- Laboratorio Nacional de Genómica para la Biodiversidad (Langebio), Unidad de Genómica Avanzada, Centro de Investigación y de Estudios Avanzados del Instituto Politécnico Nacional (CINVESTAV-IPN), Guanajuato, Mexico
Alterations in the timing of developmental programs during evolution, that lead to changes in the shape, or size of organs, are known as heterochrony. Heterochrony has been widely studied in animals, but has often been neglected in plants. During plant evolution, heterochronic shifts have played a key role in the origin and diversification of leaves, roots, flowers, and fruits. Heterochrony that results in a juvenile or simpler outcome is known as paedomorphosis, while an adult or more complex outcome is called peramorphosis. Mechanisms that alter developmental timing at the cellular level affect cell proliferation or differentiation, while those acting at the tissue or organismal level change endogenous aging pathways, morphogen signaling, and metabolism. We believe that wider consideration of heterochrony in the context of evolution will contribute to a better understanding of plant development.
The Different Types of Heterochrony
In the 1870s, Ernst Haeckel identified temporal and spatial changes in development in a descendant relative to its ancestor as the two mechanisms most important for evolution (Haeckel, 1875). Haeckel named spatial changes heterotopy, and temporal changes heterochrony.
However, the meaning of heterochrony has changed since Haeckel first coined the term. Haeckel used the term heterochrony to refer to deviations from his well-known “Biogenetic Law,” which states that the sequence of developmental events (ontogeny) largely recapitulates the sequence of events in the evolutionary history of the species (phylogeny) (Haeckel, 1875). Thus, heterochrony originally referred to a change in the timing of appearance of a feature in a developmental sequence of an organism, relative to the sequence that occurred in the organism’s phylogeny. In the middle of the 20th century, De Beer (1951) uncoupled heterochrony from recapitulation. He used heterochrony to denote differences in the timing of developmental events when comparing two related species, to explain how heterochrony could generate diversity among organisms. Gould (1977) re-associated the concept of heterochrony to recapitulation, defining heterochrony as “changes in the relative time of appearance and rate of development for characters already present in ancestors,” emphasizing changes in relative size and shape, rather than in the timing of developmental events, to detect heterochrony. At the turn of the 21st century, Smith proposed that it would be more useful to define two types of heterochrony: ‘growth heterochrony,’ which emphasizes changes in final size and shape; and ‘sequence heterochrony,’ which is closer to the original usage of Haeckel and de Beer, and allows explanation of phenotypic variation by changes in the timing of developmental events (Smith, 2002, 2003; Keyte and Smith, 2014).
Sequence heterochrony (hereafter referred to as ‘heterochrony’) can be classified in two categories: paedomorphosis and peramorphosis. When compared to ancestral development, paedomorphosis results in a juvenile or simple outcome, whereas peramorphosis results in an adult or more complex phenotype. Each of these two categories of heterochrony can result from variation in timing of the onset, offset or rate of a developmental process, as proposed by Alberch et al. (1979). This variation can result in 6 different types of heterochrony (Figure 1). Paedomorphosis can result from the precocious end of a developmental process (progenesis), from a delayed start of the process (post-displacement), or from a slower rate of development (neoteny). Peramorphosis is the result of an extended period of development due to a later termination (hypermorphosis) or an earlier onset (pre-displacement), or of a higher rate of development (acceleration) (Figure 1A). Hypothetical examples for peramorphosis and paedomorphosis in plant embryogenesis and vegetative development are shown in Figures 1B,C.
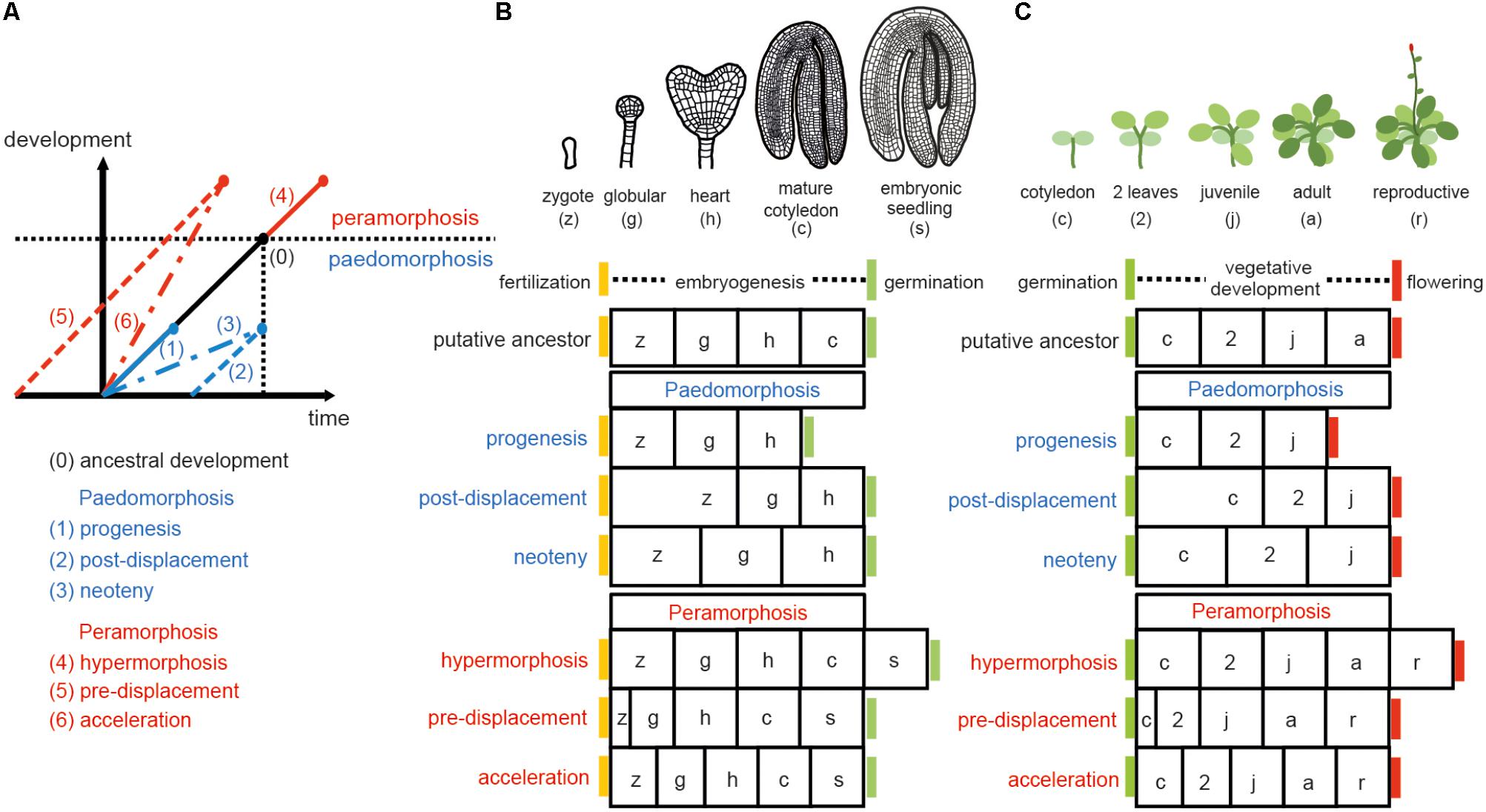
FIGURE 1. Types of heterochrony. (A) Schematic representation of the 6 types of heterochrony. The black line represents the time required to reach a certain developmental stage in the ancestral ontogeny. Blue lines show the 3 types of paedomorphosis: progenesis (precocious offset), post-displacement (delayed onset) and neoteny (slower developmental rate). Red lines show the 3 types of peramorphosis: hypermorphosis (delayed offset), pre-displacement (precocious onset), and acceleration (higher developmental rate). Drawing in (A) is based on Alberch et al. (1979) and Geuten and Coenen (2013). (B) Heterochrony scenarios for embryogenesis. Arabidopsis embryogenesis is taken here as the hypothetical ancestral development, divided in four stages for illustrative purposes. A fifth stage, where the embryo has produced the first two leaves plus the two cotyledons (denoted embryonic seedling), is proposed as the final stage in peramorphic embryogenesis, whereas paedomorphic embryogenesis is expected to conclude at the heart stage. (C) Heterochrony scenarios for vegetative development. In this case, Arabidopsis vegetative development, where the plant continues producing cauline/reproductive leaves on the stem before flowering (drawn on the top), represents a hypothetical case of peramorphic vegetative development, compared to a hypothetical ancestor which flowers at the adult stage without producing cauline leaves. Paedomorphic vegetative development is predicted to result in plants flowering at the juvenile stage.
While these classifications are useful to illustrate changes in developmental timing, when considering real life examples, it is often difficult to distinguish between different kinds of heterochrony. In many cases, evolution can result in distinct types of heterochrony, each occurring at a discrete stage of a developmental process (reviewed in plants by Li and Johnston, 2000).
Heterochrony in the Evolution and Diversification of Plants
Land plants (embryophytes) have undergone many morphological innovations since their emergence in the mid-Ordovician period, about 470 million years ago. Early diverging lineages originated over a period of more than 100 million years, during the Silurian and early Devonian periods (Kenrick and Crane, 1997; Pires and Dolan, 2012; Harrison and Morris, 2018). Embryophytes evolved from a freshwater algae ancestor that was related to the extant charophyte groups Charales, Coleochaetales, and Zygnematales. The transition to growth on land, as observed in bryophytes (hornworts, mosses, and liverworts), involved the origin of spores, alternation of gametophyte and sporophyte generations, uniaxial forms, and three-dimensional growth. Innovations of terrestrial vascular plants (lycophytes, monilophytes, and spermatophytes) included bifurcation, indeterminacy, sporophytic dominance, axillary branching, and the formation of meristems, leaves, and roots (Harrison, 2017).
Several of the evolutionary steps above have been linked to heterochrony. For the origin of sporogenesis, bryophyte data suggest that spores were produced directly from zygotes in a process involving precocious cytokinesis, acceleration of meiosis and delayed wall deposition from the zygote to the meiospores (Brown and Lemmon, 2011). The branched sporophyte (polisporangiophyte) has been hypothesized to have evolved by extended vegetative growth of the apical cell. This longer period of vegetative growth was proposed to result in a prolonged embryonic axis, shoot branching, and a delay in the transition to reproductive growth, producing the sporangium (Rothwell et al., 2014; Tomescu et al., 2014). Additional studies of plant fossils should provide more evidence for ancient plant morphologies, which would allow comparison between contemporary and extinct forms (Rothwell et al., 2014).
In extant plants, heterochronic changes have been identified in gametophyte development, embryogenesis, vegetative development, shoot maturation, and floral morphogenesis. The female gametophyte of Gnetum is structurally divergent from other plants because of differences in the timing of fertilization and somatic development. Temporal alterations in cell cycle progression have contributed to diversified temporal patterns of spermatogenesis and gamete fusion during fertilization (Friedman, 1999; Tian et al., 2005). In a case of progenesis, fertilization occurs at a free nuclear stage of somatic development, a juvenile stage compared to the ancestral somatic ontogeny, precluding the differentiation of egg cells (Friedman and Carmichael, 1998). The apomictic development of Boechera ovules has been associated with heterochronic gene expression patterns compared to non-apomictic (sexual) ovules (Sharbel et al., 2010). The development of Rafflesiaceae, a holoparasitic plant family which infects grapevines, shows two heterochronic shifts: an arrest at the proembryonic stage, which can be considered an example of neoteny, and acceleration of the transition from the undifferentiated endophyte to flowering, skipping vegetative shoot maturation (Nikolov et al., 2014).
A Quantitative Trait Loci (QTL) analysis comparing Eucalyptus globulus populations with precocious vegetative phase change and populations in which vegetative phase change is delayed several years identified the expression of the microRNA EglMIR156.5 as responsible for heterochronic variation in vegetative phase change in E. globulus (Hudson et al., 2014). Another QTL analysis concluded that heterochrony underlies natural variation in Cardamine hirsuta leaf form (Cartolano et al., 2015). QTL mapping determined that the effect is caused by cis-regulatory variation in the floral repressor ChFLC such that populations with low-expressing ChFLC alleles show both early flowering and accelerated acquisition of adult leaf traits, particularly increased leaflet number. Morphometric and QTL analyses have determined that heterochronic mutations contribute to natural variation in Antirrhinum and to grapevine heteroblasty (Costa et al., 2012; Chitwood et al., 2016). A Principal Component Analysis (PCA) of the ontogenetic trajectories of leaf form among the three genera of marsileaceous ferns (Marsilea, Regnellidium, and Pilularia) suggested that they show a paedomorphic phenotype, compared to the more complex ancestral development, caused by accelerated growth rate and early termination at a simplified leaf form (Pryer and Hearn, 2009).
Evolutionary diversity in inflorescence architecture of the Solanaceae is modulated by heterochronic shifts in the acquisition of floral fate (Lippman et al., 2008; Park et al., 2012). A comparison of transcriptomes of meristem maturation from five domesticated and wild Solanaceae species revealed a peak of expression divergence, resembling the “inverse hourglass” model for animal embryogenesis, which states that a mid-development period of divergence drives morphological variation (Lemmon et al., 2016). In grasses, a delay in the shoot meristem (SM) to floral meristem (FM) transition results in more complex panicles (Kyozuka et al., 2014). Poplars (Populus sp.) and willows (Salix sp.) bear compact unisexual inflorescences known as “catkins,” which have been proposed to be evolved from a simplification of the panicle form by an early SM to FM transition (Cronk et al., 2015). Heterochronic changes have also contributed to natural variation in flowering time and shoot architecture among Mimulus guttatus populations (Baker and Diggle, 2011).
Morphological diversity of the perianth in Dipsacoideae is caused by heterochronic changes in organ initiation, specifically in the number of sepals (Naghiloo and Claßen-Bockhoff, 2017). The great shape diversity of sepals among Iris species is due more to heterochrony than to heterotopic changes (Guo, 2015). A study in Brassicaceae showed that evolution of corolla monosymmetry from the polysymmetrical ancestral flower involved a heterochronic shift in the expression of CYC2 genes (a clade of TCP transcription factors) from early adaxial expression in ancestral floral meristems, to a later adaxial expression in petal development (Busch et al., 2012). Heterochronic, but not heterotopic, CYC2 expression has also been associated with a loss of papillate conical cells in petals and a shift to bird-pollination system in Lotus (Ojeda et al., 2017). A paedomorphic morphology, in which the flowers hold mature pollen in unopened bud-like structures, led to specialized pollination in a clade of Madagascar vines (Euphorbiaceae) (Armbruster et al., 2013). The evolution of cleistogamous capitulum from a chasmogamous ancestral state is a classic example of paedomorphosis, since the cleistogamous shape shows juvenile traits (Lord and Hill, 1987). Cleistogamy in Asteraceae specifically evolved by pre-displacement and progenesis of floral development, as well as neoteny of all whorls other than the gynoecium (Porras and Muñoz, 2000). The diversity of floral morphologies within Jaltomata, a Solanaceae genus, is due to hypermorphosis and acceleration of some corolla traits (Kostyun et al., 2017). The diversity of Azorean butterfly orchids is also caused by floral heterochronic shifts (Bateman et al., 2014). Recently, Ronse de Craene (2018) emphasized the importance of heterochrony in three developmental processes: phyllotaxis, the development of common stamen-petal primordia and obdiplostemony, linking changes in the growth rate with delayed organ initiation. Heterochronic growth rates of the perianth and style, and early hypanthium elongation, are responsible for the great species diversity within the morphologically homogeneous Eugenia genus (Vasconcelos et al., 2018). Finally, heterochronic expression of the fw2.2 allele, which affects cell division in early fruit development, is responsible for natural variation in tomato fruit size (Cong et al., 2002).
Studying Heterochronic Mutants to Elucidate Genetic Control of Timing
The study of mutants affected in developmental timing has shed light on genetic pathways controlling morphogenesis and developmental transitions. Heterochrony can be caused by earlier or later activation or repression of these pathways.
leafy cotyledon (lec), dicer-like1 (dcl1), and extra cotyledon (xtc) mutants of Arabidopsis thaliana represent heterochronic phenotypes that have helped to define seed maturation programs. lec mutants produce cotyledons with features of leaf identity (Meinke, 1992), a clear example of homeosis: the replacement of one structure by another. However, it is often difficult to distinguish between homeosis and heterochrony, since homeosis can be the result of both heterochrony and heterotopy (Li and Johnston, 2000; Geuten and Coenen, 2013). During late embryogenesis, LEC2 promotes seed maturation and represses postembryonic identity (Stone et al., 2008). Like lec mutants, dcl1 mutants show peramorphic phenotypes during embryogenesis, as chloroplast development and seed storage protein gene expression occur earlier than in wild type embryos. DCL1 is required for biogenesis of microRNAs, which repress seed maturation through the master regulators LEC2 and FUSCA3 (Nodine and Bartel, 2010; Willmann et al., 2011). The xtc1, xtc2, and altered meristem programming1 (amp1) mutants show a homeotic phenotype where the first one or two leaves are transformed into cotyledons (Conway and Poethig, 1997). In these three mutants, the globular to heart transition is delayed, causing an enlarged shoot meristem, which leads to extra organ formation during embryogenesis. This phenotype can be interpreted as hypermorphosis, a type of peramorphosis, since embryo development is extended to a more developed shape (like the peramorphic scenario in Figure 1B). However, if vegetative development is chosen as a point of reference, lec mutants would represent a case of pre-displacement (peramorphosis), and xtc1, xtc2, and amp1 would represent post-displacement (paedomorphosis) in the acquisition of leaf identity. AMP1 is required for miRNA-mediated translational repression on the ER membrane (Li et al., 2013).
Genetic regulation of the juvenile to adult transition, also called vegetative phase change, has been studied using both paedomorphic and peramorphic mutants. Screenings for mutants showing an early adult (peramorphic) phenotype produced alleles of genes related to small RNA biogenesis like zippy/ago7, sgs3, and rdr6 (Hunter et al., 2003; Peragine et al., 2004), while mutants with a late adult (paedomorphic) phenotype are due to increased miRNA levels (Gillmor et al., 2014; Xu M. et al., 2016, 2018; Guo et al., 2017). The genetic basis for these phenotypes is explained by altered expression of the closely related microRNAs miR156 and miR157, the main regulators of vegetative phase change that act by repressing SPL transcription factors at both transcriptional and translational levels, in a threshold-dependent manner (Wu and Poethig, 2006; Chuck et al., 2007; Poethig, 2013; He et al., 2018). Both MIR156 and SPL gene families are directly regulated by epigenetic marks: MIR156 is activated by H3K4me3 and H3K4ac, which are promoted by the SWR1-C complex and the chromatin remodeler BRAHMA, and is repressed by H3K27me3, which is promoted by the Polycomb proteins SWINGER and CURLY LEAF and by the chromatin remodeler PICKLE (Xu M. et al., 2016; Xu Y. et al., 2016; Xu M. et al., 2018). SPL genes are activated by histone acetylation mediated by the SAGA-like complex and repressed by H2AUb mediated by the Polycomb proteins RING1A and RING1B (Kim et al., 2015; Li et al., 2017). The study of plants with loss- or gain-of function of SPL genes has also defined an endogenous flowering pathway in which some SPL genes promote the expression of miR172, which in turn promotes flowering by repressing the APETALA2 family flowering repressors (Wang et al., 2009; Wu et al., 2009; Huijser and Schmid, 2011).
Besides major developmental transitions, organ morphogenesis can also be affected by heterochronic activation of genetic regulators. For instance, early germination of Brassica rapa embryos results in organs with mosaics of cotyledon and leaf identity (Fernandez, 1997), and differential temporal expression of the class II of TEOSINTE BRANCHED1/CYCLOIDEA/PROLIFERATION CELL FACTOR (TCP) genes results in leaves with different size and shape (Efroni et al., 2008).
Mechanisms Driving Changes in Developmental Timing in Plants (Transcriptional, Metabolic, and Cellular Heterochrony)
Transcriptional heterochrony refers to a change in the timing of activation or repression of gene expression, and is often caused by changes in cis-regulatory gene regions (Pham et al., 2017). Transcriptional heterochrony in the genetic pathways mentioned above is a common way of producing heterochronic phenotypes. Metabolic control of pathways regulating developmental transitions (referred to here as “metabolic heterochrony”), and temporal control of cell proliferation, cell expansion and cell differentiation (referred to here as “cellular heterochrony”) are other mechanisms driving heterochrony in plants (Figure 2). Transcriptional, metabolic and cellular processes are interconnected, so the molecular origin of heterochrony can be due to a combination of mechanisms.
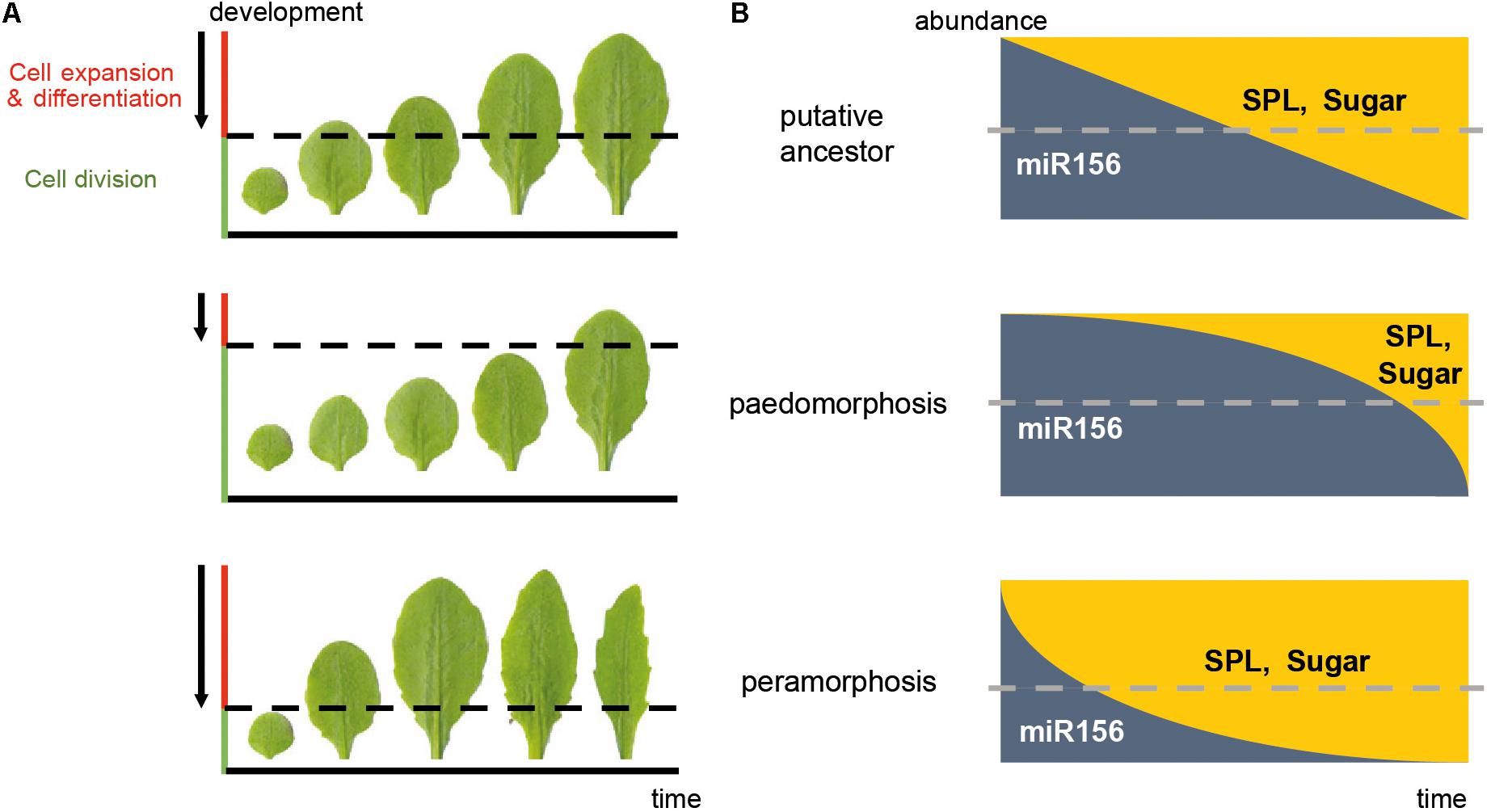
FIGURE 2. Mechanisms driving heterochrony in plants. (A) Cellular heterochrony. The sequence of rosette leaves produced during vegetative development is shown. The phenotype of Arabidopsis Columbia wild type plants is taken as a hypothetical ancestral state; a paedomorphic scenario (neoteny) and a peramorphic scenario (acceleration) are also shown. Arrows represent the direction of the wave of the cell division arrest in a basipetal gradient, where cells below the dotted line are still actively dividing whereas the cells above are expanding and/or differentiating. (B) Transcriptional and metabolic heterochrony. A graphic representation of the predicted abundance of the microRNA miR156/7 and its targets, the SPL genes, as well as sugar abundance along the time of vegetative development, corresponding to the phenotypes in (A). Dotted lines represent the threshold of miR156/7 and SPL abundance which leads to juvenility (above the threshold) or adulthood (below the threshold). In this case, a delay in the repression of miR156 and activation of SPL expression and sugar production results in paedomorphosis, whereas a precocious decay of miR156 and early activation of SPL expression and sugar production results in peramorphosis. These drawings are simplified representations, and do not reflect the exact abundances in nature. The actual pattern of miR156/SPL abundance is closer to that depicted for “peramorphosis” state.
Metabolic heterochrony can be influenced by hormones, sugars, and redox signals (Jia et al., 2017). Differential biosynthesis, transport and perception of hormones such as auxin, jasmonic acid (JA), gibberellin and abscisic acid influence heterochrony by controlling regulators of developmental processes. For instance, mutants in the AUXIN RESPONSE FACTORS (ARF) ARF3 and ARF4 delay the adult transition (Fahlgren et al., 2006; Hunter et al., 2006), and auxin homeostasis controls the transition from floral stem cell maintenance to gynoecium formation (Yamaguchi et al., 2017). Exogenous JA can delay the adult transition by postponing the decline of miR156 expression (Beydler et al., 2016), and gibberellin accelerates flowering by releasing SPL genes from repression by DELLA proteins (Yu et al., 2012). Nutritional status has been associated with the control of vegetative phase change since the early 20th century (Goebel, 1908). Sugar produced by photosynthesis is necessary for the acquisition of adult traits and is partially responsible for the decrease in miR156 expression in late vegetative development (Yang et al., 2013; Yu et al., 2013; Buendía-Monreal and Gillmor, 2017). HEXOKINASE1 (HXK1) and Trehalose-6-phosphate (T6P) are important for the sugar-mediated repression of miR156, thereby promoting vegetative phase change and flowering (Wahl et al., 2013; Yang et al., 2013).
At the cellular level, organogenesis consists of a sequence of three stages: the establishment of polarity, cell proliferation, and cell expansion (Walcher-Chevillet and Kramer, 2016). The timing of initiation and termination of these stages is crucial for the size and shape of organs, and heterochrony in this sequence results in diversification of organ size and shape (Figure 2). The transition from cell proliferation to cell expansion and differentiation requires coordination between the cell cycle and cell growth (Sablowski and Carnier Dornelas, 2014). In leaves of model plants, this transition moves as a basipetal wave of cell cycle arrest that begins at the distal part of the primordium and moves to the base. Cells behind the mitotic arrest front become highly vacuolated and begin to expand (Donnelly et al., 1999; Czesnick and Lenhard, 2015). However, other plant species can show diffuse growth, and acropetal or bidirectional cell cycle arrest gradients (Das Gupta and Nath, 2015). The acquisition of photosynthetic capacity is required for the shift from cell division to cell expansion (Andriankaja et al., 2012). This shift correlates with the role of sugar in promoting the Target of Rapamycin (TOR) pathway and repressing the Sucrose-non-fermenting1-related kinase 1 (SnRK1): TOR and T6P induce cell expansion by promoting macromolecular synthesis, whereas SnRK1 promotes catabolism (Tsai and Gazzarrini, 2014; Sablowski, 2016). Two microRNAs play opposite roles in this cellular shift: miR319 represses the expression of class II TCP factors, which are inhibitors of cell proliferation, whereas miR396 restricts the expression of GROWTH REGULATING FACTORS (GRFs), which delay differentiation (Das Gupta and Nath, 2015; Maugarny-Calés and Laufs, 2018). The transition from an indeterminate shoot apical meristem to a determinate floral meristem also involves temporal regulation of the cellular identity. The timing of AGAMOUS activation of KNUCKLES, which in turn represses WUSCHEL, defines the temporal window of indeterminacy and consequently the size and the number of organs (Sun et al., 2014).
Conclusion
An understanding of temporal regulation of plant development is necessary to better appreciate the diversity of plant forms that we see in nature, to explain plant morphological evolution, and to manipulate plant architecture for the benefit of agriculture. As outlined above, interrelated transcriptional, metabolic, and cellular mechanisms drive heterochrony in extant species. Further research on these pathways in angiosperms and basal plant lineages should reveal more about the changes in developmental timing that have driven the evolution of development in plants.
Author Contributions
MB-M conceived and wrote the manuscript. CSG edited and revised the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Alberch, P., Gould, S., Oster, G., and Wake, D. (1979). Size and shape in ontogeny and phylogeny. Paleobiology 5, 296–317. doi: 10.1017/S0094837300006588
Andriankaja, M., Dhondt, S., DeBodt, S., Vanhaeren, H., Coppens, F., DeMilde, L., et al. (2012). Exit from proliferation during leaf development in Arabidopsis thaliana: a not-so-gradual process. Dev. Cell 22, 64–78. doi: 10.1016/j.devcel.2011.11.011
Armbruster, W. S., Lee, J., Edwards, M. E., and Baldwin, B. G. (2013). Floral paedomorphy leads to secondary specialization in pollination of Madagascar Dalechampia (Euphorbiaceae). Evolution 67, 1196–1203. doi: 10.1111/evo.12002
Baker, R. L., and Diggle, P. K. (2011). Node-specific branching and heterochronic changes underlie population-level differences in Mimulus guttatus (Phrymaceae) shoot architecture. Am. J. Bot. 98, 1924–1934. doi: 10.3732/ajb.1100098
Bateman, R. M., Rudall, P. J., Bidartondo, M. I., Cozzolino, S., Tranchida-Lombardo, V., Carine, M. A., et al. (2014). Speciation via floral heterochrony and presumed mycorrhizal host switching of endemic butterfly orchids on the Azorean archipelago. Am. J. Bot. 101, 979–1001. doi: 10.3732/ajb.1300430
Beydler, B. D., Osadchuk, K., Cheng, C.-L., Manak, J. R., and Irish, E. E. (2016). The juvenile phase of maize sees upregulation of stress-response genes and is extended by exogenous JA. Plant Physiol. 171, 2648–2658. doi: 10.1104/pp.15.01707
Brown, R. C., and Lemmon, B. E. (2011). Spores before sporophytes: hypothesizing the origin of sporo- genesis at the algal – plant transition. New Phytol. 190, 875–881. doi: 10.1111/j.1469-8137.2011.03709.x
Buendía-Monreal, M., and Gillmor, C. S. (2017). Convergent repression of miR156 by sugar and the CDK8 module of Arabidopsis Mediator. Dev. Biol. 423, 19–23. doi: 10.1016/j.ydbio.2017.01.007
Busch, A., Horn, S., Mühlhausen, A., Mummenhoff, K., and Zachgo, S. (2012). Corolla monosymmetry: evolution of a morphological novelty in the Brassicaceae family. Mol. Biol. Evol. 29, 1241–1254. doi: 10.1093/molbev/msr297
Cartolano, M., Pieper, B., Lempe, J., Tattersall, A., Huijser, P., Tresch, A., et al. (2015). Heterochrony underpins natural variation in Cardamine hirsuta leaf form. Proc. Natl. Acad. Sci. U.S.A. 112, 10539–10544. doi: 10.1073/pnas.1419791112
Chitwood, D. H., Klein, L. L., O’Hanlon, R., Chacko, S., Greg, M., Kitchen, C., et al. (2016). Latent developmental and evolutionary shapes embedded within the grapevine leaf. New Phytol. 210, 343–355. doi: 10.1111/nph.13754
Chuck, G., Cigan, A. M., Saeteurn, K., and Hake, S. (2007). The heterochronic maize mutant corngrass1 results from overexpression of a tandem microRNA. Nat. Genet. 39, 544–549. doi: 10.1038/ng2001
Cong, B., Liu, J., and Tanksley, S. D. (2002). Natural alleles at a tomato fruit size quantitative trait locus differ by heterochronic regulatory mutations. Proc. Natl. Acad. Sci. U.S.A. 99, 13606–13611. doi: 10.1073/pnas.172520999
Conway, L. J., and Poethig, R. S. (1997). Mutations of Arabidopsis thaliana that transform leaves into cotyledons. Proc. Natl. Acad. Sci. U.S.A. 94, 10209–10214. doi: 10.1073/pnas.94.19.10209
Costa, M. M. R., Yang, S., Critchley, J., Feng, X., Wilson, Y., Langlade, N., et al. (2012). The genetic basis for natural variation in heteroblasty in Antirrhinum. New Phytol. 196, 1251–1259. doi: 10.1111/j.1469-8137.2012.04347.x
Cronk, Q. C. B., Needham, I., and Rudall, P. J. (2015). Evolution of catkins: inflorescence morphology of selected Salicaceae in an evolutionary and developmental context. Front. Plant Sci. 6:1030. doi: 10.3389/fpls.2015.01030
Czesnick, H., and Lenhard, M. (2015). Size control in plants — lessons from leaves and flowers. Cold Spring Harb. Perspect. Biol. 7:a019190. doi: 10.1101/cshperspect.a019190
Das Gupta, M., and Nath, U. (2015). Divergence in patterns of leaf growth polarity is associated with the expression divergence of miR396. Plant Cell 27, 2785–2799. doi: 10.1105/tpc.15.00196
Donnelly, P. M., Bonetta, D., Tsukaya, H., Dengler, R. E., and Dengler, N. G. (1999). Cell cycling and cell enlargement in developing leaves of Arabidopsis. Dev. Biol. 215, 407–419. doi: 10.1006/dbio.1999.9443
Efroni, I., Blum, E., Goldshmidt, A., and Eshed, Y. (2008). A protracted and dynamic maturation schedule underlies Arabidopsis leaf development. Plant Cell 20, 2293–2306. doi: 10.1105/tpc.107.057521
Fahlgren, N., Montgomery, T. A., Howell, M. D., Allen, E., Dvorak, S. K., Alexander, A. L., et al. (2006). Regulation of AUXIN RESPONSE FACTOR3 by TAS3 ta-siRNA Affects Developmental Timing and Patterning in Arabidopsis. Curr. Biol. 16, 939–944. doi: 10.1016/j.cub.2006.03.065
Fernandez, D. E. (1997). Developmental basis of homeosis in precociously germinating Brassica napus embryos: phase change at the shoot apex. Development 124, 1149–1157.
Friedman, W. E. (1999). Expression of the cell cycle in sperm of Arabidopsis: implications for understanding patterns of gametogenesis and fertilization in plants and other eukaryotes. Development 126, 1065–1075.
Friedman, W. E., and Carmichael, J. S. (1998). Heterochrony and developmental innovation: evolution of female gametophyte ontogeny in gnetum, a highly Apomorphic seed plant. Evolution 52, 1016–1030. doi: 10.1111/j.1558-5646.1998.tb01830.x
Geuten, K., and Coenen, H. (2013). Heterochronic genes in plant evolution and development. Front. Plant Sci. 4:381. doi: 10.3389/fpls.2013.00381
Gillmor, C. S., Silva-Ortega, C. O., Willmann, M. R., Buendía-Monreal, M., and Poethig, R. S. (2014). The Arabidopsis Mediator CDK8 module genes CCT (MED12) and GCT (MED13) are global regulators of developmental phase transitions. Development 141, 4580–4589. doi: 10.1242/dev.111229
Goebel, K. (1908). Einleitung in die Experimentelle Morphologie der Pflanzen. Leipzig: B.G. Teubner.
Guo, C., Xu, Y., Shi, M., Lai, Y., Wu, X., Wang, H., et al. (2017). Repression of miR156 by miR159 regulates the timing of the juvenile-to-adult transition in Arabidopsis. Plant Cell 29, 1293–1304. doi: 10.1105/tpc.16.00975
Guo, J. (2015). Mechanisms for the evolution of complex and diversely elaborated sepals in Iris identified by comparative analysis of developmental sequences. Am. J. Bot. 102, 819–832. doi: 10.3732/ajb.1400519
Harrison, C. J. (2017). Development and genetics in the evolution of land plant body plans. Philos. Trans. R. Soc. B Biol. Sci. 372:20150490. doi: 10.1098/rstb.2015.0490
Harrison, C. J., and Morris, J. L. (2018). The origin and early evolution of vascular plant shoots and leaves. Philos. Trans. R. Soc. B Biol. Sci. 373:20160496. doi: 10.1098/rstb.2016.0496
He, J., Xu, M., Willmann, M. R., Mccormick, K., Hu, T., Yang, L., et al. (2018). Threshold-dependent repression of SPL gene expression by miR156/miR157 controls vegetative phase change in Arabidopsis thaliana. PLoS Genet. 14:e1007337. doi: 10.1371/journal.pgen.1007337
Hudson, C. J., Freeman, J. S., Jones, R. C., Potts, B. M., Wong, M. M. L., Weller, J. L., et al. (2014). Genetic control of heterochrony in Eucalyptus globulus. G3 4, 1235–1245. doi: 10.1534/g3.114.011916
Huijser, P., and Schmid, M. (2011). The control of developmental phase transitions in plants. Development 138, 4117–4129. doi: 10.1242/dev.063511
Hunter, C., Sun, H., and Poethig, R. S. (2003). The Arabidopsis heterochronic gene ZIPPY Is an ARGONAUTE family member. Curr. Biol. 13, 1734–1739. doi: 10.1016/j.cub.2003.09.004
Hunter, C., Willmann, M. R., Wu, G., Yoshikawa, M., de la Luz Gutiérrez-Nava, M., and Poethig, R. S. (2006). Trans-acting siRNA-mediated repression of ETTIN and ARF4 regulates heteroblasty in Arabidopsis. Development 133, 2973–2981. doi: 10.1242/dev.02491
Jia, X. L., Chen, Y. K., Xu, X. Z., Shen, F., Zheng, Q. B., Du, Z., et al. (2017). miR156 switches on vegetative phase change under the regulation of redox signals in apple seedlings. Sci. Rep. 7, 1–13. doi: 10.1038/s41598-017-14671-8
Kenrick, P., and Crane, P. R. (1997). The origin and early evolution of plants on land. Nature 389, 33–39. doi: 10.1038/37918
Keyte, A. L., and Smith, K. K. (2014). Heterochrony and developmental timing mechanisms: changing ontogenies in evolution. Semin. Cell Dev. Biol. 34, 99–107. doi: 10.1016/j.semcdb.2014.06.015
Kim, J. Y., Oh, J. E., Noh, Y. S., and Noh, B. (2015). Epigenetic control of juvenile-to-adult phase transition by the Arabidopsis SAGA-like complex. Plant J. 83, 537–545. doi: 10.1111/tpj.12908
Kostyun, J. L., Preston, J. C., and Moyle, L. C. (2017). Heterochronic developmental shifts underlie floral diversity within Jaltomata ( Solanaceae ). Evodevo 8:17. doi: 10.1186/s13227-017-0080-z
Kyozuka, J., Tokunaga, H., and Yoshida, A. (2014). Control of grass inflorescence form by the fine-tuning of meristem phase change. Curr. Opin. Plant Biol. 17, 110–115. doi: 10.1016/j.pbi.2013.11.010
Lemmon, Z. H., Park, S. J., Jiang, K., Van Eck, J., Schatz, M. C., and Lippman, Z. B. (2016). The evolution of inflorescence diversity in the nightshades and heterochrony during meristem maturation. Genome Res. 26, 1676–1686. doi: 10.1101/gr.207837.116
Li, J., Wang, Z., Hu, Y., Cao, Y., and Ma, L. (2017). Polycomb group proteins RING1A and RING1B regulate the vegetative phase transition in Arabidopsis. Front. Plant Sci. 8:867. doi: 10.3389/fpls.2017.00867
Li, P., and Johnston, M. O. (2000). Heterochrony in plant evolutionary studies through the twentieth century. Bot. Rev. 66, 57–88. doi: 10.1007/BF02857782
Li, S., Liu, L., Zhuang, X., Yu, Y., Liu, X., Cui, X., et al. (2013). MicroRNAs inhibit the translation of target mRNAs on the endoplasmic reticulum in Arabidopsis. Cell 153, 562–574. doi: 10.1016/j.cell.2013.04.005
Lippman, Z. B., Cohen, O., Alvarez, J. P., Abu-Abied, M., Pekker, I., Paran, I., et al. (2008). The making of a compound inflorescence in tomato and related nightshades. PLoS Biol. 6:e288. doi: 10.1371/journal.pbio.0060288
Lord, E. M., and Hill, J. P. (1987). “Evidence for heterochrony in the evolution of plant form,” in Development as an Evolutionary Process, eds R. A. Raff and E. C. Raff (New York, NY: Alan R. Liss), 47–70.
Maugarny-Calés, A., and Laufs, P. (2018). Getting leaves into shape: a molecular, cellular, environmental and evolutionary view. Development 145:dev161646. doi: 10.1242/dev.161646
Meinke, D. W. (1992). A homoetic mutant of Arabidopsis thaliana with leafy cotyledons. Science 258, 1647–1649. doi: 10.1126/science.258.5088.1647
Naghiloo, S., and Claßen-Bockhoff, R. (2017). Developmental changes in time and space promote evolutionary diversification of flowers: a case study in Dipsacoideae. Front. Plant Sci. 8:1665. doi: 10.3389/fpls.2017.01665
Nikolov, L. A., Tomlinson, P. B., Manickam, S., Endress, P. K., Kramer, E. M., and Davis, C. C. (2014). Holoparasitic Rafflesiaceae possess the most reduced endophytes and yet give rise to the world’s largest flowers. Ann. Bot. 114, 233–242. doi: 10.1093/aob/mcu114
Nodine, M. D., and Bartel, D. P. (2010). MicroRNAs prevent precocious gene expression and enable pattern formation during plant embryogenesis. Genes Dev. 24, 2678–2692. doi: 10.1101/gad.1986710
Ojeda, D. I., Jaén-Molina, R., Santos-Guerra, A., Caujape-Castells, J., and Cronk, Q. (2017). Temporal, but not spatial, changes in expression patterns of petal identity genes are associated with loss of papillate conical cells and the shift to bird pollination in Macaronesian Lotus (Leguminosae). Plant Biol. 19, 420–427. doi: 10.1111/plb.12551
Park, S. J., Jiang, K., Schatz, M. C., and Lippman, Z. B. (2012). Rate of meristem maturation determines inflorescence architecture in tomato. Proc. Natl. Acad. Sci. U.S.A. 109, 639–644. doi: 10.1073/pnas.1114963109
Peragine, A., Yoshikawa, M., Wu, G., Albrecht, H. L., and Poethig, R. S. (2004). SGS3 and SGS2/SDE1/RDR6 are required for juvenile development and the production of trans-acting si RNAs in Arabidopsis. Genes Dev. 18, 2368–2379. doi: 10.1101/gad.1231804.nism
Pham, T., Day, S. M., Glassford, W. J., Williams, T. M., and Rebeiz, M. (2017). The evolutionary origination of a novel expression pattern through an extreme heterochronic shift. Evol. Dev. 19, 43–55. doi: 10.1111/ede.12215
Pires, N. D., and Dolan, L. (2012). Morphological evolution in land plants: new designs with old genes. Philos. Trans. R. Soc. B Biol. Sci. 367, 508–518. doi: 10.1098/rstb.2011.0252
Poethig, R. S. (2013). Vegetative Phase Change and Shoot Maturation in Plants, 1st Edn. New York, NY: Elsevier Inc. doi: 10.1016/B978-0-12-396968-2.00005-1
Porras, R., and Muñoz, J. M. (2000). Cleistogamous capitulum in Centaurea (Asteraceae): heterochronic origin. Am. J. Bot. 87, 925–933. doi: 10.2307/2656991
Pryer, K. M., and Hearn, D. J. (2009). Evolution of leaf form in marsileaceous ferns: evidence for heterochrony. Evolution 63, 498–513. doi: 10.1111/j.1558-5646.2008.00562.x
Ronse de Craene, L. (2018). Understanding the role of floral development in the evolution of angiosperm flowers: clarifications from a historical and physico-dynamic perspective. J. Plant Res. 131, 367–393. doi: 10.1007/s10265-018-1021-1
Rothwell, G. W., Wyatt, S. E., and Tomescu, A. M. F. (2014). Plant evolution at the interface of paleontology and developmental biology: an organism-centered paradigm. Am. J. Bot. 101, 899–913. doi: 10.3732/ajb.1300451
Sablowski, R. (2016). Coordination of plant cell growth and division: collective control or mutual agreement? Curr. Opin. Plant Biol. 34, 54–60. doi: 10.1016/j.pbi.2016.09.004
Sablowski, R., and Carnier Dornelas, M. (2014). Interplay between cell growth and cell cycle in plants. J. Exp. Bot. 65, 2703–2714. doi: 10.1093/jxb/ert354
Sharbel, T. F., Voigt, M. L., Corral, J. M., Galla, G., Kumlehn, J., Klukas, C., et al. (2010). Apomictic and sexual ovules of boechera display heterochronic global gene expression patterns. Plant Cell 22, 655–671. doi: 10.1105/tpc.109.072223
Smith, K. K. (2002). Sequence heterochrony and the evolution of development. J. Morphol. 252, 82–97. doi: 10.1002/jmor.10014
Smith, K. K. (2003). Time ’ s arrow: heterochrony and the evolution of development. Int. J. Dev. Biol. 47, 613–621.
Stone, S. L., Braybrook, S. A., Paula, S. L., Kwong, L. W., Meuser, J., Pelletier, J., et al. (2008). Arabidopsis leafy cotyledon2 induces maturation traits and auxin activity: implications for somatic embryogenesis. Proc. Natl. Acad. Sci. U.S.A. 105, 3151–3156. doi: 10.1073/pnas.0712364105
Sun, B., Looi, L. S., Guo, S., He, Z., Gan, E. S., Huang, J., et al. (2014). Timing mechanism dependent on cell division is invoked by Polycomb eviction in plant stem cells. Science 343:1248559. doi: 10.1126/science.1248559
Tian, H. Q., Yuan, T., and Russell, S. D. (2005). Relationship between double fertilization and the cell cycle in male and female gametes of tobacco. Sex. Plant Reprod. 17, 243–252. doi: 10.1007/s00497-004-0233-9
Tomescu, A. M. F., Wyatt, S. E., Hasebe, M., and Rothwell, G. W. (2014). Early evolution of the vascular plant body plan — the missing mechanisms. Curr. Opin. Plant Biol. 17, 126–136. doi: 10.1016/j.pbi.2013.11.016
Tsai, A. Y.-L., and Gazzarrini, S. (2014). Trehalose-6-phosphate and SnRK1 kinases in plant development and signaling: the emerging picture. Front. Plant Sci. 5:119. doi: 10.3389/fpls.2014.00119
Vasconcelos, T. N. C., Lucas, E. J., Faria, J. E. Q., and Prenner, G. (2018). Floral heterochrony promotes flexibility of reproductive strategies in the morphologically homogeneous genus Eugenia (Myrtaceae). Ann. Bot. 121, 161–174. doi: 10.1093/aob/mcx142
Wahl, V., Ponnu, J., Schlereth, A., Arrivault, S., Langenecker, T., Franke, A., et al. (2013). Regulation of flowering by trehalose-6-phosphate signaling in Arabidopsis thaliana. Science 339, 704–707. doi: 10.1126/science.1230406
Walcher-Chevillet, C. L., and Kramer, E. M. (2016). Breaking the mold: understanding the evolution and development of lateral organs in diverse plant models. Curr. Opin. Genet. Dev. 39, 79–84. doi: 10.1016/j.gde.2016.06.005
Wang, J., Czech, B., and Weigel, D. (2009). miR156-regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell 138, 738–749. doi: 10.1016/j.cell.2009.06.014
Willmann, M. R., Mehalick, A. J., Packer, R. L., and Jenik, P. D. (2011). MicroRNAs regulate the timing of embryo maturation in Arabidopsis. Plant Physiol. 155, 1871–1884. doi: 10.1104/pp.110.171355
Wu, G., Park, M. Y., Conway, S. R., Wang, J., Weigel, D., and Poethig, R. S. (2009). The sequential action of miR156 and miR172 regulates developmental timing in Arabidopsis. Cell 138, 750–759. doi: 10.1016/j.cell.2009.06.031
Wu, G., and Poethig, R. S. (2006). Temporal regulation of shoot development in Arabidopsis thaliana by miR156 and its target SPL3. Development 133, 3539–3547. doi: 10.1242/dev.02521
Xu, M., Hu, T., Smith, M. R., and Poethig, R. S. (2016). Epigenetic regulation of vegetative phase change in Arabidopsis. Plant Cell 28, 28–41. doi: 10.1105/tpc.15.00854
Xu, Y., Guo, C., Zhou, B., Li, C., Wang, H., Zheng, B., et al. (2016). Regulation of vegetative phase change by SWI2/SNF2 chromatin remodeling ATPase BRAHMA. Plant Physiol. 172, 2416–2428. doi: 10.1104/pp.16.01588
Xu, M., Leichty, A. R., Hu, T., and Poethig, R. S. (2018). H2A.Z promotes the transcription of MIR156A and MIR156C in Arabidopsis by facilitating the deposition of H3K4me3. Development 145:dev.152868. doi: 10.1242/dev.152868
Yamaguchi, N., Huang, J., Xu, Y., Tanoi, K., and Ito, T. (2017). Fine-tuning of auxin homeostasis governs the transition from floral stem cell maintenance to gynoecium formation. Nat. Commun. 8:1125. doi: 10.1038/s41467-017-01252-6
Yang, L., Xu, M., Koo, Y., He, J., and Poethig, R. S. (2013). Sugar promotes vegetative phase change in Arabidopsis thaliana by repressing the expression of MIR156A and MIR156C. eLife 2:e00260. doi: 10.7554/eLife.00260
Yu, S., Li, C., Zhou, C. M., Zhang, T. Q., Lian, H., Sun, Y., et al. (2013). Sugar is an endogenous cue for juvenile-to-adult phase transition in plants. eLife 2:e00269. doi: 10.7554/eLife.00269
Keywords: heterochrony, developmental timing, plant development, plant evolution, cell cycle, miR156
Citation: Buendía-Monreal M and Gillmor CS (2018) The Times They Are A-Changin’: Heterochrony in Plant Development and Evolution. Front. Plant Sci. 9:1349. doi: 10.3389/fpls.2018.01349
Received: 30 June 2018; Accepted: 27 August 2018;
Published: 18 September 2018.
Edited by:
Natalia Pabón-Mora, Universidad de Antioquía, ColombiaReviewed by:
Jianfei Zhao, University of Pennsylvania, United StatesJill Christine Preston, The University of Vermont, United States
Copyright © 2018 Buendía-Monreal and Gillmor. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Manuel Buendía-Monreal, bWFudWVsLmJ1ZW5kaWFAY2ludmVzdGF2Lm14
 Manuel Buendía-Monreal
Manuel Buendía-Monreal C. Stewart Gillmor
C. Stewart Gillmor