ways to achieve this pharmacological effect. In particular, the effects of most TCMs on the microbiota were related to modulating the microbiota structure, increasing the abundance of beneficial bacteria, increasing the concentration of intestinal butyrate, and inhibiting the proliferation of opportunistic pathogens. Notwithstanding, this review has the following limitations. First, most of the research conclusions listed in this review are based on animal experiments, and there is an urgent need for further evidence from human studies. Second, there is a complex interaction between the gut microbiota and host, and the causal relationship between disease improvement and the recovery of disrupted gut microbiota after intervention with TCM remains unclear. Therefore, the preliminary conclusion of TCM treating UC and T2DM by improving the gut microbiota warrants further investigation, such as the use of sterile animal models.
In fact, there are many examples of certain herbal medicines treating different diseases simultaneously. For example, Coptis chinensis can treat hyperglycemia, dyslipidemia, and metabolic-related fatty liver disease simultaneously, and through bidirectional regulation of gastrointestinal motility, Astragalus can effectively treat diarrhea and constipation. The impact of gut dysbiosis on health is multifaceted, and alterations in certain bacteria are associated with several different clinical phenotypes. Modulation of the gut microbiota potentially has therapeutic effects on a variety of diseases. The comprehensive regulatory effect of the intestinal flora on health is consistent with the “holistic view of traditional Chinese medicine”, and it also provides a new idea for research on the scientific connotation of “treating different diseases with the same method”.
Author Contributions
Overall design and manuscript revision: LZ. Manuscript draft preparation, BZ and KL. Publication retrieval: HY, ZJ, and QD. BZ and KL contributed equally to this work. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Key Research and Development Program of China (2019YFC1709904); the National Natural Science Foundation of China (82104835); the China Postdoctoral Science Foundation (2021M693542). The CACMS Innovation Fund (CI2021A01605).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
The figure of the article was drawn by Figdraw (www.figdraw.com).
References
Abenavoli, L., Scarpellini, E., Colica, C., Boccuto, L., Salehi, B., Sharifi-Rad, J., et al. (2019). Gut Microbiota and Obesity: A Role for Probiotics. Nutrients 11, 2690. doi: 10.3390/nu11112690
Agus, A., Planchais, J., Sokol, H. (2018). Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease. Cell Host Microbe 23 (6), 716–724. doi: 10.1016/j.chom.2018.05.003
Amyot, J., Semache, M., Ferdaoussi, M., Fontés, G., Poitout, V. (2012). Lipopolysaccharides Impair Insulin Gene Expression in Isolated Islets of Langerhans via Toll-Like Receptor-4 and NF-κb Signalling. PloS One 7, e36200. doi: 10.1371/journal.pone.0036200
Ananthakrishnan, A. N. (2015). Epidemiology and Risk Factors for IBD. Nature Reviews. Gastroenterol. Hepatol. 12, 205–217. doi: 10.1038/nrgastro.2015.34
Arafa, E. A., Mohamed, W. R., Zaher, D. M., Omar, H. A. (2020). Gliclazide Attenuates Acetic Acid-Induced Colitis via the Modulation of Pparγ, NF-κb and MAPK Signaling Pathways. Toxicol. Appl. Pharmacol. 391, 114919. doi: 10.1016/j.taap.2020.114919
Arora, T., Tremaroli, V. (2021). Therapeutic Potential of Butyrate for Treatment of Type 2 Diabetes. Front. Endocrinol. 12, 761834. doi: 10.3389/fendo.2021.761834
Ballan, R., Saad, S. M. I. (2021). Characteristics of the Gut Microbiota and Potential Effects of Probiotic Supplements in Individuals With Type 2 Diabetes Mellitus. Foods (Basel Switzerland) 10, 2528. doi: 10.3390/foods10112528
Balvers, M., Deschasaux, M., van den Born, B. J., Zwinderman, K., Nieuwdorp, M., Levin, E. (2021). Analyzing Type 2 Diabetes Associations With the Gut Microbiome in Individuals From Two Ethnic Backgrounds Living in the Same Geographic Area. Nutrients 13, 3289. doi: 10.3390/nu13093289
Ben-Neriah, Y., Karin, M. (2011). Inflammation Meets Cancer, With NF-κb as the Matchmaker. Nat. Immunol. 12, 715–723. doi: 10.1038/ni.2060
Bromke, M. A., Krzystek-Korpacka, M. (2021). Bile Acid Signaling in Inflammatory Bowel Disease. Int. J. Mol. Sci. 22, 9096. doi: 10.3390/ijms22169096
Cani, P. D., Osto, M., Geurts, L., Everard, A. (2012). Involvement of Gut Microbiota in the Development of Low-Grade Inflammation and Type 2 Diabetes Associated With Obesity. Gut Microbes 3, 279–288. doi: 10.4161/gmic.19625
Cannon, A. S., Nagarkatti, P. S., Nagarkatti, M. (2021). Targeting AhR as a Novel Therapeutic Modality Against Inflammatory Diseases. Int. J. Mol. Sci. 23 (1), 288. doi: 10.3390/ijms23010288
Cao, M., Xue, J., Dong, Q., Yu, D., L, Z. (2019). Effect of Huangqin Decoction and Metformin on Intestinal Flora and Insulin Signal Transduction Molecular in Diabetes. Chin. Arch. Of Traditional Chin. Med. 37, 2792–2795. doi: 10.13193/j.issn.1673-7717.2019.11.058 (In chinese).
Chang, H., Meng, H., Wang, Y., Liu, S. (2018). Scientific Connotation of TCM Theory of “Homotherapy for Heteropathy” in Treatment of Liver and Pulmonary Fibrosis Based on Metabonomics. Chin. Traditional Herbal Drugs 49, 5116–5124. doi: 10.7501/j.issn.0253-2670.2018.21.024
Chen, N., Duan, S., Tan, W., Zeng, L., Cui, G., Liu, Z. (2020). Effect of Fuzi Lizhong Pills on Intestinal Flora of Type 2 Diabetic Patients With Spleen Deficiency. J. Baotou Med. Coll. 36, 64–70. doi: 10.16833/j.cnki.jbmc.2020.05.022. (In chinese).
Cheng, H., Liu, J., Tan, Y., Feng, W., Peng, C. (2021). Interactions Between Gut Microbiota and Berberine, a Necessary Procedure to Understand the Mechanisms of Berberine. J. Pharm. Anal. doi: 10.1016/j.jpha.2021.10.003
Chen, M., Liao, Z., Lu, B., Wang, M., Lin, L., Zhang, S., et al. (2018). Huang-Lian-Jie-Du-Decoction Ameliorates Hyperglycemia and Insulin Resistant in Association With Gut Microbiota Modulation. Front. Microbiol. 9, 2380. doi: 10.3389/fmicb.2018.02380
Chen, J., Zhang, L. K., Gu, W. C., Zhang, X. S., Li, L., Han, T., et al. (2021). Effect of Banxia Xiexin Decoction on Intestinal Flora of Mice With Ulcerative Colitis Induced by Dextran Sodium Sulfate. China J. Chin. Materia Med. 46, 2871–2880. doi: 10.19540/j.cnki.cjcmm.20210119.401
Cho, N. H., Shaw, J. E., Karuranga, S., Huang, Y., da Rocha Fernandes, J. D., Ohlrogge, A. W., et al. (2018). IDF Diabetes Atlas: Global Estimates of Diabetes Prevalence for 2017 and Projections for 2045. Diabetes Res. Clin. Pract. 138, 271–281. doi: 10.1016/j.diabres.2018.02.023
Coppola, S., Avagliano, C., Calignano, A., Berni Canani, R. (2021). The Protective Role of Butyrate Against Obesity and Obesity-Related Diseases. Molecules 26, 682. doi: 10.3390/molecules26030682
Cui, L., Guan, X., Ding, W., Luo, Y., Wang, W., Bu, W., et al. (2021). Scutellaria Baicalensis Georgi Polysaccharide Ameliorates DSS-Induced Ulcerative Colitis by Improving Intestinal Barrier Function and Modulating Gut Microbiota. Int. J. Biol. Macromolecules 166, 1035–1045. doi: 10.1016/j.ijbiomac.2020.10.259
Cui, H. X., Zhang, L. S., Luo, Y., Yuan, K., Huang, Z. Y., Guo, Y. (2019). A Purified Anthraquinone-Glycoside Preparation From Rhubarb Ameliorates Type 2 Diabetes Mellitus by Modulating the Gut Microbiota and Reducing Inflammation. Front. Microbiol. 10, 1423. doi: 10.3389/fmicb.2019.01423
Dai, Z. F., Ma, X. Y., Yang, R. L., Wang, H. C., Xu, D. D., Yang, J. N., et al. (2021). Intestinal Flora Alterations in Patients With Ulcerative Colitis and Their Association With Inflammation. Exp. Ther. Med. 22, 1322. doi: 10.3892/etm.2021.10757
de Melo, I. S. V., Dos Santos, A. F., Bueno, N. B. (2018). Curcumin or Combined Curcuminoids Are Effective in Lowering the Fasting Blood Glucose Concentrations of Individuals With Dysglycemia: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Pharmacol. Res. 128, 137–144. doi: 10.1016/j.phrs.2017.09.010
Deng, J., Zeng, L., Lai, X., Li, J., Liu, L., Lin, Q., et al. (2018). Metformin Protects Against Intestinal Barrier Dysfunction via Ampkα1-Dependent Inhibition of JNK Signalling Activation. J. Cell. Mol. Med. 22, 546–557. doi: 10.1111/jcmm.13342
Everard, A., Belzer, C., Geurts, L., Ouwerkerk, J. P., Druart, C., Bindels, L. B., et al. (2013). Cross-Talk Between Akkermansia Muciniphila and Intestinal Epithelium Controls Diet-Induced Obesity. Proc. Natl. Acad. Sci. U. S. A. 110, 9066–9071. doi: 10.1073/pnas.1219451110
Fei, N., Zhao, L. (2013). An Opportunistic Pathogen Isolated From the Gut of an Obese Human Causes Obesity in Germfree Mice. ISME J. 7, 880–884. doi: 10.1038/ismej.2012.153
Feng, W., Liu, J., Tan, Y., Ao, H., Wang, J., Peng, C. (2020). Polysaccharides From Atractylodes Macrocephala Koidz. Ameliorate Ulcerative Colitis via Extensive Modification of Gut Microbiota and Host Metabolism. Food Res. Int. (Ottawa Ont.) 138, 109777. doi: 10.1016/j.foodres.2020.109777
Gan, J., Fan, H., Zhang, Z. (2020). Effects of Chlorhexidine Hydrochloride on Intestinal Flora and Immune Status in Patients With Ulcerative Colitis. Chin. J. Integr. Trad West Med. Dig 28, 949–53. doi: 10.3969/j.issn.1671-038X.2020.12.10
Gao, Y., Li, J., Wang, J., Li, X., Li, J., Chu, S., et al. (2020). Ginsenoside Rg1 Prevent and Treat Inflammatory Diseases: A Review. Int. Immunopharmacol. 87, 106805. doi: 10.1016/j.intimp.2020.106805
Gao, Z., Li, Q., Wu, X., Zhao, X., Zhao, L., Tong, X. (2017). New Insights Into the Mechanisms of Chinese Herbal Products on Diabetes: A Focus on the “Bacteria-Mucosal Immunity-Inflammation-Diabetes” Axis. J. Immunol. Res. 2017, 1813086. doi: 10.1155/2017/1813086
Geng, J., Ni, Q., Sun, W., Li, L., Feng, X. (2022). The Links Between Gut Microbiota and Obesity and Obesity Related Diseases. Biomed. Pharmacother. = Biomed. Pharmacother. 147, 112678. doi: 10.1016/j.biopha.2022.112678
Gong, P., Xiao, X., Wang, S., Shi, F., Liu, N., Chen, X., et al. (2021). Hypoglycemic Effect of Astragaloside IV via Modulating Gut Microbiota and Regulating AMPK/SIRT1 and PI3K/AKT Pathway. J. Ethnopharmacol. 281, 114558. doi: 10.1016/j.jep.2021.114558
Gurung, M., Li, Z., You, H., Rodrigues, R., Jump, D. B., Morgun, A., et al. (2020). Role of Gut Microbiota in Type 2 Diabetes Pathophysiology. EBioMedicine 51, 102590. doi: 10.1016/j.ebiom.2019.11.051
Gu, J.-F., Su, S.-L., Guo, J.-M., Zhu, Y., Zhao, M., Duan, J.-A. (2017). The Aerial Parts of Salvia Miltiorrhiza Bge. Strengthen Intestinal Barrier and Modulate Gut Microbiota Imbalance in Streptozocin-Induced Diabetic Mice. J. Funct. Foods 36, 362–374. doi: 10.1016/j.jff.2017.06.010
Habtemariam, S. (2020). Berberine Pharmacology and the Gut Microbiota: A Hidden Therapeutic Link. Pharmacol. Res. 155, 104722. doi: 10.1016/j.phrs.2020.104722
Halling, M. L., Kjeldsen, J., Knudsen, T., Nielsen, J., Hansen, L. K. (2017). Patients With Inflammatory Bowel Disease Have Increased Risk of Autoimmune and Inflammatory Diseases. World J. Gastroenterol. 23, 6137–6146. doi: 10.3748/wjg.v23.i33.6137
Hampe, C. S., Roth, C. L. (2017). Probiotic Strains and Mechanistic Insights for the Treatment of Type 2 Diabetes. Endocrine 58, 207–227. doi: 10.1007/s12020-017-1433-z
Hong, F., Xu, P., Zhai, Y. (2018). The Opportunities and Challenges of Peroxisome Proliferator-Activated Receptors Ligands in Clinical Drug Discovery and Development. Int. J. Mol. Sci. 19, 2189. doi: 10.3390/ijms19082189
Hotamisligil, G. S. (2006). Inflammation and Metabolic Disorders. Nature 444, 860–867. doi: 10.1038/nature05485
Huang, J., Guan, B., Lin, L., Wang, Y. (2021). Improvement of Intestinal Barrier Function, Gut Microbiota, and Metabolic Endotoxemia in Type 2 Diabetes Rats by Curcumin. Bioengineered. doi: 10.1080/21655979.2021.2009322
Huang, G., Ye, L., Du, G., Huang, Y., Wu, Y., Ge, S., et al. (2017). Effects of Curcumin Plus Soy Oligosaccharides on Intestinal Flora of Rats With Ulcerative Colitis. Cell. Mol. Biol. (Noisy-le-Grand France) 63, 20–25. doi: 10.14715/cmb/2017.63.7.3
Hyun, C. K. (2021). Molecular and Pathophysiological Links Between Metabolic Disorders and Inflammatory Bowel Diseases. Int. J. Mol. Sci. 22, 9139. doi: 10.3390/ijms22179139
Illiano, P., Brambilla, R., Parolini, C. (2020). The Mutual Interplay of Gut Microbiota, Diet and Human Disease. FEBS J. 287, 833–855. doi: 10.1111/febs.15217
Jess, T., Jensen, B. W., Andersson, M., Villumsen, M., Allin, K. H. (2020). Inflammatory Bowel Diseases Increase Risk of Type 2 Diabetes in a Nationwide Cohort Study. Clin. Gastroenterol. Hepatol: Off. Clin. Pract. J. Am. Gastroenterological Assoc. 18, 881–888.e881. doi: 10.1016/j.cgh.2019.07.052
Jurjus, A., Eid, A., Al Kattar, S., Zeenny, M. N., Gerges-Geagea, A., Haydar, H., et al. (2016). Inflammatory Bowel Disease, Colorectal Cancer and Type 2 Diabetes Mellitus: The Links. BBA Clin. 5, 16–24. doi: 10.1016/j.bbacli.2015.11.002
Kang, E. A., Han, K., Chun, J., Soh, H., Park, S., Im, J. P., et al. (2019). Increased Risk of Diabetes in Inflammatory Bowel Disease Patients: A Nationwide Population-Based Study in Korea. J. Clin. Med. 8, 343. doi: 10.3390/jcm8030343
Kappelman, M. D., Galanko, J. A., Porter, C. Q., Sandler, R. S. (2011). Association of Paediatric Inflammatory Bowel Disease With Other Immune-Mediated Diseases. Arch. Dis. Childhood 96, 1042–1046. doi: 10.1136/archdischild-2011-300633
Ke, H., Li, F., Deng, W., Li, Z., Wang, S., Lv, P., et al. (2021). Metformin Exerts Anti-Inflammatory and Mucus Barrier Protective Effects by Enriching Akkermansia Muciniphila in Mice With Ulcerative Colitis. Front. Pharmacol. 12, 726707. doi: 10.3389/fphar.2021.726707
Lamas, B., Richard, M. L., Leducq, V., Pham, H. P., Michel, M. L., Da Costa, G., et al. (2016). CARD9 Impacts Colitis by Altering Gut Microbiota Metabolism of Tryptophan Into Aryl Hydrocarbon Receptor Ligands. Nat. Med. 22 (6), 598–605. doi: 10.1038/nm.4102
Lees, C. W., Barrett, J. C., Parkes, M., Satsangi, J. (2011). New IBD Genetics: Common Pathways With Other Diseases. Gut 60, 1739–1753. doi: 10.1136/gut.2009.199679
Lewis, J. D., Lichtenstein, G. R., Deren, J. J., Sands, B. E., Hanauer, S. B., Katz, J. A., et al. (2008). Rosiglitazone for Active Ulcerative Colitis: A Randomized Placebo-Controlled Trial. Gastroenterology 134, 688–695. doi: 10.1053/j.gastro.2007.12.012
Li, C., Ai, G., Wang, Y., Lu, Q., Luo, C., Tan, L., et al. (2020). Oxyberberine, a Novel Gut Microbiota-Mediated Metabolite of Berberine, Possesses Superior Anti-Colitis Effect: Impact on Intestinal Epithelial Barrier, Gut Microbiota Profile and TLR4-MyD88-NF-κb Pathway. Pharmacol. Res. 152, 104603. doi: 10.1016/j.phrs.2019.104603
Li, X. W., Chen, H. P., He, Y. Y., Chen, W. L., Chen, J. W., Gao, L., et al. (2018). Effects of Rich-Polyphenols Extract of Dendrobium Loddigesii on Anti-Diabetic, Anti-Inflammatory, Anti-Oxidant, and Gut Microbiota Modulation in Db/Db Mice. Molecules 23, 3245. doi: 10.3390/molecules23123245
Li, H., Chen, X., Liu, J., Chen, M., Huang, M., Huang, G., et al. (2021a). Ethanol Extract of Centella Asiatica Alleviated Dextran Sulfate Sodium-Induced Colitis: Restoration on Mucosa Barrier and Gut Microbiota Homeostasis. J. Ethnopharmacol 267, 113445. doi: 10.1016/j.jep.2020.113445
Li, Q., Cui, Y., Xu, B., Wang, Y., Lv, F., Li, Z., et al. (2021a). Main Active Components of Jiawei Gegen Qinlian Decoction Protects Against Ulcerative Colitis Under Different Dietary Environments in a Gut Microbiota-Dependent Manner. Pharmacol. Res. 170, 105694. doi: 10.1016/j.phrs.2021.105694
Li, C., Dong, N., Wu, B., Mo, Z., Xie, J., Lu, Q. (2021). Dihydroberberine, an Isoquinoline Alkaloid, Exhibits Protective Effect Against Dextran Sulfate Sodium-Induced Ulcerative Colitis in Mice. Phytomedicine 90, 153631. doi: 10.1016/j.phymed.2021.153631
Li, H., Fan, C., Lu, H., Feng, C., He, P., Yang, X., et al. (2020). Protective Role of Berberine on Ulcerative Colitis Through Modulating Enteric Glial Cells-Intestinal Epithelial Cells-Immune Cells Interactions. Acta Pharm. Sinica. B 10, 447–461. doi: 10.1016/j.apsb.2019.08.006
Li, M. Y., Luo, H. J., Wu, X., Liu, Y. H., Gan, Y. X., Xu, N., et al. (2019a). Anti-Inflammatory Effects of Huangqin Decoction on Dextran Sulfate Sodium-Induced Ulcerative Colitis in Mice Through Regulation of the Gut Microbiota and Suppression of the Ras-PI3K-Akt-HIF-1α and NF-κb Pathways. Front. Pharmacol. 10, 1552. doi: 10.3389/fphar.2019.01552
Li, Z., Qiao, L., Yun, X., Du, F., Xing, S., Yang, M. (2021c). Increased Risk of Ischemic Heart Disease and Diabetes in Inflammatory Bowel Disease. Z. fur Gastroenterologie 59, 117–124. doi: 10.1055/a-1283-6966
Li, X., Song, Y., Wang, L., Kang, G., Wang, P., Yin, H., et al. (2021b). A Potential Combination Therapy of Berberine Hydrochloride With Antibiotics Against Multidrug-Resistant Acinetobacter Baumannii. Front. Cell. Infect. Microbiol. 11, 660431. doi: 10.3389/fcimb.2021.660431
Liang, Y., Xu, X., Yin, M., Zhang, Y., Huang, L., Chen, R., et al. (2019). Effects of Berberine on Blood Glucose in Patients With Type 2 Diabetes Mellitus: A Systematic Literature Review and a Meta-Analysis. Endocr J. 66, 51–63. doi: 10.1507/endocrj.EJ18-0109
Liao, Z., Xie, Y., Zhou, B., Zou, B., Xiao, D., Liu, W., et al. (2020). Berberine Ameliorates Colonic Damage Accompanied With the Modulation of Dysfunctional Bacteria and Functions in Ulcerative Colitis Rats. Appl. Microbiol. Biotechnol. 104, 1737–1749. doi: 10.1007/s00253-019-10307-1
Liu, J., Tan, Y., Cheng, H., Zhang, D., Feng, W., Peng, C. (2022). Functions of Gut Microbiota Metabolites, Current Status and Future Perspective. Aging Dis. doi: 10.14336/AD.2022.0104
Liu, X., Sun, Z., Wang, H. (2021). Metformin Alleviates Experimental Colitis in Mice by Up-Regulating TGF-β Signaling. Biotechnic Histochem: Off. Publ. Biol. Stain Commission 96, 146–152. doi: 10.1080/10520295.2020.1776896
Liu, T., Wang, Y., Cui, H., Tang, T. (2020). Brain Material Basis and Relevant Mechanism of Acute Stroke Treated With Rhei Radix Et Rhizoma Based on Homotherapy for Heteropathy Using Proteomics. Chin. J. Exp. Traditional Med. Formulae 26, 160–168. doi: 10.13422/j.cnki.syfjx.20201923 (In chinese).
Long, J., Liu, X. K., Kang, Z. P., Wang, M. X., Zhao, H. M., Huang, J. Q., et al. (2022). Ginsenoside Rg1 Ameliorated Experimental Colitis by Regulating the Balance of M1/M2 Macrophage Polarization and the Homeostasis of Intestinal Flora. Eur. J. Pharmacol. 917, 174742. doi: 10.1016/j.ejphar.2022.174742
Lund, J. L., Stürmer, T., Porter, C. Q., Sandler, R. S., Kappelman, M. D. (2011). Thiazolidinedione Use and Ulcerative Colitis-Related Flares: An Exploratory Analysis of Administrative Data. Inflammatory bowel Dis. 17, 787–794. doi: 10.1002/ibd.21348
Lu, P. D., Zhao, Y. H. (2020). Targeting NF-κb Pathway for Treating Ulcerative Colitis: Comprehensive Regulatory Characteristics of Chinese Medicines. Chin. Med. 15, 15. doi: 10.1186/s13020-020-0296-z
Maconi, G., Furfaro, F., Sciurti, R., Bezzio, C., Ardizzone, S., de Franchis, R. (2014). Glucose Intolerance and Diabetes Mellitus in Ulcerative Colitis: Pathogenetic and Therapeutic Implications. World J. Gastroenterol. 20, 3507–3515. doi: 10.3748/wjg.v20.i13.3507
Miraghajani, M., Dehsoukhteh, S. S., Rafie, N., Hamedani, S. G., Sabihi, S., Ghiasvand, R. (2017). Potential Mechanisms Linking Probiotics to Diabetes: A Narrative Review of the Literature. Sao Paulo Med. J. = Rev. Paulista Medicina 135, 169–178. doi: 10.1590/1516-3180.2016.0311271216
Monaco, A., Ovryn, B., Axis, J., Amsler, K. (2021). The Epithelial Cell Leak Pathway. Int. J. Mol. Sci. 22 (14), 7677. doi: 10.3390/ijms22147677
Naito, Y., Uchiyama, K., Takagi, T. (2018). A Next-Generation Beneficial Microbe: Akkermansia Muciniphila. J. Clin. Biochem. Nutr. 63, 33–35. doi: 10.3164/jcbn.18-57
Nyavor, Y., Estill, R., Edwards, H., Ogden, H., Heideman, K., Starks, K., et al. (2019). Intestinal Nerve Cell Injury Occurs Prior to Insulin Resistance in Female Mice Ingesting a High-Fat Diet. Cell Tissue Res. 376, 325–340. doi: 10.1007/s00441-019-03002-0
Pandey, A., Verma, S., Kumar, V. L. (2017). Metformin Maintains Mucosal Integrity in Experimental Model of Colitis by Inhibiting Oxidative Stress and Pro-Inflammatory Signaling. Biomed. Pharmacother. = Biomed. Pharmacother. 94, 1121–1128. doi: 10.1016/j.biopha.2017.08.020
Patterson, E., Ryan, P. M., Cryan, J. F., Dinan, T. G., Ross, R. P., Fitzgerald, G. F., et al. (2016). Gut Microbiota, Obesity and Diabetes. Postgrad. Med. J. 92, 286–300. doi: 10.1136/postgradmedj-2015-133285
Pavel, F. M., Vesa, C. M., Gheorghe, G., Diaconu, C. C., Stoicescu, M., Munteanu, M. A., et al. (2021). Highlighting the Relevance of Gut Microbiota Manipulation in Inflammatory Bowel Disease. Diagnostics (Basel Switzerland) 11, 1090. doi: 10.3390/diagnostics11061090
Pawłowska, B., Sobieszczańska, B. M. (2017). Intestinal Epithelial Barrier: The Target for Pathogenic Escherichia Coli. Adv. Clin. Exp. Med: Off. Organ Wroclaw Med. Univ. 26, 1437–1445. doi: 10.17219/acem/64883
Peng, L., Gao, X., Nie, L., Xie, J., Dai, T., Shi, C., et al. (2020). Astragalin Attenuates Dextran Sulfate Sodium (DSS)-Induced Acute Experimental Colitis by Alleviating Gut Microbiota Dysbiosis and Inhibiting NF-kappaB Activation in Mice. Front. Immunol. 11, 2058. doi: 10.3389/fimmu.2020.02058
Prosberg, M., Bendtsen, F., Vind, I., Petersen, A. M., Gluud, L. L. (2016). The Association Between the Gut Microbiota and the Inflammatory Bowel Disease Activity: A Systematic Review and Meta-Analysis. Scand J. Gastroenterol. 51, 1407–1415. doi: 10.1080/00365521.2016.1216587
Qin, J., Li, Y., Cai, Z., Li, S., Zhu, J., Zhang, F., et al. (2012). A Metagenome-Wide Association Study of Gut Microbiota in Type 2 Diabetes. Nature 490, 55–60. doi: 10.1038/nature11450
Qiu, J. (2007). ‘Back to the Future’ for Chinese Herbal Medicines. Nat. Rev. Drug Discov. 6, 506–507. doi: 10.1038/nrd2350
Radhakrishnan, S. T., Alexander, J. L., Mullish, B. H., Gallagher, K. I., Powell, N., Hicks, L. C., et al. (2022). Systematic Review: The Association Between the Gut Microbiota and Medical Therapies in Inflammatory Bowel Disease. Alimentary Pharmacol. Ther. 55, 26–48. doi: 10.1111/apt.16656
Régnier, M., Rastelli, M., Morissette, A., Suriano, F., Le Roy, T., Pilon, G., et al. (2020). Rhubarb Supplementation Prevents Diet-Induced Obesity and Diabetes in Association With Increased Akkermansia Muciniphila in Mice. Nutrients 12, 2932. doi: 10.3390/nu12102932
Saber, S., El-Kader, E. M. A. (2021). Novel Complementary Coloprotective Effects of Metformin and MCC950 by Modulating HSP90/NLRP3 Interaction and Inducing Autophagy in Rats. Inflammopharmacology 29, 237–251. doi: 10.1007/s10787-020-00730-6
Sahukari, R., Punabaka, J., Bhasha, S., Ganjikunta, V. S., Ramudu, S. K., Kesireddy, S. R. (2020). Plant Compounds for the Treatment of Diabetes, a Metabolic Disorder: NF-κb as a Therapeutic Target. Curr. Pharm. Design 26, 4955–4969. doi: 10.2174/1381612826666200730221035
Samman, F. S., Elaidy, S. M., Essawy, S. S., Hassan, M. S. (2018). New Insights on the Modulatory Roles of Metformin or Alpha-Lipoic Acid Versus Their Combination in Dextran Sulfate Sodium-Induced Chronic Colitis in Rats. Pharmacol. Reps: PR 70, 488–496. doi: 10.1016/j.pharep.2017.11.015
Shan, C. Y., Yang, J. H., Kong, Y., Wang, X. Y., Zheng, M. Y., Xu, Y. G., et al. (2013). Alteration of the Intestinal Barrier and GLP2 Secretion in Berberine-Treated Type 2 Diabetic Rats. J. Endocrinol. 218, 255–262. doi: 10.1530/JOE-13-0184
Shen, L., Ao, L., Xu, H., Shi, J., You, D., Yu, X., et al. (2019). Poor Short-Term Glycemic Control in Patients With Type 2 Diabetes Impairs the Intestinal Mucosal Barrier: A Prospective, Single-Center, Observational Study. BMC Endocr. Disord. 19, 29. doi: 10.1186/s12902-019-0354-7
Sheng, Y., Zheng, S., Ma, T., Zhang, C., Ou, X., He, X., et al. (2017). Mulberry Leaf Alleviates Streptozotocin-Induced Diabetic Rats by Attenuating NEFA Signaling and Modulating Intestinal Microflora. Sci. Rep. 7, 12041. doi: 10.1038/s41598-017-12245-2
Shen, Y., Wang, Z., Zheng, H., Zhong, J., Xu, L., Jiang, X., et al. (2018). Protective Effects of Berberine Hydrochloride on the Intestinal Mucosal Mechanical Barrier in Mice With Ulcerative Colitis. Chin. J. Mod Appl. Pharm. 35, 1765–1770. doi: 10.13748/j.cnki.issn1007-7693.2018.12.003
Shen, Y., Zhong, J., Xu, L., Li, S., Wang, Z., Zheng, H. (2017). Effects of Berberine Hydrochloride on the Expression of TNF-α, IL-1β and IL-10 of Colon Tissue in Mice With Ulcerative Colitis. Chin. J. Mod Appl. Pharm. 34, 1094–1098. doi: 10.13748/j.cnki.issn1007-7693.2017.08.005
Shen, Y., Zou, J., Chen, M., Zhang, Z., Liu, C., Jiang, S., et al. (2020). Protective Effects of Lizhong Decoction on Ulcerative Colitis in Mice by Suppressing Inflammation and Ameliorating Gut Barrier. J. Ethnopharmacol 259, 112919. doi: 10.1016/j.jep.2020.112919
Shi, Y., Dong, Y., Huang, W., Zhu, D., Mao, H., Su, P. (2016). Fecal Microbiota Transplantation for Ulcerative Colitis: A Systematic Review and Meta-Analysis. PloS One 11, e0157259. doi: 10.1371/journal.pone.0157259
Si, Q. (2016). Protective Effect and Mechanism of Alcohol Extract of Centella Asiatica on Islets of ZDF Rats (Beijing: PhD Dissertation, Beijing University of Chinese Medicine).
Silva, J. P. B., Navegantes-Lima, K. C., Oliveira, A. L. B., Rodrigues, D. V. S., Gaspar, S. L. F., Monteiro, V. V. S., et al. (2018). Protective Mechanisms of Butyrate on Inflammatory Bowel Disease. Curr. Pharm. design 24, 4154–4166. doi: 10.2174/1381612824666181001153605
Singer-Englar, T., Barlow, G., Mathur, R. (2019). Obesity, Diabetes, and the Gut Microbiome: An Updated Review. Expert Rev. Gastroenterol. Hepatol. 13, 3–15. doi: 10.1080/17474124.2019.1543023
Su, H., Wu, G., Zhan, L., Xu, F., Qian, H., Li, Y., et al. (2022). Exploration of the Mechanism of Lianhua Qingwen in Treating Influenza Virus Pneumonia and New Coronavirus Pneumonia With the Concept of “Different Diseases With the Same Treatment” Based on Network Pharmacology. Evid Based Complement Alternat Med. 2022, 5536266. doi: 10.1155/2022/5536266
Tang, X. (2020). In Vitro Fermentation Characteristics of Mulberry Leaf Albumin and Enzymatic Hydrolysates and the Alleviating Effect of Enzymatic Hydrolysates on DSS-Induced Ulcerative Colitis (MA thesis, Jinan University). doi: 10.27167/d.cnki.gjinu.2020.000911
Tian, J., Bai, B., Gao, Z., Yang, Y., Wu, H., Wang, X., et al. (2021). Alleviation Effects of GQD, A Traditional Chinese Medicine Formula, on Diabetes Rats Linked to Modulation of the Gut Microbiome. Front. Cell. Infect. Microbiol. 11, 740236. doi: 10.3389/fcimb.2021.740236
Tian, J., Jin, D., Bao, Q., Ding, Q., Zhang, H., Gao, Z., et al. (2019). Evidence and Potential Mechanisms of Traditional Chinese Medicine for the Treatment of Type 2 Diabetes: A Systematic Review and Meta-Analysis. Diabetes Obes. Metab. 21, 1801–1816. doi: 10.1111/dom.13760
Tiderencel, K. A., Hutcheon, D. A., Ziegler, J. (2020). Probiotics for the Treatment of Type 2 Diabetes: A Review of Randomized Controlled Trials. Diabetes/Metabolism Res. Rev. 36, e3213. doi: 10.1002/dmrr.3213
Tseng, C. H. (2021). Metformin Use Is Associated With a Lower Risk of Inflammatory Bowel Disease in Patients With Type 2 Diabetes Mellitus. J. Crohn’s colitis 15, 64–73. doi: 10.1093/ecco-jcc/jjaa136
Usuda, H., Okamoto, T., Wada, K. (2021). Leaky Gut: Effect of Dietary Fiber and Fats on Microbiome and Intestinal Barrier. Int. J. Mol. Sci. 22, 7613. doi: 10.3390/ijms22147613
Valdes, A. M., Walter, J., Segal, E., Spector, T. D. (2018). Role of the Gut Microbiota in Nutrition and Health. BMJ (Clinical Res. ed.) 361, k2179. doi: 10.1136/bmj.k2179
Vancamelbeke, M., Vermeire, S. (2017). The Intestinal Barrier: A Fundamental Role in Health and Disease. Expert Rev. Gastroenterol. Hepatol. 11, 821–834. doi: 10.1080/17474124.2017.1343143
Vives-Pi, M., Somoza, N., Fernández-Alvarez, J., Vargas, F., Caro, P., Alba, A., et al. (2003). Evidence of Expression of Endotoxin Receptors CD14, Toll-Like Receptors TLR4 and TLR2 and Associated Molecule MD-2 and of Sensitivity to Endotoxin (LPS) in Islet Beta Cells. Clin. Exp. Immunol. 133, 208–218. doi: 10.1046/j.1365-2249.2003.02211.x
Wang, S. Q., Cui, S. X., Qu, X. J. (2019). Metformin Inhibited Colitis and Colitis-Associated Cancer (CAC) Through Protecting Mitochondrial Structures of Colorectal Epithelial Cells in Mice. Cancer Biol. Ther. 20, 338–348. doi: 10.1080/15384047.2018.1529108
Wang, J., Ghosh, S. S., Ghosh, S. (2017). Curcumin Improves Intestinal Barrier Function: Modulation of Intracellular Signaling, and Organization of Tight Junctions. Am. J. Physiol. Cell Physiol. 312, C438–C445. doi: 10.1152/ajpcell.00235.2016
Wang, W., Wang, X., Liu, L., Liu, Z., Han, T., Sun, C., et al. (2022). Dietary Tryptophan and the Risk of Obesity and Type 2 Diabetes: Total Effect and Mediation Effect of Sleep Duration. Obes. (Silver Spring Md.) 30, 515–523. doi: 10.1002/oby.23343
Wang, K., Yang, Q., Ma, Q., Wang, B., Wan, Z., Chen, M., et al. (2018a). Protective Effects of Salvianolic Acid A Against Dextran Sodium Sulfate-Induced Acute Colitis in Rats. Nutrients 10, 791. doi: 10.3390/nu10060791
Wang, R., Zang, P., Chen, J., Wu, F., Zheng, Z., Ma, J., et al. (2018b). Gut Microbiota Play an Essential Role in the Antidiabetic Effects of Rhein. Evid Based Complement Alternat Med. 2018, 6093282. doi: 10.1155/2018/6093282
Wei, Y., Yang, H., Zhu, C., Deng, J., Fan, D. (2020). Hypoglycemic Effect of Ginsenoside Rg5 Mediated Partly by Modulating Gut Microbiota Dysbiosis in Diabetic Db/Db Mice. J. Agric. Food Chem. 68, 5107–5117. doi: 10.1021/acs.jafc.0c00605
Wu, X., Chen, H., Gao, X., Gao, H., He, Q., Li, G., et al. (2022). Natural Herbal Remedy Wumei Decoction Ameliorates Intestinal Mucosal Inflammation by Inhibiting Th1/Th17 Cell Differentiation and Maintaining Microbial Homeostasis. Inflamm. Bowel Dis, izab348. doi: 10.1093/ibd/izab348
Wu, J., Wei, Z., Cheng, P., Qian, C., Xu, F., Yang, Y., et al. (2020). Rhein Modulates Host Purine Metabolism in Intestine Through Gut Microbiota and Ameliorates Experimental Colitis. Theranostics 10, 10665–10679. doi: 10.7150/thno.43528
Xiao, S., Liu, C., Chen, M., Zou, J., Zhang, Z., Cui, X., et al. (2020). Scutellariae Radix and Coptidis Rhizoma Ameliorate Glycolipid Metabolism of Type 2 Diabetic Rats by Modulating Gut Microbiota and Its Metabolites. Appl. Microbiol. Biotechnol. 104, 303–317. doi: 10.1007/s00253-019-10174-w
Xie, C., Huang, W., Young, R. L., Jones, K. L., Horowitz, M., Rayner, C. K., et al. (2021). Role of Bile Acids in the Regulation of Food Intake, and Their Dysregulation in Metabolic Disease. Nutrients 13 (4), 1104. doi: 10.3390/nu13041104
Xie, Q., Li, H., Ma, R., Ren, M., Li, Y., Li, J., et al. (2022b). Effect of Coptis Chinensis Franch and Magnolia Officinalis on Intestinal Flora and Intestinal Barrier in a TNBS-Induced Ulcerative Colitis Rats Model. Phytomedicine 97, 153927. doi: 10.1016/j.phymed.2022.153927
Xie, J., Liu, L., Li, H., Che, H., Xie, W. (2022a). Ink Melanin From Sepiapharaonis Ameliorates Colitis in Mice via Reducing Oxidative Stress, Andprotecting the Intestinal Mucosal Barrier. Food Res. Int. (Ottawa Ont.) 151, 110888. doi: 10.1016/j.foodres.2021.110888
Xue, M., Zheng, Y., Zhang, Y., Tao, Q., Yang, Y., Yin, D. (2022). Synergistic Effect of Crude Coptis Polysaccharide and Berberine on Restoring the Intestinal Mucosal Barrier Damage in Ulcerative Colitis. Chin. J. Exp. Traditional Med. Formulae. doi: 10.13422/j.cnki.syfjx.20220702
Xu, X., Gao, Z., Yang, F., Yang, Y., Chen, L., Han, L., et al. (2020). Antidiabetic Effects of Gegen Qinlian Decoction via the Gut Microbiota Are Attributable to Its Key Ingredient Berberine. Genomics Proteomics Bioinf. 18, 721–736. doi: 10.1016/j.gpb.2019.09.007
Xu, H. M., Huang, H. L., Liu, Y. D., Zhu, J. Q., Zhou, Y. L., Chen, H. T., et al. (2021a). Selection Strategy of Dextran Sulfate Sodium-Induced Acute or Chronic Colitis Mouse Models Based on Gut Microbial Profile. BMC Microbiol. 21, 279. doi: 10.1186/s12866-021-02342-8
Xu, H. M., Huang, H. L., Xu, J., He, J., Zhao, C., Peng, Y., et al. (2021b). Cross-Talk Between Butyric Acid and Gut Microbiota in Ulcerative Colitis Following Fecal Microbiota Transplantation. Front. Microbiol. 12, 658292. doi: 10.3389/fmicb.2021.658292
Xu, J., Lian, F., Zhao, L., Zhao, Y., Chen, X., Zhang, X., et al. (2015). Structural Modulation of Gut Microbiota During Alleviation of Type 2 Diabetes With a Chinese Herbal Formula. ISME J. 9, 552–562. doi: 10.1038/ismej.2014.177
Yang, M. (2019). The Clinical Efficacy Evaluation and Mechanism Research of ‘Different Diseases With Same Treatment’ of Bushen Jianpi Jiedu Treatment Combined With Chemotherapy on Postoperative Primary Liver Cancer and Colorectal Cancer (Shanghai: MA Thesis, Shanghai University of Chinese Medicine).
Yang, Y., Chen, G., Yang, Q., Ye, J., Cai, X., Tsering, P., et al. (2017). Gut Microbiota Drives the Attenuation of Dextran Sulphate Sodium-Induced Colitis by Huangqin Decoction. Oncotarget 8 (30), 48863–48874. doi: 10.18632/oncotarget.16458
Yang, M. Y., Hu, Z. P., Yue, R. S., Chen, Y., Zhang, B. X. (2021b). Effects of Banxia Xiexin Decoction on Islet Function of Type 2 Diabetic Rats by Regulating Intestinal Microbiota - Host Metabolism. Chin. J. Traditional Chin. Med. 36, 2025–2032.
Yang, L., Song, Y., J, N. (2021). The Influence of Ginseng and Poria and White Atractylodes Powder on Intestinal Flora of Patients With Ulcerative Colitis. Henan traditional Chin. Med. 41 (09), 1003–5028. doi: 10.16367/j.issn.1003-5028.2021.09.0308
Yang, L., Yu, H., Hou, A., Man, W., Wang, S., Zhang, J., et al. (2021a). A Review of the Ethnopharmacology, Phytochemistry, Pharmacology, Application, Quality Control, Processing, Toxicology, and Pharmacokinetics of the Dried Rhizome of Atractylodes Macrocephala. Front. Pharmacol. 12, 727154. doi: 10.3389/fphar.2021.727154
Yao, Y., Chen, H., Yan, L., Wang, W., Wang, D. (2020). Berberine Alleviates Type 2 Diabetic Symptoms by Altering Gut Microbiota and Reducing Aromatic Amino Acids. Biomed. Pharmacother. = Biomed. Pharmacother. 131, 110669. doi: 10.1016/j.biopha.2020.110669
Yao, S., Zhao, Z., Wang, W., Liu, X. (2021). Bifidobacterium Longum: Protection Against Inflammatory Bowel Disease. J. Immunol. Res. 2021, 8030297. doi: 10.1155/2021/8030297
Youssef, M. E., Abd El-Fattah, E. E., Abdelhamid, A. M., Eissa, H., El-Ahwany, E., Amin, N. A., et al. (2021). Interference With the Ampkα/mTOR/NLRP3 Signaling and the IL-23/IL-17 Axis Effectively Protects Against the Dextran Sulfate Sodium Intoxication in Rats: A New Paradigm in Empagliflozin and Metformin Reprofiling for the Management of Ulcerative Colitis. Front. Pharmacol. 12, 719984. doi: 10.3389/fphar.2021.719984
Yuan, Z., Yang, L., Zhang, X., Ji, P., Hua, Y., Wei, Y. (2019). Huang-Lian-Jie-Du Decoction Ameliorates Acute Ulcerative Colitis in Mice via Regulating NF-κb and Nrf2 Signaling Pathways and Enhancing Intestinal Barrier Function. Front. Pharmacol. 10, 1354. doi: 10.3389/fphar.2019.01354
Yuan, Z., Yang, L., Zhang, X., Ji, P., Wei, Y. (2020b). Therapeutic Effect of N-Butanol Fraction of Huang-Lian-Jie-Du Decoction on Ulcerative Colitis and Its Regulation on Intestinal Flora in Colitis Mice. Biomed. Pharmacother. = Biomed. Pharmacother. 121, 109638. doi: 10.1016/j.biopha.2019.109638
Yuan, T., Yin, Z., Yan, Z., Hao, Q., Zeng, J., Li, L., et al. (2020a). Tetrahydrocurcumin Ameliorates Diabetes Profiles of Db/Db Mice by Altering the Composition of Gut Microbiota and Up-Regulating the Expression of GLP-1 in the Pancreas. Fitoterapia 146, 104665. doi: 10.1016/j.fitote.2020.104665
Zatorski, H., Sałaga, M., Fichna, J. (2019). Role of Glucagon-Like Peptides in Inflammatory Bowel Diseases-Current Knowledge and Future Perspectives. Naunyn-Schmiedeberg’s Arch. Pharmacol. 392, 1321–1330. doi: 10.1007/s00210-019-01698-z
Zhang, C., Jiang, M., Lu, A. (2013). Considerations of Traditional Chinese Medicine as Adjunct Therapy in the Management of Ulcerative Colitis. Clin. Rev. Allergy Immunol. 44, 274–283. doi: 10.1007/s12016-012-8328-9
Zhang, L., Liu, C., Jiang, Q., Yin, Y. (2021a). Butyrate in Energy Metabolism: There Is Still More to Learn. Trends Endocrinol. Metabolism: TEM 32, 159–169. doi: 10.1016/j.tem.2020.12.003
Zhang, X., Wu, J., Ye, B., Wang, Q., Xie, X., Shen, H. (2016). Protective Effect of Curcumin on TNBS-Induced Intestinal Inflammation Is Mediated Through the JAK/STAT Pathway. BMC Complement Altern. Med. 16, 299. doi: 10.1186/s12906-016-1273-z
Zhang, J., Xu, X., Li, N., Cao, L., Sun, Y., Wang, J., et al. (2022). Licoflavone B, an Isoprene Flavonoid Derived From Licorice Residue, Relieves Dextran Sodium Sulfate-Induced Ulcerative Colitis by Rebuilding the Gut Barrier and Regulating Intestinal Microflora. Eur. J. Pharmacol. 916, 174730. doi: 10.1016/j.ejphar.2021.174730
Zhang, W., Xu, J. H., Yu, T., Chen, Q. K. (2019). Effects of Berberine and Metformin on Intestinal Inflammation and Gut Microbiome Composition in Db/Db Mice. Biomed. Pharmacother. = Biomed. Pharmacother. 118, 109131. doi: 10.1016/j.biopha.2019.109131
Zhang, Y., Xu, Y., Zhang, L., Chen, Y., Wu, T., Liu, R., et al. (2022b). Licorice Extract Ameliorates Hyperglycemia Through Reshaping Gut Microbiota Structure and Inhibiting TLR4/NF-κb Signaling Pathway in Type 2 Diabetic Mice. Food Res. Int. (Ottawa Ont.) 153, 110945. doi: 10.1016/j.foodres.2022.110945
Zhang, B., Yue, R., Chen, Y., Huang, X., Yang, M., Shui, J., et al. (2020). The Herbal Medicine Scutellaria-Coptis Alleviates Intestinal Mucosal Barrier Damage in Diabetic Rats by Inhibiting Inflammation and Modulating the Gut Microbiota. Evid Based Complement Alternat Med. 2020, 4568629. doi: 10.1155/2020/4568629
Zhang, W.-Y., Zhang, H.-H., Yu, C.-H., Fang, J., Ying, H.-Z. (2017). Ethanol Extract of Atractylodis Macrocephalae Rhizoma Ameliorates Insulin Resistance and Gut Microbiota in Type 2 Diabetic Db/Db Mice. J. Funct. Foods 39, 139–151. doi: 10.1016/j.jff.2017.10.020
Zhang, L., Zhan, L., Hang, T., Luo, J., C, Z. (2021). Shenling Baizhu Powder Alleviates Chronic Inflammation to Prevent Type 2 Diabetes in ZDF Rats via Intestinal Flora China. J. Chin. Materia Med 47, 988–1000. doi: 10.19540/j.cnki.cjcmm.20210907.401
Zhao, L., Ma, P., Peng, Y., Wang, M., Peng, C., Zhang, Y., et al. (2021). Amelioration of Hyperglycaemia and Hyperlipidaemia by Adjusting the Interplay Between Gut Microbiota and Bile Acid Metabolism: Radix Scutellariae as a Case. Phytomedicine 83, 153477. doi: 10.1016/j.phymed.2021.153477
Zhong, Y.-B., Kang, Z.-P., Wang, M.-X., Long, J., Wang, H.-Y., Huang, J.-Q., et al. (2021). Curcumin Ameliorated Dextran Sulfate Sodium-Induced Colitis via Regulating the Homeostasis of DCs and Treg and Improving the Composition of the Gut Microbiota. J. Funct. Foods 86, 104716. doi: 10.1016/j.jff.2021.104716
Zhong, H. J., Yuan, Y., Xie, W. R., Chen, M. H., He, X. X. (2016). Type 2 Diabetes Mellitus Is Associated With More Serious Small Intestinal Mucosal Injuries. PloS One 11, e0162354. doi: 10.1371/journal.pone.0162354
Zhou, G., Wu, F., Zhu, J., Gao, Y., Wan, H. (2020). Effect of Wumeiwan on Intestinal Microflora, Inflammatory Factor and Short Chain Fatty Acids in Type 2 Diabetic Rats. Chin. J. Exp. Traditional Med. Formulae 26 (10), 8–15. doi: 10.13422/j.cnki.syfjx.20200605. (In chinese).
Zhuang, X., Liu, C., Zhan, S., Tian, Z., Li, N., Mao, R., et al. (2021). Gut Microbiota Profile in Pediatric Patients With Inflammatory Bowel Disease: A Systematic Review. Front. Pediatr. 9, 626232. doi: 10.3389/fped.2021.626232
Keywords: gut microbiota, diabetes mellitus, ulcerative colitis, intestinal barrier, inflammation.
Citation: Zhang B, Liu K, Yang H, Jin Z, Ding Q and Zhao L (2022) Gut Microbiota: The Potential Key Target of TCM’s Therapeutic Effect of Treating Different Diseases Using the Same Method—UC and T2DM as Examples. Front. Cell. Infect. Microbiol. 12:855075. doi: 10.3389/fcimb.2022.855075
Received: 14 January 2022; Accepted: 04 March 2022;
Published: 30 March 2022.
Edited by:
Leticia Reyes, University of Wisconsin-Madison, United StatesReviewed by:
Wuwen Feng, Chengdu University of Traditional Chinese Medicine, ChinaJianye Yuan, Shanghai University of Traditional Chinese Medicine, China
Copyright © 2022 Zhang, Liu, Yang, Jin, Ding and Zhao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Linhua Zhao, bWVsb256aGFvQDE2My5jb20=
†These authors have contributed equally to this work
 Boxun Zhang
Boxun Zhang Ke Liu
Ke Liu Haoyu Yang
Haoyu Yang Zishan Jin
Zishan Jin Qiyou Ding
Qiyou Ding Linhua Zhao
Linhua Zhao